Introduction
Ovarian steroid cell tumor is a rare subtype of
sex-cord tumor, accounting for ~0.1% of all ovarian neoplasms
(1). Among all types of ovarian
steroid cell tumor, steroid cell tumor, not otherwise specified
(NOS), is the predominant type, accounting for ~80% of all cases
(2). This type of tumor may occur
at any age, with a mean age of 43 years. Steroid cell tumors most
commonly secrete androgens, thus resulting in virilization and
hirsutism (1). Although the
majority of these tumors are benign, approximately one-third of
them may display a malignant behavior (2). Ultrasound and magnetic resonance
imaging (MRI) are the most commonly applied imaging methods for
steroid cell tumors. However, histopathology remains the gold
standard technique for tumor diagnosis. A recently used imaging
method, namely contrast-enhanced ultrasound (CEUS), uses
microbubble contrast agents for imaging. It has been reported that
CEUS may be used to improve the diagnostic accuracy of ovarian
tumor ultrasonography (3). In the
current study, a case of a female patient with ovarian steroid cell
tumor diagnosed using CEUS was reported.
Case report
A 61-year-old female was admitted to the China-Japan
Friendship Hospital (Beijing, China) with complaints of a male
patterned voice and hirsutism for >3 years. The patient also
reported unexpected hyperhidrosis that persisted since it developed
two years previously. The patient's BMI was normal but after
walking for 10 min, the patient was sweating profusely. Physical
examination revealed normal blood pressure, obvious facial hair and
low voice. In addition, laboratory tests indicated high
testosterone (21.690 nmol/l; normal range, 0.35-2.60 nmol/l) and
normal dehydroepiandrosterone (39.70 µg/dl; normal range, 12–133
µg/dl) levels in the serum. The other laboratory tests, including
cortisol and thyroid-stimulating hormone levels, and tumor markers,
such as serum CA125, carcinoembryonic antigen, CA199, CA153 and
alpha-fetoprotein levels, were in the normal range. Finally, pelvic
MRI revealed that the right ovary was normal.
The patient underwent conventional ultrasound for
routine examination. A high-resolution ultrasound instrument
(Siemens Acuson Sequoia; Siemens Medical Solutions) equipped with a
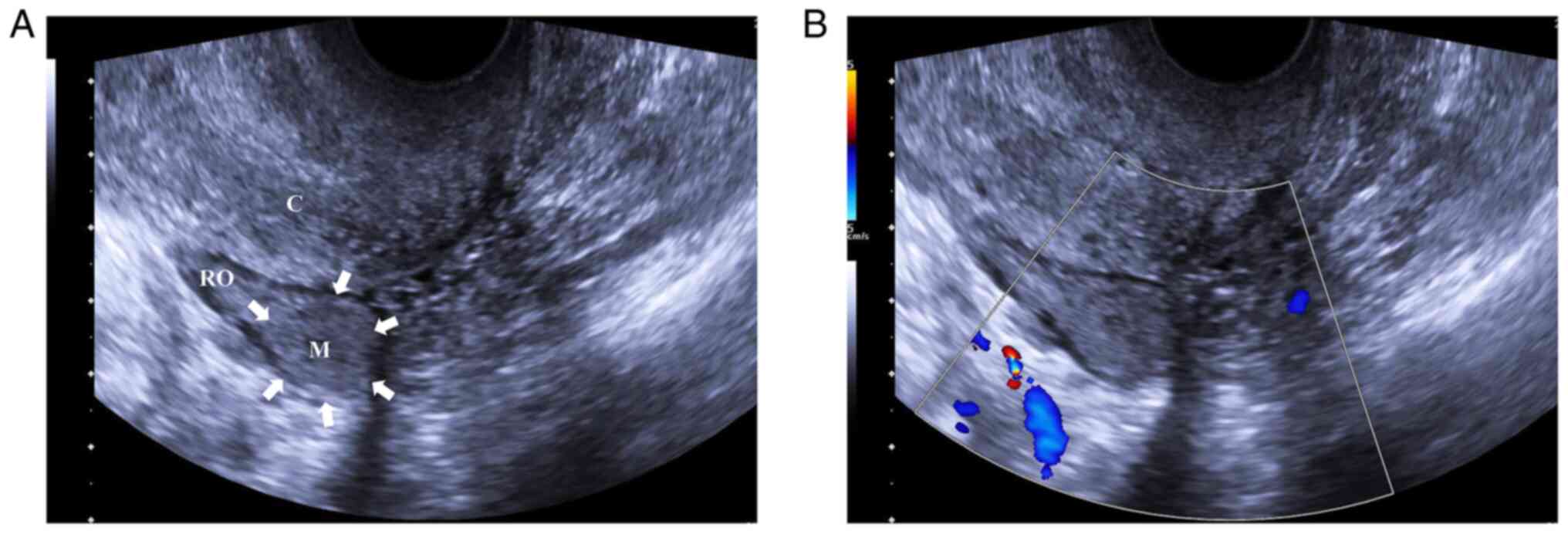
10 MHz linear probe was used. A suspicious homogeneous iso-echoic
lesion with a size of 1.7×1.5 cm was detected in the right ovary on
the gray-scale ultrasound (Fig.
1A). However, color Doppler ultrasound indicated no blood flow
signal in the lesion (Fig.
1B).
To verify the diagnosis, the same ultrasound system
(Siemens Acuson Sequoia; Siemens Medical Solutions) equipped with a
10 MHz linear probe was used for CEUS examination. Low mechanical
index CEUS was performed after a bolus intravenous injection of 1.2
ml SonoVue® (Bracco) followed by a flush of 5 ml saline.
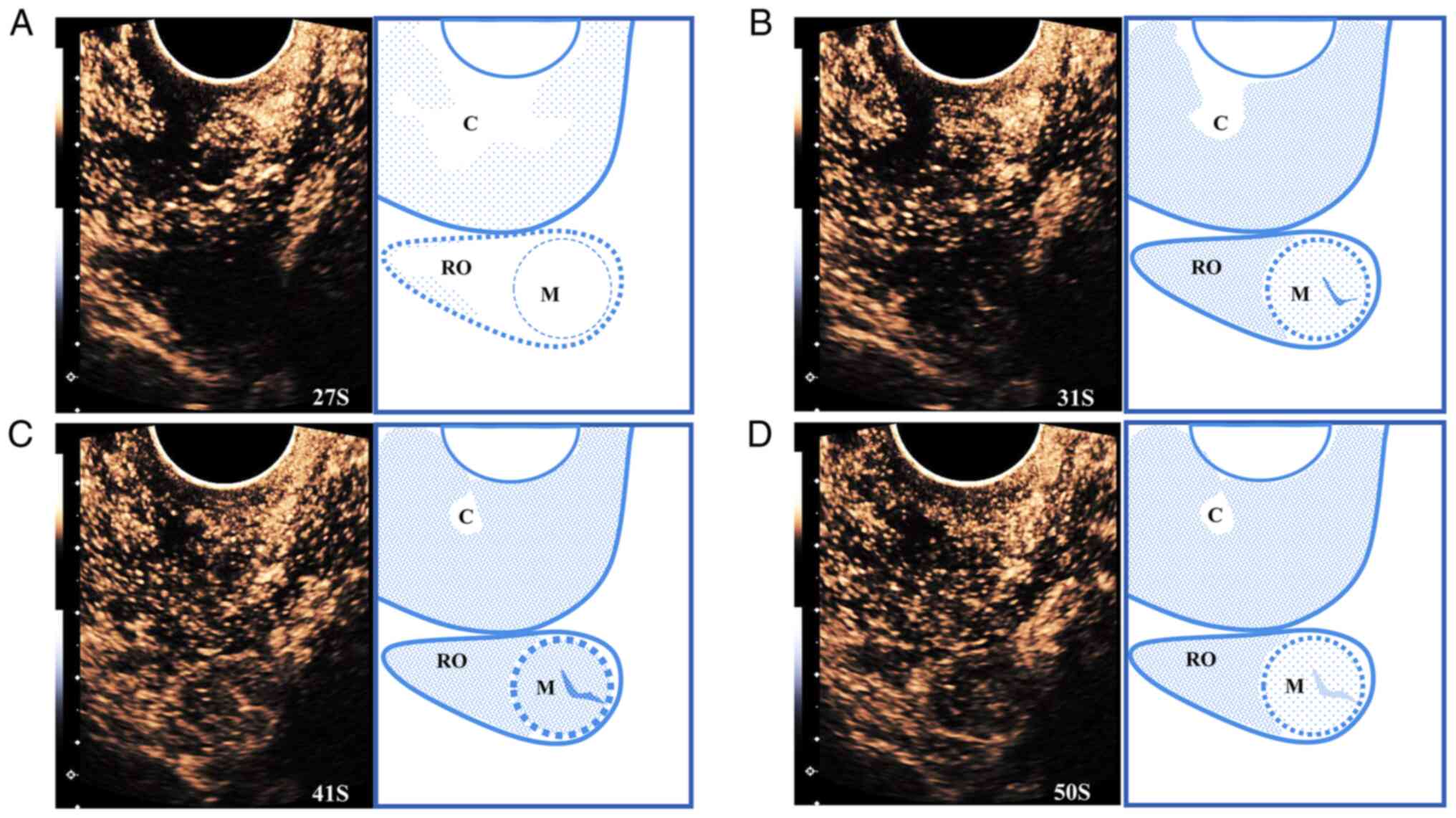
Following injection, a timer was started. During the arterial
phase, the masses appeared iso-homogeneous compared with the
peripheral ovarian parenchyma and maintained their iso-enhancement
in the late phase. The lesion was enhanced at the beginning of the
arterial phase from the center to the periphery, characterized by
ring-shaped enhancement (Fig.
2).
Due to the findings of CEUS, a
testosterone-producing ovarian tumor was suspected. Therefore, the
patient underwent laparoscopic bilateral salpingo-oophorectomy.
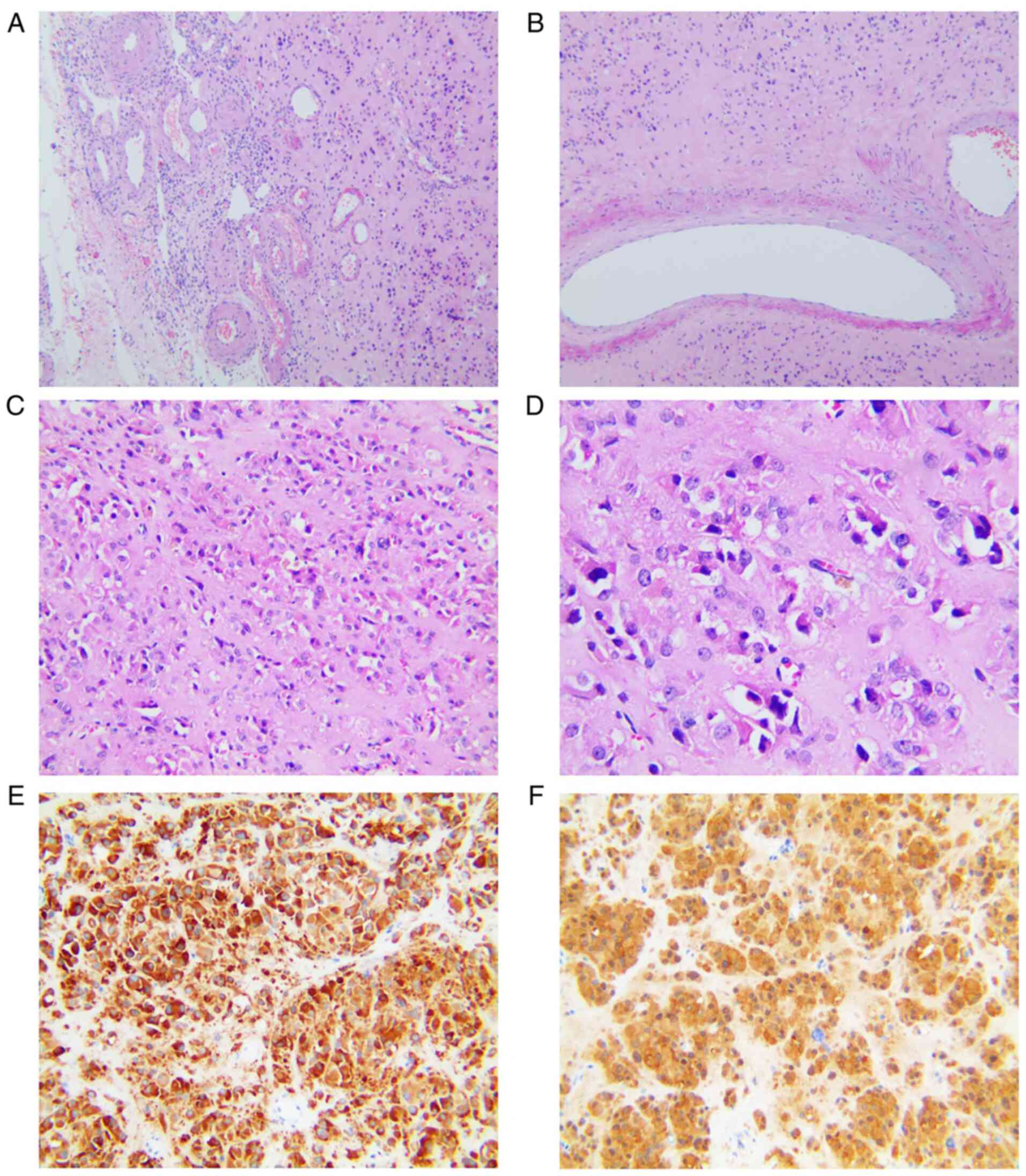
Histopathological assessment confirmed the presence of a steroid
cell tumor in the right ovary with a diameter of 1.5 cm. The
analysis also revealed mild atypia and no significant necrosis or
mitotic activity. In addition, immunohistochemical staining
indicated strong and diffuse staining for inhibin and calretinin
(Fig. 3). The analyses were
performed at the pathology department according to standard
procedures using the following reagents according to the
manufacturer's protocol: Inhibin alpha mouse monoclonal antibody
(cat. no. ZM-0460; Origene); and calretinin mouse monoclonal
antibody (cat. no. MAB-0716; MAB Biotechnologies).
Postsurgical evaluation after one month revealed
normalization of testosterone levels to 0.910 nmol/l, while the
patient experienced complete symptomatic relief. At 12 months
postoperatively, the female patient had no evidence of
recurrence.
Discussion
The incidence of steroid cell tumors is estimated to
be <0.1% among all types of ovarian tumor (1). Steroid cell tumor is an ovarian
parenchymal tumor composed of steroid cells (2). This type of tumor may occur at any
age, with an average age of 43 years (1). The clinical manifestations of the
disease are associated with the type of hormones secreted by the
tumors. The majority of them secrete testosterone. Patients with
steroid cell tumors have virilization symptoms such as hirsutism,
acne, amenorrhea, deep voice, abnormal facial hair growth and at
times alopecia (1,4,5). In
addition, in postmenopausal females, manifestations of
estrogen-secreting tumors include endometrial hyperplasia and
bleeding. Rarely, progestational effects or Cushing's syndrome may
occur (1,6). Steroid cell tumors have also been
reported in patients with von Hippel-Lindau syndrome (7–9).
Steroid cell tumors are most commonly solid, benign,
unilateral and well-circumscribed. However, mixed solid and cystic
masses may also be seen (10).
Histologically, steroid cell tumors consist of polygonal cells
(2), while calcification, necrosis
and hemorrhage may be occasionally observed. Nuclear atypia and
mitotic activity are rare. As mentioned above, ovarian steroid cell
tumors are benign. However, approximately a third of these tumors
may exhibit malignant behavior. Hayes and Scully (1) defined five pathological predictive
characteristics of the malignant behavior of these tumors: size
>7 cm; >2 mitoses/10 high-power fields; necrosis; hemorrhage;
and grade 2–3 nuclear atypia (11,12).
Immunohistochemically, inhibin and calretinin are considered
sensitive markers for diagnosing steroid cell tumors.
In the clinic, surgery is the basic treatment
approach for ovarian steroid tumors. Treatment is individualized
and depends on several factors, including tumor stage, the presence
of malignant features, age and fertility status (13,14).
Following surgery, testosterone levels may return to normal, while
virilization symptoms disappear. The prognosis of ovarian steroid
tumor is good, while metastasis and recurrence rarely occur.
The most commonly used imaging techniques for this
type of tumor are ultrasound and MRI. As demonstrated by
transvaginal ultrasound, ovarian steroid cell tumors are solid
tumors with a hypo-/iso-echoic, homogeneous or heterogeneous
texture (10,15–17),
characterized by abundant blood flow signals (18). On MRI, steroid cell tumors appear
heterogeneous and as medium-intensity solid mass interspersed with
small cysts. The tumor is significantly enhanced following
Gd-diethylene triamine pentaacetate injection, thus indicating
hypervascularity of the tumor (6,16,19).
Certain tumors may be so small that they may not be
diagnosed even after careful radiological scrutiny (17,20).
In the case of the present study, no obvious mass was observed on
the right ovary on MRI. However, a suspicious mass was observed on
gray-scale ultrasound. The tumor was not definitively diagnosed by
conventional ultrasonography due to its small size and isoechoic
appearance on gray-scale ultrasonography compared with the ovarian
tissue. Finally, CEUS revealed a solid mass in the ovary with clear
boundaries. Furthermore, color Doppler imaging indicated no blood
flow signals in and around the tumor. However, using CEUS, internal
dendritic and peripheral annular enhancements were observed, which
may have been due to the feeding vessels within the tumor and the
abundant vascular structures around it. The CEUS findings were
consistent with the histopathological results. To the best of our
knowledge, the present study was the first case study to describe
the contrast-enhanced appearance of a steroid cell tumor. It has
been reported that the use of a contrast agent may improve the
clarity of the power Doppler signal and help identify the
vascularized areas of a tumor. CEUS is able to clarify the location
of a tumor, whose lesions cannot be displayed by gray-scale
ultrasound and MRI. CEUS is not a specialized imaging method for
NOS. However, it is able to improve the specificity of ultrasound
in the differential diagnosis through dynamic microvascular
features. Therefore, the features of CEUS may improve the
diagnostic accuracy. At present, the definitive diagnosis of
ovarian steroid tumors is made based on histopathological
evaluation and immunohistochemistry (21).
Steroid cell tumors are characterized by a slightly
abundant blood supply. The present case study reported the CEUS
appearance of an ovarian steroid tumor and highlighted the
significance of CEUS in the detection of these types of tumors.
Furthermore, the imaging features of CEUS may provide useful
information regarding the tumor location, density and enhancement
pattern.
Acknowledgements
Not applicable.
Funding
The present study was supported by the Scientific Research
Foundation of the China-Japan Friendship Hospital (grant no.
2019-RC-2).
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
MS and BZ performed examinations and recorded data.
MS collected clinical information and drafted the manuscript. MS
and BZ approved the final version of the manuscript for
publication. MS and BZ checked and approved the authenticity of the
raw data. All authors read and approved the final manuscript.
Ethics approval and consent to
participate
The study was approved by the Ethics Committee of
the China-Japan Friendship Hospital (Beijing, China). Written
informed consent was provided by the patient.
Patient consent for publication
The patient provided written informed consent for
the publication of her data and images.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Hayes MC and Scully RE: Ovarian steroid
cell tumors (not otherwise specified). A clinicopathological
analysis of 63 cases. Am J Surg Pathol. 11:835–845. 1987.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Herrington CS: WHO Classification of
Tumours Female Genital Tumours. International Agency for Research
on Cancer; 2020
|
|
3
|
Ma X, Zhao Y, Zhang B, Ling W, Zhuo H, Jia
H and Li P: Contrast-enhanced ultrasound for differential diagnosis
of malignant and benign ovarian tumors: Systematic review and
meta-analysis. Ultrasound Obstet Gynecol. 46:277–283. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Liu AX, Sun J, Shao WQ, Jin HM and Song
WQ: Steroid cell tumors, not otherwise specified (NOS), in an
accessory ovary: A case report and literature review. Gynecol
Oncol. 97:260–262. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Dinc G, Saygin I, Kart C, Mungan S, Guven
S and Seda Güvendağ; Güven E, : A rare case of postmenopausal
severe virilization: Ovarian steroid cell tumour, not otherwise
specified. J Cases Obstet Gynecol. 3:19–21. 2016.
|
|
6
|
Wang PH, Chao HT, Lee RC, Lai CR, Lee WL,
Kwok CF, Yuan CC and Ng HT: Steroid cell tumors of the ovary:
Clinical, ultrasonic, and MRI diagnosis-a case report. Eur J
Radiol. 26:269–273. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Morani AC, Mubarak AI, Bhosale HR, Ramani
NS, Waguespack SG and Ying A: Steroid cell ovarian tumor in a case
of von Hippel-Lindau disease: Demonstrating lipid content of the
mass with MR imaging. Magn Reson Med Sci. 18:251–252. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Wagner M, Browne HN, Linehan WM, Merino M,
Babar N and Stratton P: Lipid cell tumors in two women with von
Hippel-Lindau syndrome. Obstet Gynecol. 116:535–539. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Marques A and Portugal R: Ovarian steroid
cell tumor in an adolescent with Von Hippel-Lindau syndrome: A case
report and review of the literature. Int J Gynecol Pathol.
39:473–477. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Patil VS, VemiReddy PR, Taqdees A and
Arakeri SU: Steroid cell tumor of the ovary-a rare case report and
review of literature. Int J Appl Basic Med Res. 9:185–187. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
del Valle Rubido C: Ovarian steroid cell
tumor associated to endometrial hyperplasia and presenting as
post-menopausal vaginal bleeding. Gynecol Obstet (Sunnyvale).
5:3162015.
|
|
12
|
Yoshimatsu T, Nagai K, Miyawaki R,
Moritani K, Ohkubo K, Kuwabara J, Tatsuta K, Kurata M, Fukushima M,
Kitazawa R, et al: Malignant ovarian steroid cell tumor, not
otherwise specified, causes virilization in a 4-year-old girl: A
case report and literature review. Case Rep Oncol. 13:358–364.
2020. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Jiang W, Tao X, Fang F, Zhang S and Xu C:
Benign and malignant ovarian steroid cell tumors, not otherwise
specified: Case studies, comparison, and review of the literature.
J Ovarian Res. 6:532013. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Ismail S, Hraib M, Issa R, Alassi T and
Alshehabi Z: A large ovarian steroid cell tumor-not otherwise
specified with a unique combination of benign and malignant
features as a challenging cause of oligomenorrhea and hirsutism in
a 21-year-old Syrian female: A case report. BMC Womens Health.
21:952021. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Matsuda S, Yamaguchi Y, Kaseki H, Watanabe
K, Ono S, Yamamoto A, Ichikawa M, Akira S and Takeshita T: Case of
ovarian steroid cell tumor diagnosed after presenting acute heart
failure. J Obstet Gynaecol Res. 46:1211–1215. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Matsukawa J, Takahashi T, Hada Y, Kameda
W, Ota K, Fukase M, Takahashi K, Matsuo K, Mizunuma H and Nagase S:
Successful laparoscopic resection of virilizing ovarian steroid
cell tumor, not otherwise specified, in a 22-year-old woman: A case
report and evaluation of the steroidogenic pathway. Fukushima J Med
Sci. 65:133–139. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Faten H, Dorra G, Slim C, Wajdi S, Nadia
C, Kais C, Tahia B and Mohamed A: Ovarian steroid cell tumor (Not
Otherwise Specified): A case report of ovarian hyperandrogenism.
Case Rep Oncol Med. 2020:69708232020.PubMed/NCBI
|
|
18
|
Jiang MJ, Le Q, Yang BW, Yuan F and Chen
H: Ovarian sex cord stromal tumours: Analysis of the clinical and
sonographic characteristics of different histopathologic subtypes.
J Ovarian Res. 14:532021. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Saida T, Tanaka YO and Minami M: Steroid
cell tumor of the ovary, not otherwise specified: CT and MR
findings. AJR Am J Roentgenol. 188:W393–W394. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Scheker EV, Kathuria A, Esnakula A, Sasano
H, Yamazaki Y, Tevosian S, Auchus RJ, Ghayee HK and Dhir G:
Expression of key androgen-activating enzymes in ovarian steroid
cell tumor, not otherwise specified. J Investig Med High Impact
Case Rep. Jun 26–2020.(Epub ahead of print). PubMed/NCBI
|
|
21
|
Sconfienza L, Perrone N, Delnevo A,
Lacelli F, Murolo C, Gandolfo N and Serafini G: Diagnostic value of
contrast-enhanced ultrasonography in the characterization of
ovarian tumors. J Ultrasound. 13:9–15. 2010. View Article : Google Scholar : PubMed/NCBI
|