Introduction
Synovial sarcoma (SS), a relatively rare malignant
soft tissue tumor, has been progressively understood since its
first description in 1936 (1,2).
Although it most frequently develops in the extremities, cases with
a primary thoracic origin, referred to as primary thoracic SS
(PTSS), are exceedingly rare, with only a few incidences reported
in the literature (3–6). PTSS often shows nonspecific clinical
features, with common symptoms such as cough, chest pain and
shortness of breath, which could be easily misdiagnosed as
frequently reported thoracic diseases, such as lung cancer or
benign lung conditions.
PTSS poses major diagnostic challenges because of
its rarity and its ability to mimic other malignancies. Its
diagnosis often relies on the combination of imaging modalities,
histopathological examination and molecular analysis. A key
diagnostic feature of SS is the chromosomal translocation
t(X;18)(p11.2;q11.2), detectable by techniques such as fluorescence
in situ hybridization (FISH) and reverse
transcription-polymerase chain reaction. However, in certain cases,
the preoperative diagnosis can be difficult, as noted in the
present case, where the tumor was initially misdiagnosed as a
solitary fibrous tumor (SFT) based on histological and
immunohistochemical (IHC) findings.
The origin of PTSS within the thoracic cavity
remains controversial, with certain reports suggesting the
involvement of the pleura, chest wall, mediastinum or a combination
of these structures (7). Accurate
localization of the tumor's primary site is crucial for appropriate
staging and treatment planning; however, in numerous cases,
including the present case, the precise origin cannot be
definitively determined (8).
Given the limited number of PTSS cases and the lack
of high-level evidence for standardized treatment protocols,
therapeutic strategies often involve a combination of surgery,
chemotherapy and radiation therapy customized according to the
patient's health condition. However, the prognosis for PTSS remains
poor, with a high risk of recurrence and metastasis (9).
The current study presents the case of a patient
diagnosed with PTSS, who, despite receiving surgical tumor
resection and subsequent adjuvant chemotherapy, died due to
widespread metastasis and associated complications.
Case report
A 45-year-old male presented to The Second Forestry
Hospital of Heilongjiang Province (Yichun, China) in April 2021
with persistent chest pain and dyspnea. The patient's medical
history was notable for chronic smoking, with no other significant
comorbidities. On initial examination, the patient appeared normal
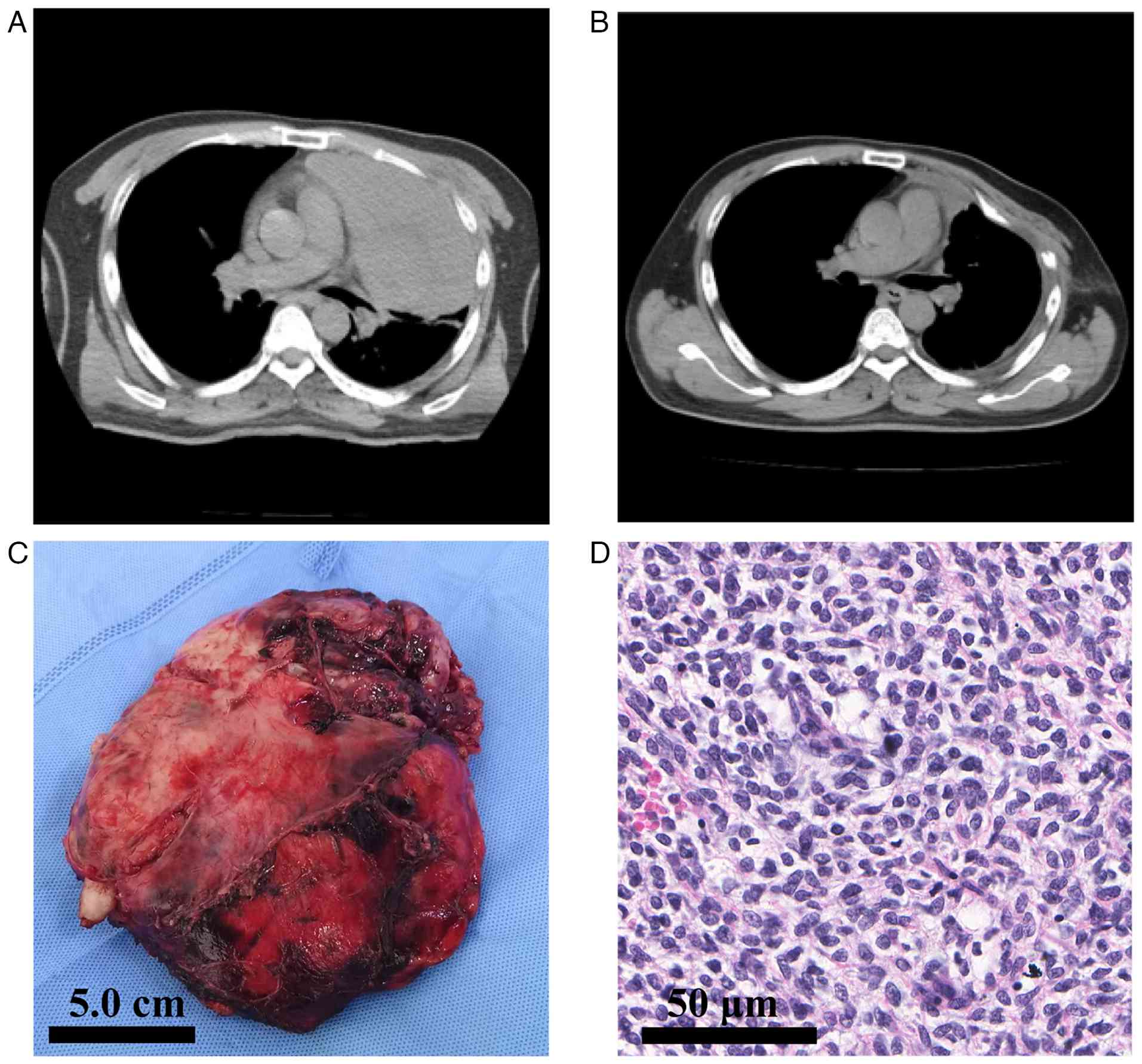
but exhibited mild respiratory distress (10). Chest X-ray revealed a large mass in
the left hemithorax. Additional imaging with a computed tomography
(CT) scan confirmed the presence of a tumor mass measuring
18.5×16.4×9.3 cm, localized to the left thoracic cavity, with broad
contact to the parietal pleura but no clear chest wall invasion
(Fig. 1A). The preoperative staging
work-up included contrast-enhanced chest CT, non-contrast head CT
and abdominal ultrasonography. Based on these examinations, no
evidence of distant metastasis was detected. A positron emission
tomography-CT scan, which could have provided a more comprehensive
metastatic survey, was not performed due to the patient's financial
constraints. All laboratory tests, including complete blood count;
renal, liver and coagulation profiles; and tumor markers (cancer
antigen 125, cancer antigen 153 and carcinoembryonic antigen), were
within their normal ranges.
CT-guided percutaneous biopsy was performed. The
specimens were fixed in 10% neutral buffered formalin, embedded in
paraffin and cut at 4-µm thickness. Histological examination
(H&E staining; Fuzhou Maixin Biotechnology Development Co.,
Ltd.) was performed according to standard clinical protocols and
revealed randomly arranged spindle-like cells with indistinct cell
borders. All immunohistochemical staining in this case was
performed on an automated staining system (Benchmark XT; Roche
Diagnostics) strictly according to the manufacturer's protocols.
Primary antibodies were obtained from Fuzhou Maixin Biotechnology
Development Co., Ltd. as ready-to-use reagents (prediluted 1:100).
The initial panel focused on the most common differential diagnosis
and showed positive staining for vimentin (cat. no. MAB-0735),
cluster of differentiation 34 (CD34; cat. no. MAR-1076) and B-cell
lymphoma 2 (BCL-2; cat. no. MAB-0711) (Fig. 2A and B). Because this profile
typically represents SFT, additional staining was not initially
performed, as the findings were deemed sufficient for a
preoperative diagnosis. Following a multidisciplinary discussion,
the team decided to proceed with direct surgical intervention on
the basis of the pathological findings (11,12).
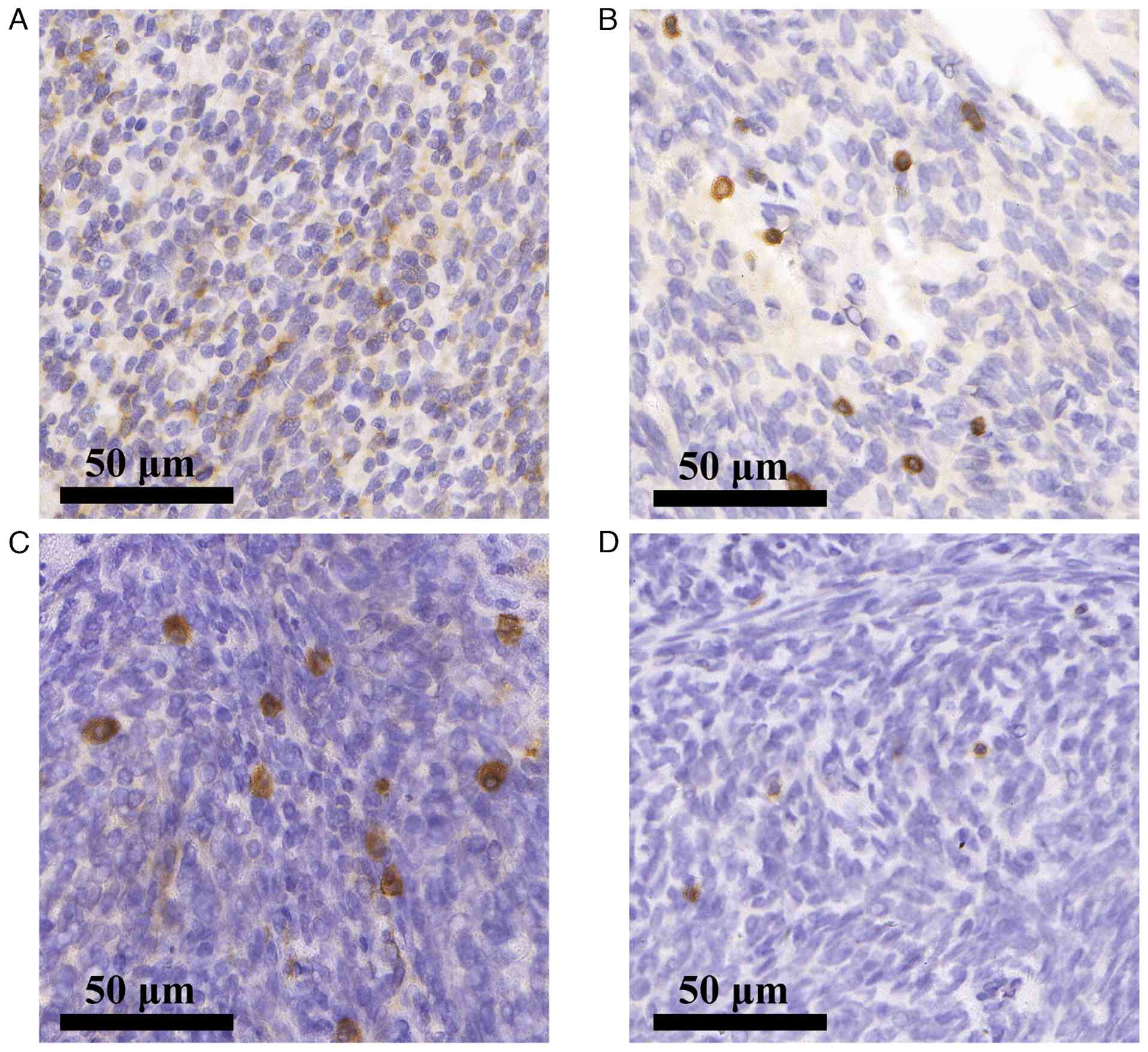
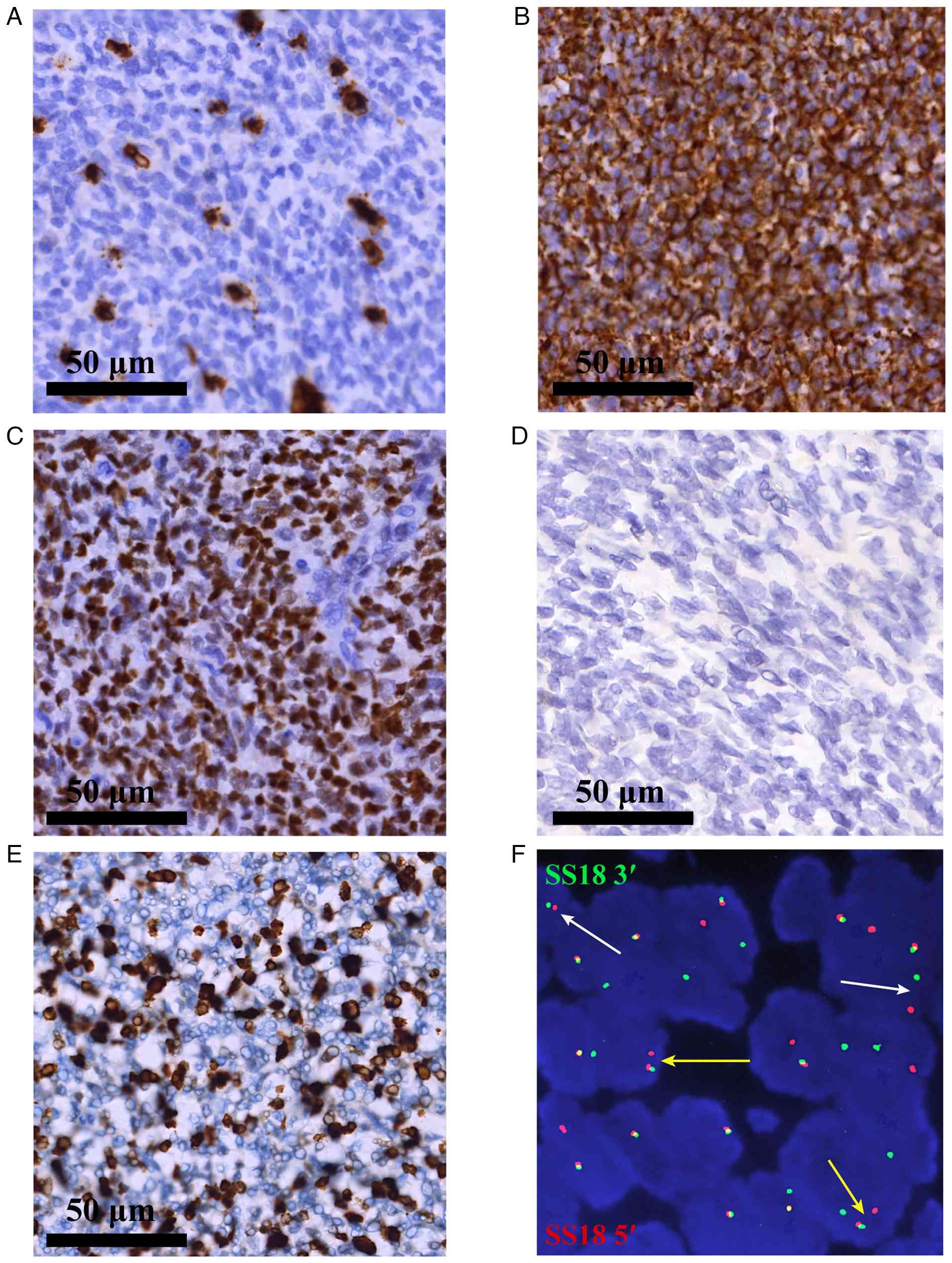 | Figure 2.IHC findings and confirmatory
molecular cytogenetic profiling. (A and B) Initial IHC staining
(original magnification, ×400; scale bar, 50 µm) of the biopsy
specimen shows positive staining for (A) CD34 and (B) BCL-2, a
profile that initially raised consideration for solitary fibrous
tumor. IHC and molecular cytogenetic profiling confirming the
diagnosis of monophasic synovial sarcoma. (C-E) IHC staining
(original magnification, ×400; scale bar, 50 µm) shows (C) strong
and diffuse nuclear positivity for transducin-like enhancer of
split 1, supporting the diagnosis; (D) negative nuclear staining
for STAT6, effectively excluding solitary fibrous tumor; and (E) a
high Ki-67 proliferation index of ~30% (scale bar, 50 µm). (F) SS18
gene fluorescence in situ hybridization analysis using
break-apart probes (original magnification, ×1,000) reveals
multiple positive signal patterns indicative of SS18 gene
rearrangement. The 5′ and 3′ends of the SS18 gene are
labeled with red and green fluorescence, respectively. One pattern
shows an abnormal signal (a single red SS18 5′ signal accompanied
by normal red-green fusion signals, indicated by yellow
arrowheads), while another shows a typical break-apart pattern
(separated green SS18 3′ and red SS18 5′signals, indicated by white
arrowheads). IHC, immunohistochemical. |
Because of the large tumor size, the patient
underwent open thoracotomy. The procedure, which lasted for 4 h
with an estimated blood loss of 600 ml, revealed a large soft
tissue mass occupying the thoracic cavity. The tumor's epicenter
was located on the parietal pleura, with firm adhesions and direct
invasion into the adjacent mediastinum and chest wall. Scattered
satellite lesions were also noted on the lung surface. Because the
preoperative diagnosis of SFT suggested a benign nature of the
mass, macroscopic complete resection of the tumor mass was planned.
Therefore, intraoperative frozen section analysis was not
performed, and an en bloc resection of the main tumor mass,
along with the involved mediastinal pleura, chest wall and all
identifiable satellite nodules, was conducted. Macroscopically, the
resection appeared complete. However, postoperative pathological
examination of the entire specimen revealed deep tumor infiltration
and the surgical margins were microscopically positive (R1
resection) at the mediastinal involvement site. The R1 resection,
with positive surgical margins, was a significant factor
influencing the high risk of recurrence and metastasis in this
case.
Gross examination of the resected specimen (Fig. 1C) revealed a well-circumscribed, but
unencapsulated, greyish-white soft tissue mass measuring
18.5×16.4×9.3 cm in the greatest dimension. The cut surface was
solid and tan-white, with focal hemorrhagic areas and ~20%
necrosis. Histological examination (Fig. 1D) demonstrated a hypercellular tumor
composed of relatively uniform, spindle-shaped cells arranged in
intersecting fascicles. The tumor cells exhibited oval nuclei,
scant cytoplasm and predominantly monophasic fibrous morphology.
Mitotic activity, assessed on H&E-stained sections, was
elevated, with an average of 12 mitoses per 10 high-power fields.
An extensive IHC panel was performed as described above, using
ready-to-use primary antibodies (prediluted 1:100; Fuzhou Maixin
Biotechnology Development Co., Ltd.), which revealed diffuse
nuclear positivity for transducin-like enhancer of split 1 (TLE1;
cat. no. MAB-0686) (Fig. 2C) and
membranous positivity for CD117 (c-kit; cat. no. Kit-0029) as well
as focal immunoreactivity for epithelial membrane antigen (EMA;
cat. no. MAB-1101) and low-molecular-weight cytokeratins [CK (cat.
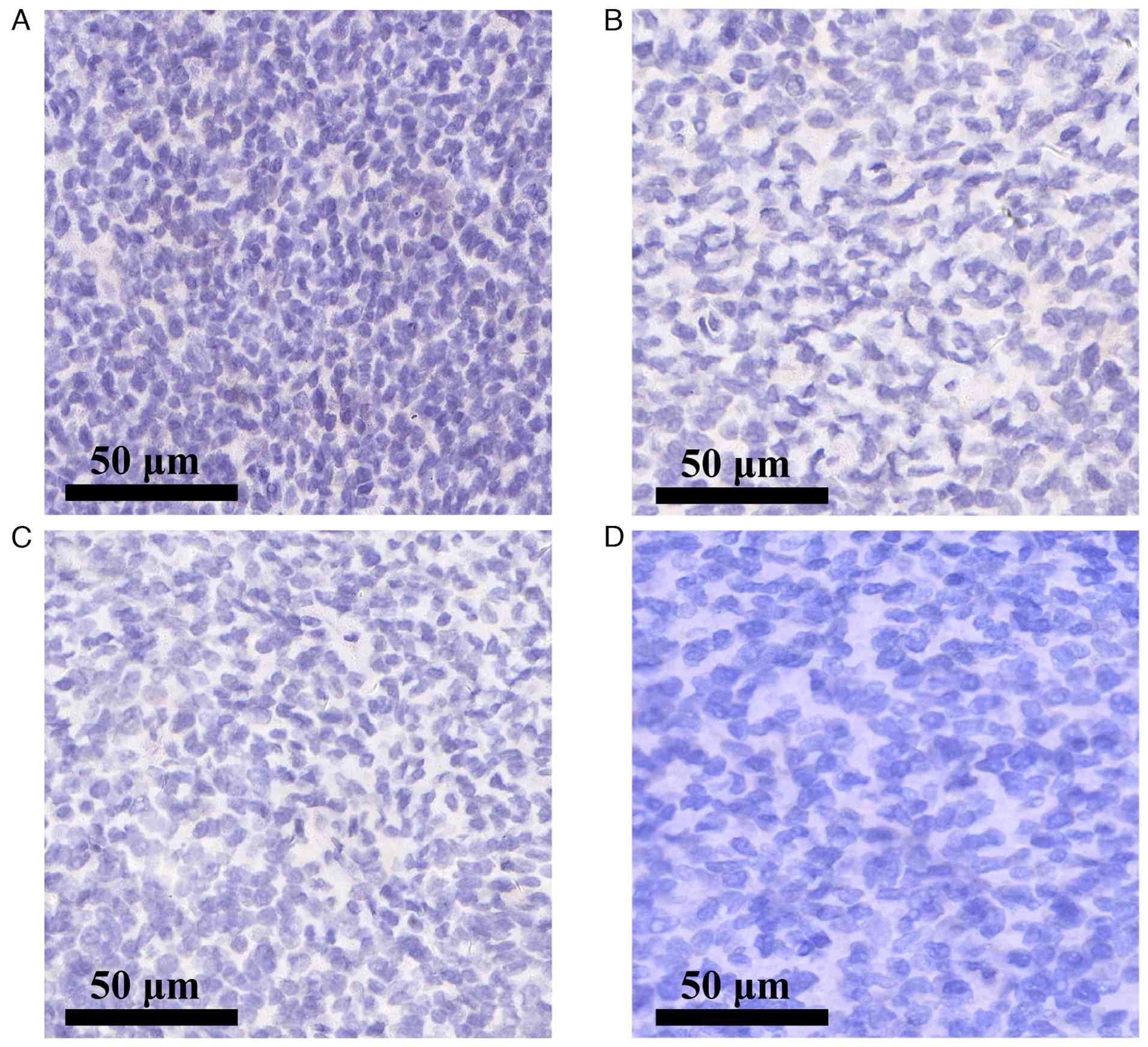
no. RAB-0050) and CK19 (cat. no. MAB-0829)] (Fig. 3A-D). The tumor was entirely negative
for STAT6 (nuclear; cat. no. RMA-1066) (Fig. 2D), which effectively ruled out the
diagnosis of SFT. A mesothelial marker panel [Calretinin (cat. no.
MAB-0716), Wilms tumor-1 (cat. no. RMA-1151), D2-40 (cat. no.
MAB-0567) and CK5/6 (cat. no. RMA-1144)] also yielded a negative
result, excluding the diagnosis of sarcomatoid mesothelioma
(Fig. 4A-D). The Ki-67 (cat. no.
RMA-0542) proliferative index was ~30%, indicating a moderately
high rate of cell proliferation (Fig.
2E) (13,14). The surgical margins were positive
for tumor cells (R1 resection), with the closest margin measuring
<1 mm at the mediastinal aspect. The diagnosis was confirmed
cytogenetically by fluorescence in situ hybridization (FISH)
using the Vysis SS18 Break Apart FISH Probe Kit (Abbott Molecular),
performed strictly according to the manufacturer's protocols. The
analysis revealed a rearrangement of the SS18 (SYT) gene locus in
80% of the nuclei assessed, and thus, fusion-specific SS18-SSX IHC
was not performed (Fig. 2F).
Pathological staging of the tumor was T4G3NxM0, which is classified
as stage III according to the American Joint Committee on Cancer
staging system (15,16).
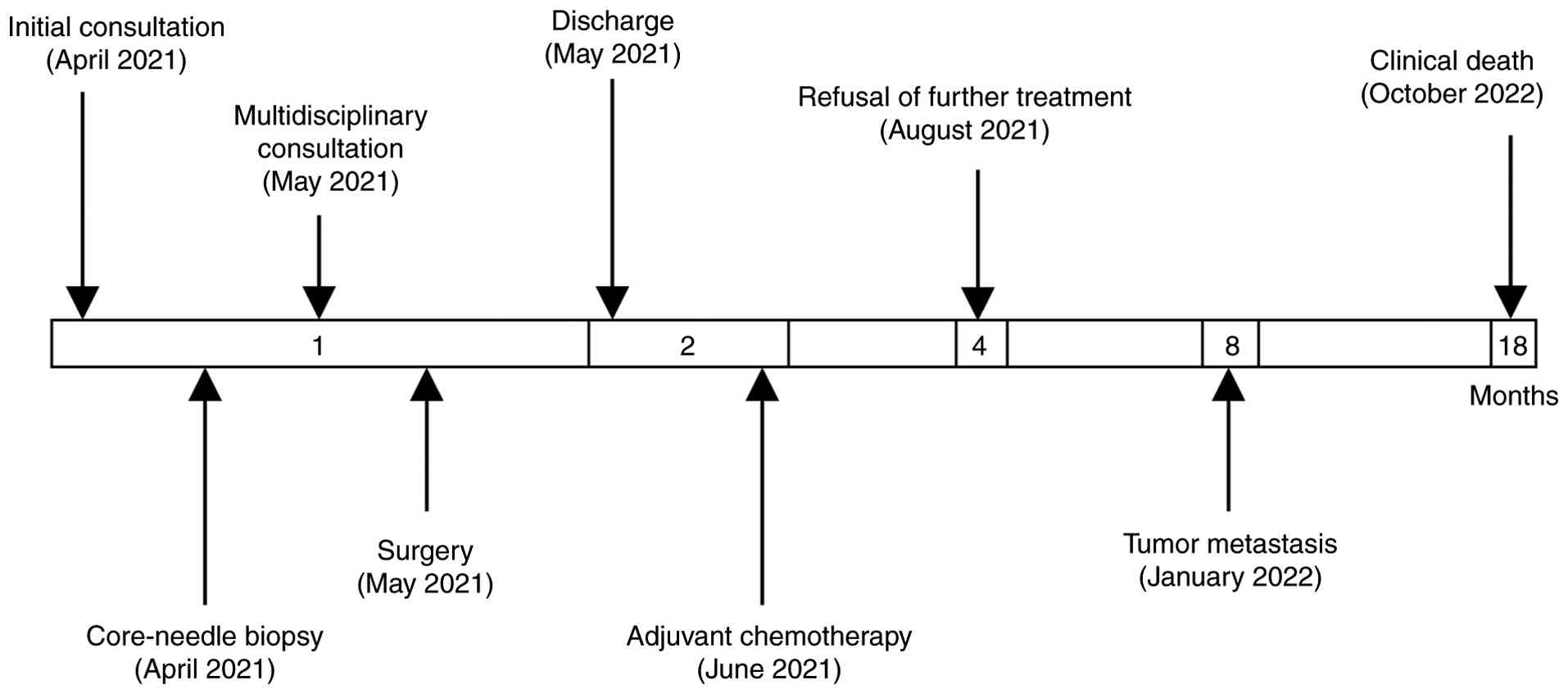
The patient's postoperative recovery was uneventful.
A follow-up chest CT scan demonstrated improved expansion of the
left lung (Fig. 1B), and the chest
tube was removed on postoperative day 5 once drainage decreased to
<100 ml per day. In light of the high-risk pathological features
(M0 by preoperative imaging, T4G3NxM0 and stage III disease with R1
resection), the multidisciplinary tumor board recommended a
comprehensive adjuvant strategy consisting of radiotherapy to the
tumor bed followed by systemic chemotherapy (17). Chemotherapy was commenced on
postoperative day 21 with a 6-cycle regimen of doxorubicin (70
mg/m2) and nedaplatin (50 mg/m2). However,
the patient discontinued treatment after 3 cycles due to grade 2
nausea and fatigue, exacerbated by personal and financial
constraints. The total cost of the initial surgical resection was
contained under $3,000, reflecting these substantial financial
limitations. The rationale for adjuvant radiotherapy was thoroughly
explained to the patient to mitigate the high risk of local
recurrence; however, despite counseling, the patient declined this
intervention. At 8 months after surgery, the patient was
re-admitted with persistent cough and cachexia; repeat CT imaging
revealed multiple bilateral pulmonary nodules, indicative of
widespread metastatic progression (data not shown). The accumulated
financial burden was a major factor contributing to the patient's
reluctance towards further therapies. The multidisciplinary team
discussed evidence-based salvage options, including palliative
radiotherapy, second-line systemic agents (such as ifosfamide,
trabectedin or pazopanib) and best supportive care. After
considering the limited prognosis and potential treatment-related
burdens, the patient elected to forgo all further anticancer
therapy. The patient died 18 months postoperation. The overall
clinical course is summarized in Fig.
5.
Discussion
PTSS is a rare and diagnostically challenging
malignancy (10), typically
presenting with nonspecific symptoms that complicate and often
delay accurate diagnosis. To summarize recent findings, a
comprehensive literature search of the PubMed (https://pubmed.ncbi.nlm.nih.gov/), Embase
(https://www.embase.com/) and Web of Science
(https://www.webofscience.com/) databases
from 2020 to the present was conducted. The search strategy
utilized the following keywords: ‘mediastinal synovial sarcoma’,
‘chest wall synovial sarcoma’, ‘thoracic synovial sarcoma’, and
‘pulmonary synovial sarcoma’. As summarized in Table I (11,18–37),
which shows a comprehensive literature review from 2020 to the
present, this entity primarily affects young to middle-aged adults
and often appears as sizable tumors at the time of identification.
A key issue related to its characterization is the controversy over
its primary anatomic site-whether it originates from the pleura,
chest wall or mediastinum. This diagnostic uncertainty is
compounded by the frequent finding of extensive tumor infiltration,
which can obscure the true anatomic epicenter. Consequently,
attempting to define the tumor origin based solely on proximity to
surrounding structures is an oversimplification that can be
misleading (10). In the present
case, preoperative imaging revealed the mass to have its broadest
and most intimate contact with the parietal pleura.
Intraoperatively, while the tumor showed adhesions to both the
parietal pleura and mediastinum, the most extensive and firmest
attachment was observed to the parietal pleura. Therefore,
synthesizing imaging and surgical findings, the parietal pleura is
considered the most probable site of origin. However, this
assessment is made with caution due to notable inconsistencies: A
primary tumor of the parietal pleura would typically be expected to
show direct infiltration into the adjacent chest wall musculature,
which was not observed. Similarly, a primary mediastinal origin
fails to adequately explain the overall growth pattern and adhesion
findings. Given these unresolved questions and in the interest of
utmost pathological and clinical rigor, it may be concluded that,
while the parietal pleura is the most likely origin, a definitive
anatomic epicenter cannot be assigned. Consequently, the tumor is
pragmatically classified as a PTSS arising within the thoracic
cavity, consistent with the literature describing cases with
widespread involvement where the precise origin remains
indeterminate (10,11,23,25,33,38–41).
 | Table I.Summary of the clinical
characteristics of primary thoracic synovial sarcoma from selected
literature. |
Table I.
Summary of the clinical
characteristics of primary thoracic synovial sarcoma from selected
literature.
| First author,
year | Number of
cases | Median age (range),
years | Gender (M/F) | Common
symptoms | Origin | Median tumor size,
cm | Primary
treatment | 5-year
survival | (Refs.) |
|---|
| Pieropan, 2022 | 20 | 41 (28–54) | 13/7 | Chest pain: 11
(55%), Dyspnea: 5 (25%), Hemoptysis: 3 (15%), Cough: 3 (15%),
Dysphagia: 2 (10%), Chest bulge: 1 (5%), Arm swelling: 1 (5%);
Incidental finding: 2 (10%) | Thoracic
Cavity | 11.0 (IQR:
8–15) | Surgery: 20 (100%),
Chemotherapy: 2 (10%), Radiotherapy: 11 (55%) | 22% (OS) 1 | (11) |
| Abdulghaffar,
2020 | 1 | 39 | M | Chest pain | Chest wall | 17.5 | Surgery +
chemotherapy | - | (18) |
| Wan, 2020 | 1 | 67 | M | Incidental
finding | Pleura | 3.8 | Surgery +
chemotherapy | - | (19) |
| Choudhary,
2020 | 1 | 71 | M | Dry cough,
right-sided chest pain, dyspnea | Pleura | - | Chemotherapy | - | (20) |
| Kagawa, 2020 | 1 | 61 | F | Right-sided chest
pain | Lung | 13 | Surgery +
chemoradiotherapy | Death | (21) |
| Kim, 2020 | 1 | 26 | F | Chest pain | Chest wall | 3 | Surgery | - | (22) |
| Khalili, 2021 | 2 | 24, 29 | F | Coughing, shortness
of breath | Pleura | 9.5/- | Surgery ±
chemotherapy | - | (23) |
| Kumar, 2021 | 1 | 26 | M | Dyspnea, cough,
fever, left-sided chest pain | Pleura | - | Palliative
chemotherapy and radiotherapy | - | (24) |
| Saad, 2021 | 1 | 14 | F | Dyspnea | Mediastinum | 1.4 | Not specified | Death | (25) |
| He, 2021 | 13 | 48 (IQR:
27–58) | 7/6 | Chest pain: 5
(38.46%), Coughing: 8 (61.54%), Dyspnea: 6 (46.15%), Blood in
sputum: 3 (23.08%), Yellow sputum: 2 (15.38%), Body examination: 1
(7.69%) | Thoracic
cavity | 6.0 (IQR:
5.5–9.0) | Surgery: 10
(76.92%), Chemotherapy: 10 (76.92%), Radiotherapy: 4 (30.77%),
Immunotherapy: 2 (15.38%), Targeted therapy: 1 (7.69%), Knife
therapy: 1 (7.69%) | Death: 7 (53.85%),
Survival: 6 (46.15%) | (26) |
| Vishnoi, 2021 | 1 | 39 | M | Chest pain | Chest wall | 6 | Surgery +
chemoradiotherapy | Death | (27) |
| Patel, 2022 | 1 | 9 | M | Pneumothorax | Pleura | 1.5 | Surgery | - | (28) |
| Oneglia, 2022 | 1 | 12 | M | Dyspnea, cough,
chest pain, pneumothorax | Lung | <5 | Surgery +
chemoradiotherapy | - | (29) |
| Shilo, 2023 | 1 | 29 | F | Pneumothorax,
cough | Lung | 1.3 | Surgery | - | (30) |
| Wu, 2023 | 1 | 38 | F | Hemoptysis | Pleura | 13.0 | Surgery +
anlotinib | Death | (31) |
| Hozain, 2023 | 1 | 36 | F | Chest pain,
syncope | Mediastinum | 12.5 | Surgery +
chemoradiotherapy | - | (32) |
| Zang, 2023 | 1 | 18 | M | Dry cough,
wheezing | Pleura | 7.8 | Surgery +
chemotherapy | - | (33) |
| Do, 2023 | 1 | 60 | F | Pneumothorax | Pleura | - | Surgery | - | (34) |
| Idrees, 2024 | 1 | 23 | F | Pain, chest wall
mass | Chest wall | 12.0 | Surgery +
chemotherapy | - | (35) |
| Imen, 2024 | 1 | 54 | M | Left-sided chest
pain, dyspnea | Pleura | 6.8 | Surgery | Death | (36) |
| Vega-Gonzalez,
2025 | 1 | 58 | F | Rapid weight loss,
dyspnea | Pleura | - | Chemotherapy | - | (37) |
| Present case,
2025 | 1 | 45 | M | Cough, dyspnea | Thoracic
cavity | 18.5 | Surgery +
chemotherapy | Death | - |
In the present case, the diagnostic challenge of
PTSS was further complicated by the discrepancy between the
findings of preoperative biopsy and postoperative pathological
examination. Initially, the tumor was misdiagnosed as an SFT,
leading to the missed opportunity for initiating neoadjuvant
chemotherapy. The definitive diagnosis of SS was made
postoperatively after identifying the characteristic
t(X;18)(p11.2;q11.2) translocation (15,16).
This underscores the importance of preoperative molecular analysis.
The initial biopsy findings, based on cellular morphology and an
IHC profile of CD34- and BCL-2-positive spindle cells, were highly
suggestive of SFT. At this stage, these findings were considered
sufficient for a preoperative diagnosis, and the rarity of PTSS
lowered the level of clinical suspicion. Consequently, neither
additional IHC staining (such as STAT-6) nor molecular analysis was
performed preoperatively. The clinical treatment approach primarily
focused on obtaining a definitive diagnosis and achieving local
control through surgical resection. This case highlights a critical
diagnostic pitfall: For large or atypical thoracic spindle cell
tumors, a higher index of suspicion for rare sarcomas and broader
preoperative diagnostic panels can help avoid misdiagnosis and
facilitate neoadjuvant treatment planning (42,43).
Complete surgical resection (R0) remains the
cornerstone of PTSS treatment, strongly correlating with improved
survival outcomes (11). However,
in the present case, achieving clear surgical margins was
particularly challenging because of the tumor's extensive
involvement of the mediastinum, chest wall and lung surface. The
tumor had to be bluntly dissected from these structures during
surgery, making it impossible to confirm whether residual tumor
tissue remained. Given the uncertainty regarding margin clearance,
adjuvant therapies such as radiation and chemotherapy are essential
to reduce the risk of recurrence, particularly in cases with a high
likelihood of residual disease (9).
In the broader context of sarcoma management,
ongoing efforts aim to optimize systemic therapy, particularly for
rare subtypes where high-level evidence is scarce (44). For the present patient with PTSS,
the established first-line adjuvant regimen is doxorubicin combined
with ifosfamide (42,43). However, following a
multidisciplinary tumor board (MDT) discussion, a modified regimen
of doxorubicin plus nedaplatin was opted for. This decision was
primarily driven by concerns regarding the potential nephrotoxicity
and neurotoxicity of ifosfamide, with the aim of preserving the
patient's quality of life during recovery. It must be acknowledged
that high-level evidence specifically supporting the efficacy of
nedaplatin in SS is lacking. The consideration of a platinum-based
agent in this case was grounded in evidence suggesting class
activity of platinum drugs in SS. This is supported by studies
reporting the treatment activity of cisplatin in SS (45), a finding corroborated by another
report (46). Furthermore,
cisplatin-containing regimens have demonstrated activity in
advanced thoracic sarcomas (45,46).
Based on this pharmacological rationale and its potentially more
favorable toxicity profile, nedaplatin was selected as an
alternative. This case highlights that the management of complex
thoracic sarcomas often requires individualized decision-making
within an MDT framework, where efficacy, toxicity and
patient-specific factors must be carefully balanced (47).
Despite the benefits of conventional chemotherapy,
there is a critical need to develop novel therapeutic strategies
for treating advanced SS. Immunotherapy, particularly with
programmed cell death-1/programmed death ligand 1 inhibitors, has
shown modest efficacy as a monotherapy, and combination strategies
are currently under investigation (48). Adoptive T-cell therapy, particularly
targeting antigens such as NY-ESO-1 and MAGE-A4, shows promising
potential for treating SS. Clinical trials have demonstrated
positive results, and Food and Drug Administration-approved
therapies have shown remarkable progress in treating advanced
cases. Despite challenges such as tumor heterogeneity, these
therapies are becoming more effective, and future treatments may
improve with more potent strategies, including dual antigen
targeting and checkpoint inhibitors (49). Additionally, targeted therapies such
as ATR inhibitors, which target key kinases in the DNA damage
response pathway, as well as BCL-2 inhibitors, are being actively
explored (46,50). These emerging therapies represent
the priority areas of SS research and provide hope for improving
future treatment outcomes.
In the present case, the pathological staging of the
tumor was T4NxM0G3 (stage III), which typically necessitates
aggressive adjuvant therapy to improve both local control and
overall survival (12,51–53).
Regrettably, because of incomplete adherence to adjuvant therapy,
primarily driven by treatment-related toxicities and, crucially, by
the patient's significant financial constraints, the prognosis was
adversely affected. This case illustrates the gap between optimal
multimodal treatment plans and their feasible execution,
highlighting that socioeconomic factors are critical determinants
of outcome alongside medical strategies. Several key insights can
be drawn from this case: Improving preoperative diagnostic accuracy
is crucial for considering neoadjuvant chemotherapy; early
intervention, ideally at the localized disease stage, may increase
the likelihood of achieving complete resection; and adherence to a
full course of adjuvant therapies, which may require addressing
underlying socioeconomic barriers, is essential to enhance survival
outcomes.
In conclusion, PTSS is a rare and aggressive
malignancy with significant diagnostic challenges. Accurate
preoperative diagnosis, timely surgical intervention and
comprehensive multimodal adjuvant therapy are essential for
improving patient outcomes. The present case report highlights the
importance of maintaining a high index of suspicion for rare
sarcomas and the need for a broader diagnostic approach, including
molecular analysis, to avoid misdiagnosis. Given the limited
evidence and rarity of PTSS, further research is necessary to
refine treatment strategies and improve patient survival.
Additional multicenter studies, particularly involving neoadjuvant
and targeted therapies, are required to establish more effective
treatment protocols and improve survival outcomes for patients with
PTSS.
Acknowledgements
Not applicable.
Funding
This research was supported by the President Foundation of The
Fifth Affiliated Hospital, Southern Medical University (grant nos.
YZ2022ZX08, YZ2022ZX10 and YZ2022ZX18). Additionally, this study
was supported by the Clinical Research Special Fund of the
Guangdong Medical Association (grant nos. 2024QC-A1017 and
2024QC-A1018).
Availability of data and materials
The data generated in the present study may be
requested from the corresponding authors.
Authors' contributions
BL designed the study, analyzed the data and drafted
the original manuscript. JZ, MS and CL were responsible for data
acquisition and the literature search. YL, MH and QC contributed to
the analysis and interpretation of the data and critically revised
the manuscript for important intellectual content. BL and QC
confirmed the authenticity of all the raw data. All authors have
read and agreed to the published version of the manuscript.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
As the patient is deceased, written informed consent
was obtained from the patient's next of kin for the publication of
this case report and any accompanying clinical information and
images.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Salah S and Salem A: Primary synovial
sarcomas of the mediastinum: A systematic review and pooled
analysis of the published literature. ISRN Oncol.
2014:4125272014.PubMed/NCBI
|
|
2
|
Blay JY, von Mehren M, Jones RL,
Martin-Broto J, Stacchiotti S, Bauer S, Gelderblom H, Orbach D,
Hindi N, Dei Tos A and Nathenson M: Synovial sarcoma:
Characteristics, challenges, and evolving therapeutic strategies.
ESMO Open. 8:1016182023. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Eisenberg RB and Horn RC: Synovial sarcoma
of the chest wall; report of a case. Ann Surg. 131:281–286. 1950.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Zhou Y, Dong W, Zou F, Zhou DA and Ma JA:
Primary giant mediastinal synovial sarcoma of the neck: A case
report and review of the literature. Oncol Lett. 7:140–144. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Kara HV, Javidfar J and D'Amico TA:
Surgical excision for mediastinal synovial sarcoma with limited
response to chemoradiotherapy. Ann Thorac Surg. 98:e69–e70. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Kang MK, Cho KH, Lee YH, Han IY, Yoon YC,
Park KT, Kang DK and Kim BM: Primary synovial sarcoma of the
parietal pleura: A case report. Korean J Thorac Cardiovasc Surg.
46:159–161. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Satoh H, Ohara G and Hizawa N: Primary
synovial sarcoma of the chest wall. J Thorac Oncol. 2:10602007.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Liang Y, Wang H, Yang J, Li X, Dai C, Shao
P, Tian G, Wang B and Wang Y: A deep learning framework to predict
tumor Tissue-of-Origin based on copy number alteration. Front
Bioeng Biotechnol. 8:7012020. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Dewi KP, Dewi IP, Iswanto I and Wulandari
L: A review on pulmonary and mediastinal synovial sarcoma. J Basic
Clin Physiol Pharmacol. 34:169–175. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Duran-Mendicuti A, Costello P and Vargas
SO: Primary synovial sarcoma of the chest: Radiographic and
clinicopathologic correlation. J Thorac Imaging. 18:87–93. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Pieropan S, Mercier O, Mitilian D, Pradère
P, Fabre D, Ion DI, Mir O, Galbardi B, Thomas De Montpreville V and
Fadel E: Feasibility and long-term outcomes of surgery for primary
thoracic synovial sarcoma. Interact Cardiovasc Thorac Surg.
35:ivac2382022. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Endara SA, De la Torre JS, Terán FJ,
Alarcón JP and Tovar CE: Multidisciplinary management of recurrent
synovial sarcoma of the chest wall. N Am Spine Soc J.
15:1002432023.PubMed/NCBI
|
|
13
|
Suster S and Moran CA: Primary synovial
sarcomas of the mediastinum: A clinicopathologic,
immunohistochemical, and ultrastructural study of 15 cases. Am J
Surg Pathol. 29:569–578. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Zeren H, Moran CA, Suster S, Fishback NF
and Koss MN: Primary pulmonary sarcomas with features of monophasic
synovial sarcoma: A clinicopathological, immunohistochemical, and
ultrastructural study of 25 cases. Hum Pathol. 26:474–480. 1995.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Aubry MC, Bridge JA, Wickert R and
Tazelaar HD: Primary monophasic synovial sarcoma of the pleura:
Five cases confirmed by the presence of SYT-SSX fusion transcript.
Am J Surg Pathol. 25:776–781. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Takenaka S, Ueda T, Naka N, Araki N,
Hashimoto N, Myoui A, Ozaki T, Nakayama T, Toguchida J, Tanaka K,
et al: Prognostic implication of SYT-SSX fusion type in synovial
sarcoma: A multi-institutional retrospective analysis in Japan.
Oncol Rep. 19:467–476. 2008.PubMed/NCBI
|
|
17
|
Spillane AJ, A'Hern R, Judson IR, Fisher C
and Thomas JM: Synovial sarcoma: A clinicopathologic, staging, and
prognostic assessment. J Clin Oncol. 18:3794–3803. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Abdulghaffar S, AlNuaimi D, AlMulla M,
Eldoky Y, Farhan R, Kumar N and Khairi TE: A rare case of
pleuropulmonary synovial sarcoma of the chest wall: A case report
and a literature review. Radiol Case Rep. 16:175–179. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Wan JL, Lam YF, Foong KW, Abdul Ghani N
and Lachmanan K: A case of primary pleural synovial sarcoma with
endobronchial recurrence. Respirol Case Rep. 8:e005472020.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Choudhary CR, Desai G, Verma L and Yogi
SK: Synovial cell sarcoma of the pleura: An uncommon cause of a
haemorrhagic pleural effusion. Int J Adv Med. 7:704–706. 2020.
View Article : Google Scholar
|
|
21
|
Kagawa Y, Kitaguchi S, Konishi H,
Hashimoto K, Norihito O, Mizumoto T, Nishino R and Sugahara F:
Primary advanced pulmonary synovial sarcoma treated with a
multidisciplinary approach. Int Cancer Conf J. 9:72–76. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Kim M, Lee SE and Choi JH: Synovial
sarcoma of the anterior chest wall: A case report. J Korean Soc
Radiol. 81:1227–1233. 2020. View Article : Google Scholar
|
|
23
|
Khalili N, Askari E, Khalili N,
Daneshvar-Kakhki A, Sadr M, Haseli S and Pourabdollah Toutkaboni M:
Primary pleuropulmonary synovial sarcoma: Report of two cases and a
comprehensive review of the literature. Cancer Invest. 40:268–281.
2022. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Kumar S, Goyal K, Bhatt R, Bansal S and
Mishra M: Primary pleural synovial sarcoma: A rare cause of
hemorrhagic pleural effusion. Adv Respir Med. 89:60–62. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Saad A, Bouacida I, Ben Radhia B, Zribi H,
Dridi A and Marghli A: Primary synovial sarcoma of the mediastinum:
A poor prognosis in a 14-year-old girl. Respirol Case Rep.
9:e008112021. View
Article : Google Scholar : PubMed/NCBI
|
|
26
|
He H, Yang L, Peng Y, Liu L, Liu L, Xue Q
and Gao S: The value of multidisciplinary team (MDT) management in
the diagnosis and treatment of primary intrathoracic synovial
sarcomas: A single-center experience. J Thorac Dis. 13:600–612.
2021. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Vishnoi JR, Sasidhar A, Kala P, Khera S,
Pareek P, Misra S and Jain A: Primary synovial sarcoma of the
anterior chest wall: A rare entity at unusual Location-case report.
SN Compr Clin Med. 3:1066–1070. 2021. View Article : Google Scholar
|
|
28
|
Patel PB, Sankrith M and Cedeno-Rodriguez
A: Pediatric pleuropulmonary synovial sarcoma: A case report in a
recurrent spontaneous pneumothorax. Respir Med Case Rep.
36:1016222022.PubMed/NCBI
|
|
29
|
Oneglia MM, Fernández Sardá MS, Felizzia
G, Álvarez M, Barrenechea M, Giubergia V, Dicembrino M and Castaños
C: Persistent spontaneous pneumothorax as a primary manifestation
of primary synovial sarcoma of the lung: A case report. Arch Argent
Pediatr. 121:e2022027142023.PubMed/NCBI
|
|
30
|
Shilo K, Kneuertz PJ, Liebner D and Chen
W: Case report: Pulmonary synovial sarcoma in a Long-term survivor
of childhood Hodgkin lymphoma. Front Oncol. 13:10961602023.
View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Wu D, Zhang H, Li X, Liu J, Liu M, Liu H,
Dong M and Chen J: Whole-exome sequencing in primary pulmonary
synovial sarcoma misdiagnosed as mesothelioma: A case report and
literature review. Thorac Cancer. 14:1520–1529. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Hozain AE, Corvin C, Li H, Zane K,
McGinnis T, Olson DJ, Connell PP and Madariaga MLL: Mediastinal
synovial sarcoma 14 years after talc pleurodesis for spontaneous
pneumothorax. Ann Thorac Surg Short Rep. 1:515–518. 2023.
View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Zang Y, Ma C, Xing X and Li H: Primary
synovial sarcoma of the pleura in an 18-year-old male patient: A
case report. Oncol Lett. 26:4612023. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Do Q, Katiyar V, Rizzo J and Singh V:
Primary pleuropulmonary synovial sarcoma presenting with recurrent
spontaneous pneumothorax. BMJ Case Rep. 16:e2545602023. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Idrees RB, Malik M, Malik F, Rehman B,
Sarwar T, Mustansar A and Chaudhary MH: Chest wall synovial
sarcoma: A unique encounter at the breast base. Cureus.
16:e634992024.PubMed/NCBI
|
|
36
|
Imen M, Ahmed L, Wissal R, Bechir BR and
Taieb C: A case report of primary pleural synovial sarcoma, an
uncommon etiology of a thoracic parietal mass. Int J Surg Case Rep.
126:1106632025. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Vega-Gonzalez J, Toro JAC, García EL,
Ospina GM, Serrano MT, Gallardo AMC, Giménez RB, Martínez DH, Egido
AG, García LA, et al: EWSR1::SSX1 Fusion-driven synovial sarcoma: A
case presentation and review of the literature. Genes Chromosomes
Cancer. 64:e700482025. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Abu-Zaid A, AlNajjar A, Alotaibi S,
Alshawaf R, Alqeshtaini N, Alhaidar R, Mohammed S and AlKattan K:
Huge primary mediastinal synovial sarcoma fully occupying the right
hemithorax. J Cancer Res Ther. 14:682–686. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Liu Y, Cui M, Zhou X, Zhai D, Qin M, Fan G
and Cai W: A case report: Synovial sarcoma of the mediastinum in an
18-year-old teenager. Front Oncol. 14:12882132024. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Lin GQ, Li YQ, Huang LJ, Luo FY, Jiang HH
and Luo WJ: Chest wall tumors: Diagnosis, treatment and
reconstruction. Exp Ther Med. 9:1807–1812. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Ouadnouni Y, Smahi M, Bouchikh M, Achir A,
Msougar Y, Lakranbi M and Benosman A: A rare tumor of the chest
wall: The synovial sarcoma. Pan Afr Med J. 9:22011.(In French).
PubMed/NCBI
|
|
42
|
Minami Y, Matsumoto S, Ae K, Tanizawa T,
Hayakawa K, Funauchi Y, Saito M and Tsuda Y: The role of
neoadjuvant chemotherapy in resectable primary synovial sarcoma.
Anticancer Res. 40:1029–1034. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Pasquali S and Gronchi A: Neoadjuvant
chemotherapy in soft tissue sarcomas: Latest evidence and clinical
implications. Ther Adv Med Oncol. 9:415–429. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Gómez-Puerto D, Cicala CM, Valverde C and
Serrano C: Emerging treatments for sarcoma: From 2024 onward.
Expert Opin Emerg Drugs. Feb 3–2025.(Epub ahead of print). doi:
10.1080/14728214. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Rodriguez-Cid JR, Juarez-Vignon Whaley JJ,
Sánchez-Domínguez G, Guzmán-Casta J, Carrasco-CaraChards S,
Guzmán-Huesca J, Riera-Sala R, Sánchez-Ríos CP, Cruz-Zermeño M,
Seidman-Sorsby A, et al: Epirubicin, cisplatin plus ifosfamide
versus standard chemotherapeutic regimens for advanced/unresectable
primary thoracic sarcomas. J Cancer Res Clin Oncol. 149:5479–5491.
2023. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Jones SE, Fleuren EDG, Frankum J, Konde A,
Williamson CT, Krastev DB, Pemberton HN, Campbell J, Gulati A,
Elliott R, et al: ATR is a therapeutic target in synovial sarcoma.
Cancer Res. 77:7014–7026. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Shamsi ZA, Paraoan V, Kim C, Saifuddin SR,
Cosker TDA, Whitwell D, Gibbons CLMH, Stavroulias D and DiChiara F:
Management and outcomes of thoracic sarcomas-a collaboration
between Orthopaedic Oncology and cardiothoracic surgery: Seven-year
clinical data from a tertiary referral centre. J Cardiothorac Surg.
20:982025. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Nowicki TS, Akiyama R, Huang RR, Shintaku
IP, Wang X, Tumeh PC, Singh A, Chmielowski B, Denny C, Federman N
and Ribas A: Infiltration of CD8 T cells and expression of PD-1 and
PD-L1 in synovial sarcoma. Cancer Immunol Res. 5:118–126. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Ruemmele T, Macedo R, Stein MN, Chan HT,
Mapara MY, Jacquemont CF and Reshef R: Emapalumab for severe
cytokine release syndrome in solid tumor CAR-T: A case report.
Front Oncol. 15:15436222025. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Fairchild CK, Floros K, Jacob S, Coon C,
Puchalapalli M, Hu B, Dozmorov M, Koblinski J, Smith S, Domson G,
et al: Evaluation of combined BCL-2/MCL-1 inhibition as a
therapeutic approach for synovial sarcoma. J Clin Oncol.
38:e235612020. View Article : Google Scholar
|
|
51
|
Song S, Park J, Kim HJ, Kim IH, Han I, Kim
HS and Kim S: Effects of adjuvant radiotherapy in patients with
synovial sarcoma. Am J Clin Oncol. 40:306–311. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Seo SW, Kim J, Son J and Lim S: Evaluation
of conditional treatment effects of adjuvant treatments on patients
with synovial sarcoma using Bayesian subgroup analysis. BMC Med
Inform Decis Mak. 20:3202020. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Desar IME, Fleuren EDG and van der Graaf
WTA: Systemic treatment for adults with synovial sarcoma. Curr
Treat Options Oncol. 19:132018. View Article : Google Scholar : PubMed/NCBI
|