Introduction
Colorectal cancer (CRC) is the most common
malignancy of the gastrointestinal tract and a leading cause of
cancer-related mortality worldwide (1); for example ~153,020 individuals were
diagnosed with CRC and ~52,550 died from the disease in the United
States in 2022 (2). Prognosis in
patients with CRC is influenced by various clinical factors,
including tumour stage, necrosis, vascular invasion,
differentiation status, immune markers such as the Ki-67
proliferation index, serum carcinoembryonic antigen (CEA) levels
and inflammatory markers (3).
However, the tumour-node-metastasis (TNM) staging system, defined
by the American Joint Committee on Cancer (AJCC), remains the
primary framework for prognostic assessment and treatment planning
(4). In previous work, several
immunohistochemical markers with potential prognostic relevance in
CRC have been evaluated (5–8).
Caudal-type homeobox 2 (CDX2) is an intestinal
transcription factor, typically localised to the nuclei of
intestinal epithelial cells, which serves a critical role in
intestinal development and homeostasis (9,10). As
a homeobox gene, CDX2 participates in embryonic development,
contributes to the maintenance of dynamic homeostasis within the
gut and functions to inhibit proliferation and tumour formation in
colon cancer cells (11–13). More recently, CDX2, along with SATB
homeobox 2, has been increasingly associated with intestinal
inflammation. CDX2 has been shown to modulate inflammasome activity
through the expression of tripartite motif containing 31, an
inhibitor of NLR family pyrin domain containing 3 (14). In murine colonic epithelium, loss of
CDX function results in enhanced macrophage infiltration and
elevated expression of proinflammatory cytokines, including TNFα,
IL1β and IL6 (14,15). Both CDX1 and CDX2 are essential for
maintaining intestinal epithelial homeostasis. In humans, loss of
CDX2 is linked to more aggressive CRC subtypes (16). Correspondingly, in an APCmutant
murine model, concurrent loss of CDX1 and CDX2 leads to increased
polyp formation throughout the intestinal tract (16). Unlike microsatellite instability
(MSI) and Ecadherin, which demonstrate stagerestricted prognostic
effects (17,18), CDX2 has been preliminarily reported
to show stable prognostic relevance across both early and advanced
tumour stages (19). Expression
levels of CDX2 are associated with tumour differentiation, invasion
depth and lymph node metastasis in a stagedependent manner, an
association not fully captured by MSI or Ecadherin alone (20). Based on archived specimens, the
nextgeneration multitarget stool DNA assay has shown promising
performance characteristics, indicating it has high sensitivity for
CRC and advanced precancerous lesions, with excellent specificity
for CRC screening and will undergo further evaluation in a
prospective clinical validation study (21).
Genes such as solute carrier family 17 member 9
(SLC17A9) function as competitive endogenous RNAs that sequester
microRNA (miR)12263p, thereby facilitating CRC progression
(22). These findings establish
SLC17A9 as a novel prognostic biomarker and a potential therapeutic
target for CRC (21). Inhibitors of
STAT3, particularly those targeting the DNAbinding domain, have
been shown to hold therapeutic potential in CRC (23). To address this, subsequent
experiments disrupted the trefoil factor 3-Janus kinase
2/STAT3-CDX2 pathway, revealing notable downstream changes that
mechanistically link these molecular interactions to CRC
progression (24).
CRCs which lose CDX2 expression are known to display
aggressive clinical behaviour yet are frequently accompanied by a
high density of tumourinfiltrating lymphocytes (25). CDX2low CRC has been associated with
worse survival and differential responses to adjuvant chemotherapy
(26). The serrated pathway of
colorectal carcinogenesis may be linked to gastrictype metaplasia
in the colon and alterations in CDX2 expression (1). CDX2, a transcription factor involved
in gastrointestinal differentiation, has been suggested as a
biomarker for favourable outcomes (such as high CDX2 expression
being associated with well and highly differentiated tumours) and
may also help identify subtypes responsive to targeted therapies
(27). Reduced CDX2 expression is
associated with worse survival and several adverse prognostic
factors, including advanced tumour stage, higher tumour grade and
BRAF mutation (28). Variations in
CDX2 expression may also correspond to differences in treatment
outcomes. For instance, loss of CDX2 detected by
immunohistochemistry (IHC) has been associated with worse prognosis
and could help identify patients who are more likely to benefit
from chemotherapy (29). Moreover,
CDX2positive cancer of unknown primary constitutes a distinct
subgroup that tends to respond favourably fluorouracilbased
therapy, supporting the role of CDX2 as a predictive biomarker for
treatment selection (30). Although
loss of CDX2 expression has been linked to worse clinical outcomes
and appears to predict benefit from adjuvant chemotherapy in stage
II and III CRC, its prognostic relevance in stage II disease,
particularly in relation to established clinical risk factors such
as MSI status, BRAF mutation status and tumour budding, remains
inadequately validated (31).
Building on the established link between CDX2 and
CRC, the present study hypothesises that high CDX2 expression
exerts a protective effect in CRC and correlates with a favourable
prognosis. To test this hypothesis, a retrospective analysis of
patients who underwent CRC cancer surgery was performed, including
stratified analyses across different TNM stages. To date, few
studies (32,33) have comprehensively evaluated the
prognostic role of CDX2 beyond AJCC 8th edition stratification, and
investigations examining the association between CDX2 expression
and both overall survival (OS) and diseasefree survival (DFS)
remain limited. Addressing this gap represents a primary aim of the
present study.
It is well established that CDX2 is commonly
expressed in normal colorectal epithelium, whereas its loss or
reduced expression is frequently observed in colorectal tumour
tissue and is associated with unfavourable prognosis (34). To further explore this association,
the relationship between differential CDX2 expression and prognosis
in patients with stage 0-IV CRC treated within a defined period was
examined as the first objective of the present study. Additional
objectives were to determine whether CDX2 expression correlates
with TNM stage, differentiation status, tumour CEA (TCEA) levels,
tumour p53 status and other clinicopathological variables and to
evaluate whether CDX2 serves as an independent prognostic factor in
CRC.
Materials and methods
Patients
A total of 802 patients with CRC who underwent
surgery between January 2017 and January 2020 in the Colorectal
Surgery Department of Huzhou Central Hospital (Huzhou, China) were
initially identified using the medical records system (version
4.37; Hangzhou Lianzhong Medical Science Co., Ltd.) and Haitai
software (version 3.0; Nanjing Haitai Medical). After applying the
inclusion and exclusion criteria, 453 cases were included in the
final analysis. Tumour staging was performed according to the AJCC
8th edition (4). For analytical
purposes, TNM staging is presented in two formats: i) Conventional
staging (denoted as ‘TNM’), which includes subgroups not used in
the final analysis; and ii) a combined grouping that merges overall
stages and combines stages 0 and I (denoted as ‘TNM1’).
The inclusion criteria were: i) A diagnosis of CRC
confirmed by colonoscopy, computed tomography or pathological
examination, regardless of whether the initial diagnosis was made
within or outside the Colorectal Surgery Department of Huzhou
Central Hospital; ii) patients who underwent colorectal surgery
(radical or nonradical) at Huzhou Central Hospital; iii) a
diagnosis of primary tumour recurrence or mortality attributable to
the primary tumour during the present study period; iv)
availability of complete and detailed clinical and pathological
records; and v) complete followup data with accurate documentation.
The exclusion criteria were: i) The presence of severe cardiac,
cerebral, hepatic or pulmonary disease that rendered the patient
unfit for surgery; ii) mortality due to nonCRC causes, or
pathological confirmation of nonadenocarcinoma malignancies, such
as gastrointestinal stromal tumours, neural tumours, lymphomas or
melanomas; and (iii) incomplete clinicopathological data or
insufficient followup information. Based on these criteria, 349
cases were excluded.
Follow-up
Patients were routinely followed up at the
outpatient clinic 2 weeks after surgery. Subsequent follow-ups were
scheduled every 3 months during the first year, every 6 months
during the second year and annually for the following 3 years, for
a total follow-up period of 5 years postoperatively. Followup data
were obtained through telephone interviews and outpatient medical
records. The endoffollowup date was January 2025, with a median
followup duration of 58 months.
CDX2 detection by IHC (Elivision
method)
For CDX2 immunohistochemistry, formalin-fixed
paraffin-embedded tissue sections of CRC tissues (4 µm thickness)
were used. Tissue fixation was performed using 10% neutral buffered
formalin at room temperature for 24–48 h. After deparaffinization
and rehydration, antigen retrieval was performed. Pretreatment was
conducted with BOND Epitope Retrieval Solution 2 (cat. no. AR9640;
Leica Biosystems) at 100°C for 30 min. The sample was incubated
with a blocking agent containing 3% hydrogen peroxide for 5 min at
room temperature. Immunohistochemical staining for CDX2 was
performed on an automated immunostainer (BONDIII; Leica Biosystems)
using the BOND Refine detection kit (cat. no. AR9640; Leica
Biosystems). A CDX2 antibody (clone EPR2764Y; Epredia; cat. no.
RM2116; dilution 1:500) served as the primary antibody. The primary
antibody was incubated at room temperature for 20 min.
Subsequently, the BOND Refine detection kit was applied according
to the manufacturer's instructions, with the secondary antibody
incubated at room temperature for 6 min. Slides were scanned using
a NanoZoomer XR scanner (Hamamatsu Photonics K.K) under ×200 and
×400 objective magnifications. The resulting digital images were
analysed with QuPath software (35). Cellular detection was performed
using the software's builtin cell detection function. Intensity
features were incorporated to calculate Haralick's texture
features, and smoothed features (within a 20 µm radius) were
generated using the corresponding smoothing function. An object
classifier was trained to distinguish tumour cells from stromal
cells. The intensity of 3,3′diaminobenzidine (DAB) chromogen
staining was quantified for each tumour cell using QuPath, with the
DAB threshold set in accordance with the manufacturer's protocol
(BONDIII; Leica Biosystems).
CDX2 classification
For each cell, DAB staining intensity was classified
into four categories according to predefined thresholds. The CDX2
scoring cutoffs were established by the pathology department in
line with the CDX2 kit protocol (cat. no. AR9640; Leica Biosystems)
and recent literature (9): A DAB
intensity value <0.25 was classified as negative (−); 0.25–0.55
as weakly positive (+); 0.55–0.85 as moderately positive (++); and
>0.85 as strongly positive (+++). Considering that cases with
complete CDX2 negativity are comparatively uncommon and do not
encompass the full spectrum of TNM staging categories (36), CDX2 (−) samples and CDX2 (+) samples
were consolidated into a unified group, defined as CDX2 absent/low,
which classification approach has been previously utilized by other
researchers (36). For binary group
comparison and to facilitate subsequent analyses, cases classified
as CDX2 (−) and CDX2 (+) were combined into a ‘CDX2 absent/low’
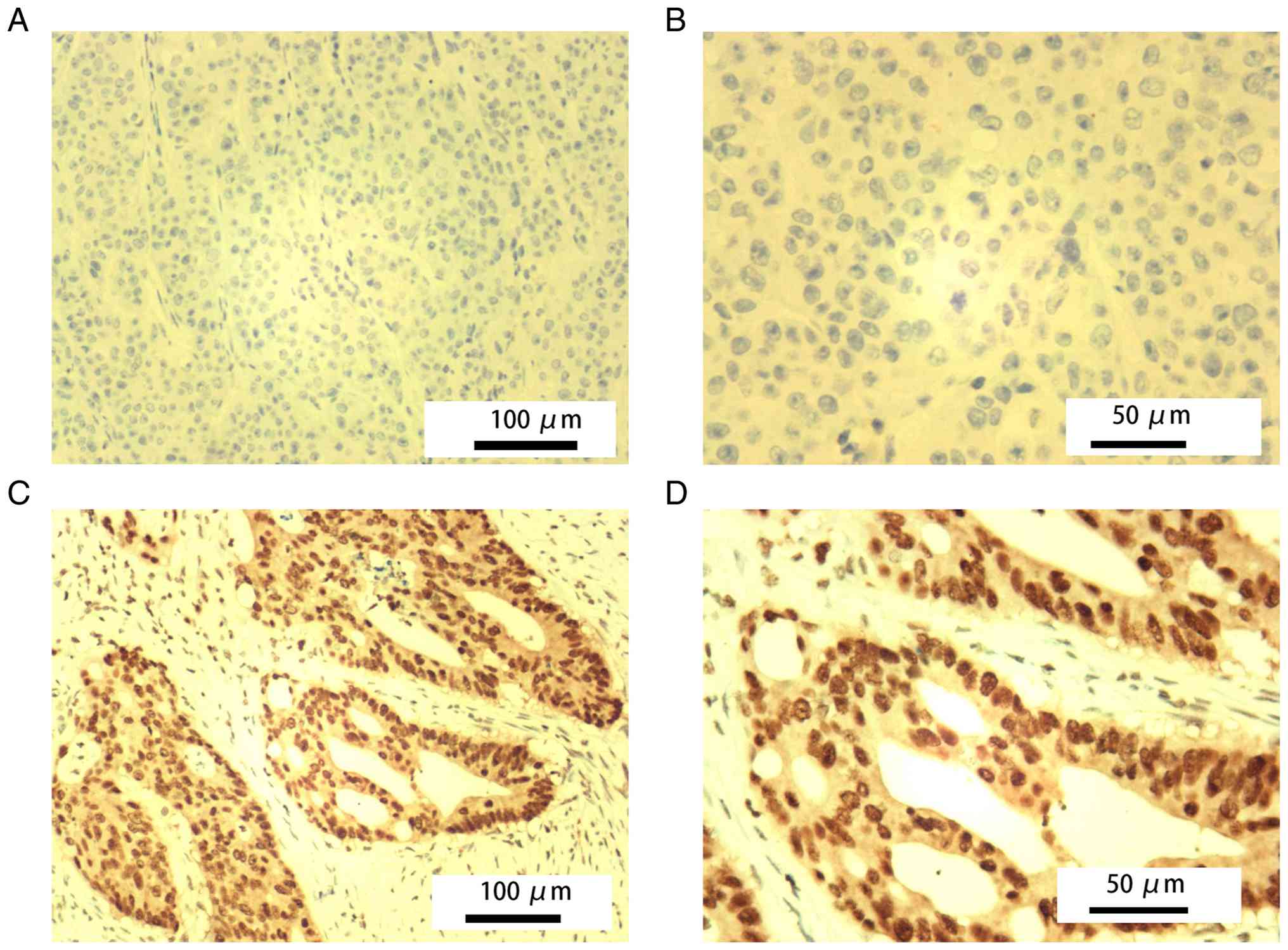
group (Fig. 1A and B), while cases
classified as CDX2 (++) and CDX2 (+++) were combined into a ‘CDX2
high’ group (Fig. 1C and D). Each
case was assessed independently by two experienced pathologists who
were blinded to clinical outcomes. In cases of discordance, a third
senior pathologist adjudicated to minimise interobserver bias.
Receiver operating characteristic curve analysis was performed to
evaluate the diagnostic efficacy of this classification. The
resulting area under the curve values were 0.75 for OS and 0.72 for
DFS (both >0.5), confirming its discriminative power in terms of
sensitivity and specificity (Fig.
S1).
Statistical analysis
All clinical and followup data were entered into
SPSS version 29.0 (IBM Corp.). Missing values were imputed with the
series mean prior to analysis. Continuous clinicopathological
variables were compared between the CDX2 absent/low and CDX2 high
expression groups using independentsamples Student's ttests.
Categorical data were analysed with crosstabulation and Pearson's
χ2 test. Ordinal categorical variables were assessed
using Spearman's correlation. The primary endpoints were defined as
the time from surgery to mortality or until 5-years
postoperatively, which served as the study cut-off. Analysis of 5
year OS and DFS was performed using Kaplan-Meier curves with
logrank tests; risk tables were generated according to CDX2
expression levels. Correlation analyses and relation maps were
performed in SPSS 29, while correlation heatmaps were visualised
using R 4.4.1 R (Posit Software, PBC). The R packages ‘corrplot’
(https://cran.r-project.org/package=corrplot),
‘ggplot2’ (https://cran.r-project.org/package=ggplot2),
‘survival’ (https://cran.r-project.org/package=survival) and
‘survminer’ (https://cran.r-project.org/package=survminer) were
employed for visualisation. Univariate and multivariate Cox
regression analyses were conducted incorporating clinical,
pathological and biochemical variables. Omnibus tests and the enter
method were used in the Cox regression analysis. In the
multivariate analysis, all variables were initially included using
the enter method. To minimize bias, continuous variables were
supplemented with the statistically recognized sequence mean
imputation for missing values. Additionally, cases with missing
values of the categorical variable CDX2 were excluded to reduce
bias. Fisher's exact test was used if expected values in cells were
<5. P<0.05 was considered to indicate a statistically
significant difference.
Results
General data
The present study included 453 patients, of whom 266
were men (58.7%) and 187 women (41.3%). Participant age ranged from
25 to 90 years, with a median of 66 years. The CDX2 absent/low
group consisted of 217 cases (47.9%), including 123 men (27.2%) and
94 women (20.8%). The CDX2 high group included 236 cases (52.1%),
with 143 men (31.6%) and 93 women (20.5%). No significant
difference in sex distribution was observed between the two groups
(χ2=0.713; P=0.398). The mean ages were 64.9±11.2 years
in the CDX2 absent/low group and 65.9±10.7 years in the CDX2 high
group; this difference was not statistically significant (t=−0.97;
P=0.334). Baseline characteristics are summarised in Table I.
 | Table I.Clinicopathological features of CDX2
classifications. |
Table I.
Clinicopathological features of CDX2
classifications.
| Variables | CDX2 absent/low
(n=217) | CDX2 high
(n=236) |
t/χ2 | P-value
(two-sided) |
|---|
| Sex |
|
| 0.713 | 0.398 |
|
Men | 123 (27.2) | 143 (31.6) |
|
|
|
Women | 94 (20.8) | 93 (20.5) |
|
|
| Age, years | 64.9±11.2 | 65.9±10.7 | −0.97 | 0.334 |
| Symptom duration,
days | 23.3±11.8 | 22.1±8.2 | 1.30 | 0.193 |
| Laparoscopy |
|
| 3.466 | 0.063 |
|
Yes | 125 (27.6) | 156 (34.4) |
|
|
| No | 92 (20.3) | 80 (17.7) |
|
|
| Lymph nodes
harvested | 15.7±6.6 | 15.7±9.7 | 0.001 | 0.999 |
| Tumour size,
cm |
|
|
|
|
|
Min | 3.3±1.4 | 3.2±1.5 | 0.816 | 0.415 |
|
Max | 4.6±1.8 | 4.6±1.8 | −0.128 | 0.898 |
| CA72-4, U/ml | 5.7±11.4 | 6.5±24.4 | −0.450 | 0.653 |
| CA19-9, U/ml | 96.9±500.3 | 45.3±143.4 | 1.466 | 0.144 |
| CA15-3, U/ml | 9.2±4.4 | 9.5±7.7 | −0.636 | 0.525 |
| CA125, U/ml | 17.9±17.8 | 18.1±4.7 | −0.061 | 0.951 |
| CEA, ng/ml | 16.3±63.1 | 16.3±54.7 | 0.000 | 1.000 |
| Preoperative CRP,
mg/l | 7.8±16.2 | 7.7±21.3 | 0.069 | 0.945 |
| Preoperative album,
g/l | 37.6±5.3 | 37.8±4.7 | −0.558 | 0.578 |
| Cost, thousand
RMB | 43.4±9.8 | 43.0±10.5 | 0.435 | 0.664 |
| Chemotherapy |
|
| 0.261 | 0.610 |
|
Yes | 142 (31.3) | 149 (32.9) |
|
|
| No | 75 (16.6) | 87 (19.2) |
|
|
| TNM1 |
|
| 11.011 | 0.012a |
| 0 and
I | 28 (6.2) | 45 (9.9) |
|
|
| II | 71 (15.7) | 96 (21.2) |
|
|
|
III | 103 (22.7) | 88 (19.4) |
|
|
| IV | 15 (3.3) | 7 (1.5) |
|
|
|
Differentiation |
|
| 1.223 | 0.543 |
| Poor
and undifferentiated | 92 (20.3) | 94 (20.8) |
|
|
|
Moderate | 121 (26.7) | 134 (29.6) |
|
|
|
High | 4 (0.9) | 8 (1.8) |
|
|
| MSI |
|
| 0.183 | 0.668 |
|
Low | 162 (35.8) | 172 (38.0) |
|
|
|
High | 55 (12.1) | 64 (14.1) |
|
|
| TP53 |
|
| 10.186 | 0.017a |
|
Negative | 55 (12.1) | 40 (8.8) |
|
|
| + | 31 (6.8) | 46 (10.2) |
|
|
| ++ | 32 (7.1) | 54 (11.9) |
|
|
|
+++ | 99 (21.9) | 96 (21.2) |
|
|
| TCEA |
|
| 3.107 | 0.389b |
|
Negative | 2 (0.4) | 1 (0.2) |
|
|
| + | 141 (31.1) | 165 (36.4) |
|
|
| ++ | 11 (2.4) | 16 (3.5) |
|
|
|
+++ | 63 (13.9) | 54 (11.9) |
|
|
| E-cadherin |
|
| 3.639 | 0.303b |
|
Negative | 1 (0.2) | 2 (0.4) |
|
|
| + | 79 (17.4) | 99 (21.9) |
|
|
| ++ | 55 (12.1) | 65 (14.3) |
|
|
|
+++ | 82 (18.1) | 70 (15.5) |
|
|
| Top2 |
|
| 0.678 | 0.712 |
| + | 37 (8.2) | 34 (7.5) |
|
|
| ++ | 162 (35.8) | 180 (39.7) |
|
|
|
+++ | 18 (4.0) | 22 (4.9) |
|
|
Clinicopathological features between
CDX2 absent/low and CDX2 high groups
No significant differences were found between the
CDX2 absent/low and CDX2 high groups in the following continuous
variables: Symptom duration (23.3±11.8 vs. 22.1±8.2 months;
P=0.193), number of lymph nodes harvested (15.7±6.56 vs. 15.7±9.69;
P=0.999), minimum tumour size (3.3±1.5 vs. 3.2±1.5cm; P=0.415) and
maximum tumour size (4.6±1.8 vs. 4.6±1.8 cm; P=0.898). Similarly,
no significant differences were found in levels of carbohydrate
antigen (CA) 72–4 (CA72-4), CA19-9, CA15-3, CA125, CEA,
preoperative C-reactive protein (CRP), preoperative albumin,
treatment cost, receipt of chemotherapy, surgical approach
(laparoscopy), TCEA, differentiation grade, MSI status, E-cadherin
expression or topoisomerase II α (Top2) expression (all P>0.05).
However, significant differences between the two groups were
identified in TNM1 stage (P=0.012) and tumour p53 (tp53) expression
(P=0.017). Detailed results are presented in Table I.
Correlations between CDX2 and other
variables
Pairwise associations between all categorical
variables listed in Table II were
analysed using Spearman's rank correlation. Significant
correlations were found between tp53 and differentiation (P=0.016)
and between tp53 and TNM1 stage (P=0.024). Significant correlations
were also observed between TCEA and E-cadherin (P<0.001),
between E-cadherin and MSI (P<0.001), between tumour
differentiation and TNM1 stage (P<0.001) and between TNM1 stage
and CDX2 expression (P=0.001). Correlation coefficients and
P-values are provided in Table II
and visualised in a correlation matrix (Fig. 2A). To further clarify these
relationships, crosstabulation analyses confirmed strong
associations between CDX2 expression (absent/low vs. high) and
moderate differentiation, TNM1 stages II and III and
poor/undifferentiated differentiation. A strong association was
also observed between moderate differentiation and TNM1 stages II
and III (Fig. 2B).
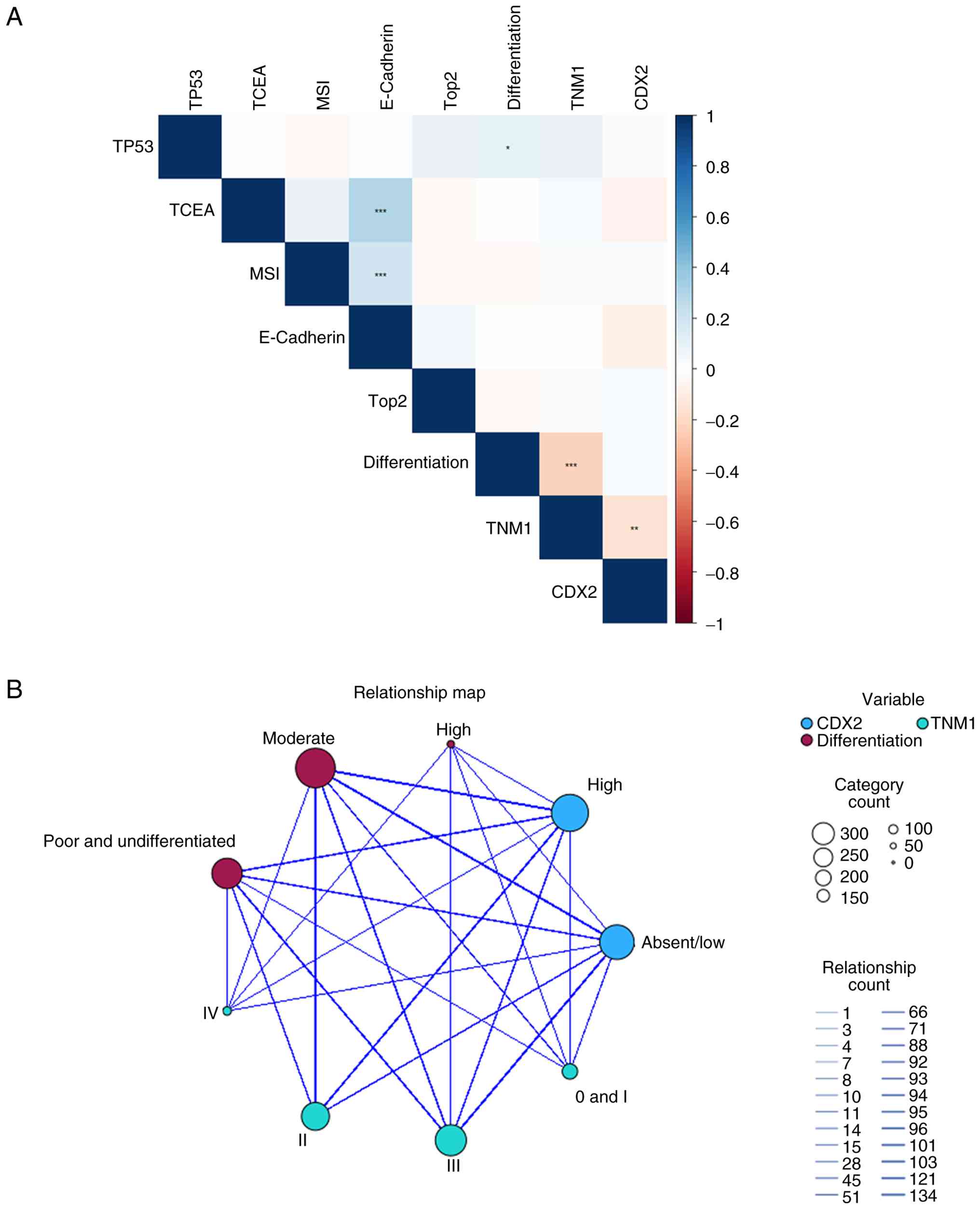 | Figure 2.Correlation and relational analysis
of CDX2. (A) Spearman correlation heatmap between CDX2 and
variables including TP53, TCEA, MSI, Ecadherin, Top2,
differentiation and TNM1 stage, generated using R 4.4.1. Blue
indicates positive correlations, red indicates negative
correlations and white indicates nearzero correlation. Asterisks
denote statistical significance: *P<0.05; **P<0.01;
***P<0.001. (B) Relation map between CDX2 expression (absent/low
vs. high), TNM1 stage and differentiation, generated using SPSS 29.
Circle size corresponds to the number of cases; line thickness
indicates the strength of association. CDX2, caudal-type homeobox
2; MSI, microsatellite instability; TCEA, tumour carcinoembryonic
antigen; TP53, tumour p53; TNM, tumour-node-metastasis; Top2,
topoisomerase II α. |
 | Table II.Spearman (two tailed) correlation
analysis matrix between CDX2 and other variables. |
Table II.
Spearman (two tailed) correlation
analysis matrix between CDX2 and other variables.
| Variable | Result | TP53 | TCEA | MSI | E-cadherin | Top2 |
Differentiation | TNM1 | CDX2 |
|---|
| TP53 | Coef. | 1.000 | 0.019 | −0.043 | 0.036 | 0.090 | 0.113a | 0.106a | 0.012 |
|
| P-value | / | 0.688 | 0.357 | 0.444 | 0.055 | 0.016a | 0.024a | 0.806 |
| TCEA | Coef. | 0.019 | 1.000 | 0.074 | 0.298c | −0.040 | −0.017 | 0.048 | −0.062 |
|
| P-value | 0.688 | / | 0.117 |
<0.001c | 0.391 | 0.718 | 0.308 | 0.188 |
| MSI | Coef. | −0.043 | 0.074 | 1.000 | 0.191c | −0.041 | −0.035 | 0.011 | 0.020 |
|
| P-value | 0.357 | 0.117 | / |
<0.001c | 0.384 | 0.453 | 0.814 | 0.669 |
| E-cadherin | Coef. | 0.036 | 0.298b | 0.191c | 1.000 | 0.051 | −0.003 | −0.015 | −0.075 |
|
| P-value | 0.444 | <0.01 |
<0.001c | / | 0.277 | 0.951 | 0.757 | 0.110 |
| Top2 | Coef. | 0.090 | −0.040 | −0.041 | 0.051 | 1.000 | −0.003 | 0.018 | 0.038 |
|
| P-value | 0.055 | 0.391 | 0.384 | 0.277 | / | 0.947 | 0.696 | 0.423 |
|
Differentiation | Coef. | 0.113a | −0.017 | −0.035 | −0.003 | −0.003 | 1.000 | −0.212c | 0.034 |
|
| P-value | 0.016a | 0.718 | 0.453 | 0.951 | 0.947 | / |
<0.001c | 0.470 |
| TNM1 | Coef. | 0.106a | 0.048 | 0.011 | −0.015 | 0.018 | −0.212c | 1.000 | −0.151b |
|
| P-value | 0.024a | 0.308 | 0.814 | 0.757 | 0.696 |
<0.001c | / | 0.001b |
| CDX2 | Coef. | 0.012 | −0.062 | 0.020 | −0.075 | 0.038 | 0.034 | −0.151b | 1.000 |
|
| P-value | 0.806 | 0.188 | 0.669 | 0.110 | 0.423 | 0.470 | 0.001b | / |
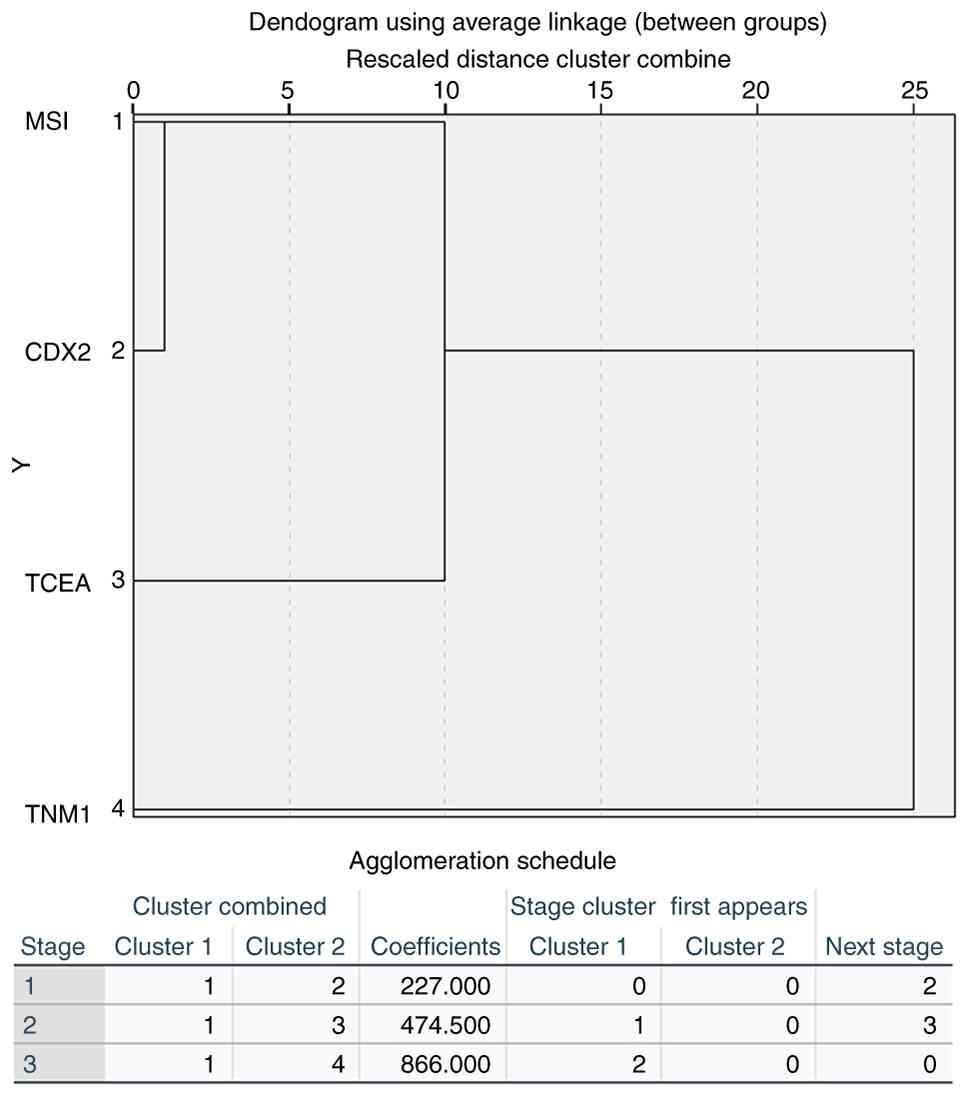
Systematic cluster analysis of CDX2,
TNM1, TCEA and MSI
A systematic cluster analysis was conducted due to
the correlations identified between the four variables analyzed.
The results demonstrated that CDX2 clustered most closely with MSI
(coefficient=258), followed by MSI with TCEA (coefficient=495), and
finally MSI with TNM1 (coefficient=876). As illustrated in Fig. 3, CDX2 exhibited a high degree of
similarity with MSI.
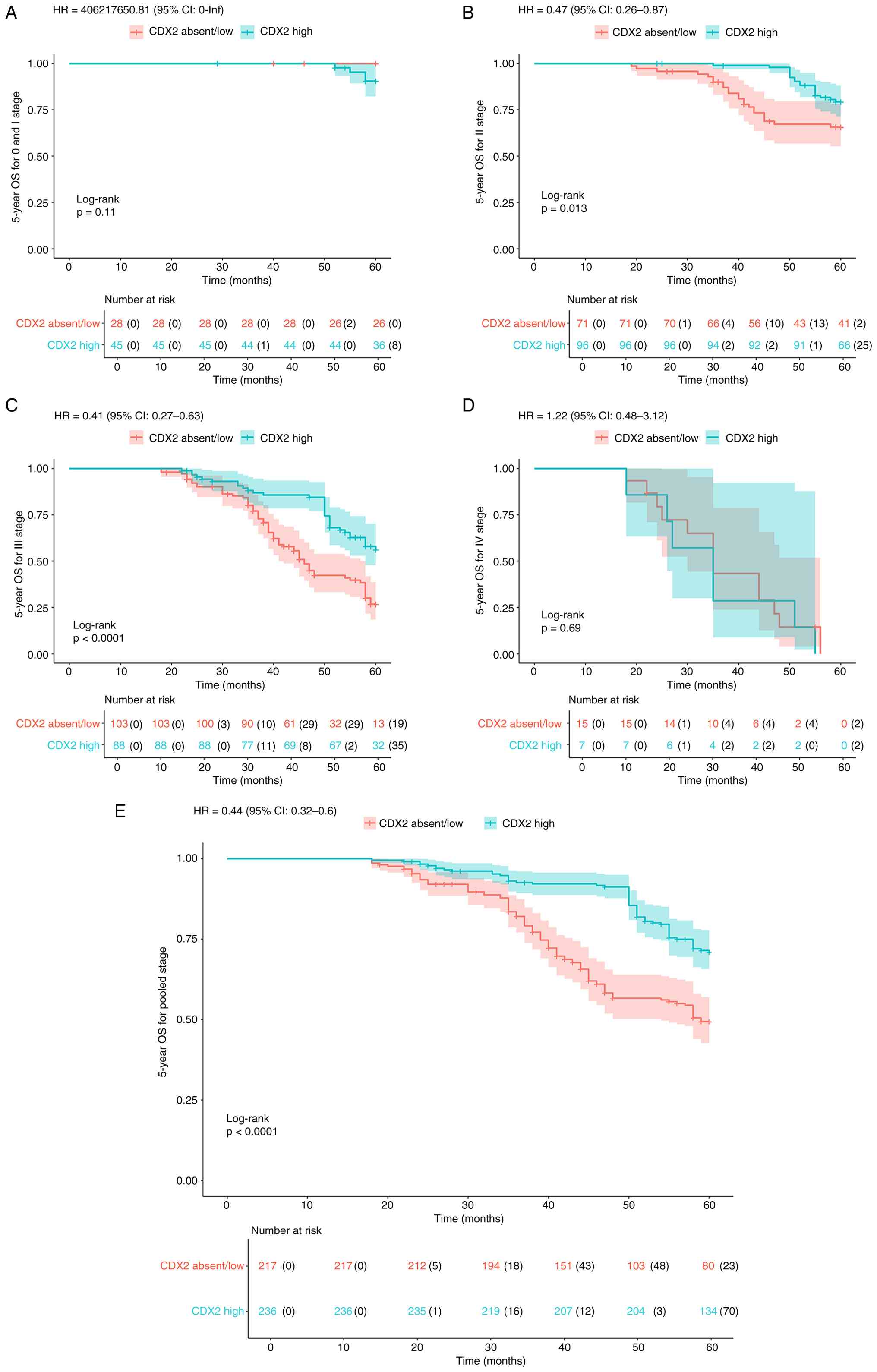
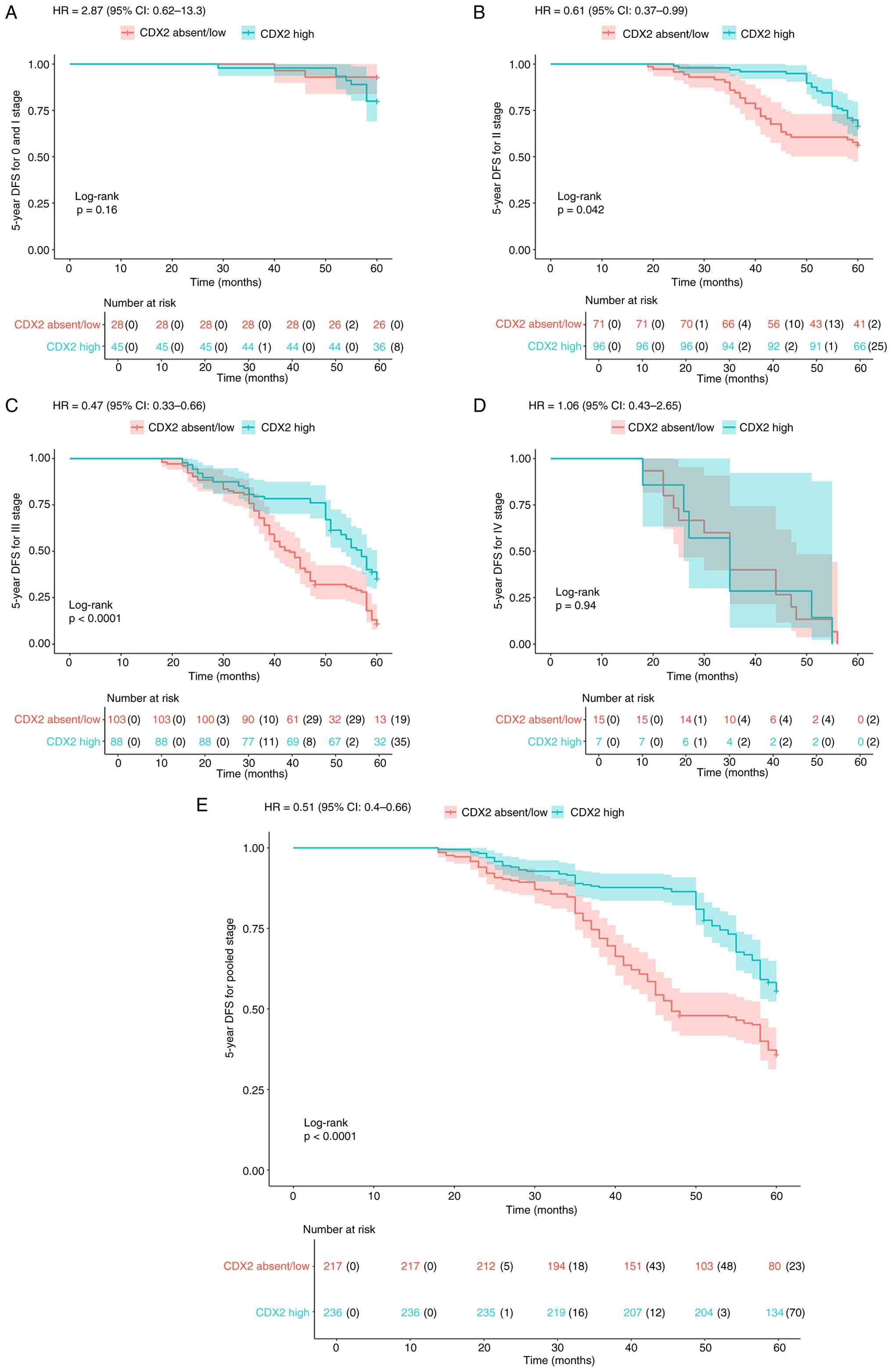
OS and DFS analysis by CDX2 expression
across CRC stages 0-IV
Kaplan-Meier survival analysis with logrank tests
was used to compare 5year OS and DFS between CDX2 expression
groups. For OS, high CDX2 expression was associated with
significantly improved survival in stage II (P=0.013) and stage III
CRC (P<0.001; Fig. 4B and C),
but not in stage I (P=0.11) or stage IV (P=0.69; Fig. 4A and D). In the pooledstage analysis
(all stages combined), the CDX2 high group showed significantly
improved OS compared with the CDX2 absent/low group (P<0.001;
Fig. 4E). Similarly, for DFS, high
CDX2 expression was associated with significantly improved outcomes
in stage II (P=0.042) and stage III (P<0.001) disease (Fig. 5B and C), but not in stage I (P=0.16)
or stage IV (P=0.94) (Fig. 5A and
D). In the pooledstage analysis, the CDX2 high group exhibited
significantly improved DFS compared with the CDX2 absent/low group
(P<0.001; Fig. 5E).
Univariate and multivariate Cox
regression analyses of OS and DFS
Cox regression models were used for univariate and
multivariate analyses. Univariate analysis identified the following
variables as significantly associated with worse OS: Not
laparoscopic surgery (vs. laparoscopy), no chemotherapy, higher
TNM1 stage, worse differentiation, MSIstable status, negative and
positive Ecadherin expression and low CDX2 expression (all
P<0.05). Sex, TCEA and Top2 expression were not significant
predictors of OS (all P>0.05). For DFS, significant variables
included not laparoscopic surgery, no chemotherapy, higher TNM1
stage, worse differentiation, negative and positive Ecadherin
expression and low CDX2 expression (all P<0.001). Notably, MSI
status was significant for OS (P=0.015) but not for DFS
(P>0.05). Detailed results are presented in Table III. Variables that were
significant in both univariate OS and DFS analyses were included in
the multivariate models. Laparoscopy was not retained as an
independent factor in the multivariate models (OS, P=0.393; DFS,
P=0.087). The remaining factors, chemotherapy, TNM1 stage,
differentiation grade, Ecadherin expression and CDX2 expression,
were statistically significant independent prognostic factors for
both OS and DFS (all P<0.05), confirming their role in CRC
prognosis. Detailed multivariate results are shown in Table IV.
 | Table III.Univariate analysis for OS and DFS by
COX regression. |
Table III.
Univariate analysis for OS and DFS by
COX regression.
|
| OS | DFS |
|---|
|
|
|
|
|---|
| Variables | HR | 95% CI | P-value | HR | 95% CI | P-value |
|---|
| Sex |
|
| 0.836 |
|
| 0.352 |
|
Men | 1 |
|
| 1 |
|
|
|
Women | 0.968 | 0.709–1.321 |
| 1.128 | 0.875–1.453 |
|
| Laparoscopy |
|
|
<0.001c |
|
|
<0.001c |
|
Yes | 1 |
|
| 1 |
|
|
| No | 1.683 | 1.238–2.287 |
| 1.690 | 1.313–2.177 |
|
| Chemotherapy |
|
|
<0.001c |
|
|
<0.001c |
| No | 1 |
|
| 1 |
|
|
|
Yes | 1.846 | 1.304–2.614 |
| 1.897 | 1.423–2.528 |
|
| TNM1 |
|
|
<0.001c |
|
|
<0.001c |
| 0 and
I | 1 |
|
| 1 |
|
|
| II | 5.336 | 1.913–14.882 |
| 2.959 | 1.559–5.615 |
|
|
III | 15.451 | 5.682–42.035 |
| 8.905 | 4.817–16.461 |
|
| IV | 54.387 | 18.452–160.302 |
| 25.218 | 12.1.2–52.551 |
|
|
Differentiation |
|
|
<0.001c |
|
|
<0.001c |
| Poor
and undifferentiated | 1 |
|
| 1 |
|
|
|
Moderate | 0.400 | 0.292–0.547 |
| 0.466 | 0.361–0.602 |
|
|
High | 0.221 | 0.054–0.896 |
| 0.154 | 0.038–0.621 |
|
| MSI |
|
| 0.015a |
|
| 0.165 |
|
Low | 1 |
|
| 1 |
|
|
|
High | 0.888 | 0.625–1.261 |
| 0.811 | 0.604–1.090 |
|
| TCEA |
|
| 0.545 |
|
| 0.498 |
|
Negative | 1 |
|
| 1 |
|
|
| + | 0.568 | 0.140–2.301 |
| 0.838 | 0.208–3.380 |
|
| ++ | 0.769 | 0.170–3.472 |
| 1.202 | 0.278~5.202 |
|
|
+++ | 0.678 | 0.165–2.795 |
| 0.946 | 0.232–3.863 |
|
|
E-cadherind |
|
| 0.030a |
|
| 0.019a |
|
Negative and + | 1 |
|
| 1 |
|
|
| ++ | 1.447 | 1.007–2.081 |
| 1.337 | 0.990–1.806 |
|
|
+++ | 0.882 | 0.650–1.285 |
| 0.848 | 0.624–1.154 |
|
| Top2 |
|
| 0.201 |
|
| 0.398 |
| + | 1 |
|
| 1 |
|
|
| ++ | 0.705 | 0.481–1.034 |
| 0.808 | 0.582–1.121 |
|
|
+++ | 0.777 | 0.429–1.408 |
| 0.759 | 0.452–1.275 |
|
| CDX2 |
|
|
<0.001c |
|
|
<0.001c |
|
Absent/low | 1 |
|
| 1 |
|
|
|
High | 0.437 | 0.319–0.599 |
| 0.515 | 0.399–0.665 |
|
 | Table IV.Multivariate analysis by Cox
regression of OS and DFS for colorectal cancer. |
Table IV.
Multivariate analysis by Cox
regression of OS and DFS for colorectal cancer.
|
| OS | DFS |
|---|
|
|
|
|
|---|
| Variables | HR | 95% CI | P-value | HR | 95% CI | P-value |
|---|
| Laparoscopy |
|
| 0.393 |
|
| 0.087 |
|
Yes | 1 |
|
| 1 |
|
|
| No | 1.151 | 0.833–1.589 |
| 1.259 | 0.967–1.641 |
|
| Chemotherapy |
|
| 0.015a |
|
| 0.018a |
| No | 1 |
|
| 1 |
|
|
|
Yes | 0.616 | 0.417–0.910 |
| 0.672 | 0.483–0.935 |
|
| TNM1 |
|
|
<0.001b |
|
|
<0.001b |
| 0 and
I | 1 |
|
| 1 |
|
|
| II | 5.588 | 1.947–16.037 |
| 2.987 | 1.520–5.868 |
|
|
III | 16.575 | 5.688–48.30 |
| 9.283 | 4.646–18.549 |
|
| IV | 53.232 | 16.747–169.198 |
| 24.222 | 10.746–54.598 |
|
|
Differentiation |
|
|
<0.001b |
|
|
<0.001b |
| Poor
and undifferentiated | 1 |
|
| 1 |
|
|
|
Moderate | 0.534 | 0.386–0.740 |
| 0.613 | 0.470–0.799 |
|
|
High | 0.292 | 0.069–1.228 |
| 0.221 | 0.054–0.909 |
|
|
E-cadherinc |
|
| 0.043a |
|
| 0.040a |
|
Negative and + | 1 |
|
| 1 |
|
|
| ++ | 1.476 | 1.019–2.138 |
| 1.348 | 0.993–1.828 |
|
|
+++ | 0.926 | 0.632–1.357 |
| 0.895 | 0.656–1.222 |
|
| CDX2 |
|
|
<0.001b |
|
|
<0.001b |
|
Absent/low | 1 |
|
| 1 |
|
|
|
High | 0.482 | 0.347–0.669 |
| 0.557 | 0.427–0.725 |
|
Discussion
CDX2, a nuclear homeobox transcription factor
encoded by the CDX homeobox gene (10,37),
is established as a specific and sensitive immunohistochemical
biomarker in colorectal adenocarcinoma (38). Its loss of expression is associated
with highgrade tumours and advanced disease stage in CRC (38–41).
Foundational research across the past decades has characterised
CDX2 as a tumour suppressor (42–44),
while more recent clinical studies link high CDX2 expression to a
favourable prognosis in CRC (1,9).
However, these prior investigations have predominantly focused on
OS, without examining DFS or conducting stagespecific analyses
across TNM stages. Moreover, they have not conclusively established
CDX2 as an independent prognostic factor in CRC. The relationships
between CDX2 expression and other clinical or immunohistochemical
variables also remain underexplored.
A previous study has reported notable associations
between tumour histological differentiation and various factors,
including perineural invasion, vascular invasion, mismatchrepair
deficiency, p53 status, CDX2 expression and Ecadherin (41). The influence of CDX2 on prognosis
merits further investigation, particularly as biological and
molecular heterogeneity may confound the assessment of other
prognostic markers (45).
CDX2negative phenotypes are associated with poorly differentiated
histological subtypes (46).
Addressing these gaps was a primary aim of the present study.
As negative CDX2 expression occurs in ~7.8% of cases
(47), CDX2 (−) and CDX2 (+) cases
were combined into a ‘CDX2 absent/low’ group, and CDX2 (++) and
CDX2 (+++) cases into a ‘CDX2 high’ group to enable more robust
statistical comparisons.
In the present cohort, CDX2 absent/low expression
accounted for 47.9% of cases, and CDX2 high expression for 52.1%.
This incidence of CDX2 absent/low (47.9%) aligns with the reported
rate of <48% for CDX2low expression alone in prior literature
(48), a concordance attributable
to the inclusive classification combining negative and low cases.
No significant differences were observed between the CDX2
expression groups regarding sex, age, tumour size, lymph node
yield, differentiation grade or preoperative serum levels of
CA72-4, CA19-9, CA15-3, CA125, CEA, CRP and albumin. However, a
significant association was identified between CDX2 expression and
pooled TNM1 stage, with a higher prevalence of CDX2 absent/low
expression in stages III and IV, a relationship not clearly
detailed in existing literature (49).
CRC prognostication traditionally relies on the
Union for International Cancer Control/AJCC TNM staging system.
Nonetheless, stageindependent survival variations observed in
clinical practice suggest that prognosis, risk stratification and
guidance for neoadjuvant/adjuvant therapy are governed by a complex
interplay of stage, pathological features and biomarkers.
Histological characteristics, such as tumour budding, perineural
invasion and lymph node metrics, alongside molecular markers such
as MSI, KRAS, BRAF and CDX2, can enhance prognostic assessment and
optimise adjuvant treatment (50).
Although a higher frequency of CDX2 absent/low
expression was noted in tissues with elevated p53 expression and an
initial analysis suggested significance, subsequent Spearman
correlation analysis did not confirm a statistically significant
association, a finding consistent with a prior report indicating
p53 has no association with CDX2 expression (45). Nevertheless, strong relationships
were observed between CDX2 expression (absent/low vs. high) and
moderate tumour differentiation; between CDX2 expression and TNM1
stages II–III; and between CDX2 expression and poor
differentiation/undifferentiated. A pronounced association was also
evident between moderate differentiation and TNM1 stages II–III.
These specific interrelationships have not been thoroughly examined
in previous studies (45,49–57).
Systematic cluster analysis in the present investigation revealed
that CDX2 clustered closely with MSI, yet TNM1 stage retained the
dominant influence. This finding challenges suggestions that
prognostic markers for CRC could supplant TNM staging in
therapeutic decision-making (58)
and reinforces the role of TNM as the cornerstone for clinical
management in CRC (49).
Correlation analysis demonstrated an inverse
relationship between CDX2 expression and TNM stage: Elevated CDX2
expression was associated with more favourable outcomes, whereas
advanced TNM stage predicted worse prognosis, results that align
with the aforementioned survival data. In the pooled analysis of
TNM1 stages, high CDX2 expression was associated with greater OS
and DFS, partially corroborating previous findings indicating that
high CDX2 expression may be associated with a good prognosis of CRC
(1,9). However, stagespecific stratification
revealed that the prognostic benefit of high CDX2 expression for
both OS and DFS was confined to stages II and III. This may be
explained by the limited discriminative power of prognostic markers
in earlystage (I) disease and the universally adverse prognosis
characteristic of stage IV CRC (59).
The present study demonstrates that established
factors such as chemotherapy, TNM1 stage, differentiation grade and
immunohistochemical markers including MSI, Ecadherin and CDX2 are
notable prognostic determinants in CRC (7,49,60–62).
TCEA, by contrast, did not emerge as an independent prognostic
factor in the present study cohort, contradicting a finding from an
earlier study (6). This discrepancy
highlights the limitation of relying on singlemarker assessment and
highlights the necessity for a multimodal prognostic approach. In
this context, the combined utility of preoperative serum CEA and
postoperative TCEA has recently been explored (8). Other serum tumour markers (such as
CA724, CA199, CA153 and CA125) showed no significant variation
across CDX2 expression groups and were therefore not included in
the univariate or multivariate Cox regression models. As the
prognostic value of these markers is welldocumented, they were not
the focus of the present analysis. Nevertheless, the potential
synergistic prognostic value of combining these markers with CDX2
and CEA warrants future investigation, as a panel of complementary
biomarkers could enhance prognostic precision.
The emphasis on CDX2 expression and its correlations
aligns with the increasing focus on molecular subtyping in CRC
management. As a key intestinal differentiation marker, CDX2 could
serve as an anchor for constructing multimodal prognostic models
that integrate clinical, pathological and molecular data.
Validation of combined biomarker panels and refinement of
riskstratification strategies will require future studies with
larger, multicentre cohorts.
Treatment stratification based on CDX2 expression
levels represents a promising strategy for personalising therapy in
gastrointestinal malignancies. CDX2 exerts contextdependent effects
on tumour biology: High expression is often associated with
favourable responses to chemotherapy and immunotherapy, whereas low
expression is associated with drug resistance and aggressive
phenotypes (20,63). Stratifying patients by CDX2 status
could therefore guide therapeutic selection, with highCDX2 tumours
potentially benefiting from conventional cytotoxic agents and
lowCDX2 tumours necessitating combinatorial approaches targeting
compensatory pathways such as epithelialmesenchymal transition.
This stratified paradigm not only aims to optimise outcomes by
minimising ineffective treatments and toxicity but also addresses
tumour heterogeneity, thereby advancing precision oncology
approaches grounded in specific molecular profiles rather than
empirical algorithms (64–66).
The present study has several limitations. External
validation in an independent cohort or publicly available dataset
would substantially enhance the robustness and reproducibility of
the prognostic role of CDX2 identified in present research. In the
present study, the analysis was based on a single-centre cohort
with strict inclusion and exclusion criteria to ensure the
homogeneity of the study population. Publicly available datasets,
including The Cancer Genome Atlas and Gene Expression Omnibus
databases (https://www.cancer.gov/ccg/research/genome-sequencing/tcga;
http://www.ncbi.nlm.nih.gov/geo/) were
used to attempt to validate the present findings (data not shown);
however, a majority of these datasets lack standardised CDX2 IHC
data (the primary detection method of CDX2 in the present study) or
consistent follow-up endpoints, which limits the direct
comparability of data. External validation is a key step to confirm
the prognostic value of CDX2 and this should be prioritised in
future research agendas. To ensure balanced datasets, CDX2 (−) and
CDX2 (+) cases were combined into a composite ‘CDX2 absent/low’
group. Although this approach deviates from the reported prevalence
of CDX2low expression (≤48%) in contemporary literature (1), it was methodologically required to
avoid excluding key subgroups during TNMstage analyses, especially
in a largescale study where such exclusions could affect
feasibility. Two specific statistical limitations should be noted:
First, no correction for multiple testing was applied across the
comparisons performed; and second, formal protocols for handling
missing data were not implemented. Further limitations include the
relatively modest sample size, the singlecentre design and the
classification of CDX2 expression based on IHC rather than genetic
testing. These factors increase potential type I error, risk bias
and limit generalizability. Consequently, findings require
validation in larger, multi-centre studies using standardized IHC
and prospective data. Serum markers CA724, CA199, CA153, CA125 and
CEA were recorded as continuous variables in the present study and
showed no significant association with CDX2 expression groups; they
were therefore not included in the Cox regression analysis. As a
potential direction for future research, these markers could be
categorised into normal and elevated groups according to
established reference values, creating categorical variables
suitable for Cox regression modelling.
In conclusion, elevated CDX2 expression is
associated with significantly improved OS and DFS in patients with
stage II and III CRC, as well as in pooledstage analyses. CDX2
represents an independent prognostic biomarker in CRC. These
findings, however, require validation in largescale, multi-centre
prospective studies. Future research should focus on the following:
i) Evaluating the combined prognostic utility of CDX2 with serum
biomarkers such as CEA, CA199 and CA724; ii) assessing its
synergistic effects with immunohistochemical markers including p53
status, MSI and Ecadherin expression; and iii) establishing
standardised scoring systems for CDX2 expression.
Supplementary Material
Supporting Data
Acknowledgements
The authors would like to thank Professor Zhaozheng
Zheng, Professor Yan Chen and Dr Chong Zhu (Department of
Colorectal Surgery, Huzhou Central Hospital, Affiliated Central
Hospital of Huzhou University, Huzhou, Zhejiang, P.R. China) for
contributing clinical data to the present study.
Funding
Funding: No funding was received.
Availability of data and materials
The data generated in the present study may be
requested from the corresponding author.
Authors' contributions
GT, YF and HQ contributed to conceptualisation,
formal analysis, investigation, methodology, supervision and the
writing of the original draft, as well as reviewing and editing the
manuscript. SYu, KG, JL, WY and SYa were involved in
conceptualisation, methodology and supervision. GT, YF and HQ were
responsible for data curation and validation. GT and YF confirm the
authenticity of all the raw data. All authors read and approved the
final version of the manuscript.
Ethics approval and consent to
participate
The present study was conducted in accordance with
the ethical principles of the Declaration of Helsinki and was
approved by the Ethics Committee of Huzhou Central Hospital
(approval no. 20250901702). The need for written informed consent
from all participants for the use of their tissue samples and
medical records for research purposes was waived.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Cazacu SM, Iordache S, Iovănescu VF,
Streba L, Neagoe CD, Busuioc CJ, Cârţu D and Florescu MM: The role
of CDX2, MUC5AC, and p53 in the evaluation of the progression of
serrated lesions toward colorectal carcinoma. Rom J Morphol
Embryol. 66:99–109. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Siegel RL, Wagle NS, Cercek A, Smith RA
and Jemal A: Colorectal cancer statistics, 2023. CA Cancer J Clin.
73:233–254. 2023.PubMed/NCBI
|
|
3
|
Tong G, Wang Y, Qian H, Tan Z, Shen Y and
Li H: The effects of two combined methods of P53 expression and
preoperative serum CEA detection on the prognosis of colorectal
cancer. Front Oncol. 15:15908362025. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Tong GJ, Zhang GY, Liu J, Zheng ZZ, Chen
Y, Niu PP and Xu XT: Comparison of the eighth version of the
American Joint Committee on Cancer manual to the seventh version
for colorectal cancer: A retrospective review of our data. World J
Clin Oncol. 9:148–161. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Tong G, Shen Y, Li H, Qian H and Tan Z:
NLRC4, inflammation and colorectal cancer (Review). Int J Oncol.
65:992024. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Tong G, Xu W, Zhang G, Liu J, Zheng Z,
Chen Y, Niu P and Xu X: The role of tissue and serum
carcinoembryonic antigen in stages I to III of colorectal cancer-A
retrospective cohort study. Cancer Med. 7:5327–5338. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Tong G, Zhang G, Hu Y, Xu X and Wang Y:
Correlation between mismatch repair statuses and the prognosis of
stage I–IV colorectal cancer. Front Oncol. 13:12783982024.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Tong G, Li H, Shen Y, Tan Z and Qian H:
The combined evaluation of preoperative serum CEA and postoperative
tissue CEA as a prognostic factor in stages 0-IV colorectal cancer:
A retrospective cohort study. Front Med (Lausanne). 11:14470412025.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Sirniö P, Elomaa H, Tuomisto A, Äijälä VK,
Karjalainen H, Kastinen M, Tapiainen VV, Sirkiä O, Ahtiainen M,
Helminen O, et al: CDX2 and SATB2 loss are associated with myeloid
cell infiltration and poor survival in colorectal cancer. Cancer
Immunol Immunother. 74:1112025. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Beck F: The role of Cdx genes in the
mammalian gut. Gut. 2004.53:1394–1396. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Sweetser S, Smyrk TC and Sinicrope FA:
Serrated colon polyps as precursors to colorectal cancer. Clin
Gastroenterol Hepatol. 11:760–767; quiz e54-5. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Balbinot C, Armant O, Elarouci N, Marisa
L, Martin E, De Clara E, Onea A, Deschamps J, Beck F, Freund JN and
Duluc I: The Cdx2 homeobox gene suppresses intestinal tumorigenesis
through non-cell-autonomous mechanisms. J Exp Med. 215:911–926.
2018. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Yu J, Liu D, Sun X, Yang K, Yao J, Cheng
C, Wang C and Zheng J: CDX2 inhibits the proliferation and tumor
formation of colon cancer cells by suppressing Wnt/β-catenin
signaling via transactivation of GSK-3β and Axin2 expression. Cell
Death Dis. 10:262019. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Jahan S, Awaja N, Hess B, Hajjar S, Sad S
and Lohnes D: The transcription factor Cdx2 regulates inflammasome
activity through expression of the NLRP3 suppressor TRIM31 to
maintain intestinal homeostasis. J Biol Chem. 298:1023862022.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Wang YS, Kou Y, Zhu RT, Han BW, Li CH,
Wang HJ, Wu HB, Xia TM and Che XM: CDX2 as a predictive biomarker
involved in immunotherapy response suppresses metastasis through
EMT in colorectal cancer. Dis Markers. 2022:90256682022. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Zhu Y, Hryniuk A, Foley T, Hess B and
Lohnes D: Cdx2 Regulates Intestinal EphrinB1 through the Notch
Pathway. Genes (Basel). 12:1882021. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Mehmood MS and Danaf N: Bacteroides
fragilis toxin in colorectal tumors activates STAT3 and drives
microsatellite instability. Ann Med Surg (Lond). 88:957–958. 2025.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Arnold A, Tronser M, Sers C, Ahadova A,
Endris V, Mamlouk S, Horst D, Möbs M, Bischoff P, Kloor M and
Bläker H: The majority of β-catenin mutations in colorectal cancer
is homozygous. BMC Cancer. 20:10382020. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Leskela S, Romero I, Cristobal E,
Pérez-Mies B, Rosa-Rosa JM, Gutierrez-Pecharroman A, Santón A,
Gonzalez BO, López-Reig R, Hardisson D, et al: The frequency and
prognostic significance of the histologic type in early-stage
ovarian carcinoma: A reclassification study by the spanish group
for ovarian cancer research (GEICO). Am J Surg Pathol. 44:149–161.
2020. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Chan WY, Chua W, Wilkinson K, Epitakaduwa
C, Mandaliya H, Descallar J, Roberts TL, Becker TM, Ng W, Lee CS
and Lim SH: The Prognostic and Predictive Utility of CDX2 in
Colorectal Cancer. Int J Mol Sci. 25:86732024. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Kamal R, Paul P and Diksha Awasthi A:
Exploring gene therapy: The next generation of colorectal cancer
treatment. Curr Gene Ther. 25:195–198. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Xu Z, Yu Y, Ni H, Sun W and Kuang Y:
LINC01836 promotes colorectal cancer progression and functions as
ceRNA to Target SLC17A9 by Sponging miR-1226-3p. Protein Pept Lett.
31:43–60. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Suleman M, Khan SU, Ali S, Alghamdi A,
Alissa M, Mushtaq RY and Crovella S: Probing the Depths of
Molecular Complexity: STAT3 as a Key Architect in Colorectal Cancer
Pathogenesis. Curr Gene Ther. 25:433–452. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Wang Y, Qian F, Shan B, Zhang L, Zhong J,
Chen S, Yao M, Li S, Cao X, Fei S and Pang X: TFF3 initiates
gastric intestinal metaplasia by activating JAK2 and STAT3 under
high salt conditions. Sci Rep. 15:235762025. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Lee JA, Park HE, Jin HY, Jin L, Yoo SY,
Cho NY, Bae JM, Kim JH and Kang GH: The combination of CDX2
expression status and tumor-infiltrating lymphocyte density as a
prognostic factor in adjuvant FOLFOX-treated patients with stage
III colorectal cancers. J Pathol Transl Med. 59:50–59. 2025.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Mukohyama J, Koizumi M, Yamashita K,
Yoshimi A, Shida D and Kakeji Y: Knockdown of CDX2 Induces
microRNA-221 Up-regulation in Human colon cancer cells. Anticancer
Res. 44:3553–3556. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Voutsadakis IA: CDX2-Suppressed colorectal
cancers possess potentially targetable alterations in receptor
tyrosine kinases and other colorectal-cancer-associated pathways.
Diseases. 12:2342024. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Bae JM, Lee TH, Cho NY, Kim TY and Kang
GH: Loss of CDX2 expression is associated with poor prognosis in
colorectal cancer patients. World J Gastroenterol. 21:1457–1467.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Cecchini MJ, Walsh JC, Parfitt J,
Chakrabarti S, Correa RJ, MacKenzie MJ and Driman DK: CDX2 and Muc2
immunohistochemistry as prognostic markers in stage II colon
cancer. Hum Pathol. 90:70–79. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Fuentes Bayne HE, Suleiman R, Eiring RA,
McGarrah PW, Thome SD, Graham RP, Garcia JJ and Halfdanarson TR:
CDX2 expression as a predictive and prognostic biomarker of 5-FU
response in cancer of unknown primary. ESMO Open. 10:1055152025.
View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Slik K, Turkki R, Carpén O, Kurki S,
Korkeila E, Sundström J and Pellinen T: CDX2 loss with
microsatellite stable phenotype predicts poor clinical outcome in
stage II colorectal carcinoma. Am J Surg Pathol. 43:1473–1482.
2019. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Wouters VM, Helderman RFCPA, Cameron K,
van der Hooff SR, Torang A, van den Bergh S, Jackstadt R, Sansom
OJ, van Neerven SM and Medema JP: CDX2 downregulation regulates
intrinsic WNT pathway activation, dictating metastasis in APC and
CTNNB1 wildtype colorectal cancer. Oncogene. 44:2091–2102. 2025.
View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Arcos M, Goodla L, Kim H, Desai SP, Liu R,
Yin K, Liu Z, Martin DR and Xue X: PINK1-deficiency facilitates
mitochondrial iron accumulation and colon tumorigenesis. Autophagy.
21:737–753. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Hassan S, Mirza T, Khatoon A, Bukhari U,
Shaikh F and Karim A: BRAF mutations and the association of V600E
with CD133 and CDX2 expression in a Pakistani colorectal carcinoma
cohort. BMC Cancer. 24:11622024. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Dzung A, Saltari A, Tiso N, Lyck R, Dummer
R and Levesque MP: STK11 Prevents invasion through signal
transducer and activator of transcription 3/5 and FAK repression in
cutaneous melanoma. J Invest Dermatol. 142:1171–1182.e10. 2022.
View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Konukiewitz B, Schmitt M, Silva M, Pohl J,
Lang C, Steiger K, Halfter K, Engel J, Schlitter AM, Boxberg M, et
al: Loss of CDX2 in colorectal cancer is associated with
histopathologic subtypes and microsatellite instability but is
prognostically inferior to hematoxylin-eosin-based morphologic
parameters from the WHO classification. Br J Cancer. 125:1632–1646.
2021. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Saad RS, Ghorab Z, Khalifa MA and Xu M:
CDX2 as a marker for intestinal differentiation: Its utility and
limitations. World J Gastrointest Surg. 3:159–166. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Kaimaktchiev V, Terracciano L, Tornillo L,
Spichtin H, Stoios D, Bundi M, Korcheva V, Mirlacher M, Loda M,
Sauter G and Corless CL: The homeobox intestinal differentiation
factor CDX2 is selectively expressed in gastrointestinal
adenocarcinomas. Mod Pathol. 17:1392–1399. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Werling RW, Yaziji H, Bacchi CE and Gown
AM: CDX2, a highly sensitive and specific marker of adenocarcinomas
of intestinal origin: An immunohistochemical survey of 476 primary
and metastatic carcinomas. Am J Surg Pathol. 27:303–310. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Choi BJ, Kim CJ, Cho YG, Song JH, Kim SY,
Nam SW, Lee SH, Yoo NJ, Lee JY and Park WS: Altered expression of
CDX2 in colorectal cancers. APMIS. 114:50–54. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Sayar I, Akbas EM, Isik A, Gokce A, Peker
K, Demirtas L and Gürbüzel M: Relationship among mismatch repair
deficiency, CDX2 loss, p53 and E-cadherin in colon carcinoma and
suitability of using a double panel of mismatch repair proteins by
immunohistochemistry. Pol J Pathol. 66:246–253. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Davidsen J, Jessen SB, Watt SK, Larsen S,
Dahlgaard K, Kirkegaard T, Gögenur I and Troelsen JT: CDX2
expression and perioperative patient serum affects the adhesion
properties of cultured colon cancer cells. BMC Cancer. 20:4262020.
View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Hryniuk A, Grainger S, Savory JG and
Lohnes D: Cdx1 and Cdx2 function as tumor suppressors. J Biol Chem.
289:33343–33354. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Platet N, Hinkel I, Richert L,
Murdamoothoo D, Moufok-Sadoun A, Vanier M, Lavalle P, Gaiddon C,
Vautier D, Freund JN and Gross I: The tumor suppressor CDX2 opposes
pro-metastatic biomechanical modifications of colon cancer cells
through organization of the actin cytoskeleton. Cancer Lett.
386:57–64. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
da Silva JL, de Albuquerque LZ, Rodrigues
FR, Bastos NC, Small IA, Barroso EBC, Cordero FL, Fernandes DS,
Paulino E and de Melo AC: Exploring biomarkers and prognostic
factors in uterine carcinosarcoma: An insight into L1CAM, CDX2,
p53, and MSI status. PLoS One. 18:e02854472023. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Kato S, Koshino A, Lasota J, Komura M,
Wang C, Ebi M, Ogasawara N, Kojima K, Tsuzuki T, Kasai K, et al:
Use of SATB2 and CDX2 Immunohistochemistry to Characterize and
Diagnose Colorectal Cancer. Appl Immunohistochem Mol Morphol.
32:362–370. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Caldas ÁMC, Nunes WA, Taboada R, Cesca MG,
Germano JN and Riechelmann RP: Loss of CDX2 and high COX2 (PTGS2)
expression in metastatic colorectal cancer. Ecancermedicalscience.
18:16662024. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Yu J, Li S, Xu Z, Guo J, Li X, Wu Y, Zheng
J and Sun X: CDX2 inhibits epithelial-mesenchymal transition in
colorectal cancer by modulation of Snail expression and β-catenin
stabilisation via transactivation of PTEN expression. Br J Cancer.
124:270–280. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Chen K, Collins G, Wang H and Toh JWT:
Pathological features and prognostication in colorectal cancer.
Curr Oncol. 28:5356–5383. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Jain R, Sansoni ER, Angel J, Gleysteen JP,
Hayes DN and Owosho AA: salivary duct carcinoma with rhabdoid
features of the parotid gland with No E-Cadherin Expression: A
Report with Anti-HER2 therapy and review of the literature. Dent J
(Basel). 11:2292023. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Veraguas-Dávila D, Saéz-Ruíz D, Álvarez
MC, Saravia F, Castro FO and Rodríguez-Alvarez L: Analysis of
trophectoderm markers in domestic cat blastocysts cultured without
zona pellucida. Zygote. 30:841–848. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Bahrami A, Jafari A and Ferns GA: The dual
role of microRNA-9 in gastrointestinal cancers: OncomiR or tumor
suppressor? Biomed Pharmacother. 145:1123942022. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Perna C, Navarro A, Ruz-Caracuel I,
Caniego-Casas T, Cristóbal E, Leskelä S, Longo F, Caminoa A, Santón
A, Ferreiro R, et al: Molecular heterogeneity of high grade
colorectal adenocarcinoma. Cancers (Basel). 13:2332021. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Melincovici CS, Boşca AB, Şuşman S, Cutaş
A, Mărginean M, Ilea A, Moldovan IM, Jianu EM, Neag MA, Bulboacă AE
and Mihu CM: Assessment of mismatch repair deficiency, CDX2,
beta-catenin and E-cadherin expression in colon cancer: Molecular
characteristics and impact on prognosis and survival-an
immunohistochemical study. Rom J Morphol Embryol. 61:715–727. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Zhang JF, Qu LS, Qian XF, Xia BL, Mao ZB
and Chen WC: Nuclear transcription factor CDX2 inhibits gastric
cancercell growth and reverses epithelialtomesenchymal transition
in vitro and in vivo. Mol Med Rep. 12:5231–5238. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Ilie-Petrov AC, Cristian DA, Diaconescu
AS, Chitul A, Blajin A, Popa A, Mandi DM, Negreanu R, Vieru C,
Vrîncianu R and Ardeleanu CM: Molecular deciphering of colorectal
cancer: Exploring molecular classifications and analyzing the
interplay among molecular biomarkers MMR/MSI, KRAS, NRAS, BRAF and
CDX2-A comprehensive literature review. Chirurgia (Bucur).
119:136–155. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
da Silveira Santos JPL, Machado CJ, Junior
EP, Rodrigues J, Vidigal PT and Resende V: Immunohistochemical
predictors for intestinal and pancreatobiliary types of
adenocarcinoma of the ampulla of vater. J Gastrointest Surg.
22:1171–1178. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Puccini A, Berger MD, Zhang W and Lenz HJ:
What we know about stage II and III colon cancer: It's still not
enough. Target Oncol. 12:265–275. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Bruun J, Sveen A, Barros R, Eide PW,
Eilertsen I, Kolberg M, Pellinen T, David L, Svindland A,
Kallioniemi O, et al: Prognostic, predictive, and pharmacogenomic
assessments of CDX2 refine stratification of colorectal cancer. Mol
Oncol. 12:1639–1655. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Shi X, Lu L, Wang Z, Dai Y, Hu S, Wu Z, Yu
R, Liu T, Jiang Y, Ma Y, et al: The potential role of tumor
deposits in the prognosis and TNM staging for colorectal cancer. J
Gastrointest Oncol. 15:2473–2495. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Zeng J, Fan W, Li J, Wu G and Wu H:
KRAS/NRAS mutations associated with distant metastasis and
BRAF/PIK3CA mutations associated with poor tumor differentiation in
colorectal cancer. Int J Gen Med. 16:4109–4120. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
62
|
Dong XF, Wang Y, Tang CH, Huang BF, Du Z,
Wang Q, Shao JK, Lu HJ and Wang CQ: Upregulated WTAP expression in
colorectal cancer correlates with tumor site and differentiation.
PLoS One. 17:e02637492022. View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Delhorme JB, Bersuder E, Terciolo C, Vlami
O, Chenard MP, Martin E, Rohr S, Brigand C, Duluc I, Freund JN and
Gross I: CDX2 controls genes involved in the metabolism of
5-fluorouracil and is associated with reduced efficacy of
chemotherapy in colorectal cancer. Biomed Pharmacother.
147:1126302022. View Article : Google Scholar : PubMed/NCBI
|
|
64
|
Mochizuki K, Kudo SE, Kato K, Kudo K,
Ogawa Y, Kouyama Y, Takashina Y, Ichimasa K, Tobo T, Toshima T, et
al: Molecular and clinicopathological differences between depressed
and protruded T2 colorectal cancer. PLoS One. 17:e02735662022.
View Article : Google Scholar : PubMed/NCBI
|
|
65
|
Badia-Ramentol J, Gimeno-Valiente F,
Duréndez E, Martínez-Ciarpaglini C, Linares J, Iglesias M,
Cervantes A, Calon A and Tarazona N: The prognostic potential of
CDX2 in colorectal cancer: Harmonizing biology and clinical
practice. Cancer Treat Rev. 121:1026432023. View Article : Google Scholar : PubMed/NCBI
|
|
66
|
Aydin SG, Olmez OF, Selvi O, Geredeli C,
Ozden F, Bilici A, Acikgoz O, Karci E, Kutlu Y, Hamdard J and Aydin
A: The prognostic role of mismatch repair status and CDX-2
expression with inflammatory markers and pathological risk factors
in stage II and III colon cancer: Multicenter real-life data. J
Gastrointest Cancer. 55:227–236. 2024. View Article : Google Scholar : PubMed/NCBI
|