Introduction
Metastatic and chemoresistant melanoma remains
intractable and very difficult to treat. Based on the remarkable
antitumor efficacy of dendritic cell (DC)-based vaccines in animal
experiments (1,2), clinical trials of a DC-based
immunotherapeutic approach have been conducted against mainly human
leukocyte antigen (HLA)-A2+ advanced melanomas in
Western counties (3–9). Since Nestle et al(3) first reported the efficacy of a DC
vaccine against metastatic melanoma in a clinical trial, DC
vaccines have become one of the main investigational therapeutic
approaches against solid tumors. With regard to metastatic
melanoma, it can be summarized that DC vaccine showed good safety
and a low clinical response, but did not indicate a clear overall
survival benefit (10–12). Moderate achievements were obtained
in a phase I–II study, however unfortunately government-approved
melanoma vaccines have yet to be developed because of the shortage
of enrolled cases and a lack of double-blind, randomized
(placebo-controlled) phase III trials.
Since sipuleucel-T (Provenge, Dendreon), an
autologous cellular immunotherapy, was approved by the USA Food and
Drug Administration (FDA), a significant benefit of DC-based
vaccines on overall survival in metastatic castration-resistant
prostate cancer patients had attracted much attention despite a low
rate of clinical response (13–15).
Like previous DC vaccine studies, in early phase trials,
sipuleucel-T showed high safety, but a weak antitumor response
which was not impressive compared with chemotherapeutic regimens.
However, the last double-blind, placebo-controlled, multicenter
phase III trial of the sipuleucel-T vaccine clearly demonstrated a
significant survival benefit for metastatic prostate cancer.
Previously, we reported a phase I clinical trial of
a DC-based vaccine against HLA-A24+ metastatic melanoma,
and obtained several clinical responders (16). Based on these promising results, we
have performed a phase II trial enrolling 24 metastatic melanoma
patients. We summarize the results of a phase II non-randomized
clinical trial and analyze various prognostic factors linked to an
increase in overall survival.
Materials and methods
Patients and study design
Twenty-four patients with metastatic melanomas were
enrolled in a phase II clinical trial of a peptide cocktail-pulsed
DC-based vaccine from 2004 to 2010 approved by the Institutional
Review Board (IRB) of Shizuoka Cancer Center, Japan. All patients
gave written informed consent. Eligibility and exclusion criteria
were similar to those for the previous phase I trial (16). The patients received the vaccine
subcutaneously (s.c.) every week for 4 weeks, then once 2 weeks
later and every month for 5 months. DCs were injected in the dose
range of 1–5×107/body/shot. Clinical response was rated
as maximal through the DC vaccinations. The patients received up to
10 injections on the condition that one or more measurable lesion
showed at lease a stable disease (SD) response and/or that an
ELISPOT assay performed after 4 injections indicated a positive
response against >1 melanoma-associated peptide. Adverse effects
were evaluated according to the NCI Common toxicity criteria.
Measurable lesions and clinical responses were evaluated by RECIST
(17).
With regard to overall survival, as a retrospective
control, survival data from 37 patients with metastatic melanoma
given best supportive care without a DC vaccine from 2004 to 2008
were utilized with approval by the IRB of Shizuoka Cancer
Center.
Preparation of the DC vaccine
The methods used to produce the DC vaccine were
described previously (16).
Briefly, monocyte-enriched fractions were separated from
leukapheresis products using OptiPrep™, and cultured in the
presence of granulocyte macrophage-colony-stimulating factor
(GM-CSF) and interleukin (IL)-4 in X-VIVO15 serum-free medium.
After 7 days, harvested cells were pulsed with a cocktail of 5
melanoma-specific synthetic peptides restricted to HLA-A2 or A24
and keyhole limpet hemocyanin (KLH). DC-enriched cells were washed
and cryopreserved in a Cryocyte bag until used. The following
peptides restricted to HLA-A2 or A24 were synthesized according to
good manufacturing practice (GMP) standards by Multiple Peptide
Systems, CA: HLA-A2: MART-1, gp100, tyrosinase, MAGE-A2, and
MAGE-A3 and HLA-A24: gp100, tyrosinase, MAGE-A1, MAGE-A2 and
MAGE-A3.
Characterization of tumor specimens using
RT-PCR and immunohistochemistry
Specimens of primary tumors or skin metastatic
lesions were obtained from 12 patients in a phase II study. The
expression of melanoma tumor antigens was investigated using
RT-PCR. HLA-class I protein expression and the phenotypes of
lymphocytes infiltrating the tumor site were examined using
immunohistochemistry (IHC). The monoclonal antibodies (mAbs)
against human HLA-class I (Hokudo Co., Ltd., Sapporo, Japan), CD8
(Thermo Scientific, Flemont, CA), Foxp3 (Abcam, Cambridge, MA) and
IL-17 (Abcam) were all purchased commercially.
Immunological monitoring
The ELISPOT assay, intracellular cytokine staining
using anti-human interferon (IFN)-λ and anti-IL-4 antibodies and
delayed-type hypersensitivity (DTH) skin tests were described
previously (16). Briefly, the
HLA-A2 or A24 peptide cocktail solution diluted to a dose of 5
μg/ml and KLH were injected intradermally into the forearm for DTH
measurements after 48 h.
Serum autoantibody against melanoma
antigens
The ELISA for detecting human antibodies and the
control reaction system were described previously (18). MAGE antigen proteins with a
glutathione S-transferase (GST) tag were all purchased from Abnova
Corp., Taipei, Taiwan. Recombinant MAGE-A1 and -A2 were full-length
constructs and MAGE-A3 was a fragment, comprising amino acids
1–135. Briefly, recombinant human MAGE antigens were added to a
96-well microplate, and sequentially diluted rabbit anti-human IgG
antibody was added. After blocking with 3% bovine serum albumin
(BSA), diluted patient serum (before or after vaccine) or human IgG
at 100 μg/ml was added to antigen-coated wells or anti-human
IgG-coated wells, respectively. After incubation, sheep
horse-HRP-conjugated anti-human IgG antibody, then substrate
solution was added and the absorbance at 450 nm was read. A serum
antibody index was calculated using the control calibration curve
as follows; index = [capture antibody dose (μg/ml)/control OD ×
sample OD/dilution ratio of sera].
Statistical analysis
The overall survival of metastatic melanoma patients
was examined by comparing differences in mean survival time (MST)
via the Kaplan-Meier method. A comparative analysis of survival
times between groups was then performed using the log-rank test.
Values of P<0.05 were considered statistically significant.
Results
Patient characteristics
Patient details are summarized in Table I. Most of the patients were in good
performance status (PS), were HLA-A24+ and had received
prior chemotherapy. As to the number of target lesions, half of the
patients had >2 metastatic lesions, the average being 1.7±0.7
(Table II).
 | Table ICharacteristics of metastatic
melanoma patients in the phase II study. |
Table I
Characteristics of metastatic
melanoma patients in the phase II study.
| Total no.
enrolled | 24 |
| Age | 58.2±12.2 |
| Gender |
| M | 12 |
| F | 12 |
| Performance
status |
| PS0 | 22 |
| PS1 | 2 |
| HLA-typing |
| A2 | 3 |
| A24 | 21 |
| Previous
therapy |
| ST | 1 |
| CT | 3 |
| ST+CT | 19 |
| ST+CT+RT | 1 |
 | Table IIPhase II study of DC-based therapy
against melanoma. |
Table II
Phase II study of DC-based therapy
against melanoma.
| | | | | | DTH | |
|---|
| | | | | |
| |
|---|
| Case | Age | Gender | Measurable
lesions | DC no. (times) | Side effects | DC | KLH | Response
(ST)a |
|---|
| MEL-1 | 53 | F | Liver |
4.2×107(10) | Fever (II) | + | + | SD (7.5) |
| MEL-2 | 50 | F | Lung |
5.2×107(10) | ND | + | + | PD (7) |
| MEL-3 | 46 | F | Lung |
0.5×107(11) | ND | + | + | SD (8.5) |
| MEL-4 | 62 | M | Lung, liver,
LN |
7.3×107(8) | Hepatic (I) | − | + | PD (6) |
| MEL-5 | 63 | M | Lung, liver |
3.1×107(8) | Hepatic (II) | − | − | PD (6) |
| MEL-6 | 45 | F | Lung |
0.7×107(10) | Leucopenia
(II) | + | + | SD (30) |
| MEL-7 | 57 | F | Lung |
0.3×107(10) | ND | − | + | PD (12) |
| MEL-8 | 68 | M | Skin |
0.6×107(14) | ND | + | + | PR (12) |
| MEL-9 | 56 | M | Lung, LN | NE (15) | Hepatic (I) | + | + | SD (32) |
| MEL-10 | 73 | F | Lung, liver | NE (5) | ND | − | − | PD (3.5) |
| MEL-11 | 68 | F | Liver, LN | NE (6) | ND | − | − | PD (5.5) |
| MEL-12 | 53 | M | Skin, LN | NE (6) | Hepatic (II) | − | + | PD (9) |
| MEL-13 | 63 | F | Liver, LN |
0.6×107(10) | ND | − | − | PD (12) |
| MEL-14 | 73 | F | Lung |
1.5×107(3) | ND | ND | ND | PD (1.5) |
| MEL-15 | 32 | M | Lung, LN |
1.0×107(10) | ND | − | + | PD (7) |
| MEL-16 | 62 | F | Lung |
1.1×107(8) | ND | − | − | PD (6) |
| MEL-17 | 60 | M | Lung, LN |
1.2×107(12) | Fever (I) | − | + | PD (10) |
| MEL-18 | 82 | F | Nasal cavity |
0.5×107(10) | ND | − | − | PD (9.5) |
| MEL-19 | 63 | M | Lung |
0.9×107(25) | Hepatic (I) | + | + | SD (60) |
| MEL-20 | 40 | M | Lung, bone |
1.7×107(4) | ND | ND | ND | PD (3) |
| MEL-21 | 36 | M | Lung |
1.2×107(20) | ND | + | + | PD (46) |
| MEL-22 | 63 | F | LN |
3.9×107(6) | ND | + | + | PD (7.5) |
| MEL-23 | 70 | M | Gingiva, lung |
1.9×107(20) | ND | − | − | SD (18.5) |
| MEL-24 | 59 | M | Lung, skin |
1.5×107(13) | ND | − | − | PD (12.5) |
DC processing and characterization
The mean number of peripheral blood mononuclear
cells (PBMCs) collected by apheresis in phase II patients was
8.5±2.5×109. The CD14 frequency increased from 19.5±10.6
to 44.7±15.3% after OptiPrep density-gradient centrifugation. The
mean percentage of DCs rated as
lin−CD11c+HLA-DR+ was 38.1±13.3%,
not different from that in the phase I trial (data not shown). The
frequencies of the DC marker including CD83, CD80, CD86, DC sign,
DEC205 and CMRF56, and DC1/DC2 ratio did not differ from the
previous report either (data not shown).
Clinical responses and adverse
effects
Among 24 cases, 1 case of partial remission (PR), 7
cases of SD and 16 cases of progressive disease (PD) were verified
(Table II). The injected mean DC
number was up to 5.0×107/body at a maximum, averaging
2.4×107/body/shot. DC injection times were 10.5 on
average, and 15 cases were completed with 10 injections. No
significant side effects of more than grade III were seen.
Immunological monitoring
Eighteen of 24 cases (75%) showed positive ELISPOT
reactions against all melanoma antigen-related peptides (Table III). Six exhibited reaction
against >3 peptides. As to the Th1/Th2 balance after vaccines,
12 of 19 evaluable cases had a ratio of >1, which indicated a
shift to Th1 development. With regard to skin tests, DTH reactions
against peptide-pulsed DC and KLH were detected in 41 and 64%,
respectively, of vaccinated patients (Table II).
 | Table IIIImmunological monitoring in melanoma
patients (phase II). |
Table III
Immunological monitoring in melanoma
patients (phase II).
| Case | HLA typing | Tumor antigen
expressiona | HLA-class I
expression | DC1/DC2 ratio | ELISPOT | Th1/Th2
balanceb |
|---|
| MEL-1 |
A*2402 | ND | ND | 192 | 1 (MAGE1) | 1.24 |
| MEL-2 |
A*2404 | 5/5 | + to ++ | 7.6 | 0 | NE |
| MEL-3 |
A*2402 | ND | ND | 149 | 3 (MAGE1-3) | 1.3 |
| MEL-4 |
A*2402 | 2/5 | + to ++ | 45.4 | 1 (Tyr) | 0.83 |
| MEL-5 |
A*2420 | ND | ND | 13.8 | 0 | 0.91 |
| MEL-6 |
A*2402 | 2/5 | ++ | 104 | 3 (MAGE1-3) | 1.12 |
| MEL-7 |
A*2402 | 4/5 | ++ | 125 | 3 (MAGE1-3) | 1 |
| MEL-8 |
A*2402 | 4/5 | ++ to +++ | 435 | 2 (MAGE1,2) | NE |
| MEL-9 |
A*0201 | ND | ND | 18.1 | 0 | 1.36 |
| MEL-10 |
A*2402 | 3/5 | + | 103 | 2 (MAGE1,2) | 1.25 |
| MEL-11 |
A*2402 | 3/5 | + | 24.1 | 2 (MAGE1,2) | 0.79 |
| MEL-12 |
A*2402 | ND | ++ to +++ | 35.0 | 1 (MAGE3) | 1.17 |
| MEL-13 |
A*2402 | ND | ND | 78.0 | 0 | 0.67 |
| MEL-14 |
A*2402 | 5/5 | + | 8.8 | 0 | ND |
| MEL-15 |
A*2402 | 3/5 | ++ | 7.2 | 4
(MAGE1-3,Tyr) | 0.46 |
| MEL-16 |
A*0201 | ND | ND | 10.4 | 1 (MAGE2) | 1.25 |
| MEL-17 |
A*0201 | 5/5 | + | 34.0 | 2
(gp100,MAGE2) | 1.01 |
| MEL-18 |
A*2402 | ND | ND | 28.6 | 3 (MAGE1-3) | 1.45 |
| MEL-19 |
A*2402 | ND | ND | 12.6 | 3 (MAGE1-3) | 1.69 |
| MEL-20 |
A*2402 | ND | ND | 73.8 | ND | ND |
| MEL-21 |
A*2402 | ND | ND | 127 | 2 (MAGE1,2) | 0.36 |
| MEL-22 |
A*2402 | 4/5 | + | 16.3 | 1 (MAGE3) | ND |
| MEL-23 |
A*2402 | ND | ND | 2.2 | 2 (Tyr,MAGE3) | 0.81 |
| MEL-24 |
A*2402 | 2/5 | + | 6.3 | 1 (MAGE3) | 1.81 |
Serum antibodies against melanoma
antigens
Prior to DC vaccines, positive cases of
anti-MAGE-A1, -A2, -A3 and tyrosinase antibodies numbered 15, 10, 1
and 9, respectively among 31 evaluable cases from the phase I and
II trials. The positive rate of anti-melanoma antigens antibody was
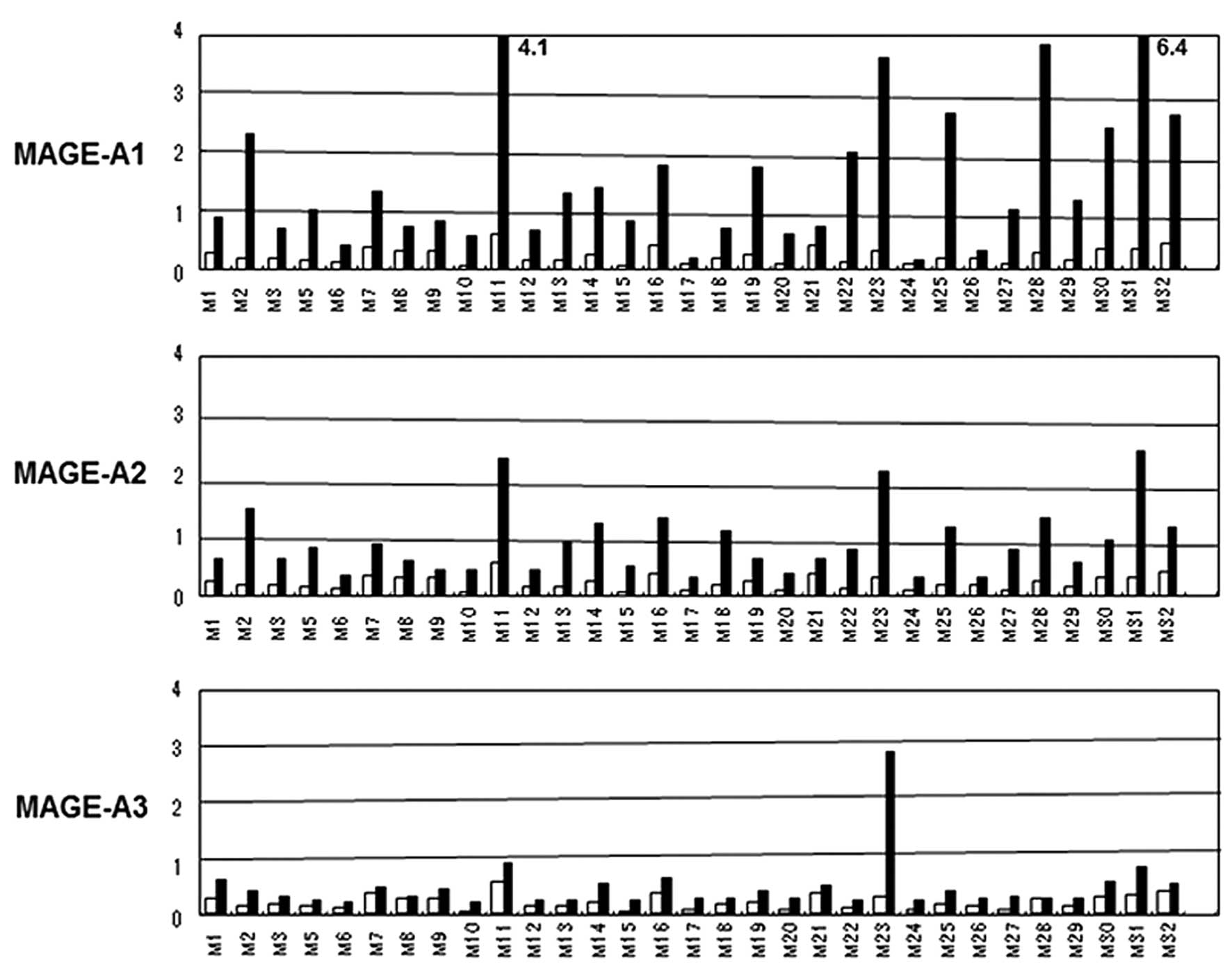
54.8% (Fig. 1, Table IV). On the other hand, the positive
rate of any antibody after vaccination was not high (40.7%)
compared with before the vaccine. The index ratio means the Ab
index after vaccine/before vaccine.
 | Table IVPositive rate of serum autoantibody
against melanoma antigens. |
Table IV
Positive rate of serum autoantibody
against melanoma antigens.
| Antigens | MAGE-A1 | MAGE-A2 | MAGE-A3 | Tyrosinase | Any antigen |
|---|
| Pre (Index
>1) | 15/31 | 10/31 | 1/31 | 9/27 | 17/31 |
| Post (Index
ratioa >2) | 4/19 | 4/27 | 5/25 | 7/27 | 11/27 |
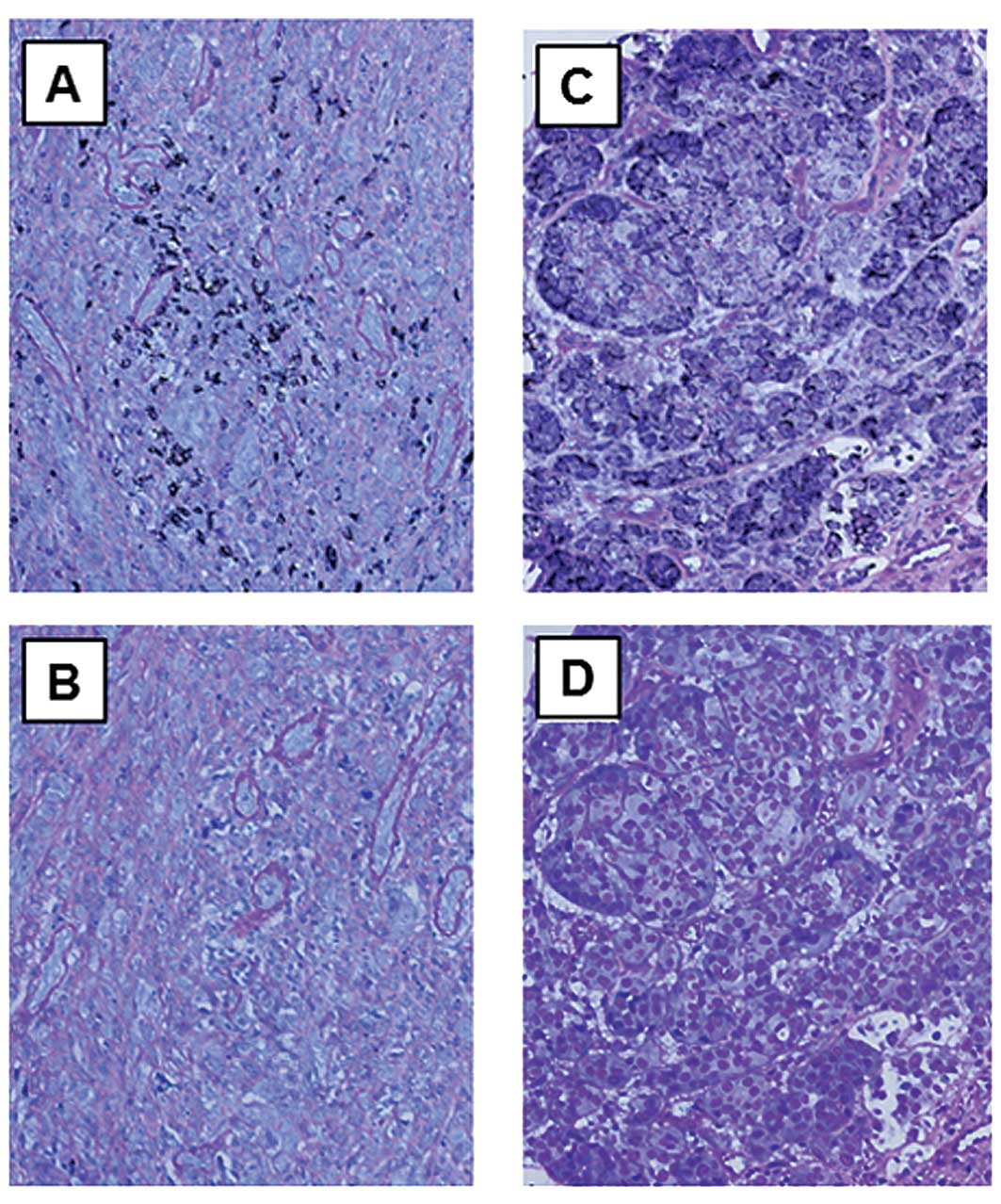
Characterization of tumor tissues
IHC was performed using tumor tissue sections
derived from 22 melanoma patients of phase I and II trials. Fifteen
tumor tissues in which melanoma antigen expression was analyzed
using RT-PCR, showed >2 antigens (Table III, phase I data not shown).
Meanwhile, IHC analysis demonstrated that most (82%) of the 22
evaluable tumor tissue specimens showed HLA-class I expression, and
that CD8 and IL-17 positive staining was seen in 60% and 53%,
respectively (Table V, Fig. 2).
 | Table VImmunohistochemical features of
melanomas: phase I, II study. |
Table V
Immunohistochemical features of
melanomas: phase I, II study.
| Antigens | HLA-class I | CD8 | Foxp3 | IL-17 |
|---|
| Melanoma tumors
(positive %) | 18/22 (82) | 9/15 (60) | 4/15 (27) | 8/15 (53) |
Overall survival analysis
Based on survival data from all metastatic melanoma
patients including these of the phase I and II study, various
clinical, immunological and DC-processing-related parameters were
analyzed in terms of the relationship to the prognosis of melanoma
patients.
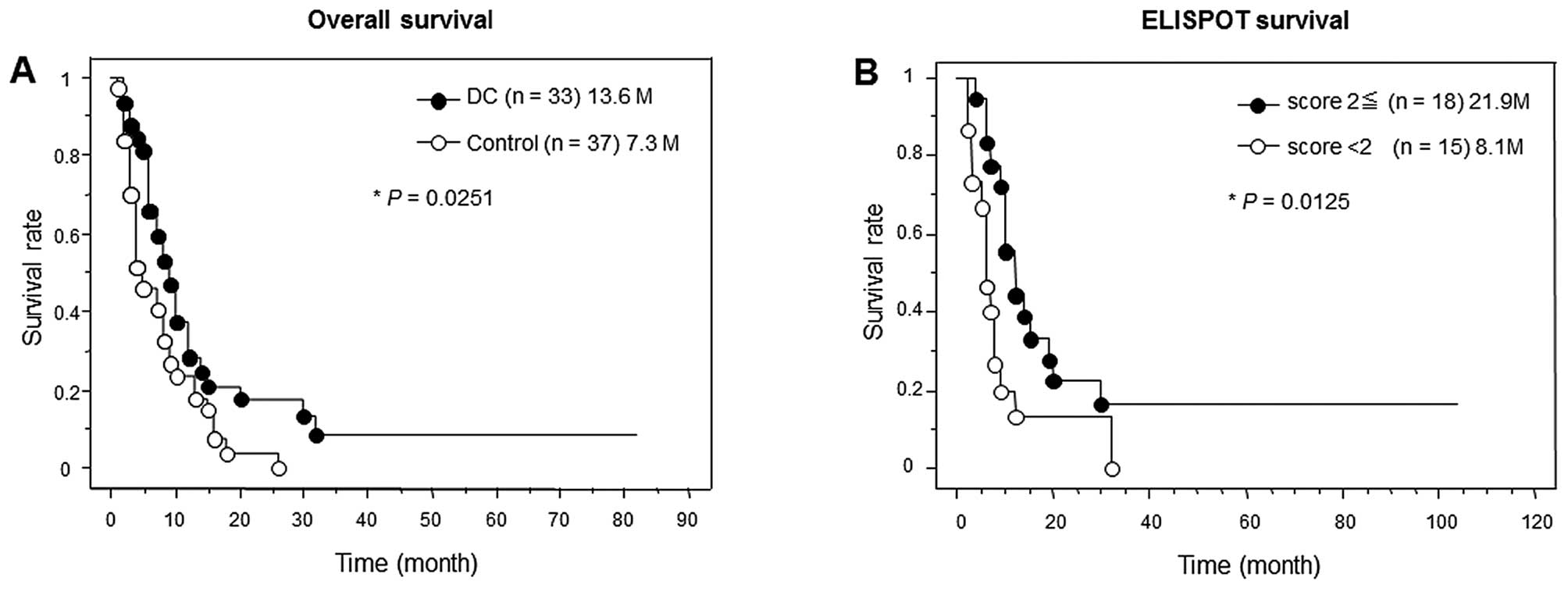
First of all, overall survival analyses were
performed between the vaccinated and non-vaccinated patients, and
between high (≥2) and low (<2) ELISPOT score groups (Fig. 3). The ELISPOT score indicates the
number of peptides with a positive cytotoxic T-cell (CTL) response.
The vaccinated group and ELISPOT high score group demonstrated
significantly longer mean survival times (13.6 M in vaccinate vs.
7.3 M in non-vaccinated; 21.9 M in high ELISPOT score vs. 8.1 M in
low).
Second, DC processing-related parameters including
injected DC numbers, DC ratio, and surface markers such as CD83 and
CCR7 did not demonstrate any relationship to overall survival
(Table VI). Third, various
immunological parameters were analyzed in terms of survival
prolongating effects (Table VII).
The number of target lesions and the number of DC injections showed
a significant correlation with survival time. Interestingly, the
anti-MAGE-A1 antibody titer before the vaccination was shown to be
a good prognostic factor, but anti-melanoma antigen antibody
induction after vaccination was not. Immune response parameters
such as ELISPOT, DTH reaction against peptide and KLH also showed a
significant survival benefit. However, IHC parameters such as CD8
and IL-17 stain levels were not relevant to survival possibly due
to the shortage of case numbers where a tumor specimen was
obtained.
 | Table VIPrognostic factors for melanoma DC
vaccines-1. |
Table VI
Prognostic factors for melanoma DC
vaccines-1.
| Factors | Cases | Mean ± SD | Groups (MST) | Statistical
analysis |
|---|
| DC nos.
(×107) | 29 | 2.4±1.8 | <2 (16.4) vs. ≥2
(15.5) | NS |
| DC ratio (%) | 33 | 38.1±13.3 | <40 (15.9) vs.
≥40 (15.3) | NS |
| DC1/DC2 ratio | 33 | 107±129 | <100 (12.6) vs.
≥100 (20.3) | NS |
| CD40 (%) | 33 | 66.8±18.9 | <70 (15.3) vs.
≥70 (15.9) | NS |
| CD83 (%) | 33 | 25.7±20.8 | <25 (14.5) vs.
≥25 (16.9) | NS |
| CD83+ DC
no. (×106) | 33 | 5.5±6.4 | <5 (15.4) vs. ≥5
(16.8) | NS |
| CCR7 (%) | 33 | 29.1±21.8 | <25 (14.3) vs.
≥25 (17.0) | NS |
 | Table VIIPrognostic factors for melanoma DC
vaccines-2. |
Table VII
Prognostic factors for melanoma DC
vaccines-2.
| Factors | Cases | Mean ± SD | Groups (MST) | Statistical
analysis |
|---|
| No. of target
lesions | 33 | 1.7±0.7 | <2 (24.1) vs. ≥2
(8.6) | 0.029g |
| No. of DC
injections | 33 | 9.8±5.5 | <10 (6.2) vs.
≥10 (24.5) | <0.0001h |
| Anti-melanoma
antigen Aba (Pre-DC) | 31 | 1.6±1.4b | <1 (3.8) vs. ≥1
(22.9) | 0.002h |
| Anti-melanoma
antigen Ab (Post-DC) | 31 | 11/27c (35%) | neg. (16.9) vs.
pos. (18.5) | NS |
| ELISPOT assay | 33 | 1.5±1.1d | <2 (8.1) vs. ≥2
(21.9) | 0.0125g |
| DTH (peptide or
DC) | 31 | 12/31e (39%) | neg. (8.9) vs. pos.
(28.3) | 0.0105g |
| DTH (KLH) | 31 | 18/31f (58%) | neg. (8.2) vs pos.
(22.4) | 0.0244g |
| Th1/Th2
balance | 29 | 1.2±0.6 | ≥1 (12.2) vs. >1
(24.1) | NS |
| CD8+ T
cell in tumors | 16 | 9/16 (56%) | neg. (6.5) vs. pos.
(10.9) | NS |
| IL17 stain in
tumors | 16 | 8/16 (50%) | neg. (10.5) vs.
pos. (7.5) | NS |
Discussion
Many clinical trials of immunotherapy using DC-based
vaccines against metastatic melanoma have been performed, since
Nestle et al(3) reported the
efficacy of a melanoma lysate or peptide-treated DC vaccine in
1998. In most trials, the number of registered cases was around 20
with mainly the HLA-A2 genotype, and only a few cases of clinical
response [partial remission (PR) and complete remission (CR)] were
reported. The difference of antigen source like synthetic peptide
or tumor lysate and maturation status did not seem to be closely
related to the clinical response (19). Additionally, a randomized phase III
study of melanoma DC vaccines has not yet been performed based on
early phase I, II studies. We have been testing a peptide-based DC
vaccine against HLA-A24+ metastatic melanoma in phase I
and II clinical trials. The ethnicity of HLA genotyping revealed a
significant difference of CTL epitope sequence and immunological
responses, which suggested that HLA-A24+-based immune
response is unique and should be investigated more intensively.
In the present study, based on survival data from
all metastatic melanoma patients from phase I and II trials,
potential prognostic factors were investigated among various
clinical, immunological and DC-processing-related parameters in
terms of the prolongating effect on overall survival.
Unfortunately, DC processing-related parameters did not show any
effect on overall survival. Interestingly, the anti-MAGE-A1
antibody titer before the vaccination was shown to be a possible
prognostic factor, but anti-melanoma antigen antibody induction
after the vaccination was not. Recently, an autoantibody
signature-based approach has been used to discover novel tumor
antigens (20–22). Especially, in melanoma
patient-derived serum, novel biomarkers involved in lymph node
metastasis prediction were identified (23). As to MAGE antigens, Stockert et
al reported that an autoantibody against MAGE-A1 was detected
in only 3 of 234 cancer patients (24), which was a very low frequency
compared with ours (48.4% in metastatic melanoma patients).
Impressively, our study demonstrated that the anti-MAGE-A1
autoantibody was positively correlated with overall survival, which
seems to be a novel observation. Meanwhile, the number of target
lesions and immune response parameters such as ELISPOT, DTH
reaction against peptide and KLH showed a prolongation effect on
overall survival, which was reasonable because tumor load and
immunological responses are known to be closely linked to prognosis
in melanoma patients (3,8).
The infiltration of CD8+ and TH17 cells
at the tumor site is reported to be closely involved in the
prognosis of solid cancer patients (25–27).
In our study, the positive rate of CD8 and IL-17 was 60 and 53%,
respectively, in 15 resected tumors. However, a significant
correlation to prognosis was not seen because of the small number
of cases.
Since sipuleucel-T (Provenge) immunotherapy was
approved by the FDA, DC-based cancer vaccine studies have been
encouraged and enhanced to develop the advanced stage of clinical
trials (28,29). As is the case with sipuleucel-T,
there will be some problems with DC-based cancer vaccines. One is
that the time for clinical evaluation might be too short to expect
prolongation of survival time because the optimal immune response
would have several weeks to operate and pass the cancer
progression. A conventional clinical evaluation based on RECIST
criteria is incompatible with overall survival benefit obtained
only by the continual administration of vaccine despite clinical
progression. To define the progression precisely in prostate
cancer, the Prostate Cancer Working Group recently devised
progression guidelines (30).
Very recently, studies of novel cancer vaccines like
sipuleucel-T and MAGE-A3 and other long peptides with conjugation
were activated at subclinical levels, which demonstrates the coming
of a new era for cancer vaccines (31–33).
The bottom line is that sequentially to the success of sipuleucel-T
trials, more phase III randomized studies of specific
peptide-pulsed DC vaccines should be performed. Additionally, a
world-wide network of translational research facilities which can
perform high-grade clinical immunotherapeutic research has to be
constructed. These efforts could lead to more efficient cancer
vaccines in the near future.
Acknowledgements
This study was supported by a grant from the
Cooperation of Innovative Technology and Advanced Research in
Evolutional Area (CITY AREA) program from the Ministry of
Education, Culture, Sports, Science and Technology, Japan.
Abbreviations:
|
DC
|
dendritic cell
|
|
HLA
|
human leukocyte antigen
|
|
FDA
|
food and drug administration
|
|
IRB
|
institutional review board
|
|
GM-CSF
|
granulocyte
macrophage-colony-stimulating factor
|
|
IL
|
interleukin
|
|
KLH
|
Keyhole limpet hemocyanin
|
|
IHC
|
immunohistochemistry
|
|
CTL
|
cytotoxic T cell
|
|
DTH
|
delayed-type hypersensitivity
|
|
CR
|
complete remission
|
|
PR
|
partial remission
|
|
SD
|
stable disease
|
|
PD
|
progressive disease
|
|
ELISA
|
enzyme-linked immunosorbent assay
|
|
IFN
|
interferon
|
|
HRP
|
horseradish peroxydase
|
|
PS
|
performance status
|
|
PBMC
|
peripheral blood mononuclear cell
|
References
|
1
|
Mayordomo JI, Zorina T, Storkus WJ,
Zitvogel L, Celluzzi C, Falo LD, Melief CJ, Ildstad ST, Kast WM and
Deleo AB: Bone marrow-derived dendritic cells pulsed with synthetic
tumor peptides elicit protective and therapeutic antitumor
immunity. Nat Med. 1:1297–1302. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Zitvogel L, Mayordomo JI, Tjandrawan T,
DeLeo AB, Clarke MR, Lotze MT and Storkus WJ: Therapy of murine
tumors with tumor peptide-pulsed dendritic cells: dependence on T
cells, B7 costimulation, and T helper cell 1-associated cytokines.
J Exp Med. 183:87–97. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Nestle FO, Alijagic S, Gilliet M, Sun Y,
Grabbe S, Dummer R, Burg G and Schadendorf D: Vaccination of
melanoma patients with peptide-or tumor lysate-pulsed dendritic
cells. Nat Med. 4:328–332. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Ranieri E, Kierstead LS, Zarour H,
Kirkwood JM, Lotze MT, Whiteside T and Storkus WJ: Dendritic
cell/peptide cancer vaccine: clinical responsiveness and epitope
spreading. Immunol Invest. 29:121–125. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Thurner B, Haendle I, Roder C, Dieckmann
D, Keikavoussi P, Jonuleit H, Bender A, Maczek C, Schreiner D, von
den Driesch P, et al: Vaccination with Mage-3a1 peptide-pulsed
mature, monocyte-derived dendritic cells expands specific cytotoxic
T cells and induces regression of some metastases in advanced stage
IV melanoma. J Exp Med. 190:1669–1678. 1999. View Article : Google Scholar
|
|
6
|
Panelli MC, Wunderlich J, Jeffries J, Wang
E, Mixon A, Rosenberg SA and Marincola FM: Phase 1 study in
patients with metastatic melanoma of immunization with dendritic
cells presenting epitopes derived from the melanoma-associated
antigens MART-1 and gp100. J Immunother. 23:487–498. 2000.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Jonuleit H, Giesecke-Tuettenberg A, Tuting
T, Thurner-Schuler B, Stuge TB, Paragnik L, Kandemir A, Lee PP,
Schuler G, Knop J and Enk AH: A comparison of two types of
dendritic cell as adjuvants for the induction of melanoma-specific
T-cell responses in humans following intranodal injection. Int J
Cancer. 93:243–251. 2001. View
Article : Google Scholar : PubMed/NCBI
|
|
8
|
Banchereau J, Palucka AK, Dhodapkar M,
Burkeholder S, Taquet N, Rolland A, Taquet S, Coquery S, Wittkowski
KM, Bhardwaj N, et al: Immune and clinical responses in patients
with metastatic melanoma to CD34+ progenitor-derived
dendritic cell vaccine. Cancer Res. 61:6451–6458. 2001.PubMed/NCBI
|
|
9
|
Lau R, Wang F, Jeffery G, Marty V,
Kuniyoshi J, Bade E, Ryback ME and Weber J: Phase I trial of
intravenous peptide-pulsed dendritic cells in patients with
metastatic melanoma. J Immunother. 24:66–78. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Chakraborty NG, Sporn JR, Tortora AF,
Kurtzman SH, Yamase H, Ergin MT and Mukherji B: Immunization with a
tumor-cell-lysate-loaded autologous-antigen-presenting-cell-based
vaccine in melanoma. Cancer Immunol Immunother. 47:58–64. 1998.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Smithers M, O’Connell K, MacFadyen S,
Chambers M, Greenwood K, Boyce A, Abdul-Jabbar I, Barker K,
Grimmett K, Walpole E and Thomas R: Clinical response after
intradermal immature dendritic cell vaccination in metastatic
melanoma is associated with immune response to particulate antigen.
Cancer Immunol Immunother. 52:41–52. 2003.
|
|
12
|
Chang AE, Redman BG, Whitfield JR,
Nickoloff BJ, Braun TM, Lee PP, Geiger JD and Mule JJ: A phase I
trial of tumor-lysate-pulsed dendritic cells in the treatment of
advanced cancer. Clin Cancer Res. 8:1021–1032. 2002.PubMed/NCBI
|
|
13
|
Burch PA, Croghan GA, Gastineau DA, Jones
LA, Kaur JS, Kylstra JW, Richardson RL, Valone FH and Vuk-Pavlovic
S: Immunotherapy (APC8015, Provenge) targeting prostatic acid
phosphatase can induce durable remission of metastatic
androgen-independent prostate camcer: a Phase 2 trial. Prostate.
60:197–204. 2004. View Article : Google Scholar
|
|
14
|
Cheever MA and Higano CS: PROVENGE
(Sipuleucel-T) in prostate cancer: the first FDA-approved
therapeutic cancer vaccine. Clin Cancer Res. 17:3520–3526. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Kantoff PW, Higano CS, Shore ND, Berger
ER, Small EJ, Penson DF, Redfern CH, Ferrari AC, Dreicer R, Sims
RB, et al: IMPACT Study Investigators: Sipuleucel-T immunotherapy
for castration-resistant prostate cancer. N Engl J Med.
363:411–422. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Akiyama Y, Tanosaki R, Inoue N, Shimada M,
Hotate Y, Yamamoto A, Yamazaki N, Kawashima I, Nukaya I, Takesako
K, et al: Clinical response in Japanese metastatic melanoma
patients treated with peptide cocktail-pulsed dendritic cells. J
Transl Med. 3:42005. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Therasse P, Arbuck SG, Eisenhauer EA,
Wanders J, Kaplan RS, Rubinstein L, Verweij J, van Glabbeke M, van
Oosterom AT, Christian MC and Gwyther SG: New guidelines to
evaluate the response to treatment in solid tumors European
Organization for Research and Treatment of Cancer, National Cancer
Institute of the United States, National Cancer Institute of
Canada. J Natl Cancer Inst. 92:205–216. 2000. View Article : Google Scholar
|
|
18
|
Iizuka A, Komiyama M, Tai S, Oshita C,
Kurusu A, Kume A, Ozawa K, Nakamuya Y, Ashizawa T, Yamamoto A, et
al: Identification of cytomegalovirus (CMV) pp65 antigen-specific
human monoclonal antibodies using single B cell-based antibody gene
cloning from melanoma partients. Immunol Lett. 135:64–73. 2011.
View Article : Google Scholar
|
|
19
|
Hersey P, Menzies SW, Halliday GM, Nguyen
T, Farrelly ML, DeSilva C and Lett M: Phase I/II study of treatment
with dendritic cell vaccines in patients with disseminated
melanoma. Cancer Immunol Immunother. 53:125–134. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Desmetz C, Maudelonde T, Mange A and
Solassol J: Identifying autoantibody signature in cancer: a
promising challenge. Expert Rev Proteomics. 6:377–386. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Desmetz C, Cortijo C, Mange A and Solassol
J: Humoral response to cancer as a tool for biomarker discovery. J
Proteomics. 72:982–988. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Tan HT, Low J, Lim SG and Chung MC: Serum
autoantibodies as biomarkers for early cancer detection. FEBS J.
276:6880–6904. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Liu Y, He J, Xie X, Su G, Teitz-Tennenbaum
S, Sabel MS and Lubman DM: Serum autoantibody profiling using a
natural glycoprotein microarray for the prognosis of early
melanoma. J Proteome Res. 9:6044–6051. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Stockert E, Jäger E, Chen YT, Scanlan MJ,
Gout I, Karbach J, Arand M, Knuth A and Old LJ: A survey of the
humoral immune response of cancer patients to a panel of human
tumor antigens. J Exp Med. 187:1349–1354. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Tosolini M, Kirilovsky A, Mlecnik B,
Fredriksen T, Mauger S, Bindea G, Berger A, Bruneval P, Fridman WH,
Pages F and Galon J: Clinical impact of different classes of
infiltrating T cytotoxic and helper cells (Th1, Th2, Treg, Th17) in
patients with colorectal cancer. Cancer Res. 71:1263–1271. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Galon J, Costes A, Sanchez-Cabo F,
Kirilovsky A, Mlecnik B, Lagorce-Pages C, Tosolini M, Camus M,
Berger A, Wind P, et al: Type, density, and location of immune
cells within human colorectal tumors predict clinical outcome.
Science. 313:1960–1964. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Dieu-Nosjean MC, Antoine M, Danel C,
Heudes D, Wislez M, Poulot V, Rabbe N, Laurans L, Tartour E, de
Chaisemartin L, et al: Long-term survival for patients with
non-small cell lung cancer with intratumoral lymphoid structures. J
Clin Oncol. 26:4410–4417. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Rini BI, Weinberg V, Fong L, Conry S,
Hershberg RM and Small EJ: Combination immunotherapy with prostatic
acid phosphatase pulsed antigen-presenting cells (provenge) plus
bevacizumab in patients with serologic progression of prostate
cancer after definitive local therapy. Cancer. 107:67–74. 2006.
View Article : Google Scholar
|
|
29
|
Cheever MA: Twelve immunotherapy drugs
that could cure cancers. Immunol Rev. 222:357–368. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Scher HI, Halabi S, Tannock I, Morris M,
Sternberg CN, Carducci MA, Eisenberger MA, Higano C and Bubley GJ:
Dreicer R, et al; Prostate Cancer Clinical Trials Working
Group: Design amd end points of clinical trials for patients with
progressive prostate cancer and castrate levels of testosterone:
recommendations of the Prostate Cancer Clinical Trials Working
Group. J Clin Oncol. 26:1148–1159. 2008.PubMed/NCBI
|
|
31
|
Buonerba C, Ferro M and Di Lorenzo G:
Sipuleucel-T for prostate cancer: the immunotherapy era has
commenced. Expert Rev Anticancer Ther. 11:25–28. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Finke LH, Wentworth K, Blumenstein B,
Rudolph NS, Levitsky H and Hoos A: Lessons from randomized phase
III studies with active cancer immunotherapies-outcomes from the
2006 meeting of the Cancer Vaccine Consortium (CVC). Vaccines.
25(Suppl 2): S97–S109. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Tyagi P and Mirakhur B: MAGRIT: the
largest-ever phase III lung cancer trial aims to establish a novel
tumor-specific approach to therapy. Clin Lung Cancer. 10:371–374.
2009. View Article : Google Scholar : PubMed/NCBI
|