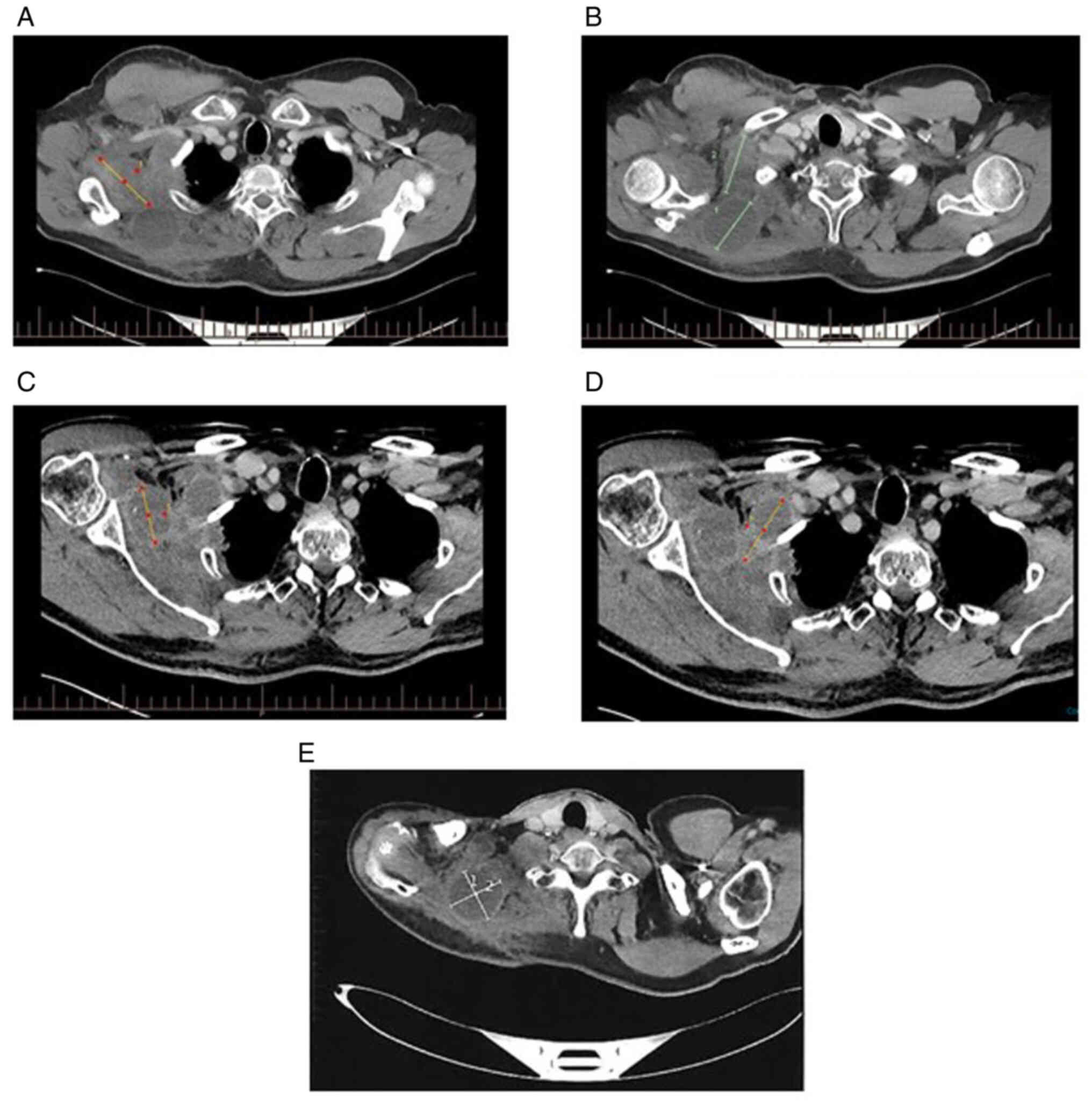
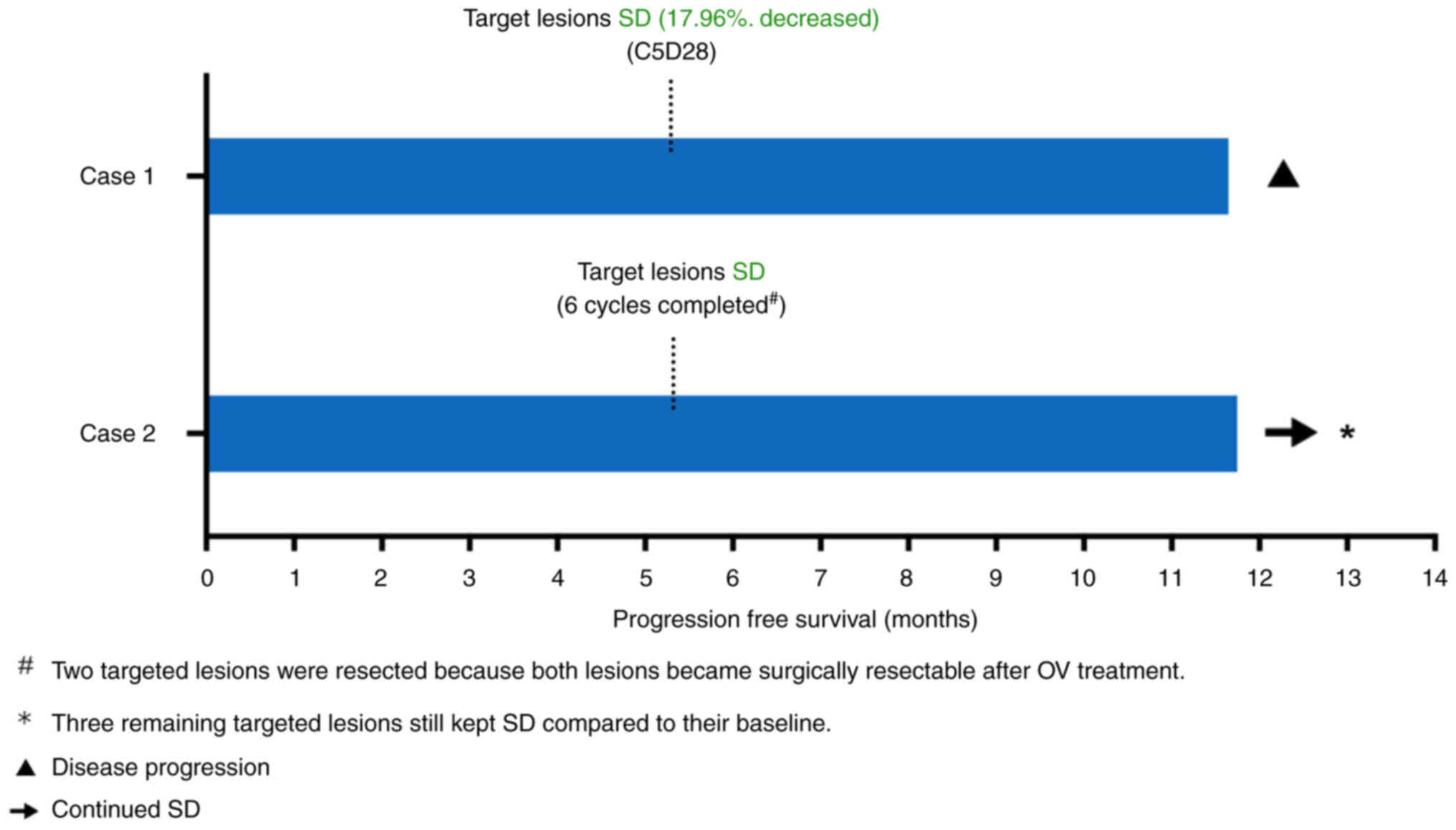
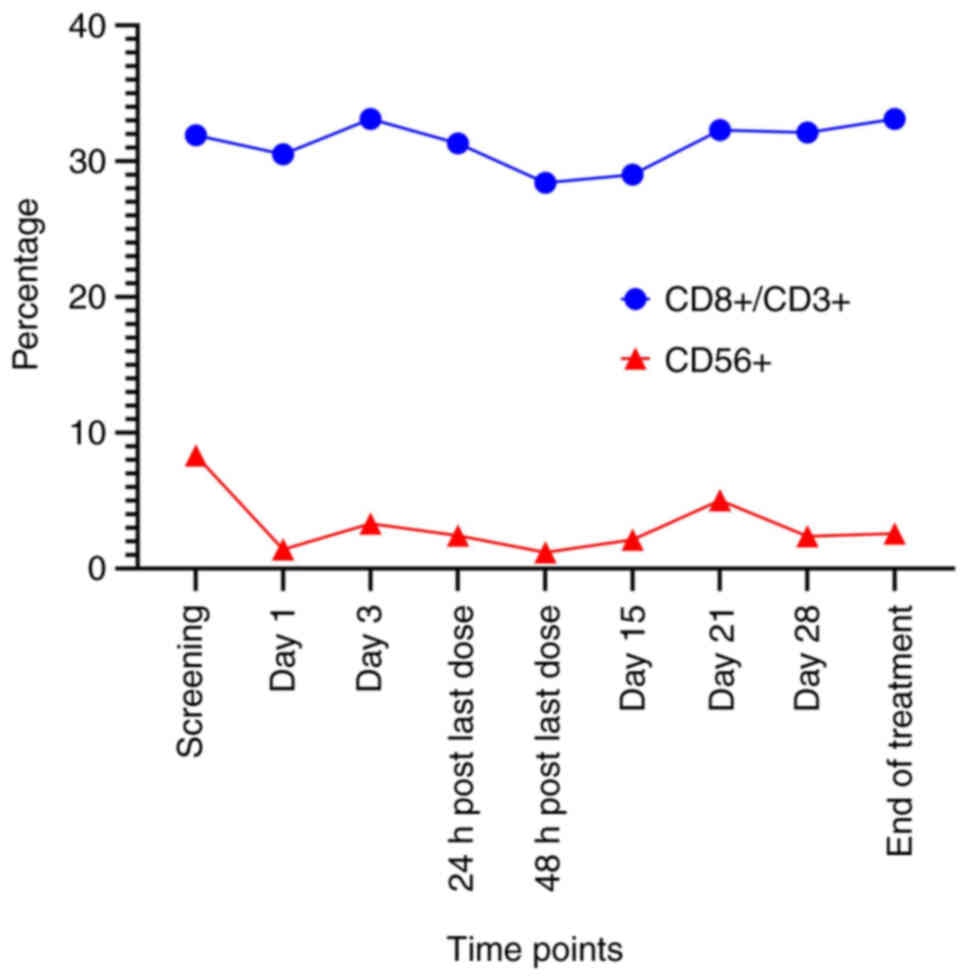
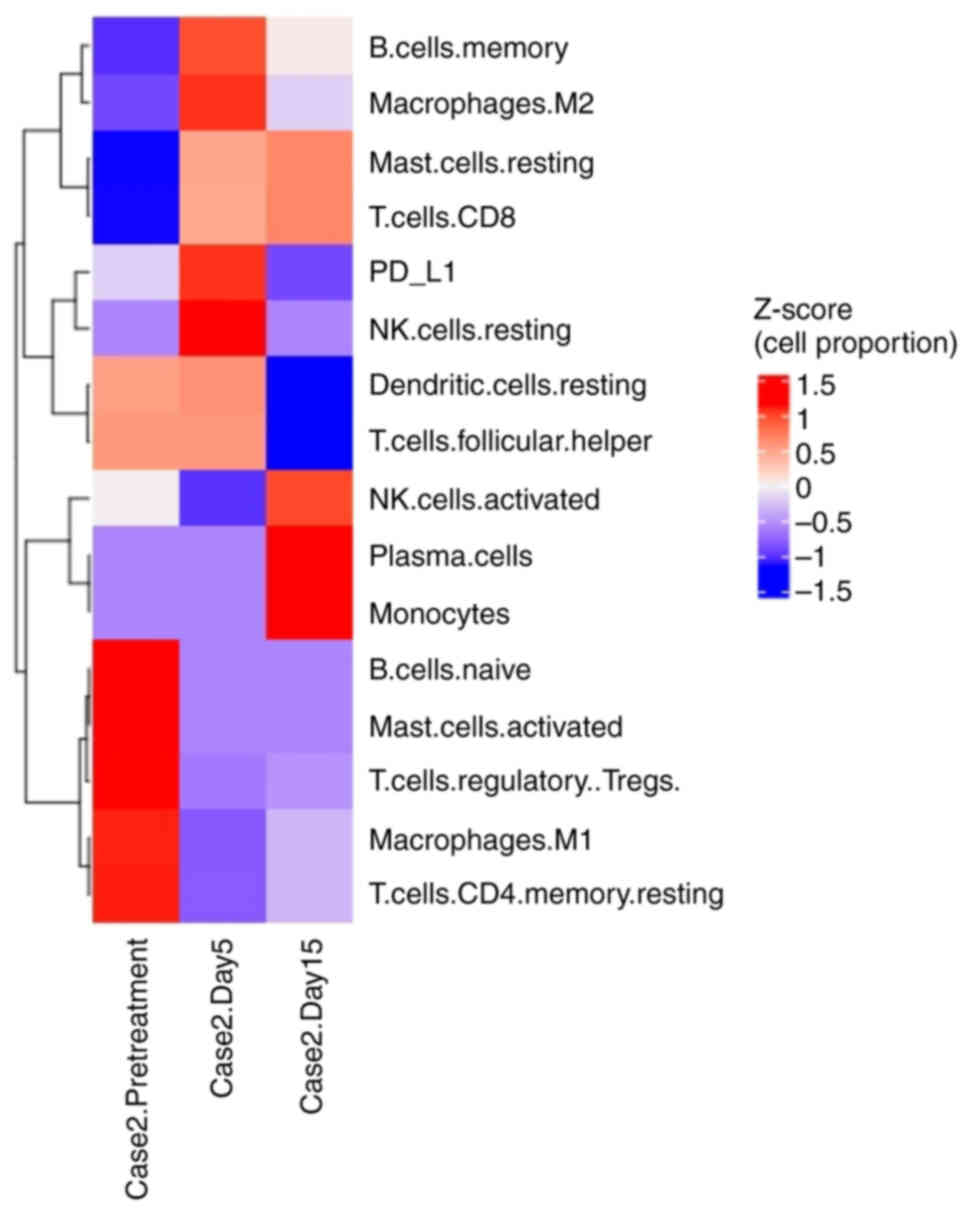
|
1
|
NCCN, . NCCN Clinical Practice Guidelines
in Oncology (NCCN Guidelines®) Soft Tissue Sarcoma
version 2.2023. https://www.nccn.org/guidelines/guidelines-detail?category=1&id=1464August
3–2023
|
|
2
|
Damerell V, Pepper MS and Prince S:
Molecular mechanisms underpinning sarcomas and implications for
current and future therapy. Signal Transduct Target Ther.
6:2462021. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Cocco E, Scaltriti M and Drilon A: NTRK
fusion-positive cancers and TRK inhibitor therapy. Nat Rev Clin
Oncol. 15:731–747. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Albarrán V, Villamayor ML, Pozas J,
Chamorro J, Rosero DI, San Román M, Guerrero P, Pérez de Aguado P,
Calvo JC, García de Quevedo C, et al: Current landscape of
immunotherapy for advanced sarcoma. Cancers. 15:22872023.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Macedo N, Miller DM, Haq R and Kaufman HL:
Clinical landscape of oncolytic virus research in 2020. J
Immunother Cancer. 8:e0014862020. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Su Y, Su C and Qin L: Current landscape
and perspective of oncolytic viruses and their combination
therapies. Transl Oncol. 25:1015302022. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Chouljenko DV, Ding J, Lee IF, Murad YM,
Bu X, Liu G, Delwar Z, Sun Y, Yu S, Samudio I, et al: Induction of
durable antitumor response by a novel oncolytic herpesvirus
expressing multiple immunomodulatory transgenes. Biomedicines.
8:4842020. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Ding J, Murad YM, Sun Y, Lee IF, Samudio
I, Liu X, Jia WW and Zhao R: Pre-existing HSV-1 immunity enhances
anticancer efficacy of a novel immune-stimulating oncolytic virus.
Viruses. 14:23272022. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Hokland P and Heron I: Analysis of the
lymphocyte distribution during Isopaque-Ficoll isolation of
mononuclear cells from human peripheral blood. J Immunol Methods.
32:31–39. 1980. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
National Institues of Health National
Cancer Institute, . Common Terminology Criteria for Adverse Events
(CTCAE). Version 5.0. https://ctep.cancer.gov/protocoldevelopment/electronic_applications/docs/CTCAE_v5_Quick_Reference_8.5×11.pdfNovember
27–2017
|
|
11
|
Eisenhauer EA, Therasse P, Bogaerts J,
Schwartz LH, Sargent D, Ford R, Dancey J, Arbuck S, Gwyther S,
Mooney M, et al: New response evaluation criteria in solid tumours:
Revised RECIST guideline (version 1.1). Eur J Cancer. 45:228–247.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Seymour L, Bogaerts J, Perrone A, Ford R,
Schwartz LH, Mandrekar S, Lin NU, Litière S, Dancey J, Chen A, et
al: iRECIST: Guidelines for response criteria for use in trials
testing immunotherapeutics. Lancet Oncol. 18:e143–e152. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Amin MB, Greene FL, Edge SB, Compton CC,
Gershenwald JE, Brookland RK, Meyer L, Gress DM, Byrd DR and
Winchester DP: The eighth edition AJCC cancer staging manual:
continuing to build a bridge from a population-based to a more
‘personalized’ approach to cancer staging. CA Cancer J Clin.
67:93–99. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Yao T, Liu JJ, Zhao LJ, Zhou JY, Wang JQ,
Wang Y, Wang ZQ, Wei LH, Wang JL and Li XP: Identification of new
fusion genes and their clinical significance in endometrial cancer.
Chin Med J (Engl). 132:1314–1321. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Monga V, Mani H, Hirbe A and Milhem M:
Non-conventional treatments for conventional chondrosarcoma.
Cancers. 12:1–12. 2020. View Article : Google Scholar
|
|
16
|
Xie L, Xu J, Sun X, Liu K, Li X, He F, Liu
X, Gu J, Lv Z, Yang R, et al: Apatinib for treatment of inoperable
metastatic or locally advanced chondrosarcoma: What we can learn
about the biological behavior of chondrosarcoma from a two-center
study. Cancer Manag Res. 12:3513–3525. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Ayodele O and Abdul Razak AR:
Immunotherapy in soft-tissue sarcoma. Curr Oncol. 27 (Suppl
1):S17–S23. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Bergsma EJ, Elgawly M, Mancuso D, Orr R,
Vuskovich T and Seligson ND: Atezolizumab as the first systemic
therapy approved for alveolar soft part sarcoma. Ann Pharmacother.
19:106002802311874212023.
|
|
19
|
D'Angelo SP, Mahoney MR, Van Tine BA,
Atkins J, Milhem MM, Jahagirdar BN, Antonescu CR, Horvath E, Tap
WD, Schwartz GK and Streicher H: Nivolumab with or without
ipilimumab treatment for metastatic sarcoma (Alliance A091401): Two
open-label, non-comparative, randomised, phase 2 trials. Lancet
Oncol. 19:416–426. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Kelly CM, Antonescu CR, Bowler T, Munhoz
R, Chi P, Dickson MA, Gounder MM, Keohan ML, Movva S, Dholakia R,
et al: Objective response rate among patients with locally advanced
or metastatic sarcoma treated with talimogene laherparepvec in
combination with pembrolizumab: A phase 2 clinical trial. JAMA
Oncol. 6:402–408. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Toulmonde M, Cousin S, Kind M, Guegan JP,
Bessede A, Le Loarer F, Perret R, Cantarel C, Bellera C and
Italiano A: Randomized phase 2 trial of intravenous oncolytic virus
JX-594 combined with low-dose cyclophosphamide in patients with
advanced soft-tissue sarcoma. J Hematol OncolJ Hematol Oncol.
15:1492022. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Barber GN: Host defense, viruses and
apoptosis. Cell Death Differ. 8:113–126. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Goldstein D and Laszlo J: The role of
interferon in cancer therapy: A current perspective. CA Cancer J
Clin. 38:258–277. 1988. View Article : Google Scholar : PubMed/NCBI
|