Introduction
Cigarette smoking is a leading contributor to cancer
development and mortality, accounting for an estimated 28.5% of all
cancer-related deaths worldwide (1). Although the established link between
smoking and cancer incidence is well-recognized, the impact of
continued smoking during cancer treatment remains an active area of
investigation. Tobacco smoke contains a number of carcinogens,
including polycyclic aromatic hydrocarbons and nitrosamines, which
not only initiate malignant transformation but also actively
promote tumor progression (2).
Moreover, smoking-induced mutagenesis can lead to the emergence of
more aggressive tumor phenotypes that are characterized by
accelerated growth, metastatic spread and resistance to therapy
(3,4). Recent findings have also indicated
that smoking can exacerbate genomic instability and epigenetic
alterations, further driving tumor aggressiveness (5). Additionally, smoking-induced chronic
inflammation and immune dysregulation create a tumor
microenvironment that promotes progression, immune evasion and
therapy resistance (6).
A concerning aspect of the impact of smoking is the
associated increased risks of recurrence, metastasis and secondary
malignancies (7,8). Smoking has also been shown to
compromise the effectiveness of established cancer therapies
including conventional chemotherapy and radiotherapy (3). Despite these well-documented risks, a
significant number of patients continue to smoke post-cancer
diagnosis, often due to psychological factors such as emotional
regulation, habitual dependence and cognitive distortions including
fatalistic thinking or self-blame. A number of patients use smoking
as a coping mechanism for stress, anxiety and depressive symptoms,
which often intensify after a cancer diagnosis (9). Additionally, nicotine dependence and
depressive symptoms have been identified as significant predictors
of relapse after cancer treatment, highlighting the challenge of
quitting smoking (10). These
barriers emphasize the urgent need for targeted interventions that
address both the physical and psychological aspects of smoking
addiction in this vulnerable population. By integrating
psychological support, such as cognitive behavioral strategies and
tailored counseling, smoking cessation programs can more
effectively assist patients with cancer to overcome these
challenges and improve their prognosis.
While the detrimental effects of smoking on
traditional cancer treatments are well documented, its influence on
immune checkpoint inhibitor (ICI) therapy is more nuanced and
paradoxical. Certain studies have suggested that smokers have an
improved response to ICIs, potentially owing to a higher tumor
mutational burden (TMB), which could enhance immune recognition and
response (11,12). Supporting this notion, a
meta-analysis by Mo et al (13) of phase II/III randomized controlled
trials demonstrated that smokers experienced a significant overall
survival (OS) benefit from programmed cell death protein 1
(PD-1)/programmed death-ligand 1 (PD-L1) inhibitors compared with
chemotherapy, while non-smokers did not exhibit statistically
significant improvements from ICI monotherapy. Similarly, Kim et
al (14) reported that ICIs
significantly prolonged the OS of ever-smokers but not of
never-smokers among patients with non-small cell lung cancer
(NSCLC). By contrast, Luo and Stent (4) found that immunotherapy benefited both
smokers and non-smokers with lung cancer; however, the relationship
between smoking status and ICI efficacy remains complex and
multifaceted. These findings suggest that a smoking history may be
associated with improved responses to ICI therapy, potentially due
to the higher TMB in smokers, which enhances immune recognition.
The complexity of these interactions highlights the need for
further investigation to clarify the mechanisms underlying these
observations and to refine the treatment strategies for patients
with cancer who smoke. Therefore, the present study aimed to
investigate the prognostic impact of smoking status and other
characteristics in patients with metastatic or advanced cancer
undergoing ICI therapy.
Patients and methods
Study population
Patients with predetermined solid tumors who
received ICI (pembrolizumab or nivolumab) therapy, either as a
monotherapy or in combination regimens in a palliative treatment
setting, were retrospectively evaluated at a single-center
institution (University Medical Center Hamburg-Eppendorf, Hamburg,
Germany). Tumor types were chosen for the present study based on
frequent ICI use. Therefore, the solid tumor types included in the
analysis were lung cancer, including NSCLC and small cell lung
cancer (SCLC), renal cell carcinoma (RCC), melanoma, urothelial
carcinoma (UC) and head and neck cancer, including head and neck
squamous cell carcinoma (HNSCC) and head and neck adenosquamous
carcinoma.
Inclusion criteria
Eligible patients included those diagnosed with the
specified tumor types, those who had received >1 dose of ICI
therapy at our center and those for whom data on the smoking status
were available. Smoking status was categorized into three groups:
Never, former and active smokers. Former smokers were defined as
those who had quit smoking prior to ICI initiation. Informed
consent and ethics approval were waived due to the retrospective
design of the study. This approach was approved by the local ethics
committee of the Hamburg Chamber of Physicians (2025-300686-WF) and
was in accordance with the Hamburg Hospital Act (HmbKHG) §12.
Exclusion criteria
Patients were excluded from the analysis if they met
any of the following criteria: i) Unclear smoking status; ii) the
disease did not correspond to the predefined tumor types; or iii)
insufficient data, such as missing follow-up information,
incomplete medical records or an absence of key baseline
characteristics.
Objectives and endpoints
The primary outcomes, OS and progression-free
survival (PFS), were analyzed using Kaplan-Meier estimates to
assess the impact of smoking status on treatment outcomes.
Differences between survival curves were evaluated using the
log-rank test. If no deaths were recorded, the survival time was
censored at the date of the last follow-up. PFS was defined as the
time from the initiation of ICI therapy to disease progression,
whereas OS was defined as the time from the initiation of ICI
therapy to death from any cause.
In addition to the primary outcomes, baseline
patient characteristics, including age, sex, Eastern Cooperative
Oncology Group (ECOG) performance status, C-reactive protein (CRP)
and lactate dehydrogenase (LDH) levels and neutrophil-to-lymphocyte
ratio (NLR), were examined for their prognostic significance. Age,
CRP, LDH and NLR were analyzed as continuous variables, to examine
whether the increasing levels correlated with an impact on the
outcomes; a hazard ratio (HR) of 1.00 reflects the per-unit
increase.
Furthermore, the objective response rate (ORR) and
disease control rate (DCR) were analyzed as secondary endpoints.
ORR was defined as partial remission (PR) or complete remission
(CR) and DCR was defined as stable disease, PR or CR.
To compare baseline characteristics between smoking
groups, the Kruskal-Wallis rank sum test was applied for continuous
variables, Pearson's χ2 test was used for categorical
variables with sufficient sample sizes, and Fisher's exact test was
performed for categorical variables with small counts or sparse
categories. Univariate and multivariate Cox regression models were
used to identify independent predictors of OS and PFS. Variables
with P<0.05 in the univariate analysis were included in the
multivariate Cox model. All statistical analyses were performed
using R (v4.4.2) (15) with the
‘gtsummary’ (v2.0.4) (16),
‘survival’ (v3.8–3) (17),
‘finalfit’ (v1.0.8) (18) and
‘survminer’ (v0.5.0) (19)
packages. Plots were generated using ‘ggplot2’ (v3.5.1) (20).
Results
Patient characteristics
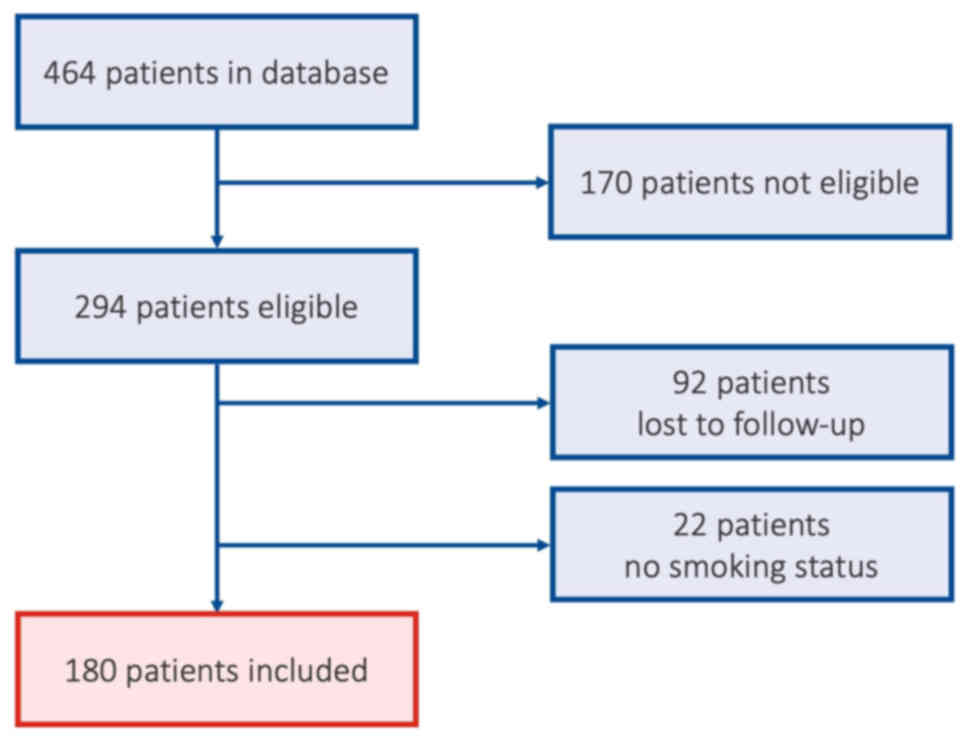
Among the 464 patients who received ICI treatment at
our tertiary center, University Medical Center Hamburg-Eppendorf,
and were initially evaluated in the present study, 170 did not meet
the predefined tumor types criteria or were treated in a clinical
trial. Of the 294 patients with the intended tumor types, 92
patients were lost to follow-up as most of these patients continued
treatment externally. Additionally, the smoking status could not be
clearly evaluated in 22 patients. Therefore, 180 patients met the
eligibility criteria for analysis (Fig.
1). The cohort included patients with NSCLC or SCLC (n=94), RCC
(n=18), UC (n=21), head and neck cancer (n=44) and melanoma (n=3).
The majority (93%) of patients presented with metastatic disease at
the initiation of ICI therapy. Patients without metastatic disease
had locally advanced stage disease and were treated for palliative
reasons. Of these patients, 143 (79%) received ICI treatment
without chemotherapy. The median age of the cohort was 68 years
(Q1-Q3, 59–74 years). Male patients constituted the predominant
demographic group (71%), while female patients comprised 29% of the
study population. Most patients demonstrated a favorable baseline
performance status, with 76% having an ECOG score of 0 or 1.
Notably, 83% of patients received ICI treatment in either the
first- or second-line setting. Based on a detailed smoking history,
the cohort was further stratified into never-smokers (n=45), former
smokers (n=81) and active smokers (n=54). The median follow-up time
for the entire cohort was 13 months (range, 5–34 months). The
baseline characteristics of the study population are summarized in
Table I.
 | Table I.Patient characteristics. |
Table I.
Patient characteristics.
| Characteristic | Never (n=45) | Former (n=81) | Active (n=54) | P-valuea |
|---|
| Sex, n (%) |
|
|
| 0.008 |
| Male | 24 (53%) | 64 (79%) | 40 (74%) |
|
|
Female | 21 (47%) | 17 (21%) | 14 (26%) |
|
| Median age, years
(SD) [IQR] | 73.0 (11.7) [13] | 68.0 (9.4) [14] | 64.0 (10.9) [13] | 0.002 |
| ECOG, n (%) |
|
|
| 0.406 |
| 0 | 11 (26%) | 18 (25%) | 12 (24%) |
|
| 1 | 24 (57%) | 40 (55%) | 21 (42%) |
|
| 2 | 5 (12%) | 12 (16%) | 10 (20%) |
|
| 3 | 2 (4.8%) | 3 (4.1%) | 7 (14%) |
|
|
N/A | 3 | 8 | 4 |
|
| BMI category, n
(%) |
|
|
| 0.032 |
| Healthy
weight | 29 (64%) | 37 (49%) | 28 (56%) |
|
|
Obesity | 3 (6.7%) | 10 (13%) | 5 (10%) |
|
|
Overweight | 13 (29%) | 25 (33%) | 9 (18%) |
|
|
Underweight | 0 (0%) | 4 (5.3%) | 8 (16%) |
|
|
N/A | 0 | 5 | 4 |
|
| Tumor, n (%) |
|
|
| <0.001 |
|
NSCLC | 16 (36%) | 43 (53%) | 31 (57%) |
|
|
SCLC | 0 (0%) | 1 (1.2%) | 3 (5.6%) |
|
|
RCC | 12 (27%) | 5 (6.2%) | 1 (1.9%) |
|
|
Melanoma | 2 (4.4%) | 1 (1.2%) | 0 (0%) |
|
|
Urothelial | 5 (11%) | 13 (16%) | 3 (5.6%) |
|
|
HNSCC | 9 (20%) | 18 (22%) | 16 (30%) |
|
|
HNAdSC | 1 (2.2%) | 0 (0%) | 0 (0%) |
|
| Metastasis, n
(%) |
|
|
| 0.049 |
| No | 4 (8.9%) | 2 (2.5%) | 7 (13%) |
|
|
Yes | 41 (91%) | 79 (98%) | 47 (87%) |
|
| Treatment, n
(%) |
|
|
| 0.034 |
| ICI
mono | 30 (67%) | 66 (81%) | 41 (76%) |
|
| ICI +
chemotherapy | 8 (18%) | 14 (17%) | 12 (22%) |
|
| ICI +
CTLA4 | 4 (8.9%) | 1 (1.2%) | 1 (1.9%) |
|
| ICI +
VEGFR | 3 (6.7%) | 0 (0%) | 0 (0%) |
|
| Line of treatment,
n (%) |
|
|
| 0.600 |
| 1 | 21 (47%) | 33 (41%) | 29 (54%) |
|
| 2 | 16 (36%) | 34 (42%) | 16 (30%) |
|
| 3+ | 8 (17.8%) | 14 (17%) | 9 (16.8%) |
|
| Median LDH, U/l
(SD) [IQR] | 248.0 | 224.5 | 214.0 | 0.309 |
|
| (187.8)
[107.5] | (132.5) [97.0] | (118.8)
[100.8] |
|
|
N/A | 2 | 1 | 2 |
|
| Median CRP, mg/l
(SD) [IQR] | 9.5 | 18.5 | 18.0 | 0.011 |
|
| (51.2) [27.8] | (50.6) [58.3] | (54.5) [56.5] |
|
|
N/A | 1 | 1 | 3 |
|
| Median NLR (SD)
[IQR] | 3.4 (2.0)
[2.2] | 4.4 (2.6)
[3.5] | 4.0 (4.3)
[3.3] | 0.031 |
|
N/A | 9 | 13 | 12 |
|
| Median doses, n
(SD) [IQR] | 7.0 (20.3)
[15.0] | 7.5 (17.0)
[10.3] | 6.0 (15.5)
[7.8] | 0.335 |
|
N/A | 0 | 1 | 0 |
|
| Median follow-up,
months (SD) [IQR] | 17.0 (25.7)
[43.0] | 14.0 (25.3)
[26.0] | 7.5 (24.3)
[22.3] | 0.025 |
| ORR, n (%) |
|
|
| >0.900 |
|
PD/stable disease | 33 (77%) | 63 (78%) | 37 (76%) |
|
|
PR/CR | 10 (23%) | 18 (22%) | 12 (24%) |
|
|
N/A | 2 | 0 | 5 |
|
| DCR, n (%) | 25 (58%) | 43 (53%) | 21 (43%) | 0.316 |
|
N/A | 2 | 0 | 5 |
|
When comparing patients with lung cancer (n=94) to
those with other solid tumors (n=86) treated with ICIs, significant
differences in the baseline characteristics and treatment outcomes
were observed. Notably, the distribution of the smoking status
differed significantly between the groups (P=0.022), with a higher
prevalence of former (47 vs. 43%) and active (36 vs. 23%) smokers
and a lower proportion of never-smokers (17 vs. 34%) in the lung
cancer cohort compared with other malignancies. While the overall
ORR, defined as PR or CR, did not significantly differ between the
lung cancer group and the other cancer types group (26 vs. 20%;
P=0.3), the DCR was significantly higher among patients with lung
cancer (59 vs. 43%; P=0.029). These findings underscore the
distinct patient characteristics, particularly the smoking history,
and differential treatment responses to ICIs in lung cancer vs.
other solid tumors.
Univariate and multivariate analyses
of OS
The median OS and PFS times for the entire cohort
from the initiation of ICI treatment were 39.1 and 5.5 months,
respectively. Among the smoking subgroups, the median OS time was
66.4 months for never-smokers, 31.6 months for former smokers and
13.9 months for active smokers (P=0.15). When comparing active
smokers to the combined former and never-smokers cohort, the median
OS times were 13.9 vs. 41.1 months, respectively (P=0.069)
(Fig. 2).
Further exploratory univariate analysis identified
several factors associated with a poorer OS including ECOG
performance status ≥3 [HR, 7.78; 95% confidence interval (CI),
3.22–18.80; P<0.001], baseline CRP levels (HR, 1.01; 95% CI,
1.00–1.01; P<0.001), NLR (HR, 1.12; 95% CI, 1.04–1.21; P=0.002)
and LDH levels (HR, 1.00; 95% CI, 1.00–1.00; P<0.001).
Multivariate analysis confirmed the significant negative prognostic
impact of ECOG performance status ≥3 (HR, 5.85; 95% CI, 1.90–18.04;
P=0.002), NLR (HR, 1.11; 95% CI, 1.02–1.21; P=0.017), LDH ≥ mean
(HR, 1.00; 95% CI, 1.00–1.00; P<0.001) and CRP levels (HR, 1.01;
95% CI, 1.00–1.01; P=0.002). All parameters from the univariate and
multivariate analyses are summarized in Table II. As CRP, NLR and LDH were
analyzed as continuous variables, hazard ratios represent the
relative risk per unit increase of the respective parameter.
 | Table II.Results of univariate and
multivariate analyses of overall survival. |
Table II.
Results of univariate and
multivariate analyses of overall survival.
| Variable | Value | HR
(univariate) | HR
(multivariate) |
|---|
| Smoking status, n
(%) |
|
|
|
| Not
active | 126 (70.0) | - | - |
|
Active | 54 (30.0) | 1.52 (95% CI
0.97–2.40, P=0.069) | - |
| Sex, n (%) |
|
|
|
|
Male | 128 (71.1) | - | - |
|
Female | 52 (28.9) | 0.95 (95% CI
0.59–1.51, P=0.818) | - |
| Mean age (SD),
years | 66.6 (10.7) | 1.00 (95% CI
0.98–1.02, P=0.804) | - |
| ECOG status, n
(%) |
|
|
|
| 0 | 41 (24.8) | - | - |
| 1 | 85 (51.5) | 0.86 (95% CI
0.50–1.51, P=0.609) | 0.62 (95% CI
0.33–1.14, P=0.124) |
| 2 | 27 (16.4) | 1.88 (95% CI
0.92–3.85, P=0.082) | 1.03 (95% CI
0.43–2.46, P=0.948) |
| ≥3 | 12 (7.3) | 7.78 (95% CI
3.22–18.80, P<0.001) | 5.85 (95% CI
1.90–18.04, P=0.002) |
| Metastasis, n
(%) |
|
|
|
| No | 13 (7.2) | - | - |
|
Yes | 167 (92.8) | 1.03 (95% CI
0.45–2.35, P=0.953) | - |
| Mean NLR (SD) | 4.6 (3.1) | 1.12 (95% CI
1.04–1.21, P=0.002) | 1.11 (95% CI
1.02–1.21, P=0.017) |
| Mean LDH, U/l
(SD) | 262.9 (144.9) | 1.00 (95% CI
1.00–1.00, P<0.001) | 1.00 (95% CI
1.00–1.00, P<0.001) |
| Mean CRP, mg/l
(SD) | 39.5 (52.2) | 1.01 (95% CI
1.00–1.01, P<0.001) | 1.01 (95% CI
1.00–1.01, P=0.002) |
Univariate and multivariate analyses
of PFS
Univariate analysis of PFS showed that female sex
(HR, 0.69; 95% CI, 0.48–1.00; P=0.049), age (HR, 0.98; 95% CI,
0.97–1.00; P=0.026) and ECOG performance status ≥3 (HR, 5.83; 95%
CI, 2.60–13.09; P<0.001) were significantly associated with PFS.
Multivariate analysis confirmed that age (HR, 0.98; 95% CI,
0.96–1.00; P=0.014) and ECOG performance status ≥3 (HR, 5.55; 95%
CI, 2.37–12.99; P<0.001) were independent predictors of a
shorter PFS time. Regarding the smoking status, no statistical
differences were detected in terms of PFS. Table III provides a complete overview of
all parameters from both analyses.
 | Table III.Results of univariate and
multivariate analyses regarding PFS. |
Table III.
Results of univariate and
multivariate analyses regarding PFS.
| Variable | Value | HR
(univariate) | HR
(multivariate) |
|---|
| Smoking status, n
(%) |
|
|
|
| Not
active | 126 (70.0) | - | - |
|
Active | 54 (30.0) | 1.26 (95% CI
0.88–1.80, P=0.216) | - |
| Sex, n (%) |
|
|
|
|
Male | 128 (71.1) | - | - |
|
Female | 52 (28.9) | 0.69 (95% CI
0.48–1.00, P=0.049) | 0.68 (95% CI
0.45–1.03, P=0.071) |
| Mean age (SD),
years | 66.6 (10.7) | 0.98 (95% CI
0.97–1.00, P=0.026) | 0.98 (95% CI
0.96–1.00, P=0.014) |
| ECOG status, n
(%) |
|
|
|
| 0 | 41 (24.8) | - | - |
| 1 | 85 (51.5) | 1.00 (95% CI
0.67–1.51, P=0.986) | 1.00 (95% CI
0.66–1.52, P=0.983) |
| 2 | 27 (16.4) | 1.76 (95% CI
1.04–2.99, P=0.036) | 1.42 (95% CI
0.81–2.49, P=0.217) |
| ≥3 | 12 (7.3) | 5.83 (95% CI
2.60–13.09, P<0.001) | 5.55 (95% CI
2.37–12.99, P<0.001) |
| Metastasis |
|
|
|
| No | 13 (7.2) | - | - |
|
Yes | 167 (92.8) | 0.95 (95% CI
0.50–1.80, P=0.867) | - |
| Mean NLR (SD) | 4.6 (3.1) | 1.01 (95% CI
0.94–1.08, P=0.797) | - |
| Mean LDH, U/l
(SD) | 262.9 (144.9) | 1.00 (95% CI
1.00–1.00, P=0.006) | 1.00 (95% CI
1.00–1.00, P<0.001) |
| Mean CRP, U/l
(SD) | 39.5 (52.2) | 1.00 (95% CI
1.00–1.01, P=0.007) | 1.00 (95% CI
1.00–1.01, P=0.281) |
Overall outcome and response rate
Among the evaluable patients, the ORR was 23% and
the DCR was 51%, which were comparable to the reported rates for
NSCLC [19% (21)] and HNSCC [30%
(22)]. However, subgroup analysis
stratified by the smoking status demonstrated no statistically
significant differences in either the ORR or DCR (Table I).
Subgroup analysis on lung cancer
Subgroup analysis based on the smoking status was
conducted for the 94 patients with lung cancer treated with ICIs.
In this subgroup analysis, 16 patients were never-smokers, 44 were
former smokers and 34 were active smokers. Regarding the baseline
characteristics, a statistically significant difference was
observed in terms of age, with never-smokers being the oldest
(median, 72.5 years), followed by former smokers (median, 69.0
years) and active smokers being the youngest (median, 65.0 years)
(P=0.034). The DCR also showed a significant difference (P=0.039),
with the highest DCR observed in never-smokers (80%), followed by
former smokers (64%) and then active smokers (44%). While a trend
towards significance was noted in the sex distribution (P=0.052),
with a higher proportion of female patients in the never-smokers
group (56%) compared with the former (23%) and active (32%) smokers
groups, other characteristics did not show statistically
significant differences. These included the ORR (P=0.5), ECOG
performance status (P=0.3), BMI category (P=0.6), tumor type
(predominantly NSCLC across all groups), presence of metastasis
(P=0.3), high-risk metastasis (P=0.12), type of ICI treatment
(P=0.3), line of therapy (P=0.3), LDH levels (P=0.6), CRP levels
(P=0.2), NLR (P=0.4), number of ICI doses (P=0.11), incidence of
immune-related adverse events (irAEs; P=0.5), type of irAEs (P=0.7)
and median follow-up time (P=0.3). Regarding the survival analysis,
the OS time was 25.5 months for never-smokers, 63 months for former
smokers and 14 months for active smokers (P=0.61).
Immune-related adverse events
Based on the analysis of 180 patients receiving ICI
therapy, 26% experienced irAEs overall. When stratified by smoking
status, the frequency of any irAE did not differ significantly
between never-smokers (31%; n=14/45), former smokers (22%; n=18/81)
and active smokers (28%; n=15/54), with P=0.5. Similarly, the
distribution of the specific type of irAE did not show a
statistically significant difference across the smoking groups
(P=0.15).
Discussion
The present study analyzed the prognostic influence
of smoking status in patients with metastatic or advanced solid
tumors undergoing ICI therapy. Contrary to the initial hypothesis
that smokers might exhibit improved outcomes due to a higher TMB,
active smokers demonstrated a reduced OS time compared with both
never-smokers and former smokers in the present study. These
results align with existing evidence linking active smoking to more
aggressive tumor biology, increased therapeutic resistance and
poorer clinical outcomes (3).
In the present study, while the univariate analysis
results suggested an association between smoking status and OS,
this relationship narrowly missed statistical significance. The
lack of significance likely reflects the impact of other prognostic
factors, such as the baseline performance status and systemic
inflammation markers, such as CRP and LDH, which have emerged as
independent predictors of survival in previous studies. This
suggests that the negative effects of active smoking may be
mediated, at least in part, by its interactions with these factors.
However, the patient selection in the present study was heavily
weighted towards lung and head and neck cancer, which may limit
generalizability.
Paradoxical reports of improved outcomes with ICI
therapy in smokers (13,23–26)
merit careful evaluation. Several meta-analyses and cohort studies
have shown that patients with a history of smoking often derive a
greater clinical benefit from ICI therapy than never-smokers,
particularly in NSCLC (14). This
phenomenon is partly attributed to the higher TMB typically
observed in smokers, which results in an increased neoantigen load
and may enhance immune recognition and the responsiveness to ICIs.
For instance, smokers with NSCLC treated with ICIs have
demonstrated a longer PFS time and higher response rates than
never-smokers, with the most notable effects observed in patients
receiving anti-PD-1/PD-L1 monotherapy. Furthermore, the tumor
immune microenvironment in smokers is often more inflamed and
immunogenic, characterized by the increased infiltration of
activated immune cells, further supporting improved responsiveness
to ICI therapy (13,25,26).
While the present study confirms the detrimental impact of active
smoking, it does not entirely refute the hypothesis that a higher
TMB in smokers enhances tumor immunogenicity, potentially improving
ICI efficacy (26,27). However, the poorer outcomes observed
in active smokers in the present study suggest that the negative
effects of smoking, such as heightened systemic inflammation,
impaired immune function and a greater burden of comorbidities, may
outweigh any theoretical benefits of a higher mutational load or
higher PD-L1 tumor proportion score (12,26–31).
In the present study, biomarkers of systemic inflammation,
including CRP, NLR and LDH, emerged as strong independent
predictors of a reduced OS time, irrespective of smoking status.
These findings imply that the inferior outcomes in active smokers
are likely driven, at least in part, by the overriding negative
impact of systemic inflammation.
This complex interplay underscores the need for
novel therapeutic strategies that can better navigate and modulate
the tumor microenvironment. In this context, emerging research into
advanced biomaterials offers potential avenues; for instance,
nano-biomaterials are being explored for their ability to regulate
the tumor microenvironment and enhance immunotherapy (32).
In the present study, the multivariate analysis
identified baseline ECOG performance status ≥3 and elevated CRP,
NLR and LDH levels as the strongest independent predictors of a
diminished OS time, irrespective of smoking status. These
biomarkers are crucial as they reflect both the functional status
of the patient and the presence of systemic inflammation, which are
known to drive tumor progression and resistance to therapy. In
clinical practice, integrating these parameters into prognostic
models could help stratify patients for tailored treatment
approaches. Moreover, active smoking can increase the inflammatory
environment in tumors and promote tumor progression and therapeutic
resistance. A poor ECOG performance status is a well-established
marker of overall patient fitness and the capacity to tolerate
systemic therapies (33).
Similarly, an elevated CRP level, a biomarker of systemic
inflammation, correlates with unfavorable outcomes in various
malignancies (34).
Although smoking status did not emerge as an
independent predictor of survival in the present study, it remains
a factor in prognosis and treatment planning. However, a number of
patients with cancer struggle to quit due to inadequate
psychological and behavioral support within oncology care.
Addressing the emotional and cognitive barriers associated with
smoking in patients with cancer is crucial, as psychological
distress, fatalistic thinking and nicotine dependence are key
factors hindering smoking cessation (35). Additionally, incorporating smoking
cessation support into routine oncology care, including regular
follow-ups and tailored cessation programs, has been shown to
improve cessation rates and significantly enhance overall patient
outcomes (36). Providing
structured psychological interventions, such as cognitive
behavioral therapy and pharmacological treatments, allows for a
more patient-centered approach that acknowledges the complex
interplay between cancer-related stress and smoking behavior
(37). Therefore, integrating
comprehensive smoking cessation programs into routine oncology care
is essential for improving treatment outcomes and overall patient
well-being.
The present study has several limitations that
should be acknowledged. Firstly, the heterogeneity of the solid
tumor types included, with a notable predominance of lung and head
and neck cancer, may limit the generalizability of the findings to
other less represented malignancies. Different tumor entities
exhibit distinct biological characteristics and immune
microenvironments, which may interact variably with the smoking
status and ICI efficacy. Secondly, although the outcomes related to
ICI therapy were analyzed, the limited number of patients prevented
a detailed analysis of the specific ICI regimens. This lack of
granularity in treatment specifics could mask differential effects
and contribute to the observed variability in outcomes. Thirdly,
the categorization of smoking status, while standard, did not
capture more nuanced details such as pack-years, duration of
smoking or precise time since cessation for former smokers. Such
granularity, if available, might reveal more complex dose-dependent
or time-dependent effects of smoking on ICI outcomes and could
explain some of the inconsistencies noted in the broader
literature, especially considering the long-lasting immune
alterations potentially induced by smoking. Regarding irAEs, this
analysis faces potential limitations, primarily due to possible
underreporting, given the retrospective setting. Finally, as a
retrospective single-center analysis with a limited number of
included patients, the present study is susceptible to inherent
biases, including the potential for unmeasured confounding
variables and selection bias, despite efforts to control for known
prognostic factors. These limitations underscore the need for
future prospective studies with more detailed data collection to
further elucidate the complex interplay between tumor type,
specific ICI regimens, detailed smoking history and clinical
outcomes.
In conclusion, the findings of the present study
underscore the need for a comprehensive approach to patient
evaluation before initiating ICI therapy. Future efforts should
focus on developing personalized treatment strategies that account
for both tumor characteristics and host factors such as the
inflammatory microenvironment. By adopting this comprehensive
approach, the full therapeutic potential of ICIs can be fully
realized, ultimately improving outcomes in patients with advanced
cancer. Moreover, further studies are needed to elucidate
mechanisms and optimize cessation strategies.
Acknowledgements
Not applicable.
Funding
The present study was supported by the Bristol Myers Squibb
Stiftung Immunonkologie.
Availability of data and materials
The data generated in the present study may be
requested from the corresponding author.
Authors' contributions
CB, CSc, CSe and FB designed the study. CSc, CSe and
FB wrote the manuscript. CSc, AP, AF and CSe collected data. CSc
and CSe analyzed and interpreted data. All authors read and
approved the final manuscript. CSc and CSe confirm the authenticity
of all the raw data.
Ethics approval and consent to
participate
The study protocol was in line with the Hamburg
Hospital Act (Hamburgisches Krankenhausgesetz, HmbKHG), and the
Ethics Committee of the Hamburg Chamber of Physicians confirmed
that consultation was not required (no. 2025-300686-WF).
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Islami F, Marlow EC, Thomson B, McCullough
ML, Rumgay H, Gapstur SM, Patel AV, Soerjomataram I and Jemal A:
Proportion and number of cancer cases and deaths attributable to
potentially modifiable risk factors in the United States, 2019. CA
Cancer J Clin. 74:405–432. 2024.PubMed/NCBI
|
|
2
|
Alberg AJ, Brock MV and Samet JM:
Epidemiology of lung cancer: Looking to the future. J Clin Oncol.
23:3175–3185. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Schaefers C, Seidel C, Bokemeyer F and
Bokemeyer C: The prognostic impact of the smoking status of cancer
patients receiving systemic treatment and radiation therapy,
surgery: A systematic review and meta-analysis. Eur J Cancer.
172:130–137. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Luo Y and Stent S: Smoking's lasting
effect on the immune system. Nature. 626:724–725. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Kuśnierczyk P: Genetic differences between
smokers and never-smokers with lung cancer. Front Immunol.
14:10637162023. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
de la Iglesia JV, Slebos RJC, Martin-Gomez
L, Wang X, Teer JK, Tan AC, Gerke TA, Aden-Buie G, van Veen T,
Masannat J, et al: Effects of tobacco smoking on the tumor immune
microenvironment in head and neck squamous cell carcinoma. Clin
Cancer Res. 26:1474–1485. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Foerster B, Pozo C, Abufaraj M, Mari A,
Kimura S, D'Andrea D, John H and Shariat SF: Association of smoking
status with recurrence, metastasis, mortality among patients with
localized prostate cancer undergoing prostatectomy or radiotherapy:
A systematic review and meta-analysis. JAMA Oncol. 4:953–961. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Masui Y, Shukuya T, Kataoka S, Shiozaki H,
Kurokawa K, Nakamura I, Miyawaki T, Koinuma Y, Asao T, Kanemaru R,
et al: Second malignancy in advanced or recurrent non-small cell
lung cancer after the advent of molecular targeted drugs and
immunotherapy. Thorac Cancer. 15:2291–2297. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Choi SH, Chan RR and Lehto RH:
Relationships between smoking status and psychological distress,
optimism, and health environment perceptions at time of diagnosis
of actual or suspected lung cancer. Cancer Nurs. 42:156–163. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Guimond AJ, Croteau VA, Savard MH, Bernard
P, Ivers H and Savard J: Predictors of smoking cessation and
relapse in cancer patients and effect on psychological variables:
An 18-Month observational study. Ann Behav Med. 51:117–127. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
McLean L, Leal JL, Solomon BJ and John T:
Immunotherapy in oncogene addicted non-small cell lung cancer.
Transl Lung Cancer Res. 10:2736–2751. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Sun LY, Cen WJ, Tang WT, Long YK, Yang XH,
Ji XM, Yang JJ, Zhang RJ, Wang F, Shao JY and Du ZM: Smoking status
combined with tumor mutational burden as a prognosis predictor for
combination immune checkpoint inhibitor therapy in non-small cell
lung cancer. Cancer Med. 10:6610–6617. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Mo J, Hu X, Gu L, Chen B, Khadaroo PA,
Shen Z, Dong L, Lv Y, Chitumba MN and Liu J: Smokers or
non-smokers: Who benefits more from immune checkpoint inhibitors in
treatment of malignancies? An up-to-date meta-analysis. World J
Surg Oncol. 18:152020. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Kim JH, Kim HS and Kim BJ: Prognostic
value of smoking status in non-small-cell lung cancer patients
treated with immune checkpoint inhibitors: A meta-analysis.
Oncotarget. 8:93149–93155. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
The R Core Team, . A Language and
Environment for Statistical Computing. R Foundation for Statistical
Computing; Vienna: 2024
|
|
16
|
Sjoberg DD, Whiting K, Curry M, Lavery JA
and Larmarange J: Reproducible summary tables with the gtsummary
package. R J. 13:570–594. 2021. View Article : Google Scholar
|
|
17
|
Terry Thernea, . A Package for Survival
Analysis in R. 2024.
|
|
18
|
Harrison E, Drake T and Pius R: Finalfit:
Quickly Create Elegant Regression Results Tables and Plots when
Modelling. 2025.
|
|
19
|
Kassambara A, Kosinski M and Biecek P:
Survminer: Drawing Survival Curves using ‘ggplot2’. 2024.PubMed/NCBI
|
|
20
|
Wickham H: ggplot2: Elegant Graphics for
Data Analysis. 2016.
|
|
21
|
Garon EB, Rizvi NA, Hui R, Leighl N,
Balmanoukian AS, Eder JP, Patnaik A, Aggarwal C, Gubens M, Horn L,
et al: Pembrolizumab for the treatment of Non-small-cell lung
cancer. N Engl J Med. 372:2018–2028. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Hobday SB, Brody RM, Kriegsman B, Basu D,
Newman J, Cohen RB, Lukens JN, Singh A, D'Avella CA and Sun L:
Outcomes among patients with mucosal head and neck squamous cell
carcinoma treated with checkpoint inhibitors. JAMA Otolaryngol Neck
Surg. 148:918–926. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Hu X, Liu Y, He Y, Wang Z, Zhang H, Yang W
and Lu J: Relationship between Patients' Baseline characteristics
and survival benefits in Immunotherapy-treated Non-small-cell lung
cancer: A systematic review and Meta-analysis. J Oncol.
2022:36019422022. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Gainor JF, Rizvi H, Jimenez Aguilar E,
Skoulidis F, Yeap BY, Naidoo J, Khosrowjerdi S, Mooradian M, Lydon
C, Illei P, et al: Clinical activity of programmed cell death 1
(PD-1) blockade in never, light, and heavy smokers with
non-small-cell lung cancer and PD-L1 expression ≥50. Ann Oncol.
31:404–411. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Sun Y, Yang Q, Shen J, Wei T, Shen W,
Zhang N, Luo P and Zhang J: The effect of smoking on the immune
microenvironment and immunogenicity and its relationship with the
prognosis of immune checkpoint inhibitors in Non-small cell lung
cancer. Front Cell Dev Biol. 9:7458592021. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Desrichard A, Kuo F, Chowell D, Lee KW,
Riaz N, Wong RJ, Chan TA and Morris LGT: Tobacco Smoking-associated
alterations in the immune microenvironment of squamous cell
carcinomas. J Natl Cancer Inst. 110:1386–1392. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Wang X, Ricciuti B, Nguyen T, Li X, Rabin
MS, Awad MM, Lin X, Johnson BE and Christiani DC: Association
between smoking history and tumor mutation burden in advanced
Non-small cell lung cancer. Cancer Res. 81:2566–2573. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Fox P, Hudson M, Brown C, Lord S, Gebski
V, De Souza P and Lee CK: Markers of systemic inflammation predict
survival in patients with advanced renal cell cancer. Br J Cancer.
109:147–153. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Norum J and Nieder C: Tobacco smoking and
cessation, PD-L1 inhibitors in non-small cell lung cancer (NSCLC):
A review of the literature. ESMO Open. 3:e0004062018. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Luetragoon T, Rutqvist LE,
Tangvarasittichai O, Andersson BÅ, Löfgren S, Usuwanthim K and
Lewin NL: Interaction among smoking status, single nucleotide
polymorphisms and markers of systemic inflammation in healthy
individuals. Immunology. 154:98–103. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Shiels MS, Katki HA, Freedman ND, Purdue
MP, Wentzensen N, Trabert B, Kitahara CM, Furr M, Li Y, Kemp TJ, et
al: Cigarette smoking and variations in systemic immune and
inflammation markers. J Natl Cancer Inst. 106:dju2942014.
View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Xu G, Li J, Zhang S, Cai J, Deng X, Wang Y
and Pei P: Two-dimensional nano-biomaterials in regulating the
tumor microenvironment for immunotherapy. Nano TransMed.
3:1000452024. View Article : Google Scholar
|
|
33
|
West H and Jin JO: Performance status in
patients with cancer. JAMA Oncol. 1:998. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
McMillan DC: The systemic
inflammation-based glasgow prognostic score: A decade of experience
in patients with cancer. Cancer Treat Rev. 39:534–540. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Wells M, Aitchison P, Harris F, Ozakinci
G, Radley A, Bauld L, Entwistle V, Munro A, Haw S, Culbard B and
Williams B: Barriers and facilitators to smoking cessation in a
cancer context: A qualitative study of patient, family and
professional views. BMC Cancer. 17:3482017. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Gritz ER, Toll BA and Warren GW: Tobacco
use in the oncology setting: Advancing clinical practice and
research. Cancer Epidemiol Biomarkers Prev. 23:3–9. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Scholten PR, Stalpers LJA, Bronsema I, van
Os RM, Westerveld H and van Lonkhuijzen LRCW: The effectiveness of
smoking cessation interventions after cancer diagnosis: A
systematic review and meta-analysis. J Cancer Policy.
39:1004632024. View Article : Google Scholar : PubMed/NCBI
|