|
1
|
Chang JT, Lee YM and Huang RS: The impact
of the cancer genome atlas on lung cancer. Transl Res. 166:568–585.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Girard L, Rodriguez-Canales J, Behrens C,
Thompson DM, Botros IW, Tang H, Xie Y, Rekhtman N, Travis WD,
Wistuba II, et al: An expression signature as an aid to the
histologic classification of non-small cell lung cancer. Clin
Cancer Res. 22:4880–4889. 2016. View Article : Google Scholar : PubMed/NCBI
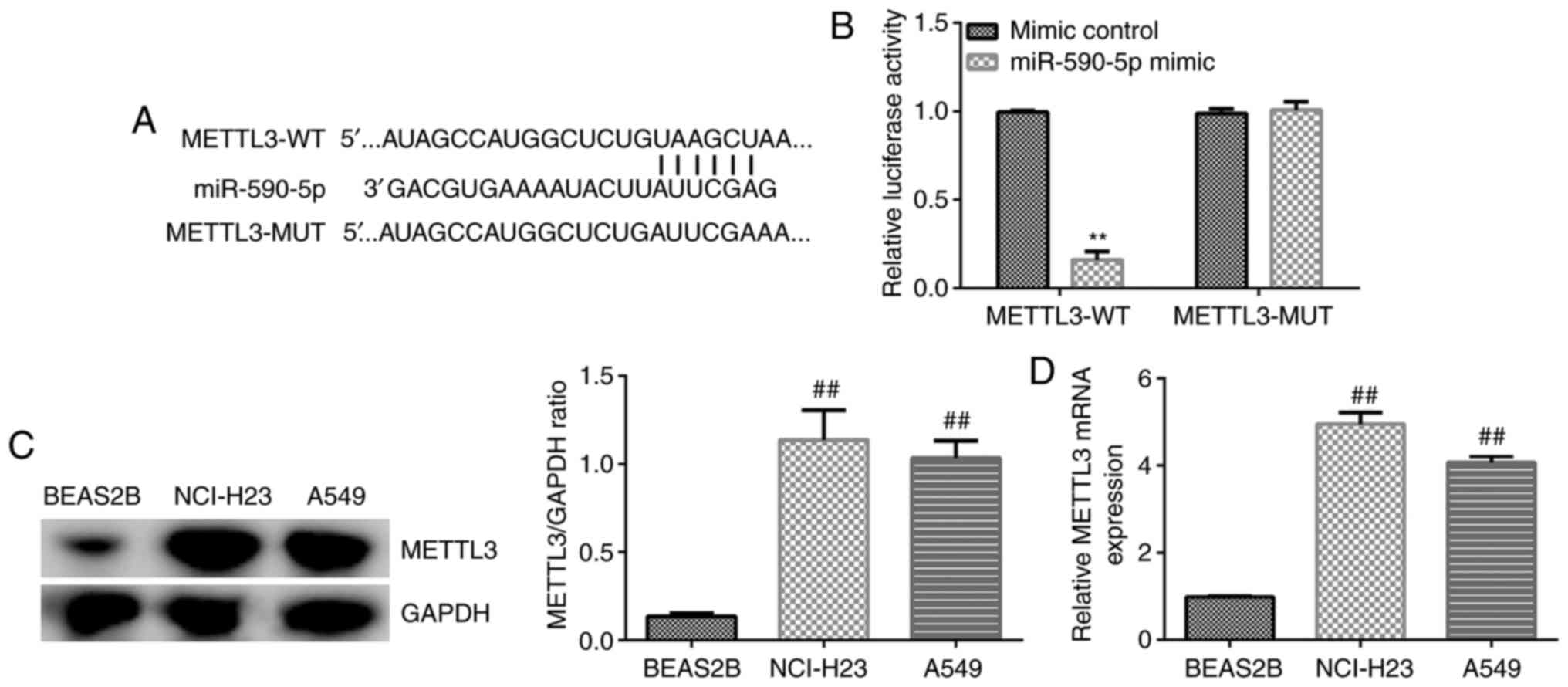
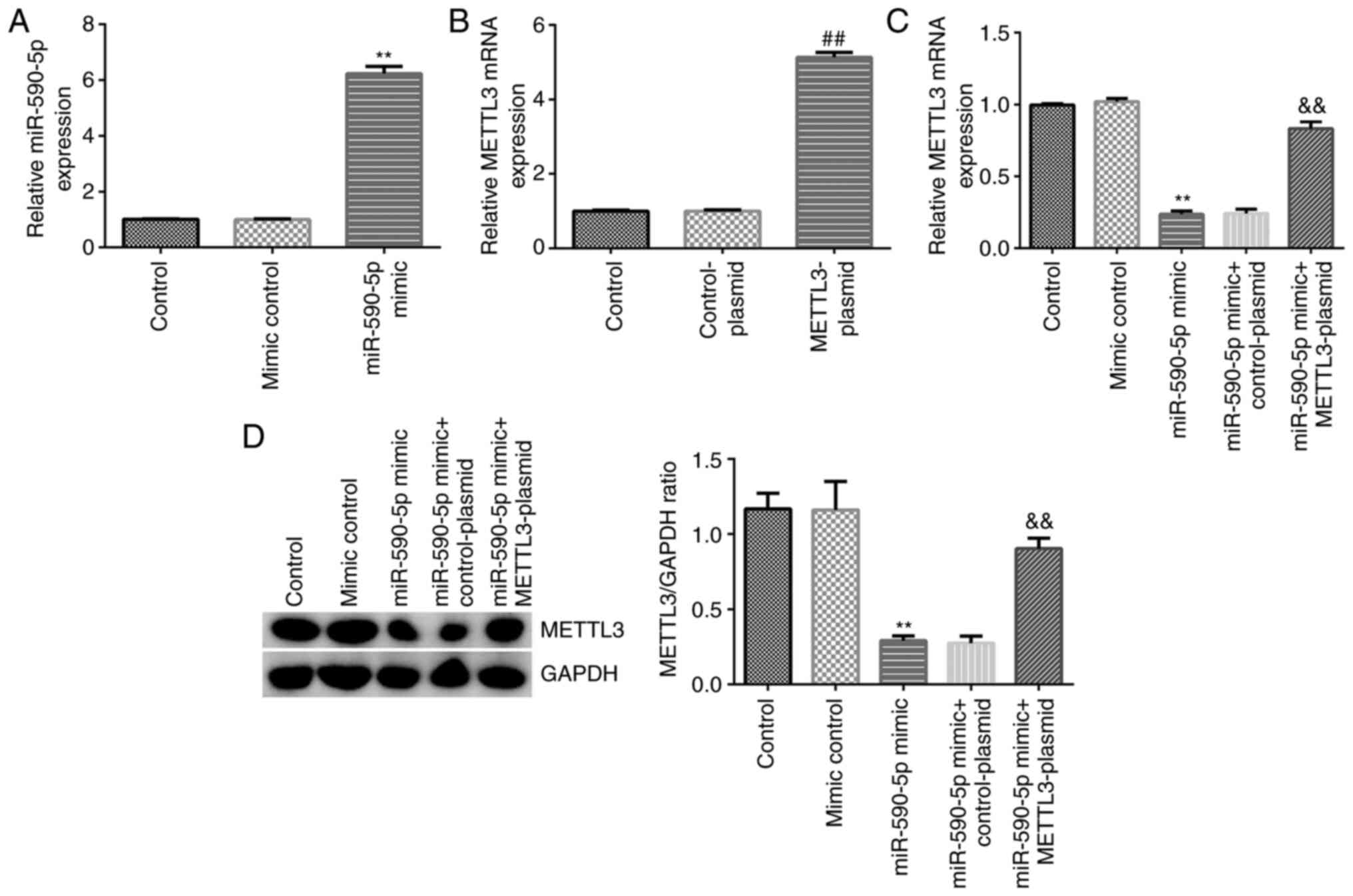
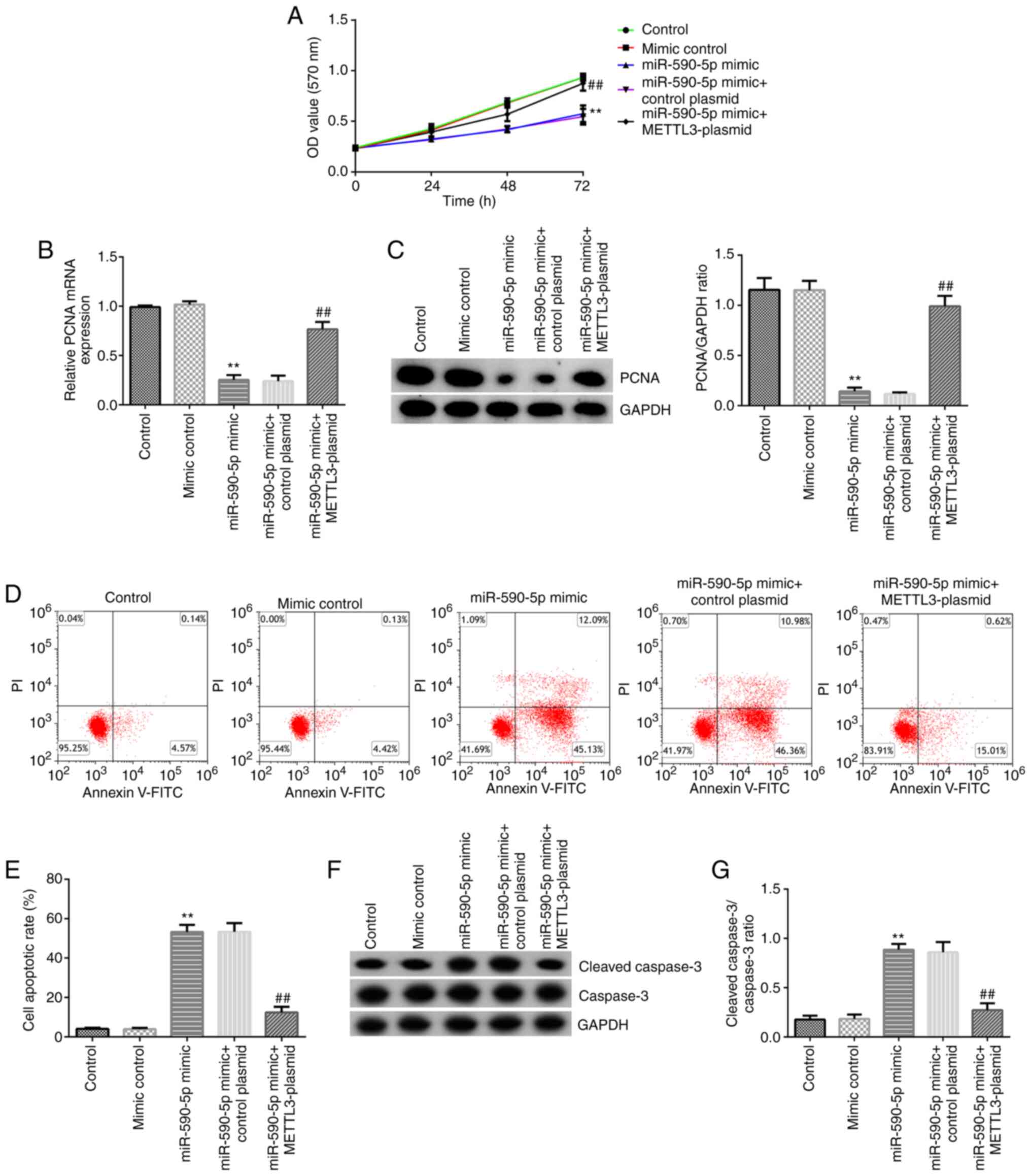
|
|
3
|
Sardenberg RA, Pinto C, Bueno CA and
Younes RN: Non-small cell lung cancer stage IV long-term survival
with isolated spleen metastasis. Ann Thorac Surg. 95:1432–1434.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Travis WD: Pathology of lung cancer. Clin
Chest Med. 32:669–692. 2011. View Article : Google Scholar : PubMed/NCBI
|
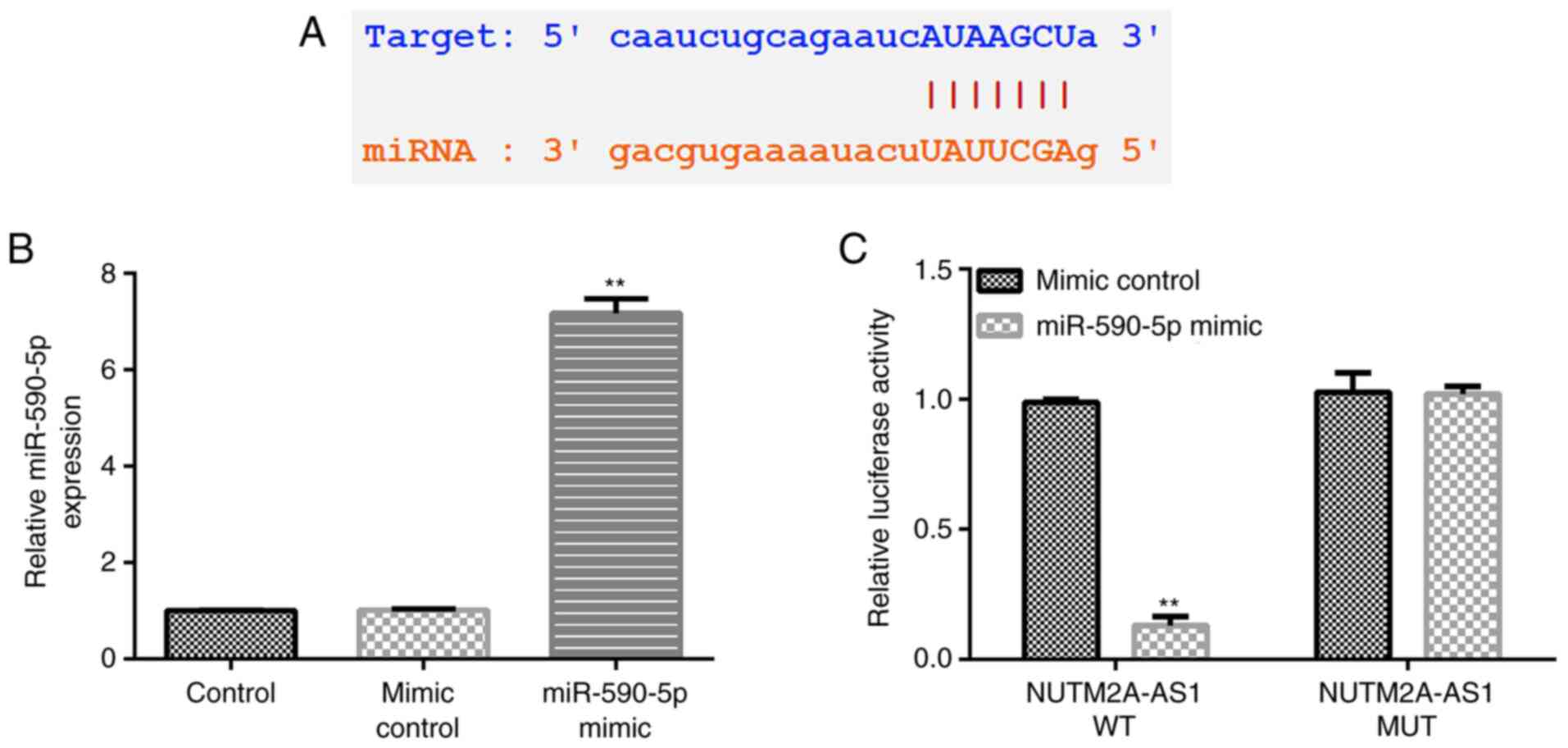
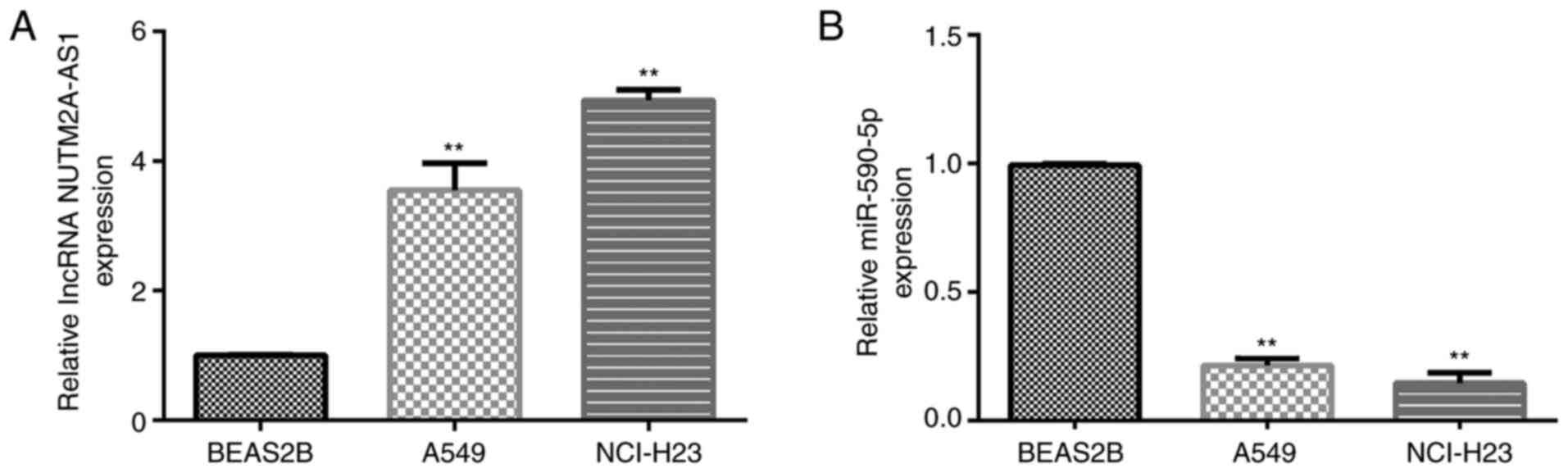
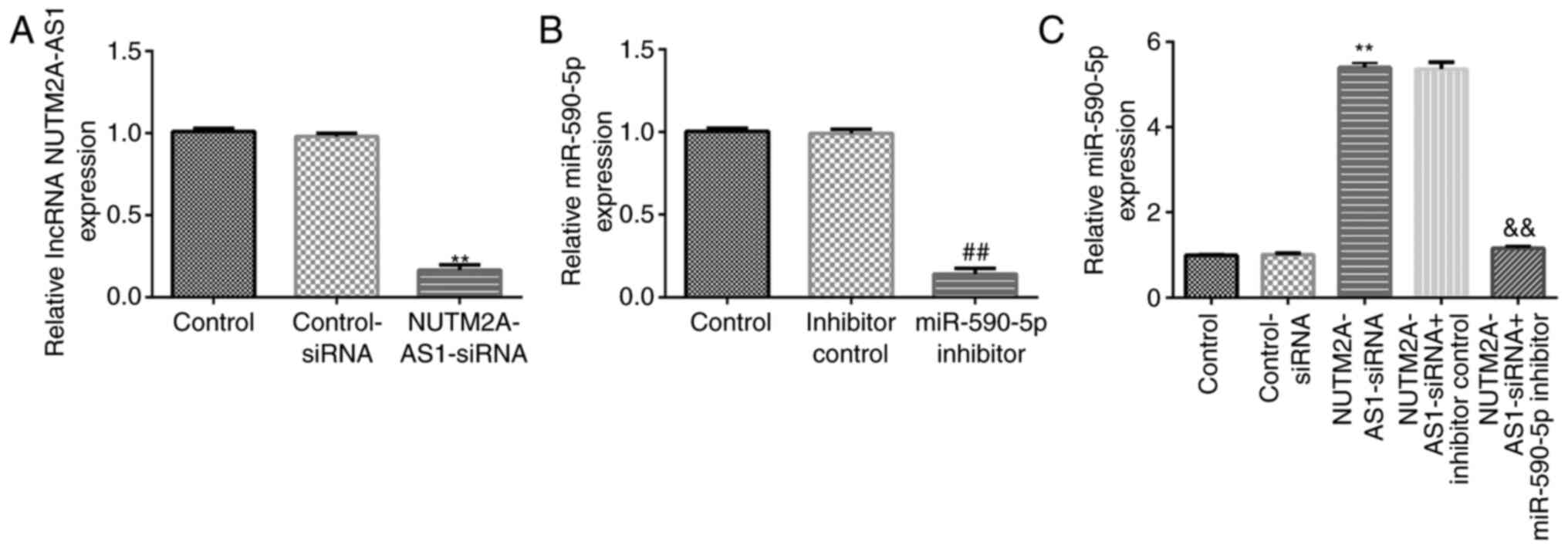
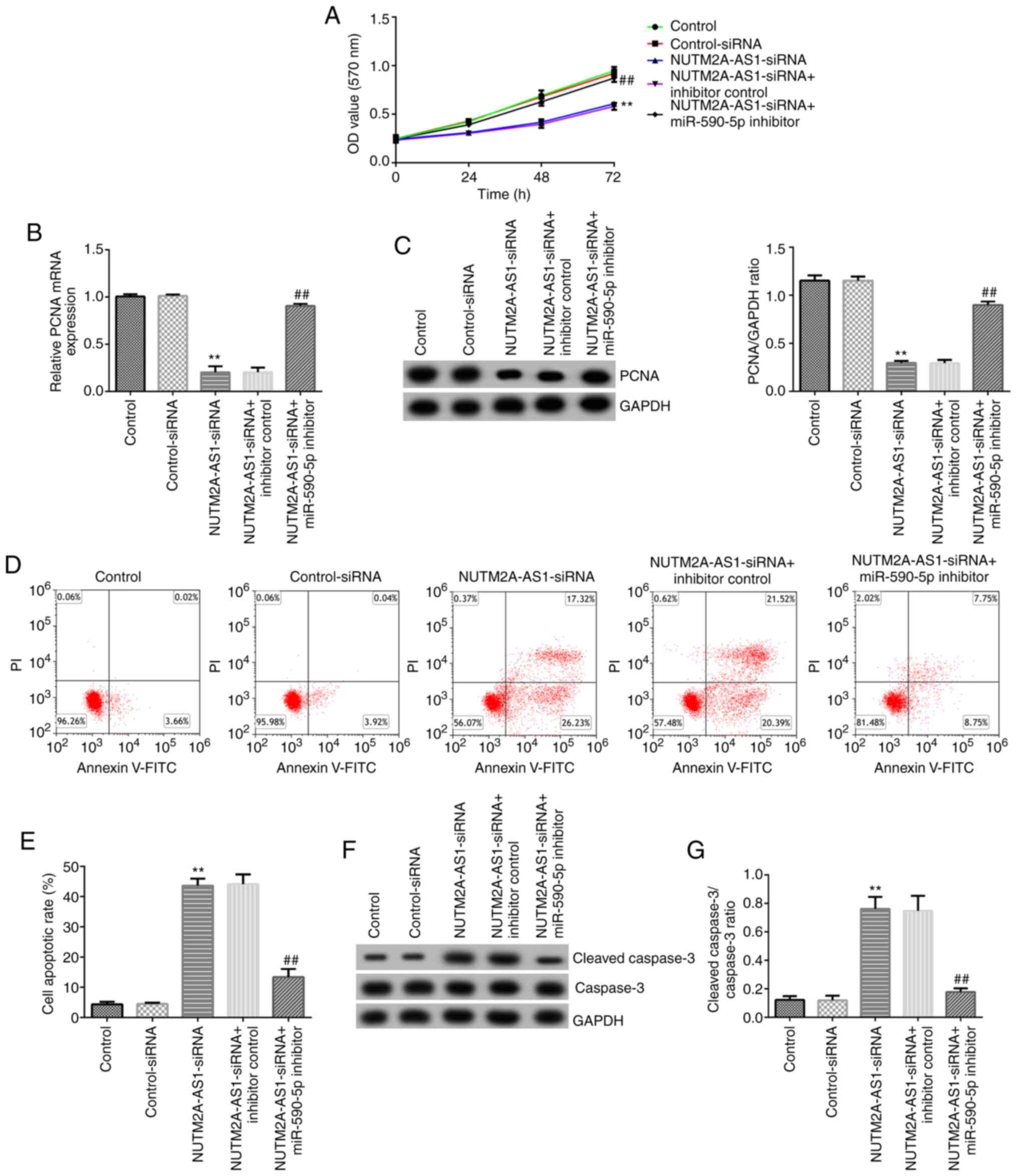
|
5
|
Cancer Genome Atlas Research Network, .
Comprehensive molecular profiling of lung adenocarcinoma. Nature.
511:543–550. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Lin JJ, Cardarella S, Lydon CA, Dahlberg
SE, Jackman DM, Jänne PA and Johnson BE: Five-year survival in
EGFR-mutant metastatic lung adenocarcinoma treated with EGFR-TKIs.
J Thorac Oncol. 11:556–565. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Peng FD, Wang R, Zhang Y, Zhao Z, Zhou W,
Chang Z, Liang H, Zhao W, Qi L, Guo Z and Gu Y: Differential
expression analysis at the individual level reveals a lncRNA
prognostic signature for lung adenocarcinoma. Mol Cancer.
16:982017. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Fatica A and Bozzoni I: Long non-coding
RNAs: New players in cell differentiation and development. Nat Rev
Genet. 15:7–21. 2014. View
Article : Google Scholar : PubMed/NCBI
|
|
9
|
Bhan A, Soleimani M and Mandal SS: Long
noncoding RNA and cancer: A new paradigm. Cancer Res. 77:3965–3981.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Li J, Meng H, Bai Y and Wang K: Regulation
of lncRNA and its role in cancer metastasis. Oncol Res. 23:205–217.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Acha-Sagredo A, Uko B, Pantazi P, Bediaga
NG, Moschandrea C, Rainbow L, Marcus MW, Davies MPA, Field JK and
Liloglou T: Long non-coding RNA dysregulation is a frequent event
in non-small cell lung carcinoma pathogenesis. Br J Cancer.
122:1050–1058. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Dehghanzadeh R, Jadidi-Niaragh F, Gharibi
T and Yousefi M: MicroRNA-induced drug resistance in gastric
cancer. Biomed Pharmacother. 74:191–199. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Zhao K, Cheng J, Chen BJ, Liu Q, Xu D and
Zhang Y: Circulating microRNA-34 family low expression correlates
with poor prognosis in patients with non-small cell lung cancer. J
Thorac Dis. 9:3735–3746. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Moretti F, D'Antona P, Finardi E, Barbetta
M, Dominioni L, Poli A, Gini E, Noonan DM, Imperatori A, Rotolo N,
et al: Systematic review and critique of circulating miRNAs as
biomarkers of stage I–II non-small cell lung cancer. Oncotarget.
8:94980–94996. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Khandelwal A, Seam RK, Gupta M, Rana MK,
Prakash H, Vasquez KM and Jain A: Circulating microRNA-590-5p
functions as a liquid biopsy marker in non-small cell lung cancer.
Cancer Sci. 111:826–839. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Wanna-Udom S, Terashima M, Lyu H, Ishimura
A, Takino T, Sakari M, Tsukahara T and Suzuki T: The m6A
methyltransferase METTL3 contributes to Transforming Growth
Factor-beta-induced epithelial-mesenchymal transition of lung
cancer cells through the regulation of JUNB. Biochem Biophys Res
Commun. 524:150–155. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Wang X, Feng J, Xue Y, Guan ZY, Zhang DL,
Liu Z, Gong Z, Wang Q, Huang JB, Tang C, et al: Structural basis of
N (6)-adenosine methylation by the METTL3-METTL14 complex. Nature.
534:575–578. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Shen C, Xuan B, Yan T, Ma Y, Xu P, Tian X,
Zhang X, Cao Y, Ma D, Zhu X, et al: m6A-dependent
glycolysis enhances colorectal cancer progression. Mol Cancer.
19:722020. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Li J, Han Y, Zhang H, Qian Z, Jia W, Gao
Y, Zheng H and Li B: The m6A demethylase FTO promotes the growth of
lung cancer cells by regulating the m6A level of USP7 mRNA. Biochem
Biophys Res Commun. 512:479–485. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Du M, Zhang Y, Mao Y, Mou J, Zhao J, Xue
Q, Wang D, Huang J, Gao S and Gao Y: MiR-33a suppresses
proliferation of NSCLC cells via targeting METTL3 mRNA. Biochem
Biophys Res Commun. 482:582–589. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Jin D, Guo J, Wu Y, Du J, Yang L, Wang X,
Di W, Hu B, An J, Kong L, et al: m6A mRNA methylation
initiated by METTL3 directly promotes YAP translation and increases
YAP activity by regulating the MALAT1-miR-1914-3p-YAP axis to
induce NSCLC drug resistance and metastasis. J Hematol Oncol.
12:1352019. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
He RZ, Jiang J and Luo DX: The functions
of N6-methyladenosine modification in lncRNAs. Genes Dis.
7:598–605. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Schmitz SU, Grote P and Herrmann BG:
Mechanisms of long noncoding RNA function in development and
disease. Cell Mol Life Sci. 73:2491–2509. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Khandelwal A, Bacolla A, Vasquez K and
Jain A: Long non-coding RNA: A new paradigm for lung cancer. Mol
Carcinog. 54:1235–1251. 2015. View
Article : Google Scholar : PubMed/NCBI
|
|
26
|
Nakagawa T, Endo H, Yokoyama M, Abe J,
Tamai K, Tanaka N, Sato I, Takahashi S, Kondo T and Satoh K: Large
noncoding RNA HOTAIR enhances aggressive biological behavior and is
associated with short disease-free survival in human non-small cell
lung cancer. Biochem Biophys Res Commun. 436:319–324. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Wu Y, Liu HB, Shi XF, Yao YW, Yang W and
Song Y: The long non-coding RNA HNF1A-AS1 regulates proliferation
and metastasis in lung adenocarcinoma. Oncotarget. 6:9160–9172.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Li N, Gao WJ and Liu NS: LncRNA BCAR4
promotes proliferation, invasion and metastasis of non-small cell
lung cancer cells by affecting epithelial-mesenchymal transition.
Eur Rev Med Pharmacol Sci. 21:2075–2086. 2017.PubMed/NCBI
|
|
29
|
Schmidt LH, Spieker T, Koschmieder S,
Schäffers S, Humberg J, Jungen D, Bulk E, Hascher A, Wittmer D,
Marra A, et al: The long noncoding MALAT-1 RNA indicates a poor
prognosis in non-small cell lung cancer and induces migration and
tumor growth. J Thorac Oncol. 6:1984–1992. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Wang J, Yu ZY, Wang J, Shen Y, Qiu JL and
Zhuang Z: LncRNA NUTM2A-AS1 positively modulates TET1 and HIF-1A to
enhance gastric cancer tumorigenesis and drug resistance by
sponging miR-376a. Cancer Med. 9:9499–9510. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Tay Y, Rinn J and Pandolfi PP: The
multilayered complexity of ceRNA crosstalk and competition. Nature.
505:344–352. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Carthew RW and Sontheimer EJ: Origins and
mechanisms of miRNAs and siRNAs. Cell. 136:642–655. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Zheng GD, Xu ZY, Hu C, Lv H, Xie HX, Huang
T, Zhang YQ, Chen GP, Fu YF and Cheng XD: Exosomal miR-590-5p in
serum as a biomarker for the diagnosis and prognosis of gastric
cancer. Front Mol Biosci. 8:6365662021. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Yan TY, Wang WH, Yang L, Chen KW, Chen R
and Han Y: Rich club disturbances of the human connectome from
subjective cognitive decline to Alzheimer's disease. Theranostics.
8:3237–3255. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Shen B, Yu S, Zhang Y, Yuan Y, Li XY,
Zhong J and Feng J: miR-590-5p regulates gastric cancer cell growth
and chemosensitivity through RECK and the AKT/ERK pathway. Onco
Targets Ther. 9:6009–6019. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Jiang XF, Xiang G, Wang Y, Zhang L, Yang
X, Cao L, Peng H, Xue P and Chen D: MicroRNA-590-5p regulates
proliferation and invasion in human hepatocellular carcinoma cells
by targeting TGF-β RII. Mol Cells. 33:545–551. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Cai W, Xu Y, Yin J, Zuo W and Su Z:
miR-590-5p suppresses osteosarcoma cell proliferation and invasion
via targeting KLF5. Mol Med Rep. 18:2328–2334. 2018.PubMed/NCBI
|
|
38
|
Mou K, Ding M, Han D, Zhou Y, Mu X, Liu W
and Wang L: miR-590-5p inhibits tumor growth in malignant melanoma
by suppressing YAP1 expression. Oncol Rep. 40:2056–2066.
2018.PubMed/NCBI
|
|
39
|
Jiang Z, Zhang J, Chen F and Sun Y:
MiR-148b suppressed non-small cell lung cancer progression via
inhibiting ALCAM through the NF-κB signaling pathway. Thorac
Cancer. 11:415–425. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Wang J, Wang Y, Sun D, Bu J, Ren F, Liu B,
Zhang S, Xu Z, Pang S and Xu S: miR-455-5p promotes cell growth and
invasion by targeting SOCO3 in non-small cell lung cancer.
Oncotarget. 8:1149562017. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Pang HY, Zheng YR, Zhao Y, Xiu XQ and Wang
JJ: miR-590-3p suppresses cancer cell migration, invasion and
epithelial-mesenchymal transition in glioblastoma multiforme by
targeting ZEB1 and ZEB2. Biochem Biophys Res Commun. 468:739–745.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Xu BB, Gu ZF, Ma M, Wang JY and Wang HN:
MicroRNA-590-5p suppresses the proliferation and invasion of
non-small cell lung cancer by regulating GAB1. Eur Rev Med
Pharmacol Sci. 22:5954–5963. 2018.PubMed/NCBI
|
|
43
|
Wang TY, Kong S, Tao M and Ju SQ: The
potential role of RNA N6-methyladenosine in Cancer progression. Mol
Cancer. 19:882020. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Zheng ZQ, Li ZX, Zhou GQ, Lin L, Zhang LL,
Lv JW, Huang XD, Liu RQ, Chen FP, He XJ, et al: Long noncoding RNA
FAM225A promotes nasopharyngeal carcinoma tumorigenesis and
metastasis by acting as ceRNA to sponge miR-590-3p/miR-1275 and
upregulate ITGB3. Cancer Res. 79:4612–4626. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Wei WW, Huo BS and Shi XL: miR-600
inhibits lung cancer via downregulating the expression of METTL3.
Cancer Manag Res. 11:1177–1187. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Zhang P, He Q, Lei Y, Li Y, Wen X, Hong M,
Zhang J, Ren X, Wang Y, Yang X, et al: m6A-mediated
ZNF750 repression facilitates nasopharyngeal carcinoma progression.
Cell Death Dis. 9:11692018. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Chao YH, Shang J and JI WD:
ALKBH5-m6A-FOXM1 signaling axis promotes proliferation
and invasion of lung adenocarcinoma cells under intermittent
hypoxia. Biochem Biophys Res Commun. 521:499–506. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Lin S, Choe J, Du P, Triboulet R and
Gregory R: The m(6)A methyltransferase METTL3 promotes translation
in human cancer cells. Mol Cell. 62:335–345. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Wang P, Doxtader KA and Nam Y: Structural
basis for cooperative function of Mettl3 and Mettl14
methyltransferases. Mol Cell. 63:306–317. 2016. View Article : Google Scholar : PubMed/NCBI
|