Introduction
The morbidity of pancreatic cancer has been
increased globally in recent years. Moreover, the prognosis of
pancreatic cancer is very poor. With the development of research in
molecular biology, oncology and the related subjects, the gene
therapy of pancreatic cancer is regarded as a new mode of cancer
treatment subsequently following traditional therapy such as
surgical operation, radiotherapy and chemotherapy. Cyclooxygenase-2
(COX-2), is a key enzyme of the metabolic process of arachidonic
acid, which plays an important role in tumorigenesis, apoptosis and
angiogenesis (1). Our previous
study found that COX-2 was highly expressed in pancreatic cancer
tissue and associated with other genes regulating cell growth of
pancreatic cancer cells (2–4). Therefore, COX-2 is implicated as one
of the molecular targets of gene therapy for pancreatic cancer. RNA
interference (RNAi) is a post-transcriptional gene silencing
process. RNAi is initiated by the enzyme Dicer which cleaves long
double-stranded RNA (dsRNA) molecules into short fragments of 21–25
nt. These short double-stranded fragments are called small
interfering RNAs (siRNAs). These siRNAs are then separated into
single strands and integrated into an active RNA-induced silencing
complex (RISC). After integration into the RISC, siRNAs base-pair
to their target mRNA and induce cleavage of the mRNA, thereby
leading to post-transcriptional gene silencing. Artificial
synthetic siRNAs have become an important tool for research on gene
function after gene silencing. siRNAs could effectively and
specifically suppress the target gene expression, which implicate
their application in the treatment of many refractory diseases
including pancreatic cancer. In this study, we investigated the
effects of COX-2 gene silencing by siRNA on cell proliferation,
cell apoptosis, cell cycle and tumorigenicity of Capan-2 human
pancreatic cancer cells. This study may provide experimental
evidence for gene therapy in pancreatic cancer.
Materials and methods
Materials and reagents
COX-2 siRNAs labeled by fluorescence Cy3
(Cy3-siRNAs) were synthesized by Guangzhou Ruibo Biotechnology Co.,
Ltd. (Guangzhou, China). The RPMI-1640 medium, 10% FBS and
Lipofectamine 2000 (Lipo) were obtained from the Gibco-Invitrogen
Corporation (USA). The cDNA synthesis kit was obtained from Dalian
Bao Bioengineering Co., Ltd. (Dalian, China). COX-2 mAb was
purchased from the Cayman Chemical Corportation (USA). β-actin mAb
was purchased from the Shanghai Kangcheng Biotechnology Corporation
(Shanghai, China). HRP-conjugated goat anti-mouse IgG was purchased
from the Shanghai Lianke Biotechnology Corporation. ECL™ Western
Blotting Detection system was obtained from Thermo Corporation
(USA). BCA™ protein assay kit was purchased from Shanghai Shenneng
Lottery Biotechnology Corporation (Shanghai, China). Annexin V
FITC/PI assay kit was purchased from Invitrogen Corporation
(USA).
COX-2 siRNA sequences
The COX-2 siRNA001, target sequence was
5′-GCTGGGAAGCCTTCTCTAA-3′ the sense strand was
5′-GCUGGGAAGCCUUCUCUAAdTdT-3′ and the antisense strand was
5′-dTdTCGACCCUUCGGAAGAGAUU-3′. COX-2 siRNA002, target sequence was
5′-GCAGCTTCCTGATTCAAAT-3′ the sense strand was
5′-GCAGCUUCCUGAUUCAAAUdTdT-3′ and the antisense strand,
5′-dTdTCGUCGAAGGACUAAGUUUA-3′. The COX-2 siRNA003, target sequence
was 5′-GAATCATTCACCAGGCAAA-3′ the sense strand was
5′-GAAUCAUUCACCAGGCAAAdTdT-3′ and the antisense strand was
5′-dTdTCUUAGUAAGUGGUCCGUUU-3′. The COX-2 siRNA004, target sequence
was 5′-AACACCGGAATTTTTGACAAG-3′ the sense strand was
5′-AACACCGGAAUUUUUGACAAGdTdT-3′ and the antisense strand,
5′-dTdTUUGUGGCCUUAAAAACUGUUC-3′. The COX-2 siRNA005 target sequence
was 5′-GATTGAAGATTATGTGCAA-3′ the sense strand was
5′-GAUUGAAGAUUAUGUGCAAdTd-3′ and the antisense strand was
5′-dTdTCUAACUUCUAAUACACGUU-3′. The COX-2 siRNA006, target sequence
was 5′-GGACTTATGGGTAATGTTA-3′ the sense strand was
5′-GGACUUAUGGGUAAUGUUAdTdT-3′ and the antisense strand was
5′-dTdTCCUGAAUACCCAUUACAAU-3′.
PCR primers
PCR primers were synthesized by the Shanghai Yingjun
Corporation (Shanghai, China). The RT-PCR primers were, COX-2
primers, forward, TTCAAATGAGATTGTGGGAAAAT and reverse,
AGATCATCTCTGCCTGAGTATCTT. β-actin primer, forward,
AAGGAAGGCTGGAAGAGTGC and reverse, CTACAATGAGCTGCGTGTGG. The
real-time PCR primers were COX-2 primers, forward,
CTGGAACATGGAATTACCCAGTTTG and reverse, TGGAACATTCCTACCACCAGCA;
β-actin primers, forward, TGGCACCCAGCACAATGAA and reverse,
CTAAGTCATAGTCCGCCTAGAAGCA.
Cell culture
The Capan-2 human pancreatic cancer cell line was
provided by the California University and the cells were cultured
in RPMI-1640 medium supplemented with 10% FBS without addition of
antibiotics at 37°C in a humidified incubator containing 5%
CO2.
siRNA transfection
Capan-2 cells were seeded at 1×106/well
in 6-well plates 1 day prior to transfection and 1.5 ml of medium
without antibiotics was added into each well so that the cells grew
to 30–50% confluence when the transfection was performed. The
siRNA-Lipo mixture was prepared according to the manufacturer's
instructions. The siRNA-Lipo mixture was uniformly added into each
well containing cells and medium. The plate was shaken back and
forth for mixing. The cells were then cultured at 37°C in a
humidified incubator containing 5% CO2, and were
collected after 24, 48 and 72 h of culture, respectively. Total-RNA
was isolated by using the TRIzol reagent. Total protein was
extracted by the RIPA assay. The RNA samples and protein samples
were stored at −30°C.
Screening test of Cy3-siRNAs and Lipo for
transfection efficiency
To test the transfection efficiency of Lipo at
different concentrations, 50 nM of Cy3-siRNAs and different
concentrations of Lipo including 3, 4, 5 and 6 μl of Lipo were
used. To test the transfection efficiency of Cy3-siRNAs at
different concentrations, 5 μl of Lipo and different concentrations
of Cy3-siRNAs including 30, 50 and 100 nM of Cy3-siRNAs were
used.
Screening efficient sequences of COX-2
siRNAs by using RT-PCR and real-time PCR
Single stranded cDNA was synthesized from 1 μg RNA
using a cDNA synthesis kit. The conditions for reverse
transcription were as follows: 65°C for 5 min, then 42°C 30 min and
95°C for 5 min to inactivate the enzyme. The newly synthesized cDNA
was amplified by PCR using the GeneAmp2700 PCR instrument (GeneAmp,
Germany). Cycling conditions were as follows: 94°C for 2 min to
denature cDNA and primers, then followed by 30 cycles at 94°C for
30 sec, 55°C (COX-2) or 61°C (β-actin) for 30 sec and 72°C for 1
min. Real-time PCR was performed by using LightCycler 480 real-time
PCR instrument (Roche, Switzerland). Cycling conditions for
real-time PCR were as follows: 95°C for 30 sec to denature cDNA and
primers, followed by 40 cycles at 95°C for 5 sec and 60°C for 20
sec.
Western blotting
Protein expression of COX-2 was measured by Western
blotting. β-actin was used as an internal control. Cellular
proteins were dissolved in sample loading buffer and run on 7.5%
sodium dodecyl sulfate-polyacrylamide gel electrophoresis
(SDS-PAGE) gels (100 V, constant voltage, 60 min). COX-2 protein
was electrotransferred onto PVDF membranes (4°C, 200 mA, 100 min).
The membranes were rinsed with PBS and blocked with 10% nonfat milk
in PBS for 3 h at room temperature. Membranes were then incubated
with the primary antibody of COX-2 (1:1,000) or β-actin (1:5,000)
in 3% nonfat milk overnight at 4°C. After primary antibody
incubation, membranes were rinsed in TBS-T wash buffer for 10 min 3
times each. Membranes were then incubated with secondary antibody
(HRP-conjugated goat anti-mouse IgG, 1:5,000) for 2 h at room
temperature and rinsed in TBS-T wash buffer for 10 min 3 times
each. The protein-antibody complexes were visualized by ECL™
Western Blotting Detection system.
Experiment grouping in vitro
Capan-2 human pancreatic cancer cells were cultured
in 6-well plates. The efficient COX-2 siRNA was transfected into
Capan-2 cells. Capan-2 cells were harvested respectively at 24, 48
and 72 h after transfection. Cell counting and flow cytometry (FCM)
were performed. Capan-2 cells without transfection were set as
blank control group. Capan-2 cells treated with negative siRNA
transfection were set as negative control group. Capan-2 cells
treated with only Lipo were set as the liposome control group.
Cell proliferation analysis
Cell proliferation analysis was performed by the
cell counting method. Capan-2 human pancreatic cancer cells were
seeded at 2×105 cells/well in 6-well plates. After 24 h
incubation, siRNA transfection was performed. Subsequently, cells
were incubated at 37°C in 5% CO2. Cells were harvested
by digestion of 0.25% trypsin at 24, 48 and 72 h respectively after
transfection. Cells of each well were counted three times. Cell
growth curves were drawn based on the average cell count of each
group.
Analysis of cell apoptosis and cell
cycle
Capan-2 human pancreatic cancer cells were seeded at
2×105 cells/well in 6-well plates. After 24 h
incubation, siRNA transfection was performed. Then cells were
incubated at 37°C in 5% CO2. Cells were harvested at 24,
48 and 72 h respectively after transfection and washed twice with
cold PBS. Cells were resuspended with 1 ml of PBS to be
1–5×106/ml of cell suspension and 0.5 ml of cell
suspension was used for cell apoptosis analysis. The remaining
portions were used for cell cycle analysis. Analysis of cell
apoptosis and cell cycle were performed by FCM using the
FACSCalibur™ flow cytometer (Becton-Dickinson, San Jose, CA).
Experiment in vivo
Animal care and euthanasia were approved by the Sun
Yat-Sen University animal studies committee. Eighteen BALB/c nu/nu
nude mice (experimental animal center, Sun Yat-Sen University,
China) were randomly divided into three groups including the
control group, negative control group and COX-2 siRNA group. Each
group consisted of 6 nude mice. As the most efficient COX-2 siRNA,
COX-2 siRNA006 was selected for transfection in vivo. Cells
including parent Capan-2 cells, Capan-2 cells transfected with
negative siRNA and Capan-2 cells transfected with COX-2 siRNA006
were harvested in the exponential growth phase and washed with cold
PBS for three times. The cells were resuspended with PBS to a 0.2
ml single cell suspension (5×106 cells/ml). The cell
suspensions were inoculated subcutaneously into flanks of
4–6-week-old BALB/C nu/nu nude mice. Xenografts in each group were
observed periodically after inoculation. The tumorigenicity of
xenografts was evaluated. The length and width of xenografts were
measured. The volume of the xenograft was calculated according to
the following formula: V=ab2/2 (V, volume; a, length; b,
width). The inhibition rate of the xenograft was calculated based
on the average volume of the xenograft. Nude mice were sacrificed
by cervical dislocation at 6 weeks after inoculation. The
xenografts were peeled off subcutaneously. The weight of the
xenografts in each group was compared.
Statistical analysis
All measurement data were present as mean ± standard
deviation (SD). The differences among multiple mean values were
evaluated using analysis of variance (ANOVA). The differences
between two mean values were estimated using independent-samples
t-test. Values for counting data were present as a rate or ratio.
The differences among the groups were analyzed using the
χ2 test. All of the statistical analyses were processed
with the statistical analysis software SPSS, version 10.0 (SPSS,
Chicago, IL).
Results
Transfection efficiency and the most
optimal transfection concentration of Lipo and siRNA
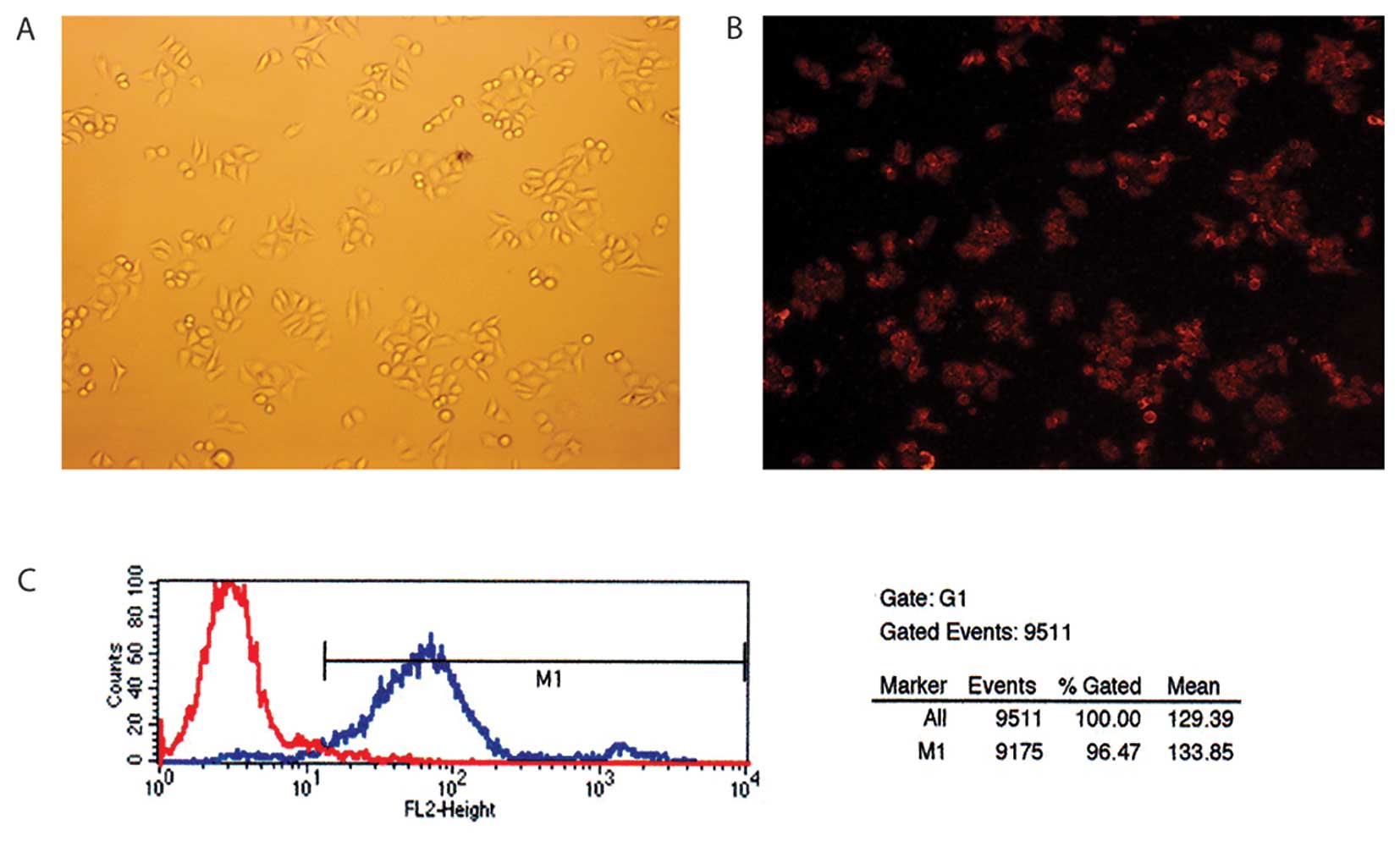
Cy3 is a red fluorescent molecule with an excitation
wavelength of 480 or 550 nm. The red fluorophore could be seen in
the cells successfully transfected with siRNA under the fluorescent
microscope (Fig. 1A and B). The
transfection efficiency was measured by FCM. Fig. 1C showed that the transfection
efficiency was 96.47%. We found that the most optimal transfection
concentration of siRNA was 50 nM and the most optimal transfection
dosage of Lipo was 5 μl/2 ml after screening the different
concentrations of siRNA and Lipo.
Screening for the most optimal sequence
of COX-2 siRNAs silencing COX-2 gene expression in Capan-2
cells
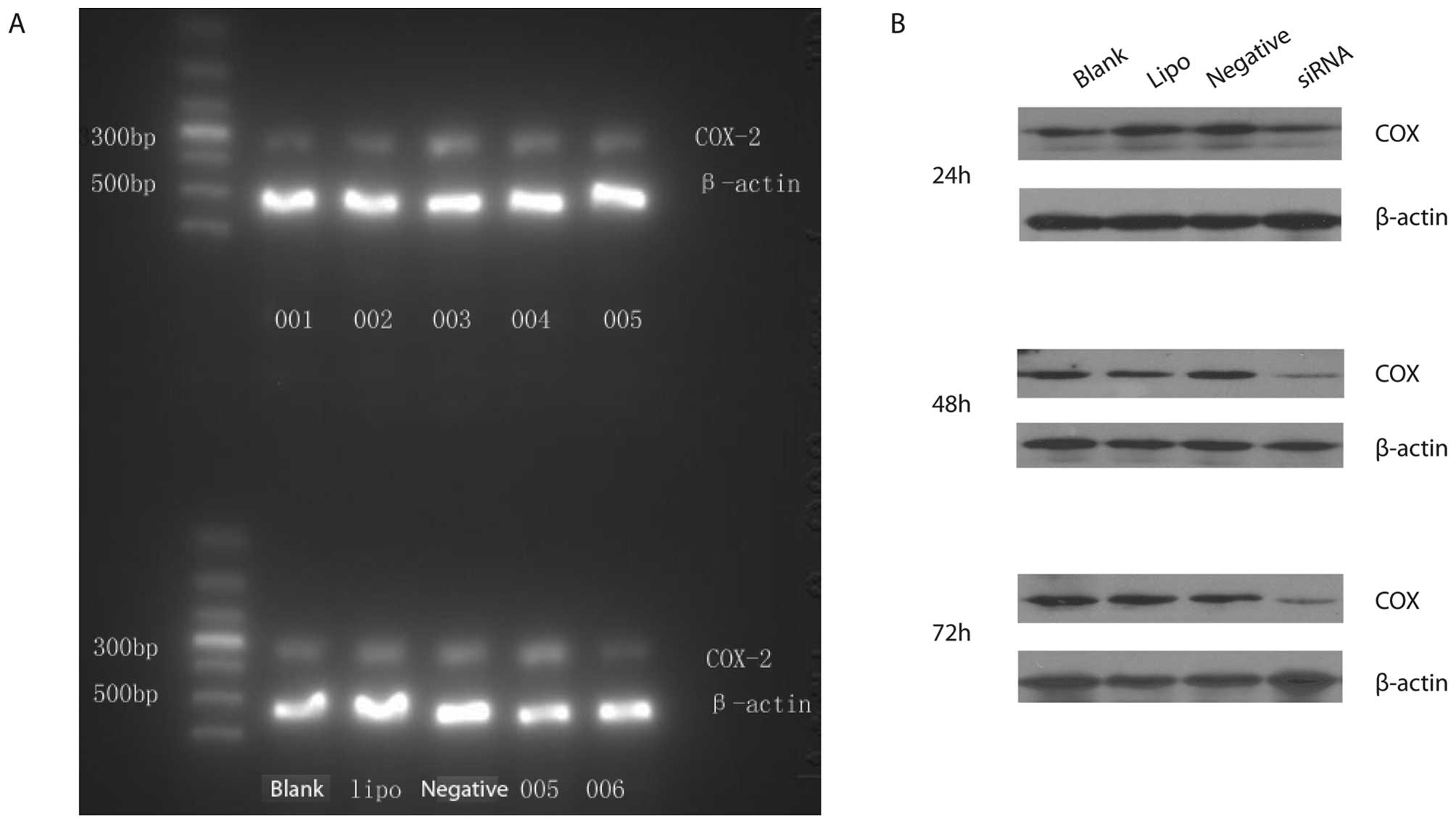
Six sequences of COX-2 siRNAs were used to determine
the silencing efficiency of COX-2 siRNAs on COX-2 mRNA expression
in Capan-2 cells. COX-2 siRNA001, COX-2 siRNA002 and COX-2 siRNA006
were found to be the most optimal sequences. However, COX-2
siRNA003, COX-2 siRNA004 and COX-2 siRNA005 were the invalid
sequences (Fig. 2A). COX-2 siRNA006
was found to have the most powerful silencing effect on COX-2 gene.
The silencing efficiency of COX-2 siRNA006 was significantly higher
than the blank control group (P<0.05). The silencing efficiency
of COX-2 siRNA006 on COX-2 mRNA was 73% at 24 h after transfection.
Moreover, protein expressions of COX-2 were down-regulated by 67
and 61% respectively at 48 and 72 h after COX-2 siRNA006
transfection. However, there was no significant inhibition effect
on protein expression of COX-2 at 24 h after COX-2 siRNA006
transfection. There were no significant differences among the Lipo
control group, negative control group and blank control group
(P>0.05) (Fig. 2B).
Cell viability of Capan-2 cells treated
with COX-2 siRNA006 at different time points
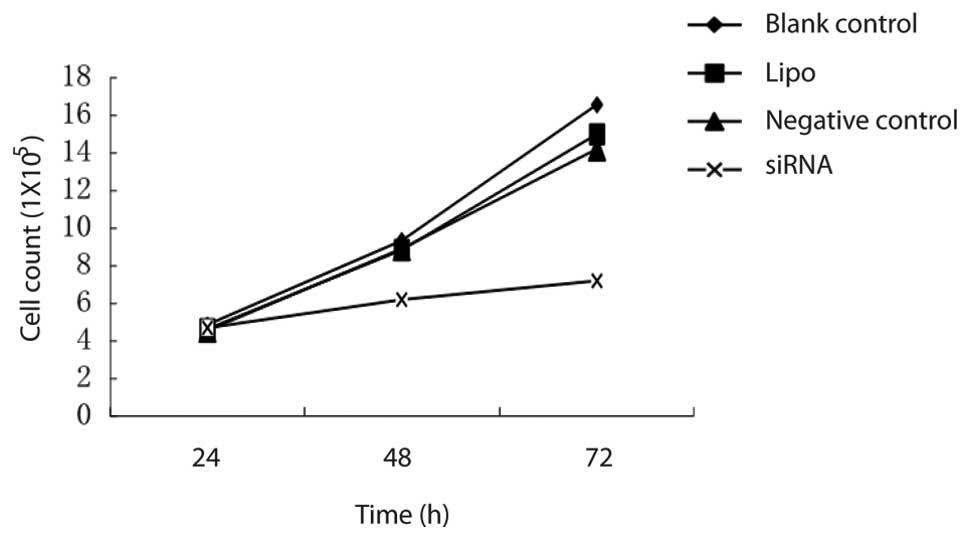
There was no significant difference in cell
viability among the different groups at 24 h after transfection
(P<0.05). Cell viability of Capan-2 cells in the COX-2 siRNA006
group was significantly decreased at 48 h after transfection. The
inhibition rate of cell proliferation was 35.48 and 56.32%
respectively at 48 and 72 h after transfection. However, there was
no significant difference between the negative group and the Lipo
control group (Fig. 3).
The influence of COX-2 siRNA006 on cell
apoptosis of Capan-2 cells
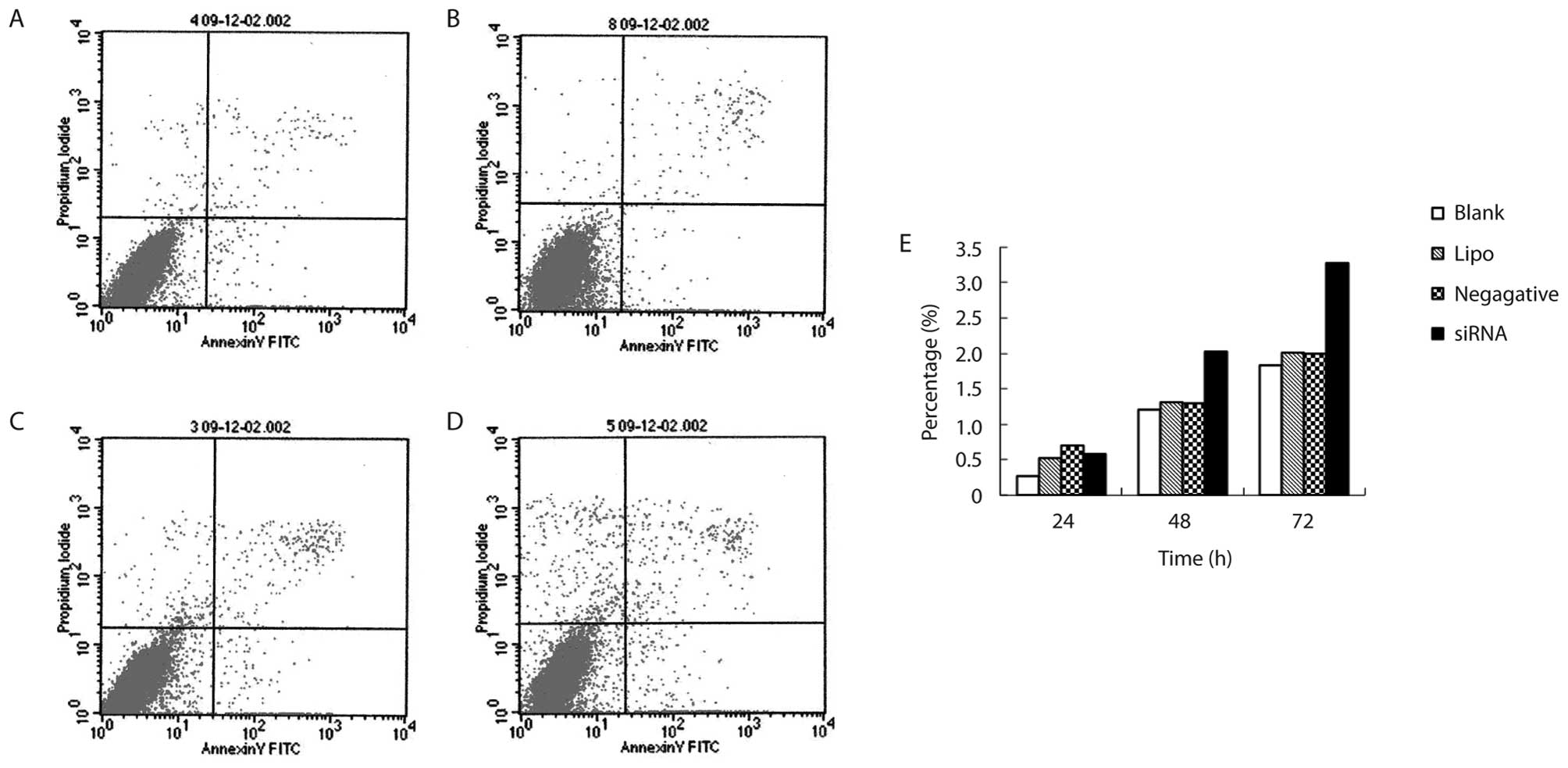
The apoptotic cells were increased as the culture
time of cells transfected with COX-2 siRNA006 was increased
(P<0.05). This was more obvious at 48 h after transfection.
There was a significant difference between the COX-2 siRNA group
and the negative control group (P<0.05). There was a significant
difference between the COX-2 siRNA group and the Lipo control group
(P<0.05). However, there were no significant difference among
negative control group, the Lipo control group and the blank
control group (Table I and Fig. 4).
 | Table IThe effect of COX-2-siRNA on apoptosis
of Capan-2 cells (n=3). |
Table I
The effect of COX-2-siRNA on apoptosis
of Capan-2 cells (n=3).
| Group | Time (h) | Apoptosis (%) |
|---|
| COX-2 siRNA | 24 | 0.58±0.32 |
| 48 | 2.03±0.07a |
| 72 | 3.27±0.29a |
| Negative control | 24 | 0.70±0.16 |
| 48 | 1.29±0.23 |
| 72 | 1.99±0.23 |
| Lipo control | 24 | 0.52±0.41 |
| 48 | 1.31±0.15 |
| 72 | 2.01±0.21 |
| Blank control | 24 | 0.27±0.28 |
| 48 | 1.20±0.16 |
| 72 | 1.83±0.28 |
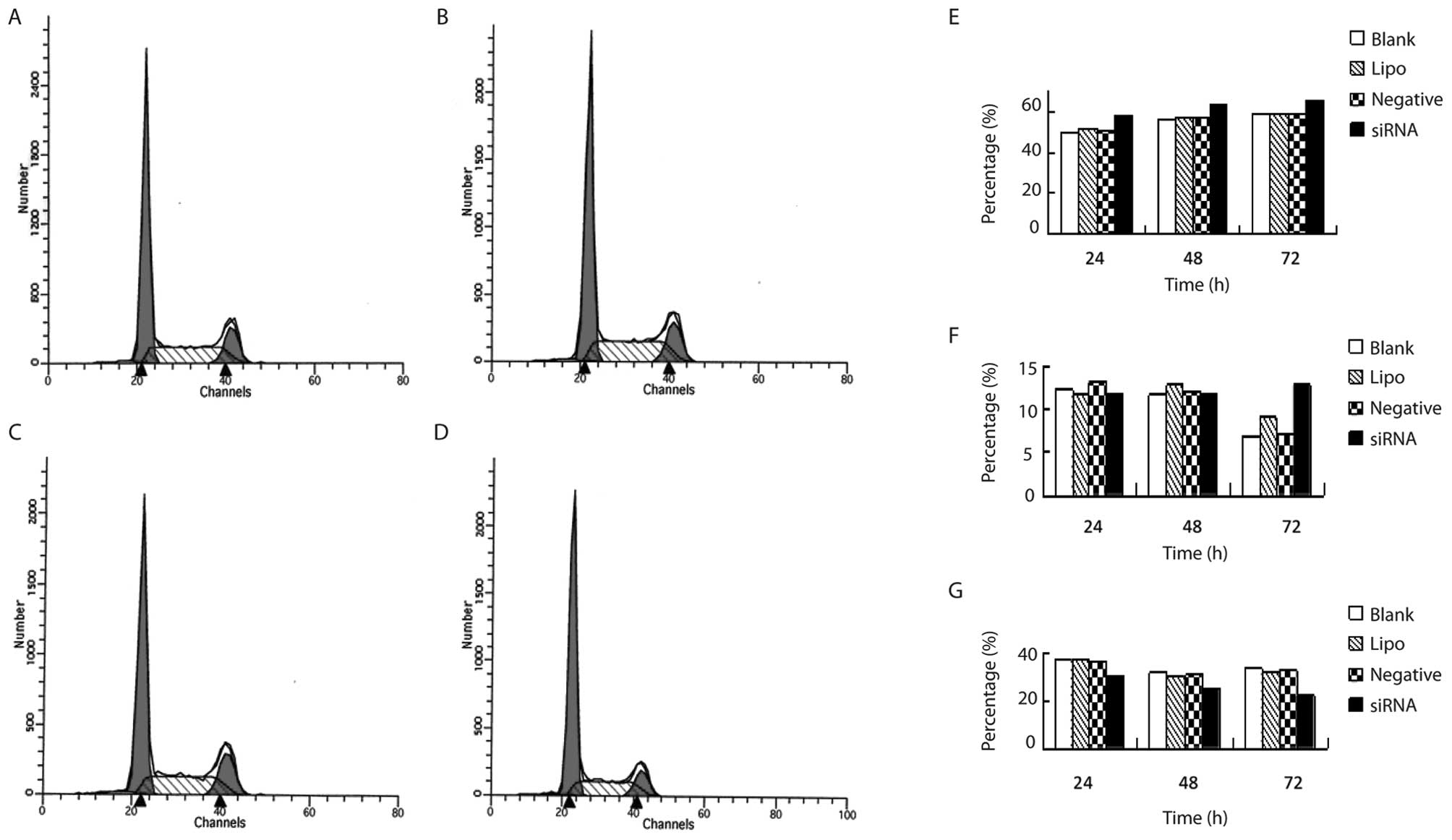
The influence of COX-2 siRNA006 on the
cell cycle of Capan-2 cells
The cells in G0/G1 phase were significantly
increased as the culture time of cells transfected with COX-2
siRNA006 increased (P<0.05). However, the cells in the S phase
were significantly decreased (P<0.05) (Table II and Fig. 5).
 | Table IIThe influence of COX-2 siRNA006 on the
cell cycle of Capan-2 cells (%) (n=3). |
Table II
The influence of COX-2 siRNA006 on the
cell cycle of Capan-2 cells (%) (n=3).
| Group | Culture time (h) | G0/G1 | G2/M | S |
|---|
| COX-2 siRNA | 24 | 58.03±1.72a | 11.70±1.19 | 30.27±0.53b |
| 48 | 63.31±1.92a | 11.81±1.72 | 24.87±1.03a |
| 72 | 65.66±0.56a | 12.81±3.22a | 22.2±2.24b |
| Negative control | 24 | 50.63±0.68 | 13.32±0.64 | 36.04±1.09 |
| 48 | 57.08±0.47 | 12.09±1.58 | 30.83±1.53 |
| 72 | 59.53±3.19 | 7.08±1.88 | 33.06±2.11 |
| Lipo control | 24 | 51.38±3.62 | 11.66±2.61 | 36.95±1.35 |
| 48 | 57.09±3.19 | 12.88±0.49 | 30.08±2.68 |
| 72 | 58.97±3.15 | 9.06±0.91 | 31.97±2.41 |
| Blank control | 24 | 49.92±3.54 | 12.46±1.35 | 37.61±2.53 |
| 48 | 56.75±3.66 | 11.70±3.30 | 31.56±2.97 |
| 72 | 59.21±2.82 | 6.76±2.21 | 34.03±4.50 |
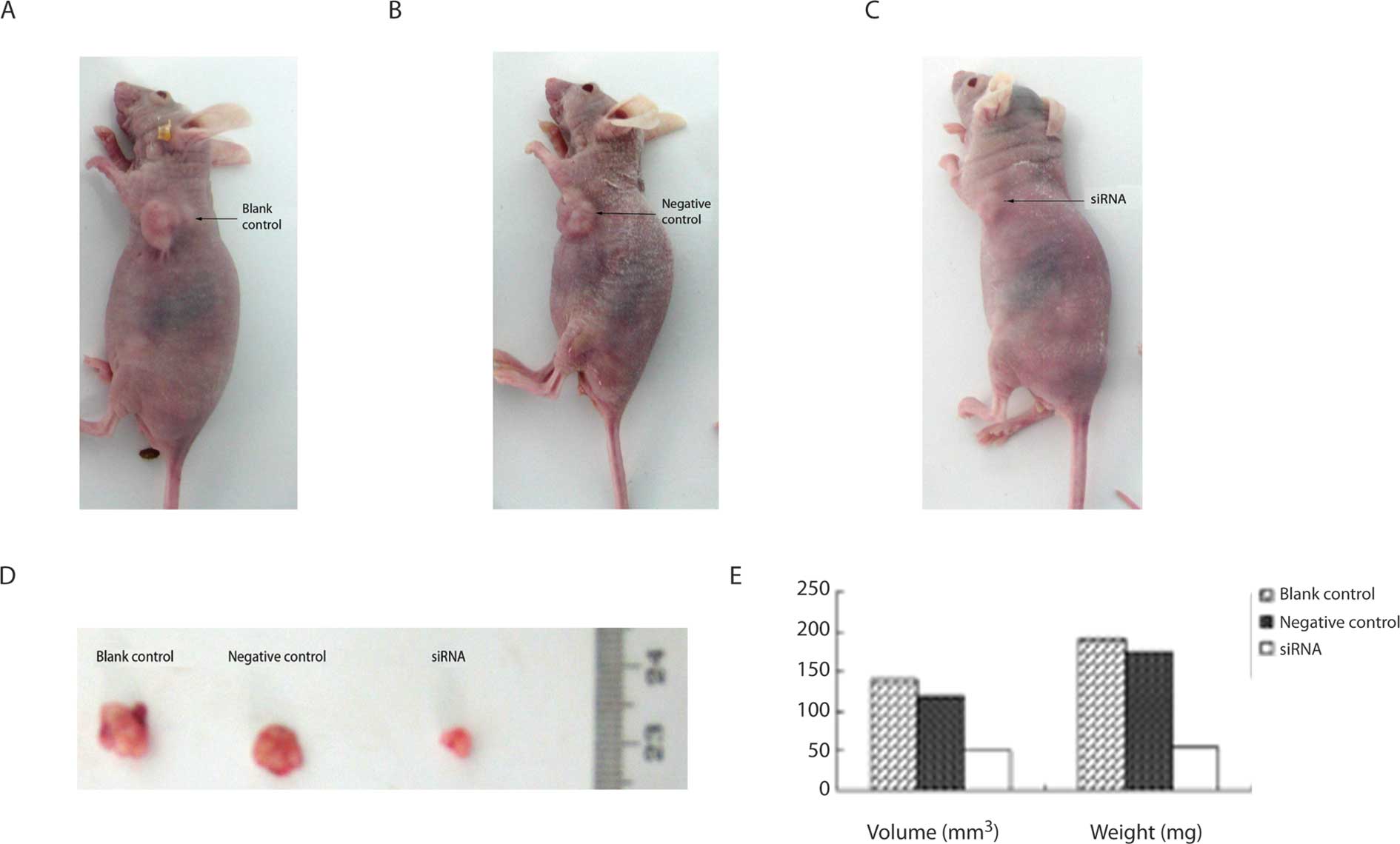
The influence of COX-2 siRNA on
subcutaneous tumorigenicity of Capan-2 cells in nude mice
Subcutaneous xenografts could be seen in the control
group and the negative control group about 1 week after
inoculation. However, those could not be seen in COX-2 siRNA group
although subcutaneous xenografts could be seen in COX-2 siRNA group
about 2 weeks after inoculation. The xenografts grew significantly
more slowly in the COX-2 siRNA group than those in the blank
control group and the negative control group (Fig. 6A-C). The size of the xenografts in
the COX-2 siRNA group were significantly smaller than those in the
blank control group and the negative control group (P<0.05).
However, there was no significant difference between the blank
control group and the negative control group (P>0.05). Nude mice
were sacrificed by cervical dislocation. The xenografts were peeled
off and weighed. The data show that the mean weight of xenografts
in the COX-2 siRNA group were significantly lower than those in the
blank control group and negative control group (P<0.05)
(Table III and Fig. 6D-E).
 | Table IIIThe effect of COX-2 siRNA on the
volume and weight of subcutaneous xenografts in nude mice. |
Table III
The effect of COX-2 siRNA on the
volume and weight of subcutaneous xenografts in nude mice.
| Group | Volume
(mm3) | Weight (mg) |
|---|
| COX-2 siRNA | 50.00±0.01a | 54.70±5.35a |
| Negative control | 119.30±0.02 | 175.53±14.36 |
| Blank control | 141.00±0.05 | 191.76±19.25 |
Discussion
COX-2 could play a role in tumorigenesis via various
pathways. First, cyclooxygenase is an enzyme with a dual function
of both a cyclooxydase and a peroxidase. The latter could directly
activate an oncogene or inhibit mutation of tumor suppressor gene.
Second, as COX-derived metabolites, prostaglandins (PGs),
especially PGE2 could promote proliferation of normal cells and
cancer cells (5–7). Third, overexpression of COX-2 could
prolong the lifetime of cancer cells and inhibit cell apoptosis.
Fourth, the expression of COX-2 lead to the production of PGE2. The
activation of the cell membrane receptor of PGE2 could increase
intracellular cAMP, which could induce the synthesis of vascular
endothelial growth factor (VEGF). Thus the increased VEGF could
promote neovascularization (8).
Fifth, the increased PGE2 could inhibit the activation of natural
killer cells and cytotoxic T cells, the production of TNF and
IL-15, hyperplasia of T cells and B cells, and induce the
production of IL-10 which is an immunosuppressive cytokine. This
may lead to down-regulated surveillant immunity and cytolytic
activity. Therefore, cancer cells could escape immunologic
surveillance; Sixth, expression of COX-2 could enhance the
migration and invasion of cancer cells, which is related to the
direct up-regulation of the expression of matrix metalloproteinase
urokinase-type plasminogen activator by COX-2 (9).
COX-2 is not expressed in the exocrine tissue of the
normal pancreas, but is slightly expressed in islet cells of
pancreas. Many previous studies showed that the expression of COX-2
in pancreatic cancer tissue was markedly increased (2,10,11).
Tucker et al reported that the expression of COX-2 mRNA in
pancreatic cancer tissue was higher by 60 times than that in
adjacent non-tumor pancreatic tissue (12). The expression of COX-2 was notably
increased in some human pancreatic cancer cell lines (13). However, the mechanism is still
unclear. Our previous studies found that the high expression of
COX-2 was related to the high expression of catalyst component of
telomerase, hTERT (3,4). That indicated that COX-2 played a
crucial role in the tumorigenesis, development and metastasis of
pancreatic cancer, which suggests that COX-2 was one of the
important targets of gene therapy in pancreatic cancer.
The COX-2 inhibitor could inhibit proliferation of
pancreatic cancer cells via down-regulation of the expression of
COX-2. However, why do we apply RNAi targeting COX-2 gene to
inhibit proliferation of pancreatic cancer cells? A great deal of
epidemiological data showed that although long-term use of NSAID
may decrease the risk of cancer, it may lead to complications of
the gastrointestinal tract, renal, cardiovascular and
cerebrovascular system. In addition, the inhibition rate of cell
proliferation on pancreatic cancer by the COX-2 inhibitor was
limited with a range of 40–62% depending on the dosage of COX-2
inhibitor (14,15). However, the inhibition rate of cell
proliferation was increased only by about 10% when the dosage of
COX-2 inhibitor was doubled (16).
Moreover, the risk of cardiovascular events may be increased when
the dosage of COX-2 inhibitor was increased. COX-2 selective
inhibitors or COX-2 non-selective inhibitors applied in the
previous studies all post-translationally regulated COX-2
expression with a dose-dependent COX-2 inhibition effect and
limited inhibition efficiency. As the COX-2 inhibitor does not
result in specific inhibition, it more or less inhibited COX-1
which maintained the normal physiological function in body.
Therefore, it may increase the risk of hypertension and
cardiovascular diseases (17). The
safety of COX-2 inhibitor in anticancer research needs further
clinical investigation.
RNAi could be used to promote cell apoptosis,
inhibit proliferation of cancer cells, invasion and metastasis of
cancer and drug resistance by suppressing the expression of
oncogene and genes that are related to carcinogenesis and
development (3,4). RNAi has become a powerful tool in
cancer research as it has the advantage of good reliability, high
specificity, low cytotoxicity and long and strong effect so on.
However, the key point of its successful application is that the
high efficiency silencing sequence (silencing rate >70%) and
high-throughput vector could be screened in advance.
Our data showed that liposome Lipofactamine 2000 had
as high as 96.47% of a transfection rate in Capan-2 cancer cells.
In the meanwhile, COX-2 siRNA006, the most efficient silencing
sequence was screened from six COX-2 siRNAs sequences. COX-2
siRNA006 targeting COX-2 gene was used to investigate its effect on
cell proliferation, cell cycle and cell apoptosis in Capan-2
pancreatic cancer cells. We found that there was no significant
difference in cell viability among different groups at 24 h after
transfection. Cell viability of Capan-2 cells in the COX-2 siRNA006
group was significantly decreased at 48 h after transfection. The
inhibition rate of cell proliferation was 35.48 and 56.32%
respectively at 48 and 72 h after transfection. The cells in G0/G1
phase were significantly increased as the culture time of cells
transfected with COX-2 siRNA006 was increased. However, the cells
in the S phase were significantly decreased. The apoptotic cells
were increased as the culture time of cells transfected with COX-2
siRNA006 was increased.
To further verify the effect of COX-2 siRNA
silencing COX-2 gene on cell growth of Capan-2 human pancreatic
cancer cells, Capan-2 cells transfected with COX-2 siRNA were
subcutaneously inoculated into BALB/c-nu/nu nude mice to
investigate the effect of COX-2 siRNA on tumorigenicity of Capan-2
cells. Our data showed that COX-2 siRNA could significantly inhibit
the tumorigenicity of Capan-2 human pancreatic cancer cells in nude
mice.
The above mentioned data in vitro and in
vivo all indicated that COX-2 siRNA could silence expression of
the COX-2 gene in Capan-2 cells and influence cell proliferation,
cell cycle, cell apoptosis and tumorigenicity of Capan-2 cells,
which may supply gene therapy of pancreatic cancer in clinical
practice with therapeutic target and theoretical evidence.
Acknowledgements
This study was supported by the Guangdong Natural
Science Foundation (no. 07001607) and Guangdong Science-Technology
Plan Program (no. 2008B030301115).
References
|
1
|
Eschwège P, de Ledinghen V, Camilli T, et
al: Arachidonic acid and prostaglandins, inflammation and oncology.
Presse Med. 30:508–510. 2001.(In French).
|
|
2
|
Zhong YQ, Shen XM, Li HG, et al:
Expression of hTERT and its correlation with expression of P-gp,
COX-2, Bcl-2 and the clinical pathological features of patients
with pancreatic cancer. Zhonghua Linchuang Yishi Zazhi. 2:14–18.
2008.
|
|
3
|
Zhong YQ, Xia ZS, Fu YR, et al: Knockdown
of hTERT by siRNA suppresses growth of Capan-2 human pancreatic
cancer cell via the inhibition of expressions of Bcl-2 and COX-2. J
Dig Dis. 11:176–184. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Zhong YQ, Huang HR, Fu YR, et al: Effect
of silencing expression of hTERT gene by siRNA on the expression of
Bcl-2 and COX-2 genes of human pancreatic cancer cells in vitro.
Zhonghua Linchuang Yishi Zazhi. 4:34–38. 2010.
|
|
5
|
Pai R, Soreghan B, Szabo IL, et al:
Prostaglandin E2 transactivates EGF receptor: a novel mechanism for
promoting colon cancer growth and gastrointestinal hypertrophy. Nat
Med. 8:289–293. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Fujino H, West KA and Regan JW:
Phosphorylation of glycogen synthase kinase-3 and stimulation of
T-cell factor signaling following activation of EP2 and EP4
prostanoid receptors by prostaglandin E2. J Biol Chem.
277:2614–2619. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Liu XH, Kirschenbaum A, Lu M, et al:
Prostaglandin E (2) stimulates prostatic intraepithelial neoplasia
cell growth through activation of the interleukin-6/GP130/STAT-3
signaling pathway. Biochem Biophys Res Commun. 290:249–255. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Eibl G, Bruemmer D, Okada Y, et al: PGE
(2) is generated by specific COX-2 activity and increases VEGF
production in COX-2-expressing human pancreatic cancer cells.
Biochem Biophys Res Commun. 306:887–897. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Ito H, Duxbury M, Benoit E, et al:
Prostaglandin E2 enhances pancreatic cancer invasiveness through an
Ets-1-dependent induction of matrix metalloproteinase-2. Cancer
Res. 64:7439–7446. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Juuti A, Louhimo J, Nordling S, et al:
Cyclooxygenase-2 expression correlates with poor prognosis in
pancreatic cancer. J Clin Pathol. 59:382–386. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Xu L, Li YM, Yu CH, et al: Expression of
p53, p16 and COX-2 in pancreatic cancer with tissue microarray.
Hepatobiliary Pancreat Dis Int. 5:138–142. 2006.PubMed/NCBI
|
|
12
|
Tucker ON, Dannenberg AJ, Yang EK, et al:
Cyclooxygenase-2 expression is upregulated in human pancreatic
cancer. Cancer Res. 59:987–990. 1999.PubMed/NCBI
|
|
13
|
Ding XZ, Tong WG and Adrian TE: Blockade
of cyclooxygenase-2 inhibits proliferation and induces apoptosis in
human pancreatic cancer cells. Anticancer Res. 20:2625–2631.
2000.PubMed/NCBI
|
|
14
|
Yao M, Zhou W, Sangha S, et al: Effects of
nonselective cyclooxygenase inhibition with low-dose ibuprofen on
tumor growth, angiogenesis, metastasis, and survival in a mouse
model of colorectal cancer. Clin Cancer Res. 11:1618–1628. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Shaik MS, Chatterjee A and Singh M: Effect
of a selective Cyclooxygenase-2 inhibitor, nimesulide, on the
growth of lung tumors and their expression of cyclooxygenase-2 and
peroxisome proliferator-activated receptor-gamma. Clin Cancer Res.
10:1521–1529. 2004. View Article : Google Scholar
|
|
16
|
Chen G and Duan HJ: Therapeutic effect of
different doses of celecoxib on human lung cancer xenograft in nude
mice and its action mechanism. Tumor. 28:837–841. 2008.
|
|
17
|
Bresalier RS, Sandler RS, Quan H, et al:
Cardiovascular events associated with rofecoxib in a colorectal
adenoma chemoprevention trial. N Engl J Med. 352:1092–1102. 2005.
View Article : Google Scholar : PubMed/NCBI
|