Introduction
Glioma is the most common primary brain tumor in
adults and the median survival for patients without therapy is up
to 3 months (1). Currently the
traditional treatment such as resection, chemotherapy and
radiotherapy has not greatly improved the median survival for
patients. Malignant glioma is notorious for its behavior of rapid
proliferation, great invasion and metastasis and it is also among
the best vascularized tumors in humans. It is well known that
angiogenesis plays a critical role in tumor progression. Therefore
new anti-angiogenic treatment strategies are required (2–6).
Revealing of various signaling pathways that lead to activation of
angiogenesis will provide molecular insight into developing
therapeutic agents to treat glioma. In the past decades,
researchers have discovered many molecular mechanisms related to
tumor angiogenesis (7–9).
Traditional rodent cancer models have disadvantages,
e.g., the high cost, long developmental phase, and limited
availability for high-throughput assays (10), which limited effective ways to find
the new molecular mechanisms and monitor the tumor in vivo
in real time.
The teleost zebrafish (Danio rerio), a promising
alternative vertebrate cancer model has attracted considerable
attention in recent years because of compelling advantages of
transparency of the embryo, rapid development, fecundity, tractable
genetics, great screening efficiency, high levels of physiologic
and genetic homology with higher vertebrates (11–13).
Moreover, the zebrafish embryo within 1 week is immune-free, and
showed no immunosuppression to transplanted human glioma cells
(14). The transgenic zebrafish
strains, e.g., casper transparent mutant line (15), and VEGFR2:G-RCFP line with green
fluorescence specifically in blood vessels (16), offered new tools for cancer
research.
In this study, we microinjected human U87 glioma
cells into zebrafish embryos and developed a xenograft zebrafish
glioma model. The U87 cells are first labeled with a red
fluorescence protein and then microinjected into perivitelline
space of VEGFR2:G-RCFP transgenic zebrafish embryos at 48 h
post-fertilization. Then in order to investigate the angiogenesis
mechanisms in details, we make use of the staining of endogenous
alkaline phosphatase, fluorescent microscopy monitoring, in
situ hybridization and quantitive RT-PCR to investigate the
change of glioma related molecules in a zebrafish embryo
angiogenesis glioma model.
Materials and methods
Glioma cell culture and transfection
The human malignant glioma cell line U87 (American
Type Culture Collection) was maintained in 90% DMEM containing 100
U/ml penicillin, 100 μg/ml streptomycin (Gibco, NY, USA) and 10%
fetal bovine serum (Invitrogen, CA, USA) with 5% CO2 at
37°C. Plasmid DNA labeled with red fluorescent protein DNA fragment
(pcDNA3.0-DsRed) was provided by Dr Ben-Ping Luo and is amplified
using Endo-Free Plasmid Mini kit I (Omega, USA). One day before
transfection, we plated 0.5–2×105 U87 cells in 500 μl of
growth medium without antibiotics in each well of a 24-well plate
so that cells are 90–95% confluent at the time of transfection.
Then these cells were transfected with pcDNA3.0-DsRed vector using
Lipofectamine 2000 (Invitrogen) as per the instructions with slight
modification. The U87 glioma cells expressing red fluorescent
protein (U87-DsRed glioma cells) were selected using 300 μg/ml G418
and cultured for future use.
Zebrafish incubation
The adult AB zebrafish and VEGFR2:G-RCFP (provided
by integrated laboratory of Sun Yat-Sen University) transgenic
zebrafish were maintained in zebrafish breeding system as
previously described (17). The
zebrafish embryos were incubated in petri dishes with E3 buffer (5
mM NaCl, 0.17 mM KCl, 0.33 mM CaCl2, 0.33 mM
MgSO4) at 28°C until they were ready for use. If the
zebrafish embryos are used after 48 hpf, it should be placed in E3
buffer with 1-phenyl-2-thiourea (PTU) (Sigma-Alrich, USA) at 24 hpf
and the buffer should be changed every day in order to eliminate
melanin. The embryos containing chorion should be dechorioned with
1 mg/ml pronase (Sigma-Alrich) and then anesthetized with 0.04
mg/ml tricaine (Sigma-Alrich) before they are microinjected with
cells( 18).
The establishment of zebrafish embryo
glioma model
Firstly, the prepared U87-DsRed glioma cells were
trypsinized and suspended in Matrigel (R&D, USA) at a density
of ~108 cells/ml. Then Narishige microinjector was used
to inject 10–30 nl suspended U87-DsRed cells into the perivitelline
space near SIV of the VEGFR2:G-RCFP transgenic zebrafish embryos at
48 hpf (18,19) to establish this zebrafish embryo
glioma model. The blank matrigel injected zebrafish embryos were
used as a negative control.
Fluorescent microscopy monitoring
When the zebrafish embryo glioma model was set up a
Nikon epifluorescent microscope was used to monitor the progression
of glioma cells and the subintestinal vessel (SIV) changes of
transgenic VEGFR2-RCFP zebrafish in real time continually for 2
days.
Whole-mount endogenous alkaline
phosphatase staining
In order to further investigate the angiogenesis
ability of glioma cells, we fixed the U87 injected zebrafish
embryos at 1 day post-injection (dpi) and 2 dpi in phosphate
buffered saline (PBS)-4% paraformaldehyde (PFA) for 2 h at room
temperature and then stained for endogenous alkaline phosphatase
activity following the protocol of Serbedzija et al(20). Then embryos were observed and
photographed under a Nikon stereomicroscope.
Whole-mount in situ hybridization
We further investigated the molecular mechanisms of
glioma angiogenesis by in situ hybridization test of
zebrafish VEGF A and VEGFR2/KDR mRNA expression in zebrafish embryo
glioma model using VEGF A and KDR digoxigenin-labeled antisense RNA
probes. At first, we cloned part of the zebrafish VEGF and KDR
sequence which were ~1 kb into the pcDNA3.0 plasmid. The primers
were as follows: VEGF (forward:
5′-ttggaattcagcgactcaccgcaacactc-3′, reverse:
5′-ataaagcttcattcgttgttccgctcctg-3′); KDR (forward:
5′-gcagaattcattcccatgccgaacattac-3′, reverse: 5′-gttaagcttagtctg
aggcgatcttgagg-3′). Then by using the DIG System nucleic acid
labeling kit (Roche, USA), we prepared these two probes with T7
promoter in vitro and then observed the signals by using an
NBT/BCIP staining solution (Roche). The details of the
high-resolution in situ hybridization protocol have been
reported (21). We used a blank
Matrigel injected zebrafish as a negative control for zebrafish
angiogenesis glioma model in whole-mount in situ
hybridization. The experiments were repeated 3 times.
Quantitive RT-PCR analysis
To determine the exact change of zebrafish VEGF A
and VEGFR2 mRNA expression in zebrafish embryo glioma model we also
carried out quantitive RT-PCR tests of zebrafish VEGF and VEGFR2
mRNA expression. The qRT-PCR primers were as follows: VEGF
(forward: 5′-tgctcctgcaaattcacacaa-3′, reverse:
5′-atcttggcttttcacatctgcaa-3′); KDR (forward:
5′-tggagttccagcacccttta-3′, reverse: 5′-cgtccttcttcaccctttca-3′);
β-actin (forward: 5′-cgtgacatcaa ggagaagct-3′, reverse:
5′-tcgtggataccgcaagattc-3′). Total RNA was extracted from 40 glioma
angiogenesis zebrafish embryos and then treated with DNase I.
Promega reverse transcription kit was used to get the zebrafish
single-stranded cDNA. Then we used the SYBR GreenER™ qPCR SuperMix
Universal kits (Applied Biosystems Inc.) to carry out quantitive
PCR in Applied Biosystems 7500 Real-Time PCR system (Applied
Biosystems Inc.) according to the manufacturer’s instructions.
Data were normalized with zebrafish β-actin and
repeated 3 times with similar results. Ct-value for each sample was
calculated with the ΔΔCt-method (22) and results were expressed as
2−ΔΔCT. A blank Matrigel injected zebrafish was used as
a negative control.
Results
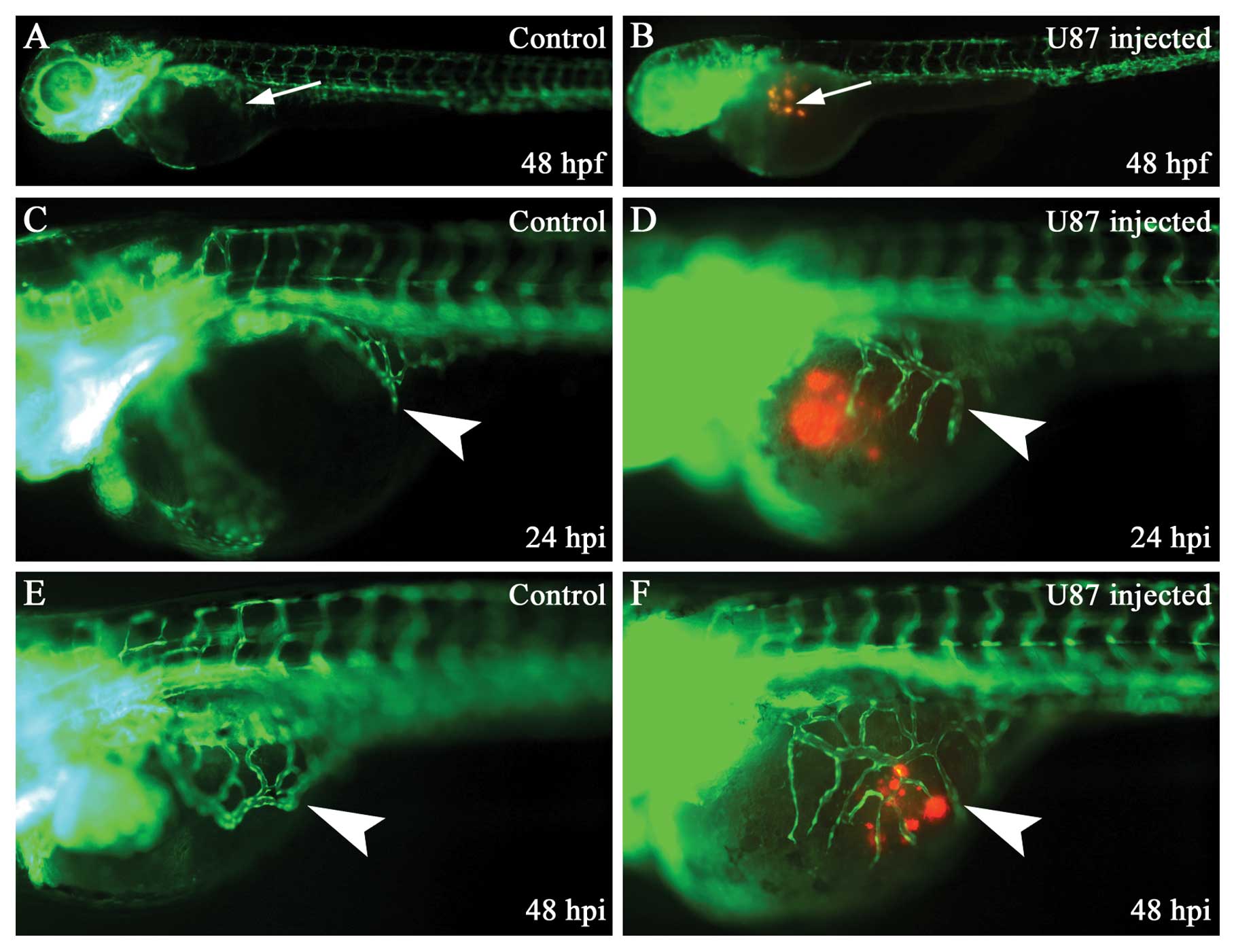
Human glioma cells U87 can survive,
proliferate and induce angiogenesis in zebrafish embryos
We established the zebrafish embryo glioma model and
we monitored the activity of U87 cells and changes of SIV in
zebrafish embryo glioma model by the use of epifluorescent
microscopy. The human U87 glioma cells can survive, proliferate and
induce angiogenesis in zebrafish embryos from 24 to 48 hpi
(Fig. 1). The angiogenesis ability
increased from 1 to 2 dpi (Fig. 1D and
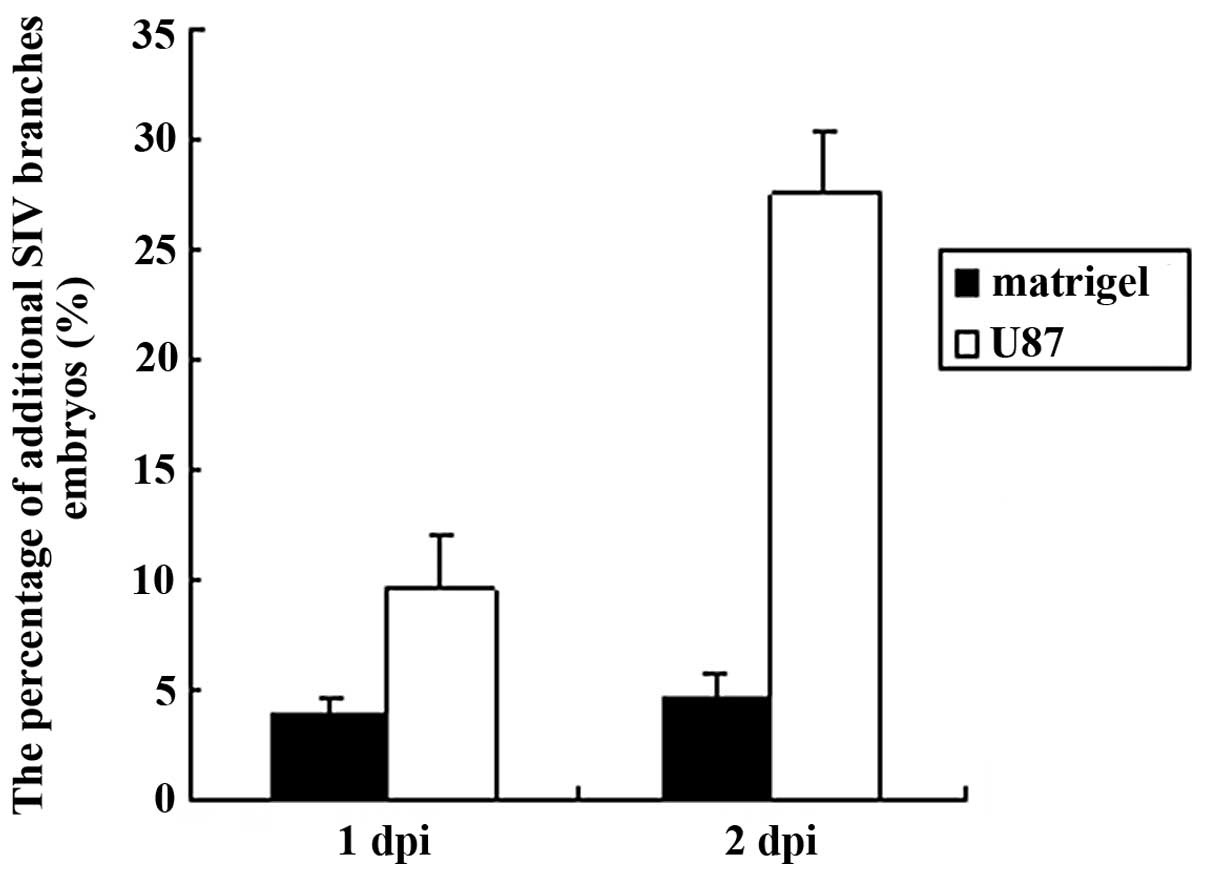
F). We calculated the percentage of additional SIV branches in
the zebrafish embryo and also found that the percentage of positive
SIV phenotype increased from 1 to 2 dpi (Fig. 2).
Endogenous alkaline phosphatase staining for
zebrafish embryo glioma model showed similar results on additional
SIV branches to that observed by the epifluorescent microscope
(Fig. 3B and D).
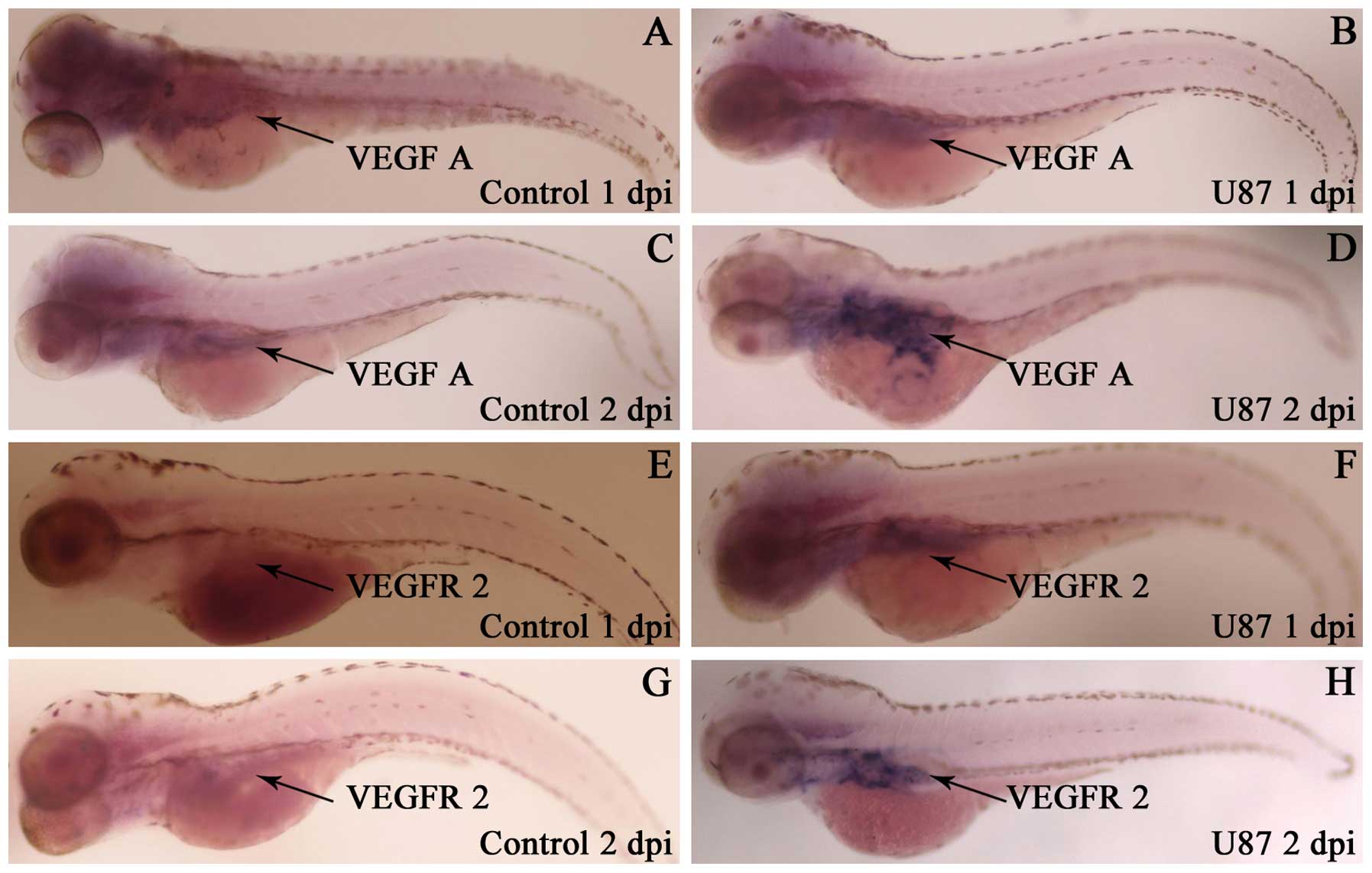
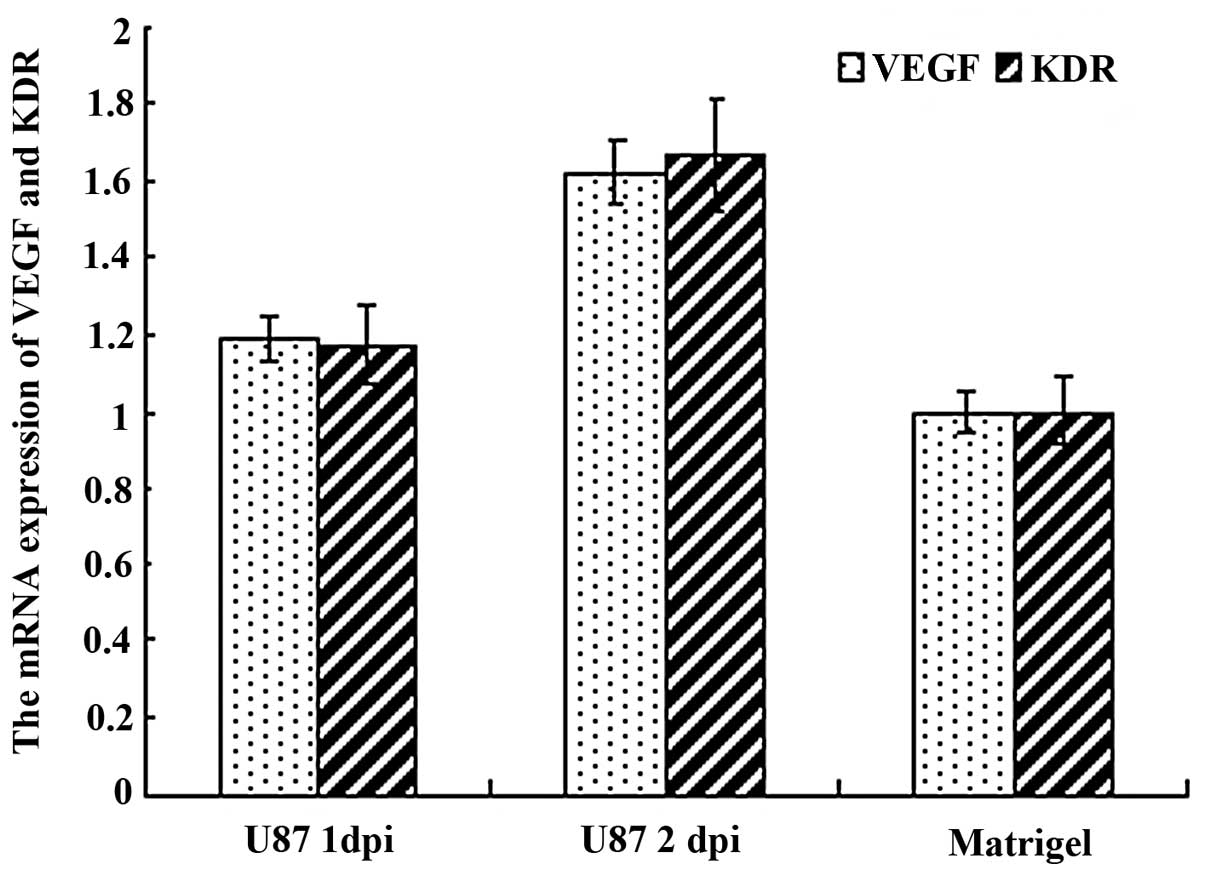
Human glioma cells U87 can induce the
ectopic zebrafish VEGF and VEGFR2 mRNA expression and increase
their expression quantity
By in situ hybridization and quantitive
RT-PCR test of zebrafish VEGF and VEGFR2 mRNA expression in
zebrafish embryo glioma model from 1 to 2 dpi, we found that the
expression area of VEGF and VEGFR2 mRNA around SIV had enlarged
(Fig. 4) and their expression was
also increased (Fig. 5).
Discussion
The zebrafish glioma model provides a convenient
in vivo system for the study of cancer cell molecular
mechanisms. Many other kinds of zebrafish cancer models have
already appeared worldwide. These models mainly include three
different types. The first type is that Spitsbergen et
al(23,24) gained by chemical induction and
acquiring several types of cancers in zebrafish through chemical
carcinogenesis such as epithelial, mesenchymal, neural neoplasia
induced by 7,12-dimethylbenz[a]anthracene and hepatic, mesenchymal
neoplasia induced by N-methyl-N′-nitro-N-nitrosoguanidine. The
second type of zebrafish cancer model is obtained by genetic
manipulation, bmyb mutation causes genome instability and increased
cancer susceptibility in zebrafish (25). The third transplanted zebrafish
glioma model is that we described herein. Amatruda et al
have also reported a study in Cancer Cell(26). A zebrafish cancer model was also
presented July 2009 in Spoleto, Italy, recapitulating a number of
new zebrafish cancer models (27),
indicating that zebrafish model could make a great contribution to
cancer research.
Glioma is a notorious malignant cancer in humans and
research has been done into its molecular mechanisms in
vitro and in vivo for many years. In this study, we
found that the U87 human glioma cells can survive, proliferate and
induce angiogenesis in a zebrafish embryo. As is known,
angiogenesis is a key factor in malignant tumor progression
(28) and here these transplanted
glioma cells U87 can also induce additional subintestinal vessels
and transfer to the distant site through intravasation and
extravasation.
Vascular endothelial growth factors (VEGFs) play an
important part in angiogenesis (29,30)
and currently include VEGF-A,-B,-C,-D,-E and placenta growth factor
(PlGF) in its family. They play a crucial role in the process of
angiogenesis by binding tyrosine kinase receptors such as VEGF
receptor-1,-2, and-3 (31). We
carried out an investigation on VEGF A and its receptor VEGFR2/KDR
(32,33), we found that the transplanted human
U87 glioma cells can not only increase the mRNA expression of
zebrafish VEGF A and VEGFR2 but also induce the secretion of VEGF A
and VEGFR2 mRNA in different anatomic sites in zebrafish, and these
molecules contribute to induction of additional zebrafish SIV
branches. This result is consistent with previous published
reports. Serbedzija et al have reported that VEGF can induce
angiogenesis by the injection of its protein directly into
zebrafish embryo (20) and Habeck
et al have also shown a very similar result to ours by
injecting VEGF plasmid into zebrafish embryo (34). The above evidence proves that VEGF
indeed plays an important role in zebrafish embryo angiogenesis.
The injected human glioma cells U87 induced zebrafish embryo
angiogenesis further confirms that zebrafish angiogenesis-related
genes show great conservation with humans, as Liang et
al(35) previously showed that
zebrafish VEGF’s functional sites.
Acknowledgements
We thank Benping Luo for providing the vector
pcDNA3.0-DsRed. This study was supported by the National Natural
Science Foundation of China (30973479 to Y. Peng), the National
High Technology Research and Development Program of China (863
Program) (2007AA021101 to Y. Peng), and Science and Technology
Planning Project of Guangdong Province, China (2009B060700040 and
2011B031800141 to Y. Peng).
Abbreviations:
|
hpf
|
hour post-fertilization
|
|
dpf
|
day post-fertilization
|
|
hpi
|
hour post-injection
|
|
dpi
|
day post-injection
|
|
DMEM
|
Dulbecco’s modified Eagle’s medium
|
|
SIV
|
subintestinal vessel
|
|
PTU
|
1-phenyl-2-thiourea
|
|
PBS
|
phosphate buffered saline
|
|
PFA
|
paraformaldehyde
|
|
qPCR
|
quantitative real-time PCR
|
References
|
1
|
Ng SS, Gao Y, Chau DH, et al: A novel
glioblastoma cancer gene therapy using AAV-mediated long-term
expression of human TERT C-terminal polypeptide. Cancer Gene Ther.
14:561–572. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Lin ZX, Yang LJ, Huang Q, et al:
Inhibition of tumor-induced edema by antisense VEGF is mediated by
suppressive vesiculo-vacuolar organelles (VVO) formation. Cancer
Sci. 99:2540–2546. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Paez-Ribes M, Allen E, Hudock J, et al:
Antiangiogenic therapy elicits malignant progression of tumors to
increased local invasion and distant metastasis. Cancer Cell.
15:220–231. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Norden AD, Drappatz J and Wen PY: Novel
anti-angiogenic therapies for malignant gliomas. Lancet Neurol.
7:1152–1160. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Vredenburgh JJ, Desjardins A, Herndon JN,
et al: Bevacizumab plus irinotecan in recurrent glioblastoma
multiforme. J Clin Oncol. 25:4722–4729. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Tuettenberg J, Friedel C and Vajkoczy P:
Angiogenesis in malignant glioma- a target for antitumor therapy?
Crit Rev Oncol Hematol. 59:181–193. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Gan HK, Lappas M, Cao DX, Cvrljevdic A,
Scott AM and Johns TG: Targeting a unique EGFR epitope with
monoclonal antibody 806 activates NF-kappaB and initiates tumour
vascular normalization. J Cell Mol Med. 13:3993–4001. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Jang FF, Wei W and De WM: Vascular
endothelial growth factor and basic fibroblast growth factor
expression positively correlates with angiogenesis and peritumoural
brain oedema in astrocytoma. J Ayub Med Coll Abbottabad.
20:105–109. 2008.
|
|
9
|
di Tomaso E, London N, Fuja D, et al:
PDGF-C induces maturation of blood vessels in a model of
glioblastoma and attenuates the response to anti-VEGF treatment.
PLoS One. 4:e51232009.PubMed/NCBI
|
|
10
|
Huse JT and Holland EC: Genetically
engineered mouse models of brain cancer and the promise of
preclinical testing. Brain Pathol. 19:132–143. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Lee LM, Seftor EA, Bonde G, Cornell RA and
Hendrix MJ: The fate of human malignant melanoma cells transplanted
into zebrafish embryos: assessment of migration and cell division
in the absence of tumor formation. Dev Dyn. 233:1560–1570. 2005.
View Article : Google Scholar
|
|
12
|
Stoletov K, Montel V, Lester RD, Gonias SL
and Klemke R: High-resolution imaging of the dynamic tumor cell
vascular interface in transparent zebrafish. Proc Natl Acad Sci
USA. 104:17406–17411. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Jesuthasan S: Genetics and development.
Zebrafish in the spotlight. Science. 297:1484–1485. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Stoletov K and Klemke R: Catch of the day:
zebrafish as a human cancer model. Oncogene. 27:4509–4520. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
White RM, Sessa A, Burke C, et al:
Transparent adult zebrafish as a tool for in vivo transplantation
analysis. Cell Stem Cell. 2:183–189. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Cross LM, Cook MA, Lin S, Chen JN and
Rubinstein AL: Rapid analysis of angiogenesis drugs in a live
fluorescent zebrafish assay. Arterioscler Thromb Vasc Biol.
23:911–912. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Westerfield M: The Zebrafish Book. A Guide
for the Laboratory Use of Zebrafish (Danio rerio) Eugene, OR:
University of Oregon Press; 2007
|
|
18
|
Nicoli S, Ribatti D, Cotelli F and Presta
M: Mammalian tumor xenografts induce neovascularization in
zebrafish embryos. Cancer Res. 67:2927–2931. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Nicoli S and Presta M: The zebrafish/tumor
xenograft angiogenesis assay. Nat Protoc. 2:2918–2923. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Serbedzija GN, Flynn E and Willett CE:
Zebrafish angiogenesis: a new model for drug screening.
Angiogenesis. 3:353–359. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Thisse C and Thisse B: High-resolution in
situ hybridization to whole-mount zebrafish embryos. Nat Protoc.
3:59–69. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Adhikary S and Eilers M: Transcriptional
regulation and transformation by Myc proteins. Nat Rev Mol Cell
Biol. 6:635–645. 2005. View
Article : Google Scholar : PubMed/NCBI
|
|
23
|
Spitsbergen JM, Tsai HW, Reddy A, et al:
Neoplasia in zebrafish (danio rerio) treated with
7,12-dimethylbenz[a]anthracene by two exposure routes at different
developmental stages. Toxicol Pathol. 28:705–715. 2000.PubMed/NCBI
|
|
24
|
Spitsbergen JM, Tsai HW, Reddy A, et al:
Neoplasia in zebrafish (Danio rerio) treated with
N-methyl-N′-nitro-N-nitrosoguanidine by three exposure routes at
different developmental stages. Toxicol Pathol. 28:716–725.
2000.PubMed/NCBI
|
|
25
|
Shepard JL, Amatruda JF, Stern HM, et al:
A zebrafish bmyb mutation causes genome instability and
increased cancer susceptibility. Proc Natl Acad Sci USA.
102:13194–13199. 2005.
|
|
26
|
Amatruda JF, Shepard JL, Stern HM and Zon
LI: Zebrafish as a cancer model system. Cancer Cell. 1:229–231.
2002. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Mione MC and Trede NS: The zebrafish as a
model for cancer. Dis Model Mech. 3:517–523. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Chi A, Norden AD and Wen PY: Inhibition of
angiogenesis and invasion in malignant gliomas. Expert Rev
Anticancer Ther. 7:1537–1560. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Leung DW, Cachianes G, Kuang WJ, Goeddel
DV and Ferrara N: Vascular endothelial growth factor is a secreted
angiogenic mitogen. Science. 246:1306–1309. 1989. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Plate KH, Breier G, Weich HA and Risau W:
Vascular endothelial growth factor is a potential tumour
angiogenesis factor in human gliomas in vivo. Nature. 359:845–848.
1992. View
Article : Google Scholar : PubMed/NCBI
|
|
31
|
Roy H, Bhardwaj S and Yla-Herttuala S:
Biology of vascular endothelial growth factors. FEBS Lett.
580:2879–2887. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Neufeld G, Tessler S, Gitay-Goren H, Cohen
T and Levi BZ: Vascular endothelial growth factor and its
receptors. Prog Growth Factor Res. 5:89–97. 1994. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Neufeld G, Cohen T, Gengrinovitch S and
Poltorak Z: Vascular endothelial growth factor (VEGF) and its
receptors. FASEB J. 13:9–22. 1999.PubMed/NCBI
|
|
34
|
Habeck H, Odenthal J, Walderich B,
Maischein H and Schulte-Merker S: Analysis of a zebrafish VEGF
receptor mutant reveals specific disruption of angiogenesis. Curr
Biol. 12:1405–1412. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Liang D, Xu X, Chin AJ, et al: Cloning and
characterization of vascular endothelial growth factor (VEGF) from
zebrafish, Danio rerio. Biochim Biophys Acta. 1397:14–20. 1998.
View Article : Google Scholar : PubMed/NCBI
|