Introduction
Gastric cancer (GC) remains one of the most common
and deadly malignancies worldwide, posing a significant challenge
to global public health. Although existing therapeutic approaches,
including surgical resection, chemotherapy, radiotherapy and
targeted therapy, have improved patient survival to some degree,
several major challenges continue to limit their effectiveness,
including high rates of recurrence and metastasis and
treatment-associated adverse effects (1).
One major factor underlying these limitations is the
significant heterogeneity of GC. Tumors arising in different
patients frequently harbor diverse genetic alterations and
molecular subtypes, leading to substantial variability in
therapeutic responses and clinical outcomes (2). Conventional treatment approaches are
often accompanied by considerable toxicities. For example,
chemotherapeutic agents frequently cause adverse effects such as
nausea, vomiting and myelosuppression, which may limit their
clinical application and adversely affect patients' quality of life
(3). Therefore, identifying novel
therapeutic targets and developing more effective and safer
treatment strategies have become key priorities in GC research
(4).
To this end, clarifying the molecular pathogenesis
and core molecular drivers of GC is essential for identifying new
therapeutic breakthroughs. The occurrence and development of GC is
a complex multi-step process, mainly involving the abnormal
activation of key signaling pathways including phosphatidylinositol
3-kinase (PI3K)-protein kinase B (Akt), hypoxia-inducible factor-1
(HIF-1) and tumor necrosis factor (TNF), as well as mutations of
critical genes such as tumor protein p53 (TP53) and AKT
serine/threonine kinase 1 (AKT1) (5,6). Core
molecular drivers include the activation of oncogenes, such as
epidermal growth factor receptor (EGFR) (7), the inactivation of tumor suppressor
genes, such as TP53 (6), the
abnormal activation of key signaling pathways, and the regulatory
effects of epigenetic modifications and non-coding RNAs (8). However, the complex interactions among
these drivers remain unclear, hindering the development of
effective targeted therapies.
Traditional Chinese medicine (TCM) offers several
advantages in the management of GC. Owing to its multi-component,
multi-target and multi-pathway characteristics, TCM formulations
can exert antitumor effects while also alleviating common clinical
symptoms, including loss of appetite, nausea, vomiting and pain,
improving patients' overall quality of life (9). Certain TCM-based therapies that focus
on strengthening the body and restoring systemic balance may help
enhance immune function and improve patients' tolerance to both the
disease and subsequent therapeutic interventions (10). For example, curcumin from the TCM
herb Curcuma longa L. exerts anti-GC effects by
downregulating and blocking the Gli1/β-catenin pathway, suppressing
GC cell proliferation and migration (11). The classic TCM formula Sijunzi
Decoction modulates and acts against GC by regulating
N6-methyladenosine modification to inhibit epithelial-mesenchymal
transition (EMT) (12).
Eclipta prostrata L
is the dried aerial part of a plant belonging to the
Asteraceae family. In TCM, it is described as having a sweet taste
and a cold nature and is believed to act on the liver and kidney
meridians. Clinically, it is commonly used to nourish the liver and
kidneys, cool the blood, and promote hemostasis. Phytochemical
studies have revealed that Eclipta prostrata L. contains a
wide variety of bioactive constituents, including triterpenoid
saponins, flavonoids, thiophenes and coumarin ethers. Modern
pharmacological research has demonstrated that this plant possesses
multiple biological activities, such as hemostatic,
hepatoprotective, immunomodulatory, antitumor, anti-inflammatory,
lipid-lowering, anti-hypoxic and anti-snake venom effects (13).
Despite these reported pharmacological activities,
the specific bioactive constituents and molecular mechanisms by
which Eclipta prostrata L. exerts therapeutic effects
against GC remain incompletely understood, and systematic
experimental validation remains limited. Therefore, the present
study combines network pharmacology with experimental verification
to identify the active components of Eclipta prostrata L.
and clarify their potential mechanisms of action in the treatment
of GC, providing a theoretical basis for its clinical
application.
Materials and methods
Reagents and antibodies
The GC AGS cell line and gastric epithelial GES-1
cells were kindly donated by Dr Zhu Anqing (Jiangsu Medical
College). Fetal bovine serum (FBS; cat. no. AB-FBS-1050S) was
purchased from ShangHai ABW Company. RPMI-1640 medium (cat. no.
22400089), DMEM (cat. no. 11960-044) and trypsin-EDTA (0.25%; cat.
no. 25200-056) were obtained from Gibco; Thermo Fisher Scientific,
Inc. Luteolin (cat. no. HY-N0162; purity 99.51%) was obtained from
ShangHai MedChemExpress.
The Cell Counting Kit-8 (CCK-8; cat. no. C0038),
Annexin V-FITC cell apoptosis detection kit (cat. no. C1062M) and
BeyoAOF™ serum-free cell cryopreservation solution (cat. no.
C0210B) were purchased from Beyotime Institute of Biotechnology.
Antibodies against HIF-1A (cat. no. sc-13515) were obtained from
Santa Cruz Biotechnology, Inc., while antibodies against protein
kinase B (AKT; cat. no. 4961) and phosphorylated AKT (p-AKT,
Ser473; cat. no. 4060) were purchased from Cell Signaling
Technology, Inc. β-actin (cat. no. 20536-1-AP) was purchased from
Wuhan Sanying Biotechnology. The secondary antibodies were
purchased from Boster Biological Technology, including goat
anti-rabbit IgG (cat. no. BA1054) and goat anti-mouse IgG (cat. no.
BA1050).
Screening of active components and
target proteins of Eclipta prostrata L
The active ingredients of Eclipta prostrata
L. were identified using the Traditional Chinese Medicine Systems
Pharmacology Database and Analysis Platform (TCMSP; http://www.tcmsp-e.com/), with screening criteria set
at oral bioavailability (OB) ≥30% and drug-likeness (DL) ≥0.18. The
SMILES structures of the selected compounds were subsequently
obtained from PubChem (https://pubchem.ncbi.nlm.nih.gov/).
These SMILES identifiers were then submitted to the
SwissTargetPrediction platform (with species limited to humans;
http://swisstargetprediction.ch/) to
predict potential target genes associated with the active
components of Eclipta prostrata L. Target gene names and
related information were extracted based on the corresponding
SMILES data. The predicted targets were further filtered using a
probability threshold of ≥0.7. After removing duplicate entries, a
total of 290 valid target genes associated with Eclipta
prostrata L. were obtained.
Acquisition and screening of
GC-related targets
Targets associated with GC were retrieved from the
widely used disease-related databases GeneCards (https://www.genecards.org) and Online Mendelian
Inheritance in Man (http://www.omim.org) using ‘GC’ as the search key
word. The gene datasets obtained from these two databases were
subsequently combined, and duplicate entries were removed to
generate a unified list of GC-related genes.
To minimize potential interference from unvalidated
targets and to improve the reliability of the subsequent analyses,
the initial gene set was further refined by retaining only
well-established GC driver genes that have been experimentally
validated and reported in authoritative studies (1,4). These
included core oncogenes, tumor suppressor genes, and key regulators
involved in GC-specific signaling pathways.
Construction of TCM
component-target-disease network and protein-protein interaction
(PPI) network
The intersection between drug-related targets and
disease-associated genes identified through the aforementioned
approaches was determined using Venny (Venny 2.1.0; http://bioinfogp.cnb.csic.es/tools/venny/index.html),
which generated both a Venn diagram and a list of overlapping
genes. The active components, corresponding drug targets, and
GC-related targets were then imported into Cytoscape (version
3.10.2; http://cytoscape.org/) for visualization
and network analysis. Based on these datasets, a traditional
Chinese medicine active component-target-disease interaction
network was constructed to illustrate the potential therapeutic
mechanisms of Eclipta prostrata L. in the treatment of
GC.
The common targets shared between the drug and the
disease were then uploaded to the STRING database (https://string-db.org) to construct a PPI network. The
species parameter was limited to Homo sapiens, and the
interaction confidence score was set to the highest confidence
level (>0.900), while all other parameters were maintained at
their default settings. The resulting protein interaction data were
downloaded in TSV format and imported into Cytoscape 3.10.2 for
visualization. Proteins that did not interact with any other nodes
in the network were excluded from the analysis.
Network topology was further analyzed using the
Network Analyzer tool within Cytoscape. The Degree value, defined
as the number of direct connection edges linked to a given node,
was calculated for each target. This metric reflects the extent of
interaction between a node and other nodes in the network, with
higher Degree values indicating greater biological importance.
Using the ‘Analyze Network’ function in the ‘Tools’ menu, degree
values were calculated for all nodes, and a corresponding PPI
network diagram was generated. Based on these topological
parameters, key targets closely associated with GC were
subsequently identified.
Gene Ontology (GO) analysis and Kyoto
Encyclopedia of Genes and Genomes (KEGG) enrichment pathway
analysis
The shared targets between the drug and the disease
identified in the aforementioned analyses were uploaded to the
Metascape platform (http://metascape.org/gp/index.html), with the species
parameter set to Homo sapiens. The ‘Custom Analysis’ option
was selected, and the significance threshold for both GO analysis
and KEGG pathway enrichment analysis was set at P<0.01.
The GO enrichment analysis included three functional
categories: Biological processes (BP), cellular components (CC) and
molecular functions (MF). All relevant results were exported and
saved for subsequent interpretation. The enrichment outcomes from
the GO and KEGG analyses were then visualized using a
bioinformatics visualization tool (https://www.bioinformatics.com.cn/), which generated
column and bubble charts to illustrate the distribution and
significance of the enriched terms and pathways.
Molecular docking verification of
active components and key targets of the drug
Molecular docking was conducted to simulate
intermolecular interactions between ligands and receptors, enabling
the prediction of potential binding modes and affinities between
candidate drug molecules and the three-dimensional structures of
target proteins. This method enabled the identification of key
active compounds and their potential therapeutic targets. Based on
the Degree values of drug components in the drug-target-disease
network and those of nodes in the PPI network, representative
active ingredients and core target proteins were selected for
molecular docking validation.
The molecular structures of the active ingredients
were obtained from the TCMSP database, while the three-dimensional
structures of the corresponding target proteins were retrieved from
the Protein Data Bank (RCSB; http://www.rcsb.org/). Before docking, the molecular
structures were prepared using PyMOL (version 3.1.8; http://pymol.org/), including the addition of hydrogen
atoms and removal of water molecules. Molecular docking between the
selected compounds and target proteins was then performed using
AutoDock Vina (version 1.1.2; http://vina.scripps.edu/). Finally, the docking
conformations and interaction results were visualized and analyzed
using PyMOL.
Culture of GC AGS and gastric
epithelial GES-1 cell line
AGS cells were cultured in RPMI-1640 medium
supplemented with 10% FBS and maintained in a humidified incubator
at 37°C with 5% CO2. The culture medium was replaced
every two days, and the cells were passaged using 0.25% trypsin for
enzymatic digestion. The gastric epithelial cell line GES-1 was
cultured in DMEM supplemented with 10% FBS. When the cell
confluence reached 80–90%, the cells were digested with 0.25%
trypsin-EDTA for subculture. Cells in the logarithmic growth phase
were used for all experiments.
CCK-8 assay
AGS cells in the logarithmic growth phase were
digested and resuspended to prepare a single-cell suspension, which
was then seeded into 96-well plates at a density of 5,000 cells per
well. A blank control group, a DMSO control group, and treatment
groups with different drug concentrations were established, with
six replicate wells for each condition. Based on the network
pharmacology analysis, the representative active compounds
quercetin, acacetin and luteolin were selected for further
investigation.
After overnight incubation to allow cell adhesion,
the treatment groups were exposed to different concentrations of
the selected compounds (5, 12.5, 25, and 50 µM) for 24 and 48 h.
CCK-8 reagent (20 µl) was then added to each well, and the cells
were incubated for another 1 h at 37°C. Cell viability was then
determined by measuring the absorbance at 450 nm using a microplate
reader. To determine the sub-toxic concentrations of luteolin that
do not affect cell proliferation, concentrations that exerted no
significant inhibitory effect (inhibition rate <5%) were defined
as sub-toxic. Accordingly, 5 and 12.5 µM luteolin were selected as
sub-toxic concentrations for wound healing and Transwell migration
assays to avoid interference from cell proliferation. The CCK-8
assay was used to assess the viability of normal gastric epithelial
GES-1 cells after 48 h of incubation. Luteolin showed no
significant cytotoxicity in GES-1 cells at 5, 12.5, 25 and 50 µM
(inhibition rate <5%), indicating that these concentrations are
safe.
Detection of cell migration rate by
cell scratch test and migration test
Scratch assay
AGS cells in logarithmic growth phase were digested
to obtain a single-cell suspension, then seeded into 6-well plates.
The cells were divided into a blank control group, a DMSO control
group, and luteolin treatment groups (5 and 12.5 µM). After
reaching full confluence, cells were serum-starved in RPMI-1640
medium (cat. no. 22400089; Gibco; Thermo Fisher Scientific, Inc.)
for 12 h to minimize proliferation interference. During the
subsequent scratch assay, cells were maintained in serum-free
RPMI-1640 medium corresponding to each treatment group. A linear
scratch was then made across the cell monolayer using a sterile
20-µl pipette tip. Following washing with PBS to remove detached
cells, the corresponding treatments were added, and the plates were
incubated at 37°C. Wound closure was observed under an inverted
light microscope at 24 and 48 h, and the cell migration rate was
calculated using the formula: Migration rate=(scratch area at 0
h-scratch area at 24 or 48 h)/scratch area at 0 h.
Transwell migration assay
AGS cells in the logarithmic growth phase were
adjusted to a density of 2×105 cells/ml and suspended in
serum-free RPMI-1640 medium, and then seeded into the upper
chambers of Transwell inserts with 8-µm pores. Luteolin (5 and 12.5
µM) or an equal volume of DMSO was added to the upper chamber
medium, while the lower chambers were filled with RPMI-1640 medium
supplemented with 10% FBS. The chambers were placed in 24-well
plates and cells were incubated at 37°C for 24 h. After incubation,
the chambers were removed, and the non-migratory cells on the upper
surface of the membrane were carefully removed using a cotton swab.
The migratory cells were subsequently fixed with 4%
paraformaldehyde at 37°C for 15 min and stained with 0.1% crystal
violet at room temperature for 30 min. Images were captured using
an inverted fluorescence microscope (Olympus Corporation). A total
of five random microscopic fields were then selected for imaging
and quantitative assessment of the migratory cells.
Detection of AGS cell apoptosis by
flow cytometry
AGS cells were collected and transferred to
centrifuge tubes, then washed twice with PBS. A cell suspension
containing 5×104 cells was prepared and centrifuged at
1,000 × g for 5 min, after which the supernatant was carefully
discarded. Subsequently, 195 µl of Annexin V-FITC binding buffer
was added to resuspend the cell pellet gently.
Next, 5 µl of Annexin V-FITC reagent was added,
followed by 10 µl of propidium iodide staining solution, with
gentle mixing after each addition. The cells were incubated at room
temperature in the dark for 10–20 min, then placed on ice. Finally,
apoptosis was assessed by flow cytometry using a cyFlow Space
Sorter (Sysmex Corporation), allowing quantification of early and
late apoptotic cells and necrotic cells. The resulting data were
recorded and analyzed using FlowJo software (version 10.8.1; FlowJo
LLC).
Western blotting
AGS cells in the logarithmic growth phase were
digested to obtain a single-cell suspension and then seeded into
6-well plates. The cells were divided into a DMSO control group and
luteolin treatment groups (25 and 50 µM). Following treatment, the
cells were washed twice with PBS, and total cellular proteins were
extracted using RIPA lysis buffer (cat. no. R0010; Beijing Solarbio
Science & Technology Co., Ltd.). Protein concentrations were
measured using the BCA assay.
Protein loading buffer was subsequently added, and
the samples were denatured at 95°C for 5 min before
electrophoresis. A total of 30 µg of protein was loaded per lane.
Proteins were separated by 10% SDS-PAGE (cat. no. P1200; Beijing
Solarbio Science & Technology Co., Ltd.) and transferred onto
0.45-µm PVDF membranes (cat. no. IPVH00010; MilliporeSigma). The
membranes were then blocked with 5% skim milk for 2 h at 37°C and
incubated overnight at 4°C with the corresponding primary
antibodies at the following dilutions: AKT and p-AKT at 1:1,000,
HIF-1A at 1:250, and β-actin at 1:5,000. On the following day, the
membranes were washed with PBS containing 0.1% Tween-20 (cat. no.
T8220; Beijing Solarbio Science & Technology Co., Ltd.) and
incubated with HRP-conjugated secondary antibodies at a dilution of
1:5,000 at room temperature for 2 h. Protein bands were detected
using enhanced chemiluminescence (ECL; cat. no. WBKLS0500;
MilliporeSigma).
The grayscale intensity of each band was quantified
using ImageJ (version 2.x; National Institutes of Health), with
β-actin serving as the internal reference. Relative changes in the
expression levels of the target proteins were then calculated.
Statistical analysis of data
All data are presented as the mean ± standard
deviation (SD). The CCK-8 cell proliferation assay was performed in
six independent replicates, while all other experiments were
conducted in triplicate. Statistical analyses were conducted using
SPSS (version 19.0; IBM Corp.). Differences among groups were
assessed by one-way analysis of variance, followed by the LSD-t
post hoc test for pairwise comparisons. P<0.05 was considered to
indicate a statistically significant difference.
Results
Acquisition and screening of active
components and drug-related targets of Eclipta prostrata L
The active ingredients of Eclipta prostrata
L. were obtained from the TCMSP database. Applying the
screening criteria of OB ≥30% and DL ≥0.18, a total of 10 active
compounds were identified (Table
I). The SMILES structures of these compounds were then used to
predict potential target genes through the SwissTargetPrediction
platform, with the species limited to humans. The predicted targets
were subsequently filtered by probability, and duplicate entries
were removed to generate the final set of targets associated with
the drug's active components.
 | Table I.Information on the active ingredients
of Eclipta prostrata L. |
Table I.
Information on the active ingredients
of Eclipta prostrata L.
| Mol ID | Molecule name | Chemical
formula | Molecular
weight | Oral
bioavailability (%) | Drug likeness | Content in
Eclipta prostrata L. (%, dry weight) |
|---|
| MOL003404 | Wedelolactone |
C16H10O7 | 314.26 | 49.6 | 0.48 | 0.045–0.062 |
| MOL003402 | Demethyl
wedelolactone |
C17H8O7 | 302.25 | 72.13 | 0.43 | 0.032–0.041 |
| MOL003398 | Pratensein |
C16H12O6 | 299.27 | 39.06 | 0.28 | 0.015–0.020 |
| MOL003389 |
3′-O-METHYLOROBOL |
C16H12O6 | 300.28 | 57.41 | 0.27 | 0.012–0.016 |
| MOL003378 |
1,3,8,9-tetrahydroxybenzofurano
[3,2-c]chromen-6-one |
C17H8O7 | 300.23 | 33.94 | 0.43 | 0.009–0.013 |
| MOL002975 | Butin |
C17H12O7 | 272.27 | 69.94 | 0.21 | 0.022–0.028 |
| MOL001790 | Linarin |
C28H32O14 | 592.6 | 39.84 | 0.71 | 0.010–0.015 |
| MOL001689 | Acacetin |
C16H12O7 | 284.28 | 34.97 | 0.24 | 0.008–0.011 |
| MOL000098 | Quercetin |
C17H10O7 | 302.25 | 46.43 | 0.28 | 0.015–0.018 |
| MOL000006 | Luteolin |
C17H10O6 | 286.25 | 36.16 | 0.25 | 0.021–0.035 |
Acquisition of potential targets of
Eclipta prostrata L. in intervening GC
Using ‘GC’ and ‘gastric carcinoma’ as search
keywords, disease-related targets were retrieved from the GeneCards
and Online Mendelian Inheritance in Man databases. After removing
duplicate entries, a consolidated list of GC-related target genes
was obtained. The initial gene set was then further refined by
retaining only well-established driver genes reported in
authoritative GC studies (1,4),
excluding unvalidated candidate genes and reducing potential
analytical bias.
Subsequently, the intersection between the
drug-related targets and disease-associated genes was identified
using Venny (Venny 2.1.0). A total of 166 common targets shared
between Eclipta prostrata L. and GC were obtained. The
overlap of these targets is illustrated in a Venn diagram (Fig. 1).
TCM component-target-disease
network
The identified active ingredients of Eclipta
prostrata L. and their shared targets with GC were imported
into Cytoscape (version 3.10.2) to construct the TCM
component-target-disease interaction network. In the resulting
network diagram, the purple polygon represents Eclipta prostrata
L., blue rectangles indicate its active components, green
ellipses correspond to the 166 shared targets, and the orange node
represents GC (Fig. 2A).
Topological analysis of the network was conducted
using the Network Analyzer tool in Cytoscape. The Degree value
represents the number of direct interactions between a component
and its targets; thus, a higher Degree value indicates greater
influence within the network. Based on this metric, the three
compounds with the highest Degree values were quercetin, luteolin
and acacetin. To delineate the core bioactive constituents
underlying the anti-GC effects of Eclipta prostrata L.,
simplified component-target-disease networks for the major active
ingredients were constructed using Cytoscape 3.10.2. As summarized
in Fig. 2B-D, network analysis
revealed robust and intricate target connectivity profiles for
quercetin, luteolin and acacetin. These data collectively validate
that the three flavonoids are the principal active components of
Eclipta prostrata L., and that a sophisticated multi-target
regulatory mechanism underpins their anti-GC activities.
Construction of the PPI network and
analysis of core targets
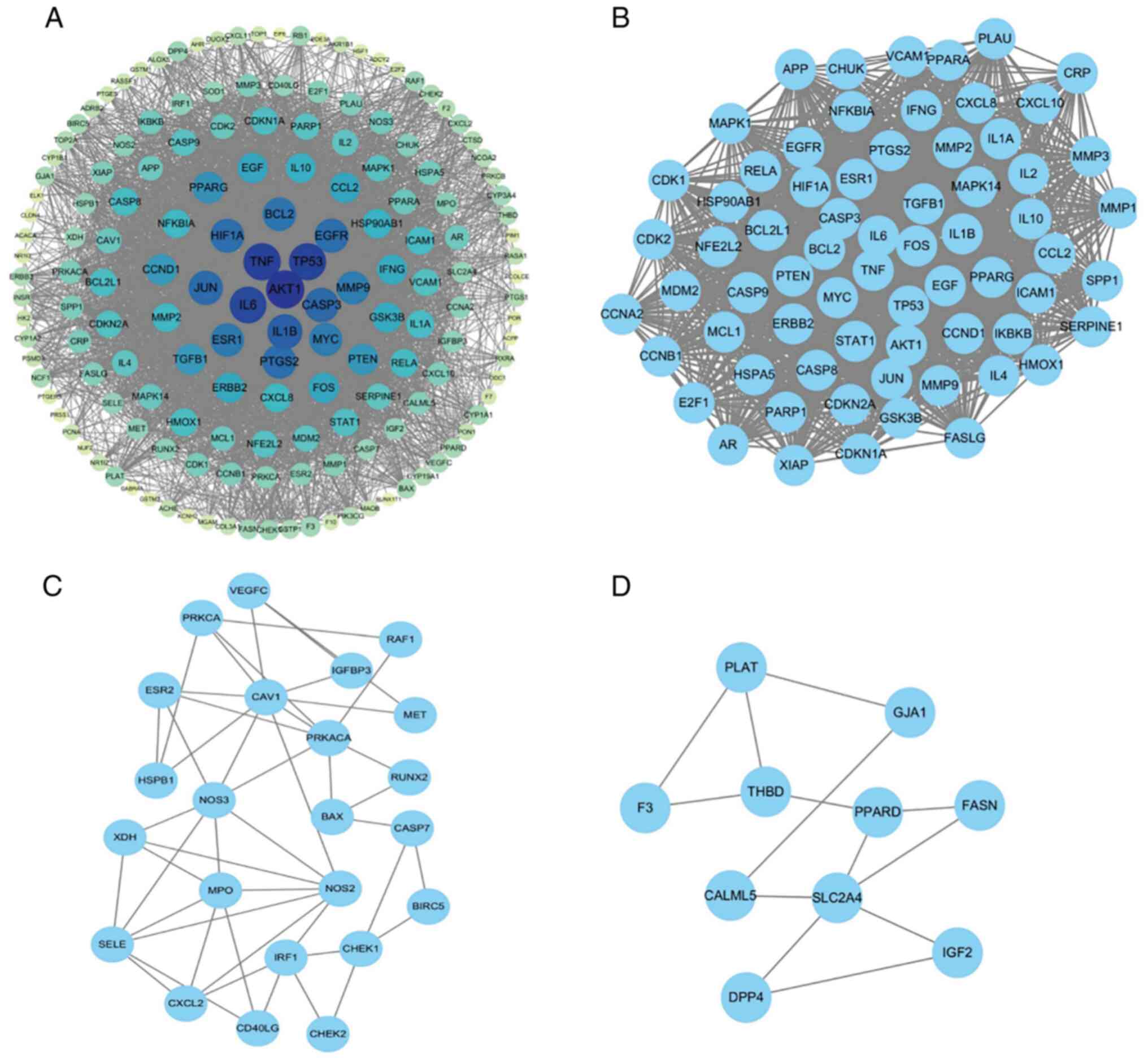
The PPI network comprised 166 nodes. As shown in
Fig. 3A, the size and color
intensity of each node reflected its Degree value, with larger and
darker nodes indicating a higher level of connectivity within the
network. The top 10 target proteins, ranked by Degree, are
presented in Table II.
 | Table II.Target proteins with the top 10
degree values in the protein-protein interaction network
diagram. |
Table II.
Target proteins with the top 10
degree values in the protein-protein interaction network
diagram.
| Serial number | Target | Degree value |
|---|
| 1 | AKT1 | 130 |
| 2 | TNF | 126 |
| 3 | TP53 | 125 |
| 4 | IL6 | 123 |
| 5 | IL1B | 115 |
| 6 | CASP3 | 112 |
| 7 | JUN | 111 |
| 8 | PTGS2 | 110 |
| 9 | EGFR | 110 |
| 10 | HIF1A | 109 |
MCODE clustering analysis identified three
significant subnetworks, with scores of MCODE Cluster 1=54.866,
MCODE Cluster 2=4.273 and MCODE Cluster 3=2.889 (Fig. 3B-D). Analysis of the PPI network
indicated that several key targets, including AKT1, TNF, TP53, IL6,
IL1B, CASP3, JUN, PTGS2, EGFR and HIF-1A, are closely associated
with GC. These findings suggest that these proteins may play
important roles in mediating the therapeutic effects of Eclipta
prostrata L. in GC.
Results of molecular docking
verification
The three compounds with the highest Degree values
in the ‘TCM component-target-disease network’, quercetin, luteolin
and acacetin, together with the 10 potential core targets
demonstrating the highest Degree values, namely AKT1, TNF, TP53,
IL6, IL1B, CASP3, JUN, PTGS2, EGFR and HIF-1A, were selected for
molecular docking analysis.
According to the binding energy criterion, where
values <-5.0 kcal·mol−¹ indicate favorable
interactions, the docking results showed that quercetin, luteolin,
and acacetin all displayed binding energies below this threshold
(Table III), suggesting stable
and favorable interactions with their predicted targets. Subsequent
cellular experiments revealed that luteolin exerted a stronger
inhibitory effect on AGS cell proliferation than quercetin or
acacetin. To further clarify the molecular basis underlying this
enhanced activity, the binding modes of luteolin with the 10 core
targets were examined using both three-dimensional structural
alignment and two-dimensional interaction analysis. As illustrated
by the 3D docking conformations (Fig.
4), luteolin successfully docked into the active sites of all
10 potential core targets, forming stable conformations stabilized
by key molecular interactions. Furthermore, the high-resolution 2D
interaction maps (Fig. S1)
provided atomic-level insights, revealing that luteolin binding was
primarily anchored by conventional hydrogen bonds and extensive
hydrophobic interactions. Notably, luteolin specifically bound to
critical functional domains of key GC-related targets, including
the ATP-binding catalytic pocket of AKT1 and the transactivation
domain of HIF-1A, essential structural regions required for their
biological activities. These combined 3D and 2D analyses validate
the robust binding capacity of luteolin and elucidate the specific
molecular basis by which it modulates the functions of these core
targets.
 | Table III.Molecular docking results of active
ingredients of Eclipta prostrata L. and common targets. |
Table III.
Molecular docking results of active
ingredients of Eclipta prostrata L. and common targets.
|
|
Affinity/(kcal·mol-1) |
|---|
|
|
|
|---|
| Gene | Acacetin | Luteolin | Quercetin |
|---|
| TP53 | −7.4 | −7.5 | −6.8 |
| IL6 | −7.7 | −7.8 | −7.7 |
| PTGS2 | −9.1 | −9.2 | −9.7 |
| CASP3 | −7.2 | −7.1 | −7 |
| JUN | −8.1 | −8.4 | −8.5 |
| TNF | −7 | −6.8 | −6.7 |
| AKT1 | −6.5 | −6.4 | −6.5 |
| EGFR | −9.4 | −9.3 | −8.7 |
| HIF1A | −6.9 | −6.9 | −6.5 |
| IL1B | −7.2 | −7.5 | −6.9 |
Results of GO and KEGG enrichment
analyses
To further clarify the potential core targets and
pathological mechanisms through which Eclipta prostrata L.
exerts therapeutic effects against GC, GO and KEGG pathway
enrichment analyses were performed on the shared targets identified
between Eclipta prostrata L. and GC.
The GO enrichment analysis results (Fig. 5A) showed that the major BP
associated with the therapeutic effects of Eclipta prostrata
L. in GC included responses to lipopolysaccharide,
bacterial-derived molecules, xenobiotic stimuli, cellular chemical
stress and oxidative stress. The enriched CC were mainly associated
with membrane rafts, membrane microdomains, protein kinase
complexes, vesicle lumens and serine/threonine protein kinase
complexes. Regarding MF, the identified targets were primarily
involved in DNA-binding transcription factor binding, RNA
polymerase II-specific DNA-binding transcription factor binding,
cytokine receptor binding, ubiquitin-like protein ligase binding,
and nuclear receptor activity (Fig.
5A).
KEGG pathway enrichment analysis further indicated
that the potential mechanisms may involve several cancer-related
signaling pathways, including the TNF signaling pathway, PI3K-Akt
signaling pathway, p53 signaling pathway, MAPK signaling pathway
and HIF-1 signaling pathway (Fig.
5B). These results suggest that multiple signaling networks may
collectively contribute to the anti-GC effects of Eclipta
prostrata.
Effect of luteolin, the main component
of Eclipta prostrata L., on the proliferation and migration of GC
AGS cell line
The CCK-8 assay was conducted to assess the effects
of the three principal active compounds, luteolin, acacetin and
quercetin, on the proliferation of AGS cells. The results indicated
that quercetin (25 and 50 µM) did not significantly influence cell
viability after either 24 or 48 h of treatment. Similarly, acacetin
(25 and 50 µM) showed no significant effect on cell viability after
24 h of exposure; however, treatment with 50 µM acacetin for 48 h
resulted in an excessively high inhibitory rate on cell
proliferation (69.8%) (Fig. S2A and
B).
In comparison, treatment with 50 µM luteolin for 24
h significantly suppressed AGS cell proliferation (P<0.001).
After 48 h of exposure to 25 and 50 µM luteolin, the inhibitory
rates in the treatment groups were significantly higher than those
observed in the DMSO control group (P<0.001) (Fig. 6C).
 | Figure 6.Effects of luteolin on the
proliferation and migration of gastric cancer AGS cell line. (A and
D) Detection of the migration rate by the cell scratch assay, cells
were treated with luteolin (12.5, 25 µM) for 24 or 48 h. Values are
expressed as the mean ± SD (n=3). (B and E) Detection of the cell
migration ability by the Transwell assay, cells were treated with
luteolin (12.5, 25 µM) for 24 or 48 h. Values are expressed as the
mean ± SD (n=3). (C) Detection of the cell proliferation activity
by the Cell Counting Kit-8 assay, cells were treated with luteolin
(0, 5, 12.5, 25, 50 µM) for 24 or 48 h. Values are expressed as the
mean ± SD (n=6). Compared with DMSO control, **P<0.01. |
Therefore, luteolin was selected for further
investigation because it exhibited a more potent and moderate
inhibitory effect on AGS cell proliferation compared with quercetin
and acacetin. In addition, luteolin is a well-known flavonoid with
established safety and potent antitumor activity in various cancer
models, supporting its rationality as the key component for
mechanistic research (14,15).
To identify sub-toxic concentrations of luteolin
that would not affect cell proliferation, an additional CCK-8 assay
was performed. Concentrations producing no significant inhibitory
effect on cell viability (inhibition rate <5%) were defined as
sub-toxic. Based on this analysis, 5 and 12.5 µM luteolin were
selected for subsequent wound healing and Transwell migration
assays to ensure that any observed changes in migration were
independent of effects on cell proliferation (Fig. 6C). The wound healing (scratch) assay
demonstrated that after 48 h of incubation, the migration rate of
cells treated with luteolin (12.5 µM) was significantly reduced
compared with that of the DMSO control group (P<0.05) (Fig. 6A and D).
To further evaluate cell migration, a Transwell
assay using 8-µm pore chambers was performed at 24 and 48 h of
culture. In the DMSO group, a large number of crystal
violet-stained cells were observed on the lower surface of the
membrane, indicating substantial migratory activity. In comparison,
the number of migratory cells in the luteolin-treated group (12.5
µM) was significantly reduced compared with the control group,
showing a statistically significant difference (P<0.01)
(Fig. 6B and E).
Cytotoxicity of luteolin on normal
gastric epithelial GES-1 cells
To ensure the safety of the working concentrations
used in migration assays, the cytotoxicity of luteolin on normal
gastric epithelial GES-1 cells was detected by CCK-8 assay. As
shown in Fig. S2C, treatment with
5, 12.5, 25, or 50 µM luteolin for 48 h showed no significant
effect on GES-1 cell viability, with inhibition rates <5%. These
results clearly indicated that luteolin exerted no obvious toxic
effect on normal gastric mucosal epithelial cells at the
concentrations used for subsequent migration experiments (Fig. S2C).
Effect of luteolin, the main component
of Eclipta prostrata L., on the apoptosis of GC cell line
Flow cytometry was performed to determine the
combined proportion of early- and late-apoptotic AGS cells after
treatment with luteolin (25 and 50 µM) for 48 h. The results showed
that the average apoptotic rate in the DMSO control group was
17.7%, whereas the rates in the 25 and 50 µM luteolin treatment
groups were 16.7 and 18.02%, respectively. No statistically
significant differences were detected among the three groups
(Fig. 7).
Effect of luteolin, the main component
of Eclipta prostrata L., on the core targets AKT and HIF-1A of GC
cell line
GO and KEGG enrichment analyses revealed that the
serine/threonine protein kinase complex and the HIF-1 signaling
pathway were among the major pathways associated with the shared
targets of Eclipta prostrata L. and GC. To further explore
this mechanism, western blot analysis was conducted to evaluate the
effects of luteolin on the expression of the core target proteins
AKT1 and HIF-1A in GC cells.
The results showed that, compared with the DMSO
control group, treatment with 25 or 50 µM luteolin significantly
reduced AKT phosphorylation at Ser473, and it decreased the protein
expression level of HIF-1A in GC cells (Fig. 8A-D). These findings suggest that
luteolin may inhibit the proliferation and migration of GC cells by
modulating the key regulatory proteins AKT1 and HIF-1A.
Discussion
GC remains the fifth most frequently diagnosed
malignancy and the fourth leading cause of cancer-related death
worldwide. Despite ongoing advances in therapeutic strategies,
several significant limitations remain. Conventional treatments,
including surgery, chemotherapy and radiotherapy, can effectively
eliminate tumor cells; however, their clinical effectiveness is
often limited by chemotherapy resistance, considerable toxicity and
adverse effects, high rates of postoperative recurrence and
treatment-related immunosuppression (1). In comparison, TCM provides a
complementary therapeutic approach characterized by multi-component
and multi-target synergistic effects. Through these mechanisms, TCM
has shown potential to inhibit tumor cell proliferation, promote
apoptosis, suppress angiogenesis, and regulate the tumor immune
microenvironment (9).
Eclipta prostrata L., a traditional herbal
medicine known for its yin-nourishing and blood-cooling properties,
has attracted increasing attention for its potential antitumor
activity. Phytochemical investigations have revealed that its major
bioactive constituents include alkaloids, flavonoids and
polysaccharides (13). Previous
studies have shown that ethanol extracts of Eclipta prostrata
L. can exert anticancer effects by suppressing tumor cell
proliferation, inducing apoptosis, and regulating oncogene
expression (16,17). Furthermore, the classical herbal
formulation Erzhi Pill, composed of Eclipta prostrata L. and
Ligustrum lucidum, has demonstrated improved efficacy in
tumor therapy (18,19). Nevertheless, the specific active
components and molecular mechanisms through which Eclipta
prostrata L. exerts therapeutic effects against GC remain
inadequately defined.
In the present study, the potential mechanisms
underlying the anti-GC effects of Eclipta prostrata L. were
investigated by combining network pharmacology with in vitro
cellular experiments. The network pharmacology analysis identified
several key bioactive compounds, including quercetin, luteolin and
acacetin, which may regulate core targets such as AKT1, TNF, TP53,
IL6, IL1B, CASP3, JUN, PTGS2, EGFR and HIF-1A. These targets are
closely associated with critical biological processes involved in
tumor progression, including cell proliferation, apoptosis and
migration (12,20). Furthermore, MCODE clustering
analysis indicated that Cluster 1 represents a major functional
module within the interaction network.
Molecular docking analysis confirmed strong binding
affinities between the identified compounds and the predicted core
targets. Specifically, 3D structural conformations and 2D
interaction profiling revealed that luteolin forms a stable network
of conventional hydrogen bonds with all 10 core targets, indicating
high target compatibility and favorable binding stability.
Consistent with these structural observations, luteolin has been
reported to show antitumor activity in various cancer models. For
example, luteolin has been shown to inhibit prostate cancer cell
proliferation by suppressing the AKT/mTOR signaling pathway
(21). It can also reduce the
proliferation and migration of esophageal cancer cells by
regulating EMT (17). Similarly,
luteolin has demonstrated synergistic effects when combined with
chemotherapeutic agents or targeted therapies, helping to reverse
drug resistance, enhance therapeutic efficacy, and reduce systemic
toxicity (22,23). It has been reported that luteolin
shows relatively low toxicity toward normal cells; concentrations
effective against tumor cells produce minimal effects on normal
gastric epithelial cells, and animal studies have shown no
significant organ toxicity (24,25).
Therefore, consistent with previous findings, luteolin may be the
main active component of Eclipta prostrata L. against GC.
Luteolin formed hydrogen bonds with all 10 core targets and
specifically bound to the key functional domains of GC-critical
AKT1 and HIF-1A. Binding to AKT1′s ATP-binding pocket may hinder
its phosphorylation and activation, consistent with the present
western blot results, which revealed reduced p-AKT levels. For
HIF-1A, binding to its transactivation domain may impair its
transcriptional activity, as supported by reduced in vitro
HIF-1A expression (26). This
aligns with reported luteolin regulatory mechanisms in gastric and
other solid tumors, further confirming specific binding to
functional domains as the key molecular basis for its anti-GC
activity.
To further explore the potential bioactive
components and therapeutic effects of Eclipta prostrata L.
against GC, a CCK-8 assay was conducted to assess the effects of
the key active compounds, quercetin, luteolin and acacetin,
identified through network pharmacology analysis, on AGS cell
proliferation. The results demonstrated that treatment with
luteolin at appropriate concentrations significantly inhibited the
proliferation and migration of AGS cells after 24–48 h of exposure,
and its inhibitory effect was more pronounced than that of
quercetin or acacetin. Of particular note, luteolin at
concentrations that effectively inhibited the migration of GC cells
displayed no cytotoxicity toward normal gastric mucosal epithelial
GES-1 cells. This selective inhibitory effect suggests that
luteolin can suppress the malignant migration of GC cells without
damaging normal gastric mucosa, thus presenting favorable
biosafety. Flow cytometric analysis revealed that treatment with
luteolin (25 and 50 µM) for 48 h did not produce a statistically
significant change in the apoptotic rate of AGS cells compared with
the control group. These results suggest that luteolin, a major
active constituent of Eclipta prostrata L., inhibits GC
cells primarily by suppressing proliferation and migration rather
than inducing apoptosis. Yang et al (17) isolated luteolin from Eclipta
prostrata L. with a content of 0.21~0.35 mg/g dry weight and
verified its potent antiproliferative activity against cancer
cells, which supports the selection of luteolin as the
representative component for in vitro mechanistic
verification in the present study.
KEGG pathway enrichment analysis identified several
signaling pathways, including TNF signaling, PI3K-Akt signaling,
p53 signaling and HIF-1 signaling, as potential mechanisms
underlying the anti-GC effects of Eclipta prostrata L. The
involvement of the PI3K-Akt pathway in regulating pathological
processes has also been demonstrated in other disease models. For
instance, studies on silica-induced pulmonary fibrosis have shown
that Galectin-3 can regulate endothelial-to-mesenchymal transition
through the PI3K/AKT signaling pathway, underscoring the conserved
role of this pathway in controlling cellular phenotypic transitions
and disease progression (27).
Extensive evidence indicates that aberrant
activation of AKT1 contributes to tumor initiation and progression
by regulating a range of cellular processes, including cell
survival, proliferation, migration and metabolism (28–30).
HIF-1A plays a critical role in tumor development. In the hypoxic
tumor microenvironment, HIF-1A expression is significantly
increased (31). As a key regulator
of cellular adaptation to hypoxia, HIF-1A controls angiogenesis and
metabolic reprogramming, particularly glucose metabolism, by
regulating the transcription of numerous downstream target genes,
promoting tumor progression (32,33).
The present study confirmed that luteolin not only significantly
inhibits the phosphorylation-dependent activation of AKT (the
functionally active form of AKT1) but also significantly
downregulates HIF-1A protein expression. These findings initially
confirm that luteolin exerts a synergistic inhibitory effect on the
occurrence and development of GC through dual regulation of AKT1
activity and the protein expression of HIF-1A, directly validating
that these two core targets are key mediators of the anti-GC effect
of Eclipta prostrata L. Notably, a previous study by the
authors demonstrated that luteoloside (luteolin-7-O-glucoside), a
key flavonoid glycoside from Eclipta prostrata L., exerted
anti-GC effects via the p53/p21 signaling pathway (20). The present study further identified
that luteolin, the aglycone of luteoloside, inhibits GC primarily
through the AKT1/HIF-1A signaling pathway. These results indicate
that different flavonoid components of Eclipta prostrata L.
can suppress GC through distinct molecular mechanisms and signaling
pathways. Building on the aforementioned analysis of cell-intrinsic
signaling pathways, it is important to note that beyond
cell-intrinsic signaling, tumors actively manipulate systemic
homeostasis through neuroendocrine and metabolic pathways, creating
a permissive environment for progression (34,35).
Future studies should examine whether Eclipta prostrata L.
or its components influence these systemic axes, potentially
amplifying their therapeutic impact.
The present study integrated network pharmacology
with in vitro experimental validation to explore the
pharmacological basis and potential mechanisms underlying the
anti-GC activity of Eclipta prostrata L. Network
pharmacology analysis identified quercetin, luteolin and acacetin
as the main active constituents. These compounds were predicted to
interact with several key molecular targets, including AKT1, TNF
and TP53, suggesting that the therapeutic effects of Eclipta
prostrata L. may be achieved through the coordinated regulation
of multiple targets and signaling pathways. As the major
representative active component of Eclipta prostrata L. with
the strongest anti-GC activity in the current screening, luteolin
is a key material basis for the herb's antitumor effects, and its
regulatory role in cancer-related signaling pathways is consistent
with the anti-GC mechanisms of Eclipta prostrata L. extracts
reported in existing literature. In vitro experiments
further demonstrated that luteolin significantly inhibited the
proliferation and migration of GC cells, reduced AKT1
phosphorylation, and decreased the protein expression level of
HIF-1A. These findings provide direct experimental evidence
supporting the identification of the key active component and
molecular targets through which Eclipta prostrata L. exerts
its therapeutic effects (Fig. 9).
Several limitations in the present study should be noted. It is
acknowledged that GeneCards and OMIM are comprehensive but not
GC-specific databases that may include some unvalidated
cancer-related genes; however, multiple screening strategies were
applied to minimize this bias. In addition, only the purified
component, luteolin, was investigated, without evaluating the
synergistic antitumor effects of multiple components in the herb.
Furthermore, no in vivo animal models or GC cell lines of
different molecular subtypes were included. Future studies will
explore the synergistic effects of key herbal components, validate
efficacy and safety in in vivo models and diverse GC cell
lines, and investigate crude extracts to clarify further the
anti-GC activity and mechanism of Eclipta prostrata L.
Supplementary Material
Supporting Data
Acknowledgements
Not applicable.
Funding
The present study was supported by High-End Foreign Experts
Project (grant no. G2022014132L) and Jiangsu Higher Education
Institution Innovative Research Team For Science and Technology
(2023) and Jiangsu Medical College Scientific and Technological
Innovation Team (2024) and Medical Research General Program of
Jiangsu Provincial Health Commission (grant no. M2024063) and
Yancheng Science and Technology Basic Research Program (grants nos.
YCBK202219 and YCBK2024046) and College-local collaborative
innovation research project of Jiangsu Medical college (grant nos.
20239401, 202490403 and 202490408).
Availability of data and materials
The data generated in this study are included in the
figures and/or tables of this article.
Authors' contributions
WY made substantial contributions to the conception
and design of the study, and acquisition of funding, and
participated in drafting the manuscript and revising it critically
for important intellectual content. YC and FQ participated in
research design and revised the manuscript. YC also conducted
experiments, and contributed to the drafting, writing and revision
of the manuscript. WX, XS and JW conducted experiments. XN
performed data analysis and interpretation of data. YC and JW
confirmed the authenticity of all the raw data. All authors read
and approved the final version of the manuscript.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
Glossary
Abbreviations
Abbreviations:
|
AKT1
|
AKT serine/threonine kinase 1
|
|
BP
|
biolo-gical process
|
|
CC
|
cellular component
|
|
DL
|
drug likeness
|
|
EGFR
|
epidermal growth factor receptor
|
|
GC
|
gastric cancer
|
|
MF
|
molecular function
|
|
OB
|
oral bioavailability
|
|
PPI
|
protein--protein interaction
|
|
TCM
|
Traditional Chinese medicine
|
|
TNF
|
tumor necrosis factor
|
|
TP53
|
tumor protein p53
|
|
PI3K-Akt
|
phosphatidylinositol 3-kinase-protein
kinase B
|
|
HIF-1
|
hypoxia--inducible factor-1
|
References
|
1
|
Smyth EC, Nilsson M, Grabsch HI, van
Grieken NC and Lordick F: Gastric cancer. Lancet. 396:635–648.
2020. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Agnarelli A, Vella V, Samuels M,
Papanastasopoulos P and Giamas G: Incorporating immunotherapy in
the management of gastric cancer: Molecular and Clinical
Implications. Cancers (Basel). 14:43782022. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
He Y, Zheng J, Ye B, Dai Y and Nie K:
Chemotherapy-induced gastrointestinal toxicity: Pathogenesis and
current management. Biochem Pharmacol. 216:1157872023. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Joshi SS and Badgwell BD: Current
treatment and recent progress in gastric cancer. CA Cancer J Clin.
71:264–279. 2021.PubMed/NCBI
|
|
5
|
Morgos DT, Stefani C, Miricescu D, Greabu
M, Stanciu S, Nica S, Stanescu-Spinu II, Balan DG,
Balcangiu-Stroescu AE, Coculescu EC, et al: Targeting PI3K/AKT/mTOR
and MAPK signaling pathways in gastric cancer. Int J Mol Sci.
25:18482024. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Gu Y, Sun M, Fang H, Shao F, Lin C, Liu H,
Li H, He H, Li R, Wang J, et al: Impact of clonal TP53 mutations
with loss of heterozygosity on adjuvant chemotherapy and
immunotherapy in gastric cancer. Br J Cancer. 131:1320–1327. 2024.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Ding J, Gao W, Yang H, Duan L, Sun D, Liu
L, Qu X, Yu H, Xu B, Zhao S, et al: KBTBD2 promotes proliferation
and migration of gastric cancer via activating the EGFR signaling
pathway. Pathol Res Pract. 254:1550952024. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Riquelme I, Pérez-Moreno P, Mora-Lagos B,
Ili C, Brebi P and Roa JC: Long non-coding RNAs (lncRNAs) as
regulators of the PI3K/AKT/mTOR pathway in gastric carcinoma. Int J
Mol Sci. 24:62942023. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Lu C, Ke L, Li J, Wu S, Feng L, Wang Y,
Mentis AFA, Xu P, Zhao X and Yang K: Chinese medicine as an
adjunctive treatment for gastric cancer: Methodological
investigation of meta-analyses and evidence Map. Front Pharmacol.
12:7977532022. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Xie Z, Jiang N, Lin M, He X, Li B, Dong Y,
Chen S and Lv G: The mechanisms of polysaccharides from tonic
chinese herbal medicine on the enhancement of immune function: A
review. Molecules. 28:73552023. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Zhang X, Zhang C, Ren Z, Zhang F, Xu J,
Zhang X and Zheng H: Curcumin affects gastric cancer cell
migration, invasion and cytoskeletal remodeling through
Gli1-β-catenin. Cancer Manag Res. 12:3795–3806. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Li X, Zhao L, Wang J, Ma T, Zhou J, Bian Y
and Guo J: The Mechanism of Sijunzi decoction suppresses gastric
cancer metastasis via the m6A methyltransferase METTL14 based on
untargeted metabolomics studies and network pharmacology analysis.
Drug Des Devel Ther. 19:2369–2392. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Timalsina D and Devkota HP: Eclipta
prostrata (L.) L. (Asteraceae): Ethnomedicinal uses, chemical
constituents, and biological activities. Biomolecules. 11:17382021.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Qin T, Zhao J, Liu X, Li L, Zhang X, Shi
X, Ke Y, Liu W, Huo J, Dong Y, et al: luteolin combined with
low-dose paclitaxel synergistically inhibits epithelial-mesenchymal
transition and induces cell apoptosis on esophageal carcinoma in
vitro and in vivo. Phytother Res. 35:6228–6240. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Wang R, Li X, Xu Y, Li Y, Zhang W, Guo R
and Song J: Progresspharmacokinetics and future perspectives of
luteolin modulating signaling pathways to exert anticancer effects:
A review. Medicine (Baltimore). 103:e393982024. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Zou YX, Mu ZQ, Wang J, Tian S, Li Y and
Liu Y: Wedelolactone, a Component from Eclipta prostrata (L.) L.,
inhibits the proliferation and migration of head and neck squamous
cancer cells through the AhR pathway. Curr Pharm Biotechnol.
23:1883–1892. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Yang J, Kim JS, Kwon YS, Seong ES and Kim
MJ: Antioxidant and antiproliferative activities of eclipta
prostrata (L.) L. Extract and isolated compounds. Molecules.
28:73542023. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Fang Z, Xue Y, Leng Y, Zhang L, Ren X,
Yang N, Chen J, Chen L and Wang H: Erzhi pills reverse
PD-L1-mediated immunosuppression in melanoma microenvironment.
Heliyon. 10:e249882024. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Qi Y, Li P, Li B, Lv P, Hu H, Tan C, Wang
W, Jiang Y and He J: Integrating of serum pharmacochemistry,
metabolomics, and experimental validation to explore the mechanism
of Erzhi Pill against Triple-negative breast cancer. J
Ethnopharmacol. 353((Pt A)): 1202872025. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Lin XX, Yang PQ, Li XJ, Xu ZZ, Wu HT, Hu
SM, Yang XL, Ding Y and Yu WZ: Network pharmacology-based analysis
and in vitro experimental verification of the inhibitory role of
luteoloside on gastric cancer cells via the p53/p21 pathway. Oncol
Lett. 29:762024. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Li H, Wang X, Lu A, Pan Y, Zhang W and Hao
L: Luteoloside inhibiting the prostate cancer cells growth and
promoting the tumor cells autophagy through the AKT/mTOR pathway. J
Men's Health. 19:49–55. 2023.
|
|
22
|
Dukel M and Zarzour F: luteolin enhances
anticancer effects of PX-478 during hypoxic response in metastatic
breast cancer cells. Anticancer Agents Med Chem. 25:1–10. 2025.
|
|
23
|
Ye S, Pan X, Zou L, Ni S, Zhang L, Hong Y
and Hu K: HepG2 exosomes coated luteolin nanoparticles remodeling
hepatic stellate cells and combination with sorafenib for the
treatment of hepatocellular carcinoma. Cancer Nano. 15:152024.
View Article : Google Scholar
|
|
24
|
Ganai SA, Sheikh FA, Baba ZA, Mir MA,
Mantoo MA and Yatoo MA: Anticancer activity of the plant flavonoid
luteolin against preclinical models of various cancers and insights
on different signalling mechanisms modulated. Phytother Res.
35:3509–3532. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Çetinkaya M and Baran Y: Therapeutic
potential of luteolin on cancer. Vaccines (Basel). 11:5542023.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Zhao Y, Xing C, Deng Y, Ye C and Peng H:
HIF-1α signaling: Essential roles in tumorigenesis and implications
in targeted therapies. Genes Dis. 11:234–251. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Cheng D, Lian W, Jia X, Wang T, Sun W, Liu
Y and Ni C: LGALS3 regulates endothelial-to-mesenchymal transition
via PI3K/AKT signaling pathway in silica-induced pulmonary
fibrosis. Int J Mol Sci. 509:1539622024.
|
|
28
|
Degan SE and Gelman IH: Emerging roles for
akt isoform preference in cancer progression pathways. Mol Cancer
Res. 19:1251–1257. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Zhu Y, Yang J, Li Y, Xu J and Fang Z:
Demethylase FTO enhances the PI3K/Akt signaling to promote gastric
cancer malignancy. Med Oncol. 40:1302023. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Miricescu D, Totan A, Stanescu-Spinu II,
Badoiu SC, Stefani C and Greabu M: PI3K/AKT/mTOR signaling pathway
in breast cancer: From molecular landscape to clinical aspects. Int
J Mol Sci. 22:1732020. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Shi S, Ou X, Liu C, Wen H and Ke J:
Research progress of HIF-1a on immunotherapy outcomes in immune
vascular microenvironment. Front Immunol. 16:15492762025.
View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Rashid M, Zadeh LR, Baradaran B, Molavi O,
Ghesmati Z, Sabzichi M and Ramezani F: Up-down regulation of HIF-1α
in cancer progression. Gene. 798:1457962021. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Jin X, Dai L, Ma Y, Wang J and Liu Z:
Implications of HIF-1α in the tumorigenesis and progression of
pancreatic cancer. Cancer Cell Int. 20:2732020. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Wang YF, Dong ZK and Jin WL: Hijacking
homeostasis: The brain-body neural circuitry in tumor pathogenesis
and emerging therapeutic frontiers. Mol Cancer. 24:2062025.
View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Francis N and Borniger JC: Cancer as a
homeostatic challenge: The role of the hypothalamus. Trends
Neurosci. 44:903–914. 2021. View Article : Google Scholar : PubMed/NCBI
|