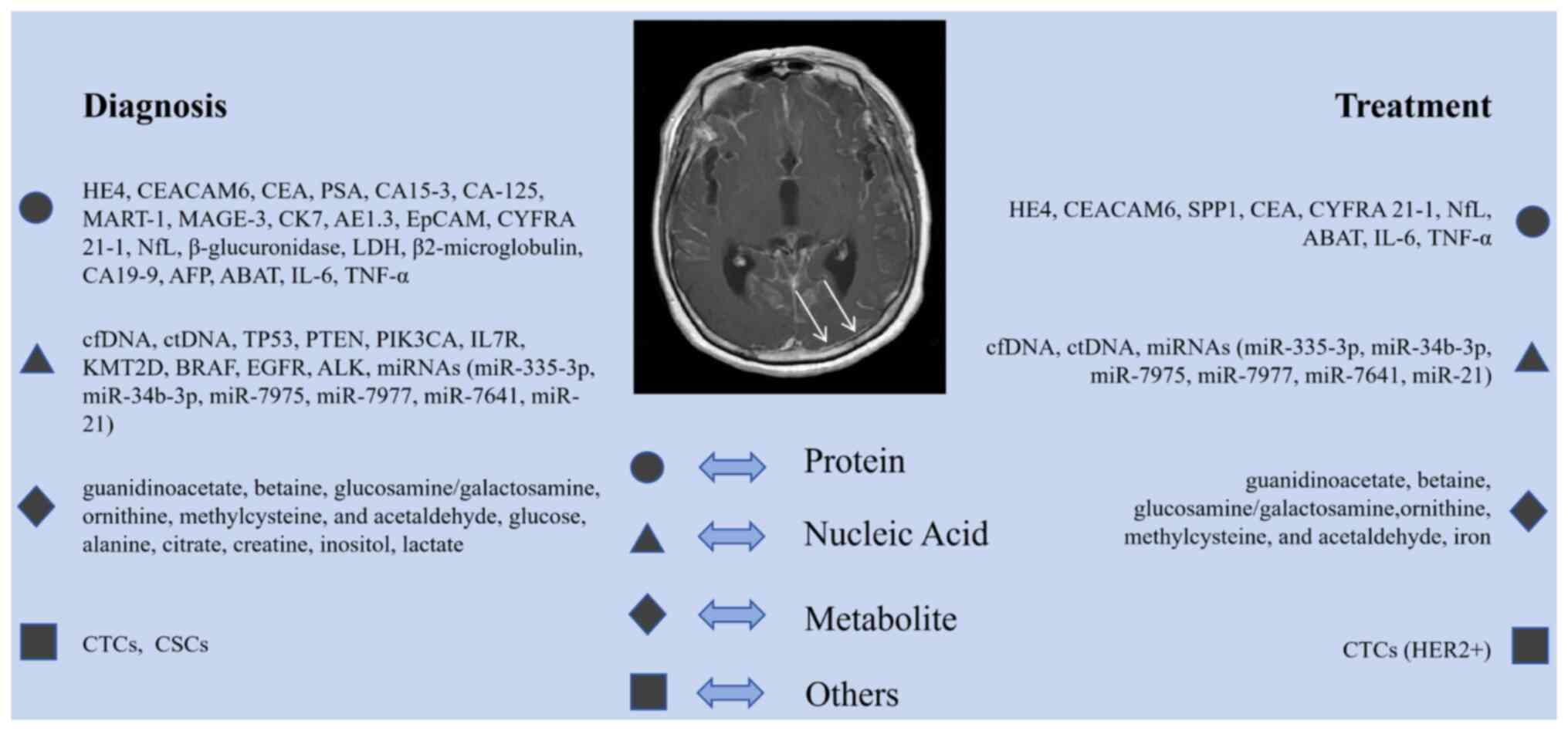
Leptomeningeal metastasis (LM) is a severe
complication characterized by the dissemination of malignant cells
to the leptomeninges and cerebrospinal fluid (CSF) (1). LM occurs in ~5% of patients with solid
tumors, most commonly lung cancer, breast cancer and melanoma
(1–3). Notably, biological behaviors and CSF
biomarker profiles of LM may vary substantially depending on the
primary tumor origin (4). The
mechanisms of LM metastasis involve diverse pathways, including
direct extension from the primary tumor, hematogenous
dissemination, perineural and perivascular spread and iatrogenic
introduction (5,6). Prognosis remains poor, with a median
overall survival time of 3.6–12.0 months despite aggressive
multimodal therapy (7). In
untreated patients, the median overall survival time is limited to
4–6 weeks (8), underscoring the
urgent need for effective diagnosis and management. Clinically, LM
presents with non-specific neurological symptoms, such as headache,
cognitive impairment, cranial nerve palsies and radiculopathy,
which frequently lead to a delayed diagnosis.
CSF circulates within the central nervous system
(CNS), providing mechanical protection and maintaining biochemical
homeostasis. Secreted by the choroid plexus, CSF contains water,
electrolytes, glucose and proteins; it facilitates nutrient
transport and metabolic waste elimination, processes essential for
normal CNS physiology. Additionally, CSF accumulates bioactive
substances (such as proteins, metabolites and RNA) secreted by CNS
cells, as well as small molecules diffused from systemic
circulation (9). Given the
limitations of tissue biopsy in CNS disorders, CSF profiling
represents a promising strategy for biomarker discovery (10). Consequently, identifying effective
CSF biomarkers is critical for the diagnosis and therapeutic
monitoring of LM.
Despite advancements in LM diagnosis using CSF, the
existing methods lack sufficient sensitivity and specificity. CSF
cytology remains the gold standard but has a limited sensitivity of
45–67% (11). Although circulating
tumor cells (CTCs) and circulating tumor DNA (ctDNA) offer higher
sensitivity, they are limited by potential false positives and
false negatives (12,13). Standard magnetic resonance imaging
often fails to detect early or small metastatic foci (14) and tissue biopsy is too invasive for
routine monitoring. Considering these challenges, developing novel
CSF-based detection techniques is of paramount importance. As CSF
directly contacts the leptomeninges, it captures biomolecules
released by tumor cells. These biomarkers offer critical insights
for early diagnosis, precision treatment and longitudinal
monitoring of LM, potentially improving patient outcomes.
Diagnostic performance and clinical relevance of CSF
biomarkers vary depending on the primary tumor type (15). CSF biomarker detection is critical
for the clinical diagnosis and management of LM. As shown in
Fig. 1, these biomarkers are
categorized into proteins, nucleic acids, metabolites and others.
Each category utilizes distinct detection methodologies to support
early diagnosis, disease monitoring and treatment evaluation.
Numerous biomarkers have been identified through integrative
multi-omics and single-cell analyses of CSF, which enable
comprehensive profiling of tumor and microenvironmental components.
Protein biomarkers, reflecting tumor biological activity, are
typically detected via immunoassays such as ELISA and
immunohistochemistry (16,17). Nucleic acid biomarkers, particularly
ctDNA and microRNAs (miRNAs/miRs), reveal molecular characteristics
and are quantified using PCR and high-throughput sequencing
(18,19). Metabolic biomarkers indicate tumor
metabolic reprogramming and are identified through gas
chromatography-mass spectrometry and liquid chromatography-mass
spectrometry (20). Additionally,
emerging biomarkers such as exosomes and cytokines are analyzed
using flow cytometry (21).
Integrating these diverse methodologies facilitates a comprehensive
strategy for early detection and personalized treatment of LM. The
major CSF biomarkers and their clinical relevance are summarized in
Table I. At present, ctDNA and
cell-free DNA (cfDNA) are the most widely applied and clinically
validated CSF biomarkers, whereas most other markers listed in
Table I remain investigational.
Altered expression of specific protein biomarkers in
CSF offers potential for the early detection and monitoring of LM.
For example, carcinoembryonic antigen-related cell adhesion
molecule 6 (CEACAM6) is a critical biomarker for diagnosing lung
adenocarcinoma (LUAD)-related LM. Elevated CSF CEACAM6 levels
correlate closely with LM incidence (22). Moreover, combined detection of human
epididymis protein 4 (HE4) and CEACAM6 enhances diagnostic
sensitivity and specificity for this subgroup. HE4, a secretory
protein associated with LM progression in LUAD, serves as a
promising adjunctive biomarker (16,23).
Additionally, elevated levels of CEA, prostate-specific antigen,
cancer antigen (CA)-153, CA-125, melanoma antigen recognized by
T-cells 1 and melanoma-associated antigen 3 are frequently used to
diagnose LM (16,24,25).
Although CSF expression of these markers is increased, definitive
diagnosis still requires corroboration via clinical and imaging
assessments.
Secreted phosphoprotein 1 (SPP1) is another notable
protein in LM pathology. Studies show that markedly elevated CSF
SPP1 levels augment the migratory capacity of tissue-resident
boundary-associated macrophages, thereby facilitating meningeal
metastasis (26,27). Notably, SPP1 is associated with
matrix metalloproteinase 14 in CSF, suggesting its potential as a
therapeutic target. Furthermore, a panel of proteins, including
α1-antichymotrypsin, apolipoprotein A-I, apolipoprotein E,
haptoglobin, hemopexin, prostaglandin D2 synthase, transthyretin
and serotransferrin, exhibits altered CSF profiles in patients with
LM (28). These proteins are
primarily involved in the immune response, inflammation and lipid
metabolism. Their detection in CSF aids in identifying LM and
provides insights into disease progression and immune dynamics.
LM diagnosis is further supported by
immunohistochemical staining for cytokeratin 7 and AE1.3. Positive
expression of these markers is linked to specific malignancies,
notably cholangiocarcinoma-related meningeal metastasis (29). These biomarkers facilitate
conclusive diagnosis via immunocytochemical analysis, particularly
when conventional CSF cytology is indeterminate. Additionally,
epithelial cell adhesion molecule (EpCAM), detected via flow
cytometry, exhibits high sensitivity for breast cancer-derived LM
(30). Efficient detection of
EpCAM-positive cells improves early diagnostic capabilities,
especially when imaging and traditional cytology are
non-diagnostic.
CYFRA 21-1, a widely employed tumor marker, shows
diagnostic utility in CSF for LM, particularly in lung cancer cases
(22,31). Combined analysis of CSF CYFRA 21-1
and cytology markedly improves diagnostic accuracy, facilitating
earlier identification and therapeutic assessment (32). Neurofilament light chain (NfL) is
also elevated in patients with LM and correlates with disease onset
and progression (33). As a marker
of neuronal injury, NfL offers prognostic value, particularly for
monitoring treatment response and drug resistance.
Cytokines, especially tumor necrosis factor-α
(TNF-α) and interleukin-6 (IL-6), are central to LM initiation and
progression. Elevated CSF concentrations of TNF-α and IL-6 reflect
inflammatory responses and immune dysregulation within the tumor
microenvironment (34). These
cytokines stimulate tumor cell proliferation and metastasis and may
participate in immune evasion. Monitoring CSF cytokine levels
facilitates early diagnosis and provides information on immune
status, making them useful biomarkers for assessing therapeutic
response.
Alterations in non-specific biomarkers such as
β-glucuronidase, lactate dehydrogenase (LDH) and β2-microglobulin
can also observed in CSF. While their elevation is not specific to
LM, they retain value for adjunctive diagnosis and monitoring
(35,36). Using multiplex immunoassay
technology facilitates concurrent detection of these markers,
augmenting diagnostic accuracy (37). Profiling CSF proteins via this
approach yields broader biological insights and enhances the
understanding of LM mechanisms.
Several candidate CSF biomarkers have been proposed
for LM, including protein tyrosine phosphatase receptor C, serpin
peptidase inhibitor clade C member 1, soluble CD44, soluble CD14,
aminopeptidase, SPP1, Fc γ receptor 1A, complement component C9,
soluble CD34 and soluble CD19. These biomarkers can be detected via
multiplex immunoassay, furnishing information relevant to diagnosis
and prognosis (36). Additionally,
GABA transaminase (ABAT) has been identified as a key player in the
CSF microenvironment. Elevated ABAT expression contributes to
neuronal survival and promotes LM (38). ABAT upregulation may be linked to
metabolic and epigenetic traits of tumor cells, presenting novel
targets for intervention. Identifying these novel biomarkers
expands the diagnostic toolkit for CSF analysis.
In summary, CSF protein biomarkers offer crucial
biological insights for the management of LM. Concurrent detection
of multiple biomarkers can improve diagnostic accuracy and
sensitivity. While technological advancements will likely uncover
additional biomarkers, it should be noted that most current protein
biomarkers are supported by limited studies and require further
validation in larger, prospective cohorts before routine clinical
application.
Liquid biopsy technologies focusing on nucleic acid
detection have created new avenues for the early diagnosis and
therapeutic monitoring of LM. Analysis of cfDNA, particularly its
ctDNA fraction, has become a critical tool for tumor surveillance
and efficacy evaluation (39,40).
Compared with conventional cytology and neuroimaging, CSF-derived
ctDNA offers superior sensitivity and specificity, particularly in
early diagnosis, detection of resistance mutations and monitoring
treatment response. Studies consistently demonstrate that CSF ctDNA
outperforms peripheral blood ctDNA in identifying driver mutations
in leptomeningeal metastases derived from non-small cell lung
cancer (NSCLC), specifically for EGFR and ALK mutations (41,42).
In patients with NSCLC-LM, the higher detection rate of driver
mutations in CSF ctDNA compared with plasma provides critical
molecular data for guiding precision therapy (43).
Exosomal miRNAs in CSF also represent a vital class
of biomarkers with diagnostic promise. Specific miRNAs, such as
miR-21, miR-483-5p and miR-342-5p, participate in the metastatic
cascade by regulating proliferation, migration and drug resistance.
Notably, miR-483-5p and miR-342-5p display differential expression
patterns between the serum and CSF of patients with NSCLC with and
without LM, suggesting their utility as targets for early diagnosis
and monitoring (44). Additionally,
CSF miR-21 levels are correlated with LM prognosis, particularly
regarding drug resistance and therapeutic response (45). Liquid biopsy emphasizing miRNA
analysis offers a sensitive, reproducible method for detecting
tumor microenvironmental alterations to inform clinical
decision-making.
Progress in molecular diagnostics has led to the
identification of novel small non-coding RNAs, including
PIWI-interacting RNA, Y RNA and small nucleolar RNA, as potential
LM biomarkers (46). Using
next-generation sequencing (NGS), researchers have delineated
altered miRNA profiles in the CSF of patients with LM, including
miR-335-3p and miR-34b-3p (9).
These miRNAs, likely originating from CTCs, are linked to LM
initiation and offer potential for early diagnosis and targeted
therapy. Furthermore, upregulation of specific miRNAs, such as
miR-7975, miR-7977 and miR-7641, shows diagnostic utility for
monitoring LM (47). Longitudinal
monitoring of these miRNA alterations allows clinicians to assess
treatment response more precisely.
Beyond miRNAs and ctDNA, chromosomal aneuploidy in
CSF is increasingly recognized as a biomarker (48,49).
Aneuploidy, defined as an abnormal chromosome number, is associated
with tumor malignancy, metastatic potential and prognosis (50). Angus et al (48) reported a strong correlation between
CSF aneuploidy and both LM risk and overall survival in patients
with NSCLC-LM. Detecting aneuploidy supports early diagnosis and
aids in predicting treatment outcomes. NGS technology facilitates
efficient aneuploidy detection, elucidating tumor genetic
signatures to guide clinical management.
In conclusion, various nucleic acid biomarkers in
CSF, including cfDNA, ctDNA, exosomal miRNAs and small non-coding
RNAs, have shown promise for diagnosing, assessing prognosis and
monitoring treatment responses in LM. Among these, ctDNA and cfDNA
currently offer the most robust clinical utility, particularly for
molecular profiling and resistance mutation detection. Other
nucleic acid biomarkers remain investigational and require
validation in large-scale studies before routine
implementation.
CSF metabolites are emerging as potential biomarkers
for the diagnosis and monitoring of LM. Previous metabolomics
studies have identified distinct metabolic profiles in the CSF of
patients with LM. These metabolites reflect tumor-specific
metabolic alterations and offer insights for early diagnosis and
therapeutic strategy formulation. For example, metabolomic analysis
of CSF from patients with breast cancer and CNS involvement
revealed the upregulation of 20 metabolites in LM cases, including
guanidinoacetate, betaine, glucosamine/galactosamine, ornithine,
methylcysteine and acetaldehyde (51). These distinct metabolic deviations
from normal cellular activity highlight metabolic reprogramming as
a key characteristic of tumor survival and proliferation, providing
a rationale for novel therapeutic interventions.
Reduced CSF glucose levels are consistently observed
in patients with LM, a phenomenon common across diverse CNS
malignancies (52–54). Notably, CSF glucose remains
subnormal even after intravenous glucose administration (5). This persistent hypoglycemia suggests a
hypoxic microenvironment, a hypothesis further supported by
elevated CSF LDH levels (35,55).
As an intracellular enzyme released upon membrane disruption, LDH
facilitates anaerobic glycolysis under hypoxic conditions (56). Collectively, these alterations
reflect the metabolic reprogramming inherent to LM and serve as
supportive diagnostic evidence.
Further metabolomic analyses indicate that patients
with LM exhibit significantly elevated CSF levels of lactate,
alanine and citrate, alongside decreased creatine and myo-inositol
(57). These profiles reflect tumor
cell adaptation to the CSF environment. Specifically, elevated
lactate is linked to anaerobic metabolism, while increased alanine
and citrate suggest enhanced glycolysis and tricarboxylic acid
cycle activity. Conversely, diminished creatine and myo-inositol
may indicate altered energy metabolism and cell membrane
perturbations (58,59). These metabolic shifts identify
potential biomarkers and underscore the complexity of tumor
metabolism in LM.
In conclusion, CSF metabolite research provides a
theoretical framework for improving LM management. Quantitative
analysis of CSF metabolites elucidates tumor metabolic signatures
and aids in identifying novel biomarkers to enhance diagnostic
precision. While metabolomics technologies continue to advance,
most current metabolite-based biomarkers remain exploratory and
require clinical validation before routine application.
Beyond proteins, nucleic acids and metabolites, CTCs
and cancer stem cells (CSCs) have emerging roles in LM diagnosis
and monitoring. CTCs are valuable for early detection, prognosis
and treatment response monitoring (63–66).
In HER2-positive breast cancer with LM, CTCs serve as biomarkers to
track therapeutic efficacy (63,65).
Integrating CTC detection with liquid biopsy technologies, such as
exosomal miRNA and ctDNA analysis, provides a comprehensive profile
that enhances diagnostic sensitivity and specificity (21,67).
Crucially, CTCs facilitate the identification of tumor-associated
genetic abnormalities, including mutations in TP53, PTEN, PIK3CA,
IL7R and KMT2D (68–70). These mutations correlate with LM
pathogenesis, providing molecular insights to guide personalized
treatment strategies.
CSCs are also implicated in LM initiation and
progression. Possessing self-renewal and multipotent
differentiation capabilities, CSCs drive tumor recurrence and
metastasis. Evidence suggests the presence of CSCs in the CSF of
patients with LM, presenting potential targets for early diagnosis
and therapeutic intervention (71).
Although detection technologies for CSCs remain developmental,
their utility in LM is increasingly recognized. Identifying CSCs in
CSF could support early diagnosis and inform personalized
immunotherapy and targeted therapies. While methodology refinement
is required, progress in this field supports the development of
innovative diagnostic and therapeutic approaches. However, both CTC
and CSC analyses in CSF currently remain primarily research tools,
with clinical application limited by technical challenges and the
need for large-scale validation.
LM management necessitates a multidisciplinary
approach combining systemic and local therapies. Systemic options
include cytotoxic chemotherapy, targeted therapy and immunotherapy,
selected based on primary tumor efficacy, molecular
characteristics, CNS penetration, treatment history and patient
factors (5). Intrathecal
administration elevates CSF drug concentrations while minimizing
systemic toxicity, although it is limited to agents capable of
crossing the blood-brain barrier, such as methotrexate, cytarabine
and thiotepa (72,73). For specific malignancies such as
breast cancer and NSCLC, targeted agents (such as osimertinib) and
immune checkpoint inhibitors (such as nivolumab) have shown
efficacy (74,75). Radiotherapy, including stereotactic
and focal techniques, is employed for symptom relief and CSF flow
improvement, although survival benefits remain limited (76). Ultimately, LM treatment requires
individualization guided by multidisciplinary evaluation and, where
feasible, inclusion in clinical trials. Given the complexity of
assessing therapeutic response, CSF biomarkers play a critical role
in monitoring efficacy and prognosis. Key biomarkers for this
purpose are summarized in Table
II.
CSF biomarkers are critical for assessing
therapeutic response in LM. ctDNA is widely utilized for monitoring
tumor burden and efficacy (40,77). A
post-treatment decrease in CSF ctDNA levels typically indicates
tumor suppression. For example, in patients with NSCLC harboring
EGFR mutations, reduced EGFR-mutant ctDNA in CSF following targeted
therapy correlates with a positive response (78,79).
Similarly, changes in CSF protein markers, such as CEA, reflect
treatment outcomes: A decline in CEA levels signals reduced tumor
burden (80,81). In breast cancer-associated LM,
decreased CSF CEACAM6 levels also indicate therapeutic efficacy
(22). Collectively, dynamic
monitoring of these biomarkers allows clinicians to gauge treatment
success promptly and adjust strategies as needed.
Advances in liquid biopsy enable the use of cfDNA
methylation profiles and cell-free CSF cytological scoring (CNB)
for efficacy assessment. CSF cfDNA methylation profiles provide
molecular insights into disease progression (82,83),
while CNB scoring offers an objective, efficient metric for
diagnosing and evaluating LM status (84). These minimally invasive techniques
facilitate real-time monitoring of therapeutic response.
Consequently, CSF-based liquid biopsy, including methylation and
CNB analysis, holds significant potential for optimizing
therapeutic regimens (85,86).
Extracellular vesicle (EV)-derived miR-21 in CSF
offers another novel metric for treatment evaluation. Research
suggests that EV-mediated miR-21 modulates methotrexate resistance,
particularly in patients with NSCLC-LM, identifying miR-21 as a
potential target for overcoming resistance (45). Monitoring alterations in CSF EV
miR-21 levels helps inform decisions regarding treatment
continuation or modification. Furthermore, variations in EV
concentration and miR-21 expression correlate with prognosis and
serve as effective biomarkers for monitoring intrathecal
chemotherapy response (87). These
findings expand the repertoire of biomarkers available for precise
patient assessment during LM follow-up.
Monitoring CSF biomarkers is critical for predicting
disease relapse, which remains a risk despite initial clinical
remission. Dynamic fluctuations in CSF ctDNA are key indicators of
progression. Specifically, sustained low or undetectable
post-treatment ctDNA levels suggest remission (88), whereas a resurgence in ctDNA
indicates tumor reactivation and elevated relapse risk (89,90).
For example, in breast cancer-LM, elevated post-treatment CEACAM6
levels strongly correlate with relapse (22). Detecting these biomarker changes
prior to the onset of clinical symptoms provides an early warning
system for disease progression.
Alterations in protein biomarkers such as SPP1 and
HE4 are also linked to progression. Post-treatment elevations in
these proteins frequently signal tumor cell reactivation (27). Specifically, in breast cancer LM,
increased HE4 levels serve as a risk indicator for relapse
(23). Consequently, longitudinal
CSF biomarker monitoring facilitates the early detection of
progression, enabling intervention before symptomatic
deterioration, thereby potentially improving patient outcomes.
CSF biomarker monitoring allows clinicians to adjust
treatment regimens based on therapeutic response and disease
status. Persistently high or rebounding biomarker levels may
necessitate treatment modification. For instance, in patients with
EGFR-mutant NSCLC, failure of CSF EGFR-mutant ctDNA to decline
during targeted therapy may indicate resistance to first-generation
EGFR-tyrosine kinase inhibitors (TKIs). This finding supports
transitioning to second- or third-generation EGFR-TKIs or
incorporating chemotherapy or immunotherapy (91). This biomarker-guided strategy
ensures patients receive optimal therapy.
Additionally, CSF biomarkers aid in assessing
treatment tolerance and adverse effects. Abnormally elevated
inflammatory markers or cytokines, accompanied by significant
clinical toxicity, may suggest an exaggerated immune response or
adverse reaction (92–94). In such scenarios, dose adjustment or
temporary discontinuation may be required to mitigate side effects.
Dynamic monitoring thus facilitates precise treatment
customization.
In summary, CSF biomarkers are indispensable for
assessing efficacy, predicting relapse and guiding therapeutic
adjustments in LM. Integrating comprehensive biomarker monitoring
into clinical practice enables precise management, ultimately
aiming to improve patient survival and quality of life.
Despite notable progress in CSF biomarker research
for LM, critical challenges persist. First, validating biomarkers
with higher sensitivity and specificity is essential to enhancing
diagnostic precision. Second, standardization of detection
methodologies is a requisite to ensure clinical reproducibility.
Third, further research must elucidate the biological mechanisms
linking biomarker expression to LM pathogenesis. Notably, while
ctDNA and cfDNA are clinically validated, most other biomarkers
remain investigational.
Looking forward, advancements in multi-omics and
single-cell sequencing will deepen the understanding of tumor
heterogeneity and the CSF microenvironment. These technologies
facilitate novel biomarker discovery and clarify tumor-immune
interactions, enabling precision diagnosis and treatment
stratification. Ultimately, enhanced clinical collaboration and
data sharing are imperative for translating these findings into
practice and improving patient outcomes.
Not applicable.
This study was supported by the CAMS Innovation Fund for Medical
Sciences (grant no. 2022-I2M-C&T-B-063), National Natural
Science Foundation of China (grant no. 82472722) and Beijing Hope
Run Special Fund of Cancer Foundation of China (grant no.
LC2022B18).
Not applicable.
HC, JW and TX conceived and designed the review and
revised the manuscript. HL, WF and FM drafted the manuscript. CF
critically revised and edited the manuscript. All authors read and
approved the final version of the manuscript. Data authentication
is not applicable.
Not applicable.
Not applicable.
The authors declare that they have no competing
interests.
|
1
|
Rinehardt H, Kassem M, Morgan E, Palettas
M, Stephens JA, Suresh A, Ganju A, Lustberg M, Wesolowski R,
Sardesai S, et al: Assessment of leptomeningeal carcinomatosis
diagnosis, management and outcomes in patients with solid tumors
over a decade of experience. Eur J Breast Health. 17:371–377. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Sharma A, Low JT and Kumthekar P: Advances
in the diagnosis and treatment of leptomeningeal disease. Curr
Neurol Neurosci Rep. 22:413–425. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Brastianos PK, Strickland MR, Lee EQ, Wang
N, Cohen JV, Chukwueke U, Forst DA, Eichler A, Overmoyer B, Lin NU,
et al: Phase II study of ipilimumab and nivolumab in leptomeningeal
carcinomatosis. Nat Commun. 12:59542021. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Nguyen A, Nguyen A, Dada OT, Desai PD,
Ricci JC, Godbole NB, Pierre K and Lucke-Wold B: Leptomeningeal
metastasis: A review of the pathophysiology, diagnostic
methodology, and therapeutic landscape. Curr Oncol. 30:5906–5931.
2023. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Le Rhun E, Weller M, van den Bent M,
Brandsma D, Furtner J, Rudà R, Schadendorf D, Seoane J, Tonn JC,
Wesseling P, et al: Leptomeningeal metastasis from solid tumours:
EANO-ESMO clinical practice guideline for diagnosis, treatment and
follow-up. ESMO Open. 8:1016242023. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Remsik J and Boire A: The path to
leptomeningeal metastasis. Nat Rev Cancer. 24:448–460. 2024.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Lamba N, Wen PY and Aizer AA: Epidemiology
of brain metastases and leptomeningeal disease. Neuro Oncol.
23:1447–1456. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Li Y and Li X: Advances in clinical
application of cerebrospinal fluid circulating tumor DNA in
leptomeningeal metastasis of non-small cell lung cancer. Zhongguo
Fei Ai Za Zhi. 27:376–382. 2024.(In Chinese). PubMed/NCBI
|
|
9
|
Im JH, Kim TH, Lee KY, Gwak HS, Lin W,
Park JB, Kim JH, Yoo BC, Park SM, Kwon JW, et al: Exploratory
profiling of extracellular MicroRNAs in cerebrospinal fluid
comparing leptomeningeal metastasis with other central nervous
system tumor statuses. J Clin Med. 10:48602021. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Lin Y, Yu T, Li H, Yin Z and Guo A:
Diagnostic value of locally produced tumor markers and blood brain
barrier integrity in lung cancer patients with leptomeningeal
metastasis. Zhongguo Fei Ai Za Zhi. 24:567–576. 2021.(In Chinese).
PubMed/NCBI
|
|
11
|
Fitzpatrick A, Iravani M, Mills A, Childs
L, Alaguthurai T, Clifford A, Garcia-Murillas I, Van Laere S, Dirix
L, Harries M, et al: Assessing CSF ctDNA to improve diagnostic
accuracy and therapeutic monitoring in breast cancer leptomeningeal
metastasis. Clin Cancer Res. 28:1180–1191. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Glitza IC, Smalley KSM, Brastianos PK,
Davies MA, McCutcheon I, Liu JKC, Ahmed KA, Arrington JA, Evernden
BR, Smalley I, et al: Leptomeningeal disease in melanoma patients:
An update to treatment, challenges, and future directions. Pigment
Cell Melanoma Res. 33:527–541. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Stetson D, Ahmed A, Xu X, Nuttall BRB,
Lubinski TJ, Johnson JH, Barrett JC and Dougherty BA: Orthogonal
comparison of four plasma NGS tests with tumor suggests technical
factors are a major source of assay discordance. JCO Precis Oncol.
3:1–9. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Mohammadi M, Mohammadi S, Hadizadeh H,
Olfati M, Moradi F, Tanzifi G and Ghaderi S: Brain metastases from
breast cancer using magnetic resonance imaging: A systematic
review. J Med Radiat Sci. 71:133–141. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Zhu JW, Shum M, Qazi MA, Sahgal A, Das S,
Dankner M, Menjak I, Lim-Fat MJ and Jerzak KJ: Cerebral spinal
fluid analyses and therapeutic implications for leptomeningeal
metastatic disease. J Neurooncol. 172:31–40. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Li X, Chen K, Li J, Tang X, Ruan H and
Guan M: Diagnostic value of cerebrospinal fluid human epididymis
protein 4 for leptomeningeal metastasis in lung adenocarcinoma.
Front Immunol. 15:13399142024. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Kharel Z, Stanford S, Hemminger LE,
Schmidt T, Hardy SJ, Zittel J, Mohile NA and Dhaka A: Efficacy of
trastuzumab deruxtecan in treating HER2-low breast cancer
leptomeningeal metastasis: A case report. Per Med. 21:335–339.
2024. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Lyman KA, Madill E, Thatikunta P,
Threlkeld ZD, Banaei N and Gold CA: An electronic health record
intervention to limit viral testing of cerebrospinal fluid.
Neurohospitalist. 13:173–177. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Miao Q, Zheng X, Li L, Zheng X, Zhang L,
Jiang K, Wu S, Wang H, Wu B, Xu Y, et al: Cerebrospinal fluid
circulating tumor DNA contributes to the detection and
characterization of leptomeningeal metastasis in non-small cell
lung cancer. J Neurooncol. 165:517–525. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Im JH, Yoo BC, Lee JH, Lee KY, Kim KH, Kim
JH, Park HJ, Park M, Lee SH, Kwon JW, et al: Experimental
assessment of leptomeningeal metastasis diagnosis in
medulloblastoma using cerebrospinal fluid metabolomic profiles.
Metabolites. 11:8512021. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
van Bussel MTJ, Pluim D, Kerklaan BM, Bol
M, Sikorska K, Linders DTC, van den Broek D, Beijnen JH, Schellens
JHM, Brandsma D, et al: Circulating epithelial tumor cell analysis
in CSF in patients with leptomeningeal metastases. Neurology.
94:e521–e528. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Wang X, Tang X, Gu J, Sun Z, Yang S, Mu Y,
Guan M, Chen K, Liu W, Ruan H and Xu J: CEACAM6 serves as a
biomarker for leptomeningeal metastasis in lung adenocarcinoma.
Cancer Med. 12:4521–4529. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Ruan H, Zhou Y, Shen J, Zhai Y, Xu Y, Pi
L, Huang R, Chen K, Li X, Ma W, et al: Circulating tumor cell
characterization of lung cancer brain metastases in the
cerebrospinal fluid through single-cell transcriptome analysis.
Clin Transl Med. 10:e2462020. View
Article : Google Scholar : PubMed/NCBI
|
|
24
|
Durrani U, Rifai K, Arshad M, Paracha A,
Deol E, Paracha M, Waheed F, Siddiqui Z, Abid A, Mustafic E, et al:
Leptomeningeal metastases in prostate cancer: A review of the
current literature. Curr Urol. 18:159–166. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Benito-Martín A, Jasiulionis MG and
García-Silva S: Extracellular vesicles, melanoma: New perspectives
on tumor microenvironment and metastasis. Front Cell Dev Biol.
10:10619822022. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Kilian M and Quintana FJ:
Immunosuppressive dura-derived macrophages in leptomeningeal
metastasis. Nat Cancer. 5:1791–1792. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Zhao J, Zeng R, Li X, Lu Y, Wang Z, Peng
H, Chen H, Fu M, Zhang Y, Huang Y, et al: Dura immunity configures
leptomeningeal metastasis immunosuppression for cerebrospinal fluid
barrier invasion. Nat Cancer. 5:1940–1961. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Galicia N, Díez P, Dégano RM, Guest PC,
Ibarrola N and Fuentes M: Proteomic biomarker identification in
cerebrospinal fluid for leptomeningeal metastases with neurological
complications. Adv Exp Med Biol. 974:85–96. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Novegno F, Umana G, Granaroli P, Borri F,
Orlandi A and Lunardi P: Current management of central nervous
system metastasis from cholangiocarcinoma: The neurosurgical
perspective. Literature review. Br J Neurosurg. 34:575–583. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Wang Y, Yang X, Li NJ and Xue JX:
Leptomeningeal metastases in non-small cell lung cancer: Diagnosis
and treatment. Lung Cancer. 174:1–13. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Hyun JW, Shin HS, Kim SH, Kong SY, Yoo H,
Gwak HS and Kim HJ: CYFRA 21-1 levels in cerebrospinal fluid as a
putative therapeutic monitoring biomarker for patients with
leptomeningeal carcinomatosis: A pilot study. Cancer Biomark.
28:81–89. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Hyun JW, Park JH, Kang BG, Park EY, Park
B, Joo J, Kim JK, Kim SH, Jeong JH, Lee HW, et al: Diagnostic and
prognostic values of cerebrospinal fluid CYFRA 21-1 in patients
with leptomeningeal carcinomatosis. Oncotarget. 8:53326–53335.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Hyun JW, Kim Y, Kim KH, Kim SH, Park EY,
Youn JH, Yoo H, Gwak HS and Kim HJ: Cerebrospinal fluid
neurofilament light chain as a potential prognostic biomarker for
leptomeningeal metastasis. Oncol Lett. 24:4282022. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Walker JG, Armstrong TS, O'Brien BJ,
Gilbert MR, Casarez RL, Fagundes C, Heijnen CJ, Andersen CR, Yuan
Y, Wu J and LoBiondo-Wood G: Associations of meaning of illness
with psychosocial, clinical, and immunological characteristics in
patients with Leptomeningeal metastasis. Compreh
Psychoneuroendocrinol. 8:1000992021. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Hasanov M, Milton DR, Davies AB, Sirmans
E, Saberian C, Posada EL, Opusunju S, Gershenwald JE, Torres-Cabala
CA, Burton EM, et al: Changes in outcomes and factors associated
with survival in melanoma patients with brain metastases. Neuro
Oncol. 25:1310–1320. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Twijnstra A, van Zanten AP, Nooyen WJ and
de Visser BW: Sensitivity and specificity of single and combined
tumour markers in the diagnosis of leptomeningeal metastasis from
breast cancer. J Neuro Neurosurg Psychiatry. 49:1246–1250. 1986.
View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Brandsma D, Voest EE, de Jager W, Bonfrer
H, Algra A, Boogerd W, Korse T, Reijneveld JC, Verbeek MM, Rijkers
G and Taphoorn MJ: CSF protein profiling using Multiplex
Immuno-assay: A potential new diagnostic tool for leptomeningeal
metastases. J Neurol. 253:1177–1184. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Martirosian V, Deshpande K, Zhou H, Shen
K, Smith K, Northcott P, Lin M, Stepanosyan V, Das D, Remsik J, et
al: Medulloblastoma uses GABA transaminase to survive in the
cerebrospinal fluid microenvironment and promote leptomeningeal
dissemination. Cell Rep. 35:1093022021. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Liang SK, Liao WY, Shih JY, Hsu CL, Yang
CY, Wu SG, Lin YT, Wen YF, Chen LC, Chen YF, et al: Clinical
utility and predictive value of cerebrospinal fluid cell-free DNA
profiling in non-small cell lung cancer patients with
leptomeningeal metastasis. Neoplasia. 60:1011132024. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Pentsova E: Applications of cerebrospinal
fluid circulating tumor cells and circulating tumor-derived DNA in
diagnosis, prognosis, and personalized treatment of CNS metastases.
Front Oncol. 14:14093832024. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Li H, Xie Y, Lin Y, Yu T and Yin Z:
Different gene mutation spectrum of the paired csf and plasma
samples in lung adenocarcinoma with leptomeningeal metastases: The
liquid biopsy based on circulating tumor DNA. Zhongguo Fei Ai Za
Zhi. 23:646–654. 2020.PubMed/NCBI
|
|
42
|
Nie N, Zhou H, Zhang K, Liu L, Luo N, Wang
R, Li X, Zhu M, Hu C, Wang Y, et al: Genotyping of cerebrospinal
fluid in lung cancer patients with leptomeningeal metastasis.
Thorac Cancer. 13:2574–2583. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Yang H, Wen L, Zhao C, Chen J, Zhou Z and
Zhou C, Cai L and Zhou C: Cerebrospinal fluid-derived circulating
tumor DNA is more comprehensive than plasma in NSCLC patients with
leptomeningeal metastases regardless of extracranial evolution.
Heliyon. 8:e123742022. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Xu Q, Ye L, Huang L, Zhou L, Chen X, Ye M,
Wu G, Zhan P, Lv T and Song Y: Serum Exosomal miRNA might be a
novel liquid biopsy to identify leptomeningeal metastasis in
non-small cell lung cancer. Onco Targets Ther. 14:2327–2335. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Im JH, Lee KY, Seo Y, Rhim J, Dho YS, Yoo
BC, Park JB, Shin SH, Yoo H, Kim JH and Gwak HS: Extracellular
vesicles from cerebrospinal fluid of leptomeningeal metastasis
patients deliver MiR-21 and induce methotrexate resistance in lung
cancer cells. Int J Mol Sci. 25:3124–2024. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Lee KY, Seo Y, Im JH, Rhim J, Baek W, Kim
S, Kwon JW, Yoo BC, Shin SH, Yoo H, et al: Molecular signature of
extracellular vesicular small non-coding RNAs derived from
cerebrospinal fluid of leptomeningeal metastasis patients:
Functional implication of miR-21 and other Small RNAs in cancer
malignancy. Cancers (Basel). 13:2092021. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Jin J, Cui Y, Niu H, Lin Y, Wu X, Qi X,
Bai K, Zhang Y, Wang Y and Bu H: NSCLC extracellular vesicles
containing miR-374a-5p promote leptomeningeal metastasis by
influencing blood-brain barrier permeability. Mol Cancer Res.
22:699–710. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Angus L, Deger T, Jager A, Martens JWM, de
Weerd V, van Heuvel I, van den Bent MJ, Smitt PAE, Kros JM, Bindels
EMJ, et al: Detection of aneuploidy in cerebrospinal fluid from
patients with breast cancer can improve diagnosis of leptomeningeal
metastases. Clin Cancer Res. 27:2798–2806. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Zhang L, Fang K, Ren H, Fan S, Wang J and
Guan H: Comparison of the diagnostic significance of cerebrospinal
fluid metagenomic next-generation sequencing copy number variation
analysis and cytology in leptomeningeal malignancy. BMC Neurol.
24:2232024. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Lukow DA and Sheltzer JM: Chromosomal
instability and aneuploidy as causes of cancer drug resistance.
Trends Cancer. 8:43–53. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Cohen-Nowak AJ, Hill VB and Kumthekar P:
Diagnostics and screening in breast cancer with brain and
leptomeningeal metastasis: A review of the literature. Cancers
(Basel). 16:36862024. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Pattanaik J, Goel V, Sehrawat P, Rathore
R, Singh RK, Garg A and Biswas A: Leptomeningeal carcinomatosis in
a patient with recurrent unresectable squamous cell carcinoma of
the retromolar trigone-a brief report. J Egypt Natl Cancer Inst.
34:462022. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Cuadra JD, de Vera MT, Carrasco EG and
Martín MI: Adenoid cystic carcinoma with a leptomeningeal
dissemination in a ‘wash’ pattern. BMJ Case Rep. 14:e2471552021.
View Article : Google Scholar
|
|
54
|
Wlock R, Patel K, Patel M and Tanase A:
Leptomeningeal carcinomatosis secondary to a gastrointestinal
stromal tumor. Cureus. 13:e162122021.PubMed/NCBI
|
|
55
|
Serra E, Abarzua-Araya Á, Arance A,
Martin-Huertas R, Aya F, Olondo ML, Rizo-Potau D, Malvehy J, Puig
S, Carrera C and Podlipnik S: Predictive and prognostic factors in
melanoma central nervous system metastases-a cohort study. Cancers
(Basel). 16:22722024. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Legrand C, Bour JM, Jacob C, Capiaumont J,
Martial A, Marc A, Wudtke M, Kretzmer G, Demangel C and Duval D:
Lactate dehydrogenase (LDH) activity of the cultured eukaryotic
cells as marker of the number of dead cells in the medium
[corrected]. J Biotechnol. 25:231–243. 1992. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
An YJ, Cho HR, Kim TM, Keam B, Kim JW, Wen
H, Park CK, Lee SH, Im SA, Kim JE, et al: An NMR metabolomics
approach for the diagnosis of leptomeningeal carcinomatosis in lung
adenocarcinoma cancer patients. Int J Cancer. 136:162–171. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Paisana E, Cascão R, Custódia C, Qin N,
Picard D, Pauck D, Carvalho T, Ruivo P, Barreto C, Doutel D, et al:
UBE2C promotes leptomeningeal dissemination and is a therapeutic
target in brain metastatic disease. Neuro Oncol Adv. 5:vdad0482023.
View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Bönig L, Möhn N, Ahlbrecht J, Wurster U,
Raab P, Puppe W, Sühs KW, Stangel M, Skripuletz T and
Schwenkenbecher P: Leptomeningeal metastasis: The role of
cerebrospinal fluid diagnostics. Front Neurol. 10:8392019.
View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Chi Y, Remsik J, Kiseliovas V, Derderian
C, Sener U, Alghader M, Saadeh F, Nikishina K, Bale T,
Iacobuzio-Donahue C, et al: Cancer cells deploy lipocalin-2 to
collect limiting iron in leptomeningeal metastasis. Science.
369:276–282. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Lupica-Tondo GL, Arner EN, Mogilenko DA
and Voss K: Immunometabolism of ferroptosis in the tumor
microenvironment. Front Oncol. 14:14413382024. View Article : Google Scholar : PubMed/NCBI
|
|
62
|
Sivanand S, Gultekin Y, Winter PS,
Vermeulen SY, Tchourine KM, Abbott KL, Danai LV, Gourgue F, Do BT,
Crowder K, et al: Cancer tissue of origin constrains the growth and
metabolism of metastases. Nat Metab. 6:1668–1681. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Malani R, Fleisher M, Kumthekar P, Lin X,
Omuro A, Groves MD, Lin NU, Melisko M, Lassman AB, Jeyapalan S, et
al: Cerebrospinal fluid circulating tumor cells as a quantifiable
measurement of leptomeningeal metastases in patients with HER2
positive cancer. J Neurooncol. 148:599–606. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
64
|
Malhotra J, Muddasani R, Fricke J,
Mambetsariev I, Reyes A, Babikian R, Dingal ST, Kim P, Massarelli
E, Feldman L, et al: Clinical utility of a circulating tumor
cell-based cerebrospinal fluid assay in the diagnosis and molecular
analysis of leptomeningeal disease in patients with advanced
non-small cell lung cancer. JCO Precis Oncol. 8:e24003732024.
View Article : Google Scholar : PubMed/NCBI
|
|
65
|
Darlix A, Cayrefourcq L, Pouderoux S, de
Champfleur NM, Bievelez A, Jacot W, Leaha C, Thezenas S and
Alix-Panabières C: Detection of circulating tumor cells in
cerebrospinal fluid of patients with suspected breast cancer
leptomeningeal metastases: A prospective study. Clin Chem.
68:1311–1322. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
66
|
Diaz M, Singh P, Kotchetkov IS, Skakodub
A, Meng A, Tamer C, Young RJ, Reiner AS, Panageas KS, Ramanathan LV
and Pentsova E: Quantitative assessment of circulating tumor cells
in cerebrospinal fluid as a clinical tool to predict survival in
leptomeningeal metastases. J Neuro Oncol. 157:81–90. 2022.
View Article : Google Scholar
|
|
67
|
Nevel KS, Wilcox JA, Robell LJ and Umemura
Y: The utility of liquid biopsy in central nervous system
malignancies. Curr Oncol Rep. 20:602018. View Article : Google Scholar : PubMed/NCBI
|
|
68
|
Congur I, Koni E, Onat OE and Keskin ZT:
Meta-analysis of commonly mutated genes in leptomeningeal
carcinomatosis. PeerJ. 11:e152502023. View Article : Google Scholar : PubMed/NCBI
|
|
69
|
Cho JH, Sim MH, Kim SY, Kim K, Lee T, Lee
J, Kang WK and Kim ST: Analysis of intrapatient heterogeneity of
circulating tumor cells at the single-cell level in the
cerebrospinal fluid of a patient with metastatic gastric cancer. J
Cancer Res Ther. 17:1047–1051. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
70
|
Ma C, Yang X, Xing W, Yu H, Si T and Guo
Z: Detection of circulating tumor DNA from non-small cell lung
cancer brain metastasis in cerebrospinal fluid samples. Thorac
Cancer. 11:588–593. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
71
|
Cordone I, Masi S, Summa V, Carosi M,
Vidiri A, Fabi A, Pasquale A, Conti L, Rosito I, Carapella CM, et
al: Overexpression of syndecan-1, MUC-1, putative stem cell markers
in breast cancer leptomeningeal metastasis: A cerebrospinal fluid
flow cytometry study. Breast Cancer Res. 19:462017. View Article : Google Scholar : PubMed/NCBI
|
|
72
|
Marowsky M, Müller V, Schmalfeldt B,
Riecke K, Witzel I and Laakmann E: Intrathecal therapy options for
meningeal carcinomatosis. Geburtshilfe Frauenheilk. 84:59–67. 2024.
View Article : Google Scholar : PubMed/NCBI
|
|
73
|
Le Rhun E, Wallet J, Mailliez A, Le Deley
MC, Rodrigues I, Boulanger T, Lorgis V, Barrière J, Robin YM,
Weller M and Bonneterre J: Intrathecal liposomal cytarabine plus
systemic therapy versus systemic chemotherapy alone for newly
diagnosed leptomeningeal metastasis from breast cancer. Neuro
Oncol. 22:524–538. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
74
|
Bian DJH, Lazaratos AM, Maritan SM,
Quaiattini A, Zeng Z, Zhu Z, Sener U, Malani R, Kim YJ, Ichihara E,
et al: Osimertinib is associated with improved outcomes in
pre-treated non-small cell lung cancer leptomeningeal metastases: A
systematic review and meta-analysiss. Heliyon. 10:e296682024.
View Article : Google Scholar : PubMed/NCBI
|
|
75
|
Sherman WJ, Romiti E, Michaelides L,
Moniz-Garcia D, Chaichana KL, Quiñones-Hinojosa A and Porter AB:
Systemic therapy for melanoma brain and leptomeningeal metastases.
Curr Treat Options Oncol. 24:1962–1977. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
76
|
Mizumatsu S, Wakabayashi K and Terashima
Y: Palliative treatment of leptomeningeal carcinomatosis from renal
cell carcinoma with local cyberknife radiotherapy and systemic
pazopanib therapy: A case report. Cureus. 16:e540252024.PubMed/NCBI
|
|
77
|
Chiang CL, Ho HL, Yeh YC, Lee CC, Huang
HC, Shen CI, Luo YH, Chen YM, Chiu CH and Chou TY: Efficacy of
different platforms in detecting EGFR mutations using cerebrospinal
fluid cell-free DNA from non-small-cell lung cancer patients with
leptomeningeal metastases. Thorac Cancer. 14:1251–1259. 2023.
View Article : Google Scholar : PubMed/NCBI
|
|
78
|
Yu C, Xu T, Fang H, Wang X, Liu N, Yang L
and Fang S: High-dose furmonertinib combined with intraventricular
chemotherapy as salvage therapy for leptomeningeal metastasis from
EGFR exon 20 insertion-mutated lung cancer. J Neurooncol.
169:203–213. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
79
|
Bai K, Chen X, Qi X, Zhang Y, Zou Y, Li J,
Yu L, Li Y, Jiang J, Yang Y, et al: Cerebrospinal fluid circulating
tumour DNA genotyping and survival analysis in lung adenocarcinoma
with leptomeningeal metastases. J Neurooncol. 165:149–160. 2023.
View Article : Google Scholar : PubMed/NCBI
|
|
80
|
Zhong W, Wu L, Huang L, Wang J, Shi H and
Wu S: Double-dose osimertinib combined with intrathecal injection
of pemetrexed improves the efficacy of EGFR-mutant non-small cell
lung cancer and leptomeningeal metastasis: Case report and
literature review. Front Oncol. 14:13774512024. View Article : Google Scholar : PubMed/NCBI
|
|
81
|
Dekker LJ, Boogerd W, Stockhammer G,
Dalebout JC, Siccama I, Zheng P, Bonfrer JM, Verschuuren JJ,
Jenster G, Verbeek MM, et al: MALDI-TOF mass spectrometry analysis
of cerebrospinal fluid tryptic peptide profiles to diagnose
leptomeningeal metastases in patients with breast cancer. Mol Cell
Proteomics. 4:1341–1349. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
82
|
Xu Y, Huang Z, Yu X, Chen K and Fan Y:
Integrated genomic and DNA methylation analysis of patients with
advanced non-small cell lung cancer with brain metastases. Mol
Brain. 14:1762021. View Article : Google Scholar : PubMed/NCBI
|
|
83
|
Xing L, Pan Y, Shi Y, Shu Y, Feng J, Li W,
Cao L, Wang L, Gu W, Song Y, et al: Biomarkers of osimertinib
response in patients with refractory and EGFR-T790M-positive
non-small cell lung cancer and central nervous system metastases:
The APOLLO study. Clin Cancer Res. 26:6168–6175. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
84
|
Chen H, Yang S, Wang L, Wu Y, Wu Y, Ma S,
He Z, Zhang C, Liu Y, Tang H, et al: High-Dose furmonertinib in
patients with EGFR-Mutated NSCLC and leptomeningeal metastases: A
prospective real-world study. J Thorac Oncol. 20:65–75. 2025.
View Article : Google Scholar : PubMed/NCBI
|
|
85
|
Wang Y, Deng H, Xin S, Zhang K, Shi R and
Bao X: Prognostic and predictive value of three DNA methylation
signatures in lung adenocarcinoma. Front Genet. 10:3492019.
View Article : Google Scholar : PubMed/NCBI
|
|
86
|
Xia S, Ye J and Chen Y, Lizaso A, Huang L,
Shi L, Su J, Han-Zhang H, Chuai S, Li L and Chen Y: Parallel serial
assessment of somatic mutation and methylation profile from
circulating tumor DNA predicts treatment response and impending
disease progression in osimertinib-treated lung adenocarcinoma
patients. Transl Lung Cancer Res. 8:1016–1028. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
87
|
Lee KY, Im JH, Lin W, Gwak HS, Kim JH, Yoo
BC, Kim TH, Park JB, Park HJ, Kim HJ, et al: Nanoparticles in 472
human cerebrospinal fluid: Changes in extracellular vesicle
concentration and miR-21 expression as a biomarker for
leptomeningeal metastasis. Cancers (Basel). 12:27452020. View Article : Google Scholar : PubMed/NCBI
|
|
88
|
Sawada H, Taniguchi Y, Iizuka S, Ikeda T,
Aga M, Hamakawa Y, Miyazaki K, Misumi Y, Agemi Y, Nakamura Y, et
al: Entrectinib response to ROS1-fusion-positive non-small-cell
lung cancer that progressed on crizotinib with leptomeningeal
metastasis: A case report. Case Rep Oncol. 16:1558–1567. 2023.
View Article : Google Scholar : PubMed/NCBI
|
|
89
|
Wijetunga NA, Goglia AG, Weinhold N,
Berger MF, Cislo M, Higginson DS, Chabot K, Osman AM, Schaff L,
Pentsova E, et al: Dynamic mutational landscape of cerebrospinal
fluid circulating tumor DNA and predictors of survival after proton
craniospinal irradiation for leptomeningeal metastases. Clin Cancer
Res. 29:775–783. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
90
|
Boire A, Brandsma D, Brastianos PK, Le
Rhun E, Ahluwalia M, Junck L, Glantz M, Groves MD, Lee EQ, Lin N,
et al: Liquid biopsy in central nervous system metastases: A RANO
review and proposals for clinical applications. Neuro Oncol.
21:571–584. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
91
|
Park S, Lee MH, Seong M, Kim ST, Kang JH,
Cho BC, Lee KH, Cho EK, Sun JM, Lee SH, et al: A phase II,
multicenter, two cohort study of 160 mg osimertinib in EGFR
T790M-positive non-small-cell lung cancer patients with brain
metastases or leptomeningeal disease who progressed on prior EGFR
TKI therapy. Ann Oncol. 31:1397–1404. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
92
|
Smalley I, Law V, Wyatt C, Evernden B,
Fang B, Koomen JM, Welsh EA, Macaulay RJB, Forsyth PA and Smalley
KSM: Proteomic analysis of CSF from patients with leptomeningeal
melanoma metastases identifies signatures associated with disease
progression and therapeutic resistance. Clin Cancer Res.
26:2163–2175. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
93
|
Li G, Fang M, Zhou Y, Liu X, Tian P and
Mei F: Afatinib overcoming resistance to icotinib and osimertinib
in NSCLC with leptomeningeal metastasis in patients with acquired
EGFR L858R/T790M or L858R/S768I mutations: Two case reports.
Heliyon. 9:e206902023. View Article : Google Scholar : PubMed/NCBI
|
|
94
|
Koyuncuer A, Varol E, Yağcioğlu BS, Bükte
Y and Sakcı Z: Cerebrospinal fluid-dissemination of a ovarian clear
cell carcinoma: A leptomeningial carcinomatosis with diagnostic
challenges. Diagn Cytopathol. 51:E228–E231. 2023. View Article : Google Scholar : PubMed/NCBI
|