Introduction
Gastric cancer is one of the most common cancers,
with specific geographical, ethnic and socioeconomic differences in
incidence. These disparities are associated with key risk factors:
High-incidence regions like Eastern Asia correlate with prevalent
Helicobacter pylori infection and high-salt dietary
patterns; ethnically, certain populations exhibit genetic
susceptibility to gastric mucosal damage; socioeconomically,
low-status groups face elevated risk due to limited access to
screening and higher exposure to risk factors such as smoking and
alcohol consumption (1). A total of
>70% of gastric cancer cases occur in developing countries and
most patients come from Eastern Asia (1). Furthermore, the age-standardized
incidence rate of gastric cancer is 13.0 per 100,000 in male and
6.1 per 100,000 in female patients (2).
Pregnancy-associated gastric cancer is defined as a
diagnosis of gastric cancer during pregnancy or ≤1 year after
delivery, and it is estimated to complicate 0.026–0.1% of all
pregnancies (3). Gastric cancer
detected during pregnancy is usually diagnosed in advanced stages
with a poor maternal and fetal prognosis (4). Most cases of pregnancy-associated
gastric cancer are diagnosed at an advanced stage as special
gastrointestinal symptoms are generally overlooked during
pregnancy, and there are several limitations and contraindications
for using diagnostic tools during pregnancy. Current treatment
adopts a multidisciplinary approach balancing maternal and fetal
interests (5). Surgical resection
is the primary curative option, though resectability is limited by
late diagnosis. Chemotherapy is considered for advanced cases in
the second/third trimesters (agents like 5-fluorouracil), while
radiotherapy is avoided due to teratogenicity (3). Survival rates in cases of
gastrointestinal cancer are strictly associated with the early
diagnosis. The maternal prognosis is poor, with 1- and 2-year
survival rates of 18.0 and 15.1%, respectively (6).
The present report describes a case of a 35-year-old
female patient, who was diagnosed with an advanced gastrointestinal
cancer. At the time of admission, the patient was 18 weeks
pregnant. The patient died 1 month after onset of the disease.
Case report
A 35-year-old female patient (gravida 2, para 1)
presented for in vitro fertilization and embryo transfer due
to tubal obstruction. During routine prenatal checkups in May 2019
at the Affiliated Heping Hospital of Changzhi Medical College
(Shanxi, China the patient reported lower abdominal fullness,
without cervical shortening, vaginal bleeding, fluid discharge or
uterine contractions noted 1 month prior to admission. The patient
was initially diagnosed with threatened abortion and treated with
10 mg dydrogesterone twice daily; however, the medication was
discontinued after 2 weeks due to a lack of clinical
improvement.
A total of 2 weeks before admission to the
Affiliated Heping Hospital of Changzhi Medical College, the patient
developed progressive nausea, abdominal distension and anorexia. Up
to this time, the patient had undergone three ultrasound
examinations: i) A scan at 6 weeks of gestation confirming an
intrauterine pregnancy, with clear visualization of the fetal
heartbeat and fetal pole; ii) a nuchal translucency ultrasound; and
iii) an ultrasound performed due to lower abdominal bloating
(original images not available). None of these examinations
revealed the presence of ascites. However, 1 week before admission,
the abdominal distension notably worsened, leading to an ultrasound
that revealed substantial ascites (data not shown). Paracentesis
was performed, draining 1,000 ml ascitic fluid for analysis.
Despite this intervention, the abdominal pain and distension
worsened 5 days later, prompting transfer to Peking University
First Hospital, Beijing, China.
The local pathology report of the ascitic fluid
showed a predominance of mesothelial cells, along with scattered
lymphocytes, neutrophils and atypical cells (data not shown).
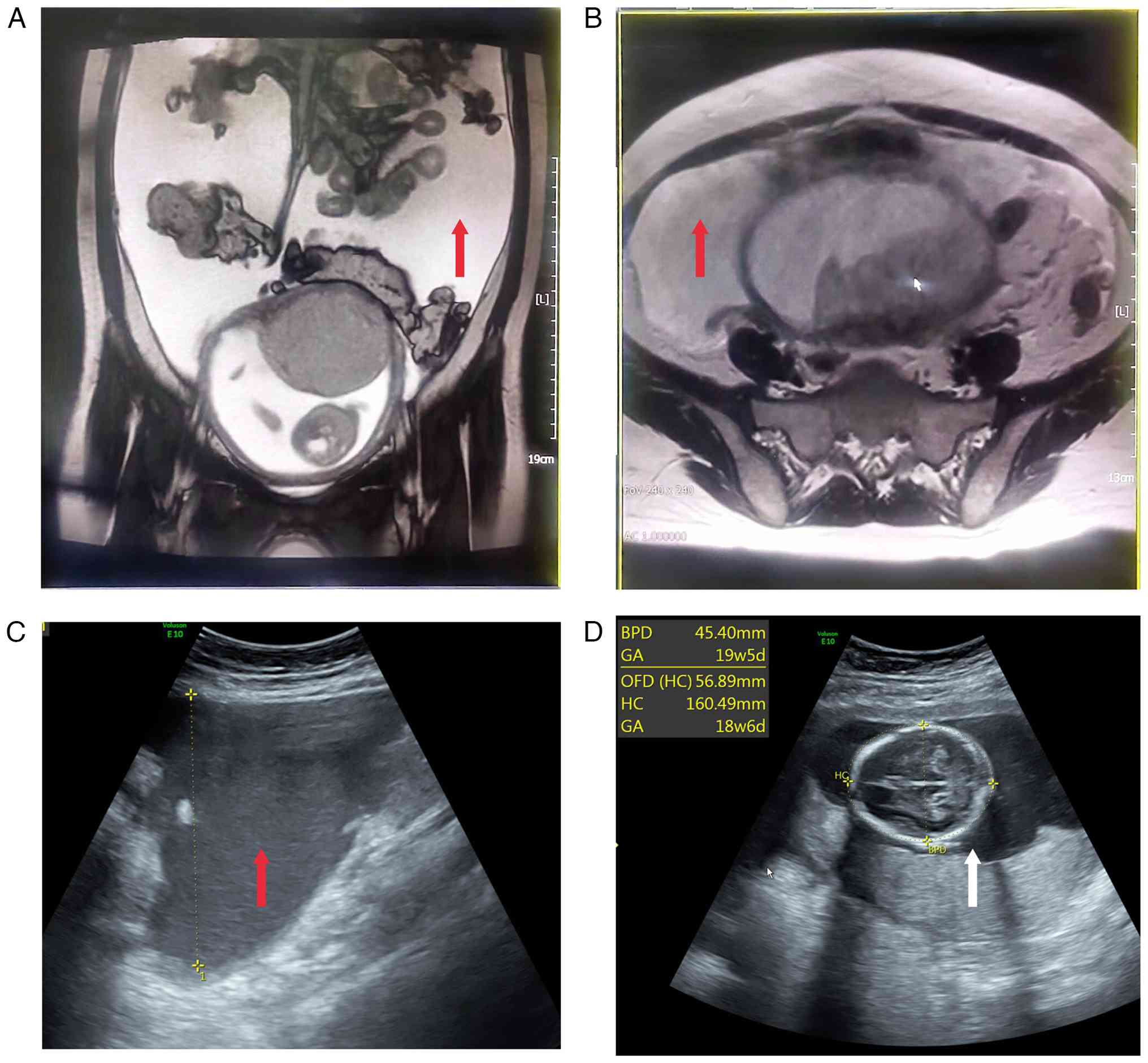
Moreover, non-enhanced pelvic MRI at the local hospital revealed
nodular thickening of the peritoneum, raising concerns about tumor
metastasis or peritoneal tuberculosis, along with notable ascites
showing fluid stratification (Fig. 1A
and B). Due to limitations in diagnostic and treatment options
at the local facility, the patient was referred to Peking
University First Hospital for further management.
Since the onset of symptoms, the patient experienced
anorexia, mental fatigue, oliguria, constipation and an 8 kg weight
loss. The medical history of the patient included severe anemia in
2003, which was diagnosed as M3 leukemia following a bone marrow
aspiration. After undergoing chemotherapy, the patient remained in
remission without recurrence. The patient denied any history of
tuberculosis or other malignancy. In June 2003, the patient
delivered a healthy female infant via spontaneous vaginal delivery.
The patient and family had not undergone any screening or
examination for Helicobacter pylori (H. pylori)
previously, and the family history was negative for genetic
disorders, cancers or infectious diseases.
On physical examination, the patient presented with
abdominal distension, dullness on percussion, an abdominal
circumference of 104 cm, an indistinct uterine fundus and a fetal
heart rate of 150 bpm. Laboratory investigations revealed a normal
hemoglobin level (136 g/l) (ref. 115–150 g/l) but notable ketosis
(+4) (ref. negative) on urinalysis. Biochemical tests showed an
albumin level of 35.5 g/l (ref. 35–50 g/l). Notably, tumor markers
were elevated, including α-fetoprotein at 36.74 ng/ml (ref. ≤10
ng/ml), cancer antigen 15–3 at 17.49 U/ml (ref. ≤30 U/ml), cancer
antigen 72–4 at 14.64 U/ml (ref. ≤6.9 U/ml), cancer antigen 19–9 at
>1,000 U/ml (ref. ≤37 U/ml), cancer antigen 125 at 1,040 U/ml
(ref. ≤35 U/ml), carcinoembryonic antigen at 180.30 ng/ml (ref. ≤5
ng/ml) and neuron-specific enolase at 11.51 ng/ml (ref. ≤16.3
ng/ml). Tests for tuberculosis were negative. Obstetric ultrasound
confirmed an intrauterine pregnancy with a viable fetus and showed
a large amount of free fluid in the pelvic and abdominal cavities,
~90 mm in depth and with no abnormalities in the bilateral adnexa.
Abdominal ultrasound further showed thickened greater omentum,
notable ascites with a maximum depth of ~93 mm (Fig. 1C and D).
After abdominal paracentesis and tube placement, the
patient experienced an improvement in symptoms, and the aspirated
ascitic fluid was sent for further analysis. Supportive therapies,
including intravenous nutrition, fluid replenishment and ketone
reduction, were also initiated. During ultrasound-guided
paracentesis, a gastric mass was noted, raising suspicion for
gastric cancer. Yellow ascitic fluid was drained. Subsequent
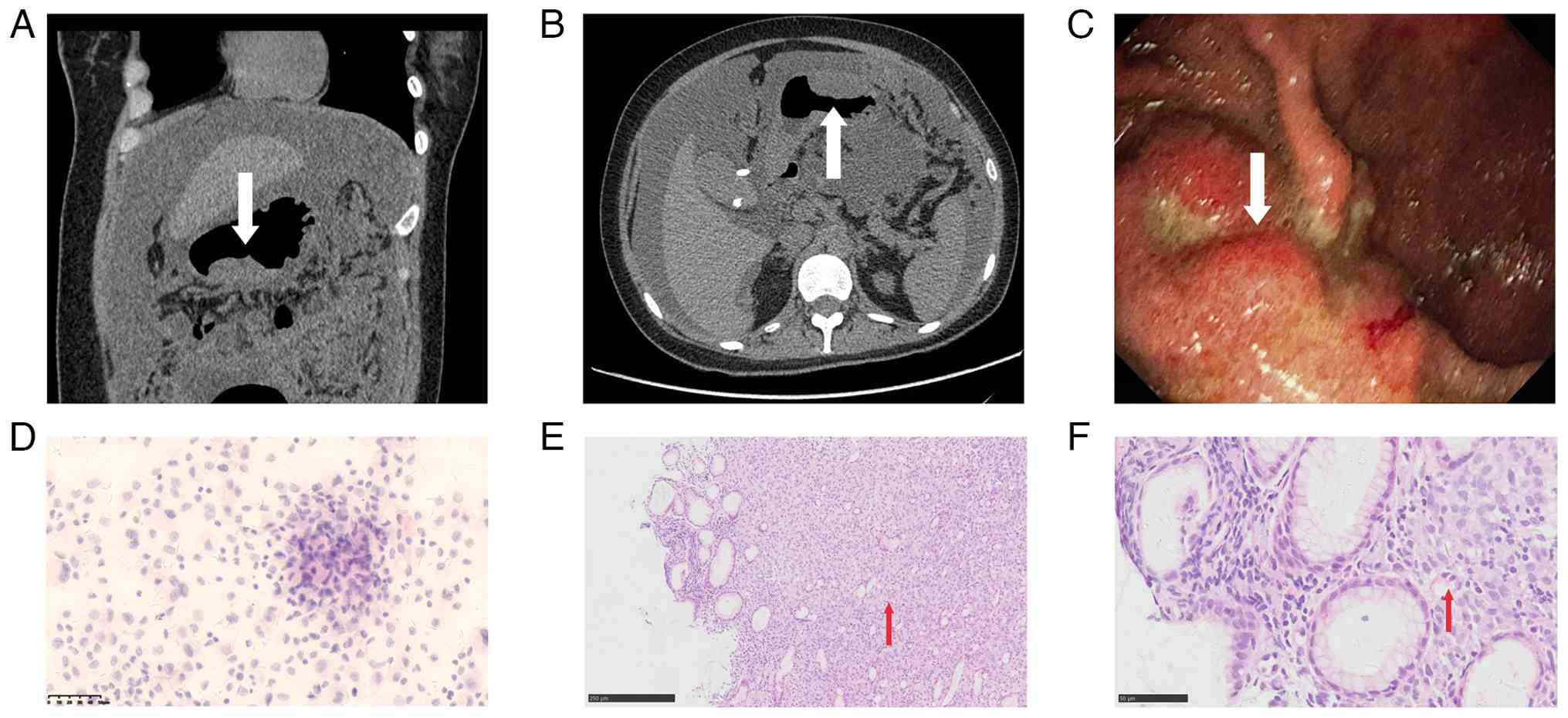
abdominal CT with contrast enhancement revealed a mass at the
greater curvature of the gastro-pyloric junction, highly suspicious
for malignancy, with alterations in the peritoneum and omentum,
suggesting metastatic seeding (Fig. 2A
and B).
The patient was informed of the findings, and an
esophagogastroduodenoscopy was performed. The examination revealed
a 5×4 cm raised mass near the greater curvature of the lower
gastric body, adjacent to the gastro-pyloric junction, with two
ulcers in the center, surrounded by irregular, nodular mucosa
(Fig. 2C). Biopsy samples were
taken from the ulcer edges for pathological examination, with
gastric cancer highly suspected. Biopsy and ascitic fluid samples
were fixed in 10% neutral-buffered formalin at room temperature for
12–24 h. Paraffin-embedded sections were cut at 4 µm. Routine
H&E staining was performed at RT (hematoxylin 5 min, eosin 2
min). Cytospin smears of ascitic fluid were prepared by
centrifugation at 1,500 × g for 10 min at room temperature, then
fixed in 95% ethanol for 10 min and stained with H&E under the
same as aforementioned. All slides were examined with an Olympus
BX43 light microscope.
Pathological results from the gastroscopic biopsy
indicated poorly differentiated gastric carcinoma, partially
composed of signet ring cell carcinoma, classified as Lauren's
mixed type. Concurrently, ascitic fluid cytology revealed
proliferated mesothelial cells, small lymphocytes, macrophages and
scattered atypical cells with irregular nuclei and mitotic figures,
suggesting a possible tumor origin (Fig. 2D-F).
Given the confirmed diagnosis of gastric cancer, the
patient and family were fully informed of the condition.
Termination of pregnancy and specialized treatment were
recommended; however, after discussion, the family of the patient
opted for discharge and palliative care at a local hospital. The
patient passed away 3 days after discharge as confirmed by
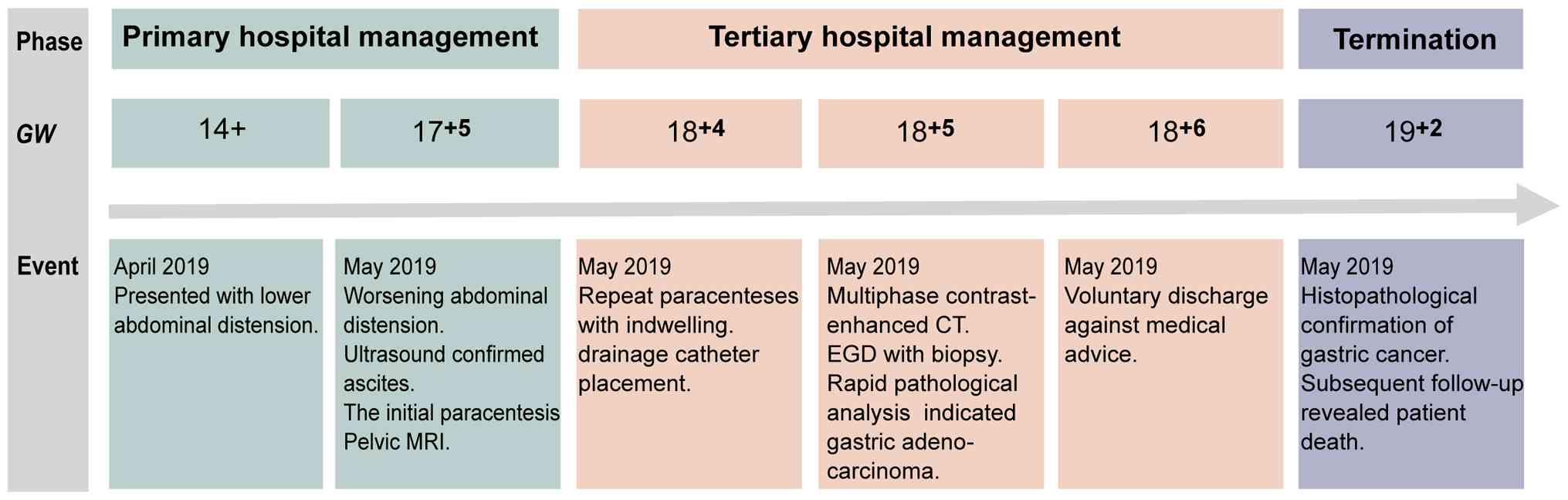
telephone follow-up with the family. Fig. 3 presents a timeline of the medical
journey of the patient.
Discussion
The incidence of gastric cancer during pregnancy is
low (0.026–0.100% of all pregnancies); however, the maternal
prognosis is poor, with reported 1- and 2-year survival rates of
18.3 and 15.1%, respectively (3).
Due to the non-specificity of symptoms and the limitations of
diagnostic methods during pregnancy, most patients are diagnosed at
an advanced stage. However, early detection is crucial for
prognosis, as poor outcomes are common when diagnosis is delayed.
Therefore, prompt attention to maternal symptoms and targeted
examinations are crucial for early diagnosis (7).
The symptoms of gastric cancer during pregnancy are
atypical, which markedly interferes with clinical diagnosis and
treatment. Symptoms such as nausea, vomiting, anorexia and weight
loss in early pregnancy are often attributed to pregnancy itself
and treated symptomatically. However, nausea and vomiting occurring
after 9 weeks of gestation warrant exclusion of other causes, such
as common hepatitis, pancreatitis or acute abdominal conditions.
Most of these can be diagnosed through routine laboratory tests and
ultrasonography (8). As early
diagnosis is beneficial for improving the prognosis of the patient,
attention should be paid to the onset and duration of
gastrointestinal symptoms (7).
Typically, pregnancy-related nausea and vomiting markedly improve
after 20 weeks of gestation (9,10).
Therefore, women with persistent gastrointestinal symptoms should
undergo active examination to rule out gastrointestinal diseases.
Ultimately, nausea and vomiting during pregnancy are diagnoses of
exclusion.
Imaging for malignant tumors in the digestive system
primarily relies on gastroscopy and CT (1); however, due to safety considerations
during pregnancy and the extremely low incidence of malignant
tumors in the digestive system, these two methods are rarely
employed, often leading to delays in diagnosis and treatment and
missed opportunities for optimal therapy (11). Currently, gastric window
ultrasonography and gastroscopy are the primary diagnostic methods
available for pregnant women. Compared with gastroscopy,
ultrasonography offers advantages such as being non-invasive,
cost-effective, highly repeatable and capable of clearly displaying
intra-gastric cavities, gastric walls and perigastric lesions with
the aid of gastrointestinal contrast agents. It can also observe
the depth of gastric cancer infiltration, making it more acceptable
to patients (12). Nevertheless,
its diagnostic significance for lesions at the gastric fundus,
micro-gastric cancers and small gastric cancers warrants further
investigation (13).
Concerns about the safety of gastroscopy, including
the risks of hypoxemia or hypotension during the procedure that may
lead to fetal death, miscarriage or preterm birth, as well as the
potential impact of medications on the fetus, contribute to the low
utilization rate of gastroscopy in pregnant women. Nevertheless,
gastroscopy holds unique significance in the diagnosis of gastric
cancer (12). A Swedish nationwide
cohort study reported that women undergoing any form of endoscopy
during pregnancy faced an increased risk of preterm birth and
delivery of small for gestational age infants (14). However, when focusing on women
without inflammatory bowel disease, celiac disease or liver
disease, this association disappeared, indicating that under
certain conditions, endoscopy may be safe and feasible during
pregnancy (14). In 2012, the
American Society for Gastrointestinal Endoscopy released guidelines
for the use of gastrointestinal endoscopy in pregnant and lactating
women, providing guidance for the safe use of gastrointestinal
endoscopy in pregnant women. Strict adherence to endoscopy
indications during pregnancy includes: Obvious or persistent
gastrointestinal bleeding; severe or refractory nausea, vomiting or
abdominal pain; dysphagia or odynophagia; strong suspicion of
colonic masses; severe diarrhea with negative test results;
gallstone pancreatitis, gallstone cholangiopathy or cholangitis;
and bile duct or pancreatic duct injury. General principles
include: Strictly adhering to indications; scheduling endoscopy
during the second trimester if not an emergency; using the lowest
dose of sedative drugs; preferring Class A or B drugs; minimizing
procedure duration; lying on the left side to avoid supine
hypotensive syndrome; obstetric consultation for fetal heart rate
monitoring before and after anesthesia; and avoiding use in certain
obstetric complications such as placental abruption, rapid labor,
rupture of membranes or eclampsia (12). CT scans are not absolutely
contraindicated during pregnancy. According to the Diagnostic
Imaging Guidelines in Pregnancy and Lactation (no. 723) issued by
the American College of Obstetricians and Gynecologists (15), the radiation dose for pelvic CT is
50 mGy; however, with the help of low-exposure techniques, the dose
can be reduced to 2.5 mGy to achieve diagnostic efficacy, which is
far below the threshold for teratogenic radiation dose before 25
weeks of gestation.
The large-scale case series reviews on gastric
cancer during pregnancy include a study of 137 cases of
pregnancy-associated gastric cancer in Japan published in 2009
(16) and a study involving 65
Chinese patients published in 2015 (17). These studies reported that
approximately one-third of the cases were detected postnatally, and
>90% of the cases were advanced stage gastric cancer. The 1- and
2-year survival rates for the tumors were both <20%. Moreover,
after adjusting for age, sex and cancer stage, a retrospective
study in Korea reported that the median survival time during
pregnancy was ~7 months, which was lower than the 15-month median
survival time observed in non-pregnant women; however, this
difference was not statistically significant. The study also
reported that the time to diagnosis during pregnancy was markedly
longer than that in non-pregnant individuals (18). Therefore, persistent
gastrointestinal symptoms in pregnant women require thorough
evaluation to facilitate early diagnosis.
Pregnancy-associated gastric cancer presents two
conflicting issues: On the one hand, it is ideal to initiate
surgical intervention as early as possible to increase the chances
of maternal recovery from gastric cancer; but on the other hand,
there is a viewpoint that the pregnancy should be continued as long
as safely possible to ensure fetal safety. Treatment plans depend
on the cancer stage and fetal development at diagnosis. In line
with current practices in China, it is generally agreed that for
pregnancies at ≥28 weeks, surgical intervention should follow
cesarean or vaginal delivery (16).
For pregnancies at <28 weeks, decisions regarding whether to
continue the pregnancy and proceed with surgery should be made
after considering the cancer stage, specialized principles for
managing gastrointestinal tumors and thorough communication with
the patient (16).
The most common metastatic sites for gastric
carcinoma include peritoneal surfaces, the liver, spleen, ovaries,
lung and brain. Gastric cancer infrequently metastasizes to the
bone (1). A literature search of
English publications over the past 5 years identified the
metastatic sites of gastric cancer during pregnancy as the placenta
(19–21), ovaries (22–25),
lungs (26), bones (27,28)
and breasts (29). Moreover, a
notable clinical concern associated with placental metastasis is
fetal metastasis (20), which is
not always obvious at birth; however, to date, no case reports of
fetal metastasis have been documented, to the best of our
knowledge. A total of one case of uterine metastasis during
pregnancy has been reported (30).
The present case has certain limitations that
require acknowledgment. Specifically, H. pylori status
remained undetermined due to the discharge of the patient before
diagnostic confirmation, precluding timely microbial evaluation.
This gap is clinically significant given the well-documented role
of H. pylori in gastric carcinogenesis. The World Health
Organization International Agency for Research on Cancer classifies
H. pylori as a Group I carcinogen, with cohort studies
demonstrating its association with ~89% of non-cardia gastric
carcinomas (31). Whilst pregnancy
necessitates careful diagnostic modality selection, with
¹3C-urea breath tests and serological assays as
preferred safety considerations, the inability to ascertain
infection status in the present case limits etiological
interpretation. This underscores the need for protocols ensuring
pre-discharge microbial testing in similar cases. Furthermore, the
association between gestational gastric cancer and H. pylori
infection remains poorly characterized, highlighting a critical
research gap.
In the present case, ascitic cytology slides were
prepared at an outside institution and could not be retrieved for
in-house review. As a result, representative images of ascitic
cytology and the corresponding ultrasound were unavailable, which
may limit the completeness of our imaging and cytological
documentation.
In conclusion, the present case report highlights
the rarity and notable maternal mortality rate associated with
gastric cancer diagnosed during pregnancy. The presented case
exemplifies the difficulties in early detection due to non-specific
symptoms and diagnostic constraints during pregnancy. Despite an
extensive diagnostic workup, the condition of the patient
deteriorated swiftly, underscoring the necessity for prompt
assessment of maternal symptoms and focused diagnostic procedures.
The present paper delineates the poor prognosis associated with
pregnancy-associated gastric cancer, with most cases identified at
an advanced stage. Additionally, the report emphasizes the critical
importance of addressing the unique challenges presented by the
coexistence of cancer and pregnancy when strategizing treatment
plans. The metastatic capacity of gastric cancer during pregnancy
complicates clinical management further. Collectively, the present
case underscores the imperative for increased awareness, timely
diagnosis and personalized treatment approaches to enhance outcomes
for both the mother and fetus in instances of pregnancy-associated
gastric cancer.
Acknowledgements
Not applicable.
Funding
Funding: No funding was received.
Availability of data and materials
The data generated in the present study may be
requested from the corresponding author.
Authors' contributions
JN and HY designed the study and analyzed patient
data. YS and YG advised on patient treatment. JN, YG, YS and HY
confirm the authenticity of all the raw data. All authors agree to
be accountable for all aspects of the work. All authors read and
approved the final manuscript.
Ethics approval and consent to
participate
The present study was approved by the Peking
University First Hospital Medical Science Research Ethics Committee
(approval no. 164-002) and performed according to the 1964 Helsinki
Declaration and its later amendments or comparable ethical
standards.
Patient consent for publication
Written informed consent was obtained from the
family of the patient for publication of the present case
report.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Smyth EC, Nilsson M, Grabsch HI, van
Grieken NC and Lordick F: Gastric cancer. Lancet. 396:635–648.
2020. View Article : Google Scholar
|
|
2
|
Bray F, Laversanne M, Sung H, Ferlay J,
Siegel RL, Soerjomataram I and Jemal A: Global cancer statistics
2022: GLOBOCAN estimates of incidence and mortality worldwide for
36 cancers in 185 countries. CA Cancer J Clin. 74:229–263.
2024.PubMed/NCBI
|
|
3
|
Maggen C, Lok CA, Cardonick E, van Gerwen
M, Ottevanger PB, Boere IA, Koskas M, Halaska MJ, Fruscio R, Gziri
MM, et al: Gastric cancer during pregnancy: A report on 13 cases
and review of the literature with focus on chemotherapy during
pregnancy. Acta Obstet Gynecol Scand. 99:79–88. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Liu H, Xie W and Gong W: Gastric cancer in
pregnancy: A review. Future Oncol. 20:1851–1860. 2024. View Article : Google Scholar
|
|
5
|
Kodama M, Moeini A, Machida H, Blake EA,
Grubbs BH and Matsuo K: Feto-maternal outcomes of pregnancy
complicated by Krukenberg tumor: A systematic review of literature.
Arch Gynecol Obstet. 294:589–598. 2016. View Article : Google Scholar
|
|
6
|
Constantin A, Constantin R, Achim F, Socea
B and Predescu D: Pregnancy and gastric cancer: A narrative review.
Diagnostics (Basel). 13:19092023. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Bozkurt M, Antonoff M, Jaramillo S,
Sagebiel T and Murphy MB: Gastroesophageal cancer during pregnancy:
A case report and review of the literature. J Gastrointest Cancer.
50:634–640. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Erick M, Cox JT and Mogensen KM: ACOG
practice bulletin 189: Nausea and vomiting of pregnancy. Obstet
Gynecol. 131:9352018. View Article : Google Scholar
|
|
9
|
Eliakim R, Abulafia O and Sherer DM:
Hyperemesis gravidarum: A current review. Am J Perinatol.
17:207–218. 2000. View Article : Google Scholar
|
|
10
|
Dean CR, Shemar M, Ostrowski GAU and
Painter RC: Management of severe pregnancy sickness and hyperemesis
gravidarum. BMJ. 363:k50002018. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Marbun VMG and Putranto AS: Diagnosis and
management of gastric cancer in pregnancy-An evidence-based case
report. Int J Surg Case Rep. 75:338–344. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
ASGE Standard of Practice Committee, .
Shergill AK, Ben-Menachem T, Chandrasekhara V, Chathadi K, Decker
GA, Evans JA, Early DS, Fanelli RD, Fisher DA, et al: Guidelines
for endoscopy in pregnant and lactating women. Gastrointest Endosc.
76:18–24. 2012. View Article : Google Scholar
|
|
13
|
Pectasides M, Sekhar A, Dighe MK, Schwartz
G, Shah SN, Mulcahy MF and Horowitz JM: Gastrointestinal
malignancies in pregnancy. Abdom Radiol (NY). 48:1709–1723. 2023.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Ludvigsson JF, Lebwohl B, Ekbom A, Kiran
RP, Green PH, Höijer J and Stephansson O: Outcomes of pregnancies
for women undergoing endoscopy while they were pregnant: A
nationwide cohort study. Gastroenterology. 152:554–563.e9. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Jain C: ACOG committee opinion No. 723:
Guidelines for diagnostic imaging during pregnancy and lactation.
Obstet Gynecol. 133:1862019. View Article : Google Scholar
|
|
16
|
Sakamoto K, Kanda T, Ohashi M, Kurabayashi
T, Serikawa T, Matsunaga M and Hatakeyama K: Management of patients
with pregnancy-associated gastric cancer in Japan: A mini-review.
Int J Clin Oncol. 14:392–396. 2009. View Article : Google Scholar
|
|
17
|
Zeng H, Zhou X, Xie H, Zhao Y and Fu W:
Gastric cancer in pregnancy in China: Case reports and a
mini-review. J Sung. 11:165–168. 2015.
|
|
18
|
Song MJ, Park YS, Song HJ, Park SJ, Ahn
JY, Choi KD, Lee GH, Jung HY, Yook JH and Kim BS: Prognosis of
pregnancy-associated gastric cancer: An age-, sex-, and
stage-matched case-control study. Gut Liver. 10:731–738. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Kleijn TG, Scholten I, Knol HM, Zwart JJ
and Hoogland AM: A pregnant woman with gastric cancer and placental
involvement. Ned Tijdschr Geneeskd. 166:D66812022.(In Dutch).
PubMed/NCBI
|
|
20
|
Oga S, Hachisuga M, Hidaka N, Fujita Y,
Tomonobe H, Yamamoto H and Kato K: Gastric cancer during pregnancy
with placental involvement: Case report and review of published
works. Obstet Gynecol Sci. 62:357–361. 2019. View Article : Google Scholar
|
|
21
|
Patan S, Benzar T, Sanford G and Kavanaugh
M: Advanced metastatic gastric adenocarcinoma identified within the
placenta: A case report with literature review. J Gastrointest
Oncol. 11:127–132. 2020. View Article : Google Scholar
|
|
22
|
Zhang Y, Du H, Li T, Li H, Deng Y and Wu
R: Krukenberg tumor of gastric origin in pregnant women with
preeclampsia. Case Rep Oncol. 16:718–727. 2023. View Article : Google Scholar
|
|
23
|
Chen HY, Lee CN and Lin SY: The
challenging presentation of gastric cancer during pregnancy with
Krukenberg tumor: A case report. Ann Med Surg (Lond). 85:2056–2058.
2023. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Mendoza-Rosado F, Nunez-Isaac O,
Espinosa-Marrón A, Lopez-Arjona K and Davila-Martinez F: Krukenberg
tumor as an incidental finding in a full-term pregnancy: A case
report. J Med Case Rep. 15:3042021. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Eckel F, Carlin G, Mayer S, Polterauer S
and Chalubinski K: Krukenberg progression of gastric carcinoma in
pregnancy: Is early diagnosis possible? Case report and review of
the literature. J Clin Med. 12:53972023. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Pacheco S, Norero E, Canales C, Martínez
JM, Herrera ME, Muñoz C and Jarufe N: The rare and challenging
presentation of gastric cancer during pregnancy: A report of three
cases. J Gastric Cancer. 16:271–276. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Kinoshita S, Yamashita K, Iwatsuki M, Sato
H, Matsumoto C, Matsumoto T, Shiraishi Y, Yoshida N and Baba H:
Disseminated carcinomatosis of the bone marrow from gastric cancer
during pregnancy. Clin J Gastroenterol. 12:447–452. 2019.
View Article : Google Scholar
|
|
28
|
Chellan D, Senthamizhselvan K, Nair A,
Mohan P, Ramkumar G and Badhe B: Skull base metastasis and
Krukenberg tumor in a pregnant woman: An unusual presentation of
metastatic gastric cancer. ACG Case Rep J. 11:e012832024.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Basoglu T, Telli TA, Demircan NC, Arikan
R, Ercelep O, Ozguven S, Soysal S, Memisoglu A, Dane F and Yumuk
PF: A rare case of gastric cancer with bilateral breast metastasis
during pregnancy. J Oncol Pharm Pract. 27:220–226. 2021. View Article : Google Scholar
|
|
30
|
Jeong B, Shim JY, Kim CJ, Won HS, Lee PR
and Kim A: Massive perivillous fibrin deposition in the placenta
and uterine metastasis of gastric adenocarcinoma during pregnancy.
J Obstet Gynaecol Res. 40:1150–1153. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Liou JM, Malfertheiner P, Lee YC, Sheu BS,
Sugano K, Cheng HC, Yeoh KG, Hsu PI, Goh KL, Mahachai V, et al:
Screening and eradication of Helicobacter pylori for gastric
cancer prevention: the Taipei global consensus. Gut. 69:2093–2112.
2020. View Article : Google Scholar
|