Introduction
Breast cancer is the most common malignancy in
women, accounting for ~30% of all new cancer cases among women
(1). Epidemiological data indicated
that since the early 2000s, its incidence has been increasing at an
average annual rate of ~0.6%, posing a major global health
challenge for women (1,2). Although advances in comprehensive
treatment strategies have markedly improved patient survival,
recurrence and metastasis remain notable obstacles in clinical
management. Therapeutic decisions in breast cancer are typically
guided by clinical stage, histological grade and hormone receptor
status. However, breast cancer is inherently heterogeneous
(3); thus, patients with identical
staging and pathological features may experience markedly divergent
outcomes despite receiving the same therapeutic regimen. The
identification of reliable prognostic biomarkers is therefore key
to enabling personalized and precise treatment.
Tumor angiogenesis is key to the growth, invasion
and metastasis of malignancies. In addition to endothelial
cell-driven angiogenesis, an alternative vascular-like structure
originating directly from tumor cells has been identified in the
tumor microenvironment, termed vasculogenic mimicry (VM). First
described by Maniotis et al (4) in uveal melanoma, VM is characterized
by channels formed through the cooperation of tumor cells and
extracellular matrix, mimicking the functional properties of blood
vessels and providing nutrients through a non-endothelium-dependent
mechanism. Subsequent studies have confirmed the presence of VM in
an extensive spectrum of malignancies, including breast (5) and urological cancer (6), ovarian (7) and hepatocellular carcinoma (8).
VM not only contributes to neovascularization and
sustains tumor growth but also serves a pivotal role in tumor cell
migration during its formation (9).
Therefore, VM is implicated not only in proliferation but also in
metastatic dissemination. Multiple studies on breast cancer have
demonstrated that VM is associated with unfavorable prognosis:
Patients with VM+ tumors tend to present with a large
tumor size, advanced stage and short disease-free survival (DFS)
and overall survival (5,10–12).
Further studies have revealed two principal aspects
of the VM-breast cancer relationship. First, VM is strongly
associated with clinicopathological characteristics. VM is
positively associated with tumor size, lymph node metastasis and
clinical stage (13), but
negatively associated with hormone receptor expression, suggesting
that VM+ breast cancer cases often represent more
aggressive subtypes. Of note, VM occurs at a markedly higher
frequency in triple-negative breast cancer (TNBC) compared with
that in non-TNBC cases (14). In
addition, VM has been reported to be positively associated with
human epidermal growth factor receptor 2 (HER2) upregulation
(15), reinforcing its association
with poor prognosis. Secondly, VM is closely associated with
metastatic potential. Cancer stem cells (CSCs) serve a key role in
tumor invasion and dissemination, and previous studies indicated
that CSCs contribute to VM formation (14,16),
suggesting that CSCs may promote metastasis through this mechanism.
In a cohort of 331 surgically treated patients with breast cancer,
Shirakawa et al (17)
reported that VM+ cases were more prone to hematogenous
metastasis and exhibited markedly lower 5-year survival.
Experimental models further demonstrated that VM is strongly
associated with distant metastasis, particularly pulmonary spread
(18).
Due to the central role of tumor vasculature in
disease progression, anti-angiogenic therapies targeting
endothelial cell-mediated angiogenesis have been developed to
starve tumors of nutrients. However, these agents often exhibit
limited efficacy or induce resistance. Previous research suggested
that VM may increase following anti-angiogenic therapy and may
represent a major mechanism of therapeutic resistance (5,19).
Beyond anti-angiogenic agents, resistance to targeted therapies may
also involve VM. For instance, HER2+ breast cancer
exhibits distinct aggressive clinical behavior. The introduction of
HER2-targeted therapies has markedly improved patient outcomes;
trastuzumab reduces recurrence by up to 40% within 10 years from
surgery in early-stage breast cancer (20). In metastatic HER2+
disease, combining trastuzumab with chemotherapy significantly
prolongs the median time to disease progression from 4.6 to 7.4
months (21). Nevertheless,
recurrence persists in 26% of early-stage HER2+ patients
within 10 years despite trastuzumab and >70% of metastatic
HER2+ tumors progress within 1 year under continuous
therapy (20). VM has been
implicated in this resistance, as trastuzumab-insensitive
HER2+ breast cancer cells have been reported to generate
VM structures within angiogenic microenvironments (22).
Collectively, notable evidence supports a strong
association between VM and adverse outcomes in breast cancer.
Elucidating the relationship between VM and clinicopathological
features, and integrating VM with other independent prognostic
factors, may improve the accuracy of predictive models. Such models
could refine postoperative risk stratification, enhance prognostic
evaluation and ultimately inform individualized therapeutic
strategies.
Based on this rationale, the present study
hypothesized that VM was closely associated with an unfavorable
prognosis in breast cancer. The present study systematically
analyzed VM expression alongside clinicopathological parameters,
explored its prognostic significance and constructed a predictive
model combining VM with additional independent risk factors. The
aim of the present study was to more accurately predict the risk of
recurrence and metastasis following surgery, and to potentially
provide a foundation for personalized treatment and clinical
decision-making in the future.
Patients and methods
Clinical data
Study population
Patients who underwent surgery for breast cancer at
the Department of Breast Surgery, Affiliated Hospital of Guangdong
Medical University (Zhanjiang, China), between January 2020 and
April 2022, and met the below inclusion criteria, were included in
the present study. All patients had received standardized surgical
treatment and postoperative paraffin-embedded pathological
specimens were available. The mean age of the overall study cohort
was 49.9±9.2 years (range, 31–73 years). The inclusion criteria
were as follows: i) Female patients newly diagnosed with invasive
breast carcinoma who underwent surgical resection; ii) complete
clinical and follow-up data available; iii) no history of
neoadjuvant chemotherapy, radiotherapy or endocrine therapy prior
to surgery; iv) absence of other concurrent malignancies and no
prior history of cancer; v) availability of intact
paraffin-embedded pathological specimens suitable for
histopathological analysis; and vi) receipt of standardized
adjuvant therapy following surgery.
The exclusion criteria were as follows: i) History
of other malignant tumors; ii) bilateral breast cancer or carcinoma
in situ; iii) male breast cancer; iv) distant metastasis at
diagnosis or lack of standardized adjuvant therapy; and v) poorly
preserved postoperative specimens unsuitable for pathological
assessment.
The histopathological analysis of breast cancer
tissue sections, as well as the use of clinical data in the present
study, were approved by the Ethics Committee of the Affiliated
Hospital of Guangdong Medical University (approval no.
PJKT2025-067; Zhanjiang, China).
Observational parameters
Clinical and pathological data, including age, body
mass index (BMI), menstrual status (premenopausal or
postmenopausal), tumor location (upper outer, lower outer, upper
inner or lower inner quadrant), T stage (T1-T4), N stage (N0, N1-N2
or N3), overall tumor stage (I–III) (23), histological grade (G1-G3) (24), estrogen receptor (ER) status,
progesterone receptor (PR) status, HER2 expression (negative or
positive), Kiel-67 antigen (Ki-67) index, breast surgical approach
(breast-conserving surgery or mastectomy) and axillary surgery type
(sentinel lymph node biopsy or axillary lymph node dissection),
were collected. Laboratory indices included carbohydrate antigen
15-3 (CA153), carcinoembryonic antigen (CEA), serum total
cholesterol, triglycerides, uric acid, alkaline phosphatase,
lactate dehydrogenase and albumin-to-globulin ratio.
Postoperative follow-up data were obtained through
inpatient records, outpatient visits and telephone consultations.
The primary endpoint was DFS, defined as the time from completion
of surgery to the first occurrence of tumor recurrence, distant
metastasis or mortality from any cause. For patients without
events, follow-up was censored at the last contact, with the
cut-off date set at March 2025. Local recurrence or metastasis was
confirmed through enhanced computed tomography (CT), positron
emission tomography-CT, biopsy or surgical pathology, bone
scintigraphy or magnetic resonance imaging.
Methods
Primary reagents
The primary reagents used in the present study are
summarized in Table I.
 | Table I.Primary reagents used in the present
study. |
Table I.
Primary reagents used in the present
study.
| Reagent | Source | cat. no. |
Dilution/concentration |
|---|
| CD34 antibody | Wuhan Servicebio
Technology Co., Ltd. | GB11169 | 1:200 |
| Horseradish
peroxidase-conjugated goat anti-rabbit IgG | Wuhan Servicebio
Technology Co., Ltd. | GB23303 | 1:200 |
| Periodic
acid-Schiff staining kit | Beyotime Institute
of Biotechnology | C0142 | Ready-to-use |
Immunohistochemical and histochemical
double staining for VM
Breast cancer tissues were fixed in 10%
neutral-buffered formalin at room temperature for 24–48 h, and then
paraffin-embedded. Tissues were obtained from the Department of
Pathology, Affiliated Hospital of Guangdong Medical University
(Zhanjiang, China). Sections (5 µm thick) were prepared using a
Leica 2155 rotary microtome (Leica Microsystems GmbH) and baked at
60°C for 1 h prior to staining. VM was identified using CD34
immunohistochemistry combined with periodic acid-Schiff (PAS)
histochemistry, according to the following procedure: i)
Deparaffinization and rehydration: Sections were sequentially
immersed in xylene I/II (10 min each), followed by a graded ethanol
series (100, 95, 80 and 70%) and rinsed in distilled water; ii)
antigen retrieval: Slides were heated to ~95-100°C in citrate
buffer (pH, 6.0) using a microwave oven (8 min at medium power, 8
min pause, then 7 min at medium-low power), cooled to room
temperature and washed three times with PBS (pH, 7.4); iii)
blocking of endogenous peroxidase: Sections were incubated in 3%
hydrogen peroxide for 25 min at room temperature in the dark,
followed by PBS washing; iv) serum blocking: Tissues were covered
with 3% bovine serum albumin and incubated for 30 min at room
temperature; v) primary antibody incubation: CD34 antibody (cat.
no. GB11169; dilution 1:200; Wuhan Servicebio Technology Co., Ltd.)
diluted in blocking buffer was applied and slides were incubated
overnight at 4°C in a humid chamber; vi) secondary antibody
incubation: Following PBS washing, slides were incubated with
HRP-conjugated goat anti-rabbit IgG (cat. no. GB23303; dilution
1:200; Wuhan Servicebio Technology Co., Ltd.) for 50 min at room
temperature; vii) diaminobenzidine (DAB) chromogenic detection:
Visualization was achieved with freshly prepared DAB solution. The
reaction was monitored microscopically and terminated by rinsing
with tap water. Positive staining appeared as brown deposits; viii)
PAS staining: Following CD34 staining, slides were treated with
periodic acid for 10 min, rinsed and incubated with Schiff's
reagent for 30 min at 37°C in the dark. Counterstaining was
performed with hematoxylin for 30 sec at room temperature; and ix)
mounting: Sections were dehydrated through a graded ethanol series,
cleared in xylene and mounted with neutral balsam for evaluation
under a light microscope.
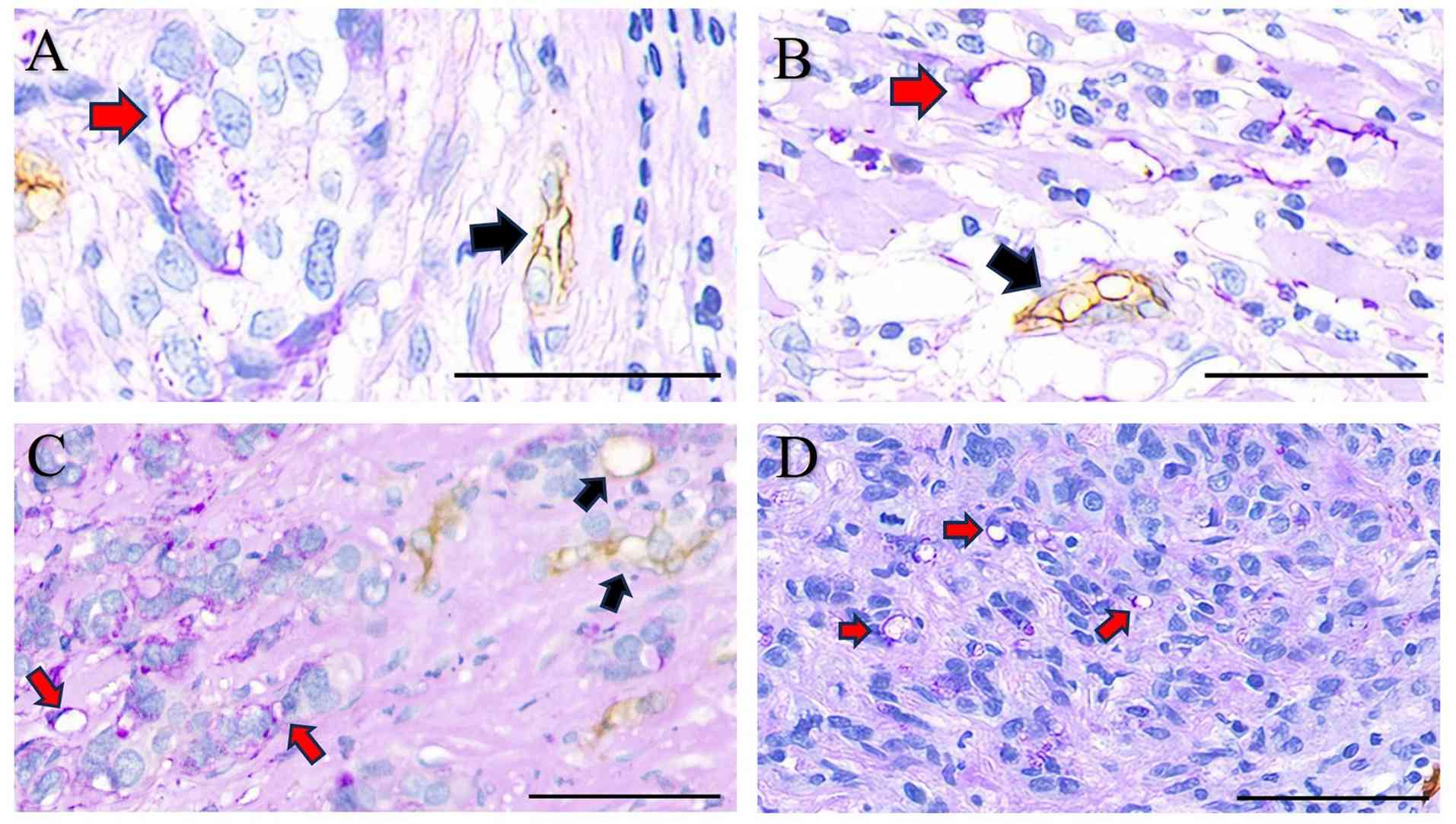
The criteria to identify VM+ and
VM− cases were pre-defined. For each case, five
representative tumor fields were examined under a high
magnification (×200) following CD34/PAS double staining. A
VM+ case was defined as the presence of PAS+,
CD34− patterned networks or vessel-like structures
containing red blood cells, distinct from CD34+
endothelial-lined vessels (4). To
minimize subjective bias, two experienced pathologists
independently assessed all sections in a blinded manner. Discordant
cases were resolved by consensus review. Representative images of
the stained sections were captured under light microscopy.
Statistical analysis
All analyses were conducted using R software
(version 4.2.1; R Foundation for Statistical Computing; http://www.R-project.org/). All immunohistochemical
staining experiments were independently repeated three times to
ensure reproducibility. Patients were categorized into
VM+ and VM− groups. Continuous variables were
expressed as the mean ± SD if normally distributed or as median
with interquartile range if non-normally distributed; categorical
variables were reported as counts and percentages. Group
comparisons were performed using an unpaired Student's t-test or
Wilcoxon rank-sum test for continuous variables and χ2
test or Fisher's exact test for categorical variables. P<0.05
was considered to indicate a statistically significant
difference.
Univariate Cox proportional hazards regression was
used to identify variables associated with DFS. Significant
variables (P<0.05) were entered into a multivariate Cox
regression model with forward stepwise selection used to determine
independent prognostic factors. A nomogram was constructed based on
the multivariate model using the ‘rms’ package (version 6.7–1;
Frank E. Harrell Jr; http://CRAN.R-project.org/package=rms). The prognostic
risk score was computed using the ‘Predict()’ function in R
software, with the following formula: Risk score=exp (2.289 × VM +
1.491 × N stage + 0.057 × Ki-67 index). Internal validation was
performed via bootstrap resampling, with a sample size of 40 per
iteration and 800 repetitions. Model performance was assessed using
the concordance index (C-index), time-dependent receiver operating
characteristic (ROC) curves (using ‘timeROC’ package, version 0.4,
Paul Blanche, https://CRAN.R-project.org/package=timeROC; and
‘ggplot2’ package, version 3.4.4, Hadley Wickham, http://CRAN.R-project.org/package=ggplot2) and
calibration plots. Kaplan-Meier survival curves with log-rank tests
were used to compare DFS between risk groups defined by independent
prognostic factors and the composite risk score derived from the
multivariate model (using ‘survival’ and ‘survminer’ packages,
version 0.4.9, Alboukadel Kassambara, http://CRAN.R-project.org/package=survminer).
DFS was defined as the time from surgery to the
first documented recurrence, metastasis or mortality from any
cause. Patients without events were censored at the last follow-up
date (March 2025).
Results
Histopathological findings
In accordance with the predefined inclusion and
exclusion criteria, a total of 120 patients with breast cancer from
between January 2020 and April 2022 with complete clinical records
were retrospectively identified from the medical database of the
Affiliated Hospital of Guangdong Medical University. All surgical
specimens had been routinely formalin-fixed and paraffin-embedded
in the Department of Pathology.
Dual CD34/PAS immunohistochemical and histochemical
staining was performed on the paraffin sections. VM was detected in
29 cases, defined as PAS+ and CD34− channels
(PAS+/CD34−), whereas 91 cases were classified as
VM− (PAS+/CD34+) (Fig.
1).
Baseline characteristics and
clinicopathological associates of VM
A total of 120 patients were included in the present
study. Clinical information was extracted from initial surgical
admission records. Laboratory indices, except for CA153 and CEA
(measured within 1 week following standardized postoperative
therapy) were obtained within 1 week prior to surgery. Pathological
parameters were derived from postoperative histopathological
assessments conducted in the Department of Pathology, Affiliated
Hospital of Guangdong Medical University.
Of the 120 patients, 29 (24.17%) were VM+
and 91 (75.83%) were VM−. Patients were stratified into
VM+ and VM− cohorts accordingly. The detailed
demographic and clinicopathological characteristics of the cohort
are summarized in Table II.
Comparative analyses revealed significant intergroup differences in
T, N and TNM stages and serum total cholesterol (all P<0.05),
whereas no significant associations were observed for other
variables. The median Ki-67 index among the entire study cohort
(n=120) was 32% (interquartile range, 18–45%), while the median
values for the VM- and VM+ subgroups were 30% (20–50%) and 40%
(20–50%), respectively.
 | Table II.Comparison of the clinicopathological
features of patients with breast cancer according to VM status. |
Table II.
Comparison of the clinicopathological
features of patients with breast cancer according to VM status.
|
Characteristics | Reference
range | Total no. of
patients | VM− | VM+ | P-value |
|---|
| Patients | - | 120 (100) | 91 (75.8) | 29 (24.2) | - |
| Age, years | - | 120 | 49.0±9.32 | 52.76±8.52 | 0.056a |
| BMI,
kg/m2 | - | 120 | 23.27±3.40 | 23.82±2.95 | 0.436a |
| Menstrual
status |
|
|
|
| 0.113b |
|
Premenopausal | - | 69 | 56 (46.7) | 13 (10.8) |
|
|
Postmenopausal | - | 51 | 35 (29.2) | 16 (13.3) |
|
| Tumor location |
|
|
|
| 0.667c |
| Upper
outer quadrant | - | 80 | 63 (52.5) | 17 (14.2) |
|
| Lower
outer quadrant | - | 12 | 9 (7.5) | 3 (2.5) |
|
| Upper
inner quadrant | - | 22 | 15 (12.5) | 7 (5.8) |
|
| Lower
inner quadrant | - | 6 | 4 (3.3) | 2 (1.7) |
|
| T stage |
|
|
|
| 0.030c,d |
| T1 | - | 37 | 32 (26.7) | 5 (4.2) |
|
| T2 | - | 77 | 56 (46.6) | 21 (17.5) |
|
| T3 | - | 4 | 1 (0.8) | 3 (2.5) |
|
| T4 | - | 2 | 2 (1.7) | 0 (0.0) |
|
| N stage |
|
|
|
| 0.022b,d |
|
N0-N1 | - | 101 | 81 (67.5) | 20 (16.7) |
|
|
N2-N3 | - | 19 | 10 (8.3) | 9 (7.5) |
|
| TNM stage |
|
|
|
| 0.006c,d |
| Stage
I | - | 28 | 25 (20.8) | 3 (2.5) |
|
| Stage
II | - | 70 | 55 (45.8) | 15 (12.5) |
|
| Stage
III | - | 22 | 11 (9.2) | 11 (9.2) |
|
| Histological
grade |
|
|
|
| 0.055b |
|
G1-G2 | - | 60 | 50 (41.7) | 10 (8.3) |
|
| G3 | - | 60 | 41 (34.2) | 19 (15.8) |
|
| ER, % | - | 120 | 70 (0–90) | 0 (0–90) | 0.325e |
| PR, % | - | 120 | 10 (0–70) | 0 (0–60) | 0.194e |
| HER2
expression |
|
|
|
| 0.956b |
|
Negative | - | 75 | 57 (47.5) | 18 (15.0) |
|
|
Positive | - | 45 | 34 (28.3) | 11 (9.2) |
|
| Ki-67 index, % | - | 120 | 30 (20–50) | 40 (20–50) | 0.513e |
| Type of breast
surgery |
|
|
|
| 0.580b |
|
Breast-conserving surgery | - | 65 | 48 (40.0) | 17 (14.2) |
|
|
Mastectomy | - | 55 | 43 (35.8) | 12 (10.0) |
|
| Type of axillary
surgery |
|
|
|
| 0.107b |
|
Sentinel lymph node
biopsy | - | 57 | 47 (39.2) | 10 (8.3) |
|
|
Axillary lymph node
dissection | - | 63 | 44 (36.7) | 19 (15.8) |
|
| CA153, U/ml | ≤25.0 | 120 | 13.19
(8.63–18.91) | 15.91
(11.31–19.62) | 0.098e |
| CEA, U/ml | ≤5.0 | 120 | 1.50
(0.94–2.29) | 1.65
(0.98–2.74) | 0.684e |
| Total cholesterol,
mmol/l | 3.1–5.7 | 120 | 5.33
(4.50–5.89) | 5.99
(5.13–6.67) | 0.015e |
| Triglycerides,
mmol/l | 0.4–2.0 | 120 | 1.09
(0.78–1.61) | 1.24
(0.83–1.71) | 0.552e |
| Uric acid,
µmol/l | 155.0–357.0 | 120 | 290.6
(247.2–357.1) | 279.4
(219.0–325.9) | 0.247e |
| Alkaline
phosphatase, U/l | 31.0–115.0 | 120 | 69.9
(58.2–85.8) | 76.1
(61.9–99.7) | 0.125e |
| Lactate
dehydrogenase, U/l | 109.0–245.0 | 120 | 177.9
(166.7–206.9) | 179.8
(164.3–196.0) | 0.949e |
| Albumin-to-globulin
ratio | 1.5–2.5 | 120 | 1.642
(1.487–1.786) | 1.667
(1.525–1.766) | 0.907e |
Univariate Cox proportional hazards
regression analysis of DFS in patients with breast cancer
All patients were followed for a period ranging from
7 to 61 months, with a median follow-up of 43 months. During this
period, 10 patients experienced recurrence or metastasis, whereas
110 patients remained disease-free. DFS was defined as the
dependent variable, while baseline characteristics, peripheral
blood biomarkers, pathological and clinical features, as well as
the presence of VM, were included as independent variables in a
univariate Cox proportional hazards regression model.
The analysis revealed that DFS was significantly
associated with VM status [hazard ratio (HR)=7.813; 95% CI,
2.016–30.270; P=0.003], nodal stage (HR=5.775; 95% CI,
1.670–19.969; P=0.006), ER expression (HR=0.981; 95% CI,
0.963–0.999; P=0.043), Ki-67 index (HR=1.046; 95% CI, 1.018–1.075;
P=0.001) and histological grade (HR=10.482; 95% CI, 1.323–83.029;
P=0.026). Patients with a high Ki-67 index (≥30%) demonstrated a
significantly shorter DFS compared with those with a lower Ki-67
index (P<0.001; Table
III).
 | Table III.Univariate Cox proportional hazards
regression analysis of disease-free survival in patients with
breast cancer. |
Table III.
Univariate Cox proportional hazards
regression analysis of disease-free survival in patients with
breast cancer.
| Variable | No. of
patients | HR (95% CI) | P-value |
|---|
| VM |
|
|
|
|
Negative | 91 | Reference | - |
|
Positive | 29 | 7.813
(2.016–30.270) | 0.003 |
| Age, years | 120 | 1.008
(0.943–1.078) | 0.809 |
| BMI,
kg/m2 | 120 | 1.032
(0.858–1.242) | 0.738 |
| Menstrual
status |
|
|
|
|
Premenopausal | 69 | Reference | - |
|
Postmenopausal | 51 | 3.359
(0.868–13.002) | 0.079 |
| Tumor location |
|
|
|
|
Upper outer
quadrant | 80 | Reference | - |
|
Lower outer
quadrant | 12 | 5.137
(1.149–22.963) | 0.032 |
|
Upper inner
quadrant | 22 | 1.731
(0.316–9.473) | 0.527 |
|
Lower inner
quadrant | 6 | 3.708
(0.414–33.200) | 0.241 |
| T stage |
|
|
|
|
T1 | 37 | Reference | - |
|
T2 | 77 | 2.058
(0.436–9.718) | 0.362 |
|
T3 | 4 | 0.000
(0.000-Inf) | 0.998 |
|
T4 | 2 | 0.000
(0.000-Inf) | 0.999 |
| N stage |
|
|
|
|
N0-N1 | 101 | Reference | - |
|
N2-N3 | 19 | 5.775
(1.670–19.969) | 0.006 |
| TNM stage |
|
|
|
|
I | 28 | Reference | - |
|
II | 70 | 1.658
(0.185–14.845) | 0.651 |
|
III | 22 | 7.002
(0.817–60.010) | 0.076 |
| Histological
grade |
|
|
|
|
G1-G2 | 60 | Reference | - |
|
G3 | 60 | 10.482
(1.323–83.029) | 0.026 |
| ER | 120 | 0.981
(0.963–0.999) | 0.043 |
| PR | 120 | 0.978
(0.952–1.006) | 0.117 |
| HER2
expression |
|
|
|
|
Negative | 75 | Reference | - |
|
Positive | 45 | 0.716
(0.185–2.773) | 0.628 |
| Ki-67 index | 120 | 1.046
(1.018–1.075) | 0.001 |
| Surgical
approach |
|
|
|
|
Breast-conserving
surgery | 65 | Reference | - |
|
Mastectomy | 55 | 0.585
(0.164–2.091) | 0.410 |
| Axillary
surgery |
|
|
|
|
Sentinel lymph
node biopsy | 57 | Reference | - |
|
Axillary lymph
node dissection | 63 | 1.304
(0.367–4.629) | 0.681 |
| CA15-3, U/ml | 120 | 0.986
(0.908–1.070) | 0.731 |
| CEA, U/ml | 120 | 0.707
(0.377–1.326) | 0.279 |
| Total cholesterol,
mmol/l | 120 | 1.096
(0.640–1.878) | 0.737 |
| Triglycerides,
mmol/l | 120 | 0.716
(0.273–1.876) | 0.496 |
| Uric acid,
µmol/l | 120 | 0.998
(0.991–1.006) | 0.687 |
| Alkaline
phosphatase, U/l | 120 | 1.009
(0.982–1.036) | 0.515 |
| Lactate
dehydrogenase, U/l | 120 | 1.004
(0.992–1.016) | 0.529 |
| Albumin/globulin
ratio | 120 | 1.048
(0.122–8.971) | 0.966 |
In combination, these findings indicated that VM,
nodal stage, ER expression, Ki-67 index and histological grade are
independent determinants of DFS in breast cancer. Among these, ER
expression acted as a protective factor, whereas VM, advanced nodal
stage, elevated Ki-67 index and higher histological grade were
identified as adverse prognostic factors (Table III).
Multivariate Cox proportional hazards
regression analysis of DFS in patients with breast cancer
As presented in Table
IV, five variables with statistical significance in univariate
analysis were included in the multivariate Cox regression model.
The results demonstrated that VM (HR=9.824; 95% CI=2.079–46.413;
P=0.004), N stage (HR=4.358; 95% CI=1.136–16.722; P=0.032) and
Ki-67 index (HR=1.059; 95% CI=1.015–1.105; P=0.008) were identified
as independent risk factors for DFS. By contrast, ER (P=0.479) and
histological grade (P=0.325) were not statistically
significant.
 | Table IV.Multivariate Cox proportional hazards
regression analysis of disease-free survival in patients with
breast cancer (n=120). |
Table IV.
Multivariate Cox proportional hazards
regression analysis of disease-free survival in patients with
breast cancer (n=120).
| Variables | HR (95% CI) | P-value |
|---|
| VM (positive vs.
negative) | 9.824
(2.079–46.413) | 0.004 |
| N stage (N2-N3 vs.
N0-N1) | 4.358
(1.136–16.722) | 0.032 |
| ER | 1.008
(0.985–1.032) | 0.479 |
| Ki-67 index | 1.059
(1.015–1.105) | 0.008 |
| Histological grade
(G3 vs. G1-G2) | 3.270
(0.309–34.577) | 0.325 |
Construction and evaluation of the
predictive model
Nomogram for the prognostic model
Based on the results of the multivariate Cox
proportional hazards regression analysis, three independent
prognostic risk factors, VM, N stage and the Ki-67 index, were
ultimately incorporated into the predictive model.
In the present model, continuous variables were
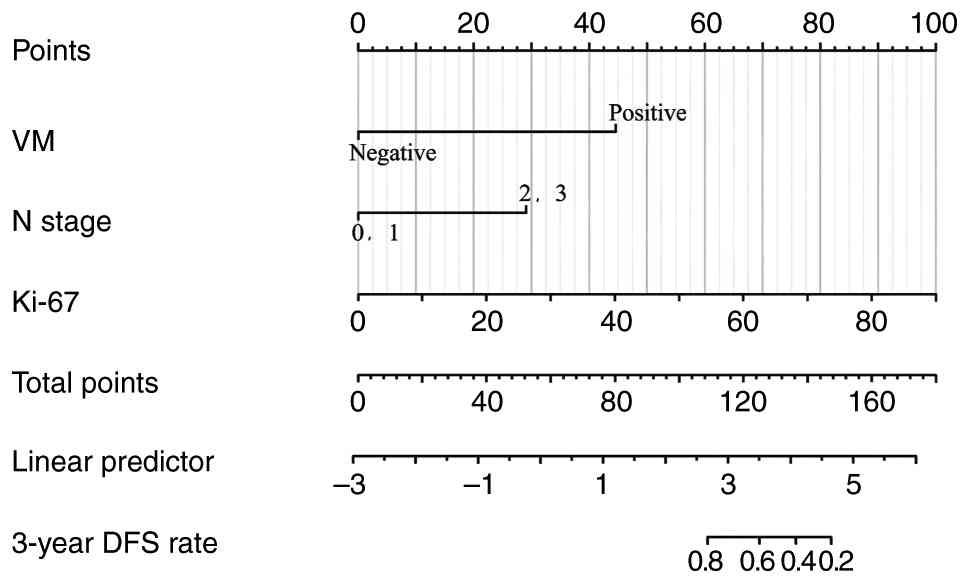
entered at their original values, while categorical variables were
coded as follows: VM, negative=0 and positive=1; N stage, N0-N1=1
and N2-N3=2.
Subsequently, a prognostic nomogram was constructed
to estimate the 3-year DFS of patients with breast cancer. Each
covariate was assigned a point value by projecting upward to the
corresponding scale. The individual scores of all variables were
then summed to generate a total score, which was further mapped
onto the probability scale at the bottom of the nomogram to yield
the predicted 3-year DFS. A higher total score corresponded to a
worse prognosis.
An investigation of the nomogram revealed that the
Ki-67 index contributed the greatest weight to the model, followed
by VM and N stage providing the smallest contribution (Fig. 2).
Evaluation of the predictive
model
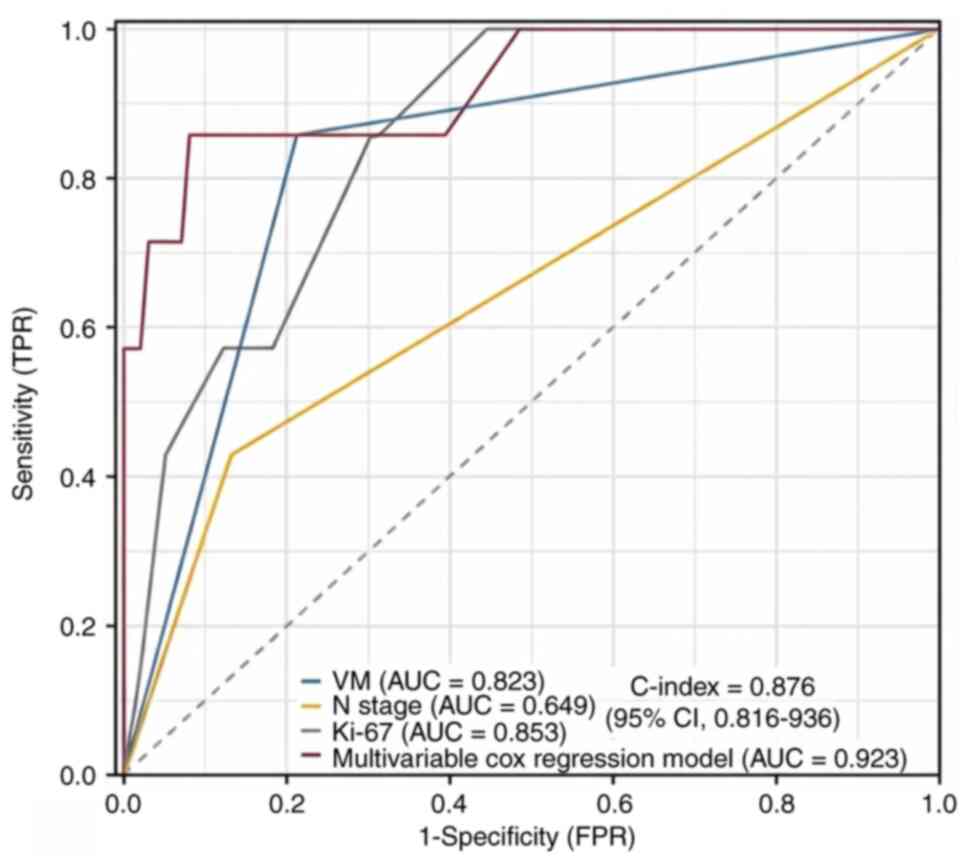
The predictive performance of the model was first
assessed using the C-index computed in R software, yielding a value
of 0.876 (95% CI, 0.816–0.936), indicative of notably enhanced
discriminative ability. Subsequently, time-dependent ROC curve
analysis was performed to compare the area under the curve (AUC)
for individual risk factors and for the multivariate Cox
proportional hazards model in predicting 3-year DFS. The results
demonstrated that the AUCs were 0.823 for VM, 0.649 for N stage,
0.853 for the Ki-67 index and 0.923 for the multivariate Cox
regression model. As demonstrated in Fig. 3, the predictive accuracy of the
multivariate model markedly outperformed that of any single factor,
with a sensitivity of 85.7% and a specificity of 91.9% at the
optimal cut-off value.
To further evaluate the prognostic reliability of
the nomogram, calibration analysis was conducted using the ‘rms’
package in R software. Internal validation was performed via
bootstrap resampling, with a sample size of 40 per iteration and
800 repetitions. As presented in Fig.
4, the calibration curve demonstrated close agreement between
predicted and observed 3-year DFS probabilities, confirming the
robust calibration and stability of the model.
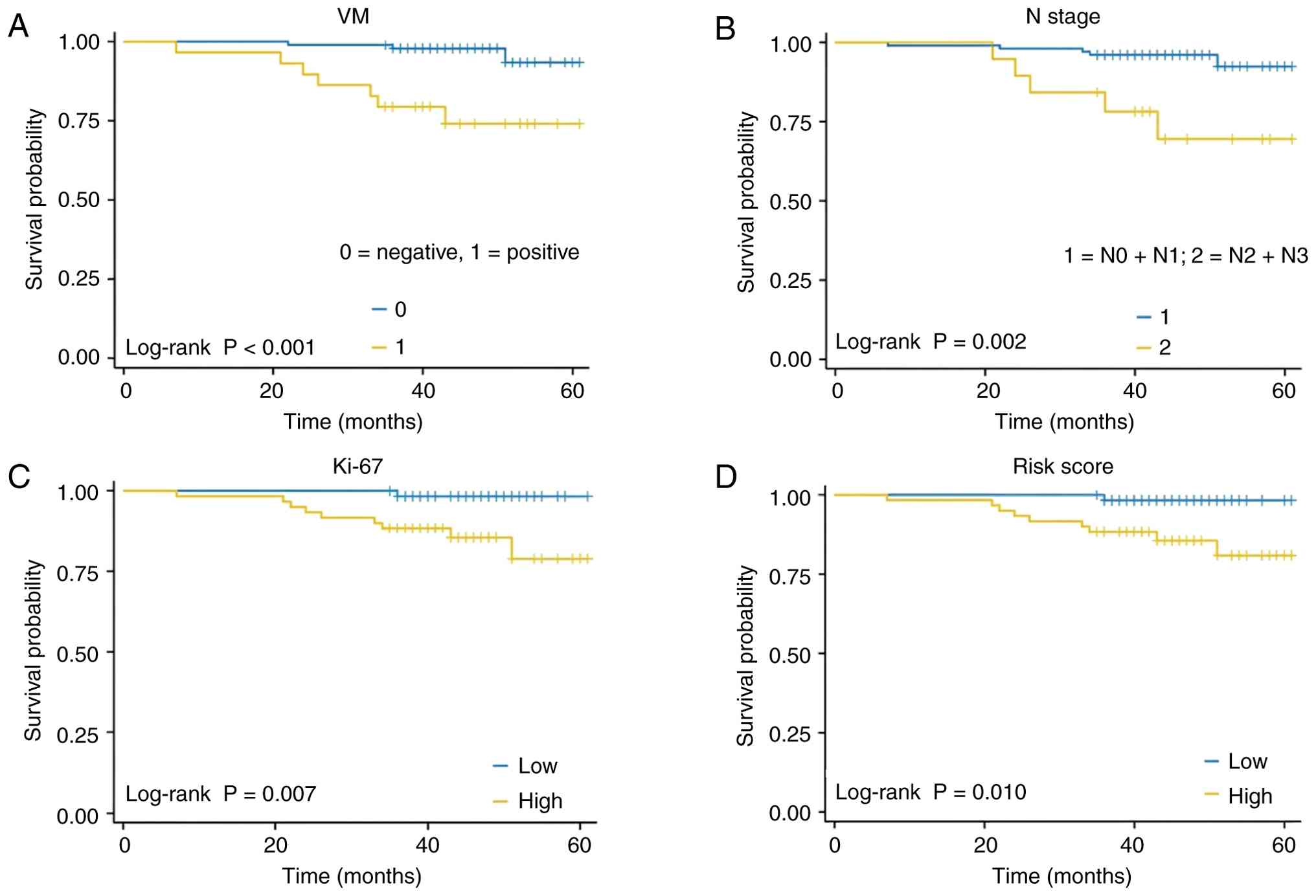
Survival analysis
Survival analysis was performed for each independent
prognostic factor, as well as for the integrated predictive model.
Kaplan-Meier survival curves demonstrated that patients with
VM+ tumors exhibited significantly lower DFS compared
with those patients with VM− tumors (log-rank test;
P<0.001). Similarly, patients with advanced N stage exhibited a
significantly reduced DFS compared with those patients in the lower
N stage group (log-rank test; P=0.002). A higher Ki-67 index (above
the median) was also associated with a significantly worse DFS
compared with a lower Ki-67 index (log-rank test; P=0.007).
Furthermore, patients stratified into the high-risk group based on
the risk score derived from the multivariate Cox regression model
had a significantly worse DFS compared with those patients in the
low-risk group (log-rank test; P=0.010). Although nodal stage and
Ki-67 index were individually associated with DFS, the composite
risk score demonstrated an improved discriminatory ability in
stratifying patients. For example, patients with high risk scores
exhibited a 3-year DFS of 58%, whereas those in the high N stage
group alone had a 3-year DFS of 67% and those with high Ki-67 alone
had a 3-year DFS of 62% (Fig. 5).
These results suggested that the integrated risk score captures
combined prognostic information beyond individual factors
alone.
Collectively, these findings underscored that VM, N
stage and the Ki-67 index represent independent prognostic
determinants of postoperative DFS in breast cancer and that the
risk score derived from the multivariate Cox proportional hazards
model provides a robust tool in identifying patients at elevated
risk of recurrence.
Discussion
Over recent decades, multiple studies have
elucidated diverse risk factors associated with the prognosis of
breast cancer (1,25). With the advent of increasingly
sophisticated therapeutic strategies, the survival outcomes of
patients have markedly improved (1,26).
Nevertheless, disease recurrence and distant metastasis remain
formidable clinical challenges, continuing to pose notable burdens
for both patients and clinicians. While previous studies have
examined the association between VM and poor prognosis in breast
cancer (10,27,28),
to the best of our knowledge, the present study was among the first
to construct and internally validate a composite prognostic model
that integrates VM status with established clinical parameters,
including nodal stage and Ki-67 index. The development of a
quantitative risk score enables more personalized risk
stratification compared with analysis based solely on individual
factors, offering a practical tool that may potentially guide
postoperative decision-making in the future.
VM represents an alternative mechanism of tumor
vascularization, in which tumor cells themselves form vascular-like
channels independent of endothelial cells, thereby facilitating
nutrient supply analogous to conventional vasculature. VM has been
demonstrated to serve a key role in tumor growth, invasion and
metastasis. Despite the intricate molecular pathways and signaling
networks involved in its formation, both basic and clinical studies
have consistently associated VM with adverse prognosis in patients
with breast cancer (5,9,10,17).
Of note, therapeutic interventions, particularly anti-angiogenic
strategies, can inadvertently promote VM. By exacerbating hypoxic
conditions within the tumor microenvironment, such treatments may
stimulate VM formation and contribute to therapeutic resistance
(29). Therefore, VM not only
augments tumor aggressiveness but also undermines treatment
efficacy, underscoring its potential value as a prognostic
biomarker and a candidate variable in risk-prediction models for
breast cancer.
In the present study, the presence of VM in invasive
breast carcinoma was observed. A retrospective analysis of 120
patients with breast cancer was conducted in the present study and
demonstrated that VM formation was reported to be associated with
tumor stage, nodal involvement and overall disease stage.
Specifically, the prevalence of VM positivity increased
progressively with higher T and N classifications, as well as
advancing clinical stage, corroborating its association with more
aggressive disease phenotypes. Of note, no VM positivity was
detected in T4 cases, likely attributable to limited sample size
and the clinical tendency to prioritize neoadjuvant therapy over
surgery in this subset. Furthermore, a significant association was
identified between VM positivity and elevated serum cholesterol
levels, a relationship that remains underexplored in the current
literature (30–33). Since the phosphoinositide
3-kinase/protein kinase B (PI3K/AKT) signaling axis has been
implicated in VM formation through the regulation of matrix
metalloproteinase-9 and epithelial-mesenchymal transition (32), and that hypercholesterolemia has
been reported to accelerate breast cancer progression through
PI3K/AKT activation in murine models (33), it is plausible that elevated
cholesterol may indirectly promote VM. This finding warrants
further investigation with larger cohorts to substantiate the
mechanistic association between dyslipidemia and VM formation in
the future.
Hypercholesterolemia is often encountered in
patients undergoing endocrine therapy and has been implicated in
tumor progression through the modulation of cell membrane dynamics
and signaling pathways (32,34,35).
Whether dyslipidemia actively contributes to VM formation remains
to be elucidated; however, these findings suggested that metabolic
factors may be considered when interpreting postoperative tumor
behavior. Routine assessment of lipid profiles may help identify
patients at risk for aggressive features such as VM, warranting
more comprehensive follow-up.
Consistent with previous studies (5,36),
VM+ patients in the present study cohort exhibited a
trend toward higher histological grade and reduced ER/PR
expression, although these associations did not reach statistical
significance, likely due to a limited sample size. Collectively,
these findings strengthened the evidence that VM is a surrogate
marker of tumor aggressiveness and an adverse prognostic
determinant in breast cancer.
Univariate Cox regression analysis incorporating
clinical, pathological and laboratory variables identified VM,
nodal stage, Ki-67 index, ER status and histological grade as
significant prognostic factors. Among these, VM, N stage and Ki-67
were confirmed as independent risk factors for reduced DFS in
multivariate Cox regression models, whereas ER expression emerged
as a protective factor. This finding aligns with existing
literature highlighting these variables as prognostically relevant
(37,38). Of note, tumor size (represented by T
stage) and age, well-established predictors in meta-analyses of
breast cancer prognostic models (39), did not reach statistical
significance in the present study, likely reflecting the relatively
small and demographically homogeneous cohort, as well as the
biological heterogeneity of molecular subtypes that may confound
conventional clinicopathological indicators.
Based on the multivariate Cox regression analysis, a
prognostic model incorporating VM, N stage and Ki-67 index was
constructed. This model demonstrated notably enhanced predictive
performance, with a C-index of 0.876 and an AUC of 0.923,
outperforming any individual factor. Calibration analysis confirmed
a close agreement between predicted and observed outcomes, further
supporting the robustness of the model. Kaplan-Meier analysis
revealed a significantly worse DFS among VM+ patients,
patients with higher N stage and patients with elevated Ki-67
indices, in line with previous studies (40–42).
Of note, the risk score derived from the present study model
effectively stratified patients into high- and low-risk groups with
distinct survival outcomes, thereby underscoring its clinical
applicability.
Despite the established prognostic value of nodal
metastasis and elevated Ki-67 index, the integrated risk score
developed in the present study provided additional stratification
by combining microanatomical features of tumor biology (namely, VM)
with conventional clinical variables. By accounting for multiple
determinants, this composite risk score may further identify
patients at the highest risk of recurrence, potentially informing
intensified surveillance or therapeutic strategies in the
future.
It is key to recognize that the present study model
currently lacks external validation and may be subject to
overfitting due to internal training on a single cohort.
Prospective validation in independent datasets is warranted to
confirm predictive performance and generalizability.
In clinical practice, a high-risk classification may
prompt consideration of more aggressive adjuvant therapy. While
standardized guidelines for additional chemotherapy based on VM
status are not yet established, patients with high composite risk
scores could be candidates for multidisciplinary review, closer
follow-up or enrollment in clinical trials evaluating adjuvant
systemic treatments tailored by molecular and microarchitectural
risk factors.
In conclusion, VM is significantly associated with
adverse clinicopathological characteristics in breast cancer,
including higher T and N stage, advanced tumor stage and elevated
serum cholesterol levels. VM constitutes an independent adverse
prognostic factor for breast cancer. N stage and Ki-67 index are
independent predictors of recurrence and survival following
surgery. A prognostic model integrating VM, N stage and Ki-67 index
demonstrated high predictive accuracy and effectively stratified
patients according to DFS risk.
Acknowledgements
Not applicable.
Funding
The present study was supported by the Jingyi Medical Research
Foundation (grant no. JV112024-020122201).
Availability of data and materials
The data generated in the present study may be
requested from the corresponding author.
Authors' contributions
ZW and YT contributed to the collection of clinical
data, statistical analysis and drafting of the manuscript. SH and
SS performed pathological evaluations and immunohistochemical
analyses. PQ and LS assisted with database management and clinical
interpretation. ML contributed to the analysis and interpretation
of data, and critically revised the manuscript. YZ conceived and
supervised the study and finalized the manuscript. All authors read
and approved the final manuscript. ZW and YZ confirm the
authenticity of all the raw data.
Ethics approval and consent to
participate
The present study protocol was reviewed and approved
by the Ethics Committee of the Affiliated Hospital of Guangdong
Medical University (approval no. PJKT2025-067; Zhanjiang, China).
Written informed consent was obtained from all participants prior
to inclusion in the present study.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Siegel RL, Giaquinto AN and Jemal A:
Cancer statistics, 2024. CA Cancer J Clin. 74:12–49.
2024.PubMed/NCBI
|
|
2
|
Han B, Zheng R, Zeng H, Wang S, Sun K,
Chen R, Li L, Wei W and He J: Cancer incidence and mortality in
China, 2022. J Natl Cancer Cent. 4:47–53. 2024.PubMed/NCBI
|
|
3
|
Turner KM, Yeo SK, Holm TM, Shaughnessy E
and Zhang X: Heterogeneity within molecular subtypes of breast
cancer. Am J Physiol Cell Physiol. 321:C343–C354. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Maniotis AJ, Folberg R, Hess A, Seftor EA,
Gardner LM, Pe'er J, Trent JM, Meltzer PS and Hendrix MJ: Vascular
channel formation by human melanoma cells in vivo and in vitro:
Vasculogenic mimicry. Am J Pathol. 155:739–752. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Andonegui-Elguera MA, Alfaro-Mora Y,
Cáceres-Gutiérrez R, Caro-Sánchez CHS, Herrera LA and Díaz-Chávez
J: An overview of vasculogenic mimicry in breast cancer. Front
Oncol. 10:2202020. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Lin X, Long S, Yan C, Zou X, Zhang G, Zou
J and Wu G: Therapeutic potential of vasculogenic mimicry in
urological tumors. Front Oncol. 13:12026562023. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Tian X, Si Q, Liu M, Shi J, Zhao R, Xiong
Y, Yu L, Cui H and Guan H: Advance in vasculogenic mimicry in
ovarian cancer (review). Oncol Lett. 26:4562023. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Zheng N, Zhang S, Wu W, Zhang N and Wang
J: Regulatory mechanisms and therapeutic targeting of vasculogenic
mimicry in hepatocellular carcinoma. Pharmacol Res. 166:1055072021.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Andreucci E, Peppicelli S, Ruzzolini J,
Bianchini F and Calorini L: Physicochemical aspects of the tumour
microenvironment as drivers of vasculogenic mimicry. Cancer
Metastasis Rev. 41:935–951. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Xing P, Dong H, Liu Q, Zhao T, Yao F, Xu
Y, Chen B, Zheng X, Wu Y, Jin F and Li J: ALDH1 expression and
vasculogenic mimicry are positively associated with poor prognosis
in patients with breast cancer. Cell Physiol Biochem. 49:961–970.
2018. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Morales-Guadarrama G, García-Becerra R,
Méndez-Pérez EA, García-Quiroz J, Avila E and Díaz L: Vasculogenic
mimicry in breast cancer: Clinical relevance and drivers. Cells.
10:17582021. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Pezzella F and Ribatti D: Vascular
co-option and vasculogenic mimicry mediate resistance to
antiangiogenic strategies. Cancer Rep (Hoboken).
5:e13182022.PubMed/NCBI
|
|
13
|
Shen R, Wu T, Huang P, Shao Q and Chen M:
The clinicopathological significance of ubiquitin-conjugating
enzyme E2C, leucine-rich repeated-containing G protein-coupled
receptor, WW domain-containing oxidoreductase, and vasculogenic
mimicry in invasive breast carcinoma. Medicine (Baltimore).
98:e152322019. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Sun H, Yao N, Cheng S, Li L, Liu S, Yang
Z, Shang G, Zhang D and Yao Z: Cancer stem-like cells directly
participate in vasculogenic mimicry channels in triple-negative
breast cancer. Cancer Biol Med. 16:299–311. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Liu T, Sun B, Zhao X, Gu Q, Dong X, Yao Z,
Zhao N, Chi J, Liu N, Sun R and Ma Y: HER2/neu expression
correlates with vasculogenic mimicry in invasive breast carcinoma.
J Cell Mol Med. 17:116–122. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Liu TJ, Sun BC, Zhao XL, Zhao XM, Sun T,
Gu Q, Yao Z, Dong XY, Zhao N and Liu N: CD133+ cells with cancer
stem cell characteristics associates with vasculogenic mimicry in
triple-negative breast cancer. Oncogene. 32:544–553. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Shirakawa K, Wakasugi H, Heike Y, Watanabe
I, Yamada S, Saito K and Konishi F: Vasculogenic mimicry and
pseudo-comedo formation in breast cancer. Int J Cancer. 99:821–828.
2002. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
D'Andrea MR, Cereda V, Coppola L, Giordano
G, Remo A and De Santis E: Propensity for early metastatic spread
in breast cancer: Role of tumor vascularization features and tumor
immune infiltrate. Cancers (Basel). 13:59172021. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Xu Y, Li Q, Li XY, Yang QY, Xu WW and Liu
GL: Short-term anti-vascular endothelial growth factor treatment
elicits vasculogenic mimicry formation of tumors to accelerate
metastasis. J Exp Clin Cancer Res. 31:162012. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Perez EA, Romond EH, Suman VJ, Jeong JH,
Sledge G, Geyer CE Jr, Martino S, Rastogi P, Gralow J, Swain SM, et
al: Trastuzumab plus adjuvant chemotherapy for human epidermal
growth factor receptor 2-positive breast cancer: Planned joint
analysis of overall survival from NSABP B-31 and NCCTG N9831. J
Clin Oncol. 32:3744–3752. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Slamon DJ, Leyland-Jones B, Shak S, Fuchs
H, Paton V, Bajamonde A, Fleming T, Eiermann W, Wolter J, Pegram M,
et al: Use of chemotherapy plus a monoclonal antibody against HER2
for metastatic breast cancer that overexpresses HER2. N Engl J Med.
344:783–792. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Hori A, Shimoda M, Naoi Y, Kagara N, Tanei
T, Miyake T, Shimazu K, Kim SJ and Noguchi S: Vasculogenic mimicry
is associated with trastuzumab resistance of HER2-positive breast
cancer. Breast Cancer Res. 21:882019. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Amin MB, Edge SB, Greene FL, Byrd DR,
Brookland RK, Washington MK, Gershenwald JE, Compton CC, Hess KR,
Sullivan DC, et al: AJCC cancer staging manual. 8th edition.
Springer; New York, NY: pp. 589–628. 2017, PubMed/NCBI
|
|
24
|
Elston CW and Ellis IO: Pathological
prognostic factors in breast cancer. I. The value of histological
grade in breast cancer: Experience from a large study with
long-term follow-up. Histopathology. 19:403–410. 1991. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Bray F, Laversanne M, Sung H, Ferlay J,
Siegel RL, Soerjomataram I and Jemal A: Global cancer statistics
2022: GLOBOCAN estimates of incidence and mortality worldwide for
36 cancers in 185 countries. CA Cancer J Clin. 74:229–263.
2024.PubMed/NCBI
|
|
26
|
Wagle NS, Nogueira L, Devasia TP, Mariotto
AB, Yabroff KR, Islami F, Jemal A, Alteri R, Ganz PA and Siegel RL:
Cancer treatment and survivorship statistics, 2025. CA Cancer J
Clin. 75:308–340. 2025.PubMed/NCBI
|
|
27
|
Wei Y, Jiao Z, Sun T, Lai Z and Wang X:
Molecular mechanisms behind vascular mimicry as the target for
improved breast cancer management. Int J Womens Health.
15:1027–1038. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Zheng S, Guo G, Yang Z, Lu Y, Lu K, Fu W
and Huang Q: Vasculogenic mimicry regulates immune infiltration and
mutational status of the tumor microenvironment in breast cancer to
influence tumor prognosis. Environ Toxicol. 39:2948–2960. 2024.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Sun H, Zhang D, Yao Z, Lin X, Liu J, Gu Q,
Dong X, Liu F, Wang Y, Yao N, et al: Anti-angiogenic treatment
promotes triple-negative breast cancer invasion via vasculogenic
mimicry. Cancer Biol Ther. 18:205–213. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Zipinotti Dos Santos D, de Souza JC,
Pimenta TM, da Silva Martins B, Junior RSR, Butzene SMS, Tessarolo
NG, Cilas PML Jr, Silva IV and Rangel LBA: The impact of lipid
metabolism on breast cancer: A review about its role in
tumorigenesis and immune escape. Cell Commun Signal. 21:1612023.
View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Gao W, Yao Y, Gao Q, Zhao T and Li H:
Impact of serum lipids on prognosis in breast cancer patients: A
systematic review and meta-analysis. World J Surg Oncol.
23:2342025. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Zhang X, Zhang J, Zhou H, Fan G and Li Q:
Molecular mechanisms and anticancer therapeutic strategies in
vasculogenic mimicry. J Cancer. 10:6327–6340. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Alikhani N, Ferguson RD, Novosyadlyy R,
Gallagher EJ, Scheinman EJ, Yakar S and LeRoith D: Mammary tumor
growth and pulmonary metastasis are enhanced in a hyperlipidemic
mouse model. Oncogene. 32:961–967. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Li Y, Deng Z, Wang Y and Shen S: Lipid
changes during endocrine therapy in early-stage breast cancer
patients: A real-world study. Lipids Health Dis. 23:92024.
View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Rillamas-Sun E, Kwan ML, Iribarren C,
Cheng R, Neugebauer R, Rana JS, Nguyen-Huynh M, Shi Z, Laurent CA,
Lee VS, et al: Development of cardiometabolic risk factors
following endocrine therapy in women with breast cancer. Breast
Cancer Res Treat. 201:117–126. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Shen Y, Quan J, Wang M, Li S and Yang J,
Lv M, Chen Z, Zhang L, Zhao X and Yang J: Tumor vasculogenic
mimicry formation as an unfavorable prognostic indicator in
patients with breast cancer. Oncotarget. 8:56408–56416. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Zhu J, Cheng J, Ma Y, Wang Y, Zou Z, Wang
W, Shi H and Meng Y: The value of inflammation-related indicators
in chemotherapy efficacy and disease-free survival of
triple-negative breast cancer. Eur J Med Res. 30:772025. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Huang X, Luo Z, Liang W, Xie G, Lang X,
Gou J, Liu C, Xu X and Fu D: Survival nomogram for young breast
cancer patients based on the SEER database and an external
validation cohort. Ann Surg Oncol. 29:5772–5781. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Phung MT, Tin Tin S and Elwood JM:
Prognostic models for breast cancer: A systematic review. BMC
Cancer. 19:2302019. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Mitra D, Bhattacharyya S, Alam N, Sen S,
Mitra S, Mandal S, Vignesh S, Majumder B and Murmu N:
Phosphorylation of EphA2 receptor and vasculogenic mimicry is an
indicator of poor prognosis in invasive carcinoma of the breast.
Breast Cancer Res Treat. 179:359–370. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Zhang Q, Peng S, Wang XM, Wang XX, Zhang
HH and Liao LQ: Analysis of clinicopathologic characteristics and
prognostic riskfactors in T1 stage breast cancer. Chin J Gen Surg.
32:761–770. 2023.(In Chinese).
|
|
42
|
Zhang ZW, Luo YB, Yang SH and Jiang BB:
Association of Ki-67 expression with prognosis in patients with
luminal B breast cancer. Matern Child Health Care China.
39:3140–3144. 2024.(In Chinese).
|