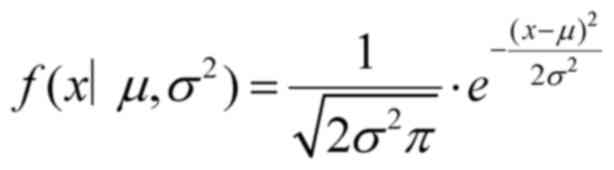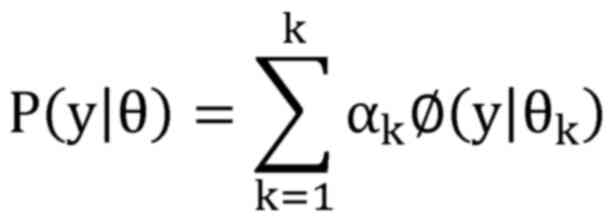Introduction
Breast cancer is the most commonly diagnosed cancer
among women worldwide, with ~2.3 million new cases each year
(1); it is also a leading cause of
cancer-related deaths, responsible for ~670,000 deaths annually
(1). Radiotherapy, as an important
component of comprehensive breast cancer treatment, is able to
effectively extend the survival times of patients with breast
cancer (2,3). Set-up errors are the differences
between the area in which the patient is actually being treated and
the area for which the treatment was planned, for example by
registering real-time images with images taken at the time of
positioning, usually through an image-guiding device. However,
set-up errors during radiotherapy may cause changes in tumor
location, leading to dose distribution deviations and affecting
treatment efficacy (4). Clinically,
image-guided monitoring and the correction of set-up errors are
employed to improve the accuracy of radiotherapy. Cone beam
computed tomography (CBCT) registration is a traditional
image-guided method; however, CBCT involves additional exposure to
radiation, which may increase the risk of secondary tumors in
patients (5). Therefore, the
rational use of image-guided methods or the search for novel
methods to obtain and correct set-up errors are important research
topics in the field of breast cancer radiotherapy.
Currently, in addition to seeking non-radiative
image-guided methods, predicting set-up errors based on past
patient data through mathematical modeling or computer deep
learning holds notable potential (6,7). The
Gaussian Mixture Model (GMM) holds significant advantages in this
area, as it is able to both describe complex error distributions
and quantify error probabilities (8). Therefore, the aim of the present study
was to use GMM to establish a predictive model for the distribution
of set-up errors in image-guided radiotherapy (IGRT) for breast
cancer. In terms of the results of the present study, the novel
aspects of this predictive model are as follows: i) Compared with
the traditional image-guided method, the set-up errors obtained via
this prediction model aids in avoiding radiation risk; ii) compared
with other predictive models such as deep learning, the GMM is
simpler, faster and capable of quantitative analysis; and iii) in
clinical applications, the GMM facilitates error intervention and
correction, reducing set-up errors and improving treatment
accuracy.
Patients and methods
Patient selection
In terms of sample size, the empirical method was
utilized and the number of patients in previous research literature
was assessed; therefore, analysis of >20 patients for the
present study was required. The maximum number of patients was
collected according to the inclusion and exclusion criteria applied
in the study period. The group sample size was then calculated
using the power analysis software PASS (version. 15; NCSS, LLC). To
collect sufficient data for analysis, 80 patients with breast
cancer who were treated in the Department of Radiation Oncology of
Fujian Medical University Union Hospital (Fuzhou, China) between
January 2021 and January 2022 were selected. The patients were
treated using a Varian 23E medical linear accelerator (Varian
Medical Systems, Inc.), the radiation dose was 40 Gy/15 fractions
and each fraction of the treatment course was image-guided using
the iSCOUT® system (version. 1.2.0; Jiangsu Rayer
Medical Technology Co., Ltd.). The present study was approved by
the Ethics Committee of Fujian Medical University Union Hospital
(Fuzhou, China; approval no. 2022WSJK017).
Inclusion and exclusion criteria
To ensure that patients could undergo
intensity-modulated radiotherapy and complete the whole course of
radiotherapy, the inclusion criteria were as follows: i) Patients
who had pathologically confirmed breast cancer; ii) patients who
received intensity-modulated radiotherapy following modified
radical mastectomy for breast cancer; iii) patients who could raise
both arms, fully exposing the affected breast; iv) patients who had
a Karnofsky Performance Status score >80; and v) patients who
had no other diseases affecting the radiotherapy. The exclusion
criteria were as follows: i) Patients who had undergone breast
valve surgery; ii) patients who had difficulty supporting their
arms and who could not meet the requirements for thermoplastic mask
location; and iii) patients who were otherwise unfit or unable to
complete the entire study process. Breast cancer was not graded in
the present study, which was independent of the study content and
results.
Radiotherapy process
Following the standard procedure, all patients were
fixed by body bracket with or without thermoplastic film, and
scanned using Philips 16-row large-aperture spiral computed
tomography (Philips Healthcare), with a slice thickness of 5 mm.
The IGRT plan was designed using a Varian Eclipse treatment
planning system (version. 15.6; Varian Medical Systems, Inc.). The
X-ray energy was 6 MV and the calculation grid was 2.5 mm.
Patients were positioned by two therapists during
treatment. Prior to each treatment process, real-time 45 and 135°
positioning images were obtained using the iSCOUT system for
position verification. The acquired images were automatically
registered with the digitally reconstructed images generated at the
same angle, performed by a senior therapist using the bone
registration mode of the iSCOUT system software. If automatic
registration could not obtain the results, manual registration was
used. The registration frame was set at the center of the treatment
target, and the chest wall, ribs, thoracic vertebrae and clavicle
were used as the regions of interest to register the set-up errors
in the lateral (LAT), longitudinal (LONG) and vertical (VERT)
direction for recording.
Data acquisition and
preprocessing
The collected three-dimensional direction data were
preprocessed. Each patient underwent 15 sessions of radiotherapy,
according to the principle that image guidance should have been
performed at least once per session, resulting in 15 sets of
three-dimensional data per patient. Any loss of data resulted as a
consequence of equipment failures, image quality issues or personal
issues of the patients. Patients with >2 sets of missing data
were excluded, whereas those with 1–2 sets of missing data had
their data completed using the mean value of their existing data to
ensure data completeness and authenticity. For each patient, based
on clinical error ranges, data with maximum and minimum deviations
>15 mm were analyzed, and data from patients with a high degree
of variability were excluded to ensure data reasonableness.
Construction of the prediction
model
Concepts used in model construction
i) From the single Gaussian model to the GMM. The
single Gaussian model, also known as the normal distribution, is
defined as follows: If a random variable x follows a
Gaussian distribution with a mean µ and a variance σ2,
it is denoted as N (µ, σ2) (9). The parameter µ represents the mean,
which corresponds to the center of the normal distribution, and the
parameter σ represents the standard deviation. The probability
density function is defined according to the following formula:
The GMM is introduced when the data distribution
consists of a linear combination of multiple Gaussian
distributions; it can theoretically fit various types of
distributions and is usually utilized to deal with data sets
containing multiple different distributions. According to the
definition, assuming that the data follow the Gaussian mixture
distribution, then the probability distribution model has the
following formula:
where αk represents the weight
coefficient, αk≥0, ∑k=1kαk=1; ø(y|θk) is
the Gaussian distribution density, θk=(μk,σkk) and the Gaussian distribution
density is as follows: ø(y|θk)=12α2πe(y-μk)22αk2.
That is, it represents the k-th Gaussian distribution density
function.
ii) Expectation maximization (EM) algorithm. The EM
algorithm is used in statistics to find the maximum likelihood
estimates of parameters in probabilistic models that depend on
unobserved latent variables; it is an effective method for solving
optimization problems involving hidden variables. Since the GMM
function is difficult to handle through partial derivatives, the EM
algorithm is commonly used to solve its parameters. The EM
algorithm is iterative, with each iteration consisting of an
expectation step and a maximization step.
iii) K-means algorithm. The K-means algorithm is
usually used in clustering, and its fundamental principle is that
the distance between the points inside the cluster is smaller than
the distance between the points outside the cluster (10). The K-means algorithm and the GMM can
express each other under certain conditions. The K-means algorithm
may be regarded as a special form of the GMM, whereas the GMM
provides stronger descriptive power. The GMM is more
computationally intensive than the K-means algorithm per iteration.
Therefore, usually in practical applications, the K-means algorithm
is used to obtain the initial clustering results first, and
subsequently, its cluster number and cluster center are passed to
the GMM as initial values for a more meticulous iteration.
The implementation of the K-means algorithm
comprises the following steps: a) K center points are randomly
selected; b) the data are traversed, and each data point is
assigned to the nearest center class; c) the mean value of each
cluster is then calculated and used as the new center; and d)
finally, steps b and c are repeated until either the convergence or
the maximum number of iterations is reached.
iv) Elbow method. There are many methods to
determine the number of clusters, including the Elbow method,
Silhouette coefficient, Gap statistic and fuzzy clustering. The
Elbow method is an intuitive and simple approach commonly used to
determine the number of clusters k of the K-means algorithm.
The method classifies the data according to different k
values by enumeration, after which the variance percentage of each
classification is obtained and a chart may be drawn. The k
value corresponding to the inflection point, the elbow, in the
curve is selected as the best cluster number. Compared with the
Elbow method, the Silhouette coefficient method considers both the
similarity within clusters and the differences between clusters,
whereas the Gap statistic method determines the appropriate number
of clusters by calculating the Gap statistic for different numbers
of clusters. The fuzzy clustering method is a membership-based
clustering method that allows data points to belong to multiple
clusters simultaneously. The k values provided by the
Silhouette score and Gap statistic method were compared with the
Elbow method used in the present study. Through comparison of the
performance of these methods and considering the data, the Elbow
method was ultimately chosen to determine the clustering parameters
(Appendix S1; Fig. S1 and S2).
Process of model building
The distribution of radiotherapy set-up errors
follows a normal distribution, which conform to the Gaussian
distribution. Given the complexity of the data distribution, a
single Gaussian model is evidently insufficient to represent the
overall dataset, making the GMM a more suitable choice. As
aforementioned, the K-means algorithm and GMM can be expressed in
terms of each other under certain conditions, with K-means being a
special case of GMM. GMM offers a stronger descriptive
capability.
To construct the prediction model, the methodology
outlined by Qiu et al (8)
was adopted. After comprehensive evaluation and analysis of the
various methods, the present study developed the error distribution
prediction model through the following process: i) The IGRT set-up
errors data of patients were recorded in three directions and
converted into three-dimensional matrix data for saving and
processing; ii) the number of clusters was determined using the
Elbow method; iii) the K-means algorithm was used to cluster the
raw set-up errors data, and both the cluster number and initial
cluster center were obtained; and iv) the number of clusters and
cluster centers obtained using the K-means algorithm were
transferred as initial values to the GMM, while the EM algorithm
was used for iteration to determine the parameters of the GMM and
the clinical significance of the model was analyzed. The model
building code is detailed in Appendix
S2.
Calculation of the PTV expansion
formula
PTV is an area expanded from the clinical target
volume that accounts for positional deviations during treatment,
respiratory motion and bladder filling changes (11,12).
The ultimate goal is to ensure the tumor receives an adequate
radiation dose. There are numerous methods available for
calculating PTV, but the most commonly used one is the PTV margin
formula recommended by Van Herk (13): M=2.5Σ + 0.7δ, where Σ is the
standard deviation of the mean of the fractionation error for each
patient, and δ is the root mean square of the standard deviation of
the fractionation error for each patient. In the present study, the
PTV margins were calculated based on the original error in each
direction, and these were then compared with the GMM parameters.
The aim was to verify whether the error range obtained through the
constructed model could replace the PTV calculation formula,
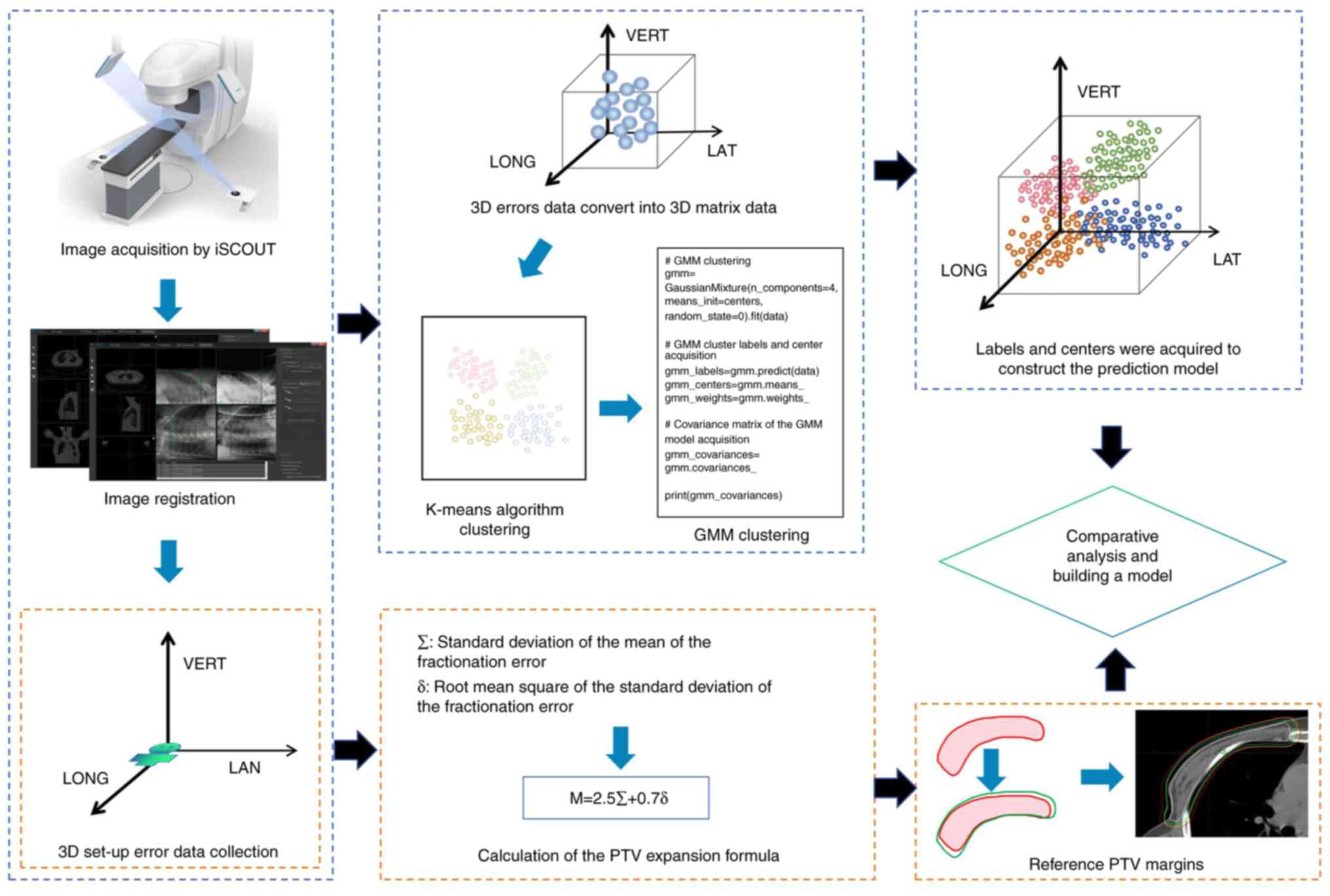
thereby simplifying the PTV calculation process. A flow chart of
the research methods employed in the present study is shown in
Fig. 1.
Data processing
In the present study, WPS Office Excel (version.
6.0.2; Kingsoft Office Software) was used for data collection and
preliminary statistics, R language programming (version. 4.0.2; R
Core Team) was used for K-means initial clustering and PyCharm
(version 2020.1.2; JetBrains) was used for construction of the GMM
and parameter solutions.
Results
Patient data
The clinical data of patients and the set-up errors
data of IGRT for each fraction were obtained. All 80 patients were
female and consisted of 36 patients with left-sided breast cancer
and 44 patients with right-sided breast cancer. The age of the
patients ranged from 26–67 years, with a median age of 47.5 years.
A total of 1,200 sets of set-up errors data were collected for
model construction.
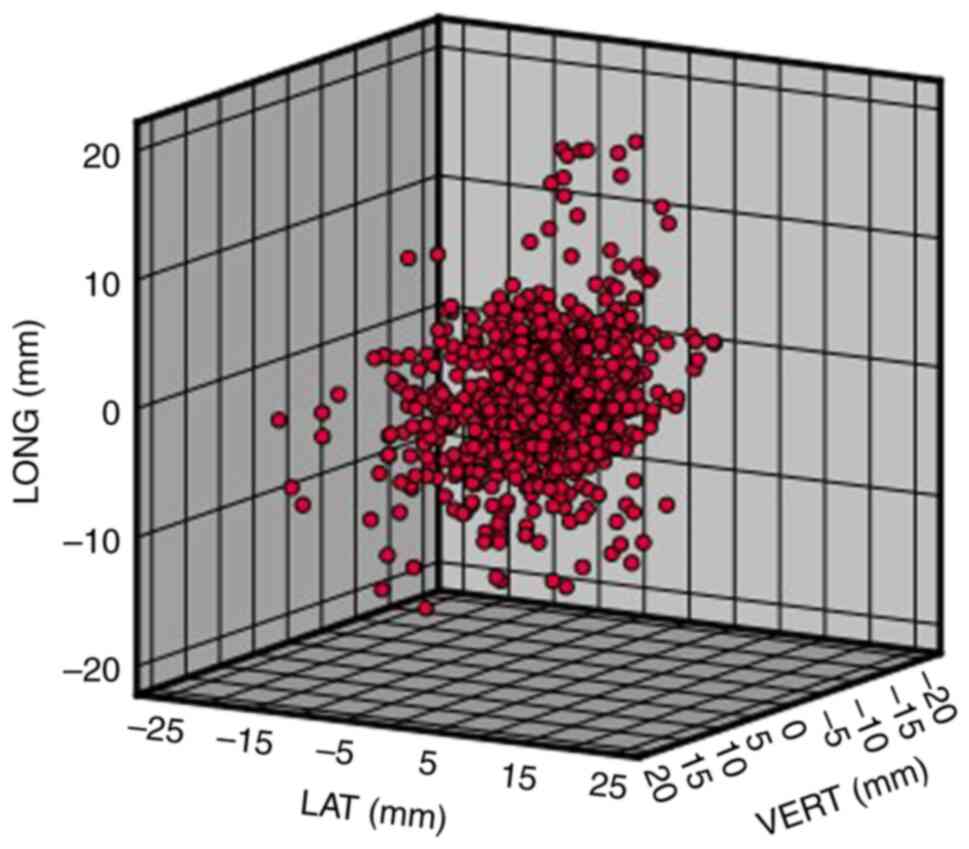
Raw error data matrix
The statistical results arising from the analysis of
the 1,200 sets of set-up errors data that were collected from 80
patients with breast cancer are shown in Table I. Each patient's IGRT set-up errors
data were recorded in the three directions of LAT, LONG and VERT,
converted into a three-dimensional matrix and the distribution of
the raw error data matrix obtained (Fig. 2).
 | Table I.Raw set-up errors statistics
(mm). |
Table I.
Raw set-up errors statistics
(mm).
| Direction | Maximum, mm | Minimum, mm | P25,
mm | P50,
mm | P75,
mm | X±S, mm |
|---|
| LAT | 23.60 | −22.70 | −2.75 | −0.90 | 1.78 | −0.90±4.81 |
| LONG | 18.10 | −17.30 | −2.60 | −0.70 | 1.30 | −0.83±3.98 |
| VERT | 15.10 | −17.30 | −2.90 | −1.30 | 0.80 | −1.18±3.42 |
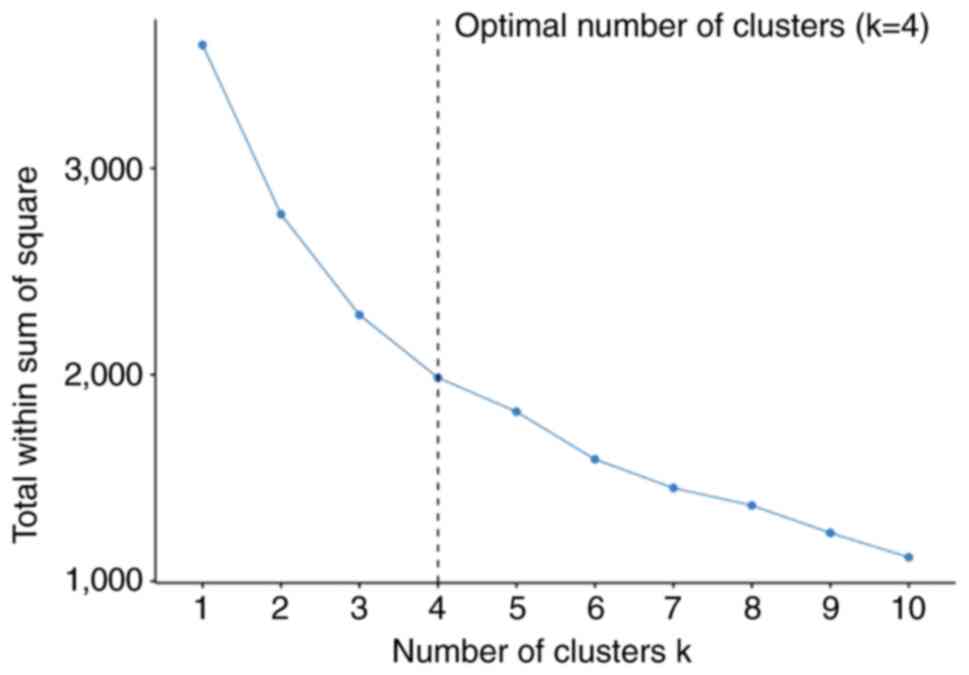
Optimal clustering number
The K-means method was used to cluster the raw
three-dimensional matrix errors data, and the optimal number of
clusters determined using the Elbow method was 4. At this point
(k=4), the internal variance decreased slowly, forming an
inflection point and the clustering effect was good (data compactly
clustered according to classification, with few discrete data)
(Fig. 3). The data were clustered
according to k=4 to obtain the initial cluster center and the
clustering effect diagram (Table
II and Fig. 4).
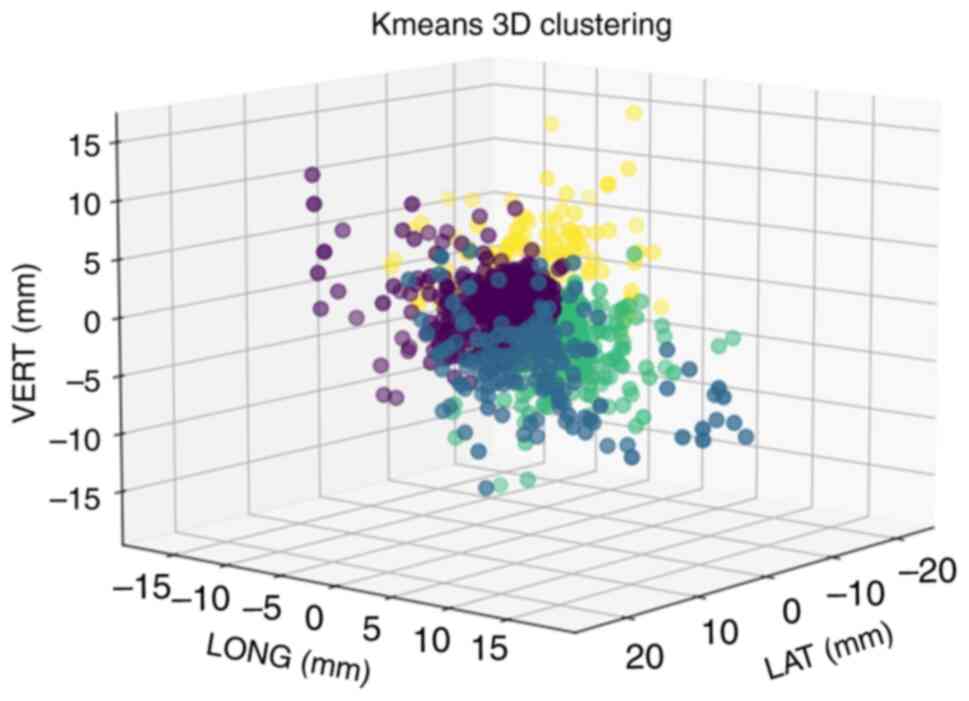 | Figure 4.Preliminary three-dimensional
clustering results by K-means. LAT, LONG and VERT were used as axes
to establish a three-dimensional clustering effect map, and the
initial cluster center points were µ1, µ2, µ3 and µ4, which are
represented by the purple, blue, green and yellow points,
respectively. LAT, lateral; LONG, longitudinal; VERT, vertical. |
 | Table II.K-means initial clustering
center. |
Table II.
K-means initial clustering
center.
| Center | LAT | LONG | VERT |
|---|
| µ1 | 1.20187967 | −2.16093058 | 0.30402782 |
| µ2 | 4.69122754 | 3.45807100 | −3.39719418 |
| µ3 | −2.87421188 | 0.22517496 | −2.87446722 |
| µ4 | −8.61301527 | −5.27360414 | 2.21768811 |
GMM for prediction of set-up errors
distribution
The cluster number and cluster centers obtained
using the K-means algorithm were passed to the GMM as initial
values, and the EM algorithm was used for iteration to solve and
determine the parameters of the GMM. The clustering effect and
cluster center distribution are shown in Fig. 5. The parameters of the GMM error
distribution prediction model obtained by solution are shown as
follows: The center coordinates of each error, namely the mean µ of
the GMM (Table III), the
covariance matrix of the errors model, namely the GMM σ (Table IV), and each error center
probability, the coefficient α of the GMM (Table V).
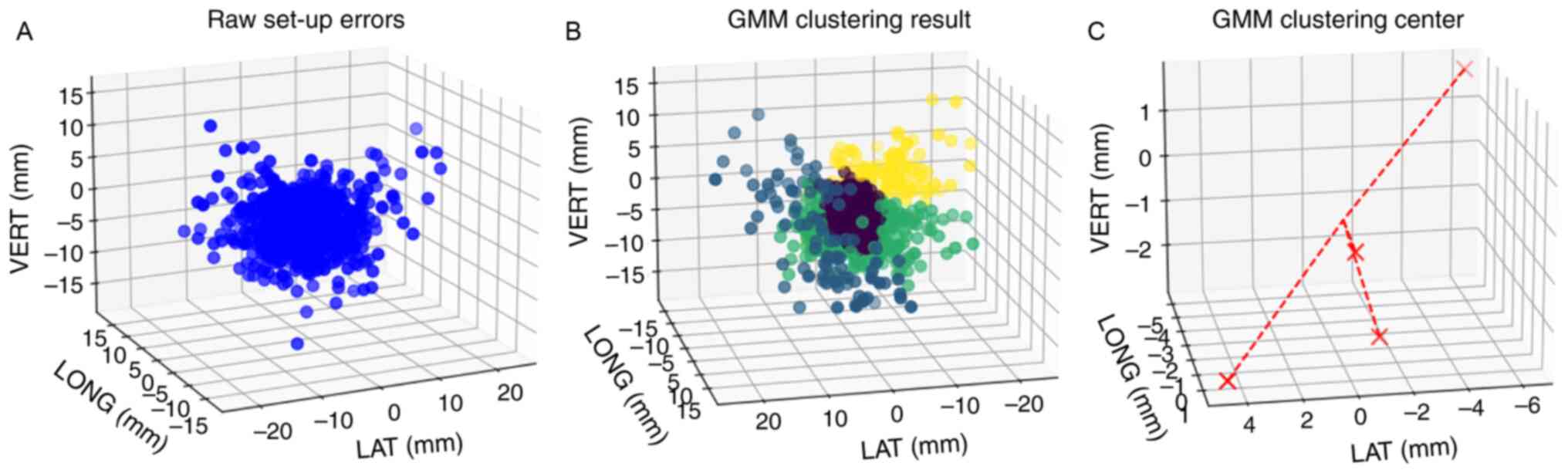 | Figure 5.Clustering results according to the
GMM. (A) The clustering effect diagram of the raw set-up errors
data is shown, where the blue dots represent the raw data. (B) The
clustering effect diagram of the GMM. Data with initial clustering
center points of µ1, µ2, µ3 and
µ4 are represented by purple, blue, green and yellow
points, respectively. (C) Distribution diagram of clustering center
points of the GMM, with the 4 red crosses representing the
clustering centers. GMM, Gaussian mixture model; LAT, lateral;
LONG, longitudinal; VERT, vertical. |
 | Table III.Center coordinates of set-up
errors. |
Table III.
Center coordinates of set-up
errors.
| Center | LAT | LONG | VERT |
|---|
| µ1 | −0.55132442 | −0.35566862 | −0.7863227 |
| µ2 | 4.59050708 | 1.47558872 | −2.75250798 |
| µ3 | −1.56319372 | −0.73935399 | −2.73943799 |
| µ4 | −6.30437674 | −5.40364623 | 1.72933048 |
 | Table IV.Covariance matrix of the Gaussian
mixture model. |
Table IV.
Covariance matrix of the Gaussian
mixture model.
| Center | LAT | LONG | VERT |
|---|
| σ1 | 4.05961107 | 0.55761847 | 1.04514747 |
|
| 0.55761847 | 3.99255928 | −0.81082324 |
|
| 1.045147478 | −0.81082324 | 3.58734632 |
| σ2 | 29.05573717 | −6.72025397 | 14.63273482 |
|
| −6.72025397 | 48.16645234 | −12.44937602 |
|
| 14.63273482 | −12.44937602 | 28.24677334 |
| σ3 | 31.2592853 | −2.90156674 | 0.63680924 |
|
| −2.90156674 | 12.48825079 | −2.49353153 |
|
| 0.63680924 | −2.49353153 | 10.83415154 |
| σ4 | 28.71612318 | −1.77487288 | −9.93513094 |
|
| −1.77487288 | 18.70292861 | 3.01798682 |
|
| −9.93513094 | 3.01798682 | 18.34102576 |
 | Table V.Each error center probability. |
Table V.
Each error center probability.
| Center | σ1 | σ2 | σ3 | σ4 |
|---|
| Probability | 0.53169465 | 0.11099651 | 0.24059103 | 0.11671781 |
Prediction of PTV expansion
The reference PTV margins based on the raw set-up
errors in each direction were calculated and compared with the GMM
parameters, where Σ is the standard deviation of the mean of the
fractionation error for each patient, and δ is the root mean square
of the standard deviation of the fractionation error for each
patient (Table VI).
 | Table VI.Reference value of the PTV margin
(mm). |
Table VI.
Reference value of the PTV margin
(mm).
| Direction | Guidance frequency,
n (%) | Σ | δ | MPTV,
mm |
|---|
| LAT | 487 (40.58) | 1.71 | 2.33 | 5.90 |
| -LAT | 713 (59.42) | 2.05 | 2.89 | 7.16 |
| LONG | 493 (41.08) | 1.83 | 1.56 | 5.67 |
| -LONG | 707 (58.92) | 1.65 | 2.43 | 5.82 |
| VERT | 399 (33.25) | 1.24 | 1.61 | 4.23 |
| -VERT | 801 (66.75) | 1.76 | 2.08 | 5.87 |
Discussion
The present study utilized the GMM to establish a
predictive model for the distribution of set-up errors in IGRT for
breast cancer. The analysis of the data obtained confirmed the
feasibility of the GMM use for quantitative description and
predictive analysis of set-up error distribution in breast-cancer
IGRT. By comparing these results with the conventional PTV margin
calculation formula, the present study demonstrated the clinical
application value of GMM. This offers a reference for controlling
set-up errors and determining PTV margins in breast cancer
radiotherapy, especially in cases without routine daily image
guidance.
Concerning the GMM parameters, it was found that the
set-up errors were mainly in the direction of four central points
(µ1-µ4). The spatial coordinates of each center could reflect the
average offset direction and offset of the points in the center.
For example, the deviation of µ1 in each of the three directions
(LAT, LONG and VERT) was within 1 mm, recorded as −0.55, −0.36 and
−0.79 mm, respectively. From the overall center distribution data,
all the raw errors in the LAT, LONG and VERT directions were
−6.3-4.60, −5.40-1.47 and −2.7-1.7 mm, respectively. According to
the probability of each center, the most possibility of the set-up
errors in the µ1 direction was 0.53, and for the other three
centers (µ2, µ3 and µ4), the highest possibilities were 0.11, 0.34
and 0.12, respectively. The covariance matrix reflects the
magnitude of the standard deviation. According to the covariance
parameters of the GMM, the maximum statistical standard deviation
of the set-up error could reach 29.06. Compared with previous
studies, Qiu et al (8)
constructed a predictive model for set-up error distribution in
pelvic tumor radiotherapy using the GMM on the Varian Novalis
Tx® linear accelerator. The center point coordinates in
the three directions were −1.85-0.72, −2.41-1.54 and −3.88-4.28 mm,
respectively. In the present study, the errors were relatively
larger, which may have been associated with greater mobility of the
breast tumors. Compared with pelvic tumors, breast tumors exhibit
greater movement, highlighting the necessity for image guidance and
set-up error prediction in breast cancer radiotherapy.
Traditional image-guided methods involve acquiring
real-time images and registering them with reference images from
the treatment plan. CBCT and MRI registration provide
high-precision 3D image guidance but come with high equipment
costs, additional radiation and long imaging times (5). Deep learning-based methods, using
Convolutional Neural Networks for image segmentation and
registration, offer high accuracy and automation, but require
large, labeled datasets and significant computational resources
(14). In contrast with traditional
image-guided methods for assessing set-up errors, the present study
investigated radiation risk using a constructed predictive model
(4). The GMM serves as the
foundation, utilizing the Gaussian probability density function
(also known as the normal distribution curve) to decompose observed
phenomena into multiple components, accurately describing their
characteristics (15).
Theoretically, regardless of the distribution pattern within the
observed dataset, the GMM can fit it through combining multiple
Single Gaussian Models linearly. In contrast to other predictive
models such as deep learning, the GMM offers simplicity, speed and
the ability for quantitative analysis (16,17).
Various techniques exist for determining the optimal number of
clusters, including the Elbow method, and the Silhouette
coefficient, the Gap statistic and fuzzy clustering methods
(18). The Elbow method involves
classifying data with different values of k, calculating the
variance percentage for each classification and plotting a graph.
The optimal number of clusters corresponds to the ‘elbow point’ on
the curve, providing an intuitive and comprehensive solution
(19). Consequently, the Elbow
method was selected for the present study to determine the
appropriate number of clusters. Notably, set-up errors during
radiotherapy significantly impact treatment accuracy (4). The set-up error during radiotherapy is
a critical factor affecting treatment accuracy. Small et al
(20) proposed a method for
analyzing the distribution of radiotherapy errors and pointed out
that different sources of errors lead to different characteristics
of the distribution of errors. Van Herk (13) focused on the effects of systematic
and random errors in radiotherapy and investigated a variety of
mathematical models to describe the errors. The systematic error is
the mean of all fractional set-up errors, whereas the random error
is the standard deviation of these errors, reflecting the diversity
and variability of these errors. However, set-up errors do not
manifest as simple three axial deviations. In the present study,
statistical and modeling methods were used to deeply study the
distribution of IGRT data. It was found that the set-up errors not
only exist in the three axes, but also tend to be concentrated
along the specific center direction, and the deviation distribution
probability of each center direction is different. This finding
suggested that the set-up errors had a more complex distribution
form, which is no longer limited to the triaxial linear deviation,
but may involve more dimensional and directional changes. This
deeper understanding of the error provides a more specific and
targeted plan for further error correction and treatment
optimization.
For validation of the prediction model and to assess
its clinical applicability, the effectiveness of the model was
assessed by comparing it with a commonly used clinical formula for
calculating the PTV expansion margin. The results indicated that
the PTV expansion margins determined using Van Herk's formula in
six directions were as follows: LAT, −7.60 and 5.90 mm; LONG, −5.82
and 5.67 mm; and VERT, −5.87 and 4.23 mm. These findings aligned
with the results derived from the GMM analysis, with deviations
ranging from 0.42-4.20 mm. The largest deviation occurred in the
VERT direction. Although the error in this direction was larger,
the distribution was more concentrated, leading to discrepancies
between the model predictions and the formula calculations.
Numerous factors are known to affect set-up errors during
radiotherapy. For breast cancer, in particular, the unique shape
and position of the breast, along with its large range of motion,
result in set-up errors that are significantly different from those
of other thoracic tissues (21). A
study performed by Chen et al (22) on 113 patients with breast cancer
undergoing radiotherapy investigated the influencing factors and
uncertainties of set-up errors. This analysis demonstrated that
body mass index, the surgical method, surgical site and
immobilization method may affect the accuracy of radiotherapy for
breast cancer. As a key image-guided device, CBCT can monitor and
correct set-up errors in real time (23). Although CBCT has a high utilization
rate and low radiation dose, it still utilizes ionizing radiation,
which may increase human radiation exposure, thereby increasing
further the risk of second primary tumors. Donovan et al
(24) reported that the doses to
the breasts, heart and lungs of patients with breast cancer during
CBCT scanning were approximately 1.7-23.2, 4.0-21.6 and 0.8-22.8
mGy, respectively. Multiple exposures increase the radiation dose
significantly, which may lead to the development of second primary
tumors (24). In order to resolve
this problem, a statistical modeling analysis of the raw error data
of patients who underwent complete IGRT treatment was performed in
the present study to present the error distribution law and predict
the error probability. The present study validated the applicative
value of error prediction ranges through calculating the PTV
expansion margins. This served to optimize the PTV boundaries,
thereby preventing issues of excessive or insufficient PTV
expansion that could lead to dosage deviations in the target area
in clinical settings (25,26). In addition, the present study
suggested that the PTV expansion should not only be limited to the
three axial errors, but that the directions and variances of the
four offset centers should also be comprehensively considered for
PTV expansion. Specifically, a non-uniform expansion strategy is
required to cover the variance offset in each center direction. The
goal of this approach is to comprehensively consider the potential
sources of error during radiotherapy, thereby providing a more
complete understanding of the requirement for PTV margins. Through
the integration of the error changes in multiple directions, not
only can the effect of the error be better controlled in planning,
but it also helps to optimize the treatment plan, thereby ensuring
adequate target coverage while minimizing the damage to the
surrounding normal tissues.
Although the present study attempted to use the GMM
to predict the distribution of errors in radiotherapy, optimize PTV
expansion margins and improve radiotherapy accuracy, certain
limitations still need to be addressed. First, regarding sample
size, 80 patients were included and 1,200 data sets collected,
which, although relatively large, still presents certain
limitations in terms of model construction and data prediction.
Future work will involve collecting more samples and conducting
in-depth analyses of the data to uncover potential differences
among subsets, with the goal of enhancing the model's robustness
and accuracy. Secondly, both the K-means algorithm and the GMM were
initialized in a random manner, so the initial state may have been
random, leading to different final results. Although
‘random_state=0’ was set for both methods in the present study to
ensure the reproducibility of the results, this only applied to
multiple runs on the same computer. Differences on different
computers, such as the implementation of the underlying libraries,
operating systems and processor architectures, can lead to
different behaviors. In addition, the library version is an
important consideration, as it must be made certain that the
libraries, such as sklearn and pandas, are installed as the same
version on all computers. Different versions of libraries may have
different default parameters, optimizations or fixes, and these
differences may affect the stability of the results. Therefore,
when replicating the present study, there may be differences in the
results using different versions. In the future, we will also test
the data on different versions and analyze the differences.
In conclusion, the present study emphasized the
importance of accurate error control in radiotherapy, and discussed
IGRT technology and its limitations in depth. GMM can be used to
quantitatively describe and predict the distribution of set-up
errors in IGRT for breast cancer, thereby providing a reference for
the set-up errors control and tumor planning target expansion of
breast cancer without routine daily image-guided radiotherapy. The
results of the present study are beneficial to reduce the extra
radiation of breast cancer radiotherapy and improve the treatment
accuracy, and can be applied to other tumors. Future studies could
introduce deep learning methods to more accurately predict and
control radiotherapy errors. Techniques such as anomaly detection
and pattern recognition may be employed for a more in-depth
analysis of images and error data (27,28).
Currently, studies are developing in the direction of multi-factor
analysis, comprehensively considering the angle error and
physiological changes, and performing more detailed data analyses
to understand the sources of errors and to change the rules more
comprehensively.
Supplementary Material
Supporting Data
Acknowledgements
Not applicable.
Funding
The present study was funded by Fujian Province Health Youth
Research Project (grant no. 2022QNA018) and Fujian Medical
University Sailing Fund General Project (grant no. 2022QH1029).
Availability of data and materials
The data generated in the present study may be
requested from the corresponding author.
Authors' contributions
FD and JC drafted the manuscript and worked on the
conception, design and interpretation of data. FD and JC confirm
the authenticity of all the raw data. ZY and FL helped with data
processing and drafting the manuscript. XL and YW reviewed the data
analysis. All authors contributed to the article. All authors read
and approved the final version of the manuscript.
Ethics approval and consent to
participate
The present study was retrospective and was approved
by the Ethics Committee of Fujian Medical University Union Hospital
(Fuzhou, China; approval no. 2022WSJK017). The data used in the
study did not interfere with the patient's treatment and the Ethics
Committee waived the requirement for informed consent. All
procedures involving human participants were in accordance with the
ethical standards of the institutional and/or national research
committee and with the 1964 Declaration of Helsinki and its later
amendments or comparable ethical standards.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Arnold M, Morgan E, Rumgay H, Mafra A,
Singh D, Laversanne M, Vignat J, Gralow JR, Cardoso F, Siesling S
and Soerjomataram I: Current and future burden of breast cancer:
Global statistics for 2020 and 2040. Breast. 66:15–23. 2022.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Long J, Fei C, Li Z and Shaojun MA: Risk
factors for neutropenia during postoperative adjuvant radiotherapy
for breast cancer. J Precision Medicine. 38:530–534. 2023.
|
|
3
|
Upadhyay R and Bazan JG: Advances in
radiotherapy for breast cancer. Surg Oncol Clin N Am. 32:515–536.
2023. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Costin IC and Marcu LG: Factors impacting
on patient setup analysis and error management during breast cancer
radiotherapy. Crit Rev Oncol Hematol. 178:1037982022. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
de Crevoisier R, Lafond C, Mervoyer A,
Hulot C, Jaksic N, Bessières I and Delpon G: Image-guided
radiotherapy. Cancer Radiother. 26:34–49. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Li G: Advances and potential of optical
surface imaging in radiotherapy. Phys Med Biol.
67:10.1088/1361–6560/ac838f. 2022. View Article : Google Scholar
|
|
7
|
Mafi M and Moghadam SM: Real-time
prediction of tumor motion using a dynamic neural network. Med Biol
Eng Comput. 58:529–539. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Qiu MM, Zhong JJ, Ouyang B, Xiao ZH and
Deng YJ: Set-up errors distribution prediction model for pelvic
tumors radiotherapy of varian NovalisTX medical linear accelerator
based on gaussian mixtures. J Sun Yat-Sen University (Medical
Sciences). 40:284–290. 2019.
|
|
9
|
Bishop CM: Pattern recognition and machine
learning. Springer; New York, NY: pp. 423–439. 2006, PubMed/NCBI
|
|
10
|
Jain AK: Data clustering: 50 years beyond
K-means. Pattern Recognition Letters. 31:651–666. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Hlavka A, Vanasek J, Odrazka K, Stuk J,
Dolezel M, Ulrych V, Vitkova M, Mynarik J, Kolarova I and Vilasova
Z: Tumor bed radiotherapy in women following breast conserving
surgery for breast cancer-safety margin with/without image
guidance. Oncol Lett. 15:6009–6014. 2018.PubMed/NCBI
|
|
12
|
Buschmann M, Kauer-Dorner D, Konrad S,
Georg D, Widder J and Knäusl B: Stereoscopic X-ray image and
thermo-optical surface guidance for breast cancer radiotherapy in
deep inspiration breath-hold. Strahlenther Onkol. 200:306–313.
2024. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
van Herk M: Errors and margins in
radiotherapy. Semin Radiat Oncol. 14:52–64. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Chrystall D, Mylonas A, Hewson E, Martin
J, Keall P, Booth J and Nguyen DT: Deep learning enables MV-based
real-time image guided radiation therapy for prostate cancer
patients. Phys Med Biol. 68:10.1088/1361–6560/acc77c. 2023.
View Article : Google Scholar
|
|
15
|
Hattel SH, Andersen PA, Wahlstedt IH,
Damkjaer S, Saini A and Thomsen JB: Evaluation of setup and
intrafraction motion for surface guided whole breast cancer
radiotherapy. J Appl Clin Med Phys. 20:39–44. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Sakurai Y, Ambo S, Nakamura M, Iramina H,
Iizuka Y, Mitsuyoshi T, Matsuo Y and Mizowaki T: Development of a
prediction model for target positioning by using diaphragm
waveforms extracted from CBCT projection images. J Appl Clin Med
Phys. 24:e141122023. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Ghorbanzadeh L, Torshabi AE, Nabipour JS
and Arbatan MA: Development of a synthetic adaptive neuro-fuzzy
prediction model for tumor motion tracking in external radiotherapy
by evaluating various data clustering algorithms. Technol Cancer
Res Treat. 15:334–347. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Li Y, Zeng X, Lin CW and Tseng GC:
Simultaneous estimation of cluster number and feature sparsity in
high-dimensional cluster analysis. Biometrics. 78:574–585. 2022.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Sammouda R and El-Zaart A: An optimized
approach for prostate image segmentation using K-means clustering
algorithm with elbow method. Comput Intell Neurosci.
2021:45538322021. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Small W Jr, Mell LK, Anderson P,
Creutzberg C, De Los Santos J, Gaffney D, Jhingran A, Portelance L,
Schefter T, Iyer R, et al: Consensus guidelines for delineation of
clinical target volume for intensity-modulated pelvic radiotherapy
in postoperative treatment of endometrial and cervical cancer. Int
J Radiat Oncol Biol Phys. 71:428–434. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Saliou MG, Giraud P, Simon L,
Fournier-Bidoz N, Fourquet A, Dendale R, Rosenwald JC and Cosset
JM: Radiotherapy for breast cancer: Respiratory and set-up
uncertainties. Cancer Radiother. 9:414–421. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Chen Q, Xi H, Gu Y, Yang XW and Jing HS:
Influencing factors and uncertainty analysis of breast cancer
set-up errors. J Med Postgraduate Students. 2:352022.(In
Chinese).
|
|
23
|
Chen SF: Research progress of cone-beam CT
guided precision radiotherapy for breast cancer. Chin J Med Phys.
36:32019.(In Chinese).
|
|
24
|
Donovan EM, James H, Bonora M, Yarnold JR
and Evans PM: Second cancer incidence risk estimates using BEIR VII
models for standard and complex external beam radiotherapy for
early breast cancer. Med Phys. 39:5814–5824. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Shen ZW, Li S, Tan X, Tian XM, Luo HL, Ji
F and Wang Y: Analysis and Verification of The Margin of Target
Volume in Radiotherapy for Breast Cancer After Radical Mastectomy.
Chin J Med Phys. 34:71–78. 2021.(In Chinese).
|
|
26
|
Batumalai V, Holloway L and Delaney GP: A
review of setup error in supine breast radiotherapy using cone-beam
computed tomography. Med Dosim. 41:225–229. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Sailunaz K, Alhajj S, Özyer T, Rokne J and
Alhajj R: A survey on brain tumor image analysis. Med Biol Eng
Comput. 62:1–45. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Ye RZ, Lipatov K, Diedrich D,
Bhattacharyya A, Erickson BJ, Pickering BW and Herasevich V:
Automatic ARDS surveillance with chest X-ray recognition using
convolutional neural networks. J Crit Care. 82:1547942024.
View Article : Google Scholar : PubMed/NCBI
|