Introduction
In recent years, more research evidence on
immunotherapy has been emerging in the field of oncology. Immune
checkpoint inhibitors (ICIs) have demonstrated notable efficacy in
both perioperative and advanced cancer treatment (1,2).
Nevertheless, the problems of hyper-progression and serious adverse
reactions after receiving immunotherapy cannot be ignored (3,4). At
present, the expression level of programmed death ligand-1 (PD-L1)
is the main predictive biomarker for immunotherapy. However, a
study has found that a small proportion of patients with negative
PD-L1 expression remain sensitive to immunotherapy (5). In addition, there is still a lack of
unified standards for the detection method of PD-L1. The prognostic
value of tumor mutational burden (TMB) as a biomarker for
immunotherapy has been validated in clinical studies and using
real-world data (6,7). However, due to the lack of a unified
detection standard, the inability to fully reflect the tumor immune
microenvironment, the differences between different tumor types and
the limitations of dynamic changes, the application of TMB is
limited (6). Therefore, PD-L1
expression and TMB cannot predict the prognosis and efficacy of
immunotherapy fully and effectively. It is therefore imperative to
explore new predictive markers for identifying patients with a
potentially poor prognosis at an earlier stage and to provide
supportive treatment to improve treatment outcomes to further
expand the population benefiting from immunotherapy.
The value of inflammatory markers and nutritional
status in evaluating tumor prognosis has gradually become a
research hotspot. Specifically, the prognostic scoring systems
based on inflammatory markers, such as the Glasgow prognostic score
(GPS), neutrophil-to-lymphocyte ratio (NLR), platelet-to-lymphocyte
ratio (PLR), immunoinflammatory index (SII), lymphocyte-to-monocyte
ratio (LMR) and prognostic nutritional index (PNI), have been shown
to have predictive value in the prognosis of antitumor therapy
(3,8–10). The
GPS, originally proposed by Forrest et al (11), is a scoring system based on a
combination of C-reactive protein (CRP) and albumin levels. Albumin
is a biomarker of nutritional status (12), whereas CRP is a sensitive indicator
of the inflammatory response and has also been shown to promote the
formation of an immunosuppressive tumor microenvironment and the
growth of tumor cells (13). CRP
can be used as an independent prognostic factor for a variety of
malignant tumors such as gastric and pancreatic cancer (14,15).
Recently, CRP has also been found to predict the efficacy of
programmed cell death protein 1 (PD-1) treatment (16). Hypoalbuminemia is a manifestation of
cachexia in patients with advanced tumors and severe inflammatory
damage can also lead to hypoalbuminemia. Decreased albumin can
weaken the ability of cells to activate immunity and antioxidant
(17,18). Thus, the GPS combines the two
indicators, CRP and albumin, to more effectively evaluate the
prognosis of patients with tumors.
The GPS criteria are as follows: CRP elevation (CRP
>10 mg/l) combined with hypoalbuminemia (albumin <35 g/l) is
assigned a score of 2, only one abnormality is assigned a score of
1 and a score of 0 is assigned when both indicators are normal
(19). In 2007, McMillan et
al (20) refined the scoring
system to create the modified GPS (mGPS). The improved scoring
system emphasizes that individuals with elevated CRP and normal
albumin are scored as 1 point. Studies have shown that the mGPS
system is more effective in reflecting systemic inflammatory
responses than isolated inflammatory indicators (21,22).
GPS and mGPS have gradually been proven to have value in evaluating
the prognosis of patients receiving immunotherapy (23,24).
However, with the continuous emergence of new research, the
predictive value of GPS and mGPS for immunotherapy deserves further
validation.
In the present study, the literature on the
association between GPS or mGPS and the prognosis of patients with
advanced cancer receiving immunotherapy was reviewed, and a
meta-analysis was conducted to demonstrate the role of GPS or mGPS
in predicting overall survival (OS) or progression-free survival
(PFS).
Materials and methods
Preferred reporting items for
systematic reviews and meta-analyses (PRISMA)
The present study was performed in accordance with
the PRISMA guidelines (25). The
protocol for the systematic review and meta-analysis was registered
in PROSPERO (https://www.crd.york.ac.uk/PROSPERO/view/CRD42023396079,
no. CRD42023396079).
Literature search
The PubMed (https://pubmed.ncbi.nlm.nih.gov/), Embase (https://www.embase.com/), Cochrane library (https://www.cochranelibrary.com/) and Web of
Science (www.webofscience.com) databases were
systematically searched by three independent researchers, searching
literature from the inception of the databases to November 2022.
The following key words were used: (‘Glasgow prognostic score’ OR
‘GPS’ OR ‘modified Glasgow prognostic score’ OR ‘mGPS’) AND
(‘immune checkpoint inhibitors’ OR ‘PD-L1 inhibitor’ OR ‘PD-1
inhibitor’) AND (‘neoplasms’ OR ‘carcinoma’). Full database search
strategies can be viewed in Supplementary materials 1. Other sources
were also included, such as relevant articles found in the review
article (26).
Study selection
The inclusion criteria were as follows: i) Studies
on patients with advanced cancer that cannot be cured by local
treatment receiving ICIs; ii) studies that calculated the GPS or
mGPS before ICI treatment; iii) studies reporting clinical outcomes
such as OS and/or PFS; and iv) studies with hazard ratio (HR) with
95% confidence interval (CI) data available. Reviews, case reports,
letters, studies not in English or with insufficient data were
excluded.
Data extraction
The following data were extracted from each
individual study: Country, cancer type, study design type, ICIs,
sample size, line of treatment, patient sex, patient age, analysis
model, GPS or mGPS, follow-up time, endpoint and HRs with 95% CIs
for OS and PFS. If both univariate and multivariate results were
available, priority was given to the multivariate results.
Quality assessment
The Newcastle-Ottawa Scale (NOS) was applied to
assess the quality of studies (27). The total score of NOS ranges from 0
to 9 based on its assessment items. In total, two reviewers
assessed each study independently and reached a consensus after
discussion.
Statistical analysis
Patients were divided into low GPS/mGPS (0) and high
GPS/mGPS (1/2/1-2) groups, with a cut-off value of 0. Studies
containing two HRs (such 0 vs. 1 and 0 vs. 2) were included in the
meta-analysis as two independent findings. The outcomes were
reported as pooled HRs with 95% CIs for OS and PFS. The pooled
results were examined by random-effects models. P<0.05 or
I2>50% was considered to indicate high heterogeneity.
Subgroup analysis was used to analyze the sources of heterogeneity.
A sensitivity analysis was performed using the leave-one-out
sensitivity method to evaluate the robustness of the combined
results and to identify the studies that contributed significantly
to heterogeneity. A graphical funnel plot and Egger's test was used
to evaluate publication bias. Statistical analyses were conducted
using Stata 14.0 (StataCorp LP).
Results
Study identification and
selection
The literature retrieval and screening process is
shown in Fig. 1. After initial
searches in the PubMed, Web of Science, Medline, Embase and
Cochrane Library databases, 71 relevant publications were screened,
and 6 additional records were identified through other sources.
Subsequently, after omitting duplicate records and eliminating by
title and abstract, detailed screening was conducted on the
remaining 43 records. In total, 15 studies that did not meet the
inclusion criteria and 10 studies with no outcome of interest were
excluded. Finally, 18 studies (28–45)
involving 1,355 patients were included in the present
meta-analysis.
Characteristics and quality
assessment
All included studies were retrospective and
published between 2020 and 2022. In 9 studies that reported GPS, 8
were from Asian countries (5 from Japan, 2 from Korea and 1 from
China), 8 reported the HR of OS and 7 reported the HR of PFS. In 9
studies that reported mGPS, 4 were from Asian countries (all from
Japan), while 5 were from European or American countries, 8
reported the HR of OS and 8 reported the HR of PFS. The NOS scores
of all included studies were ≥5. The study characteristics are
presented in Table SI.
Prognostic value of GPS on survival
outcomes
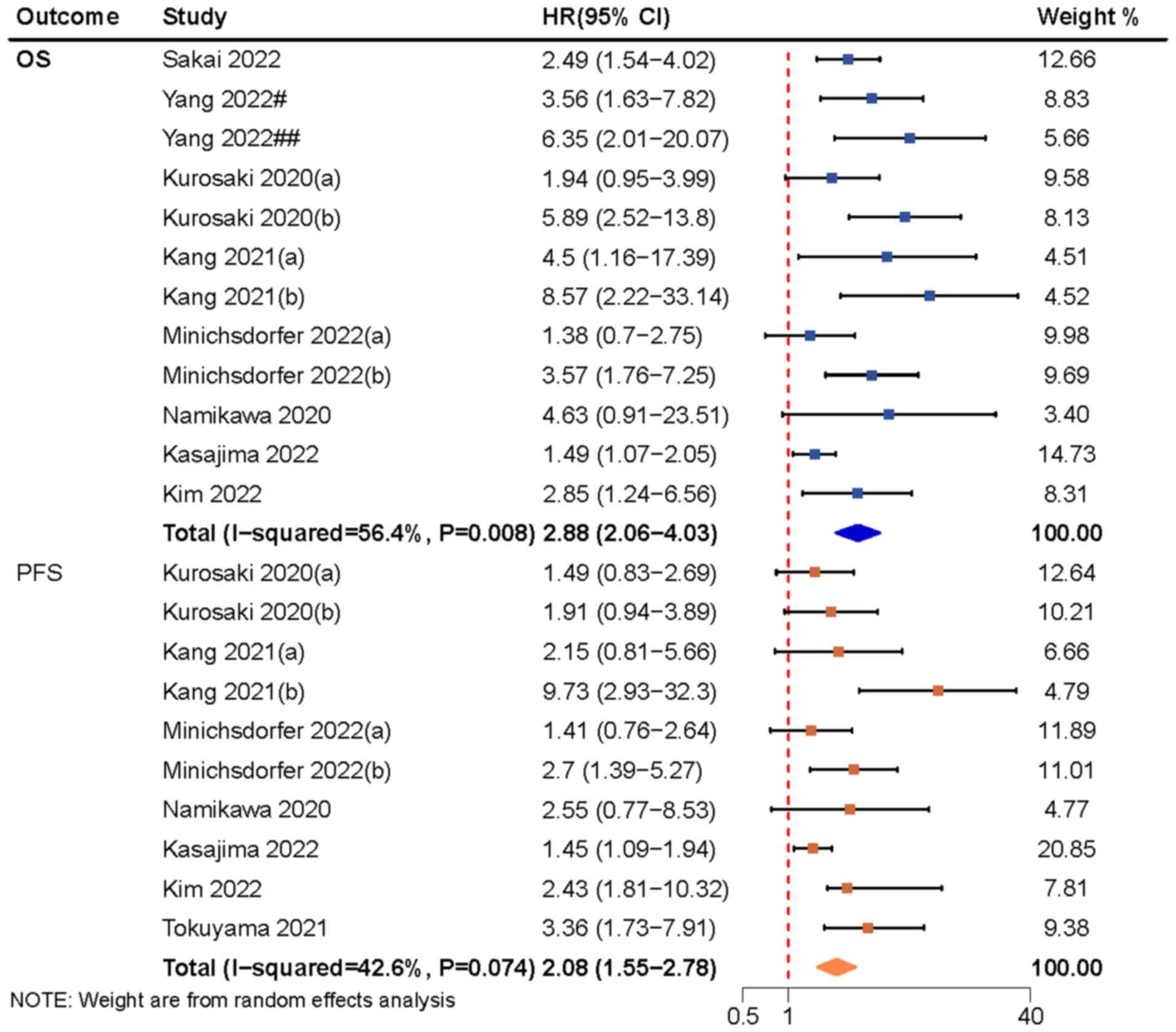
The pooled results of 8 studies involving 12 sets of
data (28–35) revealed that a high GPS in patients
receiving ICIs resulted in a poorer OS than a low GPS (HR, 2.88;
95% CI, 2.06–4.03), with high heterogeneity (Fig. 2). Sensitivity analysis showed that
heterogeneity was significantly reduced (I2 reduced from
56.4 to 30.3%) after the study by Kasajima et al (34) was excluded, indicating that this
study markedly contributed to heterogeneity (Fig. S1A). Subgroup analyses were
conducted by the GPS value. GPS 1 [from 4 studies (29–32);
HR, 2.18; 95% CI, 1.46–3.25], GPS 2 [from 4 studies (29–32);
HR, 5.03; 95% CI, 3.17–7.99] and GPS 1–2 [from 4 studies (28,33–35);
HR, 1.88; 95% CI, 1.46–2.42] were all found to be associated with a
worse OS compared with GPS 0, with low heterogeneity. No subgroup
analysis by country was performed because seven studies were from
Asian populations and only one study was from Australia. All
subgroup analyses are shown in Table
I.
 | Table I.GPS subgroup analysis for OS and mGPS
subgroup analysis for PFS. |
Table I.
GPS subgroup analysis for OS and mGPS
subgroup analysis for PFS.
| A, GPS for OS |
|---|
|
|---|
| Subgroup | Number of
studies | HR (95% CI) | P-value | Heterogeneity,
P-value (I2), % |
|---|
| Score |
|
|
|
|
| 1 vs.
0 | 4 | 2.18
(1.46–3.25) | <0.001 | 0.219 (32.1) |
| 2 vs.
0 | 4 | 5.03
(3.17–7.99) | <0.001 | 0.619 (0.0) |
| 1–2 vs.
0 | 4 | 1.88
(1.46–2.42) | <0.001 | 0.143 (44.7) |
|
| B, mGPS for
PFS |
|
|
Subgroup | Number of
studies | HR (95%
CI) | P-value | Heterogeneity,
P-value (I2), % |
|
| Region |
|
|
|
|
|
Asian | 3 | 2.60
(1.62–4.16) | <0.001 | 0.899 (0.0) |
|
Non-Asian | 5 | 2.61
(1.65–4.11) | <0.001 | 0.001 (67.9) |
| Score |
|
|
|
|
| 1 vs.
0 | 6 | 1.55
(1.17–2.05) | 0.002 | 0.391 (4.0) |
| 2 vs.
0 | 6 | 3.74
(2.02–6.91) | <0.001 | 0.011 (66.5) |
| 1–2 vs.
0 | 2 | 2.92
(1.33–6.41) | 0.007 | 0.793 (0.0) |
The pooled results of 7 studies involving 10 sets of
data (30–36) showed that a high GPS resulted in a
poorer PFS in patients receiving ICIs than a low GPS (HR, 2.08; 95%
CI, 1.55–2.78), with low heterogeneity (Fig. 2). The combined effect size was
consistent after eliminating any of the studies, indicating that
the results had a good robustness (Fig. S1B).
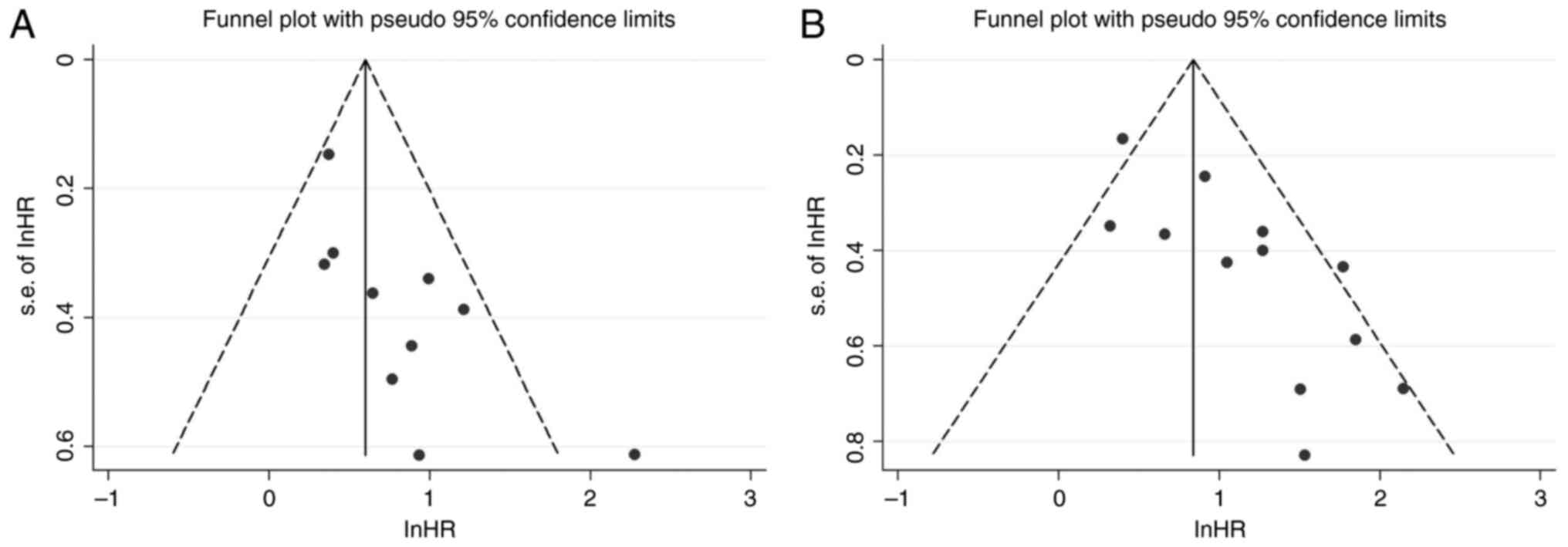
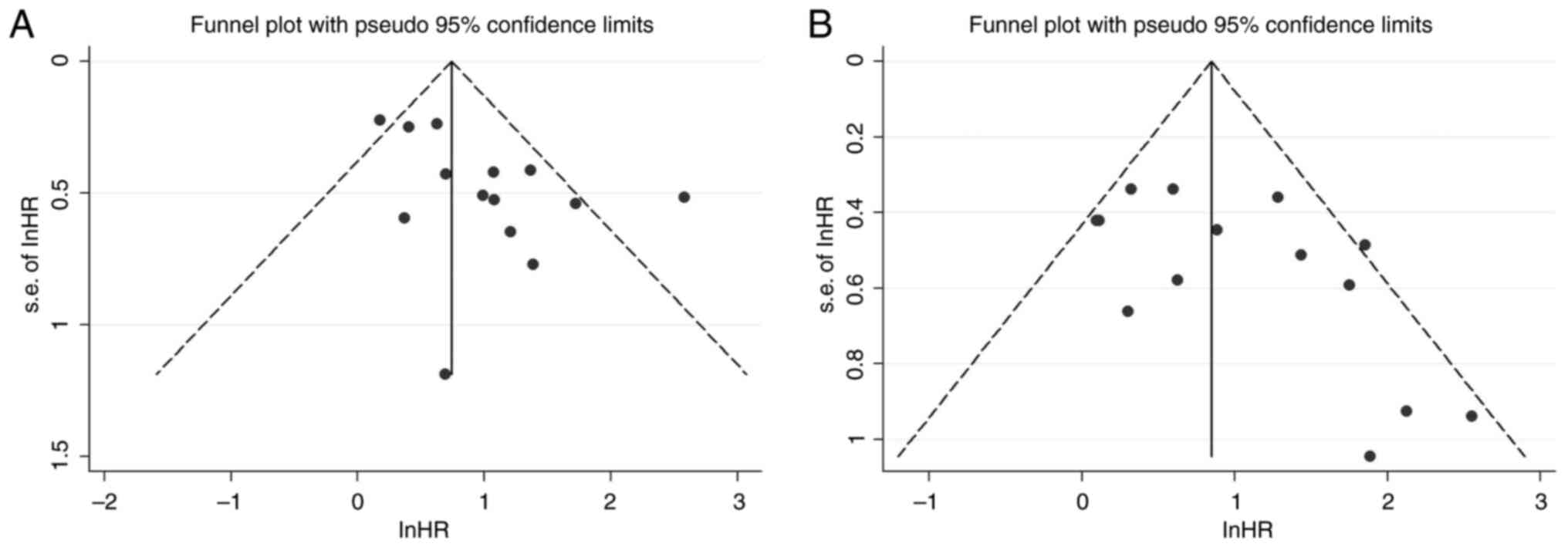
The funnel plots for GPS in OS and PFS are shown in
Fig. 3A and B. The Egger's tests
(OS, P=0.896; PFS, P=0.989) indicated that no notable publication
bias existed.
Prognostic value of mGPS on survival
outcomes
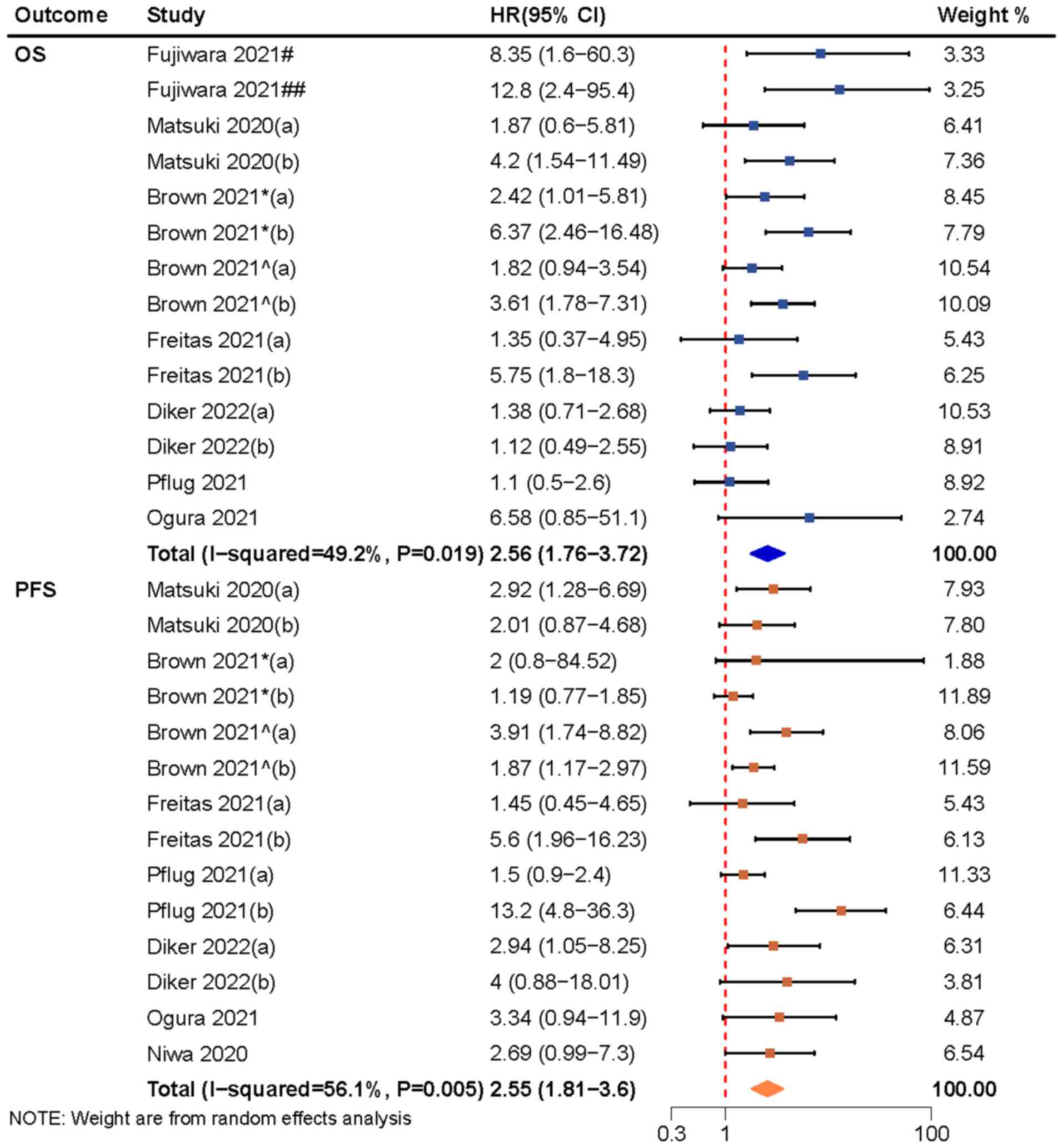
The pooled results of 8 studies involving 14 sets of
data (37–44) showed that a high mGPS resulted in a
shorter OS in patients receiving ICIs than a low mGPS (HR, 2.56;
95% CI, 1.76–3.72), with high heterogeneity (Fig. 4). The combined effect size remained
consistent after excluding any one study, which indicated that the
results were stable (Fig.
S2A).
The pooled results of 8 studies involving 14 sets of
data (38–45) showed that a high mGPS resulted in a
shorter PFS in patients receiving ICIs than a low mGPS (HR, 2.55;
95% CI, 1.81–3.60), with high heterogeneity (Fig. 4). Sensitivity analysis and subgroup
analysis by country and mGPS value were performed to analyze the
sources of heterogeneity. The combined effect size was consistent
but the heterogeneity was not eliminated after sensitivity analysis
(Fig. S2B). Subgroup analysis by
country showed that a high mGPS in Asians from 3 studies (38,44,45)
and non-Asians from 5 studies (39–43)
was associated with a shorter PFS time than a low mGPS (HR, 2.60;
95% CI, 1.62–4.16; and HR, 2.61; 95% CI, 1.65–4.11), and the
non-Asian subgroup still had high heterogeneity
(I2=67.9%). Subgroup analysis by score indicated that an
mGPS of 1 (n=6 studies) (38–43),
an mGPS of 2 (n=6 studies) (38–43)
and an mGPS of 1–2 (n=2 studies) (44,45)
was associated with a poorer PFS than mGPS 0 (HR, 1.55; 95% CI,
1.17–2.05; HR, 3.74; 95% CI, 2.02–6.91; and HR, 2.92; 95% CI,
1.33–6.41, respectively), and the mGPS 2 subgroup still had high
heterogeneity (I2=66.5%). All subgroup analysis results
are shown in Table I.
The funnel plots for mGPS in OS and PFS were
symmetrical, indicating no publication bias (OS, P=0.670; PFS,
P=0.941; Fig. 5A and B).
Discussion
GPS and mGPS have been shown to have prognostic
value in multiple cancer types and different treatment stages
(46,47). Although other inflammatory
indicators, such as NLR, PLR, PNI and SII, have some predictive
value, they lack a comprehensive assessment of nutritional status
and are less consistent across cancer types. Specifically,
Yamanouchi et al (48)
evaluated the effects of inflammatory and nutritional indicators
such as GPS, NLR and PNI on the prognosis of patients with
metastatic breast cancer, and only GPS was indicated to be an
independent predictor of OS. GPS was also shown to be an
independent indicator of prognosis in patients with hepatocellular
carcinoma, and was superior to other inflammatory prognostic scores
in terms of prognostic power (49).
Shimoyama et al (50)
evaluated the prognostic value of 17 inflammatory markers in
patients with advanced or recurrent gastric cancer who received
opdivo and found that mGPS had the strongest association with
patient outcome. GPS or mGPS also have the characteristics of being
cheap, convenient and easy to obtain, and have great application
potential (46).
The present meta-analysis, which included data from
18 studies involving 1,355 patients with cancer, assessed the
association between GPS or mGPS and survival in patients receiving
ICIs. The results indicated that a higher GPS or mGPS contributed
to poorer survival in patients with cancer, and subgroup analyses
also demonstrated that patients with GPS or mGPS 1 and GPS or mGPS
2 exhibited poorer OS and PFS than those with GPS 0. However, due
to the heterogeneity of some of the data, caution is required in
interpreting these results.
GPS, based on CRP and albumin levels, has been
proven to have independent prognostic value in various cancer types
in previous years (46,51). CRP and albumin are sensitive and
reliable indicators that reflect the inflammatory state and immune
nutritional status of a cancer population, and have been widely
confirmed as biomarkers of poor prognosis in cancer (46). CRP is an acute phase response
protein that can serve as a biomarker for infection and tissue
damage (52). CRP can promote an
inflammatory response (53),
thereby inhibiting immune function and promoting tumor
proliferation and metastasis (54).
At the same time, CRP can also suppress the immune response,
accelerate tumor migration and tumor microenvironment formation
(54), which may weaken the role of
ICIs. Previous studies have found that CRP can directly inhibit T
cells and dendritic cells (55,56),
thereby affecting the action of ICIs by impacting innate and
acquired immunity in patients with cancer. The CRP level before
treatment can not only predict the therapeutic effect of ICIs
(57), but can also predict the
survival of patients with advanced urothelial cancer or non-small
cell lung cancer receiving immunotherapy (58,59).
The serum albumin level is a notable indicator of nutritional
status, and low albumin levels are a manifestation of malnutrition
and poor general status (60).
Hypoproteinemia reflects poor nutritional status and chronic
inflammation, and can serve as a biomarker for cancer prognosis
(61). Studies have identified
serum albumin as a potential biomarker for evaluating the efficacy
of ICIs, whether alone or in combination with chemotherapy
(61,62). The results of the present study
showed that a high GPS was associated with unfavorable OS and PFS,
suggesting poor survival outcomes for patients treated with ICIs,
regardless of hypoproteinemia or elevated CRP. However, the OS
results were heterogeneous. Sensitivity analysis showed that the
study by Kasajima et al (34) contributed significantly to
heterogeneity. The study was divided into GPS 1–2 and GPS 0 groups,
with a tumor type of non-small cell lung cancer, treatment regimen
of pembrolizumab or altezomumab monotherapy and a treatment line of
second-line or subsequent-line therapy (34). There were no significant differences
in clinical features between the study by Kasajima et al
(34) and other included studies.
The heterogeneity in the OS results was reduced after subgroup
analysis based on the scores. This may be due to the subgroups
being divided reasonably, which makes the studies within each
subgroup more homogeneous in certain key features. The grouping of
different GPSs is likely to be the main cause of heterogeneity.
Extensive evidence suggests that weight loss and
poor physical fitness are related to systemic inflammatory
responses in advanced cancer (3,63,64).
Significant inflammatory response can lead to hypoalbuminemia
(65), and cancer-related
inflammation can impair albumin synthesis by altering cytokine
production and thereby increasing microvascular permeability
(66). Therefore, hypoalbuminemia
is more likely to be secondary to elevated CRP levels. There is
growing evidence that inflammation has a role in cancer
development, and that inflammation may also be accelerated by the
cancer itself due to increased catabolism and malnutrition
(67,68). McMillan et al (20) revised the GPS to mGPS to more
accurately predict the prognosis of various cancer types. mGPS
weakens the effect of albumin, emphasizing the importance of CRP.
GPS 0 is determined by the CRP and albumin levels, but mGPS 0 is
determined by CRP alone, regardless of albumin levels (20). For mGPS 1, elevated CRP is rated as
1 even if albumin levels are normal (20), but GPS 1 includes patients with
hypoalbuminemia who do not have elevated CRP levels. The rating
criteria for GPS 2 and mGPS 2 are the same and both combine high
CRP and hypoproteinemia, which may indicate strong systemic
inflammation and poor nutritional status. The results of the
present study demonstrated that mGPS, like GPS, can serve as a
predictor of OS and PFS in cancer populations receiving
immunotherapy. However, there was high heterogeneity in the PFS
data. Sensitivity analysis did not identify any study that had a
notable impact on heterogeneity. After subgroup analysis by country
and score, significant heterogeneity still existed, suggesting that
the present analysis failed to fully explain the source of
heterogeneity and further exploration and verification are
needed.
The evaluation of GPS or mGPS only requires routine
blood tests, which is low cost, highly standardized and suitable
for clinical promotion. The evaluation can be adapted to different
cancer types or population characteristics, while other static
indicators lack such optimization space. For example, mGPS further
improves the predictive sensitivity and specificity of GPS by
adjusting the critical value of CRP and albumin. The cut-off values
may vary across different diseases and even within different
cohorts of the same disease. Therefore, cut-off values can be
determined clinically according to actual disease conditions, such
as tumor type and ethnicity. Compared with PD-L1 and TMB, which
focus on the local characteristics of tumors, the core advantages
of GPS and mGPS lie in their comprehensiveness, universality and
economy, and are more suitable for widespread clinical promotion
and the construction of a comprehensive prognostic evaluation
system. GPS or mGPS can be used in combination with
immunotherapy-related markers to provide multidimensional
information. For example, a high inflammatory state may impair
efficacy in patients with high PD-L1 expression, and the
combination of GPS or mGPS and PD-L1 detection may optimize patient
stratification. Systemic inflammation may indirectly regulate
immunotherapy response by affecting intestinal flora, and GPS or
mGPS can be used as a proxy indicator of systemic inflammation in
comprehensive analysis.
The present study has some limitations. First, the
included studies were retrospective and showed notable
methodological diversity. Second, although subgroup and sensitivity
analyses were performed, heterogeneity was not completely
eliminated. Heterogeneity may be caused by multiple factors, such
as tumor type, ICI regimens, treatment lines, the diversity of the
study population and an inconsistent follow-up time. The small
number of studies within subgroups also resulted in insufficient
statistical power to accurately assess differences between
subgroups. Third, the absence of patient-level data may limit the
assessment of certain baseline characteristics and affect the
interpretation of results. Therefore, there is still a need to
design clinical studies to further validate the relevance of GPS
and mGPS in predicting the survival of each patient with cancer. In
particular, further research is needed on specific factors, such as
differences in the effectiveness of interventions in different
populations.
In conclusion, high GPS or mGPS, that is, either
high CRP or low albumin, may have adverse effects on the OS and PFS
of patients with cancer receiving ICIs. In clinical practice,
patients can be risk stratified according to GPS or mGPS prior to
immunotherapy, thereby enhancing patient management and improving
treatment outcomes. Future prospective cohort studies are needed to
validate the results of the present study, and further studies in
combination with other prognostic markers are needed to improve the
prediction accuracy.
Supplementary Material
Supporting Data
Supporting Data
Acknowledgements
Not applicable.
Funding
This study was supported by Jiangsu Pharmaceutical Society-Chia
Tai Tianqing Hospital Pharmaceutical Research Fund (grant no.
Q202211) and Research Project of Jiangsu Cancer Hospital (grant no.
ZJ202222).
Availability of data and materials
The data generated in the present study may be
requested from the corresponding author.
Authors' contributions
LZ, LH and TL conceived and designed this study. LZ,
TL and LH conducted the literature screening. TL and LL performed
the statistical analysis. LZ and LH wrote the manuscript. LH and JN
were responsible for the interpretation of data and revised the
manuscript. LZ, TL and LH checked and confirmed the authenticity of
all the raw data. All authors have read and approved the final
version of the manuscript.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
André T, Tougeron D, Piessen G, de la
Fouchardière C, Louvet C, Adenis A, Jary M, Tournigand C, Aparicio
T, Desrame J, et al: Neoadjuvant nivolumab plus ipilimumab and
adjuvant nivolumab in localized deficient mismatch
repair/microsatellite instability-high gastric or esophagogastric
junction adenocarcinoma: The GERCOR NEONIPIGA phase II study. J
Clin Oncol. 41:255–265. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
de Castro G Jr, Kudaba I, Wu YL, Lopes G,
Kowalski DM, Turna HZ, Caglevic C, Zhang L, Karaszewska B,
Laktionov KK, et al: Five-year outcomes with pembrolizumab versus
chemotherapy as first-line therapy in patients with non-small-cell
lung cancer and programmed death ligand-1 tumor proportion score
≥1% in the KEYNOTE-042 study. J Clin Oncol. 41:1986–1991. 2023.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Park JW, Chang HJ, Yeo HY, Han N, Kim BC,
Kong SY, Kim J and Oh JH: The relationships between systemic
cytokine profiles and inflammatory markers in colorectal cancer and
the prognostic significance of these parameters. Br J Cancer.
123:610–618. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Sullivan RJ and Weber JS: Immune-related
toxicities of checkpoint inhibitors: Mechanisms and mitigation
strategies. Nat Rev Drug Discov. 21:495–508. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Davis AA and Patel VG: The role of PD-L1
expression as a predictive biomarker: An analysis of all US Food
and Drug Administration (FDA) approvals of immune checkpoint
inhibitors. J Immunother Cancer. 7:2782019. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Wan L, Wang Z, Xue J, Yang H and Zhu Y:
Tumor mutation burden predicts response and survival to immune
checkpoint inhibitors: A meta-analysis. Transl Cancer Res.
9:5437–5449. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Valero C, Lee M, Hoen D, Wang J, Nadeem Z,
Patel N, Postow MA, Shoushtari AN, Plitas G, Balachandran VP, et
al: The association between tumor mutational burden and prognosis
is dependent on treatment context. Nat Genet. 53:11–15. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Zhang CL, Gao MQ, Jiang XC, Pan X, Zhang
XY, Li Y, Shen Q, Chen Y and Pang B: Research progress and value of
albumin-related inflammatory markers in the prognosis of non-small
cell lung cancer: A review of clinical evidence. Ann Med.
55:1294–1307. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Tada H, Kawabata-Iwakawa R, Takahashi H
and Chikamatsu K: Novel index based on inflammatory markers
correlates with treatment efficacy of nivolumab for
recurrent/metastatic head and neck cancer. Oncology. Nov
20–2024.(Epub ahead of print). View Article : Google Scholar
|
|
10
|
Shayimu P, Awula M, Wang CY, Jiapaer R,
Pan YP, Wu ZM, Chen Y and Zhao ZL: Serum nutritional predictive
biomarkers and risk assessment for anastomotic leakage after
laparoscopic surgery in rectal cancer patients. World J
Gastrointest Surg. 16:3142–3154. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Forrest LM, McMillan DC, McArdle CS,
Angerson WJ and Dunlop DJ: Evaluation of cumulative prognostic
scores based on the systemic inflammatory response in patients with
inoperable non-small-cell lung cancer. Br J Cancer. 89:1028–1030.
2003. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Cederholm T, Bosaeus I, Barazzoni R, Bauer
J, Van Gossum A, Klek S, Muscaritoli M, Nyulasi I, Ockenga J,
Schneider SM, et al: Diagnostic criteria for malnutrition-an ESPEN
consensus statement. Clin Nutr. 34:335–340. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Wang Y, Li Z, Huang Z, Yu X, Zheng L and
Xu J: C-reactive protein is an indicator of the immunosuppressive
microenvironment fostered by myeloid cells in hepatocellular
carcinoma. Front Oncol. 11:7748232021. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Chen Z, Sun Y, Wang J, Shen X, Chen L and
Zhuang Z: Prognostic and predictive value of serum C-reactive
protein levels in patients with metastatic or locally recurrent
gastric cancer. Int J Biol Markers. 31:e294–r299. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Nießen A, Schimmack S, Sandini M, Fliegner
D, Hinz U, Lewosinska M, Hackert T, Büchler MW and Strobel O:
C-reactive protein independently predicts survival in pancreatic
neuroendocrine neoplasms. Sci Rep. 11:237682021. View Article : Google Scholar
|
|
16
|
Zhang Y, Lu L, He Z, Xu Z, Xiang Z, Nie
RC, Lin W, Chen W, Zhou J, Yin Y, et al: C-reactive protein levels
predict responses to PD-1 inhibitors in hepatocellular carcinoma
patients. Front Immunol. 13:8081012022. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Demir O, Demirag G and Aslan G:
Prospective evaluation of hematological parameters in preoperative
renal cell cancer patients. BMC Urol. 22:2012022. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Steinhagen F, Schmidt SV, Schewe JC,
Peukert K, Klinman DM and Bode C: Immunotherapy in sepsis-brake or
accelerate? Pharmacol Ther. 208:1074762020. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Forrest LM, McMillan DC, McArdle CS,
Angerson WJ and Dunlop DJ: Comparison of an inflammation-based
prognostic score (GPS) with performance status (ECOG) in patients
receiving platinum-based chemotherapy for inoperable non-small-cell
lung cancer. Br J Cancer. 90:1704–1706. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
McMillan DC, Crozier JE, Canna K, Angerson
WJ and McArdle CS: Evaluation of an inflammation-based prognostic
score (GPS) in patients undergoing resection for colon and rectal
cancer. Int J Colorectal Dis. 22:881–886. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Watt DG, Roxburgh CS, White M, Chan JZ,
Horgan PG and McMillan DC: A survey of attitudes towards the
clinical application of systemic inflammation based prognostic
scores in cancer. Mediators Inflamm. 2015:8420702015. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Dolan RD, McSorley ST, Park JH, Watt DG,
Roxburgh CS, Horgan PG and McMillan DC: The prognostic value of
systemic inflammation in patients undergoing surgery for colon
cancer: Comparison of composite ratios and cumulative scores. Br J
Cancer. 119:40–51. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Tanaka T, Yoshida T, Masuda K, Takeyasu Y,
Shinno Y, Matsumoto Y, Okuma Y, Goto Y, Horinouchi H, Yamamoto N
and Ohe Y: Prognostic role of modified Glasgow prognostic score in
elderly non-small cell lung cancer patients treated with anti-PD-1
antibodies. Respir Investig. 61:74–81. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Kawakami H, Sunakawa Y, Inoue E, Matoba R,
Noda K, Sato T, Suminaka C, Yamaki M, Sakamoto Y, Kawabata R, et
al: Soluble programmed cell death ligand 1 predicts prognosis for
gastric cancer patients treated with nivolumab: Blood-based
biomarker analysis for the DELIVER trial. Eur J Cancer. 184:10–20.
2023. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Page MJ, McKenzie JE, Bossuyt PM, Boutron
I, Hoffmann TC, Mulrow CD, Shamseer L, Tetzlaff JM, Akl EA, Brennan
SE, et al: The PRISMA 2020 statement: An updated guideline for
reporting systematic reviews. Syst Rev. 10:892021. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Zhang Y, Chen S, Chen H and Li W: A
comprehensive analysis of Glasgow prognostic score (GPS)/the
modified Glasgow prognostic score (mGPS) on immune checkpoint
inhibitor efficacy among patients with advanced cancer. Cancer Med.
12:38–48. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Wells GA, Shea BJ, O'Connell D, Peterson
J, Welch V, Losos M and Tugwell P: The Newcastle Ottawa scale (NOS)
for assessing the quality of non-randomised studies in
meta-analyses. World J Cardiovasc Dis. May 28–2021.(Epub ahead of
print).
|
|
28
|
Sakai D, Omori T, Fumita S, Fujita J,
Kawabata R, Matsuyama J, Yasui H, Hirao M, Kawase T, Kishi K, et
al: Real-world effectiveness of third- or later-line treatment in
Japanese patients with HER2-positive, unresectable, recurrent or
metastatic gastric cancer: A retrospective observational study. Int
J Clin Oncol. 27:1154–1163. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Yang Z, Zhang D, Zeng H, Fu Y, Hu Z, Pan
Y, Chen J, Wang J, Zhang Y, Zhou Z, et al: Inflammation-based
scores predict responses to PD-1 inhibitor treatment in
intrahepatic cholangiocarcinoma. J Inflamm Res. 15:5721–5731. 2022.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Kurosaki T, Kawakami H, Mitani S, Kawabata
R, Takahama T, Nonagase Y, Fumita S, Ozaki T, Chiba Y, Tamura T and
Nakagawa K: Glasgow prognostic score (GPS) and tumor response as
biomarkers of nivolumab monotherapy in third-or later-line setting
for advanced gastric cancer. In Vivo. 34:1921–1929. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Kang HS, Shin AY, Yeo CD, Kim SK, Park CK,
Kim JS, Kim SJ, Lee SH and Kim JW: Significance of Glasgow
prognostic scores in NSCLC patients treated with immunotherapy
after platinum-based cytotoxic chemotherapy. In Vivo. 35:3423–3430.
2021. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Minichsdorfer C, Gleiss A, Aretin MB,
Schmidinger M and Fuereder T: Serum parameters as prognostic
biomarkers in a real world cancer patient population treated with
anti PD-1/PD-L1 therapy. Ann Med. 54:1339–1349. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Namikawa T, Yokota K, Tanioka N, Fukudome
I, Iwabu J, Munekage M, Uemura S, Maeda H, Kitagawa H, Kobayashi M
and Hanazaki K: Systemic inflammatory response and nutritional
biomarkers as predictors of nivolumab efficacy for gastric cancer.
Surg Today. 50:1486–1495. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Kasajima M, Igawa S, Manaka H, Yamada K,
Akazawa Y, Manabe H, Yagami Y, Yamamoto H, Ito H, Kaizuka N, et al:
The Glasgow prognostic score predicts outcomes of pembrolizumab or
atezolizumab monotherapy in patients with pretreated non-small cell
lung cancer. Oncology. 101:69–76. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Kim JH, Ahn B, Hong SM, Jung HY, Kim DH,
Choi KD, Ahn JY, Lee JH, Na HK, Kim JH, et al: Real-world efficacy
data and predictive clinical parameters for treatment outcomes in
advanced esophageal squamous cell carcinoma treated with immune
checkpoint inhibitors. Cancer Res Treat. 54:505–516. 2022.
View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Tokuyama N, Takegawa N, Nishikawa M, Sakai
A, Mimura T, Kushida S, Tsumura H, Yamamoto Y, Miki I and Tsuda M:
Pretreatment Glasgow prognostic score as a predictor of outcomes in
nivolumab-treated patients with advanced gastric cancer. PLoS One.
16:e02476452021. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Fujiwara R, Takemura K, Fujiwara M, Yuasa
T, Yasuoka S, Komai Y, Numao N, Yamamoto S and Yonese J: Modified
Glasgow prognostic score as a predictor of prognosis in metastatic
renal cell carcinoma treated with nivolumab. Clin Genitourin
Cancer. 19:e78–e83. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Matsuki T, Okamoto I, Fushimi C, Sawabe M,
Kawakita D, Sato H, Tsukahara K, Kondo T, Okada T, Tada Y, et al:
Hematological predictive markers for recurrent or metastatic
squamous cell carcinomas of the head and neck treated with
nivolumab: A multicenter study of 88 patients. Cancer Med.
9:5015–5024. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Brown JT, Liu Y, Martini DJ, Shabto JM,
Russler G, Caulfield S, Yantorni LB, Joshi SS, Kissick H, Ogan K,
et al: Baseline modified Glasgow prognostic score (mGPS) in
patients with metastatic renal cell carcinoma (mRCC) treated with
immune checkpoint inhibitors (ICI). J Clin Oncol. 39 (Suppl
15):e165462021. View Article : Google Scholar
|
|
40
|
Brown JT, Liu Y, Shabto JM, Martini D,
Ravindranathan D, Hitron EE, Russler GA, Caulfield S, Yantorni L,
Joshi SS, et al: Modified Glasgow prognostic score associated with
survival in metastatic renal cell carcinoma treated with immune
checkpoint inhibitors. J Immunother Cancer. 9:e0028512021.
View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Freitas C, Jacob M, Tavares N,
Cruz-Martins N, Souto-Moura C, Araújo D, Novais-Bastos H, Santos V,
Fernandes G, Magalhães A, et al: Modified Glasgow prognostic score
predicts survival among advanced non-small cell lung carcinoma
patients treated with anti-PD1 agents. Anticancer Drugs.
32:567–574. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Diker O and Olgun P: Association of the
immune-inflammation-nutritional parameters with immune checkpoint
inhibitor outcomes in patients with advanced non-small cell lung
cancer. J Oncol Sci. 8:43–53. 2022. View Article : Google Scholar
|
|
43
|
Pflug N, Vitus M, Knuever J, Hamacher S,
Mauch C, Schlaak M and Theurich S: Treatment-specific evaluation of
the modified Glasgow-prognostic-score in patients with advanced
cutaneous melanoma. J Eur Acad Dermatol Venereol. 35:e879–e883.
2021. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Ogura Y, Kataoka N, Kunimatsu Y, Tachibana
Y, Sugimoto T, Tani N, Sato I, Hirose K, Kato D and Takeda T:
Predictors of survival among Japanese patients receiving first-line
chemoimmunotherapy for advanced non-small cell lung cancer. Thorac
Cancer. 12:97–105. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Niwa K, Kawakita D, Nagao T, Takahashi H,
Saotome T, Okazaki M, Yamazaki K, Okamoto I, Hirai H, Saigusa N, et
al: Multicentre, retrospective study of the efficacy and safety of
nivolumab for recurrent and metastatic salivary gland carcinoma.
Sci Rep. 10:169882020. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
McMillan DC: The systemic
inflammation-based Glasgow prognostic score: A decade of experience
in patients with cancer. Cancer Treat Rev. 39:534–540. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Luo B, Zhang Y, Zhu X, Ji T, Wu K, Sun D,
Lu Y and Shi L: Prognostic impact of the high-sensitivity modified
glasgow prognostic score on patients undergoing radical surgery for
hepatocellular carcinoma: Authorship. Langenbecks Arch Surg.
409:2232024. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Yamanouchi K, Murakami S, Sato A, Ogawa S,
Shinagawa H and Kamohara Y: Integrated evaluation of inflammatory,
nutritional, and sarcopenia markers to predict survival in
metastatic breast cancer patients. In Vivo. 37:811–817. 2023.
View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Kinoshita A, Onoda H, Imai N, Iwaku A,
Oishi M, Fushiya N, Koike K, Nishino H and Tajiri H: Comparison of
the prognostic value of inflammation-based prognostic scores in
patients with hepatocellular carcinoma. Br J Cancer. 107:988–993.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Shimoyama R, Imamura Y, Uryu K, Mase T,
Ohtaki M, Ohtani K, Shiragami M, Fujimura Y, Hayashi M, Shinozaki N
and Minami H: Inflammation-based prognostic markers in patients
with advanced or recurrent gastric cancer treated with nivolumab:
Tokushukai REAl-world data project 02 (TREAD 02). Mol Clin Oncol.
21:902024. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Lorton CM, Higgins L, O'Donoghue N,
Donohoe C, O'Connell J, Mockler D, Reynolds JV, Walsh D and Lysaght
J: C-reactive protein and C-reactive protein-based scores to
predict survival in esophageal and junctional adenocarcinoma:
Systematic review and meta-analysis. Ann Surg Oncol. 29:1853–1865.
2022. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Potempa LA, Rajab IM, Olson ME and Hart
PC: C-reactive protein and cancer: Interpreting the differential
bioactivities of its pentameric and monomeric, modified isoforms.
Front Immunol. 12:7441292021. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Sproston NR and Ashworth JJ: Role of
C-reactive protein at sites of inflammation and infection. Front
Immunol. 9:7542018. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Whiteside TL: The tumor microenvironment
and its role in promoting tumor growth. Oncogene. 27:5904–5912.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Yoshida T, Ichikawa J, Giuroiu I, Laino
AS, Hao Y, Krogsgaard M, Vassallo M, Woods DM, Stephen Hodi F and
Weber J: C reactive protein impairs adaptive immunity in immune
cells of patients with melanoma. J Immunother Cancer.
8:e0002342020. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Bilen MA, Martini DJ, Liu Y, Lewis C,
Collins HH, Shabto JM, Akce M, Kissick HT, Carthon BC, Shaib WL, et
al: The prognostic and predictive impact of inflammatory biomarkers
in patients who have advanced-stage cancer treated with
immunotherapy. Cancer. 125:127–134. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Zheng X, Zhang L, Wu L, Zhao J, Sun J,
Fang Y, Zhou J, Chu Q, Shen Y, Yang Z, et al: Baseline C-reactive
protein predicts efficacy of the first-line immune checkpoint
inhibitors plus chemotherapy in advanced lung squamous cell
carcinoma: A retrospective, multicenter study. BMC Cancer.
23:12442023. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Wang H, Yang R, Zhou K, Wang S, Cheng C,
Liu D and Li W: Association between pretreatment C-reactive protein
level and survival in non-small cell lung cancer patients treated
with immune checkpoint inhibitors: A meta-analysis. Int
Immunopharmacol. 124:1109372023. View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Kijima T, Yamamoto H, Saito K, Kusuhara S,
Yoshida S, Yokoyama M, Matsuoka Y, Numao N, Sakai Y, Matsubara N,
et al: Early C-reactive protein kinetics predict survival of
patients with advanced urothelial cancer treated with
pembrolizumab. Cancer Immunol Immunother. 70:657–665. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Keller U: Nutritional laboratory markers
in malnutrition. J Clin Med. 8:7752019. View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Yoo SK, Chowell D, Valero C, Morris LGT
and Chan TA: Pre-treatment serum albumin and mutational burden as
biomarkers of response to immune checkpoint blockade. NPJ Precis
Oncol. 6:232022. View Article : Google Scholar : PubMed/NCBI
|
|
62
|
Guo Y, Wei L, Patel SH, Lopez G, Grogan M,
Li M, Haddad T, Johns A, Ganesan LP, Yang Y, et al: Serum albumin:
Early prognostic marker of benefit for immune checkpoint inhibitor
monotherapy but not chemoimmunotherapy. Clin Lung Cancer.
23:345–355. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Roxburgh CS and McMillan DC: Role of
systemic inflammatory response in predicting survival in patients
with primary operable cancer. Future Oncol. 6:149–163. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
64
|
Argilés JM, Busquets S, Stemmler B and
López-Soriano FJ: Cancer cachexia: Understanding the molecular
basis. Nat Rev Cancer. 14:754–762. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
65
|
Soeters PB, Wolfe RR and Shenkin A:
Hypoalbuminemia: Pathogenesis and clinical significance. JPEN J
Parenter Enteral Nutr. 43:181–193. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
66
|
Kose E, Wakabayashi H and Yasuno N:
Polypharmacy and malnutrition management of elderly perioperative
patients with cancer: A systematic review. Nutrients. 13:19612021.
View Article : Google Scholar : PubMed/NCBI
|
|
67
|
Daniele A, Divella R, Abbate I,
Casamassima A, Garrisi VM, Savino E, Casamassima P, Ruggieri E and
DE Luca R: Assessment of nutritional and inflammatory status to
determine the prevalence of malnutrition in patients undergoing
surgery for colorectal carcinoma. Anticancer Res. 37:1281–1287.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
68
|
Unal D, Eroglu C, Ozsoy SD, Besirli A,
Orhan O and Kaplan B: Effect on long-term survival of psychiatric
disorder, inflammation, malnutrition, and radiotherapy-related
toxicity in patients with locally advanced head and neck cancer. J
BUON. 886–893. 2015.PubMed/NCBI
|