Introduction
Mutations in the v-Ki-ras2 Kirsten rat sarcoma viral
oncogene homolog gene (KRAS) are important genetic testing
indicators for advanced-stage non-small cell lung cancer (NSCLC),
occurring in 8–10% of Asian patients with lung adenocarcinoma
(LUAD) (1). Approximately half of
KRAS-mutated NSCLCs display co-occurring genomic
alterations, mainly in cyclin dependent kinase inhibitor 2A,
PI3KA, TP53, AKT1, serine/threonine kinase 11
(STK11), kelch like ECH associated protein 1 (KEAP1)
and BRAF (2), which are
associated with an invasive phenotype, resistance to
chemoradiotherapy and poorer clinical outcomes (3). Advancements in targeting the mutant
KRAS G12C protein in LUAD include the development of sotorasib (AMG
510) and adagrasib (MRTX849), which have shown promising results
(4,5); however, current clinical guidelines
still recommend KRAS G12C inhibitors as second-line
treatments (6).
Trophoblast cell-surface antigen 2 (TROP-2) has
potential as a novel therapeutic target in lung cancer (7). High TROP-2 expression has been
observed in several human tumors, including lung, colorectal and
breast cancer, and is a negative prognostic factor in solid cancers
(8–10). TROP-2 upregulation has been detected
in 60% of squamous lung cell carcinomas, 40–60% of adenocarcinomas
and 20% of high-grade pulmonary neuroendocrine neoplasms (11,12).
TROP-2 may be considered a useful marker of poor cancer prognosis
(13). TROP-2-targeted
antibody-drug conjugates (ADCs) have demonstrated efficacy in
clinical trials of multiple types of solid cancer, such as advanced
urothelial cancer and breast cancer (14,15).
The current understanding and effective treatment of
lung cancer harboring KRAS mutation and TROP-2 upregulation
remain inadequate, and NSCLCs with both the KRAS G12C
mutation and TROP-2 upregulation have not, to the best of our
knowledge, been reported.
The present report describes a patient with advanced
LUAD who achieved favorable therapeutic outcomes with
TROP-2-targeted ADC therapy following resistance to radiotherapy,
chemotherapy and immunotherapy, without serious adverse
reactions.
Case report
A 65-year-old man, a smoker with a past medical
history of coronary artery disease and hypertension, presented to
Huangshi Hospital of Chinese Traditional Medicine (Huangshi, China)
in February 2022 with intermittent chest tightness and pain lasting
2 weeks. The patient was transferred to Hubei Cancer Hospital
(Wuhan, China) in March 2022.
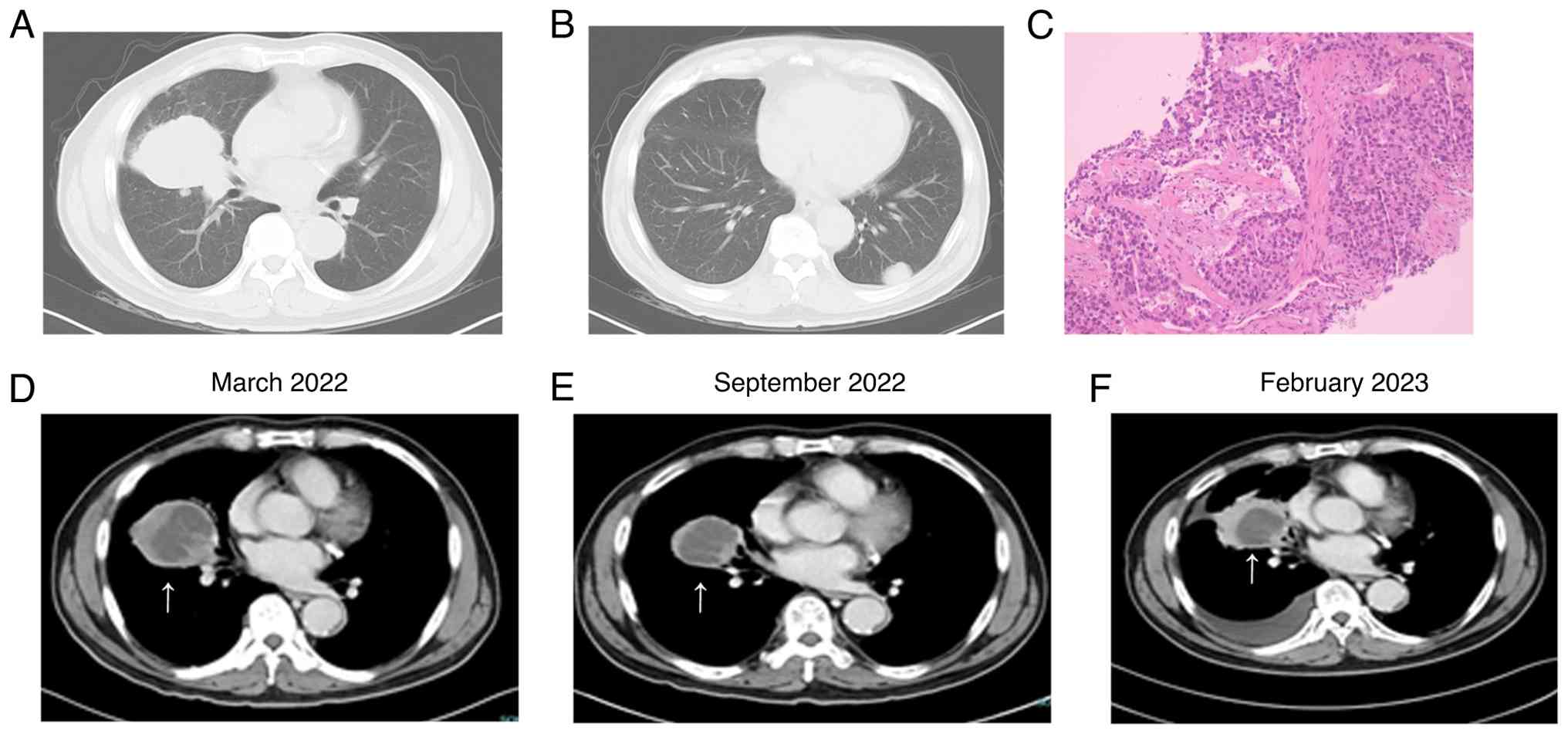
The chest computed tomography (CT) revealed a
primary malignancy in the middle lobe of the right lung (~6.7×5.7
cm) (Fig. 1A), accompanied by a
small amount of obstructive inflammation; multiple enlarged lymph
nodes in mediastinal groups 2, 4 and 7, and in the right pulmonary
hilum; and multiple metastases in both lungs and bilateral pleura
(largest measuring ~2.7×1.7 cm) (Fig.
1B). No evidence of metastasis was found elsewhere in the body.
Adenocarcinoma was diagnosed by CT-guided needle biopsy in March
2022 (Fig. 1C).
Next-generation sequencing performed in Huangshi
Hospital of Chinese Traditional Medicine revealed a KRAS
G12C mutation and a programmed cell death ligand 1 (PD-L1) tumor
proportion score of <1%, without other co-mutations. The patient
was diagnosed with right LUAD with multiple metastases to both
lungs [stage IV, cT2N0M1 according to TNM Classification and Union
for International Cancer Control (16)] harboring a KRAS G12C
mutation.
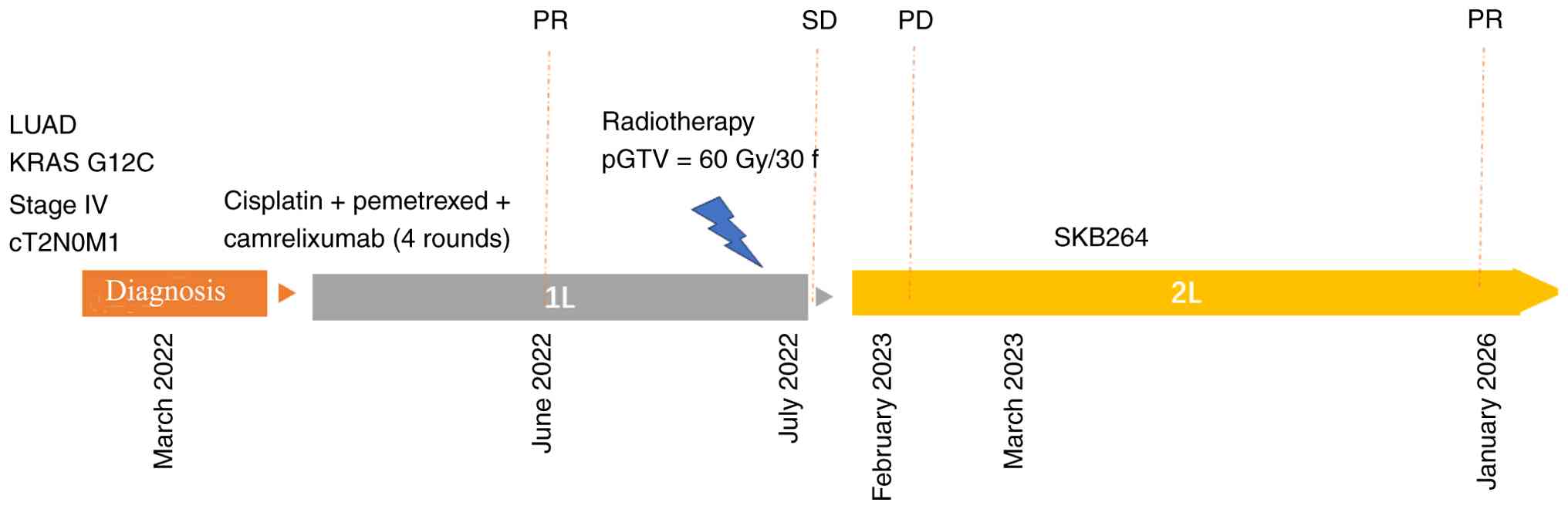
For patients with advanced NSCLC harboring
KRAS mutations, first-line therapy consists of
platinum-based dual-drug chemotherapy combined with antivascular
treatment or immunotherapy (17).
The current patient received chemotherapy (cisplatin/pemetrexed)
every 3 weeks for four cycles combined with immunotherapy
(camrelizumab). Cisplatin (25 mg/m2) was administered
for 3 days, and intravenous pemetrexed (500 mg/m2) and
camrelizumab (200 mg) were administered on day 1 of each cycle from
March 2022 to June 2022. The patient did not receive immune
maintenance therapy due to financial constraints. Evaluation
indicated partial responseon CT scan after the fourth treatment
cycle (Fig. 1D and E) based on the
Response Evaluation Criteria in Solid Tumors version 1.1 (RECIST
1.1) (18). Due to gastrointestinal
adverse effects, the patient refused to continue chemotherapy
beyond June 2022.
The addition of thoracic radiotherapy in patients
with advanced NSCLC can improve overall survival (OS) (19). To consolidate the curative effect,
chest radiotherapy was proposed in July 2022 (gross tumor volume,
60 Gy/30 F; planning target volume, 54 Gy/30 F). Follow-up CT scans
were performed every 6 weeks after radiotherapy. The patient did
not receive antitumor treatment after September 2022 and suffered
disease progression in February 2023 (half a year later).
Malignant pleural effusion and tumor growth were
observed (Fig. 1D-F). Sotorasib and
adagrasib have been approved as second-line therapies for adults
with NSCLC harboring the KRAS G12C mutation (20). The patient could not access
KRAS G12C inhibitors due to financial hardship and was
counselled for enrolment in a phase II clinical trial (KLUS Pharma,
Inc., program no. KL264-01; trial ID NCT04152499) involving the
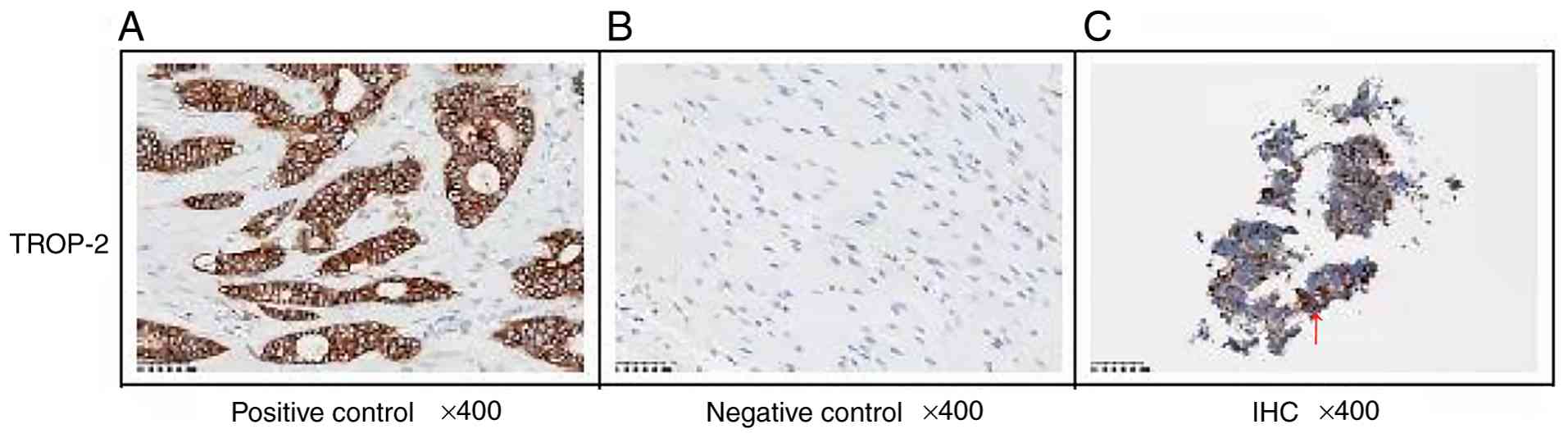
TROP-2 ADC SKB264. Standard methods were used for
immunohistochemistry (performed at the Central Laboratory of MEDx
Translational Medicine Co., Ltd.) and the evaluation of TROP-2
expression using the H-score. TROP-2 expression was observed under
the microscope by a pathologist after staining and was evaluated
according to the coloring ratio and depth. The H-score was
interpreted as follows: 0 (negative), no membrane staining of
arbitrary strength; 1+ (weakly positive), tumor cells exhibit weak
cell membrane staining; 2+ (moderately positive), tumor cells
exhibit moderate membrane staining; and 3+ (strongly positive),
tumor cells exhibit strong membrane staining. H-score=[1× (% cells
1+) + 2× (% cells 2+) + 3× (% cells 3+)] ×100. TROP-2 expression
was defined as: Weakly positive, H-score ≤100; moderately positive,
H-score ≤200; and strongly positive, H-score ≤300. Finally, TROP-2
upregulation was confirmed at the 2+ level by immunohistochemistry
(Fig. 2). The patient was
administered SKB264 (10 mg/kg, day 1 of 14-day cycles) beginning in
March 2023. The first follow-up CT scan was performed within 3
months of treatment initiation and is ongoing.
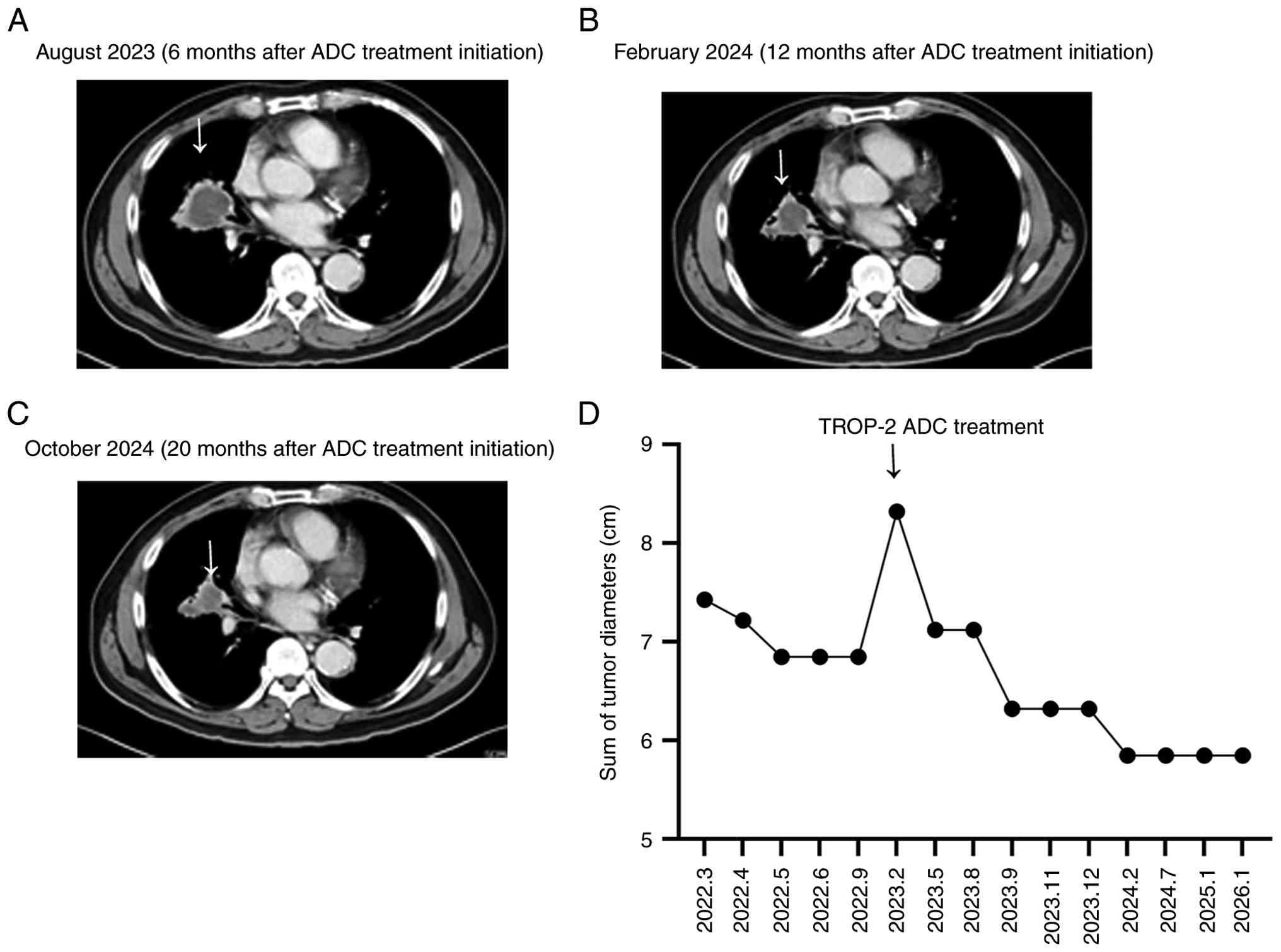
At the time of writing this report, the clinical
response of the patient has persisted for 34 months (January 2026)
after treatment initiation. The patient suffered disease
progression in February 2023 after first-line treatment (without
antitumor treatment from September 2022). There was a 42% reduction
in tumor size from the baseline (February 2023), as measured based
on RECIST 1.1 (Fig. 3). The
therapeutic course is shown in Fig.
4. The patient experienced Grade I anemia (21) in March 2023 without obvious symptoms
of ischemia, such as dizziness or fatigue, and intermittently took
oral iron supplements. In addition, the patient experienced
intermittent Grade I decreased appetite after June 2023 and
received appropriate nutritional supplementation. No other serious
adverse events were observed.
Discussion
The present case highlights the clinical potential
of ADCs targeting TROP-2 in NSCLC harboring a KRAS G12C
mutation. TROP-2 upregulation was detected, and the patient with
the KRAS G12C mutation was enrolled in a phase II clinical
trial. KRAS inhibitors were not selected due to financial
reasons. After multiline therapy, the patient benefited from a
novel ADC targeting TROP-2 (SKB264). The patient responded well to
SKB264 treatment and achieved good outcomes. Follow-up CT imaging
showed a 42% reduction in target lesions over 34 months and no
serious adverse events were observed. The survival of the patient
exceeded expectations based on published clinical trial data
(22), providing novel insights
into potential strategies for the treatment of NSCLC with
KRAS G12C mutations.
KRAS is one of the most frequently mutated
oncogenes in solid tumors. KRAS mutations occur in 20–30% of
lung cancers, with the G12C variant accounting for 35–45% of these
(23,24). Although the prevalence is lower in
Asian populations (25), nearly 3%
of Chinese patients with NSCLC harbor the KRAS G12C mutation
(26), with a higher incidence in
male smokers (27). Approximately
half of NSCLCs with KRAS mutations display co-occurring
genomic alterations (mainly in TP53, STK11 and
KEAP1). The co-occurring genomic mutations may be associated
with an invasive phenotype, resistance to chemoradiotherapy and a
worse clinical outcome (28). Based
on existing mechanistic study, TROP-2 is involved in the regulation
of several tumor signaling pathways, including the PI3K-AKT,
B-Raf-ERK, Wnt/β-catenin and other pathways (29). Genes such as TP53, STK11 and
KEAP1 are also directly or indirectly implicated. Although
abnormal expression of these genes often suggests poor prognosis or
resistance to current treatment models, targeting of TROP-2 could
serve a multi-pronged role in therapy (30). In addition, genes such as STK11,
KEAP1 and TP53, as well as biomarkers such as PD-L1 and
tumor mutation burden, are currently widely reported for predicting
the efficacy of immunotherapy (31). However, to the best of our
knowledge, whether these co-mutations or biomarkers will impact the
efficacy of ADCs is still unknown. This remains a key issue that
needs to be addressed when ADC drugs are widely used in clinical
practice, both now and in the future.
The National Comprehensive Cancer Network guidelines
(V3. 2022) recommend platinum-based chemotherapy, with or without
immunotherapy, as the first-line treatment for metastatic NSCLC
with KRAS mutations (32).
Notably, KRAS mutations may upregulate PD-L1 expression,
contributing to immune resistance (30,33),
reducing CD8+ T cell infiltration and lowering tumor
mutational burden factors that contribute to reduced benefit from
immune checkpoint inhibitors (34).
However, to the best of our knowledge, no differences have been
observed between the outcomes of patients treated with
immunotherapy or immunotherapy plus chemotherapy, with and without
KRAS mutations. A retrospective analysis of the KEYNOTE-042
trial suggests that immune checkpoint inhibitors may improve the
prognosis of KRAS-mutant NSCLC (35). According to the Flatiron database,
patients with KRAS mutations can benefit from immunotherapy
or chemoimmunotherapy; however, those with wild-type tend to
benefit from combination therapy (36).
The current patient experienced disease progression
within ~1 year of first-line chemoimmunotherapy, which is in line
with the estimated poor prognosis for KRAS G12C mutation in
patients with NSCLC. First-line chemoimmunotherapy for metastatic
NSCLC with KRAS G12C mutation results in a median
progression-free survival (PFS) and OS of 7.9 and 18.1 months,
respectively (37).
Drugs targeting KRAS G12C have been shown to
prolong the survival of patients with metastatic disease. The
multicenter phase I/II open-label CodeBreaK 100 trial reported that
the disease control rate (DCR) and objective response rate (ORR) of
sotorasib in patients with advanced and metastatic NSCLC were 38.1
and 32.2%, respectively, with tolerable toxicity (38). Compared with docetaxel, sotorasib
extended the median PFS time by 1 month (5.6 vs. 4.5 months)
(39), and received approval as the
first KRAS G12C inhibitor by the United States Food and Drug
Administration in May 2021 for the treatment of advanced NSCLC.
Similarly, in the KRYSTAL-12 study, adagrasib achieved a longer
median PFS time in patients with pretreated KRAS
G12C-mutated NSCLC than chemotherapy (5.5 vs. 3.8 months; hazard
ratio, 0.58; P<0.0001). Additionally, adagrasib demonstrated
intracranial efficacy in patients with brain metastases, with an
ORR more than twice that in the docetaxel group (24 vs. 11%)
(40). Adagrasib (trade name,
Krazati) was approved in the European Union on January 10, 2024, as
a treatment for pretreated patients with KRAS G12C-mutated
NSCLC (41). NCT05005234, a
registered phase II clinical study led by Professor Wu Yilong,
demonstrated the safety and efficacy of fluzelese monotherapy in
the treatment of advanced KRAS G12C-mutant NSCLC. Fluzelese
exhibited a DCR of 90% (95% CI, 84–95%), ORR of 49% (95% CI,
40–59%) and 12-month durable response rate of 54% (95% CI, 38–67%)
(42). Multiple phase III clinical
trials (Table I) are currently
underway to assess the effectiveness of drugs against KRAS
G12C-mutant NSCLC. Despite advances, the prognosis of patients with
KRAS G12C mutations remains poor. The development of novel
treatment models is particularly important for improving patient
prognosis.
 | Table I.Ongoing phase III trials
investigating patients with KRAS G12C-mutant lung cancer
(https://clinicaltrials.gov/; accessed
on January 30, 2025). |
Table I.
Ongoing phase III trials
investigating patients with KRAS G12C-mutant lung cancer
(https://clinicaltrials.gov/; accessed
on January 30, 2025).
| Clinical trial
ID | Phase | Setting | Total patients,
n | Treatment arms | Primary
endpoint |
|---|
| NCT06300177 | III | KRAS G12C
mutation-positive locally advanced or metastatic NSCLC after prior
standard therapy failure | 522 | D-1553 vs.
docetaxel | PFS |
| CT06497556 | III | Pretreated KRAS
G12C mutation-positive advanced or metastatic NSCLC | 320 | Divarasib vs.
sotorasib/adagrasib | PFS |
| NCT04685135 | III | Pretreated KRAS
G12C mutation-positive NSCLC patients | 453 | MRTX849 vs.
docetaxel | PFS |
| NCT02152631 | III | Patients with stage
IV NSCLC with a detectable KRAS mutation who have progressed after
platinum-based chemotherapy | 453 | Abemaciclib vs.
erlotinib | OS |
| NCT05132075 | III | Locally advanced or
metastatic KRAS G12C-mutant NSCLC | 95 | JDQ443 vs.
docetaxel | PFS |
| NCT06345729 | III | KRAS G12C-mutant,
metastatic NSCLC with PD-L1 TPS ≥50% | 600 | MK-1084 +
pembrolizumab vs. pembrolizumab | PFS |
| NCT04613596 | II/III | Advanced NSCLC with
KRAS G12C mutation | 806 | II: Adagrasib +
pembrolizumab III: Adagrasib + pembrolizumab vs. pembrolizumab | II: ORR III: PFS
OS |
| NCT06416410 | III | Advanced
non-squamous NSCLC with KRAS p.G12C mutation | 392 | JAB-21822 +
JAB-3312 vs. tislelizumab + pemetrexed + carboplatin | PFS |
| NCT01933932 | III | Locally advanced or
metastatic NSCLC (stage IIIb-IV) | 510 | Selumetinib +
docetaxel vs. placebo + docetaxel | PFS |
| NCT06119581 | III | NSCLC with KRAS
G12C mutation | 1,016 | LY3537982 +
standard therapy vs. standard therapy | TEAE, PFS |
| NCT05920356 | III | Stage IV or
advanced stage IIIB/C non-squamous NSCLC, negative for PD-L1 and
positive for KRAS p.G12C | 750 | Sotorasib +
platinum doublet chemotherapy vs. pembrolizumab + platinum doublet
chemotherapy | PFS |
| NCT06335355 | III | Patients with
advanced or metastatic NSCLC with STK11/KEAP1/KRAS mutations | 401 | Adebrelimab +
SHR-8068 + pemetrexed + carboplatin/camrelizumab + pemetrexed +
carboplatin vs. adebrelimab + pemetrexed + carboplatin | ORR, PFS |
| NCT06008093 | III | Patients with
metastatic NSCLC with non-squamous histology who have mutations
and/or co-mutations in STK11, KEAP1 or KRAS | 280 | Durvalumab +
tremelimumab + chemotherapy vs. pembrolizumab + chemotherapy | OS |
| NCT04303780 | III | Patients with NSCLC
with KRAS p.G12c mutation | 345 | AMG 510 vs.
docetaxel | PFS |
ADCs have emerged as promising therapies, combining
targeted monoclonal antibodies with cytotoxic agents to enhance
tumor-specific drug delivery and minimize systemic toxicity
(43). TROP-2, a type I
transmembrane glycoprotein, is upregulated in several malignancies
(44). Although its upregulation
mechanism is not fully understood, its role in cancer cell
proliferation, invasion and metastasis is well documented (10,45).
To the best of our knowledge, no research has explored the direct
relationship between TROP-2 and KRAS G12C. TROP-2 mainly
promotes the growth, proliferation and metastasis of tumor cells by
regulating various cellular signaling pathways, including the
calcium ion, MAPK/ERK and PI3K/AKT signaling pathways (27). The association between KRAS
mutations and TROP-2 expression has only been reported in
colorectal and pancreatic cancer (46). TROP-2 expression is generally
upregulated in patients with KRAS mutations compared with
those with wild-type KRAS, suggesting the therapeutic
potential of targeting TROP-2 in advanced tumors with KRAS
mutations (45).
TROP-2 activates MAPK signaling, ERK1/2
phosphorylation and transcription factor activator protein 1, and
may drive epithelial-mesenchymal transition through podoplanin
(47). TROP-2 upregulation is
associated with poorer differentiation, increased lymph node
metastasis and worse survival across multiple cancer types,
including NSCLC, gastric cancer and ovarian cancer (48–50).
High TROP-2 expression in NSCLC has been associated with shorter
survival, making it a compelling therapeutic target.
Several TROP-2-directed ADCs have demonstrated
clinical potential. Trodelvy (sacituzumab govitecan-hziy) is an ADC
developed by Gilead Sciences, Inc., in the United States. The
results of the IMMU-132-01 study showed reduced diameters in tumors
from different histological sources after gosatuzumab treatment,
confirming that TROP-2 is a broad target for various solid tumors
(51). According to the latest
released follow-up data, Trodelvy exhibited antitumor activity and
manageable safety in patients with SCLC in the extensive stage
(TROPiCS-03 study) (52). Dato-DXd,
an ADC combining a TROP-2 IgG1 monoclonal antibody and a Topo I
inhibitor (DXd), inhibited NSCLC growth in xenograft models
(53), and improved PFS compared
with chemotherapy (4.4 vs. 3.7 months) in the TROPION Lung 01
study. A benefit was seen across a range of TROP-2 expression
levels, although heterogeneity, downregulation or structural
changes may contribute to resistance (54). Other clinical trials (Table II) are currently underway to assess
the effectiveness of TROP-2 ADCs in NSCLC.
 | Table II.Ongoing trials involving anti-TROP-2
ADCs in lung cancer (https://clinicaltrials.gov/; accessed on October 30,
2024). |
Table II.
Ongoing trials involving anti-TROP-2
ADCs in lung cancer (https://clinicaltrials.gov/; accessed on October 30,
2024).
| Clinical trial
ID | Phase | Setting | N | Treatment arms | Primary
endpoint |
|---|
| NCT04152499 | I/II | Patients with
locally advanced unresectable/metastatic solid tumors who are
refractory to available standard therapies | 1,300 | SKB264 | ORR |
| NCT05941507 | II | Patients with
advanced solid tumors refractory to the standard of care, or for
whom no standard of care exists | 300 | LCB84 vs. LCB84 +
anti-PD-1 | ORR, DOR, PFS,
OS |
| NCT05460273
(TROPION-PanTumor02) | I/II | Advanced NSCLC | 119 | Dato-DXd | ORR |
| NCT04940325
(ICARUS-LUNG01) | II | Advanced and/or
unresectable NSCLC | 100 | DS-1062a | ORR |
| NCT06074588
(MK-2870-004) | III | Previously treated
NSCLC with EGFR mutations | 556 | SKB264 vs.
docetaxel/pemetrexed | PFS and OS |
| NCT06480136 | II | NSCLC with
previously treated | 32 | SHR-A1921 +
adebrelimab | ORR |
| NCT05609968
(KEYNOTE D46/EVOKE-03) | III | Metastatic NSCLC
with PD-L1 TPS ≥50% | 614 | Pembrolizumab +
sacituzumab govitecan vs. pembrolizumab | PFS, OS |
| NCT05687266
(AVANZAR) | III | Locally advanced or
metastatic NSCLC without actionable genomic alterations | 1,280 | Dato-DXd +
durvalumab + carboplatin vs. durvalumab + carboplatin +
cisplatin/pemetrexed/paclitaxel | PFS, OS |
| NCT06431633
(ARIAN) | II | Patients with IB to
IIIA and IIIB (N2) NSCLC who did not achieve pCR after neoadjuvant
treatment | 129 | Control vs.
zimberelimab vs. sacituzumab govitecan + zimberelimab | DFS |
Several questions remain unanswered. A phase III
clinical trial (EVOKE-01 study) failed to show that Trodelvy was
more effective than docetaxel as a second-line treatment for NSCLC
(55). It is critical to identify
the population that could benefit from TROP-2 ADC treatment. Based
on analysis of previous research, patients with EGFR mutations may
benefit more from TROP-2 ADC treatment after failure of first-line
TKI therapy (56). In addition,
patients who do not respond to anti-PD-L1 have also been shown to
benefit from TROP-2 ADC therapy (22). Therefore, the role of ADCs in lung
cancer should be comprehensively re-examined. In particular,
efficacy-predictive biomarkers are required. Positivity for TROP-2
calculated using quantitative continuous scoring effectively
predicted outcomes after Dato-DXd treatment in the TROPION Lung 01
study (57). High TROP-2 expression
may have contributed to the survival benefits in the current
patient. Whether KRAS G12C affects the efficacy of TROP-2
ADC therapy in patients requires further study. Although the
EVOKE-01 clinical study evaluated data from 14 cases with
KRAS G12C mutations, the design did not include subgroup
analysis (55). Research on the
association between TROP-2 and the activation mechanism of
KRAS mutations is still limited. However, from the results
of the aforementioned clinical and basic studies that have been
published, it can be roughly inferred that upregulation of TROP-2
likely serves an important role in the occurrence and development
of KRAS-mutant solid tumors, including NSCLC.
The present case suggests that TROP-2-targeted ADCs
may represent a viable therapeutic option for patients with
KRAS G12C-mutant NSCLC, particularly when standard
treatments are not feasible or effective. Given the generally poor
prognosis and resistance-prone nature of current targeted
therapies, TROP-2-directed strategies may offer a novel avenue for
improving outcomes. Similarly, the SKB264-II-08 clinical trial
suggested that the TROP-2 ADC regimen produced an ORR of 40%, a DCR
of 81%, a median PFS time of 6.2 months and a median OS time of
21.8 months (56). This is worse
than that recorded for the patient in the present case report. We
speculate that patients with KRAS G12C combined with high
expression of TROP-2 may form the group benefiting from TROP-ADC.
However, due to the small number of patients in this category, more
reports on similar patients are required and this hypothesis will
be further explored in future work. As aforementioned, research on
TROP-2 in KRAS-mutant solid tumors, including lung cancer,
is limited. The present case report provides indirect evidence for
the association between the two and offers novel insights and ideas
for the treatment of patients with advanced NSCLC with KRAS
mutations and high TROP-2 expression. However, the present case
report has some limitations, including the unavailability of
clearer TROP-2 immunohistochemistry and raw next-generation
sequencing data.
Acknowledgements
The authors would like to thank Mr. Minglei Cai
(Tongji Medical College, Huazhong University of Science and
Technology, Wuhan, China) for revising the manuscript.
Funding
The present study was funded by the Internal Research Grant of
Scientific Research Project of Hubei Cancer Hospital (grant nos.
2024HBCHYN03 and 2024HBCHYN09), the Clinical Scientific Research
Program of Wu Jieping Medical Funding (grant no.
320.6750.2025-10-5) and the Hubei Natural Science Foundation (grant
no. 2025AFD812).
Availability of data and materials
The TROP-2 immunohistochemistry data generated in
the present study may be requested from MEDx Translational Medicine
Co. Ltd., due to clinical trial requirements. The next-generation
sequencing data generated in the present study are not publicly
available since this was performed by Huangshi Hospital of Chinese
Traditional Medicine (Huangshi, China). The other data generated in
the present study may be requested from the corresponding
author.
Authors' contributions
YP conceived and designed the study. LS and XL
analyzed the data and wrote the manuscript. YX collected data and
images. All authors have read and approved the final version of the
manuscript. YP and LS confirm the authenticity of all the raw
data.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Written consent for publication of the images was
provided by the patient.
Competing interests
The authors declare that they have no competing
interests.
Glossary
Abbreviations
Abbreviations:
|
KRAS
|
v-Ki-ras2 Kirsten rat sarcoma viral
oncogene homolog gene
|
|
NSCLC
|
non-small cell lung cancer
|
|
LUAD
|
lung adenocarcinoma
|
|
TROP-2
|
trophoblast cell-surface antigen 2
|
|
ADC
|
antibody-drug conjugate
|
|
CT
|
computed tomography
|
References
|
1
|
El Osta B, Behera M, Kim S, Berry LD, Sica
G, Pillai RN, Owonikoko TK, Kris MG, Johnson BE, Kwiatkowski DJ, et
al: Characteristics and outcomes of patients with metastatic
KRAS-mutant lung adenocarcinomas: The lung cancer mutation
consortium experience. J Thorac Oncol. 14:876–889. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Yang Y, Shen S, Sun Y, Husain H, Zhou H,
Lu S and Li Z: The relationship between different subtypes of KRAS
and PD-L1 & tumor mutation burden (TMB) based on
next-generation sequencing (NGS) detection in Chinese lung cancer
patients. Transl Lung Cancer Res. 11:213–223. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Adderley H, Blackhall FH and Lindsay CR:
KRAS-mutant non-small cell lung cancer: Converging small molecules
and immune checkpoint inhibition. EBioMedicine. 41:711–716. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Liu J, Kang R and Tang D: Correction: The
KRAS-G12C inhibitor: Activity and resistance. Cancer Gene Ther.
30:17152023. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Canon J, Rex K, Saiki AY, Mohr C, Cooke K,
Bagal D, Gaida K, Holt T, Knutson CG, Koppada N, et al: The
clinical KRAS(G12C) inhibitor AMG 510 drives anti-tumour immunity.
Nature. 575:217–223. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Indini A, Rijavec E, Ghidini M, Cortellini
A and Grossi F: Targeting KRAS in solid tumors: Current challenges
and future opportunities of novel KRAS inhibitors. Pharmaceutics.
13:6532021. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Wu B, Yu C, Zhou B, Huang T, Gao L, Liu T
and Yang X: Overexpression of TROP2 promotes proliferation and
invasion of ovarian cancer cells. Exp Ther Med. 14:1947–1952. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Bessede A, Peyraud F, Besse B, Cousin S,
Cabart M, Chomy F, Rey C, Lara O, Odin O, Nafia I, et al: TROP2 is
associated with primary resistance to immune checkpoint inhibition
in patients with advanced non-small cell lung cancer. Clin Cancer
Res. 30:779–785. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Foersch S, Schmitt M, Litmeyer AS,
Tschurtschenthaler M, Gress T, Bartsch DK, Pfarr N, Steiger K,
Denkert C and Jesinghaus M: TROP2 in colorectal carcinoma:
Associations with histopathology, molecular phenotype, and patient
prognosis. J Pathol Clin Res. 10:e123942024. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Liu X, Deng J, Yuan Y, Chen W, Sun W, Wang
Y, Huang H, Liang B, Ming T, Wen J, et al: Advances in
Trop2-targeted therapy: Novel agents and opportunities beyond
breast cancer. Pharmacol Ther. 239:1082962022. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Inamura K, Yokouchi Y, Kobayashi M,
Ninomiya H, Sakakibara R, Subat S, Nagano H, Nomura K, Okumura S,
Shibutani T and Ishikawa Y: Association of tumor TROP2 expression
with prognosis varies among lung cancer subtypes. Oncotarget.
8:28725–28735. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Omori S, Muramatsu K, Kawata T, Miyawaki
E, Miyawaki T, Mamesaya N, Kawamura T, Kobayashi H, Nakashima K,
Wakuda K, et al: Trophoblast cell-surface antigen 2 expression in
lung cancer patients and the effects of anti-cancer treatments. J
Cancer Res Clin Oncol. 148:2455–2463. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Shimizu T, Sands J, Yoh K, Spira A, Garon
EB, Kitazono S, Johnson ML, Meric-Bernstam F, Tolcher AW, Yamamoto
N, et al: First-in-Human, phase I dose-escalation and
dose-expansion study of trophoblast cell-surface antigen 2-directed
antibody-drug conjugate datopotamab deruxtecan in non-small-cell
lung cancer: TROPION-PanTumor01. J Clin Oncol. 41:4678–4687. 2023.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Loriot Y, Balar AV, Petrylak DP, Kalebasty
AR, Grivas P, Fléchon A, Jain RK, Swami U, Bupathi M, Barthélémy P,
et al: Sacituzumab govitecan demonstrates efficacy across tumor
Trop-2 expression levels in patients with advanced urothelial
cancer. Clin Cancer Res. 30:3179–3188. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Bardia A, Hurvitz SA, Tolaney SM, Loirat
D, Punie K, Oliveira M, Brufsky A, Sardesai SD, Kalinsky K, Zelnak
AB, et al: Sacituzumab govitecan in metastatic Triple-negative
breast cancer. N Engl J Med. 384:1529–1541. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Minato H, Katayanagi K, Kurumaya H, Tanaka
N, Fujimori H, Tsunezuka Y and Kobayashi T: Verification of the
eighth edition of the UICC-TNM classification on surgically
resected lung adenocarcinoma: Comparison with previous
classification in a local center. Cancer Rep (Hoboken).
5:e14222022.PubMed/NCBI
|
|
17
|
Weinberg F and Gadgeel S: Combination
pembrolizumab plus chemotherapy: A new standard of care for
patients with advanced non-small-cell lung cancer. Lung Cancer
(Auckl). 10:47–56. 2019.PubMed/NCBI
|
|
18
|
Seymour L, Bogaerts J, Perrone A, Ford R,
Schwartz LH, Mandrekar S, Lin NU, Litière S, Dancey J, Chen A, et
al: iRECIST: Guidelines for response criteria for use in trials
testing immunotherapeutics. Lancet Oncol. 18:e143–e152. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Li W, Cao Z, Chang P, Zhang B, Li F and
Chang D: Clinical efficacy of PD-1 inhibitors plus Split-course
radiotherapy in the First-Line treatment of advanced kidney cancer:
A randomized controlled trial. J Oncol. 2022:81003232022.PubMed/NCBI
|
|
20
|
Feng S, Callow MG, Fortin JP, Khan Z, Bray
D, Costa M, Shi Z, Wang W and Evangelista M: A saturation
mutagenesis screen uncovers resistant and sensitizing secondary
KRAS mutations to clinical KRASG12C inhibitors. Proc Natl Acad Sci
USA. 119:e21205121192022. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Tan L, Liu J, Liu X, Chen J, Yan Z, Yang H
and Zhang D: Clinical research of Olanzapine for prevention of
chemotherapy-induced nausea and vomiting. J Exp Clin Cancer Res.
28:1312009. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Maurice P: TROP2-directed Antibody-drug
conjugates in advanced Non-small cell lung cancer: A fading Hope? J
Clin Oncol. 42:2839–2842. 2025.
|
|
23
|
Liu Z, Gu Y, Yu F, Zhou L, Cheng X, Jiang
H, Huang Y, Zhang Y, Xu T, Qian W and Li X: The number of
intraoperative intestinal venous circulating tumor cells is a
prognostic factor for colorectal cancer patients. Evid Based
Complement Alternat Med. 2022:41623542022. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Aredo JV, Padda SK, Kunder CA, Han SS,
Neal JW, Shrager JB and Wakelee HA: Impact of KRAS mutation subtype
and concurrent pathogenic mutations on non-small cell lung cancer
outcomes. Lung Cancer. 133:144–150. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Wu M, Zhang EW, Strickland MR, Mendoza DP,
Lipkin L, Lennerz JK, Gainor JF, Heist RS and Digumarthy SR:
Clinical and imaging features of Non-Small cell lung cancer with
G12C KRAS mutation. Cancers (Basel). 13:35722021. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
West HJ, McCleland M, Cappuzzo F, Reck M,
Mok TS, Jotte RM, Nishio M, Kim E, Morris S, Zou W, et al: Clinical
efficacy of atezolizumab plus bevacizumab and chemotherapy in
KRAS-mutated non-small cell lung cancer with STK11, KEAP1, or TP53
comutations: Subgroup results from the phase III IMpower150 trial.
J Immunother. 10:e0030272022.
|
|
27
|
Scheffler M, Ihle MA, Hein R,
Merkelbach-Bruse S, Scheel AH, Siemanowski J, Brägelmann J, Kron A,
Abedpour N, Ueckeroth F, et al: K-ras mutation subtypes in NSCLC
and Associated Co-occuring mutations in other oncogenic pathways. J
Thorac Oncol. 14:606–616. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Liu SY, Sun H, Zhou JY, Jie GL, Xie Z,
Shao Y, Zhang X, Ye JY, Chen CX, Zhang XC, et al: Clinical
characteristics and prognostic value of the KRAS G12C mutation in
Chinese non-small cell lung cancer patients. Biomark Res. 8:222020.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Upadhyay SS, Devasahayam AB, Parate SS,
Dagamajalu S, Keshava Prasad TS, Shetty R and Raju R: An assembly
of TROP2-mediated signaling events. J Cell Commun Signal.
17:1105–1111. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Gutiérrez-Babativa L, Wagner-Gutiérrez N,
Rojas L, Zuluaga J, Arrieta O and Cardona AF: Overcoming
immunotherapy resistance in non-small cell lung cancer: A narrative
review of related factors. Immunotherapy. 17:823–833. 2025.
View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Zhang Y, Sun D and Han W: SMARCA4
mutations and expression in lung adenocarcinoma: Prognostic
significance and impact on the immunotherapy response. FEBS Open
Bio. 14:2086–2103. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Ettinger DS, Wood DE, Aisner DL, Akerley
W, Bauman JR, Bharat A, Bruno DS, Chang JY, Chirieac LR, D'Amico
TA, et al: Non-small cell lung cancer, version 3.2022, NCCN
clinical practice guidelines in oncology. J Natl Compr Canc Netw.
20:497–530. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Chao YC, Lee KY, Wu SM, Kuo DY, Shueng PW
and Lin CW: Melatonin Downregulates PD-L1 expression and modulates
tumor immunity in KRAS-mutant non-small cell lung cancer. Int J Mol
Sci. 22:56492021. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Wan Y, Zhang Y, Wang G, Mwangi PM, Cai H
and Li R: Recombinant KRAS G12D protein vaccines elicit significant
anti-tumor effects in mouse CT26 tumor models. Front Oncol.
10:13262022. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Landre T, Justeau G, Assié JB, Chouahnia
K, Davoine C, Taleb C, Chouaïd C and Duchemann B: Anti-PD-(L)1 for
KRAS-mutant advanced non-small-cell lung cancers: A meta-analysis
of randomized-controlled trials. Cancer Immunol Immunother.
71:719–726. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Burns TF, Borghaei H, Ramalingam SS, Mok
TS and Peters S: Targeting KRAS-mutant non-small-cell lung cancer:
One mutation at a time, with a focus on KRAS G12C mutations. J Clin
Oncol. 38:4208–4218. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Gu G, Yu B, Wan H, Lu S, Zhu X, Zhao Y,
Fuxi Y and Liu C: Molecular characteristics and the effect of KRAS
mutation on the prognosis of immunotherapy in non-small cell lung
cancer in Xinjiang, China. Onco Targets Ther. 15:1021–1032. 2022.
View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Hong DS, Fakih MG, Strickler JH, Desai J,
Durm GA, Shapiro GI, Falchook GS, Price TJ, Sacher A, Denlinger CS,
et al: KRASG12C inhibition with sotorasib in advanced
solid tumors. N Engl J Med. 383:1207–1217. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Barlesi F, Yao W, Duruisseaux M, Doucet L,
Martínez AA, Gregorc V, Juan-Vidal O, Lu S, De Bondt C, de Marinis
F, et al: Adagrasib versus docetaxel in KRASG12C-mutated
non-small-cell lung cancer (KRYSTAL-12): A randomised, open-label,
phase 3 trial. Lancet. 406:615–626. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
de Langen AJ, Johnson ML, Mazieres J,
Dingemans AC, Mountzios G, Pless M, Wolf J, Schuler M, Lena H,
Skoulidis F, et al: Sotorasib versus docetaxel for previously
treated non-small-cell lung cancer with KRAS(G12C) mutation: A
randomised, open-label, phase 3 trial. Lancet. 401:733–746. 2023.
View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Passaro A, Jänne PA and Peters S:
Antibody-drug conjugates in lung cancer: Recent advances and
implementing strategies. J Clin Oncol. 41:3747–3761. 2023.
View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Ou SI, Janne PA, Leal TA, Rybkin II,
Sabari JK, Barve MA, Bazhenova L, Johnson ML, Velastegui KL,
Cilliers C, et al: First-in-human phase I/IB dose-finding study of
adagrasib (MRTX849) in patients with advanced KRASG12C
Solid tumors (KRYSTAL-1). J Clin Oncol. 40:2530–2538. 2022.
View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Zhou Q, Yang N, Zhao M, Huang D, Zhao J,
Yu Y, Yuan Y, Sun L, Dong X, Zhang T, et al: Potent covalent
irreversible inhibitor of KRAS G12C IBI351 in patients with
advanced solid tumors: First-in-human phase I study. Eur J Cancer.
212:1143372024. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Lucky R, Thornburg L and Halford Z:
Targeting TROP2 across solid tumors: The clinical profile and role
of datopotamab deruxtecan. Ann Pharmacother.
1:106002802513932592025.
|
|
45
|
Morgenstern-Kaplan D, Kareff SA, Trabolsi
A, Rodriguez E, Krause H, Ribeiro JR, Tan H, Antonarakis ES, Lou E,
Nagasaka M, et al: Genomic, Immunologic, and prognostic
associations of TROP2 (TACSTD2) expression in solid tumors.
Oncologist. 29:e1480–e1491. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Weng W, Meng T, Zhao Q, Shen Y, Fu G, Shi
J, Zhang Y, Wang Z, Wang M, Pan R, et al: Antibody-exatecan
conjugates with a novel Self-immolative moiety overcome resistance
in colon and lung cancer. Cancer Discov. 13:950–973. 2023.
View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Emanuela G, Marco T and Saverio A:
Targeting Trop-2 as a cancer driver. J Clin Oncol. 41:4688–4692.
2023. View Article : Google Scholar
|
|
48
|
Kushiyama S, Yashiro M, Yamamoto Y, Sera
T, Sugimoto A, Nishimura S, Togano S, Kuroda K, Yoshii M, Tamura T,
et al: Clinicopathologic significance of TROP2 and phospho-TROP2 in
gastric cancer. Mol Clin Oncol. 14:1052021. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Kalinsky K, Diamond JR, Vahdat LT, Tolaney
SM, Juric D, O'Shaughnessy J, Moroose RL, Mayer IA, Abramson VG,
Goldenberg DM, et al: Sacituzumab govitecan in previously treated
hormone receptor-positive/HER2-negative metastatic breast cancer:
Final results from a phase I/II, single-arm, basket trial. Ann
Oncol. 31:1709–1718. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Wen Y, Ouyang D, Zou Q, Chen Q, Luo N, He
H, Anwar M and Yi W: A literature review of the promising future of
TROP2: A potential drug therapy target. Ann Transl Med.
10:14032022. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Okajima D, Yasuda S, Maejima T, Karibe T,
Sakurai K, Aida T, Toki T, Yamaguchi J, Kitamura M, Kamei R, et al:
Datopotamab Deruxtecan, a novel TROP2-directed Antibody-drug
conjugate, demonstrates potent antitumor activity by efficient drug
delivery to tumor cells. Mol Cancer Ther. 20:2329–2340. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Kuo P, Elboudwarej E, Zavodovskaya M, Lin
KW, Lee CV, Diehl L, Patel J, Mekan S and Jürgensmeier JM: Trop-2
expression in non-small cell lung cancer. PLoS One.
20:e03215552025. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Paz-Ares LG, Juan-Vidal O, Mountzios GS,
Felip E, Reinmuth N, de Marinis F, Girard N, Patel VM, Takahama T,
Owen SP, et al: Sacituzumab Govitecan versus docetaxel for
previously treated advanced or metastatic non-small cell lung
cancer: The randomized, open-label phase III EVOKE-01 study. J Clin
Oncol. 42:2860–2872. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Santin AD, Corr BR, Spira A, Willmott L,
Butrynski J, Tse KY, Patel J, Mekan S, Wu T, Lin KW, et al:
Efficacy and safety of Sacituzumab Govitecan in patients with
advanced solid tumors (TROPiCS-03): Analysis in patients with
advanced endometrial cancer. J Clin Oncol. 42:3421–3429. 2024.
View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Peters S, Loi S, André F, Chandarlapaty S,
Felip E, Finn SP, Jänne PA, Kerr KM, Munzone E, Passaro A, et al:
Antibody-drug conjugates in lung and breast cancer: Current
evidence and future directions-a position statement from the ETOP
IBCSG Partners Foundation. Ann Oncol. 35:607–629. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Zhao S, Cheng Y, Wang Q, Li X, Liao J,
Rodon J, Meng X, Luo Y, Chen Z, Wang W, et al: Sacituzumab
tirumotecan in advanced non-small-cell lung cancer with or without
EGFR mutations: Phase 1/2 and phase 2 trials. Nat Med.
31:1976–1986. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Ahn MJ, Tanaka K, Paz-Ares L, Cornelissen
R, Girard N, Pons-Tostivint E, Vicente Baz D, Sugawara S, Cobo M,
Pérol M, et al: Datopotamab Deruxtecan versus docetaxel for
previously treated advanced or metastatic non-small cell lung
cancer: The randomized, open-label phase III TROPION-Lung01 study.
J Clin Oncol. 43:260–272. 2025. View Article : Google Scholar : PubMed/NCBI
|