Introduction
Intravascular large B-cell lymphoma (IVLBCL) is a
rare and aggressive subtype of diffuse LBCL (DLBCL) characterized
by non-specific clinical manifestations, which frequently lead to a
delayed diagnosis. The incidence of IVLBCL is extremely low,
occurring in fewer than 0.5 cases per million people, with a median
age at diagnosis of 70 years, and the 5-year overall survival was
46.4% (1,2). Central nervous system (CNS)
involvement occurs in 30–40% of cases and typically manifests as
neurological deficits secondary to cerebral circulatory impairment,
making it the second most common presentation of IVLBCL (3). Cavernous sinus syndrome (CSS) is
defined by dysfunction of cranial nerves III, IV, V and VI,
manifesting as ptosis, diplopia, ophthalmoplegia and facial sensory
loss, with etiologies ranging from tumors to inflammatory
conditions (4). The overlap between
IVLBCL-related CSS and other orbital pathologies creates
significant diagnostic challenges, as radiological findings may
mimic orbital masses (5,6). To the best of our knowledge, for the
first time, the present study reports an IVLBCL case presenting as
bilateral CS syndrome with diplopia, ptosis and a right orbital
mass. This study aims to highlight the rare presentation of IVLBCL
as bilateral CS syndrome, emphasize the diagnostic challenges of
its orbital and neurological manifestations and underscore the
critical role of early tissue biopsy in diagnosis.
Case report
A 69-year-old woman presented in November 2023 with
binocular diplopia 3 days after the onset of a fever. The patient
was evaluated at the Ophthalmology Clinic of Beijing Tongren
Hospital, Capital Medical University (Beijing, China). In 2020, the
patient had been diagnosed with hypertension, and their blood
pressure had since been controlled on a combination of valsartan
(80 mg/day) and amlodipine (10 mg/day). The patient also had a
4-year history of type 2 diabetes mellitus and was taking oral
empagliflozin (25 mg/day); however, suboptimal glycemic control was
being achieved, with an elevated hemoglobin A1c level at 7.5%
(normal range: 4–6%). The patient had a past history of coronary
artery disease for which isosorbide dinitrate 10 mg 3 times daily
was being prescribed. Best-corrected visual acuity was 20/60 in
both the right [oculus dexter (OD)] and left [oculus sinister (OS)]
eyes, and intraocular pressures were within normal limits.
Bilateral nuclear cataracts (grade 2) were present, while
funduscopic examination was unremarkable. The OD exhibited normal
ocular motility, with a 3-mm pupil reactive to light. By contrast,
the OS showed ptosis, with the upper eyelid margin at the
mid-pupillary level, complete ophthalmoplegia and a fixed mydriasis
measuring 6 mm. Neurological assessment revealed isolated
hypoesthesia in the left frontal dermatome. Blood tests showed
normal red blood cell, white blood cell and platelet counts, but
elevated levels of fibrinogen degradation product (5.8 µg/ml;
normal range: 0–5 µg/ml), D-dimer (2.33 mg/l; normal range: 0–0.55
mg/l) and lactate dehydrogenase (461 U/l; normal range: 120–250
U/l). A peripheral blood smear was evaluated and showed no
abnormalities.
Routine cerebrospinal fluid (CSF) tests and
biochemical indices were normal. CSF analyses, including ink
staining, Gram staining, acid-fast staining, and bacterial and
fungal cultures, were all negative. CSF pressure was 105 mm
H2O (80–180 mmH2O), and no tumor cells were
identified on CSF pathological examination. Orbital MRI revealed a
15.9×5.9 mm lesion at the right orbital apex, while
contrast-enhanced MRI showed abnormal thickening and enhancement of
bilateral CSs, extending to the adjacent dura mater and clivus
(Fig. 1A). A CT scan of the skull
base, orbit and sinuses showed no bony destruction and adjacent
hyperostosis (Fig. 1B and C). An
idiopathic orbital inflammatory pseudotumor was initially
suspected, and intravenous methylprednisolone pulse therapy was
initiated (120 mg on days 1–3 and 80 mg on days 4–6) but was
discontinued due to severe gastrointestinal symptoms. The ptosis
and complete external ophthalmoplegia of the left eye showed no
improvement. Over the subsequent week, the patient developed
complete bilateral external ophthalmoplegia, severe bilateral
blepharoptosis completely covering the pupils, and bilateral fixed
and dilated pupils (OD, 5 mm; OS, 6mm). Follow-up MRI revealed
multifocal abnormal enhancement in multiple areas, including the
bilateral CSs, right orbital compartment, sinonasal cavities,
clivus and marrow cavity of the left pterygoid process (Fig. 1D-H).
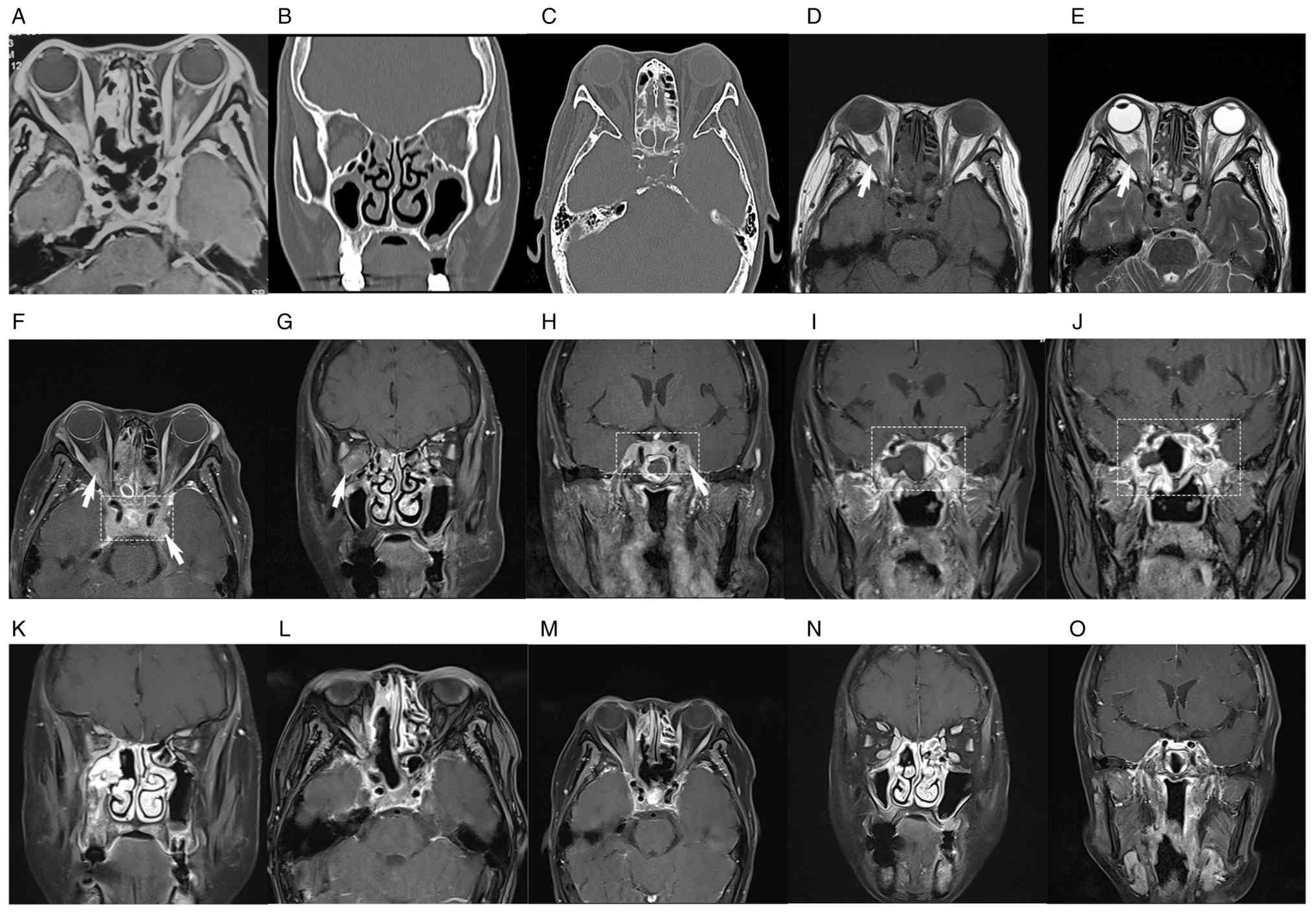 | Figure 1.MRI findings and CT scan images of the
patient. (A) MRI of patient at the first visit. (B) Coronal CT scan
of the skull base, orbit and sinuses at the first visit. (C) Axial
CT scans of the skull base, orbit and sinuses at the first visit.
(D-H) Baseline MRI before initiation of the R-CHOP regimen. (D)
Axial T1-weighted imaging showing a 1.2×1.6×0.9-cm lesion at the
right orbital apex (white arrow). (E) Axial T2-weighted imaging.
(F) Axial and (G) coronal T1 fat-suppressed gadolinium-enhanced MRI
images demonstrating masses located in the right intraconal orbital
space (white arrow), left intraorbital optic nerve sheaths, nasal
cavity, paranasal sinuses and bilateral CSs (rectangular marquee).
(H) Coronal contrast-enhanced MRI of the bilateral CSs (rectangular
marquee). (I) MRI of bilateral CSs after 3 cycles of R-CHOP
combined with a BTK inhibitor. (J) MRI revealing an enlarged CS
lesion after 4 cycles of R-CHOP combined with a BTK inhibitor
compared with that after 3 cycles of the regimen (rectangular
marquee). (K) Coronal contrast-enhanced MRI of skull base, orbit
and sinuses after six cycles of R-CHOP combined with BTK inhibitor
and two courses of HD-MTX. (L) Axial contrast-enhanced MRI of the
CS, orbit and sinuses after 6 cycles of the regimen. (M) Axial
contrast-enhanced MRI of the CS, orbit and sinuses post-treatment
corresponding to images F before treatment. (N) Coronal
contrast-enhanced MRI of the orbit and sinuses post-treatment
corresponding to images G before treatment. (O) Coronal
contrast-enhanced MRI of the CS post-treatment corresponding to
images H before treatment. After treatment, marked regression of
the lesions in the CSs and the right orbit was observed. MRI,
magnetic resonance imaging; CT, computed tomography; CS, cavernous
sinus; BTK, Bruton tyrosine kinase; HD-MTX, high-dose methotrexate;
R-CHOP, rituximab, cyclophosphamide, doxorubicin, vincristine and
prednisolone. |
Subsequently, a transnasal endoscopic sphenoid sinus
biopsy was performed, leading to a definitive diagnosis of IVLBCL.
Histopathological examination was performed on the sphenoid sinus
biopsy specimens. For histopathological analysis, the specimens
were fixed in 4% neutral formalin (Beijing Yili Fine Chemical Co.,
Ltd.) at room temperature for 24 h, embedded in paraffin and
sectioned at a thickness of 4 µm. Hematoxylin and eosin (H&E)
staining was performed at room temperature (5 min for hematoxylin,
3 min for eosin) using reagents from Beijing Yili Fine Chemical
Co., Ltd. Examination with a Nikon ECLIPSE Ci Series Upright
Clinical Microscope (Nippon Kogaku) revealed neoplastic lymphocytes
proliferating within vascular lumina, with round to oval nuclei,
coarse chromatin and prominent nucleoli (×100, ×400 magnification).
Immunohistochemical analysis was performed using the Dako EnVision
System (Agilent Technologies, Inc.). Endogenous peroxidase was
blocked with 3% hydrogen peroxide at room temperature for 10 min
(no serum blocking reagent was used). All primary antibodies were
mouse monoclonal anti-human, purchased from Beijing Zhongshan
Golden Bridge Biotechnology Co., Ltd. (OriGene Technologies, Inc.),
diluted in PBS and incubated at 25°C for 30 min. The primary
antibodies used included: CD20 (cat. no. ZM-0036; dilution 1:150),
CD19 (cat. no. ZM-0027; dilution 1:100), CD79α (cat. no. ZM-0028;
dilution 1:100), multiple myeloma oncogene 1 (MUM1; cat. no.
ZA-0511; dilution 1:150), B-cell lymphoma 2 (BCL-2; cat. no.
ZM-0024; dilution 1:100), leukocyte common antigen (LCA/CD45; cat.
no. ZM-0039; dilution 1:150), CD5 (cat. no. ZM-0037; dilution
1:150), CD3 (cat. no. ZM-0062; dilution 1:150), CD10 (cat. no.
ZM-0048; dilution 1:100), CD23 (cat. no. ZM-0050; dilution 1:100),
BCL-6 (cat. no. ZA-0512; dilution 1:150), terminal deoxynucleotidyl
transferase (TdT; cat. no. ZA-0513; dilution 1:100), CyclinD1 (cat.
no. ZA-0514; dilution 1:150), CD34 (cat. no. ZM-0046; dilution
1:150), Ki-67 (cat. no. ZA-0502; dilution 1:150), CD61 (cat. no.
ZM-0063; dilution 1:100), myeloperoxidase (MPO; cat. no. ZM-0064;
dilution 1:150), CD3 (cat. no. ZM-0062; dilution 1:150) and CD117
(cat. no. ZM-0437; dilution 1:100). The secondary antibody was
horseradish peroxidase-conjugated, provided in the Dako EnVision
System, and incubated at 25°C for 30 min. All histopathological and
immunohistochemical images were captured using a Nikon ECLIPSE-CI
light microscope (Nippon Kogaku) at ×200 magnification.
Immunohistochemistry demonstrated numerous medium-sized atypical
lymphocytes proliferating in small vessel lumina, positive for
B-lineage antigens (CD20, CD19 and CD79α), MUM1, BCL-2, LCA and
CD5, but negative for CD3, CD10, CD23, BCL-6, TdT and CyclinD1
(Fig. 2A-P). CD34
immunohistochemical staining was positive in vascular endothelial
cells, facilitating the observation of the intravascular or
extravascular distribution of lymphocytes. The Ki-67 proliferation
index was 80%. Immunohistochemistry demonstrated a typical pattern
of intravascular tumor cell infiltration, with a small subset of
lymphocytes migrating to extravascular tissues. Epstein-Barr
virus-encoded RNA (EBER) detection was performed by in situ
hybridization (ISH) using a commercial EBER probe purchased from
Beijing Zhongshan Golden Bridge Biotechnology Co., Ltd. (OriGene
Technologies, Inc.), with nasopharyngeal carcinoma tissue as
positive control and reactive lymph node hyperplasia as negative
control. The EBER ISH result was negative.
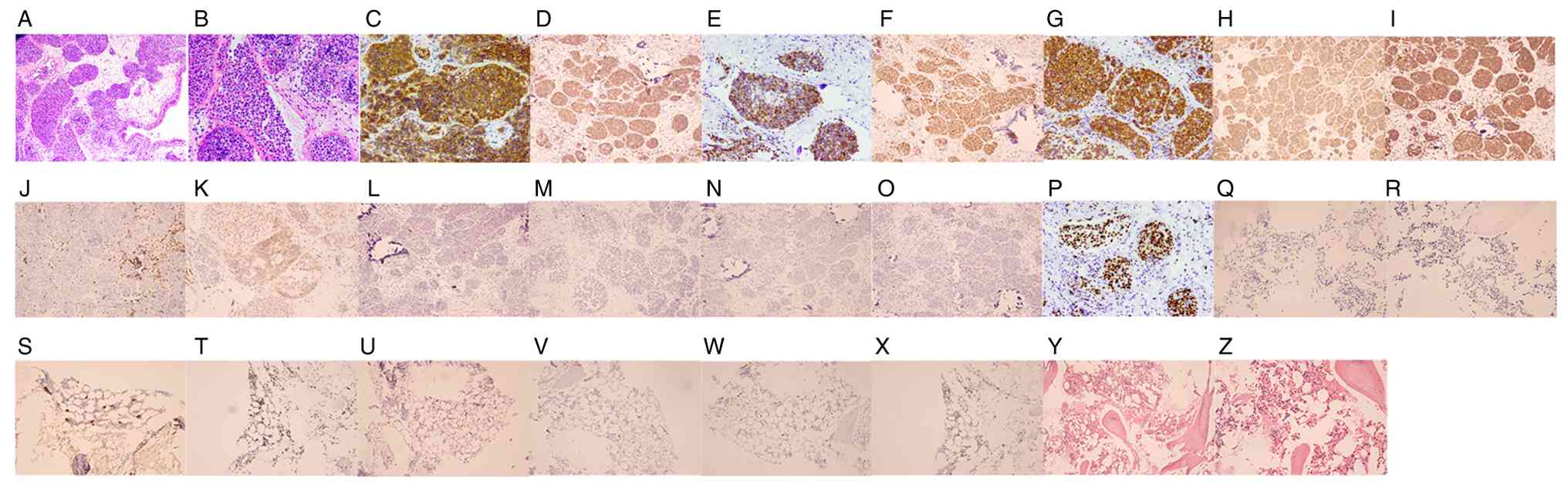 | Figure 2.Histopathological findings. (A)
H&E staining showing diffuse proliferation of neoplastic
lymphocytes within the vascular lumina (×100 magnification). (B)
H&E staining (high-power view) revealing intravascular tumor
cells with marked atypia, characterized by enlarged nuclei and a
high nuclear-to-cytoplasmic ratio (×400 magnification). (C-O) IHC
demonstrating positive staining for (C) CD20, (D) CD19, (E) CD79α,
(F) MUM1, (G) BCL-2, (H) LCA and (I) CD5 in tumor cells (×400
magnification), but negative results for (J) CD3, (K) CD10, (L)
CD23, (M) BCL-6, (N) TdT and (O) cyclin D1(×400 magnification). (P)
IHC staining demonstrating high tumor proliferative activity with a
Ki-67 index of ~80% (×100 magnification). (Q-Z) Bone marrow biopsy.
IHC staining of bone marrow smears was negative for (Q) CD20 and
(R) EBER, but positive for (S) CD61 and (T) Ki67; occasional
scattered (U) CD3-, (V) CD34-and (W) CD117-positive cells were also
noted. (X) E-cad red-positive staining. (Y and Z) Active
proliferation of bone marrow tissue, with no neoplastic cells under
(Y) low- and (Z) middle-power microscopy (×200 magnification).
H&E, hematoxylin and eosin; IHC, immunohistochemistry. |
Based on these findings, the patient was ultimately
diagnosed with IVLBCL of non-germinal center B-cell (non-GCB)
phenotype. Minimal residual disease assessment was negative.
Fluorescence in situ hybridization and cytogenetic studies
were not performed due to the patient's refusal. Bone marrow biopsy
showed mild marrow hypercellularity, a normal myeloid-to-erythroid
ratio and identifiable megakaryocytes, with no definitive tumor
cells identified. Immunohistochemical staining of bone marrow
smears was negative for CD20 and EBER, but positive for CD61, MPO
and Ki-67; occasional scattered CD3-, CD34- and CD117-positive
cells were also noted (Fig. 2Q-Z).
The results for MPO are not shown but are based on the pathology
report.
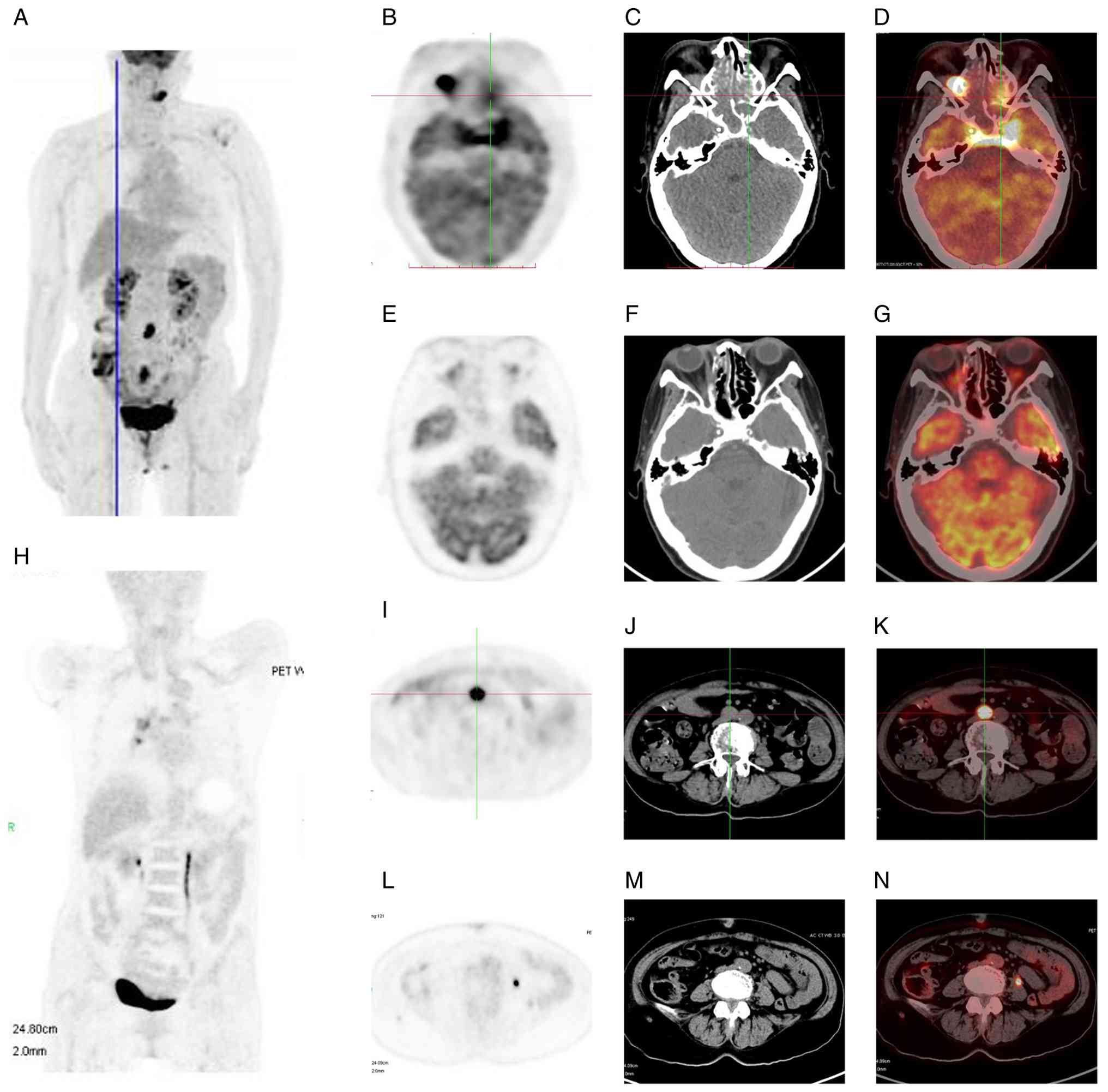
Positron emission tomography-computed tomography
(PET-CT) subsequently revealed multiple lesions with abnormally
increased fluorodeoxyglucose (FDG) uptake, involving the ethmoid
sinus, sphenoid sinus, bilateral CSs, right orbital mass,
periaortic and pelvic lymph nodes, sacral spinal canal and left
mandibular medullary cavity (Fig. 3A,
C-E and I-K). The right orbital apex and periaortic
lymphadenopathy showed the highest FDG uptake, with the maximum
standardized uptake value (SUVmax) of 11.72 and 12.77,
respectively. SUVmax values for the sphenoid sinus,
bilateral CSs, sacral spinal canal and left mandibular medullary
cavity were 6.31, 9.37, 8.69 and 8.57, respectively.
Following four initial cycles of rituximab,
cyclophosphamide, doxorubicin, vincristine and prednisolone
(R-CHOP) (600 mg rituximab on day 0, 1 g cyclophosphamide on day 1,
100 mg doxorubicin on day 1, 4 mg vincristine on day 1, 100 mg/day
prednisolone on days 0–5) combined with the Bruton tyrosine kinase
(BTK) inhibitor (320 mg/day Zanubrutinib), the frontal numbness and
pain of the patient was improved. However, MRI revealed an enlarged
CS lesion (Fig. 1I and J), and the
patient agreed to undergo high-dose methotrexate (HD-MTX) therapy
(3.5 g/m2 intravenously). After six cycles of R-CHOP
combined with BTKi and two courses of HD-MTX, MRI showed a marked
reduction in the CS lesion (Fig. 1K and
L); however, complete remission had not yet been achieved. To
further pursue complete remission, the patient agreed to receive an
additional two cycles of R-CHOP. Therefore, two cycles of the same
regimen (600 mg rituximab on day 0, 1 g cyclophosphamide on day 1,
100 mg doxorubicin on day 1, 4 mg vincristine on day 1, 100 mg/day
prednisolone on days 0–5 and 320 mg/day Zanubrutinib, each cycle
lasting 21 days) and two courses of HD-MTX (3.5 g/m2
intravenously) were administered. Following treatment, the
diplopia, frontal numbness, bilateral ptosis and ophthalmoplegia
resolved completely. Post-treatment MRI and PET-CT demonstrated
marked reduction of CS enhancement and orbital mass resolution
(Figs. 1M-O and 3B, F-H and L-N). Zanubrutinib (320 mg/day)
was administered as maintenance therapy after chemotherapy.
However, diplopia recurred again 3 months later. The patient
received volumetric-modulated arc radiotherapy at 36 Gy in 18
fractions to the elective clinical target volume, including the
bilateral orbits, bilateral nasal cavities, sphenoid sinus,
bilateral CSs and sellar region. After completing radiotherapy, the
patient declined further in-person follow-up visits. However, a
recent telephone follow-up was successfully conducted at 1 year
post-treatment, at which time the patient reported being alive,
without ptosis and with nearly normal ocular motility.
Discussion
Current diagnostic modalities for IVLBCL, including
MRI, PET-CT and plasma/CSF biomarkers, exhibit inadequate
sensitivity and specificity. Histopathological biopsy thus remains
the gold standard for definitive diagnosis. A previous study has
advocated paranasal sinus biopsy for evaluating CS lesions
(7). In the present case,
transnasal endoscopic biopsy proved advantageous, offering both
safety and efficiency while circumventing the substantial
complications associated with craniotomy-based biopsy. Although a
biopsy of the right orbital mass was deferred due to safety
concerns, its consistency with a CS tumor can be reasonably
inferred from characteristic MRI/PET/CT findings and a favorable
clinical response to R-CHOP combined with BTKi and HD-MTX.
The underlying mechanism driving the angiotropism of
neoplastic cells in IVLBCL may be associated with their
immunophenotypic and molecular features. Adhesion molecules, such
as CD29 and CD54, which are critical for lymphocyte trafficking and
transvascular migration, have been consistently absent in IVLBCL.
This deficiency likely contributes to the propensity of lymphoma
cells to localize within blood vessels (8). Besides, B-cell homeostatic chemokine
receptors [C-X-C chemokine receptor type 4 (CXCR4), CXCR5, C-C
chemokine receptor type 6 (CCR6) and CCR7] play crucial roles in
cell homing and mediate the extravascular circulation of B cells.
Downregulated expression of these receptors in IVLBCL may partly
explain the intravascular localization of tumor cells (9). Matrix metalloproteinases (MMPs) are
key enzymes that degrade the extracellular matrix during cellular
invasion and regulate blood-brain barrier (BBB) permeability under
specific conditions. Notably, MMP-2 and MMP-9 are expressed in
patients with primary CNS lymphoma (PCNSL) but are absent in IVLBCL
cases, suggesting that MMPs may modulate the biological behavior
underlying the intravascular and disseminated distribution patterns
of IVLBCL (10). However,
extravascular cell growth has been documented in IVLBCL cases with
CNS involvement (11). Consistent
with this, immunohistochemical staining in the present case
displayed both intravascular and extravascular distribution.
Both the CS and orbital lesions of the present
patient appeared isointense to cortical signal intensity on MRI T1-
and T2-weighted images with heterogeneous enhancement. MRI findings
were also consistent with an inflammatory pseudotumor, meningioma
and lymphoma (12). The initial
blood tests, CSF tests and peripheral blood smears revealed no
evidence of a neoplasm. However, intravenous methylprednisolone
pulse therapy failed to prevent disease progression, thus ruling
out the diagnosis of an inflammatory pseudotumor. On MRI,
meningiomas typically exhibit an isointense signal relative to gray
matter on all sequences, with moderate, homogeneous contrast
enhancement, and are typically accompanied by an enhancing ‘dural
tail’ sign (13). The presence of
calcifications and adjacent hyperostosis on CT is highly suggestive
of meningioma (13). However, the
present patient's MRI showed ill-defined lesion margins and
heterogeneously enhancing lesions, with no broad-based dural
attachment or enhancing ‘dural tail’ sign. Moreover, the CT scan
demonstrated no bony destruction involving the skull base or
sinuses, and no adjacent hyperostosis. The CS and orbit lesions
also exhibited no calcifications on CT. Therefore, a diagnosis of
meningioma was deemed unlikely. Furthermore, meningiomas that
encase the cavernous internal carotid artery (ICA) often narrow its
lumen, while the lesion of the present patient enlarged the CS
without compressing the ICA, a feature consistent with the typical
MRI findings of lymphoma (14).
Systemic lymphomas involving the orbit and skull
base reported in previous studies include DLBCL, primary marginal
zone BCL (MZBCL), Burkitt lymphoma (BL), mantle cell lymphoma and
natural killer/T-cell lymphoma (NKTCL) (15,16).
BL with involvement of the orbit, skull base and CS almost always
occurs in children or young adults who are aged 2 to 39 years old
(17). Furthermore, some patients
with BL plus intracranial and intraorbital involvement have
concurrent human immunodeficiency virus (HIV) and Epstein-Barr
virus (EBV) infection (18,19). The patient in the present case was
an 69-year-old woman with no evidence of HIV or EBV infection, a
clinical feature distinct from that typical of BL. Patients with
NKTCL plus orbital and CNS involvement exhibit characteristic MRI
features typified by an abscess-like appearance on neuroimaging
studies (20). This appearance
arises from the angiocentric growth pattern and associated tissue
destruction, which leads to zonal necrosis (20), a finding inconsistent with the MRI
findings of the present patient. Mucosa-associated lymphoid tissue
lymphoma, an indolent subtype of MZBCL, uncommonly involves the CNS
and typically lacks any associated extracranial involvement
(21). However, DLBCL and IVLBCL
frequently present with multisystem involvement, with CNS
involvement being a common manifestation. PET-CT confirmed that the
present patient had multisystem involvement, including the CSs, a
structure within the CNS. Within the literature, a previous study
reported a case of adult-onset DLBCL presenting as a right CS mass
extending through the superior orbital fissure and into the
intraconal space of the right orbit (17). These findings were radiologically
analogous to those of the present patient, yet without multisystem
involvement beyond the orbit and middle cranial fossa. Therefore,
it is difficult to distinguish IVLBCL from DLBCL based on clinical
and radiological manifestations alone; the gold standard for their
differentiation remains pathological analysis. In previous studies,
no cases of IVLBCL presenting with isolated orbital lesions have
been reported; all IVLBCL patients with orbital involvement exhibit
concurrent involvement of the CNS or skull base structures such as
the CS (22–24). By contrast, primary orbital lesions
may occur in patients with DLBCL in the absence of systemic
lymphoma, and orbital manifestations may precede the diagnosis of a
systemic DLBCL (15,25,26).
Therefore, orbital lesions in IVLBCL are likely secondary or
metastatic rather than primary in origin. Finally, the diagnosis of
IVLBCL in the present case was confirmed by sphenoid sinus
biopsy.
In the present case, it is hypothesized that
malignant lymphocytes initially infiltrated and propagated along
the vessel lumina of the left CS, leading to vascular infarction
secondary to complete vessel occlusion. Subsequent ischemia and
hypoxia of the cranial nerves within the CS led to the observed
neurological deficits. Over time, malignant lymphocytes extended to
the contralateral CS via the anterior and posterior intercavernous
sinuses, causing impairments of the right cranial nerves.
Histopathological analysis demonstrated that tumor cells were not
strictly confined to the intravascular space, with focal
extravascular infiltration observed under the microscope. This may
explain the orbital involvement in this patient, with tumor cells
spreading into the right orbit through the superior orbital
fissure. Notably, although the patient initially presented with
left-sided ocular symptoms, MRI already revealed abnormal signals
in the right CS and a lesion at the right orbital apex. This
clinical feature suggests that imaging changes in IVLBCL may
precede clinical manifestation, possibly as the early
ophthalmoplegia of the right eye was subtle and easily overlooked.
Another factor is that early in the disease, multiple ocular motor
cranial neuropathies in the right eye may be compressed and encased
by the orbital mass, yet malignant lymphocytes primarily infiltrate
the nerve sheath rather than the nerve fibers. Acute ischemia and
hypoxia of these nerves, caused by intravascular lymphocyte
proliferation, likely occur before compressive neuropathy. This
dissociation between imaging findings and clinical symptoms
indicates that even with early extravascular infiltration, IVLBCL
originating from the CS may not immediately produce significant
functional impairments.
IVLBCL cells display an immunophenotype consistent
with mature peripheral B cells, characterized by consistent
expression of pan-B-cell markers. The vast majority of cases
exhibit strong CD20 expression, while CD20-negative cases typically
remain positive for other pan-B-cell markers (such as CD79a and
paired box protein Pax-5). According to the Hans algorithm, 82–87%
of IVLBCL cases are classified as non-GCB, despite the absence of
gene expression profiling in these tumors (20). Immunohistochemical analysis reveals
that the expression frequencies of CD5, CD10, BCL6 and MUM1 are
22–75, 13–22, 22–26 and 75–80%, respectively (16). Despite variable frequencies, none of
these markers correlate with prognostic differences (27,28).
Previous research has shown that CD5(+)/CD10(−) IVLBCL is
correlated with a higher incidence of thrombocytopenia and bone
marrow/peripheral blood involvement, but a lower rate of
neurological abnormalities compared with the CD5(−)/CD10(−) subtype
(29). No significant differences
were observed in other clinical features or survival outcomes
between the two subtypes (29).
Notably, the present case, displaying a CD5(+)/CD10(−)
immunophenotype, presented with neurological manifestations such as
cranial nerve palsies, underscoring the need for further studies on
the heterogeneity of clinical presentation and immunophenotype in
IVLBCL.
IVLBCL is a rare malignancy with no established
standard treatment. Although the R-CHOP regimen has been shown to
significantly improve survival rates in IVLBCL patients, the
prognosis remains guarded, particularly in those with CNS
involvement (30,31). In CNS-involved IVLBCL, 1- and 2-year
overall survival rates were 44.2 and 22.2%, respectively, with
treatment approaches varying across HD-MTX plus R-CHOP, MTX,
cytarabine and intrathecal MTX (1).
Notably, BTKis, which can block the NF-κB pathway and penetrate the
BBB, have shown promising outcomes in patients with IVLBCL and CNS
involvement (31). Previous studies
have reported frequent mutations in NF-κB pathway genes in IVLBCL
and a substantial number of CNS-involved patients sensitive to
treatment with BTKis (31,32). Notably, zanubrutinib, a selective
BTKi, in combination with R-CHOP, achieved complete responses in
all patients, including those with CNS involvement (1). Additionally, a patient with primary
CNS lymphoma, who could not tolerate intensive therapy or undergo
consolidative autologous stem cell transplant (ASCT) or whole-brain
radiotherapy, remained in remission for 18 months while undergoing
monotherapy with acalabrutinib, a BTKi (33). Although no standard consolidation or
maintenance strategy has been established for conventional BCL with
CNS involvement, accumulating evidence indicates that BTKis
demonstrate favorable efficacy and tolerability in highly
aggressive lymphomas with a high risk of relapse, such as IVLBCL
and PCNSL, particularly in elderly patients or those ineligible for
ASCT (31,33–37).
Therefore, maintenance therapy with zanubrutinib was initiated for
the present patient after obtaining informed consent.
In the present case, four initial cycles of R-CHOP
combined with BTKi therapy yielded poor efficacy, suggesting that
the R-CHOP regimen may not be an effective induction regimen for
IVLBCL with CS involvement. Although R-CHOP demonstrates efficacy
in systemic DLBCL, its outcomes remain suboptimal in cases with CNS
involvement (38). Key drugs of the
R-CHOP regimen, including vincristine and doxorubicin, as well as
the large monoclonal antibody rituximab, exhibit limited
permeability across the BBB and blood-CSF barrier, leading to
inadequate drug exposure at skull base structures such as the CS.
Besides, BTKis generally demonstrate limited BBB penetration.
Zanubrutinib, a second-generation BTKi, has improved BBB
permeability compared with first-generation agents; however, its
efficacy may be restricted in IVLBCL cases that are not dependent
on the BCR/NF-κB signaling pathway (31). Furthermore, immune escape and immune
privilege may lead to the limited efficacy of R-CHOP. However, in
the present study, following two courses of HD-MTX, a marked
reduction in the size of the CS lesion was observed, highlighting
the critical role of BBB-penetrating agents in disease control.
Accordingly, for patients with IVLBCL plus orbit and CS
involvement, a treatment strategy combining R-CHOP with
BBB-penetrating agents such as HD-MTX may be more appropriate. In
the present patient, maintenance therapy with a BTKi was not
effective. These findings highlight the need to explore
individualized chemotherapy regimens for patients with IVLBCL plus
CNS and skull base involvement, as well as to develop more targeted
therapeutic strategies employing BBB-penetrating agents.
A previous study reported comparable rates of CNS
relapse in patients with DLBCL treated with HD-MTX and those
receiving intrathecal MTX (IT-MTX) (39). In a cohort of 88 patients with
aggressive BCL involving extralymphatic craniofacial sites, the
2-year rate of CNS disease was 4.2% among 88 patients who received
IT-MTX prophylaxis, compared with 2.3% among 191 patients who did
not (40). Moreover, a matched
case-control retrospective study of patients with PCNSL treated
with HD-MTX, with or without adjunctive IT-MTX, demonstrated no
significant difference in survival, disease control or
neurotoxicity between the two groups (41). Therefore, IT-MTX does not confer
superiority over HD-MTX in either the treatment or prevention of
BCL with CNS and skull involvement. In addition, an increasing
number of case reports have described adverse effects associated
with IT-MTX, including myelopathy and severe neurological
complications (42,43). Given the absence of abnormalities on
CSF examination in the present patient, HD-MTX monotherapy was
selected rather than combination therapy with IT-MTX.
In conclusion, IVLBCL is often misdiagnosed as an
inflammatory disorder, but the diagnosis should be considered when
patients sequentially develop CS syndrome, fail to respond to
steroid therapy, and particularly exhibit a dissociation between
imaging findings and clinical manifestations. A prompt biopsy is
essential to establish a definitive diagnosis. BTKi therapy in
combination with HD-MTX plus R-CHOP may provide an effective
therapeutic strategy for controlling IVLBCL with skull base and
orbital involvement. Nevertheless, more effective maintenance or
consolidation treatment strategies should be actively explored.
Acknowledgements
Not applicable.
Funding
Funding was provided by the Natural Science Foundation of China
(grant no. 82205194).
Availability of data and materials
The data generated in the present study may be
requested from the corresponding author.
Authors' contributions
QW and LBJ conceived and designed the study, and
contributed to manuscript drafting. BY and YJ collected and
analyzed the MRI and CT images from the patient records,
interpreted the radiological findings and contributed to the
discussion of imaging characteristics in the manuscript. XH
performed the histological examination of the tumor, including IHC
staining and ISH assays, and contributed to the pathological
diagnosis. HL was involved in revising the manuscript for important
intellectual content, critically reviewed the pathological and
radiological data, and provided clinical guidance for the study
design and patient management. All authors have read and approved
the final version of the manuscript. QW and LBJ have checked and
confirmed the authenticity and integrity of all raw data.
Ethics approval and consent to
participate
This study was conducted in accordance with the
principles expressed in the Declaration of Helsinki.
Patient consent for publication
Written and verbal consent was obtained from the
patient for publication of the case report and any accompanying
images.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Li Y, Li S, Liu X, Xue W, Han L, Li Y,
Zhang X and Zhang M: Clinical features and prognostic factors of
intravascular large B-cell lymphoma: A cohort study of 20 patients
from 2018 to 2024. Oncologist. 30:oyaf1562025. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Rajyaguru DJ, Bhaskar C, Borgert AJ, Smith
A and Parsons B: Intravascular large B-cell lymphoma in the United
States (US): A population-based study using Surveillance,
epidemiology, and end results program and National cancer database.
Leuk Lymphoma. 58:1–9. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Geer M, Roberts E, Shango M, Till BG,
Smith SD, Abbas H, Hill BT, Kaplan J, Barr PM, Caimi P, et al:
Multicentre retrospective study of intravascular large B-cell
lymphoma treated at academic institutions within the United States.
Br J Haematol. 186:255–262. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Kim TR, Bae KN, Son JH, Shin K, Kim H, Ko
H, Kim B and Kim MB: A case of cavernous sinus Syndrome due to
extranodal diffuse large B-cell lymphoma. Ann Dermatol. 35 (Suppl
2):S300–S303. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Wang D, Marous CL, Ozay F, Timashpolsky A,
Gulati RD, Gottesman SRS, Boruk M, Shinder R and Hodgson NM:
Intravascular large B-cell lymphoma diagnosed by nasal biopsy in a
patient presenting with bilateral ptosis and ophthalmoplegia.
Orbit. 42:450–454. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Jain R, Sawhney S, Koul RL and Chand P:
Tolosa-Hunt syndrome: MRI appearances. J Med Imaging Radiat Oncol.
52:447–451. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Zhang AS, Jonker BP, Morris CL, Campbell
RG, Alvarado R, Winder M, Sacks R, Seresirikachorn K and Harvey RJ:
Endoscopic endonasal biopsy for diagnosis of undifferentiated
lesions of the cavernous sinus. World Neurosurg. 175:e391–e396.
2023. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Ponzoni M, Arrigoni G, Gould VE, Del Curto
B, Maggioni M, Scapinello A, Paolino S, Cassisa A and Patriarca C:
Lack of CD 29 (beta1 integrin) and CD 54 (ICAM-1) adhesion
molecules in intravascular lymphomatosis. Hum Pathol. 31:220–226.
2000. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Kasuya A, Fujiyama T, Shirahama S,
Hashizume H and Tokura Y: Decreased expression of homeostatic
chemokine receptors in intravascular large B-cell lymphoma. Eur J
Dermatol. 22:272–273. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Kinoshita M, Izumoto S, Hashimoto N,
Kishima H, Kagawa N, Hashiba T, Chiba Y and Yoshimine T:
Immunohistochemical analysis of adhesion molecules and matrix
metalloproteinases in malignant CNS lymphomas: A study comparing
primary CNS malignant and CNS intravascular lymphomas. Brain Tumor
Pathol. 25:73–78. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Poropatich K, Dittmann D, Chen YH, Raparia
K, Wolniak K and Gao J: A small case series of intravascular large
B-cell lymphoma with unexpected findings: Subset of cases with
concomitant extravascular central nervous system (CNS) involvement
mimicking primary CNS lymphoma. J Pathol Transl Med. 51:284–291.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Jacobs D and Galetta S: Diagnosis and
management of orbital pseudotumor. Curr Opin Ophthalmol.
13:347–351. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Bonneville F, Jäger HR and Smirniotopoulos
JG: Differential diagnosis of intracranial masses. Diseases of the
Brain, Head and Neck, Spine 2024–2027: Diagnostic Imaging, Chapter
8. Hodler J, Kubik-Huch RA and Roos JE: Springer; Cham, CH: 2024,
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Munawar K, Nayak G, Fatterpekar GM, Sen C,
Zagzag D, Zan E and Hagiwara M: Cavernous sinus lesions. Clin
Imaging. 68:71–89. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Fritzhand SJ, Esmaeli B, Sun J and Debnam
JM: Primary disease sites and patterns of spread in cases of
neurolymphomatosis in the orbit associated with lymphoma. Cancer
Imaging. 21:392021. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Choi HK, Cheon JE, Kim IO, Youn BJ, Jung
AY, Shin SM, Kim WS and Yeon KM: Central skull base lymphoma in
children: MR and CT features. Pediatr Radiol. 38:863–867. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Ko F and Subramanian PS: Orbital and
cavernous sinus lymphoma masquerading as post-herpetic neuralgia.
Neuroophthalmology. 35:27–31. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Ruiz-Ortiz M, Azcarate-Diaz FJ,
Galindo-Rodriguez D, Torres-Calcines N and Calleja-Castano P:
Cavernous sinus syndrome as the initial symptom of Burkitt's
lymphoma: A case report and literature review. Rev Neurol.
69:249–254. 2019.(In Spanish). PubMed/NCBI
|
|
19
|
Reyneke F, Mokgoro N, Vorster M and
Sathekge M: Burkitt lymphoma and cavernous sinus syndrome with
breast uptake on 18F-FDG-PET/CT: A case report. Medicine
(Baltimore). 96:e86872017. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Chen CS, Miller NR, Lane A and Eberhart C:
Third cranial nerve palsy caused by intracranial extension of a
sino-orbital natural killer T-cell lymphoma. J Neuroophthalmol.
28:31–35. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Yang CC, Chen TY, Tsui YK and Ko CC:
Primary marginal zone B-cell lymphoma of the cavernous sinus: A
case report and review of the literature. BMC Med Imaging.
21:252021. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Roditi E, Panicker S and Fung AT:
Intravascular large B-cell lymphoma of the eye: Literature review
and new findings. Asia Pac J Ophthalmol (Phila). 13:1000532024.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Berbos ZJ, Lee MS, Zaldivar RA, Pambuccian
S and Harrison AR: Intravascular lymphoma presenting as an orbital
mass lesion: A case report. Orbit. 29:91–93. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Sato T, Goto H, Toratani A, Yamagata N,
Ashihara E, Oku N, Inaba T, Fujita N, Shimazaki C and Nakagawa M:
Neoplastic angioendotheliosis presenting Tolosa-Hunt syndrome,
intraspinal invasion and intraorbital tumor. Rinsho Ketsueki.
35:557–561. 1994.(In Japanese). PubMed/NCBI
|
|
25
|
Singh S and Ali MJ: Lymphoproliferative
tumors involving the lacrimal drainage system: A major review.
Orbit. 39:276–284. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Liu H, Ren YJ, Chen YM, Yang JR, Yang J,
Cai FM, Lei T and Liu HL: Clinical and histopathological
characteristics of 19 cases of orbital diffuse large B-cell
lymphoma. Zhonghua Yan Ke Za Zhi. 62:52–58. 2026.(Chinese).
PubMed/NCBI
|
|
27
|
Della Mura M, Sorino J, Angiuli FE,
Cazzato G, Gaudio F and Ingravallo G: Intravascular lymphoma: A
unique pattern underlying a protean disease. Cancers (Basel).
17:23552025. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Ponzoni M, Campo E and Nakamura S:
Intravascular large B-cell lymphoma: A chameleon with multiple
faces and many masks. Blood. 132:1561–1567. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Murase T, Yamaguchi M, Suzuki R, Okamoto
M, Sato Y, Tamaru J, Kojima M, Miura I, Mori N, Yoshino T and
Nakamura S: Intravascular large B-cell lymphoma (IVLBCL): A
clinicopathologic study of 96 cases with special reference to the
immunophenotypic heterogeneity of CD5. Blood. 109:478–485. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Seegobin K, Li Z, Alhaj Moustafa M, Majeed
U, Wang J, Jiang L, Kuhlman J, Menke D, Li K, Kharfan-Dabaja MA, et
al: Clinical characteristics, prognostic indicators, and survival
outcomes in intravascular lymphoma: Mayo Clinic experience
(2003–2018). Am J Hematol. 97:1150–1158. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Shao F, Su W, Zhao X, He J, Wang X, Guo F
and Xiao H: Successful treatment of hemophagocytic intravascular
large B-cell lymphoma with CNS involvement with BTK inhibitor
combined with rituximab and high-dose methotrexate. Ther Adv
Hematol. 15:204062072412707882024. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Schrader AMR, Jansen PM, Willemze R,
Vermeer MH, Cleton-Jansen AM, Somers SF, Veelken H, van Eijk R,
Kraan W, Kersten MJ, et al: High prevalence of MYD88 and CD79B
mutations in intravascular large B-cell lymphoma. Blood.
131:2086–2089. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Allison E, Campbell A, Watson AM and
Beaton B: Acalabrutinib may offer a new therapeutic approach for
consolidation and maintenance of primary CNS lymphoma with
expression of MYD88 and CD79B gene variants: A case report and
literature review of primary CNS lymphoma in the BTKi Era. Int J
Mol Sci. 26:105212025. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Yu L, Ping N, Zou R, Zhu Q, Li J, Zhang X,
Xia F, He J, Tu J, Kong D, et al: Sustained remission with PD-1 and
BTK inhibitors maintenance after chimeric antigen receptor T-cell
therapy in CNS lymphoma. Cancer Immunol Immunother. 74:3722025.
View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Du S, Bota D and Kong XT: Successful
consolidation/maintenance therapy with single agent ibrutinib for
primary CNS lymphoma after initial induction therapy. Neurol Int.
14:574–580. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Du S, Fu DB, Bota D and Kong XT: Prolonged
remission with ibrutinib maintenance therapy following radiation in
a patient with relapsed primary CNS lymphoma. CNS Oncol.
13:23455792024. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Cheng CL, Yuan CT, Fang WQ, Huang PH, Hou
HA, Tsai CH, Yao M, Chou WC and Tien HF: Both consolidation and
maintenance treatment improve outcomes in primary central nervous
system lymphoma: Real-world evidence from a tertiary medical
center. J Cancer. 16:1836–1847. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Nayak L and Batchelor TT: Is it time to
revisit R-CHOP for primary CNS lymphoma? Blood. 134:221–222. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Akimoto M, Miyazaki T, Takahashi H,
Saigusa Y, Takeda T, Hibino Y, Tokunaga M, Ohashi T, Matsumura A,
Teshigawara H, et al: Comparison of standardized prophylactic
high-dose and intrathecal methotrexate for DLBCL with a high risk
of CNS relapse. Int J Hematol. 119:164–172. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Murawski N, Held G, Ziepert M, Kempf B,
Viardot A, Hänel M, Witzens-Harig M, Mahlberg R, Rübe C,
Fleckenstein J, et al: The role of radiotherapy and intrathecal CNS
prophylaxis in extralymphatic craniofacial aggressive B-cell
lymphomas. Blood. 124:720–728. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Khan RB, Shi W, Thaler HT, DeAngelis LM
and Abrey LE: Is intrathecal methotrexate necessary in the
treatment of primary CNS lymphoma? J Neurooncol. 58:175–178. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Nelson RW and Frank JT: Intrathecal
methotrexate-induced neurotoxicities. Am J Hosp Pharm. 38:65–68.
1981.PubMed/NCBI
|
|
43
|
Rodrigues PGB, Lima TT, Duarte FB and
Nóbrega PR: Myelopathy associated with intrathecal methotrexate.
Pract Neurol. 22:141–144. 2022. View Article : Google Scholar : PubMed/NCBI
|