According to the 2020 Global Cancer Statistics
Report, there were 2.36 million newly diagnosed cases of breast
cancer, accounting for 11.7% of the novel cases of cancer in women
worldwide. This has elevated breast cancer to the most common
malignant tumors among women globally (1). A 2022 study further predicted that
breast cancer would continue to rank first in the incidence and
mortality of cancer in women, while lung cancer would become the
most common cancer type among men (2). Breast cancer imposes a notable
economic burden worldwide. The average annual direct medical
expenses for patients reaches thousands of dollars across various
countries, while indirect costs, such as loss of productivity, lead
to huge economic losses reaching billions of dollars (3). It is worth noting that the incidence
of breast cancer among young women (particularly those of working
age) is on the rise. These patients not only face the dual risks of
unemployment and disability, but also bear enormous economic and
psychological pressure (4). In
addition, breast cancer treatment may lead to chronic pain, mental
health problems such as anxiety and depression and changes in body
image, which seriously affect the quality of life of patients
(5). Although mammography has been
extensively adopted, its sensitivity is limited, the false-positive
rate is high and it is difficult to reliably detect lesions in
fast-growing interphase cancer or dense breast tissue. These
limitations highlight the urgent need to improve screening methods
(6–8). Simultaneously, biopsy for common
metastases such as lung, bone and brain is invasive, technically
difficult, expensive and dependent on skilled operators (9,10). In
summary, the limitations of existing diagnosis and monitoring
methods, as well as the high economic burden, morbidity, mortality
and disability rate of breast cancer, jointly highlight the urgent
need to develop innovative early detection and treatment
technologies. In this context, liquid biopsy technology,
particularly circulating cell-free DNA (cfDNA) analysis, as a
non-invasive, repeatable and real-time innovative tool to reflect
tumor dynamics, is changing the pattern of accurate diagnosis and
treatment of tumors at an unprecedented speed.
Targeting its advantages of non-invasiveness,
capacity for serial sampling and high patient compliance, cfDNA
analysis provides a novel breakthrough for the whole process
management of breast cancer. With further understanding of the
biological characteristics of cfDNA (such as fragmentation patterns
and methylation characteristics) and the growing maturity of
next-generation sequencing (NGS) technology, cfDNA analysis can now
detect tumor specific mutations, copy number variations (CNVs) and
epigenetic changes with high sensitivity. The present review aimed
to systematically elaborate the latest progress and transformation
path of cfDNA in the precise diagnosis and treatment of breast
cancer. In contrast to previous reviews that primarily focused on
general technical principles or pan-cancer applications (11,12),
the present review focuses on breast cancer specific scenarios,
committed to building a complete evidence chain from ‘biomarker
traits’ to ‘clinical decision support’. Specifically, the present
review aims to: i) Systematically review the frontier analysis
dimensions beyond gene mutation, including the emerging
applications of cfDNA methylation profiling and fragment omics in
early detection, molecular typing and prognosis prediction; ii)
integrate and critically evaluate the evidence-based association
between key gene mutations [such as PIK3CA, tumor protein p53
(TP53) and ESR1] and clinical endpoints [such as early recurrence,
treatment resistance and overall survival (OS)]; and iii) analyze
the transformation path and implementation challenge of cfDNA
analysis in the dynamic monitoring of therapeutic response and
tracking of drug resistance mechanism in advanced breast cancer.
Based on the aforementioned focus, the present review aims to
provide clinicians and transformation researchers with a reference
framework with clear structure, clear evidence classification and
clinical operability, thereby promoting the ultimate value of cfDNA
analysis in the practice of breast cancer precision medicine.
The present study used a narrative review method to
systematically evaluate the biological basis and clinical
translation prospects of cfDNA in breast cancer management. To
ensure the systematic and representative nature of the evidence,
the present review conducted a systematic search in the PubMed
database in September 2024, covering the period from 2016 to 2025.
The search strategy centered on three major themes: Biological
characteristics of cfDNA, detection technology and its application
in early diagnosis, treatment monitoring and prognosis assessment
of breast cancer, and combined use of subject words and free words
to optimize the recall rate. The full search strategies for all
databases are provided in Data S1.
Literature screening was completed independently by two
researchers. First, a preliminary screening was conducted based on
titles and abstracts and then the full text of eligible literature
was evaluated. Inclusion criteria were as follows: i) Original
research or high-quality reviews focusing on human breast cancer;
ii) the research content directly involves the clinical application
of cfDNA; and iii) it is published in English. The final included
literature forms the basis of the present review and supports the
preparation of each analysis table in the article (such as Table SI, Table SII, Table SIII, Table IV). These tables focus on the key
evidence of cfDNA in pathogenesis, efficacy monitoring and
prognostic gene mutations, respectively. Using the aforementioned
process, the present review aimed to build a transparent and
rigorous evidence system to provide reliable support for the
arguments and conclusions proposed.
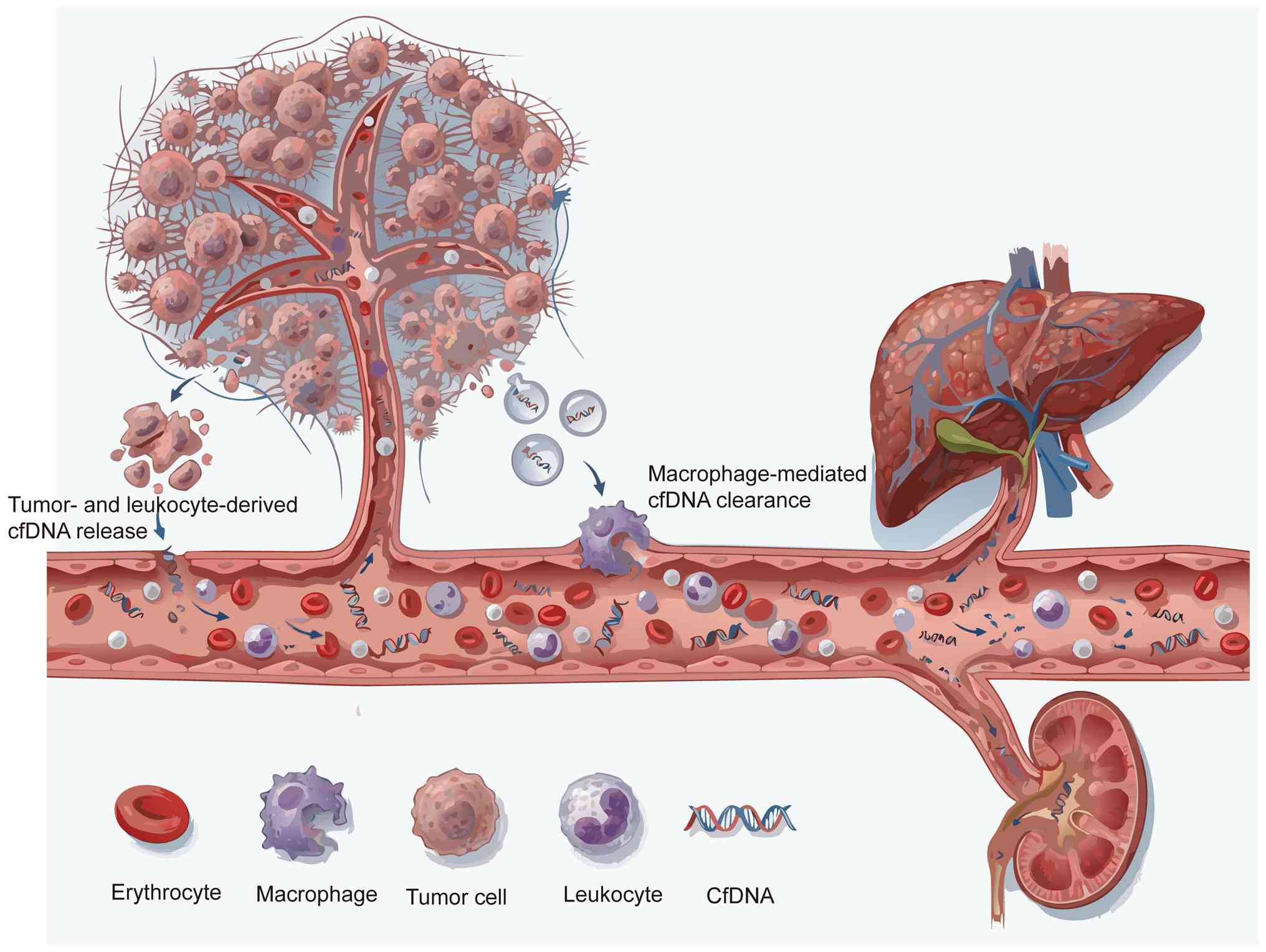
cfDNA is a key biomolecule that exists in a variety
of body fluids and serves a key role in both basic research and
clinical applications. cfDNA refers to DNA fragments released from
cells into biological fluids such as blood, ascites, pleural
effusion and urine (13). In
healthy individuals, plasma cfDNA mainly originates from leukocytes
(55%), erythrocyte precursor cells (30%), vascular endothelial
cells (10%) and hepatocytes (1%). However, in patients with cancer,
the proportion of cfDNA released by tumor cells increases markedly.
cfNDA is mainly released through extracellular vesicle pathways,
such as apoptosis, necrosis and exosomes of tumor cells, and exists
in double-stranded and single-stranded forms. It is worth noting
that the concentration of cfDNA in serum can reach 3–24-fold higher
compared with that in plasma (14–16).
Although dying cells release large amounts of DNA, <10% of it
can be detected as cfDNA in plasma. Furthermore, the detection
levels of cfDNA from different cell sources may differ from the
expected ratio by up to a 1,000-fold, typically <0.1% (17). The clearance of cfDNA involves
multiple biological pathways and organ systems, including
macrophage-mediated phagocytosis, filtration functions of the
kidney and liver, plasma nucleases and interactions with other
blood components such as serum proteins (e.g., albumin,
lipoproteins), divalent cations (Ca2+, Mg2+)
and other circulating nucleic acids. These clearance mechanisms may
work synergistically to jointly regulate the levels of cfDNA in
circulation, but their specific contributions and proportions still
warrant further study to elucidate (18,19).
The dynamic fluctuations of cfDNA in the blood result from a
complex balance between release and clearance mechanisms.
Therefore, the presence and duration of cfDNA in circulation is
regulated by the interaction of these processes, highlighting the
need to further explore its regulatory mechanisms (Fig. 1).
Recent research revealed that cfDNA fragment length
distribution is one of the key features in distinguishing patients
with cancer from healthy individuals. The length of cfDNA fragments
in plasma samples from cancer patients typically concentrate in the
range of 140–180 bp, while those from healthy individuals are
generally longer, between 160 and 265 bp (20). In tumor tissues, DNA hypomethylation
and high gene expression may promote the generation of shorter
cfDNA fragments (21). In addition,
the ends of cfDNA molecules derived from nucleosomes are shorter
and have unique end sequence characteristics. This implies that by
screening cfDNA molecules with specific end characteristics, a
higher proportion of tumor-derived cfDNA can be enriched in
patients with cancer, thereby improving diagnostic accuracy
(22). In the circulation system,
cfDNA mainly exists in two forms: Encapsulated in extracellular
vesicles (such as apoptotic bodies) or bound to proteins. In
tumor-related states, cfDNA-protein complexes have a higher
protein-DNA ratio, particularly in pathways associated with ion
channels, protein binding, transport and signal transduction
(23). Compared with healthy
individuals, the cfDNA profiles of patients with cancer exhibit a
higher proportion of short fragments and a lower C-terminal
preference (24). Concurrently,
notable variations in cfDNA fragment length exist among different
tumor types. The length variation of cfDNA fragments derived from
tumors is markedly higher compared with that of fragments derived
from healthy tissues (25,26). Although blood is the most commonly
used sample type for cfDNA analysis in clinical practice, other
body fluids such as ascites and pleural effusion can provide
supplementary diagnostic information that cannot be detected in
blood-derived cfDNA. This information is key to understanding the
tumor biology and clonal dynamics of primary tumors and metastases
(27,28). Furthermore, cfDNA sequencing
technology can also reveal clonal hematopoiesis (CH) and notable
increases in inflammation levels in the tumor microenvironment
(28).
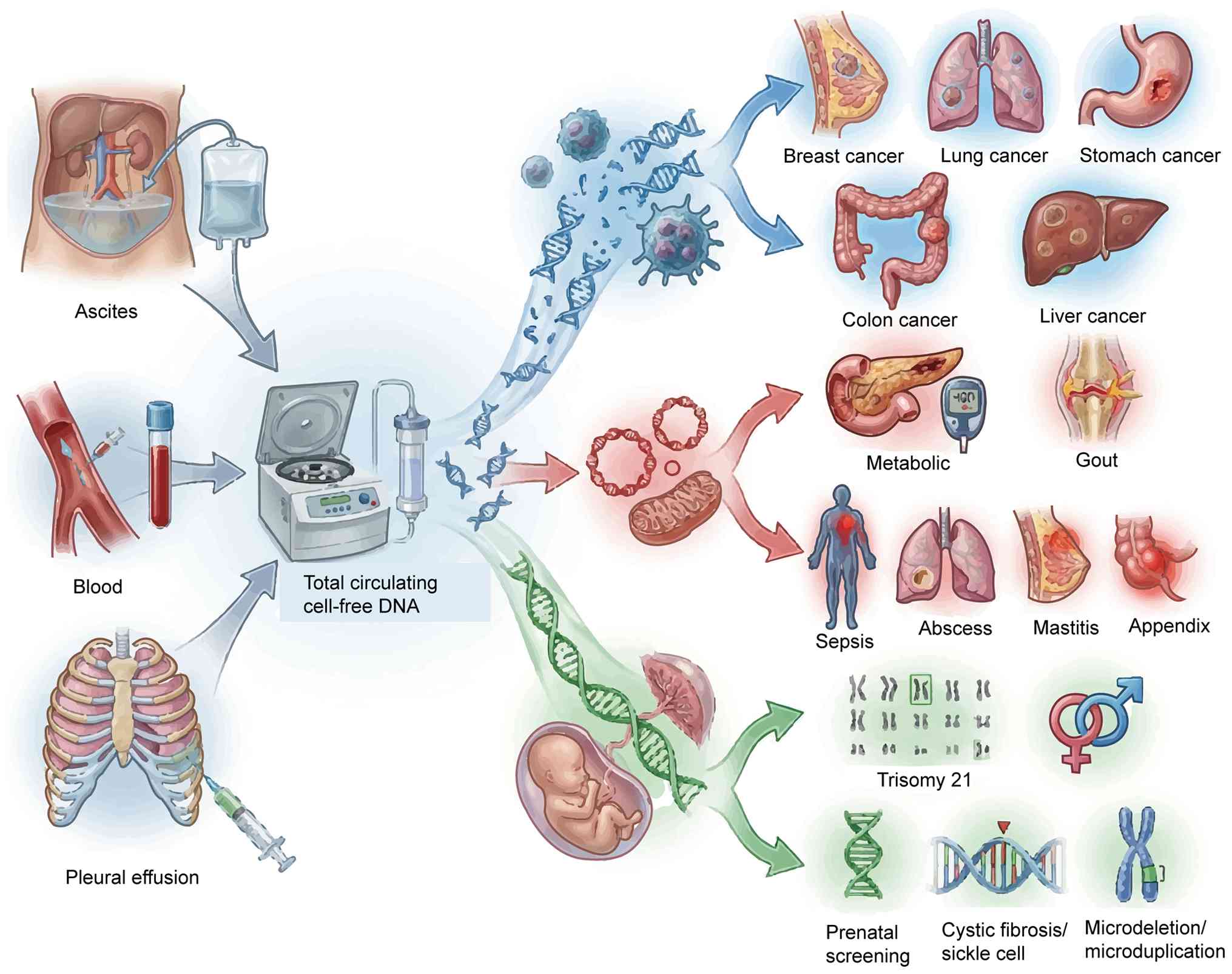
In addition to the field of oncology, cfDNA has
identified extensive applications in sepsis diagnosis, type 2
diabetes mellitus management, prenatal screening, cardiovascular
disease research and transplantation medicine (29–31).
The identification of cfDNA has provided novel avenues for
non-invasive diagnosis, enabling dynamic disease monitoring using
liquid biopsy. With increasing cfDNA research, multiple subtypes
have been identified, including circulating tumor DNA (ctDNA),
circulating mitochondrial DNA (cf-mtDNA), cell-free fetal DNA
(cf-fDNA), monocyte-derived cfDNA and nucleosome-associated cfDNA.
Among these, ctDNA has demonstrated particularly notable
application potential in oncology and has become the major focus of
current cancer research. Subsequent sections focus on the
biological properties and clinical relevance of ctDNA, cf-mtDNA and
cf-fDNA.
ctDNA refers to cfDNA fragments released by tumor
cells, accounting for ~0.01% of total cfDNA, with a concentration
range in blood samples typically between 5–1,500 ng/ml. The
cfDNA/ctDNA ratio is closely associated with tumor burden and
proliferation rate (32,33), making it a key biomarker for the
assessment of genetic and epigenetic changes in tumors (34). Most ctDNA fragments are >100 bp
in length (20) and their
concentration changes can reflect tumor burden and treatment effect
in real time. Due to its high clinical sensitivity, ctDNA is
considered a promising tool in monitoring tumor recurrence
(35,36). ctDNA fingerprinting technology
improves the specificity and sensitivity of tumor response
assessment and can detect tumor recurrence earlier than
imaging-based diagnosis (37).
However, the extremely low levels of ctDNA in circulations,
particularly in early-stage cancer, poses challenges for its
extensive clinical application. Developing advanced detection
technologies is key to overcoming this limitation.
cf-mtDNA accounts for a small proportion of the
total cfDNA in blood and other body fluids. cf-mtDNA levels are
associated with hormone receptor expression and high sensitivity to
glucocorticoids can lead to a decrease in cf-mtDNA levels (38,39).
cf-mtDNA can serve as a biomarker for metabolic disorders,
including type 2 diabetes mellitus, diabetic ketoacidosis,
hyperglycemia, hyperosmolar hyperglycemic states, hypoglycemia and
gout, and can also be used in the assessment of inflammatory
diseases such as sepsis, lung abscess, mastitis and acute
appendicitis (40). Furthermore,
previous studies have observed that cf-mtDNA levels rapidly
increase under acute psychosocial stress (41–43).
Plasma cf-mtDNA fragments exhibit a unique size distribution with a
peak at ~90 bp (44). Low cf-mtDNA
copy number is associated with increased risk of type 2 diabetes
mellitus and metabolic syndrome (45).
cf-fDNA mainly originates from the placenta and
enters the maternal circulation system. cf-fDNA has been
extensively used in non-invasive prenatal testing (NIPT) to screen
for chromosomal abnormalities such as trisomy 21 (Down syndrome),
trisomy 18 and trisomy 13. cf-fDNA is present as short fragments in
maternal plasma (46). Previous
research has reported that the peripheral circular DNA molecules of
cf-fDNA usually exhibit low methylation levels and are rapidly
cleared from the maternal circulation after delivery. Although
cf-fDNA is mainly applied in prenatal diagnosis, the extremely
short half-life, estimated to be <1 h, is similar to the
clearance rate of linear cfDNA, suggesting the general rule of
rapid metabolism of cfDNA in the body, which has reference
significance in understanding its dynamic changes (47). Selective amplification of cf-fDNA
enables targeted analysis in maternal plasma, making it a potential
tool for non-invasive prenatal paternity testing (48).
Although ctDNA, cf-mtDNA and cf-fDNA have notable
potential, their extremely low concentrations in the circulation
limit their clinical applications, particularly in the early
detection of diseases such as cancer (49,50).
This challenge is particularly pronounced in cases with low tumor
burden. The continued development of high-sensitivity detection
technology is key to promoting its extensive clinical application
and ensuring its role in precision medicine (Fig. 2).
As a carrier of molecular information for tumors,
cfDNA provides biological insights into revealing tumor-specific
mutations, drug resistance mechanisms, tumor heterogeneity, clonal
selection and epigenetic marks. Therefore, cfDNA has become a
notable tool in understanding tumor biology and disease progression
(12,51). Highly proliferative cancer cells are
prone to cell cycle disorders and apoptosis, and apoptosis induced
by their rapid proliferation may release cfDNA. This release
mechanism makes it possible to detect tumor-related genetic and
epigenetic changes, including drug resistance mechanisms, in plasma
DNA, providing a non-invasive method in monitoring tumor evolution
and treatment response (19).
DNA methylation is a heritable and reversible
epigenetic modification that is tightly regulated and highly
conserved in normal cells. However, in tumor cells, DNA methylation
patterns are markedly altered, including silencing of tumor
suppressor genes through hypermethylation, activation of
proto-oncogenes through hypomethylation and increased genomic
instability. The fragment size and terminal distribution of cfDNA
molecules will vary depending on their DNA methylation status. DNA
methylation affects cfDNA fragment size by regulating nuclease
cleavage preference during apoptosis (52). Lower DNA methylation levels lead to
a looser chromatin structure, increasing nucleosome accessibility,
thereby enhancing nuclease activity in exposed regions, resulting
in shorter DNA fragments (53).
Differential methylation analysis of multiple genes in cfDNA has
potential application in disease screening. In 2023, Manoochehri
et al (54) identified
differential methylation in cfDNA from independent plasma samples,
particularly in differentially methylated regions associated with
sperm-associated antigen 6, LINC10606, tubulin folding cofactor D
and zinc finger protein 750 genes. By analyzing the methylation
signatures of these differentially methylated regions, researchers
constructed a methylation score. This score demonstrated notable
potential in distinguishing patients with triple-negative breast
cancer (TNBC) from healthy controls, suggesting that the
methylation score could serve as an effective biomarker for TNBC
diagnosis (54). For example, in
patients with TNBC, all genes except GATA3 demonstrated markedly
higher methylation frequencies compared with that in healthy
individuals. In addition, Wnt5A and Sox17 methylation were markedly
associated with poor prognosis and shorter survival, while
kallikrein-related peptidase 10 methylation was associated with
more aggressive clinicopathological features and higher recurrence
rates. These findings highlighted the potential of gene-specific
methylation patterns as prognostic markers in breast cancer
(55). In a 2021 study, Wang et
al (56) reported that MutL
homolog 1 promoter methylation in cfDNA can lead to DNA repair
defects, microsatellite instability (MSI) and thus, promote
colorectal cancer development. This study highlighted the role of
DNA methylation in regulating gene expression and driving
tumorigenesis. Abnormal DNA methylation is one of the hallmarks of
human cancer, often occurring in the early stages, making it a key
biomarker for early detection and diagnosis. For example, in 2023,
Kim et al (57) demonstrated
that combining the cfDNA methylation markers ring finger protein
135 and lactate dehydrogenase B with α-fetoprotein can identify
~70% of hepatocellular carcinoma cases, markedly improving the
liver cancer diagnosis rate. Furthermore, cfDNA methylation
analysis can reveal tumor heterogeneity. In February 2024, Heeke
et al (58) confirmed that
the tumor phenotype of small cell lung cancer evolves with disease
progression and cfDNA methylation can identify different subtypes
at the transcriptional level. This finding provides a valuable tool
in guiding personalized treatment strategies. The role of DNA
methylation in tumors is not limited to gene regulation and tumor
initiation, but also includes identifying tumor heterogeneity,
affecting genome stability and helping tumor cells adapt to the
environment. Further understanding of these aspects is key to
advancing cancer biology research and improving treatments.
cfDNA not only reflects methylation characteristics
but also provides key clues to study acquired drug resistance.
Tumor drug resistance can emerge at any stage of cancer treatment
and is often driven by target gene mutations, drug target
amplification and adaptive changes in tumor cells at the genetic,
epigenetic and microenvironmental levels. Genetic alterations in
DNA repair pathways associated with acquired resistance can be
identified using cfDNA analysis. For example, cfDNA sequencing has
revealed complementing mutations in BRCA1 and BRCA2 in breast
cancer, elucidating potential mechanisms of resistance to
platinum-based chemotherapy drugs such as cisplatin and carboplatin
(40,59). These complementing mutations can
partially or completely restore the DNA repair function of tumor
cells, allowing them to effectively repair DNA double-strand breaks
induced by platinum drugs, ultimately leading to chemotherapy
resistance. Tumor cells can also become resistant to immunotherapy
through mutations that promote immune evasion, which can be
detected by ctDNA sequencing. In other tumor types, cfDNA has also
made notable progress in revealing the tumor microenvironment and
drug resistance mechanisms. For example, in 2024, Skoulidis et
al (60) reported that
serine/threonine kinase 11 and Kelch-like ECH-associated protein 1
mutations in lung cancer lead to immunosuppression in the tumor
microenvironment, which is characterized by CD8+ T-cell
dysfunction and an increase in myeloid-derived suppressor cells.
These findings in lung cancer suggested that cfDNA analysis has the
potential to assess the tumor immune microenvironment and predict
the efficacy of immunotherapy, which also has implications for
breast cancer. In response to the aforementioned immunosuppressive
mechanism, dual immune checkpoint inhibition can restore
CD4+ T-cell function and reprogram myeloid cells to
target the elimination of tumor cells (60). This strategy could help overcome
drug resistance and enhance the efficacy of tumor immunotherapy.
Identifying immune evasion mechanisms is key to combating
resistance to immune-based therapies. In hormone-driven cancer,
such as estrogen receptor-positive (ER+) breast cancer,
endocrine therapy resistance is often associated with mutations or
downregulation of ER genes. Estrogen promotes tumor cell
proliferation by binding to ER, causing breast gland mitosis.
Therefore, patients with ER+ breast cancer usually have
a lower risk of early recurrence and improved response to endocrine
therapy (61). However, several
studies have reported that circular RNA (circRNA)-Scm-like with
four mbt domains 2 (SFMBT2) can promote the expression of Quaking I
protein, which may contribute to endocrine resistance (62). Understanding cfDNA-based resistance
mechanisms is key to guiding precision oncology strategies,
improving treatment efficacy and developing novel therapeutic
interventions. Further studies have demonstrated that increased
expression levels of circRNA-SFMBT2 promote the growth of
ER+ breast cancer cells and exacerbate resistance to
endocrine therapies such as tamoxifen (62). ctDNA detection provides a novel
approach in studying the resistance mechanism of endocrine drugs
(63). As tumors accumulate genetic
mutations, cfDNA analysis can track mutations associated with drug
resistance (64). cfDNA analysis
can detect secondary mutations in key targeted genes [for example,
G protein-coupled receptor 155 (GPR155), ADAM metallopeptidase with
thrombospondin type 1 motif 20 (ADAMTS20), titin (TTN) and BRAF],
thereby promptly identifying the occurrence of drug resistance and
optimizing treatment strategies. For example, in 2022, Lee et
al (65) used cfDNA whole-exome
sequencing (WES) to identify secondary mutations, including GPR155
(I357S), ADAMTS20 (S1597P) and TTN (R7H), in patients with lung
adenocarcinoma and colorectal cancer following EGFR targeted
therapy and chemotherapy. These mutations occur frequently during
acquired resistance, highlighting the value of cfDNA sequencing in
detecting resistance-associated genetic variants. In breast cancer,
ESR1 gene mutations can change the structure or function of the ER,
making cancer cells independent of exogenous estrogen. The most
common cause of resistance to aromatase inhibitors in patients with
breast cancer is ESR1 mutation, which is associated with disease
recurrence, progression and resistance to endocrine therapy in
advanced breast cancer. Continuous cfDNA monitoring for ESR1
mutations serves a key role in guiding personalized treatment
decisions and adjusting therapeutic strategies (66,67).
Furthermore, in a 2023 study, Serrano et al (68) demonstrated that KIT and platelet
derived growth factor receptor α gene mutations in patients with
gastrointestinal stromal tumors can be identified using ctDNA
sequencing technology. The detection of these mutations is
particularly valuable in the context of resistance to targeted
therapy, as it is directly associated with patient prognosis.
During tumor treatment, novel mutations or gene amplifications may
appear that alter drug targets or reduce drug binding efficiency,
ultimately leading to drug resistance. In summary, cfDNA serves as
a dynamic biomarker that reflects the genetic evolution and drug
resistance mechanism of tumor cells throughout the treatment
process. By analyzing key genetic changes in cfDNA, clinicians can
potentially predict drug resistance and adjust treatment options in
a timely manner, thereby improving patient outcomes in the
future.
Obtaining tumor genomic information using
non-invasive cfDNA analysis enables early detection, treatment
monitoring and drug resistance assessment of cancer. CfDNA
fragments exhibit unique characteristics based on their source
cells, making them important biomarkers for tumor analysis. In
2024, Stanley et al (69)
analyzed cfDNA fragment patterns and identified that these patterns
could reflect the unique biological characteristics of tumors. The
study distinguished various cancer types, such as colorectal and
early breast cancer andesophageal and gastric cancer, providing a
key molecular basis for early tumor detection and screening
(69–71). Furthermore, whole-genome deep
sequencing of cfDNA has identified a large number of somatic
mutations in plasma samples with low tumor burden and has helped to
distinguish tumor-derived mutations from those associated with CH
in normal germline tissue (72).
This finding enables early detection of tumor-related gene
mutations and structural variations even when the tumor burden is
extremely low, opening up a novel way for more precise cancer
diagnosis and treatment. Furthermore, shallow whole-genome
sequencing (WGS) of cfDNA further advances clinical monitoring of
cancer treatments by accurately and consistently quantifying tumor
markers in advanced cancer (73).
The detection and analysis of cfDNA provides a solid molecular
basis for early tumor diagnosis, prognosis assessment and
personalized treatment strategies in the future.
cfDNA application extends beyond early detection and
encompasses broad dynamic monitoring of therapeutic response
assessment, recurrence warning and non-tumor disease monitoring.
Surveillance methods not only improve the specificity and
sensitivity of tumor detection but also identify tumor recurrence
earlier compared with imaging-based diagnosis. Using dynamic and
continuous monitoring of cfDNA, changes in tumor genome can be
tracked in real time, providing key support for personalized
treatment strategies and efficacy evaluation. For example, a
previous study by Tie et al (74) in 2021 reported that among patients
who underwent resection of colorectal liver metastases, those with
detectable ctDNA had lower recurrence-free survival (RFS) and OS
compared with those without detectable ctDNA. These findings
highlighted the prognostic value of cfDNA in early identification
of high-risk patients and guiding treatment adjustments. cfDNA WGS
technology has demonstrated high sensitivity and reliability in
monitoring tumor mutations and treatment response. A previous study
by Liu et al (75) in 2024
demonstrated that using ultra-low coverage genome sequencing, cfDNA
can detect the risk of second primary malignant tumors in patients
with Li-Fraumeni syndrome, providing novel biomarkers for early
intervention. Similarly, in advanced breast cancer, ESR1 and PI3K
mutations detected in plasma ctDNA are markedly associated with
prognosis and newly identified mutations can affect subsequent
treatment decisions (76). The
development of NGS technology has made cfDNA a notable tool for
longitudinal monitoring of metastatic breast cancer (77). The value of dynamic cfDNA monitoring
not only demonstrates application potential in oncology, but also
in non-tumor diseases. In type 2 diabetes mellitus management,
cfDNA levels are closely associated with glycemic control and can
serve as a key indicator to monitor disease progression and
complications (78). In organ
transplantation, cfDNA levels associate with transplant prognosis.
A previous study by the Moeller et al (79) in 2024 reported that increased plasma
cfDNA levels after heart transplantation are closely associated
with transplant rejection. Monitoring cfDNA levels can help detect
rejection early, guide timely clinical intervention and improve
patient prognosis in the future. In addition, cfDNA has also
exhibited notable monitoring value in kidney and liver
transplantation (80,81).
In summary, cfDNA has demonstrated unique advantages
in monitoring both tumor and non-tumor diseases, particularly in
early diagnosis, disease progression tracking and treatment
response assessment. With the continuous advancement of technology,
cfDNA has the potential to develop into a routine diagnostic and
monitoring tool in clinical practice. This progress holds promise
not only in enhancing treatment outcomes but also extending patient
survival and improving the quality of life.
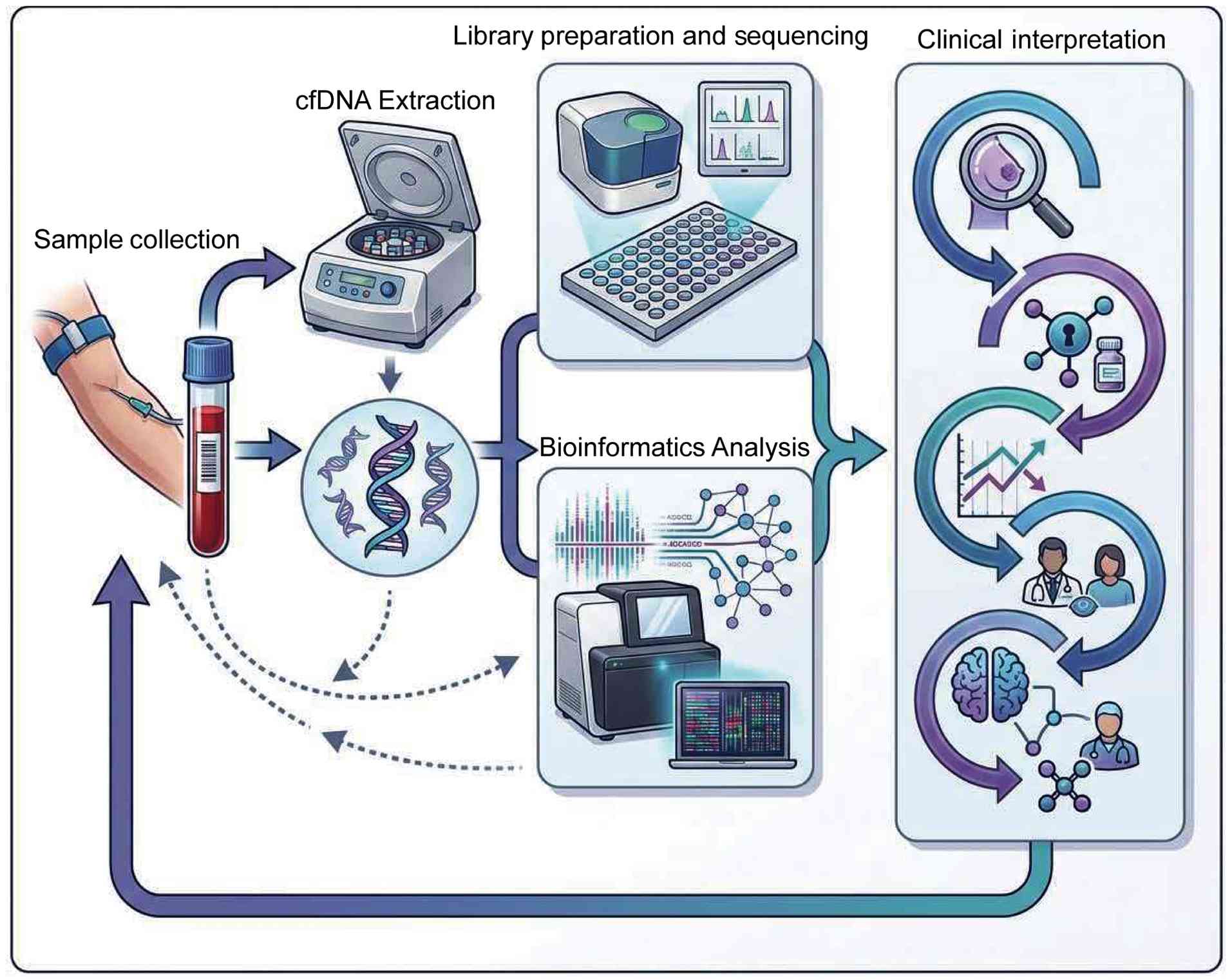
With the continuous development of biotechnology,
the extraction and analysis of cfDNA have become increasingly
efficient and convenient. Real-time monitoring of diseases, such as
breast cancer and myocardial infarction (82,83),
using simple blood samples not only markedly reduces patient trauma
and discomfort but also helps optimize the allocation of medical
resources and reduce overall medical costs (84). Currently, a variety of cfDNA
extraction and analysis technologies have been extensively used in
clinical diagnosis, physiological status assessment and basic
research. The main detection technologies include droplet digital
PCR (ddPCR), NGS and other cutting-edge emerging technologies
(85,86). The continuous improvement of these
methods has improved the sensitivity and accuracy of cfDNA
analysis, further expanding its application potential in precision
medicine and disease monitoring.
The basic principle of ddPCR involves partitioning
the sample DNA into a large number of micro-reaction units and
achieving absolute quantification of target DNA molecules by
counting the number of positive droplets after PCR amplification.
The workflow of ddPCR mainly includes key steps such as sample
preparation, droplet generation, PCR amplification, data analysis
and result reporting. Compared with traditional quantitative PCR
(qPCR), ddPCR enables high-sensitivity absolute quantification
without the need for a standard curve. The limit of detection in
ddPCR reaches 0.01–1.0% of genomic material, demonstrating notably
enhanced performance in detecting low-abundance point mutations and
CNVs. This inherently high sensitivity and precision make ddPCR
indispensable in clinical diagnostics and research applications
(87).
One of the key advantages of ddPCR is its high
sensitivity and specificity in cfDNA detection. Due to the short
half-life of cfDNA, timely and efficient purification is key to
ensuring the accuracy of analytical results (88). ddPCR enables precise quantification
of cfDNA in plasma and can effectively detect mtDNA after DNA
extraction. However, when using plasma directly as a template, the
detection process may be affected by vesicular structures such as
exosomes (89). In order to improve
the efficiency of DNA purification, researchers have increased the
cfDNA purification efficiency by up to 95-fold compared with the
original method by optimizing digestion conditions, liquid handling
parameters and magnetic bead processing procedures (90). Another major advantage of ddPCR is
its strong anti-interference ability. Traditional PCR methods are
often interfered by inhibitors present in complex biological
samples; however, ddPCR can markedly reduce such interference due
to its compartmentalized reaction unit, thereby improving the
reliability, stability and reproducibility of clinical sample
analysis (91). Furthermore, ddPCR
has demonstrated notably increased sensitivity compared with
traditional qPCR in detecting tumor-associated cfDNA and can still
accurately identify genetic variants even when the variant allele
frequency is as low as 0.1% (92,93).
Previous studies have reported that ddPCR detection of ctDNA has
high sensitivity, specificity and accuracy. For example, it has
been proven to be an effective tool in the assessment of minimal
residual disease in Ewing sarcoma and holds promise in guiding
future treatment strategies (94).
This finding provides key reference for cfDNA detection of minimal
residual disease in breast cancer. Similarly, selecting the
appropriate sample type is key to optimizing cfDNA detection.
Compared with serum, plasma can reduce the dilution effect of
non-tumor DNA, thereby improving the sensitivity of ctDNA detection
(95).
However, the automation of ddPCR technology has been
constrained by the complexity of liquid handling, particularly
during the conversion of ml-scale samples into nl droplets. To
overcome this challenge, researchers have developed a novel
integrated ddPCR microdevice, realizing a fully automated
‘sample-to-result’ detection process. The device can identify rare
tumor mutations with a detection sensitivity as low as ~1% and
requires only 2 ml of human plasma sample (96). This innovation provides a practical
solution for the automation of liquid biopsies. Further research
conducted a systematic evaluation of 15 different cfDNA extraction
and downstream analysis technologies (97,98).
The results demonstrated that automated extraction protocols and
PCR-based quantitative methods performed most notably in terms of
consistency and accuracy, with PCR-based methods demonstrating a
clear advantage. This indicated the applicability of cfDNA for
nucleic acid extraction and mutation detection and highlighted the
key role of PCR-based methods in establishing clinically feasible
workflows prior to cfDNA analysis (99).
NGS has been extensively applied in oncological
research and clinical practice. The main sequencing strategies
include WGS, WES, transcriptome sequencing (RNA-seq), genome-wide
association studies, targeted sequencing, CNVs analysis and MSI
detection (100–102). The standard workflow of NGS
includes several key steps: Sample preparation, nucleic acid
extraction, library construction, sequencing execution and
comprehensive data analysis. These steps ensure the accuracy and
reliability of genomic information, facilitating early detection of
cancer, treatment monitoring and the development of personalized
treatment strategies. Sample preparation is the first key step in
NGS and its quality directly affects data quality and reliability.
A previous study by Ritter et al (103) demonstrated that cfDNA was
successfully extracted from serum samples stored for 30 years and
24 cancer-specific mutations were identified in 25 sequenced
samples from 52 patients with breast cancer. This study highlighted
the robustness and specificity of NGS technology, confirming its
suitability for the analysis of long-term stored serum samples.
Similarly, a previous study of Jiang et al (104) in 2020 also reported that cfDNA can
maintain high quality and be suitable for NGS even after
cryopreservation at −80°C for 1–6 years. After DNA extraction and
quality assessment, library construction is a key step in the NGS
process, which directly affects sequencing accuracy and data
integrity. Using Bioanalyzer and high-sensitivity DNA chromatin
immunoprecipitation to determine the concentration and size
distribution of cfDNA can comprehensively evaluate the quantity,
quality and potential genomic DNA contamination of DNA. This step
is key to optimizing the library preparation process and ensure
high-quality sequencing results (105).
The traditional method of double stranded DNA
library preparation mainly focuses on the analysis of 167 bp double
stranded single nucleosomes and other oligonucleosomes derived from
cfDNA. However, Troll et al (106) innovatively developed an efficient
single stranded library preparation technology. This method can
construct complex libraries from as little as 1 ng of input DNA in
only 2.5 h, while completely retaining the natural ends of template
molecules. This single stranded library method not only markedly
simplifies the preparation process, but also notably expands the
potential application of cfDNA analysis. NGS serves a key role in
both cancer research and clinical applications. By continuously
optimizing the NGS workflow, clinical needs of patients can be
further met, such as faster turnaround times for timely treatment
decisions and enhanced sensitivity for detecting low-frequency
mutations in early-stage or minimal residual disease settings.
Targeted NGS of cfDNA enables dynamic monitoring of tumor
heterogeneity and its response to targeted therapies (107). This technology aids in detecting
gene mutations in ctDNA, providing valuable information in
understanding tumor evolution and precise treatment effect. In
tumor genome analysis, a key challenge faced by NGS is to
accurately distinguish tumor specific variants from sequencing
artifacts and normal germline variants. To address the problem of
false-positives, researchers have developed a machine learning
model that integrates additional filtering steps. When applied to
QIAseq® data (Qiagen GmbH), its sensitivity is 35% and
accuracy is 36%, highlighting its effectiveness in capturing tumor
specific variation. By integrating germline DNA analysis, the study
validated the somatic origin of the identified variants and lastly
detected seven variants, six of which were consistent with the
germline validation strategy (108).
In recent years, in addition to digital PCR and NGS,
a series of emerging detection technologies have also notably
enhanced cfDNA analysis capabilities. Traditional cfDNA extraction
methods often demonstrate low efficiency and poor purity. To
address this, Jeon et al (109) developed a nanostructured
conductive polymer platform for efficient capture and release of
cfDNA. They validated the effectiveness of this platform using
unprocessed plasma samples from patients with breast and lung
cancer. Research results reported that this platform could recover
tumor-specific cfDNA with high yield and purity by improving
efficiency. In addition, Raman spectroscopy technology has been
explored for cfDNA analysis. Researchers reported notable
differences in cfDNA spectral patterns in patients with breast
cancer receiving neoadjuvant therapy, suggesting that cfDNA
molecular signatures may be associated with disease status
(110,111). Similarly, by analyzing cfDNA,
researchers have also identified unique biomolecular fingerprints
that can distinguish healthy individuals from those with
prediabetes and those with type 2 diabetes mellitus (112). In metastatic breast cancer,
molecular barcoding technology (MB-NGS) is used to detect ESR1
mutations in cfDNA. Previous studies have demonstrated that MB-NGS
has higher sensitivity compared with traditional NGS, enabling the
identification of more mutations in cfDNA samples (113,114).
In summary, the continued progress of these cfDNA
analysis technologies has jointly provided strong technical support
for early detection, precise diagnosis and personalized treatment
of diseases, indicating a broad and promising future in clinical
application (Fig. 3).
Researchers hypothesize that cfDNA has notable
potential in the clinical management of breast cancer if the
following application scenarios can be realized: i) Distinguish
breast cancer patients from benign breast lesions or healthy
individuals to achieve early detection and diagnosis (115); ii) identify and monitor
micro-occult metastases to improve the sensitivity of metastasis
detection (116); iii) predict OS
based on cfDNA analysis to improve the accuracy of prognostic
assessment (117); iv) evaluate
treatment response using dynamic cfDNA monitoring before imaging
evaluation, providing real-time insight into efficacy (118); and v) detect genomic changes
associated with treatment resistance to guide the formulation of
personalized treatment strategies (119). The successful implementation of
these applications may markedly improve the diagnosis, treatment
and prognosis management of breast cancer, therefore improving
patient outcomes in the future (120).
The concentration of cfDNA, modifications (such as
methylation, nucleosome positioning and histone modifications) and
genetic mutations can all reflect the presence and progression of
tumors (121,122). Compared with imaging-based
screening and early diagnosis, cfDNA analysis may provide a more
convenient and cost-effective method, particularly for patients
with occult breast cancer. Traditionally, pathological biopsy is
considered the gold standard for breast cancer diagnosis, but it
requires imaging guidance and is an invasive procedure. As a
non-invasive alternative, cfDNA analysis can effectively meet these
diagnostic needs. Early detection of breast cancer markedly affects
treatment options and adjuvant therapy decisions, increasing the
likelihood of treatment success. Breast cancer cells, particularly
aggressive subtypes, exhibit higher mitotic and glycolytic
activities compared with normal cells and less aggressive tumor
subtypes, resulting in increased cfDNA release and higher ctDNA
proportions. This biological characteristic provides a theoretical
basis for the application of cfDNA in the screening of aggressive
breast cancer subtypes that are rapidly growing, highly invasive
and have high mortality (123).
Elevated serum cfDNA levels are closely associated
with breast cancer. Variant allele frequencies (VAF) as low as
0.08% can be detected in plasma cfDNA samples from healthy
individuals. These mutations are consistent with pathogenic
mutations that cause certain individuals to develop benign tumors
or invasive breast cancer within a decade (124,125). Furthermore, changes in DNA
methylation profiles in blood can be observed several years before
clinical detection of breast cancer, suggesting its potential as an
early biomarker (126). In
patients with breast cancer, cfDNA fragments from hypomethylated
regions were shorter compared with that in healthy individuals. In
addition, the proportion of short cfDNA fragments in hypomethylated
regions is higher compared with that in hypermethylated regions
(127). Since cfDNA is enriched in
hypomethylated genomic regions, single-base resolution analysis of
genome-wide DNA methylation in blood can effectively distinguish
early breast cancer from benign tumors (128). Analysis of cfDNA in healthy
individuals may become a promising tool for breast cancer screening
and early diagnosis, capable of detecting genomic instability and
providing key insights into early events in tumor formation.
Nucleosomes serve a key role in cancer by affecting
genome structure and gene expression. CfDNA originates from genome
regions protected by nucleosomes from enzymatic digestion, with the
majority of cfDNA fragments consisting of mononucleosomal DNA.
Longer cfDNA fragments, such as dinucleosomes and trinucleosomes,
often carry more mutational signatures and may contain additional
nucleosomes, thereby providing further insights into tumor biology
(129). cfDNA nucleosome profiling
analysis can accurately reflect the tissue origin and transcription
factor activity of different breast lesions, effectively
distinguishing benign and malignant cases (130). Of note, the dynamic changes in
transcription factor-associated nucleosomes in plasma cfDNA reveal
ER-driven status in breast cancer. The degree of enrichment of
transcription factor footprints in plasma samples corresponds to
the binding strength of transcription factors in primary tumor
tissue, making it possible to identify ER+ breast cancer
using plasma-based transcription factor footprint analysis
(131). Further understanding of
these nucleosome-related changes will help elucidate the molecular
pathological mechanisms of breast cancer and accelerate the
development of more precise cfDNA diagnostic strategies.
Breast cancer is a complex and heterogeneous disease
whose development is driven by multiple genetic and environmental
factors, including inherited mutations (e.g., BRCA1/BRCA2, TP53,
PALB2), hormonal exposure (e.g., early menarche, hormone
replacement therapy), reproductive history (e.g., nulliparity, late
age at first pregnancy), lifestyle factors (e.g., high alcohol
consumption, obesity) and ionizing radiation (136,137) Among them, 5–10% of breast cancer
cases have a clear genetic predisposition. Germline or somatic
mutations in specific genes serve a key role in the development and
early screening of breast cancer. By analyzing the genomic
sequences of cfDNA from patients with breast cancer, researchers
can detect genetic abnormalities and use bioinformatics methods to
assess the pathogenicity of these mutations. For example, a
previous study confirmed that the V465M mutation of the SMAD4 gene
is markedly associated with breast cancer. This mutation may
enhance tumor invasion and metastasis by inhibiting the TGF-β
signaling pathway (138). In
addition, cfDNA-based ESR1 mutation detection has demonstrated high
sensitivity and specificity, with an overall accuracy of 88.96%, a
positive predictive value of 56.94% and a negative predictive value
of 88.53%. This method is hypothesized to become a key diagnostic
tool in identifying ESR1 mutations in patients with breast cancer
(139). Recent studies have
identified a variety of cfDNA gene mutations that can serve as
potential biomarkers for breast cancer, which are expected to serve
a key role in breast cancer screening and diagnosis in the future
(140) [Table SI (141–149)]. The identification of these genes
not only enhances current understanding of the molecular
pathogenesis of breast cancer, but also provides novel avenues for
early diagnosis and personalized treatment. Effective use of these
genetic markers is hypothesized to notably improve the early
detection rate and diagnostic accuracy of breast cancer, thereby
providing patients with more timely intervention opportunities
[Table SI (141–149)].
Due to the special physiological status of lactating
women, there are certain limitations in the implementation of
traditional biopsy methods. As an alternative, breast milk has
emerged as a non-invasive liquid biopsy sample that exhibits
notable potential for breast cancer screening in postpartum women.
Saura et al (150) used
ddPCR technology to analyze ctDNA in breast milk, finding that
tumor mutations could be detected in 87% of cases, while in matched
plasma samples, mutations failed to be detected in 92% of cases.
This result suggested that ctDNA in breast milk is more sensitive
compared with that in plasma in detecting breast cancer. In 2
patients, ctDNA was detected in breast milk 18 and 6 months before
standard diagnosis, respectively, further highlighting its
potential as an early screening and diagnostic tool for breast
cancer (150). In addition,
Cunningham and Turner (151) used
NGS technology to simultaneously analyze 54 common breast cancer
mutated genes in breast milk and blood samples. The results
indicated that the sensitivity of ctDNA detection in breast milk
was markedly higher compared with that in plasma. This confirmed
the feasibility of breast milk ctDNA analysis and suggested that it
may become an effective strategy for breast cancer screening in the
future.
The treatment strategy for breast cancer is mainly
based on tumor stage, molecular characteristics and the overall
physical condition of the patient. Tumor staging is a key factor in
determining the direction of basic treatment, while molecular
characteristics influence the selection of targeted therapy,
endocrine therapy and chemotherapy regimens. Currently, standard
treatments for breast cancer include surgical resection,
radiotherapy, chemotherapy, targeted therapy and adjuvant therapy.
In recent years, the development of targeted therapy has made
precise intervention against cancer cell-specific molecular targets
(mainly proteins) and driver gene mutations (such as PIK3CA, ESR1
and HER2) a reality (152),
notably improving the prognosis of patients with breast cancer
(153). However, effectively
dealing with intra-tumor heterogeneity, dynamically changing
therapeutic responses, and the generation and development of drug
resistance is still a major clinical challenge (154). CDK4/6 inhibitors have become the
standard treatment for ER+/HER2− advanced
breast cancer. In this context, monitoring ctDNA levels in patients
with breast cancer aids in the real time assessment of treatment
response. Continuous ctDNA analysis is a key tool in evaluating the
efficacy of CDK4/6 inhibitors. For example, a previous study
involving 33 patients with HR+/HER2−
metastatic breast cancer demonstrated that ctDNA analysis could
detect disease progression months before radiographically visible
changes, with a sensitivity and specificity of 75 and 92%,
respectively (155). This finding
suggested that continuous ctDNA monitoring can enable early
detection of disease progression and real-time assessment of the
efficacy of CDK4/6 inhibitors. To further analyze the genetic
mechanisms associated with disease progression and drug resistance,
ESR1 mutations detected in ctDNA have been identified as notable
independent predictive markers of hormone therapy resistance.
Notably, CDK4/6 inhibitors exhibit the ability to overcome
ESR1-dependent drug resistance, which further highlights their
clinical application value in the management of drug-resistant
breast cancer (156).
CfDNA detection provides a highly sensitive and
specific non-invasive method for disease monitoring in patients
with breast cancer. Notably, ctDNA can be detected up to 2 years
before the onset of distant metastatic recurrence, with notably
increased predictive power compared with traditional imaging
examinations, CA15-3 biomarker analysis, clinical examinations and
liver function tests. Furthermore, elevated ctDNA levels are
associated with poor prognosis and detection of ctDNA mutations
before treatment often predicts unfavorable survival outcomes
(165,166). In the majority of patients with
breast cancer, ctDNA can be detected months before clinical
recurrence with extremely high specificity. This advance detection
provides a key time window for the timely introduction of
non-cross-resistant therapies, thereby effectively preventing or
delaying clinically visible metastasis and recurrence. Further
research confirmed that ctDNA analysis can effectively predict the
recurrence of all major breast cancer subtypes, with a detection
time on average 10.7 months earlier than clinical recurrence
(167). Furthermore, the failure
to clear ctDNA after treatment is a key predictor of adverse
outcomes, including distant metastasis and local recurrence.
Notably, in patients who did not achieve pCR, a higher rate of
ctDNA clearance was markedly associated with improved survival
(168). Continuous monitoring of
ctDNA enables the dynamic, real-time assessment of treatment
response, providing unique clinical opportunities for treatment
adjustments aimed at delaying metastatic recurrence and improving
patient outcomes. In brain metastatic breast cancer, ctDNA can be
detected in both cerebrospinal fluid and plasma. A previous study
involving 30 patients with breast cancer with leptomeningeal
metastases employed ultra-low-depth WGS technology to evaluate
ctDNA scores. The results demonstrated that ctDNA was present in
the cerebrospinal fluid of all patients with brain metastases,
regardless of negative cytology results or borderline MRI findings.
During intrathecal therapy, continuous ctDNA monitoring revealed
that declining ctDNA levels were markedly associated with prolonged
survival, while elevated ctDNA could be detected up to 12 weeks
before clinical progression (169). Overall, ctDNA, as a non-invasive
biomarker, exhibits strong potential in quantitatively monitoring
treatment response and disease progression in brain metastatic
breast cancer, aiding in the optimization of early intervention
strategies and guide personalized precision treatment.
In breast cancer response evaluation and disease
monitoring, cfDNA-based analysis of specific gene mutations is
increasingly becoming a key biomarker. Specific genetic mutations
in cfDNA not only provide a notable basis for personalized
treatment plans, but also enable dynamic, real-time tracking of
disease progression and treatment response, therefore optimizing
clinical decisions and improving patient outcomes. A previous study
involving 255 patients with stage IV breast cancer reported that
89% had at least one genetic mutation detected in their ctDNA
samples. Specifically, in HR+ patients, the most common
mutated genes are PIK3CA, ESR1 and TP53; in HER2+
patients, TP53, PIK3CA and Erb-b2 receptor tyrosine kinase 2
(ERBB2) mutations are the most common, with ERBB2 changes mainly
manifested as CNVs; in patients with TNBC, TP53 and PIK3CA
mutations dominate, and CNVs of Myc, cyclin E1 and PIK3CA are often
observed. Across the entire patient cohort, the mutation rates of
PIK3CA, ESR1 and ERBB2 were 39.6, 16.5 and 21.6%, respectively
(170). Recent studies have
further emphasized the key role of cfDNA genetic alterations in
breast cancer treatment monitoring, with PIK3CA and ESR1 being the
most commonly used molecular markers in assessing disease
progression and treatment response (171). Table
SII (172–182) summarizes the applications of these
cfDNA-based genetic tests in breast cancer management,
demonstrating their growing clinical value.
The prognosis assessment of breast cancer mainly
relies on clinicopathological characteristics and molecular
markers. However, accurate prognostic assessment still faces
several challenges due to the complexity of tumor heterogeneity and
difficulty in detecting early recurrence. Although the detection
rate of early-stage breast cancer is high, ~20% of patients still
experience recurrence after receiving standard treatment (187). Therefore, accurate assessment of
patient prognosis is key to optimizing subsequent management and
improving patient outcomes. In operable breast cancer, univariate
analysis results revealed that the detection of ctDNA at baseline,
after neoadjuvant therapy and during follow-up was markedly
associated with worse disease-free survival (DFS) and OS (188). In addition, a high baseline ctDNA
CNVs burden was confirmed to be associated with worse OS and RFS
(189). Sustained increases in
ctDNA levels after treatment strongly predict worse OS (190). In a cohort of patients with
primary breast cancer who underwent surgery and adjuvant
chemotherapy, ctDNA+ patients had notably shorter
progression-free survival (PFS) compared with ctDNA−
patients. Furthermore, mutational heterogeneity of ctDNA was
negatively associated with PFS. In TNBC, ctDNA was detected
positive in all relapsed patients within a median of 8 months,
whereas non-relapsed patients remained ctDNA negative during a
median follow-up of 58 months. ctDNA positivity has been reported
to be highly associated with shorter RFS and OS (191,192). A previous study involving 84
patients with high-risk early-stage breast cancer treated with NAC
or NAC combined with MK-2206 (an AKT inhibitor) demonstrated that
the ctDNA positivity rate was highest before treatment and
gradually decreased as the treatment progressed. More notably,
ctDNA clearance after treatment was closely associated with pCR and
the prognosis of ctDNA− patients who did not reach pCR
was markedly improved compared with that of ctDNA+
patients (168). Persistent ctDNA
positivity is considered a notable predictor of adverse outcomes
and metastatic recurrence, further confirming the key clinical
value of ctDNA detection in guiding treatment decisions.
Previous studies have reported that ctDNA testing
can detect recurrence earlier than imaging and clinical symptoms,
with an average lead time of 3.81 months. In patients with primary
breast cancer who receive postoperative adjuvant chemotherapy,
ctDNA is often detected before clinical or imaging confirmation of
recurrence, with a maximum predictive time window of up to 38
months (152,193). Serial postoperative ctDNA
assessment not only provides valuable prognostic information but
also creates a valuable time window for early therapeutic
intervention. The sensitivity range of ctDNA in detecting breast
cancer recurrence ranges from 0.31 to 1.0 and its specificity
ranges from 0.7 to 1.0. On average, it can confirm recurrence 10.81
months earlier than imaging (188). Changes in ctDNA levels can
indicate disease progression weeks before imaging findings, making
it a notable biomarker for real-time monitoring of disease status.
In metastatic breast cancer, ctDNA levels dynamically reflect tumor
burden and progression prior to treatment. The study has reported
that a high tumor burden index before treatment was markedly
associated with shorter OS, while a reduction to <0.02% during
treatment was associated with longer PFS and OS (194). In addition, the tumor burden index
can effectively distinguish treatment response (complete
response/partial response) from disease progression. Notably, ctDNA
tumor fraction ≥10% in metastatic breast cancer has been reported
to be markedly associated with OS and can serve as an independent
prognostic factor in multivariate analysis (195). Chromosomal instability (CIN) is a
hallmark feature of tumor development and progression. By driving
genomic instability, it endows tumor cells with a growth advantage,
enhances anti-apoptotic ability and promotes the development of
drug resistance. CIN can be quantified by somatic CNVs, MSI and
other chromosomal structural changes. In patients with breast
cancer, cfDNA CIN is associated with prognosis. Related studies
have reported that the median OS and PFS of patients with
metastatic breast cancer with high cfDNA CIN are markedly lower
compared with those with low CIN score (196). In addition, cfDNA CIN in patients
with recurrent breast cancer demonstrated high accuracy in
recurrence monitoring, with a positive rate of 77.6%, outperforming
traditional biomarkers CA15-3 and CEA. When combined with
traditional markers, the positive rate can be further increased to
88.7%. Particularly in patients with shorter DFS, cfDNA CIN
exhibits high prognostic predictive value (197). Further analysis of cfDNA in
patients with early-stage breast cancer reported that a high cfDNA
mutation burden was associated with worse RFS. Despite the
inconsistency between tumor tissue and cfDNA samples, specific
somatic variants in cfDNA are still associated with worse RFS,
suggesting that cfDNA mutation burden can serve as a prognostic
marker in patients with early-stage breast cancer (198). Among patients with metastatic
TNBC, cfDNA tumor fraction is detectable in 96.3% of cases, with
63.9% exhibiting somatic CNVs. Notably, cfDNA tumor fraction ≥10%
is markedly associated with worse metastasis-free survival.
Furthermore, specific somatic CNVs are highly expressed in patients
with metastatic TNBC and exhibit strong prognostic predictive value
(199). Overall, the detection of
high allele frequencies and a large number of non-synonymous
mutations in cfDNA of patients with metastatic breast cancer has
been confirmed to be markedly associated with poor OS (200). As key liquid biopsy biomarkers,
cfDNA and ctDNA serve a differentiated role in predicting treatment
response and evaluating prognosis of different breast cancer
subtypes. Analyzing its manifestations can provide notable insights
into breast cancer management. In patients with TNBC, cfDNA
concentration measured 3 weeks before treatment was inversely
associated with residual cancer burden (RCB), whereas ctDNA
concentration was positively associated with RCB at all time points
examined. In patients with HR+/HER2− breast
cancer, cfDNA concentration was not associated with NAC response;
however, patients with high cfDNA concentrations had lower distant
RFS compared with those with low concentrations. By contrast, in
patients with TNBC, cfDNA concentration had no notable effect on
survival, whereas ctDNA was associated with RFS at all time points
(201). In summary, compared with
cfDNA, ctDNA concentration is considered a more sensitive and
clinically practical prognostic predictor, serving as a key tool in
monitoring treatment response and guiding clinical
decision-making.
CTCs are cancer cells that detach from primary
tumors or metastatic sites and enter the blood circulation,
reflecting the invasion and metastasis potential of tumors. The
combined application of CTCs, cfDNA and ctDNA can improve the
accuracy of breast cancer prognosis assessment and provide a key
basis in formulating personalized treatment plans. Previous studies
have reported that both CTCs and ctDNA are key prognostic markers.
There were notable differences in PFS and OS based on CTC count and
ctDNA levels. Patients with CTC ≥5 or ctDNA percentage ≥0.5, as
well as those carrying ≥2 mutations, have markedly worse clinical
outcomes compared with patients below these thresholds (202). Furthermore, both total cfDNA
content and CTC count can serve as predictors of OS, while total
cfDNA levels alone can predict PFS and disease response. Notably,
the combined analysis of CTCs and cfDNA has been reported to
provide more accurate information for prognostic assessment
compared with traditional biomarkers such as CA15-3 and alkaline
phosphatase (203). Several
studies have further demonstrated that MET upregulation in
metastatic sites is markedly higher compared with that in primary
tumors and MET+ CTCs, elevated cfDNA levels and ESR1
mutations have all been reported to be closely associated with poor
prognosis (204–206). The integrated analysis confirmed
that CTC count ≥5 and high cfDNA levels are markedly associated
with shortened PFS and OS. When high CTC and high cfDNA levels
coexist, the risk of death for the patient is notably increased
(207). Furthermore, longitudinal
studies have reported that cfDNA levels can explain differences in
prognostic assessment based on CTCs. The 24-month DFS probability
was 52% for patients positive for both ctDNA and CTCs, compared
with 89% for patients negative for both ctDNA and CTCs. The mere
presence of ctDNA positivity is also associated with a lower
probability of DFS compared with ctDNA− cases (208). The combined detection of ctDNA and
CTCs improves the sensitivity and accuracy of breast cancer
prognosis assessment and provides key clinical insights for early
intervention and personalized treatment strategies in the
future.
Advances in early cancer detection technology and
the continued development of targeted anti-breast cancer therapies
have markedly increased the 5-year survival rate of breast cancer
to ~90% (209). However, despite
achieving complete remission after initial treatment, ~30% of
patients will eventually experience disease recurrence.
Furthermore, the 5-year survival rate for advanced breast cancer is
still low, at ~30% (210,211). This high recurrence rate and low
survival rate is largely attributed to the complex genetic
background of breast cancer, which continues to pose notable
challenges for its complete eradication. Current research focuses
on assessing patient survival outcomes by detecting specific
genomic alterations in cfDNA. These studies typically involve the
extraction of genomic DNA from tumor tissue or liquid biopsy
samples to identify genetic biomarkers with prognostic predictive
value. In addition to traditional clinical and histological
factors, these genetic markers are increasingly being considered as
notable adjunct tools for prognostic assessment (212–214). To gain further understanding of
the relationship between gene mutations and breast cancer
prognosis, the present review summarized recent studies on cfDNA
and ctDNA mutations and their impact on survival outcomes [Table SIII (215–225)].
The present section focused on elucidating the
detection results and clinical relevance of different gene
mutations in patients with breast cancer. These findings provide
novel perspectives and strong evidence for personalized treatment
strategies and improved prognostic assessment. From the
aforementioned studies, it is evident that the detection of genetic
mutations and their relationship with patient prognosis have become
a core area of breast cancer research. Different gene mutations can
affect the occurrence, development and response to treatment in
breast cancer, thereby affecting the survival outcome of the
patient. Table SIV (215–225) summarizes the key genes and their
functions that have been explored in multiple studies investigating
the relationship between gene mutations and various survival
indicators. The present review observed that gene mutations such as
TP53, ESR1 and PIK3CA have been confirmed to be markedly associated
with poor breast cancer survival outcomes in multiple studies. In
addition, certain less studied but equally key genes [such as
plasmacytoma variant translocation 1 (PVT1), CCCTC-binding factor
(CTCF), guanine nucleotide binding protein, α stimulating activity
polypeptide (GNAS) and Notch1] have also been identified to be
closely associated with the prognosis of patients with breast
cancer (226–229).
cfDNA, as a non-invasive liquid biopsy technology,
has demonstrated notable potential in the field of precise
diagnosis and treatment for breast cancer. Currently, the field is
at a key juncture transitioning from ‘technical verification’ to
‘clinical integration’ (115,213) The core focus lies in constructing
an integrated evidence chain that encompasses multi-dimensional
analysis of cfDNA, standardized processes and clinical utility
verification.
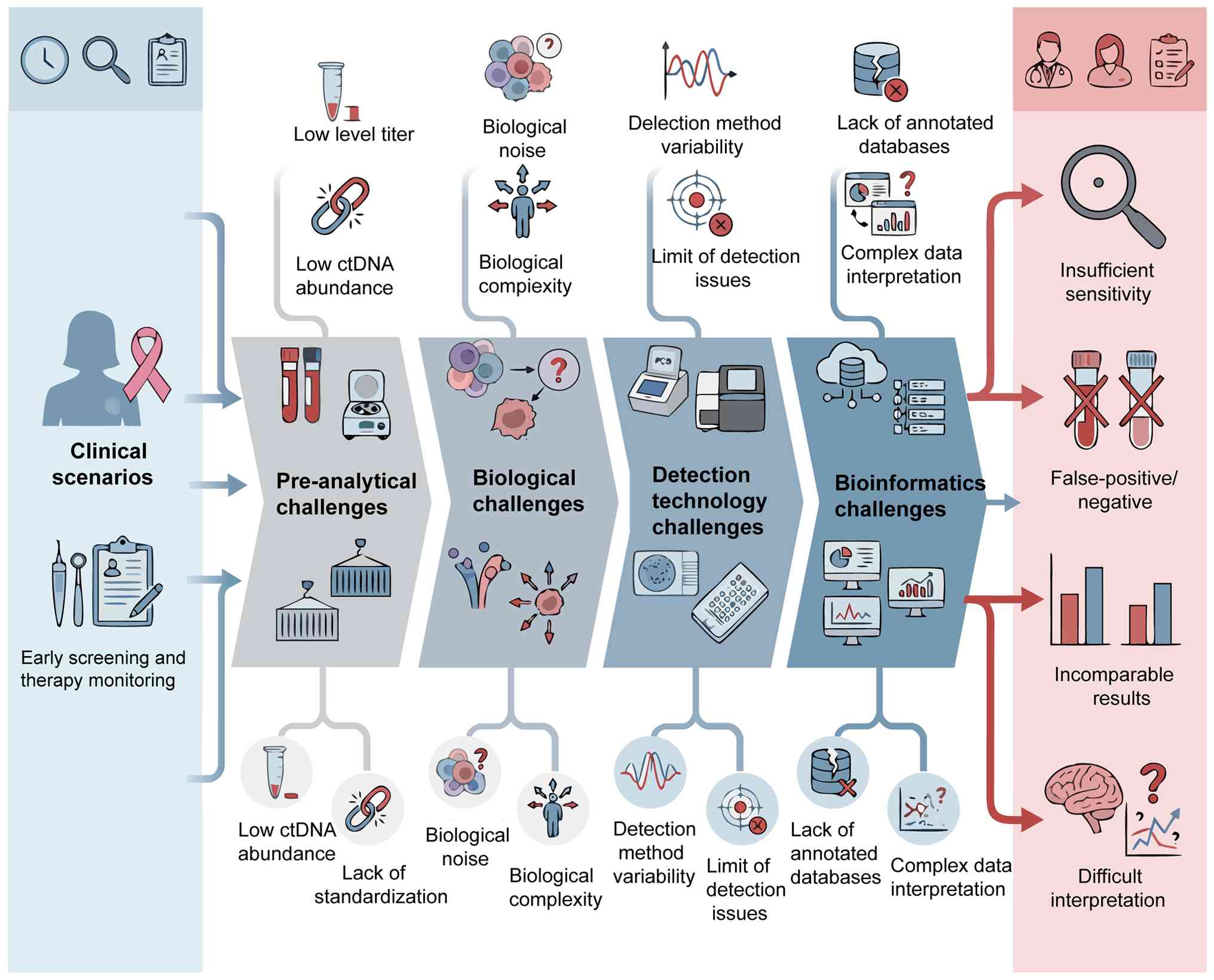
CfDNA faces multiple challenges at the biological,
technical and clinical application levels during the clinical
translation of breast cancer. These challenges are intertwined and
together constitute a complex dilemma for its integration into
clinical practice.
The presence of cfDNA in blood is a complex
biological process. In early-stage breast cancer, tumor-derived
ctDNA typically constitutes <0.01% of the total cfDNA, posing a
fundamental challenge in the detection of extremely low-abundance
signals (19,230). The release mechanism of cfDNA is
complex and diverse, including not only apoptosis and necrosis of
tumor cells, but also various factors such as active cell
secretion, dynamic changes in the tumor microenvironment and the
systemic physiological state of the host (such as inflammatory
response and strenuous exercise). These non-tumor-derived cfDNA
collectively constitute notable biological background noise,
markedly increasing the difficulty of tumor-specific cfDNA
detection (231). In addition, the
spatiotemporal heterogeneity of tumors implies that a single liquid
biopsy may not fully capture the characteristics of all subclones.
It may particularly miss tumor cells with weak hematogenous
metastasis capabilities or those located in specific
microenvironments (such as brain metastases), potentially resulting
in differences with tissue biopsy results (198). Therefore, it is necessary to
carefully evaluate the precise positioning of cfDNA analysis in
specific clinical scenarios: Whether it serves as an independent
alternative diagnostic tool or as an effective complementary
monitoring method for existing methods warrants further
research.
cfDNA analysis technology faces a dilemma of
multiple choices. dPCR has high sensitivity in detecting known
low-frequency variants but lacks the ability to explore unknown
variants (232,233). NGS enables broad-spectrum
screening, yet it still has limitations in ultra-low abundance
detection, cost control and complexity of bioinformatics analysis
(234). However, a more
fundamental and urgent challenge lies in the lack of a standardized
system for the entire cfDNA detection workflow. From sample
collection (such as anticoagulant selection), pretreatment (such as
centrifugation parameters), cfDNA extraction, library construction
and downstream bioinformatics analysis, there is a lack of unified
and extensively accepted standard operating procedures across all
key stages of the chain. This lack of standardization restricts the
reproducibility and comparability of different study results and
thus, reduces the clinical reliability of cfDNA detection (235). Therefore, establishing and
implementing an internationally recognized ‘clinical-grade’ cfDNA
testing quality management system is the cornerstone and
prerequisite for its successful translation from technology to
clinical practice.
Obtaining the cfDNA variation profile is the
starting point for analysis. The accurate interpretation of its
further clinical significance faces more complex and formidable
challenges. The core challenge lies in accurately distinguishing
background mutations from non-tumor sources such as tumor driver
mutations, non-functional passenger mutations and CH (CH of
indeterminate potential). This relies heavily on continuously
updated and improved clinical annotation databases (236). A more fundamental challenge is how
to effectively transform complex and dynamically changing cfDNA
data into specific, actionable clinical action guidelines. For
example, the scientific definition of a ‘positive’ threshold for
molecular residual disease (MRD) testing remains a key issue that
needs to be resolved. In addition, whether early or preemptive
intervention strategies based on dynamic changes in cfDNA can
markedly improve the long-term survival outcomes of patients still
needs to be verified by high-level evidence-based medical evidence.
However, there is still a lack of prospective interventional
clinical studies that can provide high-level evidence-based
evidence (237). In order to
achieve the deep integration of cfDNA data with clinical
decision-making pathways, it is key to establish a
multidisciplinary and cross-institutional collaboration mechanism
to jointly formulate rigorous and evidence-based clinical practice
guidelines b. In order to systematically explain the association
and logic between the aforementioned multi-dimensional challenges,
the present review presents a schematic diagram of the integration
dilemma of cfDNA clinical translation (Fig. 4).
To systematically promote the application of cfDNA
in breast cancer management, the present review proposed a staged
transformation strategy guided by the disease progression stage of
breast cancer and the maturity of cfDNA technology.
The short-term goal is to establish the clinical
utility of cfDNA analysis as a routine and reliable means of
disease monitoring in patients with metastatic breast cancer as a
priority. The core theoretical basis of this strategy is that
patients with metastatic breast cancer usually have a higher tumor
burden, making the cfDNA detection signal more notable.
Furthermore, there is an urgent and clear need for non-invasive,
real-time, dynamic monitoring methods in clinical practice. The key
transformation path at this stage focuses on the following three
core aspects: First, accurately verify the leadership and accuracy
of cfDNA dynamic changes in predicting imaging response and disease
progression through prospective studies; second, build a systematic
monitoring system to track the evolution of acquired mutations in
key driver genes (such as ESR1 and PIK3CA) in real time during
treatment; lastly, develop an accurate prognostic stratification
model based on cfDNA characteristics and optimize the screening
strategy for patients in clinical trials. The milestone in
achieving this stage of transformation will be the successful
completion of at least one prospective randomized controlled
clinical trial. This trial needs to demonstrate that treatment
regimen adjustments based on dynamic changes in cfDNA can markedly
and statistically improve the long-term survival outcomes of
patients.
Currently, with the sensitivity of detection
technology reaching ≤0.001% levels, the technical feasibility of
cfDNA analysis in MRD detection has been initially and
encouragingly verified. The core task of this stage is to further
integrate cfDNA detection technology into the clinical
decision-making process for adjuvant treatment and neoadjuvant
treatment in early-stage breast cancer. Its key tasks include:
First, verifying the validity and reliability of cfDNA clearance
status after neoadjuvant therapy as a surrogate endpoint for pCR;
second, establishing a standardized operating plan for
postoperative MRD monitoring and scientifically defining its
threshold and timing for clinical intervention. The key sign in
achieving transformation at this stage is confirmation, through
multiple large-scale prospective cohort studies, that adjuvant
treatment de-escalation or escalation strategies guided by ctDNA
MRD can optimize patient prognosis and reduce unnecessary treatment
toxicity.
The long-term goal is to use cfDNA technology to
build an accurate, non-invasive and efficient early screening
system for high-risk groups of breast cancer. This requires
overcoming the current challenge of insufficient sensitivity of
cfDNA in early-stage tumor detection and integrating it with deep
multi-omics analysis (such as cfDNA methylation, fragmentation
characteristics and proteomics). The main tasks of this stage
include: First, developing ultra-high-sensitivity multi-omics cfDNA
detection technology to capture weak tumor signals at an early
stage; second, establishing a large-scale prospective cohort study
to verify the specificity and sensitivity of cfDNA in early breast
cancer screening in asymptomatic high-risk groups; lastly,
systematically evaluating ethical issues, social acceptance and
economic benefits to ensure the sustainability and fairness of the
screening system. The ultimate goal of achieving this stage of
transformation is for cfDNA to potentially become a routine
component of early breast cancer screening, markedly improve the
early diagnosis rate and thereby reduce breast cancer mortality in
the future.
cfDNA has broad application prospects in the field
of precision diagnosis and treatment of breast cancer; however, it
requires continuous investment and interdisciplinary collaboration
in the future.
The future development trend will involve the
multi-omics integration of cfDNA with other liquid biopsy markers
(such as CTCs, exosomes and circulating RNA) as well as traditional
imaging and pathology data. Combining artificial intelligence and
machine learning algorithms, it is expected to extract further
biological information from massive complex data, establishing more
accurate prediction models and decision support systems. For
example, the application of deep learning to analyze cfDNA
fragmentation patterns and methylation profiles may further improve
the accuracy of early diagnosis and MRD detection.
As cfDNA becomes more extensively applied, the
possible ethical, social and economic impacts need to be addressed.
For example, issues such as over-diagnosis, patient anxiety, data
privacy protection, testing costs and allocation of medical
resources all require further discussion and regulation alongside
technological development. Establishing a sound regulatory
framework and ethical review mechanism to ensure the responsible
application of technology is key to the sustainable development of
cfDNA.
Promoting the global clinical transformation of
cfDNA technology requires strengthening international cooperation
and data sharing. Establishing a unified biobank, shared clinical
data and bioinformatics analysis platform will accelerate the
identification and validation of novel biomarkers, as well as the
development of clinical practice guidelines in the future.
CfDNA technology has exhibited notable potential in
the precise diagnosis and treatment of breast cancer; however, its
clinical translation may not be immediate. By confronting the
multi-dimensional challenges at the biological, technical and
clinical integration levels, and adopting a staged translation
strategy, focusing on the monitoring of advanced breast cancer,
decision support for adjuvant treatment of mid- and early-stage
breast cancer and thus, exploring the precise screening of
high-risk groups, can progressively transition cfDNA from the
laboratory to the clinic. In the future, with the deepening of
multi-omics integration, artificial intelligence assistance and
international cooperation, cfDNA is hypothesized to become a key
tool in breast cancer management. This progress potentially offers
patients more precise and personalized treatment plans and
therefore, improves the survival and quality of life for patients
with breast cancer worldwide.
Not applicable.
Funding: No funding was received.
Not applicable.
LF wrote the original draft under the supervision
of XN. AX performed the literature search, summarized abstracts and
prepared the initial data. YX participated in the analysis and
interpretation of the data, and created the illustrations for the
paper. XN, FY, LY reviewed, edited and discussed the manuscript,
ensuring the final content was accurate. XN reviewed and edited the
manuscript and provided funding. All authors read and approved the
final manuscript. Data authentication is not applicable.
Not applicable.
Not applicable.
The authors declare that they have no competing
interests.
|
1
|
Sung H, Ferlay J, Siegel RL, Laversanne M,
Soerjomataram I, Jemal A and Bray F: Global Cancer Statistics 2020:
GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36
Cancers in 185 Countries. CA Cancer J Clin. 71:209–249.
2021.PubMed/NCBI
|
|
2
|
Bray F, Laversanne M, Sung H, Ferlay J,
Siegel RL, Soerjomataram I and Jemal A: Global cancer statistics
2022: GLOBOCAN estimates of incidence and mortality worldwide for
36 cancers in 185 countries. CA Cancer J Clin. 74:229–263.
2024.PubMed/NCBI
|
|
3
|
Huang M, Haiderali A, Fox GE, Frederickson
A, Cortes J, Fasching PA and O'Shaughnessy J: Economic and
humanistic burden of triple-negative breast cancer: A systematic
literature review. Pharmacoeconomics. 40:519–558. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Iragorri N, de Oliveira C, Fitzgerald N
and Essue B: The out-of-pocket cost burden of cancer care-a
systematic literature review. Curr Oncol. 28:1216–1248. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Vale GC, Carvalho GAO, Hoogenkamp M,
Exterkate R, Crielaard W and Ten Cate J: Effect of high-fluoride
dentifrice on root dentine and bacterial composition in a
multispecies biofilm model. Biofouling. 38:348–354. 2022.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Burnside ES, Warren LM, Myles J, Wilkinson
LS, Wallis MG, Patel M, Smith RA, Young KC, Massat NJ and Duffy SW:
Quantitative breast density analysis to predict interval and
node-positive cancers in pursuit of improved screening protocols: A
case-control study. Br J Cancer. 125:884–892. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Glechner A, Wagner G, Mitus JW, Teufer B,
Klerings I, Böck N, Grillich L, Berzaczy D, Helbich TH and
Gartlehner G: Mammography in combination with breast
ultrasonography versus mammography for breast cancer screening in
women at average risk. Cochrane Database Syst Rev.
3:CD0096322023.PubMed/NCBI
|
|
8
|
Niraula S, Biswanger N, Hu P, Lambert P
and Decker K: Incidence, characteristics, and outcomes of interval
breast cancers compared with screening-detected breast cancers.
JAMA Netw Open. 3:e20181792020. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Pallua JD, Brunner A, Zelger B, Schirmer M
and Haybaeck J: The future of pathology is digital. Pathol Res
Pract. 216:1530402020. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Russano M, Napolitano A, Ribelli G,
Iuliani M, Simonetti S, Citarella F, Pantano F, Dell'Aquila E,
Anesi C, Silvestris N, et al: Liquid biopsy and tumor heterogeneity
in metastatic solid tumors: The potentiality of blood samples. J
Exp Clin Cancer Res. 39:952020. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Luo X, Xie S, Hong F, Li X, Wei Y, Zhou Y,
Su W, Yang Y, Tang L, Dao F, et al: From multi-omics to deep
learning: Advances in cfDNA-based liquid biopsy for multi-cancer
screening. Biomark Res. 14:32025. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Su C, Wu J, Jiang L, Lv T, Liu W, Zhang J,
Zhang Y, Peng X and Tan J: Application of cfDNA methylation in
cancer prognostic assessment: Progress and challenges (Review). Int
J Oncol. 67:982025. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Mandel P and Metais P: Nuclear acids in
human blood plasma. C R Seances Soc Biol Fil. 142:241–243. 1948.(In
French). PubMed/NCBI
|
|
14
|
Laurent D, Semple F, Starkey Lewis PJ,
Rose E, Black HA, Coe J, Forbes SJ, Arends MJ, Dear JW and Aitman
TJ: Absolute measurement of the tissue origins of cell-free DNA in
the healthy state and following paracetamol overdose. BMC Med
Genomics. 13:602020. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Stroun M, Lyautey J, Lederrey C, Mulcahy
HE and Anker P: Alu repeat sequences are present in increased
proportions compared to a unique gene in plasma/serum DNA: Evidence
for a preferential release from viable cells? Ann N Y Acad Sci.
945:258–264. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Underhill HR, Kitzman JO, Hellwig S,
Welker NC, Daza R, Baker DN, Gligorich KM, Rostomily RC, Bronner MP
and Shendure J: Fragment length of circulating tumor DNA. PLoS
Genet. 12:e10061622016. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Sender R, Noor E, Milo R and Dor Y: What
fraction of cellular DNA turnover becomes cfDNA? Elife.
12:RP893212024. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Bronkhorst AJ, Ungerer V and Holdenrieder
S: The emerging role of cell-free DNA as a molecular marker for
cancer management. Biomol Detect Quantif. 17:1000872019. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Heitzer E, Auinger L and Speicher MR:
Cell-free DNA and apoptosis: How dead cells inform about the
living. Trends Mol Med. 26:519–528. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Udomruk S, Orrapin S, Pruksakorn D and
Chaiyawat P: Size distribution of cell-free DNA in oncology. Crit
Rev Oncol Hematol. 166:1034552021. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Noë M, Mathios D, Annapragada AV, Koul S,
Foda ZH, Medina JE, Cristiano S, Cherry C, Bruhm DC, Niknafs N, et
al: DNA methylation and gene expression as determinants of
genome-wide cell-free DNA fragmentation. Nat Commun. 15:66902024.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Ju J, Zhao X, An Y, Yang M, Zhang Z, Liu
X, Hu D, Wang W, Pan Y, Xia Z, et al: Cell-free DNA end
characteristics enable accurate and sensitive cancer diagnosis.
Cell Rep Methods. 4:1008772024. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Tutanov O, Shefer A, Tsentalovich Y and
Tamkovich S: Comparative analysis of molecular functions and
biological role of proteins from cell-free DNA-protein complexes
circulating in plasma of healthy females and breast cancer
patients. Int J Mol Sci. 24:72792023. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Zhu D, Wang H, Wu W, Geng S, Zhong G, Li
Y, Guo H, Long G, Ren Q, Luan Y, et al: Circulating cell-free DNA
fragmentation is a stepwise and conserved process linked to
apoptosis. BMC Biol. 21:2532023. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Cheng J and Burwinkel B: Cell-free DNA in
breast cancer: A long way to go. Breast Cancer Res Treat.
170:201–202. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Cristiano S, Leal A, Phallen J, Fiksel J,
Adleff V, Bruhm DC, Jensen SØ, Medina JE, Hruban C, White JR, et
al: Genome-wide cell-free DNA fragmentation in patients with
cancer. Nature. 570:385–389. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Husain H, Nykin D, Bui N, Quan D, Gomez G,
Woodward B, Venkatapathy S, Duttagupta R, Fung E, Lippman SM and
Kurzrock R: Cell-free DNA from ascites and pleural effusions:
Molecular insights into genomic aberrations and disease biology.
Mol Cancer Ther. 16:948–955. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Fairchild L, Whalen J, D'Aco K, Wu J,
Gustafson CB, Solovieff N, Su F, Leary RJ, Campbell CD and Balbin
OA: Clonal hematopoiesis detection in patients with cancer using
cell-free DNA sequencing. Sci Transl Med. 15:eabm87292023.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Charoensappakit A, Sae-Khow K, Rattanaliam
P, Vutthikraivit N, Pecheenbuvan M, Udomkarnjananun S and
Leelahavanichkul A: Cell-free DNA as diagnostic and prognostic
biomarkers for adult sepsis: A systematic review and meta-analysis.
Sci Rep. 13:196242023. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Cucchiari D, Cuadrado-Payan E,
Gonzalez-Roca E, Revuelta I, Argudo M, Ramirez-Bajo MJ,
Ventura-Aguiar P, Rovira J, Bañon-Maneus E, Montagud-Marrahi E, et
al: Early kinetics of donor-derived cell-free DNA after
transplantation predicts renal graft recovery and long-term
function. Nephrol Dial Transplant. 39:114–121. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Walczak K, Grzybowska-Adamowicz J, Stawski
R, Brzezińska O, Zmysłowska A and Nowak D: Response of circulating
free cellular DNA to repeated exercise in men with type 1 diabetes
mellitus. J Clin Med. 13:58592024. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Han DSC and Lo YMD: The nexus of cfDNA and
nuclease biology. Trends Genet. 37:758–770. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Nicolini A, Ferrari P and Duffy MJ:
Prognostic and predictive biomarkers in breast cancer: Past,
present and future. Semin Cancer Biol. 52:56–73. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Tan WY, Nagabhyrava S, Ang-Olson O, Das P,
Ladel L, Sailo B, He L, Sharma A and Ahuja N: Translation of
epigenetics in cell-free DNA liquid biopsy technology and precision
oncology. Curr Issues Mol Biol. 46:6533–6565. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Driussi A, Lamaze FC, Kordahi M, Armero
VS, Gaudreault N, Orain M, Enlow W, Abbosh C, Hodgson D, Dasgupta
A, et al: Clinicopathological predictors of the presence of blood
circulating tumor DNA in early-stage non-small cell lung cancers.
Mod Pathol. 38:1007442025. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Pu X, Li Z, Wang X and Jiang H: Ascites
and serial plasma circulating tumor DNA for predicting the
effectiveness of hyperthermic intraperitoneal chemotherapy in
patients with peritoneal carcinomatosis. Front Oncol.
12:7914182022. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Li J, Jiang W, Wei J, Zhang J, Cai L, Luo
M, Wang Z, Sun W, Wang S, Wang C, et al: Patient specific
circulating tumor DNA fingerprints to monitor treatment response
across multiple tumors. J Transl Med. 18:2932020. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Blalock ZN, Wu GWY, Lindqvist D, Trumpff
C, Flory JD, Lin J, Reus VI, Rampersaud R, Hammamieh R, Gautam A,
et al: Circulating cell-free mitochondrial DNA levels and
glucocorticoid sensitivity in a cohort of male veterans with and
without combat-related PTSD. Transl Psychiatry. 14:222024.
View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Panagopoulou M, Karaglani M, Tzitzikou K,
Kessari N, Arvanitidis K, Amarantidis K, Drosos GI, Gerou S,
Papanas N, Papazoglou D, et al: Mitochondrial fraction of
circulating cell-free DNA as an indicator of human pathology. Int J
Mol Sci. 25:41992024. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Szilágyi M, Pös O, Márton É, Buglyó G,
Soltész B, Keserű J, Penyige A, Szemes T and Nagy B: Circulating
cell-free nucleic acids: Main characteristics and clinical
application. Int J Mol Sci. 21:68272020. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Trumpff C, Michelson J, Lagranha CJ,
Taleon V, Karan KR, Sturm G, Lindqvist D, Fernström J, Moser D,
Kaufman BA and Picard M: Stress and circulating cell-free
mitochondrial DNA: A systematic review of human studies,
physiological considerations, and technical recommendations.
Mitochondrion. 59:225–245. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Trumpff C, Shire D, Lee S, Stanko K, Park
A, Wilson A, Scudder MR, Gianaros PJ, Kaufman BA, Picard M and
Marsland AL: Effects of acute psychological stress on blood
cell-free mitochondrial DNA (cf-mtDNA): A crossover experimental
study. Psychoneuroendocrinology. 182:1076442025. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Limberg AS, Berg F, Köper E, Lindgraf C,
Gevers C, Kumsta R, Hummel EM and Moser DA: Cell-free DNA release
following psychosocial and physical stress in women and men. Transl
Psychiatry. 15:262025. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Liu Y, Zhou K, Guo S, Wang Y, Ji X, Yuan
Q, Su L, Guo X, Gu X and Xing J: NGS-based accurate and efficient
detection of circulating cell-free mitochondrial DNA in cancer
patients. Mol Ther Nucleic Acids. 23:657–666. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Fazzini F, Lamina C, Raftopoulou A, Koller
A, Fuchsberger C, Pattaro C, Del Greco FM, Döttelmayer P, Fendt L,
Fritz J, et al: Association of mitochondrial DNA copy number with
metabolic syndrome and type 2 diabetes in 14 176 individuals. J
Intern Med. 290:190–202. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Chiu RWK and Lo YM: Cell-free fetal DNA
coming in all sizes and shapes. Prenat Diagn. 41:1193–1201. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Sin STK, Ji L, Deng J, Jiang P, Cheng SH,
Heung MMS, Lau CSL, Leung TY, Chan KC, Chiu RWK and Lo YMD:
Characteristics of fetal extrachromosomal circular DNA in maternal
plasma: Methylation status and clearance. Clin Chem. 67:788–796.
2021. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Tan Y, Zhang L, Bai P, Li Z, Zhang R, Yang
F, Wang L and Liang W: Detection of cell-free fetal DNA in maternal
plasma using two types of compound markers. Electrophoresis.
42:1158–1167. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Esfandiari N, Arani AK, Jamshidi A, Miri O
and Schwaminger SP: Cell-free DNA in precision medicine: Overcoming
biological barriers through integrated nanoparticle platforms for
simultaneous diagnosis and therapy. J Nanobiotechnology. Feb
27–2026.(Epub ahead of print). View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Wever BMM and Steenbergen RDM: Unlocking
the potential of tumor-derived DNA in urine for cancer detection:
Methodological challenges and opportunities. Mol Oncol.
19:1918–1934. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Serrano A, Weber TS, Berthelet J, Ftouni
S, El-Saafin F, Lee S, Lim E, Charafe-Jauffret E, Ginestier C,
Williams D, et al: Genetic barcoding uncovers the clonal makeup of
solid and liquid biopsies and their ability to capture
intra-tumoral heterogeneity. Mol Syst Biol. Feb 11–2026.(Epub ahead
of print). View Article : Google Scholar
|
|
52
|
An Y, Zhao X, Zhang Z, Xia Z, Yang M, Ma
L, Zhao Y, Xu G, Du S, Wu X, et al: DNA methylation analysis
explores the molecular basis of plasma cell-free DNA fragmentation.
Nat Commun. 14:2872023. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Pessei V, Macagno M, Mariella E, Congiusta
N, Battaglieri V, Battuello P, Viviani M, Gionfriddo G, Lamba S,
Lorenzato A, et al: DNA demethylation triggers cell free DNA
release in colorectal cancer cells. Genome Med. 16:1182024.
View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Manoochehri M, Borhani N, Gerhäuser C,
Assenov Y, Schönung M, Hielscher T, Christensen BC, Lee MK, Gröne
HJ, Lipka DB, et al: DNA methylation biomarkers for noninvasive
detection of triple-negative breast cancer using liquid biopsy. Int
J Cancer. 152:1025–1035. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Panagopoulou M, Karaglani M,
Balgkouranidou I, Biziota E, Koukaki T, Karamitrousis E, Nena E,
Tsamardinos I, Kolios G, Lianidou E, et al: Circulating cell-free
DNA in breast cancer: Size profiling, levels, and methylation
patterns lead to prognostic and predictive classifiers. Oncogene.
38:3387–3401. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Wang D, O'Rourke D, Sanchez-Garcia JF, Cai
T, Scheuenpflug J and Feng Z: Development of a liquid biopsy based
purely quantitative digital droplet PCR assay for detection of MLH1
promoter methylation in colorectal cancer patients. BMC Cancer.
21:7972021. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Kim SC, Kim DW, Cho EJ, Lee JY, Kim J,
Kwon C, Kim-Ha J, Hong SK, Choi Y, Yi NJ, et al: A circulating
cell-free DNA methylation signature for the detection of
hepatocellular carcinoma. Mol Cancer. 22:1642023. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Heeke S, Gay CM, Estecio MR, Tran H,
Morris BB, Zhang B, Tang X, Raso MG, Rocha P, Lai S, et al: Tumor-
and circulating-free DNA methylation identifies clinically relevant
small cell lung cancer subtypes. Cancer Cell. 42:225–237.e5. 2024.
View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Weigelt B, Comino-Méndez I, de Bruijn I,
Tian L, Meisel JL, García-Murillas I, Fribbens C, Cutts R,
Martelotto LG, Ng CKY, et al: Diverse BRCA1 and BRCA2 reversion
mutations in circulating cell-free DNA of therapy-resistant breast
or ovarian cancer. Clin Cancer Res. 23:6708–6720. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Skoulidis F, Araujo HA, Do MT, Qian Y, Sun
X, Cobo AG, Le JT, Montesion M, Palmer R, Jahchan N, et al: CTLA4
blockade abrogates KEAP1/STK11-related resistance to PD-(L)1
inhibitors. Nature. 635:462–471. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Darrigues L, Pierga JY, Bernard-Tessier A,
Bièche I, Silveira AB, Michel M, Loirat D, Cottu P, Cabel L, Dubot
C, et al: Circulating tumor DNA as a dynamic biomarker of response
to palbociclib and fulvestrant in metastatic breast cancer
patients. Breast Cancer Res. 23:312021. View Article : Google Scholar : PubMed/NCBI
|
|
62
|
Li Z, Li Y, Han D, Wang X, Li C, Chen T,
Li W, Liang Y, Luo D, Chen B, et al: circRNA-SFMBT2 orchestrates
ERα activation to drive tamoxifen resistance in breast cancer
cells. Cell Death Dis. 14:4822023. View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Guo R and Zhang R: Dual effects of circRNA
in thyroid and breast cancer. Clin Transl Oncol. 25:3321–3331.
2023. View Article : Google Scholar : PubMed/NCBI
|
|
64
|
Werner B, Yuwono N, Duggan J, Liu D, David
C, Srirangan S, Provan P; INOVATe Investigators, ; DeFazio A, Arora
V, et al: Cell-free DNA is abundant in ascites and represents a
liquid biopsy of ovarian cancer. Gynecol Oncol. 162:720–727. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
65
|
Lee JW, Park YS, Choi JY, Chang WJ, Lee S,
Sung JS, Kim B, Lee SB, Lee SY, Choi J and Kim YH: Genetic
characteristics associated with drug resistance in lung cancer and
colorectal cancer using whole exome sequencing of cell-free DNA.
Front Oncol. 12:8435612022. View Article : Google Scholar : PubMed/NCBI
|
|
66
|
Iams WT, Mackay M, Ben-Shachar R, Drews J,
Manghnani K, Hockenberry AJ, Cristofanilli M, Nimeiri H, Guinney J
and Benson AB III: Concurrent tissue and circulating tumor DNA
molecular profiling to detect guideline-based targeted mutations in
a multicancer cohort. JAMA Netw Open. 7:e23517002024. View Article : Google Scholar : PubMed/NCBI
|
|
67
|
Razavi P, Chang MT, Xu G, Bandlamudi C,
Ross DS, Vasan N, Cai Y, Bielski CM, Donoghue MTS, Jonsson P, et
al: The genomic landscape of endocrine-resistant advanced breast
cancers. Cancer Cell. 34:427–438.e6. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
68
|
Serrano C, Bauer S, Gómez-Peregrina D,
Kang YK, Jones RL, Rutkowski P, Mir O, Heinrich MC, Tap WD,
Newberry K, et al: Circulating tumor DNA analysis of the phase III
VOYAGER trial: KIT mutational landscape and outcomes in patients
with advanced gastrointestinal stromal tumor treated with
avapritinib or regorafenib. Ann Oncol. 34:615–625. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
69
|
Stanley KE, Jatsenko T, Tuveri S,
Sudhakaran D, Lannoo L, Van Calsteren K, de Borre M, Van Parijs I,
Van Coillie L, Van Den Bogaert K, et al: Cell type signatures in
cell-free DNA fragmentation profiles reveal disease biology. Nat
Commun. 15:22202024. View Article : Google Scholar : PubMed/NCBI
|
|
70
|
Yang L, An M, Song H, Zhang X, Wang M,
Yang L, Wang X, Yang H, Hong X and Song Z: Multidimensional
cell-free DNA fragmentomics enables early detection of breast
cancer. Breast Cancer Res. 28:62025. View Article : Google Scholar : PubMed/NCBI
|
|
71
|
Cheng S, Luo Y, Dong X, Liu M, Wu Z, Xu L,
Yin H, Li X, Shi S, Zhai H, et al: Advanced ensemble staking model
employing cfDNA fragmentation for early detection of esophageal and
gastric cancer. Cancer Lett. 631:2179452025. View Article : Google Scholar : PubMed/NCBI
|
|
72
|
Ganesamoorthy D, Robertson AJ, Chen W,
Hall MB, Cao MD, Ferguson K, Lakhani SR, Nones K, Simpson PT and
Coin L: Whole genome deep sequencing analysis of cell-free DNA in
samples with low tumour content. BMC Cancer. 22:852022. View Article : Google Scholar : PubMed/NCBI
|
|
73
|
Rickles-Young M, Tinoco G, Tsuji J,
Pollock S, Haynam M, Lefebvre H, Glover K, Owen DH, Collier KA, Ha
G, et al: Assay validation of cell-free DNA shallow whole-genome
sequencing to determine tumor fraction in advanced cancers. J Mol
Diagn. 26:413–422. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
74
|
Tie J, Wang Y, Cohen J, Li L, Hong W,
Christie M, Wong HL, Kosmider S, Wong R, Thomson B, et al:
Circulating tumor DNA dynamics and recurrence risk in patients
undergoing curative intent resection of colorectal cancer liver
metastases: A prospective cohort study. PLoS Med. 18:e10036202021.
View Article : Google Scholar : PubMed/NCBI
|
|
75
|
Liu D, Yehia L, Dhawan A, Ni Y and Eng C:
Cell-free DNA fragmentomics and second malignant neoplasm risk in
patients with PTEN hamartoma tumor syndrome. Cell Rep Med.
5:1013842024. View Article : Google Scholar : PubMed/NCBI
|
|
76
|
Migliaccio I, Romagnoli D, Galardi F, De
Luca F, Biagioni C, Curigliano G, Criscitiello C, Minisini AM,
Moretti E, Risi E, et al: Mutational analysis of circulating tumor
DNA in patients with estrogen receptor-positive/human epidermal
growth factor receptor 2-negative advanced breast cancer receiving
palbociclib: Results from the trend trial. JCO Precis Oncol.
8:e23002852024. View Article : Google Scholar : PubMed/NCBI
|
|
77
|
Liu MC, MacKay M, Kase M, Piwowarczyk A,
Lo C, Schaeffer J, Finkle JD, Mason CE, Beaubier N, Blackwell KL
and Park BH: Longitudinal shifts of solid tumor and liquid biopsy
sequencing concordance in metastatic breast cancer. JCO Precis
Oncol. 6:e21003212022. View Article : Google Scholar : PubMed/NCBI
|
|
78
|
Hopkins MK, Koelper N, Bender W, Durnwald
C, Sammel M and Dugoff L: Association between cell-free DNA fetal
fraction and gestational diabetes. Prenat Diagn. 40:724–727. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
79
|
Moeller CM, Oren D, Fernandez Valledor A,
Rubinstein G, Lotan D, Mehlman Y, Slomovich S, Rahman S, Lee C,
Baranowska J, et al: Clinical utility of donor-derived cell-free
DNA in heart transplant recipients with multi-organ transplants.
Clin Transplant. 38:e154792024. View Article : Google Scholar : PubMed/NCBI
|
|
80
|
Baumann AK, Beck J, Kirchner T, Hartleben
B, Schütz E, Oellerich M, Wedemeyer H, Jaeckel E and Taubert R:
Elevated fractional donor-derived cell-free DNA during subclinical
graft injury after liver transplantation. Liver Transpl.
28:1911–1919. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
81
|
Oellerich M, Sherwood K, Keown P, Schütz
E, Beck J, Stegbauer J, Rump LC and Walson PD: Liquid biopsies:
Donor-derived cell-free DNA for the detection of kidney allograft
injury. Nat Rev Nephrol. 17:591–603. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
82
|
O'Reilly D, O'Grady A, Hayes C, Piggott L,
Keane R, Fitzpatrick OM, Conroy M, Aretz NK, Synnott D, Dillon R,
et al: Clinical benefit and cost of plasma-first next-generation
sequencing in patients with newly diagnosed advanced non-small cell
lung cancer in Ireland: The PLAN study. Eur J Cancer.
225:1155822025. View Article : Google Scholar : PubMed/NCBI
|
|
83
|
Corpuz JCG: Circulating-free DNA and
myocardial infarction: From diagnostic promise to clinical
translation. Int J Cardiol Cardiovasc Risk Prev.
27:2005172025.PubMed/NCBI
|
|
84
|
Loft M, To YH, Gibbs P and Tie J: Clinical
application of circulating tumour DNA in colorectal cancer. Lancet
Gastroenterol Hepatol. 8:837–852. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
85
|
Yi H, Youk J, Lim Y, Roh H, Kyung D, Kim
H, Bang D, Keam B, Kim T, Kim M, et al: Analytical and clinical
validation of a highly sensitive NGS-based ctDNA assay with
real-world concordance in non-small cell lung cancer. Cancer Res
Treat. 56:765–773. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
86
|
Dannebaum R, Mikhaylichenko O, Siegel D,
Li C, Hall E, Margeridon S, Herrera M, Loomis K, Riel T, Ramesh M,
et al: Digital PCR assay utilizing in-droplet methylation-sensitive
digestion for estimation of fetal cfDNA from plasma. Prenat Diagn.
45:500–509. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
87
|
Valpione S and Campana L: Detection of
circulating tumor DNA (ctDNA) by digital droplet polymerase chain
reaction (dd-PCR) in liquid biopsies. Methods Enzymol. 629:1–15.
2019. View Article : Google Scholar : PubMed/NCBI
|
|
88
|
Lee H, Park C, Na W, Park KH and Shin S:
Precision cell-free DNA extraction for liquid biopsy by integrated
microfluidics. NPJ Precis Oncol. 4:32020. View Article : Google Scholar : PubMed/NCBI
|
|
89
|
Ye W, Tang X, Liu C, Wen C, Li W and Lyu
J: Accurate quantitation of circulating cell-free mitochondrial DNA
in plasma by droplet digital PCR. Anal Bioanal Chem. 409:2727–2735.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
90
|
Ware SA, Desai N, Lopez M, Leach D, Zhang
Y, Giordano L, Nouraie M, Picard M and Kaufman BA: An automated,
high-throughput methodology optimized for quantitative cell-free
mitochondrial and nuclear DNA isolation from plasma. J Biol Chem.
295:15677–15691. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
91
|
Henriksen TV, Drue SO, Frydendahl A,
Demuth C, Rasmussen MH, Reinert T, Pedersen JS and Andersen CL:
Error characterization and statistical modeling improves
circulating tumor DNA detection by droplet digital PCR. Clin Chem.
68:657–667. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
92
|
Palacín-Aliana I, García-Romero N,
Carrión-Navarro J, Puig-Serra P, Torres-Ruiz R, Rodríguez-Perales
S, Viñal D, González-Rumayor V and Ayuso-Sacido Á: ddPCR overcomes
the CRISPR-Cas13a-based technique for the detection of the BRAF
p.V600E mutation in liquid biopsies. Int J Mol Sci. 25:109022024.
View Article : Google Scholar : PubMed/NCBI
|
|
93
|
Postel M, Roosen A, Laurent-Puig P, Taly V
and Wang-Renault SF: Droplet-based digital PCR and next generation
sequencing for monitoring circulating tumor DNA: A cancer
diagnostic perspective. Expert Rev Mol Diagn. 18:7–17. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
94
|
Seidel MG, Kashofer K, Moser T, Thueringer
A, Liegl-Atzwanger B, Leithner A, Szkandera J, Benesch M,
El-Heliebi A and Heitzer E: Clinical implementation of plasma
cell-free circulating tumor DNA quantification by digital droplet
PCR for the monitoring of Ewing sarcoma in children and
adolescents. Front Pediatr. 10:9264052022. View Article : Google Scholar : PubMed/NCBI
|
|
95
|
Lee JS, Kim M, Seong MW, Kim HS, Lee YK
and Kang HJ: Plasma vs serum in circulating tumor DNA measurement:
Characterization by DNA fragment sizing and digital droplet
polymerase chain reaction. Clin Chem Lab Med. 58:527–532. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
96
|
Geng Z, Li S, Zhu L, Cheng Z, Jin M, Liu
B, Guo Y and Liu P: ‘Sample-to-answer’ detection of rare ctDNA
mutation from 2 ml plasma with a fully integrated DNA extraction
and digital droplet PCR microdevice for liquid biopsy. Anal Chem.
92:7240–7248. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
97
|
Kresse SH, Thorkildsen EM, Brandt-Winge S,
Pharo H, Vedeld HM and Lind GE: Comparison of enzymatic and
bisulfite conversion of circulating cell-free tumor DNA for DNA
methylation analyses. Clin Epigenetics. 17:932025. View Article : Google Scholar : PubMed/NCBI
|
|
98
|
Addamo-De Nard B, Geissmann M, Akhoundova
D, Pistoni C, Brezina T, Zoche M, Weber A, Hussung S and Fritsch R:
A novel KRAS exon 2 drop-off digital PCR assay for mutation
detection in cell-free DNA of cancer patients. Diagn Pathol.
20:622025. View Article : Google Scholar : PubMed/NCBI
|
|
99
|
Lampignano R, Neumann MHD, Weber S, Kloten
V, Herdean A, Voss T, Groelz D, Babayan A, Tibbesma M,
Schlumpberger M, et al: Multicenter evaluation of circulating
cell-free DNA extraction and downstream analyses for the
development of standardized (Pre)analytical work flows. Clin Chem.
66:149–160. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
100
|
Ehman M, Punian J, Weymann D and Regier
DA: Next-generation sequencing in oncology: Challenges in economic
evaluations. Expert Rev Pharmacoecon Outcomes Res. 24:1115–1132.
2024. View Article : Google Scholar : PubMed/NCBI
|
|
101
|
Hartwich TMP, Choi S, Hwang A, Bellone S,
Palmieri L, Seo H, Kim T, Hong J, Krakstad C, Trovik J, et al:
Integrated mutational landscape analysis of endometrial stromal
sarcoma. Proc Natl Acad Sci USA. 123:e25311051232026. View Article : Google Scholar : PubMed/NCBI
|
|
102
|
Johnson A, Shen Y, Zheng X, Su F, Zeng J,
Uzunparmak B, Breuer EK, Holla V, Kahle M, Kim SH, et al: The
actionable transcriptome: A framework for incorporating RNA
sequencing into precision oncology. Nat Rev Clin Oncol. 23:213–229.
2026. View Article : Google Scholar : PubMed/NCBI
|
|
103
|
Ritter M, Paradiso V, Widmer P, Garofoli
A, Quagliata L, Eppenberger-Castori S, Soysal SD, Muenst S, Ng CKY,
Piscuoglio S, et al: Identification of somatic mutations in
thirty-year-old serum cell-free DNA from patients with breast
cancer: A feasibility study. Clin Breast Cancer. 20:413–421.e1.
2020. View Article : Google Scholar : PubMed/NCBI
|
|
104
|
Jiang X, Li H, Liu J, Sun H, Zhang L, Li
W, Yao J and Cheng Y: Feasibility analysis of cell-free DNA derived
from plasma of lung cancer patients for next-generation sequencing.
Biopreserv Biobank. 18:117–121. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
105
|
Szadkowska P, Roura AJ, Wojtas B, Wojnicki
K, Licholai S, Waller T, Gubala T, Zukowski K, Karpeta M, Wilkus K,
et al: Improvements in quality control and library preparation for
targeted sequencing allowed detection of potentially pathogenic
alterations in circulating cell-free DNA derived from plasma of
brain tumor patients. Cancers (Basel). 14:39022022. View Article : Google Scholar : PubMed/NCBI
|
|
106
|
Troll CJ, Kapp J, Rao V, Harkins KM, Cole
C, Naughton C, Morgan JM, Shapiro B and Green RE: A ligation-based
single-stranded library preparation method to analyze cell-free DNA
and synthetic oligos. BMC Genomics. 20:10232019. View Article : Google Scholar : PubMed/NCBI
|
|
107
|
Page K, Guttery DS, Fernandez-Garcia D,
Hills A, Hastings RK, Luo J, Goddard K, Shahin V, Woodley-Barker L,
Rosales BM, et al: Next generation sequencing of circulating
cell-free DNA for evaluating mutations and gene amplification in
metastatic breast cancer. Clin Chem. 63:532–541. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
108
|
Jongbloed EM, Jansen MPHM, de Weerd V,
Helmijr JA, Beaufort CM, Reinders MJT, van Marion R, van IJcken
WFJ, Sonke GS, Konings IR, et al: Machine learning-based somatic
variant calling in cell-free DNA of metastatic breast cancer
patients using large NGS panels. Sci Rep. 13:104242023. View Article : Google Scholar : PubMed/NCBI
|
|
109
|
Jeon S, Lee H, Bae K, Yoon KA, Lee ES and
Cho Y: Efficient capture and isolation of tumor-related circulating
cell-free DNA from cancer patients using electroactive conducting
polymer nanowire platforms. Theranostics. 6:828–836. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
110
|
Jia Y, Li Y, Bai X, Liu L, Shan Y, Wang F,
Yu Z and Zheng C: Raman spectroscopy and exosome-based machine
learning predicts the efficacy of neoadjuvant therapy for
HER2-positive breast cancer. Anal Chem. 97:1374–1385. 2025.
View Article : Google Scholar : PubMed/NCBI
|
|
111
|
Wu Y, Tian X, Ma J, Lin Y, Ye J, Wang Y,
Lu J and Yin W: Label-free discrimination analysis of breast cancer
tumor and adjacent tissues of patients after neoadjuvant treatment
using Raman spectroscopy: A diagnostic study. Int J Surg.
111:1788–1800. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
112
|
Papadakis VM, Cheimonidi C, Panagopoulou
M, Karaglani M, Apalaki P, Katsara K, Kenanakis G, Theodosiou T,
Constantinidis TC, Stratigi K and Chatzaki E: Label-free human
disease characterization through circulating cell-free DNA analysis
using raman spectroscopy. Int J Mol Sci. 24:123842023. View Article : Google Scholar : PubMed/NCBI
|
|
113
|
Masunaga N, Kagara N, Motooka D, Nakamura
S, Miyake T, Tanei T, Naoi Y, Shimoda M, Shimazu K, Kim SJ and
Noguchi S: Highly sensitive detection of ESR1 mutations in
cell-free DNA from patients with metastatic breast cancer using
molecular barcode sequencing. Breast Cancer Res Treat. 167:49–58.
2018. View Article : Google Scholar : PubMed/NCBI
|
|
114
|
Venetis K, Pepe F, Pescia C, Cursano G,
Criscitiello C, Frascarelli C, Mane E, Russo G, Salimbeni BT,
Troncone G, et al: ESR1 mutations in HR+/HER2-metastatic breast
cancer: Enhancing the accuracy of ctDNA testing. Cancer Treat Rev.
121:1026422023. View Article : Google Scholar : PubMed/NCBI
|
|
115
|
El-Fatah RMA, Badawy HK, Pasha HF, Mesbah
NM, Abo-Elmatty DM and Abdel-Hamed AR: Plasma cell-free DNA
biomarkers as novel diagnostic and prognostic tools in breast
cancer. Cancer Genet. 302–303. 1–7. 2026.PubMed/NCBI
|
|
116
|
Buckley DN, Kalfa AJ, Gooden G, Lewinger
JP, Pacheco M, Gayton J, Spicer D, Carpten J, Stewart D, Lenz HJ,
et al: MammaTrace-a cell-free DNA methylation plasma only assay for
minimal residual disease detection in breast cancer patients.
medRxiv [Preprint]. 2025.05.20.25328012. 2025.
|
|
117
|
Jindal A, Nijhawan S, Vijay U, Kaur A and
Dana R: Diagnostic accuracy of cfDNA levels in gallbladder cancer:
A study using qPCR & threshold evaluation. Indian J Med Res.
161:143–151. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
118
|
Lin AY, Isaeva OI, Deger T, Geurts VCM,
Vessies DCL, Voorwerk L, Slagter M, Moore KS, van der Leest P,
Martens JWM, et al: CfDNA-based copy-number dynamics during
anti-PD1 treatment in metastatic triple negative breast cancer.
Cell Rep Med. 6:1025122025. View Article : Google Scholar : PubMed/NCBI
|
|
119
|
Bharde A, Nadagouda S, Dongare M,
Hariramani K, Basavalingegowda M, Haldar S, D'Souza A, Jadhav B,
Prajapati S, Jadhav V, et al: ctDNA-based liquid biopsy reveals
wider mutational profile with therapy resistance and metastasis
susceptibility signatures in early-stage breast cancer patients. J
Liq Biopsy. 7:1002842024. View Article : Google Scholar : PubMed/NCBI
|
|
120
|
Bronte G, Ravaioli S and Bravaccini S:
What to look for in cell-free DNA from breast cancer patients.
Breast Cancer Res Treat. 170:199–200. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
121
|
Mohammad S, Ullah I, Ali A, Jan Z, Aleem
B, Khan M and Naseem W: Role of circulating tumor DNA and cell-free
DNA biomarkers in diagnosis and prognosis of oral cancer-a
systematic review. BMC Oral Health. 25:5222025. View Article : Google Scholar : PubMed/NCBI
|
|
122
|
Alemany-Cosme E, Cruz-Chamorro D, Hervas
D, Garcia D, Martinez-Tomas R, Briones-Gomez A, Galbis-Caravajal J,
Calvo A, Juan O, Lahoz A, et al: Genome-wide methylation analysis
of circulating cell-free DNA identifies an episignature in lung
cancer: validation of EMP2 as a potential biomarker. Clin
Epigenetics. 18:82025. View Article : Google Scholar : PubMed/NCBI
|
|
123
|
Bredno J, Lipson J, Venn O, Aravanis AM
and Jamshidi A: Clinical correlates of circulating cell-free DNA
tumor fraction. PLoS One. 16:e02564362021. View Article : Google Scholar : PubMed/NCBI
|
|
124
|
Alborelli I, Generali D, Jermann P,
Cappelletti MR, Ferrero G, Scaggiante B, Bortul M, Zanconati F,
Nicolet S, Haegele J, et al: Cell-free DNA analysis in healthy
individuals by next-generation sequencing: A proof of concept and
technical validation study. Cell Death Dis. 10:5342019. View Article : Google Scholar : PubMed/NCBI
|
|
125
|
Khurram I, Khan MU, Ibrahim S, Saleem A,
Khan Z, Mubeen M, Khawar A and Ali Q: Efficacy of cell-free DNA as
a diagnostic biomarker in breast cancer patients. Sci Rep.
13:153472023. View Article : Google Scholar : PubMed/NCBI
|
|
126
|
Xu Z, Sandler DP and Taylor JA: Blood DNA
methylation and breast cancer: A prospective case-cohort analysis
in the sister study. J Natl Cancer Inst. 112:87–94. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
127
|
Wang J, Niu Y, Yang M, Shu L, Wang H, Wu
X, He Y, Chen P, Zhong G, Tang Z, et al: Altered cfDNA
fragmentation profile in hypomethylated regions as diagnostic
markers in breast cancer. Epigenetics Chromatin. 16:332023.
View Article : Google Scholar : PubMed/NCBI
|
|
128
|
Liu J, Zhao H, Huang Y, Xu S, Zhou Y,
Zhang W, Li J, Ming Y, Wang X, Zhao S, et al: Genome-wide cell-free
DNA methylation analyses improve accuracy of non-invasive
diagnostic imaging for early-stage breast cancer. Mol Cancer.
20:362021. View Article : Google Scholar : PubMed/NCBI
|
|
129
|
Hasenleithner SO and Speicher MR: A
clinician's handbook for using ctDNA throughout the patient
journey. Mol Cancer. 21:812022. View Article : Google Scholar : PubMed/NCBI
|
|
130
|
Han BW, Cai GX, Liu Q, Yang X, Guo ZW,
Huang LM, Li K, Ouyang GJ, Yang XX, Ye GL and Wu YS: Noninvasive
discrimination of benign and malignant breast lesions using
genome-wide nucleosome profiles of plasma cell-free DNA. Clin Chim
Acta. 520:95–100. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
131
|
Rao S, Han AL, Zukowski A, Kopin E,
Sartorius CA, Kabos P and Ramachandran S: Transcription
factor-nucleosome dynamics from plasma cfDNA identifies ER-driven
states in breast cancer. Sci Adv. 8:eabm43582022. View Article : Google Scholar : PubMed/NCBI
|
|
132
|
Lee KH, Shin TJ, Kim WH and Cho JY:
Methylation of LINE-1 in cell-free DNA serves as a liquid biopsy
biomarker for human breast cancers and dog mammary tumors. Sci Rep.
9:1752019. View Article : Google Scholar : PubMed/NCBI
|
|
133
|
Arko-Boham B, Aryee NA, Blay RM, Owusu
EDA, Tagoe EA, Doris Shackie ES, Debrah AB and Adu-Aryee NA:
Circulating cell-free DNA integrity as a diagnostic and prognostic
marker for breast and prostate cancers. Cancer Genet. 235–236.
65–71. 2019.PubMed/NCBI
|
|
134
|
Abd El Hafeez HA, Abd El Rahman MZ, Kamel
TM, Rezk KM, Mohamed FM and Abdel-Hameed ZA: The role of
circulating cell-free DNA and its integrity as a biomarker for
diagnosis of breast cancer using ALU (247/115) bP sequences. Egypt
J Immunol. 30:44–55. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
135
|
Bortul M, Giudici F, Tierno D, Generali D,
Scomersi S, Grassi G, Bottin C, Cappelletti MR, Zanconati F and
Scaggiante B: A case-control study by ddPCR of ALU 260/111 and
LINE-1 266/97 copy number ratio in circulating cell-free DNA in
plasma revealed LINE-1 266/97 as a potential biomarker for early
breast cancer detection. Int J Mol Sci. 24:85202023. View Article : Google Scholar : PubMed/NCBI
|
|
136
|
Xiao D, Zhang M, Qu Y and Su X: Functions
of methyltransferase-like 3 in breast cancer: Pathogenesis, drug
resistance, and therapeutic target. Breast Cancer Res. 26:1102024.
View Article : Google Scholar : PubMed/NCBI
|
|
137
|
Gaughan NA, Rademacher N, Rogers C, Purdy
A, Mattingly JD, Patten CR, Cobb AN, Kong AL and Cortina CS:
Current insights into breast and ovarian cancer risk: A
contemporary review. Minerva Surg. 80:334–353. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
138
|
Balasundaram A, Kumar SU, D TK, Anil Dedge
A, R G, K SS, R S and C GPD: The targeted next-generation sequence
revealed SMAD4, AKT1, and TP53 mutations from circulating cell-free
DNA of breast cancer and its effect on protein structure-A
computational approach. J Biomol Struct Dyn. 41:15584–15597. 2023.
View Article : Google Scholar : PubMed/NCBI
|
|
139
|
Raei M, Heydari K, Tabarestani M, Razavi
A, Mirshafiei F, Esmaeily F, Taheri M, Hoseini A, Nazari H,
Shamshirian D and Alizadeh-Navaei R: Diagnostic accuracy of ESR1
mutation detection by cell-free DNA in breast cancer: A systematic
review and meta-analysis of diagnostic test accuracy. BMC Cancer.
24:9082024. View Article : Google Scholar : PubMed/NCBI
|
|
140
|
Scandino R, Nardone A, Casiraghi N,
Galardi F, Genovese M, Romagnoli D, Paoli M, Biagioni C, Tonina A,
Migliaccio I, et al: Enabling sensitive and precise detection of
ctDNA through somatic copy number aberrations in breast cancer. NPJ
Breast Cancer. 11:252025. View Article : Google Scholar : PubMed/NCBI
|
|
141
|
Decker B, Allen J, Luccarini C, Pooley KA,
Shah M, Bolla MK, Wang Q, Ahmed S, Baynes C, Conroy DM, et al:
Rare, protein-truncating variants in ATM, CHEK2 and PALB2, but not
XRCC2, are associated with increased breast cancer risks. J Med
Genet. 54:732–741. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
142
|
Li D, Li P, Wu J, Yi J, Dou Y, Guo X, Yin
Y, Wang D, Ma C and Qiu L: Methylation of NBPF1 as a novel marker
for the detection of plasma cell-free DNA of breast cancer
patients. Clin Chim Acta. 484:81–86. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
143
|
Shirkavand A, Boroujeni ZN and Aleyasin
SA: Examination of methylation changes of VIM, CXCR4, DOK7, and
SPDEF genes in peripheral blood DNA in breast cancer patients.
Indian J Cancer. 55:366–371. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
144
|
Douvdevani A, Bernstein-Molho R, Asraf K,
Doolman R, Laitman Y and Friedman E: Circulating cell-free DNA
(cfDNA) levels in BRCA1 and BRCA2 mutation carriers: A preliminary
study. Cancer Biomark. 28:269–273. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
145
|
Murillo Carrasco A, Acosta O, Ponce J,
Cotrina J, Aguilar A, Araujo J, Rebaza P, Pinto JA, Fujita R and
Buleje J: PUM1 and RNase P genes as potential cell-free DNA markers
in breast cancer. J Clin Lab Anal. 35:e237202021. View Article : Google Scholar : PubMed/NCBI
|
|
146
|
Wang SC, Liao LM, Ansar M, Lin SY, Hsu WW,
Su CM, Chung YM, Liu CC, Hung CS and Lin RK: Automatic detection of
the circulating cell-free methylated DNA pattern of GCM2, ITPRIPL1
and CCDC181 for detection of early breast cancer and surgical
treatment response. Cancers (Basel). 13:13752021. View Article : Google Scholar : PubMed/NCBI
|
|
147
|
Winter JM, Sheehan-Hennessy L, Yao B,
Pedersen SK, Wassie MM, Eaton M, Chong M, Young GP and Symonds EL:
Detection of hypermethylated BCAT1 and IKZF1 DNA in blood and
tissues of colorectal, breast and prostate cancer patients. Cancer
Biomark. 34:493–503. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
148
|
Danos P, Giannoni-Luza S, Murillo Carrasco
AG, Acosta O, Guevara-Fujita ML, Cotrina Concha JM, Guerra Miller
H, Pinto Oblitas J, Aguilar Cartagena A, Araujo JM, et al: Promoter
hypermethylation of RARB and GSTP1 genes in plasma cell-free DNA as
breast cancer biomarkers in Peruvian women. Mol Genet Genomic Med.
11:e22602023. View Article : Google Scholar : PubMed/NCBI
|
|
149
|
Grisolia P, Tufano R, Iannarone C, De
Falco A, Carlino F, Graziano C, Addeo R, Scrima M, Caraglia F,
Ceccarelli A, et al: Differential methylation of circulating free
DNA assessed through cfMeDiP as a new tool for breast cancer
diagnosis and detection of BRCA1/2 mutation. J Transl Med.
22:9382024. View Article : Google Scholar : PubMed/NCBI
|
|
150
|
Saura C, Ortiz C, Matito J, Arenas EJ,
Suñol A, Martín Á, Córdoba O, Martínez-Sabadell A, García-Ruiz I,
Miranda I, et al: Early-stage breast cancer detection in breast
milk. Cancer Discov. 13:2180–2191. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
151
|
Cunningham N and Turner NC: Liquid
biopsies in breast milk for the early detection of breast cancer.
Cancer Discov. 13:2125–2127. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
152
|
Singareeka Raghavendra A, Damodaran S,
Barcenas CH, Fuqua SA, Layman RM and Tripathy D: Personalizing
therapies over the course of hormone
receptor-positive/HER2-negative metastatic breast cancer. CA Cancer
J Clin. 76:e700552026.PubMed/NCBI
|
|
153
|
Buonaiuto R, Botticelli A, Criscitiello C,
Dieci MV, Fusco N, Gerratana L, Lambertini M, Malapelle U, Malorni
L and De Angelis C: Key decision factors in second-line therapy:
Expert insights on HR+/HER2-metastatic breast cancer post-CDK4/6
inhibitor progression. Cancer Treat Rev. 138:1029722025. View Article : Google Scholar : PubMed/NCBI
|
|
154
|
Lüönd F, Tiede S and Christofori G: Breast
cancer as an example of tumour heterogeneity and tumour cell
plasticity during malignant progression. Br J Cancer. 125:164–175.
2021. View Article : Google Scholar : PubMed/NCBI
|
|
155
|
Chin YM, Shibayama T, Chan HT, Otaki M,
Hara F, Kobayashi T, Kobayashi K, Hosonaga M, Fukada I, Inagaki L,
et al: Serial circulating tumor DNA monitoring of CDK4/6 inhibitors
response in metastatic breast cancer. Cancer Sci. 113:1808–1820.
2022. View Article : Google Scholar : PubMed/NCBI
|
|
156
|
Crucitta S, Ruglioni M, Lorenzini G,
Bargagna I, Luculli GI, Albanese I, Bilancio D, Patanè F, Fontana
A, Danesi R and Del Re M: CDK4/6 inhibitors overcome endocrine ESR1
mutation-related resistance in metastatic breast cancer patients.
Cancers (Basel). 15:13062023. View Article : Google Scholar : PubMed/NCBI
|
|
157
|
van den Ende NS, Nguyen AH, Jager A, Kok
M, Debets R and van Deurzen CHM: Triple-negative breast cancer and
predictive markers of response to neoadjuvant chemotherapy: A
systematic review. Int J Mol Sci. 24:29692023. View Article : Google Scholar : PubMed/NCBI
|
|
158
|
Jiang Y, Zeng J, Liu J, Deng W, Kahlert
UD, Tang R, Xu M, Shi W and Wang Q: Comparing efficacy of
neoadjuvant therapy of triple-negative breast cancer: A Bayesian
network meta-regression analysis. Medicine (Baltimore).
105:e469622026. View Article : Google Scholar : PubMed/NCBI
|
|
159
|
Gupta S, Nair N, Hawaldar RW, Joshi S,
Gulia S, Shet T, Sarin R, Parmar V, Desai S, Ghosh J, et al:
Addition of carboplatin to sequential taxane-anthracycline
neoadjuvant chemotherapy in triple-negative breast cancer: A phase
III randomized controlled trial. J Clin Oncol. 44:9–19. 2026.
View Article : Google Scholar : PubMed/NCBI
|
|
160
|
Cirmena G, Ferrando L, Ravera F, Garuti A,
Dameri M, Gallo M, Barbero V, Ferrando F, Del Mastro L, Garlaschi
A, et al: Plasma cell-free DNA integrity assessed by automated
electrophoresis predicts the achievement of pathologic complete
response to neoadjuvant chemotherapy in patients with breast
cancer. JCO Precis Oncol. 6:e21001982022. View Article : Google Scholar : PubMed/NCBI
|
|
161
|
Chen JH, Addanki S, Roy D, Bassett R,
Kalashnikova E, Spickard E, Kuerer HM, Meas S, Sarli VN, Korkut A,
et al: Monitoring response to neoadjuvant chemotherapy in triple
negative breast cancer using circulating tumor DNA. BMC Cancer.
24:10162024. View Article : Google Scholar : PubMed/NCBI
|
|
162
|
Parsons HA, Blewett T, Chu X, Sridhar S,
Santos K, Xiong K, Abramson VG, Patel A, Cheng J, Brufsky A, et al:
Circulating tumor DNA association with residual cancer burden after
neoadjuvant chemotherapy in triple-negative breast cancer in TBCRC
030. Ann Oncol. 34:899–906. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
163
|
Cailleux F, Agostinetto E, Lambertini M,
Rothé F, Wu HT, Balcioglu M, Kalashnikova E, Vincent D, Viglietti
G, Gombos A, et al: Circulating tumor DNA after neoadjuvant
chemotherapy in breast cancer is associated with disease relapse.
JCO Precis Oncol. 6:e22001482022. View Article : Google Scholar : PubMed/NCBI
|
|
164
|
Ortolan E, Appierto V, Silvestri M, Miceli
R, Veneroni S, Folli S, Pruneri G, Vingiani A, Belfiore A,
Cappelletti V, et al: Blood-based genomics of triple-negative
breast cancer progression in patients treated with neoadjuvant
chemotherapy. ESMO Open. 6:1000862021. View Article : Google Scholar : PubMed/NCBI
|
|
165
|
Coombes RC, Page K, Salari R, Hastings RK,
Armstrong A, Ahmed S, Ali S, Cleator S, Kenny L, Stebbing J, et al:
Personalized detection of circulating tumor DNA antedates breast
cancer metastatic recurrence. Clin Cancer Res. 25:4255–4263. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
166
|
Cullinane C, Fleming C, O'Leary DP, Hassan
F, Kelly L, O'Sullivan MJ, Corrigan MA and Redmond HP: Association
of circulating tumor DNA with disease-free survival in breast
cancer: A systematic review and meta-analysis. JAMA Netw Open.
3:e20269212020. View Article : Google Scholar : PubMed/NCBI
|
|
167
|
Garcia-Murillas I, Chopra N, Comino-Méndez
I, Beaney M, Tovey H, Cutts RJ, Swift C, Kriplani D, Afentakis M,
Hrebien S, et al: Assessment of molecular relapse detection in
early-stage breast cancer. JAMA Oncol. 5:1473–1478. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
168
|
Magbanua MJM, Swigart LB, Wu HT, Hirst GL,
Yau C, Wolf DM, Tin A, Salari R, Shchegrova S, Pawar H, et al:
Circulating tumor DNA in neoadjuvant-treated breast cancer reflects
response and survival. Ann Oncol. 32:229–239. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
169
|
Fitzpatrick A, Iravani M, Mills A, Childs
L, Alaguthurai T, Clifford A, Garcia-Murillas I, Van Laere S, Dirix
L, Harries M, et al: Assessing CSF ctDNA to improve diagnostic
accuracy and therapeutic monitoring in breast cancer leptomeningeal
metastasis. Clin Cancer Res. 28:1180–1191. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
170
|
Davis AA, Jacob S, Gerratana L, Shah AN,
Wehbe F, Katam N, Zhang Q, Flaum L, Siziopikou KP, Platanias LC, et
al: Landscape of circulating tumour DNA in metastatic breast
cancer. EBioMedicine. 58:1029142020. View Article : Google Scholar : PubMed/NCBI
|
|
171
|
Kawabata K, Nishikubo H, Kanei S, Aoyama
R, Tsukada Y, Sano T, Imanishi D, Sakuma T, Maruo K, Yamamoto Y, et
al: Significance of multi-cancer genome profiling testing for
breast cancer: A retrospective analysis of 3326 cases from Japan's
national database. Genes (Basel). 15:7922024. View Article : Google Scholar : PubMed/NCBI
|
|
172
|
Takeshita T, Yamamoto Y, Yamamoto-Ibusuki
M, Tomiguchi M, Sueta A, Murakami K and Iwase H: Clinical
significance of plasma cell-free DNA mutations in PIK3CA, AKT1, and
ESR1 gene according to treatment lines in ER-positive breast
cancer. Mol Cancer. 17:672018. View Article : Google Scholar : PubMed/NCBI
|
|
173
|
Kodahl AR, Ehmsen S, Pallisgaard N,
Jylling AMB, Jensen JD, Laenkholm AV, Knoop AS and Ditzel HJ:
Correlation between circulating cell-free PIK3CA tumor DNA levels
and treatment response in patients with PIK3CA-mutated metastatic
breast cancer. Mol Oncol. 12:925–935. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
174
|
Winn JS, Hasse Z, Slifker M, Pei J,
Arisi-Fernandez SM, Talarchek JN, Obeid E, Baldwin DA, Gong Y, Ross
E, et al: Genetic variants detected using cell-free DNA from blood
and tumor samples in patients with inflammatory breast cancer. Int
J Mol Sci. 21:12902020. View Article : Google Scholar : PubMed/NCBI
|
|
175
|
Zuo Z, Tang J, Cai X, Ke F and Shi Z:
Probing of breast cancer using a combination of plasma and urinary
circulating cell-free DNA. Biosci Rep. 40:BSR201943062020.
View Article : Google Scholar : PubMed/NCBI
|
|
176
|
Qui S, Takeshita T, Sueta A, Tomiguchi M,
Goto-Yamaguchi L, Hidaka K, Suzu I, Yamamoto Y and Iwase H:
Analysis of plasma HER2 copy number in cell-free DNA of breast
cancer patients: A comparison with HER2 extracellular domain
protein level in serum. Breast Cancer. 28:746–754. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
177
|
Sim SH, Yang HN, Jeon SY, Lee KS and Park
IH: Mutation analysis using cell-free DNA for endocrine therapy in
patients with HR+ metastatic breast cancer. Sci Rep. 11:55662021.
View Article : Google Scholar : PubMed/NCBI
|
|
178
|
Page K, Martinson LJ, Fernandez-Garcia D,
Hills A, Gleason KLT, Gray MC, Rushton AJ, Nteliopoulos G, Hastings
RK, Goddard K, et al: Circulating tumor DNA profiling from breast
cancer screening through to metastatic disease. JCO Precis Oncol.
5:PO.20.00522. 2021.PubMed/NCBI
|
|
179
|
Keup C, Suryaprakash V, Storbeck M,
Hoffmann O, Kimmig R and Kasimir-Bauer S: Longitudinal
multi-parametric liquid biopsy approach identifies unique features
of circulating tumor cell, extracellular vesicle, and cell-free DNA
characterization for disease monitoring in metastatic breast cancer
patients. Cells. 10:2122021. View Article : Google Scholar : PubMed/NCBI
|
|
180
|
Gerratana L, Davis AA, Velimirovic M,
Clifton K, Hensing WL, Shah AN, Dai CS, Reduzzi C, D'Amico P, Wehbe
F, et al: Interplay between ESR1/PIK3CA codon variants, oncogenic
pathway alterations and clinical phenotype in patients with
metastatic breast cancer (MBC): Comprehensive circulating tumor DNA
(ctDNA) analysis. Breast Cancer Res. 25:1122023. View Article : Google Scholar : PubMed/NCBI
|
|
181
|
Martens GA, Demol J, Dedeurwaerdere F,
Breyne J, De Smet K, De Jaeger P and De Smet D: Rational
thresholding of circulating tumor DNA concentration for improved
surveillance of metastatic breast cancer. ESMO Open. 9:1022352024.
View Article : Google Scholar : PubMed/NCBI
|
|
182
|
Hattori M, Serelli-Lee V, Naito Y,
Yamanaka T, Yasojima H, Nakamura R, Fujisawa T, Imai M, Nakamura Y,
Bando H, et al: Genomic landscape of circulating tumor DNA in
patients with hormone receptor-positive/human epidermal growth
factor receptor-2-negative metastatic breast cancer treated with
abemaciclib: Data from the SCRUM-Japan cancer genome screening
project. JCO Precis Oncol. 8:e23006472024. View Article : Google Scholar : PubMed/NCBI
|
|
183
|
Liu ZB, Ezzedine NE, Eterovic AK, Ensor
JE, Huang HJ, Albanell J, Choi DS, Lluch A, Liu Y, Rojo F, et al:
Detection of breast cancer stem cell gene mutations in circulating
free DNA during the evolution of metastases. Breast Cancer Res
Treat. 178:251–261. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
184
|
Silveira C, Sousa AC, Corredeira P,
Martins M, Sousa AR, Da Cruz Paula A, Selenica P, Brown DN,
Golkaram M, Kaplan S, et al: Comprehensive genomic profiling of
cell-free circulating tumor DNA detects response to ribociclib plus
letrozole in a patient with metastatic breast cancer. Biomolecules.
12:18182022. View Article : Google Scholar : PubMed/NCBI
|
|
185
|
Hunter N, Croessmann S, Cravero K, Shinn
D, Hurley PJ and Park BH: Undetectable tumor cell-free DNA in a
patient with metastatic breast cancer with complete response and
long-term remission. J Natl Compr Canc Netw. 18:375–379. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
186
|
Lenaerts L, Che H, Brison N, Neofytou M,
Jatsenko T, Lefrère H, Maggen C, Villela D, Verheecke M, Dehaspe L,
et al: Breast cancer detection and treatment monitoring using a
noninvasive prenatal testing platform: Utility in pregnant and
nonpregnant populations. Clin Chem. 66:1414–1423. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
187
|
Gidwani R, Franks JA, Enogela EM, Caston
NE, Williams CP, Aswani MS, Azuero A and Rocque GB: Survival in the
real world: A national analysis of patients treated for early-stage
breast cancer. JCO Oncol Pract. 18:e235–e249. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
188
|
Nader-Marta G, Monteforte M, Agostinetto
E, Cinquini M, Martins-Branco D, Langouo M, Llombart-Cusac A,
Cortés J, Ignatiadis M, Torri V, et al: Circulating tumor DNA for
predicting recurrence in patients with operable breast cancer: A
systematic review and meta-analysis. ESMO Open. 9:1023902024.
View Article : Google Scholar : PubMed/NCBI
|
|
189
|
Kim MH, Kim GM, Ahn JM, Ryu WJ, Kim SG,
Kim JH, Kim TY, Han HJ, Kim JY, Park HS, et al: Copy number
aberrations in circulating tumor DNA enables prognosis prediction
and molecular characterization of breast cancer. J Natl Cancer
Inst. 115:1036–1049. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
190
|
Zivanovic Bujak A, Weng CF, Silva MJ,
Yeung M, Lo L, Ftouni S, Litchfield C, Ko YA, Kuykhoven K, Van
Geelen C, et al: Circulating tumour DNA in metastatic breast cancer
to guide clinical trial enrolment and precision oncology: A cohort
study. PLoS Med. 17:e10033632020. View Article : Google Scholar : PubMed/NCBI
|
|
191
|
Chi Y, Su M, Zhou D, Zheng F, Zhang B,
Qiang L, Ren G, Song L, Bu B, Fang S, et al: Dynamic analysis of
circulating tumor DNA to predict the prognosis and monitor the
treatment response of patients with metastatic triple-negative
breast cancer: A prospective study. Elife. 12:e901982023.
View Article : Google Scholar : PubMed/NCBI
|
|
192
|
Shaw JA, Page K, Wren E, de Bruin EC,
Kalashnikova E, Hastings R, McEwen R, Zhang E, Wadsley M,
Acheampong E, et al: Serial postoperative circulating tumor DNA
assessment has strong prognostic value during long-term follow-up
in patients with breast cancer. JCO Precis Oncol. 8:e23004562024.
View Article : Google Scholar : PubMed/NCBI
|
|
193
|
La Rocca E, De Santis MC, Silvestri M,
Ortolan E, Valenti M, Folli S, de Braud FG, Bianchi GV, Scaperrotta
GP, Apolone G, et al: Early stage breast cancer follow-up in
real-world clinical practice: The added value of cell free
circulating tumor DNA. J Cancer Res Clin Oncol. 148:1543–1550.
2022. View Article : Google Scholar : PubMed/NCBI
|
|
194
|
Yi Z, Ma F, Rong G, Liu B, Guan Y, Li J,
Sun X, Wang W, Guan X, Mo H, et al: The molecular tumor burden
index as a response evaluation criterion in breast cancer. Signal
Transduct Target Ther. 6:2512021. View Article : Google Scholar : PubMed/NCBI
|
|
195
|
Reichert ZR, Morgan TM, Li G, Castellanos
E, Snow T, Dall'Olio FG, Madison RW, Fine AD, Oxnard GR, Graf RP
and Stover DG: Prognostic value of plasma circulating tumor DNA
fraction across four common cancer types: A real-world outcomes
study. Ann Oncol. 34:111–120. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
196
|
Mo H, Wang X, Ma F, Qian Z, Sun X, Yi Z,
Guan X, Li L, Liu B and Xu B: Genome-wide chromosomal instability
by cell-free DNA sequencing predicts survival in patients with
metastatic breast cancer. Breast. 53:111–118. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
197
|
Zhou H, Wang XJ, Jiang X, Qian Z, Chen T,
Hu Y, Chen ZH, Gao Y, Wang R, Ye WW and Cao WM: Plasma cell-free
DNA chromosomal instability analysis by low-pass whole-genome
sequencing to monitor breast cancer relapse. Breast Cancer Res
Treat. 178:63–73. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
198
|
Kujala J, Hartikainen JM, Tengström M,
Sironen R, Kosma VM and Mannermaa A: High mutation burden of
circulating cell-free DNA in early-stage breast cancer patients is
associated with a poor relapse-free survival. Cancer Med.
9:5922–5931. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
199
|
Stover DG, Parsons HA, Ha G, Freeman SS,
Barry WT, Guo H, Choudhury AD, Gydush G, Reed SC, Rhoades J, et al:
Association of cell-free DNA tumor fraction and somatic copy number
alterations with survival in metastatic triple-negative breast
cancer. J Clin Oncol. 36:543–553. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
200
|
Pairawan S, Hess KR, Janku F, Sanchez NS,
Mills Shaw KR, Eng C, Damodaran S, Javle M, Kaseb AO, Hong DS, et
al: Cell-free circulating tumor DNA variant allele frequency
associates with survival in metastatic cancer. Clin Cancer Res.
26:1924–1931. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
201
|
Magbanua MJM, Ahmed Z, Sayaman RW, Brown
Swigart L, Hirst GL, Yau C, Wolf DM, Li W, Delson AL, Perlmutter J,
et al: Cell-free DNA concentration as a biomarker of response and
recurrence in HER2-negative breast cancer receiving neoadjuvant
chemotherapy. Clin Cancer Res. 30:2444–2451. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
202
|
Rossi G, Mu Z, Rademaker AW, Austin LK,
Strickland KS, Costa RLB, Nagy RJ, Zagonel V, Taxter TJ, Behdad A,
et al: Cell-Free DNA and circulating tumor cells: Comprehensive
liquid biopsy analysis in advanced breast cancer. Clin Cancer Res.
24:560–568. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
203
|
Fernandez-Garcia D, Hills A, Page K,
Hastings RK, Toghill B, Goddard KS, Ion C, Ogle O, Boydell AR,
Gleason K, et al: Plasma cell-free DNA (cfDNA) as a predictive and
prognostic marker in patients with metastatic breast cancer. Breast
Cancer Res. 21:1492019. View Article : Google Scholar : PubMed/NCBI
|
|
204
|
Baek SY, Hong M, Lee S, Joo S, Jang J,
Jung C, Lee H, Yoo TK, Chung IY, Kim HJ, et al: Serial analysis of
ESR1 mutations in cell-free DNA from hormone receptor-positive,
HER2-negative metastatic breast cancer during palliative endocrine
therapy. Front Oncol. 15:17093172026. View Article : Google Scholar : PubMed/NCBI
|
|
205
|
Park J, Chang ES, Kim JY, Chelakkot C,
Sung M, Song JY, Jung K, Lee JH, Choi JY, Kim NY, et al:
c-MET-positive circulating tumor cells and cell-free DNA as
independent prognostic factors in hormone
receptor-positive/HER2-negative metastatic breast cancer. Breast
Cancer Res. 26:132024. View Article : Google Scholar : PubMed/NCBI
|
|
206
|
Kardousha A, Shehada W, Basha A, Nasser S,
Mistiri ME, Hamad A, Al-Bader SB and Elazzazy S: HER2-low
non-metastatic breast cancer in Qatar-a nationwide retrospective
cohort study to evaluate the response to neoadjuvant chemotherapy:
A real-world analysis. Front Oncol. 14:13981002024. View Article : Google Scholar : PubMed/NCBI
|
|
207
|
Ye Z, Wang C, Wan S, Mu Z, Zhang Z,
Abu-Khalaf MM, Fellin FM, Silver DP, Neupane M, Jaslow RJ, et al:
Association of clinical outcomes in metastatic breast cancer
patients with circulating tumour cell and circulating cell-free
DNA. Eur J Cancer. 106:133–143. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
208
|
Radovich M, Jiang G, Hancock BA, Chitambar
C, Nanda R, Falkson C, Lynce FC, Gallagher C, Isaacs C, Blaya M, et
al: Association of circulating tumor DNA and circulating tumor
cells after neoadjuvant chemotherapy with disease recurrence in
patients with triple-negative breast cancer: preplanned secondary
analysis of the BRE12-158 randomized clinical trial. JAMA Oncol.
6:1410–1415. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
209
|
Hu ZY, Xie N, Tian C, Yang X, Liu L, Li J,
Xiao H, Wu H, Lu J, Gao J, et al: Identifying circulating tumor DNA
mutation profiles in metastatic breast cancer patients with
multiline resistance. EBioMedicine. 32:111–118. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
210
|
Colleoni M, Sun Z, Price KN, Karlsson P,
Forbes JF, Thürlimann B, Gianni L, Castiglione M, Gelber RD, Coates
AS and Goldhirsch A: Annual hazard rates of recurrence for breast
cancer during 24 years of follow-up: Results from the international
breast cancer study group trials I to V. J Clin Oncol. 34:927–935.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
211
|
Palesh O, Kamen C, Sharp S, Golden A, Neri
E, Spiegel D and Koopman C: Physical activity and survival in women
with advanced breast cancer. Cancer Nurs. 41:E31–E38. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
212
|
Cervena K, Vodicka P and Vymetalkova V:
Diagnostic and prognostic impact of cell-free DNA in human cancers:
Systematic review. Mutat Res Rev Mutat Res. 781:100–129. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
213
|
Gao L, Zhang L, Liu J, Wang L, Fu Q, Liu
S, Li Y, Qu C, Zhao N, Tan S, et al: Discovery and validation of
cell-free DNA methylation markers for specific diagnosis,
differentiation from benign tumors, and prognosis of breast cancer.
Breast Cancer Res. 27:1082025. View Article : Google Scholar : PubMed/NCBI
|
|
214
|
Goodman CR and Barcenas CH: Liquid biopsy
in advanced and metastatic breast cancer: Translating prognostic
value to clinical implementation. Transl Cancer Res. 14:2178–2182.
2025. View Article : Google Scholar : PubMed/NCBI
|
|
215
|
Chandarlapaty S, Chen D, He W, Sung P,
Samoila A, You D, Bhatt T, Patel P, Voi M, Gnant M, et al:
Prevalence of ESR1 mutations in cell-free DNA and outcomes in
metastatic breast cancer: A secondary analysis of the BOLERO-2
clinical trial. JAMA Oncol. 2:1310–1315. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
216
|
Li X, Chen W, Wang H, Wei Q, Ding X and Li
W: Amplification and the clinical significance of circulating
cell-free DNA of PVT1 in breast cancer. Oncol Rep. 38:465–471.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
217
|
Jacot W, Dalenc F, Lopez-Crapez E,
Chaltiel L, Durigova A, Gros N, Lozano N, Lacaze JL, Pouderoux S,
Gladieff L, et al: PIK3CA mutations early persistence in cell-free
tumor DNA as a negative prognostic factor in metastatic breast
cancer patients treated with hormonal therapy. Breast Cancer Res
Treat. 177:659–667. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
218
|
Keup C, Benyaa K, Hauch S,
Sprenger-Haussels M, Tewes M, Mach P, Bittner AK, Kimmig R, Hahn P
and Kasimir-Bauer S: Targeted deep sequencing revealed variants in
cell-free DNA of hormone receptor-positive metastatic breast cancer
patients. Cell Mol Life Sci. 77:497–509. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
219
|
Muendlein A, Geiger K, Gaenger S, Dechow
T, Nonnenbroich C, Leiherer A, Drexel H, Gaumann A, Jagla W, Winder
T, et al: Significant impact of circulating tumour DNA mutations on
survival in metastatic breast cancer patients. Sci Rep.
11:67612021. View Article : Google Scholar : PubMed/NCBI
|
|
220
|
Liao H, Zhang J, Zheng T, Liu X, Zhong J,
Shao B, Dong X, Wang X, Du P, King BL, et al: Identification of
mutation patterns and circulating tumour DNA-derived prognostic
markers in advanced breast cancer patients. J Transl Med.
20:2112022. View Article : Google Scholar : PubMed/NCBI
|
|
221
|
Zhang L, Sun S, Zhao X, Liu J, Xu Y, Xu L,
Song C, Li N, Yu J, Zhao S, et al: Prognostic value of baseline
genetic features and newly identified TP53 mutations in advanced
breast cancer. Mol Oncol. 16:3689–3702. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
222
|
Tolaney SM, Toi M, Neven P, Sohn J,
Grischke EM, Llombart-Cussac A, Soliman H, Wang H, Wijayawardana S,
Jansen VM, et al: Clinical significance of PIK3CA and ESR1
mutations in circulating tumor DNA: Analysis from the MONARCH 2
study of abemaciclib plus fulvestrant. Clin Cancer Res.
28:1500–1506. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
223
|
Chen TW, Hsiao W, Dai MS, Lin CH, Chang
DY, Chen IC, Wang MY, Chang SH, Huang SM, Cheng AL, et al: Plasma
cell-free tumor DNA, PIK3CA and TP53 mutations predicted inferior
endocrine-based treatment outcome in endocrine receptor-positive
metastatic breast cancer. Breast Cancer Res Treat. 201:377–385.
2023. View Article : Google Scholar : PubMed/NCBI
|
|
224
|
Dickinson K, Sharma A, Agnihotram RV,
Altuntur S, Park M, Meterissian S and Burnier JV: Circulating tumor
DNA and survival in metastatic breast cancer: A Systematic review
and meta-analysis. JAMA Netw Open. 7:e24317222024. View Article : Google Scholar : PubMed/NCBI
|
|
225
|
Takeshita T, Iwamoto T, Niikura N,
Watanabe K, Kikawa Y, Kobayashi K, Iwakuma N, Okamura T, Tada H,
Ozaki S, et al: Identifying prognostic biomarkers for palbociclib
add-on therapy in fulvestrant-resistant breast cancer using
cell-free DNA sequencing. ESMO Open. 9:1023852024. View Article : Google Scholar : PubMed/NCBI
|
|
226
|
Sayed NH, Senousy MA, Shaker OG and Kortam
MA: Serum lncRNA PVT1 and its targets miR-146a and SIRT1 as
biomarkers for the early detection of breast cancer and its
metastasis. Biotechnol Appl Biochem. 72:1644–1657. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
227
|
Engel J, Wieder V, Bauer M, Kaufhold S,
Stückrath K, Wilke J, Hanf V, Uleer C, Lantzsch T, Peschel S, et
al: Prognostic and predictive impact of NOTCH1 in early breast
cancer. Breast Cancer Res Treat. 209:27–38. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
228
|
Kakani P, Dhamdhere SG, Pant D, Joshi R,
Mishra J, Samaiya A and Shukla S: Hypoxia-induced CTCF promotes EMT
in breast cancer. Cell Rep. 43:1143672024. View Article : Google Scholar : PubMed/NCBI
|
|
229
|
Jin X, Zhu L, Cui Z, Tang J, Xie M and Ren
G: Elevated expression of GNAS promotes breast cancer cell
proliferation and migration via the PI3K/AKT/Snail1/E-cadherin
axis. Clin Transl Oncol. 21:1207–1219. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
230
|
Kurtz DM, Scherer F, Jin MC, Soo J, Craig
AFM, Esfahani MS, Chabon JJ, Stehr H, Liu CL, Tibshirani R, et al:
Circulating tumor DNA measurements as early outcome predictors in
diffuse large B-cell lymphoma. J Clin Oncol. 36:2845–2853. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
231
|
Neuberger EWI, Sontag S, Brahmer A,
Philippi KFA, Radsak MP, Wagner W and Simon P: Physical activity
specifically evokes release of cell-free DNA from granulocytes
thereby affecting liquid biopsy. Clin Epigenetics. 14:292022.
View Article : Google Scholar : PubMed/NCBI
|
|
232
|
Sazed SA, Kibria MG and Alam MS: An
optimized real-time qPCR method for the effective detection of
human malaria infections. Diagnostics (Basel). 11:7362021.
View Article : Google Scholar : PubMed/NCBI
|
|
233
|
Abbasi A and Alexandrov LB: Significance
and limitations of the use of next-generation sequencing
technologies for detecting mutational signatures. DNA Repair
(Amst). 107:1032002021. View Article : Google Scholar : PubMed/NCBI
|
|
234
|
Hindson BJ, Ness KD, Masquelier DA,
Belgrader P, Heredia NJ, Makarewicz AJ, Bright IJ, Lucero MY,
Hiddessen AL, Legler TC, et al: High-throughput droplet digital PCR
system for absolute quantitation of DNA copy number. Anal Chem.
83:8604–8610. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
235
|
Huang AC, Postow MA, Orlowski RJ, Mick R,
Bengsch B, Manne S, Xu W, Harmon S, Giles JR, Wenz B, et al: T-cell
invigoration to tumour burden ratio associated with anti-PD-1
response. Nature. 545:60–65. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
236
|
Hanahan D: Hallmarks of cancer: New
dimensions. Cancer Discov. 12:31–46. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
237
|
Nguyen QH, Pervolarakis N, Blake K, Ma D,
Davis RT, James N, Phung AT, Willey E, Kumar R, Jabart E, et al:
Profiling human breast epithelial cells using single cell RNA
sequencing identifies cell diversity. Nat Commun. 9:20282018.
View Article : Google Scholar : PubMed/NCBI
|