Introduction
Global cancer statistics indicate that lung cancer
has the highest morbidity and mortality rates worldwide (1). Lung cancer ranks second in terms of
incidence worldwide and first in terms of mortality. Every year,
~1.6 million people die from lung cancer (2). Lung cancer is classified into small
cell lung cancer and non-small cell lung cancer (NSCLC) based on
its pathological type, with NSCLC accounting for 85% of cases
(3). Currently, chemotherapy
remains the primary treatment option for lung cancer. However, the
overall prognosis remains poor, characterized by a short median
survival time and marked toxicities and side-effects (4). A number of natural anticancer
compounds have been identified and developed for therapeutic
purposes, such as paclitaxel (5),
curcumin (6), phloretin (7), berberine (8) and artemisinin (9). The main aim of the present study was
to identify additional novel anti-NSCLC compounds with potential
for drug development.
Bulbus Fritillaria cirrhosa D.Don, also known
as chuanbeimu, is recognized as a high-quality food and medicinal
ingredient, holding the highest economic value among other beimu
products, including Fritillaria Pallidiflora Bulbus and
Fritillaria thunbergii Miq (10,11)
Alkaloids are the primary active components of F. cirrhosa
D.Don . Isosteroidal alkaloids exhibit significant anticancer,
antitussive, expectorant, anti-inflammatory and antioxidant
properties, as well as other biological properties. At present, the
experimental validation of the anticancer effects of F.
cirrhosa D.Don primarily focuses on its extracts, with a few
individual alkaloids reported to exhibit anti-proliferative and
anti-inflammatory activities (12–14).
The material basis and mechanisms of action of F. cirrhosa
D.Don against lung cancer warrant further investigation.
The precise roles and extent of the involvement of
apoptosis and autophagy in the anticancer effects of
Fritillaria remain unclear. Apoptosis refers specifically to
the orderly self-destruction of cells, a process autonomously
regulated by genes that encode proteins, such as Bax, Bcl-2,
caspase-3 and PARP-1, which help maintain the stability of the
internal environment following stimulation under physiological
conditions (15). Autophagy is the
evolutionarily conserved, lysosome-dependent degradation of
cytoplasmic contents (16,17) and is associated with various human
diseases and physiological processes, including cancer,
neurodegeneration, microbial infections and aging (18–21).
Numerous anticancer drugs simultaneously induce apoptosis and
autophagy to varying degrees, presenting novel directions for drug
discovery and development (22,23).
Chen et al (24) discovered
that berberine, by decreasing expressions of cyclooxygenase-2
(COX-2), MMP-2, and MMP-9 in A549 cells, induces upregulation of
p21 and p53 expressions, thereby exerting an inhibitory effect on
tumor development. The demethylsonodione aporphine alkaloid
isolated from Corydalis saxicola Bunting DC. can induce
apoptosis of T2 tumor cells through the ROS/p38 MAPK signaling
pathway (25). The present
investigated the effects of apoptosis and autophagy induced by
alkaloids from Fritillaria cirrhosa D.Don on the viability
of NSCLC cells.
Network pharmacology provides a powerful approach
for elucidating the multi-component, multi-target and multi-pathway
mechanisms of action of traditional Chinese medicines. The present
study aimed to identify the anticancer potential of alkaloids in
Fritillaria through network pharmacology screening and
calculation, as well as perform protein-protein interaction (PPI)
network analysis and enrichment to identify proteins with the most
significant associations for further validation, to provide a
reference for subsequent in vitro activity verification.
Materials and methods
Construction of a biological target
network of alkaloids for NSCLC
First, alkaloids from the literature were compared
in the Traditional Chinese Medicine Systems Pharmacology Database
(tcmsp-e.com/index.php). Alkaloids with an OB value of ≥30% were
selected for the next prediction and their structures were
retrieved from the PubChem (pubchem.ncbi.nlm.nih.gov/) database.
The Simplified Molecular Input Line Entry System (SMILES)
representations of these alkaloids were subsequently imported into
the Swiss Target Prediction database (swisstargetprediction.ch),
where only targets with prediction probability scores >0.1 were
retained as potential drug targets. Subsequently, the key words
‘non-small cell lung cancer’ were searched in the Genecards
database (https://www.genecards.org) to
identify relevant targets. Based on the binary relationships
between NSCLC and its targets, a target network containing 5,388
nodes was constructed. Finally, the Cytoscape (cytoscape.org/,
version 3.8.2) was applied to create a combined
‘drug-component-target-disease’ network diagram.
Construction of a drug target PPI
network
In total, 493 potential targets of 139 alkaloids and
5,387 NSCLC-related targets were imported into the Venny
(bioinfogp.cnb.csic.es/tools/venny/, version 2.1.0) software, and
the intersection of the two datasets was evaluated, which led to
the identification of 304 common targets. These targets were then
input into the STRING database (https://cn.string-db.org/) for analysis, with the
organism restricted to ‘Homo sapiens’. The minimum
interaction threshold was established at 0.4, resulting in the
generation of target protein networks. The data were subsequently
imported into Cytoscape (version 3.8.2) to construct a PPI network
map.
Screening for HUB genes
The PPI subnetwork was analyzed using the MCODE
(version 2.0.3) clustering algorithm
(apps.cytoscape.org/apps/mcode); Cytoscape (version 3.8.2) and the
MCODE plug-in were used for network topology analysis with the
following parameters: Degree cut-off=2, a node score cut-off=0.2, a
K-core of 2 and a maximum depth=100. Network topology parameters
(degree, betweenness and closeness centrality) for these targets
were obtained using the Analysis Network tool in Cytoscape. The
CytoNCA (version 2.1.6; cytoscape.org/apps/cytonca) and CytoHubba
(version 0.1) plugins (apps.cytoscape.org/apps/cytohubba) were used
to identify HUB genes. Finally, a Venn diagram was constructed to
identify the overlapping targets.
Functional enrichment analysis of the
PPI core subnetwork
The core subnetworks derived from the clustering
analysis of the PPI network were analyzed using the STRING database
and DAVID databases (david.ncifcrf.gov/summary.jsp), Reactome
(reactome.org/) and WikiPathways (wikipathways.org/) databases for
the enrichment analysis (Gene Ontology (GO; geneontology.org/) and
Kyoto Encyclopedia of Genes and Genomes (KEGG; kegg.jp/)). The
enrichment results were organized by intensity, gene count (≥15)
and P≤0.05, ranked from high to low scores, with the top 20
selected for visualization.
Experimental materials and
reagents
Imperialine, verticinone, verticine and peimisine
were purchased from Chengdu Push Bio-Technology Co., Ltd.,
imperialine-3-β-D-glucoside and delavine were obtained from Chengdu
Herbpurify Co., Ltd., and ebeiedinone and delavinone from Chengdu
Must Bio-Technology Co., Ltd. The purity of all compounds was
>98%. The Annexin V-FITC/propidium iodide (PI) apoptosis
detection kit was acquired from Dojindo Laboratories, Inc. The BCA
protein kit and RIPA buffer were from Beyotime Institute of
Biotechnology and the protease inhibitor cocktail was from Roche
Diagnostics. Phenylmethylsulfonyl fluoride and phosphatase
inhibitor were purchased from Beijing Solarbio Science &
Technology Co., Ltd. Chloroquine (CQ) and dimethyl sulfoxide were
obtained from MilliporeSigma, and the Cell Counting Kit-8 (CCK-8)
from Beijing Solarbio Science & Technology Co., Ltd. Antibodies
specific for PARP (cat. no. CY5347) and cleaved PARP (cat. no.
CY5265) were obtained from Shanghai Abways Biotechnology Co., Ltd.;
antibodies for cleaved caspase-3 (cat. no. 9664T), LC3A/B (cat. no.
12741), GAPDH (cat. no. 2118) and HRP-conjugated goat anti-rabbit
IgG secondary antibody (cat. no. ab6721) were obtained from Cell
Signaling Technology, Inc. All other chemical reagents used were of
analytical grade.
NSCLC cell lines and cultures
In total, three non-small cell lung cancer cell
lines, NCI-H1975 (CRL-5908), NCI-H1299 (CRL-5803) and A549
(CCL-185), were obtained from the American Type Culture Collection.
The cell lines were maintained in RPMI-1640 medium supplemented
with 10% (v/v) fetal bovine serum (FBS), 100 U/ml penicillin and
100 µg/ml streptomycin (all Gibco; Thermo Fisher Scientific, Inc.
at 37°C in a 5% CO2 atmosphere, and the cell culture
medium was replaced daily.
Cell viability assay
A single-cell suspension in the logarithmic growth
phase was prepared and counted. Subsequently, 100 µl of the
single-cell suspension were added to each well of a 96-well plate,
with a concentration of 4×103 cells/well for the A549
cells and 5×103 cells/well for the NCI-H1299 cells,
diluted with complete medium. Control groups included blank control
group, negative control group and experimental groups, with each
group containing ≥4 replicate wells. Following 48 h of drug
treatment, 10 µl CCK-8 regent were added to each well. The wells
were then incubated in the dark for 4 h, and the absorbance was
measured at 450 nm using a microplate reader (Bio-Rad Laboratories,
Inc.).
Annexin V-FITC/PI staining assay
Early and late apoptosis cells were evaluated using
flow cytometry. In brief, NCI-H1299 and A549 cells were treated
with ebeiedinone (10, 15, 20 and 30 µM), verticinone (15, 30 µM)
and gefitinib (15, 30 µM) for 48 h in a 37°C incubator. A blank
control without medication and a 5-fluorouracil group (15, 30 µM)
were set up. 5-Fluorouracil was used as a positive control drug for
apoptosis to determine whether cells can undergo apoptosis.
Gefitinib is used as a positive drug for comparison with
Verticinone and ebeiedinone, to determine the ability to induce
apoptosis. For the flow cytometry experiments, the cells were
stained with 5 µl annexin V-FITC and 5 µl PI at room temperature
for 15 min. The fluorescence intensity was measured using a BD
FACSCalibur flow cytometer (Becton Dickinson and Company), and the
apoptotic rates were analyzed using the NOVAexpress software
(version 1.6.2; Agilent).
Western blot analysis
A549 cells were harvested and lysed using a nuclear
and cytoplasmic protein extraction kit (Beyotime Biotechnology;
cat. no. P0028,) according to the manufacturer's instructions.
Protein concentrations were determined using the BCA protein assay
kit. A total of 20 µg of protein sample to each well. Cytoplasmic
proteins were separated by 7 and 15% SDS-PAGE and transferred to a
polyvinylidene difluoride membrane (MilliporeSigma). The membranes
were blocked with 5% skimmed milk at room temperature for 1 h,
followed by incubation with primary antibodies at 4°C overnight.
This was followed by incubation with goat anti-rabbit IgG (H&L)
(cat. no. ab6721; 1:10,000; Abcam, Cambridge, UK) at 37°C for 2 h.
Protein bands were visualized using the Immobilon Western HPR
Substrate (MilliporeSigma) and images were captured with the
GelView 6000Plus Chemiluminescence Imaging System (Guangdong
Biolight Meditech Co., Ltd.). The Image J software (version 1.54;
National Institutes of Health) was used for protein quantification.
The following are the primary antibodies used in this experiment:
GAPDH (14C10) Rabbit Monoclonal Antibody (cat. no. 2118; 1:1,000;
Cell Signaling Technology); β-tubulin (9F3) Rabbit Monoclonal
Antibody (cat. no. 2128; 1:1,000; Cell Signaling Technology, Inc.);
PARP 1 Ab (cat. no. CY5347; 1:1,000); Cleaved PARP (cat. no.
CY5265; 1:1,000; both Shanghai Abways Biotechnology Co., Ltd.);
Cleaved caspase-3 (cat. no. 9664T; 1:1,000; Cell Signaling
Technology), LC3A/B (D3U4C) Rabbit Monoclonal Antibody (cat. no.
12741; 1:1,000; Cell Signaling Technology; The United States). p62
(D1Q5S) Rabbit Monoclonal Antibody (cat. no. 39749; 1:1,000; Cell
Signaling Technology; The United States)
Statistical analysis
All data are presented as the mean ± standard
deviation. All statistical analyses were performed using GraphPad
Prism software (version 7.0; Dotmatics). Statistical analyses were
performed as follows: a one-way ANOVA was used for multi-group
comparisons, with Tukey's HSD post hoc test applied for further
pairwise analysis. The Student's t-test (unpaired) was used for
direct comparisons between two groups. P<0.05 was considered to
indicate a statistically significant difference.
Results
Alkaloid-NSCLC biological target
network
Following a comprehensive literature search and
retrieval of data from PubChem records, the SMILES representations
of alkaloids in Fritillaria were imported into the Swiss
Target Prediction program to identify drug targets that met the
specified criteria (Table SI).
Target genes for NSCLC were obtained following retrieval from the
GeneCards database. These data were integrated to construct a
‘drug-component-target-disease’ network using Cytoscape (Fig. 1). The degree-based ranking of the
top 20 pharmacologically active compounds is summarized in Table I.
 | Table I.Top 20 components in the
alkaloid-NSCLC target network. |
Table I.
Top 20 components in the
alkaloid-NSCLC target network.
| Compound | Degree | Stress | Average shortest
pathlength | Betweenness
centrality | Closeness
centrality | (Refs.) |
|---|
| Sevedinine | 84 | 228,576 | 2.289351852 | 0.028746974 | 0.436804853 | (77) |
| Hupehenidine | 61 | 87,532 | 2.395833333 | 0.018630794 | 0.417391304 | (86) |
|
(3β,5α,13α,23β)- | 57 | 108,124 | 2.414351852 | 0.011970814 | 0.414189837 | (97) |
|
7,8,12,14-tetradehydro- |
|
|
|
|
|
|
|
5,6,12,13-tetrahydro- |
|
|
|
|
|
|
|
3,23-dihydroxyveratraman-6-one |
|
|
|
|
|
|
| Yubeinine | 54 | 115,480 | 2.428240741 | 0.007531494 | 0.411820782 | (37) |
| Korseveramine | 53 | 114,668 | 2.43287037 | 0.008313714 | 0.411037108 | (72) |
|
(20R,25R)-23,26-epimino- | 53 | 47,348 | 2.43287037 | 0.014789158 | 0.411037108 | (132) |
|
3β-hydroxy-5α-cholest- |
|
|
|
|
|
|
|
23(N)-ene-6,22-dione- |
|
|
|
|
|
|
|
3-0-β-D-glucopyranoside |
|
|
|
|
|
|
| Pingbeinone | 50 | 102,036 | 2.446759259 | 0.006712711 | 0.408703879 | (64) |
| Taipaienine | 46 | 94,116 | 2.465277778 | 0.00482595 | 0.405633803 | (5) |
| Sevedinedione | 46 | 78,006 | 2.465277778 | 0.008048579 | 0.405633803 | (78) |
| Ziebeimine | 44 | 80,910 | 2.474537037 | 0.004788552 | 0.404115996 | (56) |
| Pengbeimine C | 43 | 76,382 | 2.479166667 | 0.004497064 | 0.403361345 | (104) |
| Sewertzidine | 42 | 81,864 | 2.483796296 | 0.003411146 | 0.402609506 | (73) |
| Puqienine D | 42 | 69,234 | 2.483796296 | 0.004562118 | 0.402609506 | (95) |
| Pengbeimine A | 42 | 72,362 | 2.483796296 | 0.003643213 | 0.402609506 | (101) |
| Fetisinine | 42 | 44,608 | 2.483796296 | 0.009557058 | 0.402609506 | (137) |
| N-oxide of
verticinone | 40 | 54,080 | 2.493055556 | 0.009314646 | 0.401114206 | (13) |
| Hupehenizine | 40 | 70,112 | 2.493055556 | 0.003786804 | 0.401114206 | (19) |
| Ebeienine | 40 | 54,296 | 2.493055556 | 0.008436484 | 0.401114206 | (21) |
| Delafrinone | 39 | 69,290 | 2.497685185 | 0.003396726 | 0.400370714 | (49) |
| Ussurienone | 39 | 49,010 | 2.497685185 | 0.005331877 | 0.400370714 | (63) |
Drug target PPI network screening for
HUB genes
A Venn diagram (Fig.
2A) was generated using Venny (version 2.1.0) to identify the
intersection between 493 potential targets from 139 alkaloids in
Fritillaria and 5,387 disease targets associated with NSCLC.
In total, 304 common targets were identified within the
NSCLC-related and potential alkaloid target sets. These common
targets were subsequently input into the STRING database to
retrieve target protein networks, and the results were imported
into the Cytoscape software (version 3.8.2) to create a drug target
PPI network diagram (Fig. 2B).
Cytoscape (version 3.8.2) and the MCODE plug-in were used to
analyze the topology of the PPI network. The results revealed
several targets with high degree values within this PPI network,
indicating a greater number of interactions with other proteins.
The top 20 targets, with degree values ranging from 80 to 170, are
illustrated in Fig. 2C.
In the MCODE cluster analysis, the PPI network was
divided into 11 subnetworks (Table
II), of which four contained >10 nodes (Fig. 3A and B). In addition, further
CytoHubba analysis was performed on the PPI network, yielding the
top 20 targets with the highest scores (Fig. 3C) and CytoNCA analysis was also
conducted. Finally, the intersection of the enriched Cluster 1,
CytoNCA and CytoHubba-related targets was identified using a Venn
diagram, yielding 17 common targets (Fig. 3D), namely: MDM2, EP300, PIK3CA,
STAT3, ERBB2, EZH2, GSK3B, HSP90AA1, MTOR, PARP1, JUN, AKT1, IGF1R,
ESR1, SRC, BCL2L1 and JAK2. These targets are associated with tumor
apoptosis, tumor invasion and immune evasion (26–29).
Among these, BCL2 is a typical anti-apoptotic protein in the
apoptotic pathway, providing insights for subsequent experimental
validation (30).
 | Table II.Cluster analysis of the
alkaloid-NSCLC target PPI network. |
Table II.
Cluster analysis of the
alkaloid-NSCLC target PPI network.
| Cluster | Score | Nodes | Edges | Node targets |
|---|
| 1 | 34.723 | 48 | 816 | ABL1, AKT1, AR,
AURKA, BCL2L1, BRD4, CCNE1, CDK1, CDK2, CDK4, CHEK1, CHUK, EGFR,
EP300, ERBB2, ESR1, ESR2, EZH2, FGF2, FLT1, GSK3B, HDAC1, HDAC2,
HDAC6, HSP90AA1, IGF1R, IL2, JAK2, JUN, KDR, MAP2K1, MAPK1, MAPK14,
MCL1, MDM2, MET, MTOR, PARP1, PGR, PIK3CA, PPARG, PTGS2, PTPRC,
RPS6KB1, SRC, STAT3, SYK, XIAP |
| 2 | 13.053 | 39 | 248 | ACE, AKT2, ALK,
APP, BIRC2, BIRC3, CASP3, CASP7, CDC25A, CREBBP, CYP19A1, ERBB4,
FGFR1, FLT, FYN, GRB2, HDAC3, ICAM1, IKBKB, IKBKG, JAK1, JAK3, KIT,
LCK, LGALS3, MAPK8, MDM4, NR3C1, NTRK1, PDGFRA, PIK3CB, PIK3CG,
PLK1, PRKCA, PRKCD, PRKDC, PTPN1, TP53 |
| 3 | 4.880 | 26 | 61 | SYK, IKBKB, AURKA,
LGALS3, MELK, TTK, PIK3CG, NOS2, CDC45, KIF11, ACE, CHUK, CDC25B,
IKBKG, CDK9, CDK7, INSR, NCOR2, CCNE2, YES1, HDAC3, TLR9, AURKB,
TYK2, VDR, PIK3CB |
| 4 | 4.308 | 14 | 28 | OPRM1, MAOA,
CHRNA4, F2R, ACHE, PTGER3, CHRNA3, CHRM2, HTR3A, DNM1, CYP24A1,
TBXAS1, DRD2, CYP27B1 |
| 5 | 4.000 | 4 | 6 | CTSD, CTSL, CTSB,
CTSK |
| 6 | 4.000 | 4 | 6 | SCD, ACACA, NR1H3,
FABP4 |
| 7 | 3.111 | 10 | 14 | CCR3, CHRNA7,
CHRM3, PLA2G2A, CHRM1, PTGER1, PTGFR, LTA4H, PTGER2, PTGES |
| 8 | 3.000 | 3 | 3 | PTPN22, IRAK1,
TLR7 |
| 9 | 3.000 | 3 | 3 | CYP3A4, DPP4,
NR1H4 |
| 10 | 3.000 | 3 | 3 | ADRB2, DRD4,
CRHR1 |
| 11 | 2.667 | 7 | 8 | P2RX7. ADORA1,
GRIN1, CACNA1G, KCNQ1, KCNK3, GABBR1 |
Functional enrichment analysis
The core subnetworks derived from the PPI network
clustering analysis were examined using the STRING database for
enrichment using GO, KEGG, Reactome and WikiPathways (Fig. 4). The results were organized in
descending order based on the enrichment intensity and P-value.
In the GO enrichment analysis, cluster 1 was found
to be enriched in 1,026 biological process terms and 104 molecular
function terms (P≤0.05). Additionally, a total of 58 cellular
component terms and the top 20 enriched pathways are presented in
Fig. 4A-C. KEGG pathway enrichment
analysis revealed the enrichment of 151 pathways in cluster 1,
showing association with ‘Pathways in cancer’, ‘PI3K-Akt signaling
pathway’, ‘EGFR tyrosine kinase inhibitor resistance’ and other
tumor-related pathways ranked highest in the enrichment index
(Fig. 4D).
Reactome pathway enrichment analysis demonstrated
the enrichment of 281 pathways in cluster 1, with numerous
tumor-associated pathways ranked highly in the enrichment index.
These include pathways related to ‘AKT-mediated inactivation of
FOXO1A’, ‘PTK6 Regulates cell cycle’, ‘signalling to STAT3’, ‘MET
activated STAT3’ and other tumor signal transduction pathways
(Fig. 4E). The WikiPathway
enrichment analysis revealed the enrichment of 263 pathways in
cluster 1. Among these, the ‘Robo4 and VEGF signaling pathways
crosstalk’, ‘TCA cycle nutrient use and invasiveness of ovarian
cancer’, ‘serotonin receptor 2 and STAT3 signaling’ and various
other tumor-related pathways were highly ranked (Fig. 4F).
Based on the results of pathway enrichment analysis
using different methods, it was preliminarily hypothesized that the
active components in Fritillaria cirrhosa may exert certain
effects on NSCLC. Combined with the results of core target
enrichment analysis, it was considered that these effects may be
achieved by promoting tumor cell apoptosis and autophagy and
inhibiting immune escape, providing a basis for further
verification.
Anti-proliferative ability of
isosteroid alkaloids
The enrichment analysis suggested that the
anticancer activity of alkaloids is closely related to the
regulation of autophagy and apoptosis. Previous reports have
indicated that alkaloids exert their anticancer effects through
anti-inflammatory feedback (31,32).
Furthermore, our previous research has shown that imperialine,
verticinone, verticine, imperialine-3-β-D-glucoside, delavine, and
peimisine can inhibit cigarette-induced oxidative stress and
inflammatory responses in RAW264.7 cells (33). Based on the aforementioned network
pharmacology results, this study ultimately selected imperialine,
verticinone, verticine, imperialine-3-β-D-glucoside, delavine,
peimisine, ebeiedinone, and delavinone as the subjects of
investigation (Fig. 5). The effects
of these eight distinct isosteroid alkaloids (Fig. 5A-H) derived from Fritillaria
on the viability of three types of NSCLC cells were evaluated using
CCK-8 assay. No significant effects on the viability of NCI-H1975
cells were observed for the majority of the alkaloids (Fig. 5I). By contrast, the survival rates
of the NCI-H1299 and A549 cells treated with verticinone and
ebeiedinone decreased to 75.57±4.80% and 68.9±3.06% (at 15 µM), and
65.75±6.44% and 67±3.20% (at 30 µM), respectively (Table SII, Table SII, Table SIII, Table SIV). Based on these findings,
verticinone and ebeiedinone were selected for further analyses.
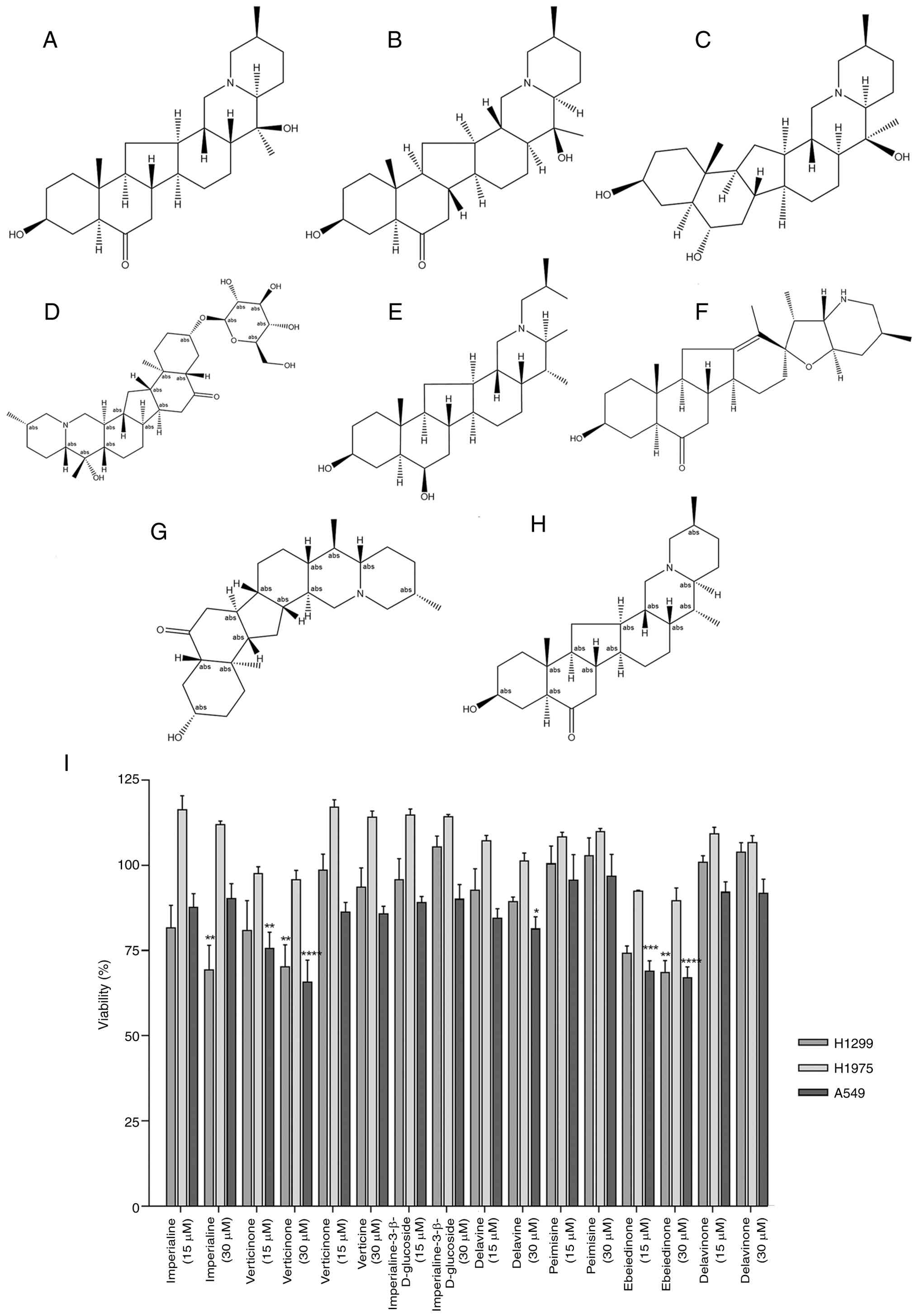 | Figure 5.Structures of isosteroid alkaloids
and their anti-proliferative effects of NSCLC cells. (A)
Imperialine, (B) verticinone, (C) verticine, (D)
imperialine-3-β-d-glucoside, (E) delavine, (F) peimisine, (G)
ebeiedinone and (H) delavinone. (I) Effects of isosteroid alkaloids
on the viability of three different cell lines, H1299, H1975 and
A549. Cells were treated with alkaloids at doses of 15 and 30 µM
for 48 h. *P<0.05, **P<0.01, ***P<0.001, ****P<0.0001
vs. control. NSCLC, non-small cell lung cancer. |
Induction of apoptosis by isosteroidal
alkaloids
To investigate the potential mechanisms underlying
the alkaloid-induced decrease in cell viability, cell death was
detected using the Annexin V-FITC/PI dual labeling assay. The
number of Annexin V-FITC-positive NCI-H1299 cells was comparable
between the groups (Fig. 6). The
group treated with gefitinib exhibited rates of 10.42 and 19.79% at
concentrations of 15 and 30 µM, respectively (Fig. 6A-G). The number of Annexin
V-FITC-positive A549 cells increased following treatment with
ebeiedinone (Fig. 7A-G).
Subsequently, experiments with a range of ebeiedinone
concentrations were conducted. The results indicated that
ebeiedinone promoted apoptosis in a concentration-dependent manner
(Fig. 8A-H). Furthermore, analysis
revealed that cells treated with dehydro-eudobelin (30 μM)
exhibited significant differences compared to the blank control
group, indicating that dehydro-eudobelin has the ability to induce
apoptosis (Fig. 8I).
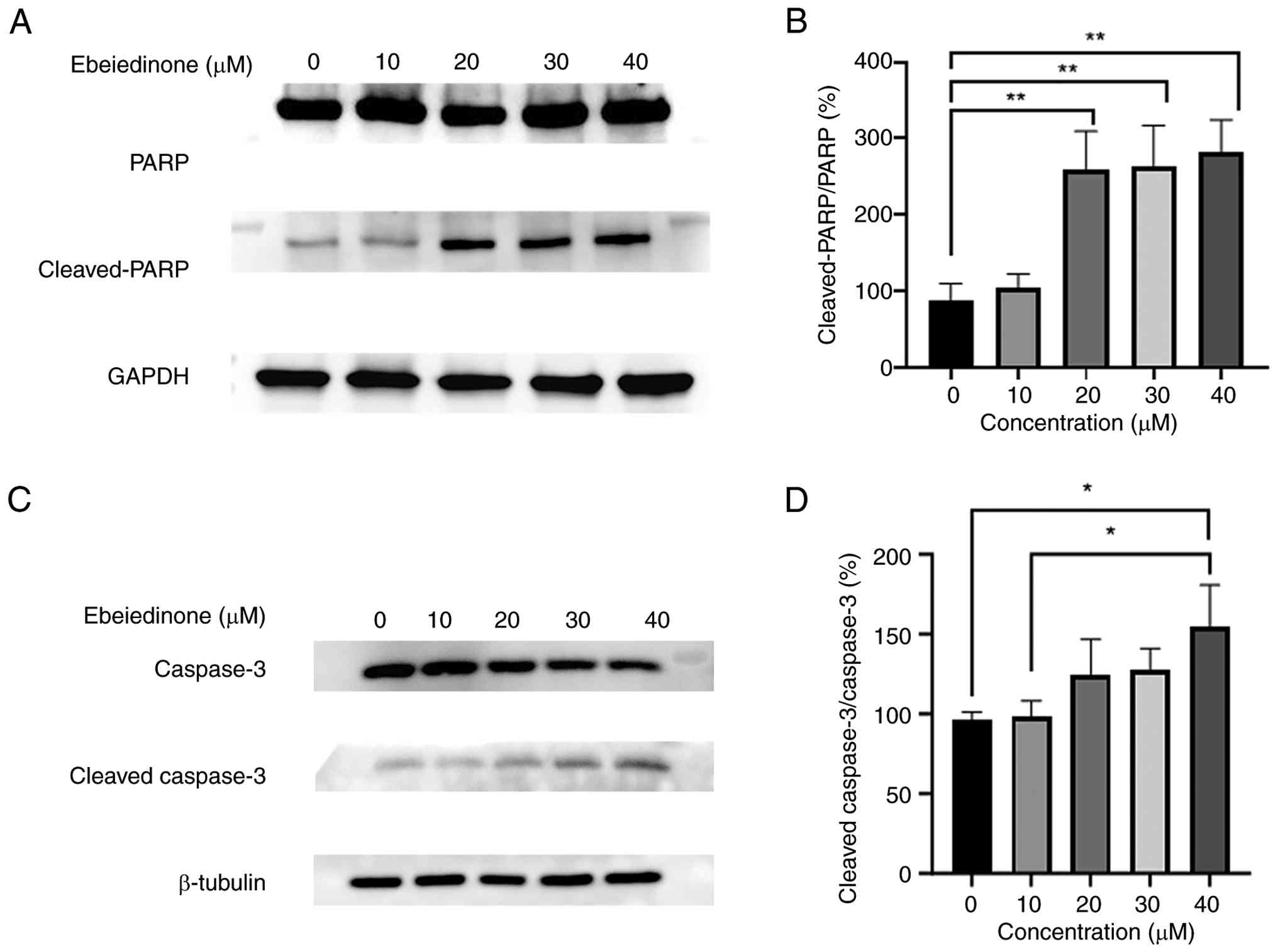
Effects of ebeiedinone on apoptosis-
and autophagy-related protein expression in A549 cells
Apoptosis is a key biological process in
multicellular organisms, characterized in part by the activation of
caspases and the induction of caspase-dependent cell death.
Caspases are regarded as the central mediators of apoptosis
(34). PARP, along with genomic
stability factors and DNA repair enzymes, was originally identified
as a key proteolytic substrate that is degraded by caspase-3 and
other cysteine proteases during apoptosis (35). Increased levels of cleaved PARP and
cleaved caspase-3 in the cells exposed to ebeiedinone were
demonstrated in the present study (Fig.
9). The increase in the level of cleaved caspase-3 was observed
across various drug concentrations and was comparable to the effect
induced by the positive control drug, gefitinib (Fig. 9A and C). Protein expression levels
of cleaved PARP/PARP and cleaved capase-3/GAPDH significantly
increased (Fig. 9B and D). In
summary, these findings demonstrated that ebeiedinone significantly
stimulated the apoptosis of NSCLC cells.
Subsequently, it was investigated whether
ebeiedinone induces autophagy in A549 cells by examining the
expression of LC3-II, a protein marker of autophagy (Fig. 10). Ebeiedinone induced LC3-II
protein expression in a concentration-dependent manner in A549
cells. Furthermore, when ebeiedinone was used in combination with
the autophagy inhibitor, CQ (10 µM, 1 h), which disrupts lysosomal
function to block autophagy (36),
LC3-II expression was further upregulated (Fig. 10A and C). Additionally, the effects
of ebeiedinone on p62 protein expression levels were assessed,
which demonstrated that the ebeiedinone reduces p62 expression,
thereby promoting cellular autophagy (Fig. 10B and D). These results further
indicated that ebeiedinone could promote autophagy in A549
cells.
Discussion
The present network-based pharmacology analysis
suggested that the therapeutic activity of alkaloids in
Fritillaria against NSCLC involves the interactions of
>300 proteins. The alkaloids may exert antitumor effects by
influencing various signaling pathways and interfering with tumor
cell components, molecular functions and biological processes. The
present study also collected relevant core targets such as MDM2,
EP300, PIK3CA and STAT3 through cluster analysis, and found that
these targets might be related to apoptosis and autophagy. Pathway
enrichment analysis of this study, it was found that the alkaloids
in Fritillaria might affect NSCLC through the PI3K-Akt
signaling pathway.
PI3K-Akt signaling pathway is an important pathway
for cell apoptosis and autophagy. In numerous types of human
cancer, the constitutively active oncoprotein, AKT1, suppresses
autophagy (37). In lung cancer,
the inhibition of AKT is also strongly associated with increased
apoptosis and autophagy. Luo et al. (38) discovered that SLL-1A-16 can induce
autophagy in NSCLC cells by inhibiting the Akt/mTOR pathway, and
simultaneously upregulate the expression of caspase-3 and Bax,
significantly inducing cell apoptosis. Mechanistically, apoptosis
is a complex, multi-step process primarily driven by a
caspase-dependent proteolytic cleavage cascade. Akt promotes cell
survival by blocking pro-apoptotic proteins. Akt negatively
regulates the function and expression of Bcl-2 homology domain 3
(BH3) proteins, which inactivate members of the Bcl-2 family by
binding to them. As a result, caspase-9, a downstream protein of
Bcl-2, cannot be cleaved, thereby inhibiting the subsequent cascade
of events and preventing cell apoptosis (39). Furthermore, the PI3K/Akt signaling
pathway is also the core pathway of autophagy (40). The inhibition of Akt can further
suppress the expression of mTOR, thereby promoting the increase of
LC3, while P62 is degraded, and eventually autophagy occurs in the
cell. Previous studies (33) have
shown that certain alkaloids in Fritillaria cirrhosa reduce
alleviate oxidative stress and inflammation in RAW264.7
macrophages. Since macrophages serve as the primary immune cells in
the tumor microenvironment, their oxidative stress and inflammatory
responses are key to polarization. Therefore, it was hypothesize
that the aforementioned alkaloids (imperialine, verticinone,
verticine, imperialine-3-β-D-glucoside, delavine, peimisine,
ebeiedinone, and delavinone) may also possess certain antitumor
effects. Concurrently, our previous work also demonstrated that
ebeiedinone exerts a protective effect on human bronchial
epithelial cells (BESA-2B) (41).
Zhang et al (42) found that
delavinone exhibits anti-colonic cancer activity. In summary, the
results of preliminary experiments, literature reviews, and network
pharmacology analyses suggest that the alkaloids in
Fritillaria may induce apoptosis and autophagy, thus
providing a foundation for subsequent in vitro validation.
Notably, in the present study, the H1299 cells did not undergo
apoptosis, even upon treatment with concentrations that induced
significant cytotoxicity. This finding may be attributed to two
possible factors. Firstly, H1299 is a cell line devoid of the p53
gene, which is highly associated with various types of human
tumors. The deletion of p53 may therefore reduce the sensitivity of
cells to initiation of the apoptotic program (15,43).
Secondly, the inhibition of H1299 cell proliferation by
isosteroidal alkaloids may not involve apoptosis and instead, may
occur through pyroptosis or iron-triggered cell death. Further
research is warranted to determine the underlying reasons and
associated mechanisms.
The present study primarily investigated the in
vitro antitumor activity of two bioactive alkaloids; however,
their in vivo pharmacokinetics and mechanisms remain
insufficiently characterized. In previous research, Wu et al
(44) and Jin et al
(45) independently conducted in
vivo pharmacokinetic analyses of verticinone and ebeiedinone,
respectively. Wu et al employed liquid chromatography-mass
spectrometry (LC-MS) to characterize the pharmacokinetic profile of
verticinone in rats, including its plasma protein binding rate
in vitro. Both oral and intravenous bioavailability were
markedly higher in male rats (oral bioavailability, 45.8% in males
vs. 2.74% in females). Tissue distribution analysis revealed
extensive penetration, with the highest affinity observed in
intestinal tissue (intestine > stomach > liver > kidney
> spleen > lung > heart > gonads > muscle > skin
> fat > brain). Notably, verticinone exhibited high plasma
protein binding rates across species (89.8–91.3% in rats vs.
89.5–94.6% in humans), suggesting negligible interspecies
variability. These findings collectively indicate that first-pass
metabolism critically governs verticinone pharmacokinetics, leading
to sex-dependent exposure in rodents. Jin et al developed a
validated ultra-performance LC-MS/MS method to evaluate ebeiedinone
pharmacokinetics in mice. Their data revealed
concentration-dependent oral bioavailability, with an average
absolute bioavailability of 30.60%. Prolonged mean residence time
and elimination half-life following oral administration compared to
intravenous injection further suggested substantial first-pass
effects.
Furthermore, as a bioactive isosteroidal alkaloid
derived from F. cirrhosae bulbus (a traditional Chinese
medicinal herb), ebeiedinone demonstrates a lower pro-apoptotic
efficacy compared with conventional agents such as cisplatin or
paclitaxel. This observation aligns with the characteristic
multi-targeted, gradual modulation of apoptotic pathways often seen
with phytochemicals.
For example, a study by Li et al (46) demonstrated that total alkaloids from
Stephania tetrandra (TAS) could induce apoptosis in A549 and
H1299 cells. However, at concentrations far below the
IC50, the proportions of early and late apoptotic cells
showed no significant difference compared with that of the control
group. This indicates that while TAS possesses pro-apoptotic
activity, its effect is not pronounced at low doses. Notably, it
was found that TAS could enhance the sensitivity of NSCLC cells to
cisplatin and exert a synergistic effect with cisplatin to
collectively increase apoptosis. Although that study did not delve
deeper into the underlying mechanism, it provides a new perspective
for the present research. Furthermore, it suggests that the limited
direct apoptotic capacity of some natural products might reveal
their potential utility in combination therapies.
The sub-IC50 dosing regimen employed in
the present study was deliberately selected to elucidate
mechanistic nuances, particularly the compound's
concentration-dependent modulation of apoptosis-related proteins,
rather than to elicit maximal cytotoxicity. Notably, western
blotting demonstrated the dose-responsive upregulation of
pro-apoptotic markers upon ebeiedinone treatment, thereby
reinforcing its apoptotic induction capacity despite the limited
Annexin V/PI signal amplitude. Moderated apoptotic effects may
reflect polypharmacological profile of ebeiedinone, which likely
engages parallel cytoprotective pathways to attenuate collateral
damage. Future studies will explore structural optimization and
synergistic combinations to amplify therapeutic potential of
ebeiedinone while preserving the favorable safety trajectory.
However, the present study had certain limitations. It is limited
to in vitro studies of the anti-NSCLC efficacy at the
cellular level and does not investigate its in vivo
efficacy. Furthermore, this study did not delve into the specific
mechanisms by which alkaloids exert their anti-NSCLC effects.
The aforementioned studies provide insight into the
pharmacokinetic behavior of verticinone and ebeiedinone,
establishing a framework for future pharmacodynamic and mechanistic
investigations of these alkaloids in vivo. To the best of
our knowledge, the present study is the first to demonstrate that
ebeiedinone combined with CQ further increases the levels of LC3-II
to promote the autophagic flux in NSCLC cells. The preliminary
findings of the present study confirm the stimulatory effects of
isosteroidal alkaloids in the bulbus Fritillaria cirrhosa on
autophagy and apoptosis in A549 cells, thus warranting further
research.
For the treatment of NSCLC, platinum-based
chemotherapy remains the primary first-line therapy. However, it
has a poor prognosis for metastatic NSCLC and is associated with
severe toxic side effects, such as hearing loss (47,48).
Bispecific antibody drugs are mainstream treatments in the
therapeutic strategy for NSCLC; for example, amivantamab can
simultaneously target both EGFR and MET (48). Nevertheless, these bispecific
antibody drugs often face challenges such as high production costs
due to manufacturing difficulties, risks of immunogenicity, and
severe side effects caused by cytokine release syndrome (CRS)
(49). Although ADC-type
small-molecule drugs are a hot research topic for NSCLC treatment,
they currently pose potential risks to in vivo biosafety
(50). Therefore, identifying new
drugs with good efficacy and low toxicity is a key focus of NSCLC
research. Natural products isolated from medicinal plants represent
a rich reservoir for anti-NSCLC agents. Increasing research has
demonstrated that natural products possess potent anti-NSCLC
activity and are being applied in clinical treatment (51,52).
Ebeiedinone, discovered in this study, warrants further
investigation into its mechanism of action and holds promise for
future application as a novel drug in the clinical treatment of
NSCLC.
Supplementary Material
Supporting Data
Acknowledgements
Not applicable.
Funding
This work was supported by the Science and Technology Projects
of Xizang Autonomous Region, China (grant nos. XZ202303ZY0007G,
XZ202401YD0022, XZ202301ZY0009G) and Noncommunicable Chronic
Diseases-National Science and Technology Major Project (grant no.
2025ZD0551812).
Availability of data and materials
The data generated in the present study may be
requested from the corresponding author.
Authors' contributions
BY and CL designed the study, BY and CL confirm the
authenticity of all the raw data. MT and MZ wrote the manuscript
and analyzed data. JH was responsible for data analysis. WMT and
KWGT performed the literature review and assisted in the
organization and analysis of the cell-related data. All authors
have read and approved the final manuscript.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Sung H, Ferlay J, Siegel RL, Laversanne M,
Soerjomataram I, Jemal A and Bray F: Global cancer statistics 2020:
GLOBOCAN estimates of incidence and mortality worldwide for 36
cancers in 185 countries. CA Cancer J Clin. 3:209–249. 2021.
|
|
2
|
Thai AA, Solomon BJ, Sequist LV, Gainor JF
and Heist RS: Lung cancer. Lancet. 10299:535–554. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Wang M, Herbst RS and Boshoff C: Toward
personalized treatment approaches for non-small-cell lung cancer.
Nat Med. 27:1345–1356. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Kubota K, Kawahara M, Ogawara M, Nishiwaki
Y, Komuta K, Minato K, Fujita Y, Teramukai S, Fukushima M and
Furuse K; Japan Multi-National Trial Organisation, : Vinorelbine
plus gemcitabine followed by docetaxel versus carboplatin plus
paclitaxel in patients with advanced non-small-cell lung cancer: A
randomised, open-label, phase III study. Lancet Oncol. 9:1135–1142.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Choi Y, Kim SA, Jung H, Kim E, Kim YK, Kim
S, Kim J, Lee Y, Jo MK, Woo J, et al: Novel insights into
paclitaxel's role on tumor-associated macrophages in enhancing PD-1
blockade in breast cancer treatment. J Immunother Cancer.
12:e0088642024. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Shafabakhsh R, Pourhanifeh MH, Mirzaei HR,
Sahebkar A, Asemi Z and Mirzaei H: Targeting regulatory T cells by
curcumin: A potential for cancer immunotherapy. Pharmacol Res.
147:1043532019. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Aliomrani M, Sepand MR, Mirzaei HR, Kazemi
AR, Nekonam S and Sabzevari O: Effects of phloretin on oxidative
and inflammatory reaction in rat model of cecal ligation and
puncture induced sepsis. Daru. 24:152016. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Kong W, Wei J, Abidi P, Lin M, Inaba S, Li
C, Wang Y, Wang Z, Si S, Pan H, et al: Berberine is a novel
cholesterol-lowering drug working through a unique mechanism
distinct from statins. Nat Med. 10:1344–1351. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Ma N, Zhang Z, Liao F, Jiang T and Tu Y:
The birth of artemisinin. Pharmacol Ther. 216:1076582020.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Wu F, Tian M, Sun Y, Wu C and Liu X:
Efficacy, chemical composition, and pharmacological effects of
herbal drugs derived from Fritillaria cirrhosa D. Don and
Fritillaria thunbergii Miq. Front Pharmacol. 13:9859352022.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Liu C, Liu S, Tse WM, Tse KWG, Erbu A,
Xiong H, Lanzi G, Liu Y and Ye B: A distinction between Fritillaria
Cirrhosa Bulbus and Fritillaria Pallidiflora Bulbus via LC-MS/MS in
conjunction with principal component analysis and hierarchical
cluster analysis. Sci Rep. 13:27352023. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Lin Q, Qu M, Patra HK, He S, Wang L, Hu X,
Xiao L, Fu Y, Gong T, He Q, et al: Mechanistic and therapeutic
study of novel anti-tumor function of natural compound imperialine
for treating non-small cell lung cancer. J Ethnopharmacol.
247:1122832020. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Wang D, Wang S, Feng Y, Zhang L, Li Z, Ma
J, Luo Y and Xiao W: Antitumor effects of Bulbus Fritillariae
cirrhosae on Lewis lung carcinoma cells in vitro and in vivo. Ind
Crops Prod. 54:92–101. 2014. View Article : Google Scholar
|
|
14
|
Wang Y, Peng M, Yang X, Tu L, Liu J, Yang
Y, Li R, Tang X, Hu Y, Zhang G, et al: Total alkaloids in
Fritillaria cirrhosa D. Don alleviate OVA-induced allergic asthma
by inhibiting M2 macrophage polarization. J Ethnopharmacol.
337:1189352025. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Nakajima Y and Kuranaga E:
Caspase-dependent non-apoptotic processes in development. Cell
Death Differ. 24:1422–1430. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Guo S, Luo W, Liu L, Pang X, Zhu H, Liu A,
Lu J, Ma DL, Leung CH, Wang Y and Chen X: Isocryptotanshinone, a
STAT3 inhibitor, induces apoptosis and pro-death autophagy in A549
lung cancer cells. J Drug Target. 10:934–942. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Debnath J, Gammoh N and Ryan KM: Autophagy
and autophagy-related pathways in cancer. Nat Rev Mol Cell Biol.
24:560–575. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Mizushima N, Levine B, Cuervo AM and
Klionsky DJ: Autophagy fights disease through cellular
self-digestion. Nature. 451:1069–1075. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Li X, He S and Ma B: Autophagy and
autophagy-related proteins in cancer. Mol Cancer. 19:122020.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Tang ZH, Zhang LL, Li T, Lu JH, Ma DL,
Leung CH, Chen XP, Jiang HL, Wang YT and Lu JJ: Glycyrrhetinic acid
induces cytoprotective autophagy via the inositol-requiring enzyme
1 α -c-Jun N-terminal kinase cascade in non-small cell lung cancer
cells. Oncotarget. 6:43911–43926. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Li T, Xu X, Tang Z, Wang Y, Leung C, Ma D,
Chen X, Wang Y, Chen Y and Lu J: Platycodin D induces apoptosis and
triggers ERK- and JNK-mediated autophagy in human hepatocellular
carcinoma BEL-7402 cells. Acta Pharmacol Sin. 36:1503–1513. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Shen J, Zhu X, Wu Z, Shi Y and Wen T:
Uvangoletin, extracted from Sarcandra glabra, exerts anticancer
activity by inducing autophagy and apoptosis and inhibiting
invasion and migration on hepatocellular carcinoma cells.
Phytomedicine. 94:1537932022. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Schaaf MB, Houbaert D, Meçe O and
Agostinis P: Autophagy in endothelial cells and tumor angiogenesis.
Cell Death Differ. 26:665–679. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Chen J, Huang X, Tao C, Wang L, Chen Z, Li
X, Zeng Q, Ma M, Zhang R and Wu Z: Berberine chloride suppresses
non-small cell lung cancer by deregulating Sin3A/TOP2B pathway in
vitro and in vivo. Cancer Chemother Pharmacol. 86:151–161. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Luo L, Luo J, Song X, Wang C, Tang D, Sun
W, Fan C, Li M and Wang H: Alkaloids from Corydalis saxicola and
their antiproliferative activity against cancer cells. Fitoterapia.
173:1057912024. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Yao Y, Zhang Q, Li Z and Zhang H: MDM2:
Current research status and prospects of tumor treatment. Cancer
Cell Int. 24:1702024. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Zeng Y, Zhou Y, Ruan J, Liu W, Fan H, Liu
N, Li L, Ma J, Jin X, Duan L, et al: EP300 compromises antitumor
immunity by increasing SOCS1 expression. J Immunother Cancer.
13:e0114882025. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Duan X, Zhang L and Liu F: The potential
value of quercetin for colorectal cancer: A systematic review and a
meta-analysis of preclinical studies. Front Pharmacol.
16:16429572025. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Takei H, Coelho-Silva JL, Leal CT, Rocha
AQ, Bianco TM, Welner RS, Mishima Y, Kobayashi IS, Mullally A, Lima
K, et al: Suppression of multiple anti-apoptotic BCL2 family
proteins recapitulates the effects of JAK2 inhibitors in JAK2V617F
driven myeloproliferative neoplasms. Cancer Sci. 113:597–608. 2022.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Vogler M, Braun Y, Smith VM, Westhoff M,
Pereira RS, Pieper NM, Anders M, Callens M, Vervliet T, Abbas M, et
al: The BCL2 family: From apoptosis mechanisms to new advances in
targeted therapy. Signal Transduct Target Ther. 10:912025.
View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Fu Y, Guo J, Xie Y, Hua J, Dai Y, Zhang W,
Lin T and Liu Y: Structural characterization, antiproliferative and
anti-inflammatory activities of alkaloids from the roots of
Zanthoxylum austrosinense. Bioorg Chem. 102:1041012020. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Zou K, Li Z, Zhang Y, Zhang HY, Li B, Zhu
WL, Shi JY, Jia Q and Li YM: Advances in the study of berberine and
its derivatives: A focus on anti-inflammatory and anti-tumor
effects in the digestive system. Acta Pharmacol Sin. 2:157–167.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Liu S, Yang T, Ming TW, Gaun TKW, Zhou T,
Wang S and Ye B: Isosteroid alkaloids with different chemical
structures from Fritillariae cirrhosae bulbus alleviate LPS-induced
inflammatory response in RAW 264.7 cells by MAPK signaling pathway.
Int Immunopharmacol. 78:1060472020. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Kumar S, Dorstyn L and Lim Y: The role of
caspases as executioners of apoptosis. Biochem Soc Trans. 50:33–45.
2022. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Carneiro BA and El-Deiry WS: Targeting
apoptosis in cancer therapy. Nat Rev Clin Oncol. 17:395–417. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Mizushima N, Yoshimori T and Levine B:
Methods in mammalian autophagy research. Cell. 140:313–326. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Kaleağasıoğlu F, Ali DM and Berger MR:
Multiple facets of autophagy and the emerging role of
alkylphosphocholines as autophagy modulators. Front Pharmacol.
11:5472020. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Luo X, Wang J, Wang R, Lian J, Guo M, Zhou
H, Zhang M, Yang Z, Li X, He X and Bi X: SLL-1A-16 suppresses
proliferation and induces autophagy in non-small-cell lung cancer
cellsvia the AKT/mTOR signaling pathway. RSC Med Chem.
15:3460–3468. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Manning BD and Cantley LC: AKT/PKB
signaling: Navigating downstream. Cell. 129:1261–1274. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Jeyamohan S, Moorthy RK, Kannan MK and
Arockiam AJV: Parthenolide induces apoptosis and autophagy through
the suppression of PI3K/Akt signaling pathway in cervical cancer.
Biotechnol Lett. 38:1251–1260. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Liu C, Zhu X, Aga E, Tse WM, Tse KWG, Liu
Y and Ye B: Ebeiedinone and peimisine inhibit cigarette smoke
extract-induced oxidative stress injury and apoptosis in BEAS-2B
cells. Cell Stress Chaperones. 29:697–708. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Zhang Y, Cai X, Ma X, Yan H, Wu Q, Tong H
and Zheng Z: Delavinone elicits oxidative stress and triggers
ferroptosis in colorectal cancer by inhibiting PKCδ-mediated
phosphorylation of Nrf2. Chem Biol Interact. 405:1113122025.
View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Hafner A, Bulyk ML, Jambhekar A and Lahav
G: The multiple mechanisms that regulate p53 activity and cell
fate. Nat Rev Mol Cell Biol. 20:199–210. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Wu X, Sun J, Peng Y, Liang Y, Wang G, Chen
H, Wu J and Zhang P: Pharmacokinetics, tissue distribution and
excretion of verticinone from F. Hupehensis in rats. Molecules.
19:20613–20626. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Jin Y, Chen Y, Liu J, Bao X, Zhi Y, Wen C
and Zhu W: Pharmacokinetics of ebeiedinone in mouse blood by
UPLC-MS/MS. Acta Chromatogr. 32:194–198. 2020. View Article : Google Scholar
|
|
46
|
Li B, Chen J, He J, Peng J, Wang Y, Liu S
and Jiang Y: Total alkaloids in Stephania tetrandra induce
apoptosis by regulating BBC3 in human non-small cell lung cancer
cells. Biomed Pharmacother. 162:1146352023. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Niu Z, Jin R, Zhang Y and Li H: Signaling
pathways and targeted therapies in lung squamous cell carcinoma:
Mechanisms and clinical trials. Signal Transduct Target Ther.
7:3532022. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Waissbluth S, Peleva E and Daniel SJ:
Platinum-induced ototoxicity: A review of prevailing ototoxicity
criteria. Eur Arch Otorhinolaryngol. 274:1187–1196. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Coleman N, Hong L, Zhang J, Heymach J,
Hong D and Le X: Beyond epidermal growth factor receptor: MET
amplification as a general resistance driver to targeted therapy in
oncogene-driven non-small-cell lung cancer. ESMO Open.
6:1003192021. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Gu Y, Wang Z and Wang Y: Bispecific
antibody drug conjugates: Making 1+1>2. Acta Pharm Sin B.
14:1965–1986. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Li BT, Smit EF, Goto Y, Nakagawa K,
Udagawa H, Mazières J, Nagasaka M, Bazhenova L, Saltos AN, Felip E,
et al: Trastuzumab Deruxtecan inHER2 -mutant non-small-cell lung
cancer. N Engl J Med. 386:241–251. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Hui Z, Wen H, Zhu J, Deng H, Jiang X, Ye
X, Wang L, Xie T and Bai R: Discovery of plant-derived anti-tumor
natural products: Potential leads for anti-tumor drug discovery.
Bioorg Chem. 142:1069572024. View Article : Google Scholar : PubMed/NCBI
|