Introduction
Transforming growth factor β (TGFβ), a well-known
angiogenic and immunosuppressive cytokine, significantly affects
mesenchymal stromal cells (MSCs) through different processes. Among
those, inhibition of their terminal differentiation into
osteoblasts (1), and regulation of
chondrogenic differentiation (2),
both of which requiring TGFβ signaling. In pathological conditions,
TGFβ signaling in MSCs contributes to the development of
myelofibrosis, a condition characterized by increased collagen
deposition in the bone marrow (3,4). On
the other hand, overexpressing TGFβ in MSCs has been shown to
enhance their therapeutic effects, particularly in reducing organ
injury and inflammation during septic conditions (5). Collectively, these effects highlight
the complex multifaceted role of TGFβ in regulating MSCs
functions.
Efforts to enhance MSCs' therapeutic potential have
increasingly focused on their response to TGFβ. For instance,
overexpressing TGFβ1 in MSCs was found to attenuate organ
dysfunction in septic mice, by reducing macrophage-driven
inflammation, and also by promoting the mobilization of MSCs for
tissue repair (5,6). Additionally, strategies exploiting
specific inhibitors of TGFβ signaling, along with genetic
engineering techniques have also been employed to overexpress or
knock down TGFβ receptors (TGFβR) or downstream signaling molecules
in MSCs (7). Hypoxic
preconditioning, a condition that mimics the low oxygen tension
present in ischemic tissues or that found within solid tumors
(8,9), has been shown to upregulate TGFβ and
other immunosuppressive factors, thereby improving MSCs' ability to
modulate immune responses (4).
These strategies help harness the beneficial effects of TGFβ while
minimizing potential adverse effects, thereby improving the
efficacy of MSC-based therapies.
MSCs play a complex role in tumor angiogenesis,
acting as both promoters and inhibitors of tumor growth [10]. This
dual role makes them a double-edged sword in cancer therapy, as
their ability to home to tumor sites and modulate the tumor
microenvironment (TME) is being explored for therapeutic purposes,
including targeted drug delivery and modulation of the immune
response (10,11). Recently, MSCs have been suggested to
be involved in vasculogenic mimicry (VM), a process where cells
form vascular-like structures without the involvement of
endothelial cells, contributing in pathological settings to tumor
blood supply and metastasis (12).
Interestingly, MSCs' ability to migrate and invade tissues is
closely linked to epithelial-to-mesenchymal transition (EMT), a
process closely linked to VM, as it enables cells to acquire
properties necessary for forming these vessel-like structures
(13). MSCs share characteristics
with cancer stem cells (CSCs), which are known to play a crucial
role in VM. CSCs can differentiate into various cell types,
including those that contribute to VM (14). Moreover, MSCs interact with the TME,
promoting conditions that support VM through their paracrine
activity, including TGFβ secretion, and that enhance local
angiogenesis and VM that support tumor growth and metastasis
(15,16). Understanding the role of MSCs in VM
could facilitate the development of targeted therapies aimed at
inhibiting this process, thereby potentially reduce tumor
progression and metastasis.
As cancer cells often adapt and develop resistance
to TGFβ's tumor-suppressive effects and start secreting TGFβ
themselves, the autocrine effects of such secretion role in MSCs'
ability to promote tumor growth, invasion, metastasis, and immune
evasion remains unknown (17). For
instance, in colorectal cancer, TGFβ secretion by tumor cells
contributes to the TME, facilitating immune suppression and
neovascularization (18).
Similarly, in breast cancer, TGFβ secreted by tumor cells and
stromal cells within the TME supports tumor maintenance and
progression (19).
Tumors have therefore a remarkable ability to
regulate their environment and influence body homeostasis through
several mechanisms including metabolic reprogramming to support
their rapid growth and survival. This involves altering their
energy production pathways, such as increasing glycolysis not only
to support the energy demands of tumor cells but to also create an
acidic and hypoxic tumor microenvironment TME (20), which can suppress the immune
response (21). In addition,
interaction with stromal cells within the TME can further promote
tumor growth and new blood vessel formation (21). Finally, tumors can hijack normal
homeostatic processes in the body as they can alter cytokine and
chemokine secretion, leading to systemic inflammation and immune
dysregulation (22,23). These mechanisms highlight the
complex interplay between tumors and their environment,
demonstrating how MSC recruitment can support their growth and
evade the body's defense mechanisms. In this study, we aim to
investigate the potential impact of TGFβ and particularly, the
interrelation between TGFβ and Smad2/3-dependant signaling on the
in vitro formation of 3D capillary-like structures.
Materials and methods
Reagents
Micro bicinchoninic acid (BCA) protein assay
reagents were from Pierce (Micro BCA™ Protein Assay Kit; Thermo
Fisher Scientific, Inc.). The polyclonal antibodies against Snail
(3879S), FOXC2 (12974S), and Fibronectin (30903S), as well as the
monoclonal antibody against GAPDH (D4C6R) were all from Cell
Signaling Technology. HRP-conjugated donkey anti-rabbit and
anti-mouse immunoglobulin (Ig) G secondary antibodies were from
Jackson ImmunoResearch Laboratories. All other reagents were from
Sigma-Aldrich; Merck KGaA.
Cell culture and capillary-like
structure formation assay
Human bone marrow-derived mesenchymal stromal/stem
cells (MSCs, PCS-500-012) were purchased from the American Type
Culture Collection. Cell culture media was from Life Technologies
Corp. Cells were plated in high glucose aMEM supplemented with 10%
FBS and 50 units/ml penicillin/streptomycin and cultured in a
humidified incubator at 37°C with 5% CO2. MSCs were kept
subconfluent and expanded for not more than 10 passages by a 1:2
split on a weekly basis. VM was assessed in vitro using
Cultrex (3432-010-01, R&D Systems) to monitor 3D capillary-like
structures formation (24). In
brief, each well of a 96-well plate was pre-coated with 50 µl of
Cultrex. MSCs suspension in culture media (104 cells/100
µl) was then seeded on top of polymerized Cultrex and incubated at
37°C in a CO2 incubator for different time points of
vascular network formations. Phase contrast pictures were taken
over time using a digital camera coupled to an inverted microscope.
For each loop and tube measurement, the pixels that belong to its
edge are considered its border or perimeter. The number of loops
and tubes, as well as tube branching formed by the cells were
quantified using the Wimasis analysis software (https://www.wimasis.com; Cordoba, Spain) or the ImageJ
software (https://imagej.net) (25).
Total RNA isolation, cDNA synthesis,
and reverse transcription-quantitative PCR (RT-qPCR)
Total RNA was extracted from cell monolayers using 1
ml of TRIzol reagent for a maximum of 3×106 cells as
recommended by the manufacturer (Life Technologies). For cDNA
synthesis, 2 µg of total RNA was reverse-transcribed using a
high-capacity cDNA reverse transcription kit (Applied Biosystems).
The cDNA was stored at −20°C prior to PCR. Gene expression was
quantified qPCR using iQ SYBR-Green Supermix (Bio-Rad
Laboratories). DNA amplification was carried out using an Icycler
iQ5 (Bio-Rad Laboratories) and product detection was performed by
measuring the binding of the fluorescent dye SYBR-Green I to
double-stranded DNA. The following primer sets were from Qiagen:
FOXC2 (Hs_FOXC2_1_SG, QT00220871), SNAI1 (Hs_SNAI1_SG, QT00010010),
Fibronectin (Hs_FN1_1_SG, QT00038024), TGFβ (Hs_TGFB1_1_SG,
QT00000728), GAPDH (Hs_GAPDH_1_SG, QT00079247) and Peptidylprolyl
Isomerase A (PPIA) (Hs_PPIA_4_SG, QT01866137). The relative
quantities of target gene mRNA were normalized against internal
housekeeping genes PPIA and GAPDH. The RNA was measured by
following a ∆CT method employing an amplification plot
(fluorescence signal vs. cycle number). The difference
(∆CT) between the mean values in the triplicate samples
of the target gene and the housekeeping genes was calculated with
the CFX manager Software version 2.1 (Bio-Rad Laboratories) and the
relative quantified value (RQV) was expressed as 2−ΔΔCq
(26). Single amplicons and
appropriate melting curves were indicative of specific qPCR
conditions and efficacy (not shown).
Transfection method and RNA
interference
For gene silencing experiments, MSCs were
transiently transfected with siRNA sequences using
Lipofectamine-2000 transfection reagent (Thermo Fisher Scientific,
Inc.). Gene silencing was performed over 24 h using 20 nM siRNA
against TGFβ (Hs_TGFB1_2 FlexiTube siRNA Geneglobe ID: SI00013601),
Smad2 (Hs_SMAD2_1 FlexiTube siRNA GeneGlobe ID: SI00082460), Smad3
(Hs_SMAD3_1 FlexiTube siRNA GeneGlobe ID: SI00082481), or scrambled
sequences (AllStar Negative Control siRNA, 1027281). The above
small interfering RNA and mismatch siRNA were all synthesized by
Qiagen and annealed to form duplexes. Gene silencing efficacy was
assessed by RT-qPCR as described above.
Nuclear extraction
Nuclear extraction was performed as described by us
previously (27). Briefly, cell
monolayers were first lysed with a cytoplasmic buffer and then with
a nuclear buffer according to the manufacturer's instructions
(Invent Biotechnologies, SC-003). In the case of the cells cultured
on Cultrex, they were first detached from the matrix using a
non-enzymatic Cultrex organoid harvesting and dissociation solution
(3700-100-01) from R&D Systems. Nucleus enrichment was assessed
upon Fibrillarin protein expression, whereas protein GAPDH protein
expression was used to assess cytosolic purity/contamination of the
nuclear fraction.
Western blot
Electrophoresis reagents origin, total cell lysis
procedure, SDS-polyacrylamide gel electrophoresis, electro transfer
to low-fluorescence polyvinylidene difluoride membranes, and
immunodetection were conducted as described in detail previously
(28). Immunoreactive material was
visualized by enhanced chemiluminescence.
Chemotactic cell migration assay
Cell migration assays were carried out using the
Real-Time Cell Analyzer (RTCA) Dual-Plate (DP) Instrument of the
xCELLigence system (Roche Diagnostics). Adherent MSC monolayers
were trypsinized and seeded (30,000 cells/well) onto CIM-Plates 16
(Roche Diagnostics). These migration plates are similar to
conventional transwells (8 µm pore size) but have gold electrode
arrays on their bottom side of the membrane to provide real-time
data acquisition of cell migration. Prior to cell seeding, the
underside of the wells from the upper chamber were coated with 25
µl of 0.15% gelatin in PBS and incubated for 1 h at 37°C. Cell
migration was continuously monitored for up to 6 h using serum-free
media, in the presence or absence of 30 ng/ml TGFβ. In all cases,
the impedance values were measured by the RTCA DP Instrument
software and were expressed as Normalized Cell Migration Index.
Each experiment was performed two times in triplicates.
Statistical data analysis
All statistical analyses were conducted using the
GraphPad Prism 7 software (Dotmatics). Data and error bars are
presented as the mean ± standard error of the mean from three or
more independent experiments, unless otherwise specified.
Hypothesis testing was performed using the Kruskal-Wallis test
followed by a Dunn Tukey's post-test (>2 groups). P<0.05 was
considered to indicate a statistically significant difference.
Results
MSCs in vitro VM triggers the
expression of EMT biomarkers and Smad2/3 phosphorylation
The ability of MSCs to form in vitro 3D
capillary-like structures mimicking VM was first assessed as
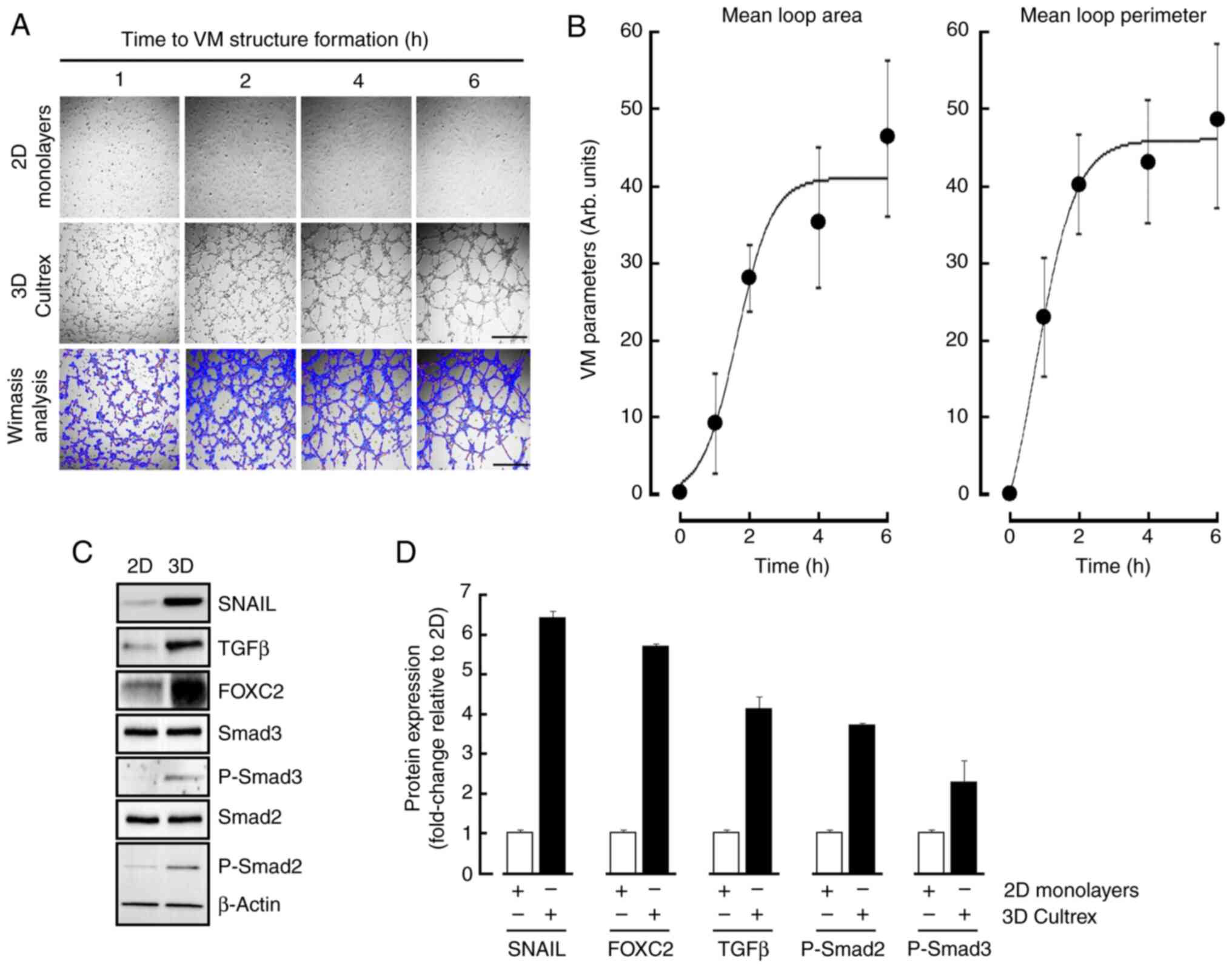
described in the Methods section. Mature structures were formed
upon 6 h (Fig. 1A, middle panels)
when compared to 2D monolayers (Fig.
1A, upper panels). VM parameters analysis, namely mean loop
area and perimeter, were performed as described in the Methods
section (Fig. 1A, lower panels) and
reflected such in vitro maturation in time (Fig. 1B). Cell lysates were isolated from
2D and 3D cultures and immunoblotting performed in an attempt to
characterize the acquisition of an epithelial-to-mesenchymal
transition (EMT) molecular phenotype as well as TGFβ signaling
(Fig. 1C). VM structures were found
to significantly trigger the expression of EMT biomarker Snail and
FOXC2 as reported elsewhere (24),
whereas inductions in TGFβ and of the phosphorylated states of
Smad2/3, but not that of total Smad2/3 or β-Actin, were also
observed (Fig. 1D, black bars).
Harmonized densitometric normalization was performed to β-Actin for
the expression of all those tested biomarkers that were found to be
changed only. Altogether, these data suggest that a potential TGFβ
signaling axis appears to correlate with EMT and be involved during
VM in MSCs.
Transient silencing of TGFβ and
pharmacological inhibition of TGFβR1 kinase activity alters in
vitro VM
The contribution of the TGFβ signaling axis
involving the TGFβR was next assessed using Galunisertib, a
pharmacological inhibitor well known to alter the kinase activity
of TGFβR (29–31). Moreover, the requirement of TGFβ, as
induced upon VM (Fig. 1), was also
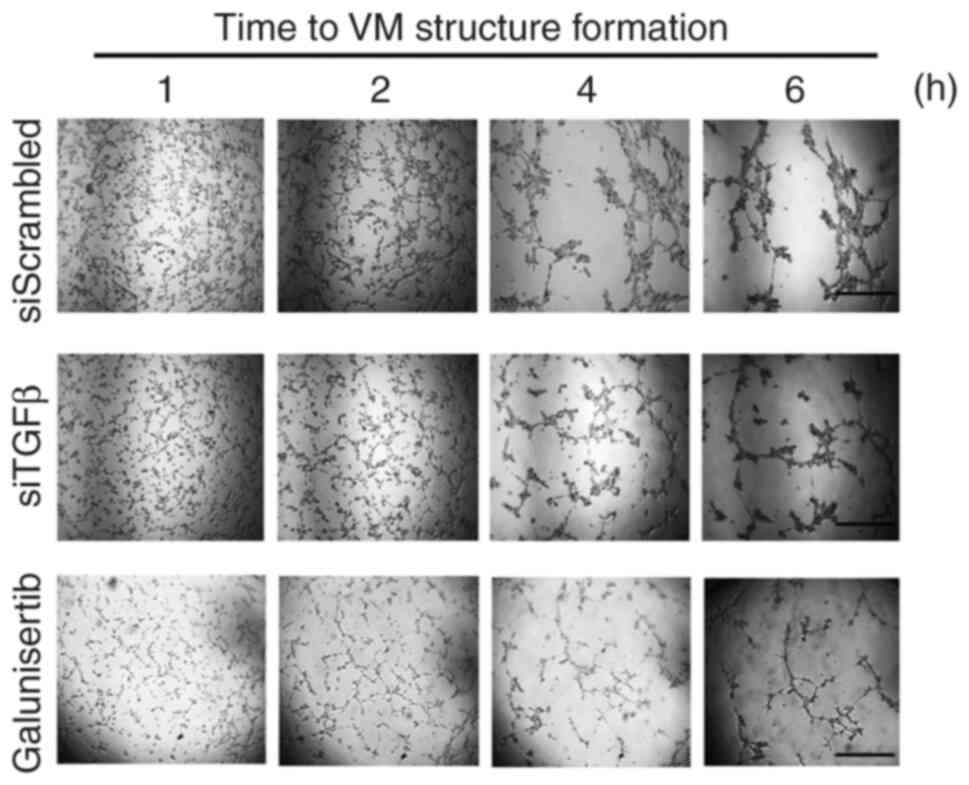
addressed. Pre-transient silencing of TGFβ was performed for 24 h,
then cells seeded on top of Cultex for 6 h. VM structures were
reduced when TGFβ was silenced (Fig.
2, middle panels). This suggests a potential requirement for an
autocrine regulation process to take place in order to trigger VM.
Similarly, when cells were treated with Galunisertib, VM formation
was also inhibited (Fig. 2, lower
panels). Altogether, these data confirm that a TGFβ signaling axis
requiring the kinase activity of the TGFβR is a prerequisite to VM
and further support the increase in downstream phosphorylation
status of Smad2/3 observed (Fig.
1). Whether TGFβ could further solely and specifically regulate
any downstream transcriptional activity was next assessed.
TGFβ triggers activation of Smad2/3
and nuclear translocation of Snail
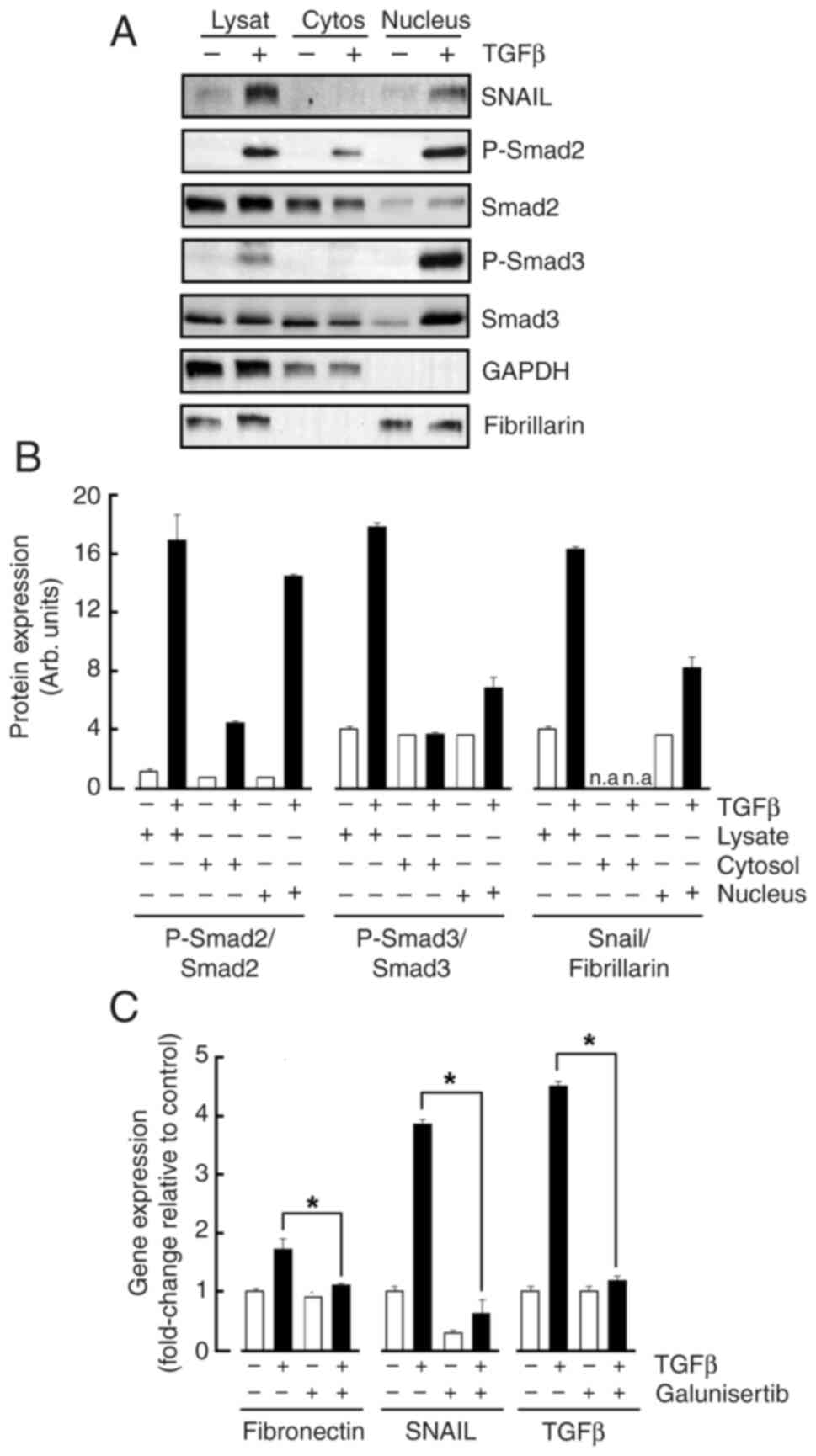
MSCs were treated or not with TGFβ and nuclear
fractionation performed as described in the Methods section. Cell
lysates were harvested along cytoplasmic and nuclear fractions and
immunoblotting performed. The purity control of the cytosolic
fraction and of the nuclear material was respectively attested when
GAPDH and Fibrillarin were immunoblotted (Fig. 3A). While TGFβ effects in total cell
lysates were confirmed (Fig. 3B,
Lysate), Snail as well as the phosphorylated Smad2/3 proteins were
found to significantly translocate to the Fibrillarin-enriched
nucleus fraction (Fig. 3B,
Nucleus). Collectively, efficient nuclear translocation in response
to TGFβ treatment prompts for the exploration of gene regulation
processes. Total RNA was therefore extracted from treated cells and
genes of interest assessed by RT-qPCR. TGFβ indeed significantly
increased Snail, Fibronectin, and TGFβ gene expression levels
confirming increased transcriptional activity in treated MSC
(Fig. 3C). Galunisertib
pharmacological inhibition of the TGFβR activity prevented those
TGFβ-mediated inductions and confirms the necessity of an active
signal transducing process (Fig.
3C).
VM triggers differential EMT and TGFβ
biomarker gene expression and requires Smad2/3 signaling
Given the active transcriptional process induced in
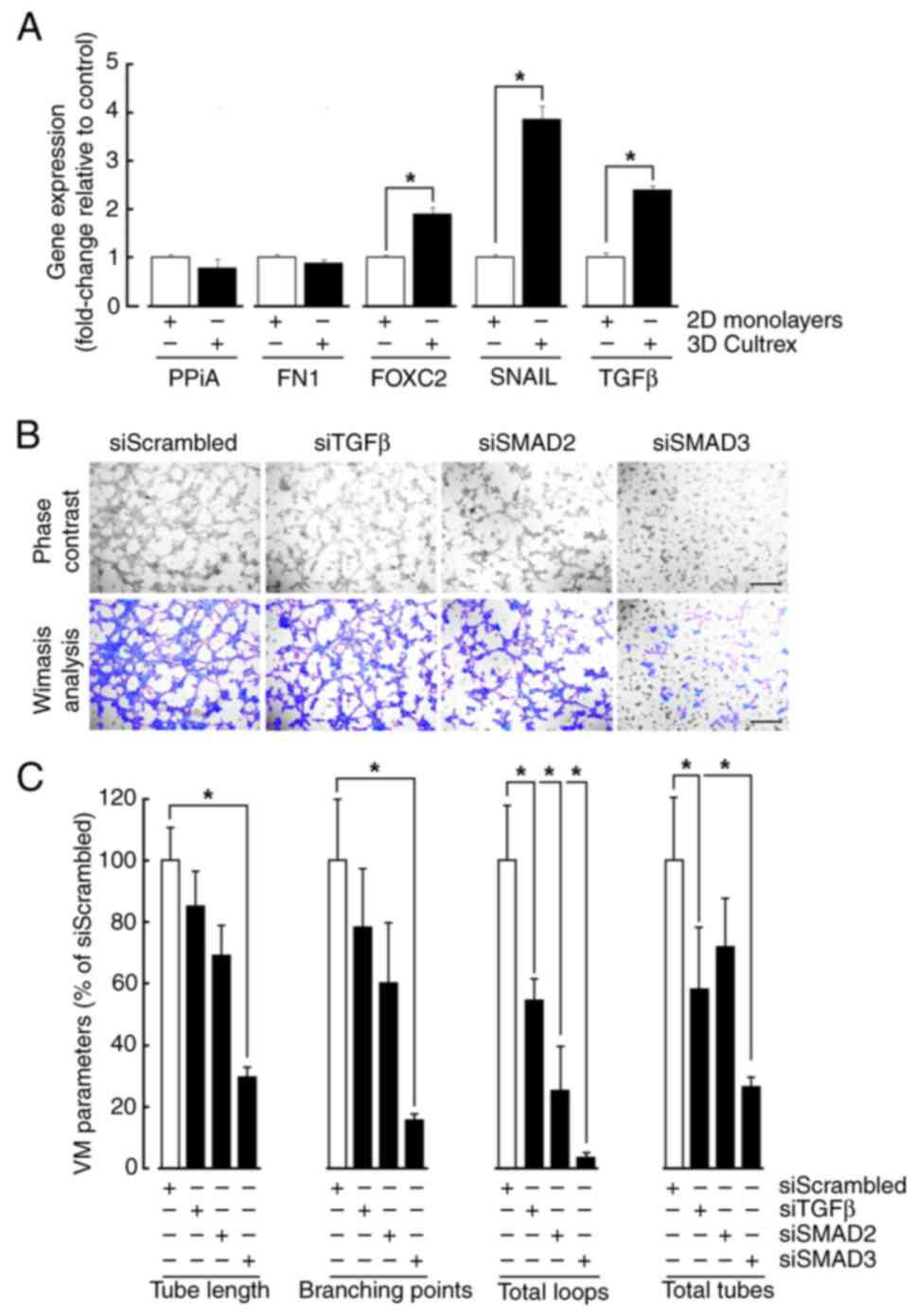
TGFβ-treated cells, total RNA was extracted from cell monolayers
and from cells forming VM on Cultrex. While increased gene
expression of Snail, FOXC2, and TGFβ upon VM recapitulated that
increase observed in response to TGFβ, gene expression of
Fibronectin remained however unchanged (Fig. 4A). As FOXC2 and SNAIL have been
previously reported to alter VM formation in MSC (24), transient gene silencing was
performed to repress TGFβ, as well as Smad2/3 and cells
subsequently seeded on top of Cultrex (Fig. 4B). Accordingly with the downstream
effect of Galunisertib on Smad2/3 phosphorylation, silencing of
Smad3 reduced all the VM parameters associated with vascular
structure formation, including tube length, branching points, total
loops, and total tubes (Fig. 4C).
This was strongly associated with significant reduction in total
loops, and a tendency to reduction of all other parameters in
Smad2-silenced cells. Intriguingly, silencing of TGFβ also only
altered total loops and tubes formation without affecting other VM
parameters (Fig. 4C). Collectively,
this evidence suggests that common signaling cues are triggered
upon either TGFβ treatment or VM formation. Moreover, given the
selective regulation of EMT biomarkers expression, namely that of
Fibronectin, complex interplay between these cues will require
further investigation although evidence suggests that possible
autocrine regulation by TGFβ may regulate VM.
Silencing of Smad2/3 in MSC inhibits
their chemotactic response to TGFβ
Given some of the common acquisition of an EMT
phenotype between TGFβ treatment and VM formation, the involvement
of the Smad2/3 signaling required for MSCs to migrate and form 3D
capillary-like structures was next assessed. Coupled to the
increased TGFβ expression and a possible autocrine regulation, MSC
chemotaxis in response to TGFβ was performed in siRNA transiently
silenced cells for Smad2 and Smad3 as described in
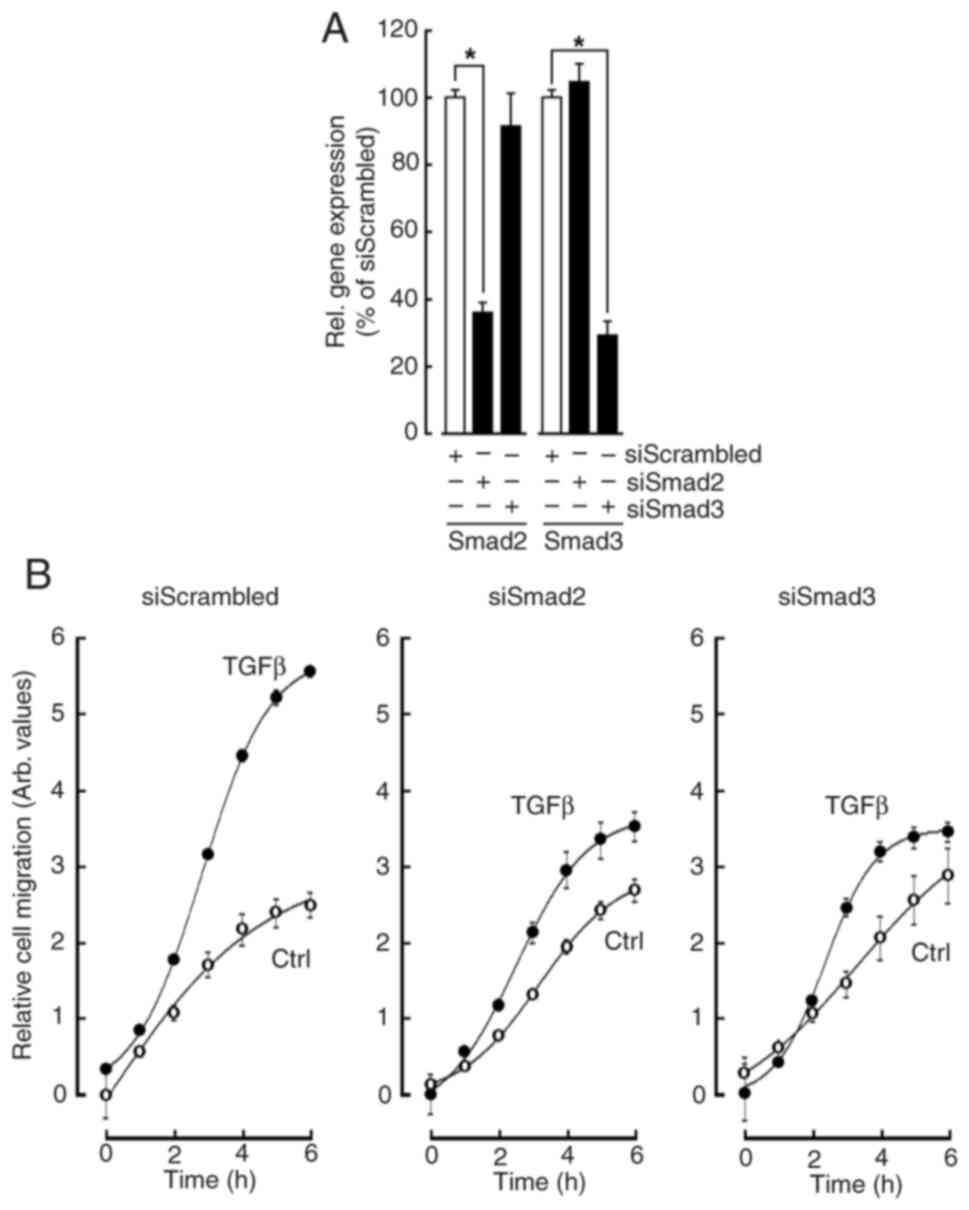
the Methods section and validated (Fig.
5A). Real-time cell migration was monitored for up to 6 h and
found to significantly increase in response to TGFβ (Fig. 5B, left panel, closed circles). When
gene silencing was performed to suppress either Smad2 or
Smad3, TGFβ chemotaxis was significantly reduced in both
conditions (Fig. 5B, middle and
right panels respectively). This evidence supports the hypothesis
that an autocrine TGFβ-mediated process could regulate in
vitro VM formation. More importantly, and along their role in
VM formation described above, this represents strong evidence for
the involvement of Smad2/3 transducing events in response to such
autocrine regulation.
Discussion
Several signaling pathways such as Wnt/β-Catenin,
Notch signaling, PI3K/Akt, MAPK/ERK, and Hedgehog pathways regulate
MSCs plasticity (32), impacting
their ability to differentiate into various cell types and
compromising their adaptive capacity within different environments
including the TME. In the current study we focused on the TGFβ/SMAD
pathway which is recognized to regulate MSCs differentiation into
3D capillary-like structures, a process also believed to maintain
stemness (33). As TGFβ signaling
through Smad2/3 is particularly important for inducing EMT
(34), one can thereafter safely
assume that these pathways collectively interact with each other,
and with the extracellular matrix (ECM) proteins to regulate MSC
plasticity and adaptability, facilitating their role in VM
(35). Understanding these
interconnected pathways can help to develop novel therapeutic
strategies in tissue regeneration repair, or in anticancer
therapies.
Cytokines' regulation that promotes MSC mobilization
include Stromal Cell-Derived Factor-1 (SDF-1), Granulocyte
Colony-Stimulating Factor (G-CSF), Granulocyte-Macrophage
Colony-Stimulating Factor (GM-CSF), Substance P (SP), and
incidentally, TGFβ (36). These
cytokines interact through complex networks to regulate MSC
motility and migration (37). While
MSCs can exhibit anti-tumor effects, such as through modulation of
the immune response within the TME (11), tumors create an inflammatory
microenvironment that releases several of these cytokines and
chemokines which enable MSCs recruitment to the tumor site
(38). Notably, vascular
progenitors derived from murine bone marrow stromal cells were
found to be avidly recruited by vascularizing tumors (39). Once recruited, MSC can interact with
tumor cells through paracrine signaling, promoting angiogenesis,
tumor growth, and metastasis (40).
Additionally, MSCs are involved in VM a process
where vessel-like structures form without endothelial cells,
providing a blood supply to the tumor. MSC can contribute to these
pseudo-vascular networks by differentiating into endothelial-like
cells (24). This involvement of
MSCs in VM highlights their role in tumor progression and
metastasis (41). While TGFβ
signaling is known to promote tumor vasculature by strengthening
the association between pericytes and endothelial cells (42), which is crucial for the formation of
stable blood vessels, the role of TGFβ-primed MSC was yet to be
explored. Here, we demonstrate that TGFβ and TGFβ-mediated
signaling play a role in regulating in vitro VM.
TGF-β also plays a crucial role in immunosuppression
within the TME through several molecular mechanisms involving, in
part, the Smad2 and Smad3 proteins (43). These mechanisms collectively help
TGFβ maintain immune homeostasis and prevent overactive immune
responses that could lead to autoimmunity or chronic inflammation
(44). Strategies, such as using
antibodies to block TGFβ receptors have shown promise in reversing
immunosuppression. These antibodies have been shown to modulate
macrophage polarization and enhance immune cell infiltration,
leading to significant anti-tumor effects (45). Since VM is associated with
immunosuppression in cancer, our study provides the first evidence
of Smad2 and Smad3 involvement in MSC-driven. These signaling
intermediates are part of the TGFβ signaling pathway and, through
this nuclear translocation, appeared to be crucial in the in
vitro formation of VM. Pharmacological evidence further
highlights the importance of TGFβR kinase activity in VM. The
nuclear translocation of phosphorylated Smad2/3 is necessary for
transcriptional regulation, reinforcing their role in MSC-driven VM
(46). While we show that TGFβ
potentially plays a significant role in regulating MSCs within the
context of cancer, in part through the acquisition of mesenchymal
properties which enhance their migratory capabilities and VM, more
work will be required to better assess how increased TGFβ secreted
by MSCs forming VM can shape the TME through the secretion of ECM
components and cytokines that would support tumor growth and immune
evasion. MSCs secretion of TGFβ and contribution to an
immunosuppressive environment may also affect immune cells within
the TME and help tumors evade immune detection. Whether MSCs can
maintain the cancer stem cells crucial for tumor initiation,
progression, and resistance to therapy will also require to be
addressed. Altogether, these multifaceted roles of TGFβ definitely
make it a critical target for therapeutic strategies aimed at
disrupting its signaling pathways to inhibit cancer
progression.
On the other hand, the contribution of MSCs to
carcinogenic processes can be relatively well exploited in clinical
settings by employing them as drug delivery vehicles (47). Thanks to their natural tumor-homing
abilities, MSCs can be engineered to deliver anticancer drugs like
doxorubicin, paclitaxel, and cisplatin directly to tumor sites
(11). This approach has the
potential to enhance treatment efficacy while minimizing side
effects. Moreover, MSCs can also be engineered to alter the TME so
that to inhibit cancer progression by, modifying them to secrete
anti-tumor cytokines or to disrupt the supportive stroma around
tumors (48). Accordingly, several
clinical trials are underway to evaluate the safety and efficacy of
MSC-based therapies in cancer treatment (49). Although these strategies are still
under investigation, they hold promise for advancing cancer
treatment by targeting the complex and diverse roles MSCs play in
tumor biology.
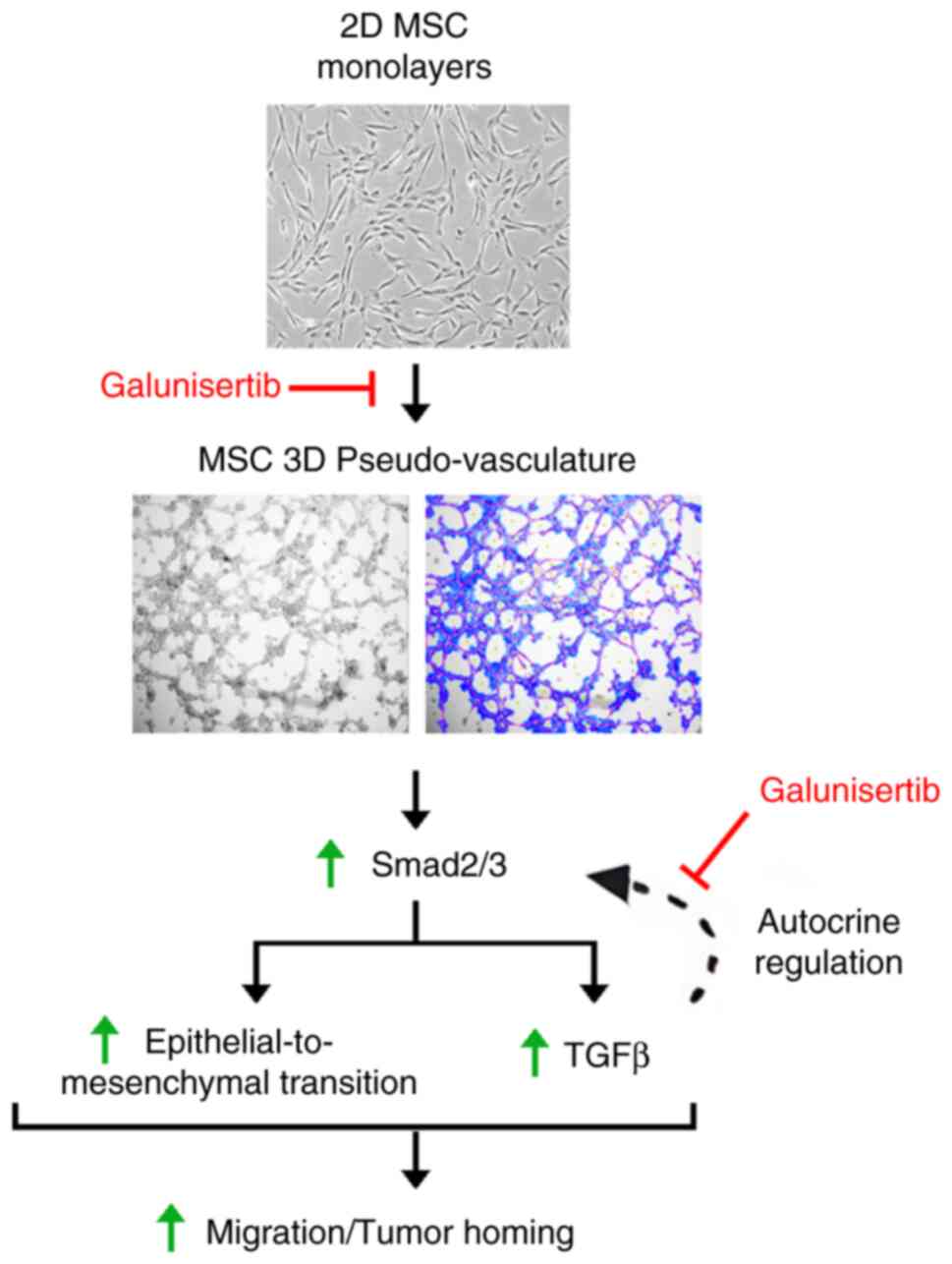
In conclusion, MSCs' key contributions and guiding
significance for future research and clinical practice include
their tumor homing ability, and capacity to modulate the immune
response, which is crucial in the TME (50). Our study highlights in vitro
an alternative mechanism involving VM and allowing MSCs to adopt a
vascular-like phenotype. The role of Smad2/3 signaling and
TGFβ-mediated autocrine regulation in MSCs mobilization and in
vitro VM is summarized (Fig.
6). The involvement of TGFβ-induced autocrine signaling in VM
could be a target for future anticancer strategies targeting
Smad2/3 signaling in MSCs. Unraveling the roles of Smad2/3 in VM
highlights their potential as therapeutic targets in cancer
treatment. Modulating this pathway could provide means to disrupt
the VM process and inhibit tumor progression. Drugs that inhibit
TGFβR activity can indirectly modulate Smad2/3 signaling. Several
of these inhibitors are currently being investigated for their
ability to disrupt TGFβ-mediated cancer progression (51,52).
While these experimental approaches are still in early stages of
research, further studies are essential to evaluate their safety
and effectiveness in cancer treatment. Despite their potential,
MSCs face several challenges in clinical applications. Among those,
combining MSC-based therapies with other treatments like
chemotherapy, radiotherapy, and immunotherapy could eventually
enhance overall treatment efficacy but may show limitations in
clinical trials (53). Importantly,
in the current study, additional research is needed to explore
their potential in specifically targeting MSCs mobilization
processes.
Acknowledgements
Not applicable.
Funding
This work was funded by a grant from the Natural Sciences and
Engineering Research Council of Canada (NSERC, grant no.
RGPIN-2024-04541).
Availability of data and materials
The data generated in the present study may be
requested from the corresponding author.
Authors' contributions
KPDG, MER, AZ, MD and BA contributed to the study
conception and design. Material preparation, data collection and
analysis were performed by KPDG, MER, AZ and MD. KPDG, AZ and BA
confirm the authenticity of all the raw data. The first draft of
the manuscript was written by KPDG and BA. All authors commented on
previous versions of the manuscript. All authors read and approved
the final version of the manuscript.
Ethics approval and consent to
participate
The need for ethics approval for the use of human
bone marrow-derived mesenchymal stromal/stem cells was waived by
the Université du Québec à Montréal ethics committee.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
Authors' information
BA holds an institutional Research Chair in Cancer
Prevention and Treatment.
Glossary
Abbreviations
Abbreviations:
|
CSCs
|
cancer stem cells
|
|
ECM
|
extracellular matrix
|
|
EMT
|
epithelial-to-mesenchymal
transition
|
|
PPIA
|
peptidylprolyl isomerase A
|
|
TGFβ
|
transforming growth factor β
|
|
TGFβR
|
transforming growth factor β
receptor
|
|
TME
|
tumor microenvironment
|
|
VM
|
vasculogenic mimicry
|
References
|
1
|
Grafe I, Alexander S, Peterson JR, Snider
TN, Levi B, Lee B and Mishina Y: TGF-β family signaling in
mesenchymal differentiation. Cold Spring Harb Perspect Biol.
10:a0222022018. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Yang X, Tian S, Fan L, Niu R, Yan M, Chen
S, Zheng M and Zhang S: Integrated regulation of chondrogenic
differentiation in mesenchymal stem cells and differentiation of
cancer cells. Cancer Cell Int. 22:1692022. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Yao JC, Oetjen KA, Wang T, Xu H, Abou-Ezzi
G, Krambs JR, Uttarwar S, Duncavage EJ and Link DC: TGF-β signaling
in myeloproliferative neoplasms contributes to myelofibrosis
without disrupting the hematopoietic niche. J Clin Invest.
132:e1540922022. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Wei E, Hu M, Wu L, Pan X, Zhu Q, Liu H and
Liu Y: TGF-β signaling regulates differentiation of MSCs in bone
metabolism: Disputes among viewpoints. Stem Cell Res Ther.
15:1562024. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Liu F, Xie J, Zhang X, Wu Z, Zhang S, Xue
M, Chen J, Yang Y and Qiu H: Overexpressing TGF-β1 in mesenchymal
stem cells attenuates organ dysfunction during CLP-induced septic
mice by reducing macrophage-driven inflammation. Stem Cell Res
Ther. 11:3782020. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Wan M, Li C, Zhen G, Jiao K, He W, Jia X,
Wang W, Shi C, Xing Q, Chen YF, et al: Injury-activated
transforming growth factor β controls mobilization of mesenchymal
stem cells for tissue remodeling. Stem Cells. 30:2498–2511. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Fan XL, Zhang Y, Li X and Fu QL:
Mechanisms underlying the protective effects of mesenchymal stem
cell-based therapy. Cell Mol Life Sci. 77:2771–2794. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Denko NC and Giaccia AJ: Tumor hypoxia,
the physiological link between Trousseau's syndrome
(carcinoma-induced coagulopathy) and metastasis. Cancer Res.
61:795–798. 2001.PubMed/NCBI
|
|
9
|
Emami Nejad A, Najafgholian S, Rostami A,
Sistani A, Shojaeifar S, Esparvarinha M, Nedaeinia R, Haghjooy
Javanmard S, Taherian M, Ahmadlou M, et al: The role of hypoxia in
the tumor microenvironment and development of cancer stem cell: A
novel approach to developing treatment. Cancer Cell Int. 21:622021.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Liang W and Chen X, Zhang S, Fang J, Chen
M, Xu Y and Chen X: Mesenchymal stem cells as a double-edged sword
in tumor growth: Focusing on MSC-derived cytokines. Cell Mol Biol
Lett. 26:32021. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Aravindhan S, Ejam SS, Lafta MH, Markov A,
Yumashev AV and Ahmadi M: Mesenchymal stem cells and cancer
therapy: Insights into targeting the tumour vasculature. Cancer
Cell Int. 21:1582021. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Andonegui-Elguera MA, Alfaro-Mora Y,
Cáceres-Gutiérrez R, Caro-Sánchez CHS, Herrera LA and Díaz-Chávez
J: An overview of vasculogenic mimicry in breast cancer. Front
Oncol. 10:2202020. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Yao J, Sun L, Gao F and Zhu W: Mesenchymal
stem/stromal cells: Dedicator to maintain tumor homeostasis. Hum
Cell. 38:212024. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
den Hollander P, Maddela JJ and Mani SA:
Spatial and temporal relationship between epithelial-mesenchymal
transition (EMT) and stem cells in cancer. Clin Chem. 70:190–205.
2024. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Hass R: Role of MSC in the tumor
microenvironment. Cancers (Basel). 12:21072020. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Hill BS, Pelagalli A, Passaro N and
Zannetti A: Tumor-educated mesenchymal stem cells promote
pro-metastatic phenotype. Oncotarget. 8:73296–73311. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Fasano M, Pirozzi M, Miceli CC, Cocule M,
Caraglia M, Boccellino M, Vitale P, De Falco V, Farese S, Zotta A,
et al: TGF-β modulated pathways in colorectal cancer: New potential
therapeutic opportunities. Int J Mol Sci. 25:74002024. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Sritharan S and Sivalingam N: Secretion of
IL-6 and TGF-β2 by colon cancer cells may promote resistance to
chemotherapy. Ind J Clin Biochem. 2024. View Article : Google Scholar
|
|
19
|
Dumont N and Arteaga CL: Transforming
growth factor-beta and breast cancer: Tumor promoting effects of
transforming growth factor-beta. Breast Cancer Res. 2:125–132.
2000. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Huang K, Han Y, Chen Y, Shen H, Zeng S and
Cai C: Tumor metabolic regulators: Key drivers of metabolic
reprogramming and the promising targets in cancer therapy. Mol
Cancer. 24:72025. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Zhang H, Li S, Wang D, Liu S, Xiao T, Gu
W, Yang H, Wang H, Yang M and Chen P: Metabolic reprogramming and
immune evasion: The interplay in the tumor microenvironment.
Biomark Res. 12:962024. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Paul D and Nedelcu AM: The underexplored
links between cancer and the internal body climate: Implications
for cancer prevention and treatment. Front Oncol. 12:10400342022.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Slominski RM, Raman C, Chen JY and
Slominski AT: How cancer hijacks the body's homeostasis through the
neuroendocrine system. Trends Neurosci. 46:263–275. 2023.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Roy ME, Veilleux C and Annabi B: In vitro
biomaterial priming of human mesenchymal stromal/stem cells:
Implication of the Src/JAK/STAT3 pathway in vasculogenic mimicry.
Sci Rep. 14:214442024. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Schneider CA, Rasband WS and Eliceiri KW:
NIH image to ImageJ: 25 Years of image analysis. Nat Methods.
9:671–675. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(−Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Roy ME, Veilleux C, Paquin A, Gagnon A and
Annabi B: Transcriptional regulation of CYR61 and CTGF by LM98: A
synthetic YAP-TEAD inhibitor that targets in-vitro vasculogenic
mimicry in glioblastoma cells. Anticancer Drugs. 35:709–719. 2024.
View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Pratt J, Haidara K and Annabi B: MT1-MMP
expression levels and catalytic functions dictate LDL
receptor-related protein-1 ligand internalization capacity in U87
glioblastoma cells. Int J Mol Sci. 23:142142022. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Holmgaard RB, Schaer DA, Li Y, Castaneda
SP, Murphy MY, Xu X, Inigo I, Dobkin J, Manro JR, Iversen PW, et
al: Targeting the TGFβ pathway with galunisertib, a TGFβRI small
molecule inhibitor, promotes anti-tumor immunity leading to
durable, complete responses, as monotherapy and in combination with
checkpoint blockade. J Immunother Cancer. 6:472018. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Sicard AA, Suarez NG, Cappadocia L and
Annabi B: Functional targeting of the TGF-βR1 kinase domain and
downstream signaling: A role for the galloyl moiety of green
tea-derived catechins in ES-2 ovarian clear cell carcinoma. J Nutr
Biochem. 87:1085182021. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Djediai S, Gonzalez Suarez N, El
Cheikh-Hussein L, Rodriguez Torres S, Gresseau L, Dhayne S,
Joly-Lopez Z and Annabi B: MT1-MMP cooperates with TGF-β
receptor-mediated signaling to trigger SNAIL and Induce
epithelial-to-mesenchymal-like transition in U87 glioblastoma
cells. Int J Mol Sci. 22:130062021. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Antoon R, Overdevest N, Saleh AH and
Keating A: Mesenchymal stromal cells as cancer promoters. Oncogene.
43:3545–3555. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Liu S, Ren J, Hu Y, Zhou F and Zhang L:
TGFβ family signaling in human stem cell self-renewal and
differentiation. Cell Regen. 13:262024. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Wang J, Peng J, Chen Y, Nasser MI and Qin
H: The role of stromal cells in epithelial-mesenchymal plasticity
and its therapeutic potential. Discov Oncol. 15:132024. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Novoseletskaya ES, Evdokimov PV and
Efimenko AY: Extracellular matrix-induced signaling pathways in
mesenchymal stem/stromal cells. Cell Commun Signal. 21:2442023.
View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Nam D, Park A, Dubon MJ, Yu J, Kim W, Son
Y and Park KS: Coordinated regulation of mesenchymal stem cell
migration by various chemotactic stimuli. Int J Mol Sci.
21:85612020. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Cottler-Fox MH, Lapidot T, Petit I, Kollet
O, DiPersio JF, Link D and Devine S: Stem cell mobilization.
Hematology Am Soc Hematol Educ Program. 419–437. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Ridge SM, Sullivan FJ and Glynn SA:
Mesenchymal stem cells: Key players in cancer progression. Mol
Cancer. 16:312017. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Annabi B, Naud E, Lee YT, Eliopoulos N and
Galipeau J: Vascular progenitors derived from murine bone marrow
stromal cells are regulated by fibroblast growth factor and are
avidly recruited by vascularizing tumors. J Cell Biochem.
91:1146–1158. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Frisbie L, Buckanovich RJ and Coffman L:
Carcinoma-associated mesenchymal stem/stromal cells: Architects of
the pro-tumorigenic tumor microenvironment. Stem Cells. 40:705–715.
2022. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Zhang J, Qiao L, Liang N, Xie J, Luo H,
Deng G and Zhang J: Vasculogenic mimicry and tumor metastasis. J
BUON. 21:533–541. 2016.PubMed/NCBI
|
|
42
|
Zonneville J, Safina A, Truskinovsky AM,
Arteaga CL and Bakin AV: TGF-β signaling promotes tumor vasculature
by enhancing the pericyte-endothelium association. BMC Cancer.
18:6702018. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Yoshimura A and Muto G: TGF-β function in
immune suppression. Curr Top Microbiol Immunol. 350:127–147.
2011.PubMed/NCBI
|
|
44
|
Konkel JE, Zhang D, Zanvit P, Chia C,
Zangarle-Murray T, Jin W, Wang S and Chen W: Transforming growth
factor-β signaling in regulatory T cells controls T helper-17 cells
and tissue-specific immune responses. Immunity. 46:660–674. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Guo C, Sun H, Du Y, Dai X, Pang Y, Han Z,
Xiong X, Li S, Zhang J, Zheng Q and Gui X: Specifically blocking
αvβ8-mediated TGF-β signaling to reverse immunosuppression by
modulating macrophage polarization. J Exp Clin Cancer Res.
44:12025. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Itoh F, Itoh S, Adachi T, Ichikawa K,
Matsumura Y, Takagi T, Festing M, Watanabe T, Weinstein M, Karlsson
S and Kato M: Smad2/Smad3 in endothelium is indispensable for
vascular stability via S1PR1 and N-cadherin expressions. Blood.
119:5320–5628. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Tashima T: Mesenchymal stem cell
(MSC)-based drug delivery into the brain across the blood-brain
barrier. Pharmaceutics. 16:2892024. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Minev T, Balbuena S, Gill JM, Marincola
FM, Kesari S and Lin F: Mesenchymal stem cells-the secret agents of
cancer immunotherapy: Promises, challenges, and surprising twists.
Oncotarget. 15:793–805. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Wu X, Jiang J, Gu Z, Zhang J, Chen Y and
Liu X: Mesenchymal stromal cell therapies: Immunomodulatory
properties and clinical progress. Stem Cell Res Ther. 11:3452020.
View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Lan T, Luo M and Wei X: Mesenchymal
stem/stromal cells in cancer therapy. J Hematol Oncol. 14:1952021.
View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Kim BG, Malek E, Choi SH, Ignatz-Hoover JJ
and Driscoll JJ: Novel therapies emerging in oncology to target the
TGF-β pathway. J Hematol Oncol. 14:552021. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Guo Y, Wang Z, Zhou H, Pan H, Han W, Deng
Y, Li Q, Xue J, Ge X, Wang S, et al: First-in-human study of
GFH018, a small molecule inhibitor of transforming growth factor-β
receptor I inhibitor, in patients with advanced solid tumors. BMC
Cancer. 24:4442024. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Margiana R, Markov A, Zekiy AO, Hamza MU,
Al-Dabbagh KA, Al-Zubaidi SH, Hameed NM, Ahmad I, Sivaraman R, Kzar
HH, et al: Clinical application of mesenchymal stem cell in
regenerative medicine: a narrative review. Stem Cell Res Ther.
13:3662022. View Article : Google Scholar : PubMed/NCBI
|