Introduction
Primary clear-cell carcinoma of the liver (PCCCL) is
an infrequent subtype of hepatocellular carcinoma (HCC), with its
incidence constituting 3–7% of all cases of HCC. Pathologically,
>50% of the tumor cells display clear cytoplasm replete with
glycogen. In comparison with common HCC, certain reports propose
that PCCCL may possess relatively lower invasiveness and a more
favorable prognosis. However, other investigations have indicated
that it is predisposed to microvascular invasion (MVI) and
extra-hepatic metastasis (1). As a
result, the overall survival outcome of PCCCL remains a subject of
contention.
While clear cell morphology can also occur in
metastatic renal cell carcinoma and other primary hepatic
neoplasms, immunohistochemical (IHC) confirmation of hepatocellular
origin using markers such as hepatocyte paraffin antigen 1
(HepPar-1), arginase-1 (Arg-1) and glypican-3 (GPC3) becomes
essential for the accurate diagnosis of PCCCL (2,3).
Furthermore, recent genomic studies have revealed that PCCCL may
harbor distinct molecular alterations compared with conventional
HCC, including a lower frequency of tumor protein 53 mutations and
enrichment of catenin β1 mutations, which could partially explain
its relatively indolent behavior in a subset of patients (4).
Currently, the treatment of advanced PCCCL primarily
adheres to the HCC diagnosis and treatment guidelines, with
surgical intervention serving as the cornerstone of treatment. The
combination of targeted therapy and immune checkpoint inhibitors,
such as programmed cell death (PD)-1 monoclonal antibodies, is
assuming an increasingly prominent role (5). Nevertheless, there is still a
substantial dearth of data regarding the differential efficacy of
this subtype in systemic treatment and its long-term survival
rates. The present case report aims to augment the diagnostic and
therapeutic approaches and experiences of PCCCL in clinical
practice.
Case report
A 37-year-old female patient was admitted to the
hospital in October 2021, presenting with persistent distending
pain in the right upper abdomen for 15 days. Throughout the disease
course, the patient did not exhibit any symptoms such as nausea,
vomiting, anorexia, fatigue, fever, chills or jaundice. The past
medical, family and personal history of the patient was
unremarkable. Upon admission, a series of laboratory examinations
were carried out (normal reference values are provided in
parentheses): Alanine aminotransferase was 27.7 U/l (7.0–40.0 U/l),
aspartate aminotransferase was 21.5 U/l (13.0–35.0 U/l) and total
bilirubin was 9.0 µmol/l (5.0–21.0 µmol/l). The results of the
hepatitis B serological profile were as follows: Hepatitis B
surface antigen (HBsAg), <0.1 index (<1 index); antibody to
hepatitis B surface antigen, 17.9 mIU/ml (<8.0 mIU/ml);
hepatitis Be antigen, 0 index (<0.8 index); hepatitis Be
antibody, 0.2 index (<0.8 index) and hepatitis B core antibody,
0.1 index (<0.5 index). The α-fetoprotein (AFP) level was 33.9
ng/ml (<25.0 ng/ml), and no elevation was detected in
carcinoembryonic antigen, ferritin or carbohydrate antigen 19-9.
Abdominal computed tomography (CT) revealed that the liver
maintained its normal shape and size. However, an irregular
mixed-density mass (containing fat density; Fig. 1A) was identified in segment S6 of
the liver. In October 2021, the patient underwent resection of this
segment S6. During the operation, the liver was found to be of
normal size, with a soft texture and a healthy red color. The
exophytic mass in segment S6 of the liver was densely adherent to
the omentum of the hepatic flexure of the colon. The tumor had two
areas of rupture, accompanied by tiny nodules in the adjacent
omentum. Postoperative pathological examination indicated clear
cell carcinoma in segment S6 of the liver. In >50% of the tumor
cells, clear cytoplasm was observed, which was rich in glycogen
(Fig. 1E). According to the
Edmondson-Steiner grading system, the mass was classified as grade
III. MVI was present (MVI grading, M2), and cancer metastasis was
evident in the omental nodules. IHC analysis showed positive
results for hepatocyte (Fig. 1F),
GPC3 (Fig. 1G) and Ki-67 (40%
positive) (Fig. 1H). The
postoperative diagnosis was PCCCL (Clinical Research Group of Liver
Cancer stage IIIa).
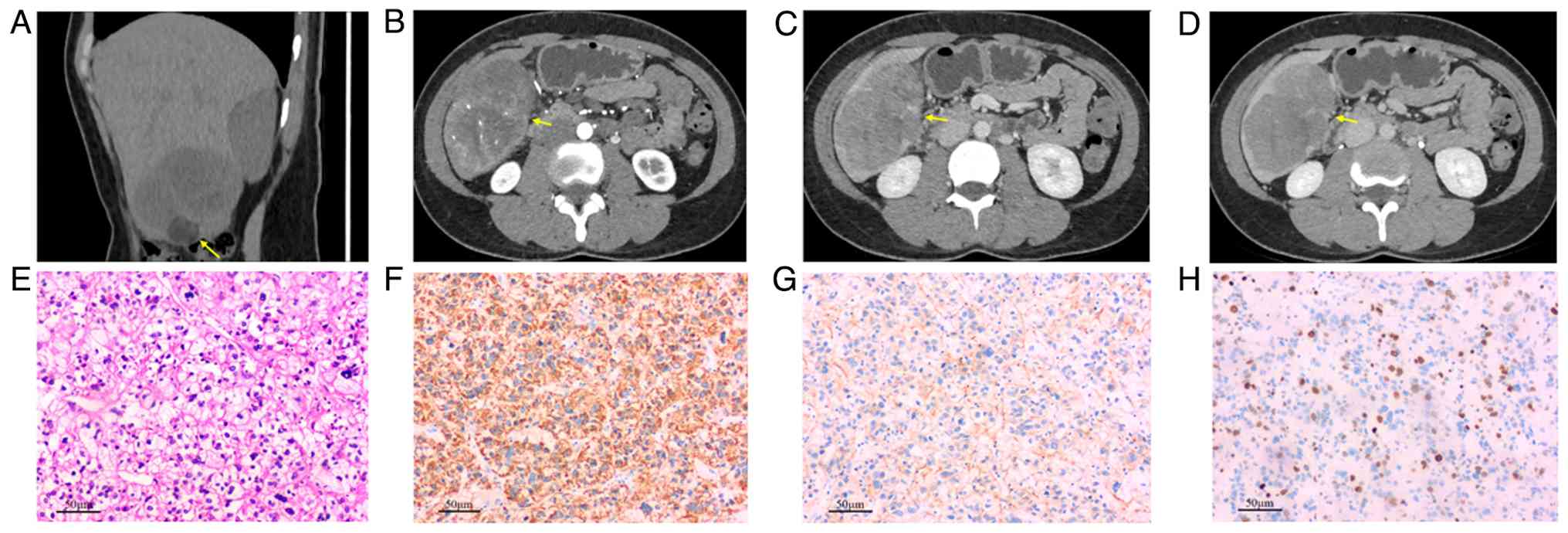 | Figure 1.(A) Preoperative non-contrast CT
revealed an irregular, mixed-density mass situated in segment S6
(as indicated by the yellow arrow in the figure, the same applies
to B-D), which contained fat components. The mass measured
7.31×4.92 cm, with CT values ranging from −11 to 34 HU. (B) During
the arterial phase of CT enhancement scanning, multiple small
vessels traverse the tumor, demonstrating moderate heterogeneous
enhancement. (C) In the venous phase and (D) the delayed phase, the
degree of enhancement diminishes, manifesting as the characteristic
of ‘fast-in and fast-out’. (E) H&E staining revealed that the
tumor tissue was predominantly constituted by a high proportion
(80%) of vacuolated clear cells. The tumor cells were arranged in a
sheet-like or nested configuration and exhibited a large cell
volume. The cytoplasm appeared transparent and vacuolated, whereas
the nuclei demonstrated irregular morphologies, suggesting marked
nuclear atypia. Notably, mitotic figures were rarely observed
(scale bar, 50 µm). (F-H) Immunohistochemistry: (F) Hepatocyte (+),
(G) glypican-3 (−) and (H) Ki-67 40% (scale bar, 50 µm). |
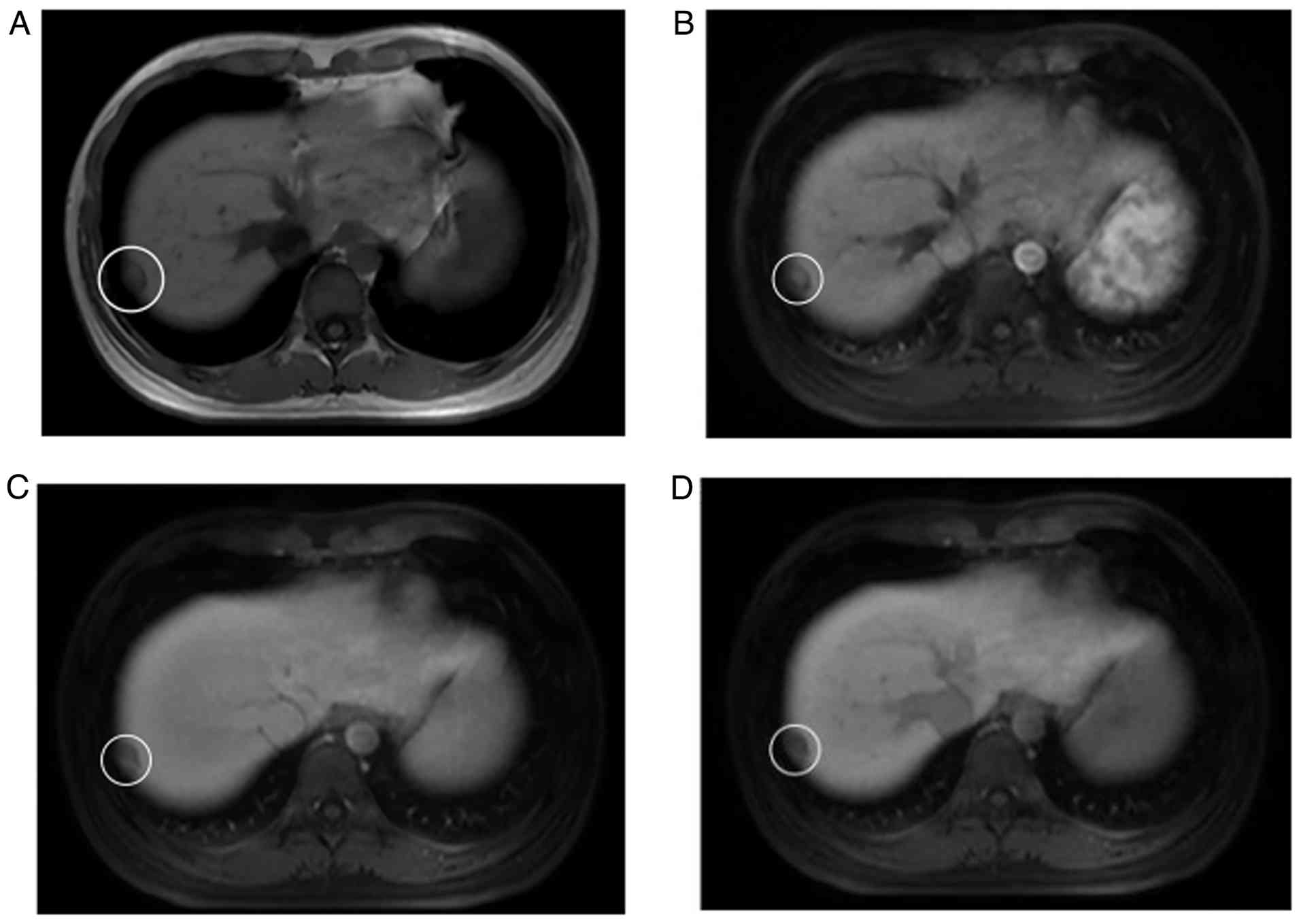
In November 2021, prior to planned further systemic
treatment, a follow-up abdominal MRI showed a newly detected
subcapsular nodule (18×10 mm) in segmen 7 (S7) of the liver. On
T1-weighted imaging, it presented as a slightly hypointense signal
lesion (Fig. 2A). Contrast-enhanced
scanning showed mild heterogeneous enhancement, and there was no
contrast agent uptake during the hepatobiliary-specific phase,
which was diagnosed as intrahepatic metastasis (Fig. 2B-D). Subsequently, the patient was
administered a combination therapy of lenvatinib (8 mg once daily)
and toripalimab (240 mg).
Following the completion of six cycles of
immunotherapy, an abdominal CT in July 2022 revealed that the
nodule in the liver had not notably diminished in size (17.0×11.0
mm). The treatment regimen was adjusted to lenvatinib at 8 mg once
daily in combination with tislelizumab at 200.0 mg every 3 weeks
for maintenance treatment. Endoscopy examination indicated antral
gastritis and inflammatory alterations in the terminal ileum and
the colon. Persistent myelosuppression was noted, with a white
blood cell count of 3.3×109/l. Thyroid function was
abnormal, with the following specific parameters: Thyroid
stimulating hormone, 22.6 mIU/l (0.5–4.7 mIU/l); FT3 and FT4 were
normal. The adrenocorticotropic hormone (ACTH) level was 32.4 pg/ml
(1.6–13.9 pg/ml between 7:00 and 10:00 a.m.). The patient was
regularly followed up in the outpatient clinic during this period
and continued to receive combination therapy with lenvatinib and
tislelizumab. In August 2023, laboratory results revealed an ACTH
concentration of 816.0 pg/ml, the patient experienced an
immune-related adverse event (irAEs). As a result, immunotherapy
was temporarily suspended. An abdominal CT scan 2 months later
indicated that the nodule in the liver had regressed to 10.0×9.0
mm. A laparoscopic resection of liver S7 was performed. The
pathological report suggested that the nodule exhibited necrosis
subsequent to the treatment for HCC. A postoperative ACTH level of
69.8 pg/ml confirmed the condition and maintenance therapy with
lenvatinib combined with tislelizumab was continued.
In June 2024, the patient was admitted to the same
hospital presenting with ‘melena accompanied by dizziness, fatigue
and transient aphasia’. Blood routine examination results showed a
hemoglobin level of 70.0 g/l (115.0–170.0 g/l) and the fecal occult
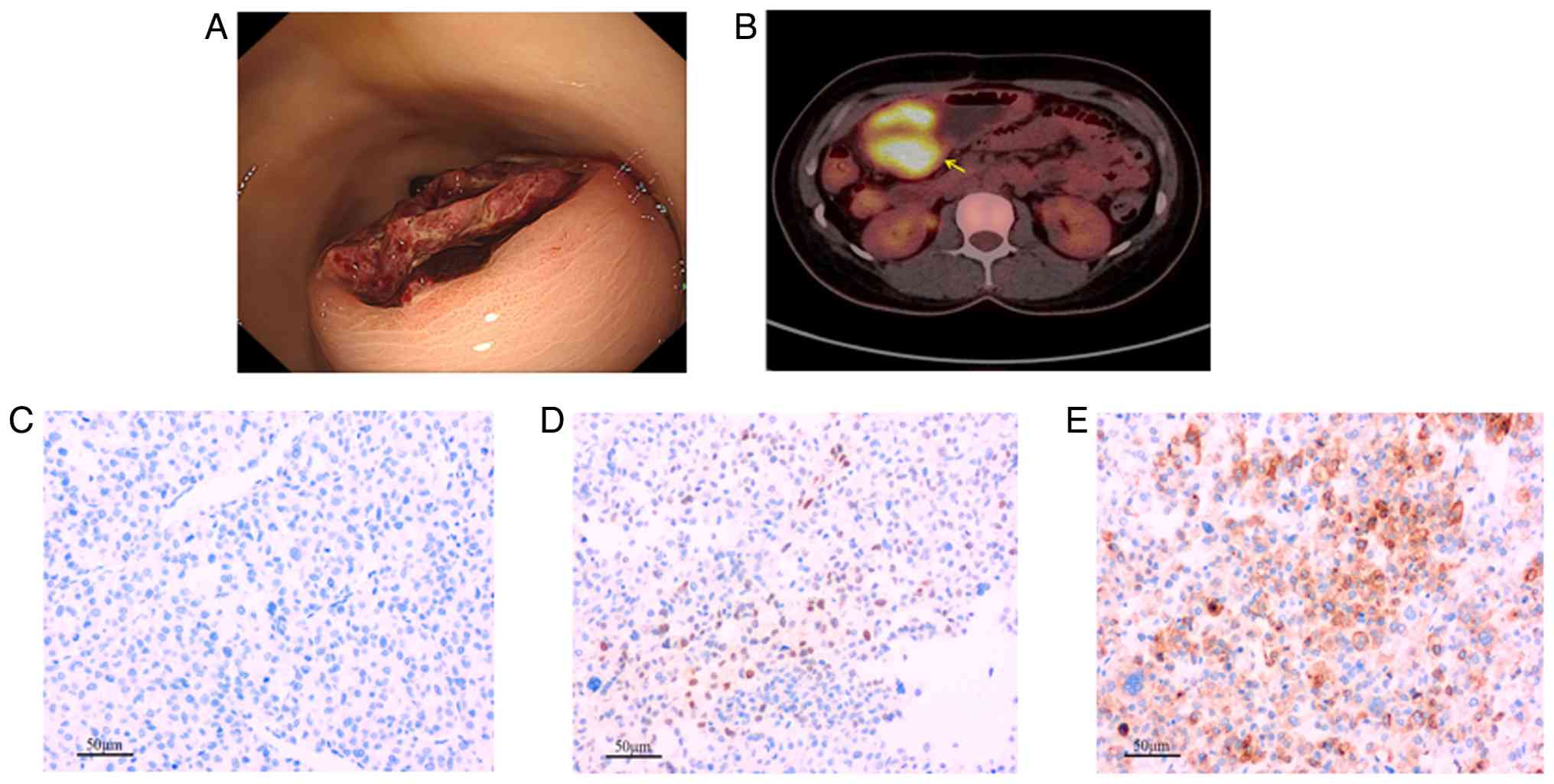
blood test was positive. Gastroscopy revealed a 2.5-cm submucosal
elevation at the greater curvature of the gastric antrum, with
ulceration and bleeding at the apex (Fig. 3A). Abdominal CT showed thickening of
the gastric antrum wall ≤21.0 mm. 18F-fluorodeoxyglucose
(18F-FDG) positron emission tomography/CT (PET/CT)
imaging indicated increased 18F-FDG uptake of the
gastric antrum mass (Fig. 3B); no
other abnormalities were detected throughout the body. After blood
transfusion and acid-suppressing and gastric-protecting therapies,
the symptoms of the patient were alleviated. In July 2024, the
patient underwent a distal gastrectomy with gastrojejunostomy.
Based on gastroscopy findings, 18F-FDG PET/CT imaging
results and pathological analysis of the gastric antral tumor
(Fig. 3), the final diagnosis was
metastatic hepatocellular carcinoma. The tumor was located in the
whole layer of the gastric wall, without penetration of the serosa
layer and the resection margins were negative. IHC analysis showed
hepatocyte (−) (Fig. 3C), Arg-1 (+)
(Fig. 3D) and GPC3 (+) (Fig. 3E). All tissue samples were fixed in
4% formaldehyde, embedded in paraffin and sectioned. Histological
assessment: Tissue samples were fixed in 4% formaldehyde at room
temperature for 24 h, then embedded in paraffin. Sections of 4 µm
thickness were cut and stained with H&E according to standard
protocols. Briefly, sections were deparaffinized, rehydrated,
stained with hematoxylin for 5 min at room temperature, washed,
stained with eosin for 2 min at room temperature, dehydrated, and
mounted. Images were captured using a light microscope (BX53;
Olympus Corp.) at ×200 magnification; scale bars are indicated in
the figure legends.
IHC analysis: IHC was performed on 4-µm paraffin
sections using a Roche Ventana Benchmark XT automated staining
system (Roche Diagnostics) following the manufacturer's
instructions. The staining protocol included deparaffinization,
antigen retrieval (CCI solution; Roche Diagnostics) and blocking of
endogenous peroxidase. Sections were incubated with primary
antibodies against HepPar-1 (cat. no. ZM-0133; 1:100 dilution),
GPC3 (cat. no. ZM-0356; 1:200 dilution), Arg-1 (cat. no. ZM-0235;
1:200 dilution) and Ki-67 (ZM-0166; 1:150 dilution). All antibodies
were obtained from Shanghai Zhongshan Jinqiao Biotechnology Co.,
Ltd. All primary antibodies were incubated at 37°C for 32 min.
Detection was performed using the UltraView Universal DAB Detection
Kit (Roche Diagnostics) according to the manufacturer's protocol,
which includes a secondary antibody (goat anti-mouse/rabbit IgG
conjugated with horseradish peroxidase). The sections were
counterstained with hematoxylin, dehydrated and mounted.
Appropriate positive and negative controls were included. Images
were captured with a light microscope (Olympus BX53) at ×200
magnification.
After 2 months of recovery, the patient was switched
to the second-line treatment for liver cancer: Apatinib 750 mg once
a day, combined with tislelizumab. As of the last follow-up in July
2025, there was no evidence indicating recurrence or
metastasis.
Discussion
There are no significant differences in age, sex,
preoperative AFP level, liver cirrhosis and clinical symptoms
between patients with PCCCL and those with common HCC (CHCC)
(6). However, the incidence of
hepatitis C infection in patients with PCCCL is higher compared
with that of patients with CHCC. The clinical diagnosis mainly
relies on pathological examination: The cancer cells of PCCCL are
characterized by cytoplasm rich in glycogen or fat, a decrease in
the number and volume of organelles and mild nuclear atypia.
Histological examination reveals a moderate degree of
differentiation (7). Therefore,
some literature reports indicate that the prognosis of this disease
is generally favorable compared with that of CHCC (8). Although rare, gastric metastasis of
HCC has been previously documented in the literature (9).
The contrast-enhanced CT images of the lesion in the
present case report demonstrate moderate heterogeneous enhancement
during the arterial phase, accompanied by multiple small arteries
traversing the lesion. In the venous and delayed phases, the
enhancement diminishes, presenting a typical ‘fast in, fast out’
enhancement pattern similar to that of CHCC. However, CHCC
typically exhibits less and milder fatty degeneration.
Additionally, the relatively distinct mass boundary and
pseudocapsule shadow differ from the infiltrative growth and
indistinct boundaries commonly observed in CHCC of the same size or
grade (10). PET/CT has limited
sensitivity in the initial diagnosis of HCC. Notably,
18F-FDG PET/CT does not offer superior detection
efficiency for small intrahepatic metastases compared to
contrast-enhanced MRI. Therefore, 18F-FDG PET/CT was not
performed prior to the initial surgery. During the histological
section examination, it was found that clear cells accounted for
>50%. Furthermore, immunohistochemistry results indicated that
both the liver-specific antigens hepatocyte and glypican-3 were
positive. Considering the absence of abnormalities in other organs
detected by imaging, the final diagnosis was confirmed as PCCCL.
During the advanced stage of the tumor, the clinical symptoms of
the patient lacked specificity. However, the lesion pattern
observed by gastroscopy is different from the typical ulcerative or
infiltrative growth patterns of primary gastric cancer. By
contrast, on enhanced CT scans, the enhancement degree of the
gastric antrum lesions was relatively lower compared with that of
the primary liver lesions and there was no fat component. This
reflected the fundamental difference in tumor cell growth in
different microenvironments. To the best of our knowledge, at
present, the mechanisms of gastric metastasis of PCCCL remain to be
elucidated. It is speculated that renal clear cell carcinoma, which
often has distant metastases, may be associated with the abundance
of tumor cells, feeding arteries and MVI. Arg-1 exhibits high
specificity for HCC; its positivity in the metastasis would
strongly confirm a hepatic origin (2). Glypican-3 was focally positive in the
gastric metastases, which supported the hepatocyte origin (3). Secondly, hepatocyte (+) in the primary
lesion suggests improved differentiation of hepatocytes, while
hepatocyte (−) in the metastatic lesion may indicate that the
degree of differentiation of metastatic tumor cells had decreased
or that they had dedifferentiated, and certain liver phenotypes had
been lost (11). This corresponds
to the features of the metastatic lesion on 18F-FDG
PET/CT imaging (SUVmax=11.3) (12).
As of present, the treatment protocol for PCCCL
remains grounded in the general treatment principles of HCC. The
patient demonstrated a stable overall health status, with all liver
function indices falling within the normal range. Integrating the
findings of preoperative imaging examinations, radical resection
was identified as the most suitable treatment approach. For the
subsequent two surgeries, both the timing and methodology were
strictly aligned with current treatment principles. For resectable
lesions, including the primary tumor, intrahepatic recurrence foci
and symptomatic solitary gastric metastases, proactive surgical
intervention is pivotal in controlling local lesions and attaining
survival benefits (13). In recent
years, clinical investigations into targeted therapy, immunotherapy
(both as monotherapy and combination regimens) for advanced HCC
have yielded promising results regarding efficacy and safety
profiles, thereby informing the selection of first-line therapeutic
strategies. For instance, the REFLECT trial demonstrated that
lenvatinib was non-inferior to sorafenib in terms of overall
survival (OS) among patients with advanced HCC, solidifying the
role of lenvatinib as a first-line treatment option for this
patient population (14). A
separate retrospective study further indicated that the combination
of toripalimab, lenvatinib and hepatic artery infusion chemotherapy
markedly improved progression-free survival, OS and tumor response
rates when compared with lenvatinib monotherapy in the management
of advanced HCC (15).
Consequently, the patient in the present case report was
administered the potent combination regimen of lenvatinib plus a
PD-1 inhibitor, which aligns with the recommendations outlined in
clinical practice guidelines. However, following the completion of
six treatment cycles, CT evaluation revealed no notable reduction
in the metastatic lesion located in S7 of the liver. In contrast
with toripalimab, tislelizumab features structural modifications to
its Fc region, which mitigate macrophage-mediated T-cell exhaustion
and theoretically enhance anti-tumor immune responses (16). As evidenced by subsequent imaging
studies, switching to the combination therapy of tislelizumab and
lenvatinib resulted in a reduction in the liver S7 lesion, thereby
facilitating subsequent surgical intervention. Nevertheless, the
patient experienced exacerbation of irAEs, necessitating the
temporary suspension of immunotherapy. After a 3-month interval
during which the irAEs subsided, the first-line treatment regimen
was reinitiated.
Given the heterogeneity of HCC, tumor progression
frequently ensues following first-line systemic therapy. The low
incidence of PCCCL has also constrained the clinical synthesis of
therapeutic strategies for PCCCL metastasis. The phenomenon of
‘hyperprogressive disease’ was first defined in 2017 by Champiat
et al (17). It refers to
disease progression that meets the Response Evaluation Criteria in
Solid Tumors at the first imaging evaluation, along with at least a
two-fold increase in the tumor growth rate. This phenomenon may be
associated with various factors, including specific genetic
alterations and immune microenvironment dysregulation. In the
present case, however, a young female patient developed gastric
antral metastasis 8 months after resuming immunotherapy following a
3-month treatment pause. It is reasonable to consider this
progression part of the natural disease course. Apatinib exhibits
high specificity for VEGFR-2 binding without antagonizing VEGFR-3,
thereby failing to impede the activation of T cells stimulated by
immune checkpoint inhibitors and preserving the lymphoid-immune
system. Apatinib has been approved for patients with HCC who have
experienced failure of or intolerance to at least one prior line of
systemic antineoplastic therapy (18). Subsequent to the re-failure of PD-1
inhibitor therapy, the patient demonstrated a favorable response to
the combination regimen of apatinib and tislelizumab, with no
evidence of tumor recurrence observed during follow-up to the
present date.
Acknowledgements
Not applicable.
Funding
Funding: No funding was received.
Availability of data and materials
The data generated in the present study may be
requested from the corresponding author.
Authors' contributions
LZ contributed to the acquisition and analysis of
data, was primarily responsible for collecting the patient's
complete clinical data, including imaging, pathology, and follow-up
records, participated in the interpretation of data regarding the
treatment response and outcome and drafted the initial version of
the case report and methods sections of the manuscript. TT
contributed to the conception and design of this case study,
contributed to the analysis and interpretation of the pathological
findings, particularly regarding the rare diagnosis of primary
clear-cell carcinoma and critically revised the manuscript for
important intellectual content, specifically the discussion
section. QL and LW conceived the concept of the article and
critically revised the manuscript. LZ and QL confirm the
authenticity of all raw data. All authors have read and agreed to
the published version of the manuscript.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
The patient provided written informed consent for
the publication of her case details, including clinical data and
images.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Dudani S, de Velasco G, Wells JC, Gan CL,
Donskov F, Porta C, Fraccon A, Pasini F, Lee JL, Hansen A, et al:
Evaluation of clear cell, papillary, and chromophobe renal cell
carcinoma metastasis sites and association with survival. JAMA Netw
Open. 4:e20218692021. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Yan BC, Gong C, Song J, Krausz T,
Tretiakova M, Hyjek E, Al-Ahmadie H, Alves V, Xiao SY, Anders RA
and Hart JA: Arginase-1: A new immunohistochemical marker of
hepatocytes and hepatocellular neoplasms. Am J Surg Pathol.
34:1147–1154. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Ligato S, Mandich D and Cartun RW: Utility
of glypican-3 in differentiating hepatocellular carcinoma from
other primary and metastatic lesions in FNA of the liver: an
immunocytochemical study. Mod Pathol. 21:626–631. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Calderaro J, Couchy G, Imbeaud S, Amaddeo
G, Letouzé E, Blanc JF, Laurent C, Hajji Y, Azoulay D, Bioulac-Sage
P, et al: Histological subtypes of hepatocellular carcinoma are
related to gene mutations and molecular tumour classification. J
Hepatol. 67:727–738. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Donne R and Lujambio A: The liver cancer
immune microenvironment: Therapeutic implications for
hepatocellular carcinoma. Hepatology. 77:1773–1796. 2023.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Liu B, Li J, Yang X, Chen F, Zhang Y and
Li H: Diagnosis of primary clear cell carcinoma of the liver based
on Faster region-based convolutional neural network. Chin Med J
(Engl). 136:2706–2711. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Liu Z, Ma W, Li H and Li Q:
Clinicopathological and prognostic features of primary clear cell
carcinoma of the liver. Hepatol Res. 38:291–299. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Li ZY, Bi XY, Zhao JJ, Zhao H, Zhou JG,
Huang Z, Cai JQ and Zheng XC: Clinicopathological and prognostic
analysis of primary clear cell carcinoma of the liver. Zhonghua
Zhong Liu Za Zhi (Chinese). 35:140–143. 2013.PubMed/NCBI
|
|
9
|
Chen WT, Huang SK, Chang ML and Liaw YF:
Hepatocellular carcinoma with gastric metastasis mimicking a 4 cm
gastrointestinal stromal tumor after a 3-year disease-free
interval. J Clin Transl Hepatol. 12:218–221. 2024.PubMed/NCBI
|
|
10
|
Wang H, Tan B, Zhao B, Gong G and Xu Z: CT
findings of primary clear cell carcinoma of liver: With analysis of
19 cases and review of the literature. Abdom Imaging. 39:736–743.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Hanif R and Mansoor S: Hep par-1: A novel
immunohistochemical marker for differentiating hepatocellular
carcinoma from metastatic carcinoma. J Coll Physicians Surg Pak.
24:186–189. 2014.PubMed/NCBI
|
|
12
|
Yao Y, Civelek AC and Li XF: The
application of 18F-FDG PET/CT imaging for human hepatocellular
carcinoma: A narrative review. Quant Imaging Med Surg.
13:6268–6279. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Yang X, Yang C, Zhang S, Geng H, Zhu AX,
Bernards R, Qin W, Fan J, Wang C and Gao Q: Precision treatment in
advanced hepatocellular carcinoma. Cancer Cell. 42:180–197. 2024.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Kudo M, Finn RS, Qin S, Han KH, Ikeda K,
Piscaglia F, Baron A, Park JW, Han G, Jassem J, et al: Lenvatinib
versus sorafenib in first-line treatment of patients with
unresectable hepatocellular carcinoma: A randomised phase 3
non-inferiority trial. Lancet. 391:1163–1173. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Li Y, Wang D, Zhang F, Zheng X, Song Y,
Ran Y and Cai X: Hepatic arterial infusion chemotherapy combined
with lenvatinib and toripalimab for large hepatocellular carcinoma
(>10 cm) with major portal vein tumor thrombosis: A multicenter
propensity score matching analysis. Front Immunol. 16:16381732025.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Zhang T, Song X, Xu L, Ma J, Zhang Y, Gong
W, Zhang Y, Zhou X, Wang Z, Wang Y, et al: The binding of an
anti-PD-1 antibody to FcγRI has a profound impact on its biological
functions. Cancer Immunol Immunother. 67:1079–1090. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Champiat S, Dercle L, Ammari S, Massard C,
Hollebecque A, Postel-Vinay S, Chaput N, Eggermont A, Marabelle A,
Soria JC and Ferté C: Hyperprogressive disease is a new pattern of
progression in cancer patients treated by anti-PD-1/PD-L1. Clin
Cancer Res. 23:1920–1928. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Yu J, Yu J, Chen Y, Yang Y and Yi P: PD-1
inhibitors improve the efficacy of transcatheter arterial
chemoembolization combined with apatinib in advanced hepatocellular
carcinoma: A meta-analysis and trial sequential analysis. BMC
Cancer. 25:5642025. View Article : Google Scholar : PubMed/NCBI
|