Introduction
Cancer remains a major global health concern and a
leading cause of mortality worldwide. It is characterized by
uncontrolled cell proliferation, driven by cancer cells that have
developed novel mechanisms to regulate their own growth and
differentiation (1). Tumor
development and metastasis are sustained by complex interactions
within the tumor microenvironment (TME), involving both
cancer-cell-intrinsic factors and stromal components. Therefore,
elucidation of these intercellular communication networks is
essential for understanding tumor biology (2).
Extracellular vesicles (EVs) are now recognized as
crucial mediators of cell-to-cell communication in cancer.
Substantial evidence demonstrates that EVs are secreted by
virtually all cells within the TME and facilitate the transfer of
various biological macromolecules. As specialized carriers of
intercellular signals, EVs contribute to critical oncogenic
processes such as cell proliferation, metastasis and
epithelial-mesenchymal transition. Given their multifaceted roles
in the modulation of tumor behavior, EVs show great promise as
clinical biomarkers and novel platforms for cancer therapeutics,
paving the way for improved diagnostic and treatment strategies
(3,4).
Previous research on EVs has largely utilized
two-dimensional (2D) monolayer cultures, with EVs derived from
three-dimensional (3D) models remaining relatively underexplored.
Organoids, as in vitro 3D culture systems, recapitulate the
complex architecture and biological processes of in vivo
tumors. This makes them a powerful tool for cancer research and
provides a novel option for clinical studies in tumor biology
(5,6). The complexity of the TME is fueling
growing interest in organoid-derived EVs. These models hold
considerable potential for simulating tumor-specific intercellular
communication and warrant further investigation (7,8).
Research into the clinical applications and challenges of
integrating organoids with EVs is expected to contribute to the
development of tumor biobanks, novel therapeutic strategies and
precision oncology.
In the present review, the biogenesis and isolation
methods of EVs are delineated, and their advantages and functions
in tumor research are described. In addition, the potential of
organoids as promising models for investigating the TME is
discussed. Furthermore, recent advances in EVs derived from tissue
organoids are summarized and the prospects and challenges of
integrating organoid and EVs models for cancer therapy are
discussed.
EVs
Origin and classification of EVs
EVs have been extensively studied over the past few
decades, but their therapeutic applications have emerged more
recently. Understanding the history of EV discovery is essential
for appreciating their biological importance and for guiding the
development of new therapeutic strategies. The discovery of EVs
dates back to 1946, when Chargaff and West (9) identified platelet-derived particles,
termed ‘platelet dust’, in the blood. This was followed by the
identification of cell-derived vesicles in mouse cartilage by
Anderson in 1969 (10). In 1996,
Raposo et al (11)
demonstrated that B-cell-derived exosomes contain MHC class II
molecules and can directly induce T-cell responses. Subsequent
studies revealed that T cells release EVs carrying bioactive
ligands, such as Fas and TNF-related apoptosis-inducing ligand,
indicating a crucial role in immune regulation. EVs perform diverse
immune functions by interacting with recipient cells via their
surface proteins or by delivering bioactive cargo, thereby altering
the functional properties of the recipient cells (12). Unlike classical signaling mechanisms
based on direct cell contact or secreted factors, EVs provide a
distinct mode of intercellular communication, with the ability to
transfer proteins and RNA to directly modulate cellular functions
(13). These findings have
highlighted the crucial importance of EVs in cell signaling and
suggested new strategies for disease diagnosis and treatment.
EVs are phospholipid bilayer-enclosed particles,
which are released by diverse cell types under both physiological
and pathological conditions and have been detected in tissues and
bodily fluids, including serum, cerebrospinal fluid, saliva and
urine (14). Cells package
signaling molecules, including proteins, lipids and nucleic acids,
into EVs. This packaging protects the molecules from degradation
and enables them to evade immune surveillance, thereby facilitating
local and long-distance intercellular communication (15).
Recent advances in EV isolation and characterization
have led to the discovery of a growing number of EV subtypes. Most
mammalian cells, which typically measure 10–100 µm in diameter,
release a heterogeneous population of EVs, along with non-vesicular
extracellular nanoparticles (NPs). The overlapping size ranges of
EVs and NPs makes it challenging to differentiate between them.
Well-characterized EVs types include exosomes, which are generated
via the endocytic pathway and are released into the extracellular
space when multivesicular endosomes fuse with the plasma membrane;
microvesicles, which form through the direct outward budding and
fission of the plasma membrane; and apoptotic bodies, which are
released from cells during programmed cell death, specifically
apoptosis (16). Among these, the
term exosome is the most widely used and studied. However, due to a
lack of well-defined, category-specific markers, exosomes and
microvesicles can typically only be definitively distinguished by
confirmation of their distinct biogenesis pathways, often using
techniques such as cryo-transmission electron microscopy (17).
EVs are classified into six main types based on
their biogenesis, size and molecular markers. Exosomes, 30–150 nm
in diameter, are formed within multivesicular vesicles (MVBs)
through the endosomal pathway and are released upon MVB fusion with
the plasma membrane. The main molecular markers for exosomes are
tetratrans proteins (such as CD9, CD63 and CD81), tumor
susceptibility gene 101 protein, alix and heat shock protein
(HSP)70/90 (18). Microvesicles,
100–1,000 nm in diameter, directly bud from the plasma membrane by
outward blebbing, which is dependent on cytoskeletal rearrangements
and calcium influx. The main molecular markers for microvesicles
are annexin A1 and A2 (19).
Apoptotic body, 500–4,000 nm in diameter, are released during
apoptosis as fragmented portions of the dying cell. The main
molecular markers for apoptotic bodies are annexin V,
phosphatidylserine and caspases (20). Migrasomes, 0.5–3.0 µm in diameter,
are a subtype of EVs that are released from the elongated
membranous structure at the tail of cells during migration. First
reported in 2015, they were named due to their close association
with cell migration. The hallmark proteins of migrasomes are
tetraspanin-4 and −7, which may help cells eliminate damaged
organelles such as mitochondria, promoting the release of cellular
contents (including protein, mRNA and miRNA) for absorption by
recipient cells (21). Large
oncosomes are atypically large EVs (1–10 µm in diameter) that arise
from non-apoptotic membrane blebbing. This process can be triggered
through silencing of diaphanous-related formin-3 and overexpression
of oncoproteins such as protein kinase B, heparin binding-epidermal
growth factor and caveolin-1 (22).
Compared with the large EVs, the small ectosomes (≤100 nm) has a
notably higher percentage of CD9- and CD81-positive particles
(23). A summary of the detailed
types and classifications of EVs is provided in Table I (18–23).
 | Table I.Classification of EVs. |
Table I.
Classification of EVs.
| Type | Category of
EVs | Size, nm | Biogenesis | Molecular
markers | (Refs.) |
|---|
| Exosomes | Small | 30-150 | Multivesicular
endosomes | Tetraspanin protein
family: CD63, CD81 and CD9; ESCRT-related proteins: Alix and
TSG101; HSPs: HSP70 and HSP90 | (18) |
| Microvesicles | Medium/large | 100-1,000 | Ectosomes | Annexin A1 and A2,
and α-actinin 4 | (19) |
| Apoptotic
bodies | Large | 500-5,000 | Apoptosis | Annexin V,
phosphatidylserine, caspases | (20) |
| Migrasomes | Large | 500-3,000 | Retraction
fibers | TSPAN4 and
TSPAN7 | (21) |
| Large
oncosomes | Large | 1,000-10,000 | Ectosomes | ARF6, V-ATPase G1,
CK18 and annexin A1 | (22) |
| Small
ectosomes | Small | 30-150 | Ectosomes | CD147 and CD9 | (23) |
Separation of EVs
EVs are secreted by cells into their surrounding
environment and exist as a variety of types, including exosomes,
microvesicles and apoptotic bodies. As different types of EVs
perform distinct biological functions, their study is inherently
dependent on the isolation and purification of specific EVs
subpopulations. This section summarizes the principles, advantages
and disadvantages of various EVs isolation methods (Table II) (24–35).
 | Table II.EVs separation methods. |
Table II.
EVs separation methods.
| Separation
method | Advantages | Disadvantages | (Refs.) |
|---|
| According to EV
size |
|
|
|
|
Ultracentrifugation | Density gradient
differences | Induce EV
aggregation and morphological alterations, potentially modifying
their composition and phenotype | (24,25) |
| Size
exclusion chromatography | Suitable for
removing protein aggregates and lipoprotein particles | Limited recovery
rate, typically reducing the total particle number by approximately
half | (26) |
|
High-performance liquid
chromatography | Maintains the
integrity and biological activity of exosomes; high sample purity
with low co-precipitation | Limited resolution
and sample capacity, and inability to completely remove specific
contaminants | (27) |
| Protein
organic solvent precipitation | Novel and
inexpensive method of rapidly isolating EVs from small volumes of
human blood plasma | Reduces cell
viability in vitro | (28) |
| According to EVs
surface protein markers |
|
|
|
|
Tangential flow
filtration | High-throughput
filtration method for the reliable and specific separation of
exosomes in biological fluids | Isolated EVs may be
contaminated with proteins and lipid droplets; often requires
additional purification steps to achieve high purity | (29,30) |
|
Tangential flow for analyte
capture | Novel method for
isolating micro- and nano-scale species | Membrane adsorption
and sample loss; inherent size-based limitations and restricted
resolution | (31) |
| Heparin
sulfate proteoglycan | Enhances the
purification efficiency of EVs with lower contamination levels | Some proteins in
media and biofluids can bind heparin | (32) |
| According to other
features |
|
|
|
|
Microfluidic technique | Standardized and
rapid method that can be adjusted for EVs from different cell
sources | Limited clinical
applicability due to low isolation throughput | (33) |
| Flow
cytometry | Primary technique
for EV analysis and detection of specific subtypes | Limited
fluorescence sensitivity | (34) |
| Polymer
precipitation method | Simple, robust and
cost-effective method; successfully yields large quantities of EVs
from natural killer cells | Non-specific
co-precipitation | (35) |
The choice of isolation and purification methods
directly determines the yield, purity and physicochemical
properties of the obtained EVs. Consequently, selecting a method
that is efficient, convenient and reliable based on the
characteristics of the target EVs is essential. However, common
isolation methods often fail to completely eliminate soluble
contaminants and non-vesicular particles from cell culture
supernatants or biological fluids. Therefore, it is necessary to
develop novel isolation strategies that offer high specificity,
purity and scalability for large-scale production. Established
methods for EVs isolation can be broadly categorized into several
categories.
Separation according to size
Ultracentrifugation remains the predominant method
for EV isolation, enabling the separation of vesicles based on size
and density, often through sucrose or iodixanol density gradients
(36). However, this technique can
introduce undesirable artifacts, including EV aggregation and
morphological alterations, which may affect vesicular integrity,
composition and biological activity (24,37). A
further limitation is that ultracentrifugation alone may not fully
eliminate contamination from non-vesicular components. To mitigate
these issues, methods combining ultracentrifugation with other
techniques have been developed. For example, combining
ultracentrifugation with cryo-electron microscopy and immunogold
labeling has been shown to preserve EVs diversity and prevent
aggregation (38). Similarly,
ultracentrifugation can be combined with filtration techniques,
including tangential flow filtration (TFF) and microfluidic
filtration, to reduce contaminants and enhance separation
efficiency (39,40).
Several complementary methods are available for EV
isolation, including density gradient centrifugation, size
exclusion chromatography (SEC), high-performance liquid
chromatography and integrated or combination strategies. SEC is
particularly effective at removing protein aggregates and
lipoprotein particles; however, its recovery rate is limited,
typically reducing the total particle number by approximately half
(41,42). Furthermore, SEC is unsuitable for
initial volume reduction during EV extraction, such as in the
isolation of EVs from cell culture supernatants (43). In addition, TFF is a high-throughput
method for the efficient and selective separation of EVs from
biological fluids. It has been used as an early purification step
following the removal of cellular debris via low-speed
centrifugation and filtration through 0.22-µm membranes (44). Another promising method for the
rapid isolation of EVs is protein organic solvent precipitation,
which effectively removes soluble protein contaminants and yields
EVs of higher purity compared with TFF, demonstrating potential for
clinical translation (45).
Separation according to surface
protein markers
The heterogeneity of EVs encompasses subpopulations
characterized by specific surface markers, enabling their selective
isolation. For example, HSPs commonly found on EVs derived from
cancerous or infected cells enable targeted capture. Virucine
peptides have been shown to exhibit high efficiency in the
isolation of HSP-bearing EVs from diverse sources, including cell
culture media, plasma and urine (46). Heparan sulfate proteoglycan, a cell
surface receptor involved in various biological processes, can also
be used to enhance EVs purification. For example, when conditioned
media from 293T cells was mixed with heparin-coated agarose beads
following ultracentrifugation, an EVs recovery rate of 60% was
achieved (47). This method also
reduces contamination while preserving EVs-associated proteins and
other biomarkers (48).
TFF operates on the principle of parallel fluid flow
across a membrane to reduce clogging. This technique is superior to
traditional ultracentrifugation as it effectively maintains both
high separation efficiency and the biological integrity of EVs.
Tangential flow for analyte capture (TFAC) is a novel method built
upon TFF. It enables the selective capture of target exosomes using
functionalized membranes modified with specific antibodies,
integrating both the capture and elution steps into a single
process. This streamlined process minimizes impurities, enables the
rapid processing of small clinical samples and yields high-purity
exosomes. Due to its efficiency, scalability and preservation of EV
bioactivity, TFAC is emerging as a promising method for EVs
isolation (31).
These advancements address challenges in the the
mass production of EVs for clinical translation and help the
transition from qualitative research to translational applications,
thereby opening new avenues for regenerative medicine, drug
delivery and liquid biopsy (44).
Separation according to other
features
Flow cytometry is a key technique for analyzing and
separating EVs. Following initial isolation by methods such as
ultracentrifugation, it enables the quantification of EVs from
various sources using fluorescent markers such as
carboxyfluorescein succinimidyl ester and lipid-specific dyes. This
technique is widely used to identify and characterize specific EVs
subtypes. It employs antibody-fluorophore conjugates that target
specific membrane antigens, allowing the detection and
classification of individual particles based on their fluorescence
characteristics, thereby enabling immunophenotyping and subgroup
classification (41,49).
Immunological methods provide an alternative
strategy for the isolation of EVs, particularly those derived from
antigen-presenting cells (APCs), which release immunomodulatory EVs
characterized by high levels of major histocompatibility complex II
(MHCII) expression. Using immunomagnetic beads, exosomes can be
efficiently enriched from cell-free supernatants, thereby
accelerating the analysis of APC-derived EVs (42). In addition, microfluidic technology
has been developed to capture CD41-positive exosomes on mica-coated
surfaces using specific antibodies. Although this method allows the
rapid and standardized isolation of EVs from various cell types,
its clinical application is limited by its small-scale processing
capacity. Furthermore, given the overlapping physical properties of
EVs and viruses, a CD45-based immunodepletion approach has been
developed to specifically remove HIV particles without affecting
CD45-positive EVs (50). Finally,
polymer precipitation represents a cost-effective and robust
technique for large-scale EV isolation. Although its use in
clinical settings is limited, it has been successfully applied to
obtain substantial quantities of functional EVs from natural killer
(NK) cells in vitro (51).
Advantage of EVs in cancer
research
The type and quantity of EVs reflect the
physiological and pathological states. A large body of research has
focused on exploring the potential benefits and therapeutic
applications of EVs in malignant tumors (Fig. 1).
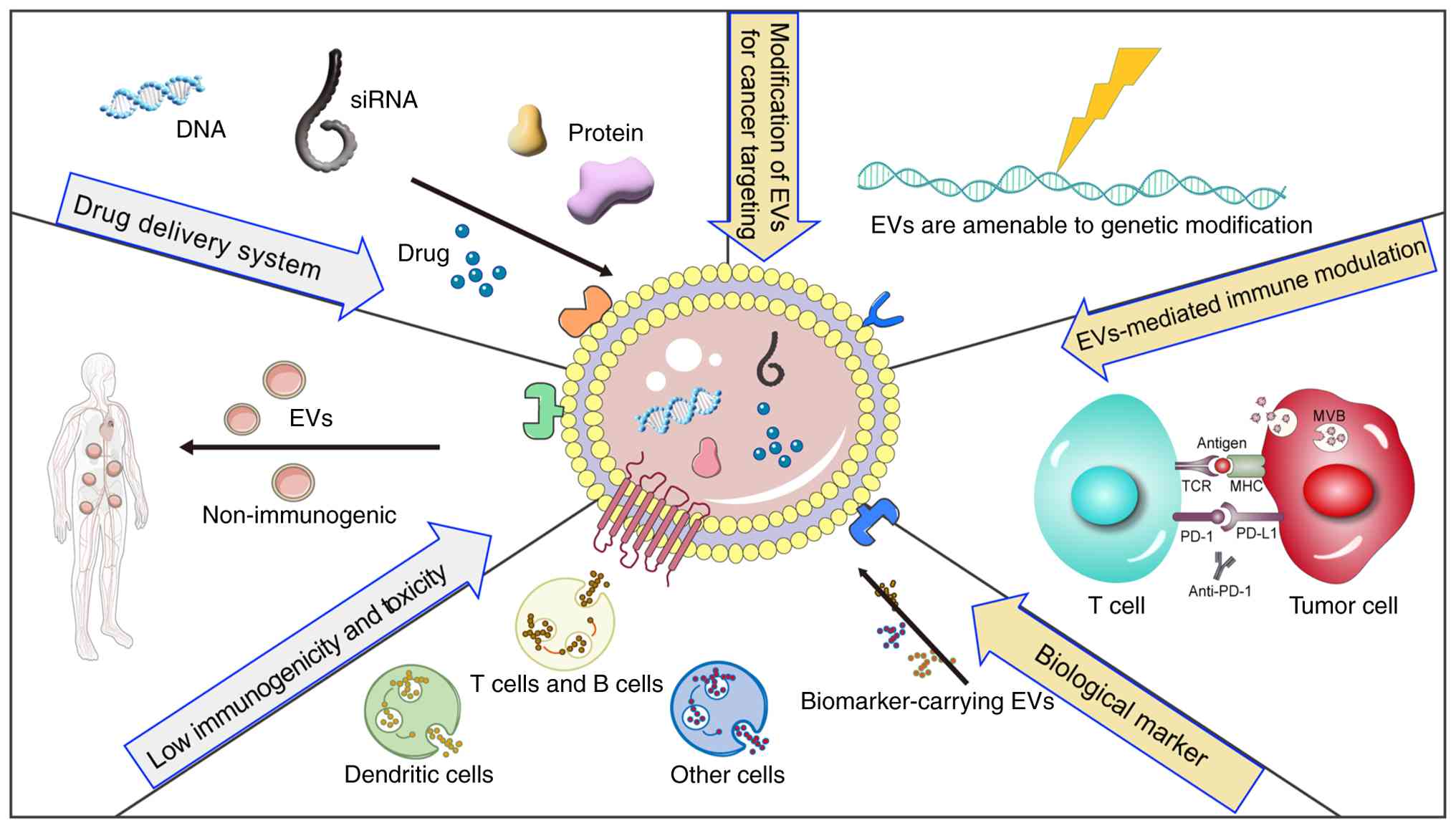 | Figure 1.Schematic overview of the advantages
and therapeutic potential of EVs. Certain nucleic acids and
proteins are transported by EVs, which can be used as biomarkers
for disease diagnosis. The targeting specificity of EVs toward
particular cell types can be enhanced through genetic engineering
or click-chemistry approaches. Also, EVs present multiple antigens
that can induce immune tolerance, positioning them as a novel
therapeutic strategy. EVs, extracellular vesicles; MHC, major
histocompatibility complex; MVB, multivesicular body; PD-1,
programmed cell death protein 1; PD-L1, programmed cell
death-ligand 1; siRNA, small interfering RNA; TCR, T cell
receptor. |
Low immunogenicity and toxicity
EVs offer key advantages over alternative delivery
systems due to their low immunogenicity and minimal cytotoxicity.
As EVs can be isolated from endogenous cellular sources, they
trigger negligible immune responses, making them highly suitable
for drug and gene delivery in tissue repair and regenerative
medicine (44). The encapsulation
of therapeutic agents within EVs not only reduces systemic toxicity
but also enhances bioavailability by minimizing the immune-mediated
clearance of the cargo prior to it reaching target tissues. For
example, in a mouse model of pancreatic cancer, small interfering
RNA (siRNA) delivered via exosomes demonstrated greater efficacy
and fewer side effects than was achieved using NP-based delivery
systems (52). By contrast,
numerous nanomedicines undergo rapid immune clearance, which limits
their clinical effectiveness. Furthermore, the low immunogenicity
and cytotoxicity of EVs enable large-scale use without typical
toxic side effects. Studies have shown that artificial EVs injected
into mice do not cause adverse reactions, highlighting their
potential as therapeutic tools and providing a strong foundation
for the clinical application of EV-based therapies (45).
Engineering EVs as potential drug
delivery vehicles
EVs outperform liposomes in terms of drug delivery
efficiency while maintaining low immunogenicity and toxicity.
Although liposomes share a similar lipid bilayer structure with EVs
and have been used as drug carriers for decades, they predominantly
accumulate in liver and spleen macrophages, resulting in rapid
clearance and reduced therapeutic efficacy. By contrast, EVs
possess endogenous surface proteins that help them to evade immune
detection, thereby extending their circulation time (53). For example, studies have shown that
EVs loaded with porphyrins via electroporation, saponin treatment
or dialysis exhibit higher loading efficiency compared with that of
liposomes, highlighting the advantages of EVs as superior drug
delivery carriers (54).
As aforementioned, EVs are membranous particles
secreted by living cells into the extracellular space, where they
play a key role in intercellular communication. Owing to their
stability, broad tissue distribution and ability to cross
biological barriers, EVs are emerging as promising drug delivery
vehicles. They can encapsulate a diverse range of biological
molecules, including microRNAs (miRNAs/miRs), siRNAs and
recombinant proteins, that are otherwise difficult to deliver
intracellularly without a carrier. Engineered EVs provide a
targeted solution for the delivery of such molecules to specific
sites (55,56).
Two primary strategies are used to direct EVs to
cancer cells and enhance their accumulation at the tumor: i)
Engineering the parent cells prior to EVs isolation and ii)
directly modifying the membrane of purified EVs. Both approaches
aim to improve the targeting specificity and efficacy of EVs-based
delivery. For example, a fusion protein composed of apoptin and the
EVs membrane protein CD9 was expressed in parent cells using
light-responsive cleavable peptide linkers. This fusion protein was
encapsulated within EVs, enabling the light-triggered release of
apoptin (57). In another example,
adipose-derived mesenchymal stem cells were transfected with a
miR-122 plasmid to produce miR-122-enriched EVs. Intratumoral
injection of these EVs increased the sensitivity of liver cancer
cells to sorafenib (58).
As lipid-bound nanostructures, EVs naturally
incorporate transmembrane proteins and surface glycans, including
glycolipids. These native components act as natural anchoring sites
for functionalizing EVs via genetic engineering or bioconjugation
techniques. A wide range of bioactive molecules, including
fluorescent probes, targeting peptides, therapeutic drugs,
nanobodies and aptamers, can be displayed at these sites. For
example, the GE11 peptide, which binds both epidermal growth factor
receptor (EGFR) and platelet-derived growth factor receptor, was
successfully expressed on the surface of engineered EVs derived
from 293 cells, enabling targeted delivery to breast cancer cells
with high levels of EGFR expression (59). In another approach, glycosylation, a
common post-translational modification, was incorporated into a
lysosome-associated membrane protein 2b-targeted peptide fusion
protein. This modification improved the stability of the peptide
and enhanced its expression in both parent cells and EVs, thereby
increasing targeting efficiency toward neuroblastoma cells
(60).
EVs in tumor immunotherapy
EVs are released by nearly all cell types and play a
crucial role in disease progression and pathogenesis by
transporting signaling molecules, including proteins, lipids,
mRNAs, miRNAs and other bioactive substances. As they can be
sampled by minimally invasive procedures, EVs show considerable
potential as clinically useful biomarkers (61). Tumor development is influenced by
dynamic interactions between tumor cells and immune cells within
the TME. Tumor cell-derived EVs (TDEVs) contribute to the
modulation of immune responses by transferring macromolecules to
recipient cells, thereby acting as carriers of immune signals and
contributing to immune surveillance (62). For example, TDEVs can promote immune
evasion by suppressing the co-stimulation of T cells and dendritic
cells (DCs), thereby conferring resistance to immune checkpoint
inhibitor (ICI) therapies (63). In
hepatocellular carcinoma, EVs secreted by hepatocytes deficient in
the gluconeogenic enzyme fructose-1,6-bisphosphatase 1 have been
shown to target infiltrating NK cells, resulting in their
dysfunction and depletion, which facilitates immune escape and
tumor progression through remodeling of the immune microenvironment
(64,65).
Conversely, EVs can also support antitumor immunity.
NK cell-derived exosomes can deliver cytotoxic agents such as
perforin and granzymes, which directly induce the lysis of melanoma
cells (66). In addition, EVs
derived from MHCII-expressing DCs can induce antigen-specific
CD4+ T-cell activation (67). Furthermore, it has been reported
that bone marrow DC-derived EVs regulate allograft rejection in
vivo, and the intravenous administration of donor DC-derived
EVs has been shown to delay acute rejection in rats receiving a
heart transplant (68).
Exosomes are nanocapsules enriched in MHCII
molecules and secreted by B-lymphoblast-like cells. They play a
crucial role in intercellular communication, particularly in
processes such as tumor angiogenesis and cell differentiation.
Advances in exosome production and purification have enhanced their
potential as versatile platforms for drug delivery, antigen
presentation and biologically targeted therapy. Consequently,
immune cell-derived exosomes are now widely regarded as efficient
and natural nanocarriers capable of trafficking to target cells and
modulating immune responses within the TME (69). The application of immune
cell-derived exosomes in immunotherapy and vaccine development has
been explored under both physiological and pathological conditions
(70). ICI therapy is a
well-established approach in tumor immunotherapy, which
significantly increases life expectancy in some patients. However,
only a minority of patients achieve clinical benefit, highlighting
the need for reliable predictive biomarkers (71). Programmed cell death protein 1
(PD-1)/programmed cell death-ligand 1 (PD-L1) inhibitors function
by restoring the ability of suppressed T cells to recognize and
kill tumor cells (71). Given the
crucial role of EVs in tumor immune regulation, a combination of
EVs and PD-L1 has been proposed as an effective biomarker for
predicting patient response to ICI therapy. Notably, a clear
dissociation has been observed between tissue-based and EV-based
PD-L1 detection: while PD-L1 in tissue shows no association with
survival, PD-L1 expression on circulating EVs reliably predicts
survival in patients with non-small cell lung cancer (NSCLC). This
dynamic, EV-based measurement offers a superior predictive model
for the identification of patients who are likely to respond to ICI
treatment and experience a survival benefit (72). In a study of 71 patients with
metastatic melanoma (MM), circulating PD-L1+ and
PD-1+ EVs were associated with responsiveness to ICI
therapy. Circulating PD-1+ EVs, in particular, were
implicated as a driving factor for resistance to anti-PD-1 therapy,
suggesting a role in the stratification in patients with MM
(73). These findings suggest that
a comprehensive understanding of the biological roles of EVs within
the TME is essential for effective cancer treatment and the
development of effective cancer immunotherapies. Furthermore, the
inhibition of EVs secretion pathways is currently being
investigated as a therapeutic strategy (74). Combining targeted therapy that
disrupt EV signaling with anti-PD-L1 therapy may potentially be
more effective than anti-PD-L1 monotherapy
3D cell cultures as prospective models to
study EVs in cancer
Potential advantages of 3D cultivation
over traditional 2D models
Historically, 2D cultures of primary cells and tumor
cell lines have been widely used in cancer research and have
provided valuable insights into tumor development and therapeutic
mechanisms. However, 2D culture systems have inherent limitations,
including an inability to recapitulate the genetic and phenotypic
heterogeneity of tumors or accurately mimic the complex in
vivo microenvironment (75). To
more accurately model human tumor biology, 3D organoid culture
systems have been developed. Organoids are self-organizing
spheroidal structures derived from cancer stem cells or
organ-specific progenitors with a 3D extracellular matrix. They
exhibit self-renewal and differentiation capacity, while
maintaining genetic and phenotypic stability over extended culture
periods (76). By more closely
recapitulating the structure and functional characteristics of
primary tumor tissues, organoids have emerged as powerful tools in
both basic and clinical cancer research. Their accessibility and
experimental versatility make them particularly valuable for
studying tumor immunity and mechanisms of drug resistance.
The limited predictive power of traditional in
vitro models presents a major obstacle to the development of
effective cancer immunotherapies. However, advances in 3D
organoid-immune cell co-culture systems have enabled the
establishment of immunocompetent tumor models that more accurately
recapitulate the patient-specific TME. These models have
transformed preclinical evaluation by enabling the functional
screening of both immunotherapeutic and chemotherapeutic agents
under conditions that mimic in vivo biology. Their
application is exemplified by a study of prostate cancer in which
the co-culture of organoids with cancer-associated fibroblasts
(CAFs) demonstrated that CAF-derived neuregulin 1 promotes
resistance to androgen therapy via HER3 activation in tumor cells
(77). Similarly, Sebrell et
al (78) showed that
co-culturing monocyte-derived DCs with gastric cancer organoids
enables patient-derived organoids (PDOs) to recruit chemokines and
DCs, thereby participating in immune surveillance during
Helicobacter pylori infection. In another study, Dijkstra
et al (79) demonstrate that
co-cultures of autologous tumor organoids and peripheral blood
lymphocytes can be used to enrich tumor-reactive T cells from the
peripheral blood of patients with mismatch repair-deficient
colorectal cancer and NSCLC. This may provide a basis for
individualized treatment of T cells in patients with cancer.
Collectively, these advances in organoid-immune cell co-culture
technology are crucial for advancing tumor immunotherapy, enabling
more precise investigation of tumor-immune interactions and
offering valuable insights to inform clinical treatment
strategies.
Applications of 3D model cultures in
EV generation
Colorectal cancer organoids have been demonstrated
to secrete significantly higher quantities of EVs than their
conventional 2D-cultured counterparts (80). In colorectal cancer organoids, APC
mutations that activate the Wnt pathway further promote EVs
secretion in Matrigel-based cultures, a process that has been
suggested to be facilitated by collagen, a key extracellular matrix
component in matrigel (81).
Another proposed mechanism involves the upregulation of specific
transporters. For example, the ATP-binding cassette transporter G1
(ABCG1), a cholesterol efflux pump, is highly expressed in colon
adenocarcinoma tumoroids with pronounced stemness features.
Silencing ABCG1 inhibits EV release and results in the
intracellular accumulation of vesicles (82).
Furthermore, the 3D architecture of organoids
supports improved cell polarization and spatial asymmetry.
Organoids derived from the LIM1863 colon carcinoma line have been
shown to secrete two spatially distinct EV subtypes. Apically
released EVs carry epithelial cell adhesion molecule (EpCAM) and
are uniquely enriched in CD63, mucin 13, sucrase-isomaltase,
dipeptidyl peptidase IV and prominin 1. By contrast, basolaterally
released EVs contain the A33 glycoprotein and are associated with
early endosome antigen 1, ADP-ribosylation factor and clathrin.
These findings demonstrate that EpCAM-positive and A33-positive EVs
are selectively sorted and secreted from the apical and basolateral
membranes, respectively (83). This
spatial organization indicates that distinct EV populations
carrying different markers and cargo proteins are released from the
apical and basal sides of cells in 3D culture (84).
Current challenges of combining EVs
with organoid models in the TME
EVs mediate crucial intercellular communication
within the TME by facilitating autocrine and paracrine signaling
between tumor cells and stromal components (85). Elevated EV levels have been observed
in numerous malignancies, where they transport molecular cargo
essential for tumor progression, and serve as potential diagnostic
and prognostic biomarkers (86).
Also, in tumor-stromal interactions, EVs coordinate complex
intercellular crosstalk, which has led to exploration of their
applications in drug delivery and targeted therapies (87).
Organoids, through their 3D structure, recapitulate
the structural and physiological complexity of in vivo
tissues more accurately than do 2D monolayer cultures, and have
emerged as powerful systems for tumor biology, regenerative
medicine and EV research (75). The
development of organoid technology has enabled the functional
evaluation of EVs in physiologically relevant 3D models, offers
alternative sources for EV production and the potential for
advancing our understanding of human diseases (88).
Several studies illustrate the promise of this
approach. For example, in an in vitro aging model, mouse
intrahepatic bile duct-derived organoids co-cultured with exosomes
from human placental mesenchymal stem cells demonstrated the
ability to delay cellular senescence (89). In another study, Taha et al
(90) revealed that matrix
metalloproteinase 3 (MMP3) regulates tumor growth and EV integrity;
MMP3 knockout disrupted EV structure and function, reduced tumor
organoid proliferation, and suppressed tumor progression,
highlighting the protumorigenic role of MMP3 in the maintenance of
organoid and EV integrity. In addition, the similarity of small RNA
profiles of EVs from 3D cervical cancer spheroids to in
vivo-derived EVs is greater than that of 2D cultures,
underscoring the physiological relevance of 3D models in EV
research (91).
Multiple studies have demonstrated that EVs loaded
with specific miRNAs can regulate immune responses, chemotherapy
resistance and metastatic processes in various cancers. For
example, esophageal adenocarcinoma (EAC)-derived EVs promote the
formation of multilayered gastric organoids through the delivery of
miR-25 and miR-210, establishing a novel co-culture model of
EAC-EVs with 3D organoids (92).
Similarly, ovarian cancer-derived EVs from malignant ascites and
plasma enhance tumor aggressiveness and organoid growth via the
transfer of miR-1246 and miR-1290 (93). In addition, in a study of organoids
derived from patients with pancreatic ductal adenocarcinoma, the
EVs miRNA profiles of the organoids revealed significantly
upregulated levels of miR-21 and miR-195, matching those in patient
plasma. This further validates tumor-derived organoids as
physiologically relevant models for the analysis of EVs cargo
(94).
Collectively, these findings indicate that
tumor-derived organoids serve as robust platform for analyzing the
molecular cargo of tumor cell-derived EVs. Emerging strategies
integrating EVs with organoid models are enhancing our ability to
recapitulate and evaluate dynamic disease processes. EV-loaded
organoid systems provide synergistic advantages for simulating
complex tumor-matrix interactions within the TME, and studies have
leveraged EV-organoid co-culture platforms to elucidate
TME-mediated mechanisms of tumor progression and therapeutic
resistance (95,96).
Overall, tumor organoids are a valuable platform for
studying EVs biology, while EVs enhance the physiological relevance
of organoids in cancer research. Consequently, the integration of
organoid and EVs technologies to generate organoid-derived EVs
(OEVs) holds considerable promise across multiple research domains
(Fig. 2).
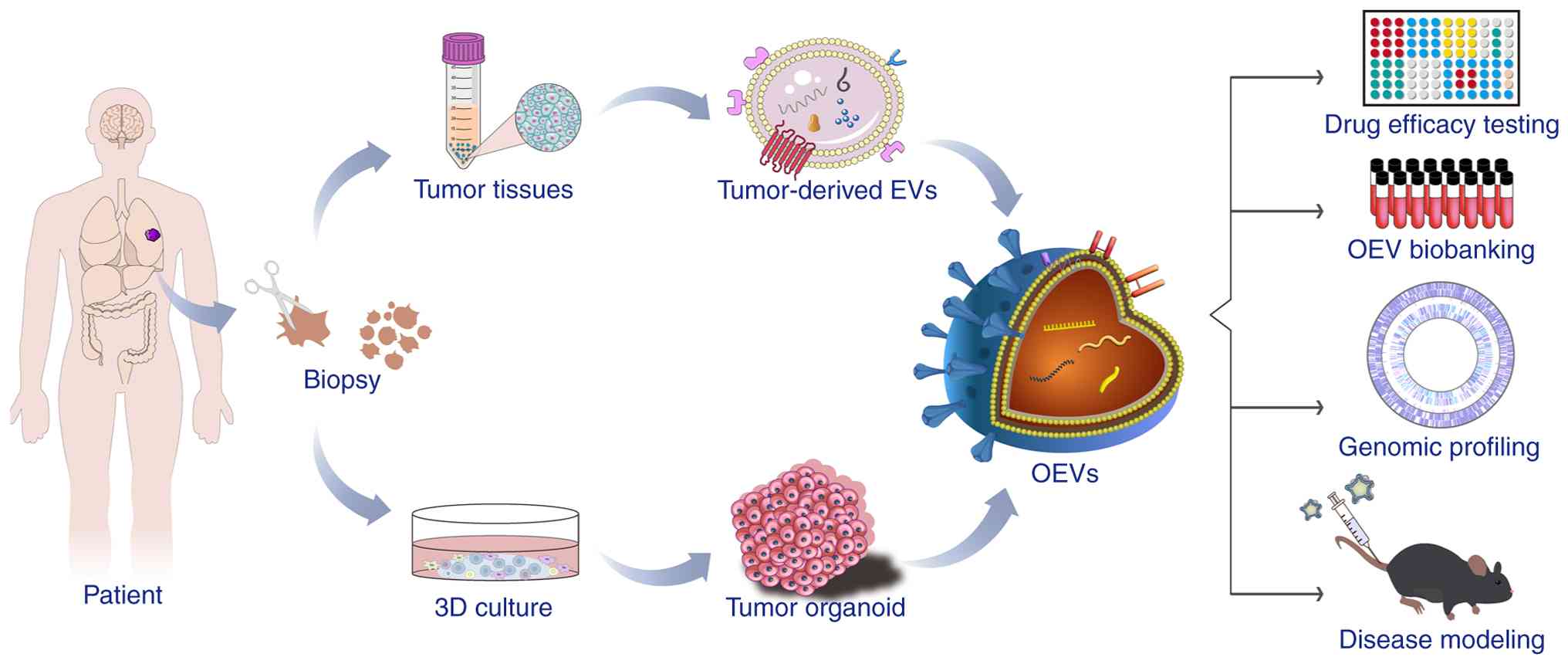 | Figure 2.Synthesis and applications of OEVs.
Tumor tissue samples are processed to obtain tissue-derived EVs and
to generate organoids via 3D culture. The EVs transport bioactive
molecules, including nucleic acids, lipids and proteins, to target
cells. The combination of organoids and EVs yields OEVs, which have
applications in diverse areas, including drug screening,
biobanking, personalized medicine, genomic profiling and animal
research. 3D, 3 dimensional; EVs, extracellular vesicles; OEVs,
organoid-derived EVs. |
Current progress and prospects of
EVs-organoid therapies
Tumor-derived EVs are closely associated with key
oncogenic processes, including drug resistance, TME remodeling,
angiogenesis and distant metastasis (83). However, EVs obtained from
conventional cell cultures may not fully capture the dynamic
changes that occur under pathological conditions. EVs isolated from
bodily fluids such as blood, urine, saliva and cerebrospinal fluid
can more accurately reflect disease-related alterations. Despite
this advantage, they only partially represent tumor-specific
changes and are subject to inter-sample variability. These
limitations can be addressed by isolating EVs directly from tissue
samples. Compared with body fluid-derived EVs, EVs isolated
directly from tumor tissues contain fewer contaminants and
originate from a more defined cellular context. They also enable
the study of specific cellular subpopulations at tumor sites,
offering a more precise representation of tumor-TME interactions
(97). Tissue-derived EVs also
enable the characterization of physiologically relevant EVs subsets
enriched within the tumor site (98). For example, one study reported the
isolation of EVs from frozen biopsy samples of primary and
metastatic melanoma, achieving high purity, sensitivity and
reproducibility. Genomic and proteomic analyses of these samples
were performed to investigate the mechanisms by which
patient-derived organoid (PDO)-derived EVs regulate the TME
(99). In another study, breast
cancer tissue-derived EVs were shown to retain genetic
characteristics of the original tumor and were capable of promoting
diabetic wound healing by suppressing oxidative stress (100).
The development of organoids is based on stem cell
biology and developmental biology principles, mimicking natural
developmental processes to generate miniature organ structures
composed of lineage-specific cell types. They replicate the spatial
architecture and physiological functions of human tissues and
organs, providing a highly relevant system for modeling both normal
physiology and pathology. As next-generation in vitro
biological models, organoids have emerged as a dynamic and
innovative platform in biomedical research (101).
Despite their considerable potential, current
organoid culture systems face several challenges that limit their
widespread application. One major limitation is the general absence
of vascularization in most organoids. As organoids increase in
size, they become constrained by inadequate oxygen supply and the
accumulation of metabolic waste, which can lead to central necrosis
(102). To address this issue,
attempts have been made to construct tumor organoids within a
microenvironment comprising vascular endothelial cells or by
co-culturing organoid tumor cells with vascular endothelial cells
to promote the generation of vascular structures (103). Another limitation is that
individual organoid models primarily assess drug effects on a
single target tissue without predicting potential side effects in
other organ systems. This underscores the requirement for
comprehensive organoid biobanks that enable the systematic
evaluation of drug efficacy and potential toxicity across multiple
tissue types (104).
Integrating multiple disciplines and model systems
is essential for advancing the study of disease progression. For
example, 3D bioprinting is an emerging manufacturing technology
that offers the potential for constructing heterogeneous cellular
microenvironments. This may accelerate organoid research by
enabling precise, scalable and reproducible fabrication of 3D
cellular structures. Therefore, the combination of organoids and 3D
bioprinting technology may facilitate the development of functional
therapies based on organoid technology (105). Zhang et al (98) ummarized the biogenesis, structure
and isolation mechanisms of OEVs. Compared with conventional EVs,
OEVs can be produced in higher yields and exhibit superior
physiological relevance. Organoids possess stem cell
characteristics, and OEVs are capable of delivering active
substances, suggesting that both have potential medical
applications. They can also be engineered to enhance their
therapeutic and targeting properties, showing promise for the
treatment of diseases including inflammatory bowel disease, cancer,
retinal disorders and brain diseases. However, substantial
challenges remain. These include the development of scaled up OEV
production methods to meet clinical demands, the elucidation of the
mechanisms by which OEVs promote tissue repair, and establishment
of standardized guidelines for their clinical application.
Conclusions and future perspectives
Intercellular communication is a fundamental
mechanism in cancer progression and metastasis. As key mediators of
cell-cell signaling within the TME, EVs regulate cellular
homeostasis, tumor development, immune regulation and other
pathological processes through their unique mode of cargo-based
information transfer. Growing evidence indicates that EVs offer
substantial advantages over traditional synthetic carriers, and
have important applications as biomarkers and drug delivery systems
in disease diagnosis and treatment.
Conventional 2D cell cultures fail to simulate the
complex cell-cell and cell-matrix interactions characteristic of
the native TME, limiting their translational relevance. By
contrast, organoids as 3D tissue analogs closely mimic the
structure and function of in vivo tissues, providing a more
physiologically representative experimental model. The development
of patient-derived 3D organoid cultures represents a major
advancement, as these self-organizing structures faithfully
replicate the architectural, genetic and phenotypic heterogeneity
of primary tumors. The use of organoids to investigate specific EVs
functions has broad application potential in regenerative medicine
and oncology. The strategic integration of EV biology with 3D
organoid technology establishes a novel and powerful research
paradigm. Supplementing organoid cultures with tumor-derived EVs,
or co-culturing tumor organoids with stromal cell-derived EVs,
creates more physiologically relevant models that capture the
paracrine and endocrine signaling networks active in cancer.
The present review has summarized the value of
organoids as promising models for studying the TME and tumor
immunity, examined research progress in the combination of organoid
and EVs approaches in malignancies, and highlighted emerging
applications and challenges associated with OEVs. However, OEV
research remains at an early stage. Further efforts are necessary
to deepen mechanistic understanding and optimize culture and
isolation methods, thereby supporting their clinical translation
and practical application.
Acknowledgements
Not applicable.
Funding
This study was supported by Ningxia Autonomous Region Key
R&D Programs (grant no. 2022BEG03124), Ningxia Natural Science
Foundation (grant no. 2025AAC030763), Ningxia Natural Science
Foundation (grant no. 2024AAC03587) and Special Talent Introduction
Project of Ningxia Autonomous Region Key R&D Programs (grant
no. 2023BSB03054).
Availability of data and materials
Not applicable.
Authors' contributions
JC and YX prepared the original draft and wrote the
main content. YG created the figures and participated in writing
the draft. XL and LW supervised the manuscript revision process and
revised the manuscript. Data authentication is not applicable. All
authors read and approved the final version of the manuscript.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Wessler S, Aberger F and Hartmann TN: The
sound of tumor cell-microenvironment communication-composed by the
cancer cluster salzburg research network. Cell Commun Signal.
15:202017. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Urabe F, Kosaka N, Ito K, Kimura T, Egawa
S and Ochiya T: Extracellular vesicles as biomarkers and
therapeutic targets for cancer. Am J Physiol Cell Physiol.
318:C29–C39. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Bebelman MP, Smit MJ, Pegtel DM and Baglio
SR: Biogenesis and function of extracellular vesicles in cancer.
Pharmacol Ther. 188:1–11. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Ortiz A: Extracellular vesicles in cancer
progression. Semin Cancer Biol. 76:139–142. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Khan NLA, Muhandiram S, Dissanayake K,
Godakumara K, Midekessa G, Andronowska A, Heath PR, Kodithuwakku S,
Hart AR and Fazeli A: Effect of 3D and 2D cell culture systems on
trophoblast extracellular vesicle physico-chemical characteristics
and potency. Front Cell Dev Biol. 12:13825522024. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Zhao H, Jiang E and Shang Z: 3D co-culture
of cancer-associated fibroblast with oral cancer organoids. J Dent
Res. 100:201–208. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Campora S and Lo Cicero A: The 3D language
of cancer: Communication via extracellular vesicles from tumor
spheroids and organoids. Int J Mol Sci. 26:71042025. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Fiorini E, Veghini L and Corbo V: Modeling
cell communication in cancer with organoids: Making the complex
simple. Front Cell Dev Biol. 8:1662020. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Chargaff E and West R: The biological
significance of the thromboplastic protein of blood. J Biol Chem.
166:189–197. 1946. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Anderson HC: Vesicles associated with
calcification in the matrix of epiphyseal cartilage. J Cell Biol.
41:59–72. 1969. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Raposo G, Nijman HW, Stoorvogel W,
Liejendekker R, Harding CV, Melief CJ and Geuze HJ: B lymphocytes
secrete antigen-presenting vesicles. J Exp Med. 183:1161–1172.
1996. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Izquierdo-Useros N, Puertas MC, Borràs FE,
Blanco J and Martinez-Picado J: Exosomes and retroviruses: the
chicken or the egg? Cell Microbiol. 13:10–17. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Mittelbrunn M and Sánchez-Madrid F:
Intercellular communication: Diverse structures for exchange of
genetic information. Nat Rev Mol Cell Biol. 13:328–335. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Abecassis MM, Burke R, Klintmalm GB, Matas
AJ, Merion RM, Millman D, Olthoff K and Roberts JP; American
Society of Transplant Surgeons, : American society of transplant
surgeons transplant center outcomes requirements-a threat to
innovation. Am J Transplant. 9:1279–1286. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Jeppesen DK, Zhang Q, Franklin JL and
Coffey RJ: Extracellular vesicles and nanoparticles: Emerging
complexities. Trends Cell Biol. 33:667–681. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Raposo G and Stoorvogel W: Extracellular
vesicles: Exosomes, microvesicles, and friends. J Cell Biol.
200:373–383. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Xu R, Greening DW, Zhu HJ, Takahashi N and
Simpson RJ: Extracellular vesicle isolation and characterization:
Toward clinical application. J Clin Invest. 126:1152–1162. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Krylova SV and Feng D: The machinery of
exosomes: Biogenesis, release, and uptake. Int J Mol Sci.
24:13372023. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Menck K, Sivaloganathan S, Bleckmann A and
Binder C: Microvesicles in cancer: Small size, large potential. Int
J Mol Sci. 21:53732020. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Xu X, Lai Y and Hua ZC: Apoptosis and
apoptotic body: Disease message and therapeutic target potentials.
Biosci Rep. 39:BSR201809922019. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Ma Y, Li T, Zhao L, Zhou D, Dong L, Xu Z,
Wang Y, Yao X and Zhao K: Isolation and characterization of
extracellular vesicle-like nanoparticles derived from migrasomes.
FEBS J. 290:3359–3368. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Minciacchi VR, You S, Spinelli C, Morley
S, Zandian M, Aspuria PJ, Cavallini L, Ciardiello C, Reis Sobreiro
M, Morello M, et al: Large oncosomes contain distinct protein cargo
and represent a separate functional class of tumor-derived
extracellular vesicles. Oncotarget. 6:11327–11341. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Mathieu M, Névo N, Jouve M, Valenzuela JI,
Maurin M, Verweij FJ, Palmulli R, Lankar D, Dingli F, Loew D, et
al: Specificities of exosome versus small ectosome secretion
revealed by live intracellular tracking and synchronized
extracellular vesicle release of CD9 and CD63. bioRXiv. 2020.
|
|
24
|
Théry C, Amigorena S, Raposo G and Clayton
A: Isolation and characterization of exosomes from cell culture
supernatants and biological fluids. Curr Protoc Cell Biol Chapter
3. Unit 3.22. 2006.PubMed/NCBI
|
|
25
|
Colombo M, Raposo G and Théry C:
Biogenesis, secretion, and intercellular interactions of exosomes
and other extracellular vesicles. Annu Rev Cell Dev Biol.
30:255–289. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
de Menezes-Neto A, Sáez MJF, Lozano-Ramos
I, Segui-Barber J, Martin-Jaular L, Ullate JME, Fernandez-Becerra
C, Borrás FE and Del Portillo HA: Size-exclusion chromatography as
a stand-alone methodology identifies novel markers in mass
spectrometry analyses of plasma-derived vesicles from healthy
individuals. J Extracell Vesicles. 4:273782015. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Huang T and He J: Characterization of
extracellular vesicles by size-exclusion high-performance liquid
chromatography (HPLC). Extracellular Vesicles: Methods and
Protocols. Springer; New York, NY: pp. 191–199. 2017, View Article : Google Scholar
|
|
28
|
Gallart-Palau X, Serra A, Wong ASW, Sandin
S, Lai MKP, Chen CP, Kon OL and Sze SK: Extracellular vesicles are
rapidly purified from human plasma by PRotein organic solvent
PRecipitation (PROSPR). Sci Rep. 5:146642015. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Heinemann ML, Ilmer M, Silva LP, Hawke DH,
Recio A, Vorontsova MA, Alt E and Vykoukal J: Benchtop isolation
and characterization of functional exosomes by sequential
filtration. J Chromatogr A. 1371:125–135. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Böing AN, van der Pol E, Grootemaat AE,
Coumans FA, Sturk A and Nieuwland R: Single-step isolation of
extracellular vesicles by size-exclusion chromatography. J
Extracell Vesicles. 3:10.3402/jev.v3.23430. 2014. View Article : Google Scholar
|
|
31
|
Dehghani M, Lucas K, Flax J, McGrath J and
Gaborski T: Tangential flow microfluidics for the capture and
release of nanoparticles and extracellular vesicles on conventional
and ultrathin membranes. Adv Mater Technol. 4:19005392019.
View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Li R, Wang C, Zhou M, Liu Y, Chen S, Chai
Z, Huang H, Zhang K, Han Z, Hua G, et al: Heparan sulfate
proteoglycan-mediated internalization of extracellular vesicles
ameliorates liver fibrosis by targeting hepatic stellate cells.
Extracell Vesicle. 1:1000182022. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Zhu L, Du J, Cheng X, Hu R, Li X, Chen X
and Xu S: Microfluidic innovations for enhanced extracellular
vesicle isolation and analysis: A comprehensive review. Anal Chem.
97:4695–4705. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Shen W, Guo K, Adkins GB, Jiang Q, Liu Y,
Sedano S, Duan Y, Yan W, Wang SE, Bergersen K, et al: A single
extracellular vesicle (EV) flow cytometry approach to reveal EV
heterogeneity. Angew Chem Int Ed Engl. 57:15675–15680. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Iannotta DAA, Lai A, Nair S, Koifman N,
Lappas M, Salomon C and Wolfram J: Chemically-induced lipoprotein
breakdown for improved extracellular vesicle purification. Small.
20:e23072402024. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Gardiner C, Di Vizio D, Sahoo S, Théry C,
Witwer KW, Wauben M and Hill AF: Techniques used for the isolation
and characterization of extracellular vesicles: Results of a
worldwide survey. J Extracell Vesicles. 5:329452016. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Issman L, Brenner B, Talmon Y and Aharon
A: Cryogenic transmission electron microscopy nanostructural study
of shed microparticles. PLoS One. 8:e836802013. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Klymiuk MC, Balz N, Elashry MI, Heimann M,
Wenisch S and Arnhold S: Exosomes isolation and identification from
equine mesenchymal stem cells. BMC Vet Res. 15:422019. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Davies RT, Kim J, Jang SC, Choi EJ, Gho YS
and Park J: Microfluidic filtration system to isolate extracellular
vesicles from blood. Lab Chip. 12:5202–5210. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Ghosh A, Davey M, Chute IC, Griffiths SG,
Lewis S, Chacko S, Barnett D, Crapoulet N, Fournier S, Joy A, et
al: Rapid isolation of extracellular vesicles from cell culture and
biological fluids using a synthetic peptide with specific affinity
for heat shock proteins. PLoS One. 9:e1104432014. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Clayton A, Court J, Navabi H, Adams M,
Mason MD, Hobot JA, Newman GR and Jasani B: Analysis of antigen
presenting cell derived exosomes, based on immuno-magnetic
isolation and flow cytometry. J Immunol Methods. 247:163–174. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Sheikh M, Bagga H, Bhojwani Y and
Telrandhe U: The role of 3D culture models and advanced
chromatography in exosome research for triple-negative breast
cancer. J Egypt Natl Canc Inst. 37:672025. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Lamichhane TN, Sokic S, Schardt JS, Raiker
RS, Lin JW and Jay SM: Emerging roles for extracellular vesicles in
tissue engineering and regenerative medicine. Tissue Eng Part B
Rev. 21:45–54. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Agrawal P, Wilkstein K, Guinn E, Mason C,
Serrano Martinez CI and Saylae J: A review of tangential flow
filtration: Process development and applications in the
pharmaceutical industry. Org Process Res Dev. 27:571–591. 2023.
View Article : Google Scholar
|
|
45
|
van der Meel R, Fens MHAM, Vader P, van
Solinge WW, Eniola-Adefeso O and Schiffelers RM: Extracellular
vesicles as drug delivery systems: Lessons from the liposome field.
J Control Release. 195:72–85. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Saito RF, Machado CML, Lomba ALO, Otake AH
and Rangel MC: Heat shock proteins mediate intercellular
communications within the tumor microenvironment through
extracellular vesicles. Appl Biosci. 3:45–58. 2024. View Article : Google Scholar
|
|
47
|
Balaj L, Atai NA, Chen W, Mu D, Tannous
BA, Breakefield XO, Skog J and Maguire CA: Heparin affinity
purification of extracellular vesicles. Sci Rep. 5:102662015.
View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Cerezo-Magaña M, Bång-Rudenstam A and
Belting M: The pleiotropic role of proteoglycans in extracellular
vesicle mediated communication in the tumor microenvironment. Semin
Cancer Biol. 62:99–107. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Navasiolava NM, Dignat-George F, Sabatier
F, Larina IM, Demiot C, Fortrat JO, Gauquelin-Koch G, Kozlovskaya
IB and Custaud MA: Enforced physical inactivity increases
endothelial microparticle levels in healthy volunteers. Am J
Physiol Heart Circ Physiol. 299:H248–H256. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Jödicke RA, Huo S, Kränkel N, Piper SK,
Ebinger M, Landmesser U, Flöel A, Endres M and Nave AH: The Dynamic
of extracellular vesicles in patients with subacute stroke: Results
of the ‘biomarkers and perfusion-training-induced changes after
stroke’ (BAPTISe) study. Front Neurol. 12:7310132021. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Jong AY, Wu CH, Li J, Sun J, Fabbri M,
Wayne AS and Seeger RC: Large-scale isolation and cytotoxicity of
extracellular vesicles derived from activated human natural killer
cells. J Extracell Vesicles. 6:12943682017. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Jang SC, Kim OY, Yoon CM, Choi DS, Roh TY,
Park J, Nilsson J, Lötvall J, Kim YK and Gho YS: Bioinspired
exosome-mimetic nanovesicles for targeted delivery of
chemotherapeutics to malignant tumors. ACS Nano. 7:7698–7710. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Buzas EI: The roles of extracellular
vesicles in the immune system. Nat Rev Immunol. 23:236–250. 2023.
View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Wang L, Wang D, Ye Z and Xu J: Engineering
extracellular vesicles as delivery systems in therapeutic
applications. Adv Sci (Weinh). 10:e23005522023. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Lou G, Song X, Yang F, Wu S, Wang J, Chen
Z and Liu Y: Exosomes derived from miR-122-modified adipose
tissue-derived MSCs increase chemosensitivity of hepatocellular
carcinoma. J Hematol Oncol. 8:1222015. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Zhao S, Di Y, Fan H, Xu C, Li H, Wang Y,
Wang W, Li C and Wang J: Targeted delivery of extracellular
vesicles: The mechanisms, techniques and therapeutic applications.
Mol Biomed. 5:602024. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Kowalczyk A, Gajda-Walczak A,
Ruzycka-Ayoush M, Targonska A, Mosieniak G, Glogowski M,
Szumera-Cieckiewicz A, Prochorec-Sobieszek M,
Bamburowicz-Klimkowska M, Nowicka AM and Grudzinski IP: Parallel
SPR and QCM-D quantitative analysis of CD9, CD63, and CD81
tetraspanins: A simple and sensitive way to determine the
concentration of extracellular vesicles isolated from human lung
cancer cells. Anal Chem. 95:9520–9530. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Cao M, Isaac R, Yan W, Ruan X, Jiang L,
Wan Y, Wang J, Wang E, Caron C, Neben S, et al:
Cancer-cell-secreted extracellular vesicles suppress insulin
secretion through miR-122 to impair systemic glucose homeostasis
and contribute to tumour growth. Nat Cell Biol. 24:954–967. 2022.
View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Ohno S, Takanashi M, Sudo K, Ueda S,
Ishikawa A, Matsuyama N, Fujita K, Mizutani T, Ohgi T, Ochiya T, et
al: Systemically injected exosomes targeted to EGFR deliver
antitumor microRNA to breast cancer cells. Mol Ther. 21:185–191.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Hung ME and Leonard JN: Stabilization of
exosome-targeting peptides via engineered glycosylation. J Biol
Chem. 290:8166–8172. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Wan M, Ning B, Spiegel S, Lyon CJ and Hu
TY: Tumor-derived exosomes (TDEs): How to avoid the sting in the
tail. Med Res Rev. 40:385–412. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
62
|
Zhao X, Yuan C, Wangmo D and Subramanian
S: Tumor-secreted extracellular vesicles regulate T-cell
costimulation and can be manipulated to induce tumor-specific
T-cell responses. Gastroenterology. 161:560–574.e11. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Rahmat JN, Liu J, Chen T, Li Z and Zhang
Y: Engineered biological nanoparticles as nanotherapeutics for
tumor immunomodulation. Chem Soc Rev. 53:5862–5903. 2024.
View Article : Google Scholar : PubMed/NCBI
|
|
64
|
Liu Z, You Y, Chen Q, Li G, Pan W, Yang Q,
Dong J, Wu Y, Bei JX, Pan C, et al: Extracellular vesicle-mediated
communication between hepatocytes and natural killer cells promotes
hepatocellular tumorigenesis. Mol Ther. 30:606–620. 2022.
View Article : Google Scholar : PubMed/NCBI
|
|
65
|
Pizzutilo EG, Romanò R, Roazzi L, Agostara
AG, Oresti S, Zeppellini A, Giannetta L, Cerea G, Signorelli D,
Siena S and Sartore-Bianchi A: Immune checkpoint inhibitors and the
exposome: Host-extrinsic factors determine response, survival, and
toxicity. Cancer Res. 83:2283–2296. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
66
|
Zhu L, Kalimuthu S, Gangadaran P, Oh JM,
Lee HW, Baek SH, Jeong SY, Lee SW, Lee J and Ahn BC: Exosomes
derived from natural killer cells exert therapeutic effect in
melanoma. Theranostics. 7:27322017. View Article : Google Scholar : PubMed/NCBI
|
|
67
|
Théry C, Duban L, Segura E, Véron P, Lantz
O and Amigorena S: Indirect activation of naïve CD4+ T cells by
dendritic cell-derived exosomes. Nat Immunol. 3:1156–1162. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
68
|
Pêche H, Heslan M, Usal C, Amigorena S and
Cuturi MC: Presentation of donor major histocompatibility complex
antigens by bone marrow dendritic cell-derived exosomes modulates
allograft rejection. Transplantation. 76:1503–1510. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
69
|
Li Q, Wang H, Peng H, Huyan T and Cacalano
NA: Exosomes: Versatile nano mediators of immune regulation.
Cancers (Basel). 11:15572019. View Article : Google Scholar : PubMed/NCBI
|
|
70
|
Zhao Y, Liu T and Zhou M:
Immune-cell-derived exosomes for cancer therapy. Mol Pharm.
19:3042–3056. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
71
|
Yang F, Wang JF, Wang Y, Liu B and Molina
JR: Comparative analysis of predictive biomarkers for PD-1/PD-L1
inhibitors in cancers: Developments and challenges. Cancers
(Basel). 14:1092021. View Article : Google Scholar : PubMed/NCBI
|
|
72
|
de Miguel-Perez D, Russo A, Arrieta O, Ak
M, Barron F, Gunasekaran M, Mamindla P, Lara-Mejia L, Peterson CB,
Er ME, et al: Extracellular vesicle PD-L1 dynamics predict durable
response to immune-checkpoint inhibitors and survival in patients
with non-small cell lung cancer. J Exp Clin Cancer Res. 41:1862022.
View Article : Google Scholar : PubMed/NCBI
|
|
73
|
Serratì S, Guida M, Di Fonte R, De Summa
S, Strippoli S, Iacobazzi RM, Quarta A, De Risi I, Guida G,
Paradiso A, et al: Circulating extracellular vesicles expressing
PD1 and PD-L1 predict response and mediate resistance to checkpoint
inhibitors immunotherapy in metastatic melanoma. Mol Cancer.
21:202022. View Article : Google Scholar : PubMed/NCBI
|
|
74
|
Abreu SC, Lopes-Pacheco M, Weiss DJ and
Rocco PRM: Mesenchymal stromal cell-derived extracellular vesicles
in lung diseases: Current status and perspectives. Front Cell Dev
Biol. 9:6007112021. View Article : Google Scholar : PubMed/NCBI
|
|
75
|
Rauner G, Gupta PB and Kuperwasser C: From
2D to 3D and beyond: The evolution and impact of in vitro tumor
models in cancer research. Nat Methods. 22:1776–1787. 2025.
View Article : Google Scholar : PubMed/NCBI
|
|
76
|
Eiraku M and Sasai Y: Self-formation of
layered neural structures in three-dimensional culture of ES cells.
Curr Opin Neurobiol. 22:768–777. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
77
|
Liu J, Li P, Wang L, Li M, Ge Z, Noordam
L, Lieshout R, Verstegen MMA, Ma B, Su J, et al: Cancer-associated
fibroblasts provide a stromal niche for liver cancer organoids that
confers trophic effects and therapy resistance. Cell Mol
Gastroenterol Hepatol. 11:407–431. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
78
|
Sebrell TA, Hashimi M, Sidar B, Wilkinson
RA, Kirpotina L, Quinn MT, Malkoç Z, Taylor PJ, Wilking JN and
Bimczok D: A novel gastric spheroid co-culture model reveals
chemokine-dependent recruitment of human dendritic cells to the
gastric epithelium. Cell Mol Gastroenterol Hepatol. 8:157–171.e3.
2019. View Article : Google Scholar : PubMed/NCBI
|
|
79
|
Dijkstra KK, Cattaneo CM, Weeber F,
Chalabi M, van de Haar J, Fanchi LF, Slagter M, van der Velden DL,
Kaing S, Kelderman S, et al: Generation of tumor-reactive T cells
by co-culture of peripheral blood lymphocytes and tumor organoids.
Cell. 174:1586–1598.e12. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
80
|
Abbas ZN, Al-Saffar AZ, Jasim SM and
Sulaiman GM: Comparative analysis between 2D and 3D colorectal
cancer culture models for insights into cellular morphological and
transcriptomic variations. Sci Rep. 13:183802023. View Article : Google Scholar : PubMed/NCBI
|
|
81
|
Szvicsek Z, Oszvald Á, Szabó L, Sándor GO,
Kelemen A, Soós AÁ, Pálóczi K, Harsányi L, Tölgyes T, Dede K, et
al: Extracellular vesicle release from intestinal organoids is
modulated by Apc mutation and other colorectal cancer progression
factors. Cell Mol Life Sci. 76:2463–2476. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
82
|
Namba Y, Sogawa C, Okusha Y, Kawai H,
Itagaki M, Ono K, Murakami J, Aoyama E, Ohyama K, Asaumi JI, et al:
Depletion of lipid efflux pump ABCG1 triggers the intracellular
accumulation of extracellular vesicles and reduces aggregation and
tumorigenesis of metastatic cancer cells. Front Oncol. 8:3762018.
View Article : Google Scholar : PubMed/NCBI
|
|
83
|
Yang Q, Xu J, Gu J, Shi H, Zhang J, Zhang
J, Chen ZS, Fang X, Zhu T and Zhang X: Extracellular vesicles in
cancer drug resistance: Roles, mechanisms, and implications. Adv
Sci (Weinh). 9:e22016092022. View Article : Google Scholar : PubMed/NCBI
|
|
84
|
Tauro BJ, Greening DW, Mathias RA,
Mathivanan S, Ji H and Simpson RJ: Two distinct populations of
exosomes are released from LIM1863 colon carcinoma cell-derived
organoids. Mol Cell Proteomics. 12:587–598. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
85
|
Maia J, Caja S, Strano Moraes MC, Couto N
and Costa-Silva B: Exosome-based cell-cell communication in the
tumor microenvironment. Front Cell Dev Biol. 6:182018. View Article : Google Scholar : PubMed/NCBI
|
|
86
|
LeBleu VS and Kalluri R: Exosomes as a
multicomponent biomarker platform in cancer. Trends Cancer.
6:767–774. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
87
|
Jurj A, Zanoaga O, Braicu C, Lazar V,
Tomuleasa C, Irimie A and Berindan-Neagoe I: A comprehensive
picture of extracellular vesicles and their contents. Molecular
transfer to cancer cells. Cancers (Basel). 12:2982020. View Article : Google Scholar : PubMed/NCBI
|
|
88
|
Bordanaba-Florit G, Madarieta I, Olalde B,
Falcón-Pérez JM and Royo F: 3D cell cultures as prospective models
to study extracellular vesicles in cancer. Cancers (Basel).
13:3072021. View Article : Google Scholar : PubMed/NCBI
|
|
89
|
Hwang WL, Lan HY, Cheng WC, Huang SC and
Yang MH: Tumor stem-like cell-derived exosomal RNAs prime
neutrophils for facilitating tumorigenesis of colon cancer. J
Hematol Oncol. 12:102019. View Article : Google Scholar : PubMed/NCBI
|
|
90
|
Taha EA, Sogawa C, Okusha Y, Kawai H, Oo
MW, Elseoudi A, Lu Y, Nagatsuka H, Kubota S, Satoh A, et al:
Knockout of MMP3 weakens solid tumor organoids and cancer
extracellular vesicles. Cancers (Basel). 12:12602020. View Article : Google Scholar : PubMed/NCBI
|
|
91
|
Thippabhotla S, Zhong C and He M: 3D cell
culture stimulates the secretion of in vivo like extracellular
vesicles. Sci Rep. 9:130122019. View Article : Google Scholar : PubMed/NCBI
|
|
92
|
Ke X, Yan R, Sun Z, Cheng Y, Meltzer A, Lu
N, Shu X, Wang Z, Huang B, Liu X, et al: Esophageal
adenocarcinoma-derived extracellular vesicle microRNAs induce a
neoplastic phenotype in gastric organoids. Neoplasia. 19:941–949.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
93
|
Wang W, Jo H, Park S, Kim H, Kim SI, Han
Y, Lee J, Seol A, Kim J, Lee M, et al: Integrated analysis of
ascites and plasma extracellular vesicles identifies a miRNA-based
diagnostic signature in ovarian cancer. Cancer Lett.
542:2157352022. View Article : Google Scholar : PubMed/NCBI
|
|
94
|
Zeöld A, Sándor GO, Kiss A, Soós AÁ,
Tölgyes T, Bursics A, Szűcs Á, Harsányi L, Kittel Á, Gézsi A, et
al: Shared extracellular vesicle miRNA profiles of matched ductal
pancreatic adenocarcinoma organoids and blood plasma samples show
the power of organoid technology. Cell Mol Life Sci. 78:3005–3020.
2021. View Article : Google Scholar : PubMed/NCBI
|
|
95
|
Zhang Y, Lu A, Zhuang Z, Zhang S, Liu S,
Chen H, Yang X and Wang Z: Can organoid model reveal a key role of
extracellular vesicles in tumors? A comprehensive review of the
literature. Int J Nanomedicine. 18:5511–5527. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
96
|
Zhou G, Li R, Sheng S, Huang J, Zhou F,
Wei Y, Liu H and Su J: Organoids and organoid extracellular
vesicles-based disease treatment strategies. J Nanobiotechnology.
22:6792024. View Article : Google Scholar : PubMed/NCBI
|
|
97
|
Crescitelli R, Lässer C and Lötvall J:
Isolation and characterization of extracellular vesicle
subpopulations from tissues. Nat Protoc. 16:1548–1580. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
98
|
Zhang C, Yang X, Jiang T, Yan C, Xu X and
Chen Z: Tissue-derived extracellular vesicles: Isolation,
purification, and multiple roles in normal and tumor tissues. Life
Sci. 321:1216242023. View Article : Google Scholar : PubMed/NCBI
|
|
99
|
Serratì S, Di Fonte R, Porcelli L, De
Summa S, De Risi I, Fucci L, Ruggieri E, Marvulli TM, Strippoli S,
Fasano R, et al: Circulating extracellular vesicles are monitoring
biomarkers of anti-PD1 response and enhancer of tumor progression
and immunosuppression in metastatic melanoma. J Exp Clin Cancer
Res. 42:2512023. View Article : Google Scholar : PubMed/NCBI
|
|
100
|
Guo J, Jiang G, Chen J, Zhang M, Xiang K,
Wang C, Jiang T, Kang Y, Sun Y, Xu X, et al: Tumor tissue derived
extracellular vesicles promote diabetic wound healing. J Diabetes
Complications. 37:1084352023. View Article : Google Scholar : PubMed/NCBI
|
|
101
|
Sprangers J, Zaalberg IC and Maurice MM:
Organoid-based modeling of intestinal development, regeneration,
and repair. Cell Death Differ. 28:95–107. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
102
|
Strobel HA, Moss SM and Hoying JB: Methods
for vascularization and perfusion of tissue organoids. Mamm Genome.
33:437–450. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
103
|
Lai Benjamin FL, Lu Rick X, Hu Y,
Davenport HL, Dou W, Wang EY, Radulovich N, Tsao MS, Sun Y and
Radisic M: Recapitulating pancreatic tumor microenvironment through
synergistic use of patient organoids and organ-on-a-chip
vasculature. Adv Funct Mater. 30:20005452020. View Article : Google Scholar : PubMed/NCBI
|
|
104
|
Zheng F, Xiao Y, Liu H, Fan Y and Dao M:
Patient-specific organoid and organ-on-a-chip: 3D cell-culture
meets 3D printing and numerical simulation. Adv Biol (Weinh).
5:e20000242021. View Article : Google Scholar : PubMed/NCBI
|
|
105
|
Hu Y, Zhu T and Cui H and Cui H:
Integrating 3D bioprinting and organoids to better recapitulate the
complexity of cellular microenvironments for tissue engineering.
Adv Healthc Mater. 14:e24037622025. View Article : Google Scholar : PubMed/NCBI
|
















