Introduction
Bladder cancer ranks as the 9th most common
malignant tumor globally and the 7th among men. Muscular invasive
bladder cancer is a particularly lethal form, ranking 13th in
mortality among malignant tumors (1). Smoking is a well-evidenced risk factor
for bladder cancer, elevating the risk by two to three times. The
risk of bladder cancer escalates with both the duration and
quantity of smoking (2–4). Common treatments for bladder cancer
include c(TURBT), bladder irrigation and radical cystectomy.
Paraneoplastic syndromes refer to a number of
symptoms that occur in patients with malignant tumors that cannot
be readily explained by local invasion or distant metastasis of the
tumor. Instead, these symptoms might be related to primary tumor
autocrine hormones or cytokines. A white blood cell (WBC) count
>50×109/l, in the absence of leukemia or infection,
is defined as a leukemoid reaction (5). The leukemoid reaction has been
reported in nearly all solid tumor types as a paraneoplastic
syndrome, with an incidence ranging from 1 to 4% (6). Furthermore, a leukemoid reaction is
frequently observed clinically in advanced stages of highly
invasive and metastatic cancer (7–9).
Bladder cancer, especially urothelial carcinoma, is rarely
associated with a leukemoid reaction. Globally, <40 cases have
been reported in the literature in the past 40 years, with most
patients experiencing poor prognosis within months; in the present
study seven of these cases were summarized in Table I.
 | Table I.Cases in the literature. |
Table I.
Cases in the literature.
| Author, year | Age/sex | Tumor type | White blood cell
count | Prognosis | (Refs.) |
|---|
| Turalic et al,
2006 | 51/woman | Poorly differentiated
bladder carcinoma with G-CSF production | 14,000/ml then
increased to 34,000/ml | Deceased before
additional treatment could be instituted | (36) |
| Kato et al,
2016 | 38/woman | Urothelial cell
carcinoma with G-CSF and PTHrP production |
107,000/mm3 | Deceased within 3
months of initial symptoms | (37) |
| Harsha et al,
2020 | 59/man | High-grade urinary
bladder cancer | Significantly
elevated, mainly eosinophils | Deteriorated rapidly,
deemed inoperable, referred for palliative management | (12) |
| Perez et al,
2009 | 63/man | High-grade
urothelial carcinoma invasive into muscularis propria | 42,000/µl with
37,000/µl neutrophils | Died nearly 4 weeks
after surgery, 4 months after initiation of neoadjuvant
chemotherapy | (19) |
| Shapiro et
al, 2014 | 43/man | Muscle-invasive
squamous cell carcinoma | 76,000/µl with
predominant polymorphonuclear cells | Died within 1 month
after hospital readmission with similar symptoms | (11) |
| He et al,
2014 | 64/man | Transitional cell
carcinoma, Grade 3 |
58,400/mm3 with neutrophil
granulocyte count of 54,900/mm3 | Died of systemic
metastasis within 6 months after cystectomy | (38) |
| Kumar et al,
2014 | 39/woman | Invasive high-grade
urothelial carcinoma | 57800/µl | Death within 10
months from first diagnosis | (7) |
In the present report, a rare case of bladder cancer
complicated by fever and a leukemoid reaction was presented. The
clinical and histological features were described, and the relevant
literature on these rare cases was reviewed.
Case report
The current patient had undergone several TURBT
procedures and experienced hematuria and decreased urine output for
2 months prior his admission in the Shanghai Municipal Hospital of
Traditional Chinese Medicine (Shanghai, China). The patient had
been treated in other hospitals before and the early examination
report was incomplete. A 62-year-old male native of Shanghai with a
40-year history of smoking, the patient presented with hematuria in
August 2016. A cystoscopy examination revealed a cauliflower-like
growth on the right wall of the bladder. Subsequently, a TURBT was
performed. Postoperative pathological diagnosis confirmed papillary
urothelial carcinoma (UC), staged as pTa (low grade) according to
the American Joint Committee on Cancer TNM classification system
(10).
Following the procedure, the patient began a
standard regimen of intravesical epirubicin instillation (50 mg
weekly for 8 weeks, then monthly for 10 months), with surveillance
cystoscopy scheduled at 3-month intervals. During the year, the
patient also underwent routine cystoscopy examinations, which
revealed no evidence of recurrent bladder cancer. However, in
October 2017, during a routine cystoscopy examination, tumors were
found on the right and top wall of the bladder. Subsequently, the
patient underwent another TURBT. Postoperative pathological
diagnosis confirmed papillary urothelial carcinoma, UC, pTa, low
grade. The same adjuvant intravesical epirubicin regimen was
administered (50 mg weekly for 8 weeks, then monthly for 10
months), alongside surveillance cystoscopy every 3 months.
In March 2019, the patient presented with visible
hematuria. Cystoscopy examination revealed tumors on the right and
top walls of the bladder. Another TURBT was performed. Pathological
diagnosis following this surgery confirmed papillary urothelial
carcinoma, high grade, non-muscle invasive. In June 2019, during a
routine cystoscopy examination, tumors were found on both walls of
the bladder and another TURBT was performed. Postoperative
pathological diagnosis confirmed papillary urothelial carcinoma,
high grade. In August 2020, during his regular cystoscopy
examination, tumors were found once again on the left, right and
top walls of the patient's bladder. The patient refused further
treatment, such as radical cystectomy, and no subsequent follow-up
visits were conducted.
In May 17 2022, the patient was admitted to Shanghai
Municipal Hospital of Traditional Chinese Medicine due to
hematuria. Cystoscopy examination revealed multiple tumors in the
urethra, trigone and both walls of the bladder. A TURBT was
performed in May 2022. Postoperative pathological diagnosis
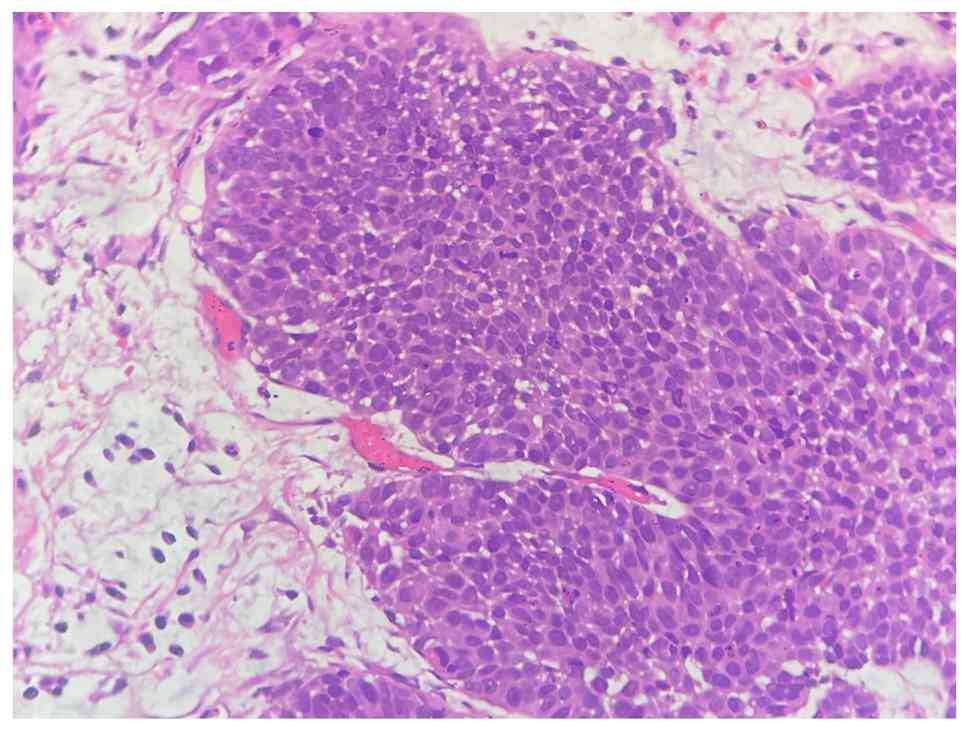
confirmed papillary urothelial carcinoma, high grade (Fig. 1). Post-surgery treatment consisted
of intravesical instillation of epirubicin at a dose of 50 mg per
session, administered once weekly for 8 weeks. In July 2022, the
patient was admitted for follow-up and underwent a cystoscopy
examination, which again revealed multiple tumors in the urethra,
trigone and both walls of the bladder. A radical cystectomy was
recommended; however, the patient refused. Instead, another TURBT
was performed the next day, with the pathological diagnosis once
more indicating high grade papillary urothelial carcinoma (Fig. 2). After the surgery, the patient did
not adhere to regular bladder irrigation or follow-up cystoscopy
examinations.
In August 2023, the patient started experiencing
painless visible hematuria and decreased urine output. The patient
was then admitted to Shanghai Municipal Hospital of Traditional
Chinese Medicine, Shanghai University of Traditional Chinese
Medicine (Shanghai, China) in October 2023. Upon admission, the
patient's body temperature was recorded at 38°C. Urinalysis
revealed 500 leukocytes/µl and 3+ leukocyte esterase. Blood
analysis revealed a WBC count of 67.83×109/l with
neutrophils at 64.5×109/l, C-reactive protein (CRP) at
150.43 mg/l, and creatinine at 1,006 µmol/l.
Urology computed tomography (CT) scan results
revealed: i) Multiple tumors in the bladder (Fig. 3A); ii) multiple lymphadenopathies in
the pelvis and along the right iliac artery (Fig. 3B); iii) bilateral ureter bladder
entrance invasion (Fig. 3C)
resulting in bilateral renal pelvis and ureter dilatation (Fig. 3D). Meropenem was administered as
anti-infective therapy at a dosage of 0.5 g via intravenous
infusion every 8 h. Cystoscopy performed under local anesthesia
identified: Multiple tumors in the posterior urethra, bladder neck
and bladder walls, with no tumors observed in the bilateral
ureters. A biopsy sample was obtained, and a urinary catheter was
placed. Pathological diagnosis confirmed high grade papillary
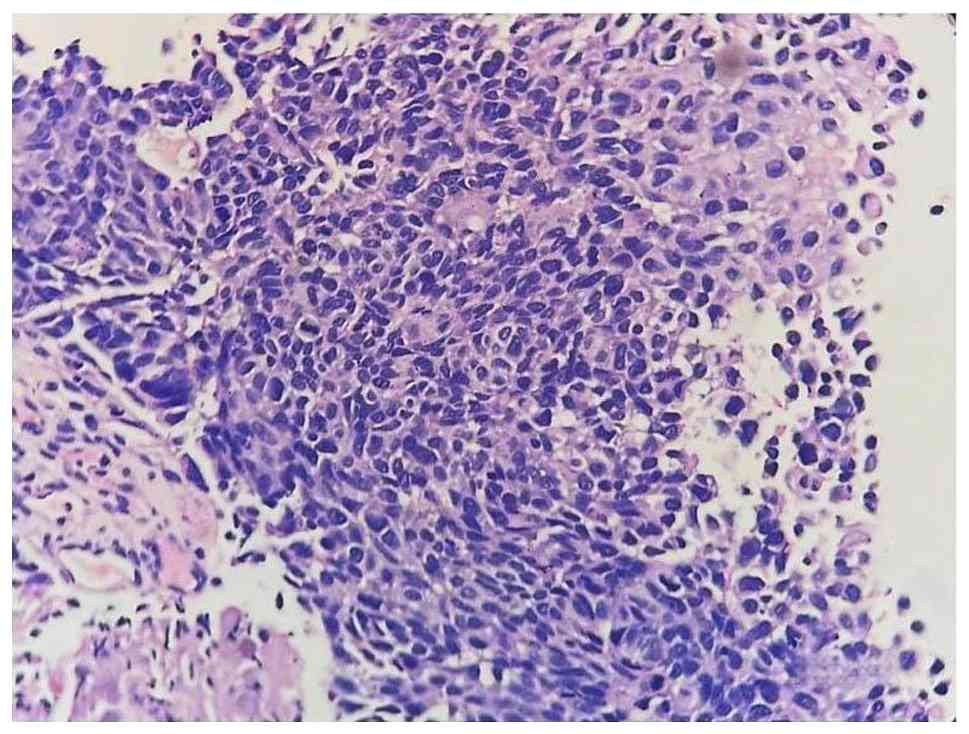
urothelial carcinoma with massive necrosis (Fig. 4).
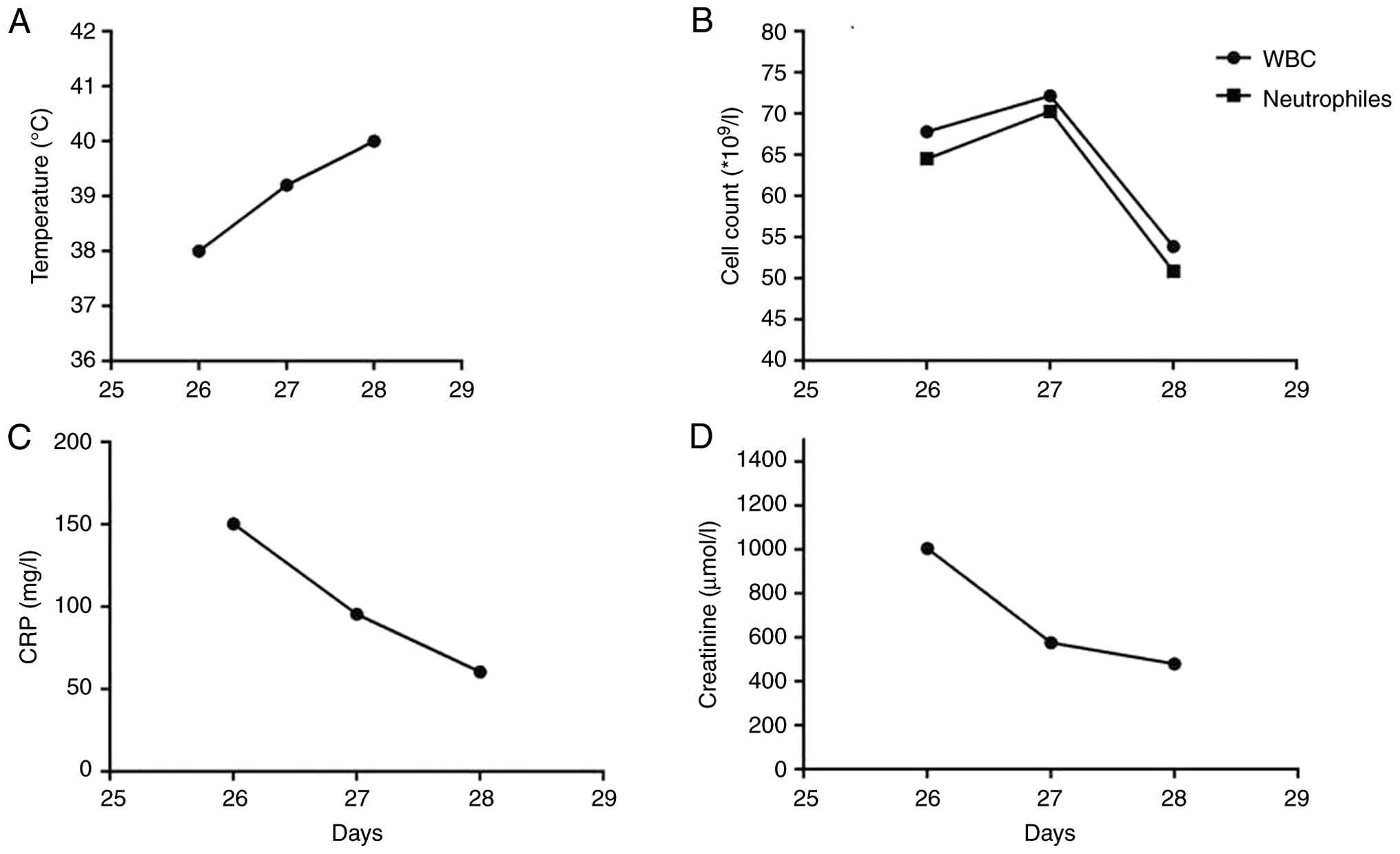
The day after admittance (October 2023), the
patient's body temperature increased to 39.2°C. Blood analysis
revealed marked leukocytosis with neutrophilia, significantly
elevated inflammatory markers, and acute kidney injury. Admission
laboratory results are presented in Table II. The patient refused a bone
marrow biopsy chest CT, bone scan and PET-CT. Considering the
patient's deteriorating health and the high risk associated with
surgery and anesthesia, palliative care was administered. The
patient's body temperature dropped to 38.6°C following an
intramuscular injection of 2 ml of compound aminophenazone and
barbital injection (containing 200 mg aminophenazone, 40 mg
antipyrine and 18 mg barbital) during nighttime. At 2 days
post-admittance (October 2023), the patient's body temperature
increased again to 40°C. Blood analysis revealed a WBC count of
53.89×109/l with neutrophils at 50.87×109/l,
CRP at 60.65 mg/l and creatinine at 480 µmol/l. The patient's white
blood cells were too high, so urosepsis was ruled out. A urine
culture was not performed following the shift to palliative care.
The patient passed away that afternoon. Because the Shanghai
Municipal Hospital of Traditional Chinese Medicine hospital has not
yet acquired the relevant technology, granulocyte-colony
stimulating factor (G-CSF) levels in specific patients cannot be
measured and tracked for a long time. The timeline of the disease
progression is summarized in Table
III, and changes in temperature, WBC count, CRP and creatinine
are illustrated in Fig. 5 [prepared
using GraphPad Prism (version 9.5; Dotmatics)].
 | Table II.Laboratory test results. |
Table II.
Laboratory test results.
| Parameter | Patient's
value | Normal reference
range |
|---|
| White blood cell
count, ×109/l | 72.17 | 4.0–10.0 |
| Neutrophil count,
×109/l | 70.27 | 2.0–7.5 |
| C-reactive protein,
mg/l | 95.65 | 0.0–5.0 |
| Creatinine,
µmol/l | 577.00 | 53-106 |
 | Table III.Timeline of the disease. |
Table III.
Timeline of the disease.
| Date | Treatment | Findings |
|---|
| 2016.8 | TURBT1 + bladder
irrigation | UC, pTa, low
grade |
| 2017.10 | TURBT2 + bladder
irrigation | UC, pTa, low
grade |
| 2019.3 | TURBT3 + bladder
irrigation | UC, pTa, low
grade |
| 2019.6 | TURBT4 + bladder
irrigation | UC, pTa, high
grade |
| 2020.8 | Cystoscopy | Bladder tumor and
patient refused further treatment |
| 2022.5 | TURBT5 + bladder
irrigation | UC, pTa, high
grade |
| 2022.7 | TURBT6 + bladder
irrigation | UC, pTa, high grade
and patient rejected total cystectomy |
| 2023.10 | Computed tomography
and cystoscopy | Multiple tumors in
the bladder, multiple lymphadenopathy, bilateral ureter bladder
entrance invasion, bilateral renal pelvis and ureter
dilatation |
| 2023.10 | Palliative
treatment | The patient passed
away |
Discussion
In urological malignancies, renal tumors are most
frequently associated with a leukemoid reaction. By contrast,
bladder tumors, particularly urothelial carcinoma, are rarely
linked to such reactions (11). The
incidence of a leukemoid reaction tends to be higher in cancer
patients who are older or have higher-grade tumors. A leukemoid
reaction, according to the type of leukocytes involved, can be
classified into: Neutrophilic, lymphocytic, monocytic or
eosinophilic, with neutrophilic leukemoid reaction being the most
prevalent (12). The leukocyte
alkaline phosphatase score is often helpful in diagnosing a
leukemoid reaction; however, its utility may vary, especially in
identifying chronic neutrophil leukemia (13). Iyengar et al (14) proposed and established a supervised
machine learning model (based on a support vector machine
algorithm) that effectively distinguishes between leukemic
reactions and myeloid malignancies using routine clinical data with
a sensitivity of 96% and a specificity of 95.9%.
The patient in the present case had a significant
smoking history and lacked regular cystoscopy follow-ups and
bladder irrigation. Presenting with hematuria, decreased urine
output and fever, along with urinalysis indicative of urinary tract
infection, the patient was initially treated with antibiotics
followed by a cystoscopy examination. Some cases report urinary
tract infection rates of 5–10% after transurethral surgery, despite
proper prophylactic antibiotics (15). However, in the present case,
antibiotic treatment failed to resolve the patient's fever and the
WBC count continued to rise, indicating that antibiotic therapy was
ineffective. The patient has no history of leukemia and no previous
history of other hematological conditions. Therefore, it was
deduced that the patient did not have severe infections; instead,
the patient exhibited a leukemoid reaction secondary to bladder
cancer, predominantly neutrophilic in nature.
The patient's August 2023 CT scan indicated the
presence of multiple tumors in the bladder and multiple
lymphadenopathies near the right iliac artery and in the pelvis.
The pathophysiological mechanism underlying fever and a leukemoid
reaction caused by urothelial carcinoma of the bladder may be
elucidated as follows: Urothelial cancer cells stimulate and
infiltrate lymph nodes, leading to the activation of macrophages,
which subsequently produce a substantial quantity of cytokines,
such as IL-6, TNF-α (16).
Additionally, necrotic malignant tumor cells release a significant
amounts of endotoxins pyrogens known as damage-associated molecular
patterns, which stimulate the production of pro-inflammatory
cytokines, contributing to the manifestation of fever in the
patient (17). Meanwhile, these
cytokines facilitate the activation and proliferation of T cells or
induce autocrine production of G-CSF, thereby promoting the
differentiation and proliferation of granulocytes and significantly
increasing peripheral leukocytes and neutrophils (18). Furthermore, urothelial carcinoma may
also lead to bone marrow microinvasion, which can stimulate
myelofibrosis and lead to extramedullary hematopoiesis, further
elevating peripheral leukocyte counts (7,19).
Chemotherapy may be considered for patients who
refuse surgical intervention or for whom surgery is not feasible.
However, in the present case, given the patient's impaired renal
function (creatinine level of 1,006 µmol/l as of October 2022) and
the rapid rise in body temperature, the patient was considered
unsuitable for gemcitabine and cisplatin chemotherapy. Surgical
resection is generally regarded as the most effective treatment for
bladder cancer complicated by a leukemoid reaction (20,21),
as the aforementioned atypical clinical features often resolve
following tumor removal (22,23).
Nevertheless, surgical resection does not consistently ensure
prolonged remission. Following the onset of a leukemoid reaction,
patients frequently succumb within a few months despite surgical
intervention (19,24–28).
In urological malignancies, a leukemoid reaction
might be associated with increased G-CSF expression in patients
with bladder cancer, a phenomenon less commonly observed in other
malignancies (11). Under normal
physiological conditions, G-CSF stimulates the proliferation and
differentiation of neutrophil colony-forming cells and alters
several functions of mature neutrophils. During a leukemoid
reaction, increased G-CSF expression is associated with heightened
leukocytosis and promotes a growth advantage to bladder cancer
cells, infiltration into the muscular layer and metastasis to
surrounding lymph nodes (29). A
total of ~9% of patients with bladder cancer exhibit increased
G-CSF levels, and these patients demonstrate a lower overall 5-year
survival rate compared with those without increased G-CSF levels
(30). Tachibana et al
(31) reported elevated G-CSF
levels and expression in the cancer cells of a 76-year-old patient
with urothelial carcinoma complicated by a leukemoid reaction.
Furthermore, this study confirmed that exogenous administration of
G-CSF can stimulate bladder cancer cell proliferation, an effect
that can be inhibited by the administration of anti-G-CSF
antibodies (31).
The autocrine and paracrine mechanisms of CSF in
bladder cancer may be associated with the re-arrangement of the
G-CSF gene occurring within one of the alleles (32) and intrinsic activation of nuclear
factors that work on the promoter region of the G-CSF gene
(33). The G-CSF/G-CSFR biological
axis promotes the expression of STAT3 protein, thereby promoting
the growth of bladder cancer cells. In addition, G-CSF can induce
the expression of β-1 integrin in bladder cancer cells, with β-1
integrin further acting on fibronectin and laminin, thereby
promoting the invasion and metastasis of bladder cancer cells
(34,35). In conclusion, cases of bladder
cancer complicated by a leukemoid reaction are rarely reported
clinically. The literature suggests that even with radical
cystectomy, the prognosis remains poor. Monitoring of G-CSF levels
in blood can help to make predictions of the patient's health
condition. Blocking the G-CSF signaling pathway might serve as a
future therapeutic target for bladder cancer complicated by a
leukemoid reaction.
Acknowledgements
Not applicable.
Funding
The present study was financially supported by the Innovation
Project of the Industrial Development Center of Shanghai University
of Traditional Chinese Medicine (grant no. YYKC-2021-01-143).
Availability of data and materials
The data generated in the present study may be
requested from the corresponding author.
Authors' contributions
JLi conceptualized and designed the present study.
TH, JX, CChu, YS, JLu, PA and CChe acquired the data. TH and YH
analyzed and interpreted the data. TH and JX drafted and critically
revised the manuscript. All authors read and approved the final
version of the manuscript. JLi and TH confirm the authenticity of
all the raw data.
Ethics approval and consent to
participate
All procedures were approved (approval no. 20230015)
by the Shanghai Municipal Hospital of Traditional Chinese Medicine,
Shanghai University of Traditional Chinese Medicine (Shanghai,
China).
Patient consent for publication
Written informed consent has been obtained from the
relative of the patient for the publication of images and data.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Gao B, Li X, Li S, Wang S, Wu J and Li J:
Pan-cancer analysis identifies RNA helicase DDX1 as a prognostic
marker. Phenomics. 2:33–49. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Burger M, Catto JW, Dalbagni G, Grossman
HB, Herr H, Karakiewicz P, Kassouf W, Kiemeney LA, La Vecchia C,
Shariat S and Lotan Y: Epidemiology and risk factors of urothelial
bladder cancer. Eur Urol. 63:234–241. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Chavan S, Bray F, Lortet-Tieulent J,
Goodman M and Jemal A: International variations in bladder cancer
incidence and mortality. Eur Urol. 66:59–73. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Van Osch FH, Jochems SH, Van Schooten FJ,
Bryan RT and Zeegers MP: Quantified relations between exposure to
tobacco smoking and bladder cancer risk: A meta-analysis of 89
observational studies. Int J Epidemiol. 45:857–870. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Yu G, Ji H, Meng C, Huang Y, Gao G, Liu C,
Wang S, Zhang L and Ju J: Esophageal adenocarcinoma with leukemoid
reaction: A case report. J Cardiothorac Surg. 14:662019. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Qing L, Xiang T, Guofu Z and Weiwei F:
Leukemoid reaction in cervical cancer: A case report and review of
the literature. BMC Cancer. 14:6702014. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Kumar AKL, Satyan MT, Holzbeierlein J,
Mirza M and Van Veldhuizen P: Leukemoid reaction and autocrine
growth of bladder cancer induced by paraneoplastic production of
granulocyte colony-stimulating factor-a potential neoplastic
marker: A case report and review of the literature. J Med Case Rep.
8:1472014. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Tachibana M and Murai M: G-CSF production
in human bladder cancer and its ability to promote autocrine
growth: A review. Cytokines Cell Mol Ther. 4:113–120.
1998.PubMed/NCBI
|
|
9
|
Ishida K, Yuhara K and Kanimoto Y: A case
of bladder tumor producing granulocyte colony-stimulating factor.
Hinyokika Kiyo. 50:253–256. 2004.(In Japanese). PubMed/NCBI
|
|
10
|
Paner GP, Stadler WM, Hansel DE, Montironi
R, Lin DW and Amin MB: Updates in the eighth edition of the
tumor-node-metastasis staging classification for urologic cancers.
Eur Urol. 73:560–569. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Shapiro DD, Potretzke A and Downs TM:
Leukemoid reaction: A rare paraneoplastic syndrome in bladder
cancer associated with a grave prognosis. Urology. 83:274–277.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Harsha BS, Manikandan R, Sreerag KS,
Dorairajan LN, Rajesh NG and Kar R: Eosinophilic leukemoid reaction
with eosinophilic tumor tissue infiltration as an extremely poor
prognostic factor in urinary bladder cancer-a known entity
revisited. Pathol Oncol Res. 26:2817–2819. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Mittal N, Gupta R, Rahman K, Singh P,
Panda I and Nityanand S: A case of paraneoplastic hyperleukocytosis
closely mimicking chronic neutrophilic leukemia. J Cancer Res Ther.
16:186–188. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Iyengar V, Meyer A, Stedman E, Casale S,
Kalsi S, Hale AJ and Freed JA: A predictive algorithm for
discriminating myeloid malignancies and leukemoid reactions. Am J
Med. 137:658–665. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Stricker PD and Grant AB: Relative value
of antibiotics and catheter care in the prevention of urinary tract
infection after transurethral prostatic resection. Br J Urol.
61:494–497. 1988. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Thompson DB, Siref LE, Feloney MP, Hauke
RJ and Agrawal DK: Immunological basis in the pathogenesis and
treatment of bladder cancer. Expert Rev Clin Immunol. 11:265–279.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Jang GY, Lee JW, Kim YS, Lee SE, Han HD,
Hong KJ, Kang TH and Park YM: Interactions between tumor-derived
proteins and Toll-like receptors. Exp Mol Med. 52:1926–1935. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Asano S: Human granulocyte
colony-stimulating factor: Its basic aspects and clinical
applications. J Pediatr Hematol Oncol. 13:400–413. 1991. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Perez FA, Fligner CL and Yu EY: Rapid
clinical deterioration and leukemoid reaction after treatment of
urothelial carcinoma of the bladder: Possible effect of granulocyte
colony-stimulating factor. J Clin Oncol. 27:e215–e217. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Gaisa NT, Braunschweig T, Reimer N,
Bornemann J, Eltze E, Siegert S, Toma M, Villa L, Hartmann A and
Knuechel R: Different immunohistochemical and ultrastructural
phenotypes of squamous differentiation in bladder cancer. Virchows
Arch. 458:301–312. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Matsuzaki K, Okumi M, Kishimoto N, Yazawa
K, Miyagawa Y, Uchida K and Nonomura N: A case of bladder cancer
producing granulocyte colony-stimulating factor and interleukin-6
causing respiratory failure treated with neoadjuvant systemic
chemotherapy along with sivelestat. Hinyokika Kiyo. 59:443–447.
2013.(In Japanese). PubMed/NCBI
|
|
22
|
Hirasawa K, Kitamura T, Oka T and
Matsushita H: Bladder tumor producing granulocyte
colony-stimulating factor and parathyroid hormone related protein.
J Urol. 167:21302002. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Stav K, Leibovici D, Siegel YI and Lindner
A: Leukemoid reaction associated with transitional cell carcinoma.
Isr Med Assoc J. 4:223–224. 2002.PubMed/NCBI
|
|
24
|
Fernández J, Clària J, Amorós A, Aguilar
F, Castro M, Casulleras M, Acevedo J, Duran-Güell M, Nuñez L, Costa
M, et al: Effects of albumin treatment on systemic and portal
hemodynamics and systemic inflammation in patients with
decompensated cirrhosis. Gastroenterology. 157:149–162. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Hoshi S, Numahata K, Morozumi K, Katumata
Y, Kuromoto A, Takai Y, Hoshi K, Bilim V and Sasagawa I: Bladder
cancer metastasis producing beta-human chorionic gonadotropin,
squamous cell carcinoma antigen, granulocyte-colony stimulating
factor, and parathyroid hormone-related protein. IJU Case Rep.
2:47–50. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Vaidyanathan S, Mansour P, Ueno M,
Yamazaki K, Wadhwa M, Soni BM, Singh G, Hughes PL, Watson ID and
Sett P: Problems in early diagnosis of bladder cancer in a spinal
cord injury patient: Report of a case of simultaneous production of
granulocyte colony stimulating factor and parathyroid
hormone-related protein by squamous cell carcinoma of urinary
bladder. BMC Urol. 2:82002. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Lin HC, Chai CY, Su YC, Wu WJ and Huang
CH: Leukemoid reaction resulting from granulocyte
colony-stimulating factor producing urothelial carcinoma of the
renal pelvis. Kaohsiung J Med Sci. 23:89–92. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Sawazaki H, Taki Y and Takeuchi H:
Granulocyte colony-stimulating factor (G-CSF) producing bladder
cancer subsequently developed from recurrent non-muscle invasive
bladder cancer. Int J Urol. 17:741–742. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Chakraborty A and Guha S: Granulocyte
colony-stimulating factor/granulocyte colony-stimulating factor
receptor biological axis promotes survival and growth of bladder
cancer cells. Urology. 69:1210–1215. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Mizutani Y, Okada Y, Terachi T, Kakehi Y
and Yoshida O: Serum granulocyte colony-stimulating factor levels
in patients with urinary bladder tumour and various urological
malignancies. Br J Urol. 76:580–586. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Tachibana M, Miyakawa A, Tazaki H,
Nakamura K, Kubo A, Hata J, Nishi T and Amano Y: Autocrine growth
of transitional cell carcinoma of the bladder induced by
granulocyte-colony stimulating factor. Cancer Res. 55:3438–3443.
1995.PubMed/NCBI
|
|
32
|
Demetri GD and Griffin JD: Granulocyte
colony-stimulating factor and its receptor. Blood. 78:2791–2808.
1991. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Nishizawa M, Tsuchiya M, Watanabe-Fukunaga
R and Nagata S: Multiple elements in the promoter of granulocyte
colony-stimulating factor gene regulate its constitutive expression
in human carcinoma cells. J Biol Chem. 265:5897–5902. 1990.
View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Chakraborty A, White SM and Guha S:
Granulocyte colony-stimulating receptor promotes
beta1-integrin-mediated adhesion and invasion of bladder cancer
cells. Urology. 68:208–213. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Chakraborty A, White SM and Lerner SP:
Granulocyte colony-stimulating factor receptor signals for
beta1-integrin expression and adhesion in bladder cancer. Urology.
63:177–183. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Turalic H, Deamant FD and Reese JH:
Paraneoplastic production of granulocyte colony-stimulating factor
in a bladder carcinoma. Scand J Urol Nephrol. 40:429–432. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Kato T, Yasuda K, Iida H, Watanabe A,
Fujiuchi Y, Miwa S, Imura J and Komiya A: Trousseau's syndrome
caused by bladder cancer producing granulocyte colony-stimulating
factor and parathyroid hormone-related protein: A case report.
Oncol Lett. 12:4214–4218. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
He H, Zhang Z, Ge J and Zhou W: Leukemoid
reaction associated with transitional cell carcinoma: A case report
and literature review. Niger J Clin Pract. 17:391–394. 2014.
View Article : Google Scholar : PubMed/NCBI
|