Introduction
Leptomeningeal metastasis (LM) is a type of central
nervous system metastasis defined by the infiltration and spread of
tumor cells in the leptomeninges or spinal leptomeninges.
Clinically, it can present with a broad spectrum of symptoms,
including headache, vomiting, mental and behavioral changes,
seizures, hearing loss, visual disturbances, limb weakness and
paresthesia (1,2). LM represents 11–20% of central nervous
system metastases, with the highest rates reported in melanoma and
lung cancer (2). LM is associated
with a notably poor prognosis and short survival time, with median
overall survival (OS) ranging from 3.6 weeks to 11 months (1). LM develops in 5–10% all patients with
solid tumors and 3–5% patients with non-small cell lung cancer
(NSCLC) (2–4). The development of LM is a complex,
multi-step process that is driven by tumor cells acquiring
capabilities for invasion and migration. These capabilities are
supported by dynamic cytoskeletal reorganization and the activation
of conserved pro-metastatic signaling pathways, such as the
SPP1-CD44 axis and CXCL2/CCL2 chemokines (5). In the absence of treatment, patients
with LM can succumb within 4–6 weeks. Conversely, the median
duration of survival with treatment ranges from 3.6 to 4.5 months
(6,7). Because of the blood-brain barrier
(BBB), the majority of drugs cannot enter the meningeal cavity;
therefore the meningeal cavity serves as a sanctuary site for tumor
cells, allowing them to evade systemic therapies. However, due to
advancements in imaging and systemic disease control, LM is
increasingly diagnosed in patients with lung adenocarcinoma
(8,9). With improvements in therapy, including
targeted therapy, radiotherapy and intrathecal therapy, OS in
patients with NSCLC and LM has improved, increasing from 1–3 to a
current range of 11.0–20.4 months (10–12).
A single-arm phase II clinical study previously
revealed that intrathecal therapy was efficient and safe in
patients with LM from epidermal growth factor receptor (EGFR)
mutation-positive NSCLC. Using the Response Assessment in
Neuro-Oncology (RANO) criteria, the response rate was high at
~80.3%, but certain patients (19.7%) did not respond to treatment
(10). In the BLOOM trial of
patients with EGFR mutation-positive NSCLC and LM, a double dose of
osimertinib produced an effective response with an objective
response rate (ORR) of 62% and a median duration of response of
15.2 months, according to a neuroradiological blinded central
independent review (11).
Similarly, in 80 patients with EGFR or anaplastic lymphoma kinase
(ALK) mutation-positive lung adenocarcinoma, median OS was 10.4
months, compared with 3.8 months in patients with wild-type lung
adenocarcinoma (13). Furthermore,
a retrospective study of patients with lung adenocarcinoma and LM
revealed that brain radiotherapy could prolong survival, with
median OS of 6.2 months (95% CI, 4.4–12.4) (13). This outcome was achieved because
brain radiotherapy could damage the BBB, thereby increasing the
intracranial antitumor drug concentration (14). Although a growing number of
therapeutic methods have led to improvements in OS and ORR among
patients with LM from lung adenocarcinoma, the existence of
non-responders highlights the need to identify the factors
influencing treatment outcomes.
The present retrospective study aimed to identify
prognostic factors for advanced lung adenocarcinoma with LM to
enhance the efficacy of novel therapies by Kaplan-Meier method.
Materials and methods
Patients
The present study was approved by the Ethics
Committee of the Ningbo Medical Center, Lihuili Hospital (approval
no. KY2024SL297-01). Requirement for informed consent was waived by
the Ethics Committee of Ningbo Medical Center Lihuili Hospital
since this is a retrospective study. All patients were treated
between November 2016 and August 2024.
The inclusion criteria were as follows: i) Age of
18–85 years; ii) confirmation of lung adenocarcinoma by pathology;
iii) LM diagnosis by cerebrospinal fluid (CSF) cytology or MRI
using previous Chinese expert consensus (15); iv) peripheral blood testing within 7
days of an LM diagnosis and before treatment; and v) receipt of
third-generation EGFR tyrosine kinase inhibitor (TKI) therapy,
intrathecal therapy or brain radiotherapy after the LM diagnosis.
The exclusion criteria were as follows: i) Immunodeficiency; ii)
diagnosis of other carcinoma; iii) presence of serious
co-morbidities (such as heart disease, infection or renal failure);
iv) inadequate laboratory information and clinical data; v)
pregnancy; vi) received corticosteroid therapy or had an infection
within the 7 days preceding enrollment; and vii) gene mutations
(including ALK, RET, HER2 and ROS1) other than EGFR exon 19 or 21
mutations, because for other mutations, either the patient
population was small or there were no definitively effective
targeted drugs established for leptomeningeal metastasis.
Clinical data collected included the following: i)
Age; ii) sex; iii) smoking status; iv) ECOG PS (16); v) Karnofsky performance status (KPS)
(17); vi) receipt of chemotherapy
or brain radiotherapy before a diagnosis of LM; vii) receipt of
brain radiotherapy after a diagnosis of LM (clinical data indicated
that there were no standard guidelines prior to 2025; the decision
to proceed with brain radiotherapy following LM was based on
physician clinical experience rather than solely on patient
condition); viii) CSF cytology; ix) brain MRI; x) driver gene
mutation status; xi) brain metastasis (BM); xii) extracranial
metastasis; xiii) receipt of third-generation EGFR TKI therapy
before a LM diagnosis; xiv) receipt of intrathecal therapy; and xv)
neurological symptoms (such as dizziness and headache). Peripheral
blood test variables included the neutrophil (×109/l),
platelet (×109/l) and lymphocyte counts
(×109/l) within 7 working days until participants
received brain radiotherapy, intrathecal therapy or targeted
therapy after the LM diagnosis.
Response evaluation
Typically, the treatment response of LM is evaluated
according to the RANO and European Association of
Neuro-Oncology-European Society for Medical Oncology guidelines
based on the clinical neurological examination, CSF pathology and
MRI (18,19). However, the relevant data were
characterized by incomplete recording, inconsistent documentation
and a lack of standardization among patients. Consequently,
LM-specific endpoints (neurological PFS, CSF cytology clearance and
intracranial control) were not reliably assessable in the present
study. Therefore, OS, defined as the time from the initial
diagnosis of LM to death or the last follow-up, was the primary
endpoint of this study.
Statistical analysis
The results were presented as the median, range and
proportion. The Kaplan-Meier method was used for survival analysis.
Univariate analysis and multivariate Cox regression analysis were
performed to identify the factors independently predictive of
survival. If predictors exhibited potential multicollinearity,
appropriate factors would be selected through association analysis
whilst considering data balance. All tests were two-sided, where
P<0.05 was considered to indicate a statistically significant
difference. The cut-offs of the neutrophil-to-lymphocyte ratio
(NLR) and platelet-to-lymphocyte ratio (PLR) were determined by
receiver operating characteristic (ROC) curve analysis and the
Youden index. Based on previous studies, the molecular graded
prognostic assessment (molGPA) score was calculated following the
established criteria (Table I),
which involved summing the individual scores for KPS, extracranial
metastasis and EGFR mutation status. This score was then
investigated as a predictive factor (20–22).
SPSS 20.0 (IBM Corp.) was used to analyze all statistical data.
 | Table I.Scoring standard of molGPA for
LM. |
Table I.
Scoring standard of molGPA for
LM.
|
| molGPA for LM |
|---|
|
|
|
|---|
| Predictor | 0 | 0.5 | 1 |
|---|
| Karnofsky
performance score | <60 | 60-70 | 80-100 |
| Extracranial
metastasis | Present | Absent | - |
| Epidermal growth
factor receptor | No sensitive
mutation | Sensitive
mutation | - |
Results
Characteristics of patients
From November 2016 to August 2024, LM from lung
adenocarcinoma was confirmed by CSF cytology or MRI in 78 patients.
The clinical information of patients is summarized in Table II. The median age of the patients
was 64 years (range, 37–83). The cohort included 39 patients <60
years old, where 44 patients were male. The majority of patients
were non-smokers (73.1%) and most patients had ECOG PS 0–2 (79.5%).
A total of 12 patients (15.4%) received brain radiotherapy before
the LM diagnosis and 24 patients (30.8%) underwent brain
radiotherapy after LM was detected. In addition, 49 patients
(62.8%) had BM and 58 patients (74.4%) exhibited extracranial
metastasis. In addition, 69 patients (88.5%) had neurological
symptoms, whilst 36 patients (46.2%) received intrathecal therapy
and 47 patients (60.3%) received third-generation EGFR TKI therapy
before LM was detected. EGFR mutational profiling identified exon
19 deletions in 26 patients (33.3%) and the L858R mutation (exon
21) in 30 patients (38.5%), whilst 22 (28.2%) were EGFR wild-type.
Furthermore, nine EGFR-wild-type patients underwent next-generation
sequencing testing, which confirmed no EGFR mutations but revealed
alterations in other genes, including KRAS, TP53 and PIK3CA. The
modalities used to diagnose LM were MRI alone (27 patients; 34.6%),
CSF cytology alone (20 patients; 25.6%) and MRI + CSF cytology (31
patients; 39.8%). The molGPA score was 0 in 13 patients (16.7%),
0.5–1.0 in 58 patients (74.4%) and 1.5–2.0 in 7 patients
(8.9%).
 | Table II.Clinical characteristics of patients
(n=78). |
Table II.
Clinical characteristics of patients
(n=78).
| Characteristic | Patients |
|---|
| Median age, years
(range) | 64.0 (37–83) |
| Age, years, N
(%) |
|
|
<60 | 39 (50.0) |
|
≥60 | 39 (50.0) |
| Sex, N (%) |
|
|
Male | 44 (56.4) |
|
Female | 34 (43.6) |
| Smoking, N (%) |
|
|
Yes | 21 (26.9) |
| No | 57 (73.1) |
| Eastern Cooperative
Oncology Group performance status, N (%) |
|
|
0-2 | 62 (79.5) |
|
3-4 | 16 (20.5) |
| Chemotherapy before
LM, N (%) |
|
|
Yes | 42 (53.8) |
| No | 36 (46.2) |
| Brain radiotherapy
before LM, N (%) |
|
|
Yes | 12 (15.4) |
| No | 66 (84.6) |
| Brain radiotherapy
after LM, N (%) |
|
|
Yes | 24 (30.8) |
| No | 54 (69.2) |
| Brain metastasis, N
(%) |
|
|
Yes | 49 (62.8) |
| No | 29 (37.2) |
| Extracranial
metastasis, N (%) |
|
|
Yes | 58 (74.4) |
| No | 20 (25.6) |
| Neurological
symptoms, N (%) |
|
|
Yes | 69 (88.5) |
| No | 9 (11.5) |
| Gene mutation, N
(%) |
|
| EGFR 19
mutation | 26 (33.3) |
| EGFR 21
mutation | 30 (38.5) |
| EGFR
wild-type | 22 (28.2) |
| Third-generation
EGFR tyrosine kinase inhibitor therapy before LM, |
|
| N (%) |
|
|
Yes | 47 (60.3) |
| No | 31 (39.7) |
| Modality of LM
diagnosis, N (%) |
|
|
MRI | 27 (34.6) |
|
Cerebrospinal fluid
cytology | 20 (25.6) |
|
Both | 31 (39.8) |
|
Neutrophil-to-lymphocyte ratio (%) |
|
|
<7.5755 | 61 (78.2) |
|
≥7.5755 | 17 (21.8) |
|
Platelet-to-lymphocyte ratio, N (%) |
|
|
<156.035 | 35 (44.9) |
|
≥156.035 | 43 (55.1) |
| Intrathecal therapy
(%) |
|
|
Yes | 36 (46.2) |
| No | 42 (53.8) |
| Molecular graded
prognostic assessment (%) |
|
|
0.0 | 13 (16.7) |
|
0.5–1.0 | 58 (74.4) |
|
1.5–2.0 | 7 (8.9) |
Optimal cut-offs for NLR and PLR
In the present study, ROC curve analysis was used to
select the appropriate cut-offs for NLR and PLR. For NLR, the
optimal cut-off was 7.5755, which had an area under curve (AUC) of
0.599. Further, the optimal cut-off for PLR was 156.035
(AUC=0.654).
Survival outcomes
Median OS for all patients was 9.0 months. OS was
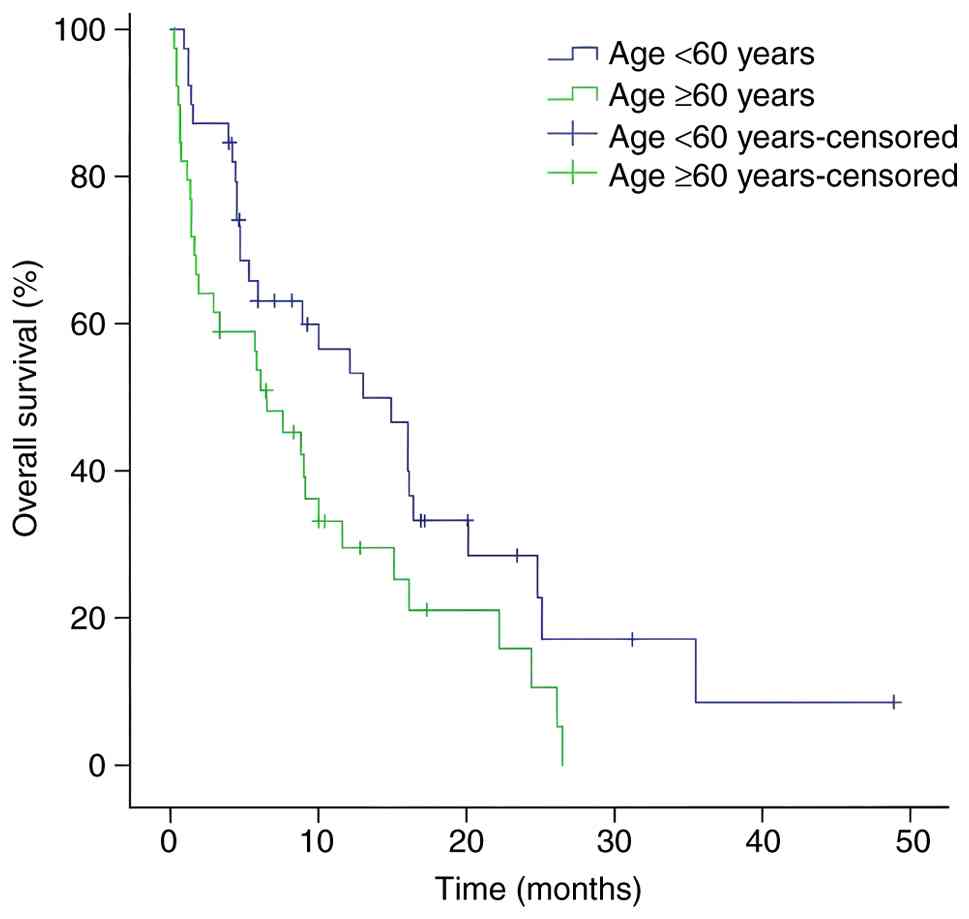
improved in patients <60 years old compared with those aged ≥60
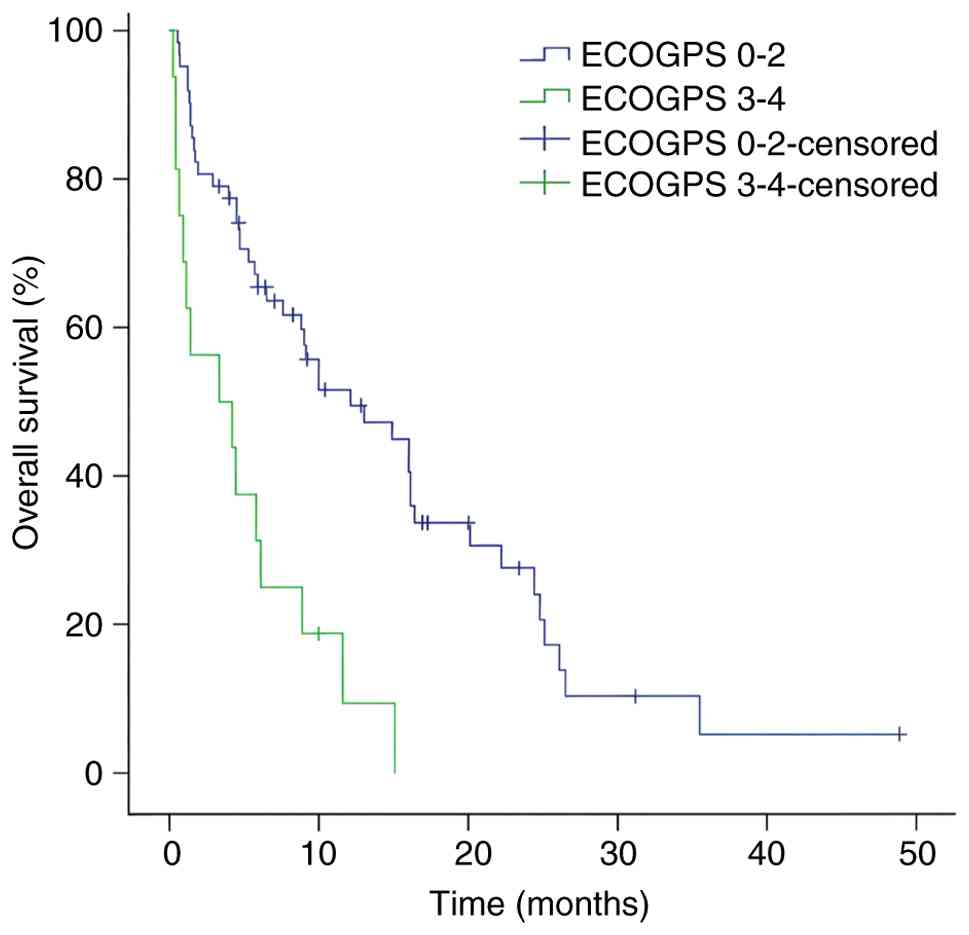
years (Fig. 1) as well as in
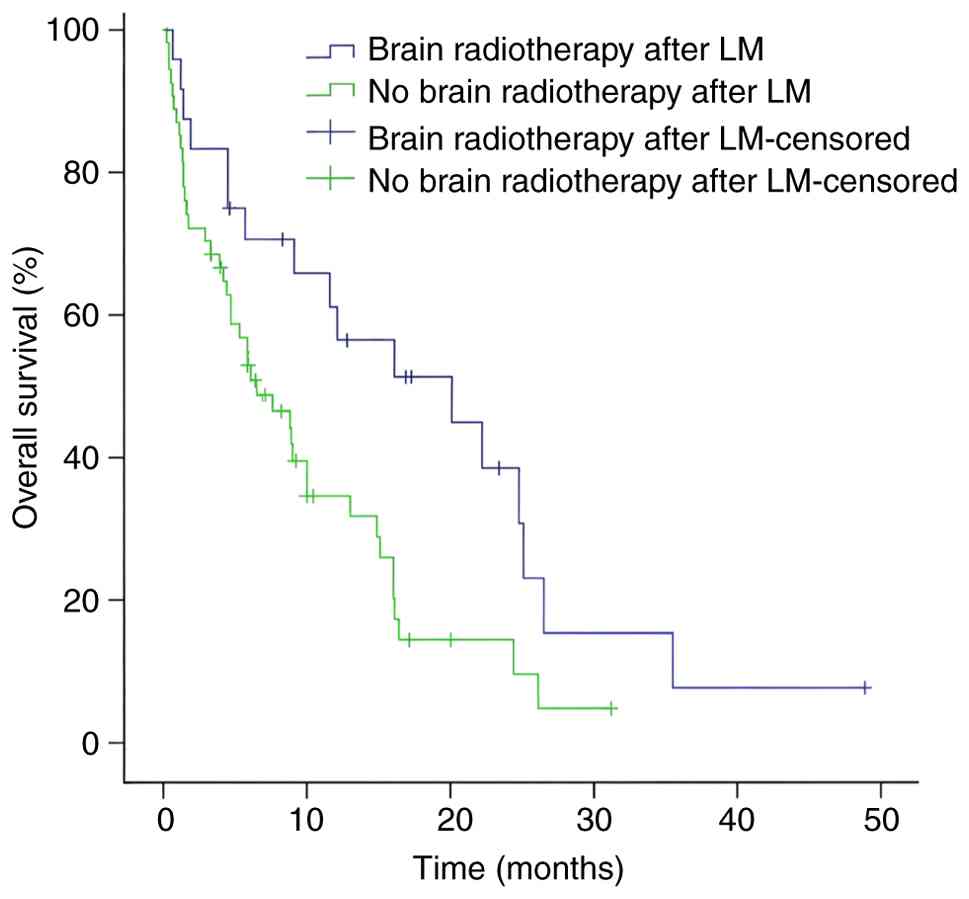
patients with an ECOG PS of 0–2 versus 3–4 (Fig. 2). The data indicated that brain
radiotherapy after the diagnosis of LM can improve the OS relative
to no brain radiotherapy (Fig. 3).
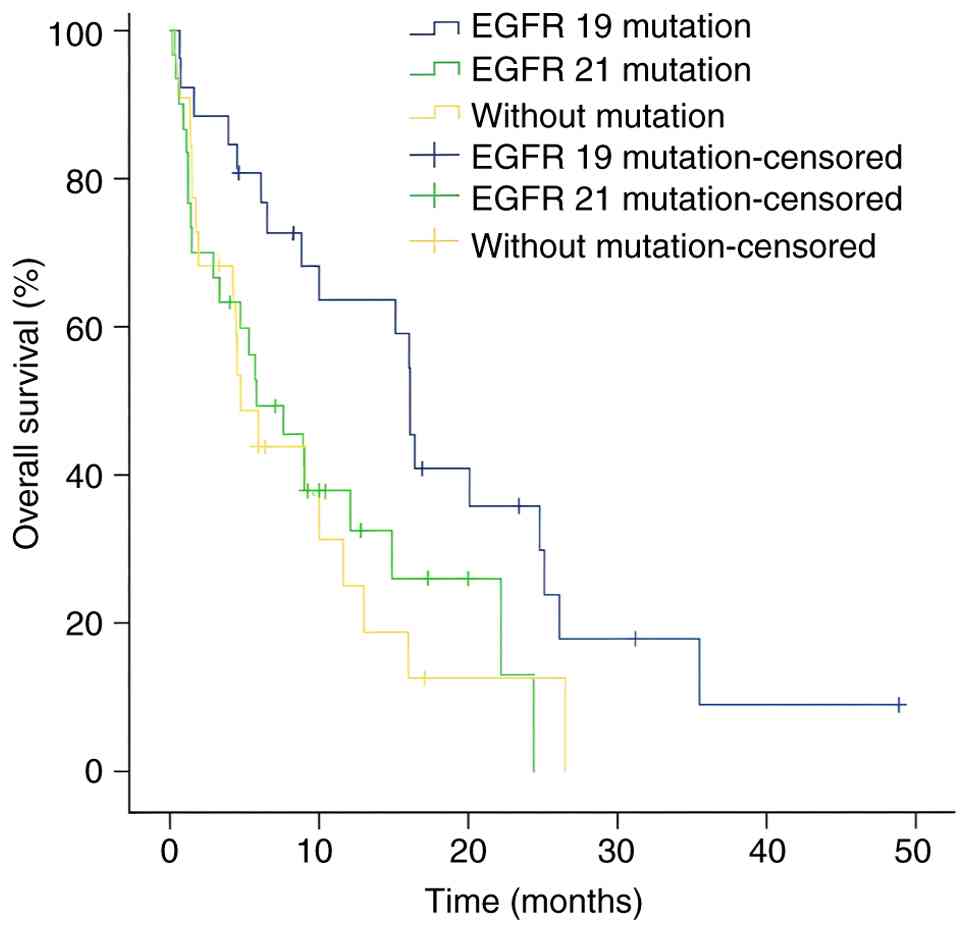
Concerning gene mutations, OS was improved in patients with EGFR 19
exon deletion compared with those with an EGFR exon 21 mutation or
EGFR wild-type (Fig. 4).
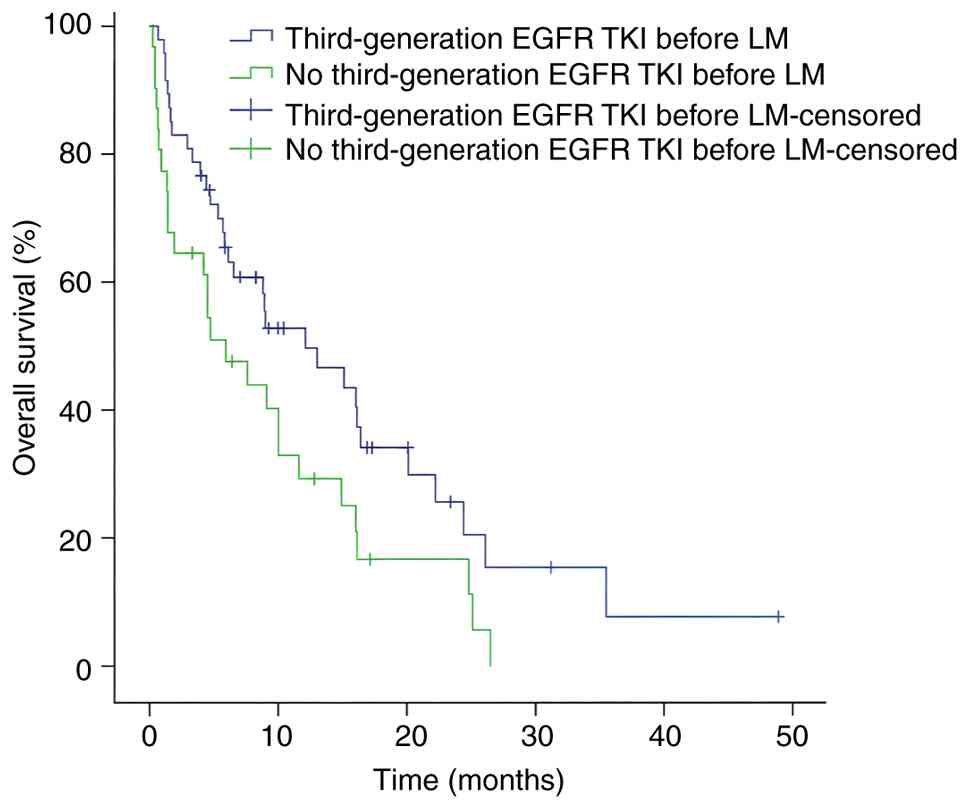
Furthermore, OS was improved among patients who received
third-generation EGFR TKI therapy before the diagnosis of LM
compared with those who received non-third-generation EGFR TKIs
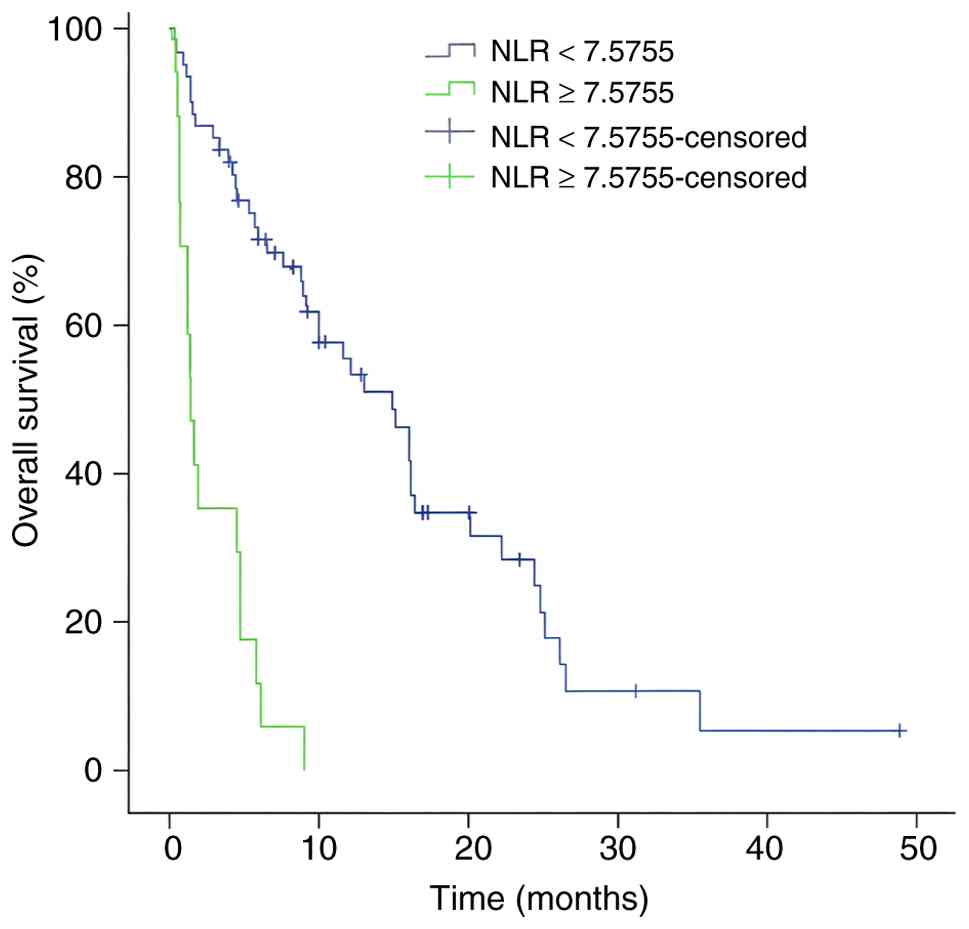
(Fig. 5). By contrast, NLR ≥7.5755
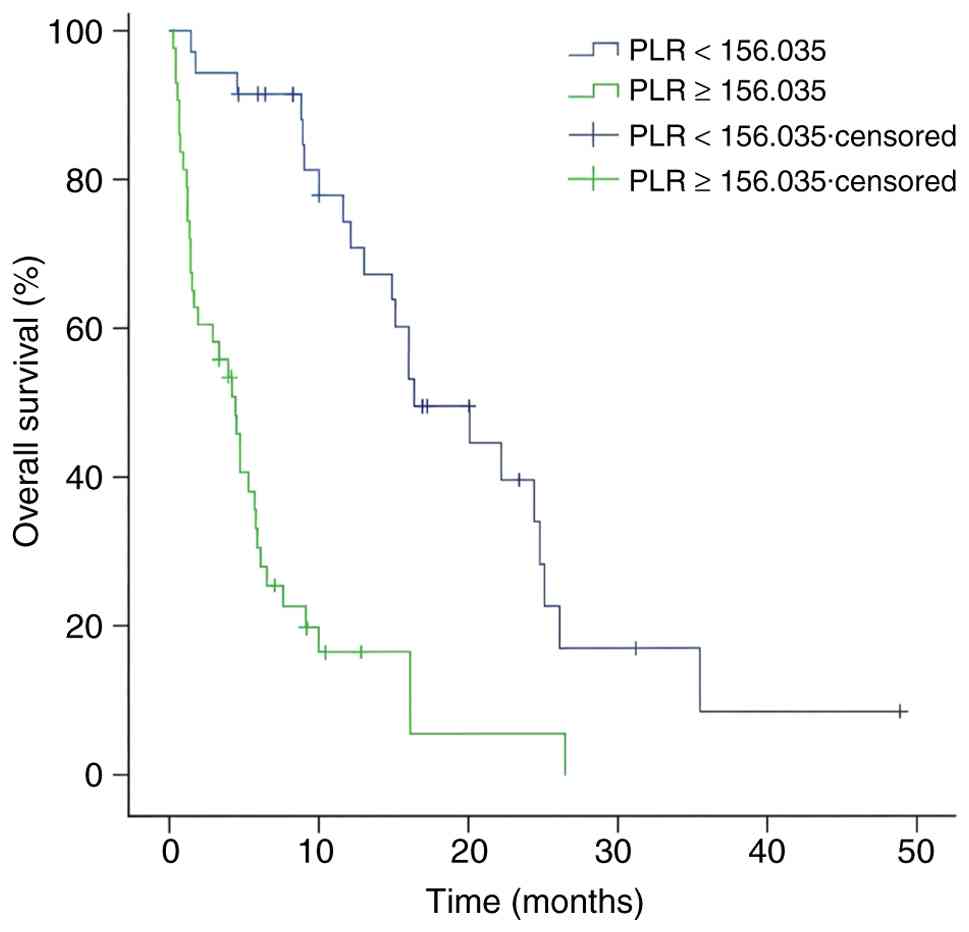
vs. <7.5755 (Fig. 6) and PLR
≥156.035 vs. <156.035 (Fig. 7)
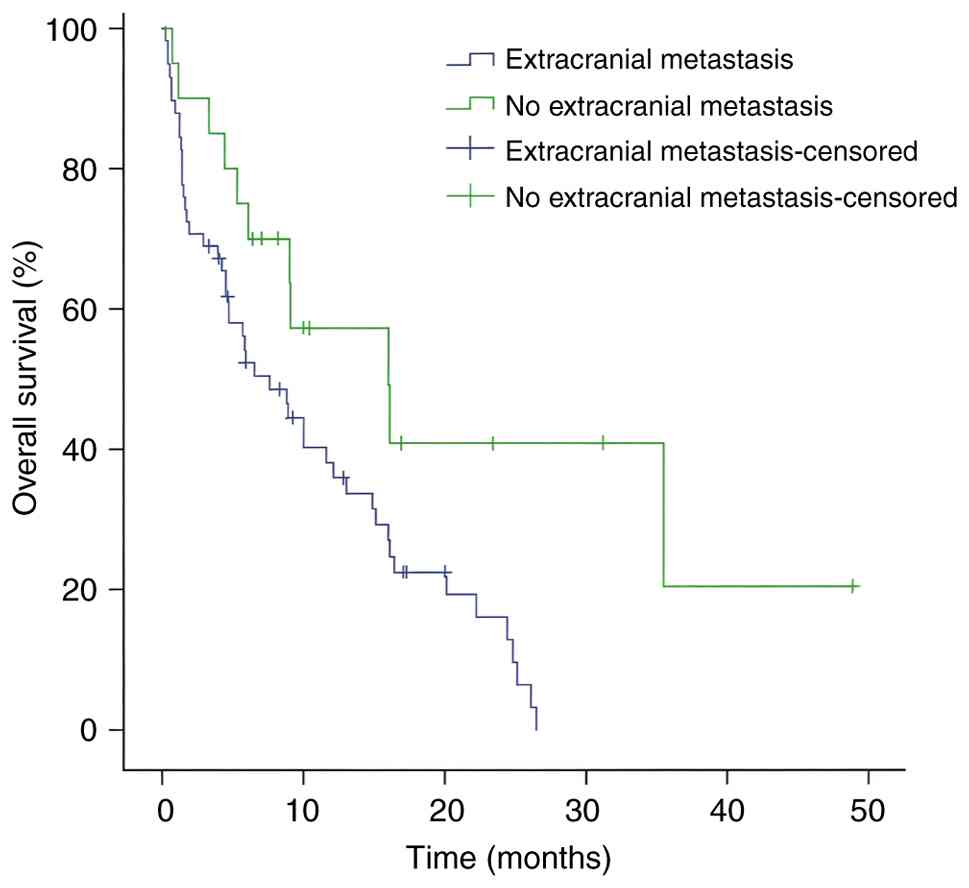
were predictive of shorter OS. In addition, extracranial metastasis
was associated with shorter OS vs. extracranial metastasis
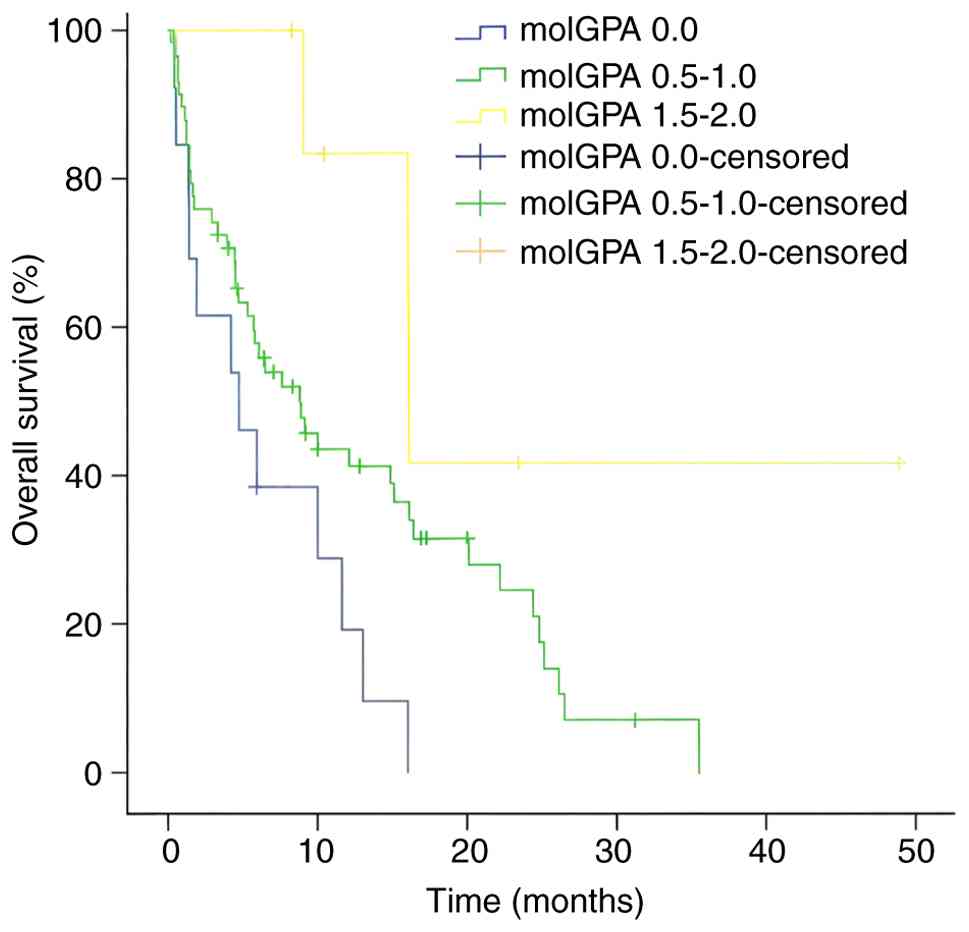
(Fig. 8). According to the molGPA
classification, patients with a score of 0.0 had worse OS compared
with those with score of 0.5–1.0 and 1.5–2.0 (Fig. 9). However, sex, smoking status, the
receipt of chemotherapy before the diagnosis of LM, the receipt of
brain radiotherapy before the diagnosis of LM, BM, neurological
symptoms, the modality used to diagnose LM diagnosis and the
receipt of intrathecal therapy were not significantly associated
with OS (Table III).
 | Table III.Uni- and multivariate analyses for
overall survival of patients with LM. |
Table III.
Uni- and multivariate analyses for
overall survival of patients with LM.
|
|
| Univariate | Multivariate |
|---|
|
|
|
|
|
|---|
| Characteristic | N | Median, months | 95% CI | P-value | Hazard ratio | 95% CI | P-value |
|---|
| Age, years |
|
|
| 0.029 | 0.844 | 0.474–1.502 | 0.563 |
|
<60 | 39 | 13.0 | 6.96–19.04 |
|
|
|
|
|
≥60 | 39 | 6.5 | 3.00–10.00 |
|
|
|
|
| Sex |
|
|
| 0.742 |
|
|
|
|
Male | 44 | 7.6 | 1.68–13.52 |
|
|
|
|
|
Female | 34 | 10.0 | 5.79–14.21 |
|
|
|
|
| Smoking |
|
|
| 0.687 |
|
|
|
|
Yes | 21 | 9.1 | 0.00–22.96 |
|
|
|
|
| No | 57 | 9.0 | 5.95–12.05 |
|
|
|
|
| Eastern Cooperative
Oncology |
|
|
| <0.001 | 0.302 | 0.152–0.599 | 0.001 |
| Group performance
status |
|
|
|
|
|
|
|
|
0-2 | 62 | 12.1 | 4.97–19.23 |
|
|
|
|
|
3-4 | 16 | 3.3 | 0.00–8.79 |
|
|
|
|
| Chemotherapy before
LM |
|
|
| 0.314 |
|
|
|
|
Yes | 42 | 11.6 | 6.61–16.59 |
|
|
|
|
| No | 36 | 7.6 | 3.25–11.95 |
|
|
|
|
| Brain radiotherapy
before LM |
|
|
| 0.514 |
|
|
|
|
Yes | 12 | 5.8 | 4.18–7.42 |
|
|
|
|
| No | 66 | 10.0 | 6.69–13.31 |
|
|
|
|
| Brain radiotherapy
after LM |
|
|
| 0.012 | 0.509 | 0.253–1.026 | 0.059 |
|
Yes | 24 | 20.1 | 5.02–35.18 |
|
|
|
|
| No | 54 | 6.5 | 2.73–10.28 |
|
|
|
|
| Brain
metastasis |
|
|
| 0.260 |
|
|
|
|
Yes | 49 | 7.6 | 3.92–11.28 |
|
|
|
|
| No | 29 | 16.0 | 9.51–22.49 |
|
|
|
|
| Extracranial
metastasis |
|
|
| 0.012 | 2.291 | 1.074–4.888 | 0.032 |
|
Yes | 58 | 7.6 | 3.99–11.21 |
|
|
|
|
| No | 20 | 16.0 | 5.47–26.53 |
|
|
|
|
| Neurological
symptoms |
|
|
| 0.110 |
|
|
|
|
Yes | 69 | 7.6 | 3.58–11.62 |
|
|
|
|
| No | 9 | 9.1 | / |
|
|
|
|
| Gene mutation |
|
|
| 0.019 | 1.050 | 0.711–1.552 | 0.806 |
| EGFR 19
mutation | 26 | 16.1 | 14.64–17.56 |
|
|
|
|
| EGFR 21
mutation | 30 | 5.8 | 1.34–10.26 |
|
|
|
|
| Without
mutation | 22 | 4.7 | 2.51–6.89 |
|
|
|
|
| Third-generation
EGFR tyrosine kinase inhibitor therapy before LM |
|
|
| 0.035 | 0.958 | 0.478–1.920 | 0.904 |
|
Yes | 47 | 12.1 | 4.20–20.00 |
|
|
|
|
| No | 31 | 5.9 | 2.39–9.41 |
|
|
|
|
| Modality of LM
diagnosis |
|
|
| 0.666 |
|
|
|
|
MRI | 27 | 9.0 | 2.18–15.83 |
|
|
|
|
|
Cerebrospinal fluid
cytology | 20 | 9.1 | 6.18–12.02 |
|
|
|
|
|
Both | 31 | 10.0 | 2.08–17.92 |
|
|
|
|
|
Neutrophil-to-lymphocyte ratio |
|
|
| <0.001 |
|
|
|
|
<7.5755 | 61 | 14.9 | 10.53–19.27 |
|
|
|
|
|
≥7.5755 | 17 | 1.4 | 0.86–1.94 |
|
|
|
|
|
Platelet-to-lymphocyte ratio |
|
|
| <0.001 | 0.233 | 0.123–0.442 | <0.001 |
|
<156.035 | 35 | 16.4 | 10.56–22.24 |
|
|
|
|
|
≥156.035 | 43 | 4.4 | 3.01–5.80 |
|
|
|
|
| Intrathecal
therapy |
|
|
| 0.880 |
|
|
|
|
Yes | 36 | 10.0 | 3.70–16.30 |
|
|
|
|
| No | 42 | 8.8 | 4.25–13.35 |
|
|
|
|
| Molecular graded
prognostic assessment |
|
|
| 0.008 | 0.625 | 0.268–1.458 | 0.277 |
|
0.0 | 13 | 4.7 | 0.00–9.40 |
|
|
|
|
|
0.5–1.0 | 58 | 8.8 | 4.61–12.99 |
|
|
|
|
|
1.5–2.0 | 7 | 16.1 | 15.89–16.31 |
|
|
|
|
Since NLR and PLR share lymphocyte counts and may
exhibit collinearity, which could lead to unstable estimates if
both were included in the multivariable model, only PLR was
selected for inclusion in the model. Multivariate analysis
identified non-extracranial metastasis [vs. extracranial
metastasis; hazard ratio (HR)=2.291; 95% CI, 1.074–4.888; P=0.032],
PLR<156.035 (vs. ≥156.035; HR=0.233; 95% CI, 0.123–0.442) and
ECOG PS 0–2 (vs. 3–4; HR=0.302; 95% CI, 0.152–0.599) as meaningful
clinical characteristics associated with OS (Table III), whereas the remaining factors
were not predictive of outcomes.
Discussion
The selection of treatment for patients with lung
adenocarcinoma and LM is a difficult clinical challenge. Although
the overall prognosis is grave, significant heterogeneity in
survival exists, underscoring the critical need for reliable
prognostic biomarkers to guide personalized management. While prior
research has established several prognostic factors, including ECOG
PS, intrathecal therapy and antiangiogenic therapy following LM,
the integrative role of routinely available hematological indices,
such as NLR and PLR, in combination with established clinical
parameters, within the framework of contemporary multimodal
treatment strategies (involving third-generation EGFR-tyrosine
kinase inhibitors and radiotherapy), remains to be further validate
(10,23). The present study aimed to address
this gap by comprehensively evaluating a panel of pre-treatment
clinical and hematological variables. The prognostic significance
of several established factors in univariate analysis were
assessed, where non-extracranial metastasis, a low PLR
(<156.035) and a good performance status (ECOG PS 0–2) were
identified as independent predictors of longer OS in the
multivariate model. This integrative approach provides a practical
and robust framework for risk stratification in this patient
population.
Neutrophils, platelets and lymphocytes are commonly
investigated as peripheral blood indicators in laboratory
examination. The independent prognostic value of NLR and PLR in the
present cohort underscores the pivotal role of the systemic
inflammatory response and tumor immune microenvironment in LM
pathogenesis. Consistent with the results of ex post facto
studies, NLR and PLR, as projected factors of lung cancer
mortality, can influence the tumor inflammatory microenvironment,
particularly in patients with BM and LM (18,19,21).
NLR >3.83 (HR=2.66; 95% CI 1.17–6.01; P=0.019) indicated poor
survival in a consecutive cohort of patients with BM (24). In a previous systematic review and
meta-analysis of patients with BM from lung cancer, 11 studies
revealed that high NLR (HR=1.84; 95% CI, 1.47–2.31; P<0.001) was
significantly associated with shorter OS, where an analysis of five
studies indicated that high PLR was predictive of shorter OS
(HR=1.53; 95% CI, 1.07–2.20; P=0.020) (25). In addition, low NLR (HR=0.49; 95%
CI, 0.28–0.86; P=0.012) was associated with longer OS in patients
with lung cancer and LM. Patients with pre-treatment PLR <145.44
(P=0.031) had longer OS according to a univariate analysis
(20). Similarly, in a multivariate
analysis of the whole cohort, a pre-treatment PLR cut-off of 209.77
was associated with a significant difference in median OS (8.0 vs.
14.5 months; P=0.047). In addition, post-treatment NLR >3.57
(HR=2.203; 95% CI, 1.060–4.578; P=0.034) was a useful prognostic
factor for patients with lung cancer and LM according to a
multivariate analysis (26).
Previously, Berghoff et al (27) reported that the brain is not a
completely independent organ. Inflammatory cells can stimulate the
release of certain substances, such as reactive oxygen species,
affecting neighboring cancer cells (28). The inflammatory microenvironment
mainly consists of neutrophils, platelets and lymphocytes. As key
factors in the tumor response, neutrophils are positioned in the
glycolytic and hypoxic tumor core, where they can promote
angiogenesis to support tumor cell proliferation (29). Neutrophils represent the most common
immune cell type in human blood, where they can regulate cancer
initiation, growth and metastasis (29). Other cells, such as T cells and
macrophages, can interact with neutrophils in the tumor
microenvironment (30). By
contrast, platelets can promote epithelial-mesenchymal transition
to facilitate cancer cell proliferation and invasiveness. Activated
platelets can help cancer cells avoid immune system surveillance
and anoikis. Therefore, platelets have an important role in tumor
metastasis (31). Lymphocytes
inhibit tumor cell proliferation and provoke cancer cell killing by
T cells (32). Studies have
identified NLR and PLR as key predictors of clinical outcomes in
various tumors, such as breast and colon cancer (33,34).
In the unique sanctuary of the central nervous system, the value of
these ratios may be amplified. Inflammatory crosstalk at the
blood-CSF barrier can modulate the permeability of this barrier and
create a permissive niche for tumor cell colonization and
proliferation. Therefore, elevated NLR and PLR likely reflect a
heightened systemic and potentially local pro-metastatic milieu
that is permissive for the establishment and progression of LM,
explaining their potent prognostic power in the present
analysis.
The persistence of ECOG PS as an independent
prognostic factor reasserts the fundamental importance of the
physiological status of the patient. A poor performance status
frequently mirrors a higher systemic tumor burden, increased
cancer-related symptomatology and diminished physiological capacity
to tolerate aggressive therapies (23). The present findings suggest that
ECOG PS captures elements of disease aggressiveness and patient
frailty that are not fully explained by laboratory values or
mutation status alone. This finding aligns with that by Wang et
al (26), who reported a
significant difference in median OS (4 vs. 15 months) based on ECOG
PS. Other studies have illustrated that patients with low ECOG PS
have longer survival, consistent with the findings in the present
study (10,20). Consequently, performance status
remains an indispensable, non-redundant tool for initial
prognostication and clinical decision-making in neuro-oncology.
The present univariate analysis suggested a survival
benefit associated with brain radiotherapy administered after LM
diagnosis. Although this effect was attenuated in the multivariate
model, it hints at the potential role of brain radiotherapy in
disrupting the BBB (14). Evidence
from the literature suggests that brain radiotherapy is beneficial
for patients with NSCLC and LM (35). In a previous retrospective study,
univariate analysis illustrated that brain radiotherapy can prolong
OS compared with non-brain radiotherapy (median OS: 11.4 vs. 5.0
months; P=0.051) in 80 patients with LM from NSCLC (36). Consistent with the present findings,
brain radiotherapy after the diagnosis of LM was associated with
longer OS (median OS=20.1 months; P=0.012) according to a
univariate analysis. Although multivariate analysis did not confirm
this finding-likely due to the limited number of patients who
received brain radiotherapy-this treatment modality following the
diagnosis of LM may still be predictive of OS in patients with
NSCLC and LM (HR=0.580; P=0.132).
According to multivariate analysis, extracranial
metastasis negatively influenced OS (P=0.032) in patients with LM
from NSCLC in the present study. Previous studies identified
extracranial metastasis as a survival biomarker. In patients with
LM from NSCLC, univariate analysis illustrated that patients with
extracranial metastasis had shorter OS (HR=2.09; P<0.01)
regardless of the EGFR status (37). Multivariate analysis in another
retrospective study also demonstrated that extracranial metastasis
was an independent predictor for poorer survival compared with the
absence of extracranial metastasis (median OS: 10.0 vs. 20.0
months; P=0.023) (38). In summary,
extracranial metastasis portended inferior outcomes among patients
with NSCLC and LM.
Different EGFR mutations have been associated with
differences in OS in patients with LM. In a study by the European
Society of Medical Oncology, patients with NSCLC and LM who
harbored EGFR exon 19 mutations had superior OS (37). There are two reasons for the
distinctions between exon 19 and 21 mutations. The tyrosine
phosphorylation structure of EGFR differs between exon 19 deletion
and exon 21 mutation in NSCLC cell lines, leading to dissimilar
alterations of downstream signaling pathways (38). The other reason is that exon 21
mutation requires asymmetric dimerization for activation and
oncogenic transformation, whereas exon 19 mutation is not
associated with dimerization activation (39). Therefore, patients with LM and EGFR
19 deletion had prolonged median OS (16.1 months; P=0.019)
according to univariate analysis in the present study.
Based on univariate analysis, the receipt of
third-generation EGFR TKI therapy before the diagnosis of LM
appeared to improve OS in the present study (OS=12.1 months;
P=0.035). Third-generation EGFR TKIs have a high rate of entry into
the BBB owing to their macrocyclic amide structure and optimized
lipophilicity (40–42). However, no difference in OS
associated with this variable was observed in the multivariate
analysis. This finding could be due to the fact that individual
third-generation EGFR TKIs, such as osimertinib, almonertinib,
furmonertinib and befotertinib, were not investigated. Therefore,
further research is needed to identify the optimal third-generation
EGFR TKIs for patients with NSCLC and LM.
Previous studies identified molGPA as a prognosis
risk factor for patients with LM (21,22).
Zhang et al (21) reported
that median OS in the low-risk molGPA group was 8.02 months (95%
CI, 0.98–38.13), which significantly exceeded the survival duration
in the medium- and high-risk groups. In addition, another study
reported median OS times of 0.9, 5.8 and 17.7 months for the low-,
medium-, and high-risk molGPA groups, respectively (P<0.001)
(22). Similarly, the median OS
times in these molGPA subgroups in the present study were 4.7, 8.8
and 16.1 months, respectively, with significant differences
identified by univariate analysis (P=0.008). These results
underscore the potential utility of the molGPA model in optimizing
patient management. However, this conclusion is tempered by the
small subgroup size (n=7) in the highest molGPA category, which may
affect the reliability of the findings. Therefore, further studies
are warranted for validation.
The current study had some limitations. It was a
retrospective study, within which the small sample size raises the
possibility of selection bias. The strong univariate association
between post-LM brain radiotherapy and survival was attenuated in
the multivariable model. This could be explained if patients
receiving radiotherapy had more favorable prognostic factors at
baseline, such as superior performance status, lower disease burden
or greater fitness for further treatment, all of which
independently predict longer survival. In addition, LM was not
pathologically confirmed by CSF cytology in all patients. There was
also no control group to verify the clinical utility of the
cut-offs. The sequence and timing of third-generation EGFR TKI
treatment or intrathecal therapy in combination with brain
radiotherapy was not standardized. PD-L1 status was also not
assessed. Comprehensive next-generation sequencing data was not
performed for all patients due to the study's retrospective nature,
which may have affected the interpretation of survival and
treatment outcomes because of the unknown status of EGFR and other
gene mutations. The measured values of NLR and PLR may have been
confounded by variations in corticosteroid regimens and occurrence
of intercurrent infections. Differences in intrathecal drugs (the
doses, fractionation and sites of brain radiotherapy) and
third-generation EGFR TKIs could lead to different results. No
internal validation was performed when selecting the ROC-based
cut-offs, where the data-driven selection of cut-offs can cause
overfitting and limited generalizability. The analysis was subject
to immortal time bias and confounding by indication due to the
retrospective design and timing of therapy administration, which
may affect the interpretation of survival benefits associated with
therapies, such as radiotherapy and pre-LM TKI use. Despite these
limitations, the present study provides evidence for predicting
outcomes in patients with lung adenocarcinoma and LM.
In conclusion, the present study identified
extracranial metastasis, PLR and ECOG PS as independent predictors
of OS in patients with lung adenocarcinoma and LM. In addition,
brain radiotherapy, EGFR 19 mutation, the receipt of
third-generation EGFR TKI therapy before the diagnosis of LM and
molGPA were significantly associated with overall survival.
Although intrathecal therapy was associated with negative survival
outcomes, EGFR mutation had a crucial influence on survival in the
majority of patients. Future studies are needed to delineate the
optimal sequencing and combination of radiotherapy, intrathecal
therapy and specific TKIs.
Acknowledgements
Not applicable.
Funding
The present study was supported by Taizhou University Medicine
Special Project (grant no. 2023XY01) and Taizhou Anti-Cancer
Association Cancer Special Research Project (grant no.
TACA2025-A02).
Availability of data and materials
The data generated in the present study may be
requested from the corresponding author.
Authors' contributions
YuL was responsible for performing the experiments
and collecting data, formal analysis of data, design of the
methodology, data curation (managing, cleaning, annotating, and
preserving data) and writing the original draft. YiL and XC
analyzed data, designing the methodology, data curation (managing,
cleaning, annotating, and preserving data), validation and writing
the original draft. YZ and HD were responsible for the software
(programming, code development, and tool testing), design of the
methodology, and reviewing and editing the manuscript. YuL and XC
confirm the authenticity of all the raw data. XC edited the
manuscript. DJ was responsible for the formal analysis of data, and
review and editing the manuscript. GC and XY were responsible for
providing resources, conceptualization of the study, reviewing and
editing the manuscript, and project administration. All authors
read and approved the final version of the manuscript.
Ethics approval and consent to
participate
The present study was approved by the Ethics
Committee of Ningbo Medical Center Lihuili Hospital (approval no.
KY2024SL297-01). This study was conducted in accordance with the
Declaration of Helsinki. Requirement for informed consent was
waived by the Ethics Committee of Ningbo Medical Center Lihuili
Hospital since this is a retrospective study.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Park S, Baldry R, Jung HA, Sun JM, Lee SH,
Ahn JS, Kim YJ, Lee Y, Kim DW, Kim SW, et al: Phase II efficacy and
safety of 80 mg Osimertinib in patients with leptomeningeal
metastases associated with epidermal growth factor receptor
mutation-positive non-small cell lung cancer (BLOSSOM). J Clin
Oncol. 42:2747–2756. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Thakkar JP, Kumthekar P, Dixit KS, Stupp R
and Lukas RV: Leptomeningeal metastasis from solid tumors. J Neurol
Sci. 411:1167062020. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Lukas RV, Thakkar JP, Cristofanilli M,
Chandra S, Sosman JA, Patel JD, Kumthekar P, Stupp R and Lesniak
MS: Leptomeningeal metastases: The future is now. J Neurooncol.
156:443–452. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Beauchesne P: Intrathecal chemotherapy for
treatment of leptomeningeal dissemination of metastatic tumours.
Lancet Oncol. 11:871–879. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Zhou C, Shan S, Wen L, Liu D, Shan C, Jin
X, Zhou Z, Li H, Li J, Wang L, et al: Immunological and
pathological characteristics of brain parenchymal and
leptomeningeal metastases from non-small cell lung cancer. Cell
Discov. 11:722025. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Liao BC, Lee JH, Lin CC, Chen YF, Chang
CH, Ho CC, Shih JY, Yu CJ and Yang JC: Epidermal growth factor
receptor tyrosine kinase inhibitors for non-small-cell lung cancer
patients with leptomeningeal carcinomatosis. J Thorac Oncol.
10:1754–1761. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Umemura S, Tsubouchi K, Yoshioka H, Hotta
K, Takigawa N, Fujiwara K, Horita N, Segawa Y, Hamada N, Takata I,
et al: Clinical outcome in patients with leptomeningeal metastasis
from non-small cell lung cancer: Okayama lung cancer study group.
Lung Cancer. 77:134–139. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Wang Y, Yang X, Li NJ and Xue JX:
Leptomeningeal metastases in non-small cell lung cancer: Diagnosis
and treatment. Lung Cancer. 174:1–13. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Ozcan G, Singh M and Vredenburgh JJ:
Leptomeningeal metastasis from non-small cell lung cancer and
current landscape of treatments. Clin Cancer Res. 29:11–29. 2023.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Fan C, Jiang Z, Teng C, Song X, Li L, Shen
W, Jiang Q, Huang D, Lv Y, Du L, et al: Efficacy and safety of
intrathecal pemetrexed for TKI-failed leptomeningeal metastases
from EGFR+ NSCLC: An expanded, single-arm, phase II clinical Trial.
ESMO Open. 9:1023842024. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Yang JCH, Kim SW, Kim DW, Lee JS, Cho BC,
Ahn JS, Lee DH, Kim TM, Goldman JW, Natale RB and Brown AP:
Osimertinib in patients with epidermal growth factor receptor
mutation-positive non-small-cell lung cancer and leptomeningeal
metastases: The BLOOM study. J Clin Oncol. 38:538–547. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Cheng H and Perez-Soler R: Leptomeningeal
metastases in non-small-cell lung cancer. Lancet Oncol. 19:e43–e45.
2018. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Oyoshi H, Hirata H, Hirano Y, Zenda S,
Zhou Y, Tomizawa K, Fujisawa T, Nakamura M, Hojo H, Motegi A, et
al: Prognostic impact of EGFR/ALK alterations in leptomeningeal
metastasis from lung adenocarcinoma treated with whole-brain
radiotherapy. Clin Exp Metastasis. 40:407–413. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Sakaguchi M, Maebayashi T, Aizawa T,
Ishibashi N and Saito T: Successful treatment of nonsmall cell lung
cancer patients with leptomeningeal metastases using whole brain
radiotherapy and tyrosine kinase inhibitors. J Cancer Res Ther.
16:930–932. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Lin G, Wang Y, Xin T, Zhang D, Zhang Q, Li
Y, Chi Y, Fan Y, Liu A, Xu H, et al: Chinese expert consensus on
leptomeningeal metastases of lung cancer. Thorac Cancer.
16:e700882025. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Oken MM, Creech RH, Tormey DC, Horton J,
Davis TE, McFadden ET and Carbone PP: Toxicity and response
criteria of the eastern cooperative oncology group. Am J Clin
Oncol. 5:649–655. 1982. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Karnofsky DA, Abelmann WH, Craver LF and
Burchenal JH: The use of the nitrogen mustards in the palliative
treatment of carcinoma-with particular reference to bronchogenic
carcinoma. Cancer. 1:634–656. 1948. View Article : Google Scholar
|
|
18
|
Le Rhun E, Weller M, van Bent M, Brandsma
D, Furtner J, Rudà R, Schadendorf D, Seoane J, Tonn JC, Wesseling
P, et al: Leptomeningeal metastasis from solid tumours: EANO-ESMO
clinical practice guideline for diagnosis, treatment and follow-up.
ESMO Open. 8:1016242023. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Hong Y, Duan P, He L, Li Q, Chen Y, Wang
P, Fu Y, Liu T and Ding Z: Systematic immunological level
determined the prognosis of leptomeningeal metastasis in lung
cancer. Cancer Manag Res. 14:1153–1164. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Le Rhun E, Nayak L, Lim-Fat MJ, Rudà R,
Pentsova E, Forsyth P, O'Brien BJ, Preusser M, Kumthekar P,
Brandsma D and Weller M: NANO-LM: An updated scorecard for the
clinical assessment of patients with leptomeningeal metastases.
Neuro Oncol. 27:455–465. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Zhang M, Tong J, Ma W, Luo C, Liu H, Jiang
Y, Qin L, Wang X, Yuan L, Zhang J, et al: Predictors of lung
adenocarcinoma with leptomeningeal metastases: A 2022
targeted-therapy-assisted molGPA model. Front Oncol. 12:9038512022.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Yin K, Li YS, Zheng MM, Jiang BY, Li WF,
Yang JJ, Tu HY, Zhou Q, Zhong WZ, Yang XN, et al: A molecular
graded prognostic assessment (molGPA) model specific for estimating
survival in lung cancer patients with leptomeningeal metastases.
Lung Cancer. 131:134–138. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Chen J, Pan L, Liu Y, Fang Y, Li R, Lu Z,
Liu A, He Y and Zeng Z: Intrathecal pemetrexed administration and
myelosuppression in patients with leptomeningeal metastases from
lung adenocarcinoma: A retrospective study. Oncol Res.
33:2107–2121. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Picarelli H, Yamaki VN, Solla DJF, Neville
IS, Santos AGD, Freitas BSAG, Diep C, Paiva WS, Teixeira MJ and
Figueiredo EG: The preoperative neutrophil-to-lymphocyte ratio
predictive value for survival in patients with brain metastasis.
Arq Neuropsiquiatr. 80:922–928. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Li Z, Zhang W and Wu S: Prognostic value
of pre-treatment neutrophil-to-lymphocyte ratio and
platelet-to-lymphocyte ratio in patients with brain metastasis from
lung cancer: A systematic review and meta-analysis. Altern Ther
Health Med. 30:108–113. 2024.
|
|
26
|
Wang JW, Yuan Q, Li L, Cao KH, Liu Q, Wang
HL, Hu K, Wu X and Wan JH: Role of systemic immunoinflammation
landscape in the overall survival of patients with leptomeningeal
metastases from lung cancer. Onco Targets Ther. 16:179–187. 2023.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Berghoff AS, Fuchs E, Richen G, Mlecnik B,
Bindea G, Spanberger T, Hackl M, Widhalm G, Dieckmann K, Prayer D,
et al: Density of tumor-infiltrating lymphocytes correlates with
extent of brain edema and overall survival time in patients with
brain metastases. Oncoimmunology. 5:e10573882015. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Hanahan D and Weinberg RA: Hallmarks of
cancer: The next generation. Cell. 144:646–674. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Ng MSF, Kwok I, Tan L, Shi C,
Cerezo-Wallis D, Tan Y, Leong K, Calvo GF, Yang K, Zhang Y, et al:
Deterministic reprogramming of neutrophils within tumors. Science.
383:eadf64932024. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Yu X, Li C, Wang Z, Xu Y, Shao S, Shao F,
Wang H and Liu J: Neutrophils in cancer: Dual roles through
intercellular interactions. Oncogene. 43:1163–1177. 2024.PubMed/NCBI
|
|
31
|
Zhou L, Zhang Z, Tian Y, Li Z, Liu Z and
Zhu S: The critical role of platelet in cancer progression and
metastasis. Eur J Med Res. 28:3852023. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Dolton G, Rius C, Wall A, Szomolay B,
Bianchi V, Galloway SAE, Hasan MS, Morin T, Caillaud ME, Thomas HL,
et al: Targeting of multiple tumor-associated antigens by
individual T cell receptors during successful cancer immunotherapy.
Cell. 186:3333–3349. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
He Y, Liu X, Wang M, Ke H and Ge C:
Neutrophil-to-lymphocyte ratio as a predictor of cardiovascular
mortality in cancer survivors. Sci Rep. 14:209802024. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Yu H, Tan L, Xue B, Feng L, Fang P, Meng X
and Luo X: Platelet-to-lymphocyte, neutrophil-to-lymphocyte and
lymphocyte-to-monocyte ratios are related to cancer-related fatigue
and quality of life in patients with cancer: A cross-sectional
study. BMJ Open. 13:e0753982023. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Barhour AB, Zaki P, McGranahan TM, Venur
V, Vellayappan B, Palmer J, Halasz LM, Yang JT, Blau M, Tseng YD,
et al: Emergent radiotherapy for brain and leptomeningeal
metastases: A narrative review. Ann Panlliat Med. 12:1405–1419.
2023. View Article : Google Scholar
|
|
36
|
Zhen J, Wen L, Lai M, Zhou Z, Shan C, Li
S, Lin T, Wu J, Wang W, Xu S, et al: Whole brain radiotherapy
(WBRT) for leptomeningeal metastasis from NSCLC in the era of
targeted therapy: A retrospective study. Radiat Oncol. 15:1852020.
View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Chiang CL, Yang HC, Luo YH, Chen CJ, Wu
HM, Chen YM, Hu YS, Lin CJ, Chung WY, Shiau CY, et al:
Leptomeningeal metastasis in patients with non-small cell lung
cancer after stereotactic radiosurgery for brain metastasis. J
Neurosurg. 139:385–392. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Liu X, Li G, Zhang H, Chang Q, Fang M, Lu
C, Tian P and Mei F: Molecular characteristics and prognostic
factors of leptomeningeal metastasis in non-small cell lung cancer.
Clin Neurol Neurosurg. 225:1075722023. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Tamura K, Yoshida T, Masuda K, Matsumoto
Y, Shinno Y, Okuma Y, Goto Y, Horinouchi H, Yamamoto N and Ohe Y:
Comparison of clinical outcomes of Osimertinib and first-generation
EGFR-tyrosine kinase inhibitors (TKIs) in TKI-untreated
EGFR-mutated non-small-cell lung cancer with leptomeningeal
metastases. ESMO Open. 8:1015942023. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Okabe T, Okamoto I, Tamura K, Terashima M,
Yoshida T, Satoh T, Takada M, Fukuoka M and Nakagawa K:
Differential constitutive activation of the epidermal growth factor
receptor in non-small cell lung cancer cells bearing EGFR gene
mutation and amplification. Cancer Res. 67:2046–2053. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Cho J, Chen L, Sangji N, Okabe T, Yonesaka
K, Francis JM, Flavin RJ, Johnson W, Kwon J, Yu S, et al: Cetuximab
response of lung cancer-derived EGF receptor mutants is associated
with asymmetric dimerization. Cancer Res. 73:6770–6779. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Chen H, Yang S, Wang L, Wu Y, Wu Y, Ma S,
He Z, Zhang C, Liu Y, Tang H, et al: High-dose furmonertinib in
patients with EGFR-mutated NSCLC and leptomeningeal metastases: A
prospective real-world study. J Thorac Oncol. 20:65–75. 2025.
View Article : Google Scholar : PubMed/NCBI
|