Introduction
Breast cancer has a high mortality and morbidity
rate among women and is therefore one of the main concerns in
global healthcare (1). In 2020,
female breast cancer accounted for 2.3 million new cases (11.7%),
surpassing lung cancer as the most commonly diagnosed cancer
(2). In the same year, 685,000
women died of breast cancer, making it the leading cause of death
in women (3). In terms of 5-year
survival, rates in developing countries are lower than in more
developed ones (2,4). Given the high survival rates in
developed countries (5),
emphasizing the identification of prognostic factors for breast
cancer is of vital importance.
The main prognostic factors of breast cancer have
been recognized for many years. These include hormone receptor
status, tumour size, lymph node status, metastasis, tumour grade,
progesterone receptor, and HER2 status (6,7). In
addition, the use of hormone replacement therapy (HRT) prior to
breast cancer diagnosis is particularly relevant, given that breast
tissue is sensitive to hormonal stimuli (8).
The use of HRT began in the 1960s and gained
widespread popularity for managing premenopausal symptoms (9). In the following decades, several
articles were published that not only supported this finding, but
also suggested that HRT could prevent chronic diseases such as
coronary heart disease (10,11) or
osteoporosis (12,13). In contrast, the use of HRT
subsequently declined after a study (14) associated its use with an increased
risk of endometrial cancer.
The main turning point for this therapy occurred at
the beginning of the 21st century. In 2002, the results of the most
important study carried out to date were published: WHI (Women's
Health Initiative). The conclusions of the study stated that
‘Overall health risks exceeded benefits from use of combined
oestrogen plus progestin’ (15).
This was followed by a dramatic decline in HRT use in most
countries (16–19), including Spain (20).
After the WHI results and the reduction in HRT use
were published, there was a decrease in the incidence of breast
cancer in some countries such as Belgium (21), France (22) or New Zealand (23). However, in other countries such as
Italy and Spain the incidence remained constant (24). Beyond uncertainty regarding
incidence, it is essential to understand how HRT is associated with
breast cancer prognosis, a debate that has persisted in the medical
and scientific community for years (25,26).
It remains unclear whether the use of HRT prior to
diagnosis is associated with a better or worse prognosis, making
the evaluation of its role as a prognostic factor necessary
(27). There is only one prior
meta-analysis aimed at evaluating the association of HRT prescribed
before cancer diagnosis (27).
The main objective of this study is to analyze the
association between pre-diagnostic HRT use and breast cancer
prognosis, while also evaluating the influence of factors
potentially modifying this relationship.
Materials and methods
Literature search
The procedures outlined in accordance with the
Preferred Reporting Items for Systematic Reviews and Meta-Analyses
statement were adhered to in the execution of this meta-analysis
(28). The protocol was registered
within the PROSPERO database (registration number:
CRD42023398582).
The following bibliographic databases were searched:
MEDLINE (https://pubmed.ncbi.nlm.nih.gov/), Cochrane
(www.cochranelibrary.com) and Web of
Science (https://www.webofscience.com/wos/alldb/basic-search).
All articles published in English or Spanish up to August 28th,
2025 were included. The search terms included (‘menopause hormonal
therapy’ or ‘hormonal replacement therapy’ or ‘hormone replacement
therapy’ or ‘postmenopausal hormone replacement therapy’ or
‘estrogen-progestin hormone replacement therapy’ or ‘combined
hormone replacement therapy’ or ‘estrogen alone hormone replacement
therapy’ or ‘hormone therapy’) and (‘breast cancer’ or ‘breast
neoplasms’ or ‘breast tumour’ or ‘breast tumours’ or ‘breast
carcinoma’ or ‘breast carcinomas’) and (‘prognosis’ or ‘survival’
or ‘prognostic factor’).
Comprehensive search criteria were used to ensure
completeness, including cumulative cohort designs,
incidence-density cohorts, case-control, and hybrid case-cohort
designs that evaluated the possible association between HRT and
breast cancer survival. Subsequently, the reference lists of all
included articles were screened to identify additional relevant
studies.
The screening and selection of articles was carried
out by two independent reviewers (JCF and JAM). These reviewers
retrieved the reference lists of each article and examined them to
identify those studies that met the specified inclusion criteria
(see below). This was carried out in multiple stages; firstly, the
reviewers identified and removed duplicates across the different
databases. Then, they selected studies by title, abstract, and
finally, by full text following the inclusion criteria.
Discrepancies were resolved by discussion until an agreement was
reached and a third researcher intervened to resolve any persisting
conflicts.
Inclusion and exclusion criteria
The studies incorporated in this analysis adhered to
the following criteria: i) Hormone Replacement Therapy (HRT) served
as the primary exposure under investigation; ii) the study design
encompassed cumulative cohort designs, incidence-density cohorts,
case-control, or hybrid case-cohort structures; iii) the key
outcomes encompassed breast cancer mortality or survival,
accompanied by risk estimates and 95% confidence intervals (CI) (or
data facilitating their computation).
Exclusion criteria comprised commentaries,
conference communications, systematic reviews, reviews, book
chapters, and letters to the editor. Additionally, articles not
written in English or Spanish and those that lacked the requisite
data for analysis and studies involving non-human samples were
excluded from consideration.
Data extraction
From the pool of eligible articles, two researchers
(JCF and JAM) systematically extracted the following information:
year of publication, authorship details, title, recruitment start
and end dates, follow-up conclusion date, interquartile range,
country of origin of the study, subpopulations examined, Hormone
Replacement Therapy use, sample size, strength of association (risk
ratio, hazard ratio or odds ratio) with its 95% confidence
intervals (CI), and the set of adjusted covariates employed in the
multivariate analysis. In instances where a study presented
multiple risk estimates, the one adjusted for the most extensive
array of confounding factors was chosen.
Study quality assessment and risk of
bias
After the study selection, methodological quality
was evaluated by two researchers (JCF and PRC) using the Newcastle
Ottawa Scale (NOS) (29). For this
evaluation, eight items were reviewed for each study. These items
were classified into three categories: selection, comparability and
outcome or exposure, depending on whether it was a cohort or a
case-control study. Studies scoring 7 or higher were deemed high
quality. Discrepancies were resolved by discussion.
Following Cochrane recommendations, we also used the
Risk of Bias in Non-randomized Studies of Exposures (ROBINS-E) tool
to assess the risk of exposure bias in each study included in our
meta-analysis (30).
Statistical analysis
Because the included studies reported different
measures of association (Hazard ratios, risk ratios and odds
ratios), all estimates were treated as generic relative effect
measures on the log scale and pooled using inverse-variance
weighting. A random-effects model was applied. Results are
presented as effect estimates accompanied by a 95% confidence
interval (CI), visually represented through Forest plots. The
assessment of heterogeneity was executed using Q and I2
statistics. Publication bias was evaluated using Begg's and Egger's
tests and by examining funnel plots (31). In addition, to account for potential
differences between time-to-event and binary effect measures, an
additional sensitivity analysis stratified by type of effect
measure (hazard ratio vs. odds ratio) was performed.
The analyses were performed considering HRT use
(yes/no), and also based on the duration of use (<5 years; 5 to
10 years; >=10 years), HRT use at the time of diagnosis
(current; past; not indicated), article quality (as per the NOS)
risk of bias (ROBINS-E tools), and year of participant recruitment.
For the latter, participants were segregated into groups: recruited
before and after 2000, due to the pivotal alteration in hormone
concentrations during treatment at that juncture (15). In all analyses, adjusted estimates
were used when available; otherwise, raw data were used.
Because the included studies used different
definitions of HRT exposure (e.g. months vs years of use, varying
cut-offs, and differing definitions of current and past use),
categories were harmonised where possible.
Data were analysed using STATA 16.0 software. Given
that the confidence intervals specified in the articles are not
totally symmetric around the effect size for all studies, the
‘civartolerance’ in STATA (defined as Confidence Interval upper
limit-effect size=effect size-Confidence Interval lower limit) was
set at 0.15. Since generic effect sizes with confidence intervals
require approximate symmetry around the effect estimate on the
appropriate scale, CI symmetry was checked using the civartolerance
option in Stata's meta suite. Two studies (32,33)
showed confidence intervals that remained markedly asymmetric even
after relaxing this tolerance, suggesting potential reporting or
calculation errors. These studies were therefore excluded from the
pooled quantitative synthesis, while their findings are
qualitatively described in the Discussion.
Results
Study characteristics
After conducting the search based on the criteria
outlined above, a total of 3,761 articles were initially
identified. Subsequently, an additional 63 articles were
incorporated through supplementary search methods. The next step
involved the removal of duplicate articles (n=839), followed by a
sequential process of filtering and exclusion guided by title
assessment (n=2,682), abstract review (n=187), and thorough
evaluation of the full articles (n=83) by the reviewers. Finally,
this meta-analysis includes 33 studies: 30 retrospective cohort
studies (8,10,32–59), 1
hybrid case-cohort study (15), 1
nested case-control study (60) and
1 prospective cohort study (61).
Given that Persson et al (32) and Hunt et al (33) reported effect sizes with asymmetric
confidence intervals, they were excluded from the quantitative
synthesis. Similarly, Christante et al (37) did not provide sufficient data for
inclusion in the meta-analysis, and was also excluded. A visual
representation of the bibliographic search process is shown in
Fig. 1. Table I summarizes the main characteristics
of the included articles, while Table
SI provides information detailing the factors included in the
multivariate analysis of each article.
 | Table I.Main characteristics of the included
articles. |
Table I.
Main characteristics of the included
articles.
| First author/s,
year | Follow
up-period | Country | Study design | Sample size | Stratum | HRT exposure | Outcome measure
(95% CI)a | Covariate
adjustment | NOS | (Refs.) |
|---|
| Manson et
al, 2018 | 1993-2014 | USA | Cohort | 26,897 | - | - | 0.55
(0.33–0.92)b | No | >7 | (38) |
| Sener et al,
2009 | 1994-2002 | USA | Cohort | 1,055 | - | - | 0.44
(0.26–0.73)b | Yes | <7 | (43) |
| Fletcher et
al, 2005 | 1993-2003 | Australia | Cohort | 4,022 | - | Current use | 0.64
(0.41–1.00)b | Yes | >7 | (39) |
| Schuetz et
al, 2007 | 1990-1999 | Germany | Cohort | 1,072 | - | Current use | 0.37
(0.24–0.57)c | No | <7 | (40) |
|
|
|
|
|
|
|
| 0.62
(0.39–1.00)c | Yes |
|
|
| Alonso-Molero et
al, | 2007-2017 | Spain | Cohort | 1,685 | Born 1940–1959 | - | 0.77
(0.38–1.55)b | Yes | >7 | (58) |
| 2022 |
|
|
|
| <5 years of
use | - | 0.71
(0.28–1.79)b |
|
|
|
|
|
|
|
|
| ≥5 years of
use | - | 0.85
(0.31–2.39)b |
|
|
|
| Rossouw et
al, 2002 | 1993-2002 | USA | Clinical trial | 16,608 | - | Past use | 1.26
(1.00–1.59)b | No | >7 | (15) |
|
|
|
|
|
| - | Past use | 1.26
(0.83–1.92)b | Yes |
|
|
|
|
|
|
|
| <5 years of
use | Past use | 2.13
(1.15–3.94)b | No |
|
|
|
|
|
|
|
| 5-10 years of
use | Past use | 4.61
(1.01–21.02)b | No |
|
|
|
|
|
|
|
| ≥10 years of
use | Past use | 1.81
(0.60–5.43)b | No |
|
|
| Godina et
al, 2020 | 2002-2016 | Sweden | Cohort | 814 | - | - | 0.68
(0.48–0.99)b | No | >7 | (8) |
|
|
|
|
|
| - | - | 0.81
(0.55–1.19)b | Yes |
|
|
| Grodstein et
al, 1997 | 1976-1994 | USA | Cohort | 121,700 | - | Current use | 0.77
(0.59–1.00)c | No | >7 | (10) |
|
|
|
|
|
| - | Current use | 0.76
(0.56–1.02)c | Yes |
|
|
| Ettinger et
al, 1996 | 1969-1992 | USA | Cohort | 454 | - | Current use | 1.89
(0.43–8.36)c | Yes | <7 | (41) |
| Jernström et
al, 1999 | 1978-1997 | Sweden | Cohort | 984 | - | - | 0.78
(0.65–0.93)c | Yes | >7 | (61) |
|
|
|
|
|
| >5 years of
use | - | 0.77
(0.56–1.08)c | Yes |
|
|
|
|
|
|
|
| <5 years of
use | - | 0.78
(0.63–0.96)c | Yes |
|
|
| Schairer et
al, 1999 | 1973-1995 | USA | Cohort | 280,000 | LNN; <5 years of
use | Past use | 0.8
(0.40–1.40)b | Yes | >7 | (42) |
|
|
|
|
|
| LNN; <5 years of
use | Current use | 0.60
(0.30–1.20)b |
|
|
|
|
|
|
|
|
| LNN; 5–10 years of
use | Past use | 0.70
(0.40–1.20)b |
|
|
|
|
|
|
|
|
| LNN; 5–10 years of
use | Current use | 0.40
(0.20–0.80)b |
|
|
|
|
|
|
|
|
| LNN; ≥10 years of
use | Past use | 1.20
(0.60–2.50)b |
|
|
|
|
|
|
|
|
| LNN; ≥10 years of
use | Current use | 0.60
(0.20–1.50)b |
|
|
|
|
|
|
|
|
| LNP; <5 years of
use | Past use | 0.50
(0.30–0.90)b |
|
|
|
|
|
|
|
|
| LNP; <5 years of
use | Current use | 0.50
(0.30–0.80)b |
|
|
|
|
|
|
|
|
| LNP; 5–10 years of
use | Past use | 1.60
(0.90–2.80)b |
|
|
|
|
|
|
|
|
| LNP; 5–10 years of
use | Current use | 1.20
(0.60–2.20)b |
|
|
|
|
|
|
|
|
| LNP; ≥10 years of
use | Past use | 1.20
(0.60–2.40)b |
|
|
|
|
|
|
|
|
| LNP; ≥10 years of
use | Current use | 0.80
(0.30–1.70)b |
|
|
|
| Hellmann et
al, 2010 | 1976-2007 | Denmark | Cohort | 12,617 | - | - | 0.64
(0.43–0.96)b | Yes | >7 | (35) |
| Newcomb et
al, 2008 | 1988-2005 | USA | Cohort | 12,269 | - | - | 0.87
(0.78–0.98)b | Yes | >7 | (36) |
|
|
|
|
|
| - | Past use | 0.92
(0.78–1.08)b |
|
|
|
|
|
|
|
|
| - | Current use | 0.85
(0.73–0.98)b |
|
|
|
|
|
|
|
|
| <5 years of
use | - | 0.89
(0.73–1.08)b |
|
|
|
|
|
|
|
|
| >5 years of
use | - | 0.89
(0.75–1.06)b |
|
|
|
| Rosenberg et
al, 2008 | 1993-2003 | Sweden | Cohort | 2,660 | - | Current use | 0.63
(0.42–0.95)b | Yes | >7 | (34) |
|
|
|
|
|
| - | Past use | 1.03
(0.68–1.54)b |
|
|
|
|
|
|
|
|
| <5 years of
use | Current use | 0.76
(0.45–1.27)b | Yes |
|
|
|
|
|
|
|
| <5 years of
use | Past use | 1.02
(0.64–1.63)b |
|
|
|
|
|
|
|
|
| >5 years of
use | Current use | 0.52
(0.29–0.93)b |
|
|
|
|
|
|
|
|
| >5 years of
use | Past use | 1.02
(0.48–2.17)b |
|
|
|
| Magnusson et
al, 1996 | 1977-1991 | Sweden | Cohort | 1,589 | - | - | 0.74
(0.49–1.14)d | No | <7 | (57) |
| Willis et
al, 1996 | 1982-1991 | USA | Cohort | 422,373 | - | - | 0.84
(0.75–0.94)c | Yes | >7 | (56) |
|
|
|
|
|
| 2-5 years of
use | - | 0.78
(0.65–0.93)c |
|
|
|
|
|
|
|
|
| 6-10 years of
use | - | 0.78
(0.62–0.98)c |
|
|
|
|
|
|
|
|
| >11 years of
use | - | 0.93
(0.75–1.15)c |
|
|
|
| Sturgeon et
al, 1995 | 1979-1989 | USA | Cohort | 49,000 | - | - | 0.70
(0.70–0.80)c | Yes | <7 | (55) |
| Bonnier et
al, 1998 | 1985-1995 | France | Cohort | 426 | - | - | 0.49
(0.24–0.98)c | No | <7 | (54) |
| Sourander et
al, 1998 | 1987-1995 | Finland | Cohort | 7,317 | - | Past use | 0.94
(0.47–1.90)c | No | >7 | (59) |
|
|
|
|
|
| - | Current use | 0.57
(0.27–1.20)c |
|
|
|
| Sellers et
al, 1997 | 1996-2002 | USA | Cohort | 41,837 | <5 years of
use | - | 1.91
(0.64–5.69)c | Yes | >7 | (53) |
|
|
|
|
|
| >5 years of
use | - | 0.92
(0.55–1.54)c |
|
|
|
| Beral et al,
2003 | 1996-2002 | UK | Cohort | 1,084,110 | - | Current use | 1.22
(1.00–1.48)c | Yes | >7 | (52) |
|
|
|
|
|
| - | Past use | 1.05
(0.85–1.29)c |
|
|
|
| Stahlberg et
al, 2005 | 1993-2004 | Denmark | Cohort | 10,874 | - | Current use | 1.97
(1.14–3.42)b | Yes | <7 | (45) |
|
|
|
|
|
| - | Past use | 1.31
(0.68–2.52)b |
|
|
|
| Norman et
al, 2010 | 1994-2004 | USA | Cohort | 2,502 | <3 years of
use | Past use | 0.79
(0.52–1.21)d | Yes | >7 | (51) |
|
|
|
|
|
| <3 years of
use | Current use | 0.86
(0.54–1.36)d |
|
|
|
|
|
|
|
|
| >3 years of
use | Past use | 0.96
(0.50–1.86)d |
|
|
|
|
|
|
|
|
| >3 years of
use | Current use | 0.81
(0.57–1.15)d |
|
|
|
| Pentti et
al, 2006 | 1994-2001 | Finland | Cohort | 11,667 | <5 years of
use | - | 0.96
(0.32–2.82)b | Yes | >7 | (50) |
|
|
|
|
|
| >5 years of
use | - | 2.62
(0.98–7.00)b |
|
|
|
| Chen et al,
2005 | 1993-2000 | USA | Cohort | 7,436 | HRT use in stage
I | - | 1.23
(0.72–2.10)b | Yes | <7 | (49) |
|
|
|
|
|
| HRT use in stage
II | - | 1.01
(0.72–1.41)b |
|
|
|
| Obi et al,
2016 | 2002-2009 | Germany | Cohort | 3,321 | - | Past use | 1.19
(0.87–1.62)b | Yes | >7 | (48) |
|
|
|
|
|
| - | Current use | 0.72
(0.53–0.97)b |
|
|
|
| Holm et al,
2014 | 1993-2008 | Denmark | Cohort | 1,064 | - | Current use | 0.55
(0.37–0.84)b | Yes | >7 | (47) |
|
|
|
|
|
| <5 years of
use | Current use | 0.64
(0.36–1.12)b |
|
|
|
|
|
|
|
|
| >5 years of
use | Current use | 0.51
(0.31–0.86)b |
|
|
|
| Chlebowski et
al, 2013 | 1993-2011 | USA | Cohort | 41,449 | - | - | 1.32
(0.90–1.93)b | Yes | >7 | (46) |
| Reding et
al, 2011 | 1993-2009 | USA | Cohort | 1,911 | - | - | 0.93
(0.68–1.27)b | Yes | >7 | (44) |
| Pocobelli et
al, 2014 | 1990-2008 | Canada | Nested | 13,823 | - | - | 1.00
(0.80–1.20)d | Yes | >7 | (60) |
|
|
|
| case-control |
|
|
|
|
|
|
|
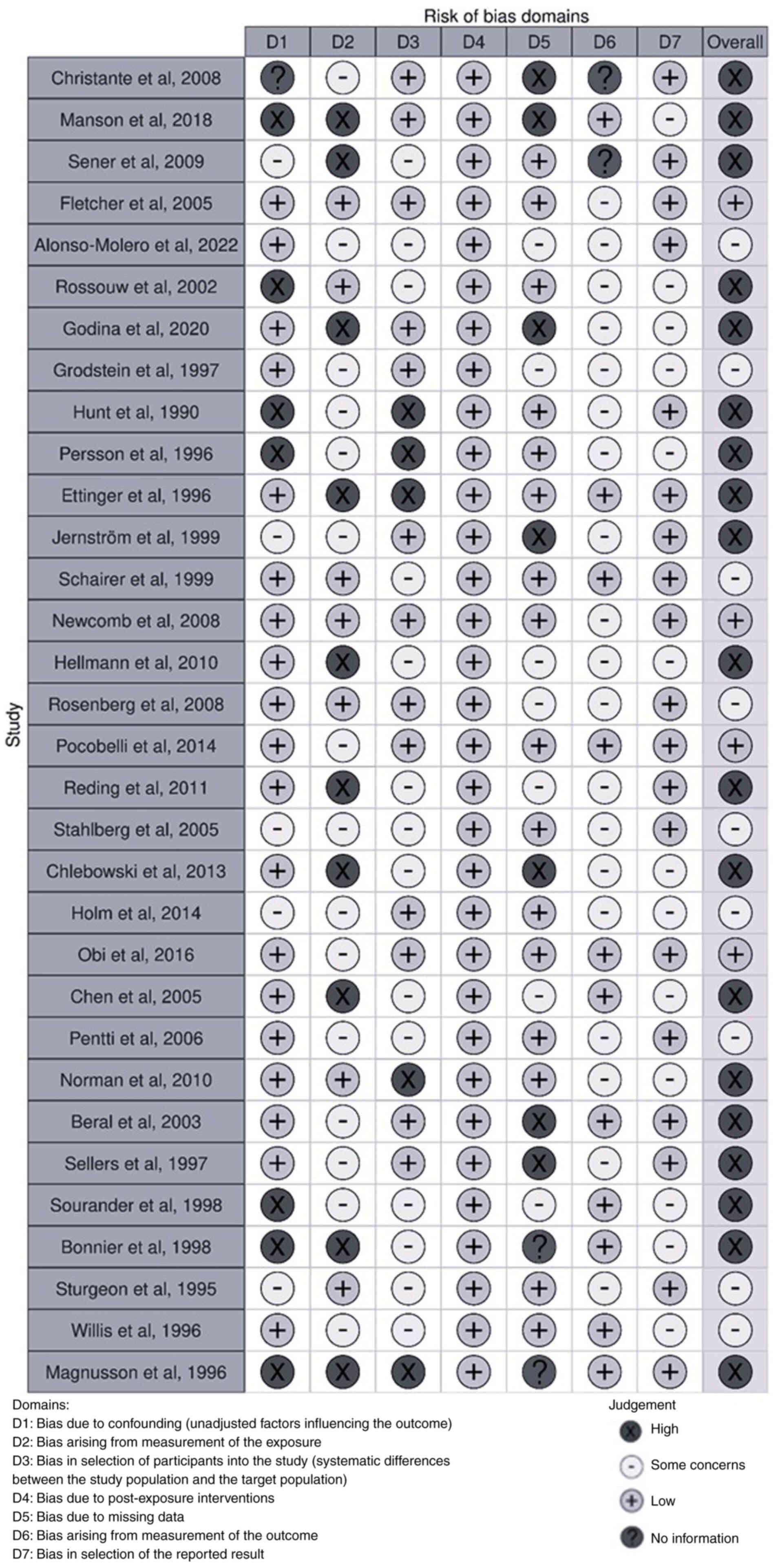
Quality and bias analysis
The results of the quality analysis conducted using
the NOS revealed that 21 of the included studies were classified as
of good or fair quality (NOS>=7) (8,10,15,34–36,38,39,42,44,46–48,50–53,56,58,60,61),
while 8 studies were identified as poor quality (NOS<7)
(40,41,43,45,49,54,55,57).
On the other hand, the application of the ROBINS-E
tool determined that 5 articles had a low risk of bias (36,39,40,48,60),
while 9 articles were identified as having some concerns (10,34,42,45,47,50,55,56,58).
In the remaining 19 studies, a high risk of bias was found
(8,15,32,33,35,37,38,41,43,44,46,49,51–54,57,59,61)
(Fig. 2).
Data analysis
Use of hormone replacement therapy vs.
non-use
First, we aggregated the findings from a total of 29
studies (8,10,15,34–36,38–58,60,61)
that comprehensively compared survival outcomes between women who
had used HRT prior to breast cancer diagnosis and those who had not
(Fig. 3).
These analyses underscored that patients using HRT
had a 14% lower risk of breast cancer mortality compared to those
who did not [OR=0.86; 95% CI: 0.79–0.94]. Heterogeneity among the
included studies was moderate (I2=57.49%, P=0.001).
Inspection of funnel plots revealed asymmetry, indicating a notable
potential for publication bias, which was further supported by
statistical testing (Egger's test: z=−3.20 P=0.001) (Fig. S1).
As a sensitivity analysis, the overall association
was re-estimated after stratifying studies by type of effect
measure. When only hazard ratios were pooled, the combined estimate
(HR 0.88, 95% CI 0.77–1.01) was very similar to the main result,
although the confidence interval was wider and crossed the null
(Fig. S2). When only risk ratios
were considered, the pooled association remained statistically
significant (RR 0.84, 95% CI 0.72–0.99) and closely aligned with
the overall estimate (Fig. S3).
Analyses restricted to the small subset of studies reporting odds
ratios (OR 0.90, 95% CI 0.78–1.03) yielded a pooled estimate
(Fig. S4) that was again of
similar magnitude but less precise. These findings indicate that
the inverse association between pre-diagnostic HRT use and breast
cancer mortality is robust across different effect measures, and
that mixing hazard ratios, risk ratios and odds ratios as generic
relative effects in the main analysis in unlikely to have
materially altered the direction or approximate size of the
association.
Next, we conducted a separate analysis comparing
crude and adjusted estimates (Figs.
S5 and S6). We observed
disparities between these two analyses: crude (OR=0.67, 95% CI:
0.49–0.93) and adjusted (OR= 0.88, 95% CI: 0.80–0.97). The strength
of the association attenuated after adjustment. Regarding
heterogeneity, the studies in the raw analysis showed high
heterogeneity (I2=81.21%, P=0.001), while the adjusted
results indicated a moderate degree (I2=57.82%,
P=0.001).
Impact of duration of HRT use
To stratify the mortality risk according to
treatment duration, two types of analyses were conducted: the first
one included the Women's Health Initiative (WHI) study (15) (Table
II), while the second excluded the Women's Health Initiative
(WHI) study, given that it specifically reported on invasive breast
cancer (Table SII).
 | Table II.Results from the present
meta-analysis. |
Table II.
Results from the present
meta-analysis.
| Subgroup | Number of
studies | Number of
patients | exp(ES) (95%
CI) | I2
(%) |
|---|
| HRT
durationa |
|
|
|
|
| <5
years | 10 | 791,147 | 0.80
(0.69–0.94) | 41.55 |
| 5-10
years | 10 | 791,147 | 0.85
(0.70–1.03) | 54.54 |
| >10
years | 3 | 718,981 | 1.33
(0.96–1.85) | 49.01 |
| HRT use status at
the time of diagnosisa |
|
|
|
|
|
Current | 14 | 1,545,188 | 0.82
(0.70–0.96) | 63.70 |
|
Past | 11 | 1,555,184 | 1.01
(0.90–1.13) | 38.90 |
| Year of
recruitment |
|
|
|
|
| Women
recruited in 2000 or later | 3 | 5,820 | 0.75
(0.60–0.95) | 0.00 |
| Women
recruited before 2000 | 26 | 2,170,399 | 0.87
(0.79–0.96) | 59.51 |
| Article
quality |
|
|
|
|
|
High-quality studies (NOS
≥7) | 21 | 2,104,313 | 0.87
(0.79–0.96) | 66.43 |
|
Moderate-quality (NOS ≤6) | 8 | 71,906 | 0.83
(0.62–1.11) | 68.02 |
| Risk of bias
(ROBINS-E tool evaluation) |
|
|
|
|
|
Low | 5 | 34,507 | 0.83
(0.71–0.96) | 43.12 |
|
Moderate | 9 | 901,023 | 0.93
(0.77–1.12) | 63.51 |
|
High | 18 | 1,275,147 | 0.87
(0.76–0.98) | 54.37 |
HRT use was categorized into three groups based on
duration of use: less than 5 years (15,34,36,42,47,50,53,56,58,61),
between 5 and 10 years (15,34,36,42,47,50,53,56,58,61),
and more than 10 years (15,42,56).
When including the WHI study (15), the analysis for duration of use of
less than 5 years showed an association with lower breast cancer
mortality (Fig. 4). This
association persisted after removing the WHI study from the
analysis (Fig. S7), although both
estimates relied on a relatively low number of contributing studies
and should be interpreted with caution.
For a 5 to 10-year period, the initial analysis
(Fig. 4) established a
non-significant association with breast cancer mortality, whereas
the second analysis, which excluded the WHI study, reached
statistical significance. Again, the WHI study (15) presented the most pronounced
association, with a Hazard Ratio (HR) of 4.61 (95% CI 1.01–21.03).
Heterogeneity was moderate in both analyses.
Finally, for treatment which exceeded 10 years,
there was virtually no difference in the odds ratio (OR) whether
including the WHI study or not (15), showing no clear association. A
distinction was noted when comparing these results with the
Schairer study (42), where
participants were treated for more than 12 years (Table II; Figs. 4 and S7).
Use at the time of diagnosis
When conducting the analysis based on HRT status at
the time of diagnosis, two distinct groups emerged: those actively
using HRT (defined as ‘Current’) (10,34,36,39–42,45,47,48,51,52,59,60)
and those who had previously used it (defined as ‘Past’) (10,15,34,36,42,45,48,51,52,59,60);
(Fig. 5; Table II).
In this analysis, current users had a lower risk of
breast cancer mortality, whereas past users did not differ from
those who had never used HRT (Table
II; Fig. 5). Removing the WHI
study did not materially change the estimates for past users
(Fig. S8).
Significant heterogeneity was observed among the
included studies, which remained similar even when the WHI Study
(15) was excluded.
Year of recruitment
For this analysis, we categorized studies based on
the year of recruitment. On the one hand, studies with participant
recruitment before the year 2000 (10,15,34–36,38–47,49–57,60,61);
on the other, studies with participant recruitment after the year
2000 (8,48,58)
(Fig. 6; Table II). That year marked a turning
point in the use of HRT following the publication of the WHI trial
results (15).
When the recruitment occurred after the year 2000,
HRT use was significantly associated with lower breast cancer
mortality, without heterogeneity across combined studies.
Conversely, when examining studies with earlier recruitment, a
decrease in the strength of the association was noted, with
moderate heterogeneity (Table II;
Fig. 6).
Exploring the influence of article
quality and risk of bias
Finally, we evaluated the impact of study quality
(Fig. S9; Table II). Separating studies by their NOS
score revealed only minor differences. In high-quality studies
(NOS≥7) (8,10,15,34–36,38,39,42,44,46–48,50–53,56,58,60,61),
the strength of the association was slightly weaker than in
moderate-quality studies (NOS<7) (40,41,43,45,49,54,55,57).
Furthermore, heterogeneity was higher in the lower-quality group.
However, the test for subgroup differences was not statistically
significant (P=0.76).
We also stratified the analyses by ROBINS-E risk of
bias (Fig. S10), classifying
studies as having low (36,39,40,48,60),
moderate (34,41,42,45,47,50,55,56,58),
or high risk (8,15,35,38,43,44,46,49,51–54,57,61).
We found a statistically significant association in both low-risk
studies and in high-risk studies, whereas the association did not
reach statistical significance in studies with moderate risk of
bias. Across all three risk-of-bias strata, heterogeneity was
moderate and the overall test for subgroup differences was
insignificant. Taken together, these findings indicate that the
results are broadly consistent across levels of study quality and
risk of bias, although estimates are less precise in studies with
lower-quality or moderate-risk.
Meta-regression analysis
We conducted a meta-regression to assess potential
sources of heterogeneity in our analyses. Predictors such as the
quality of included articles, publication year, and recruitment
year were evaluated. In none of these analyses did any of the
variables appear to influence the heterogeneity shown, as the
R2 value was always zero (Table III). Given the limited number of
studies relative to the number of potential predictors, these
analyses were likely underpowered and should be interpreted with
caution.
 | Table III.Meta-regression analysis. |
Table III.
Meta-regression analysis.
| Subgroup | Coefficient (95%
CI) | τ2 | I2,
% |
|---|
| Model | Not applicable | 0.045 | 57.49 |
| Period of
recruitment before 2000 | −0.136 (−0.260 to
0.530) | 0.065 | 65.95 |
| Year of
publication | −0.006 (−0.020 to
0.008) | 0.067 | 65.29 |
| High quality (NOS
≥7) | 0.061 (−0.200 to
0.330) | 0.066 | 66.23 |
Discussion
Our study has highlighted several key findings that
contribute to the current understanding of the relationship between
HRT and breast cancer prognosis. The results indicate that HRT use
prior to breast cancer diagnosis is associated with improved
survival in breast cancer patients, consistent with findings
reported in a previous meta-analysis (27).
The potential mechanisms that may underlie the
observed association with a better prognosis include the following
factors: 1) women taking HRT tend to adopt healthier lifestyles and
maintain a lower BMI [8], and 2) women using HRT often undergo more
frequent gynecological and breast cancer screenings, leading to
earlier detection of less aggressive (more hormone-dependent)
cancers (62).
Interestingly, our analysis also revealed potential
disparities in the impact of HRT use on breast cancer survival
based on the year of participant recruitment. Studies with
participant recruitment before the year 2000 (10,15,38–41,43,61)
showed lower odds of breast cancer mortality among HRT users than
non-users, whereas studies recruiting after 2000 showed an
attenuated difference (8,58). This temporal difference could be
attributed to changes in HRT prescription practices, formulations,
or population characteristics over time. Although an inverse
association was observed for shorter treatment durations in some
analyses, current use at diagnosis was associated with lower
mortality compared with never having taken HRT, these findings were
not fully consistent across all subgroup analyses. In addition, the
statistical power of the duration of use and use-at-diagnosis
subgroups is limited because these subgroups rely on a relatively
small number of studies. Consequently, these results should be
considered exploratory and interpreted with caution rather than as
definitive evidence of effect modification. Furthermore, to the
best of our knowledge, only three articles included women recruited
after the year 2000, which further restricts the precision of these
estimates and highlights the need for further research in this
field.
Although our pooled analysis suggests that
pre-diagnostic HRT use is associated with improved breast cancer
survival, several lines of evidence indicate that this association
may be at least partially attributable to confounding by health
behaviors, screening intensity, and selective prescribing patterns
rather than reflecting a direct causal protective effect. Multiple
observational studies included in our meta-analysis document
substantial baseline differences between HRT users and non-users.
For example, cohorts with detailed baseline information such as
Newcomb et al (36),
Rosenberg et al (34),
Godina et al (8),
Alonso-Molero et al (58),
Pocobelli et al (60), and
others (42,46–48,50,53–55,57)
consistently show that women using HRT tend to have a lower body
mass index, healthier lifestyle profiles (including lower smoking
rates and more moderate alcohol consumption), higher educational
attainment, and more frequent engagement with preventive health
care, including regular mammography and gynecological check-ups.
These women are also more likely to be diagnosed with earlier-stage
tumours and more favourable prognostic profiles at breast cancer
diagnosis. This pattern is consistent with a ‘healthy estrogen
user’ profile and suggests that residual confounding by unmeasured
or incompletely adjusted lifestyle, comorbidity, and socioeconomic
factors may persist even in studies that performed multivariable
adjustment.
In addition, several studies (8,34,36,42,48)
with detailed information on pre-diagnostic mammographic history
and mode of tumour detection report that HRT users are
significantly more likely to have screen-detected breast cancers,
and their tumours tend to be smaller and less advanced at diagnosis
compared to non-users. This raises the possibility that lead-time
bias and detection bias contribute to the observed survival
advantage, as earlier detection among HRT users may inflate
survival estimates without necessarily altering the underlying
disease course. In this context, the apparently lower breast cancer
mortality among current HRT users should be interpreted with
particular caution, as it is highly susceptible to healthy-user and
survivor biases and may not reflect a direct protective effect of
HRT.
Furthermore, evidence from the Breast Cancer
Detection Demonstration Project (42) and related cohorts indicates that HRT
is selectively prescribed to, and maintained in, healthier women,
and is often discontinued when women develop symptoms of serious
illness. This creates a ‘healthy estrogen user survivor effect’
whereby current users appear to have lower mortality rates not
because HRT confers protection, but because women who remain on HRT
are systematically healthier than those who stop or never start
therapy. In contrast to these observational findings, the
randomised Women's Health Initiative trial (15) which, by design minimizes
healthy-user bias and differential screening, did not demonstrate a
survival benefit from combined oestrogen-plus-progestin therapy.
Taken together, these considerations suggest that while our
meta-analysis demonstrates a consistent inverse association between
pre-diagnostic HRT use and breast cancer mortality across multiple
observational studies, this association should be interpreted
cautiously and may not reflect a true causal protective effect.
Our comprehensive evaluation of study quality and
potential biases using the NOS (29) and the ROBINS-E tool (30) enhances the robustness of our
findings. A substantial proportion of the included studies were
judged to have suboptimal quality or a high risk of bias according
to the NOS and ROBINS-E assessments. However, stratified analyses
showed that the direction and magnitude of the association between
HRT use and breast cancer prognosis were broadly similar across NOS
and ROBINS-E strata, and tests for subgroup differences were not
statistically significant. These findings support the robustness of
the overall association but also indicate that the high prevalence
of bias and the imprecision observed in some subgroups should
temper confidence in the exact magnitude of the association.
Furthermore, it is noteworthy that our analyses
revealed the influence of specific studies on heterogeneity. When
the WHI study (15) was removed
from the analysis of HRT use at the ‘Time of Diagnosis’, the
heterogeneity between studies decreased even further, with an
I2 of 1.05% (P=0.43). The WHI Study (15) itself demonstrated the most
pronounced association in the subgroup analysis of HRT duration of
use analysis for the period of less than 5 years, reporting an RR
of 2.13 (95% CI 1.15–3.94). However, the remaining evaluated
studies indicated an association between HRT use and breast cancer
survival. A moderate-to-high degree of heterogeneity was initially
observed, with an I2 of 63.28% (P=0.01). Nevertheless,
when the WHI study was excluded, the heterogeneity disappeared
(I2=0% and P=0.46). These observations accentuate the
potential influence of specific studies on the overall
heterogeneity of the results. One of the possible explanations that
could justify the discordant results from the WHI study could be
the advanced age of the participants or the inclusion of only those
who showed no signs of cancer on a previous mammogram (63).
The strengths of our study compared to previous
meta-analyses, include the incorporation of the ROBINS-E tool
(30), which enables the systematic
evaluation of potential biases, ensuring methodological rigor in
observational research. Additional strengths include stratification
by publication years and the inclusion of three articles published
after 2017 (8,38,58),
the publication year of the aforementioned meta-analysis. These
articles all feature follow-up periods exceeding 10 years and are
high-quality studies with NOS scores of 7 or higher.
Despite the valuable insights gained from this
meta-analysis, several limitations warrant consideration. First,
the observed heterogeneity among studies could be influenced by
differences in study design, patient populations, treatment
regimens, and methodological approaches. A longer follow-up period
and a more precise assessment of HRT use would be necessary to
provide a more accurate estimation of breast cancer survival with
HRT.
Second, the potential for publication bias, as
suggested by funnel plot asymmetry, indicates that smaller studies
with non-significant findings might be underrepresented.
Third, there could be heterogeneity in the combined
odds ratios (ORs) due to variations in study methodologies; some
studies utilized raw data without controlling for confounding
factors, whereas others adjusted for relevant confounders. However,
the sensitivity analysis restricted to adjusted estimates showed an
association (OR=0.88) very similar to the overall result (OR=0.86),
which supports the robustness of the main finding.
Fourth, the definition of HRT exposure varied
across studies, and the necessity to harmonize different exposure
categories may have introduced non-differential misclassification.
This could attenuate differences between subgroups and bias the
corresponding estimates towards the null.
Fifth, different effect measures (HRs, RRs and ORs)
were combined as generic relative effects, even though these
measures are not strictly interchangeable for time-to-event
outcomes. Nevertheless, sensitivity analyses restricted to each
effect measure separately yielded pooled estimates that were very
similar in magnitude and direction to the main analysis, suggesting
that this approximation is unlikely to have substantially biased
the overall findings.
Sixth, there is a lack of information regarding HRT
dosage, and substantial variations exist in the methods used to
group regimens across studies.
Finally, several subgroup analyses were based on a
limited number of studies in some strata, so these findings should
be interpreted with caution. From a clinical perspective, these
observational findings should not be interpreted as evidence to
initiate or continue HRT use with the aim of improving breast
cancer prognosis.
In conclusion, our systematic review and
meta-analysis suggest that HRT use prior to breast cancer diagnosis
is associated with improved breast cancer survival, although the
statistical power of some subgroup analyses is limited by the low
number of available studies. The observed association with better
survival appears to be influenced by factors such as treatment
duration, timing of discontinuing HRT use, and the year of
participant recruitment. These findings underscore the complexity
of the relationship between HRT use and breast cancer prognosis and
emphasize the need for personalized assessment of HRT use in the
context of breast cancer management. Further research is warranted
to clarify the underlying mechanisms and potential clinical
implications of these observed associations. Importantly, these
observational data do not support the use of HRT as a therapeutic
strategy to enhance breast cancer prognosis.
Supplementary Material
Supporting Data
Supporting Data
Acknowledgements
Not applicable.
Funding
Funding: No funding was received.
Availability of data and materials
The data generated in the present study are
included in the figures and/or tables of this article.
Authors' contributions
JACF contributed substantially to the
conceptualization and design of the study, conducted an independent
bibliographic search, created the database with the relevant
information extracted from each article, contributed to the
analysis and interpretation of the data, and wrote the paper. JAM
conducted an independent bibliographic search and data extraction
as part of the double-blind review process of the meta-analysis.
JACF and JAM confirm the authenticity of all the raw data,
performed the statistical analysis and participated in the critical
revision of the manuscript. JACF and PRC contributed to the
evaluation of the quality by applying the Newcastle Ottawa Scale
and the interpretation of the data. JL supervised the analysis and
interpretation of the data. IGA contributed to the
conceptualization and design of the study and participated in the
critical revision of the manuscript. TDS contributed to the
conceptualization and design of the study, performed the analysis
and contributed to the interpretation of the data, and wrote the
paper. All authors have read and approved the final version of the
manuscript.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
Glossary
Abbreviations
Abbreviations:
|
HR
|
hazard ratio
|
|
HRT
|
hormone replacement therapy
|
|
NOS
|
Newcastle Ottawa Scale
|
|
OR
|
odds ratio
|
|
WHI
|
Women's Health Initiative
|
References
|
1
|
Kashyap D, Pal D, Sharma R, Garg VK, Goel
N, Koundal D, Zaguia A, Koundal S and Belay A: Global increase in
breast cancer incidence: Risk Factors and Preventive Measures.
BioMed Res Int. 2022:96054392022. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Sung H, Ferlay J, Siegel RL, Laversanne M,
Soerjomataram I, Jemal A and Bray F: Global cancer statistics 2020:
GLOBOCAN estimates of incidence and mortality worldwide for 36
cancers in 185 countries. CA Cancer J Clin. 71:209–249.
2021.PubMed/NCBI
|
|
3
|
Breast Cancer, . https://www.who.int/es/news-room/fact-sheets/detail/breast-cancerJanuary
16–2023(In Spanish).
|
|
4
|
Wilkinson L and Gathani T: Understanding
breast cancer as a global health concern. Br J Radiol.
95:202110332022. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Sun YS, Zhao Z, Yang ZN, Xu F, Lu HJ, Zhu
ZY, Shi W, Jiang J, Yao PP and Zhu HP: Risk factors and preventions
of breast cancer. Int J Biol Sci. 13:1387–1397. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Early Breast Cancer Trialists' and
Collaborative Group (EBCTCG), . Aromatase inhibitors versus
tamoxifen in early breast cancer: Patient-level meta-analysis of
the randomised trials. Lancet. 386:1341–1352. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
DeSantis C, Jemal A and Ward E:
Disparities in breast cancer prognostic factors by race, insurance
status, and education. Cancer Causes Control. 21:1445–1450.
2010.PubMed/NCBI
|
|
8
|
Godina C, Ottander E, Tryggvadottir H,
Borgquist S, Isaksson K and Jernström H: Prognostic impact of
menopausal hormone therapy in breast cancer differs according to
tumor characteristics and treatment. Front Oncol. 10:802020.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Cagnacci A and Venier M: The controversial
history of hormone replacement therapy. Medicina (Mex). 55:6022019.
View Article : Google Scholar
|
|
10
|
Grodstein F, Stampfer MJ, Colditz GA,
Willett WC, Manson JE, Joffe M, Rosner B, Fuchs C, Hankinson SE,
Hunter DJ, et al: Postmenopausal hormone therapy and mortality. N
Engl J Med. 336:1769–1776. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Falkeborn M, Persson I, Adami HO,
Bergström R, Eaker E, Lithell H, Mohsen R and Naessén T: The risk
of acute myocardial infarction after oestrogen and
oestrogen-progestogen replacement. Br J Obstet Gynaecol.
99:821–828. 1992. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Kiel DP, Felson DT, Anderson JJ, Wilson PW
and Moskowitz MA: Hip fracture and the use of estrogens in
postmenopausal women. The Framingham study. N Engl J Med.
317:1169–1174. 1987. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Weiss NS, Ure CL, Ballard JH, Williams AR
and Daling JR: Decreased risk of fractures of the hip and lower
forearm with postmenopausal use of estrogen. N Engl J Med.
303:1195–1198. 1980. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Ziel HK and Finkle WD: Increased risk of
endometrial carcinoma among users of conjugated estrogens. N Engl J
Med. 293:1167–1170. 1975. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Rossouw JE, Anderson GL, Prentice RL,
LaCroix AZ, Kooperberg C, Stefanick ML, Jackson RD, Beresford SA,
Howard BV, Johnson KC, et al: Risks and benefits of estrogen plus
progestin in healthy postmenopausal women: Principal results From
the Women's Health Initiative randomized controlled trial. JAMA.
288:321–333. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Ravdin PM, Cronin KA, Howlader N, Berg CD,
Chlebowski RT, Feuer EJ, Edwards BK and Berry DA: The decrease in
breast-cancer incidence in 2003 in the United States. N Engl J Med.
356:1670–1674. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
de Jong-van den Berg LTW, Faber A and van
den Berg PB: HRT use in 2001 and 2004 in The Netherlands-a world of
difference. Maturitas. 54:193–197. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Silverman BG and Kokia ES: Use of hormone
replacement therapy, 1998–2007: Sustained impact of the Women's
Health Initiative findings. Ann Pharmacother. 43:251–258. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Menon U, Burnell M, Sharma A,
Gentry-Maharaj A, Fraser L, Ryan A, Parmar M, Hunter M and Jacobs
I; UKCTOCS Group, : Decline in use of hormone therapy among
postmenopausal women in the United Kingdom. Menopause. 14:462–467.
2007. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Baladé Martínez L, Montero Corominas D and
Macías Saint-Gerons D: Use of hormone replacement therapy in Spain:
Trends in the 2000–2014 period. Med Clin (Barc). 147:287–292.
2016.(In Spanish). View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Vankrunkelsven P, Kellen E, Lousbergh D,
Cloes E, Op de Beeck L, Faes C, Bruckers L, Mertens R, Coebergh JW,
Van Leeuwen FE and Buntinx F: Reduction in hormone replacement
therapy use and declining breast cancer incidence in the Belgian
province of Limburg. Breast Cancer Res Treat. 118:425–432. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Séradour B, Allemand H, Weill A and
Ricordeau P: Sustained lower rates of breast cancer incidence in
France in 2007. Breast Cancer Res Treat. 121:799–800. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Kumle M: Declining breast cancer incidence
and decreased HRT use. Lancet. 372:608–610. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Zbuk K and Anand SS: Declining incidence
of breast cancer after decreased use of hormone-replacement
therapy: Magnitude and time lags in different countries. J
Epidemiol Community Health. 66:1–7. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Gompel A and Santen RJ: Hormone therapy
and breast cancer risk 10 years after the WHI. Climacteric.
15:241–249. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Lobo RA: Hormone-replacement therapy:
Current thinking. Nat Rev Endocrinol. 13:220–231. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Yu X, Zhou S, Wang J, Zhang Q, Hou J, Zhu
L, He Y, Zhao J and Zhong S: Hormone replacement therapy and breast
cancer survival: A systematic review and meta-analysis of
observational studies. Breast Cancer. 24:643–657. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Page MJ, McKenzie JE, Bossuyt PM, Boutron
I, Hoffmann TC, Mulrow CD, Shamseer L, Tetzlaff JM, Akl EA, Brennan
SE, et al: PRISMA 2020 Statement: An updated guideline for
reporting systematic reviews. Rev Esp Cardiol (Engl Ed).
74:790–799. 2021.(In Spanish). View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Wells GA, Shea B, O'Connell D, Peterson J,
Welch V, Losos M and Tugwell P: The Newcastle-Ottawa Scale (NOS)
for assessing the quality of nonrandomised studies in
meta-analyses. 2011.https://www.ohri.ca/programs/clinical_epidemiology/oxford.aspJanuary
19–2023
|
|
30
|
Higgins JPT, Morgan RL, Rooney AA, Taylor
KW, Thayer KA, Raquel A. Silva RA, Courtney Lemeris C, Akl EA, et
al: A tool to assess risk of bias in non-randomized follow-up
studies of exposure effects (ROBINS-E). Environment International.
186:1086022024.Launch version, 20 June, 2023. https://www.riskofbias.info/welcome/robinse-toolJuly
3–2023 View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Egger M, Smith GD, Schneider M and Minder
C: Bias in meta-analysis detected by a simple, graphical test. BMJ.
315:629–634. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Persson I, Yuen J, Bergkvist L and
Schairer C: Cancer incidence and mortality in women receiving
estrogen and estrogen-progestin replacement therapy-long-term
follow-up of a Swedish cohort. Int J Cancer. 67:327–332. 1996.
View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Hunt K, Vessey M and McPherson K:
Mortality in a cohort of Long-term users of hormone replacement
therapy: An updated analysis. Br J Obstet Gynaecol. 97:1080–1086.
1990. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Rosenberg LU, Granath F, Dickman PW,
Einarsdóttir K, Wedrén S, Persson I and Hall P: Menopausal hormone
therapy in relation to breast cancer characteristics and prognosis:
A cohort study. Breast Cancer Res. 10:R782008. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Hellmann SS, Thygesen LC, Tolstrup JS and
Grønbæk M: Modifiable risk factors and survival in women diagnosed
with primary breast cancer: Results from a prospective cohort
study. Eur J Cancer Prev. 19:366–373. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Newcomb PA, Egan KM, Trentham-Dietz A,
Titus-Ernstoff L, Baron JA, Hampton JM, Stampfer MJ and Willett WC:
Prediagnostic use of hormone therapy and mortality after breast
cancer. Cancer Epidemiol Biomarkers Prev. 17:864–871. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Christante D, Pommier S, Garreau J, Muller
P, LaFleur B and Pommier R: Improved breast cancer survival among
hormone replacement therapy users is durable after 5 years of
additional follow-up. Am J Surg. 196:505–511. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Manson JE, Aragaki AK, Rossouw JE,
Anderson GL, Prentice RL, LaCroix AZ, Chlebowski RT, Howard BV,
Thomson CA, Margolis KL, et al: Menopausal hormone therapy and
Long-Term All-Cause and Cause-Specific mortality: The Women's
Health initiative randomized trials. Obstet Gynecol Surv. 73:22–24.
2018. View Article : Google Scholar
|
|
39
|
Fletcher AS, Erbas B, Kavanagh AM, Hart S,
Rodger A and Gertig DM: Use of hormone replacement therapy (HRT)
and survival following breast cancer diagnosis. Breast. 14:192–200.
2005. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Schuetz F, Diel IJ, Pueschel M, von Holst
T, Solomayer EF, Lange S, Sinn P, Bastert G and Sohn C: Reduced
incidence of distant metastases and lower mortality in 1072
patients with breast cancer with a history of hormone replacement
therapy. Am J Obstet Gynecol. 196:342.e1–e9. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Ettinger B, Friedman GD and Quesenberry CP
Jr: Reduced mortality associated with Long-Term postmenopausal
estrogen therapy. Obstet Gynecol. 87:6–12. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Schairer C, Gail M, Byrne C, Rosenberg PS,
Sturgeon SR, Brinton LA and Hoover RN: Estrogen replacement therapy
and breast cancer survival in a large screening study. J Natl
Cancer Inst. 91:264–270. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Sener SF, Winchester DJ, Winchester DP, Du
H, Barrera E, Bilimoria M, Krantz S and Rabbitt S: The effects of
hormone replacement therapy on postmenopausal breast cancer biology
and survival. Am J Surg. 197:403–407. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Reding KW, Doody DR, McTiernan A, Hsu L,
Davis S, Daling JR, Porter PL and Malone KE: Age-related variation
in the relationship between menopausal hormone therapy and the risk
of dying from breast cancer. Breast Cancer Res Treat. 126:749–761.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Stahlberg C, Lynge E, Andersen ZJ, Keiding
N, Ottesen B, Rank F, Hundrup YA, Obel EB and Pedersen AT: Breast
cancer incidence, case-fatality and breast cancer mortality in
Danish women using hormone replacement therapy-a prospective
observational study. Int J Epidemiol. 34:931–935. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Chlebowski RT, Manson JE, Anderson GL,
Cauley JA, Aragaki AK, Stefanick ML, Lane DS, Johnson KC,
Wactawski-Wende J, Chen C, et al: Estrogen plus progestin and
breast cancer incidence and mortality in the Women's health
initiative observational study. J Natl Cancer Inst. 105:526–355.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Holm M, Olsen A, Kroman N and Tjønneland
A: Lifestyle influences on the association between pre-diagnostic
hormone replacement therapy and breast cancer prognosis-Results
from the Danish ‘Diet, Cancer and Health’ prospective cohort.
Maturitas. 79:442–448. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Obi N, Heinz J, Seibold P, Vrieling A,
Rudolph A, Chang-Claude J, Berger J and Flesch-Janys D:
Relationship between menopausal hormone therapy and mortality after
breast cancer The MARIEplus study, a prospective case cohort. Int J
Cancer. 138:2098–2108. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Chen W, Petitti DB and Geiger AM:
Mortality following development of breast cancer while using
oestrogen or oestrogen plus progestin: A computer record-linkage
study. Br J Cancer. 93:392–398. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Pentti K, Honkanen R, Tuppurainen MT,
Sandini L, Kröger H and Saarikoski S: Hormone replacement therapy
and mortality in 52- to 70-year-old women: The Kuopio osteoporosis
risk factor and prevention study. Eur J Endocrinol. 154:101–107.
2006. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Norman SA, Weber AL, Localio AR,
Marchbanks PA, Ursin G, Strom BL, Weiss LK, Burkman RT, Bernstein
L, Deapen DM, et al: Hormone therapy and fatal breast cancer.
Pharmacoepidemiol Drug Saf. 19:440–447. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Beral V and Million Women Study
Collaborators, . Breast cancer and hormone-replacement therapy in
the Million Women Study. Lancet. 362:419–427. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Sellers TA, Mink PJ, Cerhan JR, Zheng W,
Anderson KE, Kushi LH and Folsom AR: The role of hormone
replacement therapy in the risk for breast cancer and total
mortality in women with a family history of breast cancer. Ann
Intern Med. 127:973–980. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Bonnier P, Bessenay F, Sasco AJ, Beedassy
B, Lejeune C, Romain S, Charpin C, Piana L and Martin PM: Impact of
menopausal hormone-replacement therapy on clinical and laboratory
characteristics of breast cancer. Int J Cancer. 79:278–282. 1998.
View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Sturgeon SR, Schairer C, Brinton LA,
Pearson T and Hoover RN: Evidence of a healthy estrogen user
survivor effect. Epidemiology. 6:227–231. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Willis DB, Calle EE, Miracle-McMahill HL
and Heath CW Jr: Estrogen replacement therapy and risk of fatal
breast cancer in a prospective cohort of postmenopausal women in
the United States. Cancer Causes Control. 7:449–457.
1996.PubMed/NCBI
|
|
57
|
Magnusson C, Holmberg L, Nordén T,
Lindgren A and Persson I: Prognostic characteristics in breast
cancers after hormone replacement therapy. Breast Cancer Res Treat.
38:325–334. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Alonso-Molero J, Gómez-Acebo I, Llorca J,
Lope-Carvajal V, Amiano P, Guevara M, Martín V, Castaño-Vinyals G,
Fernández-Ortiz M, Obón-Santacana M, et al: Effect of the use of
prediagnosis hormones on breast cancer prognosis: MCC-Spain study.
Menopause. 29:1315–1322. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Sourander L, Rajala T, Räihä I, Mäkinen J,
Erkkola R and Helenius H: Cardiovascular and cancer morbidity and
mortality and sudden cardiac death in postmenopausal women on
oestrogen replacement therapy (ERT). Lancet. 352:1965–1969. 1998.
View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Pocobelli G, Newcomb PA, Li CI, Cook LS,
Barlow WE and Weiss NS: Fatal breast cancer risk in relation to use
of unopposed estrogen and combined hormone therapy. Breast Cancer
Res Treat. 145:439–447. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Jernström H, Frenander J, Fernö M and
Olsson H: Hormone replacement therapy before breast cancer
diagnosis significantly reduces the overall death rate compared
with never-use among 984 breast cancer patients. Br J Cancer.
80:1453–1458. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
62
|
Cheek J, Lacy J, Toth-Fejel S, Morris K,
Calhoun K and Pommier RF: The impact of hormone replacement therapy
on the detection and stage of breast cancer. Arch Surg.
137:1015–1021. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Chlebowski RT, Anderson GL, Aragaki AK,
Manson JE, Stefanick ML, Pan K, Barrington W, Kuller LH, Simon MS,
Lane D, et al: Association of menopausal hormone therapy with
breast cancer incidence and mortality during Long-term Follow-up of
the Women's Health initiative randomized clinical trials. JAMA.
324:369–380. 2020. View Article : Google Scholar : PubMed/NCBI
|