Introduction
Adenomyoepithelioma (AME) of the breast, which was
initially described by Hamperl in 1970 (1), is a biphasic tumor characterized by
small epithelium-lined spaces with inner luminal ductal cells and a
proliferation of variably enlarged myoepithelial cells. AME is
rare, and one AME case (0.048%) was found in a series of 2078
consecutive breast tumors diagnosed by core needle biopsy, with a
benign clinical course (2,3). In contrast to these observations,
malignant AME (M-AME) is AME with carcinoma features, in which the
malignancy may arise from either luminal epithelial or
myoepithelial components, or from both cell types. M-AME is
extremely rare, and no data on incidence have been reported
(4) and the prognosis depends on
the histological subtype of the invasive diseases (5). The low number of reported cases and
short periods of follow-up limit the available information on
prognostic and genetic features in M-AME. Genetically, breast
cancers display complex somatic mutations and significant molecular
heterogeneity, and TP53, PIK3CA and GATA binding protein 3
(GATA3) are the only three genes recurrently mutated in
>10% of unselected breast cancers (6), while, the genetic drivers of AME
depend on the estrogen receptor (ER). ER-positive AME display
PIK3CA or AKT1 activating mutations, while
ER-negative AME harbor highly recurrent HRAS Q61R hotspot
mutations (5,7). Mitogen-activated protein kinase (MAPK)
and PI3K-AKT pathways are the two major downstream intracellular
pathways of oncogenesis (8). The
MAPK pathway is composed of RAS, RAF, MEK and extracellular
signal-regulated kinase (ERK). RAS contains 3 closely
related proteins encoded by the three following genes: HRAS,
KRAS and NRAS. The activation caused by these gene
mutations leads to uncontrolled cell growth and promotes
oncogenesis (9). The global
incidence of HRAS-mutant tumors is 7% of all RAS-mutant
tumors (10), and the
identification of the HRAS mutation aids in confirming a
diagnosis, and selecting an effective medicine. For example,
HRAS mutations are a frequent tumorigenic gene alteration in
salivary gland epithelial-myoepithelial carcinoma (EMC), which
indicates biphasic tubular structures, usually composed of tightly
coupled inner ductal and prominent outer myoepithelial cells
(11). The HRAS mutation is
useful for diagnosing salivary gland EMC, and discriminates it from
its mimics (11). Moreover, medical
drugs that target HRAS mutant tumors are available (12).
In the present study, a M-AME case with pulmonary
metastasis is reported. Genetical analysis of breast M-AME with
distant metastasis is limited, and this is the first M-AME case
with HRAS Q13R mutation, which was confirmed during the
examination of primary and metastatic lesions. Moreover, 16 cases
of M-AME are reviewed including the present case, and the incidence
of the frequent HRAS G13R mutation in M-AME is
highlighted.
Case report
Case presentation and pathology
A 53-year-old Japanese woman presented to a local
clinic with a palpable and non-tender mass in her left breast in
October 2022. The patient had no other symptoms. The mass was
located at the C region, with a maximum diameter of 2.5 cm. The
core needle biopsy was indicative of AME and malignancy was not
ruled out. Her medical and family history was unremarkable. Total
mastectomy and sentinel lymph node dissection were performed at
Osaka Medical and Pharmaceutical University in November 2022. A
gray mass (2.5×2.0×1.8 cm) was noted. For histological analysis,
the surgical specimens were fixed in 10% buffered formalin for 24 h
at room temperature (RT) and embedded in paraffin. The sections (4
µm-thickness) from the paraffin block were stained with hematoxylin
for 5 min and eosin for 1 min at RT. The stained sections were
examined under a light microscope (BX53; Olympus Corporation).
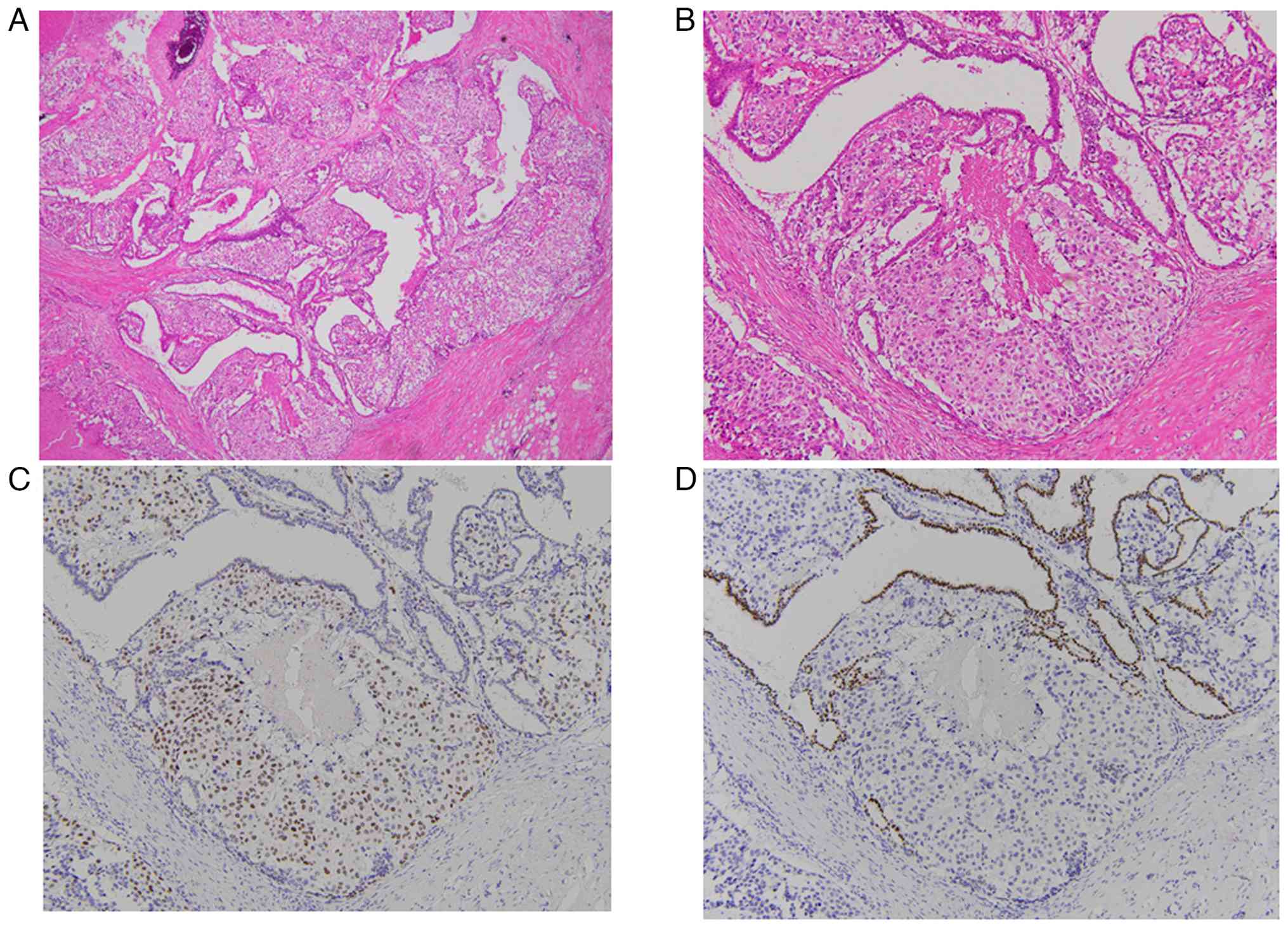
Histologically, clear and epithelioid myoepithelial cells
surrounding the papillary glandular epithelium were noted in
hematoxylin-eosin (H&E) stain (Fig.
1A and B). Myoepithelial cells exhibited mitoses (12/10 high
power fields) and moderate or severe atypia. Necrosis was also
present. Metaplastic change was not observed. Sentinel lymph nodes
indicated lack of metastasis. For subsequent analysis,
immunohistochemistry (IHC) was carried out. Briefly, 4 µm-thick
sections obtained from the paraffin block were stained with primary
antibodies. For IHC, automated immunostaining devices, VENTANA
BenchMark ULTRA (Roche Diagnostics) was used. After
deparaffinization with EZ buffer (Roche Diagnostics), blocking
endogenous peroxidases and antigen retrievals, the used antibodies
were p63 (4A4, cat. no. 518-101961), S100 (polyclonal, cat. no.
518-110109), GATA3 (L50-823, cat no. 518-111953), thyroid
transcription factor-1 (TTF-1, SP141, cat. no. 518-110871),
estrogen receptor (ER, SP1, cat. no. 518-107932), progesterone
receptor (PgR, 1E2, cat. no. 518-102463) and HER2(4B5, cat no.
790-2991). The stained sections were observed under a light
microscope (BX53). IHC revealed that myoepithelial cells were
positive for p63 (Fig. 1C) and
S-100, and epithelial cells were positive for GATA3 (Fig. 1D) and negative for TTF-1. From these
findings, malignant adenomyoepithelioma (T2N0M0) was diagnosed.
Both epithelial and myoepithelial cells were negative for ER, PgR
and HER2. After the operation, the administration of epirubicin
combined with cyclophosphamide followed by docetaxel was performed
with the patient's consent. There were no adverse and unanticipated
events.
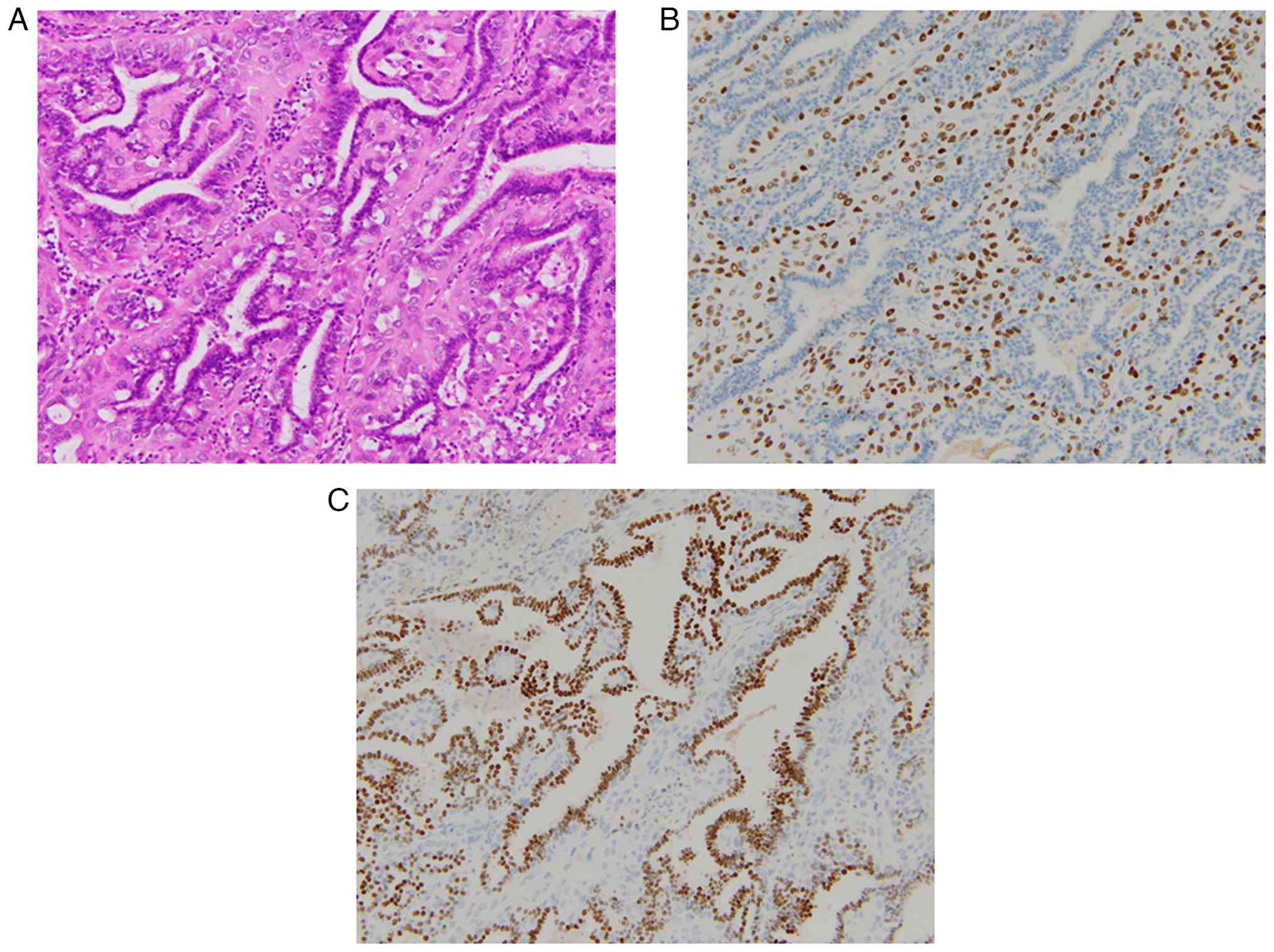
Twenty-six months later, chest computed tomography
indicated a mass in the right lung (S6), and segmentectomy of the
lung was performed. The tumor cells were 1.5 cm with a clear
margin, and histologically, myoepithelial cells had proliferated
along with bronchiolar epithelium (Fig.
2A). Myoepithelial cells with clear or epithelioid features
were positive for p63 (Fig. 2B) and
S-100, and epithelial cells were positive for TTF-1 (Fig. 2C); a low number of epithelial cells
was positive for GATA3. From these findings, pulmonary metastasis
from breast tumor was diagnosed. The patient was seen in December
2025, and complained of occasional dizziness, but exhibited no
evidence of recurrence on computed tomography. Follow-up visits are
scheduled every three months.
Molecular genetic analysis
To investigate whether the mutational status of
HRAS in breast and lung is the same, a polymerase chain
reaction (PCR) followed by Sanger's sequencing was performed
(11,13). Briefly, DNA from formalin-fixed
paraffin-embedded tissue was extracted with a QIAamp DNA FFPE
Tissue Kit (Qiagen, Inc.). The tumor component of the slides was
microdissected to increase the tumor cell ratio. PCR products were
purified using a QIAquick Spin Kit (Qiagen, Inc.). Each purified
product was directly sequenced using a forward primer with a BigDye
Terminator version 3.1 cycle sequencing kit on an ABI 3730
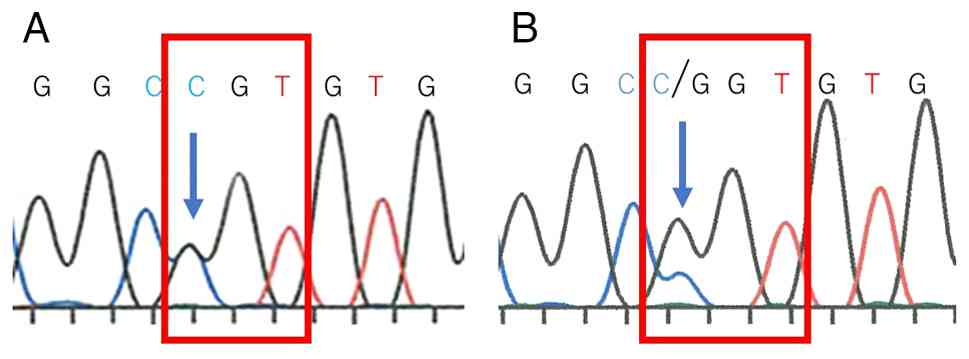
instrument (Thermo Fisher Scientific, Inc.). A mutation analysis of
HRAS (exons 2 and 3) was performed, and the primer sequences
are listed in Table I. The present
case harbored HRAS G13R mutation in breast and lung tissues
(Fig. 3).
 | Table I.PCR primers used for Sanger
sequencing. |
Table I.
PCR primers used for Sanger
sequencing.
| Gene | Direction | Sequence (5′-3′) |
|---|
| HRAS exon
2 | Forward |
CAGGCCCCTGAGGAGCGATG |
|
| Reverse |
TTCGTCCACAAAATGGTTCT |
| HRAS exon
3 | Forward |
TCCTGCAGGATTCCTACCGG |
|
| Reverse |
GGTTCACCTGTACTGGTGGA |
Literature review
Literature analysis was performed using PubMed
database (https://pubmed.ncbi.nlm.nih.gov/) using the key words
‘adenomyoepithelioma’ and ‘breast’. Within these terms, M-AME with
genetic analyses and clinical findings was selected. In total, 16
M-AME including the case reported in the present study were
selected (14–16). The clinical and pathological
characteristics of these cases are detailed in Table II. All patients were female, and
the median patient age at diagnosis was 66 years (range, 42–84
years). Metastasis to a lymph node and lung was seen in 3 and 2
cases, respectively. Two cases indicated recurrence. A total of 3
cases harbored mutations in both HRAS and PIK3CA
genes, and within the HRAS mutation, G13R, Q61K, G12D and
G12S were noted in 4 (25%), 1 (6%), 1 (6%) and 1 case (6%),
respectively. ER-negative HRAS G13R and PIK3CA H1047R
were noted in two cases.
 | Table II.Cases of malignant adenomyoepithelioma
with genetical analyses. |
Table II.
Cases of malignant adenomyoepithelioma
with genetical analyses.
| First author/s,
year | Case | Age, years | Size, cm | Follow-up,
months | Lymph node
metastasis | Metastasis | Architecture | Myoepithelial
cells | Mitosis | Necrosis | ER | HRAS | PIK3CA | AKT1 | APC | ATM | (Refs.) |
|---|
| Lubin et al,
2019 | 1 | 69 | 0.7 | 85 | NA | No | Papillary | Clear,
epithelioid | 12/10 HPF | NA | (+) | NA | NA | E17K | NA | NA | (14) |
|
| 2 | 55 | 1.4 | 8 | NA | No | Tubular | Epithelioid | 15/10 HPF | (+) | (−) | NA | NA | E17K | NA | F858L |
|
|
| 3 | 73 | 2.5 | NA | NA | NA | Tubular | Clear | 5/10 HPF | (+) | (−) | NA | NA | NA | P870S, A1564P | NA |
|
|
| 4 | 67 | 1.0 | NA | NA | NA | Lobulated | Clear | 10/10 HPF | NA | (+) | NA | NA | NA | E1317Q | NA |
|
|
| 5 | 78 | 1.5 | NA | NA | NA | Tubular | Clear | 16/10 HPF | (+) | (+) | NA | H1047R | NA | NA | NA |
|
|
| 6 | 78 | 1.6 | NA | NA | NA | Tubular | Clear | 4/10 HPF | NA | (−) | NA | NA | NA | NA | NA |
|
|
| 7 | 65 | 1.0 | 12 | NA | Chest wall | Spindle | Spindle | 10/10 HPF | NA | (−) | Q61K | H1047R, H1065L | NA | NA | NA |
|
| Ginter et
al, 2020 | 8 | 78 | 1.4 | 23 | NA | No | Papillary,
lobulated | Epithelioid,
clear | 12/10 HPF | NA | (−) | NA | H1047P | NA | NA | NA | (15) |
|
| 9 | 66 | 1.2 | 37 | (+) | No | Lobulated,
spindled | Spindled,
clear | 5/10 HPF | NA | (+) | NA | NA | E17K | NA | NA |
|
|
| 10 | 42 | 4.8 | 16 | (−) | No | Lobulated,
tubular | Clear | 20/10 HPF | NA | (−) | NA | NA | NA | NA | NA |
|
|
| 11 | 56 | 2.2 | 24 | (+) | DOD | Papillary | Epithelioid,
spindle, clear | 13/10 HPF | NA | (−) | G12D | NA | NA | NA | NA |
|
| Bièche et
al, 2021 | 12 | 84 | 2.5 | 12 | NA | Recurrence | Tubular,
lobulated | Clear |
3/mm2 | (+) | (+) | G13R | wt | wt | NA | NA | (16) |
|
| 13 | 76 | 1.8 | NA | NA | NA | Tubular,
lobulated | Clear |
3/mm2 | (+) | (−) | G13R | H1047R | wt | NA | NA |
|
|
| 14 | 60 | 1.9 | 75 | NA | No | Tubular, spindle,
cystic | Clear, spindle |
6/mm2 | (+) | (−) | G13R | H1047R | wt | NA | NA |
|
|
| 15 | 55 | 5.5 | 11 | (+) | Lung | Tubular | Clear |
10/mm2 | (−) | (+) | G12S | wt | wt | NA | NA |
|
| Present study | 16 | 53 | 2.5 | 31 | (−) | Lung | Papillary | Clear,
epithelioid | 12/10 HPF | (+) | (−) | G13R | ND | ND | ND | ND | - |
Discussion
M-AME is a rare disease, and its clinical course is
not fully clarified. Ahmed and Heller (17) indicated that the hematogenous spread
seems to be more frequent than the lymphatic spread, with the lung
and brain metastasis. The tumor diameter with distant metastasis
was 2 cm or larger, as noted in the present case. Genetic analysis
of matched classic and malignant components of M-AME has suggested
that it derives from malignant transformation of a pre-existing
classic AME (7). Malignancy of
M-AME arise from either luminal epithelial or myoepithelial
components, or from both cell types (5). In the present case, myoepithelial
cells revealed cytological atypia and mitoses, and the pulmonary
metastatic lesion indicated myoepithelial proliferation.
Myoepithelial cells seemed to be a malignant component. Although
genetic data on M-AME are limited, and PIK3CA and
HRAS Q61R hotspot mutations are noted in both AME and M-AME
(5,7). HRAS mutations are rare in
common-type breast cancers (6). The
majority of the invasive breast cancers arising in AME display a
triple-negative phenotype (ER-, PgR-, HER2-negative) (5), and the HRAS Q61R hotspot
mutation of AME and M-AME, which is related to ER-negative cases
(7). In vitro analyses
demonstrated that forced expression of mutant HRAS Q61R on
non-malignant and ER-negative breast epithelial cells, resulted in
high grade malignancy (increased proliferation and migration), and
oncogenic properties and acquisition of a myoepithelial-like
phenotype (7). HRAS
mutations are a tumorigenic gene alteration noted in salivary gland
EMC (11). AME of the breast is
identical in histological and immunohistochemical structure to EMC
of the salivary gland (18), and
the most frequent HRAS mutation in the salivary gland EMC is
Q61R(82.1%), as noted in breast AME. Other mutations noted were the
following: G13R(9.0%), Q61K(7.5%) and Q61(1.5%). Among salivary
gland tumors, HRAS mutation is 100% specific to EMC and not
to any other histological type, and the mutation is useful for the
diagnosis of salivary gland EMC (11). In addition, in breast tissues,
HRAS mutation may be useful in the diagnosis of AME and
M-AME.
The present review indicated that the HRAS
G13R was most frequently noted (4 cases, 25%) in M-AME, and 2 cases
harbored mutations in both HRAS G13R and PIK3CA
H1047R. Although HRAS Q61 mutation was seen in 4 M-AME cases
without clinical findings, the HRAS G13R mutation was not
seen in AME (7). For distinguishing
whether AME is benign or malignant, H&E stain is the most
important. In the present case, frequent mitoses, cytological
atypia and necrosis of breast myoepithelial cells were recognized,
and the diagnosis of M-AME was made. In addition to H&E stain,
HRAS G13R mutation may be an adjunctive indicator of
malignancy. Further case accumulations of M-AME are required.
HRAS mutation causes activation of the RAS-MAPK pathway, and
MEK is a downstream effector of HRAS in the MAPK
pathway. A recent study has shown that tumors with HRAS G13R
mutations may be responsive to MEK inhibitors such as trametinib.
In metastasis-derived breast M-AME xenografts with HRAS
mutations (HRAS G13R or G125), the trametinib treatment
resulted in a marked anti-tumor activity (16). Agminated Spitz nevus with
HRAS G13R indicated optimal response to trametinib (19). From these findings, it can be
deduced that the HRAS protein may be a potential target, and
the genotypic analysis is important for the treatment selection of
M-AME.
The presented patient is followed up regularly,
without medication, and exhibits no evidence of disease one year
following the lung operation. The patient underwent total
mastectomy, sentinel lymph node dissection and adjuvant
chemotherapy, but lung metastasis was seen about two years later.
Although most M-AME are cured with wide excision with negative
margins, local recurrence or hematogenous metastasis is
demonstrated (17). The 5-year
overall survival of M-AME is as high as 87.5% during median
follow-up of 55 months, and it has a relatively good prognosis
(20), but distant metastasis and
local recurrence tend to occur 4 months to 2–3 years after the
first diagnosis (21), as seen in
the present case. From these, the early detection of recurrence
seems to improve the prognosis, and regular scanning using computed
tomography and tumor marker measurements are planned in the current
case. M-AME has a high rate of triple negative subtype (5), as seen in the presented case, and
endocrine and anti-HER2 therapy is not indicated. Although there is
a little objective evidence of radiotherapy or conventional
chemotherapy (20), MEK inhibitor
may be a candidate in case of recurrence. Genotypic analysis of
M-AME is useful for making the diagnosis and selecting a
medicine.
In conclusion, a case of HRAS G13R mutated
M-AME with lung metastasis was reported. Review analyses indicated
that HRAS G13R was the popular mutation of M-AME, which may
be considered a driver mutation in M-AME.
Acknowledgements
Not applicable.
Funding
Funding: No funding was received.
Availability of data and materials
The data generated in the present study may be
requested from the corresponding author.
Authors' contributions
HK and RT interpreted and analyzed the data, and
drafted the manuscript. RT, YU, TN, YH and KK acquired the data and
revised the manuscript. MI designed the study, conducted the
treatment and revised the manuscript. MI and TN confirm the
authenticity of all the raw data. All authors read and approved the
final version of the manuscript.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
The patient provided written informed consent for
the publication of their data and images.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Hamperl H: The myothelia (myoepithelial
cells). Normal state; regressive changes; hyperplasia; tumors. Curr
Top Pathol. 53:161–220. 1970. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Cheung KL, Wong AWS, Parker H, Li VWY,
Winterbottom L, Morgan DAL and Ellis IO: Pathological features of
primary breast cancer in the elderly based on needle core
biopsies-a large series from a single centre. Crit Rev Oncol
Hematol. 67:263–267. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Foschini MP, Geyer FC, Hayes MM, Marchio C
and Nishimura R: Adenomyoepithelioma. In: WHO classification of
tumours. Breast tumours. 5th edition. IARC; Lyon: pp. 43–45.
2018
|
|
4
|
Ali RH and Hayes MM: Combined
epithelial-myoepithelial lesions of the breast. Surg Pathol Clin.
5:661–699. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Foschini MP, Geyer FC, Hayes MM, Marchio C
and Nishimura R: Malignant adenomyoepithelioma. In: WHO
classification of tumours. Breast tumours. 5th edition. IARC; Lyon:
pp. 46–48. 2018
|
|
6
|
The Cancer Genome Atlas Network, .
Comprehensive molecular portraits of human breast tumours. Nature.
490:61–70. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Geyer FC, Li A, Papanastasiou AD, Smith A,
Selenica P, Burke KA, Edelweiss M, Wen HC, Pisculoglio S,
Schultheis AM, et al: Recurrent hotspot mutations in HRAS Q61 and
PI3K-AKT pathway genes as drivers of breast adenomyoepitheliomas.
Nature Commun. 9:18162018. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Ciardiello F and Tortora G: EGFR
antagonists in cancer treatment. N Engl J Med. 358:1160–1174. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Dhillon AS, Hagan S, Rath O and Kolch W:
MAP kinase signalling pathways in cancer. Oncogene. 26:3279–3290.
2007. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Prior IA, Hood FE and Hartley JL: The
frequency of Ras mutations in cancer. Cancer Res. 80:2969–2974.
2020. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Urano M, Nakaguro M, Yamamoto Y, Hirai H,
Tanigawa M, Saigusa N, Shimizu A, Tsukahara K, Tada Y, Sakurai K,
et al: Diagnostic significance of HRAS mutations in
epithelial-myoepithelial carcinomas exhibiting a broad
histopathologic spectrum. Am J Surg Pathol. 43:984–994. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Nigam A, Krishnamoorthy GP, Chatila WK,
Berman K, Saqcena M, Walch H, Venkatramani M, Ho AL, Schltz N,
Fagin JA and Untch BR: Cooperative genomic lesions in HRAS-mutant
cancers predict resistance to farnesyltransferase inhibitors.
Oncogene. 43:2806–2819. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Shimura T, Tada Y, Hirai H, Kawakita D,
Kano S, Tsukahara K, Shimizu A, Takase S, Imanishi Y, Ozawa H, et
al: Prognostic and histogenetic roles of gene alteration and the
expression of key potentially actionable targets in salivary duct
carcinomas. Oncotarget. 9:1852–1867. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Lubin D, Toorens E, Zhang PJ, Jaffer S,
Baraban E, Bleiweiss IJ and Nayak A: Adenomyoepitheliomas of the
breast frequently harbor recurrent hotspot mutations in PIK3-AKT
pathway-related genes and a subset show genetic similarity to
salivary gland epithelial-myoepithelial carcinoma. Am J Surg
Pathol. 43:1005–1013. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Ginter PS, Mcintire PJ, Kurtis B,
Mirabelli S, Motanagh S, Hoda S, Elemento O, Shin SJ and Mosquera
JM: Adenomyoepithelial tumors of the breast: Molecular
underpinnings of a rare entity. Mod Pathol. 33:1764–1772. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Bièche I, Coussy F, El-Botty R, Vacher S,
Château-Joubert S, Dahmani A, Montaudon E, Reyes C, Gentien D,
Reyal F, et al: HRAS is a therapeutic target in malignant
chemo-resistant adenomyoepithelioma of the breast. J Hematol Oncol.
14:1432021. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Ahmed AA and Heller DS: Malignant
adenomyoepithelioma of the breast with malignant proliferation of
epithelial and myoepithelial elements. A case report and review of
the literature. Arch Pathol Lab Med. 124:632–636. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Seifert G: Are adenomyoepithelioma of the
breast and epithelial-myoepithelial carcinoma of the salivary
glands identical tumours? Virchow Arch. 433:285–287. 1998.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Parsons L, Chen A, Bertus B, Beatty C,
Stashower J, Tomboc P, Brooke S and Zinn Z: Topical trametinib for
agminated spitz nevi harboring HRAS mutation. Pediatric Dermatol.
42:841–843. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Alqudaihi HMA, Lee SB, Son BH, Ahn SH, Lee
JW, Ko BS, Kim HJ, Chung IY, Kim J and Gog G: Clinicopathological
characteristics and outcomes of malignant adenomyoepithelioma of
the breast: A single institution's experience. World J Surg Oncol.
20:1282022. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Bult P, Verwiel JM, Wobbes T, Kooy-Smits
MM, Biert J and Holland R: Malignant adenomyoepithelioma of the
breast with metastasis in the thyroid gland 12 years after excision
of the primary tumor. Case report and review of the literature.
Virchow Arch. 436:158–166. 2000. View Article : Google Scholar : PubMed/NCBI
|