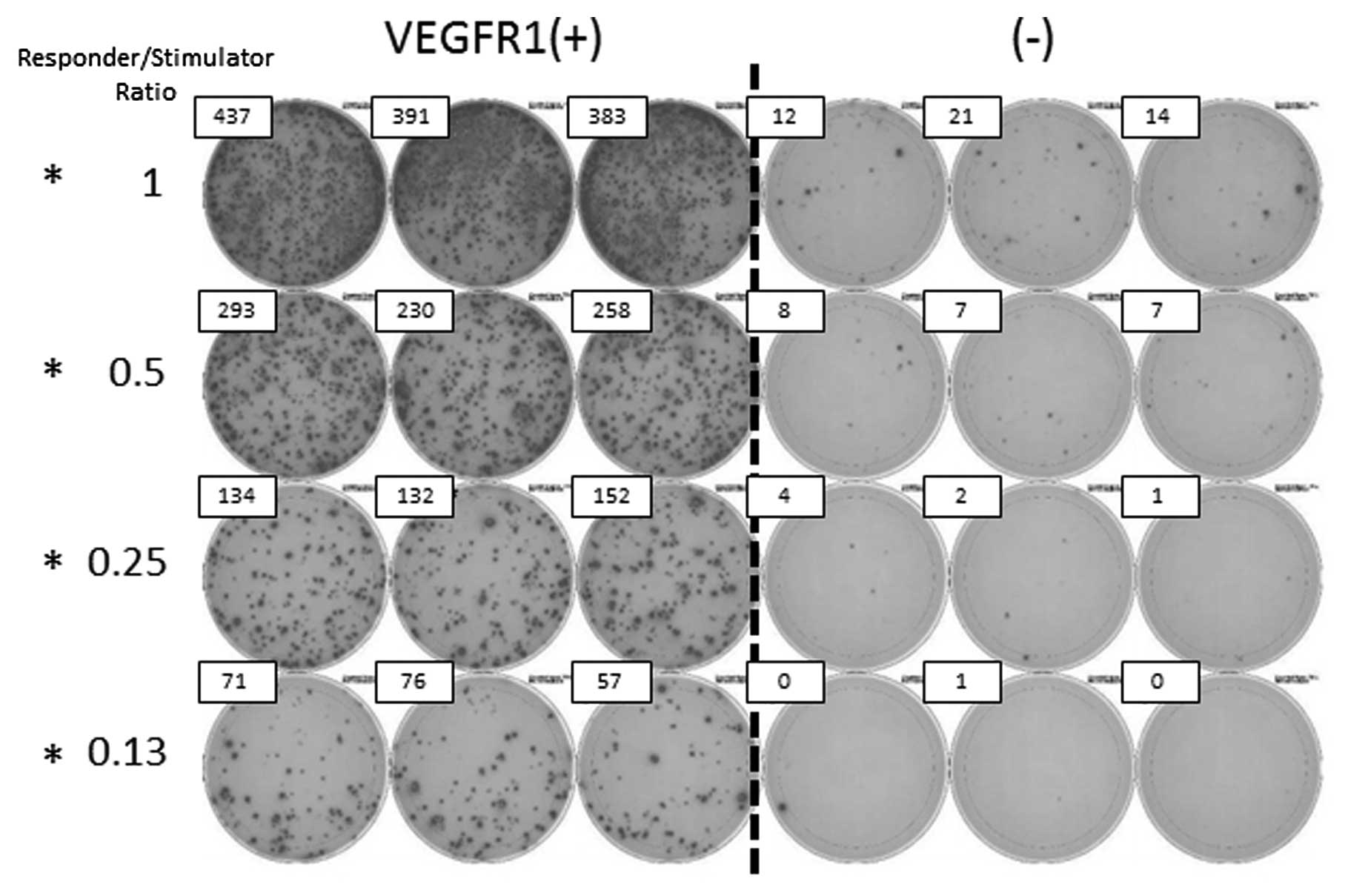
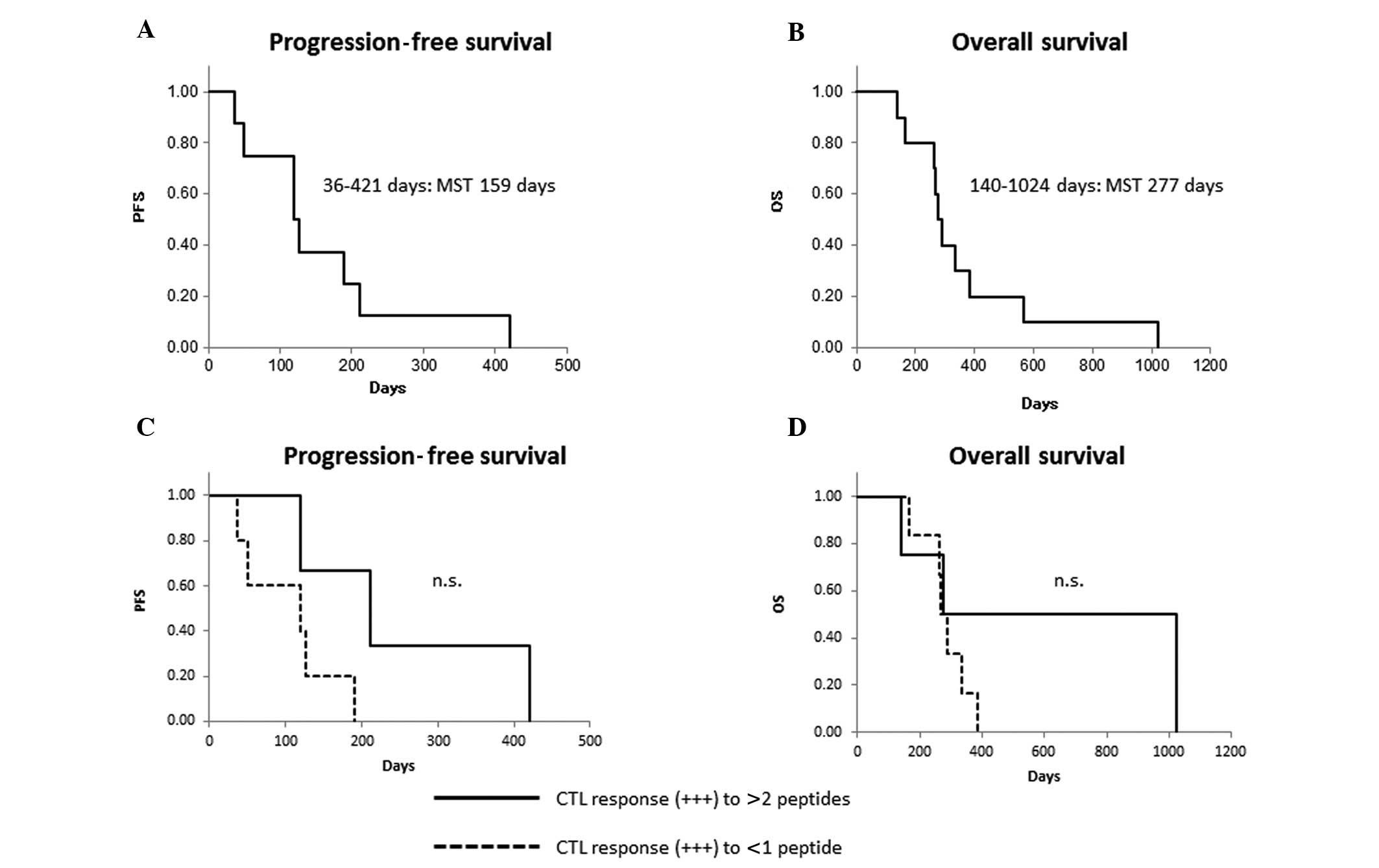
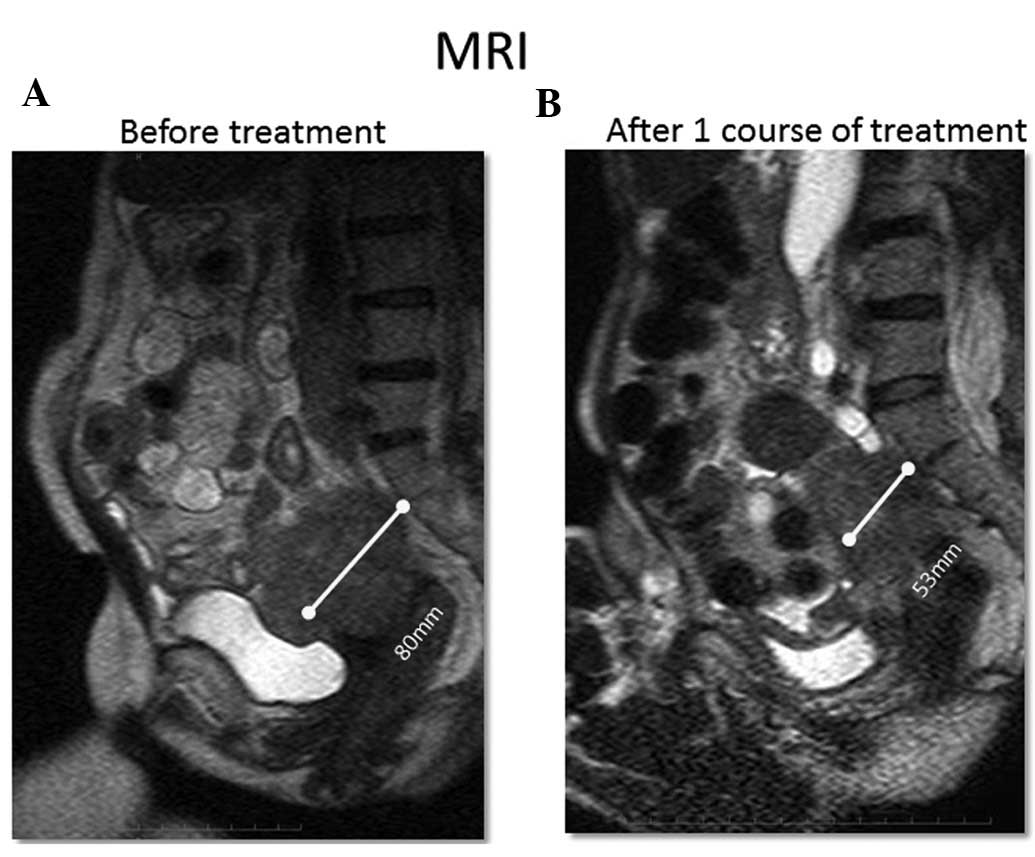
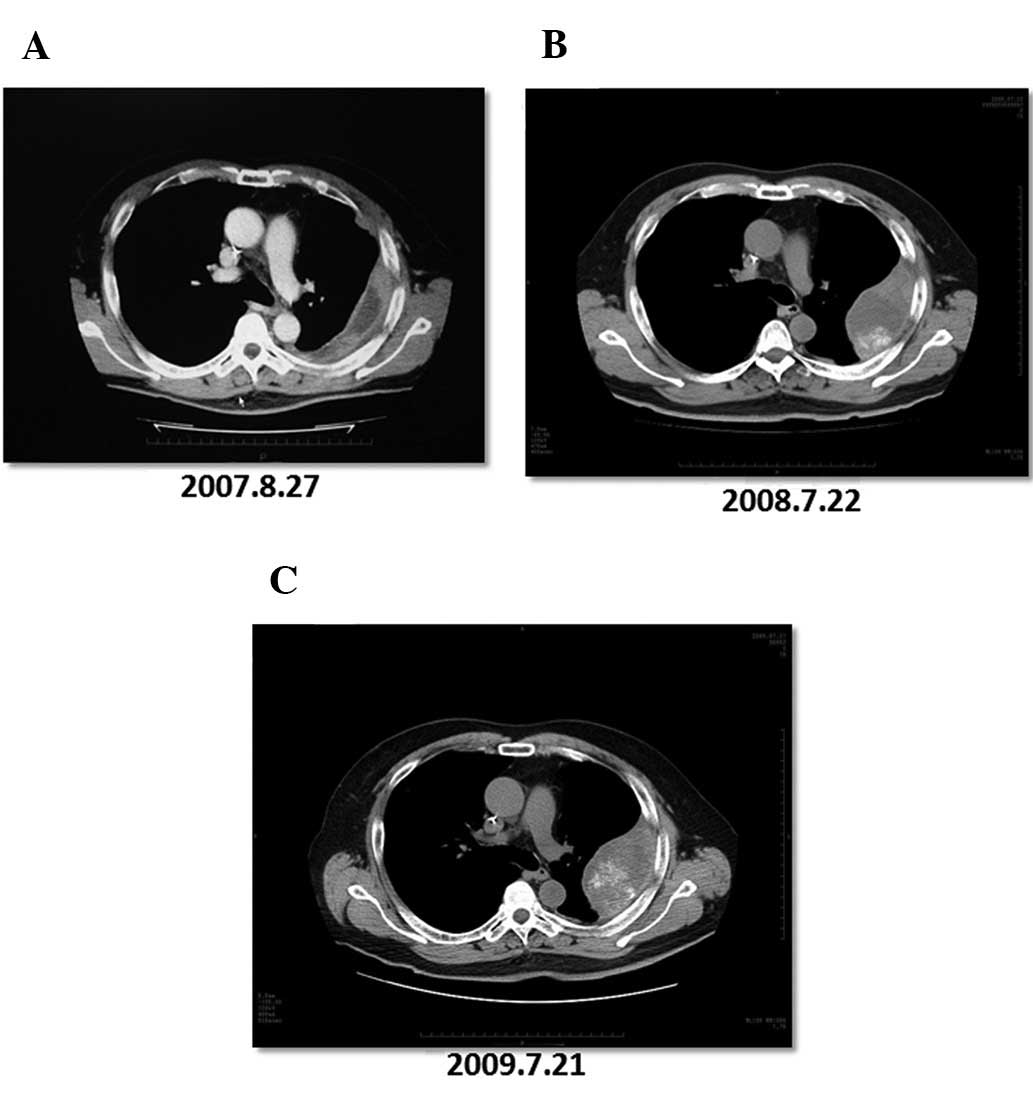
|
1
|
Shimokawa T, Matsushima S, Tsunoda T,
Tahara H, Nakamura Y and Furukawa Y: Identification of TOMM34,
which shows elevated expression in the majority of human colon
cancers, as a novel drug target. Int J Oncol. 29:381–386.
2006.PubMed/NCBI
|
|
2
|
Lin YM, Furukawa Y, Tsunoda T, Yue CT,
Yang KC and Nakamura Y: Molecular diagnosis of colorectal tumors by
expression profiles of 50 genes expressed differentially in
adenomas and carcinomas. Oncogene. 21:4120–4128. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Yagyu R, Furukawa Y, Lin YM, Shimokawa T,
Yamamura T and Nakamura Y: A novel oncoprotein RNF43 functions in
an autocrine manner in colorectal cancer. Int J Oncol.
25:1343–1348. 2004.PubMed/NCBI
|
|
4
|
Soker S, Takashima S, Miao HQ, Neufeld G
and Klagsbrun M: Neuropilin-1 is expressed by endothelial and tumor
cells as an isoform-specific receptor for vascular endothelial
growth factor. Cell. 92:735–745. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Niethammer AG, Xiang R, Becker JC, Wodrich
H, Pertl U, Karsten G, Eliceiri BP and Reisfeld RA: A DNA vaccine
against VEGF receptor 2 prevents effective angiogenesis and
inhibits tumor growth. Nat Med. 8:1369–1375. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Li Y, Wang MN, Li H, King KD, Bassi R, Sun
H, Santiago A, Hooper AT, Bohlen P and Hicklin DJ: Active
immunization against the vascular endothelial growth factor
receptor flk1 inhibits tumor angiogenesis and metastasis. J Exp
Med. 195:1575–1584. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Wada S, Tsunoda T, Baba T, Primus FJ,
Kuwano H, Shibuya M and Tahara H: Rationale for antiangiogenic
cancer therapy with vaccination using epitope peptides derived from
human vascular endothelial growth factor receptor 2. Cancer Res.
65:4939–4946. 2005. View Article : Google Scholar
|
|
8
|
Douillard JY, Hoff PM, Skillings JR,
Eisenberg P, Davidson N, Harper P, Vincent MD, Lembersky BC,
Thompson S, Maniero A and Benner SE: Multicenter phase III study of
uracil/tegafur and oral leucovorin versus fluorouracil and
leucovorin in patients with previously untreated metastatic
colorectal cancer. J Clin Oncol. 20:3605–3616. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Carmichael J, Popiela T, Radstone D, Falk
S, Borner M, Oza A, Skovsgaard T, Munier S and Martin C: Randomized
comparative study of tegafur/uracil and oral leucovorin versus
parenteral fluorouracil and leucovorin in patients with previously
untreated metastatic colorectal cancer. J Clin Oncol. 20:3617–3627.
2002. View Article : Google Scholar
|
|
10
|
Therasse P, Arbuck SG, Eisenhauer EA,
Wanders J, Kaplan RS, Rubinstein L, Verweij J, Van Glabbeke M, van
Oosterom AT, Christian MC and Gwyther SG: New guidelines to
evaluate the response to treatment in solid tumors. European
Organization for Research and Treatment of Cancer, National Cancer
Institute of the United States, National Cancer Institute of
Canada. J Natl Cancer Inst. 92:205–216. 2000. View Article : Google Scholar
|
|
11
|
Sadanaga N, Nagashima H, Mashino K, Tahara
K, Yamaguchi H, Ohta M, Fujie T, Tanaka F, Inoue H, Takesako K,
Akiyoshi T and Mori M: Dendritic cell vaccination with MAGE peptide
is a novel therapeutic approach for gastrointestinal carcinomas.
Clin Cancer Res. 7:2277–2284. 2001.PubMed/NCBI
|
|
12
|
Nagorsen D and Thiel E: Clinical and
immunologic responses to active specific cancer vaccines in human
colorectal cancer. Clin Cancer Res. 12:3064–3069. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Rosenberg SA, Yang JC and Restifo NP:
Cancer immunotherapy: moving beyond current vaccines. Nat Med.
10:909–915. 2004. View
Article : Google Scholar : PubMed/NCBI
|
|
14
|
Kono K, Mizukami Y, Daigo Y, Takano A,
Masuda K, Yoshida K, Tsunoda T, Kawaguchi Y, Nakamura Y and Fujii
H: Vaccination with multiple peptides derived from novel
cancer-testis antigens can induce specific T-cell responses and
clinical responses in advanced esophageal cancer. Cancer Sci.
100:1502–1509. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Correale P, Cusi MG, Tsang KY, Del Vecchio
MT, Marsili S, Placa ML, Intrivici C, Aquino A, Micheli L, Nencini
C, Ferrari F, Giorgi G, Bonmassar E and Francini G:
Chemo-immunotherapy of metastatic colorectal carcinoma with
gemcitabine plus FOLFOX 4 followed by subcutaneous granulocyte
macrophage colony-stimulating factor and interleukin-2 induces
strong immunologic and antitumor activity in metastatic colon
cancer patients. J Clin Oncol. 23:8950–8958. 2005.
|
|
16
|
Correale P, Aquino A, Giuliani A,
Pellegrini M, Micheli L, Cusi MG, Nencini C, Petrioli R, Prete SP,
De Vecchis L, Turriziani M, Giorgi G, Bonmassar E and Francini G:
Treatment of colon and breast carcinoma cells with 5-fluorouracil
enhances expression of carcinoembryonic antigen and susceptibility
to HLA-A(*)02.01 restricted, CEA-peptide-specific cytotoxic T cells
in vitro. Int J Cancer. 104:437–445. 2003.PubMed/NCBI
|
|
17
|
Dauer M, Herten J, Bauer C, Renner F,
Schad K, Schnurr M, Endres S and Eigler A: Chemosensitization of
pancreatic carcinoma cells to enhance T cell-mediated cytotoxicity
induced by tumor lysate-pulsed dendritic cells. J Immunother.
28:332–342. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Correale P, Cusi MG, Del Vecchio MT,
Aquino A, Prete SP, Tsang KY, Micheli L, Nencini C, La Placa M,
Montagnani F, Terrosi C, Caraglia M, Formica V, Giorgi G, Bonmassar
E and Francini G: Dendritic cell-mediated cross-presentation of
antigens derived from colon carcinoma cells exposed to a highly
cytotoxic multidrug regimen with gemcitabine, oxaliplatin,
5-fluorouracil, and leucovorin, elicits a powerful human
antigen-specific CTL response with antitumor activity in vitro. J
Immunol. 175:820–828. 2005.
|
|
19
|
Sato Y, Fujiwara T, Mine T, Shomura H,
Homma S, Maeda Y, Tokunaga N, Ikeda Y, Ishihara Y, Yamada A, Tanaka
N, Itoh K, Harada M and Todo S: Immunological evaluation of
personalized peptide vaccination in combination with a
5-fluorouracil derivative (TS-1) for advanced gastric or colorectal
carcinoma patients. Cancer Sci. 98:1113–1119. 2007. View Article : Google Scholar
|
|
20
|
Correale P, Del Vecchio MT, Di Genova G,
Savellini GG, La Placa M, Terrosi C, Vestri M, Urso R, Lemonnier F,
Aquino A, Bonmassar E, Giorgi G, Francini G and Cusi MG:
5-Fluorouracil-based chemotherapy enhances the antitumor activity
of a thymidylate synthase-directed polyepitopic peptide vaccine. J
Natl Cancer Inst. 97:1437–1445. 2005. View Article : Google Scholar
|
|
21
|
Weihrauch MR, Ansén S, Jurkiewicz E,
Geisen C, Xia Z, Anderson KS, Gracien E, Schmidt M, Wittig B, Diehl
V, Wolf J, Bohlen H and Nadler LM: Phase I/II combined
chemoimmunotherapy with carcinoembryonic antigen-derived
HLA-A2-restricted CAP-1 peptide and irinotecan, 5-fluorouracil, and
leucovorin in patients with primary metastatic colorectal cancer.
Clin Cancer Res. 11:5993–6001. 2005. View Article : Google Scholar
|
|
22
|
Harrop R, Drury N, Shingler W, Chikoti P,
Redchenko I, Carroll MW, Kingsman SM, Naylor S, Melcher A, Nicholls
J, Wassan H, Habib N and Anthoney A: Vaccination of colorectal
cancer patients with modified vaccinia Ankara encoding the tumor
antigen 5T4 (TroVax) given alongside chemotherapy induces potent
immune responses. Clin Cancer Res. 13:4487–4494. 2007. View Article : Google Scholar
|
|
23
|
Yuan L, Kuramitsu Y, Li Y, Kobayashi M and
Hosokawa M: Restoration of interleukin-2 production in
tumor-bearing rats through reducing tumor-derived transforming
growth factor beta by treatment with bleomycin. Cancer Immunol
Immunother. 41:355–362. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Backus HH, Dukers DF, van Groeningen CJ,
Vos W, Bloemena E, Wouters D, van Riel JM, Smid K, Giaccone G,
Pinedo HM and Peters GJ: 5-Fluorouracil induced Fas upregulation is
associated with apoptosis in liver metastases of colorectal cancer
patients. Ann Oncol. 12:209–216. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Sato Y, Shomura H, Maeda Y, Mine T, Une Y,
Akasaka Y, Kondo M, Takahashi S, Shinohara T, Katagiri K, Sato M,
Okada S, Matsui K, Yamada A, Yamana H, Itoh K and Todo S:
Immunological evaluation of peptide vaccination for patients with
gastric cancer based on pre-existing cellular response to peptide.
Cancer Sci. 94:802–808. 2003. View Article : Google Scholar : PubMed/NCBI
|