Introduction
Pulmonary lymphoepithelioma-like carcinoma (PLELC)
is a type of lung cancer that accounts for <1% of all lung
cancers. It belongs to non-small cell lung cancer (NSCLC). The
stage at diagnosis is usually earlier than that of other NSCLC
subtypes; thus, the prognosis is slightly better than that of
others. PLELC is highly associated with Epstein-Barr virus (EBV)
and is more common in middle-aged Asian women. Histological
classification is similar to undifferentiated nasopharyngeal
carcinoma (1), both of which are
non-keratinized squamous cell carcinomas. Early surgical resection
is the main treatment method (2)
and chemotherapy also plays an important role. In recent years, the
development of lifeomics has brought immunotherapy and targeted
therapy into the vision of clinicians (3), making the precision treatment of rare
tumors like PLELC possible.
PLELC is characterized by high programmed death
ligand 1 (PD-L1) expression and lack of a driver gene: A 2020 study
of 86 patients with PLELC showed that the PD-L1 expression rate was
up to 78.9% (4). Among them, 47
cases were tested genetically, and only PD-L1 was positive. The
proportion of PD-L1 expression in other studies with small sample
sizes may not be statistically reliable. In 2022, a real-world
study from five cancer centers in China was reported (5). A total of 770 patients with LELC were
included, among which 598 were patients with PLELC. This was the
largest sample size reported so far. Among 770 cases with LELC, 34
cases were tested for PD-L1, of which 25 cases were positive
(73.5%) and 16 cases had high expression. At the same time, 305
cases were subjected to gene detection: 10 cases had an EGFR
mutation, 3 cases had an anaplastic lymphoma kinase rearrangement,
7 cases had a tumor protein 53 gene mutation, 2 cases had a Notch
receptor mutation and >90% of patients had no driver gene.
Compared to a brief review in 2020 (6), the results of both studies are largely
similar. Notably, the former included multi-site LELC, while the
latter focused solely on PLELC and all had negative KRAS mutations.
The latter is a clearly more valuable reference, and in addition,
selection bias in retrospective studies cannot be ignored. Based on
these research findings, the relationship between high PD-L1
expression and prognosis in PLELC is yet to be fully understood.
The correlation between high expression of PD-L1 and prognosis in
PLELC remains elusive, as this is based solely on case reports,
which often contradict each other. On the one hand, a meta-analysis
in 2017 showed that PD-L1 expression is an unfavorable biomarker in
PLELC and NSCLC, mainly manifested by reducing the overall
survival. Additionally, increased PD-L1 expression is not
associated with KRAS mutations (7).
On the other hand, a 60-year-old Japanese woman with EBV-positive
PLELC also had high PD-L1 expression. And after surgery, this
patient was followed up for 30 months without recurrence (8). In clinical practice, the active use of
immune checkpoint inhibitors is generally recommended and numerous
patients appear to benefit from it (9). The impact of high PD-L1 expression on
the prognosis of PLELC remains elusive and further research is
still needed to elucidate its potential relationship. In pulmonary
PLELC, the absence of mutations (or wild-type status) in relevant
genes is a common finding. The co-occurrence of high PD-L1
expression and KRAS mutations is even rarer, with no documented
cases in recent literature. KRAS mutations often mean that
anti-EGFR treatment is ineffective, with fewer options and worse
prognosis (10). Therefore, special
efforts are needed when treating such patients, and identifying the
patients and providing appropriate treatment plans are crucial.
Case report
In February 2021 (first visit), a 58-year-old woman
was admitted to a local hospital due to cough and sputum. Chest CT
revealed a 2-cm nodule in the middle lobe of the right lung. Due to
personal reasons, no special treatment was given. In November 2021,
the chest CT found that the nodule had increased to 3.8 cm, and
thus, the patient came to Sichuan Northern Medical College
Affiliated Hospital (Nanchong, China) for treatment. During the
course of the disease, the patient occasionally coughed with white
frothy sputum, without dyspnea or other discomforts. The Eastern
Cooperative Oncology Group score was 1 (11). No significant medical, personal,
marital, reproductive, menstrual or family history was found. On
November, 2021, an enhanced chest CT scan, followed by a
high-resolution CT and reconstruction, were performed. The CT scan
showed a plain scan with enhanced imaging: Soft tissue nodules in
the medial segment of the right middle lobe of the lung, mostly
tumor-related lesions, possibly lung cancer. No metastasis was
observed in the brain or bones. In November 2021, the preoperative
screening for infectious diseases suggested that hepatitis B
surface antigen (HBsAg) was >130 IU/ml (normal, <0.05 IU/ml)
and hepatitis B E antigen was 1,322 S/CO (normal, 0.000–0.009
S/CO). Hepatitis B E antibodies, hepatitis B surface and hepatitis
B core antibody were negative, and liver function tests indicated
that aspartate aminotransferase (AST) was 37 U/l (normal range,
13–25 U/l) and alanine aminotransferase (ALT) was 27 U/l (normal
range, 7–40 U/l). The diagnosis of HBV was consistent. However, HBV
DNA testing was not performed at this time, and further diagnosis
and treatment were not performed.
Surgery was scheduled for November 2021: Under
general anesthesia, the patient underwent thoracoscopic resection
of the right middle lobe of the lung, thoracoscopic intrathoracic
lymph node dissection, thoracoscopic adhesion lysis of the pleura
and closed thoracic drainage (intraoperative frozen section
pathology report from November 2021). Standard H&E staining
procedure: After tissue was fixed with 4% formaldehyde, 4–5 µm
paraffin sections were prepared. The staining steps included
hematoxylin nuclear staining for 5–10 min, followed by
differentiation and bluing, then eosin cytoplasmic staining for 1–3
min, and finally dehydration and mounting. Observation was
performed using a brightfield microscope with typical
magnifications of 40× (scale bar, ~20 µm) or 200× (scale bar, ~5
µm). The diagnosis for the ‘right middle lobe’ sample was cancer,
suspected as squamous cell carcinoma. In December 2021 (this was
from an analysis of the previous sample from November), the
pathology report indicated the following: ‘Right middle lobe’
invasive cancer with pleural invasion, no residual cancer at the
bronchial, vascular or lung ends. Immunohistochemistry (IHC):
Thyroid transcription factor-1 (TTF-1) (−), novel aspartic
proteinase A (Napsin A) (−), cytokeratin 5/6 (CK5/6) (+), P40
protein (P40) (+), Epstein-Barr virus (EBV)-encoded small RNA
(EBER) (+), proliferation index (Ki-67) (+, ~50%), supporting a
diagnosis of PLELC of the right middle lobe (Fig. 1). IHC protocol summary: Samples were
fixed in 10% neutral buffered formalin for 24–48 h at room
temperature (RT). After paraffin-embedding, they were sectioned at
4–5 µm. Following heat-induced epitope retrieval (e.g., citrate
buffer, 95–100°C, 20 min), samples were blocked with 5% normal
serum (RT, 1 h). They were then incubated with primary antibody
(4°C overnight) and then with HRP-conjugated secondary antibody
(RT, 1 h). The following primary antibodies were used: TTF-1 (cat.
no. 2509170599d; dilution, ready-to-use); Napsin A (cat. no.
250806074d; dilution, ready-to-use); CK5/6 (cat. no. 2509110744c6;
dilution, 1:100); P40 (cat. no. 2510291006e; dilution,
ready-to-use); Ki-67 (cat. no. 2511260731C7; dilution, 1:150; all
from Fuzhou Maixin Biotechnology Development Co., Ltd) and PD-L1
(cat. no. P04565CN-03; dilution, 1:50; Dako; Agilent Technologies,
Inc.). Secondary antibodies: General-purpose (cat. no. DD23;
dilution, ready-to-use; Xiamen Talent Biomedical Technology Co.,
Ltd.). Staining was visualized with diaminobenzidine and
counterstaining with hematoxylin was performed, followed by
observation with a light microscope.
 | Figure 1.Pathology and IHC of the pulmonary
lymphoepithelioma-like carcinoma. (A and B) Histological analysis.
(A) heterologous cells were arranged in clusters (magnification,
×40); (B) the nuclei of heterologous cells were vacuolated and the
nucleoli were clear (magnification, ×200; H&E staining); the
arrows point at heterologous cells. (C-I) IHC analysis. (C)
Cytoplasmic CK5/6 was positive, the arrows point at positive CK5/6
cytoplasm; (D) nuclear P40 was positive; the arrows point at
positive P40 nuclear; (E) thyroid transcription factor-1 was
negative; (F) Napsin A was negative; (G) nuclear EBER was positive;
the arrows point at positive EBER nuclear; (H) proliferation index
Ki-67 positive ~50%, the arrows point at positive Ki-67 nuclear;
(I) PD-L1 detection microscopic image: Tumor proportion score=55%
(magnification, ×100), the arrows point at positive PD-L1 nuclear.
CK, cytokeratin; PD-L1, programmed cell death ligand 1; EBER,
Epstein-Barr virus-encoded small RNA; IHC,
immunohistochemistry. |
Hybridization in situ: EBER monoclonal
antibody (cat. no. 25080502; dilution, ready-to-use; Beijing
Zhongshan Jinqiao Biotechnology Co., Ltd.). The patient only
underwent in situ hybridization (ISH) testing on the lung
tumor tissue resected by surgery, and no fluorescence ISH (FISH)
testing was performed. ISH was performed using the EBER Detection
Kit (In Situ Hybridization; product code, ISH-7001; Beijing
Zhongshan Jinqiao Biotechnology Co., Ltd.) according to the
manufacturer's instructions. Previous experience suggests that FISH
is not a routine testing method for lung cancer (12).
Histological examination revealed infiltration by
numerous lymphocytes and plasma cells. IHC analysis confirmed
squamous cell carcinoma (positive for cytoplasmic CK5/6 and nuclear
P40) and ruled out an adenocarcinoma component (negative for TTF-1
and Napsin A; if present, the diagnosis would be adenosquamous
carcinoma). Serological testing revealed EBV antibody
seropositivity, which is consistent with an Epstein-Barr virus
infection. Together, these findings fulfill the gold-standard
criteria for diagnosing pulmonary lymphoepithelioma-like carcinoma
(PLELC) (1).
Postoperative staging was pT2aN0M0, stage IB
(American Joint Committee on Cancer, 8th edition) (13). In January 2022, a targeted drug
therapy 10 gene test (14) was
performed, revealing a KRAS gene mutation (atypical driving gene).
The PD-L1 expression volume was 55% [tumor proportion score: High
expression (15)] (Fig. 2). Based on the patient's clinical
characteristics and willingness, adjuvant chemotherapy combined
with immunotherapy was planned postoperatively. Prior to the start
of adjuvant chemoradiotherapy, the patient was considered to have
an active HBV infection and was not treated (not given entecavir
antiviral treatment). Chemotherapy combined with immunotherapy
could potentially re-activate HBV, so an HBV DNA test
(9.600×107 IU/ml) was performed in January 2022, and
entecavir antiviral therapy was initiated once daily, 0.5 mg each
time p.o. In February 2022, a follow-up HBV DNA test showed mild
elevations in transaminase levels (AST: 75 U/l; ALT: 94 U/l) and
the HBV DNA level was 9.220×105 IU/ml (normal, <20
IU/ml). Due to the persistent nature of the HBV, it can't be
completely eradicated; therefore, long-term maintenance therapy
with entecavir is required (16).
The PLELC surgery was performed in November, 2021, and an EBV DNA
test was conducted in January 2022, and the result was
<4.000×102 copies/ml. This means that the patient's
EBV DNA had become undetectable after surgery (at least
4004.000×102 copies/ml is required for detection). As
cancer treatment, from January 2022, a penpulimab 200 mg
immunotherapy combined with TC regimen (albumin-taxol 300 mg +
carboplatin 500 mg, every three weeks) was initiated, and until May
2022, six cycles were given. In June 2022, a chest plain scan with
enhanced CT revealed no definite signs of tumor recurrence nor
metastasis, with no significant changes compared to that performed
in March 2022. From June 2022 to April 2023, the patient received
nine cycles of immunomodulatory maintenance therapy with penpulimab
200 mg ivgtt q3w. During the treatment, there were no significant
adverse events, and there was no recurrence or metastasis of the
tumor. Since April 2023, regular follow-ups have been conducted,
with the last follow-up in February 2025. No recurrence or
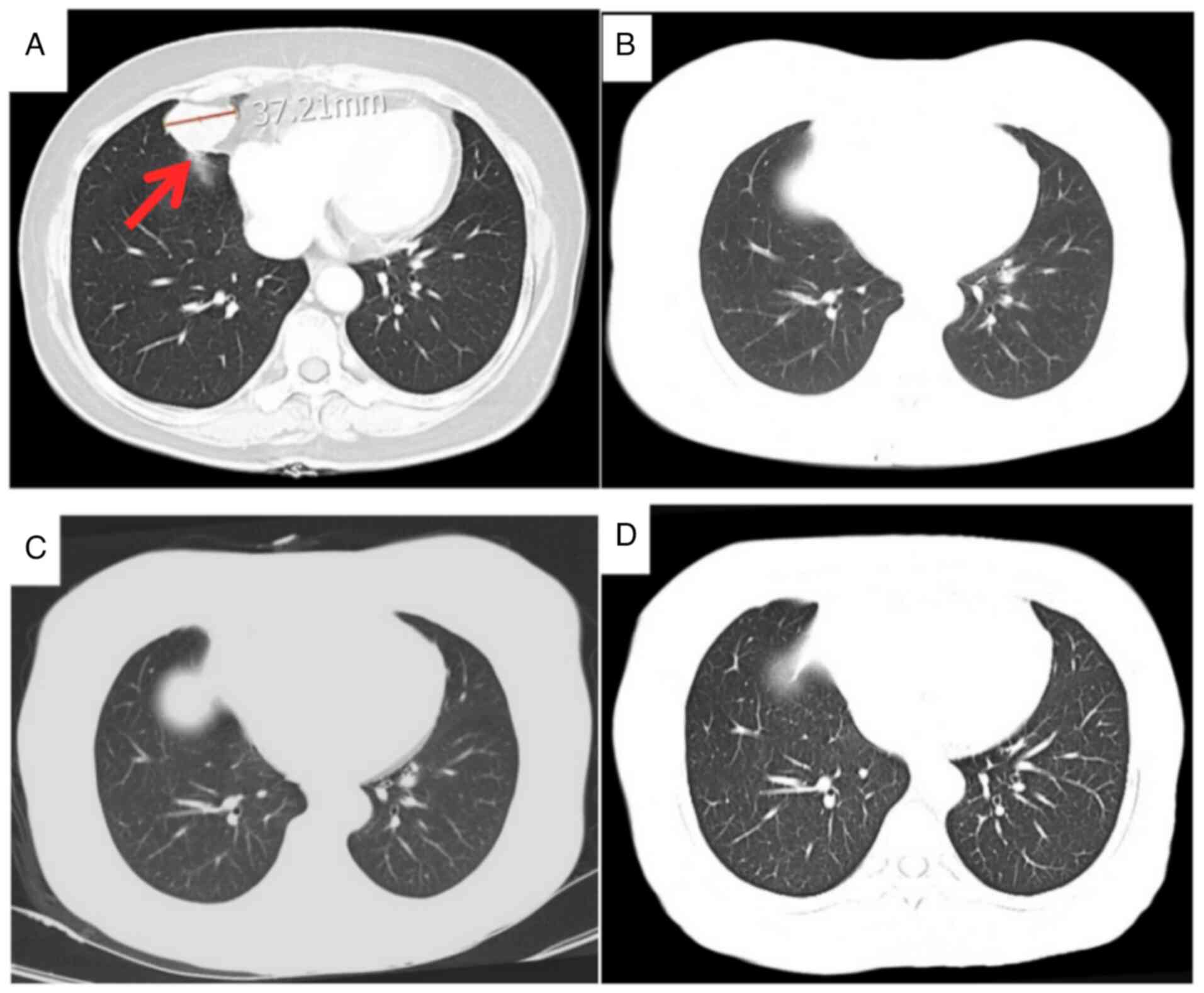
metastasis was found (representative CT images are shown in
Fig. 2). The preoperative image
from November 2021 shows a lesion measuring 37.21 mm, as indicated
by the arrow (Fig. 2A). Before the
first postoperative chemotherapy session in January 2022, the
lesion was no longer visible (Fig.
2B). Following the first postoperative chemotherapy session in
March 2022, no obvious recurrence or metastasis was observed
(Fig. 2C). By February 2025, there
remained no obvious recurrence or metastasis (Fig. 2D). For patients with pulmonary
lymphoepithelioma-like carcinoma undergoing curative treatment,
long-term follow-up plans are typically based on time staging:
during the first 3 years after treatment (high-risk period), a
medical history review, physical examination, and enhanced chest CT
scan are recommended every 6 months. Therefore, the next follow-up
appointment is expected to be in October 2025, and the patient
didn't complain of any abnormalities by telephone follow-up in July
2025.
Discussion
LELC is a rare tumor that can occur in various
locations, including the nasopharynx, tonsils, tongue, digestive
system, bladder and lungs. In 2015, the World Health Organization
noted that the lungs are one of the most common sites for this type
of cancer, with 90% of patients testing positive for EBV (1,2,5,17).
A positive EBER result in IHC is the gold standard for diagnosing
this condition and serves as a key method for differentiation
(1). The treatment and prognosis of
the disease are influenced by several factors, including the tumor
location (18) the PD-L1 expression
status and the type of gene mutation (19). The case of the present study
involved PLELC in the middle lobe of the right lung, similar to
other types of lung cancer, with the main symptom being a cough.
Due to the lack of specific imaging features (20) chest CT can only provide a
preliminary diagnosis of lung cancer. Pathology confirms the
characteristics of squamous cell carcinoma, and a definitive
diagnosis ultimately relies on IHC. The pathology results for the
present case indicated the presence of atypical cell proliferation
arranged in nests, suggesting squamous cell carcinoma. High Ki-67
expression was present, with ~50% of cell nuclei being positive.
Ki-67 is a tumor growth marker and also has prognostic significance
(21,22).
Currently, there is no specific treatment for PLELC.
Early-stage cases are typically treated with surgery, while those
in the intermediate to late stages often follow the treatment
protocols for NSCLC (23). This
case has certain key features: i) EBV was positive before surgery
but turned negative after surgery. ii) High PD-L1 expression
combined with KRAS mutation and chronic HBV infection. EBV-negative
LELC has been observed in Caucasians (5), but this case differs from traditional
clinical epidemiology, as it involves a middle-aged Asian woman
whose EBV test was positive at the preoperative, while negative at
the postoperative stage. The mechanisms of EBV infection and tumor
development are not fully understood. A 2018 study suggested that
the pathogenesis of related malignant tumors may be closely linked
to unique epigenetic dysregulation (24). Another 2025 study found that in
nasopharyngeal carcinoma, EBV infection is associated with AKT3
signaling, but this correlation has not been confirmed in PLELC
(25). Postoperative IHC confirmed
EBER positivity, which helped diagnose the patient, demonstrating
the competence of our pathologists and preventing misdiagnosis as
common lung squamous cell carcinoma (26), which could have affected treatment.
Preoperative biopsy or routine blood EBV testing for lung
malignancies can reduce misdiagnosis rates and provide more
reliable data for clinical practice and academic research. Studies
have shown that neoadjuvant immunotherapy is a viable strategy
(27), which necessitates
preoperative biopsies. One of the interesting aspects of this case
is the change in EBV. The clinical value of dynamic monitoring of
EBV (preoperative positivity → postoperative negativity) urgently
needs validation. This viral kinetic feature may reflect immune
reconstitution or a reduction in tumor burden (28), but there is currently a lack of
prospective studies to confirm its association with long-term
survival rates. The patient's PD-L1 expression level was as high as
55%; it is a key factor in improving the prognosis of
immunotherapy, which has been validated in most studies. It marks
that the patient's immune system has identified and is attempting
to attack the tumor (29). Common
first-line immunotherapeutic agents include pembrolizumab,
sintilimab and durvalumab, while second-line immunotherapy often
involves Nivolumab. Combining these with chemotherapy can enhance
treatment efficacy (30).
Penpulimab, a novel domestic PD-L1 inhibitor (31), offers similar efficacy and better
safety compared to other immunotherapeutic drugs (32). When combined with chemotherapy, it
can be used as a first-line treatment for locally advanced or
metastatic squamous cell carcinoma (33). To date, the patient of the present
study has survived for >44 months without any significant
immune-related adverse events, making it a valuable reference for
the clinical application of Penpulimab in such patients.
Considering the potential allergic reactions to paclitaxel and the
renal toxicity of cisplatin, the patient opted for albumin-bound
paclitaxel combined with carboplatin as the treatment regimen,
which is the most common and effective platinum-based double-drug
chemotherapy regimen for NSCLC (34).
Under the guidance principle of precision tumor
treatment, targeted therapy is a necessary consideration. The
patient had a KRAS mutation and the possibility of targeted therapy
exists. However, in clinical practice, drugs targeting KRAS
mutations are not satisfactory in terms of efficacy and safety
(35), which may be due to the
widespread and complex metabolic changes caused by the reduction of
guanosine triphosphate activity of the KRAS gene (36). He et al (37) compared 22 studies and found that
chemotherapy combined with immunotherapy can serve as the
first-line treatment for KRAS-mutated NSCLC, and the combination
with anti-angiogenesis may offer better outcomes. Currently, KRAS
inhibitors (targeting G12C) are in clinical trials, such as the
GFH925 study (38), the LY3537982
study (39) and the AseBreaK 101
study (40). As clinical trials
progress, it is possible that KRAS inhibitors could become the
first-line treatment for KRAS-mutated NSCLC and potentially for
PLELC. The patient refused targeted therapy due to personal
reasons. As a clinical physician, it is also important to consider
the impact of HBV on treatment and prognosis. Research data
indicate that immunotherapy can indeed lead to the reactivation of
the HBV in certain HBsAg seropositive patients (41); even for neoadjuvant immunotherapy,
it is essential to first screen for HBsAg. Recently, researchers
have developed a model to predict the prognosis of PLELC (42). This model showed that positivity for
HBsAg is significantly associated with overall survival (hazard
ratio=2.028, P=0.023), making it an independent risk factor. The
study also highlights that immunotherapy is a significant option
for improving the prognosis of PLELC. The patient in question had a
high DNA load of HBV infection, which was controlled after
treatment, but continued monitoring is necessary in subsequent
treatments. Unfortunately, current clinical studies generally lack
patient cohorts that simultaneously have KRAS mutations, high PD-L1
expression (≥50%) and HBsAg+. The immune treatment response for
this triple-feature population has remained to be clearly defined,
and there is an urgent need for prospective large-scale trials to
verify its safety and efficacy.
In summary, the present study reported a case of
PLELC with KRAS mutations, high PD-L1 expression and concurrent HBV
infection. The patient received adjuvant immunotherapy plus
chemotherapy and maintenance immunotherapy post-surgery, achieving
an ongoing progression-free survival of >44 months. This
suggests that adjuvant immunotherapy plus chemotherapy, along with
maintenance immunotherapy, could be an option for treating such
patients. To date, one study has been conducted (43), but more data and individualized
treatment are still needed.
Acknowledgements
Not applicable.
Funding
This work was financially supported by the National Natural
Science Foundation of China (grant no. 82202976) and the Dr Pioneer
Foundation of North Sichuan Medical College (grant no.
CBY21-QD18).
Data availability statements
The data generated in the present study may be
requested from the corresponding author.
Authors' contributions
JinZ was responsible for methodology, investigation
[the systematic collection, collation, and analysis of the clinical
data for this case report (including medical history, laboratory
tests, imaging, and pathological data), participation in clinical
discussions regarding the case, and review of relevant literature
to provide the core data and rationale for the report], language
editing, obtaining materials, visualization and writing-original
draft. QX, JiaZ, RZ and YZ were involved in methodology,
investigation [the systematic collection, collation, and analysis
of the clinical data for this case report (including medical
history, laboratory tests, imaging, and pathological data),
participation in clinical discussions regarding the case, and
review of relevant literature to provide the core data and
rationale for the report] and obtaining materials, writing-original
draft, language editing and providing general supervision. ZL
performed investigation [the systematic collection, collation, and
analysis of the clinical data for this case report (including
medical history, laboratory tests, imaging, and pathological data),
participation in clinical discussions regarding the case, and
review of relevant literature to provide the core data and
rationale for the report], methodology, supervision,
writing-original draft and writing-review & editing, and
obtained funding. All authors have read and approved the final
manuscript. All authors have verified and confirmed the
authenticity of the original data.
Ethics approval and consent to
participate
The study was approved by the Medical Ethics
Committee of the Affiliated Hospital of North Sichuan Medical
College (Nanchong, China; approval no. 2025ER426-1).
Patient consent for publication
The patient provided written informed consent for
the publication of the clinical data and images.
Competing interests
The authors declare that they have no competing
interests.
Use of artificial intelligence tools
During the preparation of this work, an AI tool,
Yuanbao v2.57.10.150 (https://yuanbao.tencent.com), was used to improve the
readability and language of the manuscript or to generate images,
and subsequently, the authors revised and edited the content
produced by the AI tools as necessary, taking full responsibility
for the ultimate content of the present manuscript.
References
|
1
|
Qin Y, Gao G, Xie X, Zhu Z, Guan W, Lin X,
Xie Z, Ming O, Chen R, Zhong N, et al: Clinical features and
prognosis of pulmonary Lymphoepithelioma-like carcinoma: Summary of
Eighty-five cases. Clin Lung Cancer. 20:e329–e337. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Lin Z, Fu S, Zhou Y, Zhang X, Chen C, He
LN, Li H, Wang Y, Chen T, Zhang L and Hong S: First-line
platinum-based chemotherapy and survival outcomes in locally
advanced or metastatic pulmonary lymphoepithelioma-like carcinoma.
Lung Cancer. 137:100–107. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Zhang J, Lin L, Li W and Guo J: Role of
the ‘inflammation-immunity-abolism’ network in non-small cell lung
cancer: A multi-omics analysis. Discov Oncol. 16:8472025.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Chen J, Gu C, Chen X, Dai C, Zhao S, Xie
H, Fei K and Chen C: Clinicopathological and prognostic analyses of
86 resected pulmonary lymphoepithelioma-like carcinomas. J Surg
Oncol. 123:544–552. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Chen M, Chen Y and Fang X: Clinical
features and treatment outcome of lymphoepithelioma-like carcinoma
from multiple primary sites: A population-based, multicentre,
real-world study. BMC Pulm Med. 22:3602022. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Hu Y, Ren S, Liu Y, Han W and Liu W:
Pulmonary Lymphoepithelioma-Like Carcinoma: A Mini-review. Onco
Targets Ther. 13:3921–3929. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Zhang M, Li G and Wang Y and Wang Y, Zhao
S, Haihong P, Zhao H and Wang Y: PD-L1 expression in lung cancer
and its correlation with driver mutations: A meta-analysis. Sci
Rep. 7:102552017. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Sasaki A, Kato T, Ujiie H, Cho Y, Sato M
and Kaji M: Primary pulmonary lymphoepithelioma-like carcinoma with
positive expression of Epstein-Barr virus, PD-L1: A case report.
Int J Surg Case Rep. 79:431–435. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Zhong YM, Yin K, Chen Y, Xie Z, Lv ZY,
Yang JJ, Yang XN, Zhou Q, Wang BC, Zhong WZ, et al: PD-1/PD-L1
combined with LAG3 is associated with clinical activity of immune
checkpoint inhibitors in metastatic primary pulmonary
lymphoepithelioma-like carcinoma. Front Immunol. 13:9518172022.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Yin K, Feng HB, Li LL, Chen Y, Xie Z, Lv
ZY, Guo WB, Lu DX, Yang XN, Yan WQ, et al: Low frequency of
mutation of epidermal growth factor receptor (EGFR) and arrangement
of anaplastic lymphoma kinase (ALK) in primary pulmonary
lymphoepithelioma-like carcinoma. Thorac Cancer. 11:346–352. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Sok M, Zavrl M, Greif B and Srpčič M:
Objective assessment of WHO/ECOG performance status. Support Care
Cancer. 10:3793–3798. 2019.PubMed/NCBI
|
|
12
|
Becnel D, Abdelghani R, Nanbo A, Avilala
J, Kahn J, Li L and Lin Z: Pathogenic role of Epstein-Barr virus in
lung cancers. Viruses. 13:8772021. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Kutob L and Schneider F: Lung cancer
staging. Surg Pathol Clin. 13:57–71. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Wu YX, Zhang WL, Wang TM, Liao Y, Zhang
YJ, Xiao RW, Jia YJ, Wu ZY, Deng CM, Yang DW, et al: Genomic
landscapes of Epstein-Barr virus in pulmonary
Lymphoepithelioma-Like carcinoma. J Virol. 96:e01693212022.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Zhao X, Bao Y, Meng B, Xu Z, Li S, Wang X,
Hou R, Ma W, Liu D, Zheng J, et al: From rough to precise: PD-L1
evaluation for predicting the efficacy of PD-1/PD-L1 blockades.
Front Immunol. 13:9200212022. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Shi M, Sun WL, Hua YY, Han B and Shi L:
Effects of entecavir on hepatitis B virus covalently closed
circular DNA in hepatitis B e antigen-positive patients with
hepatitis B. PLoS One. 10:e01177412015. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Bégin LR, Eskandari J, Joncas J and
Panasci L: Epstein-Barr virus related lymphoepithelioma-like
carcinoma of lung. J Surg Oncol. 36:280–283. 1987. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Samaras MG, Koufopoulos NI, Mitsos S,
Dylja E, Monokrousou A, Tomos P, Panayiotides IG and Goutas D:
Lymphoepithelial carcinoma of the lung: A case report and review of
the literature. Cureus. 16:e703092024.PubMed/NCBI
|
|
19
|
Zhai X, Liu J, Lu D and Zhou Q:
Demographics, clinical features, and prognosis of rare
lymphoepithelioma-like carcinoma across different anatomic sites. J
Egypt Natl Canc Inst. 34:52022. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Mo Y, Shen J, Zhang Y, Zheng L, Gao F, Liu
L and Xie C: Primary lymphoepithelioma-like carcinoma of the lung:
Distinct computed tomography features and associated clinical
outcomes. J Thorac Imaging. 29:246–251. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Sun X and Kaufman PD: Ki-67: More than a
proliferation marker. Chromosoma. 127:175–186. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Menon SS, Guruvayoorappan C, Sakthivel KM
and Rasmi RR: Ki-67 protein as a tumour proliferation marker. Clin
Chim Acta. 491:39–45. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Zhang L, Zheng J, Fang LJ, Zhang L and
Zhou JY: Efficacy analysis of PD-1/PD-L1 inhibitors in the
treatment of advanced primary pulmonary lymphoepithelioma-like
carcinoma: 6 case reports and literature review. Zhonghua Zhong Liu
Za Zhi. 46:590–594. 2024.(In Chinese). PubMed/NCBI
|
|
24
|
Li L, Ma BBY, Chan ATC, Chan FKL, Murray P
and Tao Q: Epstein-Barr Virus-induced epigenetic pathogenesis of
Viral-associated Lymphoepithelioma-like carcinomas and natural
Killer/T-cell lymphomas. Pathogens. 7:632018. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Tan B, Xu K, Lyu Y, Liang Y, Liang R, Lei
K, Liang J, Huang J, Wang K, Wu D, et al: Single-cell analysis
reveals transcriptomic features and therapeutic targets in primary
pulmonary lymphoepithelioma-like carcinoma. Commun Biol. 8:3942025.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Yin CJ, Wang GJ, Su XM and Li D: Primary
pulmonary lymphoepithelioma-like carcinoma misdiagnosed as lung
squamous cell carcinoma: A case report. World J Clin Cases.
11:7876–7880. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Chen J, Fan L, Deng H, Li L and Li S:
Neoadjuvant immunochemotherapy-a promising strategy for primary
pulmonary lymphoepithelioma-like carcinoma. World J Surg Oncol.
22:3382024. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Li W, Yang C, Lv Z, Li J, Li Z, Yuan X, Wu
S, Yuan Y, Cui L, Lu J, et al: Integrating pre- and post-treatment
plasma Epstein-Barr virus DNA levels for better prognostic
prediction of Nasopharyngeal Carcinoma. J Cancer. 9:2715–2722.
2021. View Article : Google Scholar
|
|
29
|
Tang L, Chen N, He W, Zhou J, Zhang J, Lin
Z, Wang Z, Hao J and Lin F: The clinicopathological features and
prognosis of primary pulmonary lymphoepithelioma-like carcinoma: A
systematic review and meta-analysis. PLoS One. 15:e02407292020.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Zhou Y, Huang J, Lan J, Hu H, Yuan Z, Dong
L, Deng H, Yue LA, Xiao Y and Yang X: Comparison of first-line
immunotherapy efficacy between advanced lung squamous cell
carcinoma and pulmonary lymphoepithelioma-like carcinoma: A
propensity score matching multicenter study. J Cancer Res Ther.
19:1011–1018. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Song Y, Zhou K, Jin C, Qian Z, Hou M, Fan
L, Li F, Ding K, Zhou H, Li X, et al: Penpulimab for relapsed or
refractory classical Hodgkin lymphoma: A multicenter, Single-Arm,
pivotal phase I/II Trial (AK105-201). Front Oncol. 12:9252362022.
View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Huang Z, Pang X, Zhong T, Qu T, Chen N, Ma
S, He X, Xia D, Wang M, Xia M and Li B: Penpulimab, an
Fc-Engineered IgG1 Anti-PD-1 antibody, with improved efficacy and
low incidence of Immune-Related adverse events. Front Immunol.
13:9245422022. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Kaplon H, Chenoweth A, Crescioli S and
Reichert JM: Antibodies to watch in 2022. MAbs. 14:20142962022.
View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Zhou C, Wu L, Fan Y, Wang Z, Liu L, Chen
G, Zhang L, Huang D, Cang S, Yang Z, et al: Sintilimab plus
platinum and gemcitabine as First-Line treatment for advanced or
metastatic squamous NSCLC: Results from a randomized, Double-blind,
Phase 3 trial (ORIENT-12). J Thorac Oncol. 16:1501–1511. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Tamiya Y, Matsumoto S, Zenke Y, Yoh K,
Ikeda T, Shibata Y, Kato T, Nishino K, Nakamura A, Furuya N, et al:
Large-scale clinico-genomic profile of non-small cell lung cancer
with KRAS G12C: Results from LC-SCRUM-Asia study. Lung Cancer.
176:103–111. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
McCormick F: KRAS as a therapeutic target.
Clin Cancer Res. 21:1797–1801. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
He Q, Liu X, Jiang L, Liu P, Xuan W, Wang
Y, Meng R, Feng H, Lv S, Miao Q, et al: First-line treatments for
KRAS-mutant non-small cell lung cancer: Current state and future
perspectives. Cancer Biol Ther. 26:24414992025. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Gregorc V, González-Cao M, Salvagni S,
Koumarianou A, Gil-Bazo I, Maio M, Viteri S, Majem M, Gutiérrez V,
Bernabe Caro R, et al: KROCUS: A phase II study investigating the
efficacy and safety of fulzerasib (GFH925) in combination with
cetuximab in patients with previously untreated advanced KRAS G12C
mutated NSCLC. J Clin Oncol. 42:LBA85112024. View Article : Google Scholar
|
|
39
|
Burns TF, Dragnev KH, Fujiwara Y,
Murciano-Goroff YR, Lee DH, Hollebecque A, Koyama T, Cassier PA,
Italiano A, Heist RS, et al: Efficacy and safety of olomorasib
(LY3537982), a second-generation KRAS G12C inhibitor (G12Ci), in
combination with pembrolizumab in patients with KRAS G12C-mutant
advanced NSCLC. J Clin Oncol. 42:85102024. View Article : Google Scholar
|
|
40
|
Kuboki Y, Fakih M, Strickler J, Yaeger R,
Masuishi T, Kim EJ, Bestvina CM, Kopetz S, Falchook GS, Langer C,
et al: Sotorasib with panitumumab in chemotherapy-refractory
KRASG12C-mutated colorectal cancer: A phase 1b trial. Nat Med.
30:265–270. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Zhang X, Zhou Y, Chen C, Fang W, Cai X,
Zhang X, Zhao M, Zhang B, Jiang W, Lin Z, et al: Hepatitis B virus
reactivation in cancer patients with positive Hepatitis B surface
antigen undergoing PD-1 inhibition. J Immunother Cancer. 7:3222019.
View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Chen X, Liu T, Mo S, Yang Y, Chen X, Hong
S, Zhou T, Chen G, Zhang Y, Ma Y, et al: A Novel
Inflammation-Marker-Based prognostic model for advanced pulmonary
Lymphoepithelioma-Like carcinoma. J Inflamm Res. 18:2433–2445.
2025. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Yang X, Xiao Y, Zhou Y, Hu H, Deng H,
Huang J, Liang M, Yuan Z, Dong L and Huang S: Efficacy and safety
of immunotherapy in locally advanced or metastatic pulmonary
lymphoepithelioma-like carcinoma: A multicenter retrospective
study. Ther Adv Med Oncol. 17:175883592513160992025. View Article : Google Scholar : PubMed/NCBI
|