|
1
|
Che Z, Fan J, Zhou Z, Li Q, Ma Z, Hu Z, Wu
Y, Jin Y, Su Y, Liang P and Li H: Activation-induced cytidine
deaminase expression facilitates the malignant phenotype and
epithelial-to-mesenchymal transition in clear cell renal cell
carcinoma. DNA Cell Biol. 39:1299–1312. 2020. View Article : Google Scholar
|
|
2
|
Zhang F, Ma X, Li H, Zhang Y, Li X, Chen
L, Guo G, Gao Y, Gu L, Xie Y, et al: FOXK2 suppresses the malignant
phenotype and induces apoptosis through inhibition of EGFR in
clear-cell renal cell carcinoma. Int J Cancer. 142:2543–2557. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Wang Y, Cong R, Liu S, Zhu B, Wang X and
Xing Q: Decreased expression of METTL14 predicts poor prognosis and
construction of a prognostic signature for clear cell renal cell
carcinoma. Cancer Cell Int. 21:462021. View Article : Google Scholar
|
|
4
|
Tacconi EM, Tuthill M and Protheroe A:
Review of adjuvant therapies in renal cell carcinoma: Evidence to
date. Onco Targets Ther. 13:12301–12316. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Su Y, Lu J, Chen X, Liang C, Luo P, Qin C
and Zhang J: Long non-coding RNA HOTTIP affects renal cell
carcinoma progression by regulating autophagy via the
PI3K/Akt/Atg13 signaling pathway. J Cancer Res Clin Oncol.
145:573–588. 2019. View Article : Google Scholar
|
|
6
|
Xu F, Guan Y, Xue L, Huang S, Gao K, Yang
Z and Chong T: The effect of a novel glycolysis-related gene
signature on progression, prognosis and immune microenvironment of
renal cell carcinoma. BMC Cancer. 20:12072020. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Czarnecka AM, Kukwa W, Kornakiewicz A,
Lian F and Szczylik C: Clinical and molecular prognostic and
predictive biomarkers in clear cell renal cell cancer. Future
Oncol. 10:2493–2508. 2014. View Article : Google Scholar
|
|
8
|
Wang ZH, Zhang YZ, Wang YS and Ma XX:
Identification of novel cell glycolysis related gene signature
predicting survival in patients with endometrial cancer. Cancer
Cell Int. 19:2962019. View Article : Google Scholar
|
|
9
|
Gray RE and Harris GT: Renal cell
carcinoma: Diagnosis and management. Am Fam Physician. 99:179–184.
2019.PubMed/NCBI
|
|
10
|
Shi J, Wang K, Xiong Z, Yuan C, Wang C,
Cao Q, Yu H, Meng X, Xie K, Cheng Z, et al: Impact of inflammation
and immunotherapy in renal cell carcinoma. Oncol Lett. 20:2722020.
View Article : Google Scholar
|
|
11
|
Zhao E, Li L, Zhang W, Wang W, Chan Y, You
B and Li X: Comprehensive characterization of immune- and
inflammation-associated biomarkers based on multi-omics integration
in kidney renal clear cell carcinoma. J Transl Med. 17:1772019.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Chen Q, Wang J, Zhang Q, Zhang J, Lou Y,
Yang J, Chen Y, Wei T, Zhang J, Fu Q, et al: Tumour cell-derived
debris and IgG synergistically promote metastasis of pancreatic
cancer by inducing inflammation via tumour-associated macrophages.
Br J Cancer. 121:786–795. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Balkwill F and Mantovani A: Inflammation
and cancer: Back to Virchow? Lancet. 357:539–545. 2001. View Article : Google Scholar
|
|
14
|
Liubomirski Y, Lerrer S, Meshel T,
Rubinstein-Achiasaf L, Morein D, Wiemann S, Körner C and Ben-Baruch
A: Tumor-stroma-inflammation networks promote pro-metastatic
chemokines and aggressiveness characteristics in triple-negative
breast cancer. Front Immunol. 10:7572019. View Article : Google Scholar
|
|
15
|
Ngabire D and Kim GD: Autophagy and
inflammatory response in the tumor microenvironment. Int J Mol Sci.
18:20162017. View Article : Google Scholar
|
|
16
|
Carraway RE and Cochrane DE: Enhanced
vascular permeability is hypothesized to promote
inflammation-induced carcinogenesis and tumor development via
extravasation of large molecular proteins into the tissue. Med
Hypotheses. 78:738–743. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Mantovani A, Bussolino F and Dejana E:
Cytokine regulation of endothelial cell function. FASEB J.
6:2591–2599. 1992. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Dominguez C, David JM and Palena C:
Epithelial-mesenchymal transition and inflammation at the site of
the primary tumor. Semin Cancer Biol. 47:177–184. 2017. View Article : Google Scholar
|
|
19
|
Koong AC, Denko NC, Hudson KM, Schindler
C, Swiersz L, Koch C, Evans S, Ibrahim H, Le QT, Terris DJ and
Giaccia AJ: Candidate genes for the hypoxic tumor phenotype. Cancer
Res. 60:883–887. 2000.PubMed/NCBI
|
|
20
|
Taher MY, Davies DM and Maher J: The role
of the interleukin (IL)-6/IL-6 receptor axis in cancer. Biochem Soc
Trans. 46:1449–1462. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Kaminska K, Czarnecka AM, Escudier B, Lian
F and Szczylik C: Interleukin-6 as an emerging regulator of renal
cell cancer. Urol Oncol. 33:476–485. 2015. View Article : Google Scholar
|
|
22
|
DiGiacomo JW and Gilkes DM: Tumor hypoxia
as an enhancer of inflammation-mediated metastasis: Emerging
therapeutic strategies. Target Oncol. 13:157–173. 2018. View Article : Google Scholar
|
|
23
|
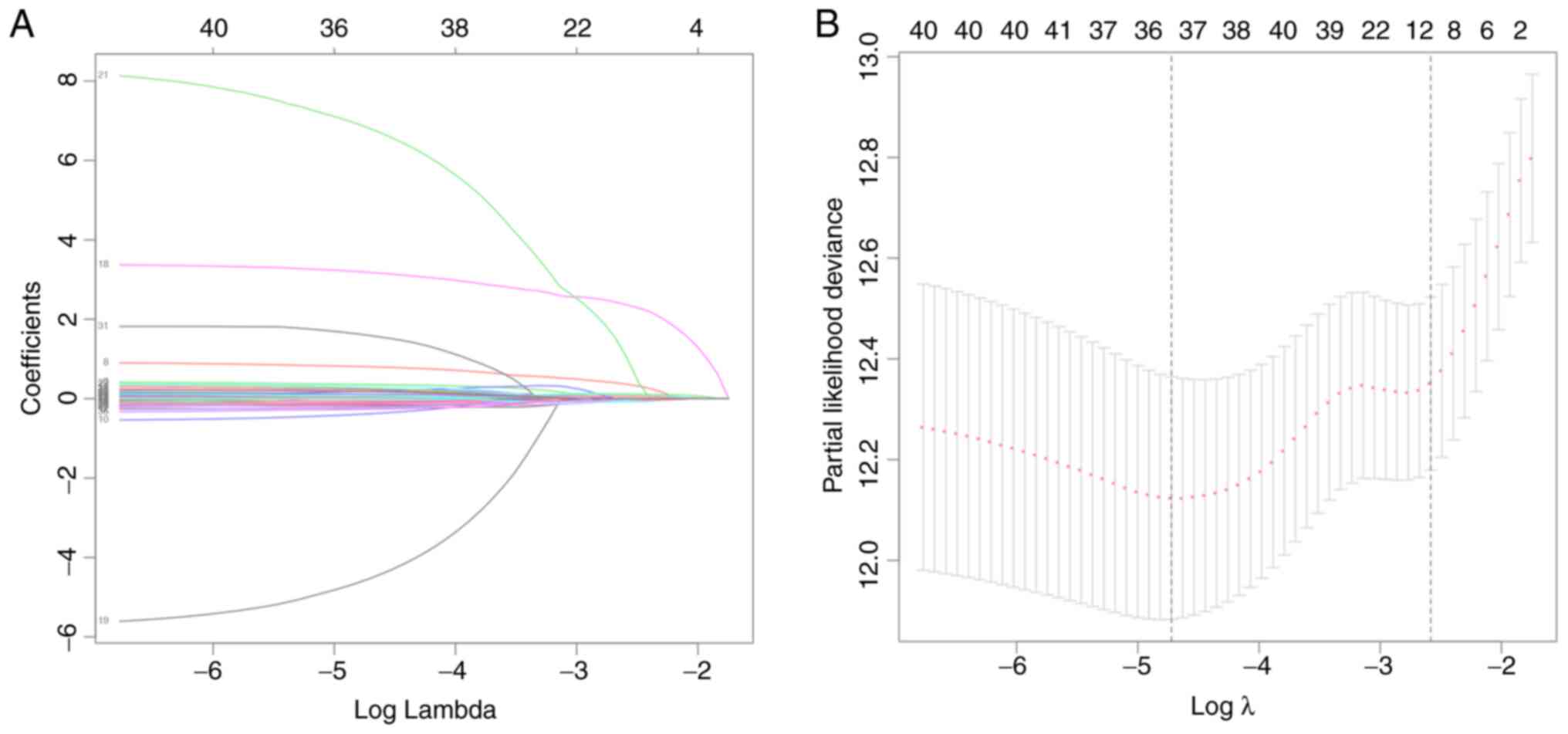
Simon N, Friedman J, Hastie T and
Tibshirani R: Regularization paths for Cox's proportional hazards
model via coordinate descent. J Stat Softw. 39:1–13. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Jin W, Zhang Y, Liu Z, Che Z, Gao M and
Peng H: Exploration of the molecular characteristics of the
tumor-immune interaction and the development of an individualized
immune prognostic signature for neuroblastoma. J Cell Physiol.
236:294–308. 2021. View Article : Google Scholar
|
|
25
|
Therneau TM and Grambsch PM: Modeling
survival data: Extending the Cox model. Springer; New York, NY: pp.
39–77. 2000
|
|
26
|
Li B, Cui Y, Diehn M and Li R: Development
and validation of an individualized immune prognostic signature in
early-stage nonsquamous non-small cell lung cancer. JAMA Oncol.
3:1529–1537. 2017. View Article : Google Scholar
|
|
27
|
Newman AM, Liu CL, Green MR, Gentles AJ,
Feng W, Xu Y, Hoang CD, Diehn M and Alizadeh AA: Robust enumeration
of cell subsets from tissue expression profiles. Nat Methods.
12:453–457. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Liu L, Huang L, Chen W, Zhang G, Li Y, Wu
Y, Xiong J and Jie Z: Comprehensive analysis of necroptosis-related
long noncoding RNA immune infiltration and prediction of prognosis
in patients with colon cancer. Front Mol Biosci. 9:8112692022.
View Article : Google Scholar
|
|
29
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(−Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Xiao Z, He L, Takemoto M, Jalanko H, Chan
GC, Storm DR, Betsholtz C, Tryggvason K and Patrakka J: Glomerular
podocytes express type 1 adenylate cyclase: Inactivation results in
susceptibility to proteinuria. Nephron Exp Nephrol. 118:e39–e48.
2011. View Article : Google Scholar
|
|
31
|
Saleh AA, Tayel SI, Shalaby AG and El
Naidany SS: Role of adiponectin gene and receptor polymorphisms and
their mRNA levels with serum adiponectin level in myocardial
infarction. Appl Clin Genet. 13:241–252. 2020. View Article : Google Scholar
|
|
32
|
Mertens TC, Hanmandlu A, Tu L, Phan C,
Collum SD, Chen NY, Weng T, Davies J, Liu C, Eltzschig HK, et al:
Switching-Off Adora2b in vascular smooth muscle cells halts the
development of pulmonary hypertension. Front Physiol. 9:5552018.
View Article : Google Scholar
|
|
33
|
Feuser K, Thon KP, Bischoff SC and Lorentz
A: Human intestinal mast cells are a potent source of multiple
chemokines. Cytokine. 58:178–185. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Berglöf E, Andre R, Renshaw BR, Allan SM,
Lawrence CB, Rothwell NJ and Pinteaux E: IL-1Rrp2 expression and
IL-1F9 (IL-1H1) actions in brain cells. J Neuroimmunol. 139:36–43.
2003. View Article : Google Scholar
|
|
35
|
Chai L, Dai L, Che Y, Xu J, Liu G, Zhang Z
and Yang R: LRRC19, a novel member of the leucine-rich repeat
protein family, activates NF-kappaB and induces expression of
proinflammatory cytokines. Biochem Biophys Res Commun. 388:543–548.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Venerito M, Helmke C, Jechorek D, Wex T,
Rosania R, Antweiler K, Weigt J and Malfertheiner P: Leukotriene
receptor expression in esophageal squamous cell cancer and
non-transformed esophageal epithelium: A matched case control
study. BMC Gastroenterol. 16:852016. View Article : Google Scholar
|
|
37
|
Liu B, Xie Y, Mei X, Sun Y, Shi W and Wu
Z: Reciprocal regulation of interleukin-17A and interleukin-22
secretion through aryl hydrocarbon receptor activation in CD4(+) T
cells of patients with vitiligo. Exp Ther Med. 21:1582021.
View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Cai X, Zhang P, Wang S, Hong L, Yu S, Li
B, Zeng H, Yang X and Shao L: lncRNA FGD5 antisense RNA 1
upregulates RORA to suppress hypoxic injury of human cardiomyocyte
cells by inhibiting oxidative stress and apoptosis via miR195. Mol
Med Rep. 22:4579–4588. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Flores-Mendoza LK, Estrada-Jimenez T,
Sedeno-Monge V, Moreno M, Manjarrez MD, González-Ochoa G,
Millán-Pérez Peña L and Reyes-Leyva J: IL-10 and socs3 are
predictive biomarkers of dengue hemorrhagic fever. Mediators
Inflamm. 2017:51975922017. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Liang Y, Qiao L, Peng X, Cui Z, Yin Y,
Liao H, Jiang M and Li L: The chemokine receptor CCR1 is identified
in mast cell-derived exosomes. Am J Transl Res. 10:352–367.
2018.PubMed/NCBI
|
|
41
|
Wu J, Lv Y, Li Y, Jiang Y, Wang L, Zhang
X, Sun M, Zou Y, Xu J and Zhang L: MCM3AP-AS1/miR-876-5p/WNT5A axis
regulates the proliferation of prostate cancer cells. Cancer Cell
Int. 20:3072020. View Article : Google Scholar
|
|
42
|
Patil I: Visualizations with statistical
details: The'ggstatsplot' approach. J Open Source Softw.
6:31672021. View Article : Google Scholar
|
|
43
|
Alba AC, Agoritsas T, Walsh M, Hanna S,
Iorio A, Devereaux PJ, McGinn T and Guyatt G: Discrimination and
calibration of clinical prediction models: Users' guides to the
medical literature. JAMA. 318:1377–1384. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
de Vivar Chevez AR, Finke J and Bukowski
R: The role of inflammation in kidney cancer. Adv Exp Med Biol.
816:197–234. 2014. View Article : Google Scholar
|
|
45
|
Zhang X, Wei X, Wang Y, Wang S, Ji C, Yao
L and Song N: Pyroptosis regulators and tumor microenvironment
infiltration characterization in clear cell renal cell carcinoma.
Front Oncol. 11:7742792021. View Article : Google Scholar
|
|
46
|
Nishida J, Momoi Y, Miyakuni K, Tamura Y,
Takahashi K, Koinuma D, Miyazono K and Ehata S: Epigenetic
remodelling shapes inflammatory renal cancer and
neutrophil-dependent metastasis. Nat Cell Biol. 22:465–475. 2020.
View Article : Google Scholar
|
|
47
|
Gorka J, Marona P, Kwapisz O, Rys J, Jura
J and Miekus K: The anti-inflammatory protein MCPIP1 inhibits the
development of ccRCC by maintaining high levels of tumour
suppressors. Eur J Pharmacol. 888:1735912020. View Article : Google Scholar
|
|
48
|
Roumenina LT, Daugan MV, Noé R, Petitprez
F, Vano YA, Sanchez-Salas R, Becht E, Meilleroux J, Clec'h BL,
Giraldo NA, et al: Tumor cells hijack macrophage-produced
complement C1q to promote tumor growth. Cancer Immunol Res.
7:1091–1105. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Hua X, Chen J, Su Y and Liang C:
Identification of an immune-related risk signature for predicting
prognosis in clear cell renal cell carcinoma. Aging (Albany NY).
12:2302–2332. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Vezzani B, Carinci M, Previati M,
Giacovazzi S, Della Sala M, Gafà R, Lanza G, Wieckowski MR, Pinton
P and Giorgi C: Epigenetic regulation: A link between inflammation
and carcinogenesis. Cancers (Basel). 14:12212022. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Özdemir BH: Tumor microenvironment:
Necroptosis switches the subtype of liver cancer while necrosis
promotes tumor recurrence and progression. Exp Clin Transplant. Mar
15–2022.(Epub ahead of print). View Article : Google Scholar
|
|
52
|
Jha NK, Arfin S, Jha SK, Kar R, Dey A,
Gundamaraju R, Ashraf GM, Gupta PK, Dhanasekaran S, Abomughaid MM,
et al: Re-establishing the comprehension of phytomedicine and
nanomedicine in inflammation-mediated cancer signaling. Semin
Cancer Biol. Feb 23–2022.(Epub ahead of print). View Article : Google Scholar
|
|
53
|
Şenbabaoğlu Y, Gejman RS, Winer AG, Liu M,
Van Allen EM, de Velasco G, Miao D, Ostrovnaya I, Drill E, Luna A,
et al: Tumor immune microenvironment characterization in clear cell
renal cell carcinoma identifies prognostic and
immunotherapeutically relevant messenger RNA signatures. Genome
Biol. 17:2312016. View Article : Google Scholar
|
|
54
|
Chevrier S, Levine JH, Zanotelli VR,
Silina K, Schulz D, Bacac M, Ries CH, Ailles L, Jewett MA, Moch H,
et al: An immune atlas of clear cell renal cell carcinoma. Cell.
169:736–749.e718. 2017. View Article : Google Scholar
|
|
55
|
Xiao GF, Yan X, Chen Z, Zhang RJ, Liu TZ
and Hu WL: Identification of a novel immune-related prognostic
biomarker and small-molecule drugs in clear cell renal cell
carcinoma (ccRCC) by a merged microarray-acquired dataset and TCGA
database. Front Genet. 11:8102020. View Article : Google Scholar
|
|
56
|
Tang X, Zhang A, Feng Y, Su Y, Wang X,
Jiang F and Ma J: A novel pyroptosis-related lncRNAs signature for
predicting the prognosis of kidney renal clear cell carcinoma and
its associations with immunity. J Oncol. 2021:99971852021.
View Article : Google Scholar
|
|
57
|
Dunn GP, Bruce AT, Ikeda H, Old LJ and
Schreiber RD: Cancer immunoediting: From immunosurveillance to
tumor escape. Nat Immunol. 3:991–998. 2002. View Article : Google Scholar
|
|
58
|
Wang H, Franco F and Ho PC: Metabolic
regulation of tregs in cancer: Opportunities for immunotherapy.
Trends Cancer. 3:583–592. 2017. View Article : Google Scholar : PubMed/NCBI
|