Introduction
Gastric-type endocervical adenocarcinoma (GEA) is a
specific subtype of cervical adenocarcinoma that is not associated
with human papillomavirus (HPV) and accounts for only 1–3% of
cervical adenocarcinomas (1). The
disease is characterized by vaginal mucin (2,3) or
watery discharge (4,5), but irregular vaginal bleeding can also
occur. However, these clinical manifestations lack specificity and
can also be seen in other types of endocervical lesions. Due to the
insidious nature of GEA and the extremely low success rate of
routine screening and biopsy, a missed or delayed diagnosis, or a
misdiagnosis are common problems (6–8). In
addition to diagnostic difficulties, the poor prognosis of GEA is a
concern. This is largely attributable to an unusual pattern of
dissemination, including lymphatic spread and invasion of the
parametrial space, which results in poor progression-free survival
and overall survival times. In 104 patients with GEA (9), the mean survival time for patients
with stage I disease was reported to be ~5 years, while stage II
was ~38.1 months, stage III was ~22.8 months and stage IV was ~5.4
months. In addition, the literature states that the 5-year specific
overall survival rate of GEA is only 30–42% (7). It is notable that even in stage I, the
disease is often poorly differentiated (high grade) (6), is resistant to standard therapy
(10) and often involves metastases
to sites such as the ovaries, peritoneum or omentum. Therefore, it
is important to improve the preoperative diagnosis of GEA.
Through careful analysis of the relevant literature
and the cases reported in the present study, it was found that
imaging examination methods have significant advantages in the
diagnosis of GEA. For example, in transvaginal ultrasound (TVUS)
examination, the lesion is multilocular cystic and communicates
with the uterine surface to form tunnel-like changes (5,8), while
in contrast-enhanced US (CEUS), a ‘fast rise and fast fall’ curve
is formed (11,12), and finally, in magnetic resonance
(MR) examination, a ‘cosmic-type’ image is noted, with a small cyst
or solid component in the center of the lesion surrounded by a
large cyst (13–15). These typical imaging findings are
essential for the diagnosis and differential diagnosis of GEA. With
the rapid development of science and technology, artificial
intelligence (AI) has been widely used in the field of medical
imaging. AI technology has made significant progress, enabling
models to automatically recognize and analyze a variety of medical
images, such as X-rays, computed tomography and magnetic resonance
(MR) imaging scans. In particular, it has great potential in tumor
detection. AI technology can identify and locate the tumor area,
and provide accurate measurement results, thereby greatly improving
the accuracy and efficiency of radiologists (16).
The present study describes two complete cases of
GEA, and the most comprehensive and reliable imaging features of
the three aforementioned imaging examinations are described in
detail, which play an important role in the diagnosis of the
disease. These imaging features can provide effective clues for
clinicians to develop more accurate and effective treatment plans
to improve the prognosis of affected patients.
Case reports
Case one
Patient A, a 46-year-old woman, presented to The
Women's Hospital, School of Medicine, Zhejiang University
(Hangzhou, China) in February 2021 with moderate vaginal discharge
that was clear in color or occasionally yellow in color, no
abdominal pain or abdominal distension, and no contact bleeding.
The patient had no family history of genetic, neurological,
psychiatric, infectious or similar diseases. A ThinPrep®
cytological test (TCT) was performed and the patient was negative
for intraepithelial lesions or malignancy (NILM), but was
HPV-positive. The results of the TVUS suggested multiple cysts in
the cervix. In June 2021, the vaginal fluid of the patient had been
greenish-yellow and a cervical biopsy had shown chronic
inflammation of the cervical mucosa with squamous cell
proliferation. The diagnosis was further confirmed by TVUS
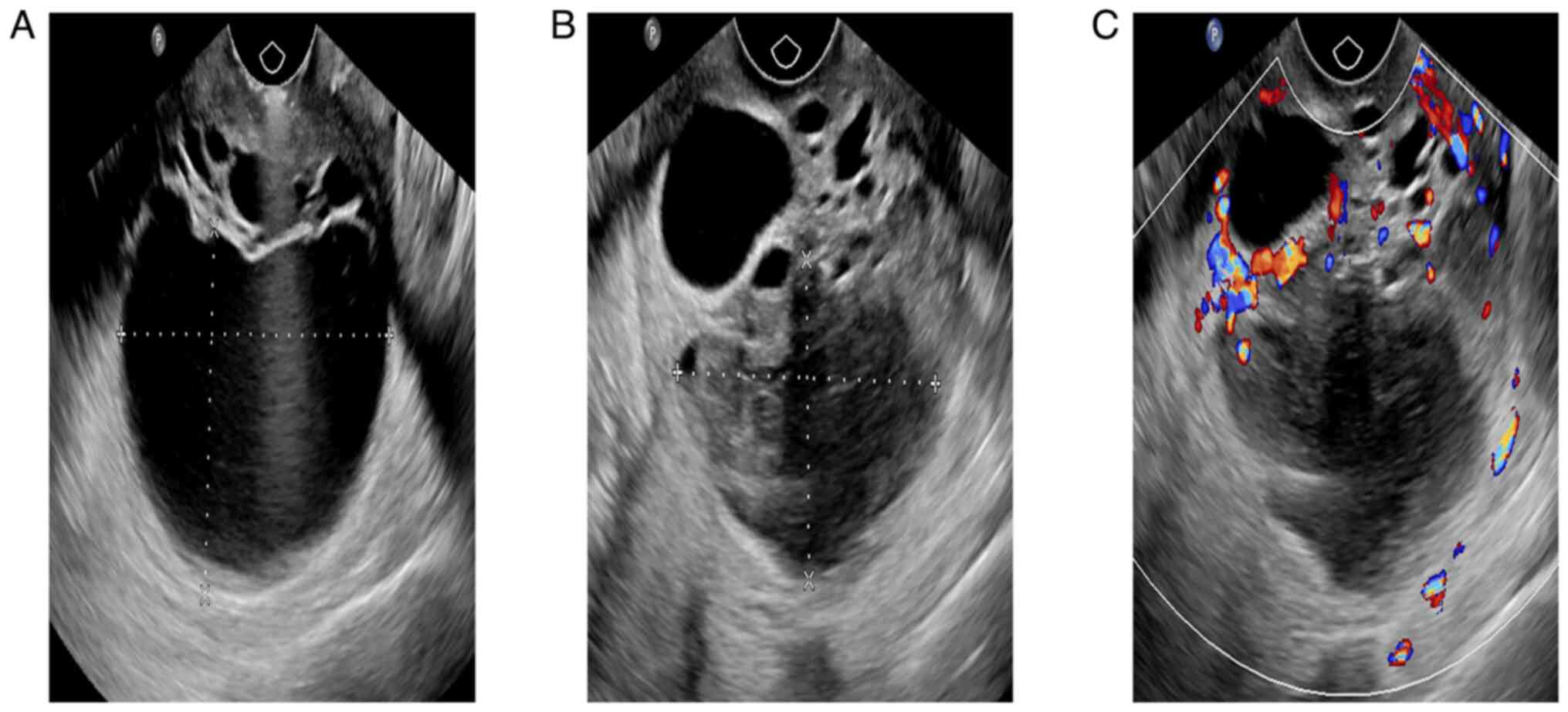
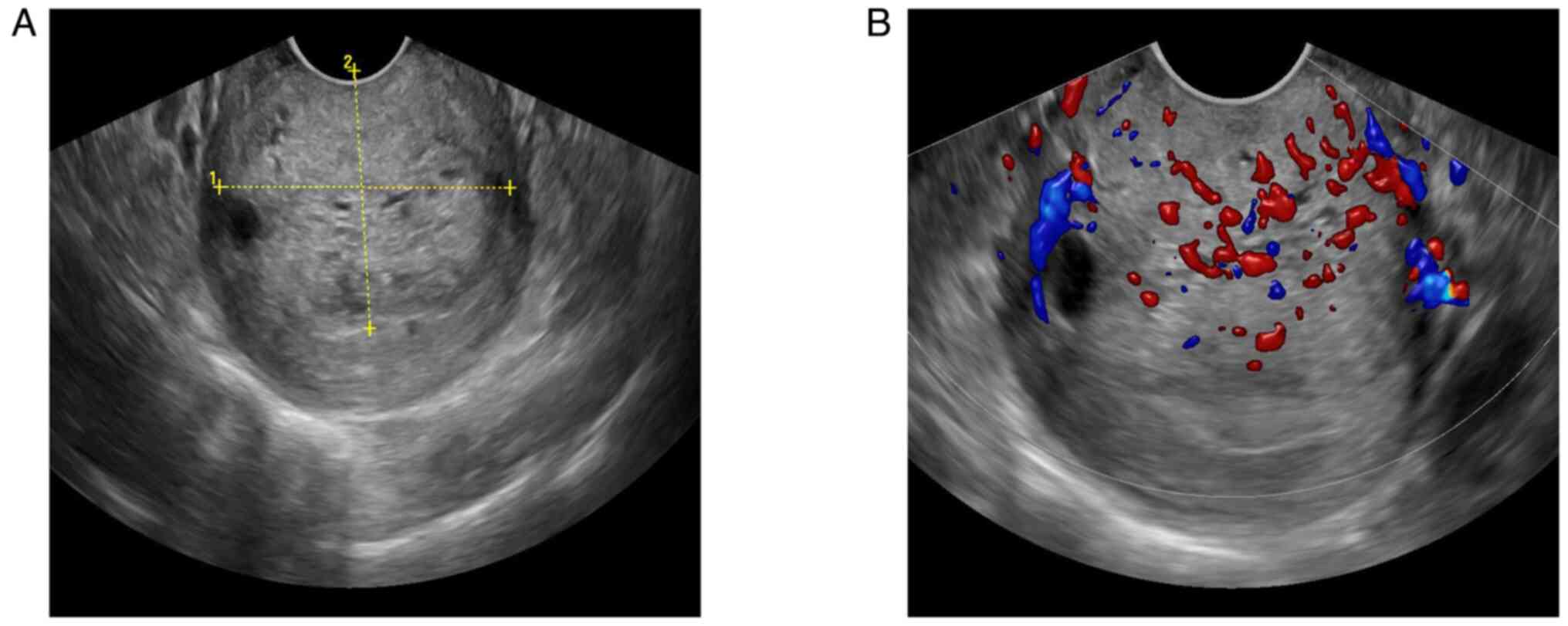
(Fig. 1A-C), which suggested a
multilocular cystic solid mass with irregular thickening of the
cyst wall in the cervix. The fluid in some of the cysts was
viscous, the solid regions were small and honeycomb-like and color
Doppler showed a rich blood supply. In August 2021, the results of
another TVUS showed that the cervical cysts had increased in size,
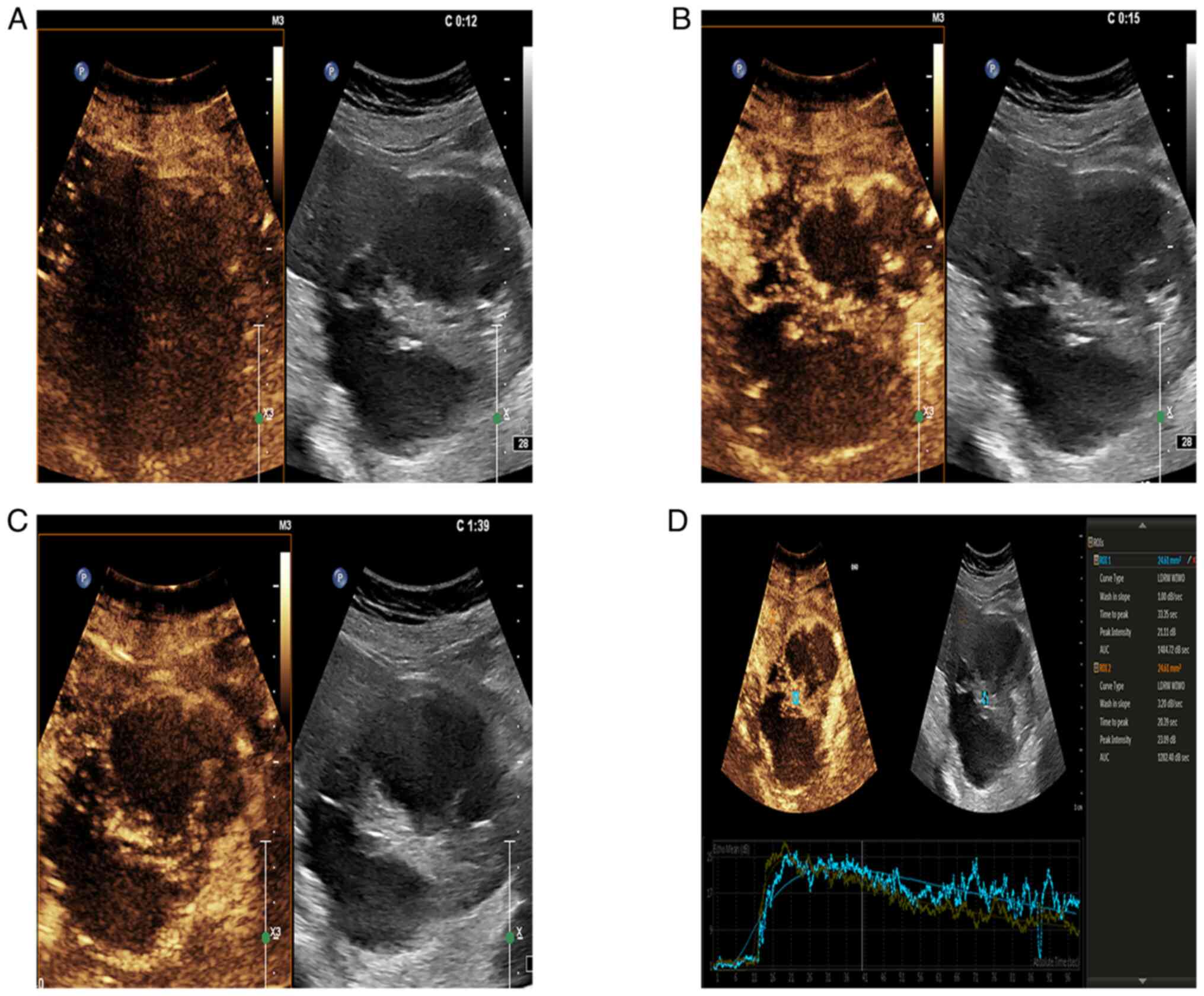
and the larger cysts were ~7.5 cm in diameter. The CEUS (Fig. 2A-D) suggested that the contrast
agent began to fill at 12 sec, reached a peak at 15 sec and began
to weaken at 1 min and 39 sec. The solid regions showed
hyperenhancement, which was indicative of cervical cancer.
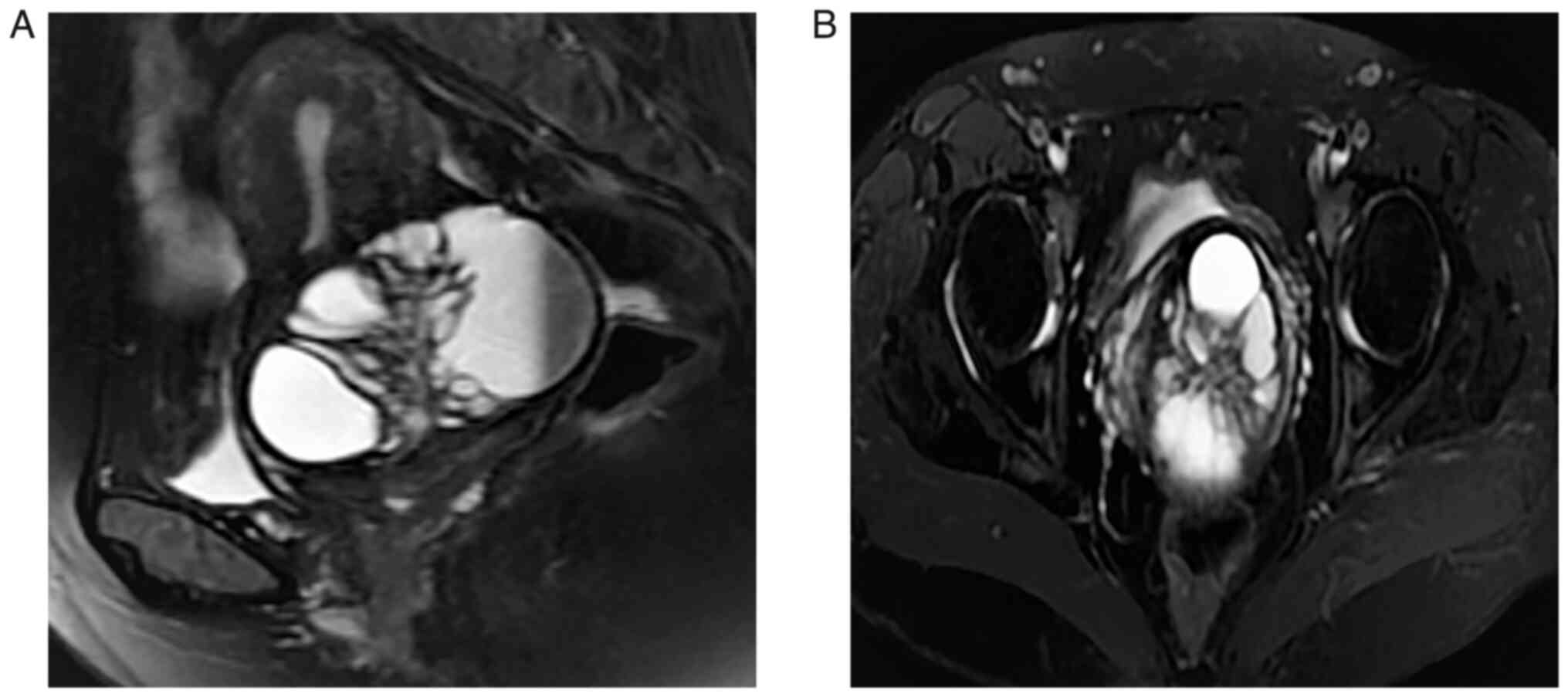
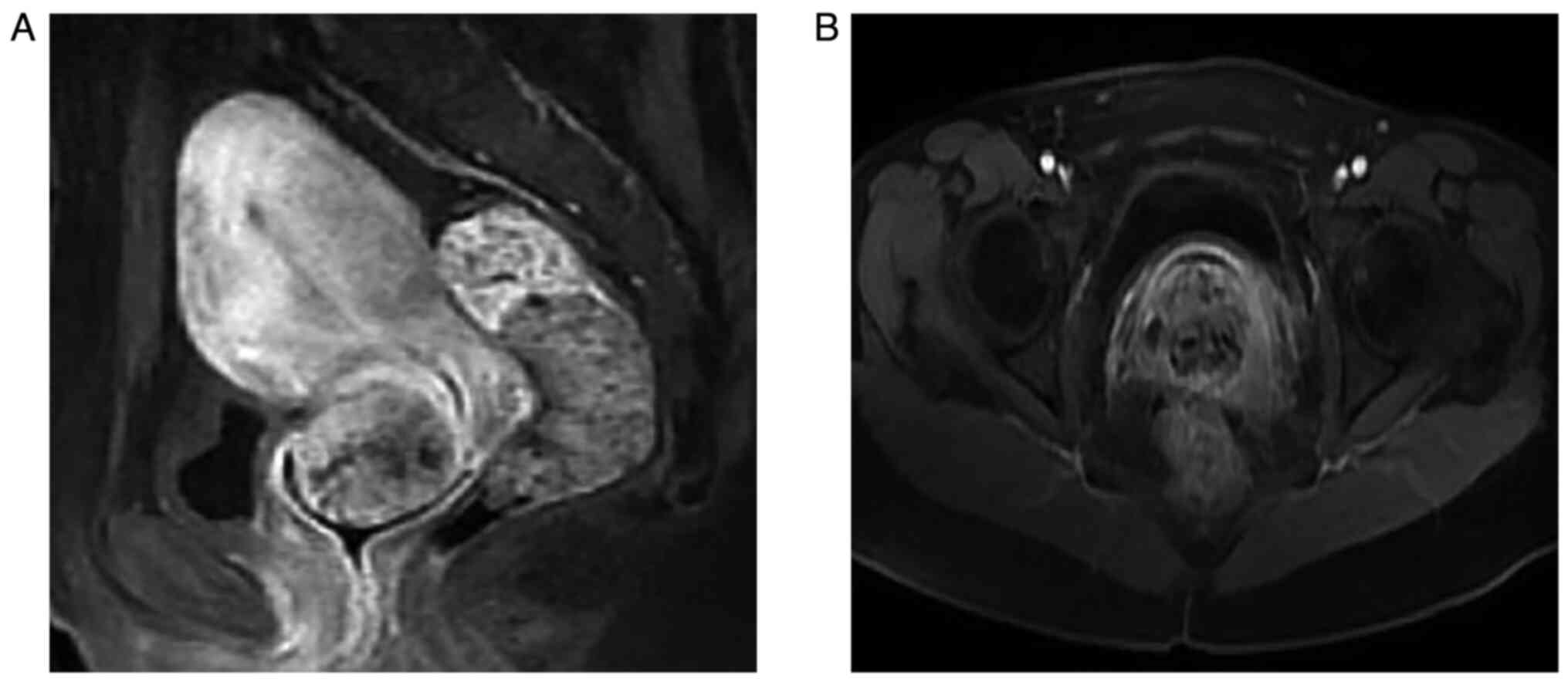
Contrast-enhanced MR of the pelvis (Fig. 3A and B) showed abnormal signal foci
in the cervix with multiple cystic lesions, hypointensity on
T1-weighted imaging (T1WI) and hyperintensity on T2WI of the cystic
fluid, and moderate enhancement of the cyst wall and cyst septum,
leading to the consideration of GEA. Furthermore, hydrocolpos were
still observed after conization. After discussion, the decision was
made to perform US-guided biopsy of the cervical mass.
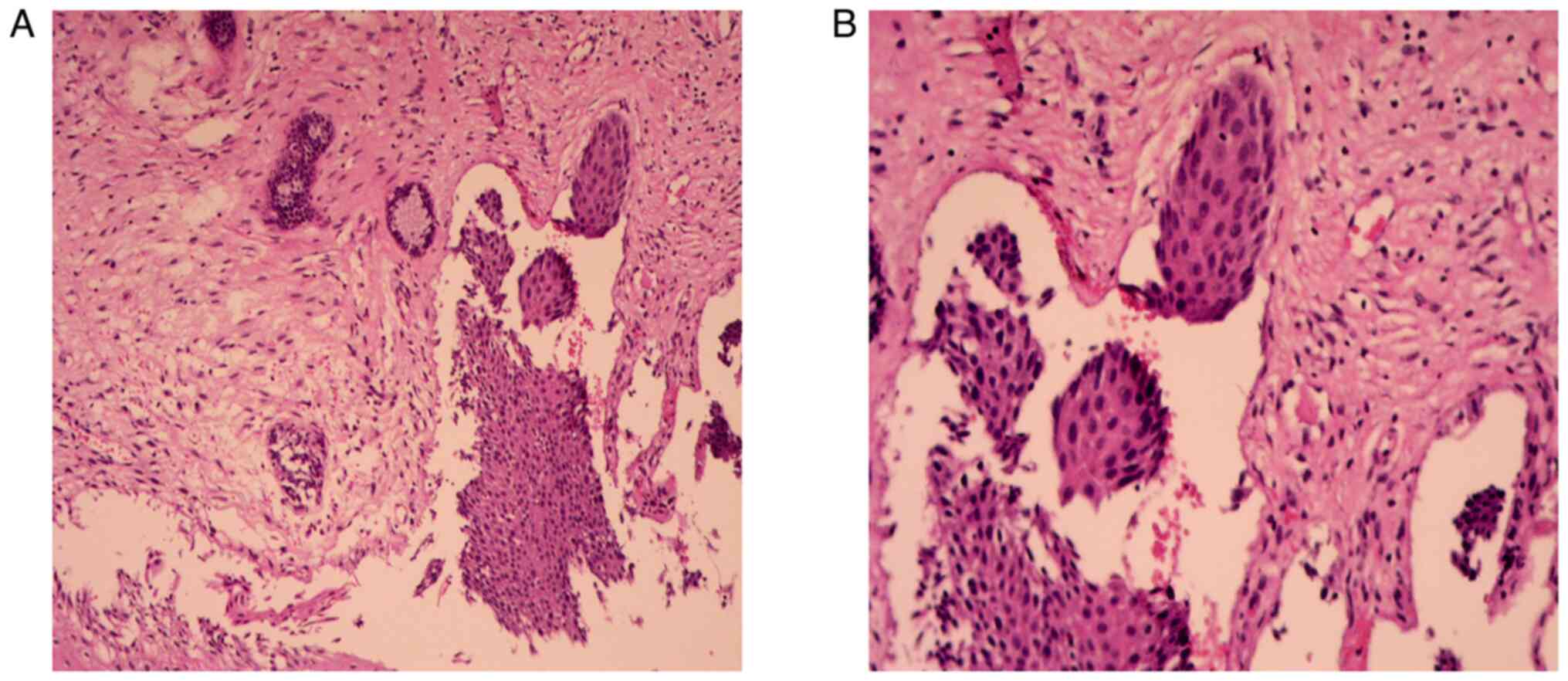
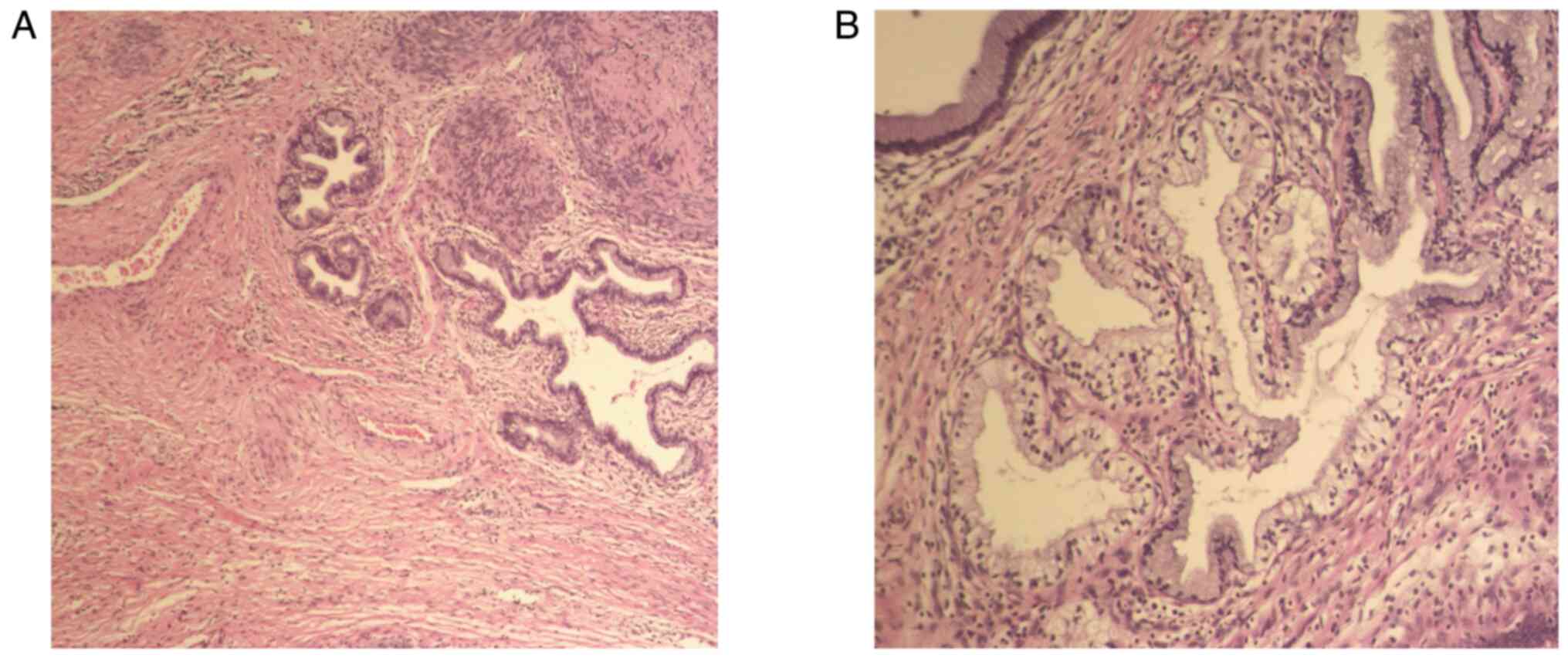
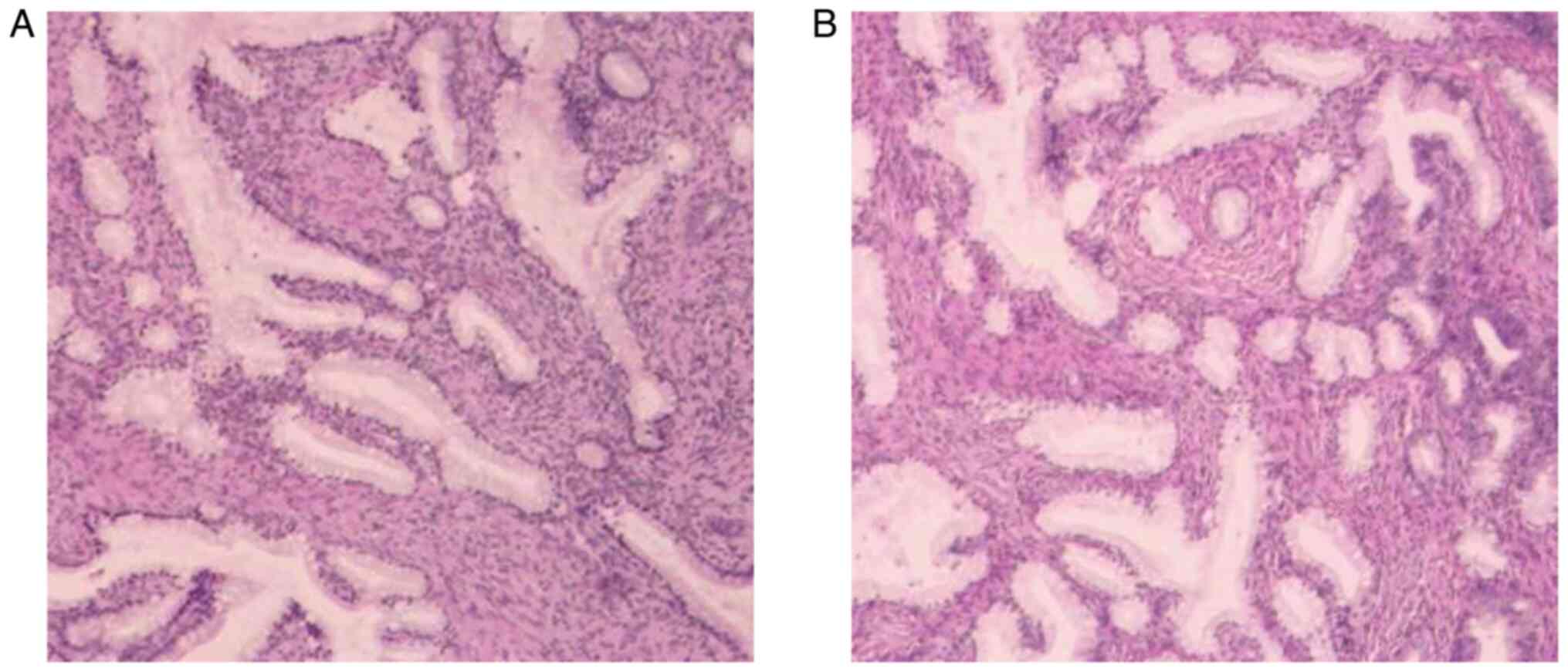
Postoperative pathological (Fig. 4A and
B) results of the cervical mass showed smooth muscle tissue and
cervical mucosa chronic inflammation. After obtaining informed
consent from the patient, an abdominal extrafascial hysterectomy,
bilateral salpingo-oophorectomy and intestinal adhesiolysis were
performed at the Women's Hospital, School of Medicine, Zhejiang
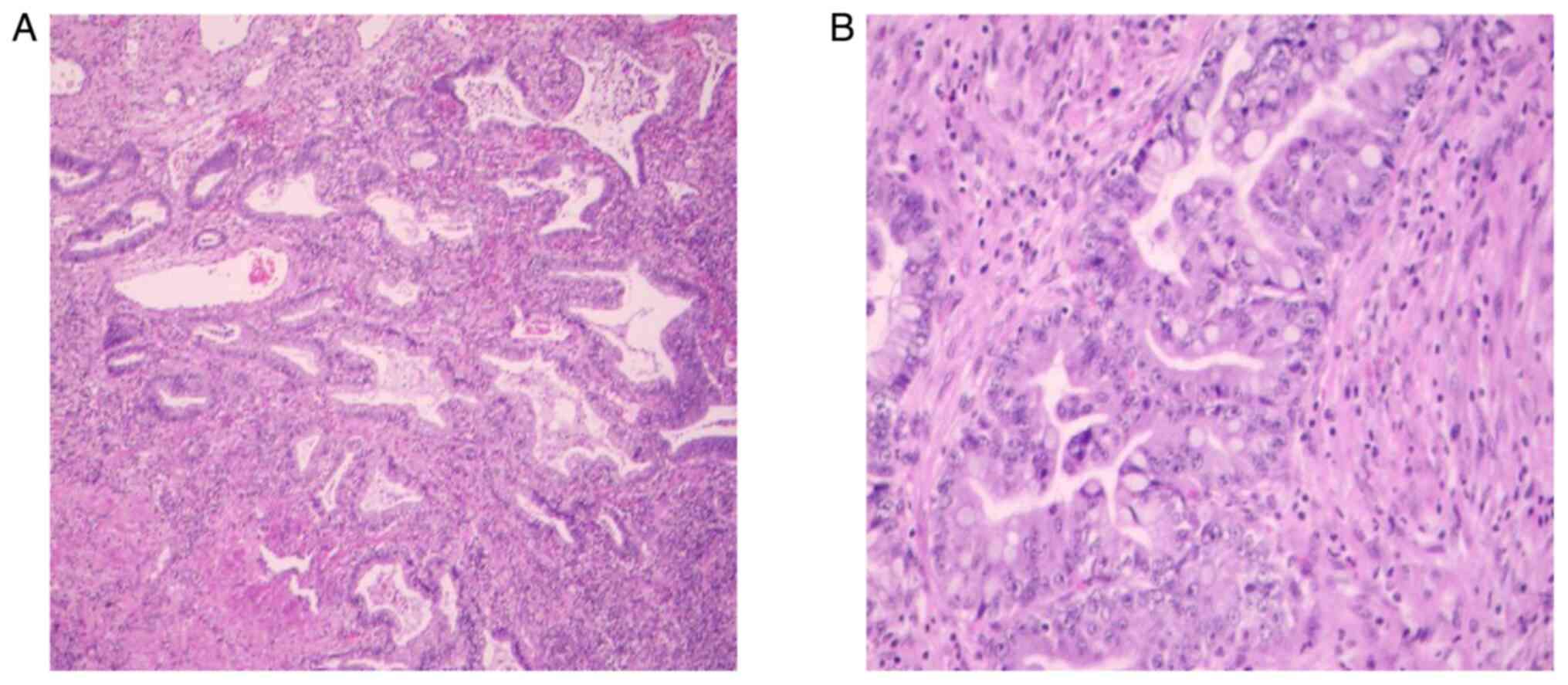
University (Hangzhou, China). Postoperative pathology (Fig. 5A and B) showed atypical hyperplasia
of the cervical lobular gland with malignant transformation
(well-differentiated gastric adenocarcinoma), invading more than
two-thirds of the stroma, involving the isthmus intima and
fibromuscular layer. The periuterine and bilateral ovarian vessels
were not invaded by the tumor. The results were in accordance with
International Federation of Gynecology and Obstetrics (FIGO) stage
IB3 disease (17).
Immunohistochemical staining results: ER (+), PR (+), CEA (+), P53
(+; wild-type) and P16 (+), Ki-67 (+ 5%), PAX2 (−), MUC-6 (+),
Mucin5AC (+) and SMA (+). Alcian blue, and periodic acid Schiff
staining results: (−/+). Postoperative adjuvant chemotherapy with
paclitaxel and carboplatin was administered four times, once a
month. After 2 months of adjuvant chemotherapy, the patient was
doing well, with pelvic contrast-enhanced MR showing no obvious
recurrence or metastasis. The patient has been followed up for
>2 years and no recurrence has been found. This case suggests
that clinical symptoms with multimodal imaging should be combined
to reduce misdiagnosis or missed diagnosis.
Case two
Patient B, a 39-year-old woman, presented to The
Women's Hospital, School of Medicine, Zhejiang University
(Hangzhou, China) in August 2022 with increased vaginal discharge
for 6 months, which was mucoid and clear in color, accompanied by
frequent urination and no abnormal uterine bleeding. The patient
had no family history of genetic, neurological, psychiatric,
infectious or similar diseases. The results of the TCT showed that
the patient was NILM and HPV-negative. The TVUS (Fig. 6A and B) showed heterogeneous echo
areas in the anterior lip of the neck with clear boundaries,
regular shapes, multiple cystic areas and color Doppler
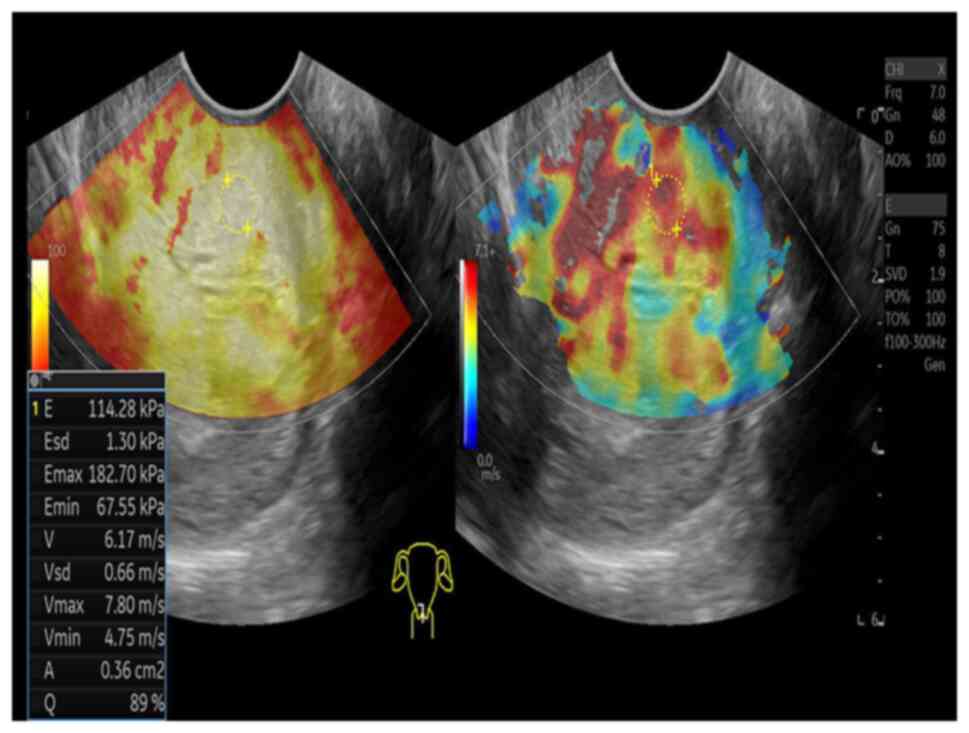
demonstrated a rich blood supply. The shear wave elasticity
(Fig. 7) showed moderately hard
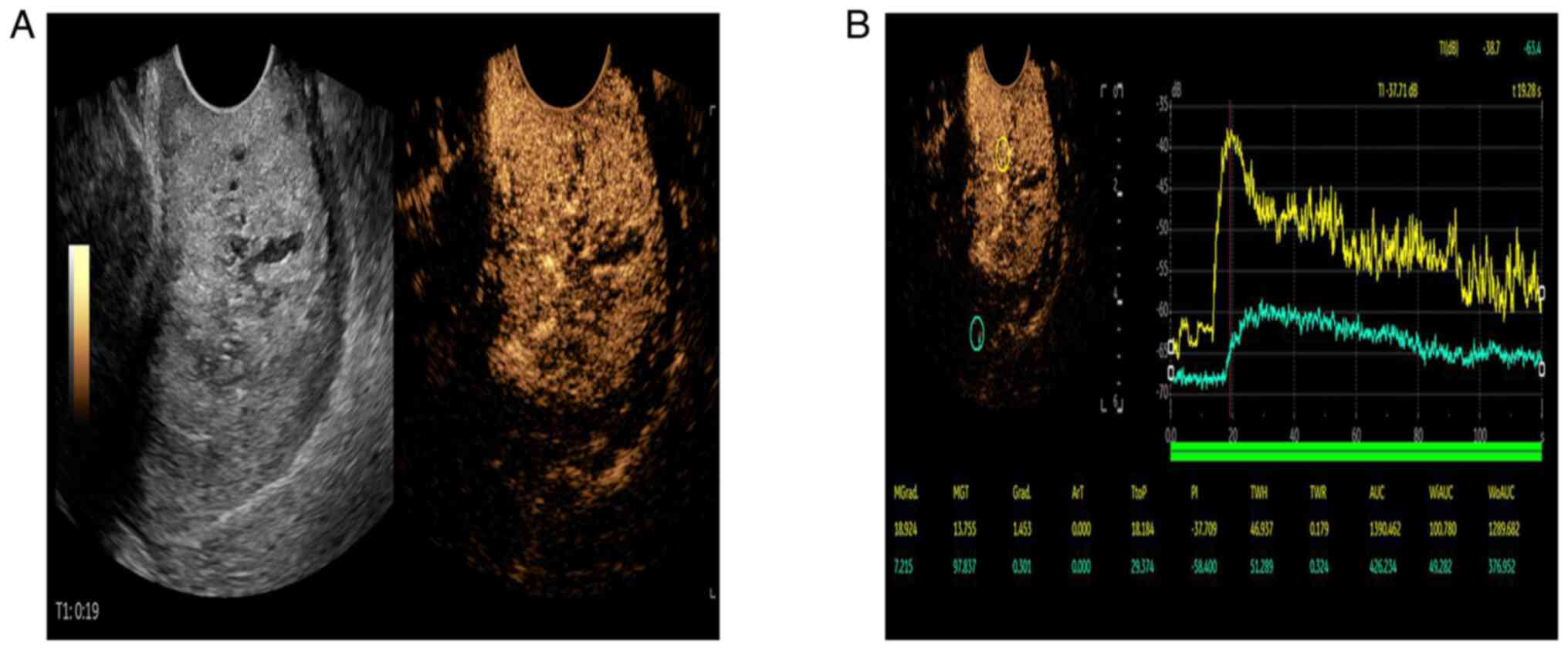
texture, indicating a diagnosis of GEA. For further diagnosis, CEUS
was performed and the results (Fig. 8A
and B) suggested that the hypoechoic area of the anterior
cervical lip began filling at 14 sec and reached the peak at 19
sec. The tumor tissue showed rapid hyperenhancement and then slowly
decreased. Pelvic contrast-enhanced MR (Fig. 9A and B) suggested the presence of a
leiomyoma lesion on the anterior lip of the cervix, with clear
boundaries and patchy long T2 signals, which showed heterogeneous
enhancement after enhanced scanning, and so cervical leiomyoma with
degeneration was considered. The cervical biopsy pathology
(Fig. 10A and B) was indicative of
GEA. After obtaining informed consent, the patient underwent an
abdominal radical hysterectomy, pelvic lymph node dissection,
bilateral salpingo-oophorectomy and intestinal adhesion lysis at
the Women's Hospital, School of Medicine, Zhejiang University. The
postoperative pathological (Fig. 11A
and B) diagnosis was GEA, which infiltrated the entire
interstitial layer and involved the fornix. The tumor did not
invade the periuterine, bilateral ovarian vessels and greater
omental tissue. A total of 33 lymph nodes were not invaded by the
tumor. The immunohistochemical staining results were: Insulin-like
growth factor II m-RNA-binding protein 3 (+), D2-40 (vascular +),
estrogen receptor (−), progesterone receptor (−), carcinoembryonic
antigen (local +), P53 (wild-type), low molecular weight
cytokeratin (+), P16 (+), Ki-67 (+ 60%), MUC-6 (+), programmed cell
death protein 1 (tumor proportion score:-; combined positive
score:<1) and programmed death ligand 1 (tumor proportion
score:-; combined positive score:<1). The results were in
accordance with FIGO stage IIA2. Preoperative examination showed
that the CA199 tumor marker level of the patient was 401.9 U/ml
(normal reference value, ≤37.0 U/ml) (18). At 2 months post-surgery, the CA199
tumor marker level decreased to 86.0 U/ml (normal reference value,
≤37.0 U/ml). At 8 months post-surgery, the CA199 tumor marker level
had recovered to 25.4 U/ml (normal reference value, ≤37.0 U/ml).
The patients were followed up every 3 months for 1–2 years
post-operation, and every 6 months for 3–5 years thereafter.
Currently, patient B has been followed up for nearly 2 years with
no evident recurrence. The key messages in this case are the need
to accurately understand the typical imaging findings of GEA and
the fact that the use of a deep biopsy or conical incision for
suspicious lesions is necessary.
Discussion
The most common TVUS finding of GEA is cervical
hypertrophy with multiple cystic lesions (5). Park et al (8) proposed that 60% of GEA lesions were
multilocular cystic, 40% were solid and 50% of cystic lesions were
accompanied by solid components. The study also indicated that
color Doppler showed a rich blood supply in GEA lesions, which is
helpful for the diagnosis of GEA. In the present study, the two
cases were similar to those observed in the literature, whereby
multiple cystic dark areas were seen in the cervix, and color
Doppler showed an abundant blood supply. By contrast, patient A
showed irregular thickening of the cyst wall, viscous fluid in the
interior and small honeycomb-like changes in the solid part of the
lesion. The cystic dark area of the cervix in patient B
communicated with the cervical surface to form a tunnel-like
change, which was helpful for the diagnosis of GEA.
Compared with TVUS alone, the combined application
of TVUS and CEUS can significantly improve the accuracy of the
diagnosis of GEA. CEUS can greatly improve the sensitivity of the
lesion blood vessels and their small blood flow signals (11), and make real-time dynamic
observations of the capillary blood flow inside the lesion to
determine its perfusion and distribution, and ultimately determine
the microcirculation status inside the lesion tissue (12). Some studies (11) have proposed that the time to peak
(TTP) and peak intensity (PI) of CEUS quantitative parameters have
statistical significance in the differentiation of benign and
malignant cervical tumors, and found that lower TTP values and
higher PI values are indicators for the identification of early
cervical cancer. In cervical cancer, CEUS shows rapid enhancement
of focal tissue, showing a ‘sharp rise and fall’ curve (12). In the present study, in patient A,
CEUS showed rapid hyperenhancement of the lesion with the results
from the tumor tissues (TTP, 20.39 sec; PI, 23.09 dB) and the
control tissue (TTP, 33.35 sec; PI, 21.11 dB). In patient B, CEUS
showed rapid hyperenhancement of the lesion followed by slow
regression with the results from the tumor tissues (TTP, 18.18 sec;
PI, −37.71 dB) and control tissues (TTP, 29.37 sec; PI, −58.40 dB).
The results of CEUS in patients A and B were consistent with the
results of previous studies. The tumor tissues had lower TTP values
and higher PI values than control tissues (11,12).
This may be as the larger the lesion infiltration area, the more
new blood vessels, the less smooth muscle cells and elastic fibers,
and the weaker or less functional the vascular endothelial cells.
As a result, the filling and fading times of the contrast agent are
accelerated, resulting in decreased TTP values and increased PI
values, forming a ‘fast rise and fast fall’ curve (11,12,19).
The difference is that the enhanced mode of CEUS can be ‘fast in
and fast out’ or ‘fast in and slow out’. To the best of our
knowledge, to date, the present study is the first to apply CEUS to
the diagnosis of GEA. Therefore, in the absence of direct
correlation studies, lessons are drawn from the application
experience of CEUS technology in other types of cervical tumors and
combined with the existing GEA-related research data for
speculation and analysis. This may aid the understanding and
evaluation of the potential value of CEUS in GEA.
Currently, MR is the most accurate imaging
examination for the diagnosis of GEA, as it can most effectively
identify the parenchymal component in the cystic lesions, which is
also the key to distinguish GEA from other cervical cystic lesions
(20). Some scholars have found
that GEA often occurs in the cervical stroma, does not form any
exophytic mass in morphology, is located in the upper part of the
cervical canal, often invades the uterine body, and is accompanied
by multiple cystic lesions (6,21). The
characteristic MR findings are multilocular cystic lesions in the
deep part of the cervix, in which the solid region is composed of
multiple cysts of varying sizes (20). In another study (22), patients with GEA exhibited an
isohypointense signal on T1WI, a slightly hyperintense signal on
T2WI, an isointense signal on diffusion-weighted imaging and
heterogeneous enhancement after contrast enhancement. In a
multicenter study (13–15), contrast-enhanced pelvic MR of
patients with GEA showed a cervical isthmic mass with multiple
small cysts and/or a solid region surrounded by large cysts. This
phenomenon was termed the ‘cosmos’ mode, and this pattern was used
to diagnose GEA with an accuracy of 67% (10/15 patients). The
present study reported the cases of two patients. The pelvic
contrast-enhanced MR images of patient A were consistent with those
described in the previous literature and also showed a typical
‘cosmos’ mode. However, the images of patient B did not show this
pattern, possibly due to the difference in presentation caused by
the different degree of invasion of the GEA mass into the cervical
stroma.
In order to improve the diagnostic accuracy of GEA,
in addition to imaging methods, other screening methods must be
used in combination, such as cytological examination (including TCT
examination and HPV examination), colposcopy and cervical biopsy
(23). However, for GEA, some
studies have found that the accuracy of detecting cervical invasive
adenocarcinoma based on Pap smears is only 45–76% (24,25).
In the present study, the preoperative workup of patient A,
including cytology, colposcopy and a biopsy by cervical conization,
was negative. There are two main reasons for the negative results.
One reason (26) is that GEA
lesions are very deep, occur in the cervical canal, do not
penetrate the cortex of the transitional zone and show a skip
infiltration, which can lead to insufficient sampling of the lesion
tissue. The other reason (27) is
that the atypia of GEA itself is easily overlooked in smears, as
its tissue is well differentiated and easily confused with benign
lesions. The retrospective analysis of Papanicolaou smears of
patients with invasive adenocarcinoma of the cervix, which appear
negative, has found false-negative rates as high as 40–60%
(25). After reviewing the
literature on GEA, the Department of Pathology of the Women's
Hospital, School of Medicine, Zhejiang University, re-studied the
previously negative Pap smears, and finally >20 cases of GEA
were diagnosed. It has also been proposed that when a patient has a
large amount of vaginal watery discharge, a vaginal US examination
and cytology smear should be performed. If cervical multiple cysts
and yellow mucus are found in the cytology smear, a HIK1083 latex
agglutination test is recommended, which can identify gastric
mucosal mucin and has a very high specificity (28), which is very helpful for the
diagnosis of GEA.
In conclusion, it is recommended that TVUS be
performed first when a patient presents with a large amount of
vaginal watery discharge. If there are multiple cystic dark areas
with tunnel-like changes in the cervix, CEUS and/or pelvic enhanced
MR should be further used to confirm the diagnosis. In addition, if
the imaging findings are not consistent with the typical imaging
findings of GEA, a biopsy or surgery is recommended to clarify the
lesion type for further treatment. The present study has some
limitations due to the small number of cases and limited clinical
data.
Acknowledgements
Not applicable.
Funding
The present study was supported by grants from the National
Natural Science Foundation of China (grant nos. 81974470 and
82272004) and the Natural Science Foundation of Zhejiang Province
(grant no. LY18H180001).
Availability of data and materials
The data generated in the present study may be
requested from the corresponding author.
Authors' contributions
GK and XF collected clinical data. MW analyzed the
data. QW and YL analyzed and interpreted the data, and wrote the
paper. GK and MW confirm the authenticity of all the raw data. JZ
obtained medical images (TVUS, CEUS and MRI scans). All authors
have read and approved the final manuscript.
Ethics approval and consent to
participate
The present study was approved by the Institutional
Ethics Committee of the Women's Hospital, School of Medicine,
Zhejiang University (approval no. IRB-20230191-R).
Patient consent for publication
As this study was retrospective, the two patients
who had been discharged from the hospital were contacted by
telephone and provided verbal consent for publication. The
requirement for written consent was waived by the Institutional
Ethics Committee of the Women's Hospital, School of Medicine,
Zhejiang University.
Competing interests
The authors declare that they have no competing
interests.
Glossary
Abbreviations
Abbreviations:
|
GEA
|
gastric-type endocervical
adenocarcinoma
|
|
HPV
|
human papillomavirus
|
|
TCT
|
ThinPrep®, cytological test
|
|
TVUS
|
transvaginal ultrasound
|
|
CEUS
|
contrast-enhanced ultrasound
|
|
MR
|
magnetic resonance
|
|
NILM
|
negative for intraepithelial lesion or
malignancy
|
|
FIGO
|
International Federation of Obstetrics
and Gynecology
|
References
|
1
|
McCluggage WG, Singh N and Gilks CB: Key
changes to the World Health Organization (WHO) classification of
female genital tumours introduced in the 5th edition (2020).
Histopathology. 80:762–778. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Holl K, Nowakowski AM, Powell N,
McCluggage WG, Pirog EC, Collas De Souza S, Tjalma WA, Rosenlund M,
Fiander A, Castro Sánchez M, et al: Human papillomavirus prevalence
and type-distribution in cervical glandular neoplasias: Results
from a European multinational epidemiological study. Int J Cancer.
137:2858–2568. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Qian XQ, Wang FF, Liang Y, Chen LL and Wan
XY: Gastric-type mucinous carcinoma with an abnormal increase of
CA199: A case report and literature review. Front Surg.
9:9459842022. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Kerwin CM, Markese M, Moroney MR, Smith LP
and Patel NU: Adenocarcinoma of the uterine cervix, gastric-type
(GAS): A review of the literature focused on pathology and
multimodality imaging. Abdom Radiol (NY). 48:713–723. 2023.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Talia KL and McCluggage WG: The developing
spectrum of gastric-type cervical glandular lesions. Pathology.
50:122–133. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Greenland NY, Wolsky RJ, Darragh TM and
Vohra P: Gastric-type endocervical adenocarcinoma and cervical
cytology: Experience at a general hospital and review of the
literature. Cytopathology. 32:75–83. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Nishio S, Mikami Y, Tokunaga H, Yaegashi
N, Satoh T, Saito M, Okamoto A, Kasamatsu T, Miyamoto T, Shiozawa
T, et al: Analysis of gastric-type mucinous carcinoma of the
uterine cervix-An aggressive tumor with a poor prognosis: A
multi-institutional study. Gynecol Oncol. 153:13–19. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Park SB, Lee JH, Lee YH, Song MJ, Lim KT,
Hong SR and Kim JK: Adenoma malignum of the uterine cervix: Imaging
features with clinicopathologic correlation. Acta Radiol.
54:113–120. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Li G, Jiang W, Gui S and Xu C: Minimal
deviation adenocarcinoma of the uterine cervix. Int J Gynecol
Obstet. 110:89–92. 2010. View Article : Google Scholar
|
|
10
|
McKelvey JL and Goodlin RR: Adenoma
malignum of the cervix. A cancer of deceptively innocent
histological pattern. Am Cancer Soc. 16:549–557. 1963.
|
|
11
|
Shi JP, Zhang DJ, Yu HY and Cui Q:
Assessing Intra-Lesion Microvascular Density in Patients with Early
and Mid-Late Stages of Cervical Squamous Cell Carcinoma by
Time-Intensity Curve of CEUS. Chin J Ultrasound Med. 3:319–322.
2022.(In Chinese).
|
|
12
|
Huang R, Wei L and Qin WH: Analysis of the
Value of Ultrasonographic Parameters and MVD for Differential
Diagnosis and Prognostic Evaluation of Cervical Cancer. The Pract J
Cancer. 38:39–41. 2023.
|
|
13
|
Wang J, Yang Q, Wang D, Li M and Zhang N:
Case report: Gastric-Type endocervical adenocarcinoma mimicking
submucosal myoma under hysteroscopy. Front Med (Lausanne).
9:8454452022. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Takatsu A, Shiozawa T, Miyamoto T,
Kurosawa K, Kashima H, Yamada T, Kaku T, Mikami Y, Kiyokawa T,
Tsuda H, et al: Preoperative differential diagnosis of minimal
deviation adenocarcinoma and lobular endocervical glandular
hyperplasia of the uterine cervix: A multicenter study of
clinicopathology and magnetic resonance imaging findings. Int J
Gynecol Cancer. 21:1287–1296. 2011.PubMed/NCBI
|
|
15
|
Tsuboyama T, Yamamoto K, Nakai G, Yamada
T, Fujiwara S, Terai Y, Ohmichi M and Narumi Y: A case of
gastric-type adenocarcinoma of the uterine cervix associated with
lobular endocervical glandular hyperplasia: Radiologic-pathologic
correlation. Abdom Imaging. 40:459–465. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Maniaci A, Fakhry N, Chiesa-Estomba C,
Lechien JR and Lavalle S: Synergizing ChatGPT and general AI for
enhanced medical diagnostic processes in head and neck imaging. Eur
Arch Otorhinolaryngol. 281:3297–3298. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Bhatla N, Aoki D, Sharma DN and
Sankaranarayanan R: Cancer of the cervix uteri: 2021 update. Int J
Gynecol Obstet. 155 (Suppl 1):S28–S44. 2021. View Article : Google Scholar
|
|
18
|
China association of professional
committee of pancreatic cancer, . China of diagnosis and treatment
of pancreatic cancer comprehensive guide (2020). Chin surg.
59:81–100. 2021.(In Chinese).
|
|
19
|
Xie YD, Xu CL, Yang B and Zhang LJ: Value
of time-intensity curve of contrast-enhanced ultrasound in early
diagnosis of cervical cancer. J Ultrasound Clin Med. 8:626–628.
2020.
|
|
20
|
Park KJ, Kim MH, Kim JK and Cho KS:
Gastric-Type adenocarcinoma of the uterine cervix: Magnetic
resonance imaging features, clinical outcomes, and prognostic
factors. Int J Gynecol Cancer. 28:1203–1210. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Kido A, Mikami Y, Koyama T, Kataoka M,
Shitano F, Konishi I and Togashi K: Magnetic resonance appearance
of gastric-type adenocarcinoma of the uterine cervix in comparison
with that of usual-type endocervical adenocarcinoma: A pitfall of
newly described unusual subtype of endocervical adenocarcinoma. Int
J Gynecol Cancer. 24:1474–1479. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Chen YP, Ho SP, Liou WS and Chen CJ:
Minimal deviation adenocarcinoma of the uterine cervix. Taiwan J
Obstet Gynecol. 54:447–449. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Ren WH, Zhao XL and Zhao FH: Global
guidelines for cervical cancer screening: A systematic review.
Zhonghua Yi Xue Za Zhi. 101:1882–1889. 2021.(In Chinese).
PubMed/NCBI
|
|
24
|
Pak SC, Martens M, Bekkers R, Crandon AJ,
Land R, Nicklin JL, Perrin LC and Obermair A: Pap smear screening
history of women with squamous cell carcinoma and adenocarcinoma of
the cervix. Aust N Z J Obstet Gynaecol. 47:504–507. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Kondo E, Takahashi A, Usami T, Yamamoto A,
Nomura H, Matoda M, Okamoto S, Omatsu K, Sugiyama Y and Takeshima
N: An analysis of Papanicolaou smear before treatment for
adenocarcinoma in situ of the uterine cervix in 65 cases. J Jpn Soc
Clin Cytol. 54:114–118. 2015. View Article : Google Scholar
|
|
26
|
Wada T, Ohishi Y, Kaku T, Aman M, Imamura
H, Yasutake N, Sonoda K, Kato K and Oda Y: Endocervical
adenocarcinoma with morphologic features of both usual and gastric
types: Clinicopathologic and immunohistochemical analyses and
High-risk HPV detection by in situ hybridization. Am J Surg Pathol.
41:696–705. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Stolnicu S, Hoang L, Chiu D, Hanko-Bauer
O, Terinte C, Pesci A, Aviel-Ronen S, Kiyokawa T, Alvarado-Cabrero
I, Oliva E, et al: Clinical Outcomes of HPV-associated and
unassociated endocervical adenocarcinomas categorized by the
International endocervical adenocarcinoma criteria and
classification (IECC). Am J Surg Pathol. 43:466–474. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
28
|
D'Alessandro P, Giudicepietro A, Della
Corte L, Arduino B, Saccone G, Iacobelli A, Insabato L and Zullo F:
A case of gastric-type mucinous endocervical adenocarcinoma in
presence of nabothian cysts. Eur J Obstet Gynecol Reprod Biol.
236:254–255. 2019. View Article : Google Scholar : PubMed/NCBI
|