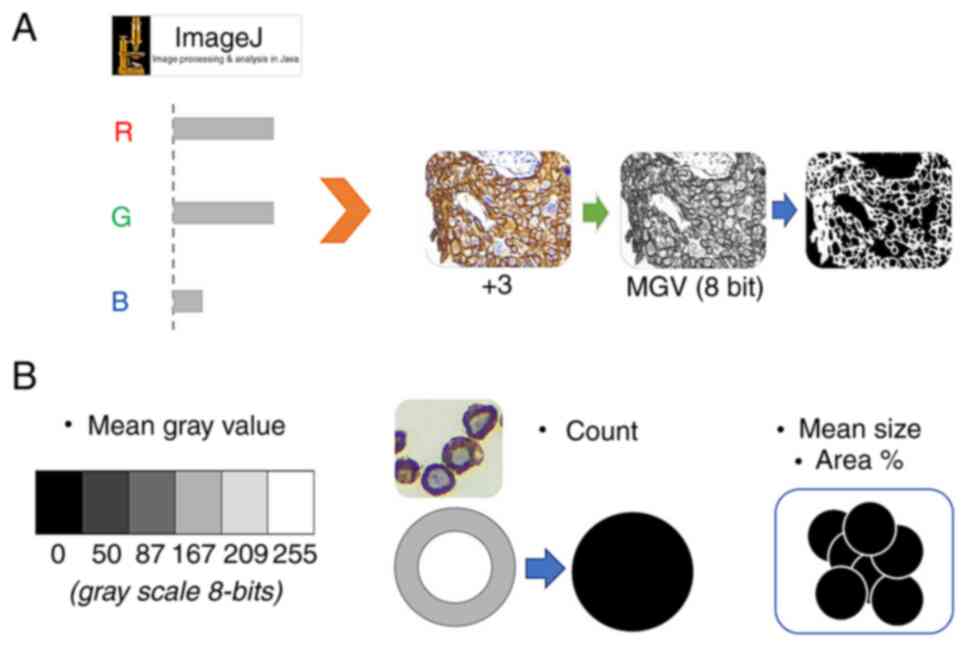
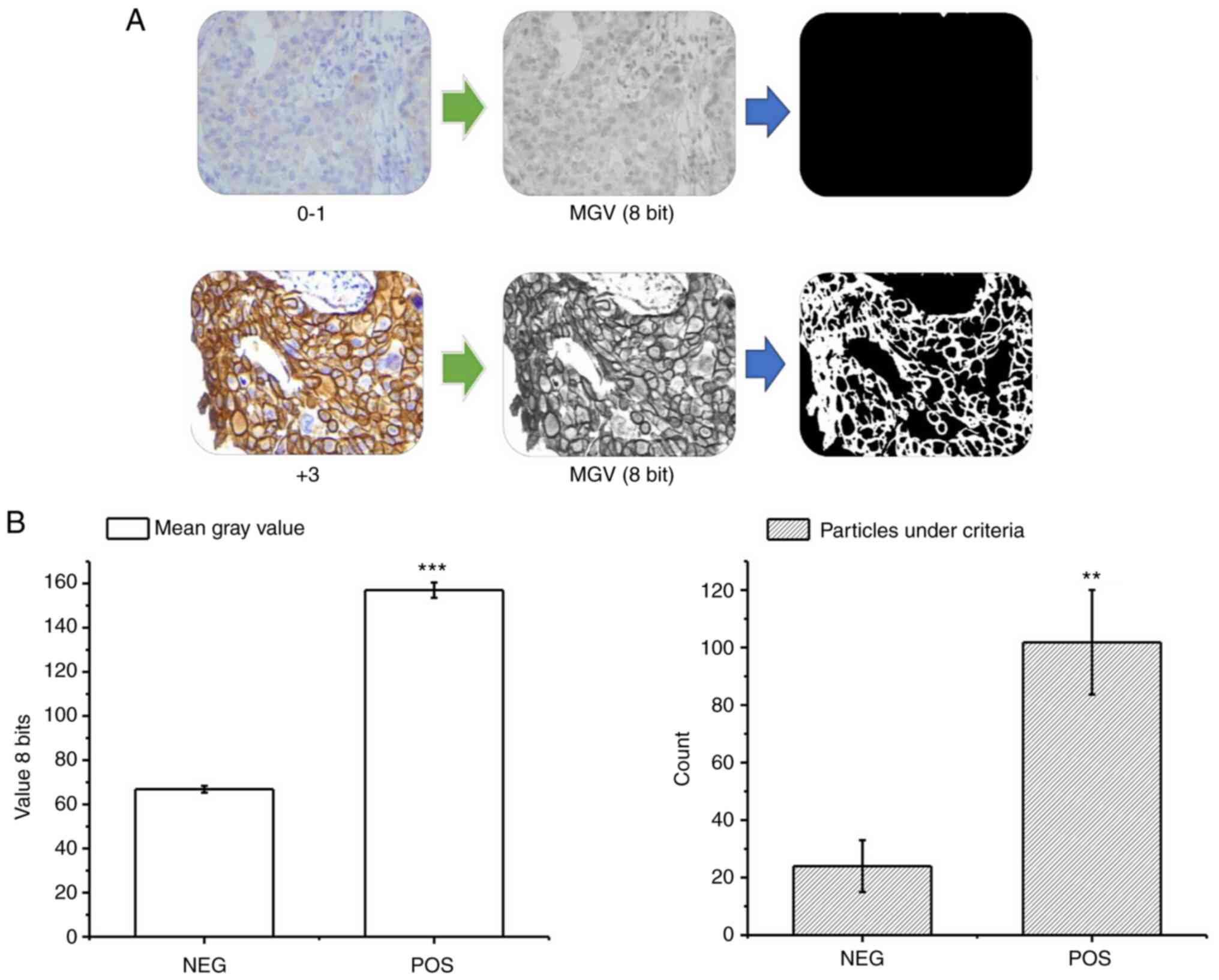
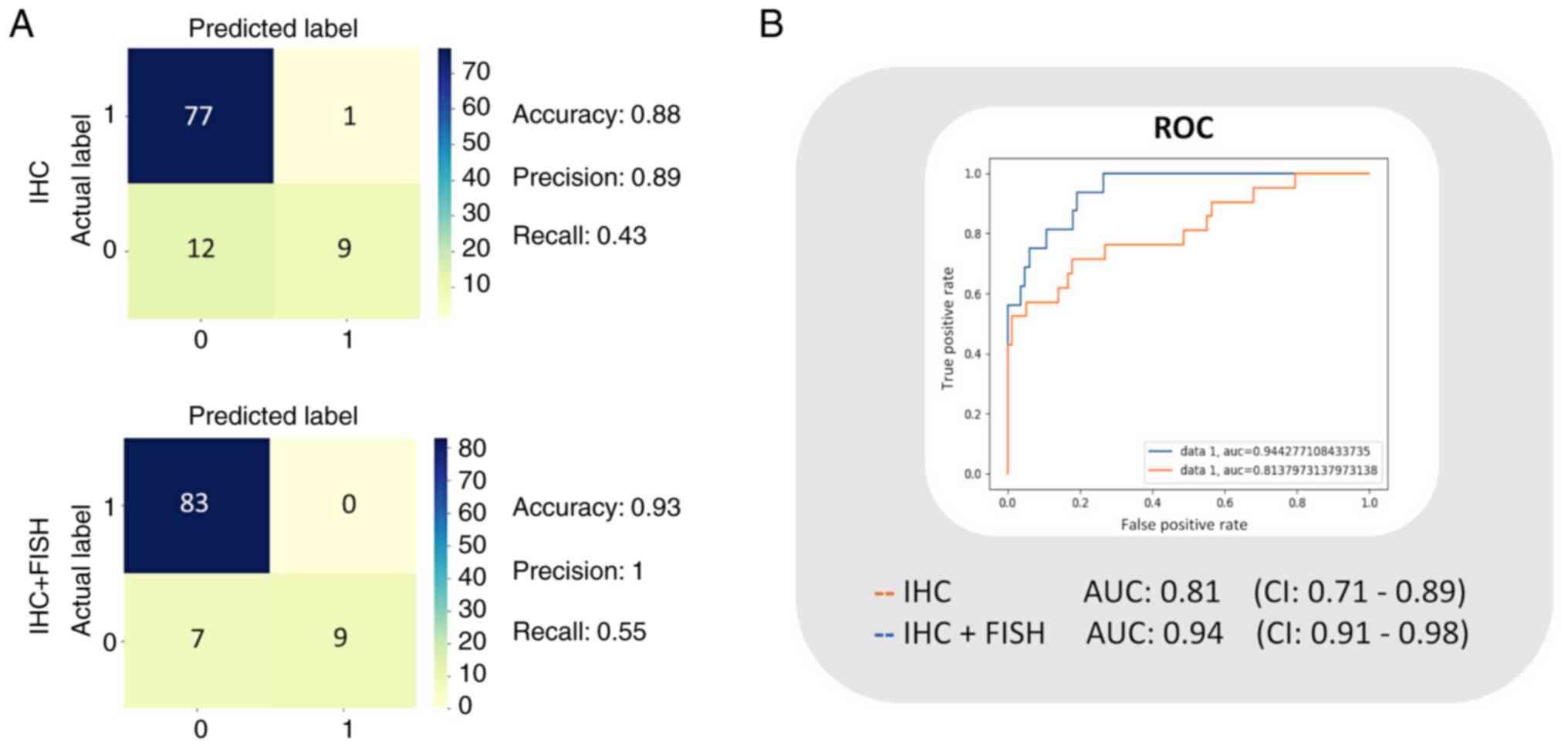
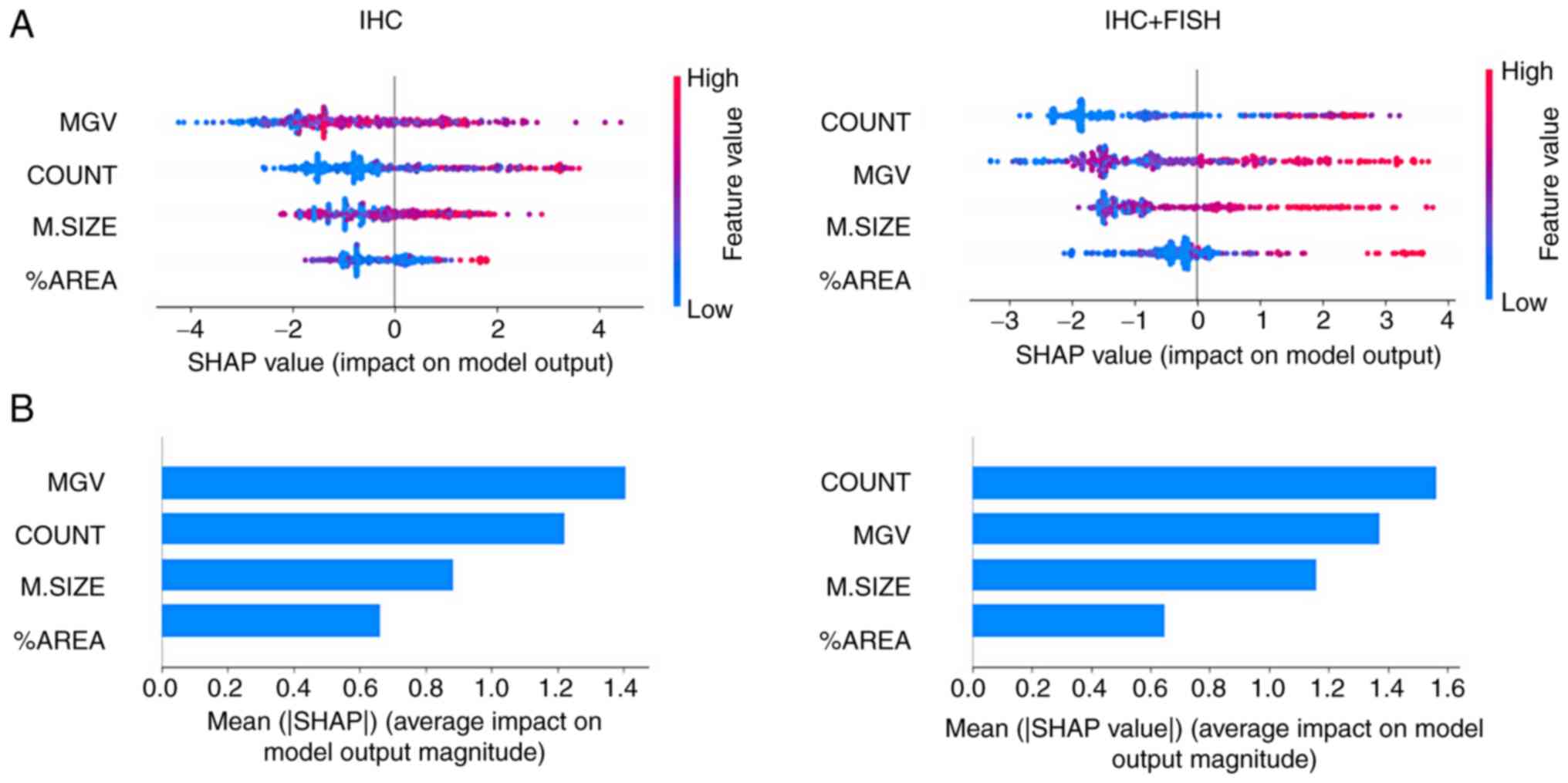
|
1
|
Bray F, Ferlay J, Soerjomataram I, Siegel
RL, Torre LA and Jemal A: Global cancer statistics 2018: GLOBOCAN
estimates of incidence and mortality worldwide for 36 cancers in
185 countries. CA Cancer J Clin. 68:394–424. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Waks AG and Winer EP: Breast cancer
treatment: A review. JAMA. 321:288–300. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Banegas MP, Püschel K, Martínez-Gutiérrez
J, Anderson JC and Thompson B: Perceived and objective breast
cancer risk assessment in Chilean women living in an underserved
area. Cancer Epidemiol Biomarkers Prev. 21:1716–1721. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Icaza G, Núñez L and Bugueño H:
Epidemiological analysis of breast cancer mortality in women in
Chile. Rev Med Chil. 145:106–114. 2017.(in Spanish). View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Thomssen C, Balic M, Harbeck N and Gnant
M: St. Gallen/Vienna 2021: A brief summary of the consensus
discussion on customizing therapies for women with early breast
cancer. Breast Care (Basel). 16:135–143. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Balic M, Thomssen C, Würstlein R, Gnant M
and Harbeck N: St. Gallen/Vienna 2019: A brief summary of the
consensus discussion on the optimal primary breast cancer
treatment. Breast Care (Basel). 14:103–110. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Li C, Bian X, Liu Z, Wang X, Song X, Zhao
W, Liu Y and Yu Z: Effectiveness and safety of pyrotinib-based
therapy in patients with HER2-positive metastatic breast cancer: A
real-world retrospective study. Cancer Med. 10:8352–8364. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Wang B, Ding W, Sun K, Wang X, Xu L and
Teng X: Impact of the 2018 ASCO/CAP guidelines on HER2 fluorescence
in situ hybridization interpretation in invasive breast cancers
with immunohistochemically equivocal results. Sci Rep. 9:167262019.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Masuda N, Lee SJ, Ohtani S, Im YH, Lee ES,
Yokota I, Kuroi K, Im SA, Park BW, Kim SB, et al: Adjuvant
capecitabine for breast cancer after preoperative chemotherapy. N
Engl J Med. 376:2147–2159. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Slomski A: Adjuvant therapy for
HER2-positive breast cancer. JAMA. 322:11342019. View Article : Google Scholar
|
|
11
|
Gown AM: Current issues in ER and HER2
testing by IHC in breast cancer. Mod Pathol. 21 (Suppl 2):S8–S15.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Press MF, Seoane JA, Curtis C, Quinaux E,
Guzman R, Sauter G, Eiermann W, Mackey JR, Robert N, Pienkowski T,
et al: Assessment of ERBB2/HER2 status in HER2-equivocal breast
cancers by FISH and 2013/2014 ASCO-CAP guidelines. JAMA Oncol.
5:366–375. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Gupta S, Neumeister V, McGuire J, Song YS,
Acs B, Ho K, Weidler J, Wong W, Rhees B, Bates M, et al:
Quantitative assessments and clinical outcomes in HER2 equivocal
2018 ASCO/CAP ISH group 4 breast cancer. NPJ Breast Cancer.
5:282019. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Díaz-Serrano A, Angulo B, Dominguez C,
Pazo-Cid R, Salud A, Jiménez-Fonseca P, Leon A, Galan MC, Alsina M,
Rivera F, et al: Genomic profiling of HER2-positive gastric cancer:
PI3K/Akt/mTOR pathway as predictor of outcomes in HER2-positive
advanced gastric cancer treated with trastuzumab. Oncologist.
23:1092–1102. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Jensen K, Krusenstjerna-Hafstrøm R, Lohse
J, Petersen KH and Derand H: A novel quantitative
immunohistochemistry method for precise protein measurements
directly in formalin-fixed, paraffin-embedded specimens: Analytical
performance measuring HER2. Mod Pathol. 30:180–193. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Goddard KAB, Weinmann S, Richert-Boe K,
Chen C, Bulkley J and Wax C: HER2 evaluation and its impact on
breast cancer treatment decisions. Public Health Genomics. 15:1–10.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Wolff AC, Hammond MEH, Hicks DG, Dowsett
M, McShane LM, Allison KH, Allred DC, Bartlett JM, Bilous M,
Fitzgibbons P, et al: Recommendations for human epidermal growth
factor receptor 2 testing in breast cancer: American society of
clinical oncology/college of American pathologists clinical
practice guideline update. Arch Pathol Lab Med. 138:241–256. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Fu R, Ma X, Bian Z and Ma J: Digital
separation of diaminobenzidine-stained tissues via an automatic
color-filtering for immunohistochemical quantification. Biomed Opt
Express. 6:544–558. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Morelli P, Porazzi E, Ruspini M and Banfi
G: Analysis of errors in histology by root cause analysis: A pilot
study. J Prev Med Hyg. 54:90–96. 2013.PubMed/NCBI
|
|
20
|
Qaiser T, Mukherjee A, Reddy Pb C,
Munugoti SD, Tallam V, Pitkäaho T, Lehtimäki T, Naughton T, Berseth
M, Pedraza A, et al: HER2 challenge contest: A detailed assessment
of automated HER2 scoring algorithms in whole slide images of
breast cancer tissues. Histopathology. 72:227–238. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Varghese F, Bukhari AB, Malhotra R and De
A: IHC profiler: An open source plugin for the quantitative
evaluation and automated scoring of immunohistochemistry images of
human tissue samples. PLoS One. 9:e968012014. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
McCabe A, Dolled-Filhart M, Camp RL and
Rimm DL: Automated quantitative analysis (AQUA) of in situ protein
expression, antibody concentration, and prognosis. J Natl Cancer
Inst. 97:1808–1815. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Larson JS, Goodman LJ, Tan Y, Defazio-Eli
L, Paquet AC, Cook JW, Rivera A, Frankson K, Bose J, Chen L, et al:
Analytical validation of a highly quantitative, sensitive,
accurate, and reproducible assay (HERmark) for the measurement of
HER2 total protein and HER2 homodimers in FFPE breast cancer tumor
specimens. Patholog Res Int. 2010:8141762010.PubMed/NCBI
|
|
24
|
Zanconati F, Cusumano P, Tinterri C, Di
Napoli A, Lutke Holzik MF, Poulet B, Dekker L and Sapino A: P205
The 70-gene expression profile, Mammaprint, for breast cancer
patients in mainly European hospitals. Breast. 20:S452011.
View Article : Google Scholar
|
|
25
|
Cronin M, Sangli C, Liu ML, Pho M, Dutta
D, Nguyen A, Jeong J, Wu J, Langone KC and Watson D: Analytical
validation of the Oncotype DX genomic diagnostic test for
recurrence prognosis and therapeutic response prediction in
node-negative, estrogen receptor-positive breast cancer. Clin Chem.
53:1084–1091. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Nielsen TO, Parker JS, Leung S, Voduc D,
Ebbert M, Vickery T, Davies SR, Snider J, Stijleman IJ, Reed J, et
al: A comparison of PAM50 intrinsic subtyping with
immunohistochemistry and clinical prognostic factors in
tamoxifen-treated estrogen receptor-positive breast cancer. Clin
Cancer Res. 16:5222–5232. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Economic Comission for Latin America and
the Caribbean, . Plan for self-sufficiency in health matters in
Latin America and the Caribbean: Lines of action and proposals
(LC/TS.2021/115). United Nations Publication; 2021, [cited 2022 Jun
7]. Available from:. https://repositorio.cepal.org/bitstream/handle/11362/47253/1/S2100556_en.pdf
|
|
28
|
Hey T, Butler K, Jackson S and
Thiyagalingam J: Machine learning and big scientific data. Philos
Trans A Math Phys Eng Sci. 378:201900542020.PubMed/NCBI
|
|
29
|
Larmuseau M, Sluydts M, Theuwissen K,
Duprezd L, Dhaenec T and Cottenier S: Race against the machine: Can
deep learning recognize microstructures as well as the trained
human eye? Scr Mater. 193:33–37. 2021. View Article : Google Scholar
|
|
30
|
Shah P, Kendall F, Khozin S, Goosen R, Hu
J, Laramie J, Ringel M and Schork N: Artificial intelligence and
machine learning in clinical development: A translational
perspective. NPJ Digit Med. 2:692019. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Dong J and Dong J: A 19-miRNA support
vector machine classifier and a 6-miRNA risk score system designed
for ovarian cancer patients. Oncol Rep. 41:3233–3243.
2019.PubMed/NCBI
|
|
32
|
Esteva A, Kuprel B, Novoa RA, Ko J,
Swetter SM, Blau HM and Thrun S: Dermatologist-level classification
of skin cancer with deep neural networks. Nature. 542:115–118.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Elsharawy KA, Gerds TA, Rakha EA and
Dalton LW: Artificial intelligence grading of breast cancer: A
promising method to refine prognostic classification for management
precision. Histopathology. 79:187–199. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Trivizakis E, Ioannidis GS, Melissianos
VD, Papadakis GZ, Tsatsakis A, Spandidos DA and Marias K: A novel
deep learning architecture outperforming ‘off-the-shelf’ transfer
learning and feature-based methods in the automated assessment of
mammographic breast density. Oncol Rep. 42:2009–2015.
2019.PubMed/NCBI
|
|
35
|
Rajkomar A, Oren E, Chen K, Dai AM, Hajaj
N, Hardt M, Liu PJ, Liu X, Marcus J, Sun M, et al: Scalable and
accurate deep learning with electronic health records. NPJ Digit
Med. 1:182018. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Wilbur DC, Smith ML, Cornell LD,
Andryushkin A and Pettus JR: Automated identification of glomeruli
and synchronised review of special stains in renal biopsies by
machine learning and slide registration: A cross-institutional
study. Histopathology. 79:499–508. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Burrell J: How the machine ‘thinks’:
Understanding opacity in machine learning algorithms. Big Data Soc.
3:1–12. 2016. View Article : Google Scholar
|
|
38
|
Rashidi HH, Tran NK, Betts EV, Howell LP
and Green R: Artificial intelligence and machine learning in
pathology: The present landscape of supervised methods. Acad
Pathol. 6:23742895198730882019. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Ahmad A and Quegan S: Analysis of maximum
likelihood classification on multispectral data. Appl Math Sci.
6:6425–6436. 2012.
|
|
40
|
Adadi A and Berrada M: Peeking inside the
black-box: A survey on explainable artificial intelligence (XAI).
IEEE Access. 6:52138–52160. 2018. View Article : Google Scholar
|
|
41
|
Arrieta AB, Díaz-Rodríguez N, Del Ser J,
Bennetot A, Tabik S, Barbado A, Garciag S, Gil-Lopez S, Molina D,
Benjamins R, et al: Explainable artificial intelligence (XAI):
Concepts, taxonomies, opportunities and challenges toward
responsible AI. Inf Fusion. 58:82–115. 2020. View Article : Google Scholar
|
|
42
|
Tosun AB, Pullara F, Becich MJ, Taylor DL,
Fine JL and Chennubhotla SC: Explainable AI (xAI) for anatomic
pathology. Adv Anat Pathol. 27:241–250. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Goodman B and Flaxman S: European union
regulations on algorithmic decision-making and a ‘right to
explanation’. AI Mag. 38:50–57. 2017.
|
|
44
|
Huang G, Huang GB, Song S and You K:
Trends in extreme learning machines: A review. Neural Netw.
61:32–48. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Zinchuk V and Zinchuk O: Quantitative
colocalization analysis of confocal fluorescence microscopy images.
Curr Protoc Cell Biol. Chapter 4: Unit 4.19. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Fereidouni F, Bader AN and Gerritsen HC:
Spectral phasor analysis allows rapid and reliable unmixing of
fluorescence microscopy spectral images. Opt Express.
20:12729–12741. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Alberts B, Johnson A, Lewis J, Raff M,
Roberts K and Walter P: Looking at the structure of cells in the
microscope. molecular Biology of the cell. 4th edition. New York:
Garland Science; 2002
|
|
48
|
Moulisová V, Jiřík M, Schindler C,
Červenková L, Pálek R, Rosendorf J, Arlt J, Bolek L, Šůsová S,
Nietzsche S, et al: Novel morphological multi-scale evaluation
system for quality assessment of decellularized liver scaffolds. J
Tissue Eng. 11:20417314209211212020. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Aguilera A, Pezoa R and Rodríguez-Delherbe
A: A novel ensemble feature selection method for pixel-level
segmentation of HER2 overexpression. Complex Intell Syst.
8:5489–5510. 2022. View Article : Google Scholar
|
|
50
|
Taylor CR and Rudbeck L:
Immunohistochemical staining methods. Sixth Edition. Agilent
Technologies; Santa Clara, CA: pp. 22–76. 2021
|
|
51
|
Dabbs DJ: Diagnostic immunohistochemistry:
Theranostic and genomic applications. Sixth Edition. Elsevier;
Amsterdam: pp. 15–54. 2021, PubMed/NCBI
|
|
52
|
Lin F and Prichard J: Handbook of
Practical Immunohistochemistry. Second Edition. Springer; New York,
NY: pp. 220–224. 2015
|
|
53
|
Untch M, Harbeck N, Huober J, von
Minckwitz G, Gerber B, Kreipe HH, Liedtke C, Marschner N, Möbus V,
Scheithauer H, et al: Primary therapy of patients with early breast
cancer: Evidence, controversies, consensus. Geburtshilfe
Frauenheilkd. 75:556–565. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Harbeck N, Penault-Llorca F, Cortes J,
Gnant M, Houssami N, Poortmans P, Ruddy K, Tsang J and Cardoso F:
Breast cancer. Nat Rev Dis Primers. 5:662019. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Koboldt DC, Fulton RS, McLellan MD,
Schmidt H, Kalicki-Veizer J, McMichael JF, Fulton LL, Dooling DJ,
Ding L, Mardis ER, et al: Comprehensive molecular portraits of
human breast tumours. Nature. 490:61–70. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Hariri N, Roma AA, Hasteh F, Walavalkar V
and Fadare O: Phenotypic alterations in breast cancer associated
with neoadjuvant chemotherapy: A comparison with baseline rates of
change. Ann Diagn Pathol. 31:14–19. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Brasó-Maristany F, Griguolo G, Pascual T,
Paré L, Nuciforo P, Llombart-Cussac A, Bermejo B, Oliveira M,
Morales S, Martínez N, et al: Phenotypic changes of HER2-positive
breast cancer during and after dual HER2 blockade. Nat Commun.
11:3852020. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Powers DMW: Evaluation: From precision,
recall and F-measure to ROC, informedness, markedness and
correlation. arXiv:. 2010:160612020.
|
|
59
|
Sharma D, Kumar S and Narasimhan B:
Estrogen alpha receptor antagonists for the treatment of breast
cancer: A review. Chem Cent J. 12:1072018. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Favretto D, Cosmi E, Ragazzi E, Visentin
S, Tucci M, Fais P, Cecchetto G, Zanardo V, Viel G and Ferrara SD:
Cord blood metabolomic profiling in intrauterine growth
restriction. Anal Bioanal Chem. 402:1109–1121. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Lokhov PG, Dashtiev MI, Moshkovskii SA and
Archakov AI: Metabolite profiling of blood plasma of patients with
prostate cancer. Metabolomics. 6:156–163. 2010. View Article : Google Scholar
|
|
62
|
Ellin J, Haskvitz A, Premraj P, Shields K,
Smith M, Stratman C and Wrenn M: Interoperability between anatomic
pathology laboratory information systems and digital pathology
systems. Madison: Digital Pathology Association; pp. 1–10. 2011
|
|
63
|
Pathology and Tissue Imaging | MetaSystems
[Internet]. [cited 2020 Aug 10]. Available from. https://metasystems-international.com/us/applications/patho/
|
|
64
|
Patología digital, . Leica Biosystems
[Internet]. [cited 2020 Aug 10]. Available from:. https://www.leicabiosystems.com/es/patologia-digital/
|
|
65
|
Dunbier AK, Anderson H, Ghazoui Z, Salter
J, Parker JS, Perou CM, Smith IE and Dowsett M: Association between
breast cancer subtypes and response to neoadjuvant anastrozole.
Steroids. 76:736–740. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
66
|
Becker S: A historic and scientific review
of breast cancer: The next global healthcare challenge. Int J
Gynecol Obstet. 131 (Suppl 1):S36–S39. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
67
|
Planes-Laine G, Rochigneux P, Bertucci F,
Chrétien AS, Viens P, Sabatier R and Gonçalves A: PD-1/PD-l1
targeting in breast cancer: The first clinical evidences are
emerging. A literature review. Cancers (Basel). 11:10332019.
View Article : Google Scholar : PubMed/NCBI
|
|
68
|
Lozano C, Córdova C, Marchant I, Zúñiga R,
Ochova P, Ramírez-Barrantes R, González-Arriagada WA, Rodriguez B
and Olivero P: Intracellular aggregated TRPV1 is associated with
lower survival in breast cancer patients. Breast Cancer (Dove Med
Press). 10:161–168. 2018.PubMed/NCBI
|
|
69
|
Campbell KJ, Dhayade S, Ferrari N, Sims
AH, Johnson E, Mason SM, Dickson A, Ryan KM, Kalna G, Edwards J, et
al: MCL-1 is a prognostic indicator and drug target in breast
cancer. Cell Death Dis. 9:192018. View Article : Google Scholar : PubMed/NCBI
|
|
70
|
Zhang Y, Zheng A, Lu H, Jin Z, Peng Z and
Jin F: The expression and prognostic significance of claudin-8 and
androgen receptor in breast cancer. Onco Targets Ther.
13:3437–3448. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
71
|
Nanda R, Liu MC, Yau C, Shatsky R, Pusztai
L, Wallace A, Chien AJ, Forero-Torres A, Ellis E, Han H, et al:
Effect of pembrolizumab plus neoadjuvant chemotherapy on pathologic
complete response in women with early-stage breast cancer: An
analysis of the ongoing phase 2 adaptively randomized I-SPY2 trial.
JAMA Oncol. 6:676–684. 2020. View Article : Google Scholar : PubMed/NCBI
|