Introduction
Breast cancer remains a notable global health
concern. In 2022, it was the most frequently diagnosed cancer among
women worldwide, with ~2.3 million novel cases and 666,000
associated mortalities (1). The
early diagnosis of breast cancer is of notable importance, as it
can increase the 5-year survival rate to 96% (2). Breast magnetic resonance imaging (MRI)
demonstrates markedly increased efficacy in detecting lesions in
women who possess either a markedly elevated risk of breast cancer
or an average risk with dense breast tissue (3). Furthermore, the utilization of breast
MRI offers advantages in a variety of additional clinical
scenarios, such as the evaluation of tumor extent (3). A common finding in dynamic
contrast-enhanced breast MRI (DCE-MRI) is non-mass enhancement
(NME). It refers to an enhancing abnormality that is separate from
background parenchymal enhancement but lacks the 3-dimensional
volume, shape or margin characteristics to be described as a
mass.
The approach to NME presents notable challenges due
to the considerable overlap in imaging characteristics of benign
and malignant etiologies (4). A
recent study reported that 51.5% of NME cases were benign and 48.5%
were malignant (5). The
morphological evaluation of NME comprises two components. The first
component pertains to the distribution, which may be classified as
linear, focal, segmental, regional, involving multiple regions or
diffuse. The second component involves the internal enhancing
patterns, which can be categorized as homogeneous, heterogeneous,
clustered ring or clumped (6). The
dynamics of contrast enhancement serves as a key secondary
diagnostic tool (7). The initial
increase typically indicates the extent of tumor angiogenesis,
while the subsequent ‘delay’ phase is indicative of the development
of stromal tumor cells (7). Early
enhancement occurs within 2 min of agent injection. During this
time, the initial rise of the enhanced curve can be divided into
three categories: ‘Slow’, ‘medium’ and ‘rapid’. Rapid enhancement
is defined as an initial peak signal intensity (SI) >90% within
90 sec, which is a strong indicator of malignancy (6,7). The
‘delayed phase’ refers to the SI of 2 min post-contrast injection.
Based on the delayed phase, kinetic curves are categorized as
persistent enhancement (Type I), plateau (Type II) or washout (Type
III) (6).
While the diagnostic value of the delayed phase in
NME has been extensively studied (8–11), the
value of initial phase in such lesions is not generally recognized.
Therefore, the present study aimed to investigate the combined use
of initial- and delayed-phase kinetics and morphological features
to improve the differentiation between benign and malignant NME
lesions.
Patients and methods
Study design and population
The present study was a prospective study conducted
in the Oncology Teaching Hospital, Medical City (Baghdad, Iraq)
from April 2022 to December 2022. The study protocol was approved
by the Ethics Committee of the Institutional Review Board at the
College of Medicine, University of Baghdad (approval no. 1458;
Baghdad, Iraq).
Inclusion criteria were as follows: i) Consecutive
female patients referred for breast MRI, either for screening
purposes or for further characterization of a previously identified
lesion; ii) presence of a pure non-mass enhancement (NME) lesion on
MRI; iii) absence of any associated mass or focal mass-like
enhancement and iv) available image-guided core biopsy for
histopathological evaluation. Excisional biopsy was performed when
indicated by core biopsy findings or at the discretion of the
treating physician.
Exclusion criteria: 1- Patients without a definitive
histopathological diagnosis (e.g., biopsy not performed, inadequate
tissue sample). 2- Cases with borderline findings on core biopsy
(such as atypical ductal hyperplasia, lobular neoplasia, or other
indeterminate lesions) were excluded if no subsequent surgical
pathology report was available.3- Patients lacking sufficient
demographic or clinical information in medical records were
excluded from final analysis.
Histopathological diagnoses were classified
according to the World Health Organization (WHO) Classification of
Breast Tumours, 5th Edition (2019) (12).
Image analysis
For each area of NME the data collected are
according to the features mentioned in the 5th edition of Breast
Imaging Reporting and Data System (BI-RADS) 2013 (12).
Initially, subtracted images were reviewed to
identify the NME lesion and evaluate its morphological features,
including maximal size, distribution (focal, linear, segmental,
regional or diffuse) and internal enhancement pattern (homogeneous,
heterogeneous, clumped or clustered ring) and the dynamic curve.
For dynamic curve, two components were assessed: i) The initial
phase representing the SI observed within the first 2 min, divided
into 3 groups as per American College of Radiology BI-RADS 5th
edition: Slow, when <50% increase in SI, medium, when 50–100%
increase in SI and rapid, when >100% increase in SI occurs
within the first 2 min; and ii) the delayed phase represents the
enhancement pattern after 2 min or when the curve starts to change.
It is grouped as: Persistent, when there is continuous signal
increase >10% over time, plateau, when SI does not change over
time after its initial rise and remains flat, and washout, when SI
decreases >10% after its highest point from its initial rise
indicating a suspicious finding.
MRI imaging protocol
A superconducting 1.5T MR imaging device (Magnetom
Aera; Siemens Healthineers) was used for the breast MRI. All
patients were examined in the prone position by applying bilateral
16-channel breast coils and using scout view sagittal protocol
localization and T1-weighted pulses.
Fast spin-echo was utilized to collect axial non-fat
saturated T1-weighted imaging (T1WI) with the following image
parameters: i) Repetition time (TR), 426 msec; ii) echo time (TE),
4.6 msec; iii) slice thickness, 4 mm; iv) field of view (FOV),
380×380 mm; and v) matrix, 307×512. The settings for axial
non-fat-suppressed T2-weighted turbo spin-echo were TR, 6,030 msec;
TE, 71 msec; FOV, 400×400 mm; matrix, 384×512; and 4 mm slice
thickness.
To get axial short T2 Transverse Dixon fat and
water, the settings were TR, 9,440 msec; TE, 72 msec; slice
thickness, 3 mm; inter-slice gap, 1 mm; FOV, 450×450 mm and matrix,
307×512.
Dynamic contrast study was performed by applying
fluoroscence-3D T1WI spectral attenuated inversion recovery
sequence with injection of a 0.2 mmol/kg gadopentetate dimeglumine
bolus by an automated injector at 3–5 ml/sec via an 18–20 gauge IV
cannula in the antecubital vein. The parameters applied were: i)
TR, 5.08 msec; ii) TE, 2.39 msec; iii) 1.8 mm slice thickness; iv)
no inter-slice gap; v) FOV, 360×360 mm; and vi) matrix, 307×512.
Next, a 20 ml saline bolus infusion at 3–5 ml/sec was
administered.
The dynamic series consisted of one pre-contrast and
five post-contrast acquisitions; each repeated every 90 sec.
DCE-MRI analysis
Initially, the subtracted images of each examination
were evaluated by RadiAnt DICOM viewer 2022.2 (Medixant) to detect
the NME lesion and assess its size, distribution and the internal
enhancement pattern. Subsequently, the non-subtracted images with
pre-contrast T1W image and the four post-contrast images were
evaluated. A region of interest was manually placed on the area of
highest enhancement within the NME on the first post-contrast
series, then time-intensity plots of dynamic images were generated
using computer-assisted diagnosis software.
The percentage of the initial enhancement was
calculated as follows: (SIpost - SIpre/SIpre) ×100, where SIpre and
SIpost are the SIs on the pre- and first post-contrast images,
respectively (13).
Statistical analysis
The SPSS software for windows (version 26; IBM
Corp.) was used for all statistical analyses. Observational data
was presented in the form of frequencies and percentages.
Associations between categorical variables were assessed using the
χ2 or Fisher's exact test, as appropriate. To estimate
malignancy likelihood for lesions exhibiting a specific enhancement
pattern (Type I with initial rise), the positive predictive value
(PPV) was calculated as the proportion of test-positive cases that
were confirmed malignant. As sensitivity and specificity assess
overall MRI performance and not sub-classification, applying them
to Type I curves, typically benign, can misrepresent diagnostic
value. PPV further captures the significance of atypical malignant
presentations within this curve type. It reflects real-world
decision-making more accurately in this context. A two-sided
P<0.05 was considered to indicate a statistically significant
difference.
Results
Patient characteristics
A total of 38 patients met the enrollment criteria;
their ages ranged from 26 to 75 years, with a mean age of 45±11.45
years. A total of 12 patients (31.6%) were in the postmenopausal
period. The majority of the patients 24 (63.2%), presented with a
breast lump, whereas 10 individuals (26.3%) were scheduled for
screening. The initial mammography revealed microcalcifications in
12 patients (41.4%) and focal asymmetry in 17 patients (58.6%).
Furthermore, 7 patients (18.4%) were referred for further imaging
due to the presence of dense breast tissue. The definitive
histopathological diagnosis was corroborated through core biopsy in
6 cases (15.8%) and via surgical excision in 32 cases (84.2%).
Histopathological evaluation confirmed the presence of malignancy
in 26 cases, representing 68.4% of the total, while the remaining
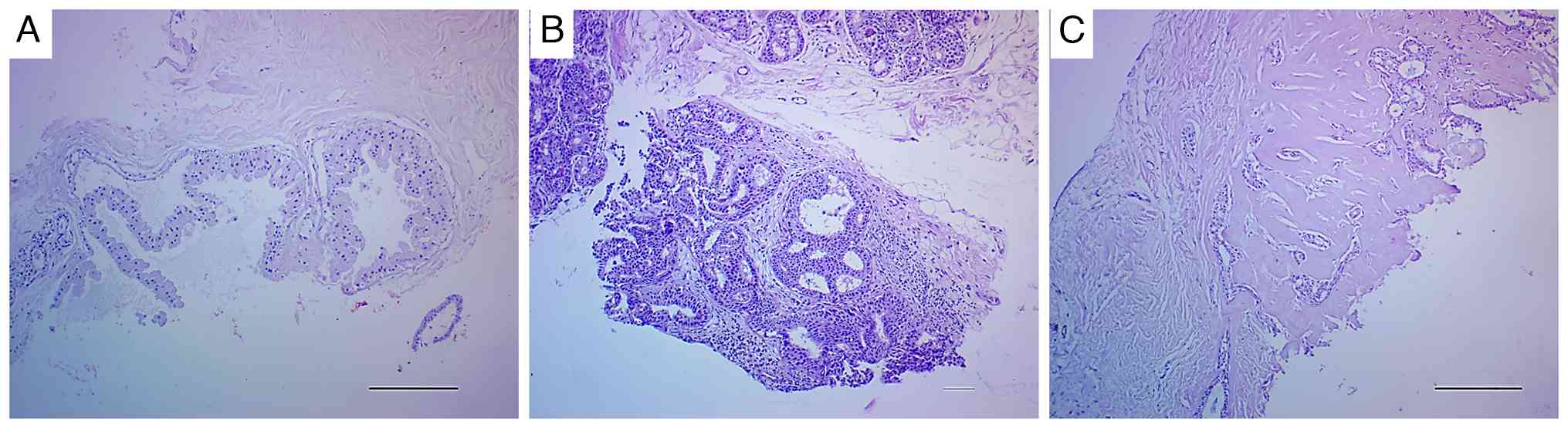
12 cases, accounting for 31.6%, were classified as benign (Fig. 1). Half of the benign lesions were
inflammatory (acute and granulomatous mastitis), 4 (33.3%)
exhibited fibrocystic changes (Fig.
1A), one of which exhibited atypical ductal hyperplasia and the
remaining 2 (16.7%) were benign intraductal papilloma. Among the
malignant lesions examined, 12 (46.2%) were classified as ductal
carcinoma in situ (DCIS; Fig.
1B), while invasive ductal carcinoma (IDC) accounted for 14
(53.8%; Fig. 1C). Details are
presented in Table I.
 | Table I.Patient characteristics. |
Table I.
Patient characteristics.
| Characteristic | n | Percentage (%) |
|---|
| Age, years |
|
|
|
<40 | 17 | 44.7 |
| ≥40 | 21 | 55.3 |
| Menopausal
status |
|
|
|
Premenopausal | 26 | 68.4 |
|
Postmenopausal | 12 | 31.6 |
| Presentation |
|
|
|
Screening | 10 | 26.3 |
| Lump | 24 | 63.2 |
| Nipple
discharge | 4 | 10.5 |
| Mammography |
|
|
|
Asymmetry | 17 | 58.6 |
|
Microcalcification | 12 | 41.4 |
| Dense
breast | 7 | 18.4 |
| Type of biopsy |
|
|
| Core
needle | 6 | 15.8 |
| Surgical
excision | 32 | 84.2 |
| Histopathology |
|
|
|
Benign | 12 | 31.6 |
|
Inflammation | 6 | 50.0 |
|
Fibrocystic changes | 3 | 25.0 |
|
Fibrocystic with atypical
ductal hyperplasia | 1 | 8.3 |
|
Intraductal papilloma | 2 | 16.7 |
|
Malignant | 26 | 68.4 |
| Ductal
carcinoma in situ | 12 | 46.2 |
| Invasive
ductal carcinoma | 14 | 53.8 |
MRI features of NME lesions and final
histopathology diagnosis
The homogeneous pattern of NME was significantly
more frequent in benign lesions (8/12; 66.7%) compared with that in
malignant lesions (8/26, 30.8%) (P=0.038). By contrast,
heterogenous and clumped patterns were more frequently observed in
malignant tumors 10 (38.5%) and 7 (26.9%), respectively compared
with 2 (16.7%) of benign lesions, although the difference was not
significant (P>0.05; Table II).
There was no significant difference between benign and malignant
tumors in terms of the enhancement distribution (P>0.05);
however, segmental enhancement was the most frequent enhancement
encountered in malignant lesion 10 (38.5%) with a PPV of 83.3%
compared with 2 (16.7%) of the benign lesions followed by the
regional type 9 (34.6%) with a PPV of 64.3%. When comparing
invasive and in situ carcinomas, regional enhancement was
significantly associated with IDC, occurring in 8 (57.1%) of IDC
cases vs. 1 (8.3%) of DCIS cases (P=0.014). The dynamic curve of
malignant lesions was significantly different compared with that of
the benign ones in terms of initial upslope and delay phase,
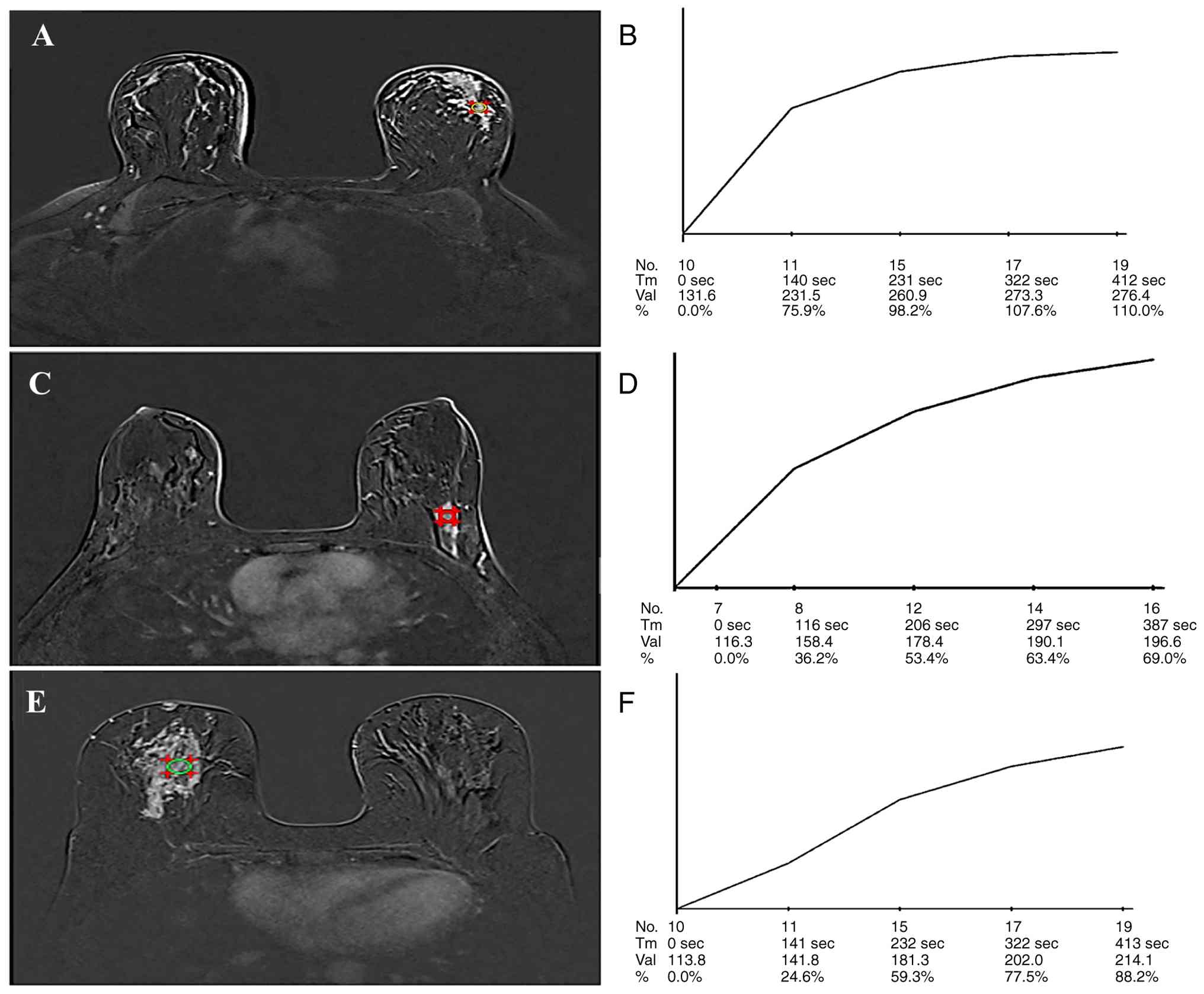
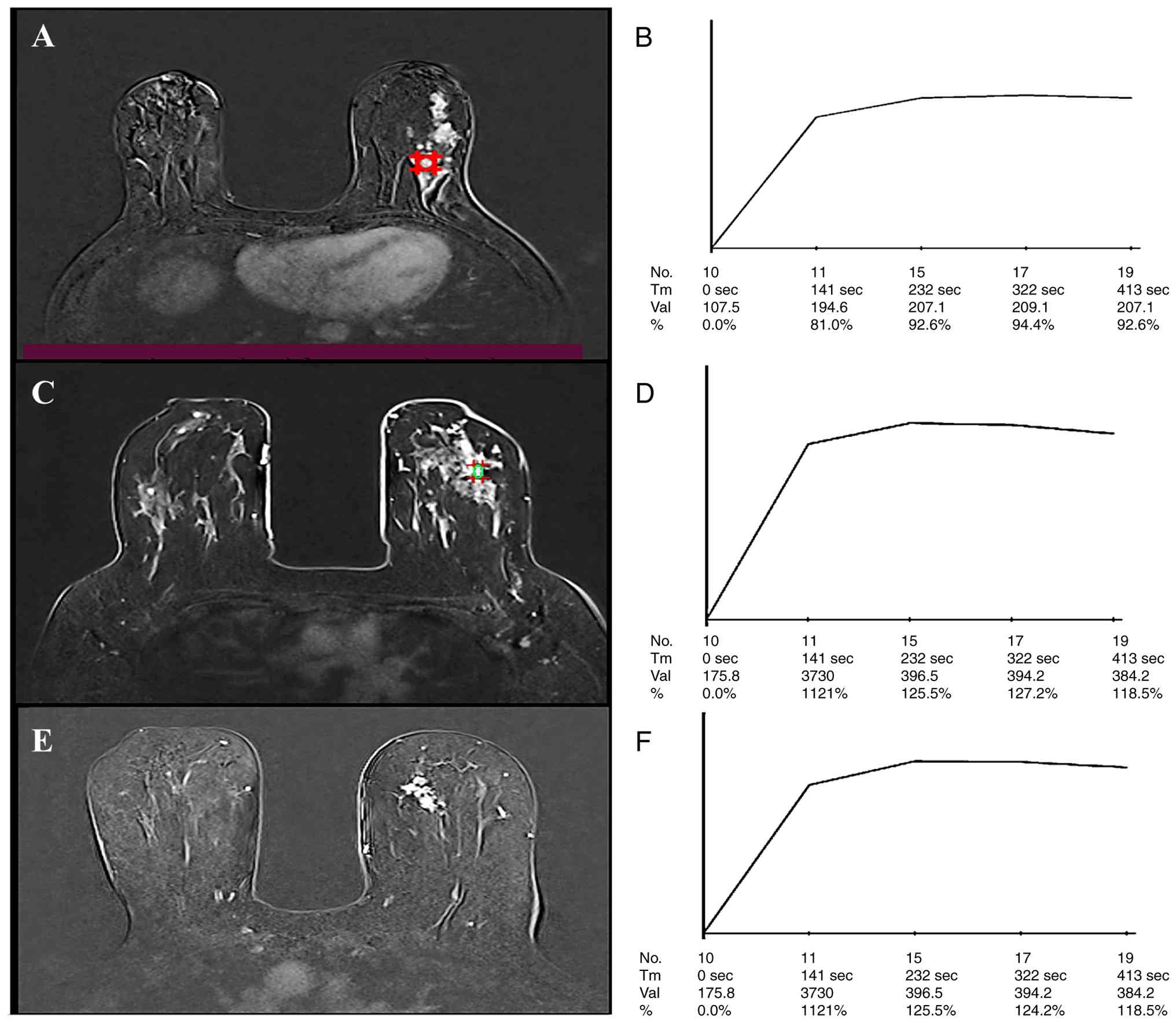
P=0.001 (Table III; Fig. 2, Fig.
3, Fig. 4). Benign lesions
exhibited slow initial upslope in 7 (58.3%) of the cases while five
(41.7%) have medium initial slope. By contrast, rapid initial slope
was the feature of 13 (50%) of malignant tumors (P=0.001; Fig. 3). A persistent delay phase was the
significant feature of 9 (75%) of the benign lesions compared with
only 7 (26.9%) malignant lesions (P=0.005; Fig. 2). None of the benign lesions
exhibited rapid initial or washout delay phase.
 | Table II.MRI of the non-mass enhancement
lesions and final histopathological outcome. |
Table II.
MRI of the non-mass enhancement
lesions and final histopathological outcome.
| A, Enhancement |
|---|
|
|---|
| Parameter | Malignant, n (%) | Benign, n (%) | P-value | PPV, % | DCIS, n (%) | IDC, n (%) | P-value |
|---|
| Homogenous | 8.0 (30.8) | 8.0 (66.6) | 0.037a | 50.0 | 5.0 (41.7) | 3.0 (21.4) | 0.401 |
| Heterogenous | 10.0 (38.5) | 2.0 (16.7) | 0.268 | 83.4 | 3.0 (25) | 7.0 (50) | 0.248 |
| Clumped | 7.0 (26.9) | 2.0 (16.7) | 0.689 | 77.8 | 3.0 (25) | 4.0 (28.6) | >0.999 |
| Cluster ring | 1.0 (3.80) | 0.0 (0.0) | >0.999 | 100.0 | 1.0 (8.3) | 0.0 | 0.462 |
|
| B,
Distribution |
|
| Parameter | Malignant, n
(%) | Benign, n
(%) | P-value | PPV, % | DCIS, n
(%) | IDC, n
(%) | P-value |
|
| Focal | 3.0 (11.5) | 2.0 (16.7) | 0.643 | 60.0 | 3.0 (25.0) | 0.0 | 0.085 |
| Linear | 1.0 (3.9) | 2.0 (16.7) | 0.229 | 33.3 | 1.0 (8.3) | 0.0 | 0.462 |
| Segmental | 10.0 (38.5) | 2.0 (16.7) | 0.268 | 83.3 | 5.0 (41.7) | 5.0 (35.7) | >0.999 |
| Regional | 9.0 (34.6) | 5.0 (41.6) | 0.675 | 64.3 | 1.0 (8.3) | 8.0 (57.1) | 0.014a |
| Diffuse | 3.0 (11.5) | 1.0 (8.3) | >0.999 | 75.0 | 2.0 (16.7) | 1.0 (7.1) | 0.580 |
 | Table III.MRI dynamic curve of non-mass
enhancement lesions and final histopathological outcome. |
Table III.
MRI dynamic curve of non-mass
enhancement lesions and final histopathological outcome.
| Parameter | Malignant, n
(%) | Benign, n (%) | P-value | PPV, % |
|---|
| Initial
upslope |
|
| 0.001a |
|
| Slow,
<50% | 3.0 (11.5) | 7.0 (58.3) |
| 30.0 |
| Medium,
50–100% | 10.0 (38.5) | 5.0 (41.7) |
| 66.7 |
| Rapid,
>100% | 13.0 (50.0) | 0.0 |
| 100.0 |
| Delay phase |
|
| 0.005a |
|
| Type
I | 7.0 (26.9) | 9.0 (75.0) |
| 46.8 |
| Type
II | 9.0 (34.6) | 3.0 (25.0) |
| 75.0 |
| Type
III | 10.0 (38.5) | 0.0 |
| 100.0 |
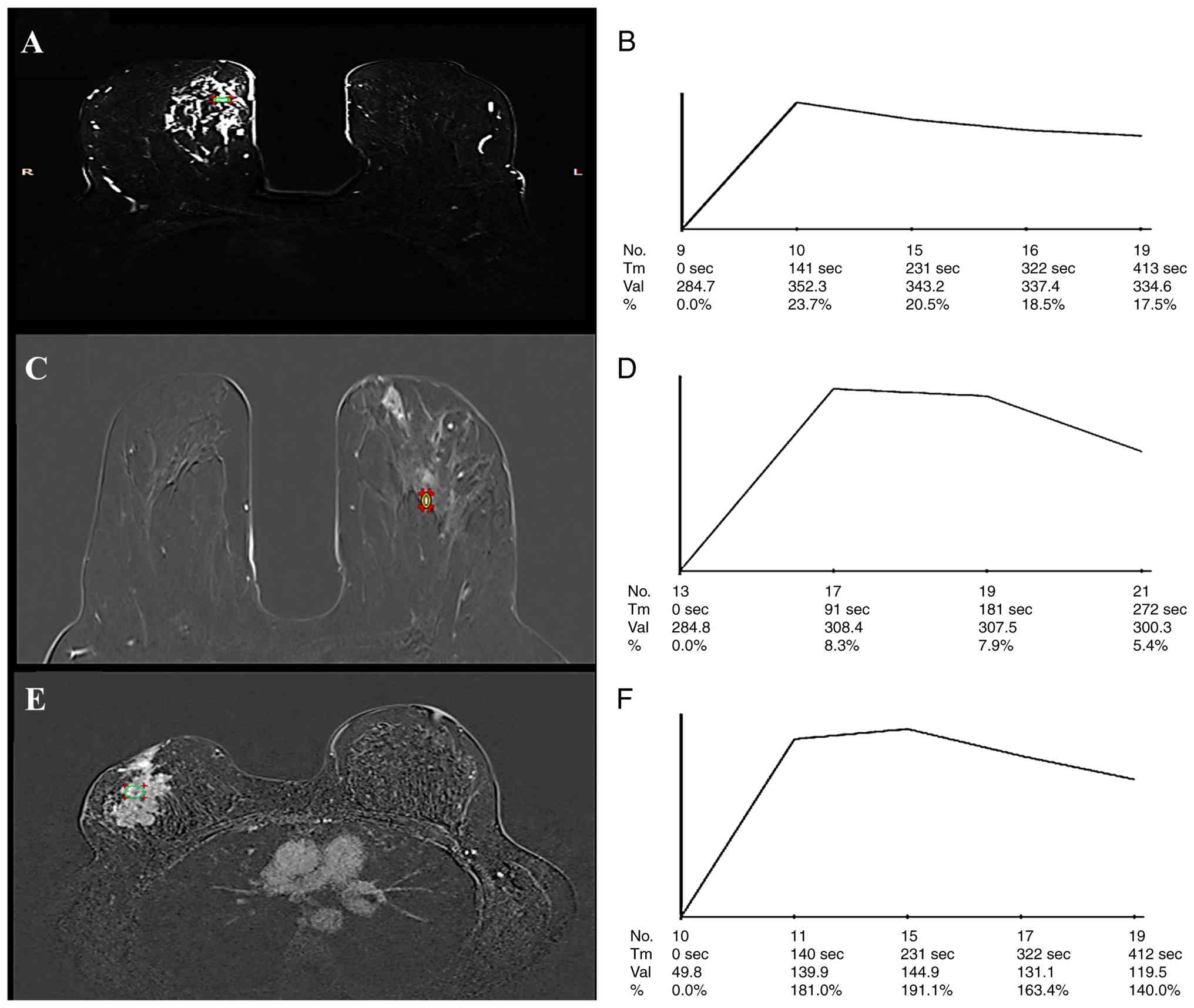
To further evaluate the 16 cases with persistent
delay phase, kinetic analysis of the initial phase was performed.
Among the benign lesions, a slow initial phase was observed in 6
cases (66.7%), while 3 (33.3%) exhibited medium initial phase.
However, a slow initial phase was also observed in 3 (42.9%) and a
medium in 4 (57.1%) of the malignant lesions (P=0.615) (Table IV; Fig.
2C and D). Among the 12 lesions with a plateau delayed phase, 1
out of the 3 benign lesions exhibited slow initial phase (Fig. 3A and B; however, none of the
malignant lesions depicted slow initial phase, yet the difference
was not significant (P=0.159; Table
IV).
 | Table IV.Initial upslope stratified in Type I
and II curve and final histopathological outcome. |
Table IV.
Initial upslope stratified in Type I
and II curve and final histopathological outcome.
| Parameter | Malignant, n
(%) | Benign, n (%) | P-value |
|---|
| Type I cases
(n=16) |
|
| 0.615 |
| Slow,
<50% | 3.0 (42.9) | 6.0 (66.7) |
|
| Medium,
50–100% | 4.0 (57.1) | 3.0 (33.3) |
|
| Rapid,
>100% | 0.0 | 0.0 |
|
| Type II cases
(n=12) |
|
| 0.159 |
| Slow,
<50% initial | 0.0 | 1.0 (33.3) |
|
| Medium,
50–100% initial | 4.0 (44.4) | 2.0 (66.7) |
|
| Rapid,
>100% initial | 5.0 (55.6) | 0.0 |
|
Combining initial phase to delay phase
curve enhances the PPV
As illustrated in Table
V, the PPV of the persistent curve is relatively notably high
at 43.8%. However, the PPV was diminished by ~10% (33.3%) upon the
inclusion of the initial phase upslope of <50%. By contrast,
adding initial upslope >50% to plateau curve increased the PPV
from 75 to 81.8%. Although the plateau curve achieved 100% PPV
enhanced with the addition of initial upslope >100% (Table V), the detection rate was 5 of 9
malignant cases confirmed by histopathology in this category
(Table III).
 | Table V.PPV of dynamic curve and with and
without including the initial curve slope cut-off 50 and 100%. |
Table V.
PPV of dynamic curve and with and
without including the initial curve slope cut-off 50 and 100%.
| Phase | MRI dynamic | Total no. | PPV, % |
|---|
| Delay | Type I | 16 | 43.8 |
|
| Type II | 12 | 75.0 |
|
| Type III | 10 | 100.0 |
| Initial, % | <50 | 10 | 30.0 |
|
| 50-100 | 15 | 66.7 |
|
| >100 | 13 | 100.0 |
| Combined | Type I and <50%
initial | 9 | 33.3 |
|
| Type II and >50%
initial | 11 | 81.8 |
|
| Type II and
>100% initial | 5 | 100.0 |
Discussion
In the present study, the MRI features of NME
lesions were evaluated with a particular focus on the initial and
delayed phases of the DCE kinetic curve. Although previous research
has extensively explored the role of enhancement patterns and
distribution in predicting malignancy (8–11),
integrated analysis of early and delayed kinetic phases remains
inadequately investigated. Of 38 NME lesions, malignancy was
verified in 26 (68.4%), with nearly half (46.2%) classified as
in situ tumors. Segmental enhancement distribution was the
main finding of malignant tumors, which accounted for 10 cases
(38.5%) with a PPV of 83.3% consistent with previous studies
reporting PPV values ranging from 67 to 100% (10,14).
The regional distribution was the predominant
pattern overall (14/38, 36.8% of NME lesions) and ranked second
among malignancies (34.6%). Notably, this distribution was
significantly associated with IDCs, occurring in 57.1% of IDCs vs.
only 8.3% of DCIS cases.
DCIS-associated NME typically exhibited segmental or
linear patterns, reflecting ductal system involvement, often with
clumped or heterogeneous enhancement (15). Consistent with this finding,
segmental distribution was the most frequent feature of DCIS lesion
in the present study, 5 (41.7%), followed by focal 3 (25%).
Homogeneous enhancement was significantly more common in benign
lesions (66.7% vs. 30.8% in malignancies).
Nonetheless, 41.7% of DCIS cases also displayed
homogeneous enhancement, highlighting the overlap between
low/intermediate-grade DCIS and benign lesions. This aligns with
the study by Yoon et al (15), which reported homogeneous
enhancement in 33.3% of DCIS cases.
Both the initial and delayed kinetic phases
exhibited significant disparities between malignant and benign
lesions. Malignancies predominantly exhibited a Type III (washout)
curve, whereas benign lesions typically demonstrated a Type I
(persistent) curve, consistent with findings by Yang et al
(8), Liu et al (9) and Aydin (10).
The washout curve attained 100% PPV while the
plateau curve reached 75%. Bluemke et al (16) reported that plateau and washout
patterns were 63.2% sensitive and 65.4% specific for malignancy
(16). However, the interpretation
of Type II (plateau) curves remains controversial. Although certain
studies associate them with malignancy, Zhou et al (11) reported that Type II curves in NME
were more frequent in benign papillary neoplasms; the present study
data suggested that integrating the initial upslope magnitude can
refine diagnostic accuracy. The present study identified that
combining type I curve with slow initial phase (<50%) reduced
the PPV by ~10% while combining type II curve with initial >50%
increased the PPV from 75 to 81.8%. A Type II curve with an initial
upslope >100% achieved 100% PPV; however, this resulted in
diminished sensitivity. These findings suggest that incorporating
the initial upslope could enhance malignancy prediction in NME
lesions. However, larger studies are warranted to validate these
observations and assess their clinical utility in the future.
The main limitation of the present study was the
limited number of enrolled cases and the loss of cases during
follow-up. As a single-center, proof-of-concept study, the primary
aim was not to provide final validation but to identify and define
promising MRI criteria (such as the combined ‘II and >100%
initial’ feature) under controlled, expert conditions. A
multi-center validation is warranted to assess the generalizability
of these criteria across radiologists of diverse experience levels
and MRI platforms. Furthermore, the present study results provided
key preliminary data necessary to power future studies. Based on
the observed effect sizes in the present study (for example, ~100
vs. ~33% PPV between groups), a sample size calculation for a
future validation study can be performed.
In conclusion, integration of initial upslope
kinetics alongside conventional delayed-phase analysis and
morphological features (for example, distribution and internal
enhancement) may improve diagnostic accuracy for malignant NME
lesions, potentially reducing unnecessary biopsies in the
future.
Acknowledgements
Not applicable.
Funding
Funding: No funding was received.
Availability of data and materials
The data generated in the present study may be
requested from the corresponding author.
Authors' contributions
TFK conceived the research idea for the present
study. TFK and MAN planned the present study and collected the
cases. AMK analyzed the data and drafted the manuscript. TFK
conceptualized and designed the present study, performed the
literature search, identified clinical studies, conducted the data
and statistical analysis, prepared and reviewed the manuscript. AMK
conceptualized and designed the present study, conducted the data
analysis, prepared and reviewed the manuscript. MAN prepared and
reviewed the manuscript. TFK and AMK confirm the authenticity of
all the raw data. All authors reviewed and edited the manuscript.
All authors read and approved the final version of the
manuscript.
Ethics approval and consent to
participate
The Ethics Committee of the Institutional Review
Board at the College of Medicine, University of Baghdad, provided
approval for the present study (approval no. 1458; Baghdad, Iraq).
Written informed consent was obtained from each patient for
participation in the present study and for the use of their samples
in scientific research, in accordance with the Declaration of
Helsinki.
Patient consent for publication
A statement of consent for publication was signed by
each patient according to the principles of the Declaration of
Helsinki.
Competing interests
The authors declare that they have no competing
interests.
Authors' information
ORCID: AK, 0000-0001-5239-9699.
References
|
1
|
Bray F, Laversanne M, Sung H, Ferlay J,
Siegel RL, Soerjomataram I and Jemal A: Global cancer statistics
2022: GLOBOCAN estimates of incidence and mortality worldwide for
36 cancers in 185 countries. CA Cancer J Clin. 74:229–263.
2024.PubMed/NCBI
|
|
2
|
Sree SV, Ng EY, Acharya RU and Faust O:
Breast imaging: A survey. World J Clin Oncol. 2:171–178. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Wekking D, Porcu M, De Silva P, Saba L,
Scartozzi M and Solinas C: Breast MRI: clinical indications,
recommendations, and future applications in breast cancer
diagnosis. Curr Oncol Rep. 25:257–267. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Gargiulo M, Dien E, Gal J, Schiappa R,
Elkind L and Lamarque M: Predictive factors for non-mass
enhancement occult in conventional breast imaging: The ‘PAMAS’
study. Eur J Radiol. 184:1119622025. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Mohamed S, Elhamd EA and Attia NM:
Non-mass enhancement on breast MRI: Clues to a more confident
diagnosis. Egypt J Radiol Nucl Med. 55:872024. View Article : Google Scholar
|
|
6
|
Spak DA, Plaxco JS, Santiago L, Dryden MJ
and Dogan BE: BI-RADS® fifth edition: A summary of
changes. Diagn Interv Imaging. 98:179–190. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Cheng L, Bai Y, Zhang J, Liu M, Li X,
Zhang A, Zhang X and Ma L: Optimization of apparent diffusion
coefficient measured by diffusion-weighted MRI for diagnosis of
breast lesions presenting as mass and non-mass-like enhancement.
Tumour Biol. 34:1537–1545. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Yang QX, Ji X, Feng LL, Zheng L, Zhou XQ,
Wu Q and Chen X: Significant MRI indicators of malignancy for
breast non-mass enhancement. J Xray Sci Technol. 25:1033–1044.
2017.PubMed/NCBI
|
|
9
|
Liu G, Li Y, Chen SL and Chen Q: Non-mass
enhancement breast lesions: MRI findings and associations with
malignancy. Ann Transl Med. 10:3572022. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Aydin H: The MRI characteristics of
non-mass enhancement lesions of the breast: Associations with
malignancy. Br J Radiol. 92:201804642019. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Zhou J, Li M, Liu D, Sheng F and Cai J:
Differential diagnosis of benign and malignant breast papillary
neoplasms on MRI with non-mass enhancement. Acad Radiol. 30 (Suppl
2):S127–S32. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
D'Orsi C, Sickles E, Mendelson E and
Morris EA: ACR BI-RADS Atlas: Breast Imaging Reporting and Data
System. American College of Radiology; Reston, VA: 2013
|
|
13
|
Cheng L and Li X: Breast magnetic
resonance imaging: Kinetic curve assessment. Gland Surg. 2:50–53.
2013.PubMed/NCBI
|
|
14
|
Asada T, Yamada T, Kanemaki Y, Fujiwara K,
Okamoto S and Nakajima Y: Grading system to categorize breast MRI
using BI-RADS 5th edition: A statistical study of non-mass
enhancement descriptors in terms of probability of malignancy. Jpn
J Radiol. 36:200–208. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Yoon GY, Choi WJ, Cha JH, Shin HJ, Chae EY
and Kim HH: The role of MRI and clinicopathologic features in
predicting the invasive component of biopsy-confirmed ductal
carcinoma in situ. BMC Med Imaging. 20:952020. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Bluemke DA, Gatsonis CA, Chen MH,
DeAngelis GA, DeBruhl N, Harms S, Heywang-Köbrunner SH, Hylton N,
Kuhl CK, Lehman C, et al: Magnetic resonance imaging of the breast
prior to biopsy. JAMA. 292:2735–2742. 2004. View Article : Google Scholar : PubMed/NCBI
|