Introduction
Peripheral blood eosinophilia is a rare condition in
patients with solid tumors, with an overall incidence of only
0.5–1.0% (1). The condition occurs
predominantly in colorectal, breast, ovarian, oral cavity and
Hodgkin's lymphomas (2), and has
rarely been reported in lung cancers, especially squamous lung
cancer. Eosinophils are non-differentiated leukocytes derived from
hematopoietic stem cells in the bone marrow, which serve roles in
immunomodulation, mediating tissue repair, maintaining the
stability of the internal environment and killing parasites
(3). High levels of eosinophils in
lung cancer are closely associated with decreased survival time and
increase the risk of the metastasis of lung cancer (4). Typically, eosinophilia of a
paraneoplastic nature can be diagnosed by excluding non-cancerous
factors, such as allergies and parasitic infections. Most patients
do not exhibit obvious clinical manifestations; however, the
persistent elevation of eosinophil levels is likely to lead to
eventual multi-organ damage.
Treatment of eosinophilia of a paraneoplastic nature
usually involves tumor resection, chemotherapy or corticosteroids.
The present report describes a case of eosinophilia in a
25-year-old female patient with squamous lung cancer. The lung
cancer suddenly deteriorated with a sudden rise in peripheral blood
eosinophil levels that did not cause significant clinical symptoms.
Glucocorticosteroids in combination with antitumor drugs were
administered to counteract the condition and prevent target organ
damage, and the peripheral blood eosinophil levels showed marked
improvement. The present report discusses the various factors that
trigger eosinophilia and the potential role of eosinophils in solid
tumors to provide a reference for the early diagnosis and treatment
of solid tumors associated with eosinophilia in the future.
Case report
A 25-year-old woman who presented with a cough and
bloody sputum without obvious triggers was admitted to The First
Affiliated Hospital of Hebei University of Traditional Chinese
Medicine (Shijiazhuang, China) in December 2023. Poorly
differentiated squamous cell carcinoma of the lower lobe of the
right lung was diagnosed after a biopsy was obtained by
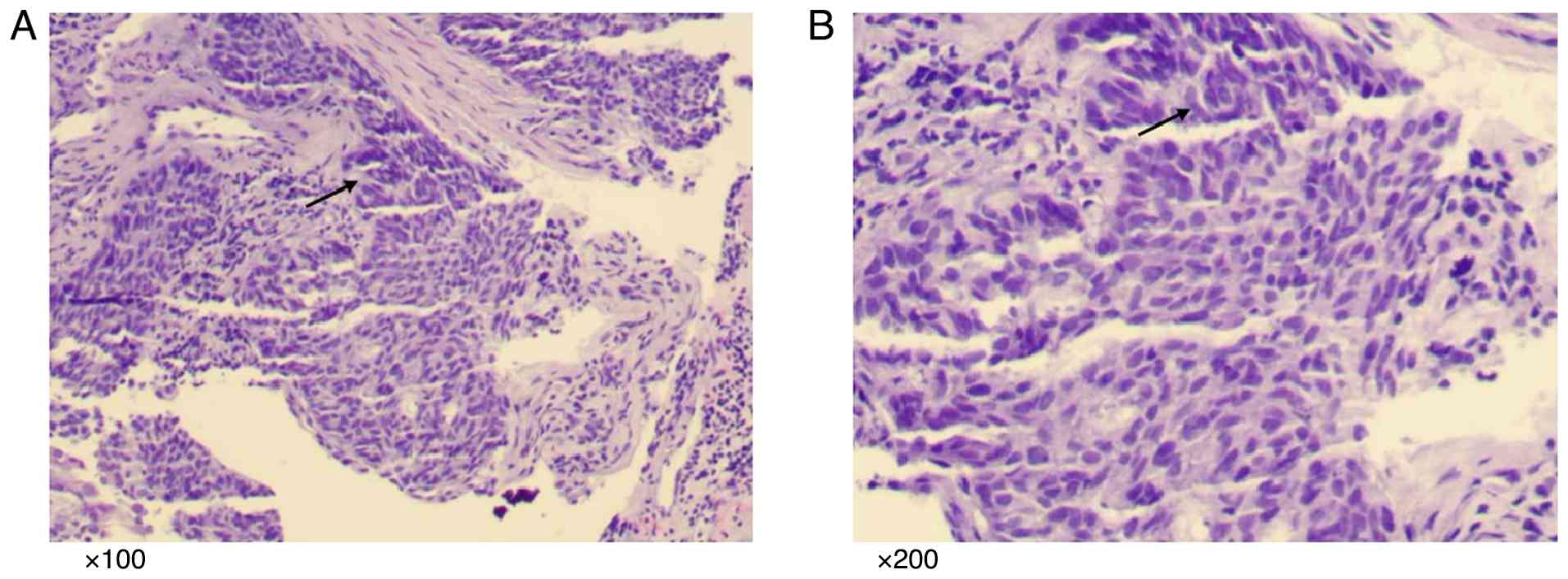
bronchoscopy. The lung histology showed a large number of
abnormally proliferated squamous cells with large and oval nuclei,
deeply stained nuclear chromatin, prominent nucleoli and
keratinization (Fig. 1; Data S1). Immunohistochemistry (Data S1) revealed the following results:
Thyroid transcription factor 1(−), NapsinA(−), AE1/AE3(+),
Vimentin(−), p40(+), cytokeratin 5/6(+), CD56(−), Synaptophysin(−),
Chromogranin A(−), CD99(+) and CD117(−) (data not shown).
Next-generation sequencing (NGS) (Data
S1) was performed on Illumina NextSeq550/NovaSeq6000 platform,
and the reference genome was GRCh37/hg19. The detection targets
include 10 core genes related to lung cancer, including single
nucleotide variation, small fragment insertion or deletion, copy
number variation and gene fusion of ERBB2 and other genes, which
can be used to assist prognosis evaluation and predict the efficacy
of targeted drugs. The results showed that the patient had a
c.2369C>T (p.T790M) mutation in the EGFR gene, and the mutation
frequency was 53.51%. Lymph nodes and distant metastases were not
detected at that time. The patient had no previous underlying
diseases, and had no history of allergies, blood transfusions or
contact with infected water, as well as no known family genetic
history. Following the detailed evaluation of the disease by the
doctor and the development of a treatment plan, the patient
received immunotherapy combined with chemotherapy for a total of 6
cycles (0.2 g tislelizumab on day 1 + 0.2 g nab-paclitaxel on day 2
+ 0.3 g carboplatin on day 2, every cycle of 21 days) at The First
Affiliated Hospital of Hebei University of Traditional Chinese
Medicine between January and April 2024. The condition of the
patient was deemed stable in May 2024, and the regimen was adjusted
to a total of three cycles of treatment with 0.2 g tislelizumab and
0.5 g carboplatin. In August 2024, an evaluation of disease
progression revealed the need for additional chemotherapy.
Nab-paclitaxel (0.2 g) were added for a total of two cycles. In
October 2024, the regimen consisted of 0.2 g tislelizumab, 30 mg
cisplatin, and 0.2 g nab-paclitaxel, administered for one cycle. In
November 2024, treatment was continued with 200 mg tislelizumab +
1.4 g gemcitabine + 400 mg carboplatin for one cycle.
In mid-December 2024, the patient was readmitted to
the hospital due to complaints of marked posterior back pain,
generalized weakness and a dry cough. Laboratory tests after
admission showed varying degrees of elevation of tumor markers as
follows: Carcinoembryonic antigen, 14.80 ng/ml (normal value,
<3.4 ng/ml); ferritin, 215.00 ng/ml (normal value, 13–150
ng/ml); glycan antigen 125, 65.70 U/ml (normal value, ≤35 U/ml);
neuron-specific enolase, 20.70 ng/ml (normal value, <16.3
ng/ml); and cytokeratin 19 fragment, 14.30 ng/ml (normal value,
<3.3 ng/ml). The blood routine showed a white blood cell count
of 14.10×109/l (normal value,
3.50–9.50×109/l), an eosinophil percentage (EOS%) of
52.8% (normal value, 0.4–8.0%), an absolute eosinophil value of
7.44×109/l (normal value, 0.02–0.52×109/l), a
platelet count of 508×109/l (normal value,
125–350×109/l), a quantitative D-dimer level of 1,195
µg/l (normal value, ≤243 µg/l) and an alkaline phosphatase level of
120 U/l (normal value, 35–100 U/l). No abnormalities were observed
in the fecal routine.
As early as the chest CT scan in January 2024, a
soft-tissue mass measuring 3.2×2.5 cm was detected in the lower
lobe of the right lung (Fig. 2A and
D). However, CT scans of the chest in December 2024 (size,
10×8.4×5.6 cm; Fig. 2C and F)
showed that the malignant space in the lower lobe of the right lung
was markedly enlarged compared with the previous scan in October
2024 (size, 5.2×3.5 cm; Fig. 2B and
E), with multiple ground-glass density nodules of varying sizes
in both lungs (the largest diameter of which was ~1.1 cm), multiple
enlarged lymph nodes in the right hilar and mediastinal regions,
and occlusion of the right lower lung bronchus with obstructive
inflammation, as well as thickening of the right lower pleural
membrane. Bone destruction of the seventh thoracic vertebra with
pathological fracture suggested bone metastasis. CT scans of the
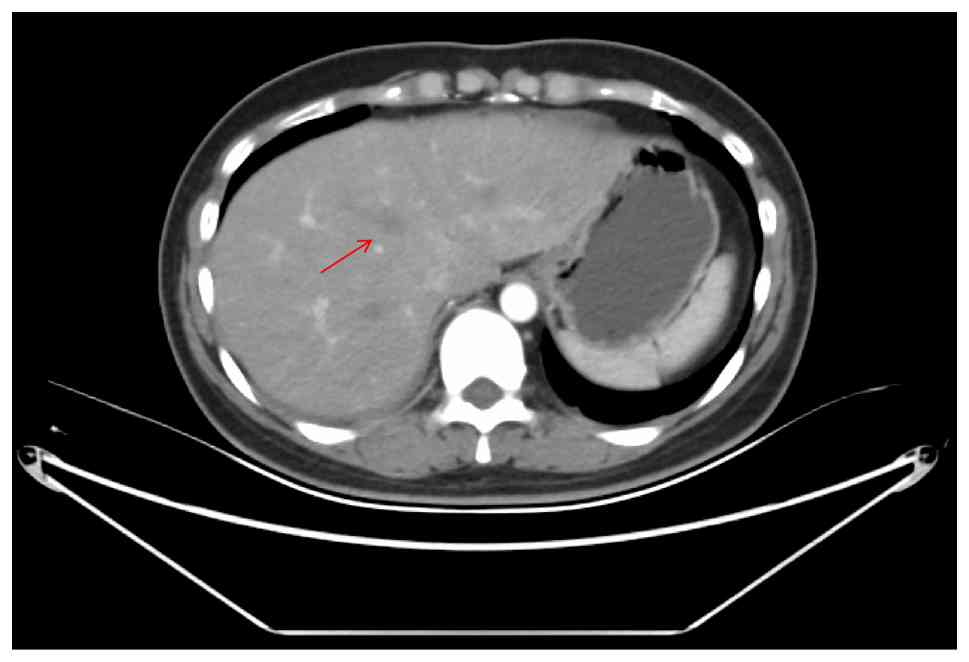
abdomen cavity (Fig. 3) showed
multiple new lesions in the liver (the largest of which was ~2.7 cm
in diameter), suggesting metastases. The imaging suggested that the
condition of the patient was in a deteriorating state.
In response to the severe pain caused by bone
metastases in the back of the patient, ibandronate injection (5 mg
intravenously) was administered to counteract the bone metastases.
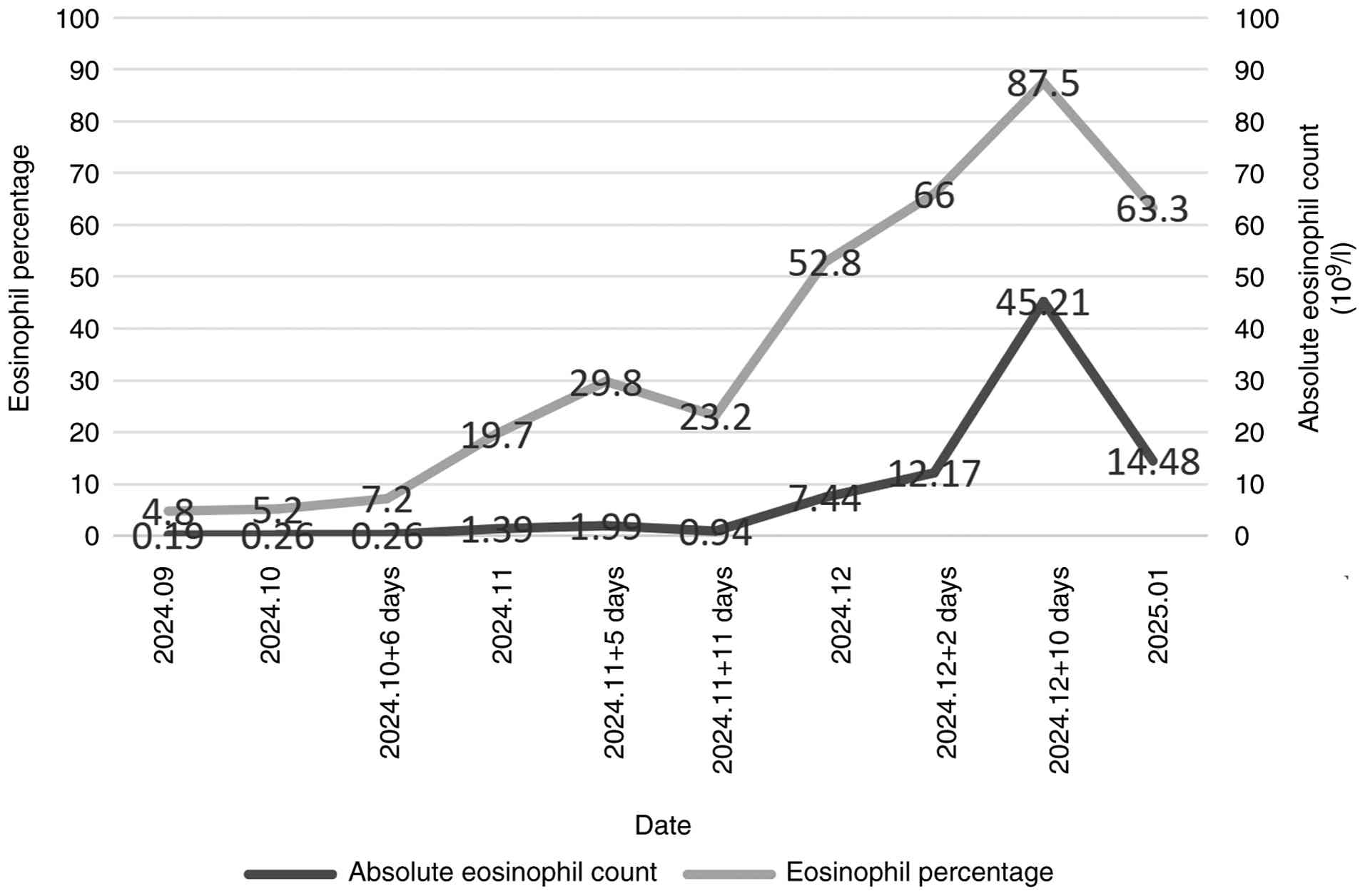
However, 10 days after admission, peripheral blood eosinophil
levels were markedly elevated, with an EOS% of 87.5% and an
absolute eosinophil value of 45.21×109/l. A blood smear
revealed 87% eosinophils, with these cells readily visible (data
not shown). An immediate review of previous test results showed
that the peripheral blood eosinophil count had fluctuated between
1.39 and 45.21×109/l during the last 2 months, with an
overall increasing trend (Fig. 4).
Based on the aforementioned observations, a joint consultation with
the Departments of Respiratory Medicine and Rheumatology was
immediately conducted, and measurements of immunoglobulin E (IgE),
rheumatoid factor, cytomegalovirus antibody, antinuclear antibody
(ANA) and antibody against systemic vasculitis were performed,
which did not show any significant abnormalities. However, the
serum total IgE level was high (79.61 IU/ml; normal value, 0–60
IU/ml). The patient was advised to undergo a bone marrow aspiration
to rule out hematological diseases. The patient's family refused to
allow this invasive operation in consideration of the physical
tolerance of the patient, and thus, evidence of leukemia could not
be obtained for the time being.
In order to avoid organ damage caused by
eosinophilia, intravenous methylprednisolone sodium succinate (20
mg; injection time, 30 min) was immediately administered for
anti-inflammatory treatment. After evaluating the condition and
tolerance of the patient, the patient was also administered
nab-paclitaxel (0.2 g) chemotherapy to treat the tumor,
diphenhydramine hydrochloride injection (20 mg) to prevent allergic
reactions before chemotherapy and tropisetron hydrochloride
injection (5 mg) as an antiemetic. Electrocardiogram, blood
pressure and blood oxygen test results were closely monitored.
During the past almost 1 year of treatment, the tumor control of
the patient had been relatively stable, but the chest CT showed
that the tumor was rapidly deteriorating. Coincidentally, the stage
of the peripheral blood eosinophilia elevation coincided with the
time of tumor deterioration (October-December 2024), and it was
hypothesized that eosinophilia was involved in the progression of
the lung cancer.
After 1 month, the patient developed numbness and
discomfort below the waist, weakness of both lower limbs and
limitations to lifting. To clarify the changes in the condition of
the patient, positron emission tomography/CT (PET/CT) was
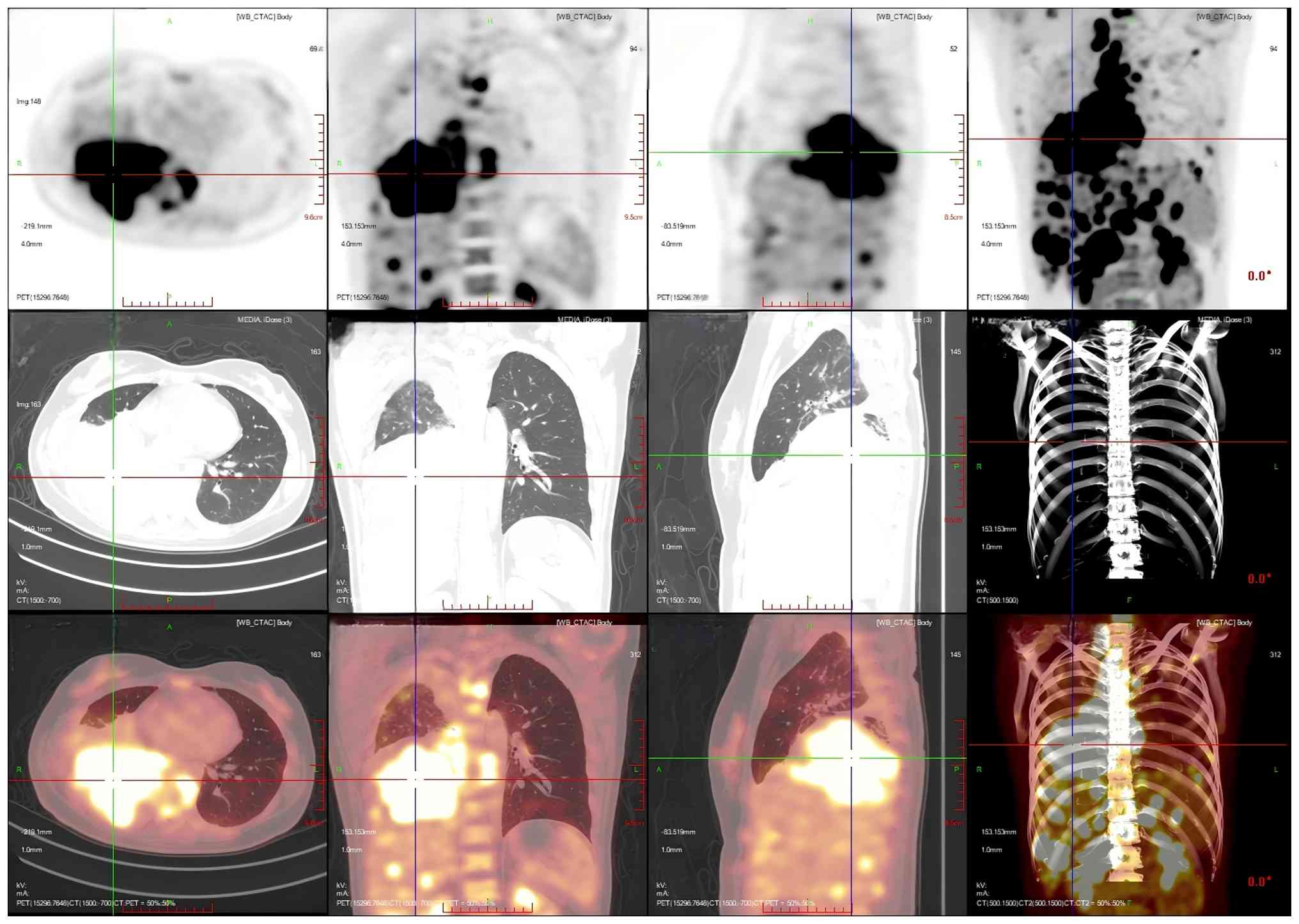
performed. PET/CT images showed that an irregular mass-like
hyperdense shadow was detected in the right lung hilar and the
middle and lower lobes of the right lung; no normal lung tissue was
seen, the lesion crossed the interlobular pleura and invaded the
upper lobe of the right lung, and the corresponding area on PET
showed a mass-like radiolucency focus with an maximum standardized
uptake value (SUVmax) of 25.8 (Fig. 5). Multiple enlarged and swollen
lymph nodes were detected in the mediastinum, bilateral pulmonary
hilar region, bilateral diaphragmatic pedicle and right cardiogenic
angle area, partially fused in the form of a mass. PET showed a
mass of radiolucent foci in the corresponding areas, and the
SUVmax of the hypermetabolic area under the bronchus was
measured to be 21.4. Thickening of the right pleura was observed,
an arcuate fluid density shadow was seen in the thoracic cavity and
mild radiolucent uptake was visible in the area of pleural effusion
on the right side of the chest cavity (Fig. 6). The liver was full, with diffuse
low-density nodules and masses of varying sizes within it, and PET
showed nodular and mass-like radioactivity uptake in the
corresponding areas. The low-density mass in the S4 segment of the
liver was measured to be ~5.0×4.4 cm in size, with an
SUVmax of 20.0. Several enlarged lymph nodes were
observed in the portal region of the liver, which had fused to form
a mass. PET scans showed mass-like radioactivity uptake in the
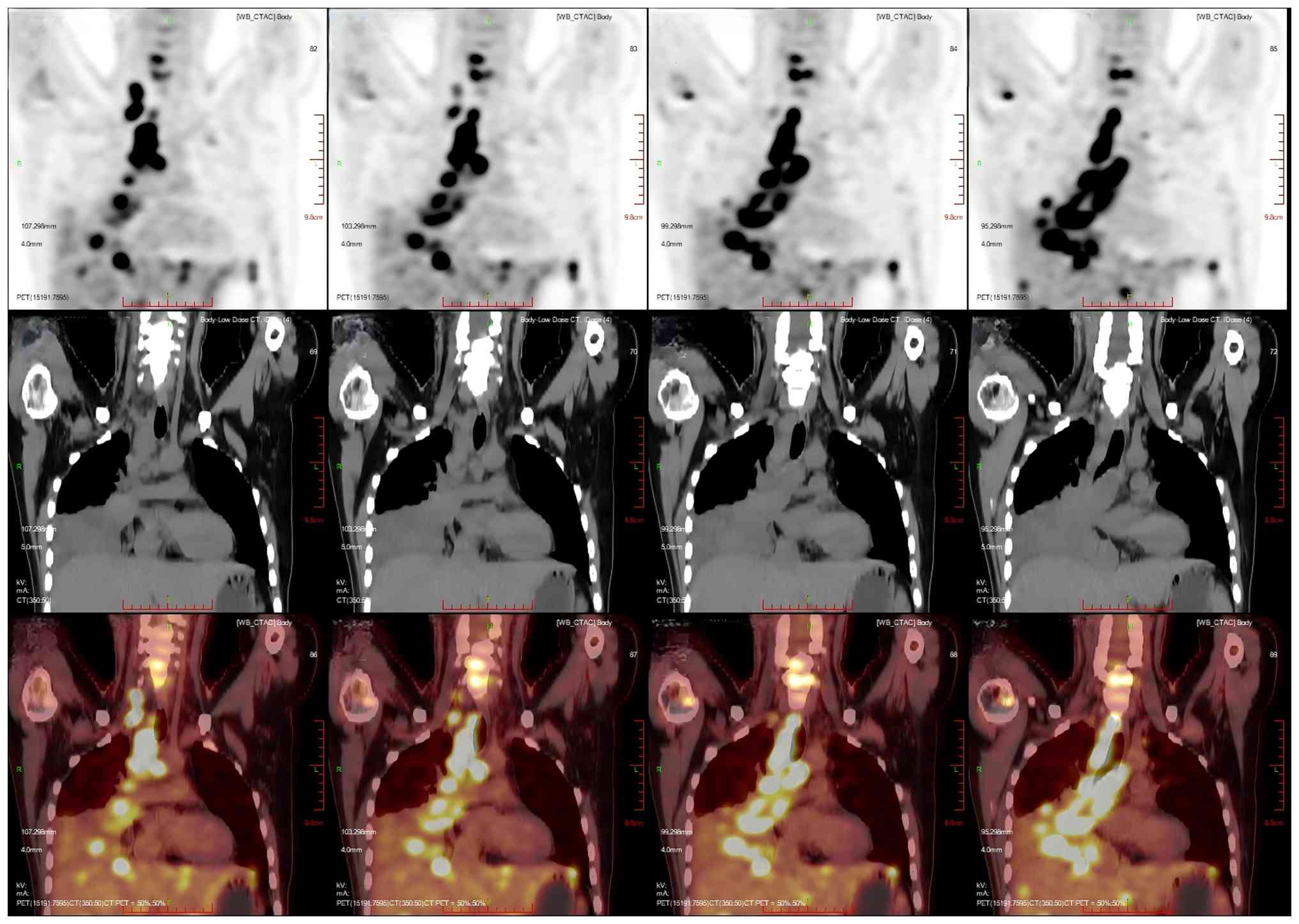
corresponding areas, with a SUVmax of 20.2 (Fig. 7). Multiple nodular and striated
radiographic uptake of the right humeral head, multiple parts of
the sternum, bilateral scapulae, cervical, thoracic and lumbar
vertebral bodies, part of the accessory bones, the two flanks of
the sacrum, multiple parts of the pelvic bone and the proximal
segment of the left femur were noted. CT demonstrated an
inhomogeneous increase in the density of the bone at the
corresponding sites, discontinuity of the cortex in some of the
neighboring bones, flattening of the seventh thoracic vertebral
body, morphological disorders and an increase in the peripheral
soft-tissue shadows. A high metabolic area was measured in the
seventh thoracic vertebral body, with an SUVmax of 20.4
(Fig. 8).
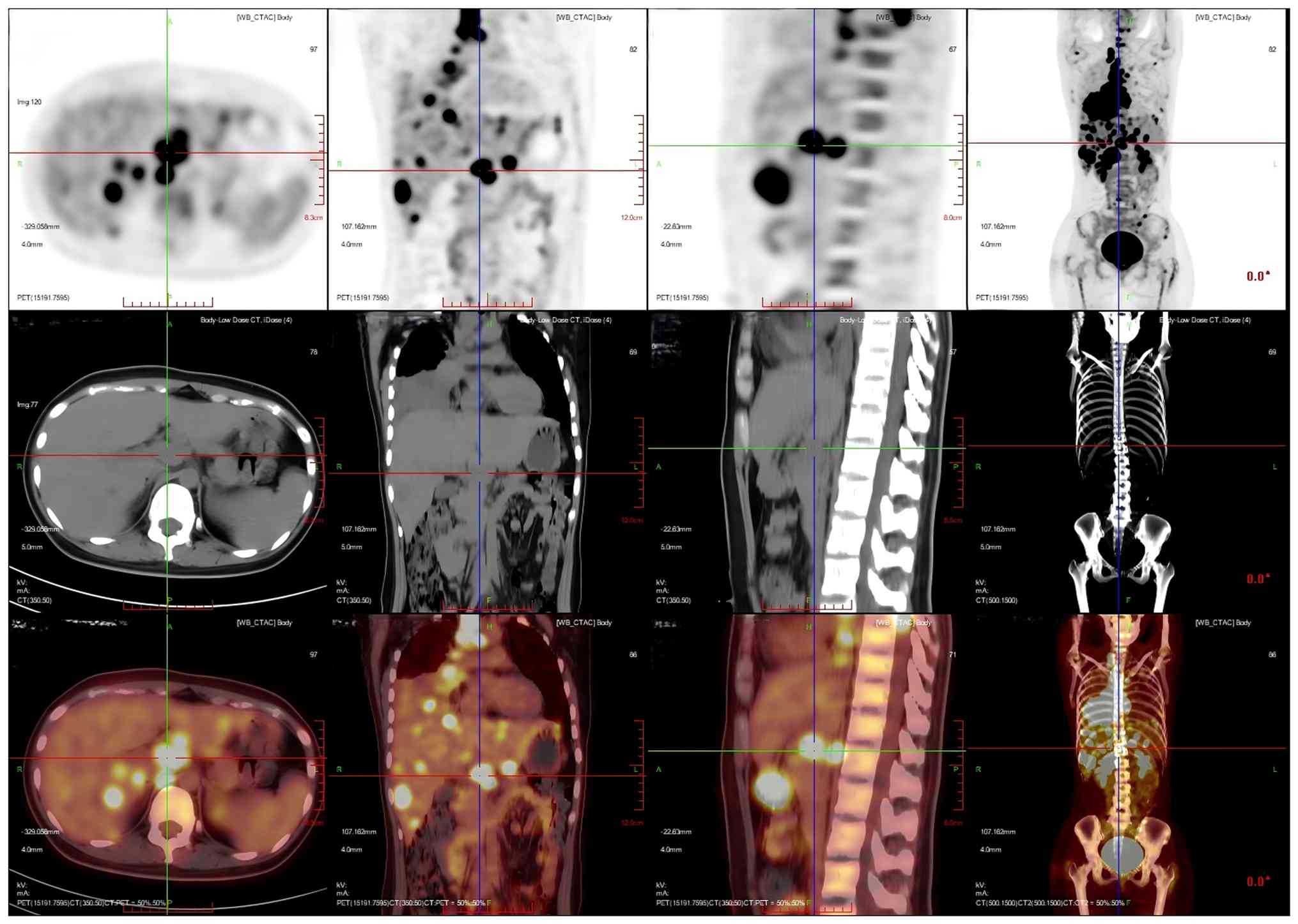 | Figure 7.Multiplanar 18F-FDG PET/CT imaging of
the patient's hilar hypermetabolic confluent lymph nodes. The liver
was full, with diffuse low-density nodules and masses of varying
sizes in the liver, and PET showed nodular and mass-like
radioactivity uptake in the corresponding areas. The low-density
mass in the S4 segment of the liver was measured to be ~5.0×4.4 cm
in size, with a SUVmax value of 20.0. Several enlarged
lymph nodes were observed in the portal region of the liver, which
had fused to form a mass. PET scans showed mass-like radioactivity
uptake in the corresponding areas, with a SUVmax value
of 20.2. Imaging modalities: PET (top row); CT (middle row); PET/CT
fusion imaging (bottom row). Imaging planes: Axial Plane (first
column): Cross-sectional views depicting transaxial details of the
hepatic hilar coalescent lymph nodes. Coronal plane (second column)
showing frontal views, illustrating the craniocaudal distribution
of the hepatic hilar lesions. Sagittal plane (third column):
Lateral views showing anteroposterior localization of the
hypermetabolic lymphadenopathy in the hepatic hilum.
Volume-rendered CT (fourth column, middle row): 3D reconstructed
whole-body CT, visualizing the spatial relationship between the
hepatic hilar lesions and thoracic/pelvic bony structures. CT,
computed tomography; PET, positron emission tomography. |
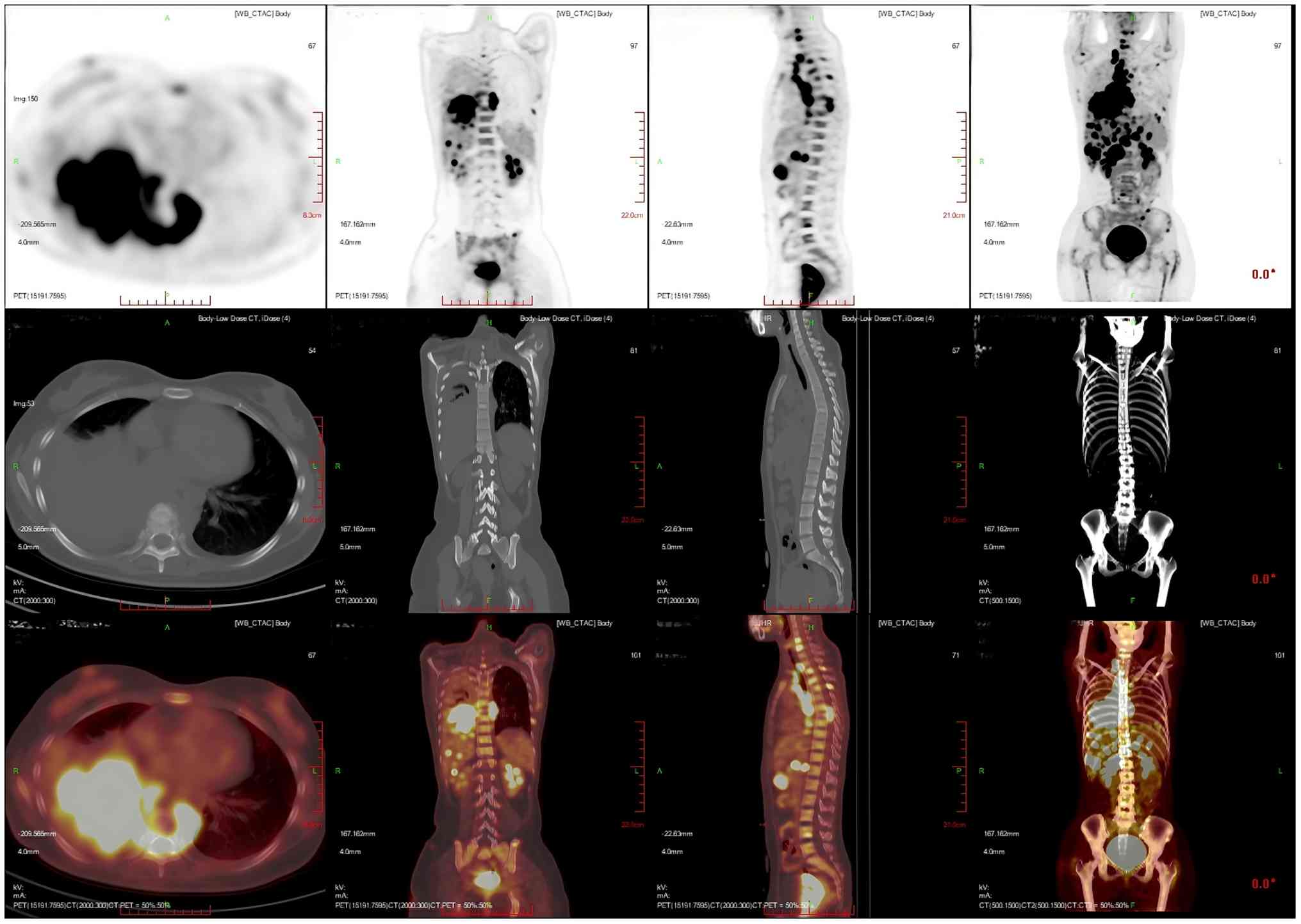 | Figure 8.Multiplanar 18F-FDG PET/CT imaging of
the patient's multifocal hypermetabolic bone lesions with
corresponding skeletal structural abnormalities. Multiple nodular
and striated radiographic uptake of the right humeral head,
multiple parts of the sternum, bilateral scapulae, cervical,
thoracic and lumbar vertebral bodies, part of the accessory bones,
the two flanks of the sacrum, multiple parts of the pelvic bone and
the proximal segment of the left femur. CT demonstrated an
inhomogeneous increase in the density of the bone at the
corresponding sites, discontinuity of the cortex in some of the
neighboring bones, flattening of the seventh thoracic vertebral
body, morphological disorders and an increase in the peripheral
soft-tissue shadows. A high metabolic area was measured in the
seventh thoracic vertebral body, with a SUVmax of 20.4.
Imaging modalities: PET (top row); CT (middle row); PET/CT Fusion
Imaging (bottom row). Imaging planes: Axial Plane (leftmost
column): Cross-sectional views (perpendicular to the body's long
axis), depicting transaxial details of focal osseous lesions.
Coronal Plane (second column): Frontal views (parallel to the
body's coronal axis), illustrating the craniocaudal distribution of
multifocal osseous lesions across the skeleton. Sagittal Plane
(third column): Lateral views (parallel to the body's sagittal
axis), showing anteroposterior localization of vertebral lesions.
3D Volume-Rendered CT (rightmost column, middle row); a 3D
reconstructed whole-body CT scan visualizing the spatial
relationship of widespread osseous lesions within the global
skeletal framework. |
The peripheral blood eosinophil level was markedly
but transiently decreased (EOS%, 63.3%; absolute eosinophil count,
14.48×109/l), but still higher than the normal range. In
response to the rapidly deteriorating condition, the situation was
explained to the family of the patient, and the treatment regimen
was changed to oral target therapy with erlotinib capsules (12 mg,
for 2 consecutive weeks, once a day). In addition, morphine
hydrochloride extended-release tablets (30 mg, once a day) were
administered orally to alleviate the pain of bone metastasis.
Lactulose oral liquid (20 ml, once a day) was administered orally,
and glycerine enema (20 ml, once a day) and multivitamins (5 ml,
once a day) were provided for nutritional support. The patient was
discharged from the hospital after symptomatic relief. During one
of the semimonthly outpatient follow-up appointments in February
2025, it was observed that the patient was unable to walk upright
due to severe bone metastases. In June 2025, the patient succumbed
to complications from the tumor.
Discussion
Eosinophils are multifunctional immune cells that
circulate in the peripheral blood and are distributed throughout
tissues. At present, eosinophils are generally considered to serve
a pleiotropic and bifacial role in the tumor microenvironment. On
the one hand, eosinophils stimulate malignant plasma cells through
an IL-6-independent mechanism, and form a synergistic support with
stromal cells (5). On the other
hand, eosinophils promote CD8+ T cell-mediated antitumor
immunity and significantly delay tumor progression (6). Although the prognosis of patients with
eosinophilic pleural effusion due to lung cancer has been reported
to be improved compared with that of patients with non-eosinophilic
malignant pleural effusion (7), in
individual cases of lung adenocarcinoma combined with eosinophilia,
this paraneoplastic syndrome is likely to reflect the
aggressiveness and progression of the lesion (8). The present report describes a rare
case of peripheral blood eosinophilia in a young woman with
squamous lung cancer during the same period as rapid tumor
progression. After treatment with glucocorticoids in combination
with antitumor agents, the peripheral blood eosinophil levels
decreased markedly. We hypothesize that the eosinophils served an
important role in promoting tumor progression.
The current diagnostic classification for
eosinophilia is mild (0.5–1.5×109/l), moderate
(1.5–5×109/l) and severe (>5×109/l)
(9). Patients who are asymptomatic
and have only mild to moderate eosinophilia may be temporarily
excluded from further testing (10). With systemic symptoms or persistent
eosinophilia (>1.5×109/l) with or without suspected
organ damage, the examination should be refined to identify or
exclude secondary causes (10,11).
Among secondary factors, allergic diseases are the most common,
followed by parasitic infections (12). Solid tumors or lymphomas may also be
associated with reactive eosinophilia (13). Drug reactions, immunodeficiency
disorders and certain skin diseases should also be taken into
consideration. The clinical history should focus on a history of
allergies, a rash or cardiopulmonary and gastrointestinal symptoms.
To consider allergic factors, serum IgE measurements may be
performed. To rule out parasitic infections, including fecal
parasites and eggs, microscopy may be performed. Considering
gastrointestinal causes, gastrointestinal endoscopy or serological
testing for serum amylase and celiac-related autoantibodies is
indicated. Imaging tests (such as chest CT scans) and fiberoptic
bronchoscopy can help rule out respiratory diseases. Rheumatologic
antibody-related tests, such as ANA or anti-double-stranded DNA
antibody analysis, should be performed in individuals predisposed
to connective tissue disease. Serological testing for
antineutrophil cytoplasmic antibodies, cytomegalovirus and human
parvovirus B19 may also be performed to rule out factors associated
with vasculitis. Eosinophilia caused by solid tumors usually has no
obvious clinical manifestations or has overlap with tumor-induced
associated symptoms, making the diagnosis challenging (10,12).
After ruling out non-cancer factors, attention may be paid to
whether the period of eosinophilia overlaps with the period of
tumor progression. Additionally, if there is suspected organ damage
due to eosinophilia (for example, to the heart, lungs, liver,
spleen, skin and nervous system), evaluation of the affected organs
should be performed (11).
Primary eosinophilia usually refers to hematological
disorders (14). In cases where no
secondary cause has been identified and eosinophilia
(>1.5×109/l) is present, a primary cause should be
considered, especially eosinophilic leukemia (15). Eosinophilic leukemia is a rare type
of leukemia characterized by abnormal eosinophilia in the
peripheral blood and bone marrow, with progressive anemia and
thrombocytopenia, and a markedly elevated white blood cell count of
up to 50–200×109/l (16,17).
The presence of naïve eosinophils in the blood and >5% of
primitive cells in the bone marrow is also required (18). The acute form of eosinophilic
leukemia is characterized by eosinophilic infiltration of multiple
organs, with cardiac, pulmonary and central nervous system
involvement, in addition to involvement of the liver, spleen and
lymph nodes. Clinical manifestations include progressive heart
failure, dyspnea, psychiatric disorders, delusions and ataxia.
Erythema and papules may appear on the skin (19). The chronic type of eosinophilic
leukemia has a slow onset, with a course of up to 2–8 years, and
manifestations such as malaise, anemia, and enlargement of the
liver, spleen and lymph nodes (20). The greatest diagnostic limitation in
the current case was the failure to perform a bone marrow
aspiration, which restricted the ability to rule out a
hematological disorder (eosinophilic leukemia). When the patient
presented with a rapid increase in blood eosinophils, the family of
the patient was immediately informed of this notable phenomenon and
the need for bone marrow aspiration was emphasized. However, the
family members believed that the patient was in the end stage of
cancer, and the quality of life of the patient was very low due to
the long-term pain. They were not willing to allow the patient to
undergo this additional invasive operation, and the main goal was
to relieve the pain. In keeping with the concept of humanistic care
for the patient and respect for the opinions of the family members,
a bone marrow puncture was not insisted on. After this, some
secondary factors were excluded by serial examination. Although a
bone marrow aspiration was not performed, there was insufficient
evidence to diagnose eosinophilic leukemia when considering the
routine blood results and the absence of apparent target organ
damage and symptomatic manifestations.
Furthermore, the immunohistochemical results
described in the manuscript are based on pathology reports rather
than retrievable image files. This is also a major limitation of
the diagnosis.
The diagnosis of eosinophilia is a diagnosis of
exclusion. Based on the exclusion of primary etiology,
understanding the clinical manifestations and laboratory tests of
various secondary etiologies and correlating them with the
progression of the disease is key for clinicians to diagnose
paraneoplastic eosinophilia.
The most important mechanism by which malignant
tumors lead to eosinophilia is the secretion of several cytokines,
including IL-5, IL-3 and granulocyte-macrophage colony-stimulating
factor, by tumor cells. Among them, IL-5 serves a key role in the
production, activation and recruitment of eosinophils into the
tumor microenvironment (21). Early
study of in vitro models of non-small cell lung cancer have
demonstrated the ability of lung cancer cells to produce IL-5-based
type 2 cytokines to recruit eosinophils (22). Eosinophils directly interact with
the tumor microenvironment, mainly via two mechanisms: First,
release of cytokines and particulate contents that affect the tumor
microenvironment and tumor cells. For example, eosinophils can
directly kill tumor cells by releasing major basic protein (MBP),
eosinophil peroxidase (EPO) and reactive oxygen species, or
eosinophils can secrete TGF-β, IL-13 and other cytokines, which
promote fibrosis, angiogenesis, immunosuppression and tumor
metastasis (23). Second,
eosinophil receptors bind to tumor cells, thereby altering the
prognosis of tumors. Eosinophils can initiate direct contact with
cancer cells and express natural killer group 2 member D, killer
markers of natural killer cells and CD8+ T cells,
thereby exerting direct antitumor effects (24,25).
Eosinophilia may be a manifestation of a poor tumor prognosis. The
coincidence of the rapid tumor progression stage and the timing of
eosinophilia in the present case also confirmed the relationship of
mutual promotion. In addition, local or systemic inflammatory
reactions triggered by cancer, the abnormal bone marrow
hematopoietic environment caused by tumor bone metastasis and other
factors will also aggravate eosinophilia, forming a vicious
circle.
Solid tumor-associated eosinophilia involves complex
immune-tumor interactions. Eosinophils can modulate inflammatory
allergic responses and interact with lymphocytes to modulate their
function in immune defense. This could be either a positive signal
of antitumor immunity or a pathological factor that promotes tumor
progression. On the one hand, activated eosinophils cause an
independent and synergistic antitumor effect of CD8+ T
cells, which can improve the tumor microenvironment to enhance the
immune response. An early study showed that eosinophils mediate
tumor rejection by promoting tumor vascular normalization (reducing
vascular leakage and alleviating hypoxia) and enhancing
CD8+ T cell infiltration, and their survival in the
tumor microenvironment may be prolonged (26). Furthermore, eosinophils can inhibit
the apoptosis of CD8+ T cells to participate in the
generation of memory CD8+ T cells and enhance the immune
defense (27). In addition,
activated eosinophils can induce the polarization of M1
macrophages, thereby improving the immunosuppressive state in
tumors. The aforementioned studies suggest that eosinophils have a
tumor-killing effect, and may enhance and activate T cells in a
type 1 immunity manner (26). This
may be an active mechanism in tumor immunotherapy. However, on the
other hand, eosinophils can promote tumor cell migration and
metastasis by secreting C-C motif ligand 6 (4). Lee et al (28) suggested that tumor-associated tissue
eosinophilia is associated with poor overall survival and that
activated eosinophils promote tumor migration primarily through C-C
motif ligand 2. Meanwhile, eosinophils can induce tumor
angiogenesis, which is related to the immunosuppressive tumor
microenvironment. Eosinophils may also promote
epithelial-mesenchymal transition, thereby enhancing tumor
invasiveness through the immunomodulatory pathway involving IL-13
and STAT6 (29). Such studies
support the idea that eosinophils promote cancer, thus confirming
the dual role of eosinophils in cancer from an immunomodulatory
perspective.
One study found that eosinophilia was positively
associated with the response to immunotherapy. However, some
patients receiving immunotherapy were prone to peripheral blood and
intra-tumor eosinophilia, which may be related to IL-5 secretion by
CD4+ T lymphocytes and IL-33-mediated eosinophil
recruitment (30). Therefore, the
altered immune status of the patient needs to be taken into
consideration. Although the current patient received tislelizumab
immunotherapy, no abnormal eosinophil manifestations were observed
in the peripheral blood during the past year of treatment. In
conjunction with the stage of the eosinophil elevation of the
patient, it was determined that pharmacogenetic factors were less
likely to be involved.
Although the total serum IgE level of the patient
was high, the patient had no history of food and drug allergies.
The patient did not exhibit any allergic symptoms, such as dyspnea,
rash, nasal congestion or a runny nose. Tumor-causing factors and
tumor cells are alloantigens or sensitizers for the human body. The
rapidly growing and spreading tumor foci of the patient stimulate
the body to produce antibodies, including IgE antibodies, which can
result in allergic reaction-like manifestations (31). In addition, both eosinophils and IgE
are typical markers of the type 2 immune response, and the
synergistic effect between them involves multiple cytokines and
signaling pathways (32–34). It has been found that IL-4, IL-5 and
IL-13 secreted by T helper 2 cells and type 2 natural lymphocytes
in the tumor microenvironment not only stimulate bone marrow
eosinophilia but also stimulate the conversion of B lymphocytes to
plasma cells, which triggers the elevation of serum total IgE
levels (35). The high-affinity IgE
receptor on the surface of tumor-associated mast cells is capable
of releasing eosinophil chemokines (such as Eotaxin-1/C-C motif
chemokine ligand 11) upon binding to antigens (36), which directly recruits eosinophils
to the tumor microenvironment. MBP and EPO secreted by eosinophils
can further stimulate mast cell degranulation, forming a positive
feedback loop (35). Therefore, it
is possible that the elevated serum total IgE level in the current
patient reflected the activation of type 2 immunity in vivo
and was closely related to the eosinophilia.
It has been noted that ~10% of patients with solid
tumors develop leukocytosis as a result of paraneoplastic
leukemia-like reactions, while eosinophilia is even rarer (37). Zhu et al (38) reported the case of an 80-year-old
man presenting with chest pain. Eosinophilia was found in the
peripheral blood (absolute value, 2.15×109/l) and
bronchial lavage fluid, accompanied by right pulmonary artery and
main bronchus invasion, and adjacent bone destruction. A subsequent
needle biopsy revealed squamous cell carcinoma of the lung. This
report highlights that elevated eosinophil counts may be associated
with metastasis and a poor prognosis in lung cancer. Ozaki et
al (39) reported a case of
cerebral infarction in a 67-year-old man with eosinophilia
(eosinophil count, 13,600/µl) due to metastasis of lung
adenocarcinoma to the bone marrow, but the precise mechanism was
unclear. In addition, Verstraeten et al (40) reported a case of non-small cell lung
cancer associated with peripheral eosinophilia. The affected
65-year-old man received a diagnosis of paraneoplastic
hypereosinophilia after ruling out anaphylaxis, leukemia,
vasculitis and parasitic infection. This is similar to the present
case. In the 8 previously reported cases of squamous cell carcinoma
of the lung combined with peripheral blood eosinophilia, the mean
age of the patients was 63 years, and only 1 case was in a female
patient (41). In the present case,
the female patient was only 25 years old, which is a notable
indication of the specificity and rarity of the case. The severity
of paraneoplastic eosinophilia can range from asymptomatic to
life-threatening. In numerous cases, markedly elevated eosinophil
counts may be accompanied by respiratory reactions and rashes, with
some patients exhibiting specific symptoms such as shortness of
breath, cognitive deficits, disorientation and speech
disorganization (42). However, in
the present case, there were no specific clinical manifestations,
which made clinical detection and identification difficult, but
also suggested that there was no apparent target organ damage.
A significant causal effect of eosinophilia,
especially with the development of squamous lung cancer, has been
found in East Asian populations. The risk of developing lung cancer
in patients with eosinophilia may be increased 1.28-fold compared
with that of patients with no eosinophilia (43). In lung squamous carcinoma biopsy
tissues, an association has been observed between the level of
tissue eosinophil infiltration and programmed cell death ligand 1
expression ≥50% (44). The present
patient had no history of smoking, blood transfusion or allergies,
and no family genetic history. Considering that the likelihood of
EGFR mutation is markedly higher in patients with these particular
types of risk factor for squamous lung cancer (for example, female,
non-smoker and Asian ethnicity), genetic testing was performed
using next-generation sequencing analysis in the present case. A
mutation in exon 20, p.T790M, of the EGFR gene (mutation frequency,
53.51%) was detected. Blood eosinophilia in patients with lung
adenocarcinoma harboring the EGFR L858R mutation and
mesenchymal-epithelial transition factor amplification has
previously been reported to parallel disease progression. Blood
eosinophilia has a negative effect on prognosis in driver-positive
non-small cell lung cancer (45),
suggesting that eosinophil counts and EGFR mutations may be
associated.
Since absolute peripheral blood eosinophil counts
are not necessarily positively associated with end-organ damage,
there is no consensus on whether treatment is needed and when to
start it in the absence of apparent organ involvement, dysfunction
and clinical manifestations. Patients with paraneoplastic
eosinophilia are typically asymptomatic, and treatment focuses on
reducing eosinophil counts and preventing eosinophil-mediated organ
damage. Resection of the tumor and chemotherapy can lead to a
decrease in paraneoplastic eosinophils (46,47).
Eosinophil counts can also be decreased by short-term use of
low-dose corticosteroids and occasionally glucocorticoids (48). The combination of corticosteroids
and hydroxyurea has been reported to enhance therapeutic effects
(29). If eosinophilia is extreme,
allopurinol should be administered concurrently (49). Glucocorticoid therapy must still be
applied when eosinophilia is severely life-threatening, and a
starting dose of 1 mg/kg/day is generally recommended, maintained
for 1–2 weeks, and tapered off over 2–3 months. If the efficacy is
not satisfactory, treatment with immunomodulators (interferon-α,
cyclosporine or azathioprine) and hydroxyurea can be added
(50,51). In the current case, nab-paclitaxel
was applied in addition to the intravenous infusion of
methylprednisolone sodium succinate to combat the primary tumor,
resulting in a marked improvement in the peripheral blood
eosinophil counts and preventing organ damage caused by further
eosinophilia.
Current research suggests that the prognosis of
eosinophilia is mainly dependent on the diagnosis of the disease,
and that patients with eosinophilia in combination with most
diseases have an improved prognosis if the diagnosis is recognized
at an early stage and treated appropriately (52). However, in the current case, the
exceptionally high eosinophil count and rapidly progressive
malignant disease suggested a poor prognosis.
In conclusion, to the best of our knowledge, this is
the first report of rapidly progressive squamous lung cancer with
eosinophilia in a young woman. Eosinophilia is a paraneoplastic
syndrome that can occur in the advanced stages of squamous lung
cancer and becomes a potential risk factor promoting the
proliferative progression of squamous lung cancer. This also
suggests the importance of recognizing the role that paraneoplastic
eosinophilia serves in the tumor and the need to take early steps
to combat it. Clinical practice should also consider the
possibility that the presence of eosinophilia in the blood may be
associated with malignancy. Corticosteroids, in combination with
anticancer drugs, should be used after early diagnosis to prevent
organ damage caused by eosinophilia and to enhance the
effectiveness of anticancer therapy to some extent. However,
eosinophilia is complicated to diagnose in both the early and late
stages, and it may also involve multiple organ systems or present
with systemic manifestations. A multidisciplinary approach should
be employed to optimize diagnosis and treatment when necessary.
Supplementary Material
Supporting Data
Acknowledgements
Not applicable.
Funding
Funding: No funding was received.
Availability of data and materials
The data generated in the present study may be found
in the National Center for Biotechnology Information under
accession number PRJNA1348469 or at the following URL: http://www.ncbi.nlm.nih.gov/bioproject/1348469. The
remaining data generated in the present study may be requested from
the corresponding author.
Authors' contributions
MZ and HF designed the study. HF, CS, DL, and JG
advised on patient treatment. MZ, DL, JG, and YH acquired the data.
HF, DL, YH, and JC analyzed and interpreted data for the work. MZ,
HF, CS, DL, JG, YH and JC confirm the authenticity of all the raw
data. All authors agree to be accountable for all aspects of the
work. All authors have read and approved the final version of the
manuscript.
Ethics approval and consent to
participate
The Ethics Committee of The First Affiliated
Hospital of the Hebei University of Chinese Medicine approved this
study, which was conducted according to the 1964 Helsinki
Declaration and its subsequent amendments, or comparable ethical
standards. The patient provided written informed consent for the
publication of their data prior to succumbing to the disease.
Patient consent for publication
The patient provided written informed consent for
the publication of their data and associated images prior to
succumbing to the disease.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Jameson JL and Johnson BE: Paraneoplastic
syndromes: Endocrinologic/hematologic. Harrison's Principles of
Internal Medicine. Fauci AS, Braunwald E, Kasper DL, Hauser SL,
Longo DL, Jameson JL and Loscalzo J: 17th edition. McGraw Hill
Medical; New York, NY: pp. 617–622. 2008
|
|
2
|
Sakkal S, Miller S, Apostolopoulos V and
Nurgali K: Eosinophils in cancer: Favourable or unfavourable? Curr
Med Chem. 23:650–566. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Kay AB: The early history of the
eosinophil. Clin Exp Allergy. 45:575–582. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Li F, Du X, Lan F, Li N, Zhang C, Zhu C,
Wang X, He Y, Shao Z, Chen H, et al: Eosinophilic inflammation
promotes CCL6-dependent metastatic tumor growth. Sci Adv.
7:eabb59432021. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Saglimbeni J, Esteva E, Canales J, Perez
OA, Eichinger A, Huntley W, Khanna KM, Dolgalev I, Klar N, Adams S
and Reizis B: Peritumoral macrophages recruit eosinophils to
promote antitumor immune responses in breast cancer. Proc Natl Acad
Sci USA. 122:e25046451222025. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Arnold IC, Artola-Boran M, Gurtner A,
Bertram K, Bauer M, Frangez Z, Becher B, Kopf M, Yousefi S, et al:
The GM-CSF-IRF5 signaling axis in eosinophils promotes antitumor
immunity through activation of type 1 T cell responses. J Exp Med.
217:e201907062020. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Takeuchi E, Okano Y, Machida H, Atagi K,
Kondou Y, Kadota N, Hatakeyama N, Naruse K and Shinohara T:
Eosinophilic pleural effusion due to lung cancer has a better
prognosis than non-eosinophilic malignant pleural effusion. Cancer
Immunol Immunother. 71:365–372. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Akkad N, Jiang Y and Shin D: Leucocytosis
and stroke in a lung cancer patient. Eur J Case Rep Intern Med.
7:0018722020.PubMed/NCBI
|
|
9
|
Tzankov A, Reichard KK, Hasserjian RP,
Arber DA, Orazi A and Wang SA: Updates on eosinophilic disorders.
Virchows Arch. 482:85–97. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Gerds AT, Gotlib J, Bose P, Deininger MW,
Dunbar A, Elshoury A, George TI, Gojo I, Gundabolu K, Hexner E, et
al: Myeloid/Lymphoid neoplasms with eosinophilia and TK Fusion
genes, version 3.2021, NCCN clinical practice guidelines in
oncology. J Natl Compr Canc Netw. 18:1248–1269. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Valent P, Klion AD, Roufosse F, Simon D,
Metzgeroth G, Leiferman KM, Schwaab J, Butterfield JH, Sperr WR,
Sotlar K, et al: Proposed refined diagnostic criteria and
classification of eosinophil disorders and related syndromes.
Allergy. 78:47–59. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Wang SA, Orazi A, Gotlib J, Reiter A,
Tzankov A, Hasserjian RP, Arber DA and Tefferi A: The international
consensus classification of eosinophilic disorders and systemic
mastocytosis. Am J Hematol. 98:1286–1306. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Shomali W and Gotlib J: World Health
Organization-defined eosinophilic disorders: 2022 update on
diagnosis, risk stratification, and management. Am J Hematol.
97:129–148. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Khoury JD, Solary E, Abla O, Akkari Y,
Alaggio R, Apperley JF, Bejar R, Berti E, Busque L, Chan JKC, et
al: The 5th edition of the World Health Organization Classification
of Haematolymphoid Tumours: Myeloid and histiocytic/dendritic
neoplasms. Leukemia. 36:1703–1719. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Gotlib J: World Health
Organization-defined eosinophilic disorders: 2017 update on
diagnosis, risk stratification, and management. Am J Hematol.
92:1243–1259. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Morsia E, Tefferi A and Pardanani A: WHO
defined chronic eosinophilic leukemia, not otherwise specified
(CEL, NOS): A contemporary series from the Mayo Clinic. Am J
Hematol. 95:e172–e174. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Vormittag-Nocito E, Khan I, Wiley E, Behm
F and Ni H: Presentation of a diagnostically challenging case of
chronic eosinophilic leukemia with marrow dysplasia and ringed
sideroblasts. SAGE Open Med Case Rep. 8:2050313X209574462020.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Savage N, George TI and Gotlib J: Myeloid
neoplasms associated with eosinophilia and rearrangement of PDGFRA,
PDGFRB, and FGFR1: A review. Int J Lab Hematol. 35:491–500. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Boxer LA and Newburger PE: Eosinophils.
Elsevier eLibrary Chapter 123; 2025
|
|
20
|
Kawashima I and Kirito K: Chronic
neutrophilic leukemia/chronic eosinophilic leukemia. Rinsho
Ketsueki. 64:1326–1334. 2023.(In Japanese). PubMed/NCBI
|
|
21
|
Kanaji N, Watanabe N, Kita N, Bandoh S,
Tadokoro A, Ishii T, Dobashi H and Matsunaga T: Paraneoplastic
syndromes associated with lung cancer. World J Clin Oncol.
5:197–223. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Alves A, Dias M, Campainha S and Barroso
A: Peripheral blood eosinophilia may be a prognostic biomarker in
non-small cell lung cancer patients treated with immunotherapy. J
Thorac Dis. 13:2716–2727. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Grisaru-Tal S, Jacobsen EA and Munitz A:
Evolving role for eosinophils in cancer: From bench to bedside.
Trends Cancer. 11:862–876. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Caruso RA, Parisi A, Quattrocchi E,
Scardigno M, Branca G, Parisi C, Lucianò R, Paparo D and Fedele F:
Ultrastructural descriptions of heterotypic aggregation between
eosinophils and tumor cells in human gastric carcinomas.
Ultrastruct Pathol. 35:145–149. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Caruso R, Irato E and Rigoli L: Eosinophil
exocytosis in a poorly differentiated tubular gastric
adenocarcinoma: Case report. Ultrastruct Pathol. 46:139–146. 2022.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Carretero R, Sektioglu IM, Garbi N,
Salgado OC, Beckhove P and Hämmerling GJ: Eosinophils orchestrate
cancer rejection by normalizing tumor vessels and enhancing
infiltration of CD8(+) T cells. Nat Immunol. 16:609–617. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Zhou J, Liu J, Wang B, Li N, Liu J, Han Y
and Cao X: Eosinophils promote CD8+ T cell memory generation to
potentiate anti-bacterial immunity. Si=-gnal Transduct Target Ther.
9:432024. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Lee TL, Chen TH, Kuo YJ, Lan HY, Yang MH
and Chu PY: Tumor-associated tissue eosinophilia promotes
angiogenesis and metastasis in head and neck squamous cell
carcinoma. Neoplasia. 35:1008552023. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Cao H, Zhang J, Liu H, Wan L, Zhang H,
Huang Q, Xu E and Lai M: IL-13/STAT6 signaling plays a critical
role in the Epithelial-mesenchymal transition of colorectal cancer
cells. Oncotarget. 7:61183–61198. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Blomberg OS, Spagnuolo L, Garner H,
Voorwerk L, Isaeva OI, van Dyk E, Bakker N, Chalabi M, Klaver C,
Duijst M, et al: IL-5-producing CD4+ T cells and eosinophils
cooperate to enhance response to immune checkpoint blockade in
breast cancer. Cancer Cell. 41:106–123.e10. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Hanahan D and Weinberg RA: Hallmarks of
cancer: The next generation. Cell. 144:646–674. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Robida PA, Puzzovio PG, Pahima H,
Levi-Schaffer F and Bochner BS: Human eosinophils and mast cells:
Birds of a feather flock together. Immunol Rev. 282:151–167. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Davoine F and Lacy P: Eosinophil
cytokines, chemokines, and growth factors: Emerging roles in
immunity. Front Immunol. 5:5702014. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Gleich GJ: Mechanisms of
eosinophil-associated inflammation. J Allergy Clin Immunol.
105:651–663. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Kovalszki A and Weller PF: Eosinophilia.
Prim Care. 43:607–617. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Ando T and Kitaura J: Tuning IgE:
IgE-associating molecules and their effects on IgE-dependent mast
cell reactions. Cells. 10:16972021. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Granger JM and Kontoyiannis DP: Etiology
and outcome of extreme leukocytosis in 758 nonhematologic cancer
patients: A retrospective, single-institution study. Cancer.
115:3919–3923. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Zhu YL, Tong ZH, Jin ML and Wang C: Lung
cancer with marked blood eosinophilia: Case report and literature
review. Zhonghua Jie He He Hu Xi Za Zhi. 32:369–372. 2009.(In
Chinese). PubMed/NCBI
|
|
39
|
Ozaki M, Mano T, Eura N, Horimoto K,
Takano M, Ohbayashi C and Sugie K: Multiple cerebral infarctions
associated with lung cancer-induced hypereosinophilia: A case
report. BMC Neurol. 21:3972021. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Verstraeten AS, De Weerdt A, van Den
Eynden G, Van Marck E, Snoeckx A and Jorens PG: Excessive
eosinophilia as paraneoplastic syndrome in a patient with
non-small-cell lung carcinoma: A case report and review of the
literature. Acta Clin Belg. 66:293–297. 2011.PubMed/NCBI
|
|
41
|
Zalewska E, Obołończyk Ł and Sworczak K:
Hypereosinophilia in solid Tumors-case report and clinical review.
Front Oncol. 11:6393952021. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Lo CH, Jen YM, Tsai WC, Chung PY and Kao
WY: Rapidly evolving asymptomatic eosinophilia in a patient with
lung adenocarcinoma causes cognitive disturbance and respiratory
insufficiency: Case report. Oncol Lett. 5:495–498. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Wang Z, Chen B, Fu Y, Ou C, Rong Q, Kong
X, Xu W, Deng Y, Jiang M and Xie J: Eosinophilia and lung cancer:
Analysis from Real-world data and mendelian randomization study.
Front Med (Lausanne). 9:8307542022. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Yuan P, Guo C, Li L, Ling Y, Guo L and
Ying J: Immune-related histologic phenotype in pretreatment tumour
biopsy predicts the efficacy of neoadjuvant anti-PD-1 treatment in
squamous lung cancer. BMC Med. 20:4032022. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Geng N, Hu W, Ge H, Xie S and Ding C:
Tumor-associated eosinophilia in a patient with EGFR-positive,
MET-amplified lung adenocarcinoma refractory to targeted therapy: A
case report and review of literature. Transl Cancer Res.
10:4988–4996. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Pandit R, Scholnik A, Wulfekuhler L and
Dimitrov N: Non-small-cell lung cancer associated with excessive
eosinophilia and secretion of interleukin-5 as a paraneoplastic
syndrome. Am J Hematol. 82:234–237. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Klimek M: Pulmonary lymphangitis
carcinomatosis: Systematic review and meta-analysis of case
reports, 1970–2018. Postgrad Med. 131:309–318. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Thomsen GN, Christoffersen MN, Lindegaard
HM, Davidsen JR, Hartmeyer GN, Assing K, Mortz CG, Martin-Iguacel
R, Møller MB, Kjeldsen AD, et al: The multidisciplinary approach to
eosinophilia. Front Oncol. 13:11937302023. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Leukemia Lymphoma Group and Chinese
Society of Hematology, Chinese Medical Association, . Guideline of
the diagnosis and treatment of eosinophilic disorders (2024).
Zhonghua Xue Ye Xue Za Zhi. 45:1–7. 2024.(In Chinese).
|
|
50
|
Shomali W and Gotlib J: World Health
Organization-defined eosinophilic disorders: 2019 update on
diagnosis, risk stratification, and management. Am J Hematol.
94:1149–1167. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Butt NM, Lambert J, Ali S, Beer PA, Cross
NC, Duncombe A, Ewing J, Harrison CN, Knapper S, McLornan D, et al:
Guideline for the investigation and management of eosinophilia. Br
J Haematol. 176:553–572. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Leru PM: Eosinophilic disorders:
evaluation of current classification and diagnostic criteria,
proposal of a practical diagnostic algorithm. Clin Transl Allergy.
9:362019. View Article : Google Scholar : PubMed/NCBI
|















![Successive chest CT scans. (A and D)
In January 2024, a soft tissue mass was detected in the lower lobe
of the right lung (size, 3.2×2.5 cm) [(A) lung window; (D)
mediastinal window]. (B and E) In October 2024, the tumor in the
lower lobe of the right lung was found to be slightly enlarged
compared with that in the previous CT scan (size, 5.2×3.5 cm) [(B)
lung window; (D) mediastinal window]. (C and F) In December 2024,
occupation in the lower lobe of the right lung was found to be
markedly enlarged compared with that in the previous CT scan (size,
10×8.4×5.6 cm) [(C) lung window; (F) mediastinal window]. The red
arrows point to the lesion.](/article_images/ol/31/6/ol-31-06-15568-g01.jpg)