Introduction
Sinonasal intestinal-type adenocarcinoma (ITAC) is a
rare category of malignant tumor, with a significantly higher
incidence among individuals exposed to wood and leather dust
(1,2). Histopathologically and
immunophenotypically, ITAC resembles intestinal adenocarcinoma and
shares the same immunophenotype [cytokeratin
(CK)20+/caudal-type homeobox 2
(CDX2)+/villin+] as colorectal cancer
(3). Histologically, ITAC is
classified into the colonic, papillary, solid, mucinous and mixed
subtypes. Well-differentiated papillary intestinal adenocarcinomas
are associated with a more favorable prognosis, whereas solid and
mucinous intestinal adenocarcinomas are associated with a poorer
prognosis (1).
Clinically, ITAC most commonly occurs in the ethmoid
sinus, followed by the nasal cavity and other sinuses (4). Early symptoms of ITAC typically
include nasal congestion, rhinorrhea and localized pain. As the
tumor progresses to an advanced stage, ocular symptoms may
manifest, such as proptosis, diplopia and blindness. In cases where
the tumor invades the cranial cavity, patients may experience
headaches and nausea, and facial ulceration may occur (5).
Surgical resection combined with adjuvant therapy
remains the cornerstone of ITAC management (6,7);
however, the selection of the surgical approach and radiotherapy
regimen varies substantially according to the stage of the tumor,
the anatomical location and the extent of invasion.
With regard to surgical approaches, the endoscopic
endonasal approach (EEA) is preferred for early-stage ITAC due to
its benefits of reduced surgical trauma, fewer complications and a
shorter period of hospitalization (8,9).
Patients with T3-T4 stage tumors may still derive benefits from
EEA, provided there is no extensive invasion of the skull base or
orbit (9). In cases with locally
advanced tumors that invade complex anatomical structures, such as
the skull base, dura mater, orbit and maxillary sinus, a more
intricate EEA, in conjunction with open surgery, is warranted
(10).
Post-operative adjuvant radiotherapy is typically
recommended to complement surgical resection for high-risk cases,
with a standard dose range of 60–70 Gy (6,7).
However, anatomical constraints often necessitate compromises in
the radiotherapy dose. The proximity of the ethmoid sinus/cranial
base area to critical structures, such as the optic nerve and
brainstem, poses challenges in achieving an optimal balance between
delivering an adequate dose to the target area and protecting
organs at risk (OAR) using conventional radiotherapy techniques.
Consequently, developing strategies to ensure sufficient dosing to
the target area, while safeguarding the OAR remains an urgent
issue.
There is currently no standardized systemic
treatment available for an ITAC that cannot be fully resected
through palliative surgery or that recurs or metastasizes
post-surgery. Given the morphological and molecular similarities
between ITAC and colorectal cancer, several studies have suggested
the use of neoadjuvant fluorouracil-based chemotherapy regimens
(4,11). Previous clinical trials have
predominantly concentrated on early-stage nasal sinus
adenocarcinoma, yielding favorable outcomes. Nonetheless, the
median progression-free survival (PFS) time for metastatic ITAC
treated with chemotherapy is only 1.2 months (12). This underscores the urgent need for
more effective therapeutic options for advanced-stage ITAC.
The tumor protein p53 (TP53) gene is the most
frequently mutated in ITAC, with a mutation incidence of 40–50%,
and the status of the p53 protein can serve as a predictor of the
tumor response to chemotherapy (13). Furthermore, KRAS proto-oncogene
GTPase mutations are present in ~43% of patients with ITAC
(14), while nuclear β-catenin
expression is observed in 31–53% of ITAC cases (15). Consequently, targeted therapies
addressing these specific mutations represent a promising avenue
for future research into the treatment of advanced-stage ITAC.
Combined immunotherapy or immunotherapy alone has
demonstrated promising outcomes in the treatment of various types
of head and neck squamous cell carcinomas, as well as colorectal
cancer (16–18). Previous research has identified that
17% of ITACs express programmed death-ligand 1 (PD-L1), suggesting
the presence of an immunosuppressive environment in this malignancy
(19). Although this evidence
suggests that immunotherapy holds therapeutic potential, the
clinical data remain limited to case reports (20,21).
The present study constitutes a preliminary
systematic validation of radiotherapy dose thresholds and describes
a potentially promising approach combining immunotherapy with
chemotherapy, followed by sequential immunotherapy as a
single-agent maintenance treatment. This strategy could provide
preliminary insights for the management of advanced-stage ITAC.
Case report
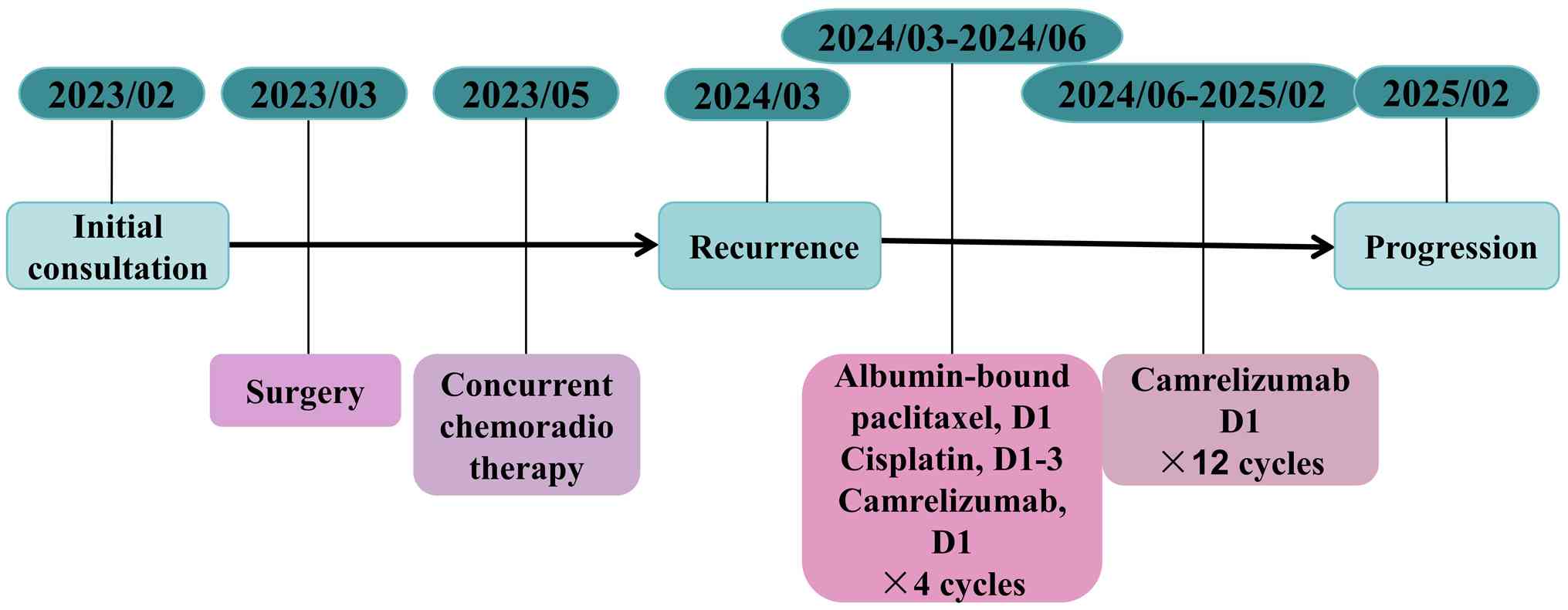
Initial treatment
A 60-year-old man presented to West China Hospital,
Sichuan University (Chengdu, China) in February 2023 with the
primary complaint of progressive loss of vision in the left eye
over the past month. Magnetic resonance imaging (MRI) revealed a
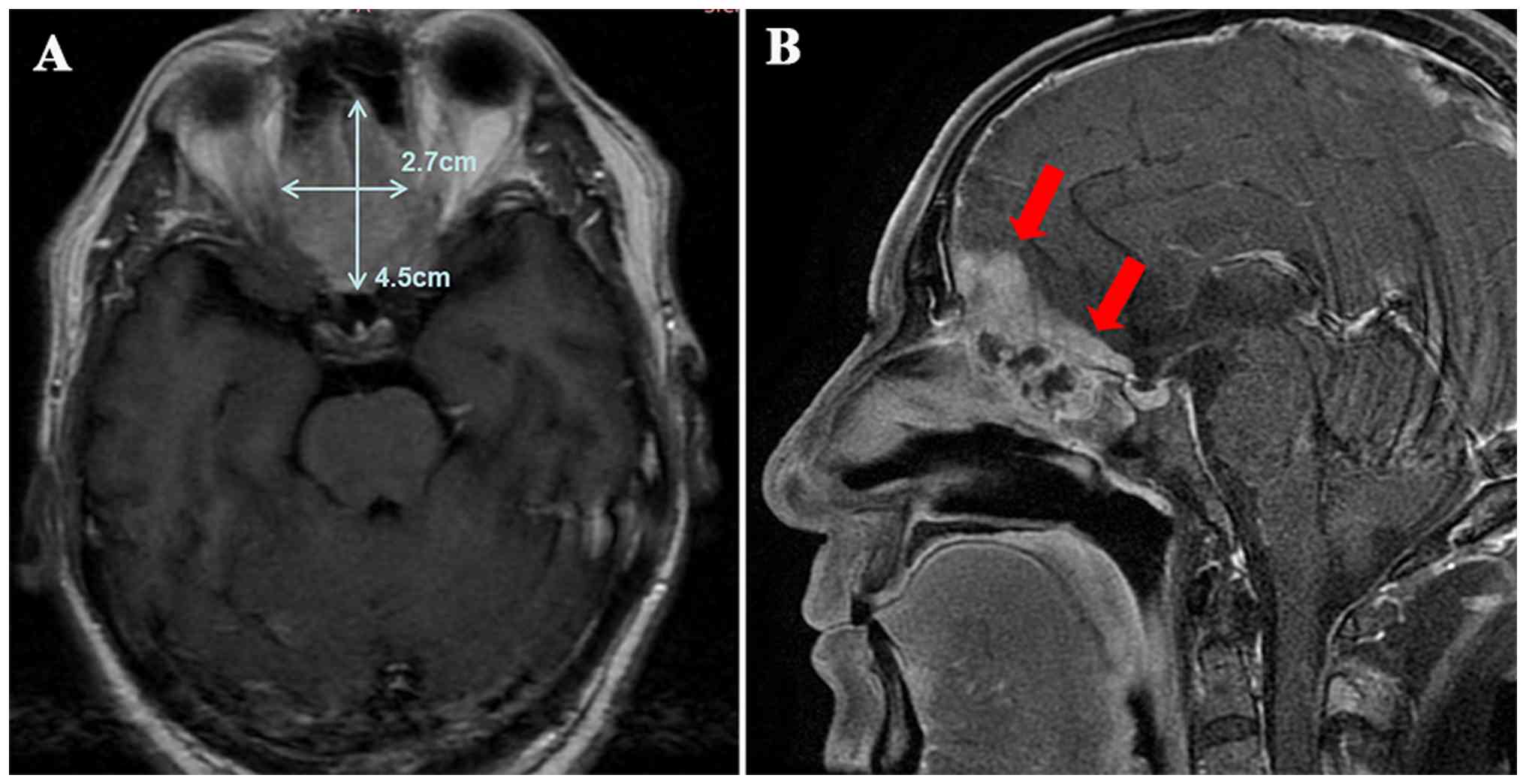
mass measuring 4.5×2.7 cm located in the region of the ethmoid and
sphenoid sinus, with invasion into the intracranial area (Fig. 1). Following a multidisciplinary team
discussion, a decision was made to proceed with surgical
intervention. The approach selected included a combination of
transnasal endoscopy and microscopic craniotomy, accompanied by
anterior skull base resection, encompassing the paranasal sinuses,
as well as the intracranial and orbital regions. A post-operative
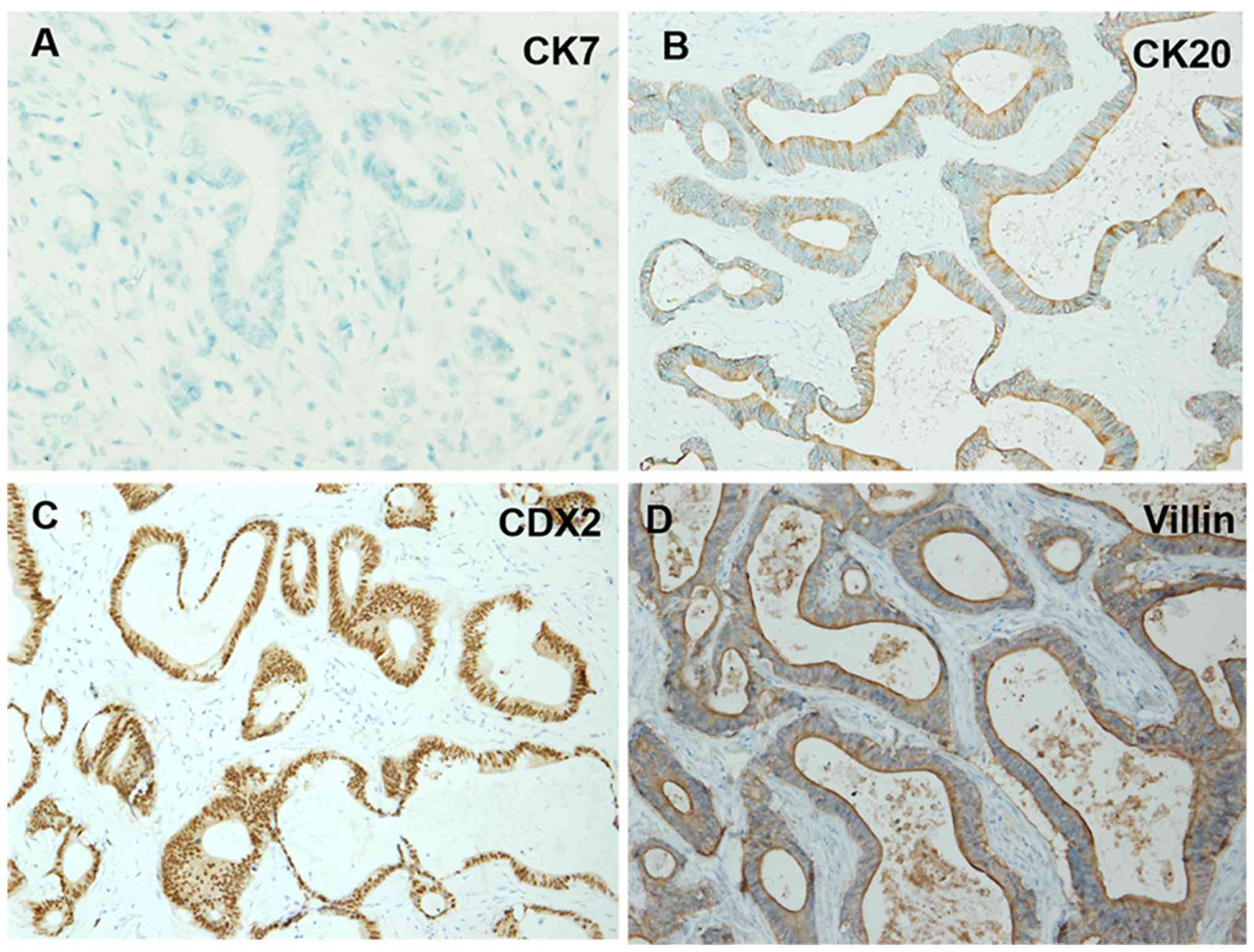
pathological analysis indicated the following immunohistochemical
findings: CK7(−), CK20(+), CDX2 (+) and villin(+) (Fig. 2), thereby confirming a diagnosis of
ITAC. Immunohistochemical staining with CK7, CK20, CDX-2 and villin
antibodies (Beijing Zhongshan Jinqiao Biotechnology Co., Ltd.) was
performed by the Department of Pathology. Due to the positive
surgical margin, adjuvant radiotherapy was administered 2 months
later at the General Hospital of Western Theater Command (Chengdu,
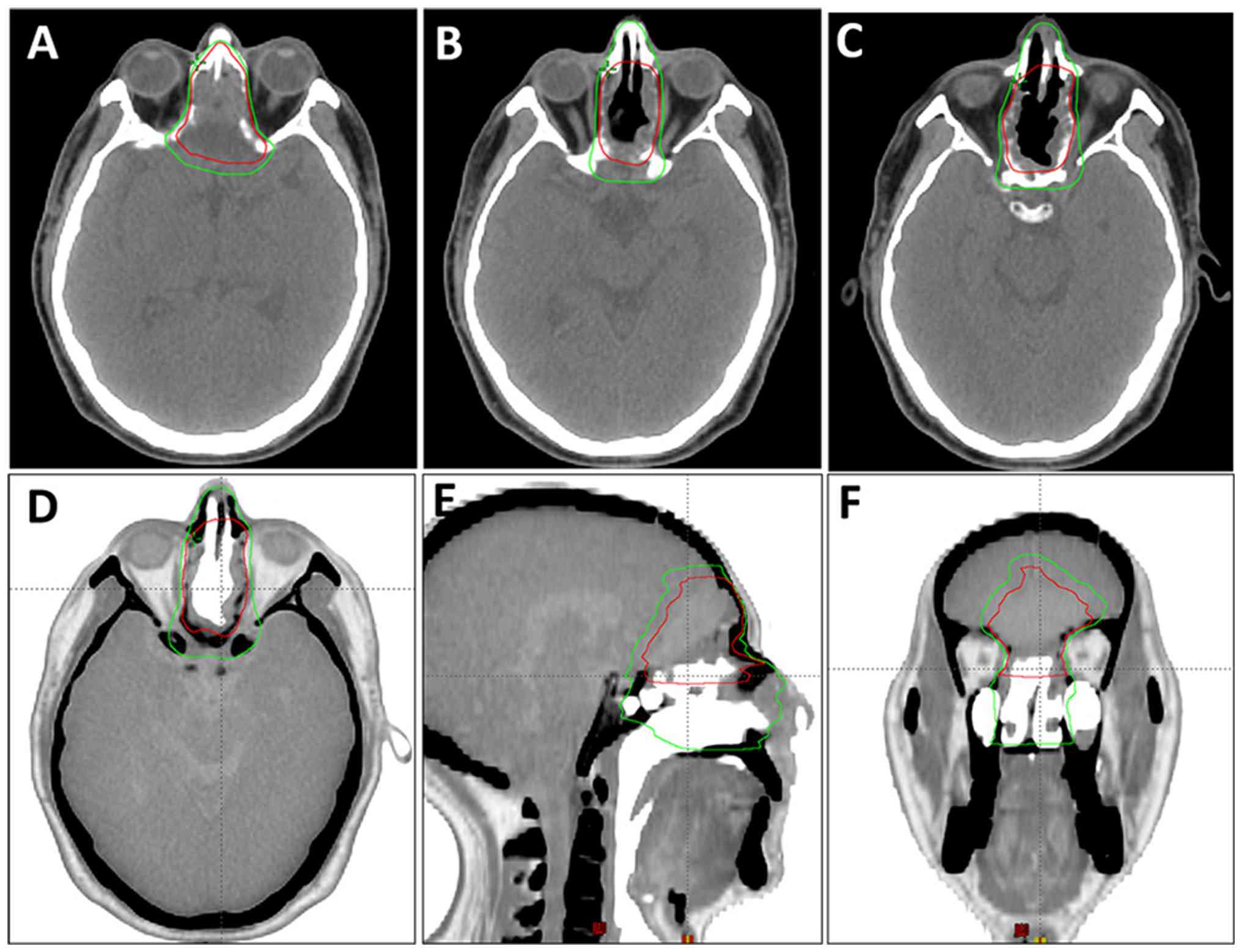
China). The radiotherapy target volume definitions were as follows:
i) Gross tumor volume of the tumor bed (GTV-tb); this encompasses
the surgical tumor bed area and any radiographically involved
margins visible on imaging. ii) Clinical target volume 1/high-risk
clinical volume (CTV-1): This includes the expansion of GTV-tb by
0.5 cm, along with the nasal vestibule, nasal cavity, turbinates
and hard palate. However, when the expansion zone approaches
critical adjacent structures (such as the optic nerves or cavernous
sinus), the margin is reduced to 0.1 cm (Fig. 3). The radiotherapy regimen included
a planned dose of 6,480 cGy in 30 fractions (216 cGy per fraction)
to the planned GTV-tb (PGTV-tb) [equivalent dose in 2-Gy fractions
(EQD2)=65.66 Gy, α/β=10], and 6,000 cGy in 30 fractions (200 cGy
per fraction) to the planning CTV-1 (PTV-1) [EQD2=60.00
Gy, α/β=10]. Due to concerns about potential injury to the optic
nerve, the patient and their family decided to shorten the
radiotherapy course to 25 sessions. The final delivered doses were
~5,400 cGy to the PGTV-tb (estimated EQD2, 54.72 Gy; α/β=10) and
5,000 cGy to the PTV-1. Concurrent chemotherapy was administered
with a regimen of cisplatin at 30 mg on days 1–2 every week for
four cycles (1 week per cycle).
Recurrence
At 12 months post-surgery, the patient experienced
bilateral vision loss, visual distortion, headaches, nausea and
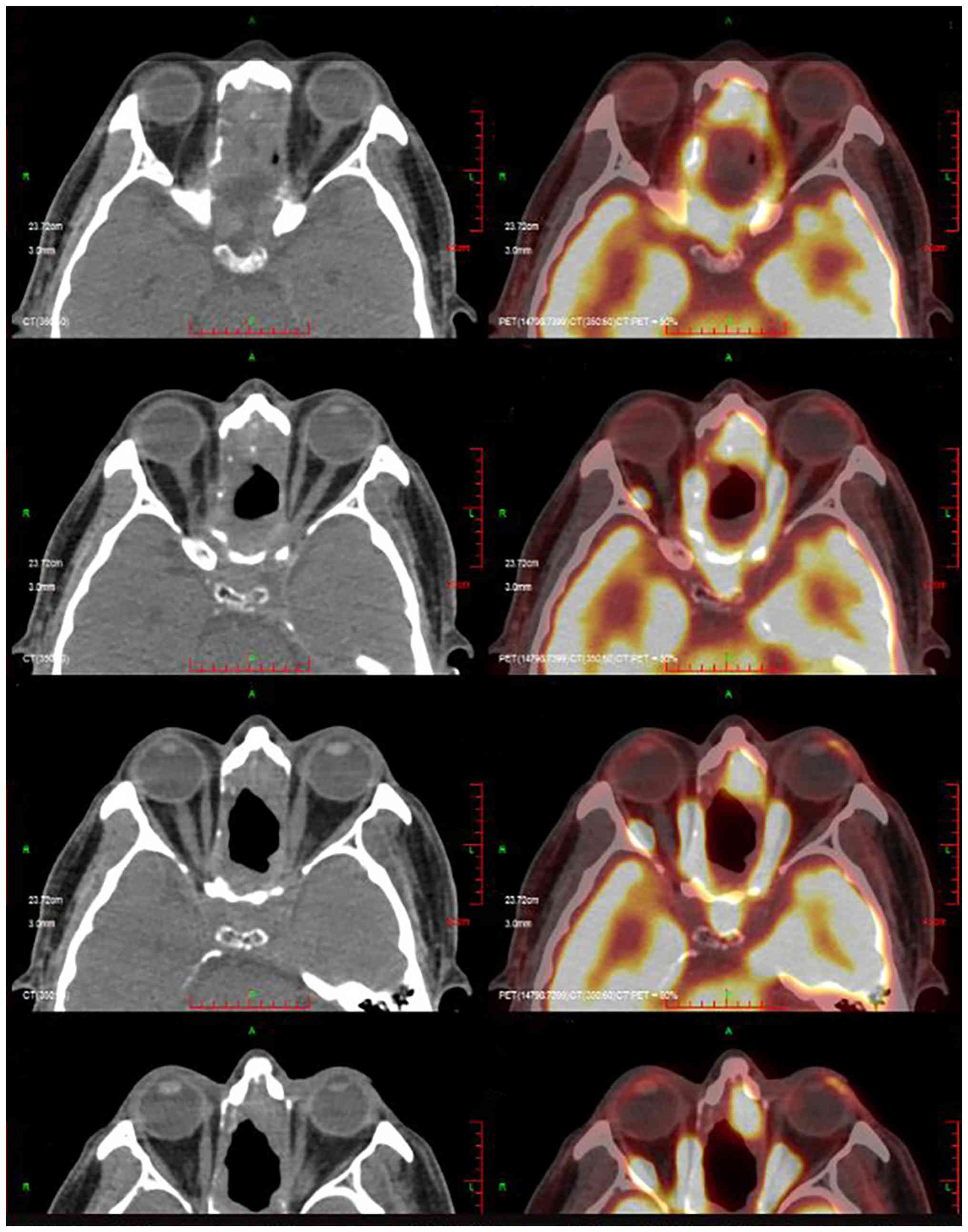
vomiting. A positron emission tomography-computed tomography
(PET-CT) scan indicated tumor recurrence in the ethmoid and
sphenoid sinuses, the anterior portion of the left nasal cavity and
the nasal septum (Fig. 4). The
biopsy results confirmed the presence of ITAC. Immunohistochemistry
revealed PD-L1 expression with a combined positive score (CPS) of 5
and programmed cell death protein 1 (PD-1) positivity at 5%
(22). The diagnosis of tumor
recurrence, including PET-CT imaging and pathological biopsy, was
confirmed at West China Hospital of Sichuan University. Systemic
therapy was initiated with a regimen consisting of albumin-bound
paclitaxel (300 mg on day 1), cisplatin (30 mg on days 1–3) and
camrelizumab (200 mg on day 1) administered intravenously every 3
weeks for four cycles (3 weeks per cycle). Following four cycles of
systemic therapy, grade II myelosuppression occurred (white blood
cell count, 2.49×109/l; normal reference range,
3.5–9.5×109/l. The physical strength of the patient also
markedly declined. The treatment regimen was adjusted to
maintenance therapy with single-agent camrelizumab. The monotherapy
schedule consisted of 200 mg camrelizumab administered once every 3
weeks. A total of 12 cycles were completed. The detailed course of
treatment, including the initiation, adjustment and the duration of
each therapeutic intervention, is summarized in a flowchart in
Fig. 5. The Systemic therapy and
the follow-up was based at the West China hospital and Dazhu
People's Hospital.
Follow-up
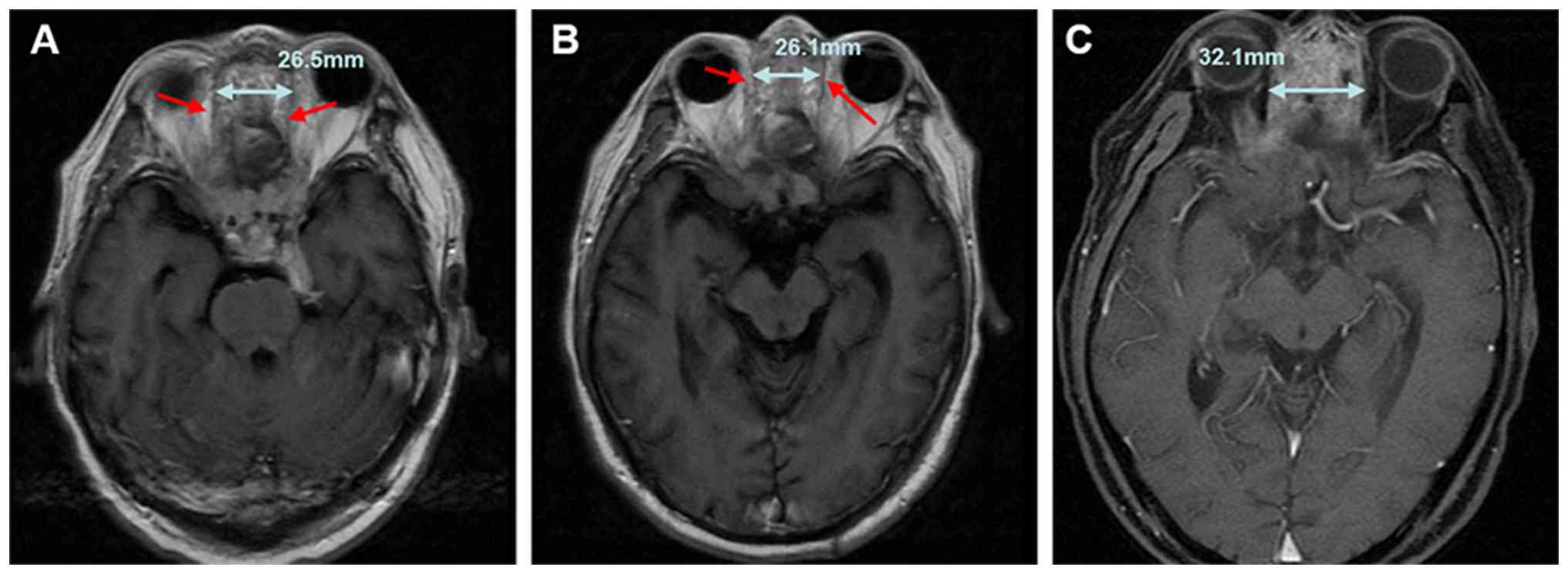
During treatment, the patient underwent MRI at each
follow-up at a fixed interval of 2–3 months (sometimes according to
their personal schedule). At the 11-month follow-up after the
initiation of chemoimmunotherapy (February 2025), MRI (T1-weighted
contrast-enhanced sequence) showed significant tumor progression
compared with the scan performed at the 7-month follow-up. The
longest diameter of the ethmoid sinus lesion had increased from
26.1 to 32.1 mm, with ill-defined boundaries and expanded invasion
into surrounding tissues, which was consistent with the progressive
disease based on the Response Evaluation Criteria in Solid Tumors
1.1 (RECIST 1.1) (Fig. 6).
Following disease progression, the patient received anlotinib (12
mg once daily for 2 weeks on and 1 week off, with 3 weeks per
cycle) and local interventional therapy to relieve symptoms, but
subsequently developed intracranial and pulmonary metastatic
progression. After that, the patient chose not to return for
regular follow-up visits and was managed with best supportive care,
being admitted to the hospital only when severe complications
(fatigue, infection and vomiting) developed.
Discussion
Sinonasal ITAC is an uncommon malignancy with a high
post-operative recurrence rate, which is a major cause of treatment
failure in numerous patients (23).
However, to date, no consensus has been reached on the recurrence
patterns of ITAC. The present case report aimed to explore the
clinical course of patients with ITAC experiencing recurrence. The
present study describes a detailed account of the management of a
60-year-old patient diagnosed with ITAC with cranial invasion. The
patient underwent an endoscopic combined transcranial tumor
resection, followed by adjuvant post-operative radiotherapy. Tumor
recurrence was observed after a 12-month follow-up period, and a
biopsy confirmed the pathology as ITAC. Immunohistochemical
analysis revealed PD-1 positivity at 5% and a PD-L1 CPS of 5.
Consequently, the treatment strategy was modified to include four
cycles of chemotherapy combined with anti-PD-1 immunotherapy,
followed by maintenance monotherapy with immunotherapy. At the
11-month follow-up, disease progression was observed.
The present case highlights two critical issues in
the treatment of advanced intracranial infiltrative ITAC, providing
valuable insight into the management of this rare neoplasm.
First, inadequate post-operative radiotherapy dosage
is likely a key factor contributing to early local recurrence.
Non-‘R0’ resection represents a high-risk factor for the early
post-operative recurrence of ITAC (24). In the present case, the tumor was
located in the ethmoid sinus and had extended to the skull and
orbit, adjacent to the optic nerve and other vital structures. The
anatomical complexity of this region rendered a complete ‘R0’
resection challenging, which may have contributed to the early
post-operative recurrence of the patient. In an observational
study, Abu-Shama et al (25)
analyzed the surgical data of 13 patients with localized recurrence
and found that difficulty was experienced in dissecting the
cribriform plate or the lateral lamina during surgery in 11 of
these patients.
An insufficient adjuvant radiotherapy dose may be
associated with early local recurrence. Based on the postoperative
radiotherapy doses for sinonasal intestinal-type adenocarcinoma
reported in the previous literature (26–31)
(Table I) and the positive surgical
margins of this patient, the radiation oncologists formulated an
initial radiotherapy plan with a total dose of 66 Gy. However, due
to concerns about potential optic nerve damage (with the optimal
maximum dose of the optic nerve recommended to be ≤54 Gy), the
patient ultimately received only 25 sessions, a dose substantially
lower than the planned regimen and the ≥66 Gy EQD2 generally
recommended for adjuvant radiotherapy in postoperative high-risk
areas (such as those with margin positivity, neural invasion or
extra-peritoneal invasion) (26).
Definite recurrence was observed at merely 12 months following
surgery, primarily involving the bilateral ethmoid sinus margins,
anterior left nasal cavity and nasal septum. These sites were
within or adjacent to the original radiotherapy target volume,
suggesting that an insufficient radiotherapy dose may be a key
factor in the early local recurrence.
 | Table I.Previous studies on postoperative
radiotherapy doses and clinical outcomes of ITAC. |
Table I.
Previous studies on postoperative
radiotherapy doses and clinical outcomes of ITAC.
| First author,
year | Literature
type | Tumor type | Radiation dose | Outcomes | (Refs.) |
|---|
| Parys et al,
2025 | Retrospective study
(200 cases) | ITAC | R1/R2 resection:
66–70 Gy/33-35 fractions; R0 resection: 60–62 Gy/30-31
fractions | - | (26) |
| Wang et al,
2025 | Retrospective study
(81 cases) | SNAC | R1/R2 resection:
66–70 Gy/33-35 fractions; R0 resection: 60–62 Gy/30-31
fractions | - | (27) |
| Hsu et al,
2015 | Case report (1
case) | ITAC | CTV-1: 70 Gy/35
fractions; CTV-2: 59.5 Gy/35 fractions | PFS: 37 months | (28) |
| Hoeben et
al, 2015 | Literature
review | ITAC | 60-66 Gy/30-33
fractions | - | (29) |
| Antognoni et
al, 2015 | Retrospective study
(30 cases) | ITAC | 50-60 Gy/25-30
fractions for CTV; 54 Gy/25-27 fractions for elective neck
irradiation | - | (30) |
| Askoxylakis et
al, 2016 | Retrospective study
(122 cases) | Sinonasal
tumors | Median total dose:
64 Gy/32-53 fractions | Patients who
received a dose of ≥60 Gy had significantly improved OS and
LRFS | (31) |
For ITACs involving complex anatomical structures
adjacent to critical organs (e.g., optic nerve and brainstem),
particularly those with high-risk factors, such as intracranial
invasion and narrow surgical margins, achieving ‘R0’ resection
margins during surgery is notably challenging. Therefore,
post-operative radiotherapy should aim to deliver a curative dose.
A dose of 66 Gy EQD2 is recommended, as per previous studies
(26,27). The application of advanced
radiotherapy modalities, including proton and heavy ion therapy, is
recommended for precise dose distribution. Proton and heavy ion
therapies, due to their distinctive Bragg peak effect, enable the
concentration of high radiation doses within the target area, while
substantially minimizing exposure to surrounding vital structures.
This capability facilitates safe dose escalation and enhances tumor
control (32,33). For instance, proton therapy has been
shown to achieve a 43% local control rate in patients with
unresectable advanced sinus tumors (34). Additionally, carbon ion therapy
permits dose escalation up to 73 Gy without an increase in acute
toxicity (35).
Secondly, PD-L1 expression-guided chemoimmunotherapy
followed by sequential immunotherapy maintenance may hold potential
clinical value for advanced recurrent sinonasal ITAC.
The pathological analysis of the biopsy tissue from
the recurrence site revealed 5% PD-1 positivity and a PD-L1 CPS of
5. Although the value is at the threshold, the presence of PD-1
positive tumor-infiltrating lymphocytes may indicate potential for
an enhanced immune response (36).
As an anti-PD-1 monoclonal antibody, camrelizumab blocks PD-1 on
immune cells to reverse PD-L1-induced immune suppression.
Consequently, a combination treatment regimen consisting of
albumin-bound paclitaxel, cisplatin and camrelizumab was
subsequently administered in the present case. This regimen was
formulated based on the following considerations: i) Chemotherapy
with paclitaxel and platinum compounds is known to induce
immunogenic tumor cell death and increase T-cell infiltration
(37,38); and ii) platinum compounds are
recognized for promoting PD-L1 expression and enhancing the
sensitivity to immune checkpoint inhibitors (39). Considering the tolerability of
long-term combination therapy, the treatment was subsequently
adjusted to camrelizumab monotherapy maintenance. This strategy,
termed ‘chemotherapy-induced combined immune maintenance’, has
shown efficacy in clinical practice for various solid tumors, such
as lung cancer (40). After the
initiation of this regimen, the patient achieved a PFS time of 11
months (followed by disease progression) with good treatment
tolerance.
Survival rates for early-stage ITAC are relatively
favorable. Dallan et al (41) reported a 5-year cancer-specific
survival rate of 81.7% in a cohort of 440 European patients with
ITAC, which was consistent with the findings in the study by Camp
et al (42). Additionally,
van de Velde et al (43)
documented a 5-year overall survival rate of 47.8% (95% CI,
39.4–55.6) among patients with ITAC in The Netherlands. By
contrast, the prognosis of patients with advanced or metastatic
ITAC remains less favorable. Sarradin et al (12) conducted a retrospective analysis on
the efficacy of chemotherapy in 6 patients with advanced ITAC,
revealing that 3 patients with meningeal involvement experienced
rapid disease progression, with a median PFS time of only 1.2
months. In the present case, the tumor of the patient had invaded
the orbit at initial diagnosis, breached the meninges, invaded the
skull and recurred swiftly post-surgery, all of which are
considered poor prognostic indicators. Nevertheless, the patient
achieved a prolonged PFS time of 11 months following
chemoimmunotherapy combined with subsequent immunotherapy
maintenance. This case provides preliminary evidence supporting the
potential of this therapeutic strategy for PD-1/PD-L1-positive
patients with advanced or recurrent ITAC.
The present case report has several limitations.
Firstly, as a single case report, the present study lacks
statistical analysis and can only provide limited reference value.
Secondly, due to the different positions of the patient during the
radiotherapy planning CT and the PET-CT scan for recurrence
diagnosis, precise registration comparison between the two could
not be performed. Nevertheless, sectional images near the optic
nerve (the main recurrence site) are included in Fig. S1. Thirdly, the recurrence diagnosis
described in this manuscript was based on pathology reports rather
than retrievable original image files, which may represent a
limitation of this study. Fourthly, due to the low incidence of
this tumor, there were virtually no established clinical guidelines
to reference during the management of this patient. The present
study merely proposes two preliminary hypotheses, and further data
from other cases and studies are required to provide stronger
evidence for the treatment of ITAC.
In conclusion, ITAC is characterized by local
invasiveness, which poses substantial challenges to treatment,
particularly when intracranial tissue is involved. The analysis of
this case provides two preliminary insights: First, an adequate
postoperative adjuvant radiotherapy dose (≥66 Gy EQD2) may be
important for controlling local recurrence. Second, for patients
with recurrent or metastatic ITAC, the assessment of PD-1/PD-L1
expression may provide a valuable reference for treatment
decision-making. In patients who are PD-1-positive or exhibit a
PD-L1 CPS ≥1, a treatment strategy involving chemotherapy followed
by sequential immune monotherapy maintenance therapy, such as
camrelizumab, may serve as a potential salvage treatment. This
approach may achieve durable disease control in some cases and
provide preliminary references for the treatment of advanced
ITAC.
Supplementary Material
Supporting Data
Acknowledgements
Not applicable.
Funding
The present study was funded by the Sichuan Provincial
Administration of Traditional Chinese Medicine Special Fund Top
Project (grant no. 2024MS191).
Availability of data and materials
The data generated in the present study may be
requested from the corresponding author.
Authors' contributions
ZL and LY contributed to the study
conceptualization. DL and ZL provided clinical advice on patient
management. QZ, SZ and QWZ participated in patient treatment and
clinical care. FY and WYG acquired and analyzed the medical imaging
data. LY and QZ participated in the analysis and interpretation of
clinical examination results, drafted and revised the manuscript.
ZL and LY confirm the authenticity of all the raw data. All authors
have read and approved the manuscript.
Ethics approval and consent to
participate
The study was approved by the Ethics Committee of
the General Hospital of Western Theater Command (approval no.
2025EC5-KY007). The patient and their family members were fully
informed about the treatment modality and provided signed informed
consent.
Patient consent for publication
Written informed consent was obtained from the
patient for the publication of this case report, including the
publication of all images, clinical data and other data included in
the manuscript.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Leivo I: Intestinal-type adenocarcinoma:
Classification, immunophenotype, molecular features and
differential diagnosis. Head Neck Pathol. 11:295–300. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Baptista Freitas M, Costa M, Freire Coelho
A, Rodrigues Pereira P, Leal M, Sarmento C, Águas L and Barbosa M:
Sinonasal adenocarcinoma: Clinicopathological characterization and
prognostic factors. Cureus. 16:e560672024.PubMed/NCBI
|
|
3
|
Kennedy MT, Jordan RCK, Berean KW and
Perez-Ordoñez B: Expression pattern of CK7, CK20, CDX-2, and villin
in intestinal-type sinonasal adenocarcinoma. J Clin Pathol.
57:932–937. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Leivo I: Sinonasal adenocarcinoma: Update
on classification, immunophenotype and molecular features. Head
Neck Pathol. 10:68–74. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Eggesbø HB: Imaging of sinonasal tumours.
Cancer Imaging. 12:136–152. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Rampinelli V, Ferrari M and Nicolai P:
Intestinal-type adenocarcinoma of the sinonasal tract: An update.
Curr Opin Otolaryngol Head Neck Surg. 26:115–121. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Siddiqui F, Smith RV, Yom SS, Beitler JJ,
Busse PM, Cooper JS, Hanna EY, Jones CU, Koyfman SA, Quon H, et al:
ACR appropriateness criteria® nasal cavity and paranasal
sinus cancers. Head Neck. 39:407–418. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Meccariello G, Deganello A, Choussy O,
Gallo O, Vitali D, De Raucourt D and Georgalas C: Endoscopic nasal
versus open approach for the management of sinonasal
adenocarcinoma: A pooled-analysis of 1826 patients. Head Neck. 38
(Suppl 1):E2267–E2274. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Vermassen T, De Keukeleire S, Saerens M,
Heerwegh S, Debacker JM, Huvenne W, Deron P, Creytens D, Ferdinande
L, Rottey S, et al: Choice of surgery in intestinal-type
adenocarcinoma of the sinonasal tract: A long-term comparative
study. Eur Arch Otorhinolaryngol. 281:2993–3004. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Keizer ME, Hovinga KE, Lacko M, Eekers
DBP, Baijens LWJ, Kremer B and Temel Y: Clinical outcome in
patients with large sinonasal tumors with intracranial extension. J
Neurol Surg B Skull Base. 85:347–357. 2023.PubMed/NCBI
|
|
11
|
Licitra L, Suardi S, Bossi P, Locati LD,
Mariani L, Quattrone P, Lo Vullo S, Oggionni M, Olmi P, Cantù G, et
al: Prediction of TP53 status for primary cisplatin, fluorouracil,
and leucovorin chemotherapy in ethmoid sinus intestinal-type
adenocarcinoma. J Clin Oncol. 22:4901–4906. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Sarradin V, Betrian S, Chaltiel L, Brac De
La Perriere C and Delord JP: 5-Fluorouracil with oxaliplatin and/or
irinotecan for advanced sinonasal intestinal-type adenocarcinoma
(ITAC). Bull Cancer. 110:168–173. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Bossi P, Perrone F, Miceli R, Cantù G,
Mariani L, Orlandi E, Fallai C, Locati LD, Cortelazzi B, Quattrone
P, et al: Tp53 status as guide for the management of ethmoid sinus
intestinal-type adenocarcinoma. Oral Oncol. 49:413–419. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Szablewski V, Solassol J, Poizat F,
Larrieux M, Crampette L, Mange A, Bascoul-Mollevi C and Costes V:
EGFR expression and KRAS and BRAF mutational status in
intestinal-type sinonasal adenocarcinoma. Int J Mol Sci.
14:5170–5181. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Hermsen MA, Riobello C, García-Marín R,
Cabal VN, Suárez-Fernández L, López F and Llorente JL:
Translational genomics of sinonasal cancers. Semin Cancer Biol.
61:101–109. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Le X, Ferrarotto R, Wise-Draper T and
Gillison M: Evolving role of immunotherapy in recurrent metastatic
head and neck cancer. J Natl Compr Cancer Netw. 18:899–906. 2020.
View Article : Google Scholar
|
|
17
|
Harrington KJ, Burtness B, Greil R,
Soulières D, Tahara M, de Castro G Jr, Psyrri A, Brana I, Basté N,
Neupane P, et al: Pembrolizumab with or without chemotherapy in
recurrent or metastatic head and neck squamous cell carcinoma:
Updated results of the phase III KEYNOTE-048 study. J Clin Oncol.
41:790–802. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
André T, Shiu KK, Kim TW, Jensen BV,
Jensen LH, Punt CJA, Smith D, Garcia-Carbonero R, Alcaide-Garcia J,
Gibbs P, et al: Pembrolizumab versus chemotherapy in microsatellite
instability-high or mismatch repair-deficient metastatic colorectal
cancer: 5-year follow-up from the randomized phase III KEYNOTE-177
study. Ann Oncol. 36:277–284. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Riobello C, Vivanco B, Reda S,
López-Hernández A, García-Inclán C, Potes-Ares S, Cabal VN, López
F, Llorente JL and Hermsen MA: Programmed death ligand-1 expression
as immunotherapeutic target in sinonasal cancer. Head Neck.
40:818–827. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Thomas Z, Jambunathan P, Jibi A, John AO
and Singh A: Low-dose nivolumab and cabozantinib in recurrent
intestinal-type papillary adenocarcinoma of the sinonasal region.
BMJ Case Rep. 16:e2550212023. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Yang L, Lu L, Ma J, Xu Z and Li N: A case
of high-grade non-intestinal paranasal sinus adenocarcinoma primary
in the maxillary sinus: Targeted therapy after postoperative
immunocombination with chemotherapy. J Cancer Res Clin Oncol.
150:3812024. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Paver EC, Cooper WA, Colebatch AJ,
Ferguson PM, Hill SK, Lum T, Shin JS, O'Toole S, Anderson L,
Scolyer RA and Gupta R: Programmed death ligand-1 (PD-L1) as a
predictive marker for immunotherapy in solid tumours: A guide to
immunohistochemistry implementation and interpretation. Pathology.
53:141–156. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Huang EI, Lu A, Tsai YT, Wang TC, Chuang
HC, Chen WC and Chen PT: Decreasing recurrence and increasing
survival rates in patients of ethmoid or sphenoid intestinal-type
adenocarcinomas: Systematic review and meta-analysis with 1126
cases. Medicine (Baltimore). 100:e273412021. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Riobello C, Sánchez-Fernández P, Córdoba
MCC, González-Gutiérrez M, Vivanco B, Cabal VN, Fernández LS,
García-Marín R, Codina-Martínez H, Lorenzo-Guerra SL, et al:
Next-generation sequencing reveals remarkable genetic stability in
primary and corresponding recurrent intestinal-type sinonasal
adenocarcinoma. Head Neck. 46:2010–2019. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Abu-Shama Y, Renard S, Nguyen DT, Henrot
P, Toussaint B, Rumeau C, Gallet P and Jankowski R: Descriptive
analysis of recurrences of nasal intestinal-type adenocarcinomas
after radiotherapy. Head Neck. 44:1356–1367. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Parys QA, De Witte M, Hauben E, Clement
PM, Hermans R, Decramer T, van Loon J, Nuyts S, Jorissen M, Vander
Poorten V and Van Gerven L: Long-term outcomes of endoscopic
resection and tailored adjuvant radiotherapy for sinonasal
intestinal-type adenocarcinoma: A historical single-center cohort
study in 200 patients. Front Oncol. 15:15221132025. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Wang J, Wang T, Zhao K, Li Y and Song X: A
retrospective analysis of patients with primary sinonasal
adenocarcinoma underwent radiotherapy running title: Radiotherapy
for sinonasal adenocarcinoma. Sci Rep. 15:371152025. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Hsu YC, Lu TY, Huang CJ and Huang MY:
Intensity-modulated radiation therapy for a sinonasal
intestinal-type adenocarcinoma patient: A rare case report. Ther
Radiol Oncol. 1:32017. View Article : Google Scholar
|
|
29
|
Hoeben A, van de Winkel L, Hoebers F,
Kross K, Driessen C, Slootweg P, Tjan-Heijnen VC and van Herpen C:
Intestinal-type sinonasal adenocarcinomas: The road to molecular
diagnosis and personalized treatment. Head Neck. 38:1564–1570.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Antognoni P, Turri-Zanoni M, Gottardo S,
Molteni M, Volpi L, Facco C, Freguia S, Mordacchini C, AlQahtani A,
Bignami M, et al: Endoscopic resection followed by adjuvant
radiotherapy for sinonasal intestinal-type adenocarcinoma:
Retrospective analysis of 30 consecutive patients. Head Neck.
37:677–684. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Askoxylakis V, Hegenbarth P, Timke C,
Saleh-Ebrahimi L, Debus J, Röder F and Huber PE: Intensity
modulated radiation therapy (IMRT) for sinonasal tumors: A single
center long-term clinical analysis. Radiat Oncol. 11:172016.
View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Dagan R, Uezono H, Bryant C, Holtzman AL,
Morris CG and Mendenhall WM: Long-term outcomes from proton therapy
for sinonasal cancers. Int J Part Ther. 8:200–212. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Mirandola A, Russo S, Bonora M, Vischioni
B, Camarda AM, Ingargiola R, Molinelli S, Ronchi S, Rossi E, Vai A,
et al: A patient selection approach based on NTCP models and DVH
parameters for definitive proton therapy in locally advanced
sinonasal cancer patients. Cancers (Basel). 14:26782022. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Saito T, Ishikawa H, Ohnishi K, Aihara T,
Mizumoto M, Fukumitsu N, Sugawara K, Okumura T and Sakurai H:
Proton beam therapy for locally advanced and unresectable (T4bN0M0)
squamous cell carcinoma of the ethmoid sinus: A report of seven
cases and a literature review. Oncol Lett. 10:201–205. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Jensen AD, Nikoghosyan AV, Ecker S,
Ellerbrock M, Debus J and Münter MW: Carbon ion therapy for
advanced sinonasal malignancies: Feasibility and acute toxicity.
Radiat Oncol. 6:302011. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Ma KL, Mitchell TC, Dougher M, Sharon CE,
Tortorello GN, Elder DE, Morgan EE, Gimotty PA, Huang AC, Amaravadi
RK, et al: Tumor-infiltrating lymphocytes in necrotic tumors after
melanoma neoadjuvant anti-PD-1 therapy correlate with pathologic
response and recurrence-free survival. Clin Cancer Res.
30:4987–4994. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Larionova I, Cherdyntseva N, Liu T,
Patysheva M, Rakina M and Kzhyshkowska J: Interaction of
tumor-associated macrophages and cancer chemotherapy.
Oncoimmunology. 8:15960042019. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Melero I, Castanon E, Alvarez M, Champiat
S and Marabelle A: Intratumoural administration and tumour tissue
targeting of cancer immunotherapies. Nat Rev Clin Oncol.
18:558–576. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Lesterhuis WJ, Punt CJA, Hato SV,
Eleveld-Trancikova D, Jansen BJ, Nierkens S, Schreibelt G, de Boer
A, Van Herpen CM, Kaanders JH, et al: Platinum-based drugs disrupt
STAT6-mediated suppression of immune responses against cancer in
humans and mice. J Clin Invest. 121:3100–3108. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Li Y, Zhao J, Li R, Yao X, Dong X, Zhang R
and Li Y: Treatment options for tumor progression after initial
immunotherapy in advanced non-small cell lung cancer: A real-world
study. Neoplasia. 57:1010432024. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Dallan I, Fiacchini G, Tricò D, Barucco M,
Turri-Zanoni M, Ferrari M, Di Girolami L, Schiavo G, Emanuelli E,
Mattavelli D, et al: Sinonasal intestinal-type adenocarcinoma:
Multi-institutional retrospective analysis based on 440 patients
with long-term follow-up. Eur J Cancer. 226:1156232025. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Camp S, Van Gerven L, Poorten VV, Nuyts S,
Hermans R, Hauben E and Jorissen M: Long-term follow-up of 123
patients with adenocarcinoma of the sinonasal tract treated with
endoscopic resection and postoperative radiation therapy. Head
Neck. 38:294–300. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
van de Velde LJ, Breimer GE, Scheurleer
WFJ, de Ridder M, Devriese LA, Braunius WW, de Bree R, van Dijk BAC
and Rijken JA: Sinonasal intestinal-type adenocarcinoma in the
Netherlands: A nationwide study (2008–2022). Head Neck.
47:2531–2540. 2025. View Article : Google Scholar : PubMed/NCBI
|