Introduction
Uterine cervical cancer (CC) is the fourth most
prevalent cancer type among women globally, underscoring the
critical need for ongoing advancements in its management (1). Despite progress in the treatment of
CC, the existing therapeutic options have persistent limitations,
including side effects and the emergence of drug resistance
(2). The histopathological
classification of CC identifies two main subtypes: Squamous cell
carcinoma (SCC) and adenocarcinoma (AC). While the treatment
approach for both subtypes is generally similar, AC is associated
with a poorer prognosis than SCC (3). Therefore, addressing the challenges
posed by AC remains an essential area for improving overall
therapeutic outcomes in CC.
Microbiota are considered to serve a pivotal role in
carcinogenesis (4,5). In the context of CC, beyond the
well-established risk factor of human papillomavirus, studies have
highlighted the contribution of the microbiome to CC development
and progression (6,7). It is widely acknowledged that the
depletion of Lactobacillus can lead to dysbiosis,
characterized by an increase in pathogenic microbial diversity,
such as Atopobium, Gardnerella vaginalis, Prevotella, Sneathia,
Streptococcus and Megasphaera (7–9). This
microbial imbalance has been associated with damage to the mucus
and cytoskeleton structures, alteration of antimicrobial peptides,
promotion of proinflammatory cytokine production, and initiation of
cervical intraepithelial neoplasia (CIN) (8,10).
Streptococcus, a prominent genus of
gram-positive bacteria, has garnered attention in the context of
patients with CC (11).
Mulato-Briones et al (12)
identified Streptococcus as a predominant bacterium in
patients with CC through the culture of cervical exocervix samples.
Streptococcus is also a potential biomarker for
distinguishing between invasive CC and CIN (13). Notably, Streptococcus
agalactiae [group B Streptococcus (GBS)] often colonizes
asymptomatically in the female genital tract and can subsequently
become a pathogen, leading to severe infections in neonates and
adults under certain conditions (14). Previous studies have indicated that
GBS stimulates proinflammatory cytokines, including IL-8, IL-1b,
IL-6, IL-17 and TNF-α, during its interaction with host epithelial
cells (15–18). GBS exhibits a notable capacity to
invade cervical epithelial cells and maintain colonization,
particularly when compared with vaginal epithelial cells (17). Zhang et al (19) revealed that GBS exerts an indirect
effect by mediating human papillomavirus infection in patients with
CIN. Additionally, GBS has a higher abundance in CIN2 than in CIN1,
and has been suggested as a crucial biomarker for predicting the
severity of CIN (20). However, to
the best of our knowledge, the underlying mechanism of the
association between GBS and CC progression, especially AC
progression, remains unclear.
Transcriptomics, through differential gene
expression and gene ontology analysis, provides a valuable tool for
evaluating mechanistic changes during the interaction between host
cells and bacteria (21–23). Molecular alterations in
cancer-related genes, coupled with the corresponding changes in
signaling pathways, guide the development of novel precision
medicine treatments for cancer (24). In alignment with this, leveraging
transcriptomic analyses also facilitates the exploration of drug
repurposing strategies, allowing the identification of existing
drugs with potential anticancer properties (25).
In the present study, transcriptomic analyses were
performed to elucidate the gene and pathway transcriptome profile
of the host CC after GBS infection, particularly for the AC type.
Building upon observed RNA sequencing (RNA-seq) data changes, the
present approach aimed to connect the findings from the
transcriptomic analysis with the potential for drug repurposing. By
identifying specific gene expression patterns and altered pathways,
this strategy provides a targeted and efficient route for
identifying existing drugs that could be repurposed as anticancer
therapies, paving the way for further investigation and clinical
development.
Materials and methods
Public data acquisition
Expression and clinical data of 304 samples from The
Cancer Genome Atlas (TCGA)-CESC were acquired from TCGA Genomic
Data Commons portal (https://portal.gdc.cancer.gov/). Metagenomic profiles
of the same cohort were sourced from TCGA metagenomic microbiome
study (https://ftp.microbio.me/pub/cancer_microbiome_analysis/TCGA).
The detailed pipeline for obtaining metagenomic profiles was
outlined in a previous study (26).
Relative abundance normalization was applied to the metagenomic
data, and Streptococcus absence was defined as a relative
abundance equal to 0.
DESeq2 was employed to investigate differentially
expressed genes (DEGs) between Streptococcus-present and
-absent groups. Significance criteria included an adjusted
P<0.05 and an absolute log2-fold change ≥0.5
(27).
Weighted gene coexpression network analysis (WGCNA)
is a computational method that organizes genes into clusters or
modules based on their coexpression patterns across different
samples. This approach reveals the complex relationships between
genes, providing insights into their roles in biological processes
and their connections to disease phenotypes (28,29).
In the present study, WGCNA was applied to identify significant
genes associated with Streptococcus in samples from patients
with AC. A soft threshold of β=7 and a scale-free topology fitting
index (R2) of 0.95 were used for matrix transformation
according to the scale-free topological criteria. The modules
exhibiting the highest correlation with Streptococcus
presence were selected. Key genes related to Streptococcus
in the AC samples were then identified if they met the following
criteria: Gene significance (GS) >0.5, module membership (MM)
>0.8 and P<0.05.
Pathway enrichment analysis was conducted using two
web-based tools, including g:Profiler (https://biit.cs.ut.ee/gprofiler/gost; version 2023)
(30) and Enrichr (https://maayanlab.cloud/Enrichr/; version June 8,
2023) (31), employing the Kyoto
Encyclopedia of Genes and Genomes pathway database (32) and Molecular Signatures Database
Hallmark 2020 (33). Statistical
significance was established with an adjusted P<0.1 and a
minimum of four genes per pathway. Significant enriched pathways
were visualized using ggplot2 version 3.4.2 (34).
HeLa cell culture
HeLa cells, also known as human AC cell lines (KCLB
number 10002; lot no. 59726; passage no. 98; Korean Cell Line Bank;
Korean Cell Line Research Foundation), were thawed, cultured and
passaged within T75 cell culture flasks using Minimum Essential
Medium (Gibco; Thermo Fisher Scientific, Inc.) supplemented with
10% fetal bovine serum (Gibco; Thermo Fisher Scientific, Inc.) in a
humidified atmosphere with 5% CO2 at 37°C.
Two subculturing instances were conducted to ensure
cellular stability for experimentation. Initially, cells were
seeded at 5–6×105 cells per flask. Media exchange
occurred every 2 days or as needed based on cellular conditions,
with harvesting conducted once cells achieved 90–95%
confluence.
Streptococcus agalactiae exposure
experiment using HeLa cells
GBS or Streptococcus agalactiae strain NCTC
818 [G19] (13813; American Type Culture Collection) was aerobically
grown at 37°C in Tryptic Soy Broth (Becton, Dickinson and Company)
according to the manufacturer's protocol.
HeLa cells were infected with GBS for 0, 2, 6 and 24
h at a multiplicity of infection equal to 100 (GBS:cell=100:1).
Right before the experiment, GBS in the bacterial medium was
precipitated and resuspended in human medium (Minimum Essential
Medium supplemented with 10% fetal bovine serum), ensuring the
concentration remained consistent with the 100:1 ratio. For the
control group, human medium without bacteria was administered.
Following this, the flasks were incubated for 0 (baseline), 2, 6
and 24 h in a humidified atmosphere with 5% CO2 at
37°C.
All experiments were conducted in triplicate.
RNA isolation and sequencing
Total RNA was isolated from HeLa cells cocultured
with GBS using the RNeasy Mini Kit (74104; Qiagen GmbH) according
to the manufacturer's protocol. Ribosomal RNA depletion was
performed using the MGIEasy rRNA Depletion Kit (1000005953; MGI
Tech Co., Ltd) and library preparation was performed using the
MGIEasy RNA Directional Library Prep Set (1000006386; MGI Tech Co.,
Ltd.). The final library concentration was 113 nM. Sample
quantification was performed using the Qubit ssDNA Assay Kit
(Q10212; Invitrogen; Thermo Fisher Scientific, Inc.) and the
Invitrogen Qubit Fluorometer (Q33216; Thermo Fisher Scientific,
Inc.). Sample quality was assessed using an Agilent 2100
Bioanalyzer (G2939AA; Agilent Technologies, Inc.). Sequencing was
conducted on the DNBSEQ-G400 sequencer (MGI Tech Co., Ltd.) using
the DNBSEQ-G400RS-High throughput sequencing FCL PE100 kit
(1000016949; MGI Tech Co., Ltd.), generating 100-bp paired-end
reads.
FASTQ files from the sequencer underwent quality
assessment using FASTQC v0.12.1 and Multiqc version 1.24.1
(35,36). Cutadapt version 4.9 was employed to
eliminate low-quality reads and sequencing adapters (37). The alignment and quantification of
RNA-seq data were performed using STAR version 2.7.11b (38). Detailed information on sequenced and
mapped samples can be found in Table
SI.
Time-series transcriptomic analysis of
GBS-HeLa cell interactions
TimeSeriesAnalysis (TiSA) is a tool including
analysis and visualization packages for RNA-seq and microarrays. It
facilitates the extraction of significant genes from time series
transcriptomic data by assessing differential gene expression along
both condition and temporal axes (39).
In the present study, TiSA was used to identify
significant genes meeting the criteria of an absolute
log2-fold change ≥1 and an adjusted P<0.05 from
longitudinal transcriptomic data, elucidating the interaction
between HeLa cells and GBS.
A principal component analysis (PCA) plot was
generated using the first and second principal components to
visualize the differences between control and GBS-exposed HeLa cell
groups at various time points. This was conducted using the
plot_PCA_TS() function from the TiSA package. The partitioning
algorithm based on recursive thresholding (PART) from the
clusterGenomics R package, version 1.0 (40), was applied for clustering. DEGs and
clusters were visualized using heatmaps, created with the
ComplexHeatmap package version 2.8.0 (http://www.bioconductor.org/packages/devel/bioc/html/ComplexHeatmap.html).
To elucidate the biological meaning of the clusters, Enrichr and
g:Profiler were used, with the criteria as aforementioned.
Reverse transcription-quantitative PCR
(RT-qPCR)
Total RNA was extracted from GBS-exposed HeLa cells
after 24 h to validate the mRNA expression of the genes belonging
to the MAPK pathway [fibroblast growth factor 21 (FGF21), nerve
growth factor (NGF), IL1A and IL1B] and mTORC1 pathway [DNA damage
inducible transcript 3 (DDIT3), cystathionine γ-lyase (CTH),
asparagine synthetase (glutamine-hydrolyzing) (ASNS) and nuclear
protein 1 (NUPR1) using the RNeasy Mini Kit (74104; Qiagen GmbH).
RT-qPCR was performed using a one-step TOPreal™ SYBR Green RT-qPCR
Kit (cat. no. RT432S; Enzynomics Co., Ltd.) on a Thermal Cycler
Dice Real Time System TP800 (Takara Bio, Inc.). This one-step kit
combines high-yield TOPscript reverse transcriptase (cat. no.
RT002; Enzynomics Co., Ltd.) with chemically modified Taq
polymerases. According to the manufacturer's instructions, reverse
transcription was performed at 50°C for 30 min, and the
thermocycling conditions were as follows: Initial denaturation at
95°C for 10 min, followed by 45 cycles of denaturation at 95°C for
5 sec, and annealing and elongation at 60°C for 30 sec. The primer
sequences utilized are presented in Table SII.
All reactions were performed in triplicate. The
relative expression levels of the target genes were normalized to
the reference gene (GAPDH) using the ΔCq method. Fold changes in
gene expression were calculated using the 2−ΔΔCq method,
with the control sample serving as the calibrator (41).
Western blotting
To validate the activation of significantly enriched
pathways in HeLa cells after 24 h of exposure to GBS, western
blotting was performed. HeLa cells were co-cultured with GBS for 24
h, washed with cold PBS and lysed using cold M-PER™ Mammalian
Protein Extraction Reagent containing 0.2% Halt™ Protease Inhibitor
Cocktail (Thermo Fisher Scientific, Inc.). Tumor cell lysates (20
µg/lane; protein concentration quantified using a Bradford assay)
were loaded onto a 4–20% Mini-PROTEAN TGX precast gel (Bio-Rad
Laboratories, Inc.) and electrophoresed for 10 min at 80 V,
followed by 60–90 min at 120 V. The proteins were transferred to
PVDF membranes for 45 min at 12 V using the semi-dry blotting
system (ATTO Corporation). After blocking with 5% skim milk for 1 h
at room temperature, the membranes were incubated overnight at 4°C
with the following primary antibodies: ERK1/2, phosphorylated
ERK1/2 (p-ERK1/2), phosphorylated mTOR at Ser2448 (p-mTOR), p38,
phosphorylated p38 (p-p38), panAKT (each at a dilution of 1:1,000),
mTOR (dilution, 1:2,000), phosphorylated AKT at Ser473 (p-AKT) and
Ki67 (each at a dilution of 1:5,000). Detailed information about
the antibodies is provided in Table
SIII.
Following incubation, the membranes were treated for
1 h at room temperature with anti-rabbit IgG HRP-linked antibody
and anti-mouse IgG HRP for β-actin antibody at a dilution of
1:5,000. The membranes were washed three times with 1X TBS with
0.1% Tween-20 for 10 min. The PVDF membranes were then processed
with a PicoEPD Western Blot Detection kit (EBP1073; ELPIS-Biotech,
Inc.). The chemiluminescence images were captured using the
Amersham Imager 600 (GE Healthcare), and the resulting bands were
semi-quantified to determine relative protein levels using ImageJ
software version 1.54 (National Institutes of Health). The
experiments were conducted in triplicate for both control and
treatment groups.
Therapeutic agents
Candidate therapeutic agents were selected through a
drug repurposing approach. One valuable tool in drug repurposing is
the Connectivity Map (CMap) tool (42). CMap is a powerful platform for
unraveling connections among drugs, genes and diseases. The
fundamental principle involves comparing the gene expression
profiles induced by a specific drug with those associated with
particular diseases (42).
Genes from TiSA clusters of interest were uploaded
to the CMap web-based tool version 1.1.1.43 (https://clue.io/). Potential anticancer drugs were
identified based on their tau score, which is converted from
normalized connectivity scores, comparing them with values of that
disease for all the drugs in the reference database (43,44).
Tau scores exceeding 90 or below-90 between two signatures indicate
strong connectivity. A negative connectivity signifies the reversal
of the disease signature by the drug (43).
Binimetinib (MEK162) was purchased from Selleck
Chemicals (cat. no. S7007). A 10 mM stock solution was generated in
DMSO and stored at −20°C.
Ridaforolimus (deforolimus; cat. no. HY-50908;
MedChemExpress) was prepared as 10 mM stock solutions with DMSO and
stored at −20°C.
Specially tailored working solutions of these drugs
were prepared for immediate use. In the case of monotherapy,
binimetinib and ridaforolimus were serially diluted to
concentrations of 0.625, 1.25, 2.5, 5, 10, 15 and 20 µM. For
combination therapy of binimetinib and ridaforolimus, a 1:1 ratio
of the drugs was applied, with serial dilutions of 1.25, 2.5, 5,
10, 15 and 20 µM used. For all treatments the cells were incubated
at 37°C with 5% CO2. Each experiment was conducted in
triplicate.
Trypan blue assay
A trypan blue assay was utilized to evaluate the
viability of HeLa cells following exposure to GBS or therapeutic
agents. After washing with PBS, adherent cells in a 90-mm plate
were dissociated using 2 ml of 0.25% trypsin-EDTA for 2 min at room
temperature. The cells were then resuspended in 8 ml cell culture
medium. A portion of this cell suspension was mixed with an equal
volume of 0.4% trypan blue (Gibco; Thermo Fisher Scientific, Inc.)
for 2 min at room temperature. The number of viable and non-viable
cells was counted in triplicate using a hemocytometer.
MTS assay
After reaching confluence in a T75 flask, HeLa cells
were subcultured into 96-well plates at a concentration of
2×104 cells/100 µl. Following a 24-h incubation period,
binimetinib and/or ridaforolimus were added into each well using
serial dilutions of 1.25, 2.5, 5, 10, 15 and 20 µM.
Subsequently, the cells were incubated at 37°C with
5% CO2 for either 24 or 48 h. Afterwards, 20 µl
CellTiter 96® AQueous One Solution (G3582; Promega
Corporation) reagent was added to each well. The plate underwent an
additional 4-h incubation at 37°C with 5% CO2, and the
absorbance at 490 nm was measured using a microplate reader
(Multiskan™ FC; Thermo Fisher Scientific, Inc.).
The absorbance intensity at 490 nm for both the
control and treatment groups was normalized to the absorbance of
the background. Subsequently, cell viability percentages for
different treatment groups were calculated using the following
formula: % viability=adjusted optical density
(OD)sample/mean (adjusted ODcontrol), where
adjusted ODsample=ODsample-mean
(ODbackground), and adjusted
ODcontrol=ODcontrol-mean
(ODbackground).
Cell apoptosis assay
To measure the induction of apoptosis,
5×105 HeLa cells were plated in 90-mm plates and allowed
to adhere and proliferate for 48 h. Cells were then treated with
either 15 µM binimetinib, 20 µM ridaforolimus, a combination of
both (5 µM binimetinib + 5 µM ridaforolimus) or DMSO (vehicle
treatment) as a control at 37°C with 5% CO2.
After 48 h, cells were harvested and transferred
into tubes at a concentration of 5×105 cells/ml. Cell
death was assessed using an annexin V-FITC apoptosis detection kit
(ab14085; Abcam) with 5 µl annexin V-FITC and 5 µl PI, followed by
a 5-min incubation at room temperature in the dark. The apoptosis
assay data were obtained using a BD FACSCalibur flow cytometer (BD
Biosciences). The graph showing the percentages of apoptotic and
necrotic cells was created using the Floreada tool (https://floreada.io; WASM version SIMD).
Dose-response curve and synergy
scores
To estimate the efficacy of anticancer drugs, the
dose-response curve R package version 3.0.1 was used to determine
the IC50 (45). The
IC50 values represent the concentrations of each drug
required to achieve a 50% inhibition of cancer cell viability.
To predict the combination (binimetinib and
ridaforolimus) effect, the synergy score was calculated using the
SynergyFinder R package version 3.8.2 (46). If the synergy score is between −10
and 10, the effect of the combination of the two drugs is additive,
if the score is >10, the effect is synergistic, and if the score
is <-10, the combination is considered antagonistic (46,47).
Effect of therapeutic agents on
GBS-exposed HeLa cells
To evaluate the effect of therapeutic agents on
GBS-exposed HeLa cells and to compare these effects with those on
HeLa cells not exposed to GBS, GBS and HeLa cells were co-cultured
at a ratio of 100:1 in a humidified atmosphere with 5%
CO2 at 37°C for 6 h. Following this exposure, the
GBS-exposed HeLa cells were treated with 15 µM binimetinib, 20 µM
ridaforolimus, a combination of both (5 µM binimetinib + 5 µM
ridaforolimus) or DMSO as a control at 37°C with 5% CO2.
The number of dead and viable cells after 24 h of treatment was
measured using a trypan blue assay in triplicate as
aforementioned.
Statistical analysis
To compare two groups, an unpaired Student's t-test
was performed. For analyses involving multiple groups, one-way
ANOVA followed by post hoc testing using the Holm-Bonferroni
correction method was performed (48). All statistical analyses were
performed using the stats package version 4.3.3 (https://search.r-project.org/R/refmans/stats/html/00Index.html)
in R version 4.3.3 (https://www.R-project.org/). The results were
visualized using the ggplot2 package version 3.4.2 (34) and ggpubr package version 0.6.0
(https://github.com/kassambara/ggpubr). Analyses were
based on three experimental repeats. In the plots, error bars were
included to represent the dispersion of the variable (standard
deviation). P<0.05 was considered to indicate a statistically
significant difference.
Results
Gene expression and pathway profiles
of Streptococcus-associated CC using TCGA-CESC public data
To investigate the relationship between
Streptococcus and CC, a cohort of 304 TCGA CC cases,
including transcriptomic data and Streptococcus abundance
profiles, were analyzed (Table
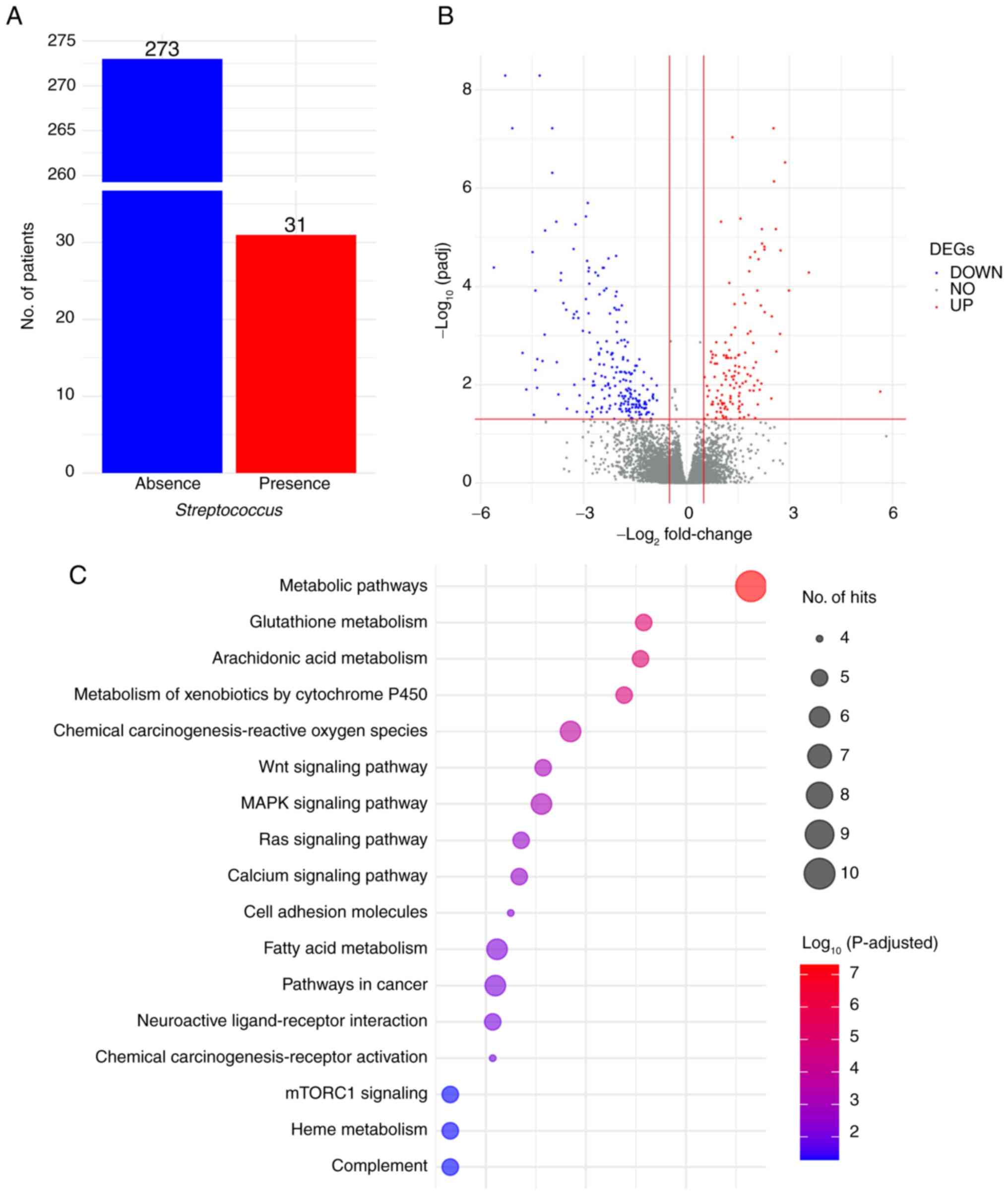
SIV). As shown in Fig. 1A, 31
cases (10.2%) exhibited the presence of Streptococcus.
DEG profiles (Fig.
1B) revealed 128 upregulated and 209 downregulated DEGs
identified using DESeq2 (Table
SV). Pathway enrichment analysis, conducted using the web-based
tools g:Profiler and Enrichr, of the list of 128 upregulated DEGs,
highlighted statistically and biologically significant pathways
associated with cancer, including ‘MAPK signaling pathway’, ‘mTORC1
signaling’, ‘Ras signaling pathway’ and ‘Wnt signaling pathway’, as
well as metabolism pathways such as ‘metabolism of xenobiotics by
cytochrome P450’, ‘fatty acid metabolism’, ‘glutathione metabolism’
and ‘arachidonic acid metabolism’ (Fig.
1C; Table SVI).
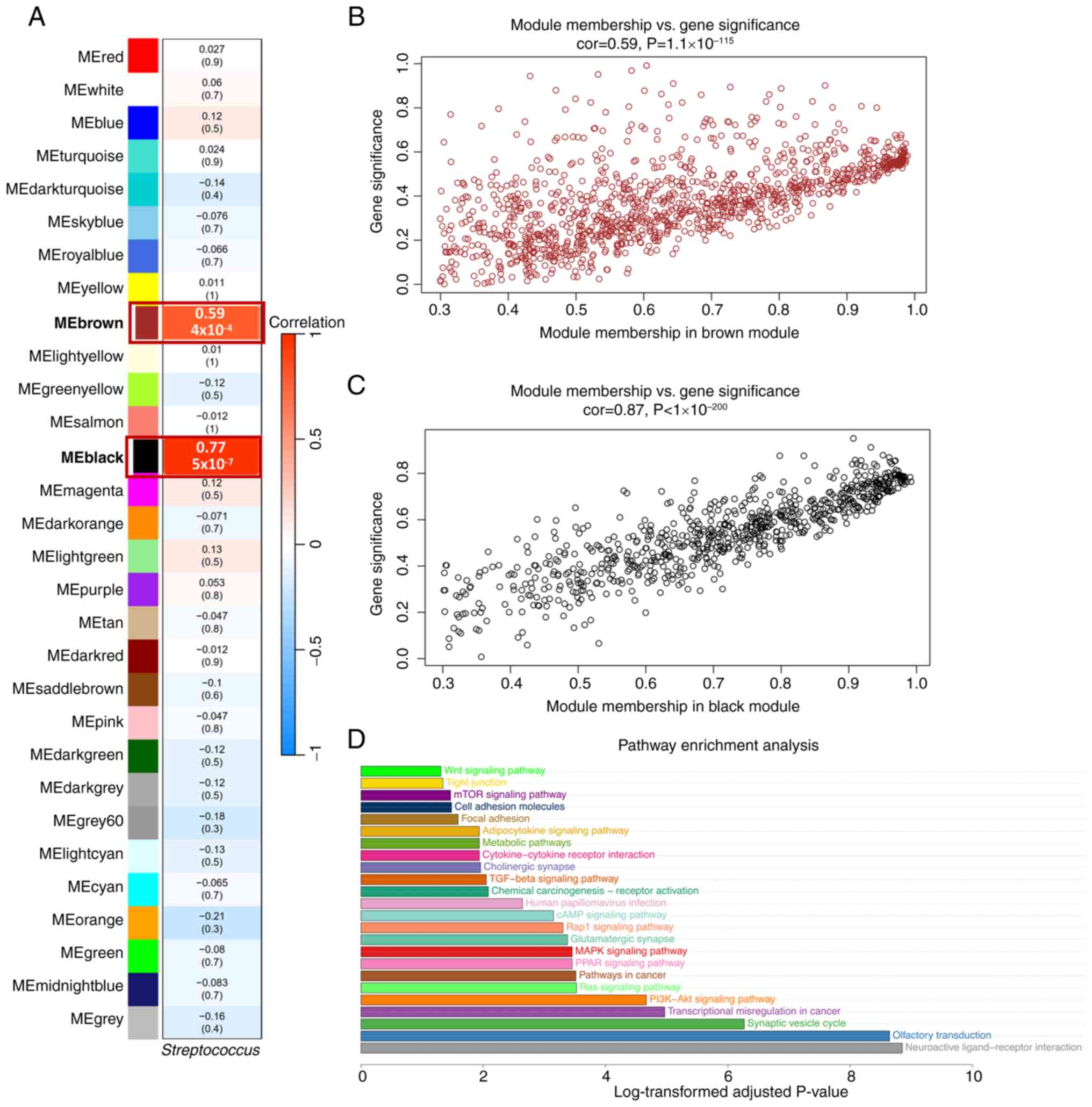
To further explore the specificity of the
relationship between Streptococcus and AC, 31 patients with
AC from a cohort of 304 patients with CC in TCGA were analyzed.
Among them, 3 patients with AC exhibited the presence of
Streptococcus (Table SIV).
Using the WGCNA tool, two significant key modules with high
correlation values, r=0.77 (P=5×10−7) for the black
module and r=0.59 (P=4×10−4) for the brown module, were
identified (Fig. 2A-C; Tables SVII and SVIII).
A total of 500 key genes related to
Streptococcus were selected from the two key modules based
on the following criteria: GS >0.5, MM >0.8 and P<0.05.
Pathway enrichment analysis showed that these genes were involved
multiple oncogenic pathways, including ‘MAPK signaling pathway’,
‘mTOR signaling pathway’, ‘PI3K-AKT signaling pathway’, ‘TGF-beta
signaling pathway’ and ‘Ras signaling pathway’ (Fig. 2D; Table
SIX).
Time-series transcriptomic analysis of
GBS-HeLa cell interaction
To further enhance the insights gained from
TCGA-CESC data, a time-series experiment was performed to
investigate HeLa cell-GBS interactions. Briefly, HeLa cells were
infected with GBS for 0, 2, 6 and 24 h, with corresponding control
groups treated solely with bacteria-free human medium.
Subsequently, the transcriptomic data generated from this
experiment were analyzed using the TiSA package.
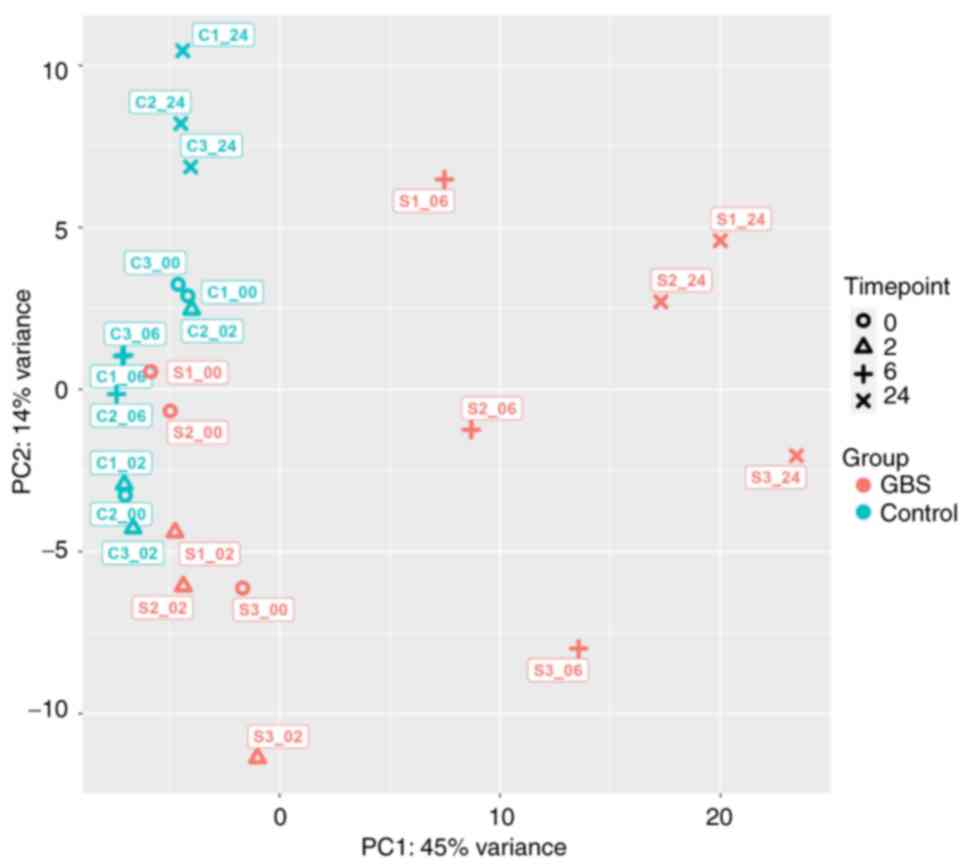
PCA plot, and DEGs between conditional
and temporal groups
The time series PCA plot demonstrated a clear
separation between the GBS-exposed HeLa cell groups at 6 and 24 h
and the other groups. PC1, representing variability over time, and
PC2, representing differences between groups, were utilized for
this analysis. As shown in Fig. 3,
the dominance of PC1, accounting for 45% of the variation, while
PC2 accounted for 14%, indicated that the variation over time was
more pronounced than the variation between groups, which was
especially evident after 6 h. Notably, the groups associated with
GBS stimulation at 6 and 24 h exhibited a distinct separation from
the other groups.
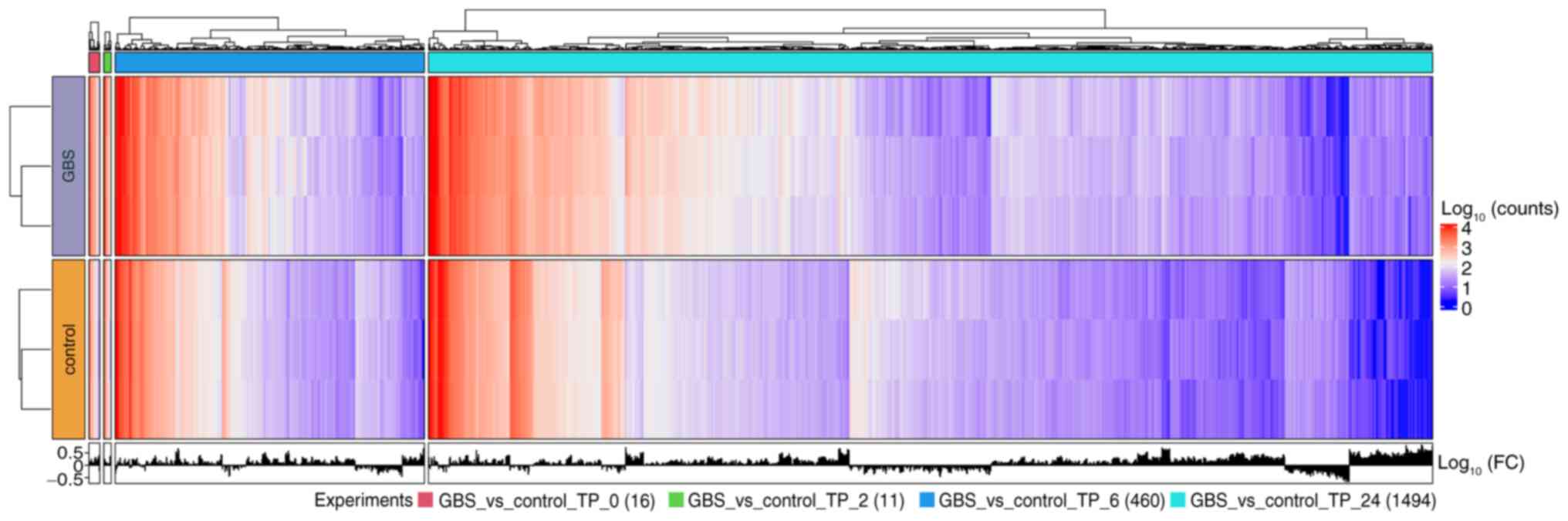
The conditional differential gene expression
analysis for four-time points is presented in Fig. 4. The number of DEGs markedly
increased after 6 h of exposure. Specifically, there were 16, 11,
460 and 1,494 DEGs after 0, 2, 6 and 24 h of exposure,
respectively.
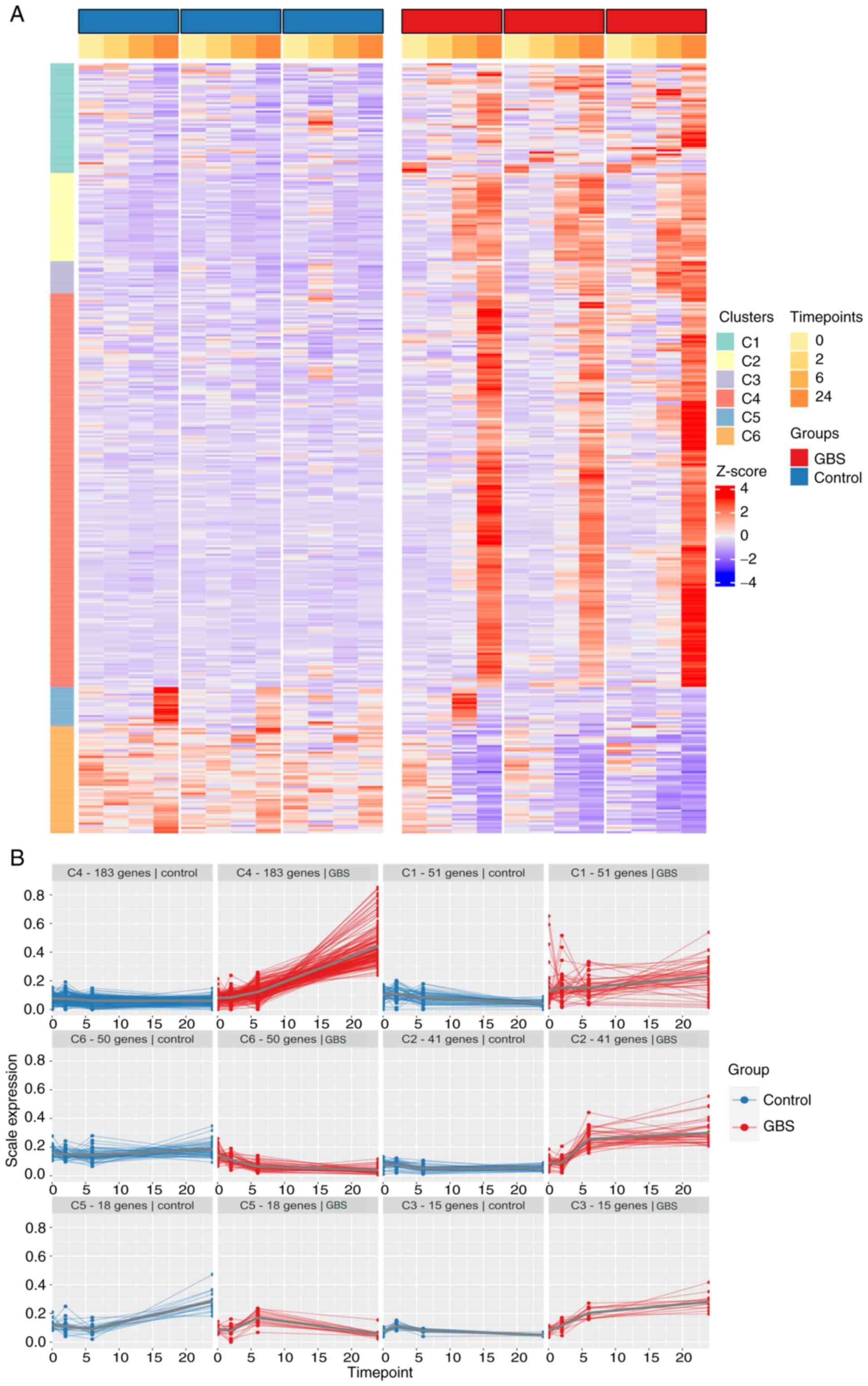
Clustering analysis
All DEGs identified from the conditional
differential gene expression analysis, totaling 1,981 genes, were
utilized for PART clustering, resulting in six clusters, as shown
in Fig. 5A and Table SX. Genes in clusters 1, 2, 3 and 4
showed an upward trajectory between 6 and 24 h, while genes in
cluster 6 decreased during the same period. Cluster 5 exhibited
mixed expression patterns (Fig.
5B).
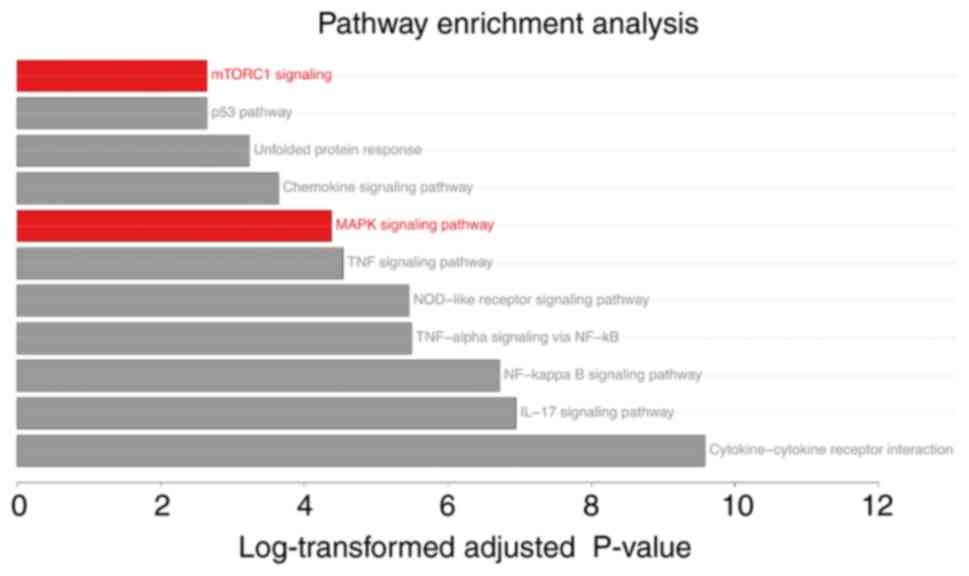
Pathway enrichment analysis
Functional analysis of each cluster was performed
using Enrichr and g:Profiler. Cluster 2 exhibited the most
significant pathways, encompassing both oncogenic pathways (‘MAPK
signaling pathway’, ‘mTORC1 signaling’ and ‘p53 pathway’) and
immune response pathways (‘TNF signaling pathway’, ‘NF-kappa B
signaling pathway’, ‘IL-17 signaling pathway’ and ‘NOD-like
receptor signaling pathway’) (Table
SXI).
The pathway enrichment analyses revealed the
upregulation of ‘MAPK signaling pathway’ and ‘mTORC1 signaling’ in
the HeLa cells-GBS exposure experiment (Fig. 6) and the TCGA-CESC public data
(Fig. 1C). Additionally, activation
of the ‘MAPK signaling pathway’ and ‘mTOR signaling pathway’ was
observed in the group of 3 patients with AC with
Streptococcus presence (Fig.
2D). These findings suggested that the MAPK and mTORC1 pathways
may serve significant roles in AC associated with GBS.
Validation of MAPK and mTORC1
signaling pathway activation after GBS exposure
To validate the activation of the MAPK and mTORC1
pathways, the upregulated expression of genes associated with the
MAPK pathway (FGF21, NGF, IL1A and IL1B) and the mTORC1 pathway
(DDIT3, CTH, ASNS and NUPR1) from cluster 2 was first confirmed
using RT-qPCR. As shown in Fig. 7C,
most of these genes exhibited higher expression levels in HeLa
cells exposed to GBS for 24 h compared with control cells, with the
exception of NGF. This discrepancy may arise from differences in
sensitivity or specificity between RNA-seq and RT-qPCR methods.
Overall, the RT-qPCR results support the activation of the MAPK and
mTORC1 pathways in response to GBS exposure.
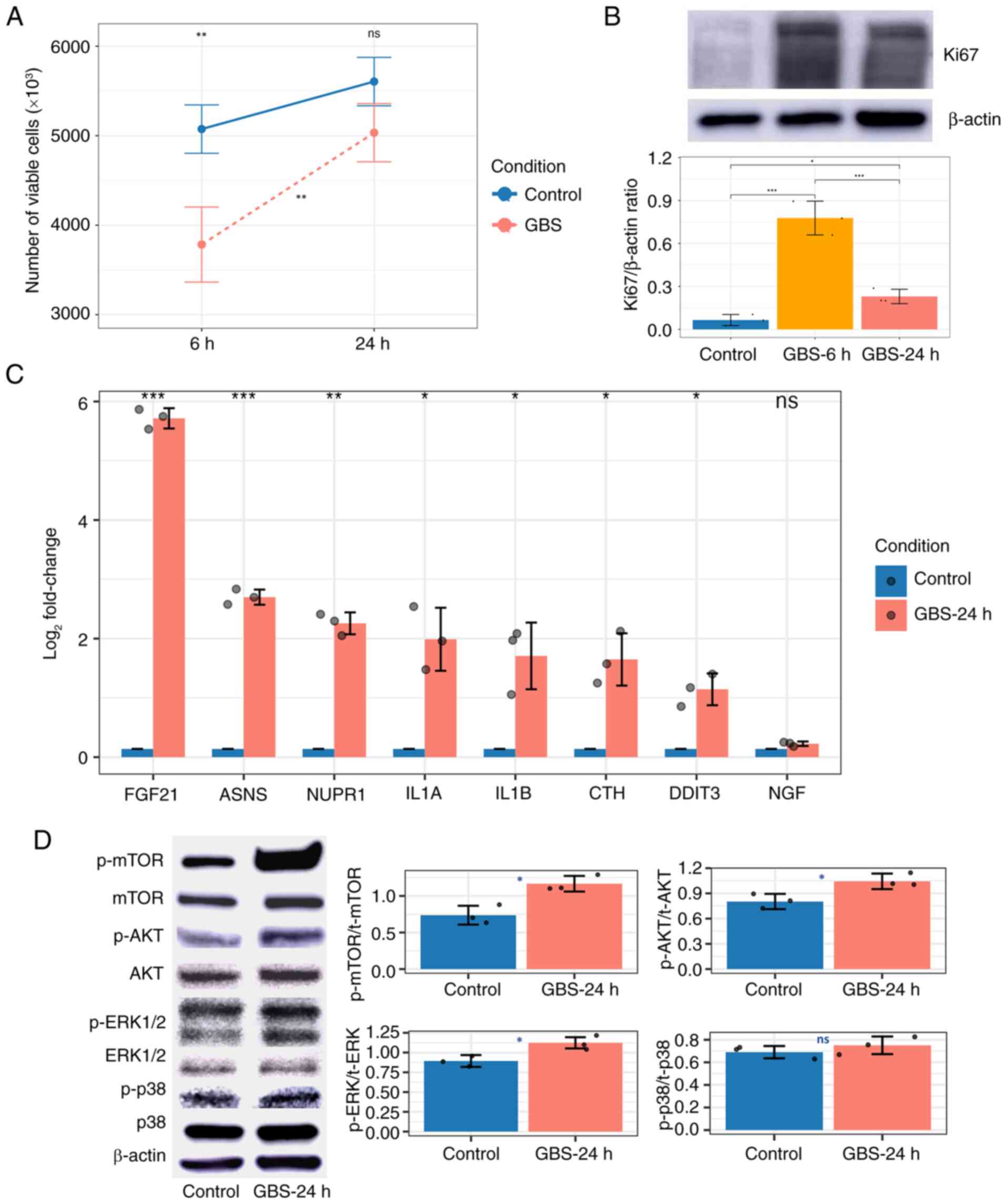 | Figure 7.Effects of GBS on HeLa cells. (A)
Line graph showing HeLa cell proliferation after 6 and 24 h of GBS
exposure, with comparisons with control cells and between two time
points, as measured by the trypan blue assay. (B) Western blot
analysis and corresponding bar plot showing Ki67 expression in HeLa
cells at 6 and 24 h post-GBS exposure. (C) Bar plot showing the
logarithm fold changes in the expression of eight targeted genes as
determined by reverse transcription-quantitative PCR. (D) Western
blot analysis and corresponding bar plots showing the levels of
proteins in HeLa cells 24 h after GBS exposure. Blue, orange and
red indicate the control group, the 6 and 24-h GBS-treated groups,
respectively. *P<0.05, **P<0.01, ***P<0.001. GBS, group B
Streptococcus; ns, not significant; p-, phosphorylated; t-,
total. |
Western blot analysis revealed a significant
increase in the ratios of p-ERK1/2 to total (t-)ERK1/2, p-AKT to
t-AKT and p-mTOR to t-mTOR in HeLa cells exposed to GBS for 24 h
compared with control cells. Although the ratio of p-p38 to t-p38
was higher in the treated HeLa cells than in the control group, the
difference was not statistically significant (Fig. 7D). The elevated phosphorylation of
ERK1/2 indicated enhanced MAPK pathway activity, while the
upregulation of p-AKT and p-mTOR suggested activation of the mTORC1
pathway, further supporting the involvement of these signaling
cascades in the cellular response to GBS exposure.
Additionally, the trypan blue assay demonstrated a
decrease in viable HeLa cell numbers at 6 h compared with control
cells, followed by a significant increase in HeLa cell numbers,
reaching levels similar to control cells by 24 h (Fig. 7A). This cell proliferation
phenomenon was confirmed by examining the expression of Ki67, a
well-known cell proliferation marker. The highest Ki67 expression
was observed after 6 h of exposure and the expression was still
increased at 24 h compared with that in the control group (Fig. 7B). This pattern may reflect cancer
cell proliferation influenced by MAPK and mTORC1 pathway activity
after 6 h of exposure to GBS.
Overall, transcriptomic analysis using public data,
time-series co-culture experiments, and validation through RT-qPCR
and western blotting assays underscored the pivotal role of the
MAPK and mTORC1 signaling pathways. These promising results led to
the hypothesis that inhibiting these pathways could be effective in
treating AC.
Therapeutic agent selection
To further investigate candidate drugs targeting the
MAPK and mTORC1 signaling pathways, the list of genes from cluster
2 was uploaded to the CMap web-based tool. Ridaforolimus (or
deforolimus) was chosen as the mTOR inhibitor due to its highest
negative range score (Table
SXII). Notably, to the best of our knowledge, it has not been
extensively studied in the context of AC or HeLa cells.
While CMap suggested several anticancer drugs to
inhibit the MAPK pathway, these drugs were previously investigated
in HeLa cells (49–51). Therefore, binimetinib, a MEK
inhibitor that has shown efficacy in other cancer types (52,53)
but has not yet been explored in AC, was selected for further
evaluation in the present study.
Evaluating the effects of therapeutic
agents on HeLa cells
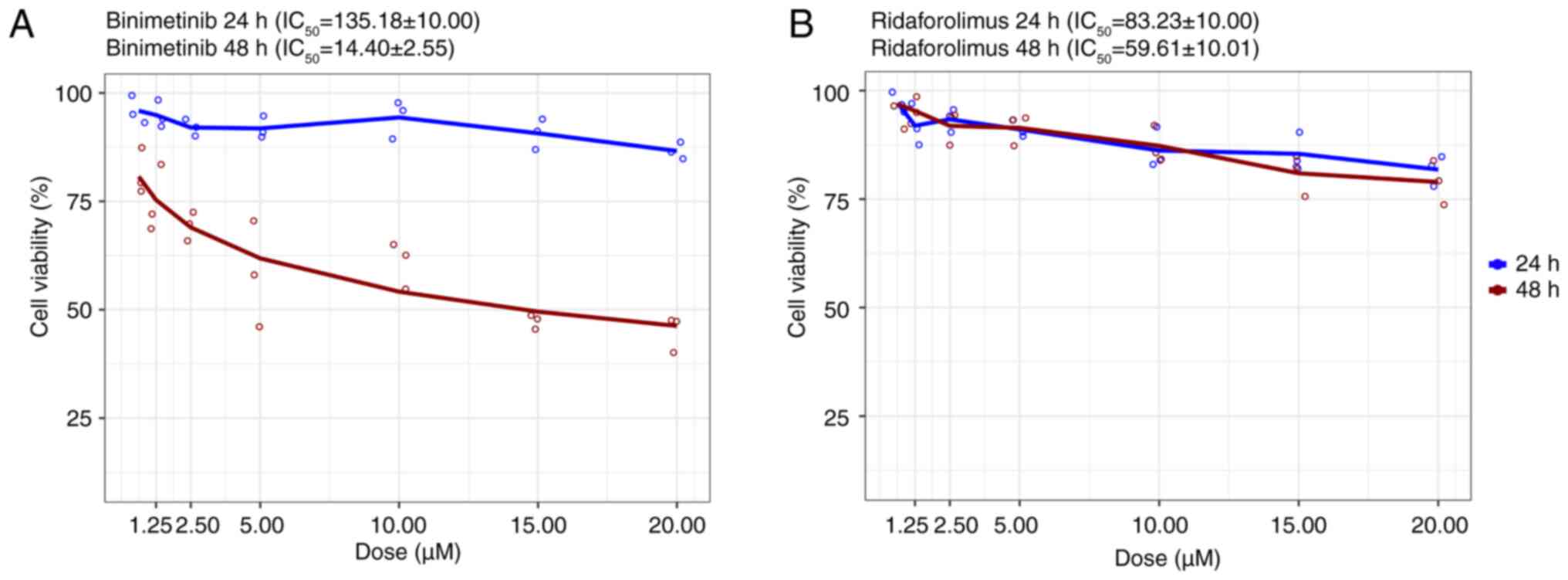
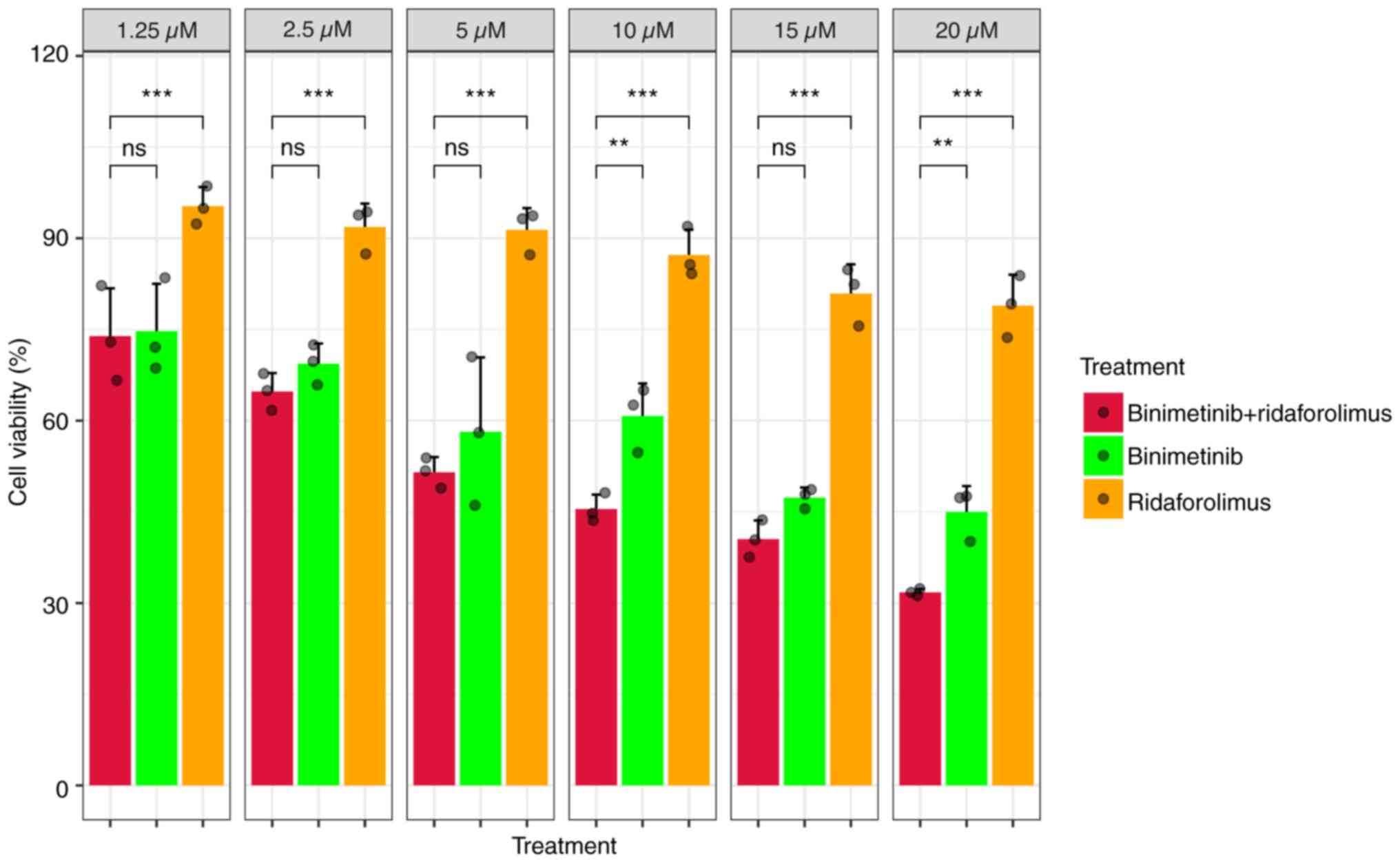
Binimetinib
Although the viability of HeLa cells only notably
decreased after 24 h of exposure to doses >20 µM compared with
the control group, a significant reduction in cell viability was
observed across all binimetinib concentrations (1.25–20 µM)
following 48 h of treatment (Figs.
8A and S1). The
IC50 was determined to be 14.40±2.55 µM (Fig. 8A).
For the apoptosis assay, binimetinib was
administered at a concentration of 15 µM, corresponding to the
IC50 value observed in the MTS assay. The apoptotic
effect after 48 h of binimetinib treatment was higher than that
observed in the control group; nevertheless, no significant
difference was detected between the two groups (Figs. 9 and S2B).
Ridaforolimus
The MTS assay revealed minimal inhibitory effects of
ridaforolimus on HeLa cell proliferation. No significant reduction
in cell viability was observed after treatment with ridaforolimus
for 24 and 48 h (Figs. 8B and
S3). The IC50 of
59.61±10.01 µM further supported this observation (Fig. 8B).
Based on this finding, the highest concentration of
ridaforolimus (20 µM) was used for the apoptosis assay. This dose
exhibited a weak cytotoxic effect (Figs. 9 and S2C).
Combination effect of binimetinib and
ridaforolimus on HeLa cells
A 1:1 ratio of binimetinib and ridaforolimus was
administered to assess the impact of combination therapy.
SynergyFinder was used to predict potential synergy, and the
results suggested that this combination may exhibit an additive
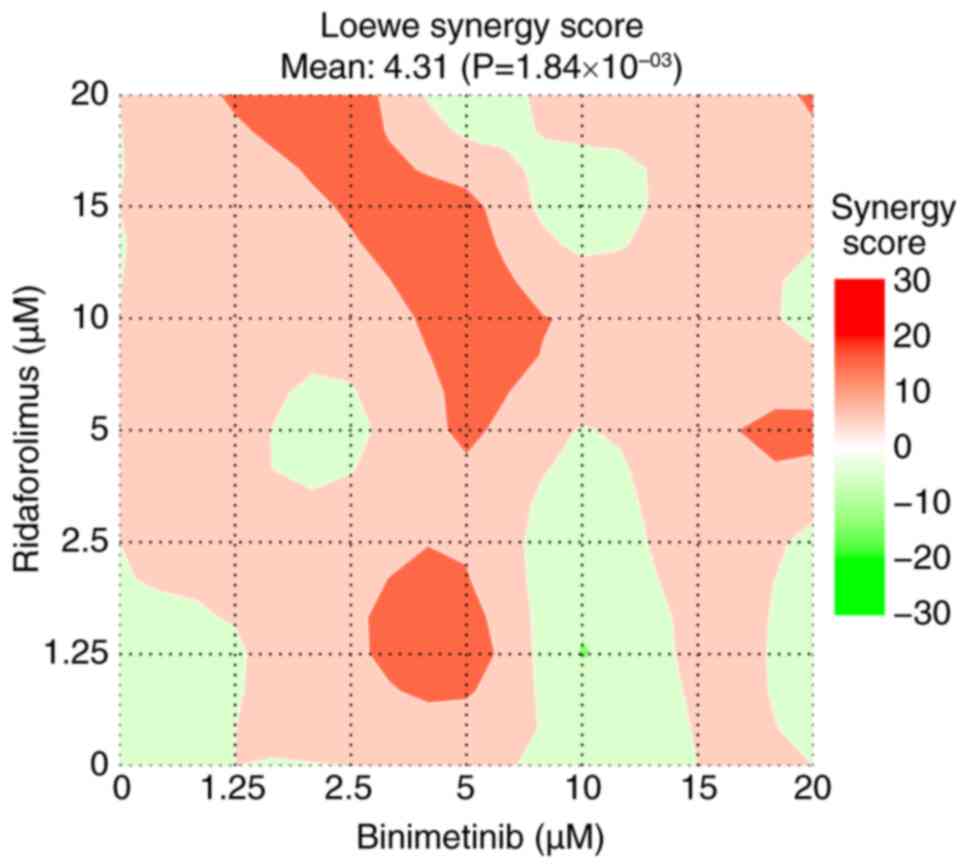
effect, as indicated by a Loewe synergy score of ~4.31, with
statistical significance at P<0.05 (Fig. 10). Compared with ridaforolimus
alone, the combination of binimetinib and ridaforolimus showed
significantly greater inhibition of cell viability in the MTS
assay. However, significant differences between the combination and
binimetinib alone were observed only at concentrations of 10 and 20
µM (Fig. 11).
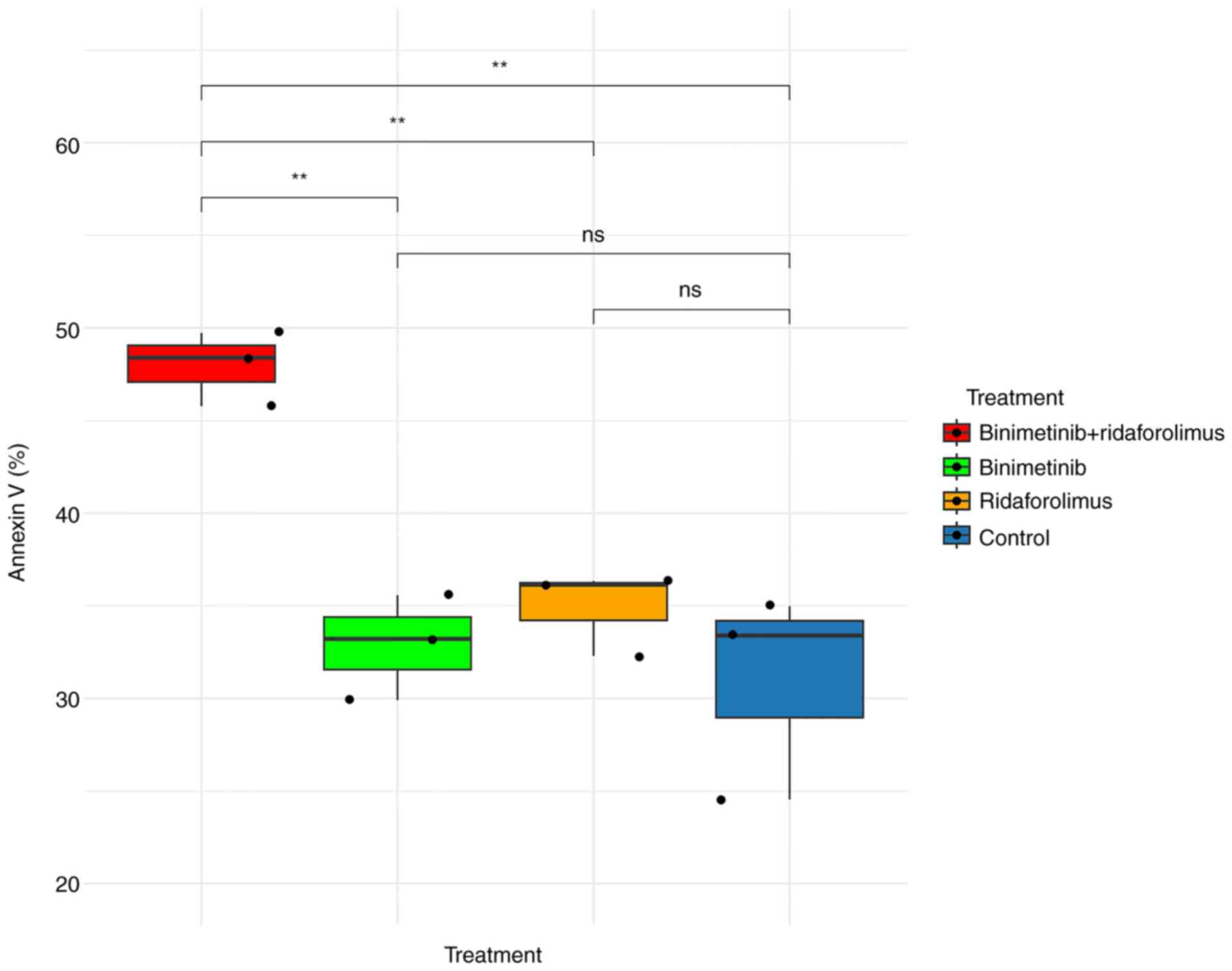
For the apoptosis assay, 5 µM binimetinib and 5 µM
ridaforolimus were used, as this combination effectively reduced
cell viability by 50% as indicated by the MTS assay. Although the
predictive tool suggested an additive effect, the combination
therapy induced notable cytotoxic effects, as evidenced by the
highest percentage of annexin V observed 48 h after treatment in
the apoptosis assay (Figs. 9 and
S2D).
Furthermore, the IC50 of this
combination, calculated using the dose-response curve package, was
determined to be 6.67±0.45 µM for each drug (Fig. S4). In practical terms, this
translates to a dose reduction of 2.16 times for binimetinib and
8.94 times for ridaforolimus to reach the IC50 when
combined, emphasizing the potent impact of this combination.
Evaluating the effects of the
therapeutic agents on HeLa cells with GBS exposure
Based on the promising therapeutic effects
observed, we hypothesized that these anticancer agents
(binimetinib, ridaforolimus and their combination) might affect
HeLa cells exposed to GBS more than the group of cells without
exposure to GBS. Experiments were conducted to confirm this
hypothesis.
After 6 h of exposure of HeLa cells to GBS, the
cells were treated with therapeutic agents (monotherapy and
combination therapy). As shown in Fig.
S5A, both monotherapies and combination therapies exhibited
enhanced effects on HeLa cells exposed to GBS compared with the
control group after 24 h of treatment, with the combination therapy
demonstrating a more pronounced effect than monotherapy.
In addition, the data specifically indicated that
combination therapy was more effective in HeLa cells exposed to GBS
than in those not exposed to GBS (Fig.
S5B).
Discussion
Numerous studies have suggested that
Streptococcus may be involved in cancer initiation,
progression and metastasis. For instance, Streptococcus
anginosus has been associated with activating gastric
tumorigenesis (54).
Streptococcus gallolyticus subsp. Gallolyticus is strongly
linked to promoting human colon cancer cell proliferation by
increasing β-catenin signaling (55). Similarly, Streptococcus
pneumoniae is related to upregulated mTOR2/AKT signaling
pathways for invasion and migration in lung cancer cells (56). Despite these observations, the
understanding of the interaction between Streptococcus and
CC remains limited. Therefore, the present study aimed to elucidate
the underlying mechanism of this interaction, with a specific focus
on the relationship between GBS and AC. The selection of GBS for
the present study was based on its prevalence as a Gram-positive
coccus, often carried asymptomatically by numerous women (57). Furthermore, it has been reported to
be associated with various types of CIN in CC (19,20).
Regarding the CC subtype selection, AC was chosen due to its poor
prognosis compared with SCC (3).
Notably, treatment strategies for AC lack specificity in clinical
settings (58,59), adding significance to the present
investigation.
The comprehensive transcriptomic analysis using
in vivo public data (TCGA-CESC) and in vitro time
series data revealed that the ‘MAPK signaling pathway’ and ‘mTORC1
signaling’ emerged as crucial elements in the host-GBS
interaction.
Elevated MAPK pathway activation is implicated in
cancer occurrence and progression, making it a pertinent target for
investigation (60,61). Research also indicates that
pathogens often target host signaling pathways to regulate a number
of cell processes in cancer, and the most common pathway observed
is MAPK signaling (62). Among the
MAPK cascades, ERK1/2 is particularly noted for its role in
regulating cell proliferation, survival, metabolism, migration and
differentiation (63,64), while p38 is more closely associated
with stress responses and inflammation (65). Nevertheless, the present study
revealed that the interaction between GBS and HeLa cells involved
MAPK signaling via ERK1/2, serving a more dominant role than the
p38 pathway.
The activation of mTORC1 pathways was confirmed by
the upregulation of p-AKT and p-mTOR in HeLa cells exposed to GBS
for 24 h, as demonstrated by western blot analysis. The activated
p-AKT Ser473 can phosphorylate and inhibit TSC complex subunit 2
(TSC2), a negative regulator of mTORC1. The inhibition of TSC2
leads to the activation of the Rheb GTPase, which in turn directly
activates the mTORC1 signaling pathway (66,67).
This pathway serves a crucial role in various cellular processes,
including cell viability, proliferation and metabolism (68,69).
Dysregulation of mTORC1 signaling has been implicated in the
development and progression of cancer (66–68).
The present study also validated the expression of
genes related to the MAPK pathway (FGF21, NGF, IL1A and IL1B) and
the mTORC1 pathway (DDIT3, CTH, ASNS and NUPR1) from cluster 2
using RT-qPCR. Most of these genes were upregulated in HeLa cells
exposed to GBS for 24 h compared with control cells at 24 h.
Previous studies have shown that these genes are associated with
enhancement of MAPK and mTORC1 pathway activation (70–77),
as well as cancer cell progression and proliferation (78–84).
To evaluate the association between cell
proliferation and GBS, HeLa cells were examined after exposure to
GBS for 6–24 h. A significant decrease in HeLa cell numbers was
observed after 6 h of GBS exposure, indicating an initial cytotoxic
effect. This phenomenon was similar to the results of a study by
Tyrrell et al (85), which
examined cell death at 6 h of direct contact between HeLa cells and
GBS. However, after 6 h of exposure, the number of viable cells
increased and reached levels similar to the control cells at 24 h.
This phenomenon was confirmed by elevated Ki67 expression, a common
cell proliferation marker, present during the active phases of the
cell cycle (G1, S, G2 and M) and absent in
resting cells (G0) (86,87).
Ki67 expression was significantly higher in GBS-treated HeLa cells
at 6 and 24 h compared with that in control cells, indicating that
GBS treatment stimulated cell proliferation. The peak in Ki67
expression at 6 h may reflect a rapid initiation of cell cycle
activity, with numerous cells entering or actively progressing
through the cycle. Since Ki67 levels typically peak at
G2 or during the mitosis phase (88), the subsequent decrease at 24 h could
suggest that fewer cells were entering the cell cycle or that cells
that initially proliferated had completed division, resulting in a
lower Ki67 signal at this time point.
According to Burnham et al (89), GBS could protect HeLa cells from
caspase-3-induced apoptosis during early exposure by triggering the
PI3K/AKT signaling pathway. Since mTORC1 signaling is a subsequent
cascade of PI3K/AKT, the present results contributed to
understanding the complex mechanisms underlying the interaction
between HeLa cells and GBS more comprehensively than before.
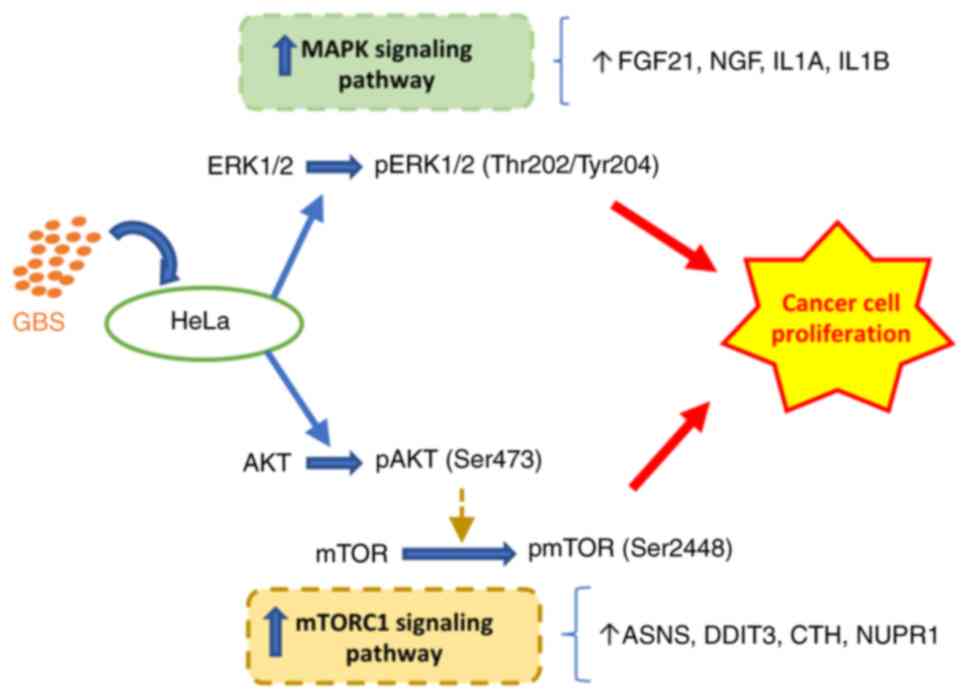
In general, while GBS may initially cause cell
stress or death, it subsequently activates survival and
proliferative pathways, specifically the MAPK and mTORC1 pathways,
leading to the recovery and proliferation of HeLa cells (Fig. 12).
While several signaling pathways, such as the
ERK/MAPK, PI3K/AKT, EGFR/VEGF and Wnt signaling pathways, are
activated in CC development, evidence for specific pathways in AC
is limited (24,90,91).
By highlighting the importance of MAPK and mTORC1 signaling
pathways in the interplay between AC cells and GBS, the present
study provided indirect evidence of their crucial role in AC
development mechanisms. Furthermore, building upon this
observation, a promising therapeutic approach for AC may be
targeting these signaling pathways through a drug repositioning
strategy.
Drug repurposing refers to the process of
identifying novel uses for existing drugs that were initially
developed for a different therapeutic indication (92,93).
This can potentially reduce the time and costs associated with drug
development. In alignment with this strategy, the present study
investigated the MEK inhibitor binimetinib and the mTOR inhibitor
ridaforolimus, which were initially developed for the treatment of
other cancer types (94–97) but have not been previously studied
in the context of AC therapy.
While binimetinib showed a modest cytotoxic effect,
it demonstrated a significant cytostatic impact on HeLa cells after
48 h, as indicated by a decrease in the absorbance at 490 nm
compared with the control group in the MTS assay. The cytostatic
effect refers to its ability to inhibit or slow down the viability
and proliferation of cancer cells, providing a means of controlling
cancer progression (98).
Conversely, ridaforolimus exhibited limited efficacy in achieving
the desired IC50 level after 48 h, accompanied by mild
cytotoxic effects. These findings prompted further investigation
into strategies to enhance the effectiveness of both binimetinib
and ridaforolimus.
Evidence suggests the efficacy of mTOR inhibitors,
such as rapamycin and its analogs, in cancer treatment (66,99).
However, the development of resistance poses a challenge to their
long-term effectiveness (100).
Targeted inhibition of mTOR can induce MAPK reactivation and lead
to resistance to single mTOR inhibition (24,101).
To address this challenge, a combinatorial approach targeting mTOR
and MAPK signaling should be considered for a more effective
response to therapies (102,103). Accordingly, a combination study
involving binimetinib and ridaforolimus was conducted. Despite the
synergy score suggesting an additive effect, the MTS and apoptosis
assays revealed that the combination outperformed using binimetinib
or ridaforolimus alone by significantly inhibiting cell
proliferation at the concentrations of 10 and 20 µM and exhibiting
a robust cytotoxic effect. This outcome underscored the importance
of exploring combination therapies, as they may yield more
effective results than monotherapy treatments in AC. In particular,
the combination (binimetinib and ridaforolimus) was more effective
in the group of HeLa cells with GBS exposure than in the group
without exposure. This indicated the important role of this
combination in treating patients with AC with GBS dominant in their
microbiome profile.
A limitation of the present study is the absence of
in vivo experiments, which restricts the ability to fully
understand the physiological relevance and systemic impact of MAPK
and mTORC1 pathway activation in response to GBS exposure. Future
in vivo studies are needed to validate the findings and
assess their implications in a more complex biological context, as
well as to assess the potential efficacy and safety of these drugs
for AC management.
In conclusion, the present transcriptomic analysis
revealed the complex molecular interaction between GBS and AC,
highlighting the crucial involvement of the ‘MAPK signaling
pathway’ and ‘mTORC1 signaling’. The combination of two anticancer
drugs targeting these pathways, binimetinib and ridaforolimus,
might become a potential therapy for the treatment of AC, offering
a promising direction for future research and clinical
investigation.
Supplementary Material
Supporting Data
Supporting Data
Supporting Data
Supporting Data
Supporting Data
Supporting Data
Supporting Data
Supporting Data
Supporting Data
Supporting Data
Supporting Data
Supporting Data
Acknowledgements
Not applicable.
Funding
This research was supported by the Brain Pool program funded by
the Ministry of Science and ICT through the National Research
Foundation of Korea (grant no. RS-2023-00283791).
Availability of data and materials
The RNA sequencing data have been deposited in the
National Center for Biotechnology Information Gene Expression
Omnibus database under accession number GSE264492 or at the
following URL: https://www.ncbi.nlm.nih.gov/geo/query/acc.cgi?acc=GSE264492.
All other data generated in the present study may be requested from
the corresponding author.
Authors' contributions
HSH and JHS were responsible for study conception.
HSH, JHS, HDTN and TML were responsible for study design. HDTN,
TML, YC, DL, DRJ and YJ performed the experiments. HDTN, TML, DL
and HSH analyzed the data. HDTN wrote the original draft. IS, JC,
NJYP, GOC, YC, OEL, YJ and DRJ contributed to data interpretation
and revised the manuscript. HDTN and TML confirm the authenticity
of all the raw data. All authors have read and approved the final
manuscript.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Sung H, Ferlay J, Siegel RL, Laversanne M,
Soerjomataram I, Jemal A and Bray F: Global cancer statistics 2020:
GLOBOCAN estimates of incidence and mortality worldwide for 36
cancers in 185 countries. CA Cancer J Clin. 71:209–249. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Burmeister CA, Khan SF, Schäfer G, Mbatani
N, Adams T, Moodley J and Prince S: Cervical cancer therapies:
Current challenges and future perspectives. Tumour Virus Res.
13:2002382022. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Couvreur K, Naert E, De Jaeghere E,
Tummers P, Makar A, De Visschere P, Van Bockstal M, Van Dorpe J, De
Neve W, Denys H and Vandecasteele K: Neo-adjuvant treatment of
adenocarcinoma and squamous cell carcinoma of the cervix results in
significantly different pathological complete response rates. BMC
Cancer. 18:11012018. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Blake SJ, Wolf Y, Boursi B and Lynn DJ:
Role of the microbiota in response to and recovery from cancer
therapy. Nat Rev Immunol. 24:308–325. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Sun J, Chen F and Wu G: Potential effects
of gut microbiota on host cancers: Focus on immunity, DNA damage,
cellular pathways, and anticancer therapy. ISME J. 17:1535–1551.
2023. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Wu S, Ding X, Kong Y, Acharya S, Wu H,
Huang C, Liang Y, Nong X and Chen H: The feature of cervical
microbiota associated with the progression of cervical cancer among
reproductive females. Gynecol Oncol. 163:348–357. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Tango CN, Seo SS, Kwon M, Lee DO, Chang HK
and Kim MK: Taxonomic and functional differences in cervical
microbiome associated with cervical cancer development. Sci Rep.
10:97202020. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Xu X, Zhang Y, Yu L, Shi X, Min M, Xiong
L, Pan J, Liu P, Wu G and Gao G: A cross-sectional analysis about
bacterial vaginosis, high-risk human papillomavirus infection, and
cervical intraepithelial neoplasia in Chinese women. Sci Rep.
12:66092022. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Audirac-Chalifour A, Torres-Poveda K,
Bahena-Román M, Téllez-Sosa J, Martínez-Barnetche J,
Cortina-Ceballos B, López-Estrada G, Delgado-Romero K,
Burguete-García AI, Cantú D, et al: Cervical microbiome and
cytokine profile at various stages of cervical cancer: A pilot
study. PLoS One. 11:e01532742016. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Nguyen HDT, Le TM, Lee E, Lee D, Choi Y,
Cho J, Park NJ, Chong GO, Seo I and Han HS: Relationship between
human papillomavirus status and the cervicovaginal microbiome in
cervical cancer. Microorganisms. 11:14172023. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Hu J, Wu Y, Quan L, Yang W, Lang J, Tian G
and Meng B: Research of cervical microbiota alterations with human
papillomavirus infection status and women age in Sanmenxia area of
China. Front Microbiol. 13:10046642022. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Mulato-Briones IB, Rodriguez-Ildefonso IO,
Jiménez-Tenorio JA, Cauich-Sánchez PI, Méndez-Tovar MDS,
Aparicio-Ozores G, Bautista-Hernández MY, González-Parra JF,
Cruz-Hernández J, López-Romero R, et al: Cultivable microbiome
approach applied to cervical cancer exploration. Cancers (Basel).
16:3142024. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Kang GU, Jung DR, Lee YH, Jeon SY, Han HS,
Chong GO and Shin JH: Potential association between vaginal
microbiota and cervical carcinogenesis in korean women: A cohort
study. Microorganisms. 294:2942021. View Article : Google Scholar
|
|
14
|
Armistead B, Oler E, Adams Waldorf K and
Rajagopal L: The double life of Group B streptococcus: Asymptomatic
colonizer and potent pathogen. J Mol Biol. 431:2914–2931. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Aksu B and Yanilmaz O: Group B
streptococci induce interleukin 8 production in human cervical
epithelial cell cultures: The role of capsule polysaccharide. Clin
Exp Health Sci. 9:49–52. 2018.
|
|
16
|
Maisey HC, Doran KS and Nizet V: Recent
advances in understanding the molecular basis of group B
Streptococcus virulence. Expert Rev Mol Med. 10:e272008. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Patras KA, Rösler B, Thoman ML and Doran
KS: Characterization of host immunity during persistent vaginal
colonization by Group B Streptococcus. Mucosal Immunol.
8:1339–1348. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Patras KA, Wang NY, Fletcher EM, Cavaco
CK, Jimenez A, Garg M, Fierer J, Sheen TR, Rajagopal L and Doran
KS: Group B Streptococcus CovR regulation modulates host immune
signalling pathways to promote vaginal colonization. Cell
Microbiol. 15:1154–1167. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Zhang C, Liu Y, Gao W, Pan Y, Gao Y, Shen
J and Xiong H: The direct and indirect association of cervical
microbiota with the risk of cervical intraepithelial neoplasia.
Cancer Med. 7:2172–2179. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Lee YH, Kang GU, Jeon SY, Tagele SB, Pham
HQ, Kim MS, Ahmad S, Jung DR, Park YJ, Han HS, et al: Vaginal
microbiome-based bacterial signatures for predicting the severity
of cervical intraepithelial neoplasia. Diagnostics (Basel).
10:10132020. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Mutz KO, Heilkenbrinker A, Lönne M, Walter
JG and Stahl F: Transcriptome analysis using Next-generation
sequencing. Curr Opin Biotechnol. 24:22–30. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Ahmed W: RNA-seq resolving host-pathogen
interactions: Advances and applications. Ecol Genet Genom.
15:1000572020.
|
|
23
|
Nathan S: Transcriptome profiling to
understand Host-bacteria interactions: Past, present and future.
ScienceAsia. 46:503–513. 2020. View Article : Google Scholar
|
|
24
|
Yip HYK and Papa A: Signaling pathways in
cancer: Therapeutic targets, combinatorial treatments, and new
developments. Cells. 10:6592021. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Kwon OS, Kim W, Cha HJ and Lee H: In
silico drug repositioning: From Large-scale transcriptome data to
therapeutics. Arch Pharm Res. 42:879–889. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Poore GD, Kopylova E, Zhu Q, Carpenter C,
Fraraccio S, Wandro S, Kosciolek T, Janssen S, Metcalf J, Song SJ,
et al: Microbiome analyses of blood and tissues suggest cancer
diagnostic approach. Nature. 579:567–574. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Love MI, Huber W and Anders S: Moderated
estimation of fold change and dispersion for RNA-seq data with
DESeq2. Genome Biol. 15:5502014. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Zhang B and Horvath S: A general framework
for weighted gene co-expression network analysis. Stat Appl Genet
Mol Biol. 4:172005. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Langfelder P and Horvath S: WGCNA: An R
package for weighted correlation network analysis. BMC
Bioinformatics. 9:5592008. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Kolberg L, Raudvere U, Kuzmin I, Adler P,
Vilo J and Peterson H: g:Profiler-interoperable web service for
functional enrichment analysis and gene identifier mapping (2023
update). Nucleic Acids Res. 51:W207–W212. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Kuleshov MV, Jones MR, Rouillard AD,
Fernandez NF, Duan Q, Wang Z, Koplev S, Jenkins SL, Jagodnik KM,
Lachmann A, et al: Enrichr: A comprehensive gene set enrichment
analysis web server 2016 update. Nucleic Acids Res. 44:W90–W97.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Kanehisa M and Goto S: KEGG: Kyoto
encyclopedia of genes and genomes. Nucleic Acids Res. 28:27–30.
2000. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Liberzon A, Birger C, Thorvaldsdóttir H,
Ghandi M, Mesirov JP and Tamayo P: The molecular signatures
database hallmark gene set collection. Cell Syst. 1:417–425. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Wickham H: ggplot2: Elegant graphics for
data analysis. second edition. Springer-Verlag; New York: 2016
|
|
35
|
Ewels P, Magnusson M, Lundin S and Käller
M: MultiQC: Summarize analysis results for multiple tools and
samples in a single report. Bioinformatics. 32:3047–3048. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Andrews S: FastQC: A Quality Control Tool
for High Throughput Sequence Data. 2010.
|
|
37
|
Martin M: Cutadapt Removes Adapter
Sequences From High-Throughput Sequencing Reads. 2011. View Article : Google Scholar
|
|
38
|
Dobin A, Davis CA, Schlesinger F, Drenkow
J, Zaleski C, Jha S, Batut P, Chaisson M and Gingeras TR: STAR:
Ultrafast universal RNA-seq aligner. Bioinformatics. 29:15–21.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Lefol Y, Korfage T, Mjelle R, Prebensen C,
Lüders T, Müller B, Krokan H, Sarno A, Alsøe L; CONSORTIUM
LEMONAID, ; et al: TiSA: TimeSeriesAnalysis-A pipeline for the
analysis of longitudinal transcriptomics data. NAR Genom Bioinform.
5:lqad0202023. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Nilsen G, Borgan Ø, LiestØl K and
Lingjærde OC: Identifying clusters in genomics data by recursive
partitioning. Stat Appl Genet Mol Biol. 12:637–652. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(−Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Lamb J, Crawford ED, Peck D, Modell JW,
Blat IC, Wrobel MJ, Lerner J, Brunet JP, Subramanian A, Ross KN, et
al: The connectivity map: Using gene-expression signatures to
connect small molecules, genes, and disease. Science.
313:1929–1935. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Samart K, Tuyishime P, Krishnan A and Ravi
J: Reconciling multiple connectivity scores for drug repurposing.
Brief Bioinform. 22:bbab1612021. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Subramanian A, Narayan R, Corsello SM,
Peck DD, Natoli TE, Lu X, Gould J, Davis JF, Tubelli AA, Asiedu JK,
et al: A next generation connectivity map: L1000 platform and the
first 1,000,000 profiles. Cell. 171:1437–1452.e17. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Ritz C, Baty F, Streibig JC and Gerhard D:
Dose-response analysis using R. PLoS One. 10:e01460212015.
View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Zheng S, Wang W, Aldahdooh J, Malyutina A,
Shadbahr T, Tanoli Z, Pessia A and Tang J: Synergy Finderplus:
Toward better interpretation and annotation of drug combination
screening datasets. Genomics Proteomics Bioinformatics. 20:587–596.
2022. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Xia T, Xu LL, Guo PY, Shi WT, Cheng YQ and
Liu AJ: Synergism of amlodipine and telmisartan or candesartan on
blood pressure reduction by using SynergyFinder 3.0 and probability
sum test in vivo. Pharmacol Res Perspect. 11:e010642023. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Holm S: A simple sequentially rejective
multiple test procedure a simple sequentially rejective multiple
test procedure. Stat Medics. 6:65–70. 1979.
|
|
49
|
Zhang Y, Zhang Y, Li M, Meng F, Yu Z, Chen
Y and Cui G: Combination of SB431542, CHIR99021 and PD0325901 has a
synergic effect on abrogating valproic acid-induced
epithelial-mesenchymal transition and stemness in HeLa, 5637 and
SCC-15 cells. Oncol Rep. 41:3545–3554. 2019.PubMed/NCBI
|
|
50
|
Ye H, Zhang Y, Wang Y, Xia J, Mao X and Yu
X: The restraining effect of baicalein and U0126 on human cervical
cancer cell line HeLa. Mol Med Rep. 16:957–963. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Yang EJ and Chang JH: PD98059 induces the
apoptosis of human cervical cancer cells by regulating the
expression of Bcl2 and ERK2. J Exp Biomed Sci. 17:291–295.
2011.
|
|
52
|
Zahmatyar M, Kharaz L, Abiri Jahromi N,
Jahanian A, Shokri P and Nejadghaderi SA: The safety and efficacy
of binimetinib for lung cancer: A systematic review. BMC Pulm Med.
24:3792024. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Tran B and Cohen MS: The discovery and
development of binimetinib for the treatment of melanoma. Expert
Opin Drug Discov. 15:745–754. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Fu K, Cheung AHK, Wong CC, Liu W, Zhou Y,
Wang F, Huang P, Yuan K, Coker OO, Pan Y, et al: Streptococcus
anginosus promotes gastric inflammation, atrophy, and
tumorigenesis in mice. Cell. 187:882–896. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Kumar R, Herold JL, Schady D, Davis J,
Kopetz S, Martinez-Moczygemba M, Murray BE, Han F, Li Y, Callaway
E, et al: Streptococcus gallolyticus subsp. gallolyticus
promotes colorectal tumor development. PLoS Pathog.
13:e10064402017. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Song X, Liu B, Zhao G, Pu X, Liu B, Ding M
and Xue Y: Streptococcus pneumoniae promotes migration and
invasion of A549 cells in vitro by activating mTORC2/AKT through
up-regulation of DDIT4 expression. Front Microbiol. 13:10462262022.
View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Choi Y, Han HS, Chong GO, Le TM, Nguyen
HDT, Lee OE, Lee D, Seong WJ, Seo I and Cha HH: Updates on Group B
streptococcus infection in the field of obstetrics and gynecology.
Microorganisms. 10:23982022. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Hsieh HY, Lu CH and Wang L: Long-term
treatment outcomes/toxicities of definite chemoradiotherapy
(intensity-modulated radiation therapy) for early-stage ‘bulky’
cervical cancer and survival impact of histological subtype. J
Formos Med Assoc. 122:221–229. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Pan X, Yang W, Wen Z, Li F, Tong L and
Tang W: Does adenocarcinoma have a worse prognosis than squamous
cell carcinoma in patients with cervical cancer? A real-world study
with a propensity score matching analysis. J Gynecol Oncol.
31:e802020. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Guo Y, Pan W, Liu S, Shen Z, Xu Y and Hu
L: ERK/MAPK signalling pathway and tumorigenesis (Review). Exp Ther
Med. 19:1997–2007. 2020.PubMed/NCBI
|
|
61
|
Burotto M, Chiou VL, Lee JM and Kohn EC:
The MAPK pathway across different malignancies: A new perspective.
Cancer. 120:3446–3456. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
62
|
Alto NM and Orth K: Subversion of cell
signaling by pathogens. Cold Spring Harb Perspect Biol.
4:a0061142012. View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Guo Y, Pan W, Liu S, Shen Z, Xu Y and Hu
L: ERK/MAPK signalling pathway and tumorigenesis. Exp Ther Med.
19:1997–2007. 2020.PubMed/NCBI
|
|
64
|
Lavoie H, Gagnon J and Therrien M: ERK
signalling: A master regulator of cell behaviour, life and fate.
Nat Rev Mol Cell Biol. 21:607–632. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
65
|
Raingeaud J, Gupta S, Rogers JS, Dickens
M, Han J, Ulevitch RJ and Davis RJ: Pro-inflammatory cytokines and
environmental stress cause p38 mitogen-activated protein kinase
activation by dual phosphorylation on tyrosine and threonine. J
Biol Chem. 270:7420–7426. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
66
|
Tian T, Li X and Zhang J: mTOR signaling
in cancer and mtor inhibitors in solid tumor targeting therapy. Int
J Mol Sci. 20:7552019. View Article : Google Scholar : PubMed/NCBI
|
|
67
|
Zou Z, Tao T, Li H and Zhu X: MTOR
signaling pathway and mTOR inhibitors in cancer: Progress and
challenges. Cell Biosci. 10:312020. View Article : Google Scholar : PubMed/NCBI
|
|
68
|
Ben-Sahra I and Manning BD: mTORC1
signaling and the metabolic control of cell growth. Curr Opin Cell
Biol. 45:72–82. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
69
|
Pópulo H, Lopes JM and Soares P: The mTOR
signalling pathway in human cancer. Int J Mol Sci. 13:1886–1918.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
70
|
Fisher FM and Maratos-Flier E:
Understanding the physiology of FGF21. Annu Rev Physiol.
78:223–241. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
71
|
Meng D, Yang Q, Wang H, Melick CH, Navlani
R, Frank AR and Jewell JL: Glutamine and asparagine activate mTORC1
independently of Rag GTPases. J Biol Chem. 295:2890–2899. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
72
|
Gobert AP, Latour YL, Asim M, Finley JL,
Verriere TG, Barry DP, Milne GL, Luis PB, Schneider C, Rivera ES,
et al: Bacterial pathogens hijack the innate immune response by
activation of the reverse transsulfuration pathway. mBio.
10:e02174–19. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
73
|
Xing J, Kornhauser JM, Xia Z, Thiele EA
and Greenberg ME: Nerve growth factor activates extracellular
Signal-regulated kinase and p38 Mitogen-activated protein kinase
pathways to stimulate CREB serine 133 Phosphorylation. Mol Cell
Biol. 18:1946–1955. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
74
|
Yang HT, Cohen P and Rousseau S:
IL-1β-stimulated activation of ERK1/2 and p38α MAPK mediates the
transcriptional up-regulation of IL-6, IL-8 and GRO-α in HeLa
cells. Cell Signal. 20:375–380. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
75
|
Li S, Deng P, Wang M, Liu X, Jiang M,
Jiang B, Yang L and Hu J: IL-1α and IL-1β promote NOD2-induced
immune responses by enhancing MAPK signaling. Lab Invest.
99:1321–1334. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
76
|
Liu W, Feng Z, Qu N, Li R and Niu Y: NUPR1
contribution to autophagy in primary bone tumor cells by regulating
the AKT/mTOR signaling pathway. Acta Medica Mediterranea.
38:1223–1228. 2022.
|
|
77
|
Yang C, Xu X, Dong X, Yang B, Dong W, Luo
Y, Liu X, Wu Y and Wang J: DDIT3/CHOP promotes autophagy in
chondrocytes via SIRT1-AKT pathway. Biochim Biophys Acta Mol Cell
Res. 1868:1190742021. View Article : Google Scholar : PubMed/NCBI
|
|
78
|
Fan T, Wang X, Zhang S, Deng P, Jiang Y,
Liang Y, Jie S, Wang Q, Li C, Tian G, et al: NUPR1 promotes the
proliferation and metastasis of oral squamous cell carcinoma cells
by activating TFE3-dependent autophagy. Signal Transduct Target
Ther. 7:1302022. View Article : Google Scholar : PubMed/NCBI
|
|
79
|
Wang Y, Huang J, Chen W, Wang RH, Kao MC,
Pan YR, Chan SH, Tsai KW, Kung HJ, Lin KT and Wang LH:
Dysregulation of cystathionine γ-lyase promotes prostate cancer
progression and metastasis. EMBO Rep. 20:e459862019. View Article : Google Scholar : PubMed/NCBI
|
|
80
|
Lin H, Liu S, Gao W and Liu H: DDIT3
modulates cancer stemness in gastric cancer by directly regulating
CEBPβ. J Pharm Pharmacol. 72:807–815. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
81
|
Krall AS, Xu S, Graeber TG, Braas D and
Christofk HR: Asparagine promotes cancer cell proliferation through
use as an amino acid exchange factor. Nat Commun. 7:114572016.
View Article : Google Scholar : PubMed/NCBI
|
|
82
|
Gelfo V, Romaniello D, Mazzeschi M, Sgarzi
M, Grilli G, Morselli A, Manzan B, Rihawi K and Lauriola M: Roles
of il-1 in cancer: From tumor progression to resistance to targeted
therapies. Int J Mol Sci. 21:60092020. View Article : Google Scholar : PubMed/NCBI
|
|
83
|
Molloy NH, Read DE and Gorman AM: Nerve
growth factor in cancer cell death and survival. Cancers (Basel).
3:510–530. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
84
|
Sui Y, Liu Q, Xu C, Ganesan K, Ye Z, Li Y,
Wu J, Du B, Gao F, Song C and Chen J: Non-alcoholic fatty liver
disease promotes breast cancer progression through upregulated
hepatic fibroblast growth factor 21. Cell Death Dis. 15:672024.
View Article : Google Scholar : PubMed/NCBI
|
|
85
|
Tyrrell GJ, Kennedy A, Shokoples SE and
Sherburne RK: Binding and invasion of HeLa and MRC-5 cells by
Streptococcus agalactiae. Microbiology (Reading).
148:3921–3931. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
86
|
Gerdes J, Lemke H, Baisch H, Wacker H,
Schwab U and Stein H: Cell cycle analysis of a cell
proliferation-associated human nuclear antigen defined by the
monoclonal antibody Ki-67. J Immunol. 133:1710–1715. 1984.
View Article : Google Scholar : PubMed/NCBI
|
|
87
|
Schlfiter C, Duchrow M, Wohlenberg C,
Becker MHG, Key G, Flad HD and Gerdes J: The cell
Proliferation-associated antigen of antibody Ki-67: A very large,
ubiquitous nuclear protein with numerous repeated elements,
representing a new kind of cell cycle-maintaining proteins. J Cell
Biol. 123:513–522. 1993. View Article : Google Scholar
|
|
88
|
Uxa S, Castillo-Binder P, Kohler R,
Stangner K, Müller GA and Engeland K: Ki-67 gene expression. Cell
Death Differ. 28:3357–3370. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
89
|
Burnham CAD, Shokoples SE and Tyrrell GJ:
Invasion of HeLa cells by group B streptococcus requires the
phosphoinositide-3-kinase signalling pathway and modulates
phosphorylation of host-cell Akt and glycogen synthase kinase-3.
Microbiology (Reading). 153:4240–4252. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
90
|
Ji J and Zheng PS: Activation of mTOR
signaling pathway contributes to survival of cervical cancer cells.
Gynecol Oncol. 117:103–108. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
91
|
Li XW, Tuergan M and Abulizi G: Expression
of MAPK1 in cervical cancer and effect of MAPK1 gene silencing on
epithelial-mesenchymal transition, invasion and metastasis. Asian
Pac J Trop Med. 8:937–943. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
92
|
Weth FR, Hoggarth GB, Weth AF, Paterson E,
White MPJ, Tan ST, Peng L and Gray C: Unlocking hidden potential:
Advancements, approaches, and obstacles in repurposing drugs for
cancer therapy. Br J Cancer. 130:703–715. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
93
|
K W To K and Cho WCS: Drug repurposing for
cancer therapy in the era of precision medicine. Curr Mol
Pharmacol. 15:895–903. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
94
|
Colombo N, McMeekin DS, Schwartz PE, Sessa
C, Gehrig PA, Holloway R, Braly P, Matei D, Morosky A, Dodion PF,
et al: Ridaforolimus as a single agent in advanced endometrial
cancer: Results of a single-arm, phase 2 trial. Br J Cancer.
108:1021–1026. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
95
|
Chon HS, Kang S, Lee JK, Apte SM, Shahzad
MM, Williams-Elson I and Wenham RM: Phase I study of oral
ridaforolimus in combination with paclitaxel and carboplatin in
patients with solid tumor cancers. BMC Cancer. 17:4072017.
View Article : Google Scholar : PubMed/NCBI
|
|
96
|
Finn RS, Ahn DH, Javle MM, Tan BR Jr,
Weekes CD, Bendell JC, Patnaik A, Khan GN, Laheru D, Chavira R, et
al: Phase 1b investigation of the MEK inhibitor binimetinib in
patients with advanced or metastatic biliary tract cancer. Invest
New Drugs. 36:1037–1043. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
97
|
Woodfield SE, Zhang L, Scorsone KA, Liu Y
and Zage PE: Binimetinib inhibits MEK and is effective against
neuroblastoma tumor cells with low NF1 expression. BMC Cancer.
16L:1722016. View Article : Google Scholar : PubMed/NCBI
|
|
98
|
Anttila JV, Shubin M, Cairns J, Borse F,
Guo Q, Mononen T, Vázquez-García I, Pulkkinen O and Mustonen V:
Contrasting the impact of cytotoxic and cytostatic drug therapies
on tumour progression. PLoS Comput Biol. 15:e10074932019.
View Article : Google Scholar : PubMed/NCBI
|
|
99
|
Hua H, Kong Q, Zhang H, Wang J, Luo T and
Jiang Y: Targeting mTOR for cancer therapy. J Hematol Oncol.
12:712019. View Article : Google Scholar : PubMed/NCBI
|
|
100
|
Formisano L, Napolitano F, Rosa R, D'Amato
V, Servetto A, Marciano R, De Placido P, Bianco C and Bianco R:
Mechanisms of resistance to mTOR inhibitors. Crit Rev Oncol
Hematol. 147:1028862020. View Article : Google Scholar : PubMed/NCBI
|
|
101
|
Carracedo A, Ma L, Teruya-Feldstein J,
Rojo F, Salmena L, Alimonti A, Egia A, Sasaki AT, Thomas G, Kozma
SC, et al: Inhibition of mTORC1 leads to MAPK pathway activation
through a PI3K-dependent feedback loop in human cancer. J Clin
Invest. 118:3065–3074. 2008.PubMed/NCBI
|
|
102
|
Li Q, Li Z, Luo T and Shi H: Targeting the
PI3K/AKT/mTOR and RAF/MEK/ERK pathways for cancer therapy. Mol
Biomed. 3:472022. View Article : Google Scholar : PubMed/NCBI
|
|
103
|
Pitts TM, Newton TP, Bradshaw-Pierce EL,
Addison R, Arcaroli JJ, Klauck PJ, Bagby SM, Hyatt SL, Purkey A,
Tentler JJ, et al: Dual pharmacological targeting of the map kinase
and pi3k/mtor pathway in preclinical models of colorectal cancer.
PLoS One. 9:e1130372014. View Article : Google Scholar : PubMed/NCBI
|