Introduction
Gastric cancer remains a formidable global health
challenge, ranking as the fifth most common malignancy and the
third leading cause of cancer-related death worldwide (1). Despite reported improvements in 5-year
relative survival trends for stage II, III and IV (up to 85, 70 and
29%, respectively), overall results remain poor (2). Actual 5-year relapse-free survival
rate has been reported as 67.4% for stage IIIA, 55.7% for stage
IIIB and 29.9% for stage IIIC, respectively (3). Gastric cancer is a complex disease,
characterized by diverse histological subtypes and varying
responses to treatment, necessitating a multidisciplinary
management approach (4). The
incidence and prognosis vary significantly across different
regions, with a particularly high burden in some countries, such as
the Ukraine or South Korea. Furthermore, gastric cancer is most
often diagnosed at advanced stages in the Ukraine, with a
predominance of locally advanced and metastatic disease (5). Malignant neoplasms with a poor
prognosis, such as gastric, liver or pancreatic cancer, are
continuously under investigation for targets that may be used to
improve outcomes (6,7).
Current trends in gastric cancer treatment are
targeting neoadjuvant chemotherapy and immunotherapy (8–10). The
optimal treatment pathway for resectable, non-metastatic tumors
consists of a combination of perioperative chemotherapy and
surgery. The rationale for neoadjuvant treatment was first
demonstrated in the MAGIC trial, particularly for stage II and III
gastric cancer. The trial showed an improvement in overall survival
(OS) rate with three preoperative and three postoperative cycles of
epirubicin, cisplatin and infusional 5-fluorouracil (ECF) compared
with surgery alone (36 vs. 23%, respectively) (11). Similarly, the ACCORD-07 trial
demonstrated that chemotherapy combined with surgery resulted in
superior OS rate compared with surgery alone (38 vs. 24%,
respectively) (12).
Neoadjuvant chemotherapy targets the tumor in its
native environment, potentially increasing the likelihood of
complete resection, reducing the risk of recurrence and ultimately
improving survival outcomes. The FLOT4-AIO clinical trial evaluated
a regimen comprising 5-fluorouracil, leucovorin, oxaliplatin and
docetaxel (FLOT), establishing preoperative chemotherapy as a
standard of care based on superior survival outcomes compared with
ECF/epirubicin, cisplatin and capecitabine (3-year OS rate: 57 vs.
48%, respectively) (13). However,
postoperative chemotherapy is associated with a marked degree of
intolerance. In the MAGIC study (11), only 41.6% of patients completed
postoperative treatment. A similar trend was observed in the FLOT4
trial (13), where only 46% of
patients completed all prescribed treatment per protocol due to
postoperative complications and therapy-related intolerance
following curative surgery. An alternative approach is total
neoadjuvant chemotherapy (TNT), which involves the complete
administration of chemotherapy prior to surgical resection. This
strategy has gained increased attention due to the limited
tolerability of adjuvant therapy in rectal cancer (14).
TNT aims to maximize the systemic effect of
chemotherapy by ensuring that the full treatment course is
delivered before potential postoperative complications can delay or
prevent further therapy. In addition, it eliminates issues related
to postoperative intolerance. This approach became more widely
applied during the COVID-19 pandemic, when interruptions in
treatment after several cycles prompted the resumption of
chemotherapy before surgery.
However, there is still an insufficient number of
studies evaluating the safety and efficacy of the FLOT regimen in a
total neoadjuvant setting for patients with advanced gastric and
gastroesophageal junction (GEJ) cancer. Yang et al (15) reported that TNT with FLOT
demonstrated an acceptable toxicity profile. While there was no
significant difference in the proportion of patients who completed
all planned chemotherapy cycles, TNT patients received a higher
proportion of cycles that included all chemotherapy agents compared
with the standard perioperative approach (93 vs. 74%). In a study
conducted by Shi et al (16), 71.4% of patients who received
neoadjuvant chemoradiation followed by consolidational neoadjuvant
chemotherapy with the S-1 + oxaliplatin regimen proceeded to
surgery, and all achieved an R0 resection. The incidence of major
pathological responses by Mandard scores 1 and 2 (17) was 95.0 and 50.0%, respectively.
Furthermore, it has been reported that TNT may contribute to higher
pathological response rates regardless of the chemotherapy regimen
used (18).
The present retrospective study preliminary
investigated the safety and feasibility of TNT using the FLOT
regimen in the treatment of resectable gastric and GEJ cancer.
Materials and methods
Inclusion criteria
The present retrospective study was conducted to
preliminarily evaluate the safety and efficacy of TNT with the FLOT
regimen in patients with gastric and GEJ cancer. A total of 76
eligible participants were enrolled at the National Cancer
Institute (Kyiv, Ukraine) between October 2017 and October 2021.
The study included 44 male and 27 female patients with locally
advanced gastric cancer (T3-T4b, N+, according to American Joint
Committee on Cancer Staging Classification, 7th edition) (19) who were technically and medically
operable at the time of diagnosis and had no major comorbidities
that might have precluded receiving TNT with FLOT. The median age
was 56 years [interquartile range (IQR), 48.0–63.5 years]. All
patients received TNT with FLOT following multidisciplinary tumor
board (MDT) evaluation. Each patient underwent standardized initial
staging laparoscopy, including biopsies of the peritoneal and
visceral peritoneum at standard anatomical points, as well as
peritoneal lavage. Detailed patient characteristics are displayed
in Table I.
 | Table I.Patients' characteristics (n=76). |
Table I.
Patients' characteristics (n=76).
| Characteristics | Value |
|---|
| Median age (IQR),
years | 56 (48.0–63.5) |
| ECOG, n (%) |
|
| 0 | 59 (77.63) |
| 1 | 17 (22.37) |
| Sex, n (%) |
|
| Male | 47 (61.84) |
|
Female | 29 (38.16) |
| Clinical T stage, n
(%) |
|
| 1b | 0 (0.00) |
| 2 | 0 (0.00) |
| 3 | 19 (25.00) |
| 4а | 39 (51.32) |
| 4b | 18 (23.68) |
| Clinical N stage, n
(%) |
|
| N0 | 1 (1.32) |
| N1 | 25 (32.89) |
| N2 | 32 (42.11) |
| N3 | 18 (23.68) |
| Median number of
lymph nodes according to CT data (IQR) | 4 (2–6.5) |
| Tumor location, n
(%) |
|
| Siewert
II | 0 (0.00) |
| Siewert
III | 4 (5.26) |
|
Cardia | 14 (18.42) |
|
Corpus | 37 (48.68) |
|
Antrum | 7 (9.21) |
| Linitis
plastica | 14 (18.42) |
| Histology type |
|
|
Adenocarcinoma | 54 (71.05) |
| Poorly
cohesive carcinoma | 21 (27.63) |
|
Adenosquamous carcinoma | 1 (1.32) |
| Malignancy grade, n
(%) |
|
| G1 | 5 (6.58) |
| G2 | 11 (14.47) |
| G3 | 38 (50.00) |
| Gx | 22 (28.95) |
| History of previous
chemotherapy, n (%) |
|
|
Yes | 4 (5.26) |
| No | 72 (94.74) |
The inclusion criteria were as follows: i)
Histologically confirmed, locally advanced gastric or GEJ Siewert
III adenocarcinoma characterized by a linitis plastica growth
pattern or the presence of bulky lymphadenopathy without distant
metastases; ii) Eastern Cooperative Oncology Group (ECOG)
performance status of 0 or 1 (20);
iii) age between 18 and 80 years; iv) no prior anticancer therapy
within the past 5 years; and v) absence of severe comorbidities.
The primary endpoint was the safety and feasibility of TNT with
FLOT, assessed by the incidence and severity of
chemotherapy-related adverse events, rates of dose reduction or
treatment discontinuation, and completion of the planned
neoadjuvant regimen. Secondary endpoints included pathological
tumor regression, R0 resection rate, postoperative morbidity,
postoperative mortality (30- and 90-day) and detailed surgical
outcomes, including type of surgery and postoperative complication
profile.
TNT
Eligibility was verified at inclusion prior to
treatment initiation. The chemotherapy regimen consisted of eight
cycles of intravenous docetaxel (50 mg/m2), oxaliplatin
(85 mg/m2), leucovorin (200 mg/m2) and
5-fluorouracil (2,600 mg/m2 as a 24-h continuous
infusion). Treatment was administered via a port system on day 1 of
each 14-day cycle. Before each cycle, patients underwent laboratory
evaluation, including complete blood count and serum biochemistry.
Chemotherapy was delivered on an outpatient basis. Supportive
measures, including prophylactic or therapeutic granulocyte
colony-stimulating factor, antiemetics, dose modifications and
treatment delays, were applied in accordance with the Health
Service Executive National Cancer Control Program (21). Adverse events were monitored and
documented at each chemotherapy cycle, using the Common Terminology
Criteria for Adverse Events, version 5.0 (22). Radiological response assessments
were conducted by a dedicated radiology team following completion
of four chemotherapy cycles and again after completion of TNT,
using contrast-enhanced thoraco-abdomino-pelvic CT scans evaluated
according to Response Evaluation Criteria in Solid Tumors (RECIST)
version 1.1 (23). Quality of life
was assessed using the EORTC QLQ-C30 questionnaire and the gastric
cancer-specific module QLQ-STO22 (24,25)
(Table II).
 | Table II.Timeline of assessments and
interventions during total neoadjuvant chemotherapy. |
Table II.
Timeline of assessments and
interventions during total neoadjuvant chemotherapy.
|
Assessment/intervention | Inclusion | Cycle 1 | Cycle 2 | Cycle 3 | Cycle 4 | Cycle 5 | Cycle 6 | Cycle 7 | Cycle 8 | Surgery |
|---|
| Eligibility
criteria check | x |
|
|
|
|
|
|
|
|
|
| ECOG performance
status | xa | x | x | x | x | x | x | x | x | xa |
| Laboratory
tests | xa | x | x | x | x | x | x | x | x | xa |
| (CBC,
biochemistry) |
|
|
|
|
|
|
|
|
|
|
| QLQ-C30,
STO-22 | xa |
|
|
| xb |
|
|
| xb | xc |
| Tumor assessment
(CT scan) |
| xd |
|
|
|
|
|
| xd |
|
| Pathological tumor
regression |
|
|
|
|
|
|
|
|
|
|
| (Becker) |
|
|
|
|
|
|
|
|
| x |
| Postoperative
complications |
|
|
|
|
|
|
|
|
|
|
| assessment |
|
|
|
|
|
|
|
|
| xe |
Curative surgery
Patients considered at high risk for peritoneal
dissemination underwent second-look staging laparoscopy following
completion of TNT. Those without distant metastases or with locally
progressive disease that remained technically and medically
resectable proceeded to curative surgery. All major surgical
procedures were performed by a single high-volume surgical team
with expertise in gastric cancer. The surgical approach was based
on tumor location, preoperative imaging, and intraoperative
findings. Postoperative complications were classified according to
the Clavien-Dindo scale (26).
Pathological tumor regression was assessed using the Becker
classification (27).
Follow-up
Routine follow-up was conducted to monitor
postoperative complications and survival. All patients underwent
computed tomography assessment every 3 months during the first
postoperative year, every 6 months during the second year and
annually thereafter, up to 5 years.
Statistical analysis
Descriptive statistics are used to summarize
baseline characteristics, treatment exposure, pathological response
and postoperative outcomes. Categorical variables are presented as
absolute numbers and percentages. Continuous variables are
expressed as median (IQR). Data analysis was performed using SPSS
Statistics for Windows, version 28.0 (IBM Corp.). Between-group
comparisons in the cT4a vs. cT4b subgroup were performed using the
Mann-Whitney U test for continuous variables and the Pearson
χ2 test for categorical variables; Fisher's exact test
was applied when assumptions for χ2 were not met due to
small expected frequencies. All tests were two-sided and P<0.05
was considered to indicate a statistically significant difference.
Given the exploratory design, P-values were interpreted
descriptively without multiplicity adjustment.
Results
Patient characteristics
A total of 76 patients with resectable gastric or
GEJ adenocarcinoma who received TNT with FLOT were included in the
present study. The median age at diagnosis was 56 years (IQR,
48–63.5) and the majority of patients (61.84%) were male. Most
patients presented with an ECOG performance status of 0 (77.63%),
while 22.37% had an ECOG performance status of 1. Adenocarcinoma of
unspecified subtype was observed in 71.05% of patients, while
27.63% had poorly cohesive carcinoma and 1.32% had adenosquamous
carcinoma. Overall, 1 patient (1.32%) was excluded due to
metastatic disease progression after four cycles of neoadjuvant
chemotherapy. Another 3 patients (3.95%) were excluded following
completion of TNT due to newly diagnosed metastases, and 1 patient
(1.32%) was excluded due to disease progression and conversion to
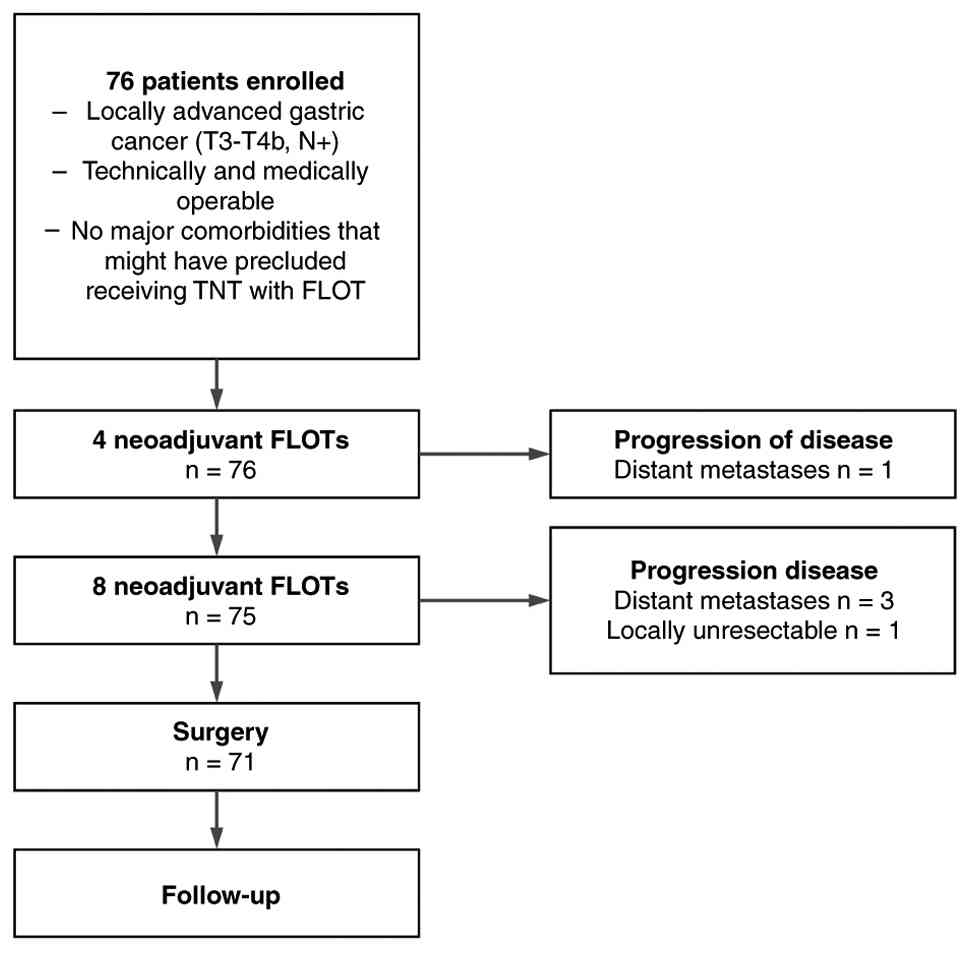
unresectable status (Fig. 1).
Regarding tumor location, the most common sites were
the corpus (48.68%), cardia (18.42%) and linitis plastica (18.42%).
Only a minority of patients had antrum (9.21%) or Siewert III
junctional tumors (5.26%) and no patients were presented with
Siewert II tumors. Clinical T4 stage was predominant, with 51.32%
of patients staged as T4a and 23.68% as T4b. Clinical T3 stage
accounted for 25.00% of cases. Extensive lymph node involvement was
common: cN1 in 32.89%, cN2 in 42.11% and cN3 in 23.68%. Only 1
patient (1.32%) had no nodal involvement (cN0). Tumor grading
revealed 50.00% of tumors as G3, 14.47% as G2, 6.58% as G1 and
28.95% as Gx. A total of 3 patients received chemotherapy in the
past for colorectal cancer and 1 patient for hematological
malignancy.
Neoadjuvant therapy completion
A total of 42 patients (55.26%) completed all 8
cycles of FLOT chemotherapy. Another 14 patients (18.42%) completed
7 cycles due to mild adverse events, 10 patients (13.16%) completed
6 cycles and 5 patients (6.58%) completed 5 cycles. The schedule of
neoadjuvant interventions is displayed in Table II.
Post-neoadjuvant assessment
After completion of TNT with FLOT, tumor response
was assessed using RECIST 1.1 criteria. A partial response was
observed in 48 patients (63.16%), stable disease in 22 patients
(28.95%) and progressive disease in 6 patients (7.89%) (Table III). Second-look staging
laparoscopy was performed based on MDT recommendations for patients
who had been considered as candidates for curative surgery,
primarily in cases where clinical or radiological findings
warranted further exploration (n=50; 70.42%). Peritoneal cytology
was obtained only in these selected patients.
 | Table III.Post-neoadjuvant assessment
investigations. |
Table III.
Post-neoadjuvant assessment
investigations.
|
Characteristics | n (%) |
|---|
| Response rate
(RECIST 1.1) (n=76) |
|
| PR | 48 (63.16) |
| SD | 22 (28.95) |
| PD | 6 (7.89) |
| Second-look staging
laparoscopy (n=71) |
|
|
Yes | 50 (70.42) |
| No | 21 (29.58) |
| Peritoneal cytology
(n=50) |
|
|
Positive | 0 (0.00) |
|
Negative | 50 (100.00) |
Safety and toxicity
Leukopenia and neutropenia were the most common
hematological toxicities, with grade 3–4 events in 21.05 and 31.58%
of patients, respectively. The remaining patients did not
experience the respective toxicity (grade 0). Febrile neutropenia
(grade 1–2) occurred in 27.63%, with 7.89% experiencing severe
(grade 3–4) episodes. Severe (grade 3–4) anemia and
thrombocytopenia were less frequent and reached 2.63 and 5.26%,
respectively (Table IV). Among the
non-hematological toxicities, grade 1–2 nausea (76.32%), peripheral
neuropathy (69.74%) and diarrhea (47.37%) were most common. For
these toxicities, grade 3–4 symptoms were reported in 10.53, 21.05
and 9.21%, respectively. Fatigue, anorexia, oral mucositis and
elevated aminotransferase levels were also noted, mostly at lower
grades. Only 2 (2.63%) patients required ICU care due to severe
metabolic complications from gastrointestinal toxicity. Dose
reductions were necessary in 40 (52.63%) patients, mainly due to
cumulative hematological, neurological or gastrointestinal
toxicity. Oxaliplatin was the most frequently adjusted drug,
followed by docetaxel and fluorouracil. Mixed or full-regimen
reductions were implemented based on severity and tolerability
(Table V). Oxaliplatin was the most
frequently reduced drug (25% reduction in 10 (13.16%) patients).
Another 5 (6.58%) patients experienced all-drugs reduction up to
50% and 5 (6.58%) more patients received 25% reduction of each
medication in the FLOT scheme. Overall, 29 patients did not manage
to tolerate the entire volume of TNT due to severe adverse events
(Table VI). The most common
reasons for not having received all 8 cycles of total NAC FLOT were
peripheral neuropathy in 10 (13.16%) patients and febrile
neutropenia in 9 (11.84%) patients.
 | Table IV.Safety and toxicity of total
neoadjuvant chemotherapy with fluorouracil, leucovorin, oxaliplatin
and docetaxel. |
Table IV.
Safety and toxicity of total
neoadjuvant chemotherapy with fluorouracil, leucovorin, oxaliplatin
and docetaxel.
|
| Hematological
toxicity | Non-hematological
toxicity |
|---|
|
|
|
|
|---|
| Toxicity | Leukopenia | Neutropenia | Febrile
neutropenia | Anemia | Platelet count
decrease | Aminotransferase
increased | Nausea | Diarrhea | Peripheral
neuropathy | Fatigue | Anorexia | Oral mucositis |
|---|
| Grades 1–2, n
(%) | 58 (76.32) | 53 (69.74) | 21 (27.63) | 11 (14.47) | 10 (13.16) | 12 (15.79) | 58 (76.32) | 36 (47.37) | 53 (69.74) | 24 (31.58) | 25 (32.89) | 17 (22.37) |
| Grades 3–4, n
(%) | 16 (21.05) | 24 (31.58) | 6 (7.89) | 2 (2.63) | 4 (5.26) | 3 (3.95) | 8 (10.53) | 7 (9.21) | 16 (21.05) | 7 (9.21) | 0 (0.00) | 3 (3.95) |
 | Table V.Dose reduction profile. |
Table V.
Dose reduction profile.
| Percentage of dose
reduction | n (%) |
|---|
| Oxaliplatin
25% | 10 (13.16) |
| All drugs 50% | 5 (6.58) |
| All drugs 25% | 5 (6.58) |
| Oxaliplatin
50% | 3 (3.95) |
| Fluorouracil
25% | 3 (3.95) |
| Docetaxel and
oxaliplatin 25% | 3 (3.95) |
| Docetaxel and
oxaliplatin 50% | 2 (2.63) |
| Oxaliplatin
discontinuation | 3 (3.95) |
| Oxaliplatin 50%,
all other drugs 25% | 1 (1.32) |
| Docetaxel 25%,
oxaliplatin 50% | 1 (1.32) |
| Oxaliplatin and
fluorouracil 25% | 1 (1.32) |
 | Table VI.Treatment discontinuation
reasons. |
Table VI.
Treatment discontinuation
reasons.
| Reason for not
having received all 8 cycles of total NAC FLOT | n (%) |
|---|
| Peripheral
neuropathy | 10 (13.16) |
| Febrile
neutropenia | 9 (11.84) |
| Hepatic
toxicity | 3 (3.95) |
| Diarrhea | 3 (3.95) |
| Oral mucositis | 2 (2.63) |
| Nausea | 1 (1.32) |
| Recurrent upper GI
bleeding | 1 (1.32) |
Surgical treatment
Second-look staging laparoscopy was performed in 50
patients (70.42%) considered high-risk for peritoneal
dissemination; all had negative peritoneal cytology (Table III). A total of 71 patients
proceeded to curative-intent surgery following a median interval of
4–6 weeks post-chemotherapy. Total gastrectomy was the most common
procedure (91.55%), with additional resections including
transhiatal total gastrectomy with distal esophagectomy (5.63%),
subtotal esophagectomy by Ivory Lewis approach (1.41%) and 1
Whipple procedure (1.41%). A D2 lymphadenectomy was performed in
all cases except for the Ivor-Lewis case, in which a 2F
lymphadenectomy was performed. The median lymph node yield was 23.8
(range, 15–42). Postoperative complications occurred in 64.79% of
patients, with major complications (Clavien-Dindo ≥III) in 33.80%.
Most frequent events included pleural effusion, pneumonia,
anastomotic leak and surgical site infection. Other complications
included lymphorrhea, fluid collection, ileus and pancreatic
fistula (data not shown). A multi-visceral resection was required
in 16 patients (22.54%) to achieve oncologically complete (R0)
margins due to suspected or confirmed invasion of adjacent
structures. The most commonly resected organs were the diaphragm
(n=5; 7.04%) and mesocolon (n=4; 5.63%). Additional resections
included: i) Left liver lobe (n=1); distal pancreas with
splenectomy (n=2); iii) spleen alone (n=1); iv) lung (n=1); v)
pericardium (n=1); and vi) left adrenal gland (n=1). Combined
resections were performed when indicated by intraoperative findings
and preoperative imaging.
The 30-day hospital readmission rate was 18.31%. The
30-day postoperative mortality rate was 4.23% and the 90-day
mortality rate reached 7.04% (Table
VII).
 | Table VII.Surgical treatment results
(n=71). |
Table VII.
Surgical treatment results
(n=71).
|
Characteristics | n (%) |
|---|
| Type of
surgery |
|
| Total
gastrectomy | 65 (91.55) |
|
Transhiatal total gastrectomy
with distal esophagectomy | 4 (5.63) |
|
Subtotal esophagectomy by
Ivory Lewis approach | 1 (1.41) |
| Whipple
procedure | 1 (1.41) |
| Multivisceral
resections |
|
|
Lung | 1 (1.41) |
|
Pericardium | 1 (1.41) |
|
Diaphragm | 5 (7.04) |
|
Mesocolon | 4 (5.63) |
| Left
adrenal gland | 1 (1.41) |
|
Liver | 1 (1.41) |
| Distal
pancreatectomy with splenectomy | 2 (2.82) |
|
Splenectomy alone | 1 (1.41) |
| Postoperative
complications |
|
|
Clavien-Dindo, any | 46 (64.79) |
|
Clavien-Dindo, III+ | 24 (33.80) |
| Readmission | 13 (18.31) |
| 30-day
mortality | 3 (4.23) |
| 90-day
mortality | 5 (7.04) |
Efficacy
Pathological tumor regression was assessed using the
Becker classification. A complete pathological response (pCR)
(Becker 1a) was observed in 2 patients (2.82%), while a
near-complete response (Becker 1b) was achieved in 10 patients
(14.08%), resulting in a major pathological response rate (Becker
1a + 1b) of 16.90%. Partial regression (Becker grade 2) was noted
in 36 patients (50.70%), and poor or no regression (Becker grade 3)
in 23 patients (32.39%). A complete (R0) resection was achieved in
66 patients (92.96%). Microscopic residual disease (R1) was
identified in 5 cases (7.04%), and no patients were found to have
macroscopic residual disease (R2). Advanced residual primary tumor
(ypT3-4) predominated after neoadjuvant therapy, accounting for
67.61% of cases, whereas complete pathological response (ypT0) was
observed in 2.82% of patients. Final pathological T staging (ypT)
is revealed in Table VIII.
 | Table VIII.Pathological response (n=71). |
Table VIII.
Pathological response (n=71).
|
Characteristics | n (%) |
|---|
| Resection
margins |
|
| R0 | 66 (92.96) |
| R1 | 5 (7.04) |
| R2 | 0 (0.00) |
| ypT stage |
|
| 0 | 2 (2.82) |
| 1a | 2 (2.82) |
| 1b | 3 (4.23) |
| 2 | 16 (22.54) |
| 3 | 30 (45.25) |
| 4a | 8 (11.27) |
| 4b | 10 (14.08) |
| ypN stage |
|
| 0 | 21 (29.58) |
| 1 | 21 (29.58) |
| 2 | 11 (15.49) |
| 3a | 7 (9.86) |
| 3b | 3 (4.23) |
| Pathohistological
regression |
|
| 1a | 2 (2.82) |
| 1b | 10 (14.08) |
| 2 | 36 (50.70) |
| 3 | 23 (32.39) |
Subgroup analysis by clinical T4
stage
To evaluate potential differences in treatment
delivery and postoperative outcomes according to the extent of
baseline tumor invasion, a subgroup analysis was performed among
patients with clinical T4 disease (n=52), comparing those staged as
cT4a (n=36) and cT4b (n=16) (Table
IX). Baseline demographic and tumor-related variables were
comparable between groups. Median age was 54.5 years (IQR, 47–63)
in the cT4a group and 61.0 years (IQR, 52–66) in the cT4b group
(P=0.38). Sex distribution did not differ significantly (male: 63.9
vs. 62.5%; P=0.92). ECOG performance status (0 vs. 1) was similarly
distributed (P=0.68). Histological subtype (adenocarcinoma vs.
poorly cohesive vs. adenosquamous) demonstrated no significant
difference between cohorts (P=0.74). Tumor location (cardia,
corpus, antrum, linitis plastica and Siewert III) showed no
statistically significant variation (P=0.57). Clinical nodal stage
(cN0-cN3) was also comparable (P=0.81). Malignancy grade showed a
non-significant trend toward differing distribution (G1-G3 vs. Gx)
(P=0.06) (Table IX).
 | Table IX.Subgroup analysis in clinical T4
disease [cT4a (n=36) vs. cT4b (n=16)]. |
Table IX.
Subgroup analysis in clinical T4
disease [cT4a (n=36) vs. cT4b (n=16)].
| Variable | cT4a | cT4b | Statistical
test | P-value |
|---|
| Baseline
characteristics |
|
|
|
|
| ECOG performance
status |
|
| Fisher's exact | 0.684 |
| 0 | 28 (77.78) | 13 (81.25) |
|
|
| 1 | 8 (22.22) | 3 (18.75) |
|
|
| Histological
subtype |
|
| Fisher's exact | 0.742 |
|
Adenocarcinoma | 26 (72.22) | 12 (75.00) |
|
|
| Poorly
cohesive | 9 (25.00) | 3 (18.75) |
|
|
|
Adenosquamous | 1 (2.78) | 1 (6.25) |
|
|
| Tumor location |
|
| Fisher's exact | 0.574 |
|
Cardia | 6 (16.67) | 4 (25.00) |
|
|
|
Corpus | 18 (50.00) | 7 (43.75) |
|
|
|
Antrum | 3 (8.33) | 2 (12.50) |
|
|
| Linitis
plastica | 7 (19.44) | 2 (12.50) |
|
|
| Siewert
III | 2 (5.56) | 1 (6.25) |
|
|
| Clinical N
stage |
|
| Fisher's exact | 0.811 |
|
cN0 | 1 (2.78) | 0 (0.00) |
|
|
|
cN1 | 12 (33.33) | 5 (31.25) |
|
|
|
cN2 | 15 (41.67) | 6 (37.50) |
|
|
|
cN3 | 8 (22.22) | 5 (31.25) |
|
|
| Malignancy
grade |
|
| Fisher's exact | 0.063 |
|
G1-G3 | 28 (77.78) | 9 (56.25) |
|
|
| Gx | 8 (22.22) | 7 (43.75) |
|
|
| Median
age (IQR), years | 54.5 (47–63) | 61.0 (52–66) | Mann-Whitney U | 0.384 |
| Male
sex, n (%) | 23 (63.89) | 10 (62.50) | Fisher's exact | >0.999 |
| Chemotherapy
delivery |
|
|
|
|
| Median
FLOT cycles administered (IQR) | 8 (7–8) | 7 (6–8) | Mann-Whitney U | 0.270 |
|
Completed 8 cycles, n (%) | 23 (63.89) | 6 (37.50) | Fisher's exact | 0.129 |
| Dose
reduction required, n (%) | 14 (38.89) | 13 (81.25) | Fisher's exact | 0.007 |
| Treatment toxicity,
n (%) |
|
|
|
|
|
Leukopenia (any grade) | 28 (77.78) | 13 (81.25) | Fisher's exact | >0.999 |
|
Leukopenia (grade 3–4) | 8 (22.22) | 3 (18.75) | Fisher's exact | >0.999 |
|
Neutropenia (any grade) | 26 (72.22) | 13 (81.25) | Fisher's exact | 0.730 |
|
Neutropenia (grade 3–4) | 12 (33.33) | 6 (37.50) | Fisher's exact | 0.763 |
| Febrile
neutropenia | 10 (27.78) | 5 (31.25) | Fisher's exact | >0.999 |
|
Anemia | 6 (16.67) | 2 (12.50) | Fisher's exact | >0.999 |
|
Thrombocytopenia | 5 (13.89) | 3 (18.75) | Fisher's exact | 0.689 |
|
Diarrhea (any grade) | 19 (52.78) | 7 (43.75) | Fisher's exact | 0.764 |
|
Diarrhea (grade 3–4) | 3 (8.33) | 2 (12.50) | Fisher's exact | 0.637 |
| Nausea
(any grade) | 29 (80.56) | 14 (87.50) | Fisher's exact | 0.704 |
| Nausea
(grade 3–4) | 4 (11.11) | 2 (12.50) | Fisher's exact | >0.999 |
|
Peripheral neuropathy | 26 (72.22) | 13 (81.25) | Fisher's exact | 0.730 |
| Oral
mucositis | 9 (25.00) | 3 (18.75) | Fisher's exact | 0.733 |
|
Elevated
aminotransferases | 6 (16.67) | 3 (18.75) | Fisher's exact | >0.999 |
| Surgical outcomes,
n (%) |
|
|
|
|
|
Multivisceral resection | 3 (8.33) | 13 (81.25) | Fisher's exact | <0.001 |
| R0
resection | 34 (94.44) | 14 (87.50) | Fisher's exact | 0.578 |
|
Postoperative complications
(any grade) | 24 (66.67) | 10 (62.50) | Fisher's exact | 0.763 |
| Major
complications (Clavien-Dindo ≥III) | 11 (30.56) | 6 (37.50) | Fisher's exact | 0.751 |
| 30-day
readmission | 7 (19.44) | 2 (12.50) | Fisher's exact | 0.704 |
| 30-day
mortality | 1 (2.78) | 1 (6.25) | Fisher's exact | 0.525 |
| 90-day
mortality | 2 (5.56) | 2 (12.50) | Fisher's exact | 0.578 |
| Pathology, n
(%) |
|
|
|
|
|
Lymphovascular invasion | 24 (66.67) | 15 (93.75) | Fisher's exact | 0.044 |
|
Perineural invasion | 17 (47.22) | 12 (75.00) | Fisher's exact | 0.077 |
| ypT stage |
|
| Fisher's exact | 0.028 |
|
ypT0 | 1 (2.78) | 0 (0.00) |
|
|
|
ypT1a | 1 (2.78) | 0 (0.00) |
|
|
|
ypT1b | 3 (8.33) | 0 (0.00) |
|
|
|
ypT2 | 9 (25.00) | 1 (6.25) |
|
|
|
ypT3 | 15 (41.67) | 7 (43.75) |
|
|
|
ypT4a | 5 (13.89) | 1 (6.25) |
|
|
|
ypT4b | 2 (5.56) | 7 (43.75) |
|
|
| ypN stage |
|
| Fisher's exact | 0.743 |
|
ypN0 | 13 (36.11) | 3 (18.75) |
|
|
|
ypN1 | 12 (33.33) | 7 (43.75) |
|
|
|
ypN2 | 5 (13.89) | 2 (12.50) |
|
|
|
ypN3a | 4 (11.11) | 3 (18.75) |
|
|
|
ypN3b | 2 (5.56) | 1 (6.25) |
|
|
Regarding chemotherapy delivery, the median number
of administered FLOT cycles was 8 (IQR, 7–8) among patients with
cT4a disease and 7 (IQR, 6–8) among those with cT4b disease
(P=0.27). Completion of all eight cycles occurred in 23 out of 36
(63.89%) patients in the cT4a group and 6 out of 16 (37.50%) in the
cT4b group (P=0.13). Dose reductions were required in 14 out of 36
(38.89%) patients with cT4a tumors and in 13 out of 16 (81.30%)
patients with cT4b tumors (P=0.007). The incidence of leukopenia
(any grade: 77.78 vs. 81.25%; P=0.77; grade 3–4: 22.22 vs. 18.75%;
P=0.79), neutropenia (any grade: 72.22 vs. 81.25%; P=0.49; grade
3–4: 33.33 vs. 37.50%; P=0.78), febrile neutropenia (27.78 vs.
31.25%; P=0.89), anemia (16.67 vs. 12.50%; P=0.70),
thrombocytopenia (13.89 vs. 18.75%; P=0.64), diarrhea (52.78 vs.
43.75%; P=0.55), nausea (80.56 vs. 87.50%; P=0.55), peripheral
neuropathy (72.22 vs. 81.25%; P=0.47), oral mucositis (25.00 vs.
18.75%; P=0.65) and elevated aminotransferases (16.67 vs. 18.75%;
P=0.85) did not differ significantly between the two groups. The
proportion of severe (grade 3–4) non-hematological toxicities was
also similar, including diarrhea (8.33 vs. 12.50%; P=0.64) and
nausea (11.11 vs. 12.50%; P=0.89).
Surgical procedures differed according to baseline T
classification. Multivisceral resection was performed in 3 out of
36 (8.33%) patients with cT4a tumors and in 13 out of 16 (81.25%)
patients with cT4b tumors (P<0.001). R0 resection was achieved
in 34 out of 36 (94.44%) cT4a patients and in 14 out of 16 (87.50%)
cT4b patients (P=0.58). Postoperative complications of any grade
occurred in 24 out of 36 (66.67%) and 10 out of 16 (62.50%)
patients, respectively (P=0.76). Major complications (Clavien-Dindo
≥III) were noted in 11 out of 36 (30.56%) cT4a patients and 6 out
of 16 (37.50%) cT4b patients (P=0.61). The 30-day readmission rates
(19.44 vs. 12.50%; P=0.54), 30-day postoperative mortality (2.78
vs. 6.25%; P=0.52) and 90-day mortality (5.56 vs. 12.50%; P=0.38)
did not differ significantly.
Pathological assessment demonstrated statistically
significant differences in residual primary tumor stage following
TNT with FLOT. Distribution of ypT0-4b differed between subgroups
(P=0.028). ypT0-2 was observed more frequently in cT4a patients.
ypT4b occurred in 2 out of 36 (5.56%) patients and 7 out of 16
(43.75%) patients in the cT4a and cT4b groups, respectively.
Post-treatment nodal stage (ypN0-N3b) did not vary significantly
between groups (P=0.75). Lymphovascular invasion was present in 24
out of 36 (66.67%) patients with cT4a disease and in 15 out of 16
(93.75%) patients with cT4b disease (P=0.04), respectively.
Perineural invasion was identified in 17 out of 36 (47.22%) and 12
out of 16 (75.00%) patients, respectively (P=0.08). Becker
regression grade distribution (1a-3) was also comparable
(P=0.64).
Discussion
The present clinical study suggests that the use of
TNT is a feasible approach for the treatment of locally advanced
resectable gastric cancer. Despite recent advances in perioperative
treatment, ≤40% of patients are unable to complete all planned
postoperative chemotherapy, as clearly shown in the MAGIC and FLOT
trials (11,13). This presents a potential limitation
in achieving long-term disease-free survival (DFS). Currently, only
a limited number of studies have investigated TNT using the FLOT
regimen in gastric cancer, and the available data remain sparse.
Moreover, the optimal number of chemotherapy cycles in the TNT
setting has not yet been established.
One of the key focuses of the present study was
chemotherapy completion. When defining TNT as eight preoperative
cycles, the present findings are comparable to those reported in
the study by Yang et al (15), which found no significant difference
in the proportion of patients completing all planned chemotherapy
cycles between TNT and perioperative groups [19/28 (67.86%) vs.
70/121 (57.85%); P=0.3]. However, TNT patients received a higher
proportion of cycles that included all chemotherapy drugs [93 vs.
74%, P<0.001]. Another study from Türkiye reported a
significantly higher completion rate in the 8-cycle FLOT (×8) group
(89.1%) compared with the four-cycle group (67.6%) (P<0.001)
(28). This difference may reflect
variability in dose reduction strategies and potential selection
bias.
Intensified preoperative chemotherapy was also
explored in the NeoFLOT trial (29), where patients in the experimental
arm received six neoadjuvant FLOT cycles. Ultimately, 48 patients
(82.7%) completed all six cycles. The median dose intensity across
all cycles was 89.2% (docetaxel, 90.4%; oxaliplatin, 89.9%;
leucovorin, 93.3%; and 5-FU, 90.7%). Dose reductions were necessary
in 25 patients (43.1%). However, since this trial included
postoperative chemotherapy, the reported toxicity profile may
reflect cumulative effects. In the present cohort, the most
frequent reasons for failure to complete all eight cycles were
peripheral neuropathy (14.08%) and febrile neutropenia (12.67%).
The most common grade 3–4 adverse events were neutropenia (31.58%),
peripheral neuropathy (21.05%) and nausea (10.53%). These toxicity
levels suggest non-inferior tolerability compared with that in the
MAGIC and FLOT trials, especially regarding life-threatening
events.
Gastrectomy was performed in 65 patients (91.55%) in
the present study. The predominance of total gastrectomy reflected
baseline characteristics of the cohort and tumor distribution, as
most lesions were located in the corpus or proximal stomach or
showed diffuse/total gastric involvement. An extended surgical
approach was chosen for 19 patients (26.76%). TNT with FLOT enabled
a relatively high rate of R0 resection (92.96%). This compares
favorably with R0 rates in the FLOT4 trial (85%) and the MAGIC
trial (81.9%). These results may be explained by more aggressive
surgical strategies aimed at ensuring complete resection in
patients who are not expected to receive adjuvant therapy.
Certain cohort studies have reported similar
surgical morbidity between TNT and perioperative groups.
Clavien-Dindo grade III–IV complication rates were comparable
(36–38%) in both arms, with no significant increase in
postoperative complications for TNT patients (13,15,30,31).
Similarly, the 30-day postoperative mortality rate was 4.23%, which
was not relatively higher compared with that in perioperative
cohort in FLOT4 (2% in the FLOT group and 3% in the ECF/EXC group)
and MAGIC (5.6% in postop chemotherapy arm and 5.9% in surgery
alone arm) trials. Multi-visceral resections may have a negative
impact on postoperative complications and mortality in affected
patients; however, this data should be validated in prospective
cohorts. One study suggested that the limited extent of resections
for gastric cancer did not compromise oncological outcomes.
However, total gastrectomy was associated with higher rates of
postoperative complications (31).
Alternative TNT strategies have also been explored.
Several trials have reported higher R0 resection and pCR rates in
patients treated with induction doublet or triplet chemotherapy
followed by chemoradiation and curative surgery (30–33).
The present study reported a relatively lower pCR rate (2.82%)
compared with those of the aforementioned trials. In the present
study, most of the patients were T4a (n=36; 50.70%) and T4b (n=16;
22.54%) with positive nodal status (cN0 only in 1 patient; 1.41%),
which may have negatively influenced the ability of TNT with FLOT
in achieving a potentially better pathological response. Moreover,
incomplete chemotherapy delivery in some patients may have
compromised oncological outcomes.
Toxicity rates associated with TNT using the FLOT
regimen were reported to be acceptable in related studies (15,24,26,30),
without a significant increase in grade 3–4 adverse events compared
with standard perioperative chemotherapy approaches. However,
survival outcomes were not consistently shown to be significantly
improved compared with conventional perioperative treatment
strategies. Given the complexity of these multimodal regimens,
further research is required to identify which patient subgroups
may derive the greatest benefit based on individualized clinical
and pathological characteristics.
The ongoing FLOT9 (PREVENT) trial aims to evaluate
3–6 cycles of preoperative FLOT followed by curative surgery, with
or without HIPEC, and adjuvant FLOT chemotherapy (34). In parallel, the OCTASUR trial is
investigating eight cycles of TNT followed by curative surgery
without adjuvant therapy (35). The
SPACE-FLOT study (n=1,887) showed that the benefit of adjuvant FLOT
depends on pathological response (36). In partial responders (n=1,207),
adjuvant FLOT improved DFS (HR, 0.68; 95% CI, 0.55–0.86;
P<0.001) and OS (HR, 0.55; 95% CI, 0.44–0.69; P<0.001) rates.
However, no survival benefit was seen in complete (n=221) or
minimal (n=459) responders, with non-significant HRs for DFS and OS
in both groups. These findings support tailoring adjuvant therapy
based on response and tolerance. Despite the findings of the
present study, other publications have reported differing results,
which may be attributed to variations in patient characteristics or
disease prevalence across study cohorts: TNT yielded higher
pathological response, with ~2-fold increase in pCR (18.9 vs. 8.1%)
in patients receiving FLOT ×8 compared to perioperative 4 + 4,
though differences did not always reach statistical significance
(P=0.29) (37).
TNT has demonstrated a trend toward improved
pathological response compared with standard perioperative FLOT.
Several studies have reported a 2-fold increase in pCR rates with
TNT compared with those when using the perioperative approach,
although these differences did not always reach statistical
significance (28,30,32).
In terms of feasibility, TNT was associated with
better treatment adherence: Up to 89% of patients completed all
eight preoperative FLOT cycles, in contrast to poor postoperative
compliance in perioperative regimens, where ~19% of patients failed
to initiate adjuvant chemotherapy (13,28).
Importantly, toxicity profiles were similar across both strategies,
with no significant increase in grade 3–4 adverse events (~27%), no
delay to surgery and no prolongation of hospital stay observed in
the TNT group.
The present study has several important limitations.
First, its retrospective design limits the ability to stratify,
randomize and prospectively compare relevant patient groups.
Second, it is a single-center study, which inherently involves a
more homogeneous patient population and may lead to overestimation
of treatment effects. Third, selection bias may be present due to
the inclusion of patients with varying tumor burden (localized vs.
locally advanced), nodal status (bulky vs. non-bulky
lymphadenopathy) and peritoneal cytology results. Additionally,
patients who were initially allocated to TNT with FLOT but received
fewer than four cycles of chemotherapy were excluded. In such
cases, upfront surgery or biological therapy may be more
appropriate for carefully selected individuals. One more critical
limitation is the absence of survival data that may be crucial in
the eventual showcasing of feasibility and survival benefits.
The absence of a control group appears as one of the
most significant limitations in the present study. The outcomes may
not be considered as equivalent of those reached in other
prospective randomized trials with the FLOT regimen. Further
multicohort investigation of TNT with FLOT for locally advanced
gastric and GEJ cancer is needed. As the aim of the present study
was to showcase the safety and feasibility of TNT with FLOT rather
than stress the issues on comparison between peri-operative,
post-operative or without chemotherapy cohorts, future direction
requires prospective randomized cohorts to obtain the statistically
significant data on TNT vs. chemotherapy + surgery + chemotherapy
that might have a relevant clinical outcome. Recurrence, OS and
relapse-free survival are being investigated and will be described
in future studies.
In the present exploratory subgroup analysis,
notable differences between cT4a and cT4b tumors emerged across the
neoadjuvant and surgical pathways. Although baseline demographics
and clinical characteristics were similar, patients with cT4b
disease required more dose reductions. Despite this, TNT with FLOT
was delivered with comparable overall toxicity, and postoperative
morbidity and mortality did not differ between groups, indicating
that treatment feasibility was maintained even in the setting of
deeper local invasion. The most pronounced distinctions were
observed at the pathological level: cT4b tumors demonstrated
significantly less primary tumor downstaging and higher rates of
lymphovascular invasion, while Becker regression and nodal status
remained similar. These findings collectively suggest that although
TNT with FLOT enables high R0 resection rates and acceptable
perioperative safety across the T4 spectrum, cT4b disease retains
more adverse biological features and exhibits a more limited
pathological response to neoadjuvant therapy, underscoring the need
for further stratified investigation in prospective cohorts.
Tumor location was the predominant rationale for
choosing a total gastrectomy as an optimal surgical approach in the
present study. Recent data on limited gastric resections has
revealed the non-inferiority of limited gastric resections in terms
of oncological outcomes, although the technique has less
postoperative complications (31,37).
As the present study is retrospective, it has no power to assess
the association between total gastrectomy, postoperative
complications and mortality rates.
The last thing to be noted is that TNT with FLOT
appeared feasible and led to downstaging in some patients; however,
due to the study's observational design, the results are hypothesis
generating rather than confirmatory.
In conclusion, the present study indicates that TNT
with FLOT may be a safe treatment strategy for resectable gastric
cancer. Further prospective studies are needed to assess long-term
survival outcomes.
Acknowledgements
Not applicable.
Funding
Funding: No funding was received.
Availability of data and materials
The data generated in the present study may be
requested from the corresponding author.
Authors' contributions
OD was responsible for conceptualization,
investigation, methodology, project administration, resources,
supervision, validation, writing the original draft, and reviewing
and editing the manuscript. MP was responsible for
conceptualization, investigation, supervision, writing the original
draft, and reviewing and editing the manuscript. AH was responsible
for conceptualization, formal analysis, investigation, methodology,
validation, visualization, writing the original draft, and
reviewing and editing the manuscript. YK was responsible for
conceptualization, methodology, project administration,
supervision, validation, and reviewing and editing the manuscript.
OD, MP and AH also performed extraction of clinical, treatment,
surgical, and pathology variables from source records;
de-identification; database entry and coding; data cleaning
(range/consistency checks and handling missing values);
cross-checking key outcomes with operative and pathology reports;
and preparation of the final locked dataset used for statistical
analysis. All authors have read and approved the manuscript. OD and
AH confirm the authenticity of all the raw data.
Ethics approval and consent to
participate
The study was conducted in accordance with the
Declaration of Helsinki and was approved by the Ethics Committee of
the National Cancer Institute (Kyiv, Ukraine; approval no. 174) on
March 14, 2024. The requirement for informed consent was
waived.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Ferlay J, Ervik M, Lam F, Laversanne M,
Colombet M, Mery L, Piñeros M, Znaor A, Soerjomataram I and Bray F:
Global Cancer Observatory: Cancer Today. International Agency for
Research on Cancer; Lyon: 2024, https://gco.iarc.fr/today/en/dataviz/maps-heatmap?mode=population&cancers=7May
5–2025
|
|
2
|
Zhang H, Yang W, Tan X, He W, Zhao L, Liu
H and Li G: Long-term relative survival of patients with gastric
cancer from a large-scale cohort: A period-analysis. BMC Cancer.
24:14202024. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Yago A, Haruta S, Ueno M, Hamada Y, Ogawa
Y, Ohkura Y, Urabe M and Udagawa H: Adequate period of surveillance
in each stage for curatively resected gastric cancer: Analyzing the
time and rates of recurrence. Gastric Cancer. 24:752–7761. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
National Comprehensive Cancer Network
(NCCN), . NCCN Clinical Practice Guidelines in Oncology. Gastric
Cancer. Version 2.2024, Published February 13, 2024. Available
from:. https://www.nccn.org/professionals/physician_gls/pdf/gastric.pdfMay
20–2025
|
|
5
|
Fedorenko ZP, Sumkina OP, Zub VO, Gorokh
EL, Gulak LO, Ryzhov AY and Yefimenko OV: Cancer in Ukraine
2021–2022, Incidence, mortality, oncology service performance
indicators. National Cancer Registry of Ukraine; http://www.ncru.inf.ua/publications/BULL_24/PDF_E/22-23-zel.pdf15–12.
2025
|
|
6
|
Tang Z, Deng L, Zhang J, Jiang T, Xiang H,
Chen Y, Liu H, Cai Z, Cui W and Xiong Y: Intelligent
hydrogel-assisted hepatocellular carcinoma therapy. Research (Wash
DC). 7:04772024.PubMed/NCBI
|
|
7
|
Chan K, Saltagi M and Choi KY: The
emerging role of sentinel lymph node biopsy in oral cavity and
oropharyngeal carcinomas. Plast Aesthet Res. 11:62024.
|
|
8
|
André T, Tougeron D, Piessen G, de la
Fouchardière C, Louvet C, Adenis A, Jary M, Tournigand C, Aparicio
T, Desrame J, et al: Neoadjuvant nivolumab plus ipilimumab and
adjuvant nivolumab in localized deficient mismatch
repair/microsatellite instability-high gastric or esophagogastric
junction adenocarcinoma: The GERCOR NEONIPIGA Phase II study. J
Clin Oncol. 41:255–265. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Janjigian YY, Van Cutsem E, Muro K,
Wainberg Z, Al-Batran SE, Hyung WJ, Molena D, Marcovitz M, Ruscica
D, Robbins SH, et al: MATTERHORN: Phase III study of durvalumab
plus FLOT chemotherapy in resectable gastric/gastroesophageal
junction cancer. Future Oncol. 18:2465–2473. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Burotto M, Wilkerson J, Stein WD, Bates SE
and Fojo T: Adjuvant and neoadjuvant cancer therapies: A historical
review and a rational approach to understand outcomes. Semin Oncol.
46:83–99. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Cunningham D, Allum WH, Stenning SP,
Thompson JN, Van de Velde CJ, Nicolson M, Scarffe JH, Lofts FJ,
Falk SJ, Iveson TJ, et al: Perioperative chemotherapy versus
surgery alone for resectable gastroesophageal cancer. N Engl J Med.
355:11–20. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Boige V, Pignon J, Saint-Aubert B, Lasser
P, Conroy T, Bouché O, Segol P, Bedenne L, Rougier P and Ychou M:
Final results of a randomized trial comparing preoperative
5-fluorouracil (F)/cisplatin (P) to surgery alone in adenocarcinoma
of stomach and lower esophagus (ASLE): FNLCC ACCORD07-FFCD 9703
trial. J Clin Oncol. 25:45102007. View Article : Google Scholar
|
|
13
|
Al-Batran SE, Homann N, Pauligk C, Goetze
TO, Meiler J, Kasper S, Kopp HG, Mayer F, Haag GM, Luley K, et al:
Perioperative chemotherapy with fluorouracil plus leucovorin,
oxaliplatin, and docetaxel versus fluorouracil or capecitabine plus
cisplatin and epirubicin for locally advanced, resectable gastric
or gastro-oesophageal junction adenocarcinoma (FLOT4): A
randomised, phase 2/3 trial. Lancet. 393:1948–1957. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Bedrikovetski S, Traeger L, Fitzsimmons T,
Perry J, Vather R, Moore JW and Sammour T: Personalized total
neoadjuvant therapy versus chemotherapy during the ‘wait period’
versus standard chemoradiotherapy for locally advanced rectal
cancer. ANZ J Surg. 93:1267–1273. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Yang J, Gold PJ, Chou JF, Coit DG, Chou
JF, Capanu M, Maron SB, Kelsen DP, Ilson DH, Janjigian YY and Ku
GY: Perioperative versus total neoadjuvant chemotherapy in gastric
cancer: A retrospective cohort study. J Gastrointest Oncol.
14:1193–1203. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Shi J, Li N, Tang Y, Jiang L, Yang L, Wang
S, Song Y, Liu Y, Fang H, Lu N, et al: Total neoadjuvant therapy
for locally advanced gastric cancer and esophagogastric junction
adenocarcinoma: Study protocol for a prospective, multicenter,
single-arm, phase II clinical trial. BMC Gastroenterol. 22:3592022.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Mandard AM, Dalibard F, Mandard JC, Marnay
J, Henry-Amar M, Petiot JF, Roussel A, Jacob JH, Segol P, Samama G,
et al: Pathologic assessment of tumor regression after preoperative
chemoradiotherapy of esophageal carcinoma. Clinicopathologic
correlations. Cancer. 73:2680–2686. 1994. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Liu S, Jiang T, Xiao L, Yang S, Liu Q, Gao
Y, Chen G and Xiao W: Total neoadjuvant therapy (TNT) versus
standard neoadjuvant chemoradiotherapy for locally advanced rectal
cancer: A systematic review and Meta-analysis. Oncologist.
26:e1555–e1566. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Edge SB, Byrd DR, Compton CC, Fritz AG,
Greene FL and Trotti A: AJCC Cancer Staging Manual. 7th edition.
Springer; London, UK: pp. 117–126. 2010
|
|
20
|
Oken MM, Creech RH, Tormey DC, Horton J,
Davis TE, McFadden ET and Carbone PP: Toxicity and response
criteria of the Eastern Cooperative Oncology Group. Am J Clin
Oncol. 5:649–655. 1982. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
NCCP Regimen, . FLOT Therapy-14 day.
Published: 08/09/2017 Review: 28/04/2026 Version number: 8b Tumour
Group: Gastrointestinal NCCP Regimen Code: 00344 ISMO Contributor:
Prof Maccon Keane. Available from:. https://www.hse.ie/eng/services/list/5/cancer/profinfo/chemoprotocols/gastrointestinal/344.pdf
|
|
22
|
Common Terminology Criteria for Adverse
Events (CTCAE) Version 5. Published, . November 27, 2027. US
Department of Health and Human Services, National Institutes of
Health, National Cancer Institute;
|
|
23
|
Eisenhauer EA, Therasse P, Bogaerts J,
Schwartz LH, Sargent D, Ford R, Dancey J, Arbuck S, Gwyther S,
Mooney M, et al: New response evaluation criteria in solid tumours:
Revised RECIST guideline (version 1.1). Eur J Cancer. 45:228–2247.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Aaronson NK, Ahmedzai S, Bergman B,
Bullinger M, Cull A, Duez NJ, Filiberti A, Flechtner H, Fleishman
SB, de Haes JC, et al: The european organization for research and
treatment of cancer QLQ-C30: A quality-of-life instrument for use
in international clinical trials in oncology. J Natl Cancer Inst.
85:365–376. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Vickery CW, Blazeby JM, Conroy T, Arraras
J, Sezer O, Koller M, Rosemeyer D, Johnson CD and Alderson D; EORTC
Quality of Life Group, : Development of an EORTC disease-specific
quality of life module for use in patients with gastric cancer. Eur
J Cancer. 37:966–971. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Dindo D, Demartines N and Clavien PA:
Classification of surgical complications: A new proposal with
evaluation in a cohort of 6336 patients and results of a survey.
Ann Surg. 240:205–213. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Becker K, Langer R, Reim D, Novotny A,
Meyer zum Buschenfelde C, Engel J, Friess H and Hofler H:
Significance of histopathological tumor regression after
neoadjuvant chemotherapy in gastric adenocarcinomas: A summary of
480 Cases. Ann Surg. 253:934–939. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Rencuzogullari A, Karahan SN,
Selcukbiricik F, Lacin S, Taskin OC, Saka B, Karahacioglu D, Gurses
B, Ozoran E, Uymaz DS, et al: The new era of total neoadjuvant FLOT
therapy for locally advanced, resectable gastric cancer: A
Propensity-matched comparison with standard perioperative therapy.
J Surg Oncol. 131:417–426. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Schulz C, Kullmann F, Kunzmann V, Fuchs M,
Geissler M, Vehling-Kaiser U, Stauder H, Wein A, Al-Batran SE,
Kubin T, et al: NeoFLOT: Multicenter phase II study of
perioperative chemotherapy in resectable adenocarcinoma of the
gastroesophageal junction or gastric adenocarcinoma-Very good
response predominantly in patients with intestinal type tumors. Int
J Cancer. 137:678–685. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Jurkowski L, Shreenivas AV, Chakrabarti S,
Kamgar M, Thomas JP, Puckett L, Shukla M and George B: Association
of total neoadjuvant therapy with favorable clinical outcomes in
patients with locally advanced esophageal and gastroesophageal
junction adenocarcinomas (LA-GEJ CA). J Clin Oncol. 39:2312021.
View Article : Google Scholar
|
|
31
|
Boubaddi M, Teixeira Farinha H, Lambert C,
Pereira B, Piessen G, Gualtierotti M, Voron T, Mantziari S, Pezet D
and Gronnier C; French Association of Surgery, : Total versus
subtotal gastrectomy for distal gastric poorly cohesive carcinoma.
Ann Surg Oncol. 31:744–752. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Ho F, Torphy RJ, Friedman C, Leong S, Kim
S, Wani S, Schefter T, Scott CD, Mitchell JD, Weyant MJ, et al:
Induction chemotherapy plus neoadjuvant chemoradiation for
esophageal and gastroesophageal junction adenocarcinoma. Ann Surg
Oncol. 28:7208–7218. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Carr RA, Hsu M, Harrington CA, Tan KS,
Bains MS, Bott MJ, Ilson DH, Isbell JM, Janjigian YY, Maron SB, et
al: Induction FOLFOX and PET-directed chemoradiation for locally
advanced esophageal adenocarcinoma. Ann Surg. 277:e538–e544. 2023.
View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Götze TO, Piso P, Lorenzen S, Bankstahl
US, Pauligk C, Elshafei M, Amato G, Reim D, Bechstein WO,
Königsrainer A, et al: Preventive HIPEC in combination with
perioperative FLOT versus FLOT alone for resectable diffuse type
gastric and gastroesophageal junction type II/III
adenocarcinoma-the phase III ‘PREVENT’-(FLOT9) trial of the
AIO/CAOGI/ACO. BMC Cancer. 21:11582021. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Pepenin M, Kondratskyi Y Dobrzhanskyi O,
Horodetskii A and Shudrak Y: FLOT Total Neoadjuvant Chemotherapy in
Locally Advanced Gastric and Gastroesophageal Junction Cancer
(OCTASUR): Randomized, single center, open label trial, Phase 2/3.
Eur J Surg Oncol. 50:1088392024. View Article : Google Scholar
|
|
36
|
SPACE-FLOT Investigators: Pathological
response guides adjuvant 5-fluorouracil, leucovorin, oxaliplatin,
and docetaxel (FLOT) chemotherapy in surgically resected
gastro-oesophageal cancer (SPACE-FLOT): International cohort study.
BJS. 112:znaf0562025. View Article : Google Scholar
|
|
37
|
Roeland E, Kanter K, Yon-Li Wo J, Fish M,
David Nipp R, Van Seventer EE, Raj Parikh A, Allen JN, Giantonio
BJ, Scott Blaszkowsky L, et al: Preliminary analysis of total
neoadjuvant therapy for patients with locally advanced gastric (G)
and gastroesophageal (GE) adenocarcinoma. J Clin Oncol. 38:393.
2020. View Article : Google Scholar
|