|
1
|
Liu YL, Yang PM, Shun CT, Wu MS, Weng JR
and Chen CC: Autophagy potentiates the anti-cancer effects of the
histone deacetylase inhibitors in hepatocellular carcinoma.
Autophagy. 6:1057–1065. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Maciejewski K, Giers M, Oleksiewicz U and
Czerwinska P: The epigenetic modifiers HDAC2 and HDAC7 inversely
associate with cancer stemness and immunity in solid tumors. Int J
Mol Sci. 25:78412024. View Article : Google Scholar
|
|
3
|
Bao Q, Li Y, Chen Y, Zheng J, Zhao J and
Hu T: Transcriptome-based network analysis related to histone
deacetylase genes and identified EMP1 as a potential biomarker for
prognosis in bladder cancer. Clin Genitourin Cancer. 23:1022622025.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Liang T, Wang F, Elhassan RM, Cheng Y,
Tang X, Chen W, Fang H and Hou X: Targeting histone deacetylases
for cancer therapy: Trends and challenges. Acta Pharm Sin B.
13:2425–2463. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Klieser E, Neumayer B, Di Fazio P, Mayr C,
Neureiter D and Kiesslich T: HDACs as an emerging target in
endocrine tumors: A comprehensive review. Expert Rev Endocrinol
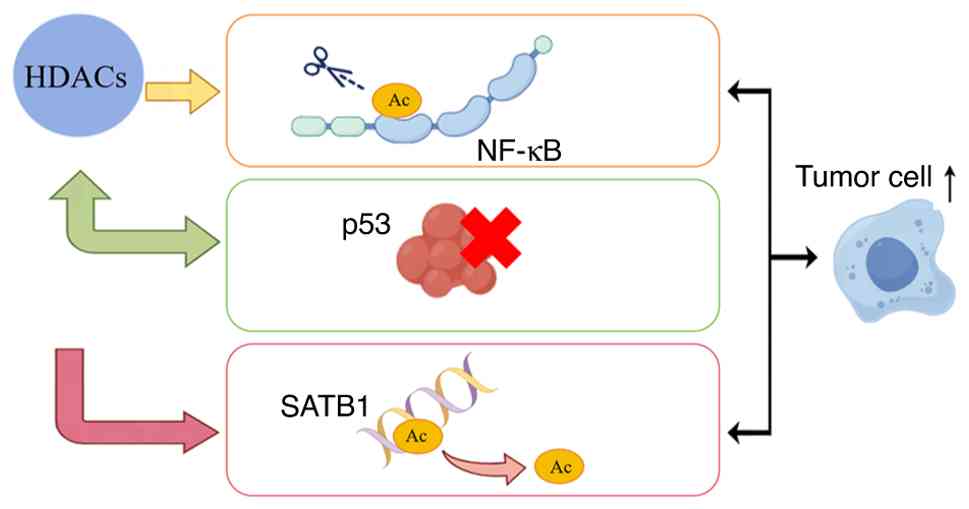
Metab. 18:143–154. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
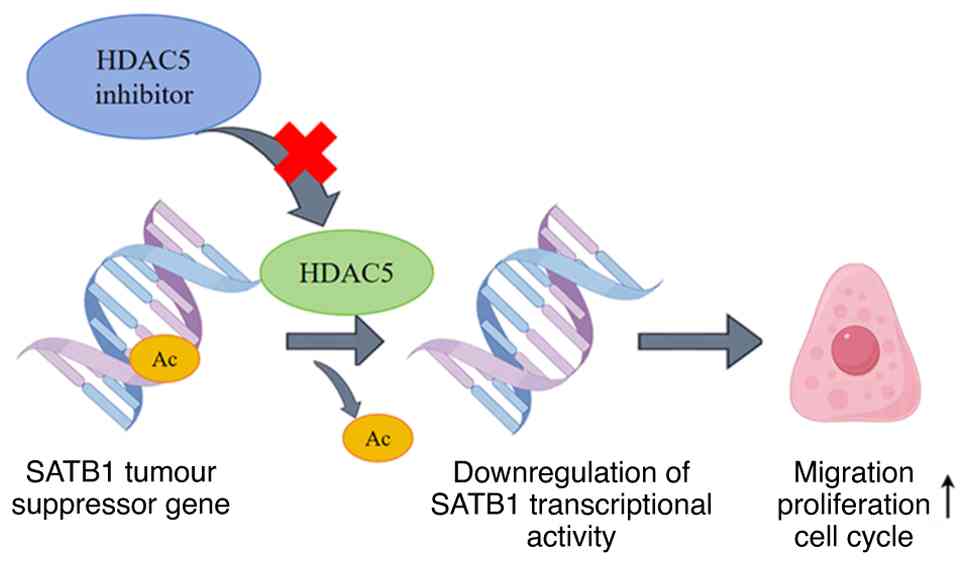
6
|
Contreras-Sanzón E, Prado-Garcia H,
Romero-Garcia S, Nuñez-Corona D, Ortiz-Quintero B, Luna-Rivero C,
Martínez-Cruz V and Carlos-Reyes Á: Histone deacetylases modulate
resistance to the therapy in lung cancer. Front Genet.
13:9602632022. View Article : Google Scholar
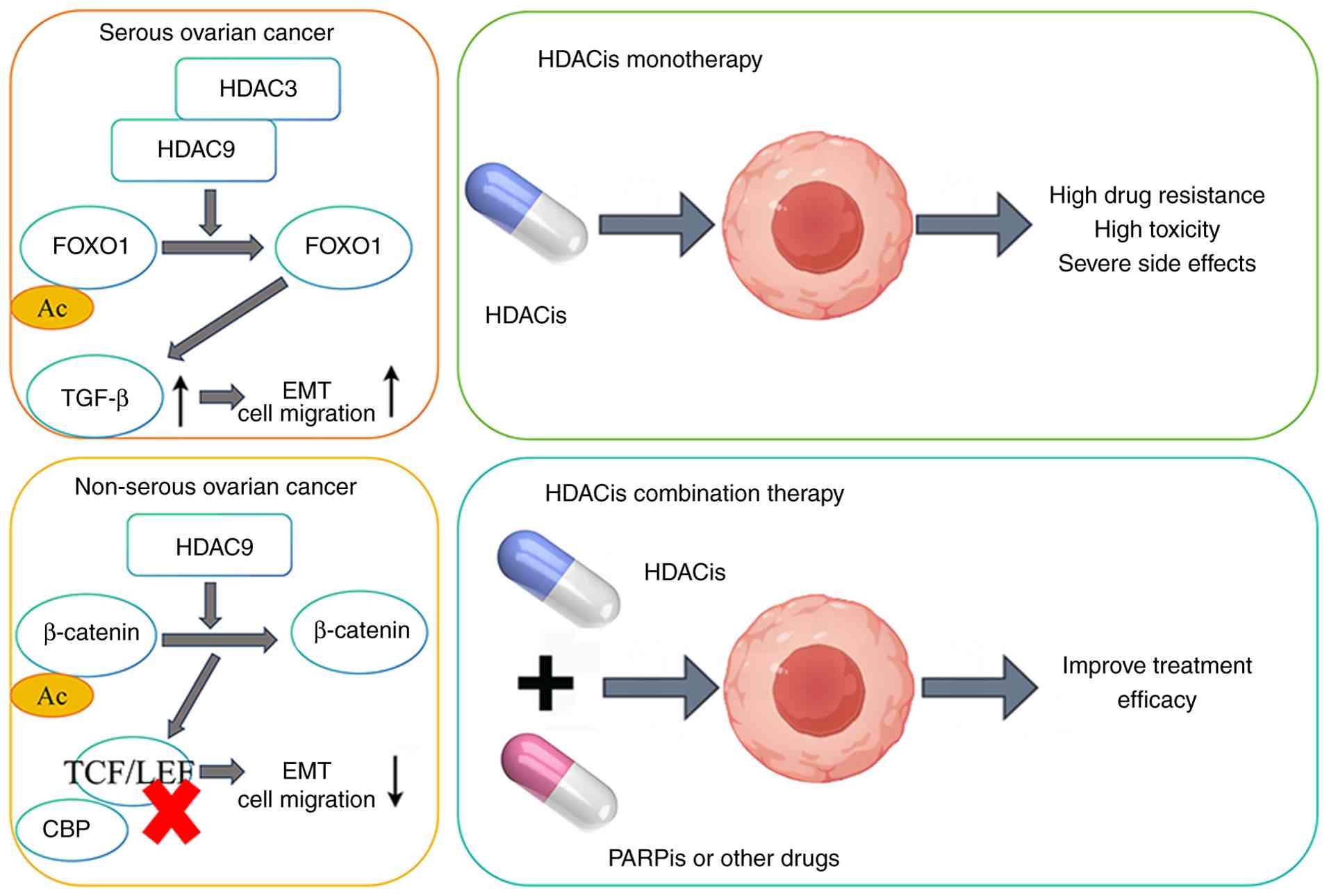
|
|
7
|
Liang R, Zhang J, Liu Z, Liu Z, Li Q, Luo
X, Li Y, Ye J and Lin Y: Mechanism and molecular network of
RBM8A-mediated regulation of oxaliplatin resistance in
hepatocellular carcinoma. Front Oncol. 10:5854522021. View Article : Google Scholar
|
|
8
|
Giordano F, Forestiero M, Leonetti AE,
Naimo GD, Marrone A, De Amicis F, Marsico S, Mauro L and Panno ML:
Valproic acid reduces invasiveness and cellular growth in 2D and 3D
glioblastoma cell lines. Int J Mol Sci. 26:66002025. View Article : Google Scholar
|
|
9
|
Zheng B, Jiang X, Liu Y, Cheng F, Zhang Y,
Niu C, Cong Z, Niu Z and He W: Elevated histone deacetylase 10
expression promotes the progression of clear cell renal cell
carcinoma by notch-1-PTEN signaling axis. Discov Oncol. 15:1562024.
View Article : Google Scholar
|
|
10
|
Jungwirth G, Yu T, Liu F, Cao J, Alaa
Eddine M, Moustafa M, Abdollahi A, Warta R, Unterberg A and
Herold-Mende C: Pharmacological landscape of FDA-approved
anticancer drugs reveals sensitivities to ixabepilone, romidepsin,
omacetaxine, and carfilzomib in aggressive meningiomas. Clin Cancer
Res. 29:233–243. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Lee H, Jung TY, Lim SH, Choi EJ, Lee J and
Min DS: Phospholipase D2 is a positive regulator of sirtuin 1 and
modulates p53-mediated apoptosis via sirtuin 1. Exp Mol Med.
53:1287–1297. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Lin Y, Jing X, Chen Z, Pan X, Xu D, Yu X,
Zhong F, Zhao L, Yang C, Wang B, et al: Histone
deacetylase-mediated tumor microenvironment characteristics and
synergistic immunotherapy in gastric cancer. Theranostics.
13:4574–4600. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Haberland M, Montgomery RL and Olson EN:
The many roles of histone deacetylases in development and
physiology: Implications for disease and therapy. Nat Rev Genet.
10:32–42. 2009. View Article : Google Scholar
|
|
14
|
Kundu R, Banerjee S, Baidya SK, Adhikari N
and Jha T: A quantitative structural analysis of AR-42 derivatives
as HDAC1 inhibitors for the identification of promising structural
contributors. SAR QSAR Environ Res. 33:861–883. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Guo K, Ma Z, Zhang Y, Han L, Shao C, Feng
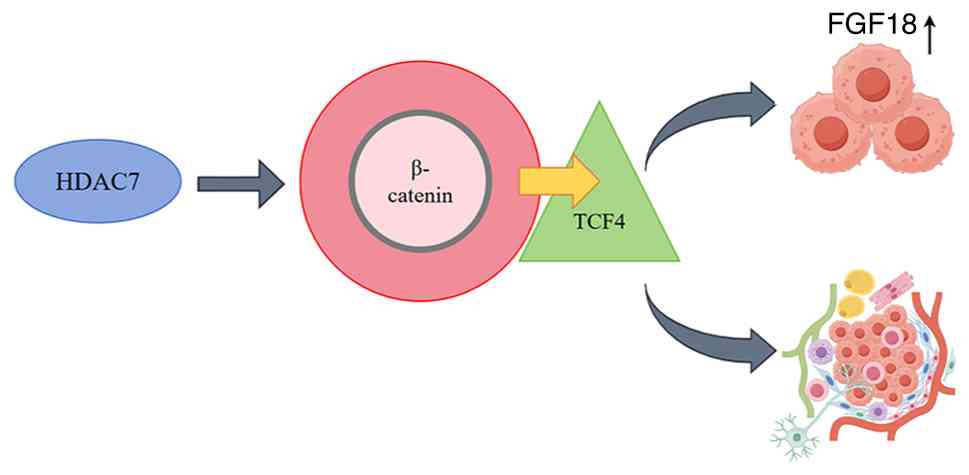
Y, Gao F, Di S, Zhang Z, Zhang J, et al: HDAC7 promotes NSCLC
proliferation and metastasis via stabilization by deubiquitinase
USP10 and activation of β-catenin-FGF18 pathway. J Exp Clin Cancer
Res. 41:912022. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Yu L, Cao H, Yang JW, Meng WX, Yang C,
Wang JT, Yu MM and Wang BS: HDAC5-mediated PRAME regulates the
proliferation, migration, invasion, and EMT of laryngeal squamous
cell carcinoma via the PI3K/AKT/mTOR signaling pathway. Open Med
(Wars). 18:202306652023. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Zhang Y, Ding P, Wang Y, Shao C, Guo K,
Yang H, Feng Y, Ning J, Pan M, Wang P, et al: HDAC7/c-Myc signaling
pathway promotes the proliferation and metastasis of choroidal
melanoma cells. Cell Death Dis. 14:382023. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Lu B, Qiu R, Wei J, Wang L, Zhang Q, Li M,
Zhan X, Chen J, Hsieh IY, Yang C, et al: Phase separation of
phospho-HDAC6 drives aberrant chromatin architecture in
triple-negative breast cancer. Nat Cancer. 5:1622–1640. 2024.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Jin J, Meng T, Yu Y, Wu S, Jiao CC, Song
S, Li YX, Zhang Y, Zhao YY, Li X, et al: Human HDAC6 senses valine
abundancy to regulate DNA damage. Nature. 637:215–223. 2025.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Pandey UB, Batlevi Y, Baehrecke EH and
Taylor JP: HDAC6 at the intersection of autophagy, the
ubiquitin-proteasome system and neurodegeneration. Autophagy.
3:643–645. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Bhagat RP, Jyotisha Dasgupta I, Amin SA,
Jakkula P, Bhattacharya A, Qureshi IA and Gayen S: First report on
analysis of chemical space, scaffold diversity, critical structural
features of HDAC11 inhibitors. Mol Divers. 29:3679–3702. 2025.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Sriramareddy SN, Faião-Flores F, Emmons
MF, Saha B, Chellappan S, Wyatt C, Smalley I, Licht JD, Durante MA,
Harbour JW and Smalley KSM: HDAC11 activity contributes to MEK
inhibitor escape in uveal melanoma. Cancer Gene Ther. 29:1840–1846.
2022. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Buglio D, Khaskhely NM, Voo KS,
Martinez-Valdez H, Liu YJ and Younes A: HDAC11 plays an essential
role in regulating OX40 ligand expression in Hodgkin lymphoma.
Blood. 117:2910–2917. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Das T, Bhattacharya A, Jha T and Gayen S:
Exploration of fingerprints and data mining-based prediction of
some bioactive compounds from Allium sativum as histone deacetylase
9 (HDAC9) inhibitors. Curr Comput Aided Drug Des. 21:270–284. 2025.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
West AC and Johnstone RW: New and emerging
HDAC inhibitors for cancer treatment. J Clin Invest. 124:30–39.
2014. View Article : Google Scholar
|
|
26
|
Debbarma M, Sarkar K and Sil SK:
Dissecting the epigenetic orchestra of HDAC isoforms in breast
cancer development: A review. Med Oncol. 42:12024. View Article : Google Scholar
|
|
27
|
Sanaei M and Kavoosi F: Histone
deacetylase inhibitors, intrinsic and extrinsic apoptotic pathways,
and epigenetic alterations of histone deacetylases (HDACs) in
hepatocellular carcinoma. Iran J Pharm Res. 20:324–336.
2021.PubMed/NCBI
|
|
28
|
Han S, Fan H, Zhong G, Ni L, Shi W, Fang
Y, Wang C, Wang L, Song L, Zhao J, et al: Nuclear KRT19 is a
transcriptional corepressor promoting histone deacetylation and
liver tumorigenesis. Hepatology. 81:808–822. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Jasim SA, Altalbawy FMA, Abohassan M,
Oghenemaro EF, Bishoyi AK, Singh RP, Kaur P, Sivaprasad GV,
Mohammed JS and Hulail HM: Histone deacetylases (HDACs) roles in
inflammation-mediated diseases; current knowledge. Cell Biochem
Biophys. 83:1375–1386. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Zhang K, Peng X, Zhou P, Li X, Shen L,
Yang L and Zhou Q: HDAC1/2-mediated deacetylation of KLF9 promotes
the malignant progression of nasopharyngeal carcinoma via CDH17.
Oncogene. 44:3183–3198. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Sahu RK, Dhakshnamoorthy J, Jain S, Folco
HD, Wheeler D and Grewal SIS: Nucleosome remodeler exclusion by
histone deacetylation enforces heterochromatic silencing and
epigenetic inheritance. Mol Cell. 84:3175–3191.e8. 2024. View Article : Google Scholar
|
|
32
|
Talom A, Barhoi A, Jirpu T, Dawn B and
Ghosh A: Clinical progress and functional modalities of HDAC
inhibitor-based combination therapies in cancer treatment. Clin
Transl Oncol. 28:71–85. 2026. View Article : Google Scholar
|
|
33
|
Sandonà M, Cavioli G, Renzini A, Cedola A,
Gigli G, Coletti D, McKinsey TA, Moresi V and Saccone V: Histone
deacetylases: Molecular mechanisms and therapeutic implications for
muscular dystrophies. Int J Mol Sci. 24:43062023. View Article : Google Scholar
|
|
34
|
Lu-Culligan WJ, Connor LJ, Xie Y, Ekundayo
BE, Rose BT, Machyna M, Pintado-Urbanc AP, Zimmer JT, Vock IW,
Bhanu NV, et al: Acetyl-methyllysine marks chromatin at active
transcription start sites. Nature. 622:173–179. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Joseph R, Dasari SK, Umamaheswaran S,
Mangala LS, Bayraktar E, Rodriguez-Aguayo C, Wu Y, Nguyen N, Powell
RT, Sobieski M, et al: EphA2- and HDAC-targeted combination therapy
in endometrial cancer. Int J Mol Sci. 25:12782024. View Article : Google Scholar
|
|
36
|
Blagitko-Dorfs N, Schlosser P, Greve G,
Pfeifer D, Meier R, Baude A, Brocks D, Plass C and Lübbert M:
Combination treatment of acute myeloid leukemia cells with DNMT and
HDAC inhibitors: Predominant synergistic gene downregulation
associated with gene body demethylation. Leukemia. 33:945–956.
2019. View Article : Google Scholar
|
|
37
|
McGuire CK, Meehan AS, Couser E, Bull L,
Minor AC, Kuhlmann-Hogan A, Kaech SM, Shaw RJ and Eichner LJ:
Transcriptional repression by HDAC3 mediates T cell exclusion from
Kras mutant lung tumors. Proc Natl Acad Sci USA.
121:e23176941212024. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Yan M, Cao H, Tao K, Xiao B, Chu Y, Ma D,
Huang X, Han Y and Ji T: HDACs alters negatively to the tumor
immune microenvironment in gynecologic cancers. Gene.
885:1477042023. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Kim YK, Lee EK, Kang JK, Kim JA, You JS,
Park JH, Seo DW, Hwang JW, Kim SN, Lee HY, et al: Activation of
NF-kappaB by HDAC inhibitor apicidin through Sp1-dependent de novo
protein synthesis: Its implication for resistance to apoptosis.
Cell Death Differ. 13:2033–2041. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Luo J, Su F, Chen D, Shiloh A and Gu W:
Deacetylation of p53 modulates its effect on cell growth and
apoptosis. Nature. 408:377–381. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Singh T, Kaur P, Singh P, Singh S and
Munshi A: Differential molecular mechanistic behavior of HDACs in
cancer progression. Med Oncol. 39:1712022. View Article : Google Scholar
|
|
42
|
Sharma S, Tyagi W, Tamang R and Das S:
HDAC5 modulates SATB1 transcriptional activity to promote lung
adenocarcinoma. Br J Cancer. 129:586–600. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Ling R, Wang J, Fang Y, Yu Y, Su Y, Sun W,
Li X and Tang X: HDAC-an important target for improving tumor
radiotherapy resistance. Front Oncol. 13:11936372023. View Article : Google Scholar
|
|
44
|
Mao C, Fan W, Liu J, Yang F, Li W, Li L,
Shi Z, Li Q, Yuan Z, Jiang Y and Chu B: Targeting HDAC and PARP
enhances STING-dependent antitumor immunity in STING-deficient
tumor. Adv Sci (Weinh). 12:e079042025. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Rodrigues Moita AJ, Bandolik JJ, Hansen
FK, Kurz T, Hamacher A and Kassack MU: Priming with HDAC inhibitors
sensitizes ovarian cancer cells to treatment with cisplatin and
HSP90 inhibitors. Int J Mol Sci. 21:83002020. View Article : Google Scholar
|
|
46
|
Li Z, Wu YH, Guo YQ, Min XJ and Lin Y:
Tasquinimod promotes the sensitivity of ovarian cancer cells to
cisplatin by down-regulating the HDAC4/p21 pathway. Korean J
Physiol Pharmacol. 29:191–204. 2025. View Article : Google Scholar
|
|
47
|
Kim M, Lu F and Zhang Y: Loss of
HDAC-mediated repression and gain of NF-κB activation underlie
cytokine induction in ARID1A- and PIK3CA-mutation-driven ovarian
cancer. Cell Rep. 17:275–288. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Feng Q, Hao S, Liu X, Yan Z, Sheng K, Li
Y, Zhang P and Sheng X: HDAC7 promotes ovarian cancer malignancy
via AKT/mTOR signalling pathway. J Cell Mol Med. 28:e701202024.
View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Xu L, Wang J, Liu B, Fu J, Zhao Y, Yu S,
Shen L, Yan X and Su J: HDAC9 contributes to serous ovarian cancer
progression through regulating epithelial-mesenchymal transition.
Biomedicines. 10:3742022. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Xu L, Yan X, Wang J, Zhao Y, Liu Q, Fu J,
Shi X and Su J: The roles of histone deacetylases in the regulation
of ovarian cancer metastasis. Int J Mol Sci. 24:150662023.
View Article : Google Scholar
|
|
51
|
Chi AJ, Hsu JL, Xiao YX, Chern JW, Guh JH,
Yu CW and Hsu LC: A novel HDAC6 inhibitor enhances the efficacy of
paclitaxel against ovarian cancer cells. Molecules. 30:27932025.
View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Thomine C, Guillemot S, Weiswald LB,
Florent R, Abeilard E, Giffard F, Brotin E, Briand M, Dolivet E,
Poulain L and Villedieu M: The anticancer effect of the HDAC
inhibitor belinostat is enhanced by inhibitors of Bcl-xL
or Mcl-1 in ovarian cancer. Mol Oncol. 19:3325–3341. 2025.
View Article : Google Scholar
|
|
53
|
Begum S, Irvin SD, Cox CK, Huang Z, Wilson
JJ, Monroe JD and Gibert Y: Anti-ovarian cancer migration and
toxicity characteristics of a platinum(IV) pro-drug with axial HDAC
inhibitor ligands in zebrafish models. Invest New Drugs.
42:644–654. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Natarajan U, Venkatesan T and Rathinavelu
A: Effect of the HDAC inhibitor on histone acetylation and
methyltransferases in A2780 ovarian cancer cells. Medicina
(Kaunas). 57:4562021. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Si L, Lai T, Zhao J, Jin Y, Qi M, Li M, Fu
H, Shi X, Ma L and Guo R: Identification of a novel pyridine
derivative with inhibitory activity against ovarian cancer
progression in vivo and in vitro. Front Pharmacol. 13:10644852022.
View Article : Google Scholar
|
|
56
|
Valdez BC, Tsimberidou AM, Yuan B, Baysal
MA, Chakraborty A, Andersen CR and Andersson BS: Synergistic
cytotoxicity of histone deacetylase and poly-ADP ribose polymerase
inhibitors and decitabine in breast and ovarian cancer cells:
Implications for novel therapeutic combinations. Int J Mol Sci.
25:92412024. View Article : Google Scholar
|
|
57
|
Qiu J, Ren T, Liu Q, Jiang Q, Wu T, Cheng
LC, Yan W, Qu X, Han X and Hua K: Dissecting the distinct tumor
microenvironments of HRD and HRP ovarian cancer: Implications for
targeted therapies to overcome PARPi resistance in HRD tumors and
refractoriness in HRP tumors. Adv Sci (Weinh). 11:e23097552024.
View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Wang Y, Wen J, Sun X, Sun Y, Liu Y, Cheng
X, Wu W, Liu Q and Ren F: CUDC-907 exhibits potent antitumor
effects against ovarian cancer through multiple in vivo and in
vitro mechanisms. Cancer Chemother Pharmacol. 93:295–306. 2024.
View Article : Google Scholar
|
|
59
|
Zheng Y and Yang X, Wang C, Zhang S, Wang
Z, Li M, Wang Y, Wang X and Yang X: HDAC6, modulated by miR-206,
promotes endometrial cancer progression through the PTEN/AKT/mTOR
pathway. Sci Rep. 10:35762020. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Li Y, Zhou W, Li L, Li JW, Li T, Huang C,
Lazaro-Camp VJ, Kavlashvili T, Zhang Y, Reyes H, et al: Enhancing
progestin therapy via HDAC inhibitors in endometrial cancer. Am J
Cancer Res. 12:5029–5048. 2022.PubMed/NCBI
|
|
61
|
Nahar S, Yu J, Lee H, Tran DN, Li R, Kim
TH, Jung JS, Kim K, Yoo JY and Jeong JW: MIG-6 regulates
HDAC1-mediated angiogenesis and tumorigenesis in PTEN-deficient
endometrioid endometrial cancer. Mol Cancer Res. Jan 21–2026.(Epub
ahead of print). View Article : Google Scholar
|
|
62
|
Psilopatis I, Pergaris A, Giaginis C and
Theocharis S: Histone deacetylase inhibitors: A promising
therapeutic alternative for endometrial carcinoma. Dis Markers.
2021:78506882021. View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Edwards K, Yao S, Pisano S, Feltracco V,
Brusehafer K, Samanta S, Oommen OP, Gazze SA, Paravati R, Maddison
H, et al: Hyaluronic acid-functionalized nanomicelles enhance SAHA
efficacy in 3D endometrial cancer models. Cancers (Basel).
13:40322021. View Article : Google Scholar : PubMed/NCBI
|
|
64
|
Pothuri B, Sawaged Z, Karpel HC, Li X, Lee
J, Musa F, Lutz K, Reese E, Blank SV, Boyd LR, et al: A phase 2
feasibility study of nab-paclitaxel and carboplatin in epithelial
carcinoma of the uterus. Gynecol Oncol. 190:209–214. 2024.
View Article : Google Scholar
|
|
65
|
Lin CK, Liu ST, Wu ZS, Wang YC and Huang
SM: Mechanisms of cisplatin in combination with repurposed drugs
against human endometrial carcinoma cells. Life (Basel).
11:1602021.PubMed/NCBI
|
|
66
|
Wu Q, Zhang W, Liu Y, Huang Y, Wu H and Ma
C: Histone deacetylase 1 facilitates aerobic glycolysis and growth
of endometrial cancer. Oncol Lett. 22:7217212021. View Article : Google Scholar
|
|
67
|
Ma M, Fei X, Jiang D, Chen H, Xie X, Wang
Z and Huang Q: Research progress on the mechanism of histone
deacetylases in ferroptosis of glioma. Oncol Rev. 18:14321312024.
View Article : Google Scholar : PubMed/NCBI
|
|
68
|
Chen X, Wang Z, Li C, Zhang Z, Lu S, Wang
X, Liang Q, Zhu X, Pan C, Wang Q, et al: SIRT1 activated by AROS
sensitizes glioma cells to ferroptosis via induction of NAD+
depletion-dependent activation of ATF3. Redox Biol. 69:1030302024.
View Article : Google Scholar
|
|
69
|
Pai P, Das I, Reddy Y, Venkidesh BS,
Bhandari P, Madalageri M, Sadashivanavar V, Pai KSR, Rao P,
Oruganti S, et al: Targeting glioblastoma with HDAC inhibitors:
Insights into hydroxamic acid-based therapeutic strategies. Acta
Neuropathol Commun. 14:92025. View Article : Google Scholar : PubMed/NCBI
|
|
70
|
Fan F, Liu P, Bao R, Chen J, Zhou M, Mo Z,
Ma Y, Liu H, Zhou Y, Cai X, et al: A dual PI3K/HDAC inhibitor
induces immunogenic ferroptosis to potentiate cancer immune
checkpoint therapy. Cancer Res. 81:6233–6245. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
71
|
Mormino A, Cocozza G, Fontemaggi G,
Valente S, Esposito V, Santoro A, Bernardini G, Santoni A, Fazi F,
Mai A, et al: Histone-deacetylase 8 drives the immune response and
the growth of glioma. Glia. 69:2682–2698. 2021. View Article : Google Scholar
|
|
72
|
Han W, Yu F, Cao J, Dong B, Guan W and Shi
J: Valproic acid enhanced apoptosis by promoting autophagy via
Akt/mTOR signaling in glioma. Cell Transplant.
29:9636897209818782020. View Article : Google Scholar
|
|
73
|
Thotala D, Karvas RM, Engelbach JA, Garbow
JR, Hallahan AN, DeWees TA, Laszlo A and Hallahan DE: Valproic acid
enhances the efficacy of radiation therapy by protecting normal
hippocampal neurons and sensitizing malignant glioblastoma cells.
Oncotarget. 6:35004–35022. 2015. View Article : Google Scholar
|
|
74
|
Mehndiratta S, Qian B, Chuang JY, Liou JP
and Shih JC: N-methylpropargylamine-conjugated hydroxamic acids as
dual inhibitors of monoamine oxidase A and histone deacetylase for
glioma treatment. J Med Chem. 65:2208–2224. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
75
|
Kunadis E, Lakiotaki E, Korkolopoulou P
and Piperi C: Targeting post-translational histone modifying
enzymes in glioblastoma. Pharmacol Ther. 220:1077212021. View Article : Google Scholar : PubMed/NCBI
|
|
76
|
Riyas Mohamed FR and Yaqinuddin A:
Epigenetic reprogramming and antitumor immune responses in gliomas:
A systematic review. Med Oncol. 42:2132025. View Article : Google Scholar
|
|
77
|
Ozair A, Bhat V, Alisch RS, Khosla AA,
Kotecha RR, Odia Y, McDermott MW and Ahluwalia MS: DNA methylation
and histone modification in low-grade gliomas: Current
understanding and potential clinical targets. Cancers (Basel).
15:13422023. View Article : Google Scholar : PubMed/NCBI
|
|
78
|
Khalid U, Simovic M, Hammann LA, Iskar M,
Wong JKL, Kumar R, Jugold M, Sill M, Bolkestein M, Kolb T, et al: A
synergistic interaction between HDAC- and PARP inhibitors in
childhood tumors with chromothripsis. Int J Cancer. 151:590–606.
2022. View Article : Google Scholar : PubMed/NCBI
|
|
79
|
He Q, Yu C, Li Y, Hao P, Mai H, Guo R,
Zhong G, Zhang K, Wong C, Chen Q and Chen Y: ERRα contributes to
HDAC6-induced chemoresistance of osteosarcoma cells. Cell Biol
Toxicol. 39:813–825. 2023. View Article : Google Scholar
|
|
80
|
Wei LH, Torng PL, Hsiao SM, Jeng YM, Chen
MW and Chen CA: Histone deacetylase 6 regulates estrogen receptor
alpha in uterine leiomyoma. Reprod Sci. 18:755–762. 2011.
View Article : Google Scholar
|
|
81
|
Stopper D, Biermann L, Watson PR, Li J,
König B, Gaynes MN, Pessanha de Carvalho L, Klose J, Hanl M,
Hamacher A, et al: Exploring alternative zinc-binding groups in
histone deacetylase (HDAC) inhibitors uncovers DS-103 as a potent
ethylhydrazide-based HDAC inhibitor with chemosensitizing
properties. J Med Chem. 68:4426–4452. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
82
|
Long J, Jia MY, Fang WY, Chen XJ, Mu LL,
Wang ZY, Shen Y, Xiang RF, Wang LN, Wang L, et al: FLT3 inhibition
upregulates HDAC8 via FOXO to inactivate p53 and promote
maintenance of FLT3-ITD+ acute myeloid leukemia. Blood.
135:1472–1483. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
83
|
McGuire JJ, Nerlakanti N, Lo CH, Tauro M,
Utset-Ward TJ, Reed DR and Lynch CC: Histone deacetylase inhibition
prevents the growth of primary and metastatic osteosarcoma. Int J
Cancer. 147:2811–2823. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
84
|
Sawai T, Yamanegi K, Nishiura H, Futani H
and Tachibana T: Sodium valproate enhances semaphorin 3A-mediated
anti-angiogenesis and tumor growth inhibition in human osteosarcoma
cells. Anticancer Res. 43:2539–2550. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
85
|
Rossi M, De Martino V, Di Giuseppe L,
Battafarano G, Di Gregorio J, Terreri S, Marampon F, Minisola S and
Del Fattore A: Anti-proliferative, pro-apototic and anti-migratory
properties of HDAC inhibitor PXD-101 on osteosarcoma cell lines.
Arch Biochem Biophys. 734:1094892023. View Article : Google Scholar : PubMed/NCBI
|
|
86
|
Magar AG, Morya VK, Koh YH and Noh KC:
Synergistic HDAC4/8 inhibition sensitizes osteosarcoma to
doxorubicin via pAKT/RUNX2 pathway modulation. Int J Mol Sci.
26:35742025. View Article : Google Scholar
|
|
87
|
Collier CD, Getty PJ and Greenfield EM:
Targeting the cancer epigenome with histone deacetylase inhibitors
in osteosarcoma. Adv Exp Med Biol. 1258:55–75. 2020. View Article : Google Scholar
|
|
88
|
Ding K, Liu H, Yang H, Zhu H, Ma J, Peng
H, Huang H, Shi W, Cao L, Wu W, et al: A prospective phase 2 study
of combination epigenetic therapy against relapsed/refractory
peripheral T cell lymphoma. Med. 5:1393–1401.e2. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
89
|
Pu J, Liu T, Wang X, Sharma A,
Schmidt-Wolf IGH, Jiang L and Hou J: Exploring the role of histone
deacetylase and histone deacetylase inhibitors in the context of
multiple myeloma: Mechanisms, therapeutic implications, and future
perspectives. Exp Hematol Oncol. 13:452024. View Article : Google Scholar
|
|
90
|
Gössl FJ, Polo P, Helmprobst F, Menzenbach
A, Visekruna A, Gress TM, Adhikary T and Lauth M: ER-phagy mediates
the anti-tumoral synergism between HDAC inhibition and
chemotherapy. Cell Commun Signal. 23:2022025. View Article : Google Scholar
|
|
91
|
Xu T, Fang Y, Gu Y, Xu D, Hu T, Yu T, Xu
YY, Shen HY, Ma P and Shu Y: HDAC inhibitor SAHA enhances antitumor
immunity via the HDAC1/JAK1/FGL1 axis in lung adenocarcinoma. J
Immunother Cancer. 12:e0100772024. View Article : Google Scholar : PubMed/NCBI
|
|
92
|
Ferro A, Graikioti D, Gezer E,
Athanassopoulos CM and Cuendet M: Entinostat-bortezomib hybrids
against multiple myeloma. Molecules. 28:14562023. View Article : Google Scholar : PubMed/NCBI
|
|
93
|
Hirano M, Imai Y, Kaito Y, Murayama T,
Sato K, Ishida T, Yamamoto J, Ito T, Futami M, Ri M, et al:
Small-molecule HDAC and Akt inhibitors suppress tumor growth and
enhance immunotherapy in multiple myeloma. J Exp Clin Cancer Res.
40:1102021. View Article : Google Scholar : PubMed/NCBI
|
|
94
|
Florean C, Lernoux M, Lorant A, Losson H,
Bormans G, Schnekenburger M and Diederich M: HDAC6 inhibitors
sensitize resistant t(11;14) multiple myeloma cells to a
combination of bortezomib and BH3 mimetics. Haematologica.
110:784–790. 2025.
|
|
95
|
García-Guerrero E, Götz R, Doose S, Sauer
M, Rodríguez-Gil A, Nerreter T, Kortüm KM, Pérez-Simón JA, Einsele
H, Hudecek M and Danhof S: Upregulation of CD38 expression on
multiple myeloma cells by novel HDAC6 inhibitors is a class effect
and augments the efficacy of daratumumab. Leukemia. 35:201–214.
2021. View Article : Google Scholar
|
|
96
|
Zhan W, Liao X, Liu J, Tian T, Yu L and Li
R: USP38 regulates the stemness and chemoresistance of human
colorectal cancer via regulation of HDAC3. Oncogenesis. 9:482020.
View Article : Google Scholar : PubMed/NCBI
|
|
97
|
Szigety KM, Liu F, Yuan CY, Moran DJ,
Horrell J, Gochnauer HR, Cohen RN, Katz JP, Kaestner KH, Seykora
JT, et al: HDAC3 ensures stepwise epidermal stratification via
NCoR/SMRT-reliant mechanisms independent of its histone deacetylase
activity. Genes Dev. 34:973–988. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
98
|
Dai B, Wang F, Wang Y, Zhu J, Li Y, Zhang
T, Zhao L, Wang L, Gao W, Li J, et al: Targeting HDAC3 to overcome
the resistance to ATRA or arsenic in acute promyelocytic leukemia
through ubiquitination and degradation of PML-RARα. Cell Death
Differ. 30:1320–1333. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
99
|
Li L, Hao S, Gao M, Liu J, Xu X, Huang J,
Cheng G and Yang H: HDAC3 inhibition promotes antitumor immunity by
enhancing CXCL10-mediated chemotaxis and recruiting of immune
cells. Cancer Immunol Res. 11:657–673. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
100
|
Han R, Luo Y, Gao J, Zhou H, Wang Y, Chen
J, Zheng G and Ling C: HDAC3: A multifaceted modulator in
immunotherapy sensitization. Vaccines (Basel). 13:1822025.
View Article : Google Scholar : PubMed/NCBI
|
|
101
|
Eichner LJ, Curtis SD, Brun SN, McGuire
CK, Gushterova I, Baumgart JT, Trefts E, Ross DS, Rymoff TJ and
Shaw RJ: HDAC3 is critical in tumor development and therapeutic
resistance in Kras-mutant non-small cell lung cancer. Sci Adv.
9:eadd32432023. View Article : Google Scholar : PubMed/NCBI
|
|
102
|
Wu X, Yang C, Sun F, Zhang Y, Wang Y, Li X
and Zheng F: Enterotoxigenic bacteroides fragilis (ETBF) enhances
colorectal cancer cell proliferation and metastasis through
HDAC3/miR-139-3p pathway. Biochem Genet. 62:3904–3919. 2024.
View Article : Google Scholar
|
|
103
|
Cassandri M, Porrazzo A, Pomella S, Noce
B, Zwergel C, Aiello FA, Vulcano F, Milazzo L, Camero S, Pajalunga
D, et al: HDAC3 genetic and pharmacologic inhibition
radiosensitizes fusion positive rhabdomyosarcoma by promoting DNA
double-strand breaks. Cell Death Discov. 10:3512024. View Article : Google Scholar : PubMed/NCBI
|
|
104
|
Zhou Y, Jin X, Ma J, Ding D, Huang Z,
Sheng H, Yan Y, Pan Y, Wei T, Wang L, et al: HDAC5 loss impairs RB
repression of pro-oncogenic genes and confers CDK4/6 inhibitor
resistance in cancer. Cancer Res. 81:1486–1499. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
105
|
Fan J, Lou B, Chen W, Zhang J, Lin S, Lv
FF and Chen Y: Down-regulation of HDAC5 inhibits growth of human
hepatocellular carcinoma by induction of apoptosis and cell cycle
arrest. Tumour Biol. 35:11523–11532. 2014. View Article : Google Scholar
|
|
106
|
Lyu H, Ishimura A, Suzuki R, Buyanbat K,
Batbayar G, Meguro-Horike M, Horike SI, Yano S and Suzuki T: HDAC5,
an early osimertinib-responsive gene, is a novel therapeutic target
for the drug resistance in EGFR-mutant lung adenocarcinoma cells.
Biochem Biophys Rep. 42:1020162025.PubMed/NCBI
|
|
107
|
Pan P, Qin G, Wang B, Yu H, Chen J, Liu J,
Bing K, Shen J, Ren D, Zhao Y, et al: HDAC5 loss enhances
phospholipid-derived arachidonic acid generation and confers
sensitivity to cPLA2 inhibition in pancreatic cancer. Cancer Res.
82:4542–4554. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
108
|
Liu C, Zheng D, Pu X and Li S: HDAC7: A
promising target in cancer. Front Oncol. 14:13279332024. View Article : Google Scholar
|
|
109
|
Lu P, Deng S, Liu J, Xiao Q, Zhou Z, Li S,
Xin J, Shu G, Yi B and Yin G: Tweety homolog 3 promotes colorectal
cancer progression through mutual regulation of histone deacetylase
7. MedComm (2020). 5:e5762024. View Article : Google Scholar : PubMed/NCBI
|
|
110
|
Gautam N, Chapagain PP, Adhikari NP and
Tiwari PB: Characterization of molecular interactions between HDAC7
and MEF2A. J Biomol Struct Dyn. 1–10. 2024.(Epub ahead of print).
View Article : Google Scholar
|
|
111
|
Lu W, Zhuang G, Guan Y, Li Y, Liu L and
Xiao M: Comprehensive analysis of HDAC7 expression and its
prognostic value in diffuse large B cell lymphoma: A review.
Medicine (Baltimore). 102:e345772023. View Article : Google Scholar : PubMed/NCBI
|
|
112
|
Jin Y, Qi X, Yu X, Cheng X, Chen B, Wu M,
Zhang J, Yin H, Lu Y, Zhou Y, et al: Discovery of a potential
hematologic malignancies therapy: Selective and potent HDAC7 PROTAC
degrader targeting non-enzymatic function. Acta Pharm Sin B.
15:1659–1679. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
113
|
Bi L, Ren Y, Feng M, Meng P, Wang Q, Chen
W, Jiao Q, Wang Y, Du L, Zhou F, et al: HDAC11 regulates glycolysis
through the LKB1/AMPK signaling pathway to maintain hepatocellular
carcinoma stemness. Cancer Res. 81:2015–2028. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
114
|
Wang W, Ding B, Lou W and Lin S: Promoter
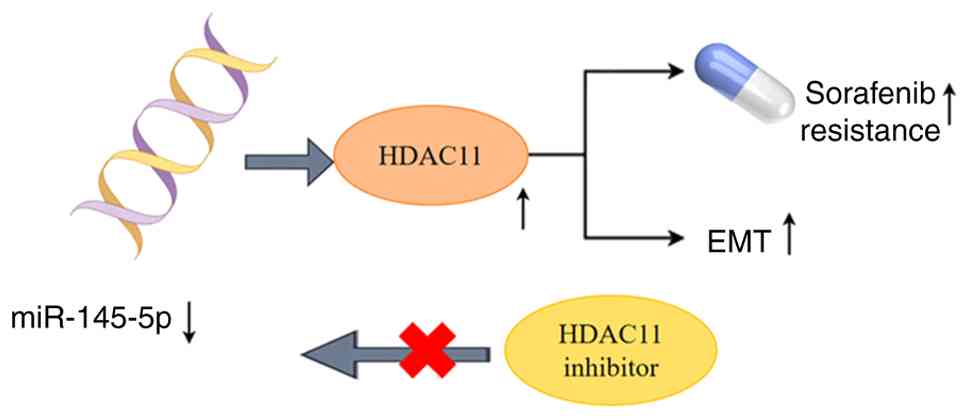
hypomethylation and miR-145-5p downregulation-mediated HDAC11
overexpression promotes sorafenib resistance and metastasis of
hepatocellular carcinoma cells. Front Cell Dev Biol. 8:7242020.
View Article : Google Scholar
|
|
115
|
Liu Y, Tong X, Hu W and Chen D: HDAC11: A
novel target for improved cancer therapy. Biomed Pharmacother.
166:1154182023. View Article : Google Scholar : PubMed/NCBI
|
|
116
|
Chen J, Cheng F, Sahakian E, Powers J,
Wang Z, Tao J, Seto E, Pinilla-Ibarz J and Sotomayor EM: HDAC11
regulates expression of C/EBPβ and immunosuppressive molecules in
myeloid-derived suppressor cells. J Leukoc Biol. 109:891–900. 2021.
View Article : Google Scholar
|
|
117
|
Villagra A, Cheng F, Wang HW, Suarez I,
Glozak M, Maurin M, Nguyen D, Wright KL, Atadja PW, Bhalla K, et
al: The histone deacetylase HDAC11 regulates the expression of
interleukin 10 and immune tolerance. Nat Immunol. 10:92–100. 2009.
View Article : Google Scholar
|
|
118
|
Li R, Wu X, Zhao P, Xue K and Li J: A
pan-cancer analysis identifies HDAC11 as an immunological and
prognostic biomarker. FASEB J. 36:e223262022. View Article : Google Scholar : PubMed/NCBI
|
|
119
|
Zhao C, Dong H, Xu Q and Zhang Y: Histone
deacetylase (HDAC) inhibitors in cancer: A patent review
(2017-present). Expert Opin Ther Pat. 30:263–274. 2020. View Article : Google Scholar
|
|
120
|
Biersack B, Polat S and Höpfner M:
Anticancer properties of chimeric HDAC and kinase inhibitors. Semin
Cancer Biol. 83:472–486. 2022. View Article : Google Scholar
|
|
121
|
Zhou WH, Luo Y, Li RX, Degrace P, Jourdan
T, Qiao F, Chen LQ, Zhang ML and Du ZY: Inhibition of mitochondrial
fatty acid β-oxidation activates mTORC1 pathway and protein
synthesis via Gcn5-dependent acetylation of raptor in zebrafish. J
Biol Chem. 299:1052202023. View Article : Google Scholar : PubMed/NCBI
|
|
122
|
Mukherjee A, Zamani F and Suzuki T:
Evolution of slow-binding inhibitors targeting histone deacetylase
isoforms. J Med Chem. 66:11672–11700. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
123
|
San-Miguel JF, Hungria VTM, Yoon SS,
Beksac M, Dimopoulos MA, Elghandour A, Jedrzejczak WW, Günther A,
Nakorn TN, Siritanaratkul N, et al: Panobinostat plus bortezomib
and dexamethasone versus placebo plus bortezomib and dexamethasone
in patients with relapsed or relapsed and refractory multiple
myeloma: A multicentre, randomised, double-blind phase 3 trial.
Lancet Oncol. 15:1195–1206. 2014. View Article : Google Scholar
|
|
124
|
Zhang L, Zhang Y, Mehta A, Boufraqech M,
Davis S, Wang J, Tian Z, Yu Z, Boxer MB, Kiefer JA, et al: Dual
inhibition of HDAC and EGFR signaling with CUDC-101 induces potent
suppression of tumor growth and metastasis in anaplastic thyroid
cancer. Oncotarget. 6:9073–9085. 2015. View Article : Google Scholar
|
|
125
|
San José-Enériz E, Gimenez-Camino N, Rabal
O, Garate L, Miranda E, Gómez-Echarte N, García F, Charalampopoulou
S, Sáez E, Vilas-Zornoza A, et al: Epigenetic-based differentiation
therapy for acute myeloid leukemia. Nat Commun. 15:55702024.
View Article : Google Scholar
|
|
126
|
Sim W, Lim WM, Hii LW, Leong CO and Mai
CW: Targeting pancreatic cancer immune evasion by inhibiting
histone deacetylases. World J Gastroenterol. 28:1934–1945. 2022.
View Article : Google Scholar
|
|
127
|
Li X, Wang Y, Deng S, Zhu G, Wang C,
Johnson NA, Zhang Z, Tirado CR, Xu Y, Metang LA, et al: Loss of
SYNCRIP unleashes APOBEC-driven mutagenesis, tumor heterogeneity,
and AR-targeted therapy resistance in prostate cancer. Cancer Cell.
41:1427–1449.e12. 2023. View Article : Google Scholar
|
|
128
|
Yang L, Qiu Q, Wang J, Wen Y, Li H, Liang
R, Feng Y, Wang F, Lin X, Tang M, et al: Preclinical and
first-in-human of purinostat mesylate, a novel selective HDAC I/IIb
inhibitor, in relapsed/refractory multiple myeloma and lymphoma.
Signal Transduct Target Ther. 10:2012025. View Article : Google Scholar : PubMed/NCBI
|
|
129
|
Bishton MJ, Harrison SJ, Martin BP,
McLaughlin N, James C, Josefsson EC, Henley KJ, Kile BT, Prince HM
and Johnstone RW: Deciphering the molecular and biologic processes
that mediate histone deacetylase inhibitor-induced
thrombocytopenia. Blood. 117:3658–3668. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
130
|
Liu YM and Liou JP: An updated patent
review of histone deacetylase (HDAC) inhibitors in cancer
(2020-present). Expert Opin Ther Pat. 33:349–369. 2023. View Article : Google Scholar
|
|
131
|
Song S, Wen Y, Tong H, Loro E, Gong Y, Liu
J, Hong S, Li L, Khurana TS, Chu M and Sun Z: The HDAC3 enzymatic
activity regulates skeletal muscle fuel metabolism. J Mol Cell
Biol. 11:133–143. 2019. View Article : Google Scholar
|
|
132
|
Bülbül EF, Robaa D, Sun P, Mahmoudi F,
Melesina J, Zessin M, Schutkowski M and Sippl W: Application of
ligand- and structure-based prediction models for the design of
alkylhydrazide-based HDAC3 inhibitors as novel anti-cancer
compounds. Pharmaceuticals (Basel). 16:9682023. View Article : Google Scholar
|
|
133
|
Ren Y, Li S, Zhu R, Wan C, Song D, Zhu J,
Cai G, Long S, Kong L and Yu W: Discovery of STAT3 and histone
deacetylase (HDAC) dual-pathway inhibitors for the treatment of
solid cancer. J Med Chem. 64:7468–7482. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
134
|
Xie X, Zhang O, Yeo MJR, Lee C, Tao R,
Harry SA, Payne NC, Nam E, Paul L, Li Y, et al: Converging
mechanism of UM171 and KBTBD4 neomorphic cancer mutations. Nature.
639:241–249. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
135
|
Gao J, Hou B, Zhu Q, Yang L, Jiang X, Zou
Z, Li X, Xu T, Zheng M, Chen YH, et al: Engineered bioorthogonal
POLY-PROTAC nanoparticles for tumour-specific protein degradation
and precise cancer therapy. Nat Commun. 13:43182022. View Article : Google Scholar : PubMed/NCBI
|
|
136
|
Sun J, Lian X, Lv C, Li H, Lin Z, Luo S,
Liu Y, Xu Y, Jiang X, Xu W, et al: Trps1 acts as a regulator of
Sf-1 transcription and testosterone synthesis in mouse Leydig
cells. Cell Biol Toxicol. 39:3141–3157. 2023. View Article : Google Scholar
|
|
137
|
Chen M, Wang C, Wang X, Tu Z, Ding Z and
Liu Z: An ‘AND’ logic-gated prodrug micelle locally stimulates
antitumor immunity. Adv Mater. 36:e23078182024. View Article : Google Scholar : PubMed/NCBI
|
|
138
|
Li S, Liu Y, Tian T, Zhang T, Lin S, Zhou
M, Zhang X, Lin Y and Cai X: Bioswitchable delivery of microRNA by
framework nucleic acids: Application to bone regeneration. Small.
17:e21043592021. View Article : Google Scholar
|
|
139
|
Xing X, Zhong W, Tang P, Tao Q, Lu X and
Zhong L: Tracking intracellular nuclear targeted-chemotherapy of
chidamide-loaded Prussian blue nanocarriers by SERS mapping.
Colloids Surf B Biointerfaces. 229:1134692023. View Article : Google Scholar : PubMed/NCBI
|
|
140
|
Hong L, Ni M, Xue F, Jiang T, Wu X, Li C,
Liang S, Chen T, Luo C and Wu Q: The role of HDAC3 in pulmonary
diseases. Lung. 203:472025. View Article : Google Scholar : PubMed/NCBI
|
|
141
|
Zhang F, Yue K, Sun S, Lu S, Jia G, Zha Y,
Zhang S, Chou CJ, Liao C, Li X and Duan Y: Targeting histone
deacetylase 11 with a highly selective inhibitor for the treatment
of MASLD. Adv Sci (Weinh). 12:e24129032025. View Article : Google Scholar : PubMed/NCBI
|
|
142
|
Pramanik SD, Kumar Halder A, Mukherjee U,
Kumar D, Dey YN and R M: Potential of histone deacetylase
inhibitors in the control and regulation of prostate, breast and
ovarian cancer. Front Chem. 10:9482172022. View Article : Google Scholar : PubMed/NCBI
|
|
143
|
Woods DM, Sodré AL, Villagra A, Sarnaik A,
Sotomayor EM and Weber J: HDAC inhibition upregulates PD-1 ligands
in melanoma and augments immunotherapy with PD-1 blockade. Cancer
Immunol Res. 3:1375–1385. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
144
|
Baretti M and Yarchoan M: Epigenetic
modifiers synergize with immune-checkpoint blockade to enhance
long-lasting antitumor efficacy. J Clin Invest. 131:e1510022021.
View Article : Google Scholar
|
|
145
|
Qian C, Lai CJ, Bao R, Wang DG, Wang J, Xu
GX, Atoyan R, Qu H, Yin L, Samson M, et al: Cancer network
disruption by a single molecule inhibitor targeting both histone
deacetylase activity and phosphatidylinositol 3-kinase signaling.
Clin Cancer Res. 18:4104–4113. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
146
|
Zhou L and Yu CW: Epigenetic modulations
in triple-negative breast cancer: Therapeutic implications for
tumor microenvironment. Pharmacol Res. 204:1072052024. View Article : Google Scholar : PubMed/NCBI
|
|
147
|
Venneker S, van Eenige R, Kruisselbrink
AB, Palubeckaitė I, Taliento AE, Briaire-de Bruijn IH, Hogendoorn
PCW, van de Sande MAJ, Gelderblom H, Mei H, et al: Histone
deacetylase inhibitors as a therapeutic strategy to eliminate
neoplastic ‘stromal’ cells from giant cell tumors of bone. Cancers
(Basel). 14:47082022. View Article : Google Scholar : PubMed/NCBI
|
|
148
|
Guo F and Wang H: Potential of histone
deacetylase inhibitors for the therapy of ovarian cancer. Front
Oncol. 12:10571862022. View Article : Google Scholar
|
|
149
|
Li X, Su X, Liu R, Pan Y, Fang J, Cao L,
Feng C, Shang Q, Chen Y, Shao C and Shi Y: HDAC inhibition
potentiates anti-tumor activity of macrophages and enhances
anti-PD-L1-mediated tumor suppression. Oncogene. 40:1836–1850.
2021. View Article : Google Scholar : PubMed/NCBI
|
|
150
|
Nguyen A, Ho L, Hogg R, Chen L, Walsh SR
and Wan Y: HDACi promotes inflammatory remodeling of the tumor
microenvironment to enhance epitope spreading and antitumor
immunity. J Clin Invest. 132:e1592832022. View Article : Google Scholar
|
|
151
|
Zhang W, Ge L, Zhang Y, Zhang Z, Zhang W,
Song F, Huang P and Xu T: Targeted intervention of tumor
microenvironment with HDAC inhibitors and their combination therapy
strategies. Eur J Med Res. 30:692025. View Article : Google Scholar : PubMed/NCBI
|
|
152
|
Jing L, Qian Z, Gao Q, Sun R, Zhen Z, Wang
G, Yang X, Li H, Guo T and Zhang W: Diffuse midline glioma treated
with epigenetic agent-based immunotherapy. Signal Transduct Target
Ther. 8:232023. View Article : Google Scholar : PubMed/NCBI
|
|
153
|
Fresquet V, Garcia-Barchino MJ, Larrayoz
M, Celay J, Vicente C, Fernandez-Galilea M, Larrayoz MJ, Calasanz
MJ, Panizo C, Junza A, et al: Endogenous retroelement activation by
epigenetic therapy reverses the warburg effect and elicits
mitochondrial-mediated cancer cell death. Cancer Discov.
11:1268–1285. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
154
|
Jenke R, Reßing N, Hansen FK, Aigner A and
Büch T: Anticancer therapy with HDAC inhibitors: Mechanism-based
combination strategies and future perspectives. Cancers (Basel).
13:6342021. View Article : Google Scholar : PubMed/NCBI
|
|
155
|
Chen Y, Su J, Li S, Chen F, Zhang Y, Wang
X, Zhang Y, Wang X, Yuan Z, Ren S, et al: Structural modifications
and prospects of histone deacetylase (HDAC) inhibitors in cancer.
Curr Med Chem. 32:8530–8555. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
156
|
Zou Q, Zhang Y, Zhou H, Lai Y, Cao Y, Li
Z, Su N, Li W, Huang H, Liu P, et al: Chidamide, a histone
deacetylase inhibitor, combined with R-GemOx in relapsed/refractory
diffuse large B-cell lymphoma (TRUST): A multicenter, single-arm,
phase 2 trial. Cancer Med. 14:e709192025. View Article : Google Scholar : PubMed/NCBI
|
|
157
|
Wang L, Gao M, Wang T, Cui P, Chen G, Han
X, Ma Z, Yang W, Jing F, Ma J, et al: First-line treatment with
HDACis plus tislelizumab combined with chemotherapy in advanced
NSCLC: A single-arm phase II study. Oncologist. 30:oyaf1552025.
View Article : Google Scholar
|
|
158
|
Wieduwilt MJ, Pawlowska N, Thomas S, Olin
R, Logan AC, Damon LE, Martin T, Kang M, Sayre PH, Boyer W, et al:
Histone deacetylase inhibition with panobinostat combined with
intensive induction chemotherapy in older patients with acute
myeloid leukemia: Phase I study results. Clin Cancer Res.
25:4917–4923. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
159
|
Gu S, Hou Y, Dovat K, Dovat S, Song C and
Ge Z: Synergistic effect of HDAC inhibitor chidamide with
cladribine on cell cycle arrest and apoptosis by targeting
HDAC2/c-Myc/RCC1 axis in acute myeloid leukemia. Exp Hematol Oncol.
12:232023. View Article : Google Scholar
|
|
160
|
Liu S, Ma S, Liu G, Hou L, Guan Y, Liu L,
Meng Y, Yu W, Liu T, Zhou L, et al: CK2B induces CD8+
T-cell exhaustion through HDAC8-mediated epigenetic reprogramming
to limit the efficacy of anti-PD-1 therapy in non-small-cell lung
cancer. Adv Sci (Weinh). 12:e24110532025. View Article : Google Scholar : PubMed/NCBI
|
|
161
|
Chen C, Li X, Zhao H, Liu M, Du J, Zhang
J, Yang X, Hou X and Fang H: Discovery of DNA-targeting HDAC
inhibitors with potent antitumor efficacy in vivo that trigger
antitumor immunity. J Med Chem. 65:3667–3683. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
162
|
Liang R, Ding D, Li Y, Lan T, Ryabtseva S,
Huang S, Ren J, Huang H and Wei B: HDACi combination therapy with
IDO1i remodels the tumor microenvironment and boosts antitumor
efficacy in colorectal cancer with microsatellite stability. J
Nanobiotechnology. 22:7532024. View Article : Google Scholar : PubMed/NCBI
|
|
163
|
Guerriero JL, Sotayo A, Ponichtera HE,
Castrillon JA, Pourzia AL, Schad S, Johnson SF, Carrasco RD, Lazo
S, Bronson RT, et al: Class IIa HDAC inhibition reduces breast
tumours and metastases through anti-tumour macrophages. Nature.
543:428–432. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
164
|
Chen KY, Kibayashi T, Giguelay A, Hata M,
Nakajima S, Mikami N, Takeshima Y, Ichiyama K, Omiya R, Ludwig LS,
et al: Genome-wide CRISPR screen in human T cells reveals
regulators of FOXP3. Nature. 642:191–200. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
165
|
Gediya P, Vyas VK, Carafa V, Sitwala N,
Della Torre L, Poziello A, Kurohara T, Suzuki T, Sanna V, Raguraman
V, et al: Discovery of novel
tetrahydrobenzo[b]thiophene-3-carbonitriles as histone deacetylase
inhibitors. Bioorg Chem. 110:1048012021. View Article : Google Scholar : PubMed/NCBI
|
|
166
|
Elmezayen AD, Al-Obaidi A and Yelekçi K:
Discovery of novel isoform-selective histone deacetylases 5 and 9
inhibitors through combined ligand-based pharmacophore modeling,
molecular mocking, and molecular dynamics simulations for cancer
treatment. J Mol Graph Model. 106:1079372021. View Article : Google Scholar
|
|
167
|
Yao K, Li Y, Wei W, Liu S, Wang X, Xu J,
Zhang R, Wu Z, Guo C, Yang L and Hu L: Synthesis and biological
evaluation of novel 4-Arylaminoquinolines derivatives as EGFR/HDAC
inhibitors. Bioorg Med Chem Lett. 122:1302142025. View Article : Google Scholar
|
|
168
|
Parag-Sharma K, Tasoulas J, Musicant AM,
do Nascimento-Filho CHV, Zhu Z, Twomey C, Liu P, Castilho RM and
Amelio AL: Synergistic efficacy of combined EGFR and HDAC
inhibitors overcomes tolerance to EGFR monotherapy in salivary
mucoepidermoid carcinoma. Oral Oncol. 115:1051662021. View Article : Google Scholar
|
|
169
|
Cao J, Zhao W, Zhao C, Liu Q, Li S, Zhang
G, Chou CJ and Zhang Y: Development of a bestatin-SAHA hybrid with
dual inhibitory activity against APN and HDAC. Molecules.
25:49912020. View Article : Google Scholar : PubMed/NCBI
|
|
170
|
Zhang Y, Wang H, Zhan Z, Gan L and Bai O:
Mechanisms of HDACs in cancer development. Front Immunol.
16:15292392025. View Article : Google Scholar
|
|
171
|
Hai R, Yang D, Zheng F, Wang W, Han X,
Bode AM and Luo X: The emerging roles of HDACs and their
therapeutic implications in cancer. Eur J Pharmacol.
931:1752162022. View Article : Google Scholar
|
|
172
|
Sanaei M and Kavoosi F: Histone
deacetylases and histone deacetylase inhibitors: Molecular
mechanisms of action in various cancers. Adv Biomed Res. 8:632019.
View Article : Google Scholar : PubMed/NCBI
|
|
173
|
Oliveira-Silva JM, de Oliveira LS,
Tagliéri JVM, Lopes LB, de Souza CVE, Batistão HKA and
Castro-Gamero AM: HDAC6: Tumor progression and beyond. Curr Cancer
Drug Targets. Jan 7–2025.(Epub ahead of print).
|
|
174
|
Jo H, Shim K and Jeoung D: Targeting HDAC6
to overcome autophagy-promoted anti-cancer drug resistance. Int J
Mol Sci. 23:95922022. View Article : Google Scholar
|
|
175
|
Kadier K, Niu T, Ding B, Chen B, Qi X,
Chen D, Cheng X, Fang Y, Zhou J, Zhao W, et al: PROTAC-mediated
HDAC7 protein degradation unveils its deacetylase-independent
proinflammatory function in macrophages. Adv Sci (Weinh).
11:e23094592024. View Article : Google Scholar : PubMed/NCBI
|