Introduction
Head and neck squamous cell carcinoma (HNSCC) is the
major pathological subtype of head and neck cancer (1). Every year, >600,000 new patients are
diagnosed worldwide, and 300,000 individuals succumb to the disease
(2). The methods for the diagnosis
and treatment of HNSCC have developed rapidly; however, the
prognosis of patients with HNSCC remains poor (3). Identifying biomarkers in body fluids is
considered important for the earlier diagnosis of HNSCC (4). For example, salivary IL-8 and −6 and
tumor necrosis factor (TNF) have been beneficial for the diagnosis
of oral squamous cell carcinoma (5).
However, the conclusions from different studies are still
incomplete (4). Therefore,
identifying novel biomarkers for HNSCC detection is important for
definitive and effective diagnosis leading to timely prevention,
precise treatment and alleviating the recurrence of HNSCC.
The COP9 signalosome (COPS) is an evolutionarily
conserved complex of 8 protein subunits (COPS1-8) (6). The known biochemical activity of the
COPS is to serve as the deneddylase to remove NEDD8 from a
neddylated cullin, in the cullin-RING-E3 ligases (CRLs) via
deneddylation, which is further enhanced by linking with the 9th
subunit, COPS9 (7,8). COPS regulates numerous cellular and
biological processes, such as embryonic development, cell cycle,
DNA damage repair, checkpoint repair control, signal transduction,
circadian rhythm, T-cell development and autophagy (9). COPS accomplishes these various
functions by controlling CRLs, the most prominent class of
E3-enzymes, which promote ubiquitination of a variety of regulatory
proteins subsequently targeted for proteasomal degradation
(10). In addition, the catalytic
center of the COPS is harbored in COPS5 (11).
The COPS subunits, which form the lid complex (19S)
of the 26S proteasome, are paralogous (12). The 19S complex can specifically
recognize ubiquitinated substrates and induce their degradation
(13). Given the characteristics of
the COPS, numerous tumor-associated genes have been identified,
including hypoxia inducible factor 1 (HIF1), p27, runt related
transcription factor 3 and p53 (14–17). The
oncoprotein Jab1/COPS5 is associated with the pathogenesis of
nasopharyngeal carcinoma (11).
COPS5 is overexpressed in thyroid carcinoma cell lines and
associated with low expression of p27 (18). Epidermal growth factor
receptor-extracellular regulated MAP kinase signaling pathway may
upregulate COPS6, which inhibited programmed cell death ligand-1
(PD-L1) degradation and subsequently maintains PD-L1 stability in
glioblastoma (19,20). In ultraviolet-irradiated cells, COPS1
is associated with both the DNA repair process and cell apoptosis
in HeLa cells (21). COPS3 is
associated with cell growth arrest mediated by myeloid leukemia
factor 1 and impairs the activation of p53, which promotes cell
proliferation and blocks constitutively photomorphogenic 1-mediated
p53 degradation (22). COPS2
overexpression upregulates the production of vascular endothelial
growth factor and promotes the stability of cyclin A (23). COPS4 deficiency facilitates the
downregulation of S-phase kinase associated protein 2, which is
associated with the degradation of p27 (24).
Compared with normal tissues, COPS5 and COPS6 have
both been found to be upregulated >40% at the mRNA level in
several types of human cancer tissues, including myeloma, lung
cancer, colon adenocarcinoma, breast cancer, glioblastoma and
leukemia (25). Using a proteome
microarray method, Yang et al (26) found that COPS2 expression was
significantly higher in the serum of patients with gastric cancer
compared with that in healthy individuals, and the mRNA expression
level of COPS2 was associated with poor prognosis. The
amplification of COPS3 was found in 31% of patients with
osteosarcoma and was significantly associated with the size of the
tumor (27). In addition, COPS3 is
upregulated in clear cell renal cell carcinoma tissues and cell
lines and patients with high expression of COPS3 have lower overall
survival (OS) rate (28). The mRNA
level of COPS8 is upregulated and predicts a poor clinical outcome
in patients with cutaneous melanoma (29). However, to the best of our best
knowledge, the association between the COPS subunits and HNSCC
remains unknown.
Therefore, the present study aimed to investigate
the association between the expression profiles of the COPS
subunits and HNSCC. It was found that the COPS subunits were highly
expressed in the HNSCC samples compared with that in normal
tissues, except for COPS9. High mRNA expression levels of COPS2,
COPS5, COPS6, COPS7A, COPS7B, COPS8 and COPS9 had prognostic
significance in patients with HNSCC. COPS5 and COPS6 could be
important for the process of HNSCC.
Materials and methods
Cell culture
The human tongue squamous cell carcinoma cell lines,
CAL27 and SCC25, were kindly provided by Professor Liang Jiang
(Tongji Hospital, Hubei, China). The CAL27 and SCC25 cell lines
were cultured in complete DMEM (Hyclone; Cytiva) containing 10% FBS
(Lonza Group, Ltd.) with 100 U/ml streptomycin and penicillin
(Thermo Fisher Scientific, Inc.). Both the cell lines were cultured
at 37°C in a humidified incubator with 5% CO2. The CAL27
and SCC25 cell lines were harvested using 0.25% trypsin-EDTA
(Thermo Fisher Scientific, Inc.) when it reached 80%
confluence.
Cell transfection
The CAL27 and SCC25 cell lines were transfected with
small interfering (si)RNA targeting COPS5
(5′-UUCUCAUACUGUCUUUCAGGUCUGAAAGACAGUAUGAGAAAA-3′) or COPS6
(5′-UUGAUUAUAUCAAUGACAGACCUGUCAUUGAUAUAAUCAAUG-3′) or a
non-specific control (50 nM) using Lipofectamine® 2000
transfection reagent (Thermo Fisher Scientific, Inc.) according to
the manufacturer's instructions. After cell transfection with siRNA
for 4 h at 37°C, the medium was replaced. After 48 h transfection,
cells were used for western blot analysis and wound healing assay.
After 24 or 48 h transfected, cell viability was tested.
Western blot analysis
Proteins from CAL27 and SCC25 cells were extracted
using RIPA lysate (Beyotime Biotechnology, China) containing 1×
protease inhibitor (Thermo Fisher Scientific, USA) at 80–90%
density. The protein concentration was determined by a BCA method
(Beyotime Institute of Biotechnology) according to the
manufacturer's instructions. Total protein (20 µg) was separated
using 10% SDS-PAGE and transferred to 0.2-µm PVDF membranes
(Bio-Rad Laboratories) at 100 V for 80 min. After being blocked
with TBS containing 0.1% Tween-20 (TBST) and 5% skimmed milk for 1
h at room temperature, the PVDF membrane was incubated with the
primary antibody overnight at 4°C. The next day, after being washed
3 times with TBST, the membrane was incubated with the goat
anti-mouse secondary antibody (cat. no. SA00001-1; 1:10,000;
ProteinTech Group, Inc.) for 1 h at room temperature, then the
bands were visualized using an ECL developing system (Beyotime
Institute of Biotechnology). The following antibodies were used:
Mouse monoclonal anti-COPS5 (cat. no. sc-13157; 1:200; Santa Cruz
Biotechnology, Inc.), mouse monoclonal anti-COPS6 (cat. no.
sc-137153; 1:200; Santa Cruz biotechnology, Inc.) and mouse
monoclonal anti-GAPDH (cat no. ab8245; 1:10,000; Abcam).
Cell viability
Following transfection with the siRNA, the cell
lines (1×104) were collected and seeded in 96-well
plates. The cells were then incubated with complete medium for 24
or 48 h and 10 µg Cell Counting Kit-8 reagent (MedChemExpress) was
added into each well. The cells were incubated in 37°C for 30 min,
then the samples were measured at 450 nm.
Wound healing assay
The CAL27 and SCC25 (1×105) cell lines
were seeded in 12-well plate. Then, cells were transfected with
siRNA or control. After the cells were cultured to 80–90%
confluence with complete DMEM medium with 10% FBS, the cells were
scratched slightly, then washed with PBS three times. Subsequently,
the cells were incubated with DMEM without FBS for 48 h and images
of the cells were captured with normal light microscope (BX51;
Olympus Corporation). The wound was quantified by ImageJ with the
Wound Healing Coherency Tool (version 1.8.0; national Institute of
Health).
Datasets and samples
The RNA expression profile and clinical data sets
were obtained from The Cancer Genome Atlas (TCGA) database
(https://gdc.cancer.gov), which included 520
patients with HNSCC and 44 adjacent normal samples. After excluding
the samples with survival times <30 days, missing clinical
information and data integration, a total of 490 samples were used
to perform the prognostic analysis.
mRNA profiles of the COPS
subunits
The ONCOMINE database (www.oncomine.org) is the world's largest oncogene chip
database and integrated data mining platform. A total of 11
datasets (Pyeon Multi-cancer, Sengupta Head-Neck, Toruner
Head-Neck, Schlingemann Head-Neck, Cromer Head-Neck, Ginos
Head-Neck, Giordano Thyroid, Peng Head-Neck, Ye Head-Neck, Vasko
Thyroid and He Thyroid) were included in the analysis (30–35). The
mRNA expression level of the COPS subunits in the clinical HNSCC
specimens were compared with that in adjacent normal controls,
using an unpaired Student's t-test. The fold change and P-value
thresholds were defined as 1.5 and 0.01, respectively. TCGA gene
expression data of the COPS subunits was determined using UALCAN
(http://ualcan.path.uab.edu) using the
‘TCGA-HNSC’ module, which provides systematical and personalized
analysis of TCGA database. University of California Santa Cruz
(UCSC) Xena (http://xenabrowser.net/) is a
comprehensive web analysis tool which provides customized analysis
methods of TCGA data. To evaluate different mRNA expression levels
of the COPS subunits and the TMN stages in primary HNSCC (n=765),
UCSC Xena was used with a one-way ANOVA analysis followed by
Bonferroni's post hoc test.
Prognosis evaluation of the COPS
subunits in HNSCC
The Kaplan-Meier plotter (www.kmplot.com) includes the 21 types of TCGA datasets
to evaluate the prognostic value of 54,000 genes in different types
of cancer. The association between mRNA expression levels of all
the COPS subunits and prognosis in patients with HNSCC was analyzed
using the Kaplan-Meier plotter. All the patients were divided into
two groups automatically using the best outcome setting. The hazard
ratio (HR) with 95% confidence intervals (CI) and log-rank were
obtained to evaluate the prognostic difference in patients with
HNSCC.
Protein-protein interaction (PPI)
network of the COPS subunits
To construct the PPI network of the COPS subunits,
Search Tool for the Retrieval of Interacting Genes/Proteins
(STRING; http://string-db.org/), which includes
both certified and predicated links, was used. In total, 10 COPS
subunits (COPS1-6, 7A and B, 8 and 9) and Homo sapiens
(organism) were selected for the analysis. Extra 50 associated
nodes were included in the analysis.
Gene ontology (GO) enrichment
analysis
GO enrichment analysis was performed using The
Database for Annotation, Visualization and Integrated Discovery
(DAVID; http://david.ncifcrf.gov/). The
catalogues of GO analysis were selected as biological process (BP),
cellular components (CC), molecular function (MF) and Kyoto
Encyclopedia of Genes and Genomes (KEGG) pathway analysis was also
performed. All associated genes (36) were included in the analysis.
P<0.05 was considered as significant. The top 12 affected
pathways were included using R version 4.1.0. (R Development Core
Team).
Cell survival analysis
To evaluate the importance of the COPS subunits in
survival of the HNSCC cell lines, Project Achilles with CERES
dependence scores was used. By accurately removing the expression
of the corresponding gene with CRISPR-Cas9 system, Project Achilles
systematically investigates and identifies the numbers of genes
which are essential for survival of the cancer cell lines. The
original data was downloaded from the Depmap portal (https://depmap.org/portal) (37). The inclusive criteria were as follow:
i) The primary disease was head and neck cancer and ii) the
pathological subtype was squamous cell carcinoma. A total of 21
cell lines were included in the analysis.
Gene set enrichment analysis (GSEA) of
COPS subunits
GSEA (http://www.broad.mit.edu/gsea), with the annotated
Hallmark effector gene sets, was performed according to the mRNA
expression level of the COPS subunits in TCGA-HNSC dataset. The
cut-off value of grouping was defined as 50%. The false discovery
rate q-value <0.25 and P<0.05 was considered to indicate a
significant difference (38). The
visual results were performed using R version 4.1.0.
Protein expression of the COPS
subunits
The protein expression of COPS subunits was
evaluated using immunohistochemistry (IHC) data downloaded from the
Human Protein Atlas (HPA) database (https://www.proteinatlas.org/). The following primary
antibodies against CCT5 and CCT6 were used CAB004242 and
HPA044315.
Statistical analysis
The HR with log-rank test was used for survival
analysis. Spearman's rank correlation test was used to evaluate the
correlation of gene expression in tumor tissues compared with
normal tissues, and the strength of the association was determined
using the absolute values. One-way ANOVA followed by Bonferroni
post hoc test was used to evaluate the difference in >2 groups.
P<0.05 was considered to indicate a statistically significant
difference.
Results
mRNA expression profiles of the COPS
subunits in HNSCC
A total 10 COPS subunits (COPS1-9) have been
identified in human cells (39,40). To
evaluate the mRNA expression levels of the COPS subunits in HNSCC,
TCGA dataset (TCGA-HNSC) was used within the UALCAN database. As
shown in Fig. 1, the expression of
COPS1-8, but not COPS9, was found to be upregulated in HNSCC
tissues compared with that in normal tissues.
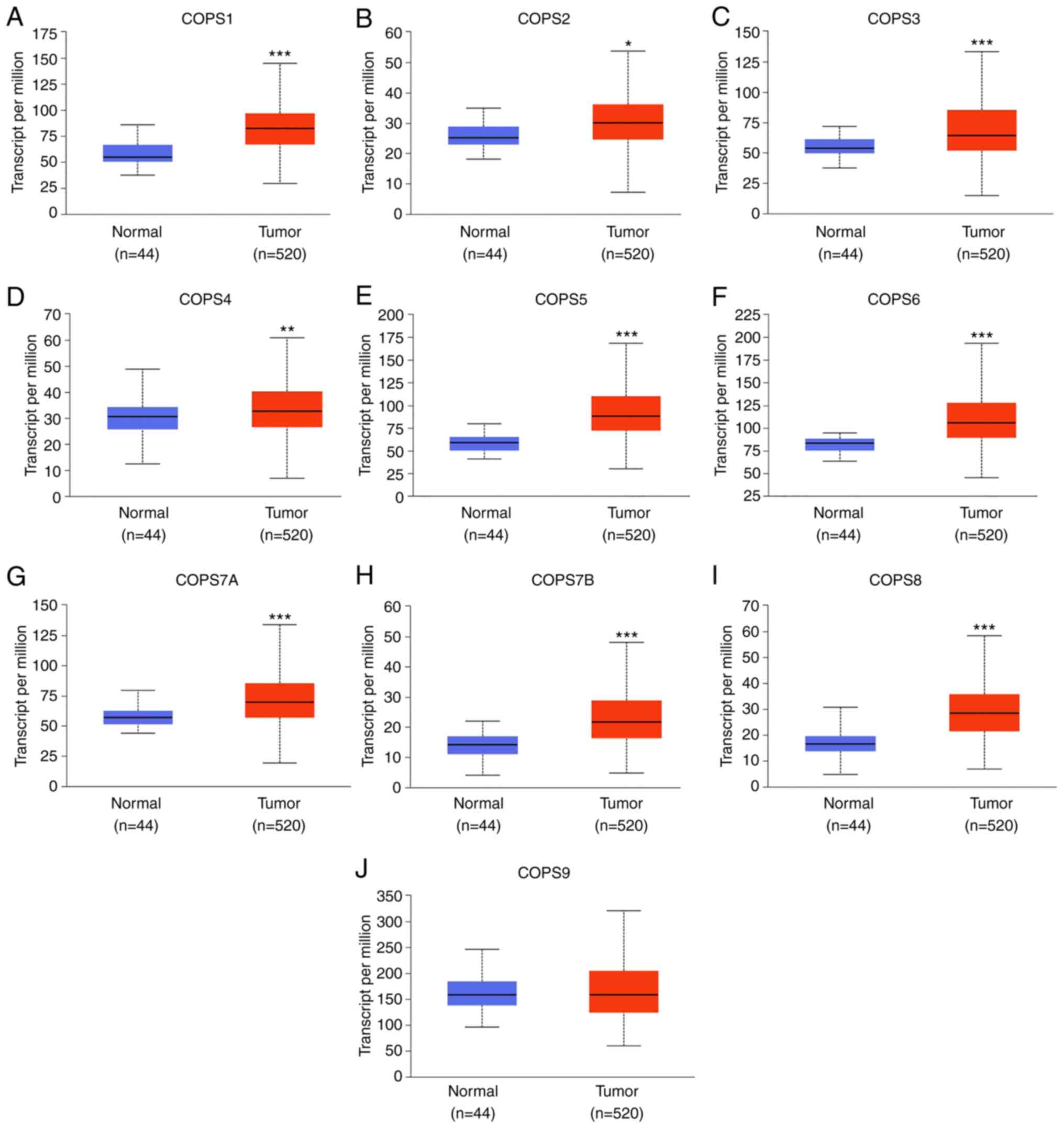 | Figure 1.Expression level of the COPS subunits
was increased in the HNSCC samples. The mRNA expression profiles of
(A) COPS1, (B) COPSS2, (C) COPS3, (D) COPS4, (E) COPS5, (F) COPS6,
(G) COPS7A, (H) COPS7B, (I) COPS8 and (J) COPS9 in HNSCC and normal
tissue samples were obtained from the UALCAN database. *P<0.05.
**P<0.01. ***P<0.001. COPS, COP9 signalosome; HNSCC, head and
neck squamous cell carcinoma. |
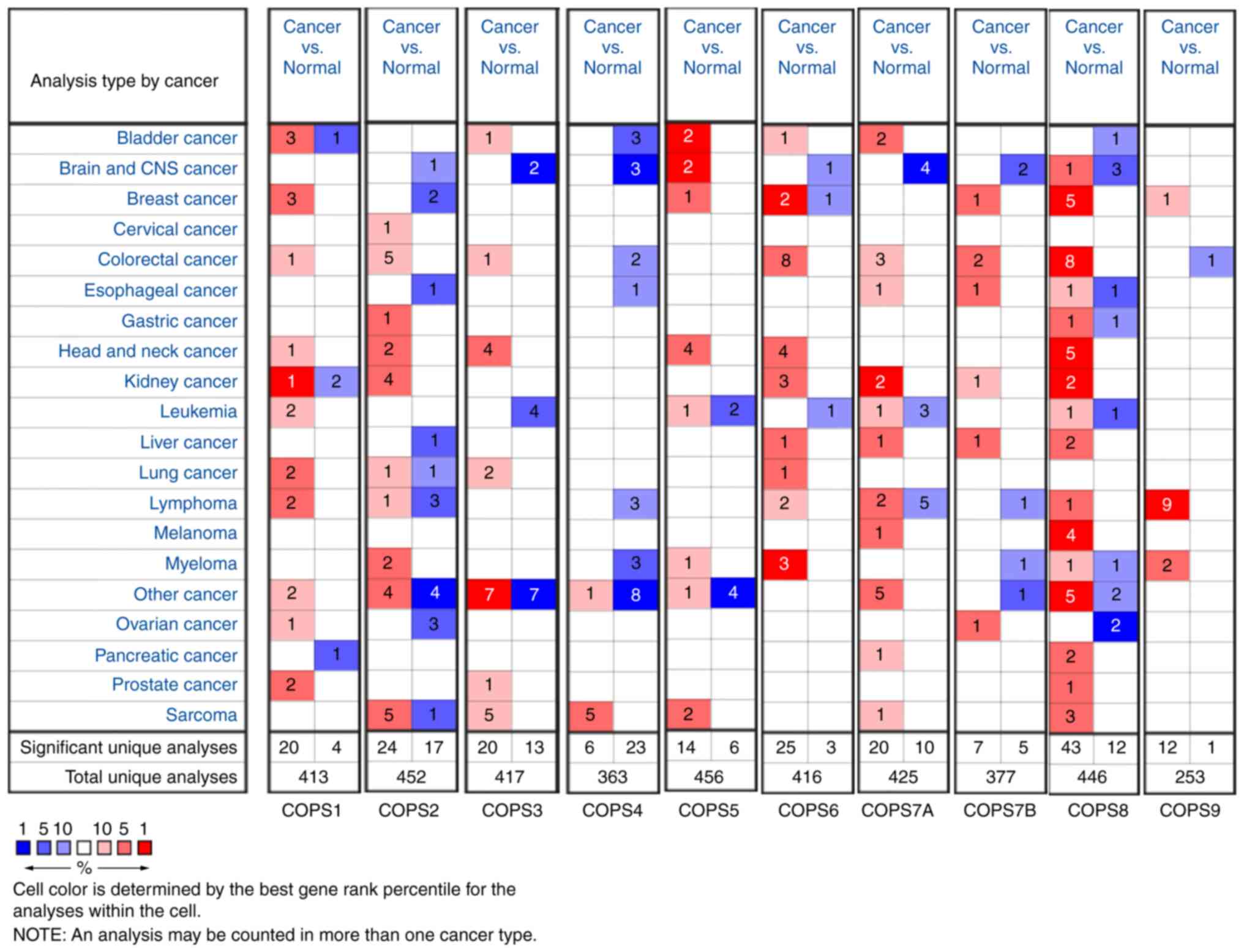
To further verify the results, the ONCOMINE database
was used to compare the transcriptional levels of the COPS subunits
in all types of cancer with the corresponding normal tissue
(Fig. 2). The red color and number
indicated the number of datasets with a statistically significant
increase (P<0.01; fold change, 1.5) in mRNA expression levels in
different types of cancers, while the blue color indicated a
decrease in mRNA expression of the COPS subunits. There was a total
of 11 HNSCC datasets in the ONCOMINE database. The overexpression
status of COPS subunits in different parts of HNSCC are presented
in Table I. In detail, one dataset
indicated COPS1 was upregulated in floor of the mouth carcinoma,
and COPS3 was increased in carcinoma from oropharyngeal, oral
cavity, floor of the mouth and tongue (Pyeon Multi-cancer)
(30). The expression of COPS2 was
found increased in both NPC (Sengupta Head-Neck) (31) and floor of the mouth carcinoma (Pyeon
Multi-cancer) (30). COPS5 (Pyeon
Multi-cancer, Sengupta Head-Neck and Toruner Head-Neck Statistics)
(30,31,34) and
COPS6 (Pyeon Multi-cancer) (30)
were upregulated in various sites of HNSCC. In addition, COPS8 was
found highly expressed in HNSCC tissues in five datasets (Pyeon
Multi-cancer, Sengupta Head-Neck, Schlingemann Head-Neck, Cromer
Head-Neck and Ginos Head-Neck) (30–33,35).
There was no significant difference in the expression of COPS7A,
COPS7B and COPS9 between HNSCC and normal tissues in the ONCOMINE
database.
 | Table I.Expression of the COPS families in
head and neck squamous cell carcinoma. |
Table I.
Expression of the COPS families in
head and neck squamous cell carcinoma.
| Gene name | Cancer type
(n) | P-value | Fold change | T-test | (Refs.) |
|---|
| COPS1 | Floor of the mouth
carcinoma (5) |
4.00×10−3 | 1.543 | 3.837 | Pyeon Multi-cancer
(50) |
| COPS2 | Nasopharyngeal
carcinoma (31) |
2.80×10−6 | 2.095 | 5.746 | Sengupta Head-Neck
(36) |
|
| Floor of the mouth
carcinoma (5) |
2.07×10−4 | 2.261 | 5.383 | Pyeon Multi-cancer
(50) |
| COPS3 | Oropharyngeal
carcinoma (6) | 3.32
×10−4 | 2.295 | 4.225 | Pyeon Multi-cancer
(50) |
|
| Oral cavity
carcinoma (4) |
3.00×10−3 | 2.206 | 3.717 | Pyeon Multi-cancer
(50) |
|
| Floor of the mouth
carcinoma (5) |
2.43×10−4 | 2.636 | 4.748 | Pyeon Multi-cancer
(50) |
|
| Tongue carcinoma
(15) |
2.01×10−4 | 2.040 | 3.913 | Pyeon Multi-cancer
(50) |
| COPS5 | Tongue carcinoma
(15) |
3.88×10−4 | 2.755 | 6.009 | Pyeon Multi-cancer
(50) |
|
| Floor of the mouth
carcinoma (5) |
1.71×10−6 | 4.533 | 8.121 | Pyeon Multi-cancer
(50) |
|
| Oral cavity
carcinoma (4) | 2.26
×10−4 | 2.401 | 4.971 | Pyeon Multi-cancer
(50) |
|
| Tonsillar carcinoma
(6) |
9.52×10−4 | 1.719 | 3.509 | Pyeon Multi-cancer
(50) |
|
| Oropharyngeal
carcinoma (6) |
1.99×10−4 | 2.762 | 4.910 | Pyeon Multi-cancer
(50) |
|
| Oral cavity
squamous cell carcinoma (16) |
3.88×10−4 | 1.823 | 4.826 | Toruner Head-Neck
Statistics (53) |
|
| Nasopharyngeal
carcinoma (31) |
1.17×10−4 | 1.595 | 4.233 | Sengupta Head-Neck
Statistics (36) |
| COPS6 | Tonsillar carcinoma
(6) |
3.01×10−4 | 1.924 | 3.915 | Pyeon Multi-cancer
(50) |
|
| Oral cavity
carcinoma (4) |
2.00×10−3 | 2.634 | 3.902 | Pyeon Multi-cancer
(50) |
|
| Oropharyngeal
carcinoma (6) |
4.81×10−4 | 2.118 | 3.820 | Pyeon Multi-cancer
(50) |
|
| Floor of the mouth
carcinoma (5) |
1.49×10−4 | 2.665 | 4.656 | Pyeon Multi-cancer
(50) |
| COPS8 | Hypopharyngeal
squamous cell carcinoma (4) |
5.00×10−3 | 2.533 | 3.794 | Schlingemann
Head-Neck (52) |
|
| Head and neck
squamous cell carcinoma (34) |
6.70×10−4 | 2.001 | 5.820 | Cromer Head-Neck
(52) |
|
| Head and neck
squamous cell carcinoma (41) |
3.48×10−9 | 1.617 | 7.125 | Ginos Head-Neck
(54) |
|
| Oropharyngeal
carcinoma (6) |
1.21×10−4 | 2.021 | 4.791 | Pyeon Multi-cancer
(50) |
|
| Nasopharyngeal
carcinoma (31) |
2.47×10−4 | 1.523 | 3.957 | Sengupta Head-Neck
(36) |
mRNA expression levels of the COPS
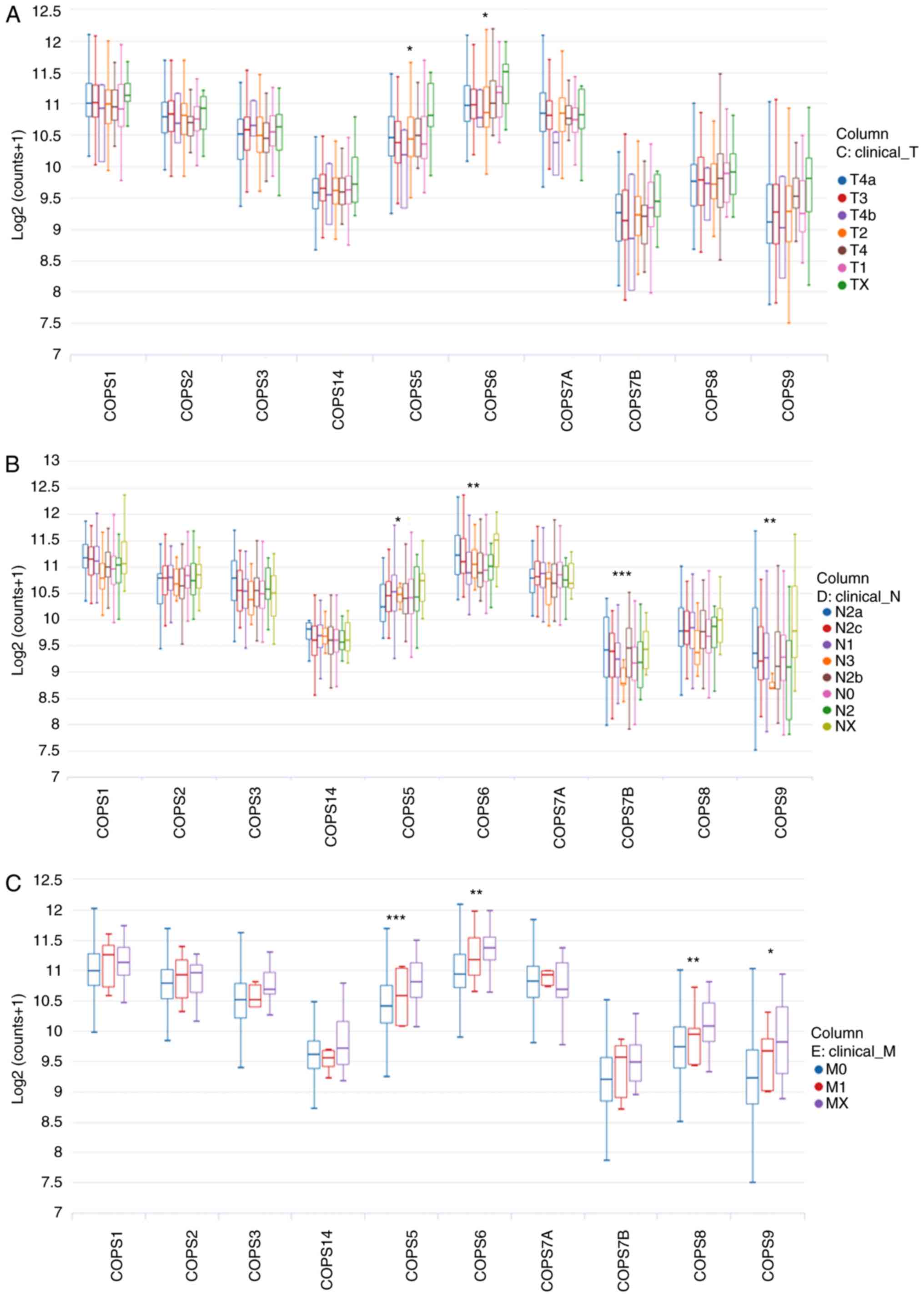
subunits are associated with TNM stage in patients with HNSCC
As most of the COPS subunits were found to be
upregulated in HNSCC tissues, the association between the mRNA
expression level of the COPS subunits and TNM stage was
investigated. The UCSC Xena web tool was used for the analysis. As
shown in Fig. 3A, the mRNA
expression levels of COPS5 and COPS6 were significantly increased
with increasing tumor size. With respect to lymph node metastasis,
an increase in the expression level of COPS5, COPS6, COPS7B and
COPS9 were found in patients with HNSCC and N stage (Fig. 3B). In addition, increased expression
of COPS5, COPS6, COPS8 and COPS9 was associated with distant
metastasis of HNSCC (Fig. 3C).
Prognostic values of the COPS subunits
in patients with HNSCC
The Kaplan-Meier plotter database and TCGA-HNSC
dataset was used to investigate whether the COPS subunits were
associated with the prognosis of HNSCC. High mRNA expression level
of COPS2 (HR, 1.44; CI, 1.09–1.9; P=0.0091; Fig. 4B), COPS5 (HR, 1.32; CI, 1.01–1.73;
P=0.042; Fig. 4E), COPS6 (HR, 1.4;
CI, 1.06–1.84; P=0.019; Fig. 4F),
COPS7A (HR, 1.56 (1.19–2.05); P=0.0011; Fig. 4G), COPS8 (HR, 1.6; CI, 1.16–2.19;
P=0.0035; Fig. 4I) and COPS9 (HR,
1.33; CI, 1.02–1.74; P=0.035; Fig.
4J) was associated with poor prognosis in patients with HNSCC.
However, high expression of COPS7B (HR, 0.75 (0.57–0.98); P=0.038;
Fig. 4H) was associated with
improved outcome in patients with HNSCC.
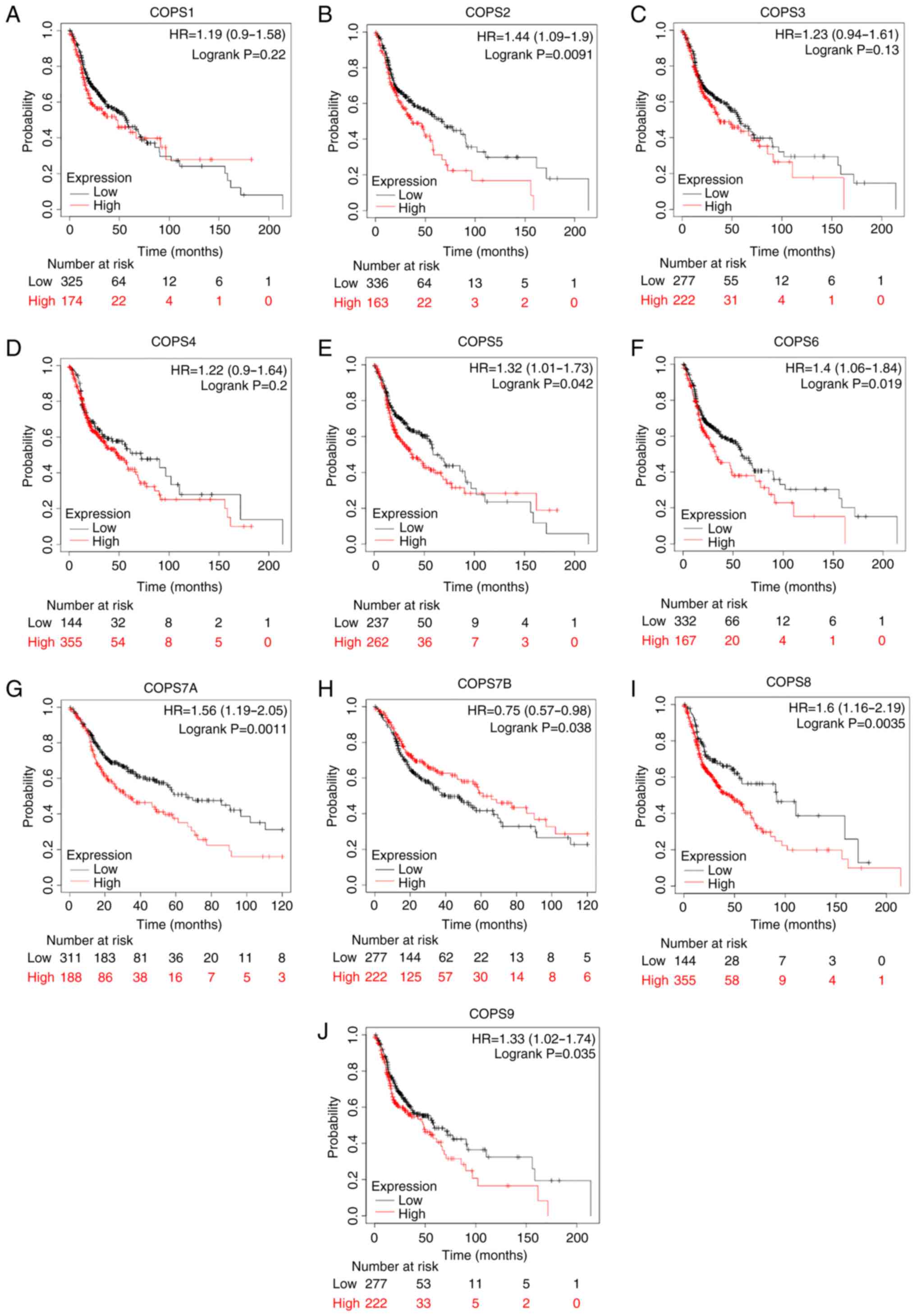 | Figure 4.COPS subunit expression and
prognostic value in patients with HNSCC. The prognostic value of
(A) COPS1, (B) COPS2, (C) COPS3, (D) COPS4, (E) COPS5, (F) COPS6,
(G) COPS7A, (H) COPS7B, (I) COPS8 and (J) COPS9 in the head and
neck squamous cell carcinoma tissues was determined using the
Kaplan-Meier plotter database and the mRNA expression profiles.
COPS, COP9 signalosome; HR, hazard ratio. |
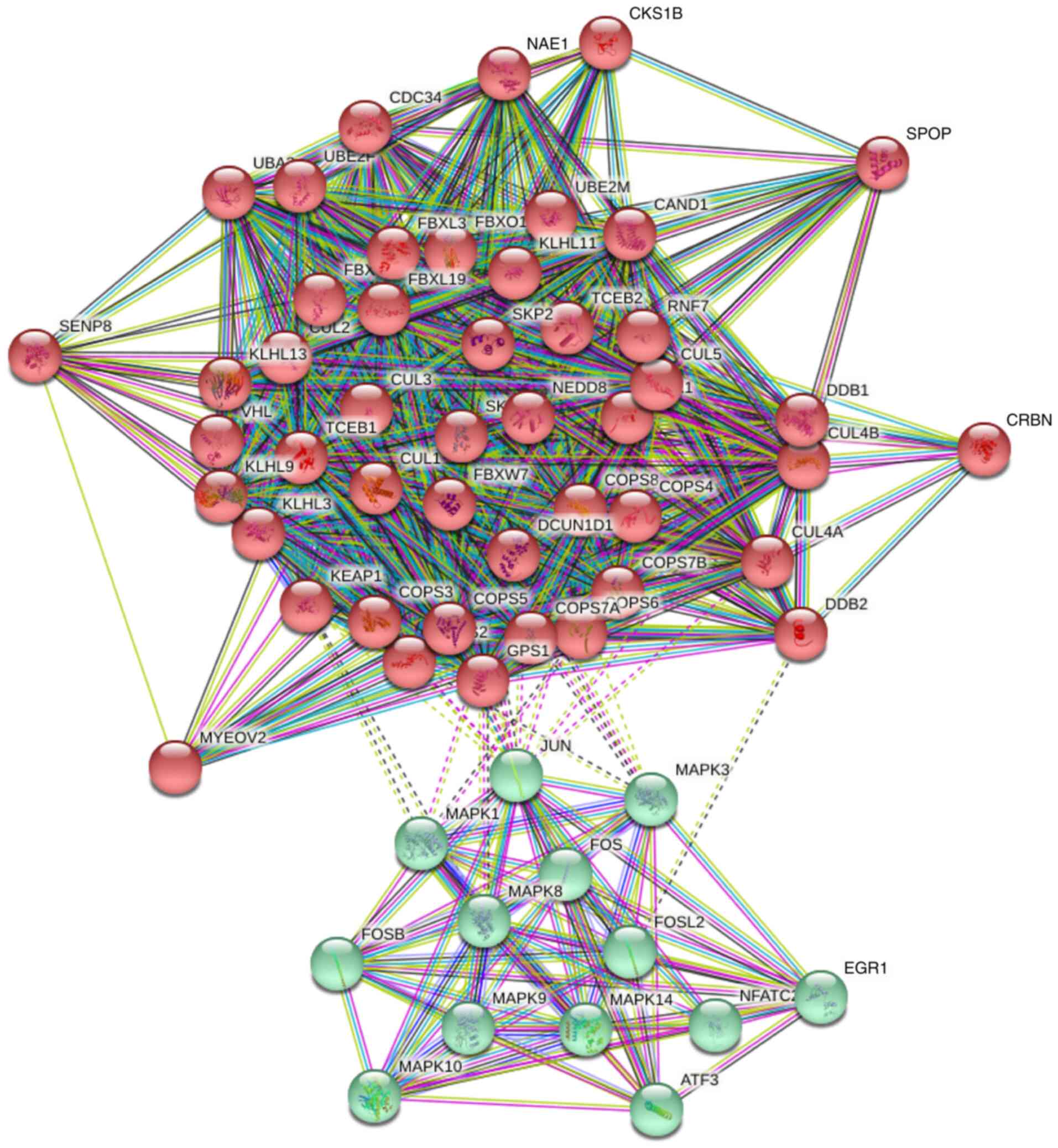
PPI network construction and GO
analysis of the COPS subunits
To predict the functions of the COPS subunits in
humans, a PPI network was constructed with the 50 most relevant
genes using STRING. The analysis included 60 nodes and 946 edges
(Fig. 5). The PPI enrichment value
was P<1.0×10−16.
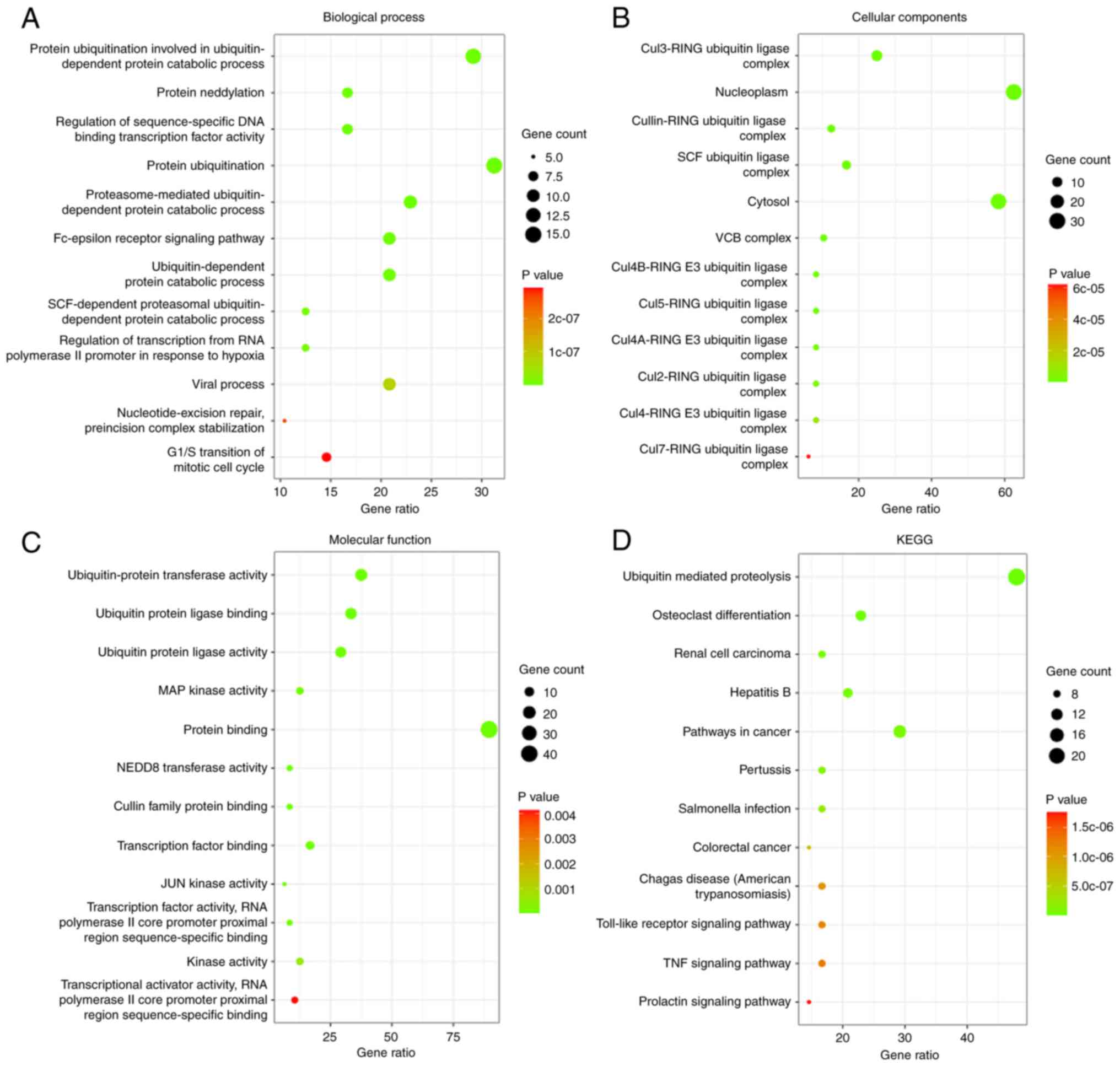
Then, the biological functions and pathways of the
associated genes with the COPS subunits were analyzed using GO and
KEGG analysis, respectively. The top 12 GO terms and KEGG pathways
are shown in Fig. 6. With respect to
BP, ‘protein ubiquitination involved in ubiquitin-dependent protein
catabolic process’, ‘Fc-epsilon receptor signaling pathway’,
‘nucleotide-excision repair, preincision complex stabilization’ and
‘G1/S transition of mitotic cell cycle’ were
significantly associated with the COPS subunits (Fig. 6A; Table
SI). For CC, the genes were associated with ‘Cul3-RING
ubiquitin ligase complex’, ‘nucleoplasm’ and ‘cytosol’ (Fig. 6B; Table
SII). The genes associated with MF were ‘ubiquitin-protein
transferase activity’, ‘MAP kinase activity’, ‘NEDD8 transferase
activity’ and ‘JUN kinase activity’, which were cancer relative
pathways (Fig. 6C; Table SIII). KEGG pathway analysis
identifies the functions of the COPS subunits and the pathways they
are involved in. The top 12 pathways are shown in Fig. 6D and Table SIV. Among these pathways, ‘ubiquitin
mediated proteolysis’, ‘renal cell carcinoma’, ‘pathways in
cancer’, ‘colorectal cancer’, ‘Toll-like receptor signaling
pathway’ and ‘TNF signaling pathway’ were observed.
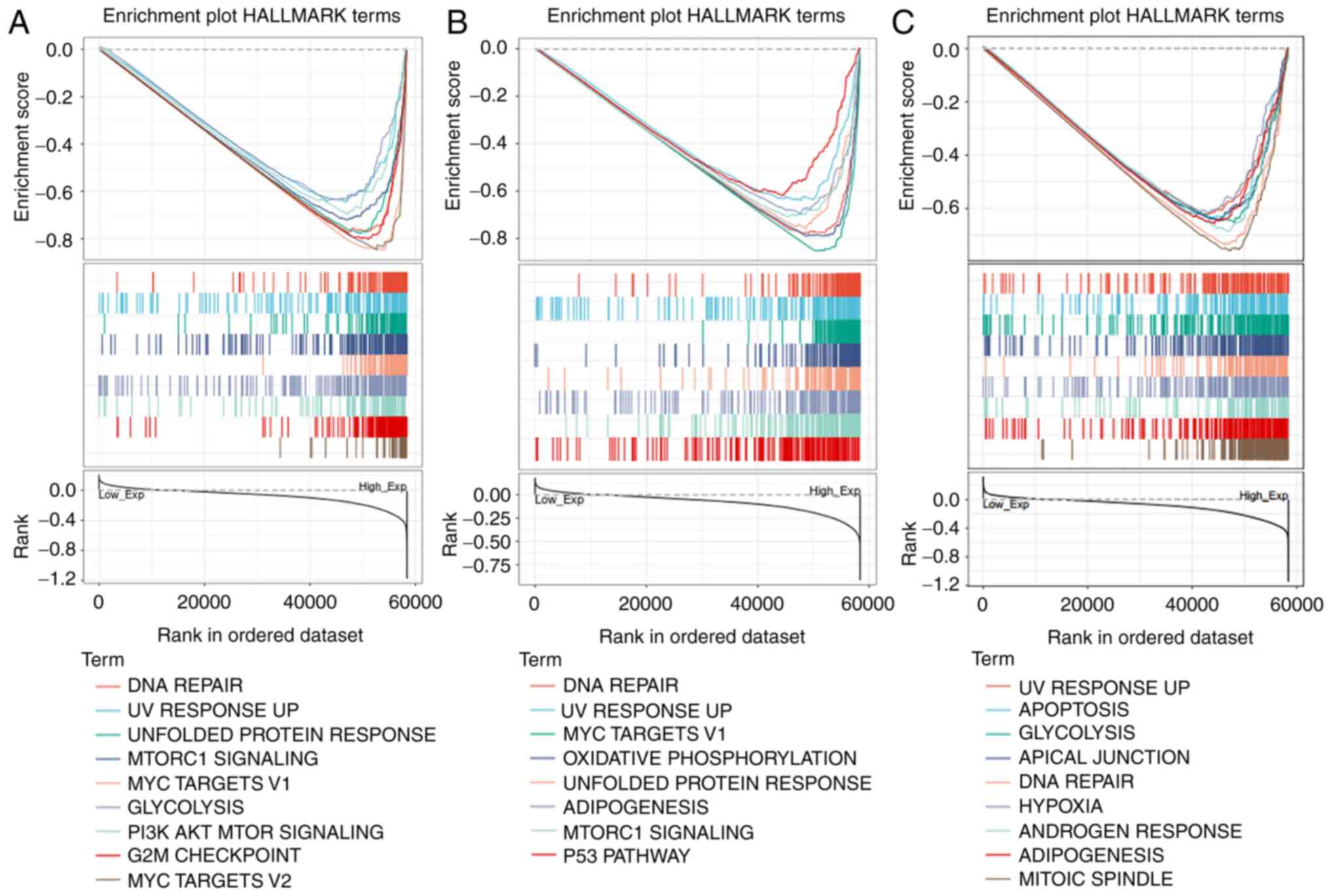
GSEA analysis of COPS5, COPS6 and
COPS8
In HNSCC, COPS5, COPS6 and COPS8 are essential for
cell growth and, as aforementioned, associated with tumor TNM
stages and prognosis. Hence, it is important to evaluate the
functions of these three genes and GSEA was performed to evaluate
Hallmark annotation. As shown in Fig.
7A and Table SV, high
expression of COPS5 was associated with tumorigenesis-associated
pathway including ‘DNA repair’, ‘unfolded protein response’,
‘MTORC1 signaling’, ‘glycolysis’ and ‘PI3K AKT mTOR signaling’. In
addition to these pathways, increased COPS6 was also associated
with ‘oxidative phosphorylation’ and ‘p53 pathway’ (Fig. 7B and Table SVI). Similarly, COPS8 was
additionally associated with ‘apoptosis’ (Fig. 7C and Table SVII).
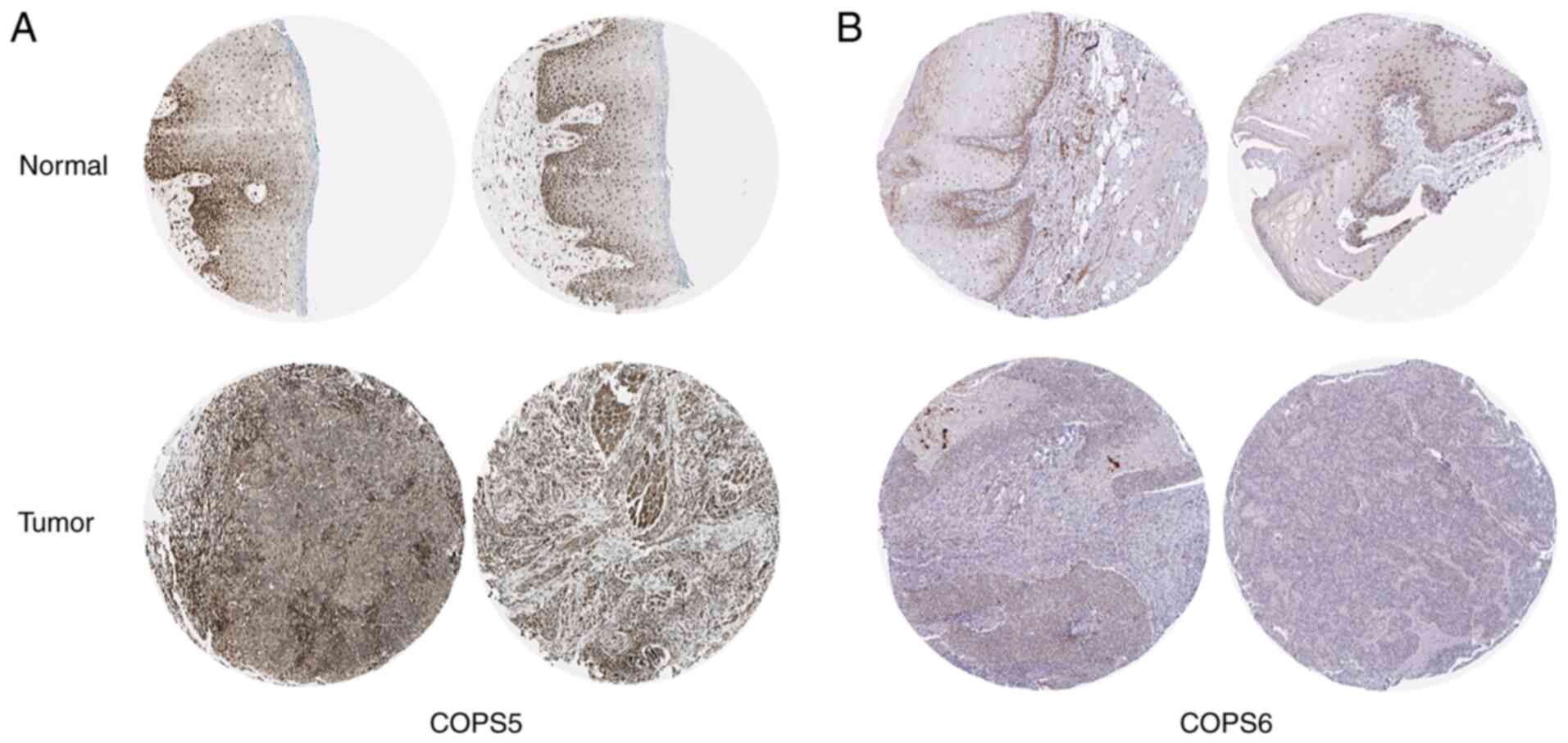
IHC analysis of the protein expression
level of the COPS subunits in HNSCC
The protein expression level of the COPS subunits in
HNSCC was investigated using the HPA database. Consistent with the
mRNA expression level, COPS2 (Fig.
S1A), COPS3 (Fig. S1B), COPS4
(Fig. S1C), COPS5 (Fig. 8A), COPS6 (Fig. 8B), COPS7A (Fig. S1D) and COPS7B (Fig. 1E) were notably upregulated in both
the cytoplasm and the nucleus in the HNSCC tissues compared with
that in normal oral epithelial tissues. COPS9 was increased in the
normal oral epithelial tissue compared with that in the tumor
tissues of HNSCC (Fig. S1F). COPS1
and COPS8 were not detected.
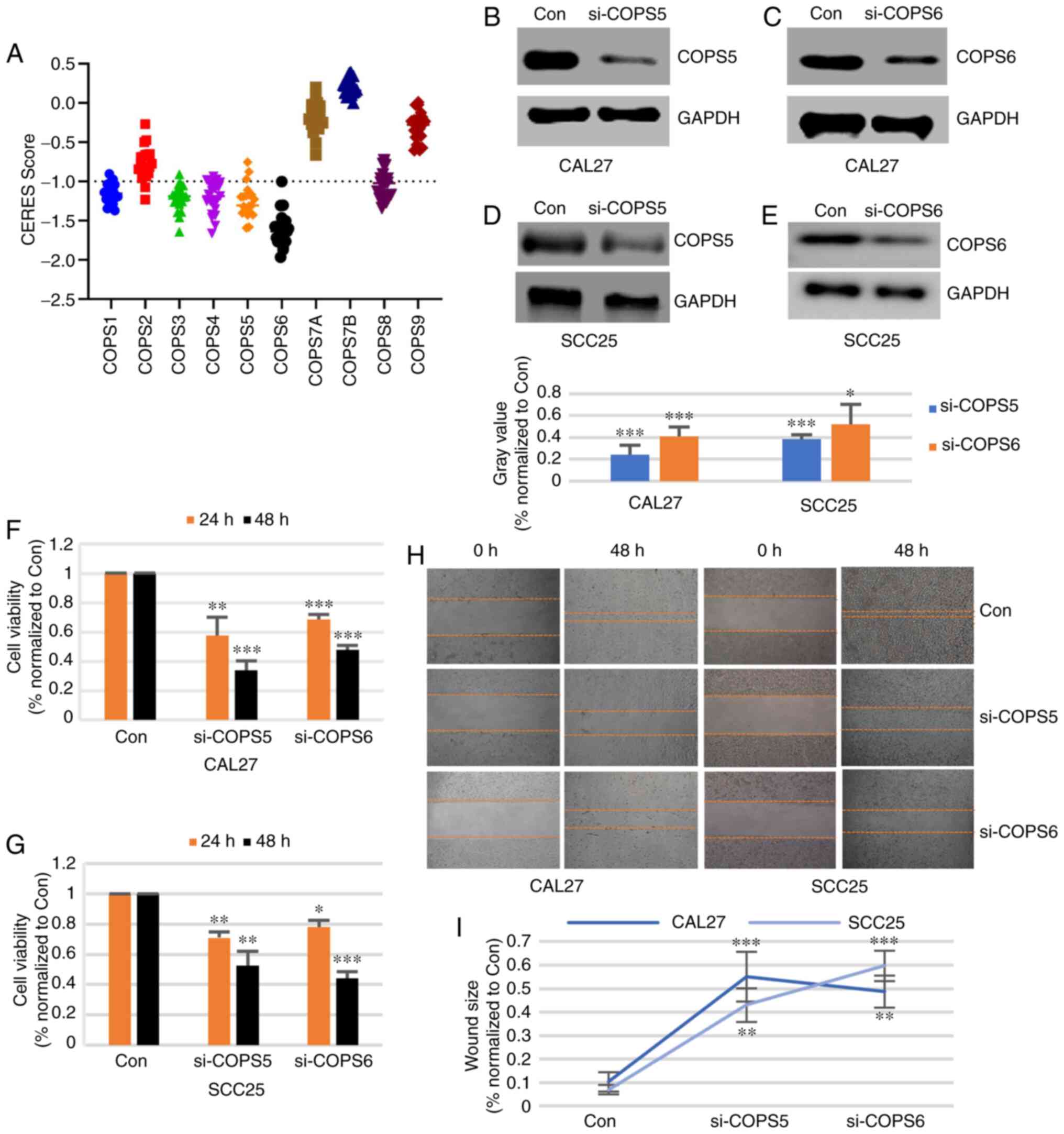
Role of COPS subunits in the survival
of the HNSCC cell lines
To evaluate the importance of the COPS subunits,
Project Achilles with CERES dependence scores was used. When the
score was >-1, it indicated that the gene was not essential for
cell survival. Otherwise, the gene was considered important for
cell survival. As shown in Fig. 9A
and Table SVIII among all the HNSCC
cell lines, COPS6 was important for cell survival. COPS1, COPS3,
COPS4, COPS5 and COPS8 were critical for cell survival in most
HNSCC cell lines. COPS2 was significantly essential for only 2
HNSCC cell lines. COPS7A, COPS7B and COPS9 were not important for
HNSCC cell survival. These results suggested that COPS1, COPS3,
COPS4, COPS5, COPS6 and COPS8 are essential for HNSCC cell
survival.
COPS5 and COPS6 are essential for cell
growth and invasion of HNSCC cells
Taken together, COPS5 and COPS6 were associated with
the survival of the HNSCC cells and could be detected in tumor
samples using IHC. To confirm the biological function of COPS5 and
COPS6 in HNSCC, the CAL27 and SCC25 cell lines were transfected
with a control siRNA, si-COPS5 and si-COPS6. Western blot analysis
showed that the protein expression level of COPS5 (Fig. 9B and D) and COPS6 (Fig. 9C and E) was notably decreased by the
siRNA. As shown in Fig. 9F and G,
the cell viabilities of the CAL27 and SCC25 cell lines were
significantly decreased by both si-COPS5 and si-COPS6 at 24 and 48
h time points (P<0.05). To detect the cell migration effect, the
CAL27 and SCC25 cell lines were transfected with control siRNA,
si-COPS5 or si-COPS6. The results from the wound healing assay
suggested that si-COPS5 and si-COPS6 inhibited the migration of the
CAL27 and SCC25 cells (Fig. 9H and
I). These results suggested that COPS5 and COPS6 could be
essential for growth and migration of the HNSCC cell lines.
Discussion
In the present study, ONCOMINE and TCGA databases
were analyzed and it was found that the COPS subunits were
upregulated in HNSCC samples compared with that in normal tissues,
except for COPS9. Using subgroup analysis, based on TNM stage, high
mRNA expression level of COPS5, COPS6, COPS7B, COPS8 and COPS9 was
associated with progression of HNSCC. Prognosis analysis, using
Kaplan-Meier plotter and TCGA-HNSC dataset suggested that mRNA
expression level of COPS2, COPS5, COPS6, COPS7A, COPS7B, COPS8 and
COPS9 could have prognostic significance. Survival analysis in the
HNSCC cell lines suggested that COPS1, COPS3, COPS4, COPS5, COPS6
and COPS8 were necessary for tumor cell survival. COPS5 and COPS6
were also found to be essential for growth and migration of the
HNSCC cell lines. Taken together, the COPS subunits may be
potential diagnostic biomarkers in the future, and COPS5 and COPS6
could be important genes in the progression of HNSCC.
COP9 is a component of a large, multi-subunit
nuclear protein complex (39). As
COPS interacts and forms supercomplexes with cullin-RING
ubiquitin-ligases, COPS could regulate the process of protein
ubiquitination and maintain protein homeostasis (39). Increasing evidence have hypothesized
that tumor cells have a large burden of unfolded proteins due to
numerous mutations in protein-coding sequences, which leads to
aberrant process of ubiquitination (41). Hence, abnormal expression of COPS
subunits could contribute to the carcinogenesis and progression of
cancer (42). The deregulated
expression of several COPS subunits has been found in different
types of tumor (14–17). However, not all the COPS subunits
were investigated in most of these studies, and it cannot be ruled
out that the expression of the COPS holo complex was changed
instead of a single subunit. To the best of our best knowledge, the
present study is the first study to investigate the expression and
prognostic values of the different COPS9 subunits in HNSCC.
Previous studies have reported that some COPS
subunits are overexpressed in various types of cancer (14–17). For
example, COPS5 and COPS6 were highly expressed in myeloma, lung
cancer, colon adenocarcinoma, breast cancer, glioblastoma and
leukemia (25). In the present
study, the conservative COPS (COPS1-COPS8) were significantly
upregulated in HNSCC tissues compared with that in normal tissues
from TCGA database. However, the COPS9 was not differentially
expressed in both TCGA and Oncomine databases. Notably, high
expression of COPS5 and COPS6 were associated with tumor size,
nodal metastasis and distant metastasis. These results suggested
that COPS5 and COPS6 may play an important role in the progression
of carcinogenesis. In fact, COPS5 and COPS6 regulate diverse
signaling pathways to promote the development of other types of
cancer. COPS5 collaborated with the oncogene, Myc, to contribute to
cell invasion in breast cancer cell lines (43). By promoting the degradation of p27,
COPS5 regulated the cytoplasm transport of p27, which assisted with
HER2-activated cell growth, CDK2 and transformation efficiently in
ovarian cancer (44,45). In addition, overexpression of COPS5
lead to reduced MDM2 self-ubiquitination and nuclear export of p53,
which caused a dysregulation of Mdm2-P53 axis (46–49).
Similarly, overexpression of COPS6 binds to MDM2, and prevents the
process of its auto-ubiquitination and degradation, which leads to
the accumulation of MDM2 and decrease of p53 in breast cancer
(49). The results from GSEA
analysis suggested that overexpression of COPS5 or COPS6 was
associated with p53 signaling pathway in HNSCC, which is consistent
with previous reports (15).
Notably, following knockdown of COPS5 and COPS6 using siRNA, these
genes were found to be essential for growth and migration of the
HNSCC cell lines. These results suggested the critical roles played
by COPS5 and COPS6 in HNSCC.
A recent study indicated that overexpression of
COPS8 induced the progression of epithelial-mesenchymal transition
in colorectal cancer cells, which lead to migration and invasion of
the cancer cells (50). By promoting
the expression of hypoxia response genes (solute carrier family 2
and HIF1) and key dormancy markers, COPS8 arrested cell
proliferation and notably enhanced cell survival under serum
deprivation, with hypoxia or the chemotherapy drug, 5-fluorouracil
(50). Consistent with these
results, in the present study, high mRNA expression level of COPS8
was found in HNSCC tissues, and was associated with distant
metastasis of cancer and indicated poor prognosis of HNSCC. In
addition, COPS8 was important for cell survival in most HNSCC cell
lines. Mechanically, high expression of COPS8 was associated with
apoptosis, DNA repair and hypoxia response. Notably, COPS8 was not
detected in HNSCC tissue using an IHC assay. We hypothesized that
the insensitive primary antibody caused the inaccurate
detection.
With respect to the other COPS subunits, the high
mRNA expression level of COPS1, COPS3 and COPS4 was found in HNSCC
tissues, but was not associated with TMN stages or prognosis.
However, high expression of COPS1 was associated with poor
prognosis in patients with advanced esophageal squamous cell
carcinoma (36). Notably, COPS1,
COPS3 and COPS4 were all necessary for cancer cell survival. A
previous study also found that knockdown of COPS3 expression with a
specific shRNA in hepatocellular carcinoma cell lines significantly
suppressed the cancer growth both in vitro and in
vivo (51). COPS4 expression was
essential for growth of breast cancer and prostate cancer cell
lines (52,53). The results from the present study
suggested that COPS2 was highly expressed in tumor tissues and
upregulated COPS2 was associated with poor prognosis in patients
with HNSCC. Zhao et al (54)
found that DDA1 promoted the progression of colorectal cancer by
activating the NFκB-COPS2-GSK3β pathway. COPS7A and COPS7B were
highly expressed in the HNSCC samples; however, the prognostic
significance was different. High mRNA expression level of COPS7A
was associated with poor prognosis in patients with HNSCC, but the
exact opposite was found with COPS7B.
There are some limitations to the present study.
First and most important, the majority of the study is
bioinformatic analysis, and a lack of clinical and functional
experiments to support the results, which may restrict the
conclusion. Second, a few results are marginally significant in
this study (HR values are close to 1), which make the results not
very convincing. Third, in Fig. 3,
TX, NX, and MX are included in the sub-analysis. These ambiguous
clinical data may lead some results unstable. Therefore, additional
clinical and experimental studies should be performed in the future
to focus on the association between the COPS subunits and
HNSCC.
In conclusion, COPS1-8 mRNA expression level was
upregulated in HNSCC tissues compared with that in normal tissues.
The expression level of COPS2, COPS5, COPS6, COPS7A, COPS7B, COPS8
and COPS9 might be associated with the prognosis of patients with
HNSCC. Notably, COPS5 and COPS6 were critical for the progression
of HNSCC.
Supplementary Material
Supporting Data
Acknowledgements
The authors would like to thank Professor Liang
Jiang (the Department of Oral and Maxillofacial Surgery, Tongji
Hospital, Hubei, China) for providing the cell lines.
Funding
This research was supported by a grant from the
Youth Science Support Project of the Central Hospital of Wuhan
(grant no. 201807632).
Availability of data and materials
All data generated or analyzed during this study are
included in this published article.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Authors' contributions
HZ designed the study and performed the
bioinformatic analysis. JZ performed the western blot, CCK-8 and
wound healing assays. WS analyzed the data. HZ and JZ wrote the
manuscript. All authors approve the final version of the
manuscript. HZ and JZ confirm the authenticity of all the raw
data.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Forastiere AA, Trotti A, Pfister DG and
Grandis JR: Head and neck cancer: Recent advances and new standards
of care. J Clin Oncol. 24:2603–2605. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Haddad RI and Shin DM: Recent advances in
head and neck cancer. N Engl J Med. 359:1143–1154. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Lo Nigro C, Denaro N, Merlotti A and
Merlano M: Head and neck cancer: Improving outcomes with a
multidisciplinary approach. Cancer Manag Res. 9:363–371. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Khurshid Z, Zafar MS, Khan RS, Najeeb S,
Slowey PD and Rehman IU: Role of salivary biomarkers in oral cancer
detection. Adv Clin Chem. 86:23–70. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Sahibzada HA, Khurshid Z, Khan RS, Naseem
M, Siddique KM, Mali M and Zafar MS: Salivary IL-8, IL-6 and TNF-α
as potential diagnostic biomarkers for oral cancer. Diagnostics
(Basel). 7:212017. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Dubiel W, Chaithongyot S, Dubiel D and
Naumann M: The COP9 signalosome: A multi-DUB complex. Biomolecules.
10:10822020. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Cavadini S, Fischer ES, Bunker RD, Potenza
A, Lingaraju GM, Goldie KN, Mohamed WI, Faty M, Petzold G, Beckwith
RE, et al: Cullin-RING ubiquitin E3 ligase regulation by the COP9
signalosome. Nature. 531:598–603. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Lingaraju GM, Bunker RD, Cavadini S, Hess
D, Hassiepen U, Renatus M, Fischer ES and Thomä NH: Crystal
structure of the human COP9 signalosome. Nature. 512:161–165. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Qin N, Xu D, Li J and Deng XW: COP9
signalosome: Discovery, conservation, activity, and function. J
Integr Plant Biol. 62:90–103. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Rao F, Lin H and Su Y: Cullin-RING ligase
regulation by the COP9 signalosome: Structural mechanisms and new
physiologic players. Adv Exp Med Biol. 1217:47–60. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Ebina M, Tsuruta F, Katoh M, Kigoshi Y,
Someya A and Chiba T: Myeloma overexpressed 2 (Myeov2) regulates
L11 subnuclear localization through Nedd8 modification. PLoS One.
8:e652852013. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Li L and Deng XW: The COP9 signalosome: An
alternative lid for the 26S proteasome? Trends Cell Biol.
13:507–509. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Karniol B and Chamovitz DA: The COP9
signalosome: From light signaling to general developmental
regulation and back. Curr Opin Plant Biol. 3:387–393. 2000.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Tomoda K, Kubota Y and Kato J: Degradation
of the cyclin-dependent-kinase inhibitor p27Kip1 is instigated by
Jab1. Nature. 398:160–165. 1999. View
Article : Google Scholar : PubMed/NCBI
|
|
15
|
Bech-Otschir D, Kraft R, Huang X, Henklein
P, Kapelari B, Pollmann C and Dubiel W: COP9 signalosome-specific
phosphorylation targets p53 to degradation by the ubiquitin system.
EMBO J. 20:1630–1639. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Kim JH, Choi JK, Cinghu S, Jang JW, Lee
YS, Li YH, Goh YM, Chi XZ, Lee KS, Wee H and Bae SC: Jab1/CSN5
induces the cytoplasmic localization and degradation of RUNX3. J
Cell Biochem. 107:557–565. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Bemis L, Chan DA, Finkielstein CV, Qi L,
Sutphin PD, Chen X, Stenmark K, Giaccia AJ and Zundel W: Distinct
aerobic and hypoxic mechanisms of HIF-alpha regulation by CSN5.
Genes Dev. 18:739–744. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Xie P, Wang H, Fang J, Du D, Tian Z, Zhen
J, Liu Y, Ding Y, Fu B, Liu F, et al: CSN5 promotes carcinogenesis
of thyroid carcinoma cells through ANGPTL2. Endocrinology.
162:bqaa2062021. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Hou J, Deng Q, Zhou J, Zou J, Zhang Y, Tan
P, Zhang W and Cui H: CSN6 controls the proliferation and
metastasis of glioblastoma by CHIP-mediated degradation of EGFR.
Oncogene. 36:1134–1144. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Su L, Guo W, Lou L, Nie S, Zhang Q, Liu Y,
Chang Y, Zhang X, Li Y and Shen H: EGFR-ERK pathway regulates CSN6
to contribute to PD-L1 expression in glioblastoma. Mol Carcinog.
59:520–532. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Dubois EL, Gerber S, Kisselev A,
Harel-Bellan A and Groisman R: UV-dependent phosphorylation of
COP9/signalosome in UV-induced apoptosis. Oncol Rep. 35:3101–3105.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Yoneda-Kato N, Tomoda K, Umehara M, Arata
Y and Kato JY: Myeloid leukemia factor 1 regulates p53 by
suppressing COP1 via COP9 signalosome subunit 3. EMBO J.
24:1739–1749. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Pollmann C, Huang X, Mall J, Bech-Otschir
D, Naumann M and Dubiel W: The constitutive photomorphogenesis 9
signalosome directs vascular endothelial growth factor production
in tumor cells. Cancer Res. 61:8416–8421. 2001.PubMed/NCBI
|
|
24
|
Denti S, Fernandez-Sanchez ME, Rogge L and
Bianchi E: The COP9 signalosome regulates Skp2 levels and
proliferation of human cells. J Biol Chem. 281:32188–32196. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Lee MH, Zhao R, Phan L and Yeung SC: Roles
of COP9 signalosome in cancer. Cell Cycle. 10:3057–3066. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Yang L, Wang J, Li J, Zhang H, Guo S, Yan
M, Zhu Z, Lan B, Ding Y, Xu M, et al: Identification of serum
biomarkers for gastric cancer diagnosis using a human proteome
microarray. Mol Cell Proteomics. 15:614–623. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Yan T, Wunder JS, Gokgoz N, Gill M,
Eskandarian S, Parkes RK, Bull SB, Bell RS and Andrulis IL: COPS3
amplification and clinical outcome in osteosarcoma. Cancer.
109:1870–1876. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Hong Y, Huang X, An L, Ye H, Ma K, Zhang F
and Xu Q: Overexpression of COPS3 promotes clear cell renal cell
carcinoma progression via regulation of Phospho-AKT(Thr308), cyclin
D1 and caspase-3. Exp Cell Res. 365:163–170. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Sun L: COPS8 in cutaneous melanoma: An
oncogene that accelerates the malignant development of tumor cells
and predicts poor prognosis. Biosci Biotechnol Biochem. 85:242–250.
2021. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Reis PP, Waldron L, Perez-Ordonez B,
Pintilie M, Galloni NN, Xuan Y, Cervigne NK, Warner GC, Makitie AA,
Simpson C, et al: A gene signature in histologically normal
surgical margins is predictive of oral carcinoma recurrence. BMC
Cancer. 11:4372011. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Dodd LE, Sengupta S, Chen IH, den Boon JA,
Cheng YJ, Westra W, Newton MA, Mittl BF, McShane L, Chen CJ, et al:
Genes involved in DNA repair and nitrosamine metabolism and those
located on chromosome 14q32 are dysregulated in nasopharyngeal
carcinoma. Cancer Epidemiol Biomarkers Prev. 15:2216–2225. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Schlingemann J, Habtemichael N, Ittrich C,
Toedt G, Kramer H, Hambek M, Knecht R, Lichter P, Stauber R and
Hahn M: Patient-based cross-platform comparison of oligonucleotide
microarray expression profiles. Lab Invest. 85:1024–1039. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Cromer A, Carles A, Millon R, Ganguli G,
Chalmel F, Lemaire F, Young J, Dembélé D, Thibault C, Muller D, et
al: Identification of genes associated with tumorigenesis and
metastatic potential of hypopharyngeal cancer by microarray
analysis. Oncogene. 23:2484–2498. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Toruner GA, Ulger C, Alkan M, Galante AT,
Rinaggio J, Wilk R, Tian B, Soteropoulos P, Hameed MR, Schwalb MN
and Dermody JJ: Association between gene expression profile and
tumor invasion in oral squamous cell carcinoma. Cancer Genet
Cytogenet. 154:27–35. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Ginos MA, Page GP, Michalowicz BS, Patel
KJ, Volker SE, Pambuccian SE, Ondrey FG, Adams GL and Gaffney PM:
Identification of a gene expression signature associated with
recurrent disease in squamous cell carcinoma of the head and neck.
Cancer Res. 64:55–63. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Okuno T, Wakabayashi M, Kato K, Shinoda M,
Katayama H, Igaki H, Tsubosa Y, Kojima T, Okabe H, Kimura Y, et al:
Esophageal stenosis and the Glasgow Prognostic Score as independent
factors of poor prognosis for patients with locally advanced
unresectable esophageal cancer treated with chemoradiotherapy
(exploratory analysis of JCOG0303). Int J Clin Oncol. 22:1042–1049.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Tsherniak A, Vazquez F, Montgomery PG,
Weir BA, Kryukov G, Cowley GS, Gill S, Harrington WF, Pantel S,
Krill-Burger JM, et al: Defining a cancer dependency map. Cell.
170:564–576.e16. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Irizarry RA, Wang C, Zhou Y and Speed TP:
Gene set enrichment analysis made simple. Stat Methods Med Res.
18:565–575. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Wolf DA, Zhou C and Wee S: The COP9
signalosome: An assembly and maintenance platform for cullin
ubiquitin ligases? Nat Cell Biol. 5:1029–1033. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Füzesi-Levi MG, Fainer I, Ivanov Enchev R,
Ben-Nissan G, Levin Y, Kupervaser M, Friedlander G, Salame TM, Nevo
R, Peter M and Sharon M: CSNAP, the smallest CSN subunit, modulates
proteostasis through cullin-RING ubiquitin ligases. Cell Death
Differ. 27:984–998. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Kuroha K, Ando K, Nakagawa R and Inada T:
The Upf factor complex interacts with aberrant products derived
from mRNAs containing a premature termination codon and facilitates
their proteasomal degradation. J Biol Chem. 288:28630–28640. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Wei N and Deng XW: The COP9 signalosome.
Annu Rev Cell Dev Biol. 19:261–286. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Adler AS, Lin M, Horlings H, Nuyten DS,
van de Vijver MJ and Chang HY: Genetic regulators of large-scale
transcriptional signatures in cancer. Nat Genet. 38:421–430. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Sui L, Dong Y, Ohno M, Watanabe Y,
Sugimoto K, Tai Y and Tokuda M: Jab1 expression is associated with
inverse expression of p27(kip1) and poor prognosis in epithelial
ovarian tumors. Clin Cancer Res. 7:4130–4135. 2001.PubMed/NCBI
|
|
45
|
Sutterlüty H, Chatelain E, Marti A,
Wirbelauer C, Senften M, Müller U and Krek W: p45SKP2 promotes
p27Kip1 degradation and induces S phase in quiescent cells. Nat
Cell Biol. 1:207–214. 1999. View
Article : Google Scholar : PubMed/NCBI
|
|
46
|
Zhou BP, Liao Y, Xia W, Zou Y, Spohn B and
Hung MC: HER-2/neu induces p53 ubiquitination via Akt-mediated MDM2
phosphorylation. Nat Cell Biol. 3:973–982. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Bernardi R, Scaglioni PP, Bergmann S, Horn
HF, Vousden KH and Pandolfi PP: PML regulates p53 stability by
sequestering Mdm2 to the nucleolus. Nat Cell Biol. 6:665–672. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Oh W, Lee EW, Sung YH, Yang MR, Ghim J,
Lee HW and Song J: Jab1 induces the cytoplasmic localization and
degradation of p53 in coordination with Hdm2. J Biol Chem.
281:17457–17465. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Zhao R, Yeung SC, Chen J, Iwakuma T, Su
CH, Chen B, Qu C, Zhang F, Chen YT, Lin YL, et al: Subunit 6 of the
COP9 signalosome promotes tumorigenesis in mice through
stabilization of MDM2 and is upregulated in human cancers. J Clin
Invest. 121:851–865. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Ju S, Wang F, Wang Y and Ju S: CSN8 is a
key regulator in hypoxia-induced epithelial-mesenchymal transition
and dormancy of colorectal cancer cells. Mol Cancer. 19:1682020.
View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Yu YS, Tang ZH, Pan QC, Chen XH, Liu XN
and Zang GQ: Inhibition of Csn3 expression induces growth arrest
and apoptosis of hepatocellular carcinoma cells. Cancer Chemother
Pharmacol. 69:1173–1180. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Bhansali M and Shemshedini L: COP9
subunits 4 and 5 target soluble guanylyl cyclase α1 and p53 in
prostate cancer cells. Mol Endocrinol. 28:834–845. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Yu TL, Cai DL, Zhu GF, Ye XJ, Min TS, Chen
HY, Lu DR and Chen HM: Effects of CSN4 knockdown on proliferation
and apoptosis of breast cancer MDA-MB-231 cells. Yi Chuan.
41:318–326. 2019.(In Chinese). PubMed/NCBI
|
|
54
|
Zhao S, Tang H, Yan D, Fan J, Sun H, Wen
Y, Yu F, Cui F, Zhang D, Xue Y, et al: DDA1 promotes stage IIB-IIC
colon cancer progression by activating NFκB/CSN2/GSK-3β signaling.
Oncotarget. 7:19794–19812. 2016. View Article : Google Scholar : PubMed/NCBI
|