Introduction
Liver cancer represents a notable global health
burden, characterized by high morbidity and mortality rates.
Hepatocellular carcinoma (HCC) is the most common type of primary
liver cancer, accounting for ~90% of all cases (1,2).
Globally, HCC ranks as the sixth most common malignancy and the
third leading cause of cancer-related death, with an estimated
830,200 deaths annually (3–5). The notable causes of liver cancer
include hepatitis B virus (HBV) infection, hepatitis C virus (HCV)
infection, non-alcoholic fatty liver disease, aflatoxin,
alcoholism, obesity and hereditary factors (6). It is estimated that by 2030,
>1,000,000 people will die annually from primary liver cancer
worldwide (7).
Despite advances in surveillance programs for HCC,
~80% of patients are diagnosed at intermediate or advanced stages
(8,9). Among the various treatment options for
liver cancer, transcatheter arterial chemoembolization (TACE) has
emerged as a primary modality for patients with unresectable
(u)HCC, owing to its distinct therapeutic advantages, including the
ability to deliver high concentrations of chemotherapeutic agents
directly into the tumor via the hepatic artery while simultaneously
inducing ischemic necrosis through embolization. This approach
maximizes local antitumor efficacy while minimizing systemic
toxicity, and can be repeated as needed based on tumor response. It
is also one of the most frequently employed locoregional therapies
for liver cancer (10,11).
The clinical efficacy of TACE has been demonstrated
in multiple aspects; compared with traditional surgical resection,
TACE is effective and associated with less physical trauma.
According to a meta-analysis by Lencioni et al (12), the 1-, 3- and 5-year overall
survival (OS) rates are 70.3, 40.4 and 32.4%, respectively, with a
median survival time of 19.4 months. A meta-analysis encompassing
4,966 patients with HCC reported that those treated with TACE
achieve improved survival outcomes, with a median OS time of 3.3
years and a 5-year OS rate of 34% (13).
The efficacy of TACE is influenced by multiple
factors, including tumor vascularity, tumor size and number and
liver function reserve (13). TACE
is generally more effective in hypervascular tumors, whereas its
therapeutic effect is relatively limited in hypovascular tumors
(14). Moreover, current evidence
indicates that TACE has inherent limitations (15). Factors such as tumor progression,
unfavorable tumor location or size and incomplete embolization
contribute to a low rate of complete necrosis and a high rate of
local recurrence; studies (14,16)
have reported a complete necrosis rate <20% following TACE.
Furthermore, whether repeated TACE sessions consistently confer
clinical benefit remains controversial (17). The present study aimed to evaluate
the clinical efficacy of TACE frequencies in patients with uHCC and
to identify factors influencing prognosis.
Materials and methods
Study design and patient
selection
The present retrospective study was conducted in
compliance with the Declaration of Helsinki. The study protocol was
approved by the Ethics Committee of Hebei General Hospital
(Shijiazhuang, China) (approval no. 2024-LW-0204). Medical records
of 159 patients diagnosed with uHCC who were admitted to Hebei
General Hospital between January 2017 and December 2023 were
retrospectively reviewed. The patients had a median age of 62 years
(range, 35–81 years) and included 125 males (78.6%) and 34 females
(21.4%). Laboratory data obtained ≤3 days before the first TACE
session were collected. Inclusion criteria were as follows: i)
Histologically, cytologically or clinically confirmed HCC based on
American Association for the Study of Liver Diseases criteria
(18); ii) Child-Pugh class A or B
(19); iii) Barcelona Clinic Liver
Cancer (BCLC) stage B or C (20),
or stage A with contraindications to surgery (severe
cardiopulmonary insufficiency); iv) no prior TACE or systemic
antitumor therapy for HCC; v) eligibility for TACE according to
established indications (15); and
vi) availability of complete follow-up data. Exclusion criteria
were as follows: i) History or presence of other malignancies; ii)
prior local or systemic therapy for HCC; iii) Child-Pugh class C;
iv) pregnancy; v) coagulation disorders; and vi) missing or
incomplete follow-up data. Liver function was assessed using the
Child-Pugh classification, which allocates 1 to 3 points for each
of five parameters (total bilirubin, serum albumin, prothrombin
time, ascites and hepatic encephalopathy), with a total score
ranging from 5 to 15 points. Scores were categorized as follows:
Class A, 5–6 points; class B, 7–9 points; and class C, ≥10 points.
Tumor staging was performed according to the BCLC system. Patient
demographics, medical history, preoperative laboratory tests
(including blood count, liver function and tumor markers), imaging
findings and other relevant data, including body mass index,
Child-Pugh class, HBV infection status, HCV infection status,
hypertension, diabetes, α-fetoprotein, carcinoembryonic antigen,
carbohydrate antigen 19-9, albumin, alanine aminotransferase and
aspartate aminotransferase levels, white blood cell count,
neutrophil-to-lymphocyte ratio, platelet count, cirrhosis, tumor
number, portal vein tumor thrombus, extrahepatic metastasis,
maximal tumor size and BCLC stage, were retrieved from the
electronic medical record system.
Treatment
All patients were hospitalized to complete
preoperative evaluations, which included comprehensive laboratory
tests (blood count, liver and renal function, coagulation profile
and tumor markers), contrast-enhanced imaging (CT or MRI) to assess
tumor burden and vascular anatomy, and evaluation of liver function
reserve using the Child-Pugh classification. Patients were then
informed of the available treatment options, underwent TACE
according to their wishes and tumor characteristics, and provided
written informed consent. TACE was performed by an experienced
interventional radiologist following clinical practice guidelines
(15). Following routine
disinfection, draping and local anesthesia, the femoral artery was
punctured and the location, number and size of tumors were
confirmed by hepatic arteriography. Based on the angiographic
findings, the catheter was advanced into the tumor-feeding artery.
Embolic agents (lipiodol, gelatin sponge, drug-eluting
microspheres) were injected to occlude the tumor blood supply,
followed by infusion of chemotherapeutic drugs (fluorouracil,
cisplatin, epirubicin) to induce tumor cell death. The dosage was
tailored based on tumor vascularity, patient body surface area,
general condition, liver function and tolerance to the procedure.
Following embolization, intraoperative digital subtraction
angiography was performed to evaluate tumor devascularization. The
procedure was considered successful when repeat imaging of the
common hepatic artery confirmed near-complete absence of tumor
staining. Following completion of the infusion, the catheter was
removed and manual compression was applied at the puncture site for
15 min to achieve hemostasis. The patient was transferred to the
Department of Hepatobiliary Surgery in stable condition, without
any intraoperative complications. Postprocedural supportive care
included hepatoprotective agents, gastric mucosal protection,
antiemetics, analgesics and nutritional support; prophylactic
antibiotics were administered as needed to prevent infection.
Follow-up imaging was repeated 4–6 weeks after TACE using
contrast-enhanced CT (Siemens SOMATOM Definition Flash; Siemens
Healthineers) or contrast-enhanced MRI (Siemens MAGNETOM Skyra
3.0T; Siemens Healthineers). CT scanning parameters were as
follows: Tube voltage, 120 kV; tube current, 200–250 mAsec; slice
thickness, 5 mm; and intravenous injection of 80–100 ml of
iodinated contrast agent (Omnipaque, 350 mg I/ml) at a rate of 3
ml/sec, with arterial, portal venous and delayed phase imaging. MRI
parameters included axial T1-weighted, T2-weighted and
diffusion-weighted sequences, with intravenous administration of
0.1 mmol/kg of gadolinium-based contrast agent (Gd-DTPA) for
dynamic contrast-enhanced imaging. The necessity for additional
TACE sessions was assessed based on imaging findings and tumor
marker levels. Patients were informed about the recommended and
alternative treatment options, including the benefits and risks of
each approach, potential treatment-related adverse effects (AEs)
and tumor-related risk factors. To account for procedural
heterogeneity, the specific embolic agents and chemotherapeutic
regimens used in each TACE session were documented. The decision to
repeat TACE was made by a multidisciplinary team according to
standardized clinical criteria as follows: i) Presence of residual
viable tumor or new intrahepatic lesions on follow-up
contrast-enhanced CT/MRI performed 4–6 weeks after the previous
TACE; ii) preserved liver function (Child-Pugh class A or B); iii)
acceptable tolerance to prior TACE sessions (absence of grade ≥3
AEs); iv) absence of contraindications, including notable tumor
progression with new extrahepatic metastases, main portal vein
invasion or deterioration of performance status to Eastern
Cooperative Oncology Group (ECOG) ≥3 (21); and v) patient preference following
discussion of risks and benefits. TACE was not repeated in cases of
disease progression with new extrahepatic metastases, notable
deterioration of liver function (Child-Pugh class C) or patient
refusal. Repeat TACE was performed at intervals of 4–8 weeks, as
determined by the multidisciplinary team to allow sufficient time
for tumor response assessment (typically 4–6 weeks post-TACE) and
for patient recovery from any post-embolization syndrome (PES).
Follow-up and assessment
Enrolled patients were followed up regularly through
review of clinical records or by telephone contact. Survival status
was documented. The final follow-up date was October 2024.
Contrast-enhanced CT/MRI and laboratory tests were performed 4–6
weeks after the first TACE session to assess treatment response.
Thereafter, imaging and laboratory assessments were repeated every
3 months on an outpatient basis. Follow-up results were reviewed by
the multidisciplinary team to determine tumor status (progression
or non-progression). Overall survival (OS) was defined as the time
from first TACE to death from any cause or the last follow-up
(October 2024). PFS was defined as the interval from first TACE to
the first documented disease progression [according to Modified
Response Evaluation Criteria in Solid Tumors (mRECIST) criteria]
(22) or death from any cause,
whichever occurred first. Patients without progression or death at
the last follow-up were censored. For patients who underwent TACE
for recurrent HCC following hepatectomy, the starting point was
similarly the date of the first TACE session for the recurrence.
AEs were graded according to the National Cancer Institute Common
Terminology Criteria for AEs, version 5.0 (23). OS was the primary endpoint of the
present study, while PFS and the incidence of AEs were secondary
endpoints. Tumor response was assessed by two diagnostic
radiologists, each with >10 years of experience, according to
mRECIST (22). Efficacy was
evaluated using mRECIST criteria as follows: i) Complete response
(CR), disappearance of intratumoral arterial enhancement in all
target lesions; ii) partial response (PR), ≥30% decrease in the sum
of diameters of viable (enhancing) target lesions; iii) progressive
disease (PD), increase of ≥20% in the sum of diameters of viable
target lesions, or appearance of new lesions; iv) stable disease
(SD), neither sufficient shrinkage to qualify for PR nor sufficient
increase to qualify for PD. Best objective response rate (ORR) was
defined as the proportion of patients achieving either CR or PR at
any time during follow-up, according to mRECIST criteria. A total
of two tumor response measures were reported: Response at the first
assessment (4–6 weeks after the initial TACE), which enables
unbiased comparison with uniform follow-up duration across groups,
and cumulative best response over the entire follow-up period,
reflecting the maximum response achieved at any time.
Statistical analysis
Statistical analyses were conducted using Microsoft
Excel (version 2019; Microsoft Corp.), SPSS (version 26.0; IBM
Corp.) and R software (version 4.3.3; R Foundation for Statistical
Computing; http://www.R-project.org/). The
propensity score model included the following baseline covariates
measured before the first TACE session: Sex (male/female), age
(<60 vs. ≥60 years), body mass index (BMI; <24 vs. ≥24
kg/m2), Child-Pugh class (A vs. B), HBV infection
(yes/no), HCV infection (yes/no), hypertension (yes/no), diabetes
(yes/no), α-fetoprotein (AFP; <400 vs. ≥400 ng/ml),
carcinoembryonic antigen (CEA; <5 vs. ≥5 ng/ml), carbohydrate
antigen 19-9 (CA199; <37 vs. ≥37 U/ml), albumin (<35 vs. ≥35
g/l), alanine aminotransferase (ALT; <40 vs. ≥40 U/l), aspartate
aminotransferase (AST; <40 vs. ≥40 U/l), white blood cell (WBC)
count (<10 vs. ≥10×109/l), neutrophil-to-lymphocyte
ratio (NLR; continuous), platelet (PLT) count (<100 vs.
≥100×109/l), cirrhosis (yes/no), tumor number (single
vs. multiple), portal vein tumor thrombus (yes/no), extrahepatic
metastasis (yes/no), maximal tumor size (<5 vs. ≥5 cm) and BCLC
stage (A/B vs. C). The number of TACE sessions was excluded from
the propensity score model, as it represented the treatment
assignment. Matching was performed using a 1:1 nearest neighbor
matching algorithm with a caliper width of 0.1 using the ‘MatchIt’
package [version 4.5.0) (24) in R
software (https://cran.r-project.org/web/packages/MatchIt/].
For the three-group comparison, a sequential pairwise matching
approach was employed. First, propensity scores were estimated
using logistic regression based on the aforementioned baseline
covariates. Second, patients in the TACE 2 group were matched 1:1
to those in the TACE 1 group using nearest neighbor matching with a
caliper of 0.1. From the remaining unmatched patients, those in the
TACE ≥3 group were matched 1:1 to patients in the TACE 1 group
using the same caliper. Standardized mean differences (SMDs) were
calculated to evaluate post-matching balance; all covariates
exhibited SMD <0.1, indicating successful matching. Quantitative
data are presented as frequencies, mean ± standard deviation or
medians with 95% confidence intervals (CIs) before and after
propensity score matching (PSM). Differences between groups were
assessed using the χ2 or Fisher's exact test, as
appropriate. Survival curves for PFS and OS were estimated using
the Kaplan-Meier method and compared using the log-rank test. Uni-
and multivariate analyses were conducted using Cox proportional
hazards models to identify prognostic factors. All variables with
P<0.05 in the univariate analysis were included in a
multivariate Cox proportional hazards model to identify independent
predictors. All statistical tests were two-tailed, and P<0.05
was considered to indicate a statistically significant difference.
The proportional hazards assumption was assessed using Schoenfeld
residuals. Receiver operating characteristic (ROC) curve analysis
was performed to determine the optimal cutoff value for the NLR.
The optimal cutoff was defined as the value maximizing the Youden
index (sensitivity + specificity - 1). Data on systemic therapy
(targeted therapy and/or immunotherapy) were collected, including
the timing of initiation relative to TACE sessions. Patients were
categorized according to the timing of systemic therapy initiation
(between TACE sessions) or after the completion of all TACE
sessions. Patients who received systemic therapy prior to any TACE
were excluded according to the inclusion criteria. In the Cox
regression models, systemic therapy was included as a binary
covariate (ever vs. never received). Sensitivity analyses were
performed excluding patients who received systemic therapy between
TACE sessions to assess potential confounding. To mitigate the risk
of overfitting given the sample size of 81 patients following
matching, a conservative modeling strategy was employed. Only
variables with P<0.10 in the univariate analysis were considered
for inclusion in the multivariate models. The final multivariate
models included seven variables for PFS and six variables for
OS.
Results
Patient characteristics
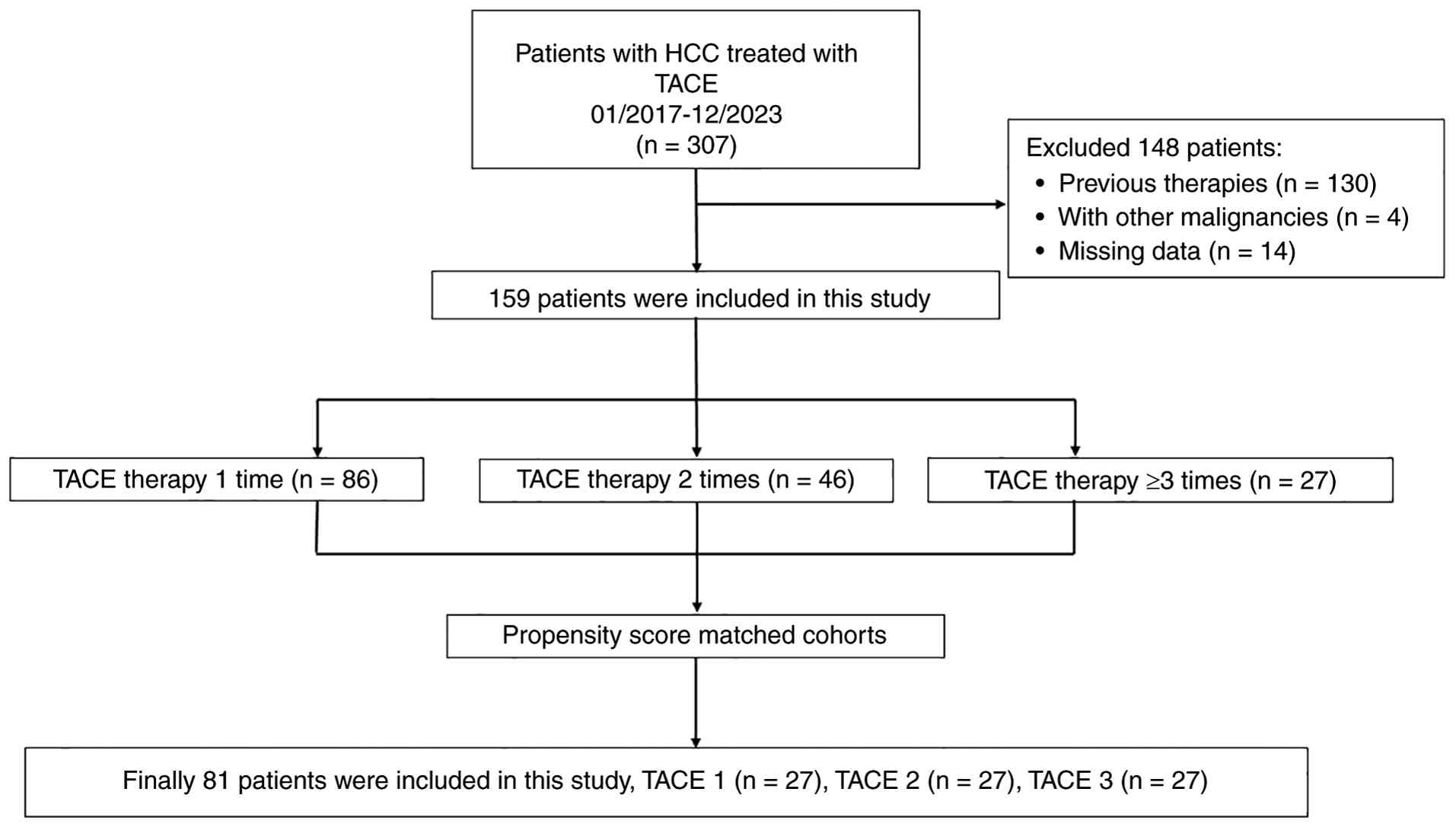
A total of 307 patients who underwent TACE for
primary HCC during the study period were initially screened
(Fig. 1). Based on the inclusion
and exclusion criteria, 159 patients were enrolled, including 86 in
the TACE 1 group, 46 in the TACE 2 group and 27 in the TACE ≥3
group. Before PSM, there was a significant difference among the
three groups in the presence of portal vein tumor thrombus (all
P<0.05), whereas no significant differences were observed for
the remaining variables (P>0.05; Table I). Through a sequential pairwise
matching process, 27 patients were successfully identified in each
group, resulting in well-balanced baseline characteristics across
all three groups (Table II). The
absence of significant differences in all baseline covariates after
matching (all P>0.05) confirmed that the three groups were well
balanced, thereby supporting the validity of subsequent
comparisons.
 | Table I.Patient demographics and baseline
characteristics before propensity score matching in the TACE 1
(n=86), TACE 2 (n=46) and TACE 3 (n=27) groups. |
Table I.
Patient demographics and baseline
characteristics before propensity score matching in the TACE 1
(n=86), TACE 2 (n=46) and TACE 3 (n=27) groups.
| Characteristic | TACE 1, n (%) | TACE 2, n (%) | TACE 3, n (%) |
χ2/Fisher's statistic | P-value |
|---|
| Sex |
|
|
| 1.316 | 0.518 |
|
Female | 17 (19.77) | 9 (19.57) | 8 (29.63) |
|
|
|
Male | 69 (80.23) | 37 (80.43) | 19 (70.37) |
|
|
| Age, years |
|
|
| 0.041 | 0.980 |
|
<60 | 31 (36.05) | 16 (34.78) | 10 (37.04) |
|
|
|
≥60 | 55 (63.95) | 30 (65.22) | 17 (62.96) |
|
|
| BMI,
kg/m2 |
|
|
| 1.154 | 0.562 |
|
<24 | 47 (54.65) | 23 (50.00) | 17 (62.96) |
|
|
|
≥24 | 39 (45.35) | 23 (50.00) | 10 (37.04) |
|
|
| Child-Pugh |
|
|
| 0.683 | 0.711 |
| A | 57 (66.28) | 33 (71.74) | 17 (62.96) |
|
|
| B | 29 (33.72) | 13 (28.26) | 10 (37.04) |
|
|
| HBV |
|
|
| 0.740 | 0.691 |
| No | 26 (39.53) | 12 (26.09) | 6 (22.22) |
|
|
|
Yes | 60 (60.47) | 34 (73.91) | 21 (77.78) |
|
|
| HCV |
|
|
| 4.192 | 0.123 |
| No | 74 (86.05) | 41 (89.13) | 27 (100.00) |
|
|
|
Yes | 12 (13.95) | 5 (10.87) | 0 (0.00) |
|
|
| Hypertension |
|
|
| 0.706 | 0.703 |
| No | 54 (62.79) | 28 (60.87) | 19 (70.37) |
|
|
|
Yes | 32 (37.21) | 18 (39.13) | 8 (29.63) |
|
|
| Diabetes |
|
|
| 0.262 | 0.877 |
| No | 69 (80.23) | 38 (82.61) | 21 (77.78) |
|
|
|
Yes | 17 (19.77) | 8 (17.39) | 6 (22.22) |
|
|
| AFP, ng/ml |
|
|
| 0.103 | 0.950 |
|
<400 | 51 (59.30) | 26 (56.52) | 16 (59.26) |
|
|
|
≥400 | 35 (40.70) | 20 (43.48) | 11 (40.74) |
|
|
| CEA, ng/ml |
|
|
| 0.952 | 0.621 |
|
<5 | 69 (80.23) | 40 (86.96) | 22 (81.48) |
|
|
| ≥5 | 17 (19.77) | 6 (13.04) | 5 (18.52) |
|
|
| CA199, U/ml |
|
|
| 0.693 | 0.707 |
|
<37 | 38 (44.19) | 23 (50.00) | 14 (51.85) |
|
|
|
≥37 | 48 (55.81) | 23 (50.00) | 13 (48.15) |
|
|
| Albumin, g/l |
|
|
| 1.235 | 0.539 |
|
<35 | 39 (45.35) | 19 (41.30) | 9 (33.33) |
|
|
|
≥35 | 47 (54.65) | 27 (58.70) | 18 (66.67) |
|
|
| ALT, U/l |
|
|
| 2.394 | 0.302 |
|
<40 | 46 (53.49) | 31 (67.39) | 16 (59.26) |
|
|
|
≥40 | 40 (46.51) | 15 (32.61) | 11 (40.74) |
|
|
| AST, U/l |
|
|
| 0.480 | 0.787 |
|
<40 | 25 (29.07) | 16 (34.78) | 8 (29.63) |
|
|
|
≥40 | 61 (70.93) | 30 (65.22) | 19 (70.37) |
|
|
| WBC
(×109/l) |
|
|
| 2.954 | 0.228 |
|
<10 | 77 (89.53) | 45 (97.83) | 25 (92.59) |
|
|
|
≥10 | 9 (10.47) | 1 (2.17) | 2 (7.41) |
|
|
| NLR |
|
|
| 4.247 | 0.120 |
|
<1.82 | 15 (17.44) | 15 (32.61) | 5 (18.52) |
|
|
|
≥1.82 | 71 (82.56) | 31 (67.39) | 22 (81.48) |
|
|
| PLT
(×109/l) |
|
|
| 2.474 | 0.290 |
|
<100 | 20 (23.26) | 10 (21.74) | 10 (37.04) |
|
|
|
≥100 | 66 (76.74) | 36 (78.26) | 17 (62.96) |
|
|
| Cirrhosis |
|
|
| 1.704 | 0.427 |
| No | 40 (46.51) | 18 (39.13) | 9 (33.33) |
|
|
|
Yes | 46 (53.49) | 28 (60.87) | 18 (66.67) |
|
|
| Tumor number |
|
|
| 1.133 | 0.567 |
|
Single | 33 (38.37) | 21 (45.65) | 13 (48.15) |
|
|
|
Multiple | 53 (61.63) | 25 (54.35) | 14 (51.85) |
|
|
| Portal vein tumor
thrombus |
|
|
| 7.700 | 0.021 |
| No | 52 (60.47) | 32 (69.57) | 24 (88.89) |
|
|
|
Yes | 34 (39.53) | 14 (30.43) | 3 (11.11) |
|
|
| Extrahepatic
metastasis |
|
|
| 2.031 | 0.362 |
| No | 64 (74.42) | 38 (82.61) | 23 (85.19) |
|
|
|
Yes | 22 (25.58) | 8 (17.39) | 4 (14.81) |
|
|
| Maximum tumor
diameter, cm |
|
|
| 0.056 | 0.973 |
|
<5 | 23 (26.74) | 13 (28.26) | 7 (25.93) |
|
|
| ≥5 | 63 (73.26) | 33 (71.74) | 20 (74.07) |
|
|
| BCLC stage |
|
|
| 4.385 | 0.356 |
| A | 20 (23.26) | 13 (28.26) | 10 (37.04) |
|
|
| B | 21 (24.42) | 12 (26.09) | 9 (33.33) |
|
|
| C | 45 (52.33) | 21 (45.65) | 8 (29.63) |
|
|
 | Table II.Patient demographics and baseline
characteristics after propensity score matching in the TACE 1
(n=27), TACE 2 (n=27) and TACE 3 (n=27) groups. |
Table II.
Patient demographics and baseline
characteristics after propensity score matching in the TACE 1
(n=27), TACE 2 (n=27) and TACE 3 (n=27) groups.
| Characteristic | TACE 1, n (%) | TACE 2, n (%) | TACE 3, n (%) |
χ2/Fisher's statistic | P-value |
|---|
| Sex |
|
|
| 1.714 | 0.424 |
|
Female | 4 (14.81) | 6 (22.22) | 8 (29.63) |
|
|
|
Male | 23 (85.19) | 21 (77.78) | 19 (70.37) |
|
|
| Age, years |
|
|
| 0.453 | 0.797 |
|
<60 | 8 (29.63) | 8 (29.63) | 10 (37.04) |
|
|
|
≥60 | 19 (70.37) | 19 (70.37) | 17 (62.96) |
|
|
| BMI,
kg/m2 |
|
|
| 1.884 | 0.390 |
|
<24 | 12 (44.44) | 14 (51.85) | 17 (62.96) |
|
|
|
≥24 | 15 (55.56) | 13 (48.15) | 10 (37.04) |
|
|
| Child-Pugh |
|
|
| 2.371 | 0.306 |
| A | 20 (74.07) | 22 (81.48) | 17 (62.96) |
|
|
| B | 7 (25.93) | 5 (18.52) | 10 (37.04) |
|
|
| HBV |
|
|
| 0.963 | 0.618 |
| No | 5 (18.52) | 8 (29.63) | 6 (22.22) |
|
|
|
Yes | 22 (81.48) | 19 (70.37) | 21 (77.78) |
|
|
| HCV |
|
|
| 5.271 | 0.072 |
| No | 22 (81.48) | 24 (88.89) | 27 (100.00) |
|
|
|
Yes | 5 (18.52) | 3 (11.11) | 0 (0.00) |
|
|
| Hypertension |
|
|
| 0.752 | 0.687 |
| No | 16 (59.26) | 17 (62.96) | 19 (70.37) |
|
|
|
Yes | 11 (40.74) | 10 (37.04) | 8 (29.63) |
|
|
| Diabetes |
|
|
| <0.001 | >0.999 |
| No | 21 (77.78) | 21 (77.78) | 21 (77.78) |
|
|
|
Yes | 6 (22.22) | 6 (22.22) | 6 (22.22) |
|
|
| AFP, ng/ml |
|
|
| <0.001 | >0.999 |
|
<400 | 16 (59.26) | 16 (59.26) | 16 (59.26) |
|
|
|
≥400 | 11 (40.74) | 11 (40.74) | 11 (40.74) |
|
|
| CEA, ng/ml |
|
|
| 2.382 | 0.304 |
|
<5 | 21 (77.78) | 25 (92.59) | 22 (81.48) |
|
|
| ≥5 | 6 (22.22) | 2 (7.41) | 5 (18.52) |
|
|
| CA199, U/ml |
|
|
| 0.100 | 0.951 |
|
<37 | 15 (55.56) | 15 (55.56) | 14 (51.85) |
|
|
|
≥37 | 12 (44.44) | 12 (44.44) | 13 (48.15) |
|
|
| Albumin, g/l |
|
|
| 0.418 | 0.811 |
|
<35 | 11 (40.74) | 11 (40.74) | 9 (33.33) |
|
|
|
≥35 | 16 (59.26) | 16 (59.26) | 18 (66.67) |
|
|
| ALT, U/l (%) |
|
|
| 0.723 | 0.697 |
|
<40 | 15 (55.56) | 18 (66.67) | 16 (59.26) |
|
|
|
≥40 | 12 (44.44) | 9 (33.33) | 11 (40.74) |
|
|
| AST, U/l |
|
|
| 0.437 | 0.804 |
|
<40 | 10 (37.04) | 10 (37.04) | 8 (29.63) |
|
|
|
≥40 | 17 (62.96) | 17 (62.96) | 19 (70.37) |
|
|
| WBC
(×109/l) |
|
|
| 0.426 | 0.808 |
|
<10 | 25 (92.59) | 26 (96.30) | 25 (92.59) |
|
|
|
≥10 | 2 (7.41) | 1 (3.70) | 2 (7.41) |
|
|
| NLR |
|
|
| 5.222 | 0.073 |
|
<1.82 | 6 (22.22) | 12 (44.44) | 5 (18.52) |
|
|
|
≥1.82 | 21 (77.78) | 15 (55.56) | 22 (81.48) |
|
|
| PLT
(×109/l) |
|
|
| 1.997 | 0.368 |
|
<100 | 6 (22.22) | 6 (22.22) | 10 (37.04) |
|
|
|
≥100 | 21 (77.78) | 21 (77.78) | 17 (62.96) |
|
|
| Cirrhosis |
|
|
| 0.418 | 0.811 |
| No | 11 (40.74) | 11 (40.74) | 9 (33.33) |
|
|
|
Yes | 16 (59.26) | 16 (59.26) | 18 (66.67) |
|
|
| Tumor number |
|
|
| 0.297 | 0.862 |
|
Single | 12 (44.44) | 14 (51.85) | 13 (48.15) |
|
|
|
Multiple | 15 (55.56) | 13 (48.15) | 14 (51.85) |
|
|
| Portal vein tumor
thrombus |
|
|
| 5.951 | 0.051 |
| No | 17 (62.96) | 17 (62.96) | 24 (88.89) |
|
|
|
Yes | 10 (37.04) | 10 (37.04) | 3 (11.11) |
|
|
| Extrahepatic
metastasis |
|
|
| 1.942 | 0.379 |
| No | 24 (88.89) | 26 (96.30) | 23 (85.19) |
|
|
|
Yes | 3 (11.11) | 1 (3.70) | 4 (14.81) |
|
|
| Maximum tumor
diameter, cm |
|
|
| 0.793 | 0.673 |
|
<5 | 9 (33.33) | 10 (37.04) | 7 (25.93) |
|
|
| ≥5 | 18 (66.67) | 17 (62.96) | 20 (74.07) |
|
|
| BCLC stage |
|
|
| 1.404 | 0.843 |
| A | 11 (40.74) | 10 (37.04) | 10 (37.04) |
|
|
| B | 6 (22.22) | 6 (22.22) | 9 (33.33) |
|
|
| C | 10 (37.04) | 11 (40.74) | 8 (29.63) |
|
|
Procedural characteristics
To account for procedural heterogeneity, the
specific embolic agents and chemotherapeutic regimens used in each
TACE session were documented. The distribution of embolic agents
was comparable across groups: Lipiodol alone (42%), lipiodol +
gelatin sponge (38%) and drug-eluting microspheres (20%), with no
significant intergroup differences (P=0.45). Chemotherapeutic
regimens included epirubicin + cisplatin + fluorouracil (55%),
epirubicin alone (25%) and other combinations (20%), with no
significant differences between groups (P=0.52). The choice of
regimen and dosage was at the discretion of the operator, guided by
tumor characteristics, liver function and patient tolerance,
reflecting real-world clinical practice.
Treatment characteristics
In the TACE ≥3 group, the number of sessions ranged
from 3 to 6, with a median of 3 [interquartile range (IQR): 3–4).
The median interval between the first and second TACE sessions was
6.2 weeks (IQR: 5.1–8.4), and between the second and third sessions
was 7.8 weeks (IQR: 6.3–10.2). These intervals did not differ
significantly between the TACE 2 and ≥3 groups for the first two
sessions (P=0.42), indicating comparable initial treatment
intensity.
Efficacy
According to mRECIST criteria, one patient in the
TACE 1 group achieved CR and seven achieved PR. In the TACE 2
group, eight patients achieved CR and 13 achieved PR, while in the
TACE 3 group, seven achieved CR and five achieved PR (Table III, Fig. 2). The ORR in the TACE 1, TACE 2, and
TACE 3 groups was 29.6, 77.8 and 44.4%, respectively (P=0.001). The
DCR in the TACE 1, TACE 2, and TACE 3 groups was 66.7, 88.9 and
96.3%, respectively (P=0.016). The differences in ORR and DCR among
the three groups were significant (both P<0.05; Table III, Fig. 2). Pairwise comparisons revealed
significant differences in both ORR and DCR between the TACE 1 and
2 groups (Table III; Fig. 2). The difference in DCR between the
TACE 1 and 3 groups was significant (P≤0.05), whereas the
difference in ORR was not (P>0.05). Conversely, the difference
in ORR between the TACE 2 and 3 groups was significant (P=0.012),
while the difference in DCR was not (P=0.61; Fig. 3).
 | Table III.Therapeutic efficacy. |
Table III.
Therapeutic efficacy.
| Variable (%) | TACE 1 (n=27) | TACE 2 (n=27) | TACE 3 (n=27) | P-value |
|---|
| CR | 1 (3.7) | 8 (29.6) | 7 (25.9) | 0.027 |
| PR | 7 (25.9) | 13 (48.1) | 5 (18.5) | 0.067 |
| SD | 10 (37.0) | 3 (11.1) | 14 (51.9) | 0.002 |
| PD | 9 (33.3) | 3 (11.1) | 1 (3.7) | 0.003 |
| Objective response
rate | 8 (29.6) | 21 (77.8) | 12 (44.4) | 0.001 |
| Disease control
rate | 18 (66.7) | 24 (88.9) | 26 (96.3) | 0.016 |
Safety analysis
Treatment-related AEs occurred in 60/81 patients
(74.1%). The majority were grade 1–2 in severity and were
manageable with symptomatic treatment. No treatment-related deaths
were observed. The incidence of AEs was 19 patients (70.4%) in the
TACE 1 group, 23 (85.2%) in the TACE 2 group and 18 (66.7%) in the
TACE 3 group, with no significant difference among groups
(P=0.259). Furthermore, no significant differences were observed in
the frequencies of specific AEs between groups (Table IV).
 | Table IV.Treatment emergent adverse
events. |
Table IV.
Treatment emergent adverse
events.
| Adverse events
(%) | TACE 1 (n=27) | TACE 2 (n=27) | TACE 3 (n=27) | P-value |
|---|
| Total | 19 (70.4) | 23 (85.2) | 18 (66.7) | 0.259 |
| Fever | 8 (29.6) | 7 (25.9) | 8 (29.6) | 0.941 |
| Abdominal pain | 9 (33.3) | 12 (44.4) | 9 (33.3) | 0.621 |
| Vomiting or
nausea | 1 (3.7) | 7 (25.9) | 2 (7.4) | 0.068 |
| Elevated ALT or
AST | 3 (11.1) | 4 (14.8) | 0 (0.0) | 0.152 |
| Decreased
appetite | 2 (7.4) | 7 (25.9) | 2 (7.4) | 0.097 |
| Rash | 0 (0.0) | 3 (11.1) | 0 (0.0) | 0.103 |
| Hypertension | 0 (0.0) | 0 (0.0) | 1 (3.7) | >0.999 |
| Hand-foot
syndrome | 0 (0.0) | 3 (11.1) | 0 (0.0) | 0.103 |
| Diarrhea | 0 (0.0) | 2 (7.4) | 0 (0.0) | 0.325 |
|
Hypoalbuminemia | 1 (3.7) | 0 (0.0) | 0 (0.0) | >0.999 |
| Hypothyroidism | 0 (0.0) | 0 (0.0) | 2 (7.4) | 0.325 |
| WBC decrease | 1 (3.7) | 0 (0.0) | 1 (3.7) | >0.999 |
Survival analysis
Follow-up duration was calculated from the date of
the first TACE to the date of death or last follow-up (October
2024). Adherence to the follow-up schedule was high: 91% of
patients underwent imaging at 3 months, 84% at 6 months and 76% at
12 months. The frequency of imaging/unit time was similar across
groups (every 3.2–3.8 months), indicating comparable follow-up
intensity. The median number of follow-up imaging assessments was
four (IQR: 2–7) in the TACE 1 group, six (IQR: 4–9) in the TACE 2
group and seven (IQR: 5–10) in the TACE 3 group, reflecting the
longer total follow-up duration in the multi-session groups rather
than a difference in follow-up intensity. The comparable frequency
of assessments/unit time across groups suggested that surveillance
bias was unlikely to account for the observed differences in
outcomes (data not shown). The median overall follow-up duration
for the entire cohort was 20 months (95% CI: 16.1–23.9). The TACE 3
group exhibited the longest median OS (26 months; 95% CI:
14.6–37.4), followed by the TACE 2 group (21 months; 95% CI:
16.4–25.6) and the TACE 1 group (8 months; 95% CI: 0–18.2;
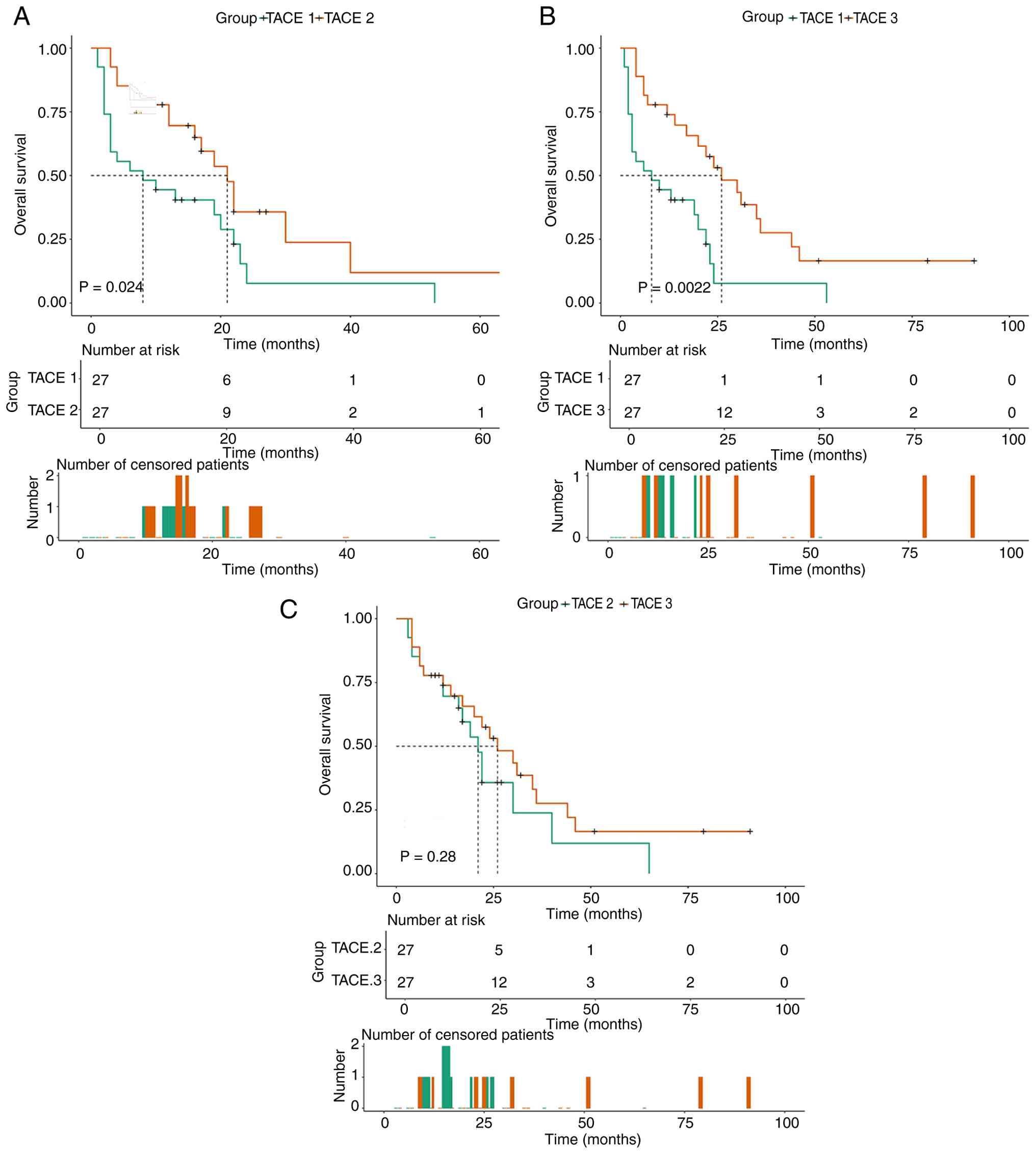
P=0.0028; Fig. 4). Compared with
the TACE 1 group, OS was significantly prolonged in both the TACE 2
and 3 groups (P<0.05; Fig. 5A and
B), whereas no significant difference was observed between the
TACE 2 and 3 groups (Fig. 5C).
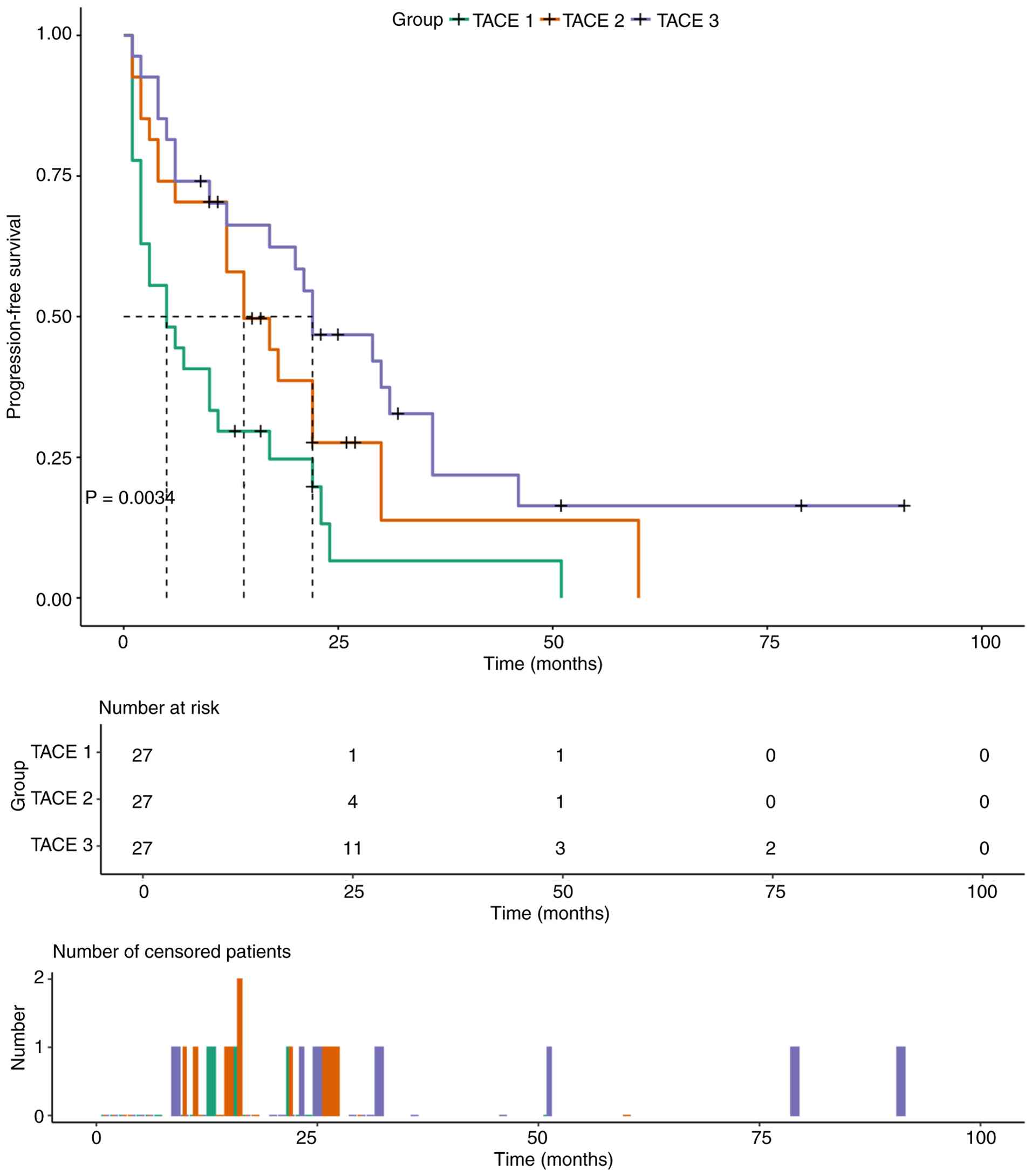
Similarly, median PFS was longest in the TACE 3 group (22 months;
95% CI: 11.5–32.5), followed by the TACE 2 group (14 months; 95%
CI: 6.9–21.1) and shortest in the TACE 1 group (5 months; 95% CI:
0.0–10.1; P=0.0034; Fig. 6).
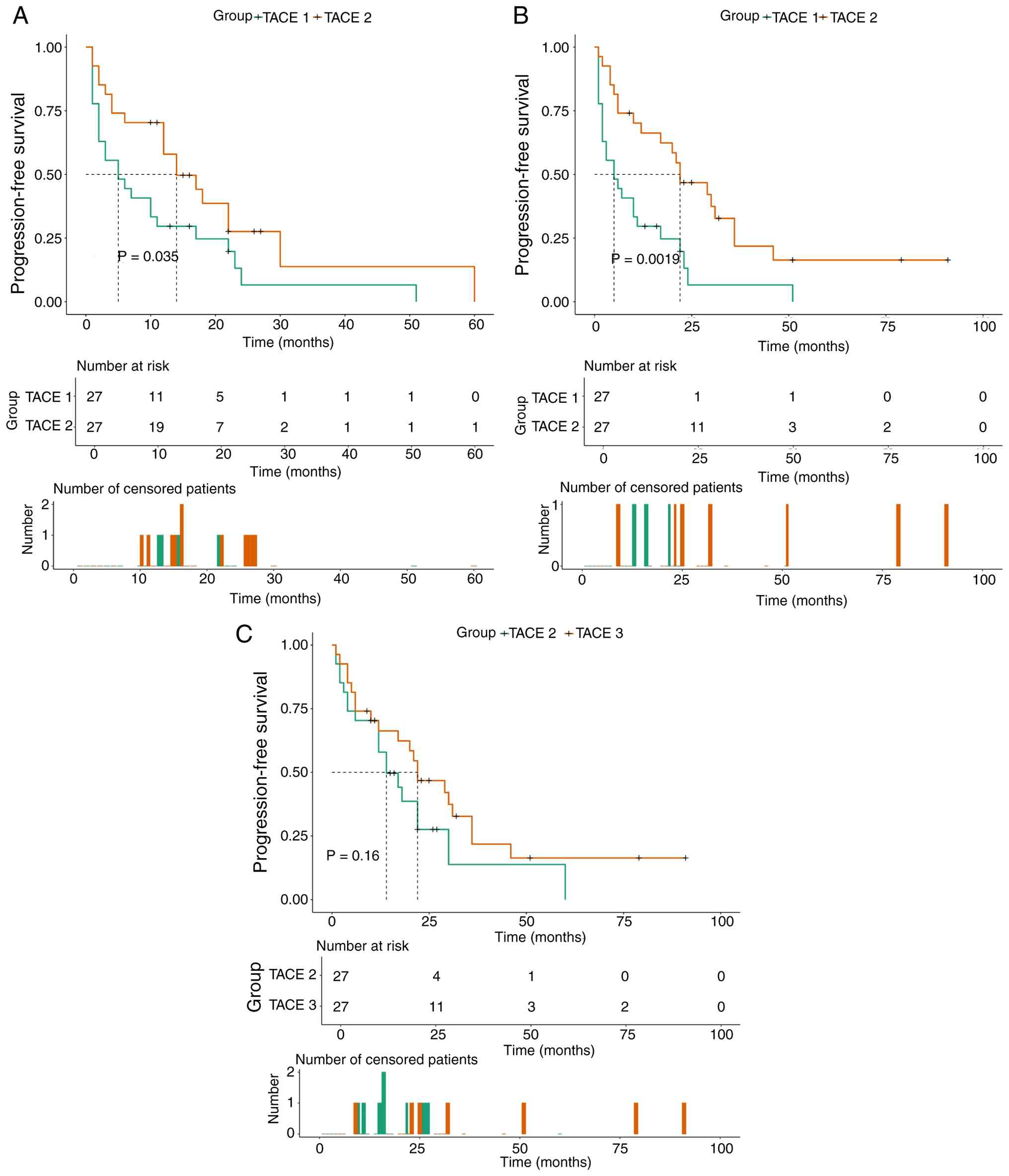
Pairwise comparisons revealed significant differences in PFS
between TACE 1 and 2 (P=0.035; Fig.
7A) and between TACE 1 and 3 (P=0.0019; Fig. 7B), but not between TACE 2 and 3
(P=0.16; Fig. 7C). In summary, both
the TACE 2 and 3 groups achieved significantly longer PFS and OS
compared with the TACE 1 group, however, no significant difference
was observed between the TACE 2 and 3 groups. Notably, the survival
benefit in the TACE 2 and 3 groups became evident within the first
6 months of follow-up (Fig. 4,
Fig. 5, Fig. 6, Fig.
7) and persisted throughout the observation period. The early
divergence of the survival curves, before notable immortal time
could accumulate, supports the interpretation that the benefits
reflect genuine treatment effects rather than bias.
Risk factor analysis
ROC curve analysis was performed to determine the
optimal cutoff value for NLR. The area under the curve was 0.673,
and the optimal cutoff value was 1.82 (P=0.003; Fig. 8). Uni- and multivariate Cox
regression analyses were performed to identify factors associated
with PFS and OS. In the matched cohort, univariate analysis
revealed that hypertension, CA199 levels, portal vein tumor
thrombus, number of TACE sessions, AEs, targeted
therapy/immunotherapy and BCLC stage were significantly associated
with PFS (all P<0.05) (Table V;
Fig. 9). Among the 81 patients
following PSM, 38 (46.9%) received targeted therapy and/or
immunotherapy during follow-up (Table
II). Of these, eight patients (21.1%) received systemic therapy
between TACE sessions (typically following documented progression
following the first TACE), whereas 30 (78.9%) initiated systemic
therapy after completing all TACE sessions. The median time from
the first TACE to initiation of systemic therapy was 9.2 months
(IQR: 5.8–14.3) (data not shown). Sensitivity analysis excluding
the eight patients who received systemic therapy between TACE
sessions yielded similar results for the association between the
number of TACE sessions and survival outcomes (data not shown),
suggesting minimal confounding by the timing of systemic therapy.
Multivariate analysis identified hypertension, number of TACE
sessions, adverse reactions, targeted therapy/immunotherapy and
BCLC stage as independent prognostic factors for PFS (Table V; Fig.
9). Univariate analysis showed that hypertension, CA199 levels,
NLR, portal vein tumor thrombus, maximal tumor size, number of TACE
sessions, adverse reactions and BCLC stage were associated with OS
(all P<0.05) (Table VI;
Fig. 10). Multivariate analysis
revealed that CA199 levels, number of TACE sessions, adverse
reactions and BCLC stage were independent prognostic factors for OS
(Table VI; Fig. 10). No significant violations were
detected for any covariate in the final models (P=0.38 for OS and
P=0.31 for PFS). Visual inspection of log-minus-log survival plots
confirmed parallel curves for categorical variables, supporting the
validity of the Cox proportional hazards models (data not
shown).
 | Figure 9.Forest plots of univariate and
multivariate predictors of progression-free survival. *P<0.05,
**P<0.01, ***P<0.001 vs. the reference category. BMI, body
mass index; HBV, hepatitis B virus; HCV, hepatitis C virus; AFP,
α-fetoprotein; CEA, carcinoembryonic antigen; ALT, alanine
aminotransferase; AST, aspartate aminotransferase; WBC, white blood
cell; NLR, neutrophil-to-lymphocyte ratio; PLT, platelet; TACE,
transcatheter arterial chemoembolization; BCLC, Barcelona Clinic
Liver Cancer. |
 | Figure 10.Forest plots of univariate and
multivariate predictors of overall survival. *P<0.05,
**P<0.01, ***P<0.001 vs. the reference category. BMI, body
mass index; HBV, hepatitis B virus; HCV, hepatitis C virus; AFP,
α-fetoprotein; CEA, carcinoembryonic antigen; ALT, alanine
aminotransferase; AST, aspartate aminotransferase; WBC, white blood
cell; NLR, neutrophil-to-lymphocyte ratio; PLT, platelet; TACE,
transcatheter arterial chemoembolization; BCLC, Barcelona Clinic
Liver Cancer. |
 | Table V.Univariate and multivariate
predictors of progression-free survival. |
Table V.
Univariate and multivariate
predictors of progression-free survival.
|
| Univariate Cox
analysis | Multivariate Cox
analysis |
|---|
|
|
|
|
|---|
| Variable | HR | 95% CI | P-value | HR | 95% CI | P-value |
|---|
| Sex
(male/female) | 0.960 | 0.712–1.294 | 0.787 |
|
|
|
| Age (<60/≥60
years) | 0.945 | 0.722–1.238 | 0.683 |
|
|
|
| BMI (<24/≥24
kg/m2) | 1.515 | 0.901–2.548 | 0.117 |
|
|
|
| Child-Pugh class
(A/B) | 0.967 | 0.736–1.269 | 0.808 |
|
|
|
| HBV (yes/no) | 0.845 | 0.477–1.499 | 0.565 |
|
|
|
| HCV (yes/no) | 1.559 | 0.700–3.471 | 0.277 |
|
|
|
| Hypertension
(yes/no) | 2.515 | 1.473–4.296 | 0.001 | 1.933 | 1.107~3.376 | 0.020 |
| Diabetes
(yes/no) | 1.289 | 0.705–2.357 | 0.409 |
|
|
|
| AFP (<400/≥400
ng/ml) | 1.273 | 0.772–2.100 | 0.344 |
|
|
|
| CEA (<5/≥5
ng/ml) | 1.504 | 0.779–2.906 | 0.224 |
|
|
|
| CA199 (<37/≥37
U/ml) | 1.703 | 1.007–2.880 | 0.047 | 1.410 | 0.802–2.481 | 0.233 |
| Albumin (<35/≥35
g/l) | 0.920 | 0.547–1.547 | 0.753 |
|
|
|
| ALT (<40/≥40
U/l) | 0.725 | 0.430–1.222 | 0.227 |
|
|
|
| AST (<40/≥40
U/l) | 0.994 | 0.586–1.688 | 0.983 |
|
|
|
| WBC
(<10/≥10×109/l) | 0.963 | 0.346–2.684 | 0.943 |
|
|
|
| NLR
(<1.82/≥1.82) | 1.599 | 0.880–2.907 | 0.124 |
|
|
|
| PLT
(<100/≥100×109/l) | 1.624 | 0.907–2.908 | 0.103 |
|
|
|
| Cirrhosis
(yes/no) | 0.805 | 0.487–1.330 | 0.396 |
|
|
|
| Tumor number
(single/multiple) | 1.243 | 0.757–2.042 | 0.390 |
|
|
|
| Portal vein tumor
thrombus (yes/no) | 2.499 | 1.446–4.320 | 0.001 | 1.150 | 0.581–2.277 | 0.687 |
| Extrahepatic
metastasis (yes/no) | 1.842 | 0.830–4.091 | 0.133 |
|
|
|
| Maximal tumor size
(<5/≥5 cm) | 1.716 | 0.966–3.049 | 0.066 |
|
|
|
| Number of TACE | 0.728 | 0.589–0.899 | 0.003 | 0.679 | 0.523–0.880 | 0.003 |
| Adverse reaction
(yes/no) | 2.119 | 1.113–4.037 | 0.022 | 2.588 | 1.301–5.147 | 0.007 |
| Targeted therapy
and immunotherapy (yes/no) | 0.593 | 0.354–0.993 | 0.047 | 0.370 | 0.204–0.674 | 0.001 |
| BCLC (A + B/C) | 2.342 | 1.364–4.022 | 0.002 | 3.833 | 1.841–7.982 | <0.001 |
 | Table VI.Uni- and multivariate predictors of
overall survival. |
Table VI.
Uni- and multivariate predictors of
overall survival.
|
| Univariate Cox
analysis | Multivariate Cox
analysis |
|---|
|
|
|
|
|---|
| Variables | HR | 95% CI | P-value | HR | 95% CI | P-value |
|---|
| Sex
(male/female) | 1.017 | 0.751–1.376 | 0.914 |
|
|
|
| Age (<60/≥60
years) | 1.034 | 0.786–1.362 | 0.809 |
|
|
|
| BMI (<24/≥24
kg/m2) | 1.425 | 0.830–2.445 | 0.199 |
|
|
|
| Child-Pugh class
(A/B) | 0.943 | 0.711–1.250 | 0.682 |
|
|
|
| HBV (yes/no) | 0.883 | 0.481–1.623 | 0.689 |
|
|
|
| HCV (yes/no) | 1.678 | 0.748–3.762 | 0.209 |
|
|
|
| Hypertension
(yes/no) | 1.910 | 1.092–3.340 | 0.023 | 1.424 | 0.791–2.561 | 0.239 |
| Diabetes
(yes/no) | 1.164 | 0.610–2.223 | 0.644 |
|
|
|
| AFP (<400/≥400
ng/ml) | 1.174 | 0.696–1.978 | 0.548 |
|
|
|
| CEA (<5/≥5
ng/ml) | 1.655 | 0.848–3.228 | 0.139 |
|
|
|
| CA199 (<37/≥37
U/ml) | 2.259 | 1.282–3.981 | 0.005 | 2.211 | 1.211–4.037 | 0.010 |
| Albumin (<35/≥35
g/l) | 0.802 | 0.468–1.374 | 0.422 |
|
|
|
| ALT (<40/≥40
U/l) | 0.897 | 0.524–1.534 | 0.690 |
|
|
|
| AST (<40/≥40
U/l) | 1.394 | 0.787–2.467 | 0.254 |
|
|
|
| WBC
(<10/≥10×109/l) | 1.116 | 0.400–3.114 | 0.834 |
|
|
|
| NLR
(<1.82/≥1.82) | 1.946 | 1.026–3.690 | 0.042 | 1.572 | 0.803–3.078 | 0.187 |
| PLT
(<100/≥100×109/l) | 1.421 | 0.786–2.569 | 0.245 |
|
|
|
| Cirrhosis
(yes/no) | 0.754 | 0.447–1.275 | 0.292 |
|
|
|
| Tumor number
(single/multiple) | 1.307 | 0.778–2.196 | 0.311 |
|
|
|
| Portal vein tumor
thrombus (yes/no) | 2.854 | 1.595–5.104 | <0.001 | 1.049 | 0.524–2.099 | 0.894 |
| Extrahepatic
metastasis (yes/no) | 1.833 | 0.775–4.335 | 0.167 |
|
|
|
| Maximal tumor size
(<5/≥5 cm) | 2.027 | 1.098–3.740 | 0.024 | 1.147 | 0.560–2.347 | 0.708 |
| Number of TACE | 0.729 | 0.585–0.908 | 0.005 | 0.628 | 0.477–0.826 | 0.001 |
| Adverse reaction
(yes/no) | 2.264 | 1.132–4.527 | 0.021 | 2.209 | 1.063–4.593 | 0.034 |
| Targeted therapy
and immunotherapy (yes/no) | 0.654 | 0.383–1.117 | 0.120 |
|
|
|
| BCLC (A + B/C) | 2.343 | 1.337–4.104 | 0.003 | 3.161 | 1.497–6.673 | 0.003 |
Discussion
By contrast with normal liver tissue, which receives
~75% of its blood supply from the portal vein and 25% from the
hepatic artery, HCC derives 85–90% of its blood supply from the
hepatic artery. This unique vascularization forms the basis for
TACE, which achieves its antitumor effect by occluding the tumor
main feeding artery (25).
According to the BCLC staging system, first-line treatment options
for intermediate and advanced HCC include TACE, transarterial
radioembolization and systemic therapy (17). Among these, TACE is the most
commonly used and preferred treatment for uHCC, offering the
advantages of decreasing tumor burden and alleviating symptoms
(17).
In China, TACE is primarily employed as a palliative
treatment for HCC (11,26). In particular, it provides a key
therapeutic option for patients with intermediate or advanced HCC
who are not candidates for surgical resection or local ablation.
TACE can achieve curative outcomes in selected patients with HCC
with small tumor burden, a relatively simple blood supply and
feasible superselective catheterization (27,28).
The clinical practice of administering multiple TACE
sessions is based on an on-demand approach (17). However, the association between the
number of TACE sessions and long-term patient prognosis has become
a notable focus of clinical research (29).
In the present study, a significant association was
observed between the number of TACE sessions and treatment efficacy
in patients with primary liver cancer. Specifically, the short-term
efficacy of a single TACE session was inferior to that of multiple
sessions. However, increasing the number of sessions beyond two did
not further improve outcomes; ORR was lower in patients receiving
≥3 sessions compared with those receiving two sessions. This may be
attributable to the multifaceted effects of TACE, which exerts its
therapeutic action through the intra-arterial infusion of
chemotherapeutic agents and embolic materials into the
tumor-feeding hepatic artery (17).
In this process, chemotherapeutic drugs directly target tumor
cells, while embolic agents occlude the tumor vascular supply,
inducing ischemia and hypoxia within the tumor tissue and leading
to necrosis. However, repeated TACE sessions may also damage normal
liver parenchyma, particularly when the number of sessions is high,
potentially exacerbating liver function impairment (17). Multiple TACE sessions may induce
alterations in the peritumoral microenvironment, including
hypoxia-induced angiogenesis, upregulation of pro-angiogenic
factors such as vascular endothelial growth factor, recruitment of
immunosuppressive cells (for example, myeloid-derived suppressor
cells and tumor-associated macrophages) and epithelial-mesenchymal
transition, potentially promoting tumor recurrence and metastasis
(30,31). Furthermore, repeated TACE may lead
to a decline in hepatic functional reserve, adversely affecting
patient overall health and quality of life (32). In the present study, two measures of
tumor response were assessed: Response at the first assessment,
which enables unbiased comparison with uniform follow-up duration
across groups, and cumulative best response over the entire
follow-up period, which reflects the maximum response achieved at
any time. The latter measure is clinically meaningful, as it
captures the benefit of additional treatments in patients who
respond to subsequent TACE sessions; however, it may be influenced
by differences in follow-up duration.
Based on previous studies (15,32),
following ≥3 consecutive standardized TACE sessions, patients
underwent contrast-enhanced CT/MRI within 1–3 months of the last
procedure to assess treatment response using mRECIST criteria. If
the intrahepatic target lesion shows progressive disease compared
with the pre-treatment status, TACE failure/refractoriness is
considered to have occurred, which negatively impacts patient
outcomes (15).
The most common complication following TACE is PES,
characterized by hepatic pain, nausea, vomiting and fever. Some
studies (33–35) have reported that PES may increase
the risk of mortality in patients with primary liver cancer by
nearly twofold and may serve as an early predictor of long-term
survival. The incidence and severity of AEs in the present study
were consistent with those reported in the literature (17,34).
Grade 1–2 AEs were predominant in all three groups and generally
well tolerated. Symptoms gradually resolved with appropriate
supportive care and no treatment-related deaths occurred. The
incidence of adverse reactions did not differ significantly between
the groups, which may be attributable to the limited sample size
and warrants validation in larger studies. The present study has
several limitations. First, as a single-center retrospective study,
it is susceptible to inherent selection bias despite the
application of PSM. The treatment protocols, patient selection
criteria and follow-up practices reflect the experience of a single
institution, potentially limiting the generalizability of findings
to other settings with different patient populations or treatment
approaches. Second, the sample size after PSM (81 patients) was
modest, which may have limited the statistical power to detect
smaller differences between the TACE 2 and 3 groups, a comparison
that showed no significant differences in PFS (P=0.16) or OS
(P=0.34) despite both groups outperforming the TACE 1 group, and to
conduct more extensive subgroup analyses. Third, the observational
design precluded definitive causal inferences regarding the optimal
number of TACE sessions. Fourth, the heterogeneity in TACE
techniques, embolic agents and chemotherapeutic regimens, although
reflective of real-world practice, introduces potential confounding
factors. Fifth, the follow-up duration, while adequate for
assessing early-to-intermediate outcomes, may not capture long-term
survival beyond 3–5 years. To address these limitations, future
research should include multicenter prospective cohort studies with
larger and more diverse patient populations, as well as
standardized treatment protocols. Randomized controlled trials
comparing different TACE strategies (on-demand vs. scheduled TACE,
fixed number of sessions vs. response-adapted approach) would
provide the highest level of evidence. Furthermore, studies
incorporating quality-of-life assessment and cost-effectiveness
analyses would provide more comprehensive information to guide
clinical decision-making. With 62 progression events and 53 death
events in the cohort, the models achieved events per variable (EPV)
ratios of ~8.9 and 8.8, respectively, which are within acceptable
limits to avoid overfitting (the commonly accepted threshold is EPV
≥10). Sensitivity analyses using stepwise selection and penalized
Cox regression (ridge regression) yielded similar hazard ratio
estimates, supporting the robustness of the findings.
The comparable distribution of these technical
factors across groups suggests that procedural heterogeneity was
unlikely to confound the comparison of outcomes among TACE
frequency groups.
Numerous factors influence the prognosis of liver
cancer. Tumor stage and diameter are significantly associated with
survival outcomes in HCC (36).
Studies (20,37,38)
have identified advanced tumor stage as an independent risk factor
for poor prognosis in HCC. In the present study, hypertension,
number of TACE sessions, adverse reactions, targeted
therapy/immunotherapy and BCLC stage were identified as independent
prognostic factors for PFS, while CA199 levels, number of TACE
sessions, adverse reactions and BCLC stage were independent
prognostic factors for OS. The BCLC staging system is one of the
most widely used staging classifications for HCC; it integrates
multiple variables, including tumor burden, liver functional
reserve and tumor aggressiveness and is essential for both
prognostic stratification and treatment selection. The BCLC system
is widely accepted in clinical practice (38,39).
In the present study, both uni- and multivariate Cox
regression analyses confirmed that BCLC stage and the occurrence of
adverse reactions were independent prognostic factors for PFS and
OS. Emerging evidence suggests that combining TACE with targeted
therapy and immunotherapy significantly improves PFS, OS and ORR in
patients with advanced HCC, with an acceptable safety profile,
compared with TACE alone (40).
However, in the present cohort, targeted therapy/immunotherapy was
associated with PFS but not with OS, and hypertension was an
independent predictor for PFS only, whereas CA199 level was
independently associated with OS only. These findings may be
attributable to the limited sample size. The potential confounding
effect of subsequent systemic therapy warrants careful
consideration. In the present cohort, the majority of patients
(78.9%) received systemic therapy after completing all TACE
sessions, typically following disease progression. This suggests
that systemic therapy was more often a consequence of disease
progression rather than a confounder of the association between
TACE frequency and survival. Sensitivity analyses excluding
patients who received systemic therapy between TACE sessions did
not notably alter the findings. Nevertheless, the present study
cannot exclude the possibility that the timing of systemic therapy
influenced outcomes; future studies should collect detailed timing
data to enable time-dependent covariate analysis. The
identification of hypertension as an independent risk factor for
PFS, but not for OS, warrants exploration of potential underlying
mechanisms. Hypertension may affect hepatic microcirculation and
sinusoidal pressure, potentially influencing drug delivery and
distribution during TACE (41).
Additionally, antihypertensive medications, particularly
β-blockers, have been implicated in altered tumor angiogenesis and
progression in some studies (42).
Chronic hypertension may also reflect underlying endothelial
dysfunction and systemic inflammation, which may promote tumor
progression (43). However, these
hypotheses require further investigation. Although CA199 is not a
classical HCC biomarker, elevated levels may indicate several
underlying conditions, such as undetected combined
hepatocellular-cholangiocarcinoma components, which are associated
with a poorer prognosis (44),
biliary obstruction or cholangitis, which may complicate the
clinical course and limit treatment options (45), or advanced liver disease with
cholestasis, reflecting more severe underlying liver dysfunction
(46). Additionally, tumor
expression of CA199 may serve as a marker of more aggressive
biological behavior (47). The
specificity of CA199 for OS, rather than PFS, suggests that it may
be more closely associated with overall disease progression and
hepatic functional deterioration than early tumor response. Future
studies incorporating histological confirmation and detailed
biliary imaging would help elucidate this association. Immortal
time bias is a theoretical concern in studies evaluating treatment
frequency, as patients must survive long enough to receive multiple
treatments. The present study evaluated treatment strategies
(single vs. multiple TACE) rather than the time-dependent effect of
each additional session. PSM balanced baseline prognostic factors
that may influence both treatment receipt and survival, decreasing
confounding by indication. All survival analyses were calculated
from the date of first TACE, ensuring identical starting points for
all groups. The relatively short intervals between TACE sessions
(median, 6–8 weeks) minimized the immortal time window and the
early divergence of survival curves (within the first 6 months)
suggested that the observed benefits are unlikely to be solely
attributable to immortal time bias. Nevertheless, the present study
cannot entirely exclude residual confounding factors; future
prospective studies employing landmark analyses or time-dependent
covariate approaches are required to validate the present
findings.
Previous studies (48,49)
have demonstrated that the number of TACE sessions is a protective
factor for prognosis in patients with HCC. The present study
indicated that repeated TACE for HCC has clinical value. Within the
range of one to three TACE sessions, an increased number of TACE
sessions was associated with prolonged PFS and OS times. However,
the indications for repeat TACE should be strictly applied; blind
expansion of its use is not advocated, as preserving liver
function, maintaining quality of life and minimizing unnecessary
procedures are of paramount clinical importance. Furthermore,
greater emphasis should be placed on comprehensive treatment
strategies, such as TACE combined with targeted therapy and
immunotherapy, to improve prognosis in patients with liver
cancer.
The number of TACE sessions significantly influences
the prognosis of patients with HCC. Prior to TACE, Child-Pugh class
and the presence of cirrhosis should be assessed to evaluate
hepatic functional reserve. During the procedure, superselective
segmental embolization should be performed when possible to
minimize damage to the peritumoral liver parenchyma. Following
TACE, active hepatoprotective therapy should be administered to
mitigate the occurrence of AEs. Additionally, the interval between
TACE sessions should be appropriately managed and other therapeutic
modalities, such as targeted therapy and immunotherapy, may be
considered in combination. In clinical practice, treatment plans
should be tailored to individual tumor characteristics and liver
function, while avoiding indiscriminate repeat treatment that may
compromise liver function and decrease survival.
In conclusion, the present study evaluated the
efficacy and safety of different numbers of TACE sessions in
patients with uHCC, with control for confounding factors through
the use of PSM. While previous studies (50,51)
have examined TACE refractoriness and repeated TACE, the present
study specifically compares outcomes across three distinct
treatment frequency groups (1, 2 and ≥3 sessions) in a
well-balanced cohort following PSM. The present findings complement
previous studies (17,52) and provide additional evidence to
inform clinical decision-making regarding the optimal number of
TACE sessions. However, indiscriminate repeat prophylactic TACE
does not necessarily enhance treatment efficacy; treatment should
be tailored to the individual clinical situation, integrating
appropriate comprehensive strategies to optimize survival outcomes.
For patients with uHCC and preserved liver function (Child-Pugh
class A or B) who demonstrate initial response to TACE, ≥2 TACE
sessions should be considered to optimize tumor response and
survival outcomes. The present data suggest that a second planned
TACE session, even in patients with an apparent good initial
response, may confer additional benefit through more complete tumor
necrosis and treatment of satellite lesions. Routine prophylactic
TACE beyond three sessions should be avoided in the absence of
clear evidence of ongoing benefit. The decision to proceed with a
third or subsequent TACE session should be individualized,
considering objective evidence of residual viable tumor on imaging,
sustained adequate liver function (Child-Pugh class A or B with
minimal decompensation), absence of TACE refractoriness
(progressive disease despite two consecutive TACE sessions) and
patient performance status and preferences. For patients with
treatment-related adverse reactions, enhanced prognostic monitoring
is warranted, as these patients exhibited significantly worse PFS
and OS in the present study. Close attention to preserving liver
function, aggressive supportive care and early consideration of
alternative or combination therapies (targeted therapy,
immunotherapy) may be beneficial in this subgroup. Patients with
hypertension may require more intensive monitoring for early
progression and optimal blood pressure control should be maintained
throughout the treatment course. For patients with elevated CA199
at baseline, clinicians should consider the possibility of combined
hepatocellular-cholangiocarcinoma or biliary pathology (53) and appropriate diagnostic workup may
be warranted. These patients may benefit from closer surveillance
and consideration of more aggressive treatment strategies.
Acknowledgements
Not applicable.
Funding
Funding: No funding was received.
Availability of data and materials
The data generated in the present study are not
publicly available due to patient privacy and ethical restrictions
associated with the institutional review board approval, but may be
requested from the corresponding author.
Authors' contributions
The study was conceptualized by LZ and XX. Formal
analysis and data interpretation were performed by LZ, XX, CL, HG,
XZ and WL. Data acquisition and clinical management were carried
out by WL, CL and HG. Literature analysis was conducted by XX, CL
and HG. Writing of the original draft was conducted by XX, LZ and
CL. Reviewing and editing of the manuscript was performed by XX,
LZ, CL, HG, XZ and WL. Supervision was provided by LZ, XZ, HG and
WL. Project administration was conducted by CL, HG and WL. All
authors have read and approved the final version of the manuscript.
LZ, XX and CL confirm the authenticity of all the raw data.
Ethics approval and consent to
participate
The present study was approved by the Medical Ethics
Committee of Hebei General Hospital (Shijiazhuang, China; approval
no. 2024-LW-0204). Written informed consent was obtained from all
patients for the TACE procedure prior to treatment. For the
retrospective analysis of medical records, the requirement for
informed consent was waived by the Ethics Committee due to the
retrospective nature of the study. The study was performed in
accordance with The Declaration of Helsinki.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Bray F, Ferlay J, Soerjomataram I, Siegel
RL, Torre LA and Jemal A: Global cancer statistics 2018: GLOBOCAN
estimates of incidence and mortality worldwide for 36 cancers in
185 countries. CA Cancer J Clin. 68:394–424. 2018.PubMed/NCBI
|
|
2
|
Llovet JM, Kelley RK, Villanueva A, Singal
AG, Pikarsky E, Roayaie S, Lencioni R, Koike K, Zucman-Rossi J and
Finn RS: Hepatocellular carcinoma. Nat Rev Dis Primers. 7:62021.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Siegel RL, Miller KD, Fuchs HE and Jemal
A: Cancer statistics, 2021. CA Cancer J Clin. 71:7–33.
2021.PubMed/NCBI
|
|
4
|
Haber PK, Puigvehí M, Castet F, Lourdusamy
V, Montal R, Tabrizian P, Buckstein M, Kim E, Villanueva A,
Schwartz M and Llovet JM: Evidence-Based management of
hepatocellular carcinoma: Systematic review and meta-analysis of
Randomized controlled trials (2002–2020). Gastroenterology.
161:879–898. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Sung H, Ferlay J, Siegel RL, Laversanne M,
Soerjomataram I, Jemal A and Bray F: Global cancer statistics 2020:
GLOBOCAN estimates of incidence and mortality worldwide for 36
cancers in 185 countries. CA Cancer J Clin. 71:209–249.
2021.PubMed/NCBI
|
|
6
|
Wild C, Weiderpass E and Stewart BW: World
cancer report: cancer research for cancer prevention. International
Agency for Research on Cancer; Lyon: 2020
|
|
7
|
Chen X, Zhong G and Gong J: Pathogenesis
and treatment progress of hepatocellular carcinoma. Int J Surg.
47:202–206. 2020.(In Chinese).
|
|
8
|
Forner A, Reig M and Bruix J:
Hepatocellular carcinoma. Lancet. 391:1301–1314. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Villanueva A: Hepatocellular carcinoma. N
Engl J Med. 380:1450–1462. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Lu J, Zhao M, Arai Y, Zhong BY, Zhu HD, Qi
XL, de Baere T, Pua U, Yoon HK, Madoff DC, et al: Clinical practice
of transarterial chemoembolization for hepatocellular carcinoma:
consensus statement from an international expert panel of
International Society of Multidisciplinary Interventional Oncology
(ISMIO). Hepatobiliary Surg Nutr. 10:661–671. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Park JW, Chen M, Colombo M, Roberts LR,
Schwartz M, Chen PJ, Kudo M, Johnson P, Wagner S, Orsini LS and
Sherman M: Global patterns of hepatocellular carcinoma management
from diagnosis to death: The BRIDGE Study. Liver Int. 35:2155–2166.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Lencioni R, de Baere T, Soulen MC, Rilling
WS and Geschwind JF: Lipiodol transarterial chemoembolization for
hepatocellular carcinoma: A systematic review of efficacy and
safety data. Hepatology. 64:106–116. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Takayasu K, Arii S, Kudo M, Ichida T,
Matsui O, Izumi N, Matsuyama Y, Sakamoto M, Nakashima O, Ku Y, et
al: Superselective transarterial chemoembolization for
hepatocellular carcinoma. Validation of treatment algorithm
proposed by Japanese guidelines. J Hepatol. 56:886–892. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Okusaka T, Okada S, Ueno H, Ikeda M,
Yoshimori M, Shimada K, Yamamoto J, Kosuge T, Yamasaki S, Iwata R,
et al: Evaluation of the therapeutic effect of transcatheter
arterial embolization for hepatocellular carcinoma. Oncology.
58:293–299. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Clinical Guidelines Committee of Chinese
College of Interventionalists, . Chinese clinical practice
guidelines for transarterial chemoembolization of hepatocellular
carcinoma (2023 edition). Zhonghua Yi Xue Za Zhi. 103:2674–2694.
2023.(In Chinese). PubMed/NCBI
|
|
16
|
Jansen MC, van Hillegersberg R, Chamuleau
RA, van Delden OM, Gouma DJ and van Gulik TM: Outcome of regional
and local ablative therapies for hepatocellular carcinoma: a
collective review. Eur J Surg Oncol. 31:331–347. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Raoul JL, Forner A, Bolondi L, Cheung TT,
Kloeckner R and de Baere T: Updated use of TACE for hepatocellular
carcinoma treatment: How and when to use it based on clinical
evidence. Cancer Treat Rev. 72:28–36. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Marrero JA, Kulik LM, Sirlin CB, Zhu AX,
Finn RS, Abecassis MM, Roberts LR and Heimbach JK: Diagnosis,
staging, and management of hepatocellular carcinoma: 2018 practice
guidance by the American association for the study of liver
diseases. Hepatology. 68:723–750. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Pugh RN, Murray-Lyon IM, Dawson JL,
Pietroni MC and Williams R: Transection of the oesophagus for
bleeding oesophageal varices. Br J Surg. 60:646–649. 1973.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Reig M, Forner A, Rimola J, Ferrer-Fàbrega
J, Burrel M, Garcia-Criado Á, Kelley RK, Galle PR, Mazzaferro V,
Salem R, et al: BCLC strategy for prognosis prediction and
treatment recommendation: The 2022 update. J Hepatol. 76:681–693.
2022. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Oken MM, Creech RH, Tormey DC, Horton J,
Davis TE, McFadden ET and Carbone PP: Toxicity and response
criteria of the Eastern Cooperative Oncology Group. Am J Clin
Oncol. 5:649–655. 1982. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Lencioni R and Llovet JM: Modified RECIST
(mRECIST) assessment for hepatocellular carcinoma. Semin Liver Dis.
30:52–60. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Freites-Martinez A, Santana N,
Arias-Santiago S and Viera A: Using the common terminology criteria
for adverse events (CTCAE - Version 5.0) to evaluate the severity
of adverse events of anticancer therapies. Actas Dermosifiliogr
(Engl Ed). 112:90–92. 2021.(In English, Spanish). View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Ho D, Imai K, King G and Stuart EA:
MatchIT: Nonparametric preprocessing for parametric causal
inference. Journal of statistical software. 42:1–28. 2011.
View Article : Google Scholar
|
|
25
|
Yamada R, Sato M, Kawabata M, Nakatsuka H,
Nakamura K and Takashima S: Hepatic artery embolization in 120
patients with unresectable hepatoma. Radiology. 148:397–401. 1983.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Shi M, Lu LG, Fang WQ, Guo RP, Chen MS, Li
Y, Luo J, Xu L, Zou RH, Lin XJ and Zhang YQ: Roles played by
chemolipiodolization and embolization in chemoembolization for
hepatocellular carcinoma: Single-blind, randomized trial. J Natl
Cancer Inst. 105:59–68. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Miyayama S, Yamashiro M, Ikeda R,
Matsumoto J, Takeuchi K, Sakuragawa N, Ueda T, Sanada T, Notsumata
K and Terada T: Efficacy of superselective conventional
transarterial chemoembolization using guidance software for
hepatocellular carcinoma within three lesions smaller than 3 cm.
Cancers (Basel). 13:63702021. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Miyayama S, Yamashiro M, Ikuno M, Okumura
K and Yoshida M: Ultraselective transcatheter arterial
chemoembolization for small hepatocellular carcinoma guided by
automated tumor-feeders detection software: Technical success and
short-term tumor response. Abdom Imaging. 39:645–656. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Wang L, Zhang J and Liu J: Re-evaluating
the survival benefit of adding camrelizumab to TACE plus
tyrosine-kinase inhibitors in unresectable hepatocellular
carcinoma: the undervalued influence of TACE frequency. Ann
Hepatol. 30:1021262025. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Sergio A, Cristofori C, Cardin R, Pivetta
G, Ragazzi R, Baldan A, Girardi L, Cillo U, Burra P, Giacomin A and
Farinati F: Transcatheter arterial chemoembolization (TACE) in
hepatocellular carcinoma (HCC): The role of angiogenesis and
invasiveness. Am J Gastroenterol. 103:914–921. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Mizukoshi E, Yamashita T, Arai K,
Sunagozaka H, Ueda T, Arihara F, Kagaya T, Yamashita T, Fushimi K
and Kaneko S: Enhancement of tumor-associated antigen-specific T
cell responses by radiofrequency ablation of hepatocellular
carcinoma. Hepatology. 57:1448–1457. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Raoul JL, Gilabert M and Piana G: How to
define transarterial chemoembolization failure or refractoriness: A
European perspective. Liver Cancer. 3:119–124. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Gao S, Yang X, Wang L, Zhang F and Li J:
Analysis of the correlation between the presence of
post-embolization syndrome after TACE for hepatocellular carcinoma
and the clinical mortality of patients. Modern Digestion and
Inttervention. 25:230–233. 2020.(In Chinese).
|
|
34
|
Mason MC, Massarweh NN, Salami A,
Sultenfuss MA and Anaya DA: Post-embolization syndrome as an early
predictor of overall survival after transarterial chemoembolization
for hepatocellular carcinoma. HPB (Oxford). 17:1137–1144. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Roehlen N, Stoehr F, Müller L, Luxenburger
H, Gairing SJ, Reincke M, Schultheiss M, Berisha F, Weinmann A,
Foerster F, et al: Prediction of postembolization syndrome after
transarterial chemoembolization of hepatocellular carcinoma and its
impact on prognosis. Hepatol Commun. 7:e02522023. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Cun J, Xu Y, Li W and Zhao X: Analysis of
factors affecting the prognosis of transcatheter arterial
chemoembolization for hepatitis B-related hepatocellular carcinoma.
J Interv Med. 4:66–70. 2021.PubMed/NCBI
|
|
37
|
Yi W, Tao H, Zhiqiang Z, Gang C, Hem L and
Henglie C: Correlation between serum microRNA-599 expression level
and prognosis of patients with hepatocellular carcinoma after
receiving transcatheter arterial chemoembolization. J Intervent
Radiol. 30:1265–1270. 2021.(In Chinese).
|
|
38
|
Tsilimigras DI, Bagante F, Sahara K, Moris
D, Hyer JM, Wu L, Ratti F, Marques HP, Soubrane O, Paredes AZ, et
al: Prognosis after resection of barcelona clinic liver cancer
(BCLC) Stage 0, A, and B hepatocellular carcinoma: A comprehensive
assessment of the current BCLC classification. Ann Surg Oncol.
26:3693–3700. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Kikuchi L, Chagas AL, Alencar R, Tani C,
Diniz MA, D'Albuquerque LAC and Carrilho FJ: Adherence to BCLC
recommendations for the treatment of hepatocellular carcinoma:
Impact on survival according to stage. Clinics (Sao Paulo).
72:454–460. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Zhu HD, Li HL, Huang MS, Yang WZ, Yin GW,
Zhong BY, Sun JH, Jin ZC, Chen JJ, Ge NJ, et al: T ransarterial
chemoembolization with PD-(L)1 inhibitors plus molecular targeted
therapies for hepatocellular carcinoma (CHANCE001). Signal
Transduct Target Ther. 8:582023. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Neves K, Alves-Lopes R, Montezano A and
Touyz R: P169 VEGF inhibition-induced PARP overactivation leads to
endothelial dysfunction and inflammation: role of sirtuin 1
signalling. J Hyper. 42:e1222024. View Article : Google Scholar
|
|
42
|
Nair SG, Benny S, Jose WM and Aneesh TP:
Beta-blocker adjunct therapy as a prospective anti-metastatic with
cardio-oncologic regulation. Clin Exp Metastasis. 41:9–24. 2024.
View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Abe M and Arima H: Hypertension-cancer
recurrence crosstalk: Implications for survivorship care. Hypertens
Res. Feb 19–2026.(Epub ahead of print). View Article : Google Scholar
|
|
44
|
Lv TR, Hu HJ, Ma WJ, Liu F, Jin YW and Li
FY: Meta-analysis of prognostic factors for overall survival and
disease-free survival among resected patients with combined
hepatocellular carcinoma and cholangiocarcinoma. Eur J Surg Oncol.
50:1072792024. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Ghallab M, Abosheaishaa H, Gupta I,
Abdelmoteleb S and Stern R: A case of choledocholithiasis and
obstructive jaundice with a very high serum carbohydrate antigen
19-9 (CA 19-9) level: A case report and review of literature.
Cureus. 14:e324472022.PubMed/NCBI
|
|
46
|
Giannini E, Borro P, Botta F, Chiarbonello
B, Fasoli A, Malfatti F, Romagnoli P, Testa E, Risso D, Lantieri
PB, et al: Cholestasis is the main determinant of abnormal CA 19-9
levels in patients with liver cirrhosis. Int J Biol Markers.
15:226–230. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Du T, Zou J, Yang Y, Xie H, Pang H, Zhuang
W, Wang S and Wei G: CA19-9-related macrophage polarization drives
poor prognosis in HCC after immune checkpoint inhibitor treatment.
Front Oncol. 14:15281382024. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Guoqi L, Xiangwei W, Hongqiang Y, Lv H,
Sun H, Chu Z, Li T, Qiu M, Ma J, Zhang S, et al: Prognostic factors
of TACE treatment in patients with primary hepatic carcinoma. J
Modern Oncol. 267–270. 2016.(In Chinese).
|
|
49
|
Zhang YF, Guo RP, OuYang HY, Shen JX, Zhao
J, Tan GS, Le Y, Wei W and Shi M: Target lesion response predicts
survival of patients with hepatocellular carcinoma retreated with
transarterial chemoembolization. Liver Int. 36:1516–1524. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Yang C, Luo YG, Yang HC, Yao ZH and Li X:
Effects of early TACE refractoriness on survival in patients with
hepatocellular carcinoma: A real-world study. J Hepatocell
Carcinoma. 9:621–631. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Zhong BY, Wang WS, Zhang S, Zhu HD, Zhang
L, Shen J, Zhu XL, Teng GJ and Ni CF: Re-evaluating transarterial
chemoembolization failure/refractoriness: A survey by Chinese
college of interventionalists. J Clin Transl Hepatol. 9:521–527.
2021.PubMed/NCBI
|
|
52
|
Kim JH, Shim JH, Lee HC, Sung KB, Ko HK,
Ko GY, Gwon DI, Kim JW, Lim YS and Park SH: New intermediate-stage
subclassification for patients with hepatocellular carcinoma
treated with transarterial chemoembolization. Liver Int.
37:1861–1868. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Huang XW, Huang Y, Chen LD, Wang Z, Yang
Z, Liu JY, Xie XY, Lu MD, Shen SL and Wang W: Potential diagnostic
performance of contrast-enhanced ultrasound and tumor markers in
differentiating combined hepatocellular-cholangiocarcinoma from
hepatocellular carcinoma and cholangiocarcinoma. J Med Ultrason
(2001). 45:231–241. 2018. View Article : Google Scholar : PubMed/NCBI
|