Introduction
Vasculogenic mimicry (VM), first described in 1999,
refers to the ability of highly aggressive melanoma cells to
generate fluid-conducting channels independent of the vascular
network of the tumor. In this distinctive phenomenon, tumor cells
imitate endothelial cells in a way that promotes tumor growth,
invasion and metastasis.
VM exhibits certain key characteristics: i) Staining
patterns. VM channels stain negative for vascular endothelial
markers such as CD34/CD31 but positive for periodic acid-Schiff,
which is specific to the extracellular matrix (ECM); ii) absence of
endothelial cells. Unlike conventional blood vessels, these
VM-derived channels lack vascular endothelial cells; iii) ECM
remodeling. These channels are essentially microcirculation
pathways reshaped by the ECM; and iv) interconnection with tumor
micro-vessels. VM channels connect with the micro-vessels of the
tumor, allowing for blood flow. Furthermore, the relatively thin
walls of these VM channels provide a conducive environment for
tumor cell infiltration and extravasation, bolstering the migratory
and metastatic potential of tumor cells. The presence of VM often
predicts a highly aggressive tumor and a worse prognosis (1). Since VM does not depend on traditional
angiogenic pathways, anti-angiogenic drugs, such as sorafenib and
bevacizumab, have a limited effect on VM-forming tumors,
potentially affecting tumor response to therapy (2).
Despite the pivotal role of anti-angiogenic
therapies in cancer management, their clinical efficacy remains
markedly limited by VM-mediated resistance. Current anti-VEGF
strategies frequently fail to target these VM channels due to their
distinct molecular regulation, creating an urgent need for
innovative therapeutic approaches. Emerging evidence positions
Traditional Chinese Medicine (TCM) monomers as multifaceted agents
capable of concurrently addressing angiogenesis and VM (3). The present systematic analysis of
preclinical studies revealed that TCM monomers demonstrate
cross-cancer efficacy in VM suppression. This multi-target capacity
circumvents resistance mechanisms and may synergize with existing
anti-angiogenic regimens.
Search and selection criteria
Web of Science (https://www.webofscience.com/), PubMed (https://pubmed.ncbi.nlm.nih.gov/) and China
National Knowledge Infrastructure (CNKI; http://www.cnki.net/) databases, employed as mining
tools, were searched for the following terms: [‘vasculogenic
mimicry’ in the database (title/abstract) and (extracted
compounds)] and [‘neoplasms’ or all its synonyms (cancer) in the
database (title/abstract)]. Publications up to April 1, 2025, were
imported into Zotero (version 6.0; Hongzhou Intelligent Technology
Co., Ltd.). After removing duplicates, 2,358 initial records were
screened, yielding 64 eligible studies based on the following
criteria: i) In vitro/in vivo experiments; ii) VM
quantification; and iii) TCM monomer intervention. Case reports and
reviews were excluded.
Inhibition of multiple cancer VM processes
by herbal monomers
The mechanisms by which various TCM monomers inhibit
VM formation in malignant tumors were summarized (Fig. 1).
These monomers potentially inhibit the
aggressiveness and metastasis of liver and breast cancer, as well
as other solid tumors, by disrupting the VM-associated tumor
microenvironment. Their multi-target mechanism of action enhances
chemotherapy sensitivity and reduces drug resistance. Due to their
natural origin, these compounds offer advantages such as low
toxicity and high efficacy, providing a novel strategy for the
treatment of tumors. Fig. 2
displays the role of herbal compounds in the formation of VM.
Table I summarizes the anti-VM
mechanisms of Chinese herbal components across different cancer
types.
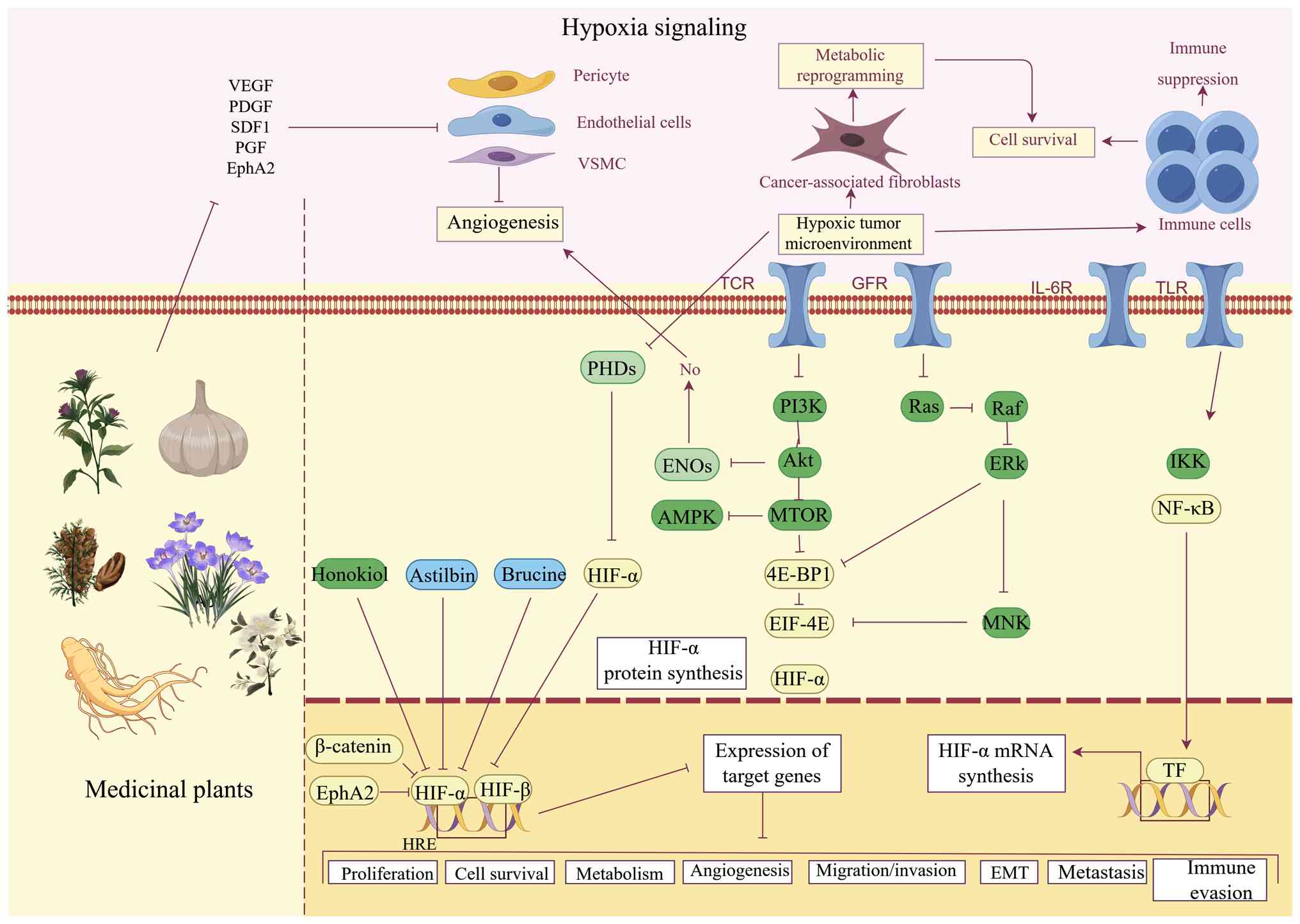 | Figure 2.Mechanisms by which herbal compounds
inhibit VM by affecting hypoxia signaling pathways and their
associated molecules. Under hypoxic conditions, PHDs activity is
inhibited, leading to enhanced stability of HIF-α protein and
formation of a complex with HIF-β, which activates downstream
target genes (e.g., VEGF, PDGF and SDF1), driving angiogenesis,
metabolic reprogramming, cell survival and immune suppression. In
addition, hypoxic conditions can activate PI3K/AKT/mTOR,
Ras/Raf/ERK, AMPK-mTOR and NF-κB axes to promote HIF-α synthesis
(e.g., blocking HIF-α degradation) and activate downstream effector
molecules (e.g., VEGF monoclonal antibody or receptor inhibitor),
and a variety of compounds isolated from herbs can reverse this
process by blocking the relevant signaling pathways and their
associated factors. VM, vasculogenic mimicry; PHDs, prolyl
hydroxylases; HIF, hypoxia inducible factor; PDGF, platelet-derived
growth factor; SDF1, stromal cell-derived factor-1; AMPK,
AMP-activated protein kinase; EMT, epithelial-mesenchymal
transition; EphA2, ephrin type-A receptor 2; VSMC, vascular smooth
muscle; TF, transcription factor; ENOs, endothelial nitric oxide
synthase; HRE, hypoxia response element; TCR, T cell receptor; GFR,
glomerular filtration rate; TLR, target lesion revascularization;
PGF, placental growth factor. |
 | Table I.Specific molecular mechanisms
underlying the inhibition of VM in multiple cancer types by TCM
monomers. |
Table I.
Specific molecular mechanisms
underlying the inhibition of VM in multiple cancer types by TCM
monomers.
| TCM monomer | Cancer models | Concentrations | Model | Key targets | Effects | (Refs.) |
|---|
| Melittin | SMMC7721, Huh7 | 2 and 4 µg/ml; | In
vitro | HIF-1α↓,
p-AKT↓, | Inhibiting VM | (10) |
|
| and Hep G2
cells | 24 h |
| VEGF↓, MMP-2↓
and | formation |
|
|
|
|
|
| MMP-9↓ |
|
|
|
| Male BALB/c
nude | 50 and 100
g/kg/d | In vivo |
|
|
|
|
| mice |
|
|
|
|
|
| Myricetin | HCC cells | 80 µmol/l; | In
vitro | E-cadherin↓, | Inhibiting HCC
cells | (12,13) |
|
|
| 48 h |
| VEGFR1↓ and | invasion,
metastasis, |
|
|
|
|
|
| VEGFR2↓ | VM formation
and |
|
|
|
|
|
|
| angiogenesis |
|
|
| Female BALB
mice | 15 and | In vivo |
|
|
|
|
|
| 30 mg/kg/d |
|
|
|
|
| Daurisoline | HCC cells | 5, 10 and | In
vitro | AKT↓,
ERK-p38↓, | Inducing
apoptosis, | (14) |
|
|
| 20 µmol/l; |
| RhoA↓ and
ROCK2↓ | inhibiting VM
and |
|
|
|
| 48 h |
|
| improving drug |
|
|
|
|
|
|
| sensitivity |
|
|
| Male BALB/c
nude | 20 mg/kg/d | In vivo |
|
|
|
|
| mice |
|
|
|
|
|
| Ethanolic extract
of | HepG2 cells | 75 and 125
µg/ml; | In
vitro | VEGFA↓, MMP-2↓ | Inhibiting
cell | (16) |
| Elephantopus
scaber |
| 48 h |
| and MMP-9↓ | proliferation, |
|
|
|
|
|
|
| migration and
VM |
|
|
Celastrusorbiculatus | MHCC97-H cells | 80 µg/ml; | In
vitro | MMP2↓, MMP9↓, | Inhibiting VM | (18) |
| extraction | and HepG2
cells | 48 h |
| Twist1↓ and
EphA2↓ | formation |
|
|
| Male BALB/c
nude | - | In vivo |
|
|
|
|
| mice |
|
|
|
|
|
| Baicalein | A549 cells | 60 µmol/l; | In
vitro | VE-CAD↓,
EphA2↓, | Inhibiting VM
formation | (22) |
|
|
| 72 h |
| MMP14↓, MMP2↓, |
|
|
|
| Male BALB/c
nude | 10 mg/kg/d | In vivo | MMP9↓ and
PI3K↓ |
|
|
|
| mice |
|
|
|
|
|
| Lycorine | SPC-A-1 and
A549 | 60 µmol/l; | In
vitro | SAV1↑ | Inhibiting
cell | (24) |
|
| cells | 72 h |
|
| proliferation, |
|
|
| Female nude
mice | 10 mg/kg/d | In vivo |
| migration and
VM |
|
| Curcumin | A549 cells | 10 µg/ml; | In
vitro | MMP-2↓ and
HIF-1α↓ | Inhibiting VM | (26) |
|
|
| 48 h |
|
| formation |
|
|
| Male BALB/c
nude | 150 µg/kg/d | In vivo |
|
|
|
|
| mice |
|
|
|
|
|
|
Tetramethylpyrazine | A549 cancer
stem- | 100 and | In
vitro | HGF↓ and
c-Met↓ | Inhibiting stem
cell- | (28) |
|
| like cells | 400 µmol/l; |
|
| like cell VM |
|
|
|
| 24 and 48 h |
|
| formation |
|
|
Dihydroartemisinin | A549 and H3255 | 50 µmol/l; | In
vitro | E-cadherin↑, | Inhibiting
cell | (32) |
|
| cells | 24 and 48 h |
| N-cadherin↓
and | growth,
proliferation, |
|
|
|
|
|
| VE-CAD↓ | migration and
VM |
|
|
|
|
|
|
| formation |
|
| Salvianolic acid
A | A549 and H1299 | 50 µmol/l; | In
vitro | EphA2↓,
VE-CAD↓, | Inhibiting
cell | (33) |
|
| cells | 24 h |
| p-PI3K↓,
p-AKT↓, | invasion and
VM |
|
|
|
|
|
| p-mTOR↓ and
MMP2↓ | formation |
|
| Matrine | CT26 SW480 and | 4 mmol/l; | In
vitro | JNK↓ and ERK↓ | Inhibiting VM | (42) |
|
| KM12 cells | 24 h |
|
| formation |
|
| Amentoflavone | HCT-116 cells | 150 µmol/l; | In
vitro | HIF-1α↓,
β-catenin↓, | Inhibiting
cell | (44) |
|
|
| 24 h |
| VEGF↓,
vimentin↓ | proliferation
and |
|
|
|
|
|
| and Snail↓ | VM formation |
|
| Delphinidin | SW620 cells | 180 µmol/l; | In
vitro | RhoA↓, ROCK↓
and | Inhibiting VM
and | (45) |
|
|
| 48 h |
| VEGF↓ | inducing
apoptosis |
|
| Evodiamine | HCT-116 cells | 0.375, 0.75
and | In
vitro | HIF-1α↓,
VE-CAD↓, | Inhibiting
tumor | (47) |
|
|
| 1.5 µmol/l; |
| VEGF↓, MMP2↓
and | growth and |
|
|
|
| 24 h |
| MMP9↓ | VM formation |
|
|
| Female BALB/c | 10 mg/kg/d | In vivo |
|
|
|
|
| nude mice |
|
|
|
|
|
| Saffronin | NCI-N87 and
Hs- | 100 µmol/l; | In
vitro | HIF-1α↓,
Notch1↓, | Inhibiting VM | (53) |
|
| 746T GC cells | 24 h |
| SHH↓, PTCH2↓
and | formation |
|
|
|
|
|
| Gli1↓ |
|
|
|
Dihydroartemisinin | HGC-27 and
SGC- | 320 µmol/l; | In
vitro | FGF2↓ and
FGFR1↓ | Inhibiting VM | (54) |
|
| 7901 cells | 24 and 48 h |
|
| formation |
|
|
| BALB/c thymus | 50 mg/kg/d | In vivo |
|
|
|
|
| nude mice |
|
|
|
|
|
| Formononetin | MGC-803 cells | 80 µmol/l; | In
vitro | HIF-1α↓ and
VEGF↓ | Inhibiting VM | (55) |
|
|
| 24 h |
|
| formation and |
|
|
|
|
|
|
| inducing
apoptosis |
|
| Ginsenoside
Rg3 | SGC7901 cells | 160 mg/l; | In
vitro | GSK-3β↓,
Wnt2B↓, | Inhibiting VM | (57) |
|
|
| 72 h |
| β-catenin↓,
MMP2↓ | formation |
|
|
|
|
|
| and MMP9↓ |
|
|
| Aucubin | MGC803 cells | 80 µmol/l; | In
vitro | RhoA↓, ROCK1↓, | Inhibiting EMT
and | (59) |
|
|
| 24 h |
| N-cadherin↓,
vimentin↓ | VM formation |
|
|
|
|
|
| and VE-CAD↓ |
|
|
| Jaceosidin | HGC-27 cells | 40 µmol/l; | In
vitro | SphK1↓ and
S1P↓ | Inhibiting VM | (60) |
|
|
| 24 h |
|
| formation |
|
| Artemether | BMVEC/U87
cells | 30 µmol/l; | In
vitro | HIF-1α↓,
MMP-2↓ | Inhibiting VM | (67) |
|
|
| 24 h |
| and PI3K↓ | formation and |
|
|
|
|
|
|
| inducing
apoptosis |
|
|
| ICR mice | 1 mg/kg/d | In vivo |
|
|
|
|
Isoliquiritigenin | SHG44 cells | 160 umol/l; | In
vitro | MMP-2↓, VEGF↓
and | Inhibiting VM | (72) |
|
|
| 24 h |
| EphA2↓ | formation |
|
| Moroidin | U87 cells and
U251 | 2 µmol/l; | In
vitro | p-ERK↓,
β-catenin↓ | Inhibiting EMT
and | (73) |
|
| cells | 24 h |
| and MMP-9↓ | VM formation |
|
| Rhizoma
Paridis | 143B and MG-63 | 1 µg/ml; | In
vitro | p-PI3K↓,
p-mTOR↓, | Inhibiting VM | (81,83) |
| saponins | cells | 24 h |
| MMP-2↓,
MMP-14↓, | formation and |
|
|
| Male BALB/c
nude | 100 mg/kg/d | In vivo | Ln5γ2↓ and
Ln5γ2×↓ | metastasis |
|
|
| mice |
|
|
|
|
|
| Curcumin | S180 cells | 20 µmol/l; | In
vitro | MMP-2↓ and
MMP-9↓ | Inhibiting VM | (82) |
|
|
| 72 h |
|
| formation |
|
| Artesunate | OCM-1 cells
and | 60 µmol/l; | In
vitro | HIF-1α↓, VEGF↓ and
PDGF↓ | Inhibiting VM | (89–92) |
|
| C918 cells | 24 h |
|
| formation |
|
| Luteolin | C918 and HUVEC | 25 µmol/l; | In
vitro | p-PI3K↓,
p-AKT↓ | Inhibiting VM
lumen | (93) |
|
| cells | 24 h |
| and VEGF↓ | formation |
|
| Brucine | MDA-MB-231
cells | 1 µmol/l; | In
vitro | MMP-2↓, MMP-9↓ | Disrupting the | (100) |
|
|
| 24 h |
| and EphA2↓ | cytoskeleton
and |
|
|
|
|
|
|
| inhibiting VM |
|
|
|
|
|
|
| formation |
|
| Triptonide | MDA-MB-231, | 10 nmol/l; | In
vitro | Twist1↓,
Notch1↓, | Inhibiting VM | (102) |
|
| MDA-MB-468 and | 72 h |
| VEGFR2↓ and | formation |
|
|
| BT-549 cells |
|
| VE-CAD↓ |
|
|
|
| NOD-SCID
female | 3 mg/kg/d | In vivo |
|
|
|
|
| mice |
|
|
|
|
|
| Astilbin | MCF-7 and MDA- | 300 µmol/l; | In
vitro | HIF-1α↓,
VEGF↓, | Inhibiting VM | (104) |
|
| MB-231 cells | 24 h |
| VE-CAD↓, | formation |
|
|
|
|
|
| N-cadherin↓, |
|
|
|
|
|
|
| MMP-2↓ |
|
|
| Ginsenoside
Rg3 | SW-1990 and
PCI- | 500 and | In
vitro | miR-204↑ and
DVL3↓ | Inhibiting VM | (111,112) |
|
| 35 cells | 750 µmol/l; 48
h |
|
| formation |
|
| Cinobufagin | SKOV3 cells | 7.5 µg/ml; | In
vitro | MMP9↓, MMP14↓ | Inhibiting | (116) |
|
|
| 24 h |
| and LAMC2↓ | macrophage |
|
|
| Female BALB/c | 1.5, 3 and | In vivo |
| polarization
and |
|
|
| nude mice | 6 mg/kg/d |
|
| inhibiting VM |
|
|
|
|
|
|
| formation |
|
| Catechins | SKOV3 and ES-2 | 100 ng/ml | In
vitro | MMP2 ↓ and | Inhibiting VM | (119) |
|
| cells | 24 h |
| TGF-β↑ | formation |
|
| Sinomenine | A2780 cells | 8 mmol/l; | In
vitro | VEGF↓, EphA2↓, | Inhibiting VM | (121) |
|
|
| 24 and 48 h |
| MMP-9↓,
MMP-2↓, | formation |
|
|
|
|
|
| CXCR4↓ and |
|
|
|
|
|
|
| p-STAT3↓ |
|
|
| Pristimerin | HeLa cells | 2.5 µmol/l; | In
vitro | SHH↓, Gli1
mRNA↓, | Inhibiting VM | (125) |
|
|
| 48 h |
| VEGF-A↓,
VE-CAD↓ | formation |
|
|
|
|
|
| and Gli1↓ |
|
|
| Honokiol | HeLa cells | 100 µg/ml; | In
vitro | p-EGFR↓,
MMP-2↓, | Inhibiting VM | (120) |
|
|
| 24 h |
| MMP-9↓,
EphA2↓, | formation |
|
|
|
|
|
| VEGF↓ and
CDH2↓ |
|
|
| Ligustilide | EC-109 cells | 200 µmol/l; | In
vitro | Cyclin-D1↓,
Bcl-2↓, | Inhibiting VM | (124) |
|
|
| 24 h |
| RhoA↓, ROCK↓, | formation |
|
|
|
|
|
| p21↑, Bax↑ and |
|
|
|
|
|
|
| caspase-3↑ |
|
|
| Atractylonin | T24 cells | 160 mg/l; | In
vitro | RhoA↓ and
ROCK1↓ | Inhibiting VM | (127) |
|
|
| 48 h |
|
| formation |
|
| Lupeol | B16-F16 cells | 100 µmol/l; | In
vitro | CD-133↓ | Inhibiting VM | (132) |
|
|
| 24 h |
|
| formation |
|
|
Epigallocatechin | PC-3 cells | 80 µmol/l; | In
vitro | VE-CAD↓,
Twist↓, | Inhibiting VM | (137) |
| gallate |
| 24 h |
| N-cadherin↓ and
AKT↓ | formation |
|
| Kaempferol | PC-3 cells | 15 µmol/l; | In
vitro | PSA↓, TMPRSS2↓ | Inhibiting VM | (139) |
|
|
| 24 h |
| and TMEPA1↓ | formation |
|
| Resveratrol | PC-3 cells | 40 µmol/l; | In
vitro | MMP-2↓,
VE-CAD↓, | Inhibiting VM | (141) |
|
|
| 24 h |
| EphA2↓ and
5γ-2↓ | formation |
|
Liver cancer
Hepatocellular carcinoma (HCC) had a global
incidence of 865,269 cases in 2022 and is the third most common
cause of mortality worldwide (4).
Metabolism-related diseases, such as alcoholic liver disease,
diabetes and non-alcoholic fatty liver disease, are emerging as a
cause of HCC (5). Anti-angiogenic
drugs, such as sorafenib and bevacizumab, have exhibited limited
long-term efficacy in HCC, with VM being associated with the
malignant phenotype and prognosis of HCC, as well as resistance to
such treatments (6). TCM monomers
inhibit VM formation through multi-target mechanisms.
Melittin
Melittin, a 26-residue cationic peptide from bee
venom, exhibits notable anticancer properties (7). Mellitin can reverse
epithelial-mesenchymal transition (EMT) in HCC induced by
CoCl2. Notably, it reduced hypoxia inducible factor
(HIF)-1α, p-AKT, VEGF, MMP-2 and MMP-9 expression. Ex vivo and
in vivo investigations underscored its robust biological
efficacy in inhibiting VM formation. This inhibitory effect was
primarily mediated through the suppression of the HIF-1α/AKT
signaling pathway (8).
Myricetin (MYR)
MYR is a flavonoid commonly found in bayberry and
other plants (9). MYR, a highly
potent antagonist of production of anthocyanin pigment 1 (PAP1),
inhibits VM formation in HCC cells. Molecular docking studies
revealed that MYR binds specifically to Leu258 and Thr261 residues
within the PAP1 protein (10,11).
The regulatory effects of MYR on EMT are mediated by upregulating
E-cadherin protein levels and downregulating intracellular
expression of VEGFR1 and VEGFR2.
Others
Daurisoline (DAS) is a bisbenzylisoquinoline
alkaloid isolated from Menispermum dauricum and rhizoma
Menispermi. DAS inactivates Ras homolog family member A
(RhoA)/Rho-associated coiled-coil containing protein kinase 2
(ROCK2), inhibits AKT and ERK-p38 MAPK signaling, induces HCC cell
apoptosis and inhibits VM formation. The combined use of DAS with
sorafenib can potentially improve the sensitivity of sorafenib
(12).
The ethanolic extract of Elephantopus scaber
(ESEE), a traditional medicinal plant, contains a diverse array of
bioactive compounds, including epifriedelinol, lupeol, flavonoids
and glucosides (13). ESEE exhibits
potent antitumorigenic effects in HepG2 HCC cells by inhibiting the
expression of MMP-2, VEGFA and MMP-9. Furthermore, it reduces the
number and area of cell-formed luminal structures in vitro,
thereby suppressing HCC cell proliferation, metastasis and VM
formation (14).
Celastrus orbiculatus (oriental bittersweet),
a member of the Celastraceae family, is a medicinal plant whose
vines and stems possess notable antitumor and anti-inflammatory
activities. Celastrus orbiculatus is also called Dragon
Grass or Yellow Vine (15). The
main target of Celastrus orbiculatus extract in liver cancer
is ephrin type-A receptor 2 (EphA2), a gene associated with VM in
liver cancer cells. Knockdown or overexpression of EphA2 markedly
affects VM in liver cancer, with the expression levels of
VE-cadherin (VE-CAD) changing with EphA2 changes (16).
Although the aforementioned natural products
demonstrate inhibitory effects on EMT and VM in combating HCC,
their mechanisms of action exhibit distinct pharmacological
profiles: Melittin and ESEE converge on the HIF-1α/AKT signaling
axis; MYR functions exclusively as a PAP1 antagonist; DAS
predominantly targets the RhoA/ROCK2-MAPK pathway; while
Celastrus orbiculatus exerts its effects through EphA2 gene
modulation. However, notable limitations persist in current
research paradigms. First, mechanistic investigations remain
insufficiently characterized, with a lack of direct target
validation studies. Second, pharmacokinetic profiles and in
vivo efficacy data remain limited, which creates key obstacles
for clinical translation. Challenges including suboptimal
bioavailability, tumor microenvironment heterogeneity and lack of
standardized extraction protocols collectively present substantial
barriers to clinical application. Therefore, the path from bench to
bedside for these compounds remains fraught with complexities
requiring systematic resolution.
Lung cancer
Lung cancer caused 1,817,172 cases of mortality
worldwide in 2022 (4). Accounting
for ~85% of cases, non-small cell lung cancer (NSCLC) is the
predominant form of lung cancer, with high mortality and low
survival rates, necessitating novel therapeutic options (17). VM is widespread in lung cancer and
associated with poor prognosis (18).
Baicalein
Derived from the medicinal plant Scutellaria
baicalensis (TCM), baicalein is a therapeutically active
flavonoid and has been extensively studied for its potent
anticancer effects across various malignancies, such as lung
cancer, liver cancer and colorectal cancer, acting at the molecular
and cellular levels (19). In A549
lung cancer cells, baicalein dose-dependently suppressed the mRNA
expression of VM-associated genes, including VE-CAD, EphA2, MMP14,
MMP2, MMP9, PI3K and laminin subunit γ-2 (LAMC2). In vitro
and in vivo investigations have demonstrated that baicalein
suppresses VM formation in NSCLC by targeting the RhoA/ROCK
signaling cascade (20).
Lycorine
Derived from the Amaryllidaceae family, the natural
alkaloid lycorine exhibits diverse pharmacological properties
encompassing anti-inflammatory, antifungal, antiviral, antimalarial
and antitumor activities (21).
Lycorine inhibits Salvador homolog-1 (SAV1) degradation and
activates mammalian Sterile 20-like kinase 1 (MST1), thereby
reversing SAV1 deficiency in lung cancer cells. MST1 activation
suppresses oncogene-driven transcription and attenuates AKT and
NF-κB signaling pathways, markedly inhibiting lung cancer cell
proliferation, metastasis and VM formation (22).
Others
Curcumin, a natural polyphenolic phytoalexin, has
attracted notable scientific interest due to its pleiotropic
biological activities (23).
Curcumin-docetaxel micelles modified with octreotide downregulated
MMP-2 and HIF-1α, exhibited robust cytotoxicity and suppressed VM
in A549 lung cancer cells (24).
Tetramethylpyrazine (TMP), a bioactive alkaloid compound isolated
from TCM herbs, has demonstrated diverse pharmacological properties
over the past few decades. The therapeutic efficacy of TMP has been
validated across various diseases, such as urinary tract
infections, cystitis, pneumonia, diarrhea and otitis media,
underscoring its clinical potential (25). TMP reduced the number of lumens and
crossovers in A549 lung cancer stem cell-like cells (CSLCs) in
vitro under hypoxia, mediated through modulation of the
hepatocyte growth factor (HGF)/cellular-mesenchymal epithelial
transition (c-Met) signaling axis (26).
Dihydroartemisinin (DHA) is an active metabolite of
artemisinin and its derivatives artesunates (ARTs) (27). DHA downregulates VE-CAD mRNA and
protein expression levels and inhibits VM formation in NSCLC A549
and H3255 cells (28). Salvianolic
acid A (Sal-A), an active component of the traditional herbal
medicine Salvia miltiorrhiza, markedly attenuates the
activity and invasiveness of NSCLC cells and suppresses
capillary-like structure formation. These effects are mediated by
the downregulation of key VM-associated proteins, including EphA2,
VE-CAD and MMP2, as well as the inhibition of phosphorylated PI3K
(p-PI3K), p-AKT and p-mTOR within tumor cells (29).
Baicalin, DHA and Sal-A demonstrate inhibitory
effects on VM formation in NSCLC through multifaceted mechanisms
involving modulation of VM-related core genes (e.g., VE-CAD, EphA2
and MMP2/9) and suppression of the PI3K/AKT/mTOR signaling pathway.
Notably, baicalin exhibits specificity for the RhoA/ROCK pathway,
while Sal-A further enhances its anti-VM activity by inhibiting
mTOR phosphorylation (30). In
parallel, lycorine reverses SAV1 deficiency through activation of
the MST1 kinase and concurrent suppression of the AKT/NF-κB axis,
whereas curcumin uniquely targets VM formation in CSLCs by
regulating the HGF/c-Met signaling cascade (22). Furthermore, nanomedicine-based
strategies synergistically augment anti-VM efficacy through dual
actions: Downregulation of MMP-2/9 and HIF-1α, modulation of
E-cadherin expression and enhanced drug delivery efficiency
(31). However, current mechanistic
investigations into VM inhibition remain predominantly constrained
to phenotypic associations, with key gaps in rigorous target
validation studies, exemplified by the proposed direct interaction
between baicalin and RhoA kinase, and insufficient in vivo
pharmacodynamic characterization. While nanoformulations
demonstrate enhanced tumor-targeting capabilities, their complex
compositions introduce potential uncontrollable experimental
variables that may compromise mechanistic clarity and therapeutic
reproducibility (31).
Colorectal cancer
In 2022, colorectal cancer accounted for 1,926,118
novel cases, exhibiting the second-highest mortality rate amongst
all cancers in the world (4).
Although the targeted therapeutic agent bevacizumab prolongs
progression-free survival (32,33),
it is associated with adverse effects such as hypertension,
proteinuria, hemorrhage, gastrointestinal perforation (34,35),
thrombophilia and neurological disorders (36). VM formation is markedly associated
with invasion, metastasis and poor prognosis (37).
Matrine (MAT)
MAT, a bioactive alkaloid derived from the
traditional Chinese herb Sophora flavescens Aiton, has
demonstrated notable anticancer properties through extensive
research. Studies have consistently reported that MAT exhibits
multifaceted antitumor effects, including the inhibition of cancer
cell proliferation, cell cycle arrest, induction of apoptosis and
suppression of metastatic potential in malignant cells (38,39).
MAT exhibited potent inhibitory effects on the proliferation and
metastasis of colon cancer CT26 cells. Furthermore, it effectively
disrupted actin cytoskeleton organization and suppressed VM
formation. These effects were mediated by the inhibition of JNK and
ERK phosphorylation within the MAPK signaling pathway, blockade of
the EMT process and synergistic interactions with claudin-9
silencing (40).
Amentoflavone (AMF)
AMF, a natural biflavonoid compound, has been
reported to exhibit multiple biological activities (41), including anticancer effects. AMF
suppresses the HIF-1α/VEGF signaling pathway in a
concentration-dependent manner, leading to the downregulation of
HIF-1α, β-catenin, VEGF, vimentin, Snail and VM-associated
tubulin-like structures. These effects are reversed by
dimethyloxalylglycine (DMOG), an activator of the HIF-1α/VEGF
pathway (42).
Others
Delphinidin, a natural anthocyanin abundant in
blue-violet plants and their fruits, exerts anti-VM effects by
suppressing the RhoA/ROCK signaling pathway. Mechanistically, it
downregulates the expression of key molecular mediators, including
RhoA, ROCK and VEGF, thereby markedly diminishing VM-associated
tubulin-based network formation. This dual regulatory mechanism
disrupts tumor vascularization and promotes apoptosis in cancer
cells through cytoskeletal reorganization (43). Evodiamine (EVO) is a quinazoline
alkaloid isolated from the herb Wuzhu [Tetradium ruticarpum
(A. Jussieu) T. G. Hartley] (44).
EVO suppresses colorectal cancer tumor growth and VM formation by
downregulating key molecular markers, including HIF-1α, VE-CAD,
VEGF, MMP2 and MMP9 (45).
AMF and EVO demonstrate anti-VM activity in
colorectal cancer by targeting HIF-1α and its downstream effectors,
including VEGF, MMP2/9 and VE-CAD. Notably, both Astragalus
and Atractylodis macrocephalae mixture and AMF exhibit
additional modulation of reactive oxygen species (ROS) homeostasis,
with the HIF-1α/VEGF axis confirmed as a key mediator through
DMOG-reversible effects. By contrast, MAT and Delphinidin employ
distinct mechanisms: MAT suppresses VM formation via MAPK pathway
inhibition and EMT disruption, demonstrating synergistic anti-VM
effects when combined with claudin-9 silencing; Delphinidin
uniquely targets RhoA/ROCK signaling to downregulate VEGF
expression while inducing apoptosis. Although HIF-1α emerges as a
convergent target for multiple compounds, current mechanistic
conclusions predominantly rely on correlational analyses rather
than rigorous direct target engagement validation. This underscores
the need for advanced methodologies such as surface plasmon
resonance binding assays, cellular thermal shift assays and
CRISPR-based genetic validation to establish causal relationships
between compound-target interactions and phenotypic outcomes.
Gastric cancer
There were 968,350 novel cases of gastric cancer in
2022, presenting a notable threat to the global health system
(4). It has been reported that
>33% of patients receiving preoperative chemotherapy followed by
surgery experienced disease recurrence, with a median follow-up
period of 27.8 months. Notably, 44% of these relapses occurred
within the 1st year post-surgery (46). Radiotherapy faces resistance issues
(47,48) and has serious side effects, such as
inflammatory skin reactions, fatigue and digestive system symptoms
(49). The identification of VM in
gastric cancer cells offers a novel therapeutic target for drug
development, presenting novel opportunities for treatment
strategies in the future (50).
Saffronin
Saffronin is an active ingredient extracted from the
stigma of saffron. Saffronin regulates several signaling pathways,
including HIF-1α, Notch1 and Sonic hedgehog (SHH), reduces the
expression levels of patched-2, glioma-associated oncogene homolog
1 (Gli1) and other related protein factors, and inhibits VM
formation and metastasis of human umbilical vein endothelial cells
(HUVEC) tubes and gastric cancer cells (51).
DHA
DHA, derived from the antimalarial drug Artemisia
annua, has potent pharmacological activity. In gastric cancer,
the fibroblast growth factor 2 (FGF2) gene is most closely
associated with angiogenesis and VM. DHA exerts similar effects as
fibroblast growth factor blockers, inhibiting VM formation and
decreasing the expression of VM-associated biologically active
factors (52).
Others
Formononetin (FMN), a naturally occurring
isoflavone, is widely distributed in numerous medicinal plants,
including Astragalus membranaceus, red clover, Trifolium
pratense and Pueraria lobata. FMN regulates the
HIF-1α/VEGF signaling pathway in a concentration-dependent manner,
destroys the lumen structure already formed by VM in gastric cancer
cells and markedly induces apoptosis (53). Ginsenoside Rg3 (Rg3), a bioactive
steroidal saponin, belongs to the ginsenoside family derived from
Panax ginseng C.A. Meyer (54). Rg3 inhibits glycogen synthase kinase
(GSK)-3β, Wnt family member 2B mRNA and β-linker protein
expression, as well as cellular VM formation, in SGC7901 gastric
cancer cells in vitro (55).
Aucubin (AU), a bioactive iridoid glycoside, is a
principal active constituent derived from Eucommia ulmoides
Oliv (56). In vitro
treatment of MGC803 gastric cancer cells with AU markedly
downregulated metastasis-associated proteins (RhoA, ROCK1,
N-cadherin, vimentin and VE-CAD) while upregulating the epithelial
marker E-cadherin. These effects collectively suppressed EMT and VM
in gastric cancer cells (57).
Jaceosidin dose-dependently inhibits VM in HGC-27 gastric cancer
cells through selective modulation of the sphingosine kinase
1/sphingosine-1-phosphate (SphK1/S1P) signaling pathway.
Experimental data demonstrated that increasing concentrations of
jaceosidin progressively inhibit tumor cell clonogenicity and
VM-associated tubular network formation. Notably, the compound
induces dose-responsive disassembly of preformed VM channels
through cytoskeletal reorganization, while simultaneously blocking
de novo VM formation by suppressing cancer cell plasticity
(58).
FMN and Rg3 demonstrate anti-VM activity in gastric
cancer through convergent targeting of the HIF-1α signaling axis,
with saffronin exhibiting additional modulation of Notch1 and SHH
pathways. Notably, FMN suppresses HIF-1α/VEGF signaling, while Rg3
inhibits the GSK-3β/Wnt/β-catenin cascade. By contrast, AU and
jaceosidin employ distinct mechanisms: AU disrupts VM formation via
RhoA/ROCK-mediated EMT inhibition and downregulation of
metastasis-related proteins, whereas jaceosidin selectively targets
the SphK1/S1P pathway to alter cytoskeletal dynamics and cancer
cell plasticity. DHA employs a unique strategy by functioning as an
FGF2 antagonist, directly inhibiting FGF-driven angiogenesis and VM
processes. While these findings underscore the multi-target
potential of natural compounds, notable limitations persist.
Current mechanistic insights rely predominantly on in vitro
models (e.g., SGC7901, MGC803 and HGC-27 cell lines) with
inadequate validation in complex in vivo tumor
microenvironments. The recurrent focus on HIF-1α as a shared target
among multiple compounds (saffronin, FMN and Rg3) suggests its role
as a convergent signaling hub but raises concerns about therapeutic
redundancy and off-target effects.
Glioma
Complete surgical resection of glioma is often
limited by its high degree of infiltration, with residual lesions
remaining in 60–70% of patients postoperatively (59). Radiotherapy has the risk of damaging
normal brain tissue (60,61) and facing local resistance (62), potentially leading to local
recurrence, whereas most chemotherapeutic agents, such as
platinum-based drugs, methotrexate and doxorubicin, have difficulty
crossing the blood-brain barrier (BBB) (63). VM is present in glioma and
represents a potential therapeutic target (64). Therefore, the development of novel
anti-VM drugs capable of crossing the BBB is key.
Artemether, a derivative of artemisinin, is rapidly
absorbed in lipids and represents a highly permeable compound that
disrupts VM by downregulating MMP-2 and HIF-α. Dual-targeted
artemether/paclitaxel micelles enhanced BBB penetration and tumor
accumulation while suppressing glioma VM through coordinated
downregulation of HIF-1α, MMP-2 and PI3K expression (65).
Isoliquiritigenin (ISL) is a bioactive chalcone
compound isolated from licorice (66). The combination of ISL and
temozolomide, when administered under hypoxic conditions, markedly
reduced the number and length of VM channels in glioma SHG44 cells.
This effect was accompanied by a marked downregulation of MMP-2,
VEGF and EphA2 protein levels (67).
Artemether and ISL demonstrate anti-VM activity in
glioma through modulation of key pro-angiogenic factors, including
MMP-2 and HIF-1α. While both compounds target these core VM
mediators, their pharmacological profiles diverge markedly.
Artemether, characterized by high lipophilicity, directly
suppresses MMP-2 and HIF-1α expression, with its therapeutic
efficacy markedly enhanced when formulated in dual-targeted
polymeric micelles co-loaded with paclitaxel. This nanoformulation
strategy not only improves BBB penetration through
permeability-glycoprotein inhibition but also synergistically
inhibits PI3K signaling to disrupt VM network formation. By
contrast, ISL exhibits hypoxia-dependent synergism with
temozolomide, demonstrating notable VM suppression in glioma
stem-like cells. The dual-targeted artemether/paclitaxel micelle
system demonstrates BBB penetration and tumor targeting capacity,
has notable engineering value but faces challenges in terms of
formulation stability and cost. The ISL-temozolomide combination
enhances VM inhibition and reverses temozolomide resistance;
however, potential drug interactions and long-term toxicity have
not yet been clarified.
Glioblastoma (GBM)
GBM, the most common primary malignant brain tumor
in adulthood, is highly aggressive and heterogeneous (68). It has been reported that 75–90% of
cases recur within 1 year after surgery (68,69).
The median survival of patients receiving standard treatment in
clinical trials is 14.6–21.1 months (70). Radiotherapy, immunotherapy and
targeted therapy do not provide satisfactory results. Targeting VM,
a key mechanism of tumor angiogenesis independent of endothelial
cells, represents a potential therapeutic breakthrough in
refractory types of cancer.
Moroidin, a cyclic peptide compound derived from
silver chickweed seeds, is a novel microtubule-targeting agent due
to its ability to inhibit microtubule polymerization. Moroidin
markedly inhibited EMT in GBM by decreasing the protein kinase
p-ERK and inhibiting the activation of β-catenin, inhibiting
angiogenesis, smooth muscle actin and MMP-9 levels (71).
Ruta graveolens L (RGWE), commonly called
rue, is a perennial herbaceous plant native to the Mediterranean
region that has become naturalized worldwide (72). RGWE markedly suppresses VM formation
in U87-MG GBM cells while concurrently inducing cytotoxicity in
GBM-derived cancer stem cells and disrupting existing VM networks
(73).
Moroidin functions as a novel
microtubule-destabilizing agent that selectively inhibits
microtubule polymerization, leading to suppression of ERK
phosphorylation and β-catenin activation. This dual inhibition
disrupts EMT, angiogenesis and MMP-9 expression, thereby
attenuating VM network formation. By contrast, RGWE exhibits
broad-spectrum VM inhibition in U87-MG cells while demonstrating
dual cytotoxicity against both bulk tumor cells and glioblastoma
stem cells, effectively dismantling pre-established VM
architectures. Notably, the stem cell-targeting capability of RGWE
suggests synergistic potential with conventional therapies, as GSCs
are known contributors to therapeutic resistance. Moroidin focuses
on microtubule polymerization inhibition, featuring a novel
mechanism and unique target; however, its direct effect on VM core
markers (such as VE-CAD) remains to be elucidated. RGWEs exhibit
potent killing effects on cancer stem cells and VM structures;
however, their complex composition obscures the core active
compounds and mechanisms.
Osteosarcoma
Osteosarcoma originates from mesenchymal tissue; the
tumor cells produce bone-like material. Surgery is the primary
method of treatment; however, it may lead to amputation, which
affects the quality of life in patients (74). The effectiveness of chemotherapy is
associated with the location of tumor growth; however, it also
faces the problem of drug resistance (75). The local recurrence rate of
osteosarcoma is often between 7 and 10% (76), and the relevant genetic features of
VM are associated with osteosarcoma prognosis (77). TCM herbal therapies can be a good
adjuvant treatment.
Rhizoma Paridis saponins (RPS)
RPS exhibit potent antitumor activity by
simultaneously suppressing angiogenesis and inhibiting cancer cell
migration and invasion through multiple molecular mechanisms
(78). RPS treatment in
osteosarcoma cells markedly downregulated migration-inducing gene-7
(MIG-7) expression while concurrently inhibiting the
PI3K/MMPs/laminin-5 γ2 chain (Ln-5γ2) signaling pathway. These
molecular alterations corresponded with marked decreases in
downstream effector protein expression, ultimately leading to
substantial inhibition of cellular protrusion formation and VM
structural development (79).
Others
Curcumin exhibits a concentration-dependent
inhibitory effect on the tubular VM structures in osteosarcoma S180
cells, as well as on the reticular networks formed through their
interaction with endothelial cells. This antitumor effect is
mechanistically associated with the notable downregulation of
MMP-2/9 at transcriptional and translational levels (80).
Paris polyphylla ethanol extract (PPEE)
demonstrates dual anti-osteosarcoma efficacy in vivo and
in vitro by modulating cell cycle regulators [upregulating
p-cyclin-dependent kinase (CDK)1, p-cell division cycle 25C and
p-checkpoint kinase 2 (Chk2)], promoting apoptosis, suppressing
metastasis-related proteins [focal adhesion kinase (FAK), MIG-7 and
MMP-2/9) and inhibiting VM formation (81).
RPS exert their anti-VM activity primarily through
modulation of the PI3K/MMPs/Ln-5γ2 signaling axis, accompanied by
reduced expression levels of MIG-7, thereby attenuating cellular
protrusion formation and VM network assembly. Similarly, PPEE
targets FAK and MIG-7 while concurrently regulating cell cycle
progression and apoptosis through modulation of p-CDK1 and p-Chk2.
By contrast, curcumin exhibits concentration-dependent disruption
of both VM tubular structures and endothelial-integrated vascular
networks, achieving MMP-2/9 suppression at both transcriptional and
translational levels. Notably, these compounds demonstrate
complementary mechanisms: RPS focuses on ECM remodeling and cell
motility, PPEE integrates anti-metastatic and cell cycle control,
while curcumin provides broad-spectrum MMP inhibition. However,
current findings remain constrained by reliance on two-dimensional
cell culture models (e.g., U2OS and MG-63), with limited validation
in three-dimensional VM assays or in vivo osteosarcoma
microenvironments. The target and specific mechanism of RPS are
clear; however, in vivo studies are warranted. Curcumin is
an old drug used in anticancer drugs and breaking through its
inherent bioavailability dilemma remains an urgent issue to be
solved. PPEE has been thoroughly studied in vivo and in
vitro; however, defining its core active ingredient is
challenging because of its mixed nature.
Choroidal melanoma (CM)
CM is the most common primary intraocular malignancy
in adults, with a unique biological behavior, susceptibility to
hepatic metastases, poor prognosis and retinal risks associated
with local radiotherapy and surgery (82,83).
VM is highly associated with the first symptoms of CM and mortality
rates (84,85).
ART has been the focus of its diverse
pharmacological actions, including anti-inflammatory,
immunoregulatory and anticancer properties (86). Artemisinin treatment markedly
suppressed HIF-1α and angiogenic factor expression in OCM-1 and
C918 melanoma cells, concurrently inhibiting VM formation through
blockade of the Wnt family member 5a/calcium/calmodulin-dependent
protein kinase II (CaMKII) signaling axis (87–90).
Luteolin (LUT), a naturally occurring flavonoid
polyphenol, is abundantly present in various plant sources,
including fruits, vegetables and medicinal herbs. This bioactive
compound demonstrates remarkable pharmacological potential,
particularly in oncology and immunomodulation, through its potent
antitumor and anti-inflammatory properties (91). LUT exerts potent anti-angiogenic and
anti-VM effects in C918 uveal melanoma cells and HUVECs by
targeting the PI3K/AKT pathway, markedly downregulating VEGF, PI3K
and AKT expression (92,93). The IC50 value of LUT in
human C918 choroidal melanoma cells was as low as 24.41 µmol/l
after 24 h. LUT markedly suppresses VEGF expression and PI3K/AKT
phosphorylation (p-PI3K/p-AKT) in C918 uveal melanoma cells and
HUVECs, inhibiting VM and mosaic vessel formation in choroidal
melanoma models (89,90,94).
ART and LUT demonstrate potent anti-VM activity in
uveal melanoma, particularly in aggressive C918 cell models,
through modulation of distinct signaling hubs. ART exerts its
anti-VM effects via suppression of the
Wnt5a/Ca2+/calmodulin-dependent protein CaMKII axis,
leading to downstream attenuation of HIF-1α and angiogenic factors
essential for VM network formation. By contrast, LUT targets the
PI3K/AKT pathway with high potency, markedly reducing VEGF
expression and PI3K/AKT phosphorylation. Notably, the dual
inhibition of VM by LUT and angiogenesis positions it as a
multifaceted therapeutic candidate, while the mechanism of ART
highlights the therapeutic potential of Wnt pathway modulation in
HIF-1α-driven malignancies. However, current findings remain
constrained by reliance on monolayer cultures and lack validation
in three-dimensional VM models or in vivo uveal melanoma
systems.
Breast cancer
There were 2,308,897 novel cases of breast cancer in
2022 (4), the second-highest
incidence in the world among cancers. Standardized treatment of
breast cancer comes with adverse effects, such as drug resistance
problems, neurotoxicity, hot flashes, mood swings, lymphedema,
fatigue and cardiotoxicity, which severely affect the quality of
life of patients (95,96). VM is associated with the malignant
phenotype of breast cancer (97).
Brucine
Brucine, an extract of the traditional herb
Strychnos nux-vomica L, disrupts the cytoskeleton and
microtubule structure of MDA-MB-231 cells and reduces the tubule
number, intersections and average tubule length in a
concentration-dependent manner, downregulates MMP-2, MMP-9 and
EphA2 and reduces VM formation. These findings provide a novel
therapeutic strategy for triple-negative breast cancer (TNBC)
(98).
Others
Triptonide, a bioactive diterpenoid tricyclic oxide
derived from Tripterygium wilfordii Hook F, exerts potent
antitumor effects in TNBC by degrading Twist1 and Notch1
oncoproteins, downregulating VEGFR2/VE-calmodulin expression,
suppressing NF-κB signaling and markedly inhibiting VM (99).
Astilbin (AST), a bioactive dihydroflavonol
glycoside, is widely distributed in medicinal plants and dietary
sources, exhibiting diverse pharmacological properties that have
garnered notable research interest (100,101). AST demonstrates
concentration-dependent anti-angiogenic and apoptosis-inducing
activities in breast cancer cell lines (MCF-7 and MDA-MB-231)
through dual inhibition of HIF-1α expression and subsequent VEGF
signaling pathway activation. This inhibition concomitantly
downregulates angiogenesis-associated markers (VE-CAD, N-cadherin
and MMP-2) and reduces VM lumen formation, while inducing tumor
cell apoptosis through HIF-1α/VEGF pathway modulation (102).
Brucine functions as a microtubule-destabilizing
agent that disrupts cytoskeletal dynamics by binding β-tubulin,
leading to concentration-dependent downregulation of MMP-2, MMP-9
and EphA2, which are key mediators of ECM remodeling and VM network
formation. By contrast, triptonide exhibits unique oncoprotein
degradation activity, promoting ubiquitination-mediated proteolysis
of Twist1 and Notch1 transcription factors. This results in
suppression of VEGFR2/VE-CAD signaling and attenuation of
NF-κB-driven pro-inflammatory responses, thereby inhibiting VM
channel assembly. Notably, AST employs a dual mechanism targeting
both HIF-1α stabilization and VEGF secretion, reducing
angiogenesis-related markers (VE-CAD, N-cadherin and MMP-2) while
inducing caspase-dependent apoptosis. These findings highlight the
therapeutic potential of combining microtubule disruption
(Brucine), oncoprotein degradation (triptonide) and
hypoxia/angiogenesis axis inhibition (AST) for VM suppression in
TNBC. Triptonide has certain clinical application potential due to
its cancer protein degradation mechanism; however, toxicity issues
need to be addressed first. AST has potential synergistic
advantages due to its simultaneous regulation of VM and EMT. The
cytoskeletal targeting of brucine warrants further thorough
research.
Pancreatic cancer
In 2022, pancreatic cancer already ranked second in
the world with 467,005 cases of mortality, associated with smoking,
obesity and diabetes (103). The
majority (80–85%) of cases are diagnosed at advanced stages
(locally advanced/metastatic), associated with dismal long-term
outcomes (5-year survival rate, <10%) (104,105). The risk of complications after
surgical treatment, the efficiency of chemotherapeutic agents and
drug resistance markedly affect the prognosis and mortality rate of
pancreatic cancer (106). The
presence of VM in pancreatic cancer may be associated with
prognosis (107).
Rg3 exerts multi-target antitumor effects in
pancreatic adenocarcinoma (PAAD) through epigenetic regulation. Rg3
upregulates microRNA-204 to suppress dishevelled segment polarity
protein 3-mediated signaling, thereby downregulating VE-CAD
expression in SW1990 cells (108).
This intervention coordinately inhibits PI3K pathway activation,
evidenced by reduced phosphorylation of PI3K and decreased
N-cadherin/VE-CAD levels (109).
The dual suppression disrupts cancer stemness maintenance and
inhibits VM through impaired 3D tube/sphere formation and
matrix-remodeling capacity (109,110).
Ginsenoside Rg3 has notable therapeutic potential
and is expected to be used in clinical applications; however, its
ability to penetrate the pancreatic cancer interstitial barrier
in vivo has not been verified. Furthermore, its lack of
regulation of hypoxic microenvironments (such as HIF-1α) may weaken
its clinical efficacy.
Ovarian cancer (OC)
The global incidence of OC has reached 32,439,815
cases (4). Emerging data reveal
rising incidence and mortality rates across diverse populations,
with notable regional disparities (4). VM networks serve a pivotal role in
disease progression by forming functional connections with host
blood vessels; this hybrid vascular system promotes hematogenous
dissemination of tumor cells, driving metastatic spread and
conferring treatment resistance (111–113). VM represents a promising
therapeutic target for OC treatment, which offers the potential to
disrupt tumor perfusion and metastatic dissemination simultaneously
(113).
Cinobufagin
Cinobufagin, a cardiotonic steroid derived from
Bufo species, demonstrates anti-metastatic mechanisms in OC
through ECM remodeling and immune microenvironment modulation. This
compound transcriptionally suppresses forkhead box S1 (FOXS1) to
inhibit MMP9/MMP14 and LAMC2, blocking VM formation in SKOV3 cells
by impairing tumor-driven matrix reorganization (114). The intervention concurrently
disrupts the C-C motif chemokine ligand 2/receptor 2 (CCL2/CCR2)
chemokine axis, inhibiting protumoral (M2-like) macrophage
polarization and reprogramming the immunosuppressive tumor
microenvironment (115). Notably,
cinobufagin intercepts TGF-β-induced EMT, a key mediator of OC
metastasis, through these coordinated multi-target actions
(114,115).
Others
Catechins are more commonly referred to as tea
polyphenols derived from tea, one of the oldest and most consumed
beverages in the world (116).
Catechins with specific acyl portions, such as catechin gallate
(CG), epi-CG (ECG), gallo-CG (GCG) and epigallocatechin gallate
(EGCG), have pharmacological effects that inhibit TGF-β-induced
cell migration, MMP-9 and Snail secretion and OC cell VM (117). Sinomenine (SIN), a bioactive
alkaloid isolated from the medicinal vine Sinomenium acutum,
exhibits well-documented anti-inflammatory and antitumor properties
with established pharmacological applications (118). SIN demonstrates dose-dependent
anti-VM activity in OC A2780 cells through multi-target
transcriptional regulation. This compound coordinately suppresses
VM-associated markers (VEGF, EphA2 and MMP-2/9) and disrupts the
C-X-C motif chemokine receptor 4 (CXCR4)/STAT3 signaling axis,
evidenced by reduced p-STAT3 levels. The dual inhibition mechanism
blocks ECM remodeling required for VM channel formation,
establishing SIN as a potent VM suppressor in OC (119).
Cinobufagin disrupts matrix remodeling and immune
microenvironment dynamics by targeting FOXS1, while concurrently
modulating the CCL2/CCR2 axis to attenuate tumor-associated
macrophage recruitment. EGCG suppresses TGF-β-induced EMT and MMP-9
secretion, thereby reducing ECM degradation and VM channel
formation. By contrast, SIN employs dual pathway inhibition: It
directly downregulates VM-related markers (VEGF, EphA2 and MMP-2/9)
while intercepting CXCR4/STAT3 signaling, a key axis for cancer
stem cell maintenance and metastatic dissemination. Notably, these
compounds converge on matrix remodeling but diverge in upstream
regulation, cinobufagin integrates immunomodulatory and
transcriptional repression, catechins focus on TGF-β blockade and
SIN targets chemokine/STAT signaling. However, these findings are
mainly based on single cell line models (such as SKOV3 and A2780)
and isolated signaling pathway analyses, lacking in vivo
validation of complex microenvironment interactions and clinical
relevance evidence.
Cervical cancer
Cervical cancer ranks among the most prevalent
malignancies in women globally and represents the primary cause of
cancer-related mortality in 37 countries (4), particularly in low- and middle-income
regions (4). Patients with
VM-positive cervical cancer exhibited markedly reduced overall
survival (OS), with strong correlations to lymph node metastasis,
advanced International Federation of Gynecology and Obstetrics
stage (III/IV) [hazard ratio (HR)=2.3; 95% CI, 1.8–3.0], tumor size
>4 cm and hypofractionation regimens (120), which markedly affects VEGF
treatment and induces recurrence (121).
Pristimerin (Pris), a quinone methide triterpenoid
derived from Celastrus and Maytenus species
(Celastraceae family), exhibits broad bioactive properties,
including anti-inflammatory, antimicrobial and antitumor effects,
with emerging potential in oncology (122). Pris inhibited the structure and
number of VM lumens in cervical cancer HeLa cells and downregulated
the expression levels of SHH, Gli1 mRNA, VEGF-A, VE-CAD and Gli1
proteins, which were associated with the inhibition of the SHH/Gli1
signaling pathway. By contrast, overexpression of SHH impaired the
physiological effects of Pris to a certain extent (123).
Honokiol, a bioactive biphenyl neolignan primarily
derived from Magnolia officinalis bark, has been
pharmacologically characterized as the principal active constituent
of this TCM herb (124). Honokiol
dose-dependently suppresses VM formation in cervical cancer HeLa
cells by inhibiting the EGFR signaling pathway, evidenced by
reduced p-EGFR and downregulation of downstream effectors (MMP-2/9,
EphA2, VEGF and cadherin-2) (125).
Notably, while both compounds disrupt VM formation,
their mechanisms diverge fundamentally: Pris targets developmental
signaling cascades, whereas honokiol focuses on receptor tyrosine
kinase-mediated oncogenic signaling. These findings highlighted the
therapeutic potential of combining HH pathway inhibitors with EGFR
antagonists for VM suppression in cervical cancer. To the best of
our knowledge, current research on Pris and honokiol lacks in
vivo experimental data and safety analysis.
Esophageal cancer
Esophageal cancer has the seventh-highest mortality
rate in the world (445,129 mortalities and a mortality rate of
4.6%) (4), with anti-angiogenic
therapy only achieving limited improvement in OS with increased
side effects (126). VM can be an
independent prognostic factor for esophageal cancer and is markedly
associated with cancer stage, lymph node metastasis and
hypo-differentiation (127).
Ligustilide (LIG), a predominant phthalide
derivative, serves as a key bioactive constituent in several TCM
herbs, notably Angelica sinensis and Ligusticum
chuanxiong (128). LIG
dose-dependently suppressed proliferative (cyclin-D1↓),
anti-apoptotic (Bcl-2↓) and metastatic (RhoA/ROCK↓) proteins in
EC-109 esophageal cancer cells while upregulating cell cycle arrest
(p21↑) and pro-apoptotic markers (Bax/Caspase-3↑). Notably, RhoA
activators partially reversed these effects, confirming pathway
specificity (129).
The reversal effect of RhoA activators suggests
that their action is limited by target dependence and lacks
verification of upstream regulatory mechanisms (such as RhoA
activating factors) and the effects of the in vivo
microenvironment.
Bladder cancer (BC)
BC is approximately four times more common in men
compared to women and has the ninth highest incidence worldwide,
with smoking being a major risk factor (4). VM-positive patients with BC have more
aggressive tumors and are more prone to poor cancer outcomes
(130).
Atractylodin (ATR) is one of the main active
ingredients of Rhizoma Atractylodis. ATR has various
pharmacological features, such as antitumor properties (131). ATR modulates the RhoA/ROCK
signaling pathway, concentration-dependently reducing lumen
formation in BC T24 cells while enhancing F-actin fluorescence
intensity, disrupting cytoskeletal dynamics, downregulating
RhoA/ROCK1 expression and ultimately inhibiting VM formation, a
process key for cytoskeletal motility and cell adhesion (132).
The effects of ATR are based solely on a single
cell line model and isolated signaling pathway analysis, lacking
validation in complex in vivo microenvironments and evidence
of clinical relevance.
Melanoma
Recently, the mortality rate of melanoma has
indicated a downward trend, with the number of novel cases having
reached 331,647 (4). Factors such
as ultraviolet radiation, genetic susceptibility and phenotypic
features combine to constitute the major risk factors for melanoma
(133). VM was first identified in
melanomas, with invasive cell lines being more likely to form VM,
which is positively associated with tumor thickness and risk of
metastasis. VM represents a key risk factor for melanoma
progression, linked with aggressive metastasis and poor patient
outcomes (134). VM can counteract
antitumorigenic therapy and be used as a prognostic marker for
melanoma.
Lupeol, a pentacyclic triterpenoid, is ubiquitously
distributed in edible plants (e.g., mango and olive) and medicinal
species (e.g., Ficus religiosa and Taraxacum
officinale), exhibiting notable pharmacological potential
(135). Lupeol demonstrates
multimodal antineoplastic efficacy in B16-F10 melanoma models,
exhibiting direct cytotoxicity alongside dual anti-angiogenic and
VM-suppressive effects. Mechanistically, this triterpenoid disrupts
tumor-stromal crosstalk by targeting cancer stem cell populations
and inhibiting endothelial progenitor cell (EPC) recruitment,
thereby attenuating VM channel formation and angiogenesis. Notably,
lupeol reverses dacarbazine resistance through epigenetic
modulation of EPC differentiation programs, potentially via
suppression of EPC-specific growth signaling pathways (136).
Lupeol has exhibited potential in inhibiting VM
formation and overcoming drug resistance in melanoma mouse models
and cell experiments. However, the causal chain between drug
resistance and VM, as well as safety assessments at clinically
feasible concentrations, has not been directly verified.
Prostate cancer
Prostate cancer accounted for 1.5 million incident
cases and 397,000 cases of mortality globally, ranking as the most
frequently diagnosed malignancy across 118 countries/territories
(4). The available standardized
treatment options, surgery and radiotherapy, as well as androgen
deprivation therapy, are associated with large adverse effects
(such as frequent urination, urgency, dysuria, diarrhea and sexual
dysfunction) and recurrence rates (overall recurrence rate
approaches 50%) (137–139). VM is frequently observed in
prostate cancer specimens and demonstrates notable associations
with aggressive clinicopathological features, including Gleason
score ≥7, lymph node involvement (odds ratio, 3.2; 95% CI, 2.1–4.9)
and distant metastasis (HR=2.8) (140).
EGCG
EGCG exerts anti-prostate cancer effects by
suppressing VM through selective downregulation of the
Twist/VE-CAD/AKT axis (Twist↓, VE-CAD↓ and p-AKT↓) while
maintaining EphA2 expression, revealing pathway-specific modulation
(141).
Others
Kaempferol (KMP), a dietary flavonol found in
fruits and vegetables, exhibits potent anticancer activity by
scavenging ROS and modulating redox-sensitive signaling pathways
(e.g., Nrf2/KEAP1 and NF-κB) (142). KMP, a dietary flavonoid, exerts
multimodal anti-prostate cancer effects by simultaneously
suppressing androgen receptor (AR) signaling, via
dihydrotestosterone antagonism and downregulation of AR-target
genes (protein-specific antigen/transmembrane serine protease
2-ETS-related gene) and inhibiting VM in PC-3 cells. Concurrently,
KMP disrupts VM formation by inhibiting matrix-remodeling
metalloproteinases and EMT-related pathways(such as Nrf2/KEAP1 and
NF-κB pathways). The coordinated suppression of the AR axis and VM
plasticity mechanistically contributes to its therapeutic potential
against castration-resistant prostate cancer progression (143).
Resveratrol (3,4′,5-trihydroxy-trans-stilbene; RES)
is a promising candidate for cancer therapy (144). RES inhibits the levels of factors
such as MMP-2, VE-CAD, EphA2 and laminin subunit 5γ-2 in human
prostate cancer PC-3 cells. Furthermore, it can inhibit
serum-induced and Twist nuclear localization and exerts antitumor
vascular effects through inhibition of EphA2/Twist-VE-CAD/AKT
signaling VM effects (145).
Notably, while all three compounds converge on
VE-CAD and AKT pathway modulation, EGCG and RES share a marked
focus on Twist1-mediated transcriptional regulation, whereas KMP
uniquely integrates anti-androgen activity into its anti-VM
repertoire. These findings highlight the therapeutic potential of
combining AR antagonists with EMT/VM inhibitors in prostate cancer.
These conclusions are highly dependent on simplified mechanistic
studies using a single cell line model (PC-3) and lack in
vivo microenvironment validation, clinical relevance evidence
and systematic analysis of pathway interactions. Whether the
anti-VM effect can be reproduced in androgen-sensitive models or
patients remains to be elucidated.
Discussion
VM in malignant tumors represents a multifaceted
biological process orchestrated by intricate molecular networks and
intersecting signaling pathways. The widespread occurrence of VM
across diverse malignancies highlights its potential as a novel
therapeutic target for cancer treatment. Although VM universally
involves tumor cells mimicking endothelial vessel formation to
sustain perfusion and evade immunity, its underlying molecular
mechanisms and signaling pathways exhibit notable heterogeneity
across cancer types.
VM in aggressive tumors, including melanoma, lung
cancer, small cell lung cancer, GBM and HCC, is regulated by
diverse signaling pathways, such as VEGF, FGF, Notch and RhoA/ROCK
cascades (146). In prostate
cancer, VM may be associated with signaling pathways such as
PI3K-AKT, VEGFR1 and VEGFR2 (147). In gastric cancer, VM formation is
closely associated with elevated expression of HIF-1α and
subsequent upregulation of hypoxia-responsive proteins (carbonic
anhydrase IX, glucose transporter type 1 and VEGF) (148). The identification of VM as a
therapeutic target enables tumor vascular disruption and nutrient
deprivation, thereby suppressing tumor growth and metastasis
(146,149).
TCM monomers, active ingredients or active
substances extracted from Chinese herbal medicines and further
isolated and purified, may have specific pharmacological effects.
The research and application of CMMs have demonstrated their
potential in various fields, including anti-inflammatory,
antitumor, anti-viral (150),
modulation of autophagy (151) and
improvement of neurological function (152–154).
Using TCM monomers to target VM for cancer therapy
offers various advantages: i) Direct inhibition of the expression
of VM-related molecules presents a clear mechanism of action
explaining their therapeutic effect; ii) VM is markedly associated
with poor prognosis in patients with cancer and TCM monomers can
suppress tumor aggressiveness and metastasis by inhibiting VM
formation; iii) targeting VM, a key contributor to anti-angiogenic
therapy resistance, using TCM monomers enhances antitumor efficacy
by overcoming this adaptive escape mechanism; iv) TCM monomers have
multi-targeting effects and can simultaneously affect multiple
molecules and signaling pathways associated with VM; v) research on
the role of TCM monomers in inhibiting VM provides key clues for
the development of novel antitumor drugs; and vi) TCM monomers
often originate from natural plants, with the cost of their
extraction and preparation being relatively low compared with that
of traditional antitumor drugs, such as chemotherapeutic drugs and
targeted drugs, which are costly and expensive to develop, produce
and evaluate in clinical trials. Monomers are more readily
available in several countries and regions, and particularly for
patients with a poorer financial situation, TCM monomers may be a
more economical option.
VM formation involves the synergistic or
independent regulation of multiple signaling pathways, with
different pathways serving context-specific roles across different
cancer types. Although HIF-1α/VEGF is central in traditional
angiogenesis, it exerts the opposite effect in VM. Hypoxia induces
VM formation via HIF-1α; nevertheless, VEGF-targeted therapy may
fail or even promote VM. Melittin, curcumin, AMF, EVO, saffronin,
FMN, artemether, ART and AST inhibit VM formation in tumor cells by
suppressing the HIF-1α/VEGF signaling pathway. Notch1 mediates
tumor cell endothelial-like transformation through Δ-like canonical
Notch ligand 4, promoting the formation of luminal structures.
Saffronin and triptonide inhibit luminal structures in tumor cells
by inhibiting Notch1. The SHH/Gli1 and SHH pathways enhance VM
capacity by activating EMT and maintaining tumor stem cell
characteristics. For instance, Pris and saffronin inhibit the
action of SHH, whereas Wnt5a/CaMKII promotes endothelial-like
differentiation through β-catenin and EMT regulation, akin to AMF,
ginsenoside Rg3 and moroidin. RhoA/ROCK is a core effector pathway
for VM. DAS, delphinidin, AU, LIG and atractylonin can directly
regulate cytoskeletal reorganization and matrix degradation,
whereas PI3K/AKT/mTOR serves as a hub, integrating hypoxia signals
(HIF-1α) and matrix remodeling (MMPs) to promote VM. Melittin,
Sal-A, artemether, RPS, LUT and EGCG inhibit this process.
Furthermore, NF-κB indirectly supports VM through the inflammatory
microenvironment, whereas CCL2/CCR2 mediates VM formation via
macrophage-dependent matrix modification. The cross-activation of
these pathways renders VM a mechanism of resistance to
anti-angiogenic therapy, necessitating targeted interventions that
combine hypoxia regulation (HIF-1α), EMT inhibition (SHH/Wnt) and
cytoskeletal blockade (ROCK) through a multi-pronged strategy.
The clinical translation of CMMs deserves
attention. For instance, natural melittin can non-specifically
destroy cell membranes, causing hemolysis and posing a risk of
hepatotoxicity (155). However,
using nanocarriers can optimize its half-life, improving its
stability in the blood (155). The
novel peptide melittin-K1 has demonstrated a favorable safety
profile in normal tissues in a nude mouse model (156). Baicalin concentrations in tumor
tissue are markedly higher compared with those in normal lung
tissue, with rapid but limited drug absorption (157,158). Furthermore, its derivatives (such
as BAL) overcome the low bioavailability of the parent compound
through structural modification (water solubility increased by
4-fold, tumor inhibition rate improved to 65.27%) (158). A pharmacokinetic study indicated
that this compound exhibits lung tissue-targeted accumulation
(lung/plasma concentration ratio of 3–5 times) and demonstrates
dose-dependent antitumor effects within the effective dose range
(50–100 mg/kg) with controllable toxicity (IC50 >200
µmol/l for normal cells) (159).
Despite the pharmacokinetic limitations of brucine, including poor
water solubility, short half-life and low oral bioavailability,
encapsulation in bioconjugate-loaded solid lipid nanoparticles or
polylactic acid-based nanoparticles enhances its sustained release
and prolongs therapeutic effects, markedly improving
bioavailability (160,161).
Although TCM monomers demonstrate multi-target VM
inhibition potential across cancer types, clinical translation
remains challenged by mechanistic complexity, tumor
microenvironment modulation gaps and unvalidated efficacy/safety
profiles. This necessitates future advances in multi-omics-guided
target discovery, precision combination regimens (e.g., mTOR/VEGF
inhibitors) and VM-specific biomarker development to bridge
preclinical findings to therapeutic applications.
Conclusion
Anti-angiogenic therapy is key in the modern
prevention and treatment of malignant tumors; however, clinical
practice over recent years has revealed limitations in its efficacy
(162). VM emergence underscores
the need for novel therapeutic strategies. In this context, the
development and application of herbal medicines have introduced
promising avenues for VM-targeted therapy. The present review
demonstrates that multiple herbal monomers, including curcumin,
ART, LUT, TMP and ginsenoside Rg3, suppress VM in cancer cells.
This effect is mediated by targeting VM-associated protein
expression (e.g., VE-CAD and MMPs), disrupting key signaling
pathways (e.g., PI3K/AKT and EphA2), inhibiting CSLC
differentiation and reprogramming macrophage polarization.
Ginsenoside Rg3 demonstrates broad-spectrum anti-VM activity across
multiple cancer types, including gastric adenocarcinoma (SGC7901
cells) and pancreatic ductal adenocarcinoma (SW1990 and PAAD
cells), where it markedly reduces luminal-like reticular structures
and VM network density. Mechanistic studies revealed that Rg3
employs a dual inhibitory strategy: In gastric cancer, it
suppresses HIF-1α/VEGF signaling and the GSK-3β/Wnt/β-catenin
pathway to disrupt ECM remodeling (55), while in pancreatic cancer, it
additionally targets EMT regulators such as Twist1 and Snail
(108). This multi-cancer efficacy
positions Rg3 as a versatile therapeutic candidate, although
current findings remain constrained by reliance on monolayer cell
models and lack of comparative studies across different tumor
microenvironments. Future studies can synergize multi-disciplinary
expertise and innovative approaches to advance the understanding of
VM inhibition. These findings need to be confirmed by more thorough
studies, including clinical and preclinical trials. Future research
should focus on optimizing drug formulations, expanding the
therapeutic window and developing combination therapy strategies.
With more thorough research, herbal monomers hold notable promise
as agents in the development of novel anti-VM drugs. The present
systematic review carries inherent limitations due to publication
bias. While rigorous inclusion criteria were employed to mitigate
this limitation, the absence of negative/null findings in published
databases leaves residual uncertainty. Readers are therefore
advised to interpret the conclusions with appropriate caution,
recognizing that the true therapeutic potential of these agents may
differ from currently reported estimates.
Acknowledgements
Not applicable.
Funding
This research was funded by the National Natural Science
Foundation of China 2023 (grant no. 82305301), Heilongjiang
Province Traditional Chinese Medicine Research Project (grant no.
ZHY2024-048), Project of Institute of Chinese Medicine Nanjing
University (grant no. ICMN2024012) and the Scientific Research
Program of Chinese Medicine in Heilongjiang Province (grant no. ZHY
2024-223).
Availability of data and materials
Not applicable.
Authors' contributions
FYL and YQF prepared the original draft. YS
reviewed and edited the manuscript. JL, DND, SXL and YSZ retrieved
the data and participated in the conceptualization and design of
the article as well as the data analysis. All authors have read and
approved the final manuscript. Data authentication is not
applicable.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
Glossary
Abbreviations
Abbreviations:
|
AMF
|
amentoflavone
|
|
ART
|
artesunate
|
|
AST
|
astilbin
|
|
ATR
|
atractylonin
|
|
AU
|
aucubin
|
|
BBB
|
blood-brain barrier
|
|
BC
|
bladder cancer
|
|
CaMKII
|
calmodulin-dependent kinase II
|
|
CCL2/CCR2
|
C-C motif chemokine ligand 2/receptor
2
|
|
CM
|
choroidal melanoma
|
|
c-Met
|
cellular-mesenchymal epithelial
transition
|
|
CSLCs
|
cancer stem cell-like cells
|
|
DHA
|
dihydroartemisinin
|
|
ECM
|
extracellular matrix
|
|
EGCG
|
epigallocatechin gallate
|
|
EMT
|
epithelial-mesenchymal transition
|
|
EVO
|
evodiamine
|
|
FMN
|
formononetin
|
|
GBM
|
glioblastoma
|
|
HCC
|
hepatocellular carcinoma
|
|
HGF
|
hepatocyte growth factor
|
|
MAT
|
Matrine
|
|
NSCLC
|
non-small cell lung cancer
|
|
PAAD
|
pancreatic adenocarcinoma
|
|
PPEE
|
Paris polyphylla ethanol
extract
|
|
Pris
|
pristimerin
|
|
RES
|
resveratrol
|
|
RGWE
|
Ruta graveolens water
extract
|
|
RPS
|
rhizoma Paridis saponins
|
|
Sal-A
|
salvianolic acid A
|
|
SAV1
|
Salvador homolog-1
|
|
SHH
|
Sonic hedgehog
|
|
TCM
|
Traditional Chinese Medicine
|
|
VM
|
vasculogenic mimicry
|
References
|
1
|
Zhang S, Guo H, Zhang D, Zhang W, Zhao X,
Ren Z and Sun B: Microcirculation patterns in different stages of
melanoma growth. Oncol Rep. 15:15–20. 2006.PubMed/NCBI
|
|
2
|
Chen J, Wang Y, Wu M, Yu K, Liu J and
Chang J: Vasculogenic mimicry triggers early recidivation and
resistance to adjuvant therapy in esophageal cancer. BMC Cancer.
24:11322024. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Miao K, Liu W, Xu J, Qian Z and Zhang Q:
Harnessing the power of traditional Chinese medicine monomers and
compound prescriptions to boost cancer immunotherapy. Front
Immunol. 14:12772432023. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Bray F, Laversanne M, Sung H, Ferlay J,
Siegel RL, Soerjomataram I and Jemal A: Global cancer statistics
2022: GLOBOCAN estimates of incidence and mortality worldwide for
36 cancers in 185 countries. CA Cancer J Clin. 74:229–263.
2024.PubMed/NCBI
|
|
5
|
Chen JG, Zhang YH, Lu JH and Kensler TW:
Liver cancer etiology: Old issues and new perspectives. Curr Oncol
Rep. 26:1452–1468. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Zheng N, Zhang S, Wu W, Zhang N and Wang
J: Regulatory mechanisms and therapeutic targeting of vasculogenic
mimicry in hepatocellular carcinoma. Pharmacol Res. 166:1055072021.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Duan X, Zou H, Yang J, Liu S, Xu T and
Ding J: Melittin-incorporated nanomedicines for enhanced cancer
immunotherapy. J Control Release. 375:285–299. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Chen Q, Lin W, Yin Z, Zou Y, Liang S, Ruan
S, Chen P, Li S, Shu Q, Cheng B and Ling C: Melittin inhibits
hypoxia-induced vasculogenic mimicry formation and
epithelial-mesenchymal transition through suppression of HIF-1α/Akt
pathway in liver cancer. Evid Based Complement Alternat Med.
2019:96029352019.PubMed/NCBI
|
|
9
|
Song X, Tan L, Wang M, Ren C, Guo C, Yang
B, Ren Y, Cao Z, Li Y and Pei J: Myricetin: A review of the most
recent research. Biomed Pharmacother. 134:1110172021. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Wang M, Ren S, Bi Z, Zhang L, Cui M, Sun
R, Bao J, Gao D, Yang B, Li X, et al: Myricetin reverses
epithelial-endothelial transition and inhibits vasculogenic mimicry
and angiogenesis of hepatocellular carcinoma by directly targeting
PAR1. Phytother Res. 36:1807–1821. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Xiao T, Zhang Q, Zong S, Zhong WL, Qin Y,
Bi Z, Chen S, Liu HJ, Wei JJ, et al: Protease-activated receptor-1
(PAR1) promotes epithelial-endothelial transition through Twist1 in
hepatocellular carcinoma. J Exp Clin Cancer Res. 37:1852018.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Zhang X, Zhang JG, Mu W, Zhou HM, Liu GL
and Li Q: The role of daurisoline treatment in hepatocellular
carcinoma: Inhibiting vasculogenic mimicry formation and enhancing
sensitivity to sorafenib. Phytomedicine. 92:1537402021. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Djati MS, Habibu H, Jatiatmaja NA and
Rifa'I M: Tapak liman (Elephantopus scaber L.)
extract-induced CD4+ and CD8+ differentiation from hematopoietic
stem cells and progenitor cell proliferation in mice (Mus
musculus L). AIP Conf Proc. Nov 29–2017.(Epub ahead of print).
View Article : Google Scholar
|
|
14
|
Wang J, Liao C, Huang G, Li W, Huang Y,
Liao Z and Huang S: Effect of Elephantopus scaber L. extract
on vasculogenic mimicry of hepatocellular carcinoma. J Chin Clin
Pharmacol. 38:652–656. 2022.(In Chinese).
|
|
15
|
Feng X, Yin Z, Ou S, Chu Z, Feng J, Luo Y,
Hu Y, Liu Y, Jiang W, Wang X and Wang H: The anti-tumor effects of
Celastrus orbiculatus Thunb. and its monomer composition: A review.
J Ethnopharmacol. 310:1163632023. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Chen J, Dai SY, Wu S, Wu MK, Yu KK, Liu
JC, Chang JY and Liu YQ: COE targets EphA2 to inhibit vasculogenic
mimicry formation induced by hypoxia in hepatocellular carcinoma.
Front Pharmacol. 15:14214702024. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Hsu JL, Leu WJ, Hsu LC, Hsieh CH and Guh
JH: Doxazosin inhibits vasculogenic mimicry in human non-small cell
lung cancer through inhibition of the VEGF-A/VE-cadherin/mTOR/MMP
pathway. Oncol Lett. 27:1702024. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Mao C, Zhang J, Yang C, Mei L, Feng Y, Dai
F, Huang Y, Xiao H and Deng B: BCAR1 facilitates the survival of
lung adenocarcinoma cells by augmenting the unfolded protein
response, autophagy, and the formation of vasculogenic mimicry.
Biochim Biophys Acta Mol Basis Dis. 1871:1675582025. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Lei C, Yu Y, Zhu Y, Li Y, Ma C, Ding L,
Han L and Zhang H: The most recent progress of baicalein in its
anti-neoplastic effects and mechanisms. Biomed Pharmacother.
176:1168622024. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Zhang Z, Nong L, Chen M, Gu X, Zhao W, Liu
M and Cheng W: Baicalein suppresses vasculogenic mimicry through
inhibiting RhoA/ROCK expression in lung cancer A549 cell line. Acta
Biochim Biophys Sin (Shanghai). 52:1007–1015. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Xiao H, Xu X, Du L, Li X, Zhao H, Wang Z,
Zhao L, Yang Z, Zhang S, Yang Y and Wang C: Lycorine and organ
protection: Review of its potential effects and molecular
mechanisms. Phytomedicine. 104:1542662022. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Zhao Z, Xiang S, Qi J, Wei Y, Zhang M, Yao
J, Zhang T, Meng M, Wang X and Zhou Q: Correction of the tumor
suppressor Salvador homolog-1 deficiency in tumors by lycorine as a
new strategy in lung cancer therapy. Cell Death Dis. 11:3872020.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Wang W, Li M, Wang L, Chen L and Goh BC:
Curcumin in cancer therapy: Exploring molecular mechanisms and
overcoming clinical challenges. Cancer Lett. 570:2163322023.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
An Q, Shi CX, Guo H, Xie SM, Yang YY, Liu
YN, Liu ZH, Zhou CZ and Niu FJ: Development and characterization of
octreotide-modified curcumin plus docetaxel micelles for potential
treatment of non-small-cell lung cancer. Pharm Dev Technol.
24:1164–1174. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Lin J, Wang Q, Zhou S, Xu S and Yao K:
Tetramethylpyrazine: A review on its mechanisms and functions.
Biomed Pharmacother. 150:1130052022. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Bin Liu, Meng Li, Jie He, Xiaoru Yan and
Zhang P: Effect of tetramethylpyrazine on vascular mimicry of A549
caner stem-like cell under hypoxia environment. Chin J Exp Tradit
Med Formulae. 28:64–70. 2022.(In Chinese).
|
|
27
|
Dai X, Zhang X, Chen W, Chen Y, Zhang Q,
Mo S and Lu J: Dihydroartemisinin: A potential natural anticancer
drug. Int J Biol Sci. 17:603–622. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Bingqi Hu, Jing Zhou, Junfeng Huang and
Chen L: A preliminary study of dihydroartemisinin inhibiting
migration invasion and vasculogenic mimicry of non-small cell lung
cancer cells. An Hui Yi Ke Da Xue. 58:766–771. 2023.(In
Chinese).
|
|
29
|
Jin L, Chen C, Huang L, Bu L, Zhang L and
Yang Q: Salvianolic acid A blocks vasculogenic mimicry formation in
human non-small cell lung cancer via PI3K/Akt/mTOR signalling. Clin
Exp Pharmacol Physiol. 48:508–514. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Si L and Lai Y: Pharmacological mechanisms
by which baicalin ameliorates cardiovascular disease. Front
Pharmacol. 15:14159712024. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Rahman M, Afzal O, Ullah SNMN, Alshahrani
MY, Alkhathami AG, Altamimi ASA, Almujri SS, Almalki WH, Shorog EM,
Alossaimi MA, et al: Nanomedicine-based drug-targeting in breast
cancer: Pharmacokinetics, clinical progress and challenges. ACS
Omega. 8:48625–48649. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
He X, Lan H, Jin K and Liu F: Can
immunotherapy reinforce chemotherapy efficacy? a new perspective on
colorectal cancer treatment. Front Immunol. 14:12377642023.
View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Choi HY and Chang JE: Targeted therapy for
cancers: From ongoing clinical trials to FDA-Approved drugs. Int J
Mol Sci. 24:136182023. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Léna H, Greillier L, Cropet C, Bylicki O,
Monnet I, Audigier-Valette C, Falchero L, Vergnenègre A, Demontrond
P, Geier M, et al: Nivolumab plus ipilimumab versus
carboplatin-based doublet as first-line treatment for patients with
advanced non-small-cell lung cancer aged ≥70 years or with an ECOG
performance status of 2 (GFPC 08-2015 ENERGY): A randomised,
open-label, phase 3 study. Lancet Respir Med. 13:141–152. 2025.
View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Randall LM and Monk BJ: Bevacizumab
toxicities and their management in ovarian cancer. Gynecol Oncol.
117:497–504. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Chong CR and Jänne PA: The quest to
overcome resistance to EGFR-targeted therapies in cancer. Nat Med.
19:1389–1400. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Walia GK, Kinra P and Akal RS: Potential
for tumor aggressiveness and its association with vasculogenic
mimicry marker in colorectal carcinomas in Indian population- A
tertiary care hospital level study. Indian J Pathol Microbiol.
68:261–266. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Zhang H, Chen L, Sun X, Yang Q, Wan L and
Guo C: Matrine: A promising natural product with various
pharmacological activities. Front Pharmacol. 11:5882020. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Peng X, Zhou D, Wang X, Hu Z, Yan Y and
Huang J: Matrine suppresses proliferation and invasion of SGC7901
cells through Inactivation of PI3K/Akt/uPA pathway. Ann Clin Lab
Sci. 46:457–462. 2016.PubMed/NCBI
|
|
40
|
Du Q, Lin Y, Ding C, Wu L, Xu Y and Feng
Q: Pharmacological activity of matrine in inhibiting colon cancer
cells VM formation, proliferation, and invasion by downregulating
claudin-9 mediated EMT process and MAPK signaling pathway. Drug Des
Devel Ther. 17:2787–2804. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Xiong X, Tang N, Lai X, Zhang J, Wen W, Li
X, Li A, Wu Y and Liu Z: Insights Into Amentoflavone: A natural
multifunctional biflavonoid. Front Pharmacol. 12:7687082021.
View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Feng N, Wang P, He M and Liu J: Effects of
Amentoflavone modulating HIF-1α/VEGF signalling pathway on
proliferation, apoptosis and angiogenic mimicry in colon cancer
cells. Chin J Gerontol. 45:701–707. 2025.(In Chinese).
|
|
43
|
Tan R, Li L and Luo D: Effectsofdel
phinidinonthebiolo gicalbehaviorandvasculo genic mimicr yformation
ofcoloncancercellsb yre gulatin gtheRhoA /ROCKsi gnalin gpathw.
Shaanxi Med J. 53:1468–1473. 2024.(In Chinese).
|
|
44
|
Lin L, Liu Y, Tang R, Ding S, Lin H and Li
H: Evodiamine: A extremely potential drug development candidate of
alkaloids from Evodia rutaecarpa. Int J Nanomedicine. 19:9843–9870.
2024. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Zeng D, Zhou P, Jiang R, Li XP, Huang SY,
Li DY, Li GL, Li LS, Zhao S, Hu L, et al: Evodiamine inhibits
vasculogenic mimicry in HCT116 cells by suppressing
hypoxia-inducible factor 1-alpha-mediated angiogenesis. Anticancer
Drugs. 32:314–322. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Mokadem I, Dijksterhuis WPM, Van Putten M,
Heuthorst L, de Vos-Geelen JM, Haj Mohammad N, Nieuwenhuijzen GAP,
van Laarhoven HWM and Verhoeven RHA: Recurrence after preoperative
chemotherapy and surgery for gastric adenocarcinoma: A multicenter
study. Gastric Cancer. 22:1263–1273. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Ramos A, Sadeghi S and Tabatabaeian H:
Battling chemoresistance in cancer: Root causes and strategies to
uproot them. Int J Mol Sci. 22:94512021. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Farkhondeh T, Pourbagher-Shahri AM,
Azimi-Nezhad M, Forouzanfar F, Brockmueller A, Ashrafizadeh M,
Talebi M, Shakibaei M and Samarghandian S: Roles of Nrf2 in gastric
cancer: Targeting for therapeutic strategies. Molecules.
26:31572021. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Montagnani F, Turrisi G, Marinozzi C,
Aliberti C and Fiorentini G: Effectiveness and safety of
oxaliplatin compared to cisplatin for advanced, unresectable
gastric cancer: A systematic review and meta-analysis. Gastric
Cancer. 14:50–55. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Kim HS, Won YJ, Shim JH, Kim HJ, Kim BS
and Hong HN: Role of EphA2-PI3K signaling in vasculogenic mimicry
induced by cancer-associated fibroblasts in gastric cancer cells.
Oncol Lett. 18:3031–3038. 2019.PubMed/NCBI
|
|
51
|
Zang M, Hou J, Huang Y, Wang J, Ding X,
Zhang B, Wang Y, Xuan Y and Zhou Y: Crocetin suppresses
angiogenesis and metastasis through inhibiting sonic hedgehog
signaling pathway in gastric cancer. Biochem Biophys Res Commun.
576:86–92. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Wang H, Ding Q, Zhou H, Huang C, Liu G,
Zhao X, Cheng Z and You X: Dihydroartemisinin inhibited
vasculogenic mimicry in gastric cancer through the FGF2/FGFR1
signaling pathway. Phytomedicine. 134:1559622024. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Lun Han and Yanfei Ma: Effects of
formononetin on proliferation and apoptosis of gastric cancer cells
and tumor vasculogenic mimicry by regulating HIF-1α/VEGF signaling
pathway. Int J Dig Dis. 43:297–303. 2023.
|
|
54
|
Nakhjavani M, Smith E, Townsend AR, Price
TJ and Hardingham JE: Anti-angiogenic properties of ginsenoside
Rg3. Molecules. 25:49052020. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Huatinng Li and Youwei Tang: Ginsenoside
Rg3 suppresses the formation of vasculogenic mimicry of gastric
cancer SGC7901 cells by inhibiting Wnt/β-catenin pathway in vitro.
Chin J Cancer Biother. 26:518–523. 2019.(In Chinese).
|
|
56
|
Yang P, Zhang Q, Shen H, Bai X, Liu P and
Zhang T: Research progress on the protective effects of aucubin in
neurological diseases. Pharm Biol. 60:1088–1094. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Song M, Zhou X, Liu J, Hu W, Zhang M and
Zheng X: Effects of aucubin on epithelial mesenchymal transition
and vasculogenic mimicry in gastric cancer MGC803 cells by
regulating the RhoA/ROCK signaling pathway. Chin J Cancer Biother.
31:365–370. 2024.(In Chinese).
|
|
58
|
Sun B, Zhou L, Wang X, Lv Y, Zhang H, Tu J
and Feng H: Effectsof jaceosidinon proliferation,
apoptosisandvasculo genicmimicr y ofgastriccancercellsa. Shaanxi
Med J. 53:1463–1467. 2024.(In Chinese).
|
|
59
|
Zhang D, Wang W, Zhou H, Su L, Han X,
Zhang X, Han W, Wang Y and Xue X: ANXA1: An important independent
prognostic factor and molecular target in glioma. Front Genet.
13:8515052022. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Ahmed R, Oborski MJ, Hwang M, Lieberman FS
and Mountz JM: Malignant gliomas: Current perspectives in
diagnosis, treatment, and early response assessment using advanced
quantitative imaging methods. Cancer Manag Res. 6:149–170.
2014.PubMed/NCBI
|
|
61
|
Nabors LB, Portnow J, Ahluwalia M,
Baehring J, Brem H, Brem S, Butowski N, Campian JL, Clark SW,
Fabiano AJ, et al: Central nervous system cancers, version 3.2020,
NCCN clinical practice guidelines in oncology. J Natl Compr Cancer
Netw. 18:1537–1570. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
62
|
Liguori GL: Challenges and promise for
glioblastoma treatment through extracellular vesicle inquiry.
Cells. 13:3362024. View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Zeng Z, Chen Y, Geng X, Zhang Y, Wen X,
Yan Q, Wang T, Ling C, Xu Y, Duan J, et al: NcRNAs: Multi-angle
participation in the regulation of glioma chemotherapy resistance
(Review). Int J Oncol. 60:762022. View Article : Google Scholar : PubMed/NCBI
|
|
64
|
Chen YS and Chen ZP: Vasculogenic mimicry:
A novel target for glioma therapy. Chin J Cancer. 33:74–79. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
65
|
Li X, Shi L, Li Y and Li Q, Duan X, Wang Y
and Li Q: The enhanced treatment efficacy of invasive brain glioma
by dual-targeted artemether plus paclitaxel micelles. Artif Cells
Nanomed Biotechnol. 48:983–996. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
66
|
Chen Z, Ding W, Yang X, Lu T and Liu Y:
Isoliquiritigenin, a potential therapeutic agent for treatment of
inflammation-associated diseases. J Ethnopharmacol. 318:1170592024.
View Article : Google Scholar : PubMed/NCBI
|
|
67
|
Sun QH, Li ZY, Sun JJ, Liang SL, Li H and
LE LM: Research advances in isoliquiritigenin against glioma.
Journal of International Neurology and Neurosurgery. 49:89–92.
2022.(In Chinese).
|
|
68
|
Van Der Hoorn A, Yan JL, Larkin TJ,
Boonzaier NR, Matys T and Price SJ: Posttreatment apparent
diffusion coefficient changes in the periresectional area in
patients with glioblastoma. World Neurosurg. 92:159–165. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
69
|
Oronsky B, Reid TR, Oronsky A, Sandhu N
and Knox SJ: A review of newly diagnosed glioblastoma. Front Oncol.
10:5740122021. View Article : Google Scholar : PubMed/NCBI
|
|
70
|
Brown NF, Ottaviani D, Tazare J, Gregson
J, Kitchen N, Brandner S, Fersht N and Mulholland P: Survival
outcomes and prognostic factors in glioblastoma. Cancers (Basel).
14:31612022. View Article : Google Scholar : PubMed/NCBI
|
|
71
|
Min P, Li Y, Wang C, Fan J, Liu S, Chen X,
Tang Y, Han F, Zhang A and Feng L: Cyclopeptide moroidin inhibits
vasculogenic mimicry formed by glioblastoma cells via regulating
β-catenin activation and EMT pathways. J Biomed Res. 38:322–333.
2024. View Article : Google Scholar : PubMed/NCBI
|
|
72
|
Campanile M, Cuomo O, Brancaccio P,
Vinciguerra A, Casamassa A, Pastorino O, Volpicelli F, Gentile MT,
Amoroso S, Annunziato L, et al: Ruta graveolens water
extract (RGWE) ameliorates ischemic damage and improves
neurological deficits in a rat model of transient focal brain
ischemia. Biomed Pharmacother. 154:1135872022. View Article : Google Scholar : PubMed/NCBI
|
|
73
|
Camerino I, Franco P, Bajetto A, Thellung
S, Florio T, Stoppelli MP and Colucci-D'Amato L: Ruta
graveolens, but not Rutin, inhibits survival, migration,
invasion, and vasculogenic mimicry of glioblastoma cells. Int J Mol
Sci. 25:117892024. View Article : Google Scholar : PubMed/NCBI
|
|
74
|
Gao B, Wang MD, Li Y and Huang F: Risk
stratification system and web-based nomogram constructed for
predicting the overall survival of primary osteosarcoma patients
after surgical resection. Front Public Health. 10:9495002022.
View Article : Google Scholar : PubMed/NCBI
|
|
75
|
Lilienthal I and Herold N: Targeting
molecular mechanisms underlying treatment efficacy and resistance
in osteosarcoma: A review of current and future strategies. Int J
Mol Sci. 21:68852020. View Article : Google Scholar : PubMed/NCBI
|
|
76
|
Shah A, Cardoso FN, Souza F, Montreuil J,
Pretell-Mazzini J, Temple HT, Hornicek F, Crawford B and Subhawong
TK: Failure modes in orthopedic oncologic reconstructive surgery: A
review of imaging findings and failure rates. Curr Oncol.
31:6245–6266. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
77
|
Yan L, Li R, Li D, Zhu Y, Lv Z and Wang B:
Development of a novel vasculogenic mimicry-associated gene
signature for the prognostic assessment of osteosarcoma patients.
Clin Transl Oncol. 25:3501–3518. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
78
|
Ke F, Zhang R, Chen R, Guo X, Song C, Gao
X, Zeng F and Liu Q: The role of Rhizoma Paridis saponins on
anti-cancer: The potential mechanism and molecular targets.
Heliyon. 10:e373232024. View Article : Google Scholar : PubMed/NCBI
|
|
79
|
Yao N, Zhou J, Jiang Y, Jin Q, Zhu H,
Zhang J and Li Z: Rhizoma Paridis saponins suppresses vasculogenic
mimicry formation and metastasis in osteosarcoma through regulating
miR-520d-3p/MIG-7 axis. J Pharmacol Sci. 150:180–190. 2022.
View Article : Google Scholar : PubMed/NCBI
|
|
80
|
Hong Fang, Qun Zhang and Longbang Chen:
The Effects of Curcumin on Vasculogenic Mimicry in Vitro of Sarcoma
Cells and Its Mechanism. Western J Trad Chin Med. 33:21–25.
2020.(In Chinese).
|
|
81
|
Ke Ren, Jing Zhou, Zhenlin Li, Shihui
Qian, He Li and Chen Wang: Effects of Paris polyphylla
ethanol extract on human osteosarcoma and its mechanism. Chin Trad
Herbal Drugs. 50:120–128. 2019.(In Chinese).
|
|
82
|
Bilmin K, Synoradzki KJ, Czarnecka AM,
Spałek MJ, Kujawska T, Solnik M, Merks P, Toro MD, Rejdak R and
Fiedorowicz M: New perspectives for eye-sparing treatment
strategies in primary uveal melanoma. Cancers (Basel). 14:1342021.
View Article : Google Scholar : PubMed/NCBI
|
|
83
|
Bai H, Bosch JJ and Heindl LM: Current
management of uveal melanoma: A review. Clin Exp Ophthalmol.
51:484–494. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
84
|
Sabazade S, Gill V, Herrspiegel C and
Stålhammar G: Vasculogenic mimicry correlates to presenting
symptoms and mortality in uveal melanoma. J Cancer Res Clin Oncol.
148:587–597. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
85
|
Yuan Y, Geng B, Xu X, Zhao H, Bai J, Dou
Z, Jia S, Yu X and Luo W: Dual VEGF/PDGF knockdown suppresses
vasculogenic mimicry formation in choroidal melanoma cells via the
Wnt5a/β-catenin/AKT signaling pathway. Acta Histochem.
124:1518422022. View Article : Google Scholar : PubMed/NCBI
|
|
86
|
Liu X, Cai Y, Zhang Y, Zhang H, Tian S,
Gong Y, Song Q, Chen X, Ma X, Wen Y, et al: Artesunate: A potential
drug for the prevention and treatment from hepatitis to
hepatocellular carcinoma. Pharmacol Res. 210:1075262024. View Article : Google Scholar : PubMed/NCBI
|
|
87
|
Ma QY, Xu XY, Zhu YZ, Yao NN, Liu YC, Gao
XD, Zhang Q and Luo WJ: Artesunate inhibits vasculogenic mimicry in
choroidal melanoma through HIF-1α/ VEGF/PDGF pathway. Acta
Histochem. 126:1521742024. View Article : Google Scholar : PubMed/NCBI
|
|
88
|
Geng B, Zhu Y, Yuan Y, Bai J, Dou Z, Sui A
and Luo W: Artesunate Suppresses Choroidal Melanoma Vasculogenic
Mimicry Formation and Angiogenesis via the Wnt/CaMKII Signaling
Axis. Front Oncol. 11:7146462021. View Article : Google Scholar : PubMed/NCBI
|
|
89
|
Li JX, Zhang J, Wei CX, Zhang H, Pei C,
Zhao F and Cai ZP: Effect of Artesunate on Experimental Choroidal
Neovascularization and Expression of HIF-1α and VEGF. Chinese
Journal of Experimental Traditional Medical Formulae. 27:83–89.
2021.(In Chinese).
|
|
90
|
Ma QY, Liu YC, Zhang Q, Yi WD, Sun Y, Gao
XD, Zhao XT, Wang HW, Lei K and Luo WJ: Integrating network
pharmacology, molecular docking and experimental verification to
reveal the mechanism of artesunate in inhibiting choroidal
melanoma. Front Pharmacol. 14483812024. View Article : Google Scholar : PubMed/NCBI
|
|
91
|
Zhu M, Sun Y, Su Y, Guan W, Wang Y, Han J,
Wang S, Yang B, Wang Q and Kuang H: Luteolin: A promising
multifunctional natural flavonoid for human diseases. Phytother
Res. 38:3417–3443. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
92
|
Chen YF, Wu S, Li X, Chen M and Liao HF:
Luteolin suppresses three angiogenesis modes and cell interaction
in uveal melanoma in vitro. Curr Eye Res. 47:1590–1599. 2022.
View Article : Google Scholar : PubMed/NCBI
|
|
93
|
Shi M, Wu W, Luo H, Yu J and Liao H:
Inhibitory effects of luteolin on vasculogenic mimicry in human
choroidal melanoma cell line C918. Recent Adv Ophthalmol.
40:731–735. 2020.
|
|
94
|
Li X, Chen M, Lei X, Huang M, Ye W, Zhang
R and Zhang D: Luteolin inhibits angiogenesis by blocking Gas6/Axl
signaling pathway. Int J Oncol. 51:677–685. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
95
|
Wu S, Xu J, Ma Y, Liang G, Wang J and Sun
T: Advances in the mechanism of CDK4/6 inhibitor resistance in
HR+/HER2- breast cancer. Ther Adv Med Oncol.
16:175883592412824992024. View Article : Google Scholar : PubMed/NCBI
|
|
96
|
Wang Y and Minden A: Current molecular
combination therapies used for the treatment of breast cancer. Int
J Mol Sci. 23:110462022. View Article : Google Scholar : PubMed/NCBI
|
|
97
|
Morales-Guadarrama G, García-Becerra R,
Méndez-Pérez EA, García-Quiroz J, Avila E and Díaz L: Vasculogenic
mimicry in breast cancer: Clinical relevance and drivers. Cells.
10:17582021. View Article : Google Scholar : PubMed/NCBI
|
|
98
|
Xu MR, Wei PF, Suo MZ, Hu Y, Ding W, Su L,
Zhu YD, Song WJ, Tang GH, Zhang M and Li P: Brucine suppresses
vasculogenic mimicry in human triple-negative breast cancer cell
line MDA-MB-231. Biomed Res Int. 2019:65432302019.PubMed/NCBI
|
|
99
|
Zhang M, Meng M, Liu Y, Qi J, Zhao Z, Qiao
Y, Hu Y, Lu W, Zhou Z, Xu P and Zhou Q: Triptonide effectively
inhibits triple-negative breast cancer metastasis through
concurrent degradation of Twist1 and Notch1 oncoproteins. Breast
Cancer Res. 23:1162021. View Article : Google Scholar : PubMed/NCBI
|
|
100
|
Yang D and Zhang QF: The natural source,
physicochemical properties, biological activities and metabolism of
astilbin. Crit Rev Food Sci Nutr. 63:9506–9518. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
101
|
Sharma A, Gupta S, Chauhan S, Nair A and
Sharma P: Astilbin: A promising unexplored compound with
multidimensional medicinal and health benefits. Pharmacol Res.
158:1048942020. View Article : Google Scholar : PubMed/NCBI
|
|
102
|
Wang Y, Gu Y, Liu Q, Ma S and Gong Z:
Astilbin inhibits proliferation, migration and vasculogenic mimicry
formation of breast cancer cells by regulating HIF-1α/VEGF axis.
Chin J Cancer Biother. 31:796–802. 2024.(In Chinese).
|
|
103
|
Haq S, Ali S, Mohammad R and Sarkar FH:
The complexities of epidemiology and prevention of gastrointestinal
cancers. Int J Mol Sci. 13:12556–12572. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
104
|
Maloney S, Clarke SJ, Sahni S, Hudson A,
Colvin E, Mittal A, Samra J and Pavlakis N: The role of diagnostic,
prognostic, and predictive biomarkers in the management of early
pancreatic cancer. J Cancer Res Clin Oncol. 149:13437–13450. 2023.
View Article : Google Scholar : PubMed/NCBI
|
|
105
|
Zhang Z, Zhou N, Guo X, Li N, Zhu H and
Yang Z: Pretherapeutic assessment of pancreatic cancer: Comparison
of FDG PET/CT plus delayed PET/MR and contrast-enhanced CT/MR.
Front Oncol. 11:7904622022. View Article : Google Scholar : PubMed/NCBI
|
|
106
|
Del Chiaro M, Sugawara T, Karam SD and
Messersmith WA: Advances in the management of pancreatic cancer.
BMJ. 383:e0739952023. View Article : Google Scholar : PubMed/NCBI
|
|
107
|
Benjakul N, Prakobphol N,
Tangshewinsirikul C, Dulyaphat W, Svasti J, Charngkaew K and
Kangsamaksin T: Notch signaling regulates vasculogenic mimicry and
promotes cell morphogenesis and the epithelial-to-mesenchymal
transition in pancreatic ductal adenocarcinoma. PLoS One.
17:e02790012022. View Article : Google Scholar : PubMed/NCBI
|
|
108
|
Zhao T, Wang Z and Lin S: Inhibitory
effect of ginsenoside Rg3 on vasculogenic mimicry in pancreatic
cancer. Chin J Trad Chin MedPharm. 39:5509–5513. 2024.
|
|
109
|
Cai X, Wang Z, Lin S, Chen H and Bu H:
Ginsenoside Rg3 suppresses vasculogenic mimicry by impairing
DVL3-maintained stemness via PAAD cell-derived exosomal miR-204 in
pancreatic adenocarcinoma. Phytomedicine. 126:1554022024.
View Article : Google Scholar : PubMed/NCBI
|
|
110
|
Zhao T, Wang Z and Lin S: Ginsenoside Rg3
inhibits epithelial-mesenchymal transition and vasculogenic mimicry
of pancreatic cancer cells through PI3K signaling. Chin Trad Herbal
Drugs. 55:6261–6268. 2024.(In Chinese).
|
|
111
|
Pandiar D, Smitha T and Krishnan RP:
Vasculogenic mimicry. J Oral Maxillofac Pathol. 27:228–229. 2023.
View Article : Google Scholar : PubMed/NCBI
|
|
112
|
Tian X, Si Q, Liu M, Shi J, Zhao R, Xiong
Y, Yu L, Cui H and Guan H: Advance in vasculogenic mimicry in
ovarian cancer (Review). Oncol Lett. 26:4562023. View Article : Google Scholar : PubMed/NCBI
|
|
113
|
Lim D, Do Y, Kwon BS, Chang W, Lee MS, Kim
J and Cho JG: Angiogenesis and vasculogenic mimicry as therapeutic
targets in ovarian cancer. BMB Rep. 53:291–298. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
114
|
Wang N, Yang Y, Wang H, Li Y, Wang M and
Li Q: Cinobufagin modulates vasculogenic mimicry and
tumor-associated macrophages to inhibit ovarian cancer progression.
Eur J Pharmacol:. 987:1771572025. View Article : Google Scholar : PubMed/NCBI
|
|
115
|
Wang XY, Zeng HQ and Hu W: Cinobufagin
alleviates bone cancer pain by inhibiting the spinal CCL2/CCR2
pathway and microglial activation. Acta Pharmaceutica Sinica.
55:67–73. 2020.(In Chinese).
|
|
116
|
Farhan M: Green tea catechins: Nature's
way of preventing and treating cancer. Int J Mol Sci. 23:107132022.
View Article : Google Scholar : PubMed/NCBI
|
|
117
|
Sicard AA, Dao T, Suarez NG and Annabi B:
Diet-derived gallated catechins prevent TGF-β-mediated
epithelial-mesenchymal transition, cell migration and vasculogenic
mimicry in chemosensitive ES-2 ovarian cancer cells. Nutr Cancer.
73:169–180. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
118
|
Jiang S, Li S, Pang S, Liu M, Sun H, Zhang
N and Liu J: A systematic review: Sinomenine. Heliyon.
10:e299762024. View Article : Google Scholar : PubMed/NCBI
|
|
119
|
Zhenyu Yan, Meng Guo, Ran Yang, Bo Su and
Haiyan Zhang: Effects of Sinomenine regulating CXCR4-STAT3 axis on
proliferation, migration and angiogenic mimicry of ovarian cancer
A2780 cells. J Mod Oncol. 32:2944–2951. 2024.
|
|
120
|
Zhou TJ, Huang XH, Gong L and Xiang L:
Vasculogenic mimicry and hypoxia-inducible factor-1α expression in
cervical squamous cell carcinoma. Genet Mol Res.
15:150173962016.PubMed/NCBI
|
|
121
|
Tang H, Chen L, Liu X, Zeng S, Tan H and
Chen G: Pan-cancer dissection of vasculogenic mimicry
characteristic to provide potential therapeutic targets. Front
Pharmacol. 15:13467192024. View Article : Google Scholar : PubMed/NCBI
|
|
122
|
Sun J, Tian Z, Wu J, Li J, Wang Q, Huang S
and Wang M: Pristimerin exerts pharmacological effects through
multiple signaling pathways: A comprehensive review. Drug Des Dev
Ther. 18:1673–1694. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
123
|
Jianwei Luo, Hongke Huang and Yanli Hu:
Effects of pristimerin on the proliferation, apoptosis and
vasculogenic mimicry of cervical cancer HeLa cells by regulating
the Shh/Gli1 signaling pathway. Chin J Cancer Biother. 31:687–693.
2024.(In Chinese).
|
|
124
|
Chen C, Zhang QW, Ye Y and Lin LG:
Honokiol: A naturally occurring lignan with pleiotropic
bioactivities. Chin J Nat Med. 19:481–490. 2021.PubMed/NCBI
|
|
125
|
Xiaocong Jiang, Xiuhua Pan, Wenjuan Cai,
Chi Zhang and Yuhong Lan: Effects of honokiol on vasculogenic
mimicry and epithelial-mesenchymal transition of cervical cancer
cells. World Clinical Drug. 44:922–932. 2023.
|
|
126
|
Li Y, Hou M, Lu G, Ciccone N, Wang X and
Zhang H: The prognosis of anti-angiogenesis treatments combined
with standard therapy for newly diagnosed glioblastoma: A
meta-analysis of randomized controlled trials. PLoS One.
11:e01682642016. View Article : Google Scholar : PubMed/NCBI
|
|
127
|
Hujanen R, Almahmoudi R, Karinen S, Nwaru
BI, Salo T and Salem A: Vasculogenic mimicry: A promising
prognosticator in head and neck squamous cell carcinoma and
esophageal cancer? A systematic review and meta-analysis. Cells.
9:5072020. View Article : Google Scholar : PubMed/NCBI
|
|
128
|
Peng D, Qiao HZ, Tan HY, Wang YX, Luo D,
Qiao LJ, Cai YF, Zhang SJ, Wang Q and Guan L: Ligustilide
ameliorates cognitive impairment via AMPK/SIRT1 pathway in vascular
dementia rat. Metab Brain Dis. 37:1401–1414. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
129
|
Hao K, Wang X, Liu Z, Liu D and Li J:
Effects of ligustilide regulating RhoA/ROCK signaling pathway on
biological behavior of esophageal cancer cells. Tianjin Medical
Journal. 52:1164–1170. 2024.(In Chinese).
|
|
130
|
Li B, Mao X, Wang H, Su G, Mo C, Cao K and
Qiu S: Vasculogenic mimicry in bladder cancer and its association
with the aberrant expression of ZEB1. Oncol Lett. 15:5193–5200.
2018.PubMed/NCBI
|
|
131
|
Zhong W and Zhang Q: Atractylodin: An
alkyne compound with anticancer potential. Am J Chin Med.
52:1729–1757. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
132
|
Lai H, Li Q, Lu Y, Liu Y, Qin X and Wei R:
Impacts of Atractylonin on Proliferation, Apoptosis and
Vasculogenic Mimicry of Bladder Cancer Cells by Regulating
RhoA/ROCK1 Signal Pathway. Chin J Cell Biol. 46:1497–1505. 2024.(In
Chinese).
|
|
133
|
Tímár J and Ladányi A: Molecular pathology
of skin melanoma: Epidemiology, differential diagnostics, prognosis
and therapy prediction. Int J Mol Sci. 23:53842022. View Article : Google Scholar : PubMed/NCBI
|
|
134
|
Zhang Z, Imani S, Shasaltaneh MD,
Hosseinifard H, Zou L, Fan Y and Wen Q: The role of vascular
mimicry as a biomarker in malignant melanoma: A systematic review
and meta-analysis. BMC Cancer. 19:11342019. View Article : Google Scholar : PubMed/NCBI
|
|
135
|
Liu K, Zhang X, Xie L, Deng M, Chen H,
Song J, Long J, Li X and Lou J: Lupeol and its derivatives as
anticancer and anti-inflammatory agents: Molecular mechanisms and
therapeutic efficacy. Pharmacol Res. 164:1053732021. View Article : Google Scholar : PubMed/NCBI
|
|
136
|
Bhattacharyya S, Mitra D, Ray S, Biswas N,
Banerjee S, Majumder B, Mustafi SM and Murmu N: Reversing effect of
Lupeol on vasculogenic mimicry in murine melanoma progression.
Microvasc Res. 121:52–62. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
137
|
Singh J, Trabulsi EJ and Gomella LG: Is
there an optimal management for localized prostate cancer? Clin
Interv Aging. 5:187–197. 2010.PubMed/NCBI
|
|
138
|
Kurganovs NJ and Engedal N: To eat or not
to eat: A critical review on the role of autophagy in prostate
carcinogenesis and prostate cancer therapeutics. Front Pharmacol.
15:14198062024. View Article : Google Scholar : PubMed/NCBI
|
|
139
|
Pender GC, Ombaka JH, Mwitari PG and Guyah
B: Common biological factors implicated in benign prostatic
hyperplasia and prostate cancer, conventional and phytotherapeutic
approaches employed in management: A review. Int J Sci Res Arch.
11:049–074. 2023. View Article : Google Scholar
|
|
140
|
Liu R, Yang K, Meng C, Zhang Z and Xu Y:
Vasculogenic mimicry is a marker of poor prognosis in prostate
cancer. Cancer Biol Ther. 13:527–533. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
141
|
Yeo C, Han DS, Lee HJ and Lee EO:
Epigallocatechin-3-gallate suppresses vasculogenic mimicry through
inhibiting the twist/VE-Cadherin/AKT pathway in human prostate
cancer PC-3 cells. Int J Mol Sci. 21:4392020. View Article : Google Scholar : PubMed/NCBI
|
|
142
|
Nejabati HR and Roshangar L: Kaempferol: A
potential agent in the prevention of colorectal cancer. Physiol
Rep. 10:e154882022. View Article : Google Scholar : PubMed/NCBI
|
|
143
|
Da J, Xu M, Wang Y, Li W, Lu M and Wang Z:
Kaempferol promotes apoptosis while inhibiting cell proliferation
via androgen-dependent pathway and suppressing vasculogenic mimicry
and invasion in prostate cancer. Anal Cell Pathol (Amst).
2019:19076982019.PubMed/NCBI
|
|
144
|
Ren B, Kwah MX, Liu C, Ma Z, Shanmugam MK,
Ding L, Xiang X, Ho PC, Wang L, Ong PS and Goh BC: Resveratrol for
cancer therapy: Challenges and future perspectives. Cancer Lett.
515:63–72. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
145
|
Han DS, Lee HJ and Lee EO: Resveratrol
suppresses serum-induced vasculogenic mimicry through impairing the
EphA2/twist-VE-cadherin/AKT pathway in human prostate cancer PC-3
cells. Sci Rep. 12:201252022. View Article : Google Scholar : PubMed/NCBI
|
|
146
|
Dudley AC and Griffioen AW: Pathological
angiogenesis: Mechanisms and therapeutic strategies. Angiogenesis.
26:313–347. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
147
|
Qin L, Ren Y, Chen AM, Guo FJ, Xu F, Gong
C, Cheng P, Du Y and Liao H: Peroxisome proliferator-activated
receptor γ ligands inhibit VEGF-mediated vasculogenic mimicry of
prostate cancer through the AKT signaling pathway. Mol Med Rep.
10:276–282. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
148
|
Pinto MP, Sotomayor P, Carrasco-Avino G,
Corvalan AH and Owen GI: Escaping Antiangiogenic Therapy:
Strategies Employed by Cancer Cells. Int J Mol Sci. 17:14892016.
View Article : Google Scholar : PubMed/NCBI
|
|
149
|
Luo Q, Wang J, Zhao W, Peng Z, Liu X, Li
B, Zhang H, Shan B, Zhang C and Duan C: Vasculogenic mimicry in
carcinogenesis and clinical applications. J Hematol Oncol.
13:192020. View Article : Google Scholar : PubMed/NCBI
|
|
150
|
Chen N, Jiang D, Shao B, Bai T, Chen J,
Liu Y, Zhang Z, Zhou Y, Wang X and Zhu Z: Anti-BVDV activity of
traditional Chinese medicine monomers targeting NS5B (RNA-Dependent
RNA Polymerase) in vitro and in vivo. Molecules. 28:34132023.
View Article : Google Scholar : PubMed/NCBI
|
|
151
|
Hu Z, Wang X, Hu Q and Chen X: Exploring
the protective effects of herbal monomers against diabetic
retinopathy based on the regulation of autophagy and apoptosis: A
review. Medicine (Baltimore). 102:e355412023. View Article : Google Scholar : PubMed/NCBI
|
|
152
|
Lv D, Liu Y, Tang R, Fu S, Kong S, Liao Q,
Li H and Lin L: Analysis of clinical trials using anti-tumor
traditional Chinese medicine monomers. Drug Des Devel Ther.
18:1997–2020. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
153
|
Li Z, Zhu J and Ouyang H: Research
progress of traditional Chinese medicine in improving hepatic
fibrosis based on inhibiting pathological angiogenesis. Front
Pharmacol. 14:13030122023. View Article : Google Scholar : PubMed/NCBI
|
|
154
|
Zheng T, Jiang T, Huang Z, Ma H and Wang
M: Role of traditional Chinese medicine monomers in cerebral
ischemia/reperfusion injury:a review of the mechanism. Front
Pharmacol. 14:12208622023. View Article : Google Scholar : PubMed/NCBI
|
|
155
|
Ye R, Zheng Y, Chen Y, Wei X, Shi S, Chen
Y, Zhu W, Wang A, Yang L, Xu Y and Peng J: Stable loading and
delivery of melittin with lipid-coated polymeric nanoparticles for
effective tumor therapy with negligible systemic toxicity. ACS Appl
Mater Interfaces. 13:55902–55912. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
156
|
Ke M, Dong J, Wu R and Lv Y: The
inhibitory effect of Melittin-K1, a mutant of Melittin, on the
growth of human hepatocellular carcinoma cell line BEL-7402. Modern
Oncology. 27:2636–2641. 2019.(In Chinese).
|
|
157
|
Sui X, Han X, Chen P, Wu Q, Feng J, Duan
T, Chen X, Pan T, Yan L, et al: Baicalin Induces Apoptosis and
Suppresses the Cell Cycle Progression of Lung Cancer Cells Through
Downregulating Akt/mTOR Signaling Pathway. Front Mol Biosci.
28:6022822021. View Article : Google Scholar : PubMed/NCBI
|
|
158
|
Wang R, Wang C, Lu L, Yuan F and He F:
Baicalin and baicalein in modulating tumor microenvironment for
cancer treatment: A comprehensive review with future perspectives.
Pharmacol Res. 199:1070322024. View Article : Google Scholar : PubMed/NCBI
|
|
159
|
Cao H, Li J, Sun S, Fang J, Wang Y, Zhang
Y and Mao W: Pharmacodynamic Effect and Mechanism of Baicalein on
Lung Adenocarcinoma A549 cells. Chinese Journal of Experimental
Traditional Medical Formulae. 23:98–103. 2017.(In Chinese).
|
|
160
|
Deng X, Ruan X and Hao H: Study on matrix
sustained release tablets of brucine solid lipid nanoparticles.
Chinese Traditional and Herbal Drugs. 49:5298–5304. 2018.(In
Chinese).
|
|
161
|
Guan Q, Wang L, Liu Z, Feng Y, Wen M, Zhao
Y, Feng W and Lyu S: Cytotoxicity and Cellular Uptake Test of
Brucine-loaded Solid Lipid Nanoparticles. Chinese Journal of
Experimental Traditional Medical Formulae. 23:1–6. 2017.(In
Chinese).
|
|
162
|
Pellerino A, Bruno F, Soffietti R and Rudà
R: Antiangiogenic therapy for malignant brain tumors: Does it still
matter? Curr Oncol Rep. 25:777–785. 2023. View Article : Google Scholar : PubMed/NCBI
|