Introduction
Triple-negative breast cancer (TNBC) represents an
aggressive molecular subtype of breast cancer, accounting for
15–20% of all cases (1,2). Despite advances in comprehensive
treatment strategies, TNBC remains characterized by early relapse
and a high propensity for distant metastasis (3–5). Owing
to the absence of estrogen receptor (ER), progesterone receptor
(PR) and HER2, patients receive limited benefits from endocrine or
targeted therapies and have been shown to experience markedly worse
overall survival (OS) compared with other breast cancer molecular
subtypes, particularly hormone receptor-positive and HER2-positive
disease (6–8). Although initially chemosensitive, TNBC
frequently develops drug resistance, contributing to early relapse,
distant metastasis and reduced disease-free survival and OS
(9). Current prognostic systems
based on tumor size, lymph node status and histologic grade
insufficiently capture the biological heterogeneity of TNBC or the
important influence of host-related factors (10,11).
Growing evidence has indicated that systemic host responses,
particularly immune and nutritional status, serve central roles in
tumor progression and treatment response (12,13).
Thus, biomarkers integrating host immunity and nutrition may offer
more accurate prognostic insight within TNBC management.
Systemic inflammation and nutritional deficiency are
key determinants of cancer initiation, progression and survival
(14). Chronic inflammation
promotes immune evasion, angiogenesis and epithelial-mesenchymal
transition through cytokine-mediated pathways including IL-6/STAT3,
TNF-α/NF-κB and TGF-β (15–17). Concurrently, malnutrition impairs
immune competence, leading to compromised T-cell activation,
macrophage dysfunction and weakened antitumor responses (18,19).
Composite indices reflecting inflammation or nutrition, such as the
modified Glasgow prognostic score (mGPS), prognostic nutritional
index (PNI), systemic immune-inflammation index (SII),
neutrophil-to-lymphocyte ratio (NLR) and platelet-to-lymphocyte
ratio (PLR), have shown prognostic utility in a number of
malignancies, including TNBC (20–23).
Elevated NLR independently predicted shorter disease-free survival
in early-stage TNBC (24), while a
meta-analysis of 819 patients showed that a high PLR was associated
with a worse overall and disease-free survival, although effect
sizes and cut-off values varied markedly across studies (25). SII demonstrated prognostic value in
both recurrent/metastatic TNBC (n=62) (26) and a large neoadjuvant cohort of 422
patients, whereby postoperative SII remained an independent
predictor of OS (27). Composite
markers combining inflammatory and nutritional domains have also
been explored. For example, the Naples Prognostic Score (NPS)
outperformed PNI, NLR, PLR and controlling nutritional status
(CONUT) in a cohort of 223 postoperative patients with TNBC
(28). However, the majority of
TNBC-specific analyses remain constrained by modest sample sizes
(frequently <100 patients) (26,29,30)
and inconsistent findings across cohorts. For example, PLR has only
exhibited OS values considered to be borderline statistically
significant before sensitivity adjustment, with numerous
neoadjuvant studies having reported that SII, system inflammation
response index (SIRI) and PLR were not consistently associated with
treatment response or long-term outcomes. Given that the majority
of indices quantify inflammatory activation or nutritional reserve
in isolation, these values fail to capture bidirectional
interactions, which are particularly relevant in TNBC, whereby
systemic inflammation, lymphocyte depletion and metabolic fragility
often coexist. These limitations highlight the need for a simple
integrative biomarker capable of concurrently reflecting
inflammatory-immune activity and nutritional status to improve
preoperative risk stratification in TNBC.
To address this gap, the present study has developed
the lymphocyte-albumin-monocyte index (LANI), a novel biomarker
combining serum albumin (ALB) and the lymphocyte-to-monocyte ratio
(LMR). Both parameters are routinely measured and inexpensive,
supporting feasibility for clinical use. ALB reflects protein
nutritional status, as well as the inflammatory response of the
host, as hypoalbuminemia is associated with cytokine-driven
catabolism, impaired hepatic synthesis and poor tolerance to
treatment (31,32). LMR represents the balance between
antitumor immunity and tumor-promoting inflammation, as lymphocytes
mediate cytotoxic surveillance (33), whereas monocytes can differentiate
into tumor-associated macrophages that foster invasion and immune
escape (34). In TNBC, accumulating
evidence has demonstrated the pronounced coexistence of immune
suppression and metabolic exhaustion. Clinically, patients with
TNBC frequently exhibit elevated systemic inflammatory activation
and impaired lymphocyte-mediated immunity. High NLR has been
repeatedly associated with reduced disease-free and OS in
early-stage and neoadjuvant TNBC cohorts (35–37),
while SII has been identified as an independent adverse prognostic
factor in recurrent/metastatic TNBC and in pre-surgery assessments
of patients who have undergone neoadjuvant treatment (38). Beyond peripheral inflammation,
studies assessing tumor-infiltrating lymphocytes have revealed that
increased NLR is associated with lower tumor-infiltrating
lymphocyte (TIL) density and an unfavorable CD8/Foxp3 ratio,
reflecting a shift toward an immunosuppressive microenvironment
(26,39). These findings collectively indicate
that TNBC is characterized by both systemic lymphocyte depletion
and heightened myeloid-driven inflammatory activity, features that
contribute to immune dysfunction and metabolic vulnerability.
Existing single-dimension inflammatory or nutritional markers
capture only certain aspects of this biology and are therefore
insufficient to reflect the dual immune-metabolic disturbance
typical of TNBC.
The present study aimed to evaluate the prognostic
importance of preoperative LANI in surgically treated TNBC and
develop a LANI-based nomogram for individualized survival
prediction. A retrospective cohort of 166 patients was analyzed to
assess the association between preoperative LANI and long-term OS.
Kaplan-Meier curves and multivariate Cox models were applied to
determine its prognostic value and a nomogram incorporating LANI,
tumor (T) stage and histologic grade was constructed for
personalized prediction. Model performance was examined using
time-dependent receiver operating characteristic (ROC) curves,
calibration plots, Brier scores and decision curve analysis (DCA).
Overall, LANI, in addition to its statistical performance, relies
on two inexpensive, readily available parameters, highlighting its
potential as a practical biomarker for preoperative risk
assessment, treatment planning and longitudinal monitoring in
TNBC.
Patients and methods
Study population
A total of 166 female patients with TNBC treated at
the Affiliated Cancer Hospital of Xinjiang Medical University
(Urumqi, China) between January 2018 and December 2019, were
included in the present retrospective cohort study. Inclusion
criteria ensured patients: i) Were newly diagnosed with primary
TNBC without prior anticancer treatment; ii) had available
preoperative baseline peripheral blood tests; iii) had undergone
curative-intent surgery with axillary evaluation; iv) exhibited
postoperative pathological confirmation of invasive carcinoma; v)
had complete clinicopathological staging data; and vi) had
undergone standardized adjuvant therapy and follow-up according to
institutional protocols. Exclusion criteria included the presence
of bilateral or multifocal breast cancer, pregnancy or lactation,
missing baseline hematological data, prior breast cancer or breast
surgery, concomitant malignancies or the absence of definitive
postoperative pathology. Patients who had received neoadjuvant
therapy were excluded to ensure comparability of baseline
laboratory parameters. According to institutional perioperative
assessment standards, candidates for curative surgery generally did
not present with uncontrolled severe hepatic or renal dysfunction,
overt systemic infections or hematologic disorders that could
markedly alter ALB or leukocyte-derived indices. All baseline
hematological parameters, including serum ALB and leukocyte
subsets, were obtained from routine peripheral blood tests
performed within 7 days prior to surgery. These measurements were
collected at the time of first hospital admission as part of
standard preoperative evaluation. No longitudinal or post-treatment
measurements of these indices were incorporated into the present
analysis. The present study was reviewed and approved by The Ethics
Committee of the Affiliated Cancer Hospital of Xinjiang Medical
University (approval no. K-2024056) and conducted in accordance
with the Declaration of Helsinki. Written informed consent was
obtained from all participants. All eligible patients were
consecutively enrolled to minimize selection bias.
Data collection
Clinical and laboratory data were extracted from the
electronic medical records. Variables included baseline
characteristics (age, BMI, menopausal status, smoking and alcohol
history), hematological parameters (ALB, lymphocyte, monocyte,
neutrophil and platelet counts), pathological features [T stage,
lymph node (N) stage, American Joint Committee on Cancer (AJCC)
stage, histologic grade and Ki-67] as well as survival outcomes.
LMR was calculated as lymphocyte count divided by monocyte count.
The LANI score was derived using prespecified thresholds for ALB
and LMR (ALB, 40 g/l; LMR, 2.97): LANI=0 when ALB <40 g/l and
LMR <2.97; LANI=1 when either ALB ≥40 g/l or LMR ≥2.97l; and
LANI=2 when both ALB ≥40 g/l and LMR ≥2.97. OS was defined as the
interval from surgery to mortality (from any cause) or last
follow-up (December 2024). Furthermore, due to the retrospective
design, a number of factors that can directly influence ALB and
lymphocyte/monocyte counts, such as hepatic or renal dysfunction,
acute or chronic infections, autoimmune or inflammatory diseases or
corticosteroid use, were not systematically documented and
therefore could not be adjusted for, leaving potential residual
confounding factors.
Statistical analysis
All analyses were performed using SPSS (version
29.0.2; IBM Corp.) and R software (version 4.4.2; Posit Software,
PBC). Baseline characteristics were summarized as counts and
percentages for categorical variables and as means ± SD or medians
with interquartile range (IQR) for continuous variables. Group
comparisons used the χ2 or Fisher's exact test for
categorical variables, an unpaired (independent samples) Student's
t-test for normally distributed continuous variables (two-group
comparisons). The Wilcoxon rank-sum test was used for non-normally
distributed variables.
ROC curve analysis was applied to determine the
optimal cut-off values for ALB and LMR using the Youden index,
yielding 39.35 g/l and 2.526, respectively. To ensure practical
clinical applicability, the ROC-derived cut-offs (39.35 g/l for ALB
and 2.526 for LMR) were converted to clinically interpretable
thresholds. Routine laboratory reports do not provide three-decimal
precision and statistically optimized values with excessive
granularity are not used in real-world decision-making. Therefore,
the ALB cut-off was rounded to 40.0 g/l, consistent with thresholds
commonly adopted in breast cancer prognostic studies (40,41).
For LMR, the ROC-derived value was converted to 2.97, which most
closely preserved the distributional characteristics of the
original value while remaining compatible with clinically
reportable formats. These refined thresholds were therefore used to
construct the LANI score. To examine whether this rounding
influenced prognostic performance, a sensitivity analysis was
conducted comparing Cox regression and time-dependent ROC results
obtained using the raw ROC cut-offs compared with the rounded
clinical cut-offs, demonstrating minimal differences in predictive
accuracy between the two approaches.
Restricted cubic spline (RCS) models were fitted to
evaluate potential non-linear associations of ALB and LMR with OS.
Kaplan-Meier survival curves were generated and compared using the
log-rank test. Univariate Cox analyses were first performed to
screen candidate prognostic variables (P<0.05). However, because
T stage, N stage and AJCC stage exhibited notable
multicollinearity, given that AJCC stage is derived from T and N,
simultaneous inclusion of these variables in the multivariable Cox
model resulted in unstable estimates. Therefore, to avoid
overadjustment and preserve model stability, only T stage was
retained among these tumor burden indicators. This decision was
supported by both statistical considerations [more stable hazard
ratios (HR) with narrower CIs] and clinical interpretability (T
stage representing the core component of anatomical tumor extent).
Thus, the final multivariate model included T stage, histologic
grade and LANI, representing three complementary dimensions of
prognosis, namely tumor burden, biological aggressiveness and host
inflammatory-nutritional status, respectively. HRs and 95% CIs were
calculated using the Wald method. A prognostic nomogram was
constructed based on these variables. Model discrimination and
calibration were assessed using time-dependent ROC curves, Brier
scores and calibration plots for 1-, 3- and 5-year OS. DCA was used
to evaluate clinical net benefit. Internal validation was conducted
in the predefined validation cohort. All statistical tests were
two-sided, with P<0.05 being considered to indicate a
statistically significant difference.
Results
Clinicopathological characteristics of
the study cohort
Within the present study, 166 patients with TNBC
were included, all of whom underwent curative-intent surgery with
axillary evaluation as clinically indicated, comprising 116 in the
training cohort and 50 in the validation cohort. Among them, 12.7%
were >45 years, 31.3% were aged 45–59 years, 44.6% were aged
60–74 years and 11.4% were aged ≥75 years. BMI distribution showed
4.2% underweight, 42.8% normal, 38.0% overweight and 15.1% obese.
With regard to tumor characteristics, 16.9, 61.4, 18.1 and 3.6%
were T1, T2, T3 and T4 stages, respectively. Nodal status showed
47.0% N0, 25.3% N1, 17.5% N2 and 10.2% N3 stages. AJCC staging
included 10.2% exhibiting stage I, 57.8% stage II and 31.9% stage
III. Histologic grade comprised 18.1% grade (G) 1, 59.0% G2 and
22.9% G3. High Ki-67 expression (>30%) was observed in 45.8% of
tumors. No significant differences were observed between the
training and validation cohorts (all P>0.05; Table I).
 | Table I.Baseline clinicopathological
characteristics of patients with triple-negative breast cancer in
the overall (n=166), training (n=116) and validation (n=50)
cohorts. |
Table I.
Baseline clinicopathological
characteristics of patients with triple-negative breast cancer in
the overall (n=166), training (n=116) and validation (n=50)
cohorts.
|
| Cohort |
|
|---|
|
|
|
|
|---|
| Characteristic | Overall, n (%) | Training, n
(%) | Validation, n
(%) | P-value |
|---|
| Age, years |
|
|
| 0.886 |
|
<45 | 21 (12.7) | 16 (13.8) | 5 (10.0) |
|
|
45-59 | 52 (31.3) | 37 (31.9) | 15 (30.0) |
|
|
60-74 | 74 (44.6) | 50 (43.1) | 24 (48.0) |
|
|
≥75 | 19 (11.4) | 13 (11.2) | 6 (12.0) |
|
| BMI,
kg/m2 |
|
|
| 0.325 |
|
<18.5 | 7 (4.2) | 3 (2.6) | 4 (8.0) |
|
|
>28 | 25 (15.1) | 18 (15.5) | 7 (14.0) |
|
|
18.5-24 | 71 (42.8) | 53 (45.7) | 18 (36.0) |
|
|
24-28 | 63 (38.0) | 42 (36.2) | 21 (42.0) |
|
| T stage |
|
|
| 0.337 |
| T1 | 28 (16.9) | 19 (16.4) | 9 (18.0) |
|
| T2 | 102 (61.4) | 68 (58.6) | 34 (68.0) |
|
| T3 | 30 (18.1) | 25 (21.6) | 5 (10.0) |
|
| T4 | 6 (3.6) | 4 (3.4) | 2 (4.0) |
|
| N stage |
|
|
| 0.832 |
| N0 | 78 (47.0) | 57 (49.1) | 21 (42.0) |
|
| N1 | 42 (25.3) | 29 (25.0) | 13 (26.0) |
|
| N2 | 29 (17.5) | 19 (16.4) | 10 (20.0) |
|
| N3 | 17 (10.2) | 11 (9.5) | 6 (12.0) |
|
| AJCC stage |
|
|
| 0.757 |
| I | 17 (10.2) | 12 (10.3) | 5 (10.0) |
|
| II | 96 (57.8) | 69 (59.5) | 27 (54.0) |
|
|
III | 53 (31.9) | 35 (30.2) | 18 (36.0) |
|
| Histologic
grade |
|
|
| 0.355 |
| G1 | 30 (18.1) | 24 (20.7) | 6 (12.0) |
|
| G2 | 98 (59.0) | 65 (56.0) | 33 (66.0) |
|
| G3 | 38 (22.9) | 27 (23.3) | 11 (22.0) |
|
| Ki-67 |
|
|
| 0.762 |
|
>30% | 76 (45.8) | 54 (46.6) | 22 (44.0) |
|
|
≤30% | 90 (54.2) | 62 (53.4) | 28 (56.0) |
|
Association of ALB and LMR with
clinicopathological features
Within the training cohort, low ALB levels (<40.0
g/l) were significantly associated with advanced N stage
(P=0.0301), higher AJCC stage (P=0.0221) and poor histologic grade
(P=0.0022). Similarly, low LMR levels (<2.97) were significantly
associated with a higher T stage (P=0.0051). However, no
significant associations were found between ALB or LMR and age,
BMI, pathological subtype or Ki-67 expression (all P>0.05;
Table II). Notably, a brief
examination of these associations revealed that patients with
decreased ALB tended to present with more extensive nodal
involvement and a more advanced T stage, whereas patients with
lower LMR were more likely to exhibit larger primary tumors (a
higher T stage). These associations underscore how both ALB and LMR
reflect underlying tumor burden and systemic
inflammatory-nutritional status, providing a biological rationale
for their integration into the LANI score. These findings suggest
that decreased ALB and LMR are associated with tumor progression
and adverse pathological characteristics in patients with TNBC.
 | Table II.Association between ALB and LMR
levels and clinicopathological features in patients with
triple-negative breast cancer. |
Table II.
Association between ALB and LMR
levels and clinicopathological features in patients with
triple-negative breast cancer.
|
| ALB | LMR |
|---|
|
|
|
|
|---|
| Characteristic | Overall
(n=116) | <40.0
(n=42) | ≥40.0 (n=74) | P-value | Overall
(n=116) | <2.97
(n=33) | ≥2.97 (n=83) | P-value |
|---|
| Age, years |
|
|
|
>0.999a |
|
|
| 0.364a |
|
<45 | 16 (13.8) | 6 (14.3) | 10 (13.5) |
| 16 (13.8) | 5 (15.2) | 11 (13.3) |
|
|
≥75 | 13 (11.2) | 5 (11.9) | 8 (10.8) |
| 13 (11.2) | 6 |(18.2) | 7 (8.4) |
|
|
45-59 | 37 (31.9) | 13 (31.0) | 24 (32.4) |
| 37 (31.9) | 11 (33.3) | 26 (31.3) |
|
|
60-74 | 50 (43.1) | 18 (42.9) | 32 (43.2) |
| 50 (43.1) | 11 (33.3) | 39 (47.0) |
|
| BMI,
kg/m2 |
|
|
| 0.552a |
|
|
| 0.651a |
|
<18.5 | 3 (2.6) | 2 (4.8) | 1 (1.4) |
| 3 (2.6) | 1 (3.0) | 2 (2.4) |
|
|
>28 | 18 (15.5) | 6 (14.3) | 12 (16.2) |
| 18 (15.5) | 3 (9.1) | 15 (18.1) |
|
|
18.5-24 | 53 (45.7) | 21 (50.0) | 32 (43.2) |
| 53 (45.7) | 17 (51.5) | 36 (43.4) |
|
|
24-28 | 42 (36.2) | 13 (31.0) | 29 (39.2) |
| 42 (36.2) | 12 (36.4) | 30 (36.1) |
|
| Pathology |
|
|
| 0.918b |
|
|
| 0.259b |
|
Invasive ductal carcinoma | 89 (76.7) | 32 (76.2) | 57 (77.0) |
| 89 (76.7) | 23 (69.7) | 66 (79.5) |
|
|
Invasive lobular
carcinoma | 27 (23.3) | 10 (23.8) | 17 (23.0) |
| 27 (23.3) | 10 (30.3) | 17 (20.5) |
|
| T stage |
|
|
| 0.357a |
|
|
| 0.005a |
| T1 | 19 (16.4) | 7 (16.7) | 12 (16.2) |
| 19 (16.4) | 6 (18.2) | 13 (15.7) |
|
| T2 | 68 (58.6) | 25 (59.5) | 43 (58.1) |
| 68 (58.6) | 20 (60.6) | 48 (57.8) |
|
| T3 | 25 (21.6) | 7 (16.7) | 18 (24.3) |
| 25 (21.6) | 3 (9.1) | 22 (26.5) |
|
| T4 | 4 (3.4) | 3 (7.1) | 1 (1.4) |
| 4 (3.4) | 4 (12.1) | 0 (0.0) |
|
| N stage |
|
|
| 0.030a |
|
|
| 0.647a |
| N0 | 57 (49.1) | 15 (35.7) | 42 (56.8) |
| 57 (49.1) | 14 (42.4) | 43 (51.8) |
|
| N1 | 29 (25.0) | 11 (26.2) | 18 (24.3) |
| 29 (25.0) | 8 (24.2) | 21 (25.3) |
|
| N2 | 19 (16.4) | 8 (19.0) | 11 (14.9) |
| 19 (16.4) | 7 (21.2) | 12 (14.5) |
|
| N3 | 11 (9.5) | 8 (19.0) | 3 (4.1) |
| 11 (9.5) | 4 (12.1) | 7 (8.4) |
|
| AJCC stage |
|
|
| 0.022a |
|
|
| 0.111a |
| I | 12 (10.3) | 6 (14.3) | 6 (8.1) |
| 12 (10.3) | 4 (12.1) | 8 (9.6) |
|
| II | 69 (59.5) | 18 (42.9) | 51 (68.9) |
| 69 (59.5) | 15 (45.5) | 54 (65.1) |
|
|
III | 35 (30.2) | 18 (42.9) | 17 (23.0) |
| 35 (30.2) | 14 (42.4) | 21 (25.3) |
|
| Histologic
grade |
|
|
| 0.002b |
|
|
| 0.271b |
| G1 | 24 (20.7) | 4 (9.5) | 20 (27.0) |
| 24 (20.7) | 6 (18.2) | 18 (21.7) |
|
| G2 | 65 (56.0) | 21 (50.0) | 44 (59.5) |
| 65 (56.0) | 16 (48.5) | 49 (59.0) |
|
| G3 | 27 (23.3) | 17 (40.5) | 10 (13.5) |
| 27 (23.3) | 11 (33.3) | 16 (19.3) |
|
| Ki-67 |
|
|
| 0.862b |
|
|
| 0.499b |
|
>30% | 54 (46.6) | 20 (47.6) | 34 (45.9) |
| 54 (46.6) | 17 (51.5) | 37 (44.6) |
|
|
≤30% | 62 (53.4) | 22 (52.4) | 40 (54.1) |
| 62 (53.4) | 16 (48.5) | 46 (55.4) |
|
Determination of optimal cut-off
values for ALB and LMR
ROC curve analysis identified 39.35 g/l as the
optimal cut-off value for ALB [area under the curve (AUC)=0.757]
and 2.526 for LMR (AUC=0.634), indicating the prognostic predictive
ability of these indices in patients with TNBC (Fig. 1A). RCS analysis further demonstrated
a continuous association between decreasing ALB, lower LMR levels
and an elevated OS risk, demonstrating their prognostic
significance (Fig. 1B and C). For
improved clinical interpretability and applicability, the present
ROC-derived cut-off values (39.35 g/l and 2.526) were rounded to
the nearest conventional clinical thresholds (40.0 g/l and 2.97,
respectively) for subsequent LANI model construction. Collectively,
these findings highlight the prognostic relevance of ALB and LMR in
patients with TNBC. To further enhance predictive accuracy, these
markers were integrated into the LANI model for comprehensive
prognostic evaluation.
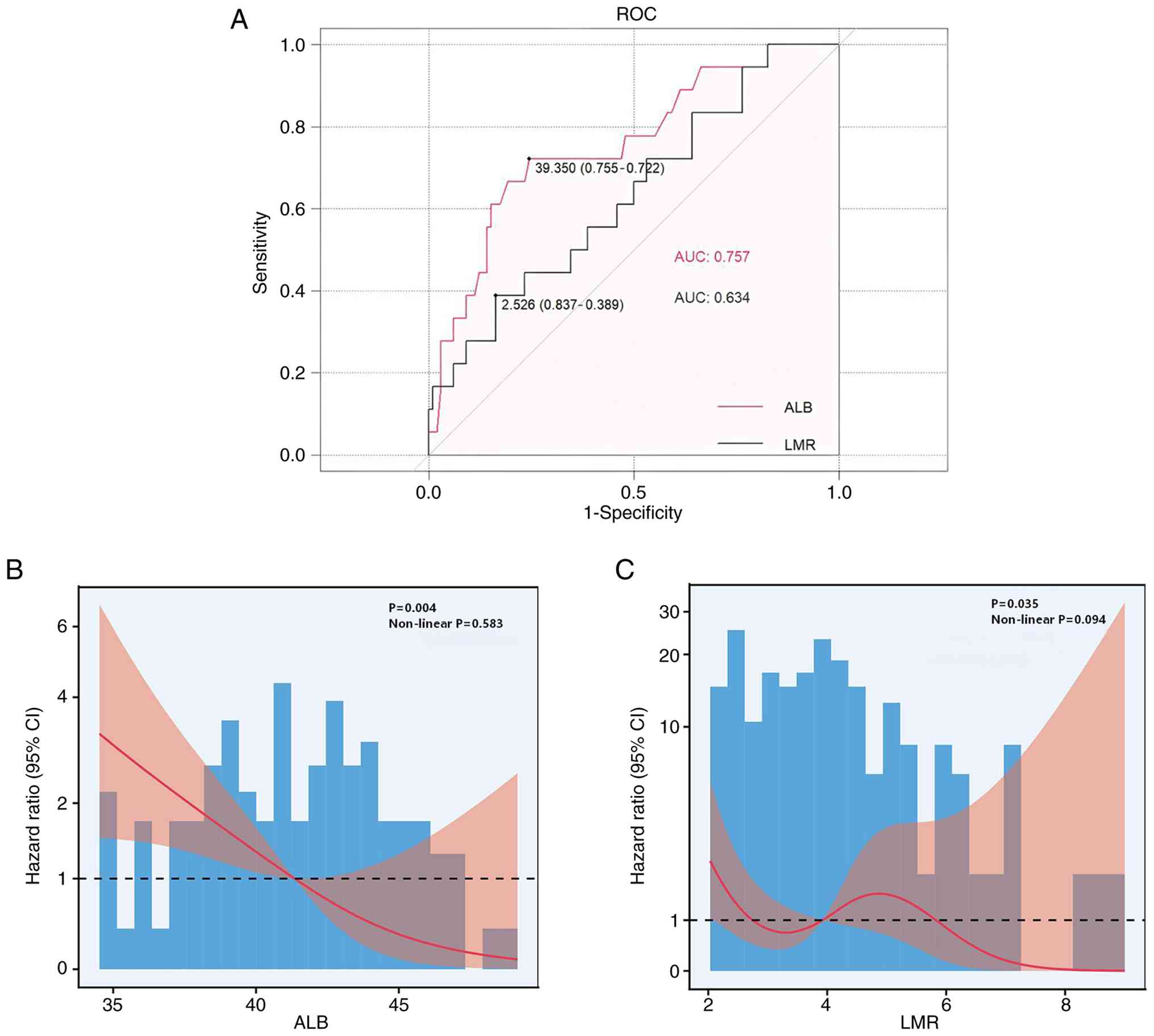 | Figure 1.Prognostic relevance of ALB and LMR
in patients with triple-negative breast cancer. (A) ROC curve
analysis for serum ALB and LMR, showing their predictive accuracy
for OS, with optimal cut-off values of 39.35 g/l for ALB
(AUC=0.757) and 2.526 for LMR (AUC=0.634). These ROC-derived
cut-offs closely approximated the prespecified clinical thresholds
for ALB and LMR (40.0 g/l and 2.97, respectively) used in the LANI
classification to enhance clinical applicability. RCS plots
illustrating the continuous relationship between (B) ALB, (C) LMR
and OS risk. ALB, albumin; LMR, lymphocyte-to-monocyte ratio; ROC,
receiver operating curve; AUC, area under the curve; LANI,
lymphocyte-albumin-monocyte index; OS, overall survival. |
Development and prognostic value of
the LANI model
To improve prognostic stratification in TNBC
management, the LANI model was developed using the rounded clinical
cut-offs for ALB and LMR (Table
III). Patients were categorized into three groups: LANI=0 (ALB
<40.0 g/l and LMR <2.97), LANI=1 (either ALB ≥40.0 g/l or LMR
≥2.97) and LANI=2 (ALB ≥40.0 g/l and LMR ≥2.97). To illustrate the
distribution of events across LANI strata, survival outcomes were
summarized in both cohorts. Within the training cohort (n=116), 18
mortalities occurred. According to the Cox regression dataset,
17/59 (28.8%) mortalities were observed in the LANI ≤1 group,
whereas only 1/57 (1.8%) mortalities occurred in the LANI=2 group
(Table IV). Within the validation
cohort (n=50), 7 mortalities were observed during follow-up. The
lower mortality rate consistently observed in the LANI=2 group
across both cohorts supports its protective effect and may explain
the wide CI exhibited by this group, which reflects a limited
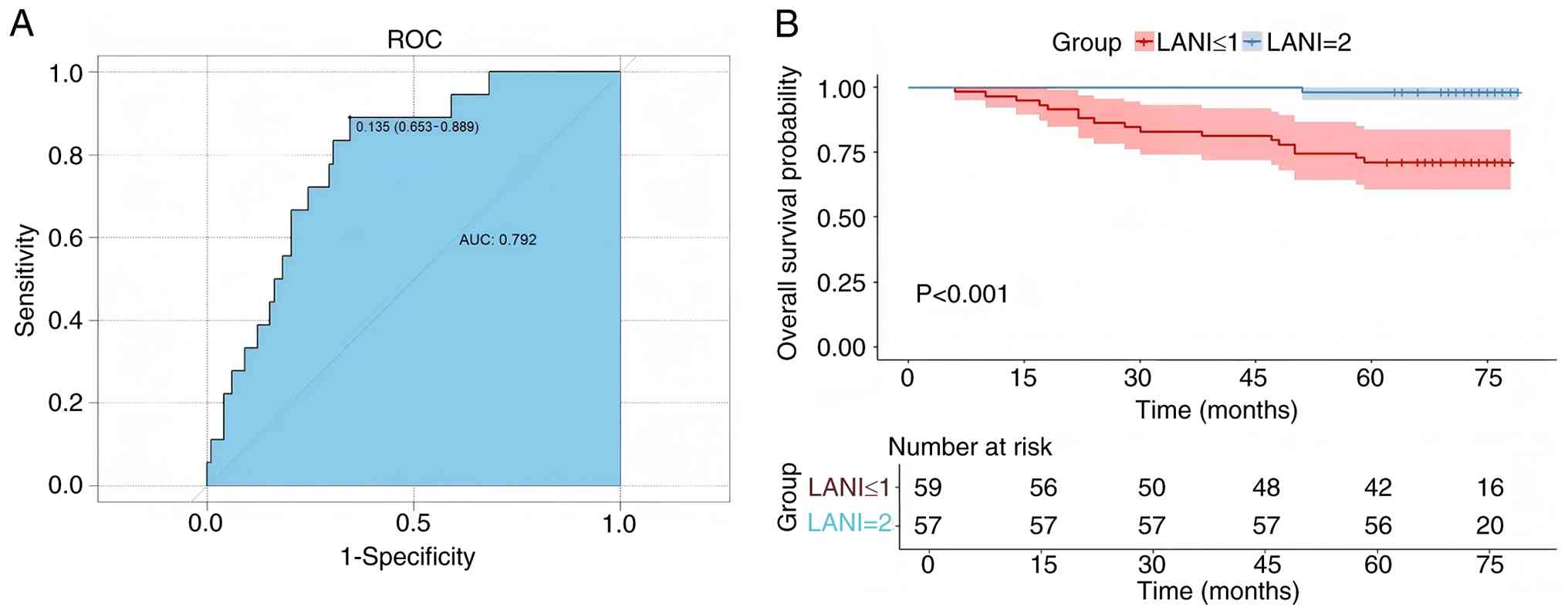
number of events rather than model instability. ROC curve analysis
demonstrated that LANI achieved a higher discriminatory ability for
OS compared with either ALB or LMR alone (AUC=0.792; Fig. 2A). Kaplan-Meier analysis further
exhibited significantly longer OS for patients in LANI=2 compared
with those of LANI ≤1 (log-rank P<0.001; Fig. 2B).
 | Table III.Development of the LANI prognostic
model based on ALB and LMR levels in patients with triple-negative
breast cancer. |
Table III.
Development of the LANI prognostic
model based on ALB and LMR levels in patients with triple-negative
breast cancer.
| LANI | Levels | No. patients |
|---|
| 0 | ALB <40.0 and
LMR <2.97 | 16 |
| 1 | ALB ≥40.0 or LMR
≥2.97 | 43 |
| 2 | ALB ≥40.0 and LMR
≥2.97 | 57 |
 | Table IV.Univariate and multivariate Cox
regression analyses of clinicopathological factors associated with
overall survival in patients with triple-negative breast
cancer. |
Table IV.
Univariate and multivariate Cox
regression analyses of clinicopathological factors associated with
overall survival in patients with triple-negative breast
cancer.
|
|
|
| Univariate | Multivariate |
|---|
|
|
|
|
|
|
|---|
| Characteristic | No. patients | Event (n) | HR | 95% CI | P-value | HR | 95% CI | P-value |
|---|
| Age, years |
|
|
|
|
|
|
|
|
|
<45 | 16 | 3 |
|
|
|
|
|
|
|
≥75 | 13 | 2 | 0.79 | 0.13-4.75 | 0.799 | 2.84 | 0.23-34.94 | 0.415 |
|
45-59 | 37 | 8 | 1.21 | 0.32-4.56 | 0.78 | 2.00 | 0.36-11.02 | 0.426 |
|
60-74 | 50 | 5 | 0.51 | 0.12-2.14 | 0.36 | 1.24 | 0.17-8.94 | 0.831 |
| BMI,
kg/m2 |
|
|
|
|
|
|
|
|
|
<18.5 | 3 | 0 |
|
|
|
|
|
|
|
>28 | 18 | 3 | 3.21 | 0.44-6.35 | 0.132 | 3.99 | 0.48-36.92 | 0.841 |
|
18.5-24 | 53 | 9 | 0.77 | 0.31-2.56 | 0.671 | 0.66 | 0.25-1.72 | 0.394 |
|
24-28 | 42 | 6 | 1.22 | 0.36-4.99 | 0.865 | 1.65 | 0.28-9.63 | 0.578 |
| T stage |
|
|
|
|
|
|
|
|
| T1 | 19 | 1 |
|
|
|
|
|
|
| T2 | 68 | 12 | 3.50 | 0.46-26.93 | 0.229 | 7.53 | 0.54-104.87 | 0.133 |
| T3 | 25 | 2 | 10.56 | 1.94-57.26 | 0.015 | 4.12 | 1.10-15.48 | 0.036 |
| T4 | 4 | 3 | 22.81 | 2.35-221.19 | 0.007 | 24.23 | 2.30-255.54 | 0.008 |
| N stage |
|
|
|
|
|
|
|
|
| N0 | 57 | 2 |
|
|
|
|
|
|
| N1 | 29 | 5 | 5.22 | 1.01-26.92 | 0.048 | 5.98 | 0.60-59.47 | 0.127 |
| N2 | 19 | 6 | 10.94 | 2.21-54.26 | 0.003 | 3.02 | 0.08-111.19 | 0.548 |
| N3 | 11 | 5 | 15.81 | 3.07-81.51 | <0.001 | 1.53 | 0.02-100.16 | 0.842 |
| AJCC stage |
|
|
|
|
|
|
|
|
| I | 12 | 1 |
|
|
|
|
|
|
| II | 69 | 4 | 0.67 | 0.07-5.98 | 0.719 |
|
|
|
|
III | 35 | 13 | 5.27 | 0.69-40.32 | 0.109 |
|
|
|
| Histologic
grade |
|
|
|
|
|
|
|
|
| G1 | 24 | 3 |
|
|
|
|
|
|
| G2 | 65 | 2 | 2.41 | 1.12-5.18 | 0.024 | 1.98 | 1.05-3.72 | 0.036 |
| G3 | 27 | 13 | 4.64 | 1.32-16.33 | 0.017 | 2.76 | 1.41-5.40 | 0.003 |
| Ki-67 |
|
|
|
|
|
|
|
|
|
>30% | 54 | 11 |
|
|
|
|
|
|
|
≤30% | 62 | 7 | 0.53 | 0.21-1.38 | 0.196 | 1.34 | 0.25-7.06 | 0.730 |
| ALB |
|
|
|
|
|
|
|
|
|
<40.0 | 42 | 13 |
|
|
|
|
|
|
|
≥40.0 | 74 | 5 | 0.18 | 0.07-0.52 | 0.001 | 1.73 | 0.23-12.77 | 0.591 |
| LMR |
|
|
|
|
|
|
|
|
|
<2.97 | 33 | 8 |
|
|
|
|
|
|
|
≥2.97 | 83 | 10 | 0.46 | 0.18-1.17 | 0.105 | 3.19 | 0.47-21.55 | 0.234 |
| LANI |
|
|
|
|
|
|
|
|
| ≤1 | 59 | 17 |
|
|
|
|
|
|
| 2 | 57 | 1 | 0.05 | 0.01-0.39 | 0.004 | 0.03 | 0.002-0.46 | 0.012 |
Within the univariate Cox analysis, a higher T
stage, N stage, histologic grade and lower ALB value were all
associated with worse survival. Furthermore, within the
multivariate model including T stage, histologic grade and LANI, T3
(HR=4.12; 95% CI: 1.10-15.48; P=0.036), T4 (HR=24.23; 95% CI:
2.30-255.54; P=0.008), G3 histologic grade (HR=2.76; 95% CI:
1.41-5.40; P=0.003) and LANI (HR=0.03; 95% CI: 0.002-0.46; P=0.012)
remained independent prognostic factors for OS (Table IV). Sensitivity analyses
demonstrated the robustness of LANI. Cox models based on raw
ROC-derived cut-offs and rounded clinical thresholds yielded
comparable estimates, indicating that rounding the cut-offs did not
notably compromise model performance (data not shown).
Nomogram construction and
calibration
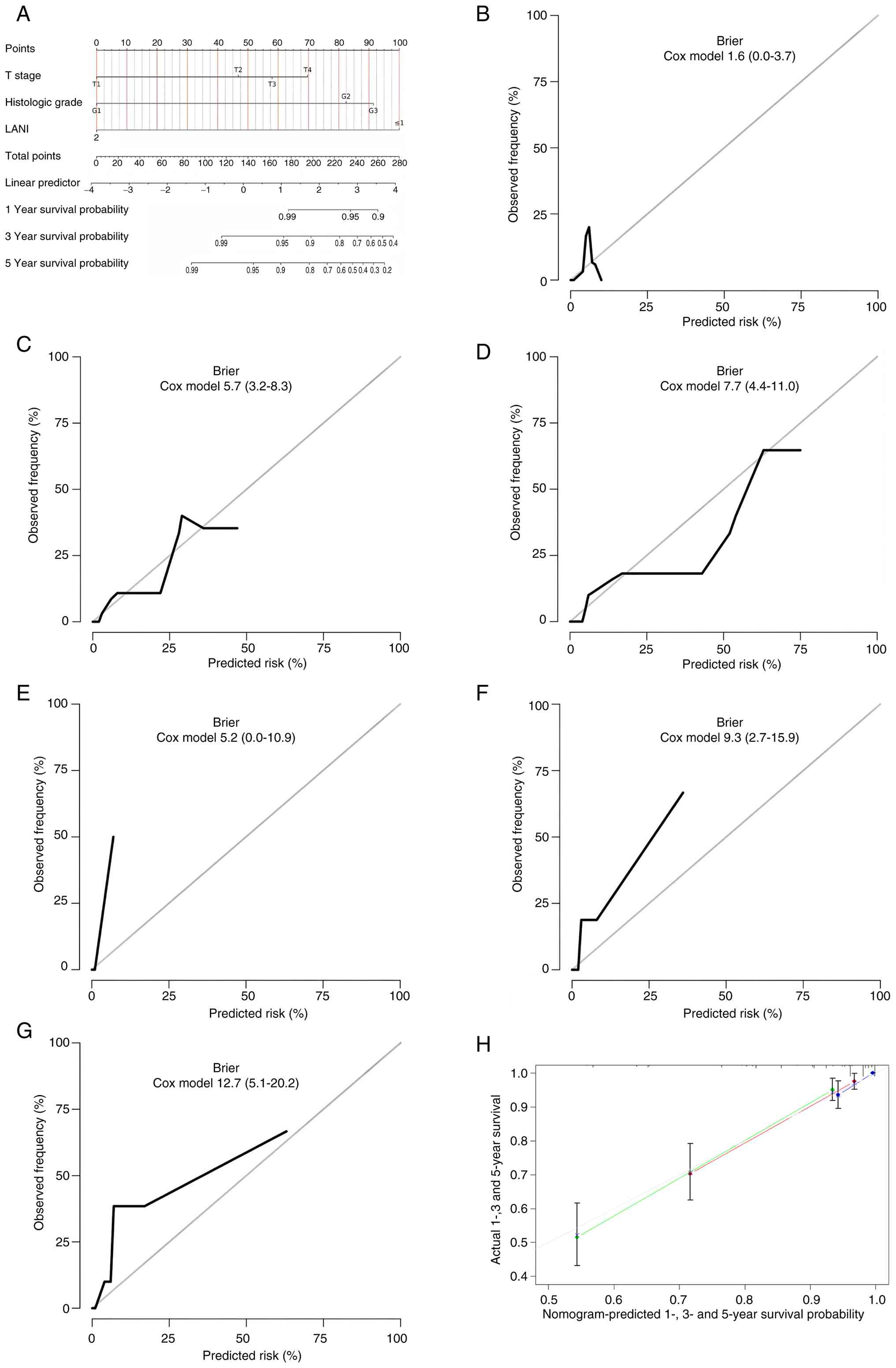
A prognostic nomogram incorporating T stage,
histologic grade and LANI was constructed to predict 1-, 3- and
5-year OS in patients with TNBC (Fig.
3A). Brier score analyses demonstrated a good predictive
performance by the model, with values of 1.6, 5.7 and 7.7 for 1-,
3- and 5-year OS in the training cohort and 5.2, 9.3 and 12.7 in
the validation cohort (Fig. 3B-G).
The relatively low Brier scores across different time points
indicated stable model accuracy. In addition, calibration plots
comparing predicted and observed OS revealed a close concordance,
particularly at 1 and 3 years, suggesting a good calibration
performance by the nomogram (Fig.
3H). These findings demonstrate that the nomogram based on LANI
and clinicopathological factors provides reliable individualized
prognostic predictions for patients with TNBC.
Clinical utility of the nomogram
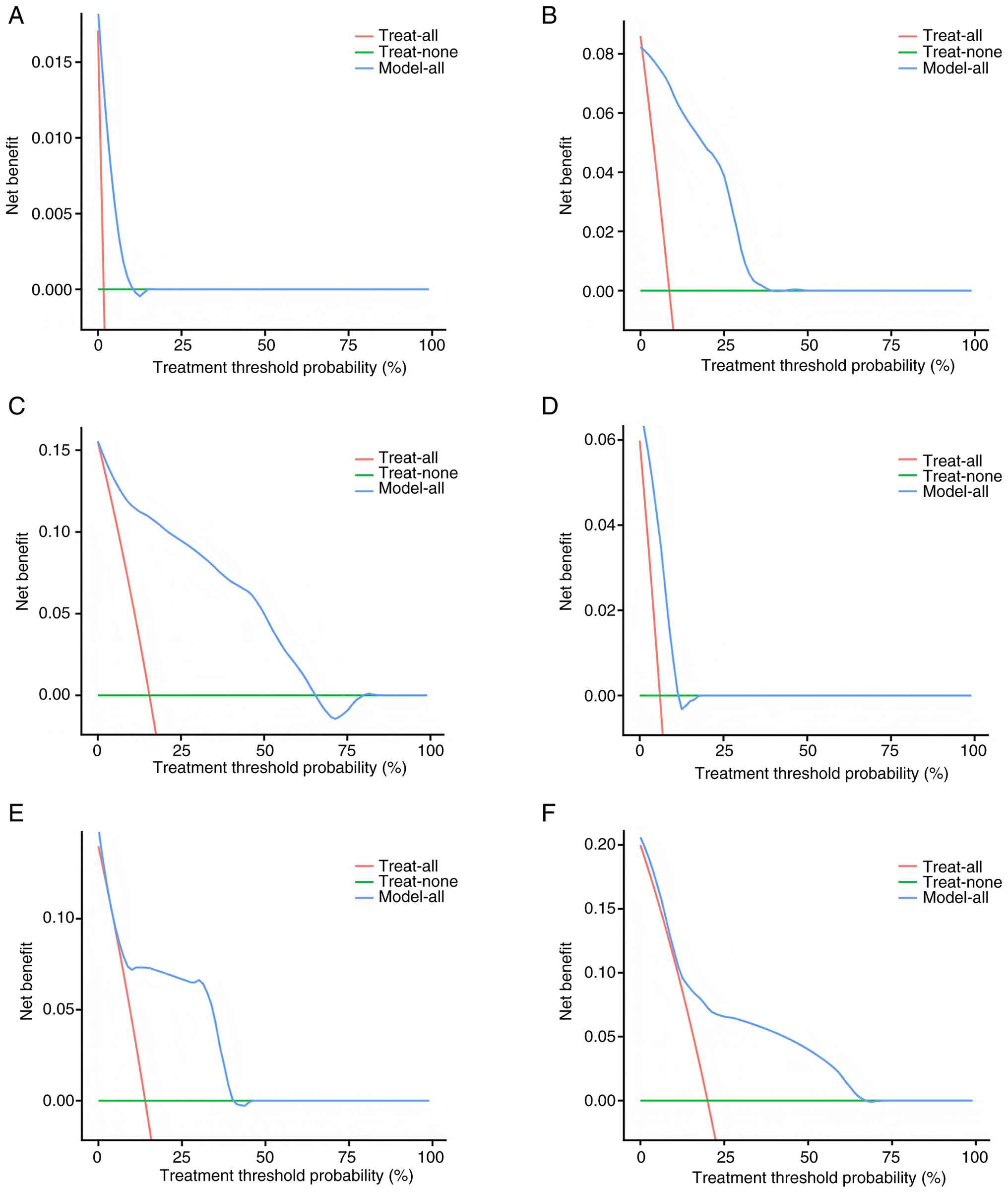
DCA was applied to assess the net clinical benefit
of the LANI-based nomogram in patients with TNBC (Fig. 4A-F). Within the training cohort, the
nomogram consistently outperformed the treat-all and treat-none
strategies across a range of threshold probabilities for 1-, 3- and
5-year OS, with the most pronounced benefit observed between 5–45%
for 3-year OS and up to 70% for 5-year OS. Within the validation
cohort, the nomogram similarly demonstrated a higher net benefit
compared with the treat-all and treat-none strategies across a
clinically relevant range of threshold probabilities (Fig. 4D-F). These findings suggest that the
nomogram may provide good value in guiding individualized clinical
decisions, especially for medium- to long-term prognostic
assessment. Collectively, the DCA results highlight the robustness
and translational potential of the LANI-based nomogram as a
reliable tool for risk stratification in TNBC.
Internal validation of the LANI
model
To further demonstrate the robustness of the LANI
model, internal validation was performed using the predefined
validation cohort. Model discrimination remained robust in the
validation cohort (Fig. 2).
Kaplan-Meier analysis demonstrated that patients with LANI=2
exhibited a significantly longer OS compared with those of LANI ≤1
(log-rank P<0.001; Fig. 2B),
consistent with findings from the training cohort. These validation
results reinforced the stability and generalizability of the
LANI-based nomogram across independent subsets. Calibration
analyses (Fig. 3B-H) showed close
agreement between predicted and observed outcomes, supporting the
calibration accuracy of the model. In addition, DCA (Fig. 4D-F) demonstrated a consistently
higher net clinical benefit of the LANI-based nomogram compared
with the treat-all or treat-none strategies. Collectively, these
results support the feasibility of integrating the LANI score into
individualized prognostic assessments and clinical decision-making
for patients with TNBC.
Discussion
Within the present study, the LANI model, an
integrative biomarker combining serum ALB and the LMR, was
developed. Furthermore, its strong prognostic value in TNBC was
demonstrated. Among 166 surgically treated patients, those with
LANI=2 (ALB ≥40.0 g/l and LMR ≥2.97) consistently showed the most
favorable OS. Multivariate Cox regression demonstrated LANI as an
independent protective factor, whereas higher T stage (T3-T4) and
poor histologic differentiation (G3) were adverse prognostic
indicators. A prognostic nomogram incorporating LANI, T stage and
histologic grade achieved robust discrimination, reliable
calibration and clear clinical net benefits, highlighting its
translational potential. The LANI thresholds were derived from
ROC-based cut-off values and rounded to commonly used clinical
reference points to enhance interpretability without impairing
predictive performance.
Beyond statistical performance, the biological
foundation of LANI provides a coherent mechanistic explanation for
its prognostic relevance in TNBC. In this cohort, low serum ALB was
significantly associated with higher nodal burden, a more advanced
AJCC stage and worse histologic differentiation, whereas a reduced
LMR was associated with deeper primary tumor invasion. These
clinical patterns suggest that an unfavorable LANI profile reflects
not only anatomical tumor advancement but also a profoundly
perturbed host milieu characterized by chronic inflammation,
metabolic stress and compromised antitumor immunity.
Serum ALB is a sensitive indicator of protein-energy
reserve and inflammatory-metabolic load, whereby in aggressive
breast cancer types, proinflammatory cytokines, such as IL-6 and
TNF-α, are frequently elevated and activate STAT3 and NF-κB
signaling in hepatocytes, shifting protein synthesis toward
acute-phase reactants at the expense of ALB production (42–44).
This negative acute-phase response accelerates proteolysis,
amplifies oxidative stress and sustains a systemic catabolic state,
consistent with studies showing that ALB-related inflammatory
ratios, such as the neutrophil percentage-to-ALB ratio (NPAR),
reflect systemic inflammation and distant metastatic potential in
breast cancer (45–47). Meanwhile, the LMR captures the
balance between lymphocyte-mediated antitumor immunity and
monocyte/macrophage-driven protumor inflammation. Accumulating
evidence has shown that breast cancer cells promote monocyte
differentiation into M2-polarized tumor-associated macrophages
through exosomal long non-coding RNAs, the
MIR4435-2HG/ubiquitin-conjugating enzyme E2 N axis and the
leupaxin/histone deacetylase 6/early growth response 2 pathway,
accompanied by upregulation of IL-10, VEGF and TGF-β, thereby
facilitating angiogenesis, epithelial-mesenchymal transition and
immune escape (48–50). Integration of these findings with
the present results suggests LANI effectively unifies two key
biological axes, protein-energy reserve and immune-cell
homeostasis, into a condensed surrogate of the host immunometabolic
state, providing a robust mechanistic basis for its association
with TNBC clinical outcomes.
Growing evidence has indicated that
inflammatory-nutritional composite indices exhibit improved
prognostic discrimination compared with single inflammatory markers
in breast cancer. For example, in a postoperative TNBC cohort
(n=223), the NPS demonstrated higher AUCs and C-indices compared
with PNI, NLR, PLR and CONUT for both OS and breast cancer-specific
survival, underscoring the value of integrating immune and
nutritional dimensions (28). In a
neoadjuvant TNBC population, Zhu et al (30) showed that a multiparametric model
incorporating IL-6, PLR, SII and Ki-67 exhibited improved
stratification of disease-free survival compared with any single
indicator. Consistently, Sun et al (38) introduced the ALB-corrected NLR ratio
(NLR-0.04 × ALB), reporting C-indices of 0.74-0.76 for disease-free
survival and OS in both the training and validation cohorts,
outperforming traditional inflammation-based parameters.
Furthermore, a large neoadjuvant study demonstrated that
pretreatment and posttreatment NLR markedly contributed to OS
prediction, with the constructed nomogram reaching a C-index of
~0.76, further supporting the rationale for embedding inflammatory
markers into composite prognostic models (51).
Based upon these breast cancer-specific findings,
the present study introduced a simple TNBC-focused composite index
(LANI) that integrates serum ALB and the LMR into a single
prognostic measure and to the best of our knowledge, this was
applied for the first time in tumor prognostic research. In the
present cohort, preoperative LANI achieved an AUC of 0.792 for OS,
outperforming ALB alone (AUC=0.757) and the LMR alone (AUC=0.634).
Previous breast cancer studies have shown that commonly used
inflammatory or inflammatory-nutritional markers, such as NLR, SII,
and SIRI, typically yield AUC values between 0.58-0.63, whereas
composite scores such as NPS, PLR or NLR-based prognostic models
frequently fall between 0.60-0.70 (28,52,53).
Within this framework, the AUC=0.792 of LANI lies toward the upper
end of the performance spectrum reported for similar indices and
reflects a comparatively strong discriminatory ability in the TNBC
setting. Although isolated studies have described higher AUCs, such
as an NLR AUC=0.827 in an overall breast cancer population
(54), these findings were
influenced by differences in study population and the incorporation
of tumor markers, limiting direct comparison. Collectively, these
observations position LANI as a high-performing, biologically
grounded inflammatory-nutritional index with potential utility for
prognostic refinement in TNBC.
Although neutrophil, lymphocyte, monocyte, platelet
and ALB values were available for all patients in the present
study, a number of biochemical indicators required for
reconstructing mGPS, PNI or CONUT, specifically C-reactive protein,
total protein and total cholesterol, were not consistently
documented. As the present study was retrospective and relied
entirely on electronic medical records, these parameters were only
obtained when clinicians suspected inflammatory activity or
metabolic-nutritional abnormalities, resulting in marked
missingness and precluding reconstruction of those indices. Despite
this limitation, the prognostic performance of LANI remained
comparable with and in some instances higher than that reported for
established inflammatory-nutritional markers, suggesting the
potential advantages of integrating immune and nutritional
components into a unified index. As LANI was newly introduced in
the present study, its prognostic utility has not been evaluated
previously in breast cancer or other malignancies. Future
prospective TNBC cohorts with complete biochemical datasets will be
key in reconstructing established indices and enabling direct,
head-to-head comparisons of ROC-based and C-index performances to
precisely determine the clinical positioning of LANI.
By jointly incorporating serum ALB and the LMR, LANI
consolidates nutritional and immune status into a quantitative
surrogate of immunometabolic exhaustion. Patients with LANI=0,
defined by concurrent hypoalbuminemia and low LMR, exhibit a
high-risk host phenotype marked by chronic inflammation, catabolic
metabolic remodeling, lymphocyte depletion and myeloid-dominant
immunosuppression. This phenotype aligns with recent studies
demonstrating that ALB-based inflammatory ratios and
lymphocyte-derived indices (including NPAR and PLR) predict distant
metastasis and unfavorable survival in breast cancer (50,55).
In TNBC, such immunometabolic exhaustion frequently coexists with
an immunosuppressive tumor microenvironment characterized by
M2-type macrophage enrichment, insufficient CD8+ T-cell
infiltration and elevated expression of checkpoint molecules
including programmed cell death protein 1/programmed death-ligand
1, T cell immunoglobulin and mucin domain-containing 3 and
lymphocyte activation gene 3, all of which promote immune evasion
and diminish therapeutic benefit (56–59).
Inflammatory cytokines, tumor-infiltrating lymphocytes and immune
checkpoint molecules were not analyzed in the present study, as
owing to the retrospective design, such measurements were not part
of the routine assessment at the time of patient enrollment.
Consequently, long-term archived serum or tissue specimens suitable
for multiplex cytokine testing or immune profiling were not
consistently available, precluding reliable quantification of these
biomarkers. Nevertheless, the clinical patterns observed in the
present cohort, including the significantly worse OS in patients
with low LANI and its association with more advanced
clinicopathological features, are consistent with established
translational evidence suggesting that IL-6/TNF-α-driven
inflammation, M2 macrophage polarization and CD8+ T-cell
dysfunction shape an immunosuppressive TNBC microenvironment and
contribute to adverse outcomes (60). These consistencies suggest that LANI
may capture an underlying immunometabolic program integral to tumor
aggressiveness and treatment responsiveness. Future investigations
should therefore aim to incorporate LANI-stratified immune
analyses, including: i) Multiplex immunohistochemistry to assess
CD8, CD68 and CD163 expression and characterize TIL density and
tumor-associated macrophage polarization; ii) serum multiplex
cytokine profiling of IL-6, TNF-α and IL-10; and iii) single-cell
or spatial transcriptomic profiling to delineate immunometabolic
niches across LANI groups (61–63).
Through such integrative approaches, LANI may evolve from a
pragmatic clinical index into a translational bridge associating
routine hematologic markers with high-dimensional immune-molecular
phenotyping, supporting its development as a mechanistically
grounded prognostic and potentially predictive biomarker in
TNBC.
In addition, a number of methodological
considerations should be acknowledged when interpreting the present
findings. The present study was retrospective and single-center in
design, with a modest sample size and a relatively limited number
of outcome events, which inherently restricts the generalizability
of the results. Although internal validation supported the
stability of the model, the absence of external, multicenter
validation precludes a full assessment of its reproducibility and
robustness in broader TNBC populations. Furthermore, despite
multivariable adjustment, residual confounding cannot be fully
excluded, particularly because comorbidities, inflammatory
conditions and medication exposures that could influence ALB,
lymphocyte or monocyte measurements were not systematically
documented. More specifically, serum ALB may be influenced by
unrecognized hepatic dysfunction (including steatosis, chronic
hepatitis or impaired synthetic capacity), renal protein loss,
inflammatory cytokine-driven acute-phase responses, perioperative
hemodilution from intravenous fluids, malnutrition, catabolic
illness and less commonly, protein-losing enteropathy (64,65).
In addition, lymphocyte and monocyte counts may fluctuate with
acute or chronic infections, autoimmune or inflammatory disorders,
hematologic abnormalities, physiologic stress responses and prior
or undocumented exposure to immunosuppressive medications or
corticosteroids. Given that such factors were not consistently
captured in the present retrospective dataset, their potential
confounding effects could not be fully assessed. Furthermore,
although G2 tumors exhibited an intermediate risk in the
multivariate analysis, the modest effect size and limited number of
events necessitate cautious interpretation and further validation
in larger cohorts. Similarly, the low HR observed in the LANI=2
subgroup must be viewed in the context of event sparsity, as only a
single mortality occurred within this category in each cohort,
which inevitably broadened the CIs, reflecting statistical
limitations rather than model instability. Despite this, the
protective direction of association remained consistent across all
cohorts and sensitivity analyses, supporting the biological
plausibility of LANI as an indicator of host resilience. The
integrated nomogram combining LANI, T stage and histologic grade
demonstrated strong discrimination, favorable calibration and
meaningful clinical utility, yet these prognostic properties also
require validation in larger, more event-rich populations.
Collectively, these sources of bias are likely non-differential in
nature and would tend to attenuate, rather than exaggerate, the
observed associations, suggesting that the estimated effect of LANI
may, if anything, be conservative.
Future prospective, large-scale, multicenter studies
with standardized documentation of comorbidities, inflammatory and
nutritional biomarkers and medication exposures will be key in
validating these findings across heterogeneous TNBC populations.
Incorporating serial measurements will allow characterization of
dynamic immuno-nutritional trajectories throughout the
perioperative period and adjuvant therapy. Further integration of
LANI with TNBC molecular subtyping, genomic and immunogenomic
features and emerging multi-omics signatures, may deepen
understanding of the immunometabolic mechanisms underlying
host-tumor interactions. Embedding LANI into clinical prediction
models or digital decision-support platforms may ultimately enhance
individualized risk stratification and guide perioperative and
systemic treatment planning within precision oncology frameworks.
Overall, LANI represents a simple, accessible and biologically
grounded biomarker with promising utility in the preoperative
evaluation of TNBC, warranting further validation and clinical
translation in future multicenter prospective studies.
In conclusion, the present study demonstrates that
the LANI, derived from serum ALB and the LMR, is a reliable and
independent predictor of OS in patients with TNBC. LANI reflects
the combined influence of nutritional status and systemic
immune-inflammatory balance, enabling clearer preoperative risk
stratification compared with either component alone. The LANI-based
nomogram showed strong discrimination, good calibration and
meaningful clinical benefits in both the training and validation
cohorts. These findings support LANI as a simple and reproducible
tool that may complement conventional pathological parameters and
assist individualized perioperative management of TNBC.
Acknowledgements
Not applicable.
Funding
The present study was supported by The National Natural Science
Foundation of China (grant no. 82260549).
Availability of data and materials
The data generated in the present study may be
requested from the corresponding author.
Authors' contributions
BM contributed to the conception and design of the
present study. BZ, KW, MA and FL collected and analyzed data. BZ
and KW wrote and revised the manuscript. BZ and FL confirm the
authenticity of all the raw data. All authors read and approved the
final version of the manuscript.
Ethics approval and consent to
participate
The present study was approved by The Ethics
Committee of the Affiliated Cancer Hospital of Xinjiang Medical
University (approval no. K-2024056) in accordance with the
Declaration of Helsinki. All patients signed a written informed
consent form, which included consent to participate in the present
study, use of their medical data for research purposes and the
publication of anonymized findings.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Dogra AK, Prakash A, Gupta S and Gupta M:
Prognostic significance and molecular classification of triple
negative breast cancer: A systematic review. Eur J Breast Health.
21:101–114. 2025.PubMed/NCBI
|
|
2
|
Bray F, Laversanne M, Sung H, Ferlay J,
Siegel RL, Soerjomataram I and Jemal A: Global cancer statistics
2022: GLOBOCAN estimates of incidence and mortality worldwide for
36 cancers in 185 countries. CA Cancer J Clin. 74:229–263.
2022.PubMed/NCBI
|
|
3
|
Chen Z, Liu Y, Lyu M, Chan CH, Sun M, Yang
X, Qiao S, Chen Z, Yu S, Ren M, et al: Classifications of
triple-negative breast cancer: insights and current therapeutic
approaches. Cell Biosci. 15:132025. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Kannan K, Srinivasan A, Kannan A and Ali
N: The underlying mechanisms and emerging strategies to overcome
resistance in breast cancer. Cancers (Basel). 17:29382025.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Martorana TM, Ricciardi G, Tralongo P,
Fiorentino V, Pizzimenti C, Franchina M, Marino MA, Santarpia M,
Tuccari G, Ieni A, et al: Targeting the tumor microenvironment in
triple-negative breast cancer: Biological insights and therapeutic
opportunities. Pathol Res Pract. 275:1562262025. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Chen Y, Anwar M, Wang X, Zhang B and Ma B:
Integrative transcriptomic and single-cell analysis reveals IL27RA
as a key immune regulator and therapeutic indicator in breast
cancer. Discov Oncol. 16:9772025. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Chen H, Gui X, Zhou Z, Su F, Gong C, Li S,
Wu W, Rao N, Liu Q and Yao H: Distinct ER and PR expression
patterns significantly affect the clinical outcomes of early
HER2-positive breast cancer: A real-world analysis of 871 patients
treated with neoadjuvant therapy. Breast. 75:1037332024. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Li Y, Zhang H, Merkher Y, Chen L, Liu N,
Leonov S and Chen Y: Recent advances in therapeutic strategies for
triple-negative breast cancer. J Hematol Oncol. 15:1212022.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Jiang C, Zhang S, Jiang L, Chen Z, Chen H,
Huang J, Tang J, Luo X, Yang G, Liu J and Chi H: Precision
unveiled: Synergistic genomic landscapes in breast
cancer-Integrating single-cell analysis and decoding drug toxicity
for elite prognostication and tailored therapeutics. Environ
Toxicol. 39:3448–3472. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Zhou Y, Tao L, Qiu J, Xu J, Yang X, Zhang
Y, Tian X, Guan X, Cen X and Zhao Y: Tumor biomarkers for
diagnosis, prognosis and targeted therapy. Signal Transduct Target
Ther. 9:1322024. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
He M, Zhang JX, Jiang YZ, Chen YL, Yang
HY, Tang LC, Shao ZM and Di GH: The lymph node ratio as an
independent prognostic factor for node-positive triple-negative
breast cancer. Oncotarget. 8:44870–44880. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Altea-Manzano P, Decker-Farrell A,
Janowitz T and Erez A: Metabolic interplays between the tumour and
the host shape the tumour macroenvironment. Nat Rev Cancer.
25:274–292. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Li B, Lin R, Hua Y, Ma B and Chen Y:
Single-cell RNA sequencing reveals TMEM71 as an immunomodulatory
biomarker predicting immune checkpoint blockade response in breast
cancer. Discov Oncol. 16:12562025. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Tripathi S, Sharma Y and Kumar D:
Unveiling the link between chronic inflammation and cancer. Metabol
Open. 25:1003472025. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Hassan AF, Kheraldine H, Abujamous L,
Al-Thawadi H and Elhissi A: Inflammation-associated drug resistance
and tumor growth in TNBC. Front Immunol. 16:16231372025. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Hua S, Wang W, Yao Z, Gu J, Zhang H, Zhu
J, Xie Z and Jiang H: The fatty acid-related gene signature
stratifies poor prognosis patients and characterizes TIME in
cutaneous melanoma. J Cancer Res Clin Oncol. 150:402024. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Li KJ, Zhang ZY, Wang K, Sulayman S, Zeng
XY, Liu J, Chen Y and Zhao ZL: Prognostic scoring system using
inflammation- and nutrition-related biomarkers to predict prognosis
in stage I-III colorectal cancer patients. World J Gastroenterol.
31:1045882025.PubMed/NCBI
|
|
18
|
Zhang M, Liu J, Zhang F, Liang Q and Guo
Z: Comprehensive characterization of neddylation related genes in
cutaneous melanoma identified a novel prognostic signature for
treatment outcomes, immune landscape. Discov Oncol. 15:7222024.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Majumdar A, Saraf SK, Sahu C, Verma K and
Vishwakarma P: Current perspectives on malnutrition and
immunomodulators bridging nutritional deficiencies and immune
health. Future J Pharm Sci. 11:502025. View Article : Google Scholar
|
|
20
|
Kanbur B, Unek IT, Uzun M, Ozturk C, Yarol
RC and Balci A: Association of systemic inflammatory response index
and prognostic nutritional index scores with sarcopenia in patients
with metastatic gastric cancer. Medicina (Kaunas). 61:7852025.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Shayimu P, Awula M, Wang CY, Jiapaer R,
Pan YP, Wu ZM, Chen Y and Zhao ZL: Serum nutritional predictive
biomarkers and risk assessment for anastomotic leakage after
laparoscopic surgery in rectal cancer patients. World J
Gastrointest Surg. 16:3142–3154. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Stotz M, Gerger A, Eisner F, Szkandera J,
Loibner H, Ress AL, Kornprat P, AlZoughbi W, Seggewies FS, Lackner
C, et al: Increased neutrophil-lymphocyte ratio is a poor
prognostic factor in patients with primary operable and inoperable
pancreatic cancer. Br J Cancer. 109:416–421. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Chen Y, Zhang B, Wang X, Chen Y, Anwar M,
Fan J and Ma B: Prognostic value of preoperative modified Glasgow
prognostic score in predicting overall survival in breast cancer
patients: A retrospective cohort study. Oncol Lett. 29:1802025.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Lee YK, Hsu CY, Wang MY, Kuo WH, Lien HC,
Lee YH, Lo C and Huang CS: Prognostic factors for early-stage
triple-negative breast cancer with a focus on
neutrophil-to-lymphocyte ratio: A single-center retrospective
study. Taiwan J Obstet Gynecol. 64:1030–1036. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Alzogaray V, Defante MLR, Celso DSG,
Torres LA, Bearse M and Lopes ACFMM: Prognostic significance of
platelet-to-lymphocyte ratio in patients with triple-negative
breast cancer: Systematic review and meta-analysis. Cancer Diagn
Progn. 5:779–787. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Zhao J, Chen X, Zhang Z, Zhao D, Cao X,
Mao F, Sun Q and Peng L: Prognostic value of the systemic
immune-inflammation index in recurrent/metastatic triple-negative
breast cancer: A retrospective cohort study. Gland Surg.
14:1858–1872. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Guo Q, Wang B, Gao X, Zhao P and Lv S:
Predicting prognosis of patients with triple-negative breast cancer
undergoing neoadjuvant chemotherapy based on inflammatory status at
different time points: A propensity score matching analysis. Oncol
Lett. 29:2522025. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Qiu Y, Chen Y, Shen H, Yan S, Li J and Wu
W: Naples prognostic score: A novel predictor of survival in
patients with triple-negative breast cancer. J Inflamm Res.
17:5253–5269. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Kotowicz B, Fuksiewicz M, Jodkiewicz M,
Makowka A and Jagiełło-Gruszfeld A: Evaluation of the prognostic
value of obesity, vitamin d concentrations, and systemic
inflammatory response indexes (SIRI, SII, PIV) in patients with
breast cancer scheduled for neoadjuvant treatment. Breast Cancer
(Auckl). 19:117822342513694632025. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Zhu J, Cheng J, Ma Y, Wang Y, Zou Z, Wang
W, Shi H and Meng Y: The value of inflammation-related indicators
in chemotherapy efficacy and disease-free survival of
triple-negative breast cancer. Eur J Med Res. 30:772025. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Eckart A, Struja T, Kutz A, Baumgartner A,
Baumgartner T, Zurfluh S, Neeser O, Huber A, Stanga Z, Mueller B
and Schuetz P: Relationship of nutritional status, inflammation,
and serum albumin levels during acute illness: A prospective study.
Am J Med. 133:713–722.e7. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Gremese E, Bruno D, Varriano V, Perniola
S, Petricca L and Ferraccioli G: Serum albumin levels: A biomarker
to be repurposed in different disease settings in clinical
practice. J Clin Med. 12:60172023. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Wang K, Li K, Zhang Z, Zeng X, Wu Z, Zhang
B, Pan Y, Lau LY, Zhao Z and Chen Y: Combined preoperative
platelet-albumin ratio and cancer inflammation prognostic index
predicts prognosis in colorectal cancer: A retrospective study. Sci
Rep. 15:295002025. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Benner B, Scarberry L, Suarez-Kelly LP,
Duggan MC, Campbell AR, Smith E, Lapurga G, Jiang K, Butchar JP,
Tridandapani S, et al: Generation of monocyte-derived
tumor-associated macrophages using tumor-conditioned media provides
a novel method to study tumor-associated macrophages in vitro. J
Immunother Cancer. 7:1402019. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Zhang H, Tang S, Biskup E, Zhang Y, Yong
L, Chen L and Cai F: Long-term survival after diverse therapeutic
modalities in malignant phyllodes tumors of the breast. Technol
Cancer Res Treat. 21:153303382211210862022. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Tokumaru Y, Oshi M, Murthy V, Tian W, Yan
L, Angarita FA, Nagahashi M, Matsuhashi N, Futamura M, Yoshida K,
et al: Low intratumoral genetic neutrophil-to-lymphocyte ratio
(NLR) is associated with favorable tumor immune microenvironment
and with survival in triple negative breast cancer (TNBC). Am J
Cancer Res. 11:5743–5755. 2021.PubMed/NCBI
|
|
37
|
Bae SJ, Cha YJ, Yoon C, Kim D, Lee J, Park
S, Cha C, Kim JY, Ahn SG, Park HS, et al: Prognostic value of
neutrophil-to-lymphocyte ratio in human epidermal growth factor
receptor 2-negative breast cancer patients who received neoadjuvant
chemotherapy. Sci Rep. 10:130782020. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Sun H, Liang J, Xue S, Zhang X, Ding M,
Zhu J, Nanding A, Liu T, Lou G, Gao Y, et al: Establishment and
clinical application of a prognostic index for inflammatory status
in triple-negative breast cancer patients undergoing neoadjuvant
therapy using machine learning. BMC Cancer. 24:15592024. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Giro A, Passildas-Jahanmohan J, Kossai M,
Bidet Y, Molnar I, Bernadach M, Penault-Llorca F, Abrial C, Durando
X and Radosevic-Robin N: Comparison of the predictive and
prognostic capacities of neutrophil, lymphocyte and platelet counts
and tumor-infiltrating lymphocytes in triple-negative breast
cancer: Preliminary RESULTS of the PERCEPTION study. Anticancer
Res. 44:4983–4994. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Fang Q, Jiang H, Deng L, Sun W, Wang M,
Liu Y and Xu J: Constructing and evaluating a predictive model for
the risk of preoperative frailty in breast cancer patients. BMC
Womens Health. 25:5832025. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Miao Y, Yang R, Zhang B, Yang J, Yao L,
Wang W, Liu X, Guo X and Jia H: Naples prognostic score (NPS) as a
novel prognostic score for stage III breast cancer patients: A
real-world retrospective study. Breast Cancer (Dove Med Press).
17:403–421. 2025.PubMed/NCBI
|
|
42
|
Hou M, Li H, He T, Hui S, Dai W, Hou X,
Zhao J, Zhao J, Wen J, Kan W, et al: Icariside I reduces breast
cancer proliferation, apoptosis, invasion, and metastasis probably
through inhibiting IL-6/STAT3 signaling pathway. J Pharm Pharmacol.
76:499–513. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Vu NT, Vu QV, Vo NT, Tanigaki R, Quach HT,
Miyake Y, Shiba T and Kataoka T: Cardamonin inhibits the nuclear
translocation and DNA binding of RelA in the tumor necrosis
factor-α-induced NF-κB signaling pathway in human lung
adenocarcinoma A549 cells. Molecules. 30:43242025. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Ma H, Liu J, Jin H, Zhang M, Liang Q and
Guo Z: Comprehensive characterization of NK cell-related genes in
cutaneous melanoma identified a novel prognostic signature for
predicting the prognosis, immunotherapy, and chemotherapy efficacy.
Discov Oncol. 16:12432025. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Liu Y, Luo R, Wu Z, Zhang W, Liang C, Liu
Z and Zhao H: Association of neutrophil percentage-to-albumin ratio
with distant metastasis and survival outcomes in breast cancer
patients: A retrospective and prospective cohort study. Sci Rep.
15:454562025. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Gruys E, Toussaint MJ, Niewold TA and
Koopmans SJ: Acute phase reaction and acute phase proteins. J
Zhejiang Univ Sci B. 6:1045–1056. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Shang Y, Duan Y, Liu J, Guo R, Fang Z, Ma
F and Guo B: Association between albumin-related inflammatory
biomarkers and breast cancer risk: A secondary analysis of NHANES
1998–2018. Medicine (Baltimore). 104:e464822025. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Zhang R, Meng Z, Wu X, Piao Z, Jin T and
Zhang M: MIR4435-2HG promotes breast cancer evolution through
miR-205-5p/UBE2N axis and exosome-mediated macrophage M2-like
polarization. Cell Signal. 139:1122872026. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
He S, Wang Q, He J, Chen Z and Xiao Y: The
Leupaxin/ HDAC6/EGR2 axis facilitates breast cancer progression by
enhancing macrophage M2 polarization. Cell Biol Toxicol.
41:1542025. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Zhang J, Huang X, Ma L, Chen Z, Li T, Wang
L, Guo Y, Xu H, Li J, Qin JJ, et al: Micro-organ chip deciphers
tumor-derived G-CSF as remote commander of lung pre-metastatic
niche via VEGFA-KDR cascade. Adv Sci (Weinh). e185842025.(Epub
ahead of print). PubMed/NCBI
|
|
51
|
Gao S, Tang W, Zuo B, Mulvihill L, Yu J
and Yu Y: The predictive value of neutrophil-to-lymphocyte ratio
for overall survival and pathological complete response in breast
cancer patients receiving neoadjuvant chemotherapy. Front Oncol.
12:10656062023. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Zhang Y, Wu J, Chen W and Liang X:
Pretreatment system inflammation response index (SIRI) is a
valuable marker for evaluating the efficacy of neoadjuvant therapy
in breast cancer patients. Int J Gen Med. 17:4359–4368. 2024.
View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Li Y and Fei Y: The predictive value of
serum NLR, SII, and OPNI for lymph node metastasis in breast cancer
patients with internal mammary lymph nodes after thoracoscopic
surgery. Open Life Sci. 19:202207632024. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Fan S, Xie X, Shen Y, Wang W, Gu X and Yao
Z: The predictive value of preoperative serum
neutrophil-to-lymphocyte ratio and tumor markers for early breast
cancer patients: A retrospective study. Medicine (Baltimore).
101:e300112022. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Zhao Z, Xu H, Ma B and Dong C: Prognostic
value of platelet to lymphocyte ratio (PLR) in breast cancer
patients receiving neoadjuvant therapy: A systematic review and
meta-analysis. Front Immunol. 16:16585712025. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Tan Z, Xin H, Chen J, Lei M, Tu G and Tang
L: SIGLEC15 modulates the immunosuppressive microenvironment and
suppresses malignant phenotypes in triple-negative breast cancer.
Genes Dis. 13:1017992025. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Liu J, Wang YT, Zeng LL and Chen CL:
Lactate-related genes signature as a novel prognostic landscape in
laryngeal squamous cell carcinoma: Insights from 156 machine
learning algorithms and in vitro validation. Int J Biol Macromol.
323:1470982025. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Zhang P, Zhang H, Tang J, Ren Q, Zhang J,
Chi H, Xiong J, Gong X, Wang W, Lin H, et al: The integrated
single-cell analysis developed an immunogenic cell death signature
to predict lung adenocarcinoma prognosis and immunotherapy. Aging
(Albany NY). 15:10305–10329. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Imani S, Farghadani R, Roozitalab G,
Maghsoudloo M, Emadi M, Moradi A, Abedi B and Jabbarzadeh Kaboli P:
Reprogramming the breast tumor immune microenvironment: Cold-to-hot
transition for enhanced immunotherapy. J Exp Clin Cancer Res.
44:1312025. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Li Y, Gao Y, Wang L and Si J: NK cell
adaptation in the tumor microenvironment: Insights for NK
cell-based immunotherapy. Cytokine Growth Factor Rev. 86:181–198.
2025. View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Chen X, Chi H, Zhao X, Pan R, Wei Y and
Han Y: Role of exosomes in immune microenvironment of
hepatocellular carcinoma. J Oncol. 2022:25210252022.PubMed/NCBI
|
|
62
|
Xiang Y, Du D, Su Y, Guo L and Chen S:
Integrated pan-cancer analysis and experimental verification of the
roles of retinoid-binding proteins in breast cancer. Cancers
(Basel). 17:37062025. View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Mardamshina M, Karagach S, Mohan V, Arad
G, Necula D, Golani O, Fellus-Alyagor L, Shenoy A, Krol K, Pirak D,
et al: Integrated spatial proteomic analysis of breast cancer
heterogeneity unravels cancer cell phenotypic plasticity. Nat
Commun. 16:104822025. View Article : Google Scholar : PubMed/NCBI
|
|
64
|
Cheng T, Wang X, Han Y, Hao J, Hu H and
Hao L: The level of serum albumin is associated with renal
prognosis and renal function decline in patients with chronic
kidney disease. BMC Nephrol. 24:572023. View Article : Google Scholar : PubMed/NCBI
|
|
65
|
Wiedermann CJ: Hypoalbuminemia as
surrogate and culprit of infections. Int J Mol Sci. 22:44962021.
View Article : Google Scholar : PubMed/NCBI
|