Introduction
Colorectal cancer (CRC) has become the third most
commonly diagnosed cancer and the second most common cause of
cancer-related death globally, as estimated by the International
Agency for Research on Cancer (1).
In 2022, the National Cancer Center of China recorded 517,100 newly
diagnosed cases and 240,000 deaths of CRC (2). Metastasis is a notable cause of
CRC-related death (3); although new
therapies, such as systemic therapy, extensive surgery and local
ablative therapies, have already been utilized for the treatment of
metastatic CRC disease and doubled the overall survival for
late-stage CRC by up to 3 years, survival rates are still markedly
higher for patients with CRC without metastasis (4). Consequently, elucidating the
mechanisms underlying the metastasis and invasion of CRC is
essential for developing effective therapeutic strategies.
Epithelial-mesenchymal transition (EMT) is a central
mechanism for diversifying the cells found in complex tissues and
is associated with the formation of the parietal endoderm (5). EMT has been shown to play a marked
role in promoting the metastasis of epithelium-derived carcinoma
(6,7). The molecular markers for EMT include
the increased expression of N-cadherin, vimentin, Snail1, Snail2
(Slug), as well as the reduced expression of E-cadherin. Phenotypic
markers for EMT include increased capacity for migration and
invasion, as well as resistance to apoptosis (8). In the first step of the
invasion-metastasis cascade, CRC cells isolate themselves from one
another and nearby normal cells by downregulating E-cadherin. The
release of pro-angiogenic factors results in the formation of new
blood vessels and the infiltration of tumor cells into the
bloodstream (9,10). Snail plays a notable role in the
development of CRC and predominantly promotes EMT by suppressing
E-cadherin. Furthermore, Snail can induce the expression of genes
related to the mesenchymal phenotype, such as fibronectin and
N-cadherin (11). It is reported
that the expression levels of Snail in the tumor stroma are
associated with distant metastasis and the lower specific survival
of patients with CRC (12).
Furthermore, the expression of N-cadherin was reported to be
upregulated in CRC tissues and negatively associated with the
expression of E-cadherin, which was associated with tumor
differentiation, metastasis and the reduced survival of patients
with CRC (13). Another study
demonstrated that the upregulation of vimentin contributed to the
progression and poor prognosis of CRC (14).
5-Lipoxygenase (5-LOX) catalyzes the oxygenation of
arachidonic acid to generate leukotrienes (15). Furthermore, the overexpression of
5-LOX is known to play a marked role in the development of CRC
(16). Clinical studies have shown
that the expression levels of 5-LOX are notably upregulated in
human colorectal tumors when compared with the adjacent normal
mucosa (17,18). Zileuton, a 5-LOX inhibitor, was
found to inhibit the growth of colon cancer xenografts in athymic
mice (18). Another 5-LOX
inhibitor, AA861, notably promotes apoptosis and inhibited the
invasion and proliferation of 5-LOX-overexpressed HCA7 cells, while
an investigation involving 5-LOX-/- ApcMin/+ mice
demonstrated a marked reduction in the numbers and sizes of the
adenomas, a notable improvement in survival rate and inhibition of
the PI3K/AKT pathway via promoting PTEN (19). Bošković et al (20) synthesized 13 COX-2 and 5-LOX
inhibitors; among these inhibitors, N-hydroxy urea derivatives of
indomethacin and diclofenac exhibited a potent antimigratory effect
in the SW620 cell line.
Collectively, these existing studies investigated
the effects of 5-LOX inhibitors on the proliferation, apoptosis and
invasion of CRC cells. However, to the best of our knowledge, no
previous study has compared the effects of 5-LOX knockdown and
overexpression on the migration and invasion of CRC cells.
Furthermore, the specific relationship between 5-LOX and EMT in CRC
cells has yet to be elucidated. To elucidate the role of 5-LOX,
small interfering RNAs (siRNAs) and recombinant lentiviruses were
specifically designed to knock down and overexpress the 5-LOX gene
in CRC cells, respectively. Angiogenesis is the beginning of tumor
development, which creates necessary conditions for tumor invasion
and metastasis (21); vascular
endothelial growth factor (VEGF) and angiogenin are two major
proangiogenic factors that play marked roles in tumor angiogenesis
(22,23). Therefore, the effects of media
collected from CRC cells with or without 5-LOX expression on the
mRNA levels of proangiogenic factors in human umbilical vein
endothelial cells (HUVECs) were also investigated. In the present
study, the role of 5-LOX in migration, invasion and EMT of CRC
cells was evaluated by overexpression and knockdown of 5-LOX.
Materials and methods
Cell lines and culture
The HCT116, HT29, RKO and SW480 human colorectal
cancer cell lines were purchased from Procell Biotechnology Co,.
Ltd. Authentication of the aforementioned cell lines was conducted
using short tandem repeat profiling. HUVECs were purchased from the
Cell Resource Center, Institute of Basic Medical Sciences,
CAMS/PUMC. HCT116 and RKO cell lines were cultured with Dulbecco's
modified Eagle's medium (Sangon Biotech Co., Ltd.) containing 10%
fetal bovine serum (FBS) (PAN-Biotech GmbH) and 1%
penicillin/streptomycin (Sangon Biotech Co., Ltd.). HT29 and SW480
cell lines were cultured in RPMI-1640 (Sangon Biotech Co., Ltd.)
containing 10% FBS and 1% penicillin/streptomycin. HUVECs were
cultured in a HUVEC specific complete medium (Procell Life Science
& Technology Co., Ltd.). All cells were maintained in a
humidified atmosphere at 37°C containing 5% CO2.
siRNA sequence and transfection in
vitro
A total of three different siRNAs (50 µM) of 5-LOX
were synthesized to knock down the expression of 5-LOX in HCT116
cells, and a non-targeting siRNA control (si-NC) was transfected to
the cells as a negative control. These sequences were synthesized
by Huzhou Hippo Biotechnology Co., Ltd. as follows: si-5-LOX-#1,
5′-CCAAAUGCCACAAGGAUUUTT-3′ (sense) and 5′-AAAUCCUUGUGGCAUUUGGCA-3′
(antisense); si-5-LOX-#2, 5′-GCAGGAAGACCUGAUGUUUTT-3′ (sense); and
5′-AAACAUCAGGUCUUCCUGCCA- 3′ (antisense); si-5-LOX-#3,
5′-CCAUUGCAAUCAACACCAATT-3′ (sense); and
5′-UUGGUGUUGAUUGCAAUGGTG-3′ (antisense); si-NC,
5′-UUCUCCGAACGUGUCACGUdTdT-3′ (sense) and
5′-ACGUGACACGUUCGGAGAAdTdT-3′ (antisense). HCT116 cells in the
logarithmic growth phase were seeded into six-well plates at a
density of 2×105 per well. Transfection with 40 nM siRNA
was performed with Lipofectamine RNAiMAX transfection reagent (cat.
no. 13778030; Thermo Fisher Scientific, Inc.) in accordance with
the manufacturer's instructions. Following transfection at 37°C for
48 h, gene knockdown efficiency was measured by quantitative PCR
(qPCR) and western blotting analysis.
Recombinant lentivirus infection
Recombinant lentiviruses overexpressing 5-LOX
(OE-5-LOX) were constructed by using the GV365 plasmid
(pUbi-MCS-3FLAG-pCMV-EGFP), with empty vector control
lentiviruses (OE-Vector) as control. All lentiviruses were produced
by Shanghai GeneChem Co., Ltd.. The lentiviruses were generated by
transfecting GV365, pHelper 1.0 and pHelper 2.0 plasmid (Shanghai
GeneChem Co., Ltd.) into 293T cells with E-trans transfection
reagents (cat. no. REVG007; Shanghai GeneChem Co., Ltd.) at 37°C
for 48 h. The titer of the lentivirus used for infection was
2×108 transducing units/ml. RKO cells in the logarithmic
growth phase were seeded into six-well plates at a density of
2×105/well. Lentiviruses were infected into the RKO
cells at a multiplicity of infection of 50, supplemented with 5
µg/ml polybrene (MilliporeSigma) to enhance infection efficiency.
After incubation at 37°C for 72 h, infection efficiency was
assessed by fluorescence microscopy. Successful overexpression of
the target 5-LOX gene was verified by qPCR and western blotting
analysis.
Incubation of HUVECs
HCT116 cells (1×106 cells/well) were
transfected with siRNA and RKO cells (1×106 cells/well)
were infected with lentivirus (as aforementioned). Following siRNA
transfection (48 h) and lentivirus infection (72 h), the culture
media was replaced with fresh media; conditioned media were
subsequently collected after 24 h of incubation. HUVECs
(1×106 cells/well) were plated in 6-well plates and
incubated with the media collected from siRNA-transfected HCT116 or
lentivirus-infected RKO cells at 37°C for 24 h. HUVECs were then
divided into four groups: si-NC group (transfected with the
non-targeting siRNA control) and si-5-LOX-#2 group (transfected
with si-5-LOX-#2), OE-Vector group (infected with empty vector
control lentiviruses) and OE-5-LOX group (infected with 5-LOX
overexpressing lentiviruses). Consequently, qPCR was used to detect
the mRNA levels of VEGF and angiogenin in HUVECs. The expression of
VEGF protein in HUVECs was determined by western blotting
analysis.
Cell viability assay
Cell viability was investigated using the cell
counting kit (CCK)-8 assay (cat. no. C0037; Beyotime
Biotechnology). HUVECs (2,500 cells/well) were cultured with media
collected from siRNA-transfected HCT116 cells or
lentivirus-infected RKO cells in a 96well culture plate at 37°C for
24, 48 and 72 h. The absorbance was detected at 450 nm after the
cells had been treated with 10% CCK-8 at 37°C for 2 h. Cell
viability was calculated as the ratio of optical density (OD)
values of medium-treated cells (24, 48 and 72 h) to those of naive
cells (0 h).
Tumor cell migration and invasion
assays
HCT116 cells transfected with siRNA were divided
into si-NC and si-5-LOX#2 groups. RKO cells infected with
lentiviruses were divided into OE-Vector group and OE-5-LOX group.
HCT116 cells (8×104 cells/well) and RKO cells
(4×104 cells/well) were suspended in serum-free medium
and deposited into the upper Transwell chamber. Upper chambers
without Matrigel or Matrigel-coated were used for migration and
invasion assays, respectively. For the invasion assay, Matrigel
(Corning, Inc.) was diluted in serum-free medium at a ratio of 1:8.
A uniform layer was applied to the upper chamber of the Transwell
inserts, and the coated inserts were incubated at 37°C for 1 h to
allow polymerization. A medium containing 10% FBS was added to the
lower chamber for the two assays. Cells were cultured at 37°C and
5% CO2 for 24 h and then cleaned with PBS. After
discarding the PBS, cells were fixed with methanol (MilliporeSigma)
for 15 min at room temperature and then stained with 0.1% crystal
violet (MilliporeSigma) for 15 min at room temperature. Stained
cells were imaged using a light microscope (×100 magnification),
and a total of five random fields per insert were captured.
Wound healing assay
Transfected HCT116 cells and infected RKO cells were
seeded at a density of 1×106 cells/well in a six-well
culture plate attaining 90% confluence, then a wound was created
using a 200 µl pipette tip. Images were captured at three random
fields of view by a light microscope (×40 magnification) at 0 and
24 h. Wound area was measured using ImageJ software (version 1.54g;
National Institutes of Health). Wound-healing assay results were
presented as migration rate (%)=(initial wound area-wound area at
24 h)/initial wound area ×100.
Apoptosis assay
Transfected HCT116 cells and infected RKO cells were
collected and suspended in Binding Buffer at a density of
1×106 cells/ml. HCT116 and RKO cells were stained with
Annexin V-FITC and Annexin V-APC Apoptosis Detection kits (cat. no.
88-8005-74/88-8007-74; Invitrogen; Thermo Fisher Scientific, Inc.)
in accordance with the manufacturer's instructions. Apoptosis was
examined by flow cytometry (cytoFLEX; Beckman Coulter, Inc.). The
apoptotic rate was assessed with CytExpert (V 2.4.0.28) software
(Beckman Coulter, Life science).
Total RNA isolation and reverse
transcription-qPCR analysis (RT-qPCR)
Total RNA of HCT116, HT29, RKO, SW480, transfected
HCT116, infected RKO and HUVECs was extracted by using RNAprep Pure
Cell/Bacteria Kit (cat. no. DP430; Tiangen Biotech Co., Ltd.) in
accordance with the manufacturer's instructions. RT was performed
with a ReverTra Ace qPCR RT kit (cat. no. FSQ-101; Toyobo Co.,
Ltd.) in a total volume of 20 µl. RT was performed as follows: 37°C
for 15 min and 98°C for 5 min. The qPCR was performed with SYBR
Green qPCR Master Mix (cat. no. HY-K0522; MedChemExpress). The
reaction protocol was as follows: 5 min initial denaturation at
95°C, followed by 40 cycles of amplification with 15 sec at 95°C,
30 sec at 60°C and 30 sec at 72°C for each cycle. This was followed
by a melt curve analysis to determine the reaction specificity.
β-actin was used as the endogenous control. The oligonucleotide
primers used were as follows: β-actin, 5′-CATGTACGTTGCTATCCAGGC-3′
(forward) and 5′-CTCCTTAATGTCACGCACGAT-3′ (reverse); 5-LOX,
5′-ACAAGCCCTTCTACAACGACT-3′ (forward) and 5′-AGCTGGATCTCGCCCAGTT-3′
(reverse); Angiogenin, 5′-GTTGGTCTTCGTGCTGGGTCTG-3′ (forward) and
5′-AGTGCTGGGTCAGGAAGTGTGT-3′ (reverse); VEGF,
5′-GGCAGAAGGAGGAGGGCAGAAT-3′ (forward) and
5′-GGGCACACAGGATGGCTTGAAG-3′ (reverse). The 2−ΔΔCq
method was used to calculate the relative RNA expression (24).
Protein extraction and western
blotting (WB)
HCT116, HT29, RKO, SW480, transfected HCT116 and
infected RKO cells were incubated and then centrifuged at 175 × g
to collect cells. Total protein was extracted using RIPA buffer
(Beyotime Biotechnology). Protein concentration was determined with
a BCA protein quantification kit (cat. no. P0009; Beyotime
Biotechnology) in accordance with the manufacturer's instructions.
Extracted proteins (20 µg) were then separated by SDS-PAGE in
10–12% acrylamide gel and transferred to a polyvinylidene fluoride
membrane (MilliporeSigma). The membrane was subsequently blocked
with 5% skimmed milk in TBS containing 0.05% Tween-20 (Beyotime
Biotechnology) for 1 h at room temperature. The membrane was then
incubated with a primary antibody (1:1,000) overnight at 4°C. The
primary antibodies were 5-LOX (cat. no. 83794-4-RR; Proteintech
Group, Inc.), E-cadherin (cat. no. 20874-1-AP; Proteintech Group,
inc.), N-cadherin (cat. no. 13116; Cell Signaling Technology,
Inc.), Snail (cat. no. 13099-1-AP; Proteintech Group, Inc.),
Vimentin (cat. no. 5741; Cell Signaling Technology, Inc.), VEGFA
(cat. no. 81323-2-RR; Proteintech Group, Inc.) and GAPDH (cat. no.
81640-5-RR; Proteintech Group, Inc.). The membrane was washed three
times in TBST and incubated with diluted secondary antibody
(1:10,000) conjugated with horseradish peroxidase solution (cat.
no. ab205718; Abcam) for 1 h at room temperature. After washing,
the membrane was visualized with ECL substrate (cat. no. SW2050;
Beijing Solarbio Science & Technology Co., Ltd.) by using
chemiluminescence (AzureC500; Azure Biosystems, Inc.). The gray
values of the bands were quantified using ImageJ (version 1.54g;
National Institutes of Health), and GAPDH was used as the loading
control.
Enzyme-linked immunosorbent assay
(ELISA)
RKO cells infected with lentiviruses (the OE-Vector
and OE-5-LOX groups) were seeded in six-well plates at a density of
5×105 cells/well. After 72 h of incubation at 37°C,
media were replaced with fresh media, and the cells were cultured
at 37°C for another 48 h before supernatant collection. The
concentrations of leukotriene E4 (LTE4) and
leukotriene B4 (LTB4) in the supernatant were
detected using a LTE4 ELISA kit (cat. no. MM-928804O1)
and a LTB4 ELISA kit (cat. no. MM-927697O1; both Jiangsu
Enzyme Immunoassay Co., Ltd.), in accordance with the
manufacturer's instructions. OD values were measured at 450 nm, and
sample concentrations were calculated based on the standard curve.
Relative expression was calculated as concentration ration of the
OE-5-LOX group relative to the OE-Vector group.
Statistical analysis
Data are expressed as the mean ± standard error of
the mean. Statistical analysis was performed using GraphPad Prism
statistical package (version 9.5; Dotmatics). Comparisons between
two groups were conducted with Student's t-test. Analysis of
more than two groups was performed with one-way ANOVA followed by
Dunnett's multiple comparison test. P<0.05 was considered to
indicate a statistically significant difference.
Results
Expression of 5-LOX in different CRC
cell lines
To select the prime candidate CRC cell line for
further 5-LOX gene knockdown and overexpression, the mRNA and
protein expression levels of 5-LOX were analyzed in four CRC cell
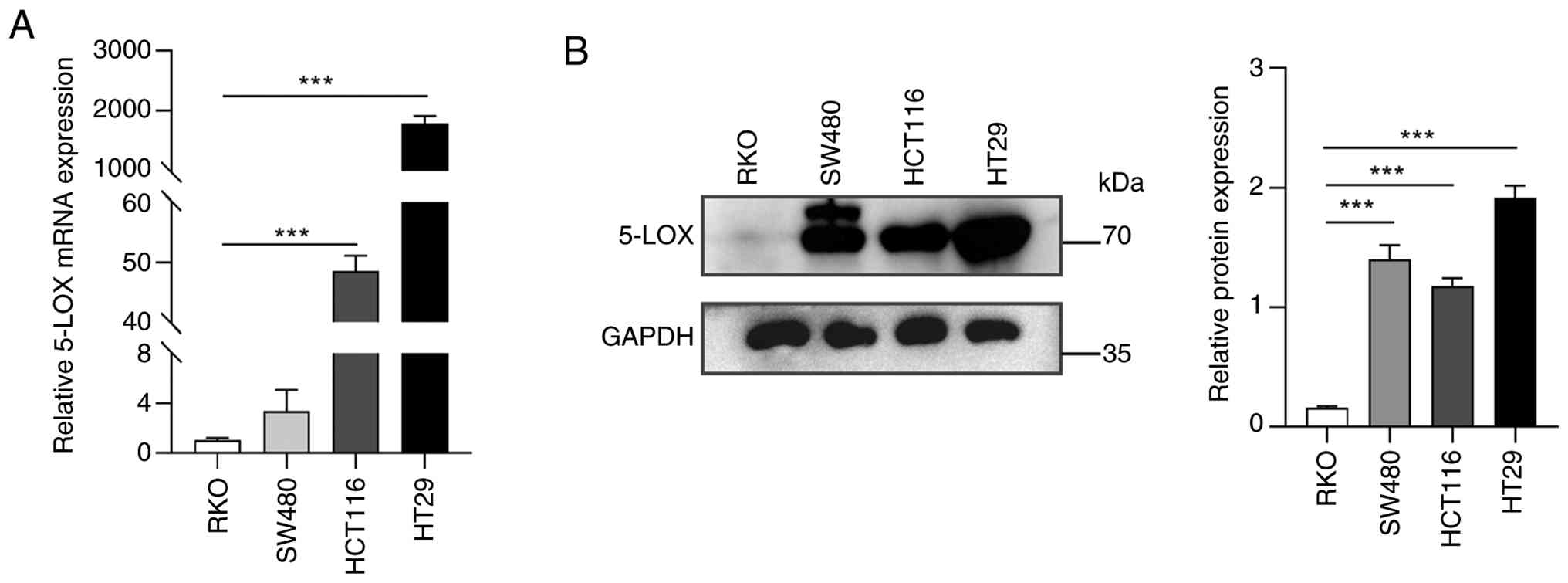
lines: HCT116, HT29, RKO and SW480. As shown in Fig. 1A, high levels of 5-LOX mRNA were
observed in HCT116 and HT29 cells while low levels of 5-LOX mRNA
were observed in RKO and SW480 cells. The 5-LOX protein was widely
expressed in HCT116, HT29 and SW480 cells other than RKO cells
(Fig. 1B). Thus, HCT116 cells,
which exhibited high levels of 5-LOX expression, were used for the
subsequent gene knockdown investigations. RKO cells, with low
levels of 5-LOX expression, were used for gene overexpression
investigations.
Inhibitory effects of siRNAs designed
to target 5-LOX
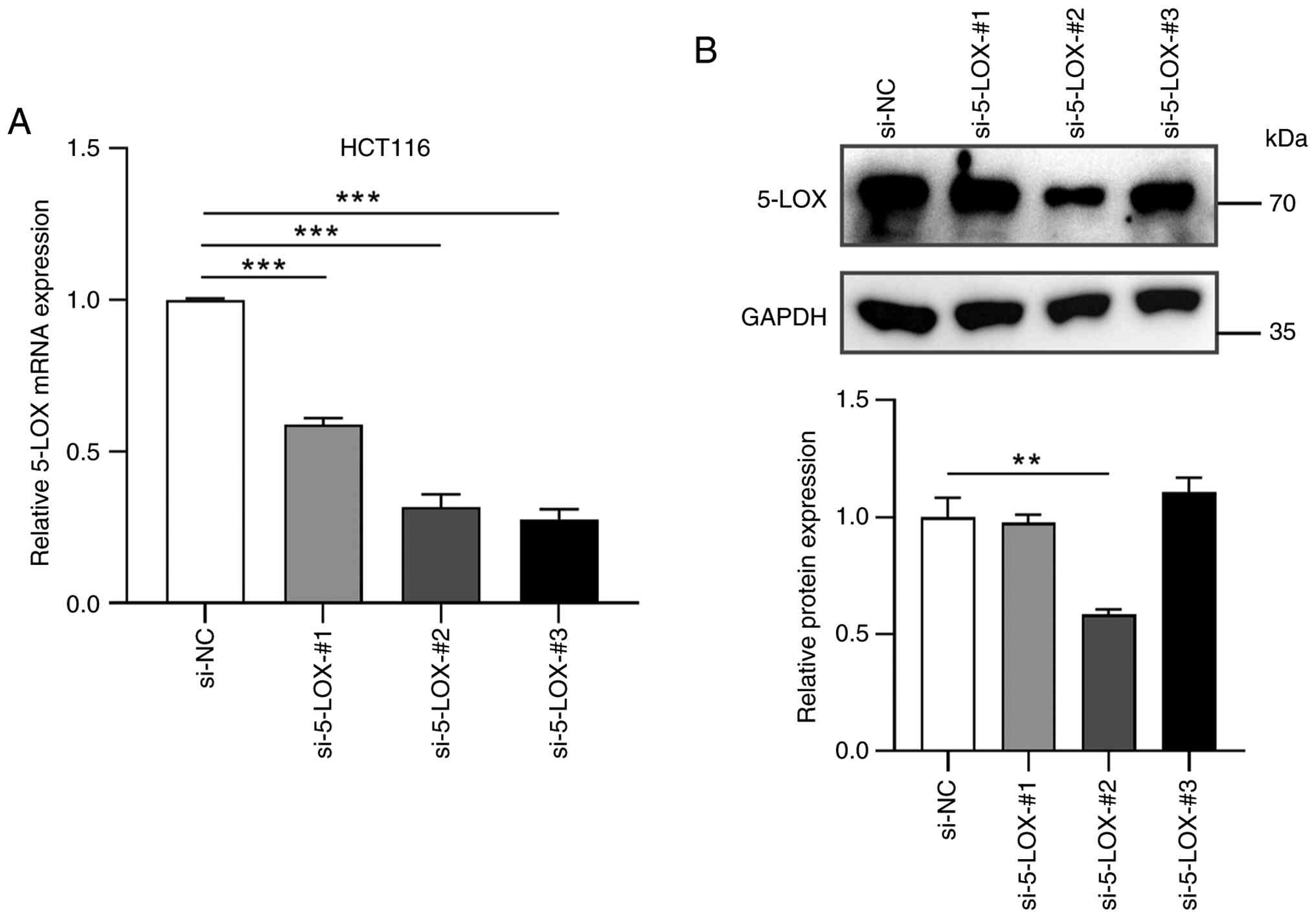
The mRNA levels of 5-LOX in HCT116 cells were
determined by RT-qPCR following treatments with siRNA for 48h. As
shown in Fig. 2A, three different
siRNAs inhibited the 5-LOX mRNA levels up to 72% compared with the
si-NC group. Of these three siRNAs, si-5-LOX-#2 inhibited the
expression of 5-LOX protein more effectively than the other siRNAs
(Fig. 2B). Therefore, the following
investigations were conducted by using si-5-LOX-#2.
Overexpression of 5-LOX in RKO
cells
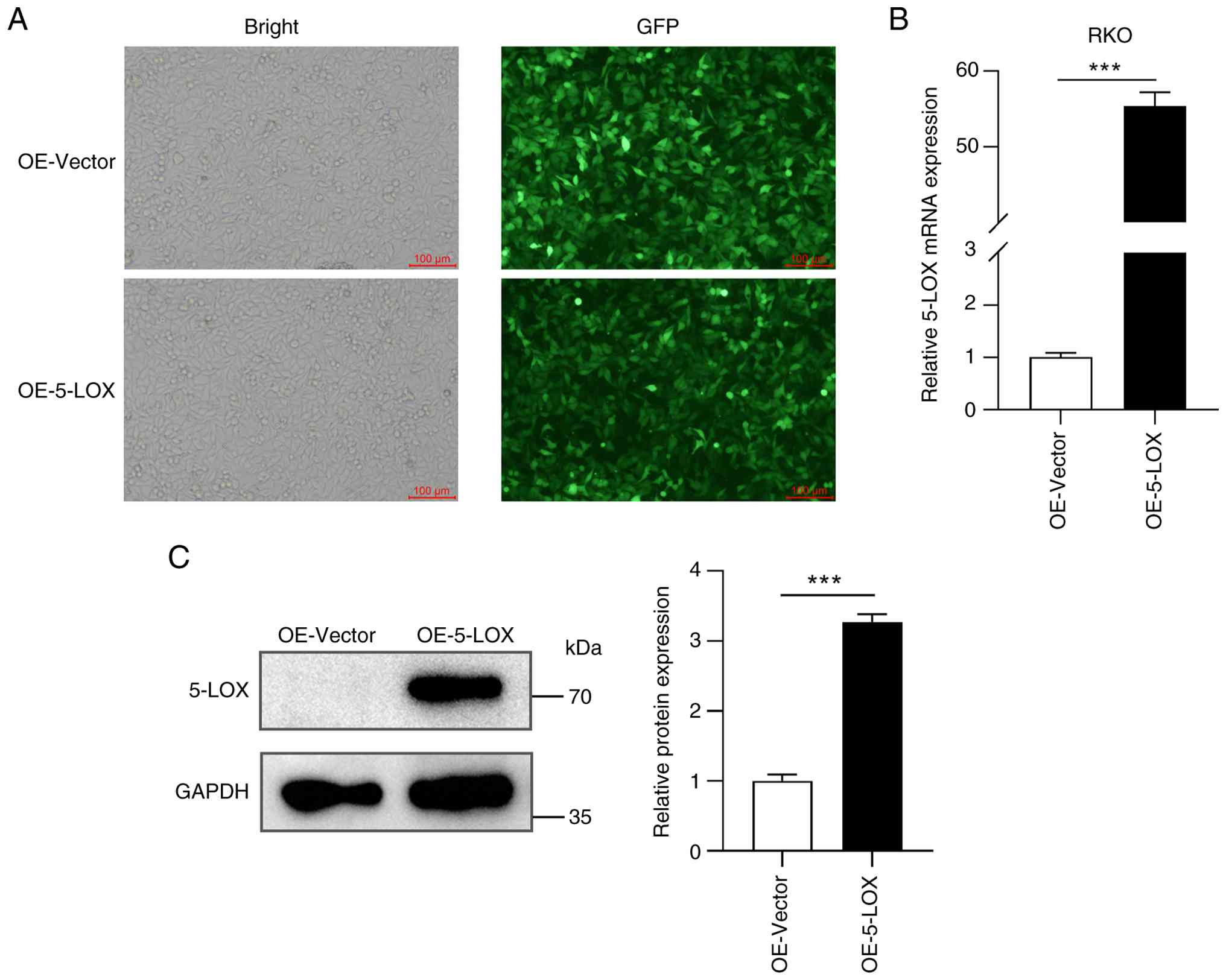
Recombinant lentiviruses expressing 5-LOX were used
to infect RKO cells. An embedded green fluorescent protein-tag was
used to visualize viral infection. As shown in Fig. 3A, RKO cells were successfully
infected with the recombinant lentiviruses after 72 h of
incubation. The mRNA level of 5-LOX in the OE-5-LOX group was
significantly higher than that in the OE-Vector group (Fig. 3B). As shown in Fig. 3C, the expression of 5-LOX protein in
the OE-Vector group remains at the basal level, consistent with the
inherently low endogenous 5-LOX expression in RKO cells. However,
the expression of 5-LOX protein in the OE-5-LOX group was
significantly higher than that in OE-Vector group. 5-LOX is a key
enzyme in leukotriene biosynthesis (25); as shown in Fig. S1, the overexpression of 5-LOX
significantly increased levels of LTB4 and
LTE4 in RKO cells.
Knockdown and overexpression of 5-LOX
impacts the migration and invasion of CRC cells
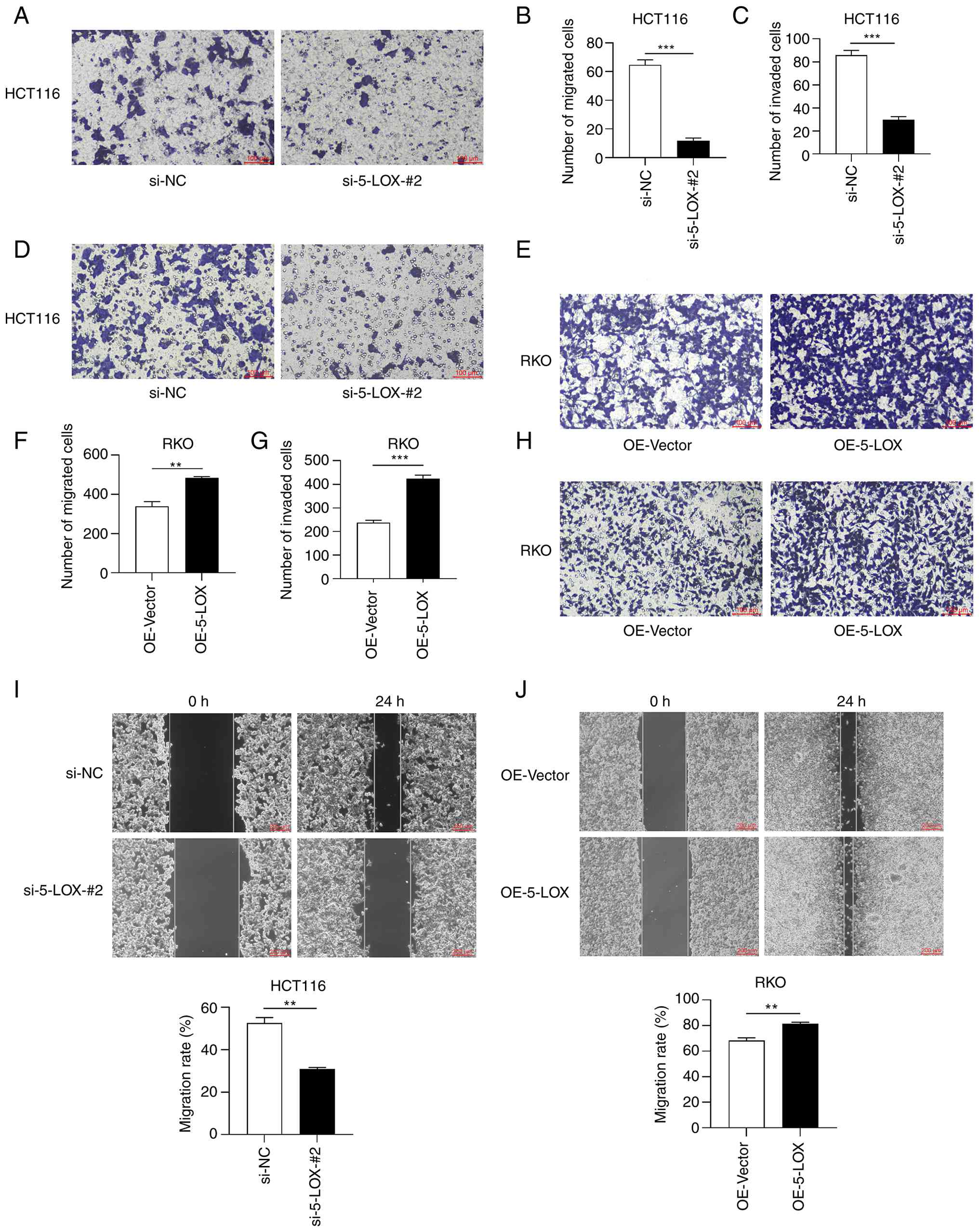
Transwell migration (Fig. 4A and B) and wound healing assays
(Fig. 4I) revealed that the
migration ability of HCT116 cells in the si-5-LOX-#2 group was
significantly reduced compared with the si-NC group. Transwell
invasion assay revealed that the si-5-LOX-#2 group had a
significantly lower number of invaded cells than the si-NC group
(Fig. 4C and D). However, the
migration ability of RKO cells in the OE-5-LOX group was
significantly higher than that in the OE-Vector group which was
determined by Transwell migration (Fig.
4E and F) and wound healing assays (Fig. 4J). RKO cells overexpressing 5-LOX
(the OE-5-LOX group) exhibited significantly higher invasion
(Fig. 4G and H) ability compared
with the cells infected with the control-lentiviruses (the
OE-Vector group).
Knockdown and overexpression of 5-LOX
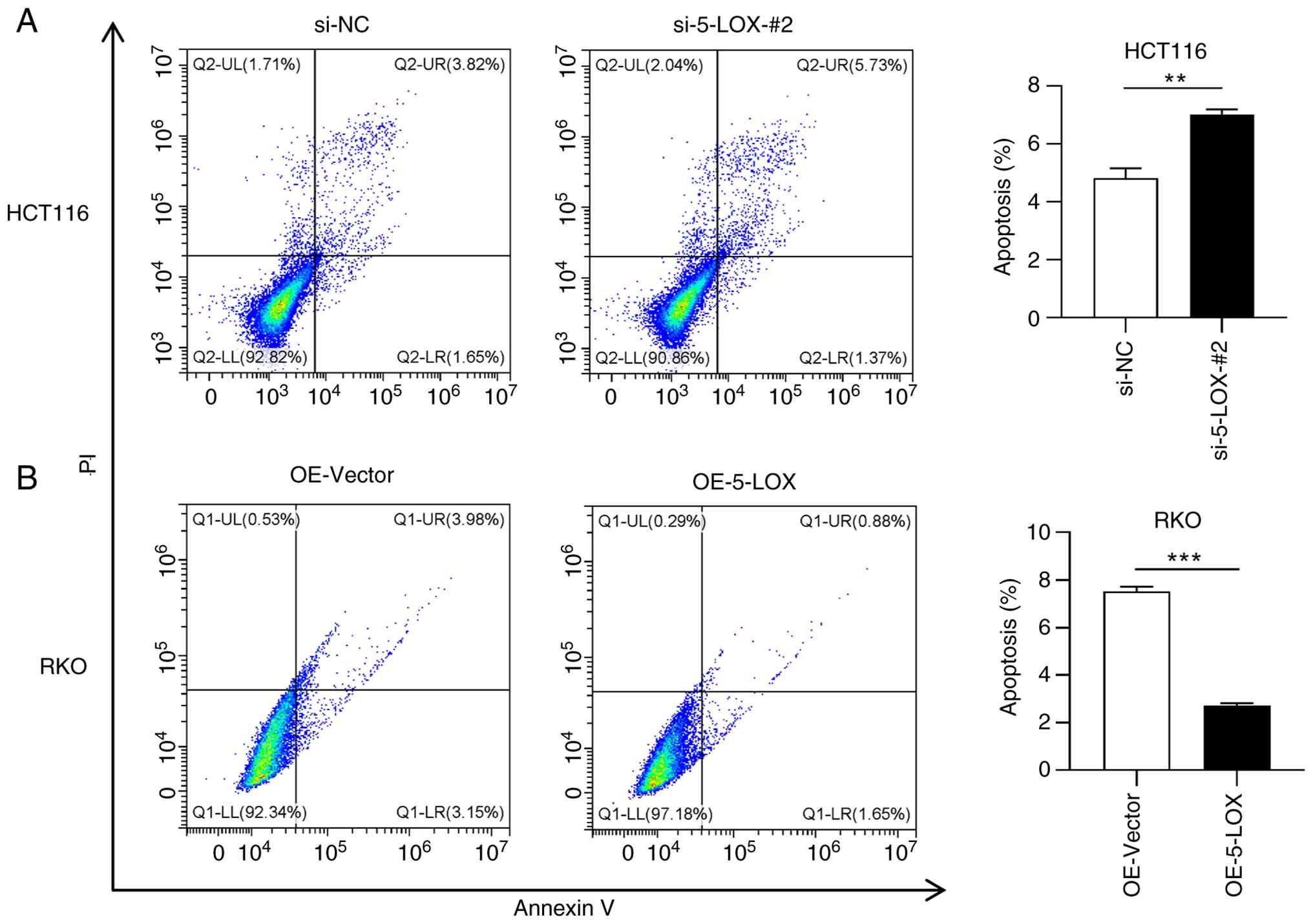
affects the apoptosis of CRC cells
As shown in Fig. 5A,
the apoptotic rate of 5-LOX-silenced HCT116 cells (the si-5-LOX-#2
group) was significantly increased compared with the cells
transfected with control siRNA (the si-NC group). Alternatively,
RKO cells overexpressing 5-LOX (the OE-5-LOX group) exhibited
significantly lower apoptotic rate in comparison to the OE-Vector
group (Fig. 5B).
Knockdown and overexpression of 5-LOX
regulates EMT
To evaluate the relationship between 5-LOX and EMT
in CRC cells, the expression of molecular markers for EMT were
analyzed by WB in 5-LOX-silenced CRC cells, 5-LOX-overexpressed CRC
cells and their corresponding control cells. WB analyses revealed
that 5-LOX knockdown significantly increased the expression of
E-cadherin and reduced the expression of N-cadherin, Snail and
Vimentin in HCT116 cells (Fig. 6A).
However, the overexpression of 5-LOX significantly downregulated
the expression of E-cadherin but upregulated the expression of
N-cadherin, Snail and Vimentin in RKO cells (Fig. 6B).
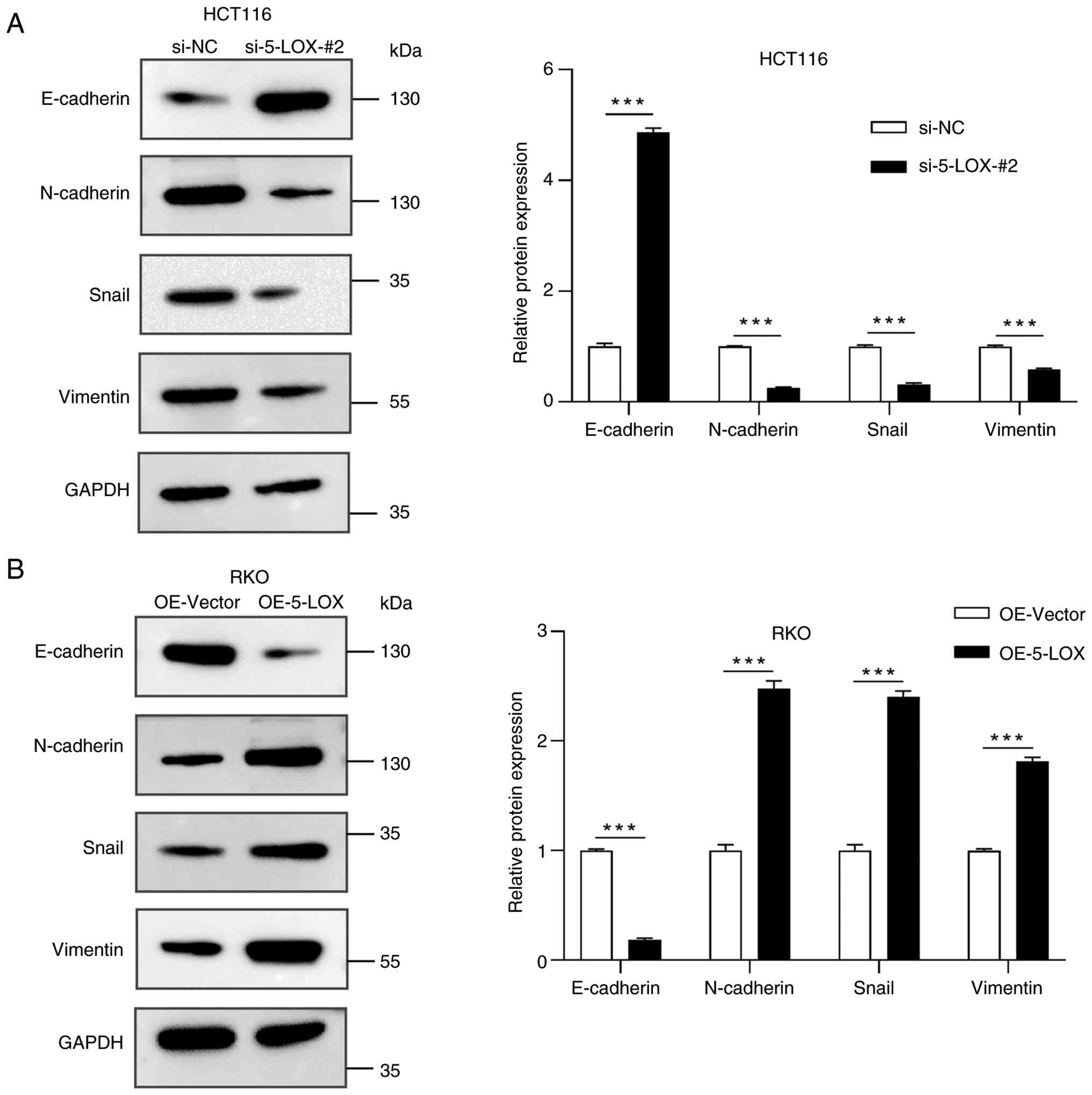 | Figure 6.Levels of EMT-associated proteins
regulated by 5-LOX knockdown and overexpression in CRC cells. (A)
The levels of EMT-associated proteins (E-cadherin, N-cadherin,
Snail and Vimentin) were determined by WB in siRNA-silenced HCT116
cells (si-NC and si-5-LOX-#2 groups). (B) The levels of
EMT-associated proteins were determined by WB in
lentivirus-overexpressed RKO cells (OE-Vector and OE-5-LOX groups).
Values are expressed as the mean ± standard error of the mean
(n=3). ***P<0.001. OE-Vector, cells infected with empty vector
control lentivirus; OE-5-LOX, cells infected with 5-LOX recombinant
lentivirus; siRNA, small interfering RNA; CRC, colorectal cancer;
5-LOX, 5-lipoxygenase; NC, negative control; WB, western blot; EMT,
epithelial-mesenchymal transition. |
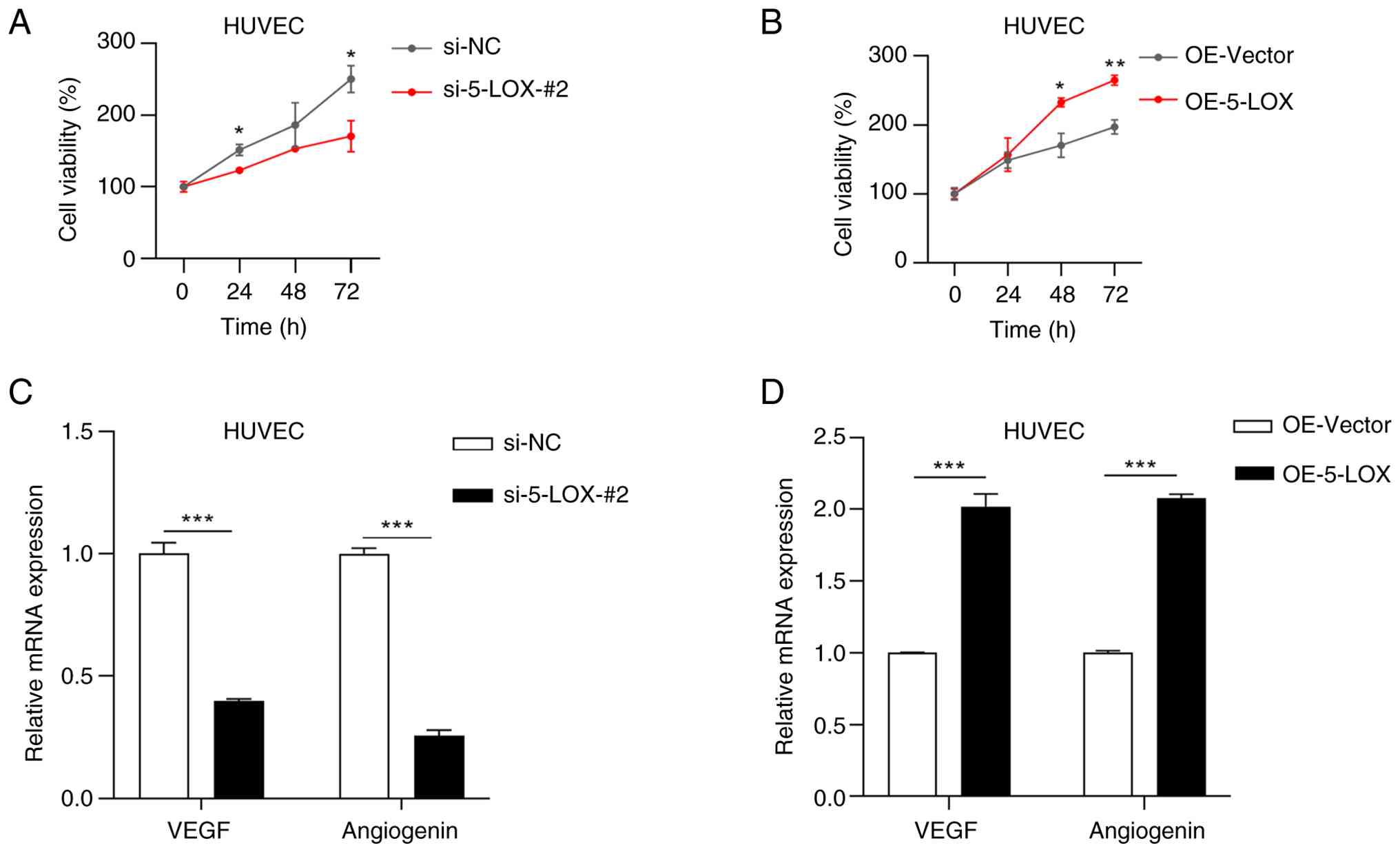
Effects of culture media from CRC
cells on HUVECs
To elucidate whether the products secreted by
5-LOX-silenced and 5-LOX-overexpressed CRC cells affect the
angiogenesis of HUVECs, HUVECs were treated with media from
5-LOX-silenced HCT116 cells, 5-LOX-overexpressed RKO cells and
their corresponding control cells in vitro. The CCK-8 assay
showed that cell viability of HUVECs was significantly suppressed
by treating with culture medium from the si-5-LOX-#2 group
(Fig. 7A), while cell viability of
HUVECs was significantly promoted by treating with culture medium
from the OE-5-LOX group (Fig. 7B).
The mRNA levels of VEGF and angiogenin were analyzed by RT-qPCR; as
shown in Fig. 7C, the mRNA levels
of VEGF and angiogenin were significantly lower in HUVECs which had
been treated with culture medium from the si-5-LOX-#2 group than in
HUVECs which had been treated with culture medium from the si-NC
group. However, the mRNA levels of VEGF and angiogenin were
significantly increased when HUVECs were treated with culture
medium from the OE-5-LOX group rather than with culture medium from
the OE-Vector group (Fig. 7D). The
expression of VEGF protein was significantly decreased in HUVECs
treated with conditioned media from 5-LOX silenced HCT116 cells but
significantly increased in HUVECs treated with conditioned media
from 5-LOX overexpressed RKO cells, compared with their respective
control groups (Fig. S2).
Discussion
5-LOX is upregulated in CRC and plays a notable role
in the development of other forms of cancer, including pancreatic
cancer (26), prostate
adenocarcinoma (27) and esophageal
adenocarcinoma (28). The
overexpression of 5-LOX gene which increase mitogenesis,
mutagenesis, angiogenesis, cell survival, immunosuppression and
metastasis in the inflammogenesis of CRC (29). In the present study, the role of
5-LOX in CRC was demonstrated by both silencing and overexpressing
5-LOX in CRC cells.
RNA interference is the process of
sequence-specific, post-transcriptional gene silencing in animals
and plants. siRNA-mediated gene silencing is an excellent tool for
studying gene function in mammalian cells and may eventually be
used for gene-specific therapeutics (30). The results of the present study
demonstrated that 5-LOX was widely expressed in both HT29 and
HCT116 CRC cell lines. Of the three siRNAs tested in the present
study, si-5-LOX-#2 exhibited notable ability to inhibit both the
mRNA and protein expression of 5-LOX in HCT116 cells. After
treating HT29 with si-5-LOX-#2, the same inhibitory effect was
observed, but the treated HT29 cells did not exhibit any migration
or invasion abilities (data not shown). Therefore, the HCT116 cell
line was considered as the best candidate for the 5-LOX knockdown
investigations. A previous study reported that the overexpression
of 5-LOX by lentivirus increased the invasion and migration
abilities of PANC-1 cells (31).
However, additional studies regarding the utilization of 5-LOX
lentivirus infection and the overexpression effects of 5-LOX in CRC
cells are needed. RKO cells with low mRNA and protein expression of
5-LOX were selected for lentiviral infection experiments. 5-LOX
lentiviral infection significantly increased the expression of
5-LOX, as determined by WB and RT-qPCR. The 5-LOX catalyzes
arachidonic acid to produce LTB4, LTD4 and
LTE4 (32), and in the
present study, the OE-5-LOX group exhibited significantly higher
levels of LTB4 and LTE4, which indicating the
increased 5-LOX enzymatic activity in RKO cells following 5-LOX
lentiviral infection.
CRC is a highly metastatic malignancy; therefore,
the impacts of 5-LOX knockdown and overexpression on the migration
and invasion of CRC cells were investigated. Downregulation of
5-LOX expression in HCT116 cells significantly decreased migration
and invasion capacities in vitro. However, the
overexpression of 5-LOX in RKO cells significantly upregulated
their migration and invasion capacities in vitro. In CRC,
EMT is a widely-known biological process that plays a marked role
in tumor progression, invasion and metastasis. Markers of EMT
(E-cadherin, N-cadherin, Vimentin and Snail) all acts as key
indicators of invasion and metastasis in CRC (33). In the present study, it was
demonstrated that the downregulation of 5-LOX expression in HCT116
cells inhibited the expression of N-cadherin, Vimentin, Snail;
however, improved the expression of E-cadherin. By contrast, high
levels of N-cadherin, Vimentin, Snail along with low level of
E-cadherin were detected in RKO cells with overexpression of 5-LOX.
Collectively, these results suggest that EMT may contribute to the
5-LOX-mediated invasion and migration of CRC cells. Moreover, the
silencing of 5-LOX significantly increased the apoptotic rate in
HCT116 cells, whereas the overexpression of 5-LOX significantly
decreased the apoptotic rate in RKO cells. However, the apoptotic
rates were relatively low, indicating a limited magnitude of
apoptosis induced by modulating 5-LOX expression. Although
siRNA-mediated 5-LOX knockdown and lentiviral 5-LOX overexpression
were used as complementary strategies in the present study,
re-overexpression of 5-LOX after knockdown requires further
validation in future work.
It was reported that F3, an inhibitor of 5-LOX,
effectively suppressed angiogenesis in endothelial cells (34). Angiogenesis in vivo usually
occurs in a complex microenvironment in which a variety of cells
interact together. Endothelial cells in the tumor microenvironment
serve a notable role in the development and progression of cancer
by regulating angiogenesis (35).
Previous research demonstrated that hypoxia (36) or tumor microenvironment stimuli
(37) could increase the VEGF
expression in HUVECs. Downregulation of angiogenin in HUVECs caused
a reduction in cell proliferation and angiogenesis (38). To understand the effect of the
microenvironment produced by CRC cells with or without the
expression of 5-LOX on HUVECs, HUVECs in vitro were treated
with culture media from CRC cells to simulate the physiological
environment in vivo. Conditioned media was collected from
5-LOX-silenced CRC cells, 5-LOX-overexpressed CRC cells and their
corresponding control cells and then incubated media with HUVECs.
The cell viability and mRNA levels of VEGF and angiogenin were
upregulated in HUVECs that had been incubated with culture medium
collected from 5-LOX-overexpressed CRC cells but downregulated in
HUVECs that had been incubated with culture medium collected from
5-LOX-silenced CRC cells. Consistent results were also observed for
VEGF protein expression in HUVECs. Conditioned medium collected
from CRC cells with 5-LOX overexpression stimulated the expression
of proangiogenic factors in HUVECs, indicating that the tumor
microenvironment in CRC cells overexpressing 5-LOX may be
angiogenically active. Further studies are needed to determine
whether 5-LOX can influence the tube formation of HUVECs in
vitro.
Taken together, the present study demonstrated that
the overexpression of 5-LOX in CRC cells promoted the migration and
invasion of CRC cells, whereas the silencing of 5-LOX inhibited the
migration and invasion of CRC cells. In addition, the knockdown but
not overexpression of 5-LOX in CRC cells inhibited EMT, which
consisted with the results of migration and invasion. Furthermore,
the mRNA levels of proangiogenic factors in HUVECs were upregulated
upon treatment with conditioned medium from 5-LOX-overexpressed CRC
cells but not medium from CRC cells in which 5-LOX had been
silenced. Collectively, the present findings suggest that 5-LOX
plays a notable role in promoting migration, invasion and EMT in
CRC cells, and that the tumor microenvironment of
5-LOX-overexpressed CRC cells may contribute to the angiogenesis in
HUVECs. Thus, 5-LOX represents a potential target for the migration
and invasion of CRC. Further investigation will focus on
establishing CRC xenograft models in nude mice with 5-LOX-knockout
and 5-LOX-overexpressing cell lines, to verify the biological
function of 5-LOX in CRC in vivo.
Supplementary Material
Supporting Data
Acknowledgements
Not applicable.
Funding
The present study was supported by the Central Guidance Fund for
Local Scientific and Technological Development (grant no.
2022ZY0161), the Public Hospital Research Joint Fund Technology
Project (grant no. 2024GLLH1008) and the Scientific Research
Projects of Higher Education Institutions in Inner Mongolia
Autonomous Region (grant no. NJZY23029).
Availability of data and materials
The data generated in the present study may be
requested from the corresponding author.
Authors' contributions
XuL, JH and XW designed and conceived the study.
XuL, JH, JZ and TS performed the experiments. XiL, CB and XG
analyzed data. XuL, TS and XW confirm the authenticity of all the
raw data. XuL wrote the manuscript. All authors read and approved
the final version of the manuscript.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Bray F, Laversanne M, Sung H, Ferlay J,
Siegel RL, Soerjomataram I and Jemal A: Global cancer statistics
2022: GLOBOCAN estimates of incidence and mortality worldwide for
36 cancers in 185 countries. CA Cancer J Clin. 74:229–263.
2024.PubMed/NCBI
|
|
2
|
Han B, Zheng R, Zeng H, Wang S, Sun K,
Chen R, Li L, Wei W and He J: Cancer incidence and mortality in
China, 2022. J Natl Cancer Cent. 4:47–53. 2024.PubMed/NCBI
|
|
3
|
Zhou H, Liu Z, Wang Y, Wen X, Amador EH,
Yuan L, Ran X, Xiong L, Ran Y, Chen W and Wen Y: Colorectal liver
metastasis: Molecular mechanism and interventional therapy. Signal
Transduct Target Ther. 7:702022. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Dekker E, Tanis PJ, Vleugels JLA, Kasi PM
and Wallace MB: Colorectal cancer. Lancet. 394:1467–1480. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Kalluri R and Neilson EG:
Epithelial-mesenchymal transition and its implications for
fibrosis. J Clin Invest. 112:1776–1784. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Kiemer AK, Takeuchi K and Quinlan MP:
Identification of genes involved in epithelial-mesenchymal
transition and tumor progression. Oncogene. 20:6679–6688. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Tsai JH and Yang J: Epithelial-mesenchymal
plasticity in carcinoma metastasis. Genes Dev. 27:2192–2206. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Lee JM, Dedhar S, Kalluri R and Thompson
EW: The epithelial-mesenchymal transition: New insights in
signaling, development, and disease. J Cell Biol. 172:973–981.
2006. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Shin AE, Giancotti FG and Rustgi AK:
Metastatic colorectal cancer: Mechanisms and emerging therapeutics.
Trends Pharmacol Sci. 44:222–236. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Lu J, Kornmann M and Traub B: Role of
epithelial to mesenchymal transition in colorectal cancer. Int J
Mol Sci. 24:148152023. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Vu T and Datta P: Regulation of EMT in
colorectal cancer: A culprit in metastasis. Cancers. 9:1712017.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Francí C, Gallén M, Alameda F, Baró T,
Iglesias M, Virtanen I and García de Herreros A: Snail1 protein in
the stroma as a new putative prognosis marker for colon tumours.
PLoS One. 4:e55952009. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Yan X, Yan L, Liu S, Shan Z, Tian Y and
Jin Z: N-cadherin, a novel prognostic biomarker, drives malignant
progression of colorectal cancer. Mol Med Rep. 12:2999–3006. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Du L, Li J, Lei L, He H, Chen E, Dong J
and Yang J: High vimentin expression predicts a poor prognosis and
progression in colorectal cancer: A study with meta-analysis and
TCGA database. Biomed Res Int. 2018:1–14. 2018. View Article : Google Scholar
|
|
15
|
Mashima R and Okuyama T: The role of
lipoxygenases in pathophysiology; New insights and future
perspectives. Redox Biol. 6:297–310. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Wang Y, Wang W, Sanidad KZ, Shih PA, Zhao
X and Zhang G: Eicosanoid signaling in carcinogenesis of colorectal
cancer. Cancer Metastasis Rev. 37:257–267. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Soumaoro LT, Iida S, Uetake H, Ishiguro M,
Takagi Y, Higuchi T, Yasuno M, Enomoto M and Sugihara K: Expression
of 5-lipoxygenase in human colorectal cancer. World J
Gastroenterol. 12:6355–6360. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Melstrom LG, Bentrem DJ, Salabat MR,
Kennedy TJ, Ding XZ, Strouch M, Rao SM, Witt RC, Ternent CA,
Talamonti MS, et al: Overexpression of 5-lipoxygenase in colon
polyps and cancer and the effect of 5-LOX inhibitors in vitro and
in a murine model. Clin Cancer Res. 14:6525–6530. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Chang J, Tang N, Fang Q, Zhu K, Liu L,
Xiong X, Zhu Z, Zhang B, Zhang M and Tao J: Inhibition of COX-2 and
5-LOX regulates the progression of colorectal cancer by promoting
PTEN and suppressing PI3K/AKT pathway. Biochem Biophys Res Commun.
517:1–7. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Bošković J, Dobričić V, Keta O, Korićanac
L, Žakula J, Dinić J, Jovanović Stojanov S, Pavić A and Čudina O:
Unveiling anticancer potential of COX-2 and 5-LOX inhibitors:
Cytotoxicity, radiosensitization potential and antimigratory
activity against colorectal and pancreatic carcinoma.
Pharmaceutics. 16:8262024. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Yang Z, Zhang X, Bai X, Xi X, Liu W and
Zhong W: Anti-angiogenesis in colorectal cancer therapy. Cancer
Sci. 115:734–751. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Tello-Montoliu A, Patel JV and Lip GY:
Angiogenin: A review of the pathophysiology and potential clinical
applications. J Thromb Haemost. 4:1864–1874. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Carmeliet P: VEGF as a key mediator of
angiogenesis in cancer. Oncology. 69:4–10. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Arocho A, Chen B, Ladanyi M and Pan Q:
Validation of the 2-DeltaDeltaCt calculation as an alternate method
of data analysis for quantitative PCR of BCR-ABL P210 transcripts.
Diagn Mol Pathol. 15:56–61. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Rådmark O, Werz O, Steinhilber D and
Samuelsson B: 5-Lipoxygenase, a key enzyme for leukotriene
biosynthesis in health and disease. Biochim Biophys Acta.
1851:331–339. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Hennig R, Ding XZ, Tong WG, Schneider MB,
Standop J, Friess H, Büchler MW, Pour PM and Adrian TE:
5-Lipoxygenase and leukotriene B(4) receptor are expressed in human
pancreatic cancers but not in pancreatic ducts in normal tissue. Am
J Pathol. 161:421–428. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Gupta S, Srivastava M, Ahmad N, Sakamoto
K, Bostwick DG and Mukhtar H: Lipoxygenase-5 is overexpressed in
prostate adenocarcinoma. Cancer. 91:737–743. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Chen X, Wang S, Wu N, Sood S, Wang P, Jin
Z, Beer DG, Giordano TJ, Lin Y, Shih WC, et al: Overexpression of
5-lipoxygenase in rat and human esophageal adenocarcinoma and
inhibitory effects of zileuton and celecoxib on carcinogenesis.
Clin Cancer Res. 10:6703–6709. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Kennedy BM and Harris RE: Cyclooxygenase
and lipoxygenase gene expression in the inflammogenesis of
colorectal cancer: Correlated expression of EGFR, JAK STAT and Src
genes, and a natural antisense transcript, RP11-C67.2.2. Cancers.
15:23802023. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Elbashir SM, Harborth J, Lendeckel W,
Yalcin A, Weber K and Tuschl T: Duplexes of 21-nucleotide RNAs
mediate RNA interference in cultured mammalian cells. Nature.
411:494–498. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Hu WM, Liu SQ, Zhu KF, Li W, Yang ZJ, Yang
Q, Zhu ZC and Chang J: The ALOX5 inhibitor Zileuton regulates
tumor-associated macrophage M2 polarization by JAK/STAT and
inhibits pancreatic cancer invasion and metastasis. Int
Immunopharmacol. 121:1105052023. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Steinhilber D, Fischer AS, Metzner J,
Steinbrink SD, Roos J, Ruthardt M and Maier TJ: 5-Lipoxygenase:
Underappreciated role of a pro-inflammatory enzyme in
tumorigenesis. Front Pharmacol. 1:1432010. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Zhang N, Ng AS, Cai S, Li Q, Yang L and
Kerr D: Novel therapeutic strategies: Targeting
epithelial-mesenchymal transition in colorectal cancer. Lancet
Oncol. 22:e358–e368. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Kim TY, Kim J, Choo HY and Kwon HJ:
Inhibition of 5-lipoxygenase suppresses vascular endothelial growth
factor-induced angiogenesis in endothelial cells. Biochem Biophys
Res Commun. 478:1117–1122. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Alvarez-García V, González A,
Alonso-González C, Martínez-Campa C and Cos S: Antiangiogenic
effects of melatonin in endothelial cell cultures. Microvasc Res.
87:25–33. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Cheng J, Yang HL, Gu CJ, Liu YK, Shao J,
Zhu R, He YY, Zhu XY and Li MQ: Melatonin restricts the viability
and angiogenesis of vascular endothelial cells by suppressing
HIF-1α/ROS/VEGF. Int J Mol Med. 43:945–955. 2019.PubMed/NCBI
|
|
37
|
Gacche RN and Meshram RJ: Targeting tumor
micro-environment for design and development of novel
anti-angiogenic agents arresting tumor growth. Prog Biophys Mol
Bio. 113:333–354. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Kishimoto K, Liu S, Tsuji T, Olson KA and
Hu GF: Endogenous angiogenin in endothelial cells is a general
requirement for cell proliferation and angiogenesis. Oncogene.
24:445–456. 2005. View Article : Google Scholar : PubMed/NCBI
|