Introduction
Pancreatic cancer (PC) accounts for >80% of
pancreatic tumors and is one of the deadliest malignancies
worldwide, with a 5-year survival rate of ~8% (1). Complete surgical resection is
currently the only potentially curative option for PC. However,
recurrence rates are high despite this intervention and long-term
survival rates remain low (2).
Unfortunately, nearly 80% of patients present with distant
metastases and are diagnosed with advanced PC, thereby missing the
opportunity for surgical intervention. Only the remaining 20% of
patients may be eligible for surgery, which remains the sole
treatment modality with curative potential (3). However, even following successful
surgery, patients face a high risk of postoperative local
recurrence or distant metastases. The liver represents the most
common site of metastasis, followed by the lungs and the peritoneum
(4,5). PC is the second most common primary
lesion leading to liver metastasis (LM) (6). Notably, ~85% of patients with
metastatic PC eventually develop LM (7), which is strongly associated with poor
prognosis. Patients diagnosed with LM, whether treated with
resection or palliative care, have an average survival of <6
months (8–10). A study by Takada et al
(10) revealed that even aggressive
combined surgical resection of both the primary tumor and LM failed
to significantly improve the prognosis of such patients.
Furthermore, a high proportion of patients developed new
intrahepatic multilocular lesions within one year. Among patients
with PC without distant metastases who undergo surgical resection,
~35–50% experience early recurrence within 12 months after surgery
and nearly 25% of these recurrences involve only LM (11,12).
Despite advances in understanding the metastatic mechanisms of PC,
diagnostic techniques for synchronous liver metastasis (SLM) have
not advanced significantly, particularly in improving diagnostic
precision. Given the high incidence of LM and their potential for
early presentation, accurate preoperative identification of LM is
paramount for optimizing patient treatment and prognosis.
Misdiagnosis or overdiagnosis can lead to unnecessary surgical
interventions or missed opportunities for radical resection.
As early as 1863, Virchow proposed the association
between malignant tumors and inflammation, noting the phenomenon of
leukocyte infiltration in tumor tissues. He speculated that the
inflammatory area might be the origin of the tumor (13). Current studies have confirmed that
tumor-associated inflammatory states affect tumor cell survival,
proliferation, metastasis, neovascularization and responsiveness to
therapy. The white blood cell (WBC) count is closely related to
metastasis, prognosis and diagnosis in pancreatic malignancies
(14–16). Additionally, tumor markers have an
essential role in the diagnosis of PC with SLM. Among these,
carbohydrate antigen 19-9 (CA199) is one of the most widely used
markers in PC. CA199 is not only helpful in evaluating prognosis
but is also valuable in diagnosing PC with SLM (17). Meanwhile, several tumor markers,
such as carcinoembryonic antigen (CEA), CA199, carbohydrate antigen
125 (CA125) and alpha-fetoprotein (AFP), are closely associated
with LM in PC (18).
This study aims to establish a diagnostic model for
the simple, fast and effective identification of SLM in patients
with PC using multicenter integrated biochemical indices and
evaluate the diagnostic value of blood biochemical indices.
Subjects and methods
Study population
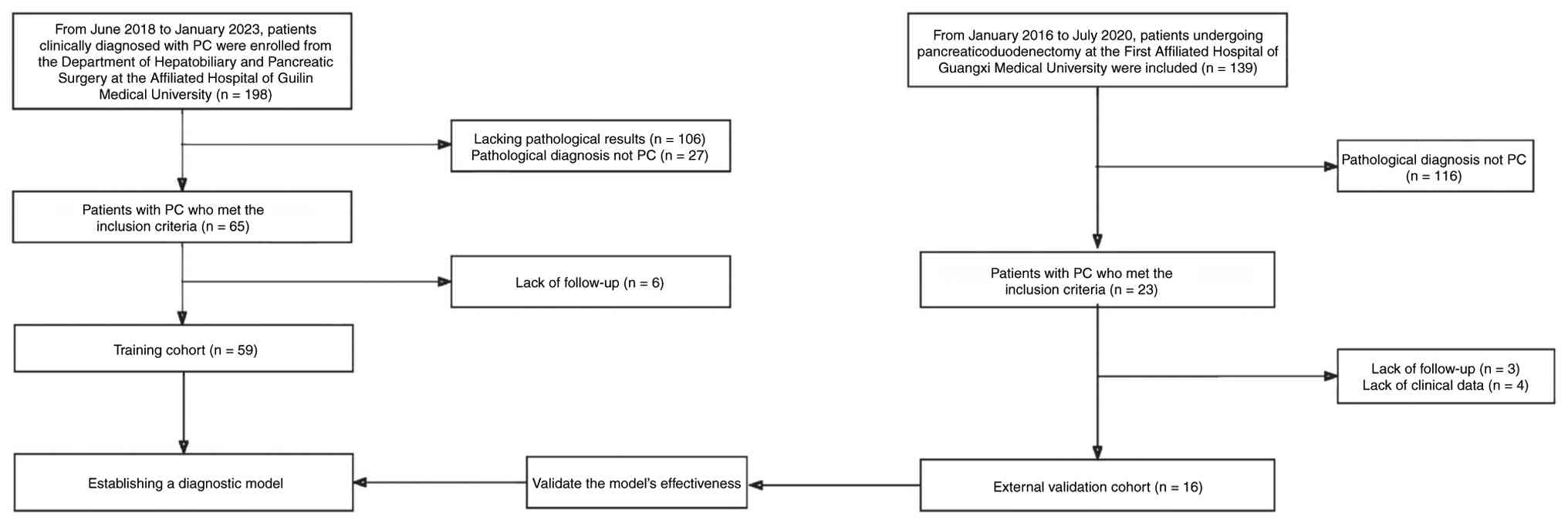
According to the inclusion and exclusion criteria,
59 patients with PC treated at the Department of Hepatobiliary and
Pancreatic Surgery of the Affiliated Hospital of Guilin Medical
University (Guilin, China), including 28 males and 31 females with
a median age of 58 years (range, 33–86 years), were continuously
included in the training cohort between June 2018 and January 2023.
Additionally, 16 patients with PC treated at the Department of
Hepatobiliary and Pancreatic Surgery of the First Affiliated
Hospital of Guangxi Medical University (Nanning, China) between
January 2016 and June 2020, comprising 8 males and 8 females with a
median age of 61.5 years (range, 39–71 years), who met the
inclusion and exclusion criteria, were included in the external
validation cohort (Fig. 1). There
were no significant differences in terms of age and gender
distribution between the training group and the validation group.
All of the cases were recruited as consecutive cases based on the
inclusion and exclusion criteria. PC was diagnosed based on tumor
pathology and LM was finally diagnosed by imaging tests,
pathological findings and follow-up results.
The inclusion criteria were as follows: i) First
diagnosis of PC by pathology and LM confirmed by imaging or
pathology; ii) in patients without LM at initial diagnosis, the
follow-up duration should be >6 months if there was no
intrahepatic metastasis during the follow-up period; and iii)
patients with pathologically confirmed PC and preoperative imaging
and blood biochemistry indicators. The following exclusion criteria
were applied: i) The nature of intrahepatic lesions is unclear; ii)
lack of follow-up data; iii) incomplete information in patient
clinical case data; iv) patients with metastases in other organs
besides LM; v) patients with other malignant tumors in combination;
and vi) patients with infectious diseases or autoimmune diseases.
This study was approved by the ethics committees of both hospitals
(ethics approval nos. 2022YJSLL-03 and 2024-E717-01) and was
prospectively registered in the Chinese Clinical Trial Registry
(registration ID: ChiCTR2200066901).
Methods
Methods of testing blood biochemical
indicators
All patients included in the study received
serologic and imaging examinations one week before surgery. Each
study subject provided 5 ml of fasting superficial venous blood,
which was centrifuged at a constant temperature of 4°C at 4,975 × g
for 15 min. The serum was retained for testing. The detection
methods used to ascertain each index were as follows: WBC,
platelets (PLTs), neutrophils (NEUT), lymphocytes (LYMPH) and
LYMPH% were measured using an automated hematology analyzer
(XN-9000; Sysmex Corp.). CEA, AFP, CA125, CA153 and CA19-9 levels
were measured using an automated chemiluminescent immunoassay
analyzer (Cobas e 801; Roche Diagnostics). Total bilirubin (TBIL),
direct bilirubin (DBIL), total protein (TP), albumin (ALB),
prealbumin, alkaline phosphatase, alanine aminotransferase,
aspartate transaminase and gamma-glutamyltranspeptidase were also
assayed.
Establishment and validation of the
diagnostic model
The 59 patients in the training cohort were divided
into the LM and non-liver metastasis (No-LM) groups based on the
presence or absence of LM. Comparative analysis was adopted to
screen for differences in clinical data between these groups.
Univariate logistic regression analysis was conducted to identify
the risk factors for SLM in patients with PC. Then, a Cox
proportional hazards model was used in the multivariate logistic
regression analysis to determine the independent risk factors for
concurrent LM. The independent risk factors identified through
multivariate analysis were integrated to construct a diagnostic
model based on blood biochemical indices. Continuous variables were
included as linear terms after assessing linearity with the
log-odds of the outcome using restricted cubic splines.
Multicollinearity among candidate variables was assessed using the
variance inflation factor (VIF). A VIF value <5 indicates that
there is no significant multicollinearity.
The performance of the model was evaluated by
plotting the receiver operating characteristic (ROC) curve and
calculating the area under the curve (AUC) and 95% confidence
intervals for each ROC curve. A nomogram was constructed to
visualize each indicator and the overall model to display the model
more intuitively and show the weights of each independent risk
factor and its influence on the diagnostic results. To further
evaluate the performance of the model, the Hosmer-Lemeshow (HL)
goodness-of-fit test was used. The model was refitted 1,000 times
using bootstrap sampling and its performance was assessed on the
original complete dataset to calculate the average diagnostic
performance and validate the model's reliability. Calibration
curves were generated to ensure that the predicted probabilities
match the actual observed rates, confirming the accuracy of the
model. Finally, decision curve analysis (DCA) was conducted to
assess the model's strengths and practical utility in clinical
settings. The validity of the model was further verified using data
from the external validation cohort.
Statistical analysis
Continuous variables were expressed as mean ±
standard deviation or median, while categorical variables were
expressed as frequencies and percentages. The independent t-test
and the Mann-Whitney U-test were employed to compare continuous
variables. The Chi-squared or Fisher's exact test was used to
analyze categorical variables. All-important variable screening,
model building and model comparison were statistically performed
using SPSS 26.0 (IBM Corp.) and R software. All statistical tests
were two-sided and P<0.05 was considered to indicate a
statistically significant difference.
Results
Patient characteristics
The general baseline information pertaining to the
training cohort and the external validation cohort is displayed in
Table I. There was a significant
difference in the levels of ALB between the training cohort and the
external validation cohort, while no significant differences were
observed for the remaining indicators.
 | Table I.Clinical characteristics compared
between the training and the external validation cohort. |
Table I.
Clinical characteristics compared
between the training and the external validation cohort.
| Indexes | Training cohort
(n=59) | Validation cohort
(n=16) | P-value |
|---|
| Sex |
|
| 0.857 |
|
Male | 28 (47.46) | 8 (50.00) |
|
|
Female | 31 (52.54) | 8 (50.00) |
|
| Age, years | 58.97±9.15 | 59.13±9.89 | 0.953 |
| CEA, ng/ml | 3.18 (1.58,
6.77) | 3.85 (2.27,
8.04) | 0.821 |
| AFP, ng/ml | 2.44 (1.63,
3.44) | 2.04 (1.71,
2.39) | 0.292 |
| CA125, U/ml | 19.45 (12.89,
29.44) | 17.30 (9.50,
34.80) | 0.628 |
| CA199, U/ml | 194.00 (22.18,
486.30) | 493.78 (51.28,
1258.04) | 0.382 |
| CA153, U/ml | 10.66 (5.99,
18.80) | 17.40 (10.00,
22.90) | 0.115 |
| TBIL, µmol/l | 73.70 (10.10,
171.20) | 128.70 (42.10,
185.30) | 0.339 |
| DBIL, µmol/l | 69.70 (4.30,
152.80) | 87.60 (36.90,
126.20) | 0.732 |
| TP, g/l | 68.14±5.24 | 67.43±7.36 | 0.664 |
| ALB, g/l | 38.17±3.87 | 35.69±3.92 | 0.028 |
| G-GT, U/l | 186.00 (22.00,
518.95) | 198.90 (94.00,
248.00) | 0.578 |
| AST, U/l | 45.30 (18.40,
91.50) | 49.00 (25.00,
83.00) | 0.923 |
| ALT, U/l | 49.70 (17.00,
131.70) | 45.00 (25.00,
128.00) | 0.974 |
| ALP, U/l | 177.00 (82.00,
339.00) | 240.00 (172.00,
279.00) | 0.157 |
| PA, mg/l | 199.17 (146.90,
230.65) | 179.40 (133.80,
194.10) | 0.129 |
| WBC, ×109/l | 6.22 (4.85,
8.40) | 6.69 (6.27,
7.95) | 0.289 |
| NEUT, ×109/l | 3.788 (2.87,
5.31) | 4.48 (3.90,
4.95) | 0.258 |
| NEUT% | 0.63±0.10 | 0.66±0.07 | 0.441 |
| LYMPH, ×109/l | 1.32 (1.14,
1.75) | 1.63 (1.03,
1.79) | 0.776 |
| LYMPH% | 0.25 (0.18,
0.30) | 0.22 (0.19,
0.26) | 0.514 |
| PLT, ×109/l | 271.00 (214.00,
313.00) | 251.00 (208.90,
269.90) | 0.453 |
| LDH, U/l | 181.35 (156.00,
231.00) | 150.00 (145.00,
212.00) | 0.252 |
| FIB, g/l | 3.68 (3.05,
4.34) | 4.17 (3.38,
4.86) | 0.247 |
| INR | 1.00 (0.93,
1.11) | 1.02 (0.93,
1.11) | 0.964 |
| SII, ×109/l | 107.50 (82.06,
162.39) | 131.98 (81.33,
175.12) | 0.995 |
| NLR | 2.58 (2.04,
3.84) | 3.00 (2.26,
4.35) | 0.348 |
| PLR | 177.96 (144.74,
237.35) | 196.99 (134.94,
248.32) | 0.933 |
| APRI | 0.43 (0.22,
0.88) | 0.50 (0.27,
0.94) | 0.872 |
| AAR | 0.971 (0.63,
1.31) | 0.92 (0.77,
1.44) | 0.887 |
| SLM |
|
| 0.916 |
|
No-LM | 45 (76.27) | 12 (75.00) |
|
| LM | 14 (23.73) | 4 (25.00) |
|
Risk factors for SLM of PC
In this study, 59 patients with PC were included in
the training cohort, comprising 12 patients in the LM group and 47
patients in the No-LM group. To avoid omitting risk factors
associated with SLM in PC, indicators with P<0.1 in the
univariate logistic regression analysis were included in the
multivariate logistic regression analysis. Univariate logistic
regression analysis identified the following indicators with
P<0.1: CEA, CA125, CA199, CA153, TBIL, DBIL, TP, WBC, NEUT,
LYMPH%, PLT, SII and NLR (Table
II). Multivariate logistic regression analysis suggested that
WBC, PLT, CEA and CA153 were independent risk factors for SLM in
patients with PC (Table III).
CA19-9 and CA125, though significant in the univariate analysis,
were not retained in the final multivariate model. There was a
strong correlation among the liver function indicators and
inflammation-related indicators. The analysis indicates that there
is little correlation between CA19-9 and other liver function
indicators as well as inflammatory markers, while there is a
certain correlation between CA125 and inflammatory markers
(Fig. S1). Therefore, their
predictive information may be covered by other included variables
in this specific cohort, or be affected by the limited sample
size.
 | Table II.Univariate logistic regression
analysis in the training cohort. |
Table II.
Univariate logistic regression
analysis in the training cohort.
| Indexes | β | S.E. | Z | P-value | OR (95% CI) |
|---|
| CEA, ng/ml | 0.02 | 0.01 | 1.75 | 0.080 | 1.02
(1.00–1.05) |
| AFP, ng/ml | −0.02 | 0.04 | −0.45 | 0.650 | 0.98
(0.91–1.06) |
| CA125, U/ml | 0.02 | 0.01 | 2.39 | 0.017 | 1.02
(1.01–1.03) |
| CA199, U/ml | 0.00 | 0.00 | 1.83 | 0.067 | 1.00
(1.00–1.00) |
| CA153, U/ml | 0.11 | 0.04 | 2.75 | 0.006 | 1.11
(1.03–1.20) |
| TBIL, µmol/l | −0.01 | 0.00 | −2.05 | 0.040 | 0.99
(0.98–0.99) |
| DBIL, µmol/l | −0.01 | 0.00 | −2.15 | 0.032 | 0.99
(0.98–0.99) |
| TP, g/l | 0.13 | 0.07 | 1.97 | 0.049 | 1.14
(1.01–1.29) |
| ALB, g/l | 0.07 | 0.08 | 0.82 | 0.413 | 1.07
(0.91–1.25) |
| GGT, U/l | 0.00 | 0.00 | 0.21 | 0.836 | 1.00
(1.00–1.00) |
| AST, U/l | −0.00 | 0.00 | −0.70 | 0.484 | 1.00
(0.99–1.01) |
| ALT, U/l | −0.00 | 0.00 | −0.83 | 0.405 | 1.00
(0.99–1.00) |
| ALP, U/l | 0.00 | 0.00 | 0.47 | 0.636 | 1.00
(1.00–1.00) |
| PA, mg/l | 0.00 | 0.00 | 1.10 | 0.272 | 1.00
(1.00–1.00) |
| WBC, ×109/l | 0.37 | 0.15 | 2.53 | 0.012 | 1.45
(1.09–1.94) |
| NEUT, ×109/l | 0.44 | 0.16 | 2.73 | 0.006 | 1.55
(1.13–2.12) |
| LYMPH, ×109/l | −0.39 | 0.66 | −0.60 | 0.550 | 0.67
(0.19–2.46) |
| LYMPH% | −8.69 | 4.19 | −2.07 | 0.038 | 0.00
(0.00–0.62) |
| PLT, ×109/l | 0.01 | 0.00 | 2.25 | 0.025 | 1.01
(1.01–1.02) |
| LDH, ×109/l | 0.00 | 0.00 | 1.09 | 0.274 | 1.00
(1.00–1.01) |
| FIB, g/l | −0.02 | 0.04 | −0.57 | 0.566 | 0.98
(0.91–1.06) |
| INR | −1.35 | 1.50 | −0.90 | 0.366 | 0.26
(0.01–4.87) |
| SII, ×109/l | 0.01 | 0.00 | 1.85 | 0.064 | 1.01
(1.00–1.02) |
| NLR | 0.32 | 0.13 | 2.38 | 0.017 | 1.38
(1.06–1.79) |
| PLR | 0.01 | 0.00 | 1.73 | 0.084 | 1.01
(1.00–1.01) |
| APRI | −0.54 | 0.52 | −1.05 | 0.292 | 0.58
(0.21–1.60) |
 | Table III.Multivariate logistic regression
analysis in the training cohort. |
Table III.
Multivariate logistic regression
analysis in the training cohort.
| Indexes | β | S.E. | Z | P-value | OR (95%CI) |
|---|
| Intercept | −12.07 | 3.34 | −3.61 | <0.001 | 0.00
(0.00–0.00) |
| CA153, U/ml | 0.17 | 0.05 | 3.08 | 0.002 | 1.18
(1.06–1.31) |
| WBC, ×109/l | 0.54 | 0.24 | 2.27 | 0.023 | 1.71
(1.08–2.72) |
| CEA, U/ml | 0.05 | 0.02 | 2.66 | 0.008 | 1.05
(1.01–1.08) |
| PLT, ×109/l | 0.01 | 0.01 | 2.00 | 0.045 | 1.01
(1.01–1.03) |
Development and validation of the
diagnostic model
Based on the results of the multivariate logistic
regression analysis, a diagnostic model integrating four blood
biochemical indicators for diagnosing SLM in patients with PC was
established as follows: Logit(P)=0.05 × (CEA, ng/ml) + 0.17 ×
(CA153, U/ml) + 0.54 × (WBC, × 109/l) + 0.01 × (PLT, ×109/l)-12.07.
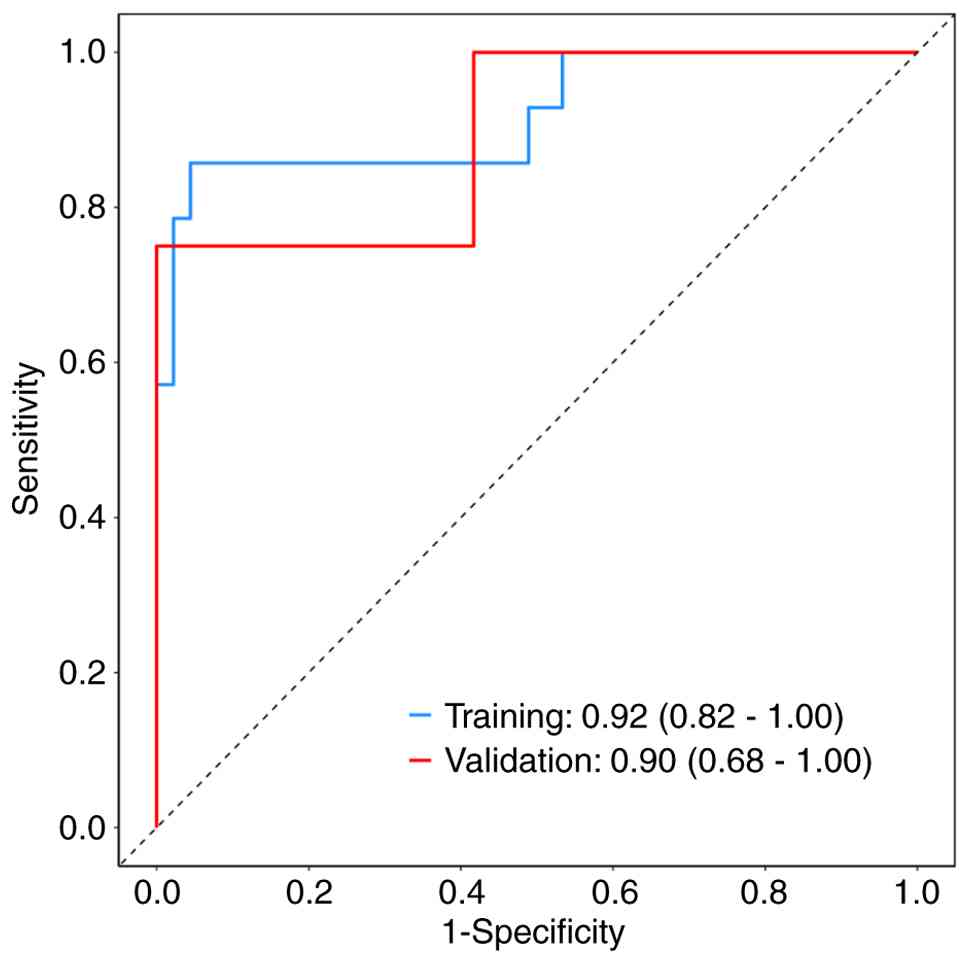
In the training cohort, the AUC of the model was 0.92 (95% CI:
0.82–1.00), and sensitivity and specificity were 0.96 and 0.86,
respectively. The AUC of the model was 0.90 (95% CI: 0.68–1.00),
and the sensitivity and specificities were 0.83 and 0.75,
respectively, in the external validation cohort. These results are
summarized in Fig. 2 and Table IV. The optimal cut-off value for
the diagnostic score was determined using the Youden index.
 | Table IV.Performance of the model. |
Table IV.
Performance of the model.
| Dataset | AUC | Accuracy | Sensitivity | Specificity | PPV | NPV | Cut-off point |
|---|
| Training
cohort | 0.92 | 0.93 | 0.96 | 0.86 | 0.96 | 0.86 | 0.457 |
| Validation
cohort | 0.90 | 0.81 | 0.83 | 0.75 | 0.91 | 0.60 | 0.457 |
Nomogram
To ascertain the value of these factors more
intuitively and understand the significance of these independent
risk factors more quickly and easily in clinical practice, a
nomogram for LM in patients with PC was constructed using R
software (Fig. 3). This nomogram
provides a visual tool to facilitate the practical application of
the diagnostic model, allowing clinicians to estimate the
probability of LM based on the CEA values, CA153, WBC and PLT. For
instance, a patient with CEA=7.3 ng/ml, CA153=15.6 U/ml,
WBC=9.4×109/l and PLT=623×109/l would have a total point score of
~2.253, corresponding to a predicted probability of LM of
~0.927.
Model fitting
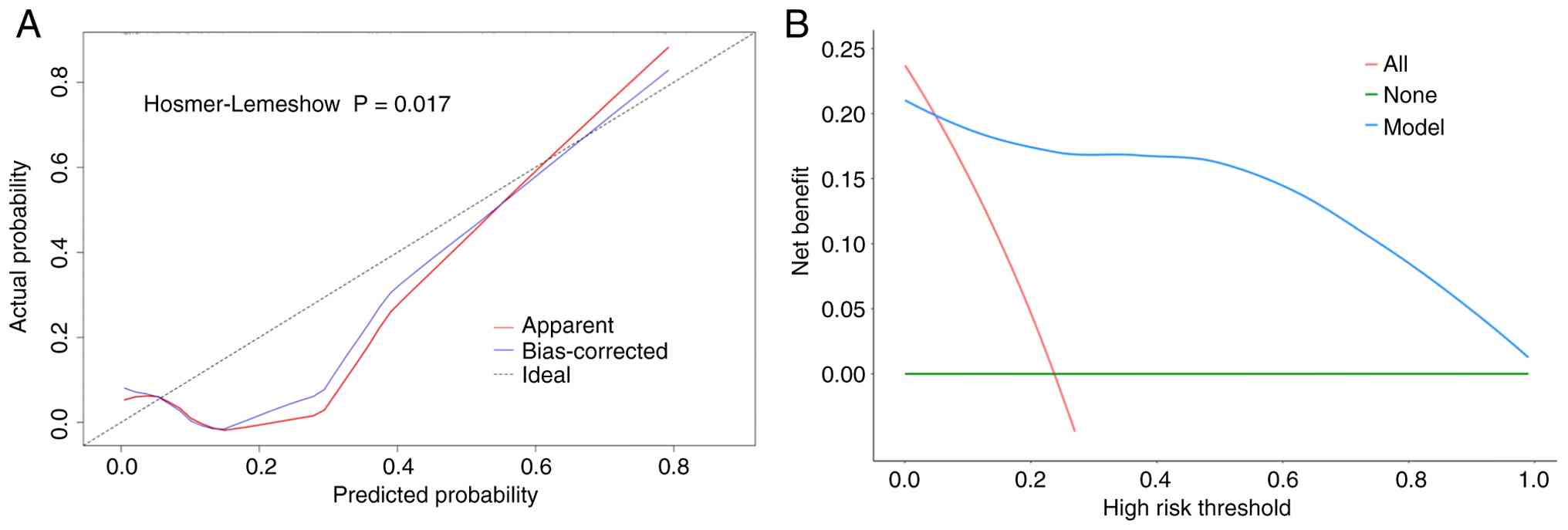
The model's goodness of fit was evaluated using the
HL test, which yielded a P-value of 0.017. Given the relatively
small sample size, the calibration was also assessed using
bootstrap resampling (1,000 replicates). The calibration curve
(Fig. 4A) shows a good agreement
between predicted and observed probabilities, with a mean absolute
error of 0.067, indicating acceptable calibration.
DCA curve
DCA was performed to determine the net benefit of
the diagnostic model, thereby evaluating its clinical
applicability. As shown in Fig. 4B,
when the threshold surpasses 0.06, the DCA curve is above the
‘None’ and ‘All’ curves, indicating that the model has a better
clinical net benefit in this range.
These evaluations prove that the diagnostic model is
not only statistically robust but also clinically applicable,
offering a valuable tool for the early identification of SLM in
patients with PC.
Discussion
PC is a highly lethal malignancy with significant
metastatic potential. Even small PCs (<20 mm in diameter) can
metastasize and ultimately lead to death (19,20).
Radical pancreatectomy is the only potentially curative treatment,
but occult metastases often diminish its efficacy (21). Although substantial progress has
been made in the diagnosis of LM through advances in imaging
techniques, minimally invasive surgery and biomarker research, the
accurate preoperative diagnosis of LM in patients with PC remains a
significant challenge. Currently, no biomarkers with both high
specificity and high sensitivity are available for the diagnosis of
LM from PC. Therefore, the exploration of effective biomarkers is
critical to improving early diagnostic accuracy for LM and guiding
individualized treatment strategies. This study revealed
significant differences in CA153, CEA, WBC and PLT levels between
the patient groups of LM and non-LM. CA153, although traditionally
used for breast cancer (22), has
shown potential value as the soluble form of mucin (MUC)-1 in PC
(23). Notably, in the multivariate
analysis of this study, CA153 emerged as an independent predictor
of LM, while CA199 and CA125 were excluded from the final model.
Statistically, in our multivariate analysis, CA153 remained an
independent predictor of liver metastasis, while CA199 and CA125
did not, suggesting it provides unique information relevant to
metastasis in the present cohort. This may be because CA153 (MUC-1)
reflects unique biological processes related to tumor invasion and
metastasis. Previous studies confirmed that the abnormal expression
and glycosylation changes of MUC-1 in PC are directly associated
with the occurrence and development of LM (24). Therefore, CA153 may serve as a
supplementary biomarker, providing additional predictive
information that is different from traditional markers for this
diagnostic model. This finding may be interpreted as CA153
potentially adding complementary value in assessing metastatic
risk, not as a replacement for conventional markers. Its clinical
utility requires validation in larger, prospective studies. CEA is
elevated in various malignant tumors, including PC (25). Currently, CEA is the second most
common serum biomarker used clinically for detecting PC, with
upregulated levels observed in 30–60% of patients with PC, and it
is associated with PC survival (26). Studies have shown that abnormally
high levels of CEA in patients with PC correlate with tumor
progression, poor prognosis and the incidence of distant metastasis
(27,28). Continuous monitoring of the changes
of CEA levels in patients with PC can aid in assessing the
treatment efficacy, predicting recurrence and guiding the selection
of subsequent treatment strategies (29,30).
This enables the optimization of therapeutic plans and disease
progression monitoring.
The WBC count is a common indicator used to assess
the body's inflammatory response, infection status and certain
hematological diseases, which significantly inhibits tumor
development (31,32). A previous study found that
tumor-secreted factors trigger inflammatory responses within the
liver microenvironment, promoting LM of PC (33). WBCs can be transformed by tumor
cells and stromal cells into tumor-associated cells, which secrete
multiple cytokines and chemokines that enhance immunosuppression in
the tumor microenvironment, thereby facilitating the proliferation
and migration of tumor cells (34).
However, the specific phenotype, function and regulatory mechanisms
of WBCs in the context of LM remain elusive in PC (35). In the present study, the WBC count
was identified as an independent risk factor for SLM in patients
with PC. It has an auxiliary role in diagnosing SLM of PC and has
the potential to improve the accuracy of such diagnoses.
A substantial amount of clinical and experimental
data indicates that the interaction between PLTs and tumor cells is
critical for tumor metastasis (36). PLTs contribute to the pre-metastatic
microenvironment by forming microthrombi that trap circulating
tumor cells, releasing growth factors (e.g., TGF-β, VEGF), and
promoting the endothelial permeability and immune evasion in the
liver (33,35,37).
Multiple studies have shown that thrombocytosis (elevated PLT
counts) is a predictor of poor prognosis in PC (38–40).
The present study demonstrates a significant difference in PLT
counts between the metastatic and non-metastatic groups.
In this study, a diagnostic model integrating CA153,
CEA, WBC and PLT was established. This model exhibited high
performance in identifying LM in patients with PC and outperformed
single biochemical indices used in other studies (41–43).
CA125 and CA199 are of certain value in diagnosing PC and its LM
(41,43–45).
The results of the present study also suggested that CA125 and
CA199 were significantly different between the LM and Non-LM
groups. However, in the multivariate logistic regression analysis,
CA199 and CA125 were not included in the diagnostic model, which
may be related to the sample size. Although the HL test suggested
certain deviations between predicted and observed outcomes
(P<0.05), bootstrap calibration and visual inspection of the
calibration curve showed a good model fit. This discrepancy may
reflect the sensitivity of the HL test to limited sample sizes
rather than poor model calibration performance.
Of note, the present study had certain limitations.
First, the main limitation of this study is the relatively small
sample size, particularly the limited number of patients with SLM
(n=14 in the training cohort). This may affect the stability of the
multivariate logistic regression model and increase the risk of
overfitting. Therefore, the present results should be interpreted
with caution, and further validation in larger, multicenter
prospective studies is warranted. Second, the external validation
cohort was small (n=16), which limits the strength of conclusions
regarding the model's generalizability. Future studies with larger,
independent cohorts are needed to confirm the robustness and
transportability of the model. In addition, the number of events
(SLM cases) per predictor variable was below the recommended
threshold of 10 events per variable for logistic regression
(46), which may elevate the risk
of overfitting. Although bootstrap validation confirmed reasonable
stability, future studies with larger samples should consider
penalized regression approaches to enhance the model's robustness.
Finally, differences in the indicator detection methods between
different medical institutions may affect the consistency of data
collection and analysis.
In conclusion, CEA, CA153, WBC and PLT are important
independent risk factors for SLM in patients with PC. The
diagnostic model integrating these blood biochemical indices
exhibits good diagnostic efficacy in identifying SLM in patients
with PC and demonstrates strong clinical applicability.
Supplementary Material
Supporting Data
Acknowledgements
Not applicable.
Funding
This study was supported by the Affiliated Hospital of Guilin
Medical University, PhD Start-up Fund (grant no. KY1303) and
Guangxi Key Laboratory of Immunology and Metabolism for Liver
Diseases (grant no. GKE-KF202505).
Availability of data and materials
The data generated in the present study may be
requested from the corresponding author.
Authors' contributions
YJ, GP, SH and JL conceived and designed the study.
YJ, GP and JL analyzed the data. YJ, GP and JL wrote the
manuscript. YJ and GP collected the data. SH and JL conceptualized
and developed an outline for the manuscript and revised it. JY and
JL confirm the authenticity of the raw data. All authors read and
approved the final manuscript.
Ethics approval and consent to
participate
The retrospective study was approved by the Ethics
Committee of the Affiliated Hospital of Guilin Medical University
(grant no. 2022YJSLL-03) and the Ethics Committee of the First
Affiliated Hospital of Guangxi Medical University (grant no.
2024-E717-01). All patients signed informed consent forms form
allowing their case and imaging data to be used anonymously for
research.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Rawla P, Sunkara T and Gaduputi V:
Epidemiology of pancreatic cancer: Global trends, etiology and risk
factors. World J Oncol. 10:10–27. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
McGuigan A, Kelly P, Turkington RC, Jones
C, Coleman HG and McCain RS: Pancreatic cancer: A review of
clinical diagnosis, epidemiology, treatment and outcomes. World J
Gastroenterol. 24:4846–4861. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Ducreux M, Cuhna AS, Caramella C,
Hollebecque A, Burtin P, Goéré D, Seufferlein T, Haustermans K, Van
Laethem JL, Conroy T, et al: Cancer of the pancreas: ESMO clinical
practice guidelines for diagnosis, treatment and follow-up. Ann
Oncol. 26 (Suppl 5):v56–v68. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Zhang XP, Gao YX, Xu S, Zhao GD, Hu MG,
Tan XL, Zhao ZM and Liu R: A novel online calculator to predict
early recurrence and long-term survival of patients with resectable
pancreatic ductal adenocarcinoma after pancreaticoduodenectomy: A
multicenter study. Int J Surg. 106:1068912022. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Kolbeinsson H, Hoppe A, Bayat A,
Kogelschatz B, Mbanugo C, Chung M, Wolf A, Assifi MM and Wright GP:
Recurrence patterns and postrecurrence survival after curative
intent resection for pancreatic ductal adenocarcinoma. Surgery.
169:649–654. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
de Ridder J, de Wilt JH, Simmer F,
Overbeek L, Lemmens V and Nagtegaal I: Incidence and origin of
histologically confirmed liver metastases: An explorative
case-study of 23,154 patients. Oncotarget. 7:55368–55376. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Hess KR, Varadhachary GR, Taylor SH, Wei
W, Raber MN, Lenzi R and Abbruzzese JL: Metastatic patterns in
adenocarcinoma. Cancer. 106:1624–1633. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Ouyang HH, Pan ZY, Ma WD, Zhao LJ, Zhang
T, Liu F and Quan MM: Multidisciplinary treatment and survival
analysis for 497 cases of pancreatic cancer with liver metastases.
Zhonghua Yi Xue Za Zhi. 96:425–430. 2016.(In Chinese). PubMed/NCBI
|
|
9
|
Ouyang H, Ma W, Liu F, Yue Z, Fang M, Quan
M and Pan Z: Factors influencing survival of patients with
pancreatic adenocarcinoma and synchronous liver metastases
receiving palliative care. Pancreatology. 17:773–781. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Takada T, Yasuda H, Amano H, Yoshida M and
Uchida T: Simultaneous hepatic resection with
pancreato-duodenectomy for metastatic pancreatic head carcinoma:
Does it improve survival? Hepatogastroenterology. 44:567–573.
1997.PubMed/NCBI
|
|
11
|
Murakawa M, Kawahara S, Takahashi D,
Kamioka Y, Yamamoto N, Kobayashi S, Ueno M, Morimoto M, Sawazaki S,
Tamagawa H, et al: Risk factors for early recurrence in patients
with pancreatic ductal adenocarcinoma who underwent curative
resection. World J Surg Oncol. 21:2632023. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Huang Y, Zhou S, Luo Y, Zou J, Li Y, Chen
S, Gao M, Huang K and Lian G: Development and validation of a
radiomics model of magnetic resonance for predicting liver
metastasis in resectable pancreatic ductal adenocarcinoma patients.
Radiat Oncol. 18:792023. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Balkwill F and Mantovani A:
Inflammationcancer: Back to virchow? Lancet. 357:539–545. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Wang DS, Luo HY, Qiu MZ, Wang ZQ, Zhang
DS, Wang FH, Li YH and Xu RH: Comparison of the prognostic values
of various inflammation based factors in patients with pancreatic
cancer. Med Oncol. 29:3092–3100. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Smith RA, Bosonnet L, Raraty M, Sutton R,
Neoptolemos JP, Campbell F and Ghaneh P: Preoperative
platelet-lymphocyte ratio is an independent significant prognostic
marker in resected pancreatic ductal adenocarcinoma. Am J Surg.
197:466–472. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Xue P, Kanai M, Mori Y, Nishimura T, Uza
N, Kodama Y, Kawaguchi Y, Takaori K, Matsumoto S, Uemoto S and
Chiba T: Neutrophil-to-lymphocyte ratio for predicting palliative
chemotherapy outcomes in advanced pancreatic cancer patients.
Cancer Med. 3:406–415. 2014. View
Article : Google Scholar : PubMed/NCBI
|
|
17
|
Raza SS, Khan H, Hajibandeh S, Hajibandeh
S, Bartlett D, Chatzizacharias N, Roberts K, Marudanayagam R and
Sutcliffe RP: Can preoperative carbohydrate antigen 19-9 predict
metastatic pancreatic cancer? Results of a systematic review and
meta-analysis. HPB (Oxford). 26:630–638. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Liu L, Xu H, Wang W, Wu C, Chen Y, Yang J,
Cen P, Xu J, Liu C, Long J, et al: A preoperative serum signature
of CEA+/CA125+/CA19-9 ≥1,000 U/ml indicates poor outcome to
pancreatectomy for pancreatic cancer. Int J Cancer. 136:2216–2227.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Hidalgo M: Pancreatic cancer. N Engl J
Med. 362:1605–1617. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Vincent A, Herman J, Schulick R, Hruban RH
and Goggins M: Pancreatic cancer. Lancet. 378:607–620. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Hartwig W, Werner J, Jäger D, Debus J and
Büchler MW: Improvement of surgical results for pancreatic cancer.
Lancet Oncol. 14:e476–e485. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Ren Z, Yang J, Liang J, Xu Y, Lu G, Han Y,
Zhu J, Tan H, Xu T and Ren M: Monitoring of postoperative
neutrophil-to-lymphocyte ratio, D-dimer, and CA153 in: Diagnostic
value for recurrent and metastatic breast cancer. Front Surg.
9:9274912023. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Beatty P, Hanisch FG, Stolz DB, Finn OJ
and Ciborowski P: Biochemical characterization of the soluble form
of tumor antigen MUC1 isolated from sera and ascites fluid of
breast and pancreatic cancer patients. Clin Cancer Res. 7 (Suppl
3):781s–787s. 2001.PubMed/NCBI
|
|
24
|
Remmers N, Anderson JM, Linde EM, DiMaio
DJ, Lazenby AJ, Wandall HH, Mandel U, Clausen H, Yu F and
Hollingsworth MA: Aberrant expression of mucin core proteins and
o-linked glycans associated with progression of pancreatic cancer.
Clin Cancer Res. 19:1981–1993. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Diehl SJ, Lehmann KJ, Sadick M, Lachmann R
and Georgi M: Pancreatic cancer: Value of dual-phase helical CT in
assessing resectability. Radiology. 206:373–378. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Swords DS, Firpo MA, Scaife CL and
Mulvihill SJ: Biomarkers in pancreatic adenocarcinoma: Current
perspectives. Onco Targets Ther. 9:7459–7467. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Basso D, Fabris C, Del Favero G, Angonese
C, Meggiato T, Infantino A, Plebani M, Piccoli A, Leandro G,
Burlina A, et al: Serum carcinoembryonic antigen in the
differential diagnosis of pancreatic cancer: Influence of tumour
spread, liver impairment, and age. Dis Markers. 6:203–207.
1988.PubMed/NCBI
|
|
28
|
Chen Y, Gao SG, Chen JM, Wang GP, Wang ZF,
Zhou B, Jin CH, Yang YT and Feng XS: Serum CA242, CA199, CA125,
CEA, and TSGF are biomarkers for the efficacy and prognosis of
cryoablation in pancreatic cancer patients. Cell Biochem Biophys.
71:1287–1291. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Lee KJ, Yi SW, Chung MJ, Park SW, Song SY,
Chung JB and Park JY: Serum CA 19-9 and CEA levels as a prognostic
factor in pancreatic adenocarcinoma. Yonsei Med J. 54:643–649.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Wu L, Huang P, Wang F, Li D, Xie E, Zhang
Y and Pan S: Relationship between serum CA19-9 and CEA levels and
prognosis of pancreatic cancer. Ann Transl Med.
3:3282015.PubMed/NCBI
|
|
31
|
Singel KL and Segal BH. Neutrophils in the
tumor microenvironment: Trying to heal the wound that cannot heal.
Immunol Rev. 273:329–343. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Yan J, Kloecker G, Fleming C, Bousamra M
II, Hansen R, Hu X, Ding C, Cai Y, Xiang D, Donninger H, et al:
Human polymorphonuclear neutrophils specifically recognize and kill
cancerous cells. Oncoimmunology. 3:e9501632014. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Costa-Silva B, Aiello NM, Ocean AJ, Singh
S, Zhang H, Thakur BK, Becker A, Hoshino A, Mark MT, Molina H, et
al: Pancreatic cancer exosomes initiate pre-metastatic niche
formation in the liver. Nat Cell Biol. 17:816–826. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Caldeira PC, Vieira ÉLM, Sousa AA,
Teixeira AL and Aguiar MCF: Immunophenotype of neutrophils in oral
squamous cell carcinoma patients. J Oral Pathol Med. 46:703–709.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Wang X, Hu LP, Qin WT, Yang Q, Chen DY, Li
Q, Zhou KX, Huang PQ, Xu CJ, Li J, et al: Identification of a
subset of immunosuppressive P2RX1-negative neutrophils in
pancreatic cancer liver metastasis. Nat Commun. 12:1742021.
View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Shi Q, Ji T, Tang X and Guo W: The role of
tumor-platelet interplay and micro tumor thrombi during
hematogenous tumor metastasis. Cell Oncol (Dordr). 46:521–532.
2023. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Schlesinger M: Role of platelets and
platelet receptors in cancer metastasis. J Hematol Oncol.
11:1252018. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Shirai Y, Shiba H, Sakamoto T, Horiuchi T,
Haruki K, Fujiwara Y, Futagawa Y, Ohashi T and Yanaga K:
Preoperative platelet to lymphocyte ratio predicts outcome of
patients with pancreatic ductal adenocarcinoma after pancreatic
resection. Surgery. 158:360–365. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Zhang SR, Yao L, Wang WQ, Xu JZ, Xu HX,
Jin W, Gao HL, Wu CT, Qi ZH, Li H, et al: Tumor-infiltrating
platelets predict postsurgical survival in patients with pancreatic
ductal adenocarcinoma. Ann Surg Oncol. 25:3984–3993. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Saito R, Kawaida H, Hosomura N, Amemiya H,
Itakura J, Yamamoto A, Takiguchi K, Maruyama S, Shoda K, Furuya S,
et al: Exposure to blood components and inflammation contribute to
pancreatic cancer progression. Ann Surg Oncol. 28:8263–8272. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Haridas D, Chakraborty S, Ponnusamy MP,
Lakshmanan I, Rachagani S, Cruz E, Kumar S, Das S, Lele SM,
Anderson JM, et al: Pathobiological implications of MUC16
expression in pancreatic cancer. PLoS One. 6:e268392011. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Shi HJ, Jin C and Fu DL: Preoperative
evaluation of pancreatic ductal adenocarcinoma with synchronous
liver metastasis: Diagnosis and assessment of unresectability.
World J Gastroenterol. 22:10024–10037. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Yuan Z, Shu Z, Peng J, Wang W, Hou J, Han
L, Zheng G, Wei Y and Zhong J: Prediction of postoperative liver
metastasis in pancreatic ductal adenocarcinoma based on
multiparametric magnetic resonance radiomics combined with
serological markers: A cohort study of machine learning. Abdom
Radiol (NY). 49:117–130. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Zhang T, Dong X, Zhou Y, Liu M, Hang J and
Wu L: Development and validation of a radiomics nomogram to
discriminate advanced pancreatic cancer with liver metastases or
other metastatic patterns. Cancer Biomark. 32:541–550. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Liu L, Xu HX, Wang WQ, Wu CT, Xiang JF,
Liu C, Long J, Xu J, Fu de L, Ni QX, et al: Serum CA125 is a novel
predictive marker for pancreatic cancer metastasis and correlates
with the metastasis-associated burden. Oncotarget. 7:5943–5956.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Peduzzi P, Concato J, Kemper E, Holford TR
and Feinstein AR: A simulation study of the number of events per
variable in logistic regression analysis. J Clin Epidemiol.
49:1373–1379. 1996. View Article : Google Scholar : PubMed/NCBI
|