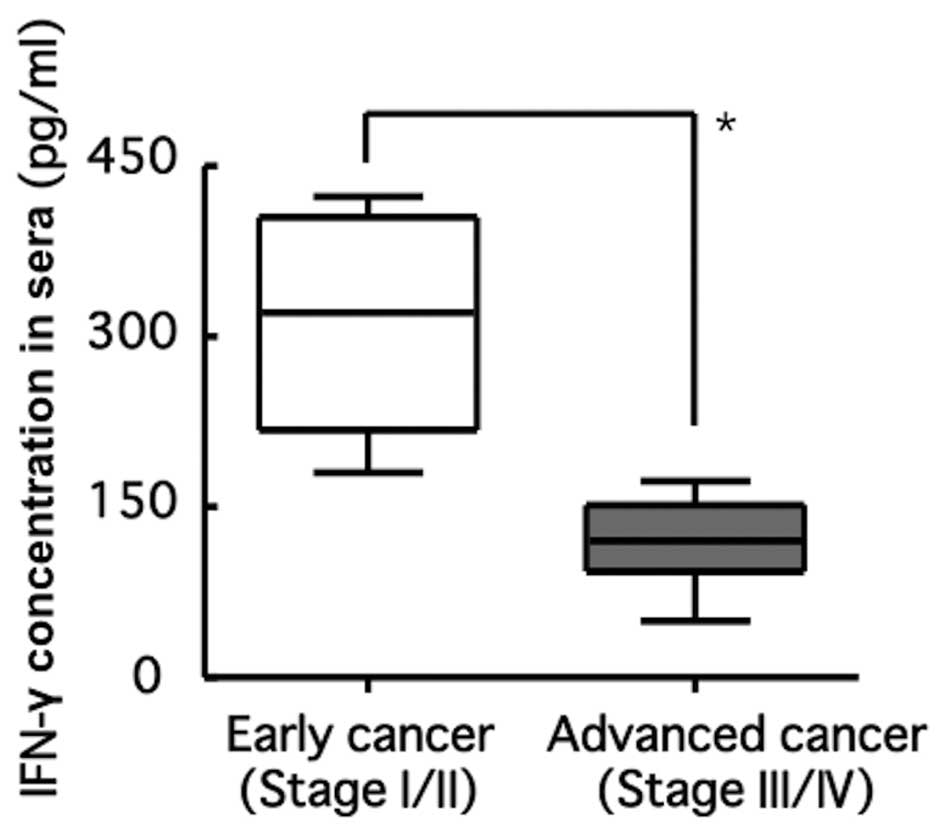
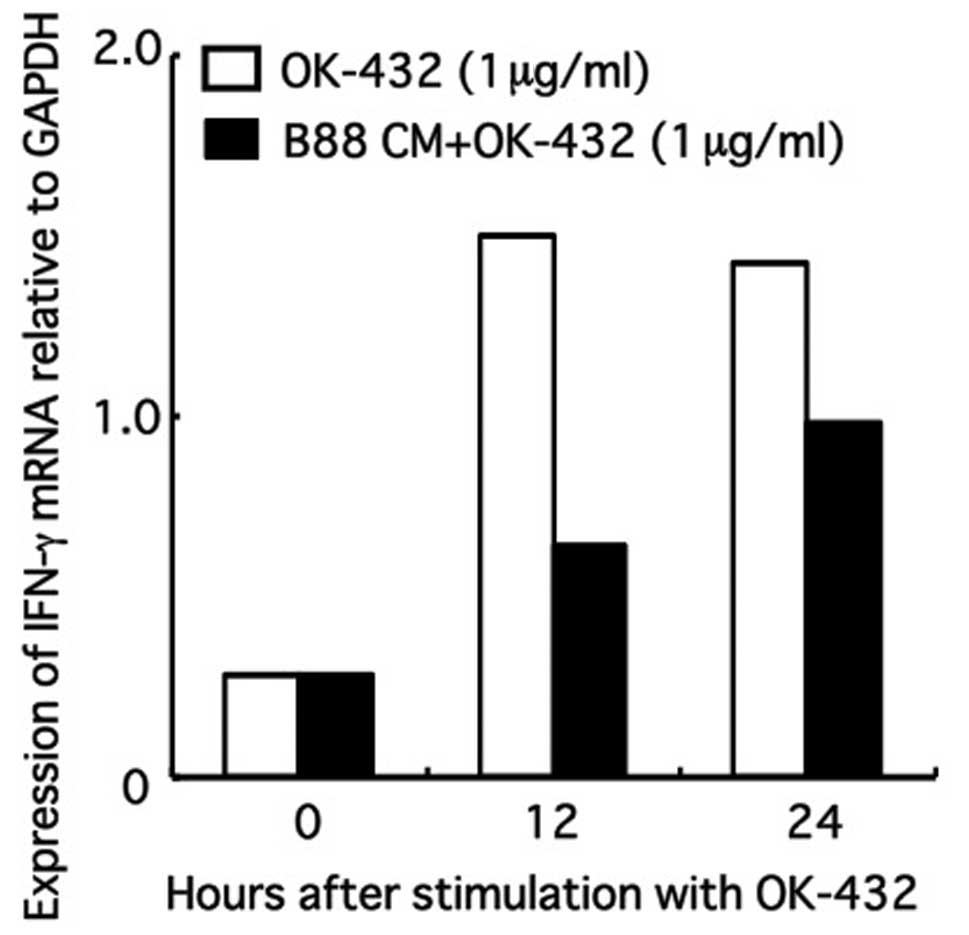
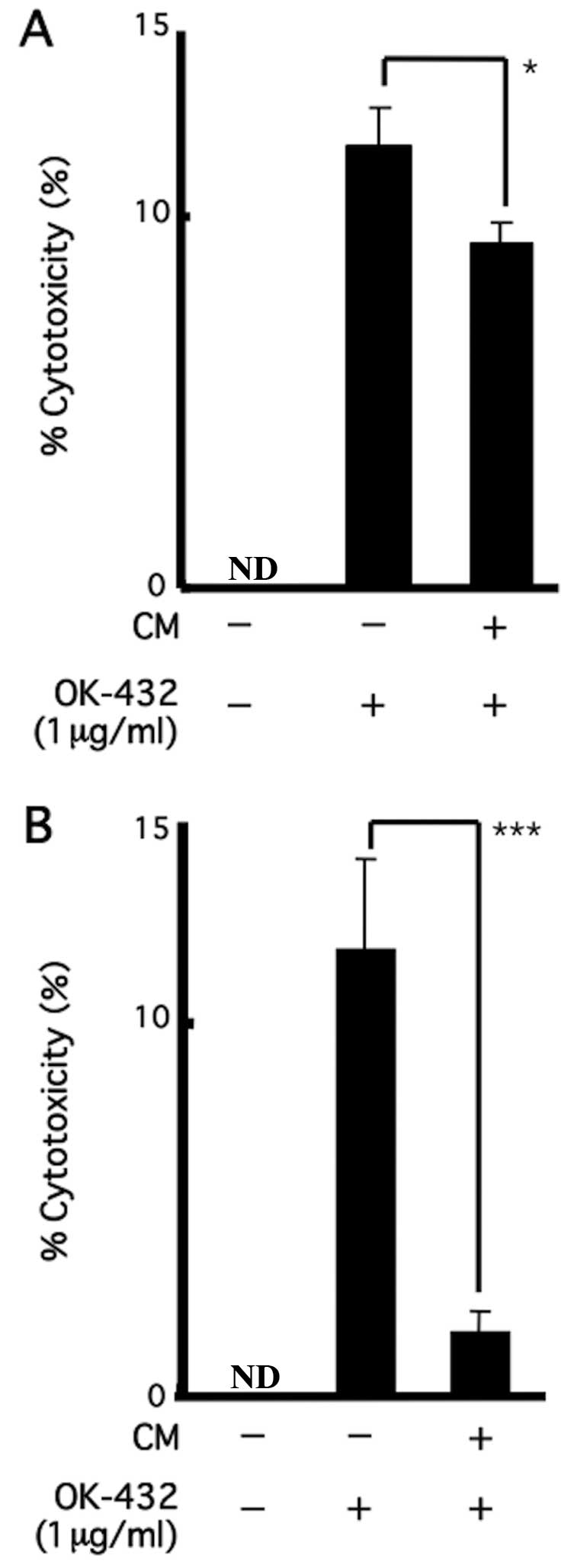
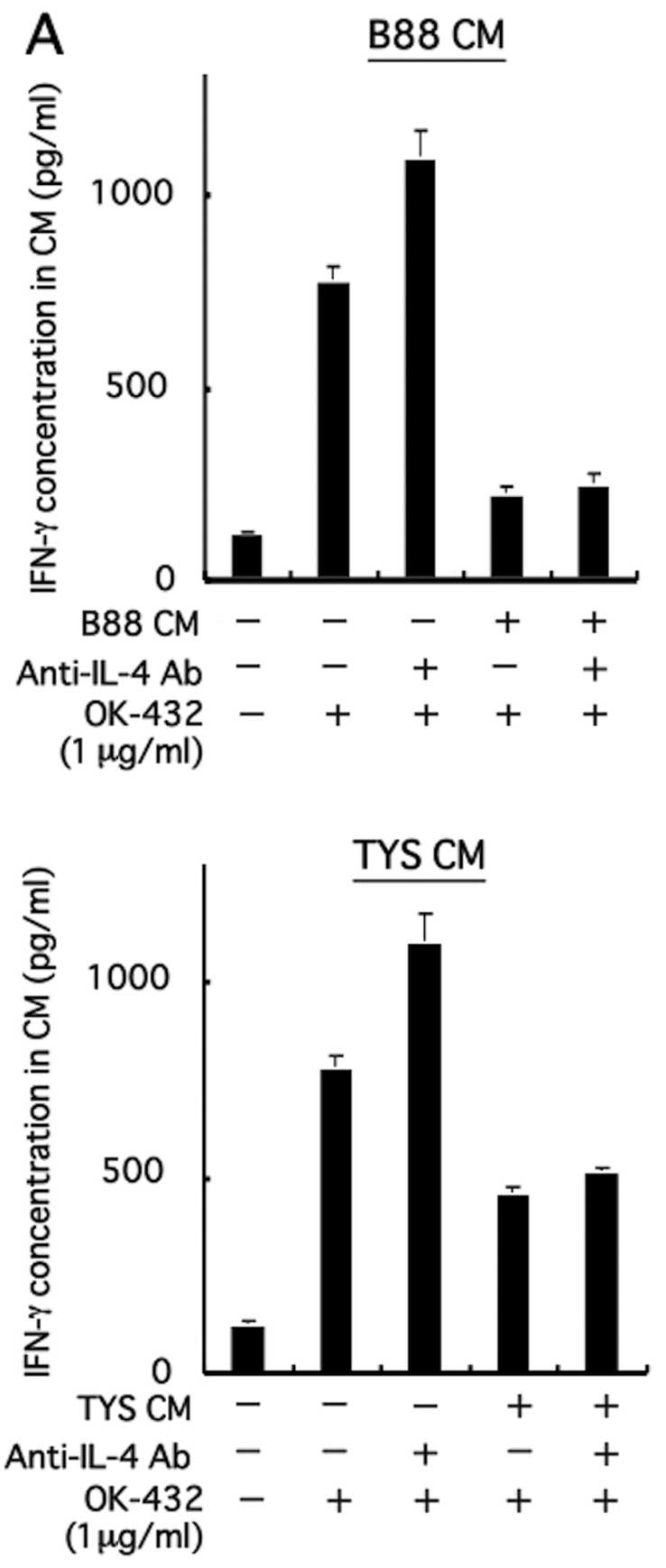
|
1
|
Okamoto H, Shoin S, Koshimura S and
Shimizu R: Studies on the anticancer and streptolysin S-forming
abilities of hemolytic streptococci. Jpn J Microbiol. 11:323–326.
1967. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Watanabe Y and Iwa T: Clinical value of
immunotherapy with the streptococcal preparation OK-432 in
non-small cell lung cancer. J Biol Response Mod. 6:169–180.
1987.PubMed/NCBI
|
|
3
|
Katano M and Torisu M: New approach to
management of malignant ascites with a streptococcal preparation,
OK-432. II Intraperitoneal inflammatory cell-mediated tumor cell
destruction. Surgery. 93:365–373. 1983.PubMed/NCBI
|
|
4
|
Uchida A and Micksche M: Intrapleural
administration of OK-432 in cancer patients: activation of NK cells
and reduction of suppressor cells. Int J Cancer. 31:1–5. 1983.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Uchida A, Micksche M and Hoshino T:
Intrapleural administration of OK-432 in cancer patients:
augmentation of autologous tumor killing activity of
tumor-associated large granular lymphocytes. Cancer Immunol
Immunother. 18:5–12. 1984. View Article : Google Scholar
|
|
6
|
Sato M, Yoshida H, Yanagawa T, Yura Y,
Urata M, Atsumi M, Hayashi Y and Takegawa Y: Effects of intradermal
administration of streptococcal preparation OK-432 on interferon
and natural killer cell activities in patients with oral cancer.
Int J Oral Surg. 13:7–15. 1984. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Sato M, Harada K, Yoshida H, Yura Y, Azuma
M, Iga H, Bando T, Kawamata H and Takegawa Y: Therapy for oral
squamous cell carcinoma by tegafur and streptococcal agent OK-432
in combination with radiotherapy: association of the therapeutic
effect with differentiation and apoptosis in the cancer cells.
Apoptosis. 2:227–238. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Oshimi K, Kano S, Takaku F and Okumura K:
Augmentation of mouse natural killer cell activity by a
streptococcal preparation, OK-432. J Natl Cancer Inst.
65:1265–1269. 1980.PubMed/NCBI
|
|
9
|
Oshimi K, Wakasugi H, Seki H and Kano S:
Streptococcal preparation OK-432 augments cytotoxic activity
against an erythroleukemic cell line in humans. Cancer Immunol
Immunother. 9:187–192. 1980. View Article : Google Scholar
|
|
10
|
Matsubara S, Suzuki F and Ishida N:
Induction of immune interferon in mice treated with a bacterial
immunopotentiator, OK-432. Cancer Immunol Immunother. 6:41–45.
1979. View Article : Google Scholar
|
|
11
|
Sato M, Hayashi Y, Yashida H, Yanagawa T,
Yura Y, Urata M and Furumoto N: Effect of immunotherapy with a
streptococcal preparation, OK-432, on the peripheral killer
lymphocyte population in patients with head and neck cancer.
Immunopharmacological Aspects of OK-432 in Humans. 1st edition.
Excerpta Medica; Tokyo: pp. 78–88. 1986
|
|
12
|
Kaji R, Yoshida H, Yanagawa T and Sato M:
Monoclonal antibody to a human salivary adenocarcinoma cell line:
augmentation of antibody-dependent cell-mediated cytotoxity
activity by streptococcal preparation OK-432 in human salivary
adenocarcinoma-bearing nude mice given the antibody. J Biol
Response Mod. 8:488–500. 1989.
|
|
13
|
Nakahara S, Tsunoda T, Baba T, Asabe S and
Tahara H: Dendritic cells stimulated with a bacterial products,
OK-432, efficiently induce cytotoxic T lymphocytes specific to
tumor rejection peptide. Cancer Res. 63:4112–4118. 2003.PubMed/NCBI
|
|
14
|
Okamoto M, Kaji R, Kasetani H, Yoshida H,
Moriya Y, Saito M and Sato M: Purification and characterization of
interferon-γ-inducing molecule of OK-432, a penicillin-killed
streptococcal preparation, by monoclonal antibody neutralizing
interferon-γ-inducing activity of OK-432. J Immunother Emphasis
Tumor Immunol. 13:232–242. 1993.
|
|
15
|
Okamoto M, Ohe G, Oshikawa T, Furuichi S,
Nishikawa N, Tano T, Ahmed SU, Yoshida H, Moriya Y, Saito S and
Sato M: Induction of Th1-type cytokines by lipoteichoic
acid-related preparation isolated from OK-432, a penicillin-killed
streptococcal agent. Immunopharmacology. 49:363–376. 2000.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Okamoto M and Sato M: Toll-like receptor
signaling in anti-cancer immunity. J Med Invest. 50:9–24. 2003.
|
|
17
|
Okamoto M and Sato M: Effective molecule
of a streptococcal preparation OK-432 and its molecular targets:
significance for cancer immunotherapy. Rec Res Dev Infect Immun.
1:29–44. 2003.
|
|
18
|
Takada H, Kawabata Y, Arakaki R, Kusumoto
S, Fukase K, Suda Y, Yoshimura T, Kokeguchi S, Kato K and Komuro T:
Molecular and structural requirements of a lipoteichoic acid from
Enterococcus hirae ATCC 9790 for cytokine-inducing,
antitumor, and antigenic activities. Infect Immun. 63:57–65.
1995.PubMed/NCBI
|
|
19
|
Okamoto M, Oshikawa T, Furuichi S,
Nishikawa N, Tano T, Ahmed SU, Yoshida H, Matsubara S, Matsuno T
and Sato M: Comparison of cytokine-inducing activity in a
lipoteichoic acid-related molecule isolated from a
penicillin-killed group A Streptococcus and from untreated
bacteria. Int Immunopharmacol. 1:1957–1968. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Oshikawa T, Okamoto M, Ohe G, Furuichi S,
Nishikawa H, Ahmed SU, Yoshida H, Moriya Y, Matsubara S, Ryoma Y,
Saito M and Sato M: Isolation of a Th1-inducing molecule from
OK-432, a streptococcal preparation, by a monoclonal antibody TS-2
that neutralizes the interferon-γ-inducing activity of OK-432:
comparison of the enhancement of anti-tumor immunity between the
TS-2-binding and TS-2-unbinding fraction. Int Immunopharmacol.
3:643–655. 2003.PubMed/NCBI
|
|
21
|
Okamoto M, Ohe G, Furuichi S, Nishikawa N,
Tano T, Ahmed SU, Yoshida H and Sato M: Enhancement of anti-cancer
immunity by a lipoteichoic-acid-related molecule isolated from a
penicillin-killed group A Streptococcus. Cancer Immunol
Immunother. 50:408–416. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Okamoto M, Ohe G, Furuichi S, Nishikawa N,
Tano T, Ahmed SU, Yoshida H and Sato M: Enhancement of anti-tumor
immunity by lipoteichoic acid-related molecule isolated from
OK-432, a streptococcal agent, in athymic nude mice bearing human
salivary adenocarcinoma: Role of natural killer cells. Anticancer
Res. 226:3229–3240. 2002.
|
|
23
|
Shimazu R, Akashi S, Ogata H, Nagai Y,
Fukudome K, Miyake K and Kimoto M: MD-2, a molecule that confers
lipopolysaccharide responsiveness on Toll-like receptor 4. J Exp
Med. 189:1777–1782. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Okamoto M, Oshikawa T, Ohe G, Furuichi S,
Nishikawa N, Tano T, Ahmed SU, Yoshida H and Sato M: Severe
impairment of anti-cancer effect of lipoteichoic acid-related
molecule isolated from a penicillin-killed Streptococcus
pyogenes in toll-like receptor 4-deficient mice. Int
Immunopharmacol. 1:1789–1795. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Okamoto M, Oshikawa T, Tano T, Ohe G,
Furuichi S, Nishikawa H, Ahmed SU, Akashi S, Miyake K, Takeuchi O,
Akira S, Moriya Y, Matsubara S, Ryoma Y, Saito M and Sato M:
Involvement of Toll-like receptor 4 signaling in interferon-γ
production and anti-tumor effect by a streptococcal agent OK-432. J
Natl Cancer Inst. 95:316–326. 2003.
|
|
26
|
UICC International Union Against Cancer.
TNM Classification of Malignant Tumours. Sobin LH and Wittekind C:
5th edition. Wiley-Liss; New York, NY: pp. 17–50. 1997
|
|
27
|
Uchida D, Begum NM, Almofti A, Nakashiro
K, Kawamata H, Tateishi Y, Hamakawa H, Yoshida H and Sato M:
Possible role of stromal cell-derived factor-1/CXCR4 signaling on
lymph-node metastasis of oral squamous cell carcinoma. Exp Cell
Res. 290:289–302. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Kawamata H, Nakashiro K, Uchida D, Harada
K, Yoshida H and Sato M: Possible contribution of active MMP2 to
lymph-node metastasis and secreted cathepsin L to bone invasion of
newly established human oral-squamous-cancer cell lines. Int J
Cancer. 70:120–127. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Yanagawa T, Hayashi Y, Yoshida H, Yura Y,
Nagamine S, Bando T and Sato M: An adenoid squamous
carcinoma-forming cell line established from an oral keratinizing
squamous cell carcinoma expressing carcinoembryonic antigen. Am J
Pathol. 124:496–509. 1986.
|
|
30
|
Anderson LC, Nilsson K and Gahmberg CG:
K-562, a human erythroleukemic cell line. Int J Cancer. 23:143–147.
1979. View Article : Google Scholar
|
|
31
|
Klein E, Klein G, Nadkarmi JS, Nadkarmi
JJ, Wigzell H and Clifford P: Surface IgM-kappa specificity on a
Burkitt lymphoma cell in vivo and in derived culture lines. Cancer
Res. 28:1300–1310. 1968.PubMed/NCBI
|
|
32
|
Boyüm A: Isolation of mononuclear cells
and granulocytes from human blood. Isolation of mononuclear cells
by one centrifugation, and of granulocytes by combining
centrifugation and sedimentation at 1g. Scand J Clin Lab Invest
Suppl. 97:77–89. 1968.PubMed/NCBI
|
|
33
|
Nachlas MM, Margulies SI, Goldberg JD and
Seligman AM: The determination of lactic dehydrogenase with a
tetrazolium salt. Anal Biochem. 1:317–326. 1960. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Korzeniewski C and Callewaert DM: An
enzyme-release assay for natural cytotoxicity. J Immunol Methods.
64:313–320. 1983. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Decker T and Lohmann-Matthes ML: A quick
and simple method for the quantitation of lactate dehydrogenase
release in measurements of cellular cytotoxicity and tumor necrosis
factor (TNF) activity. J Immunol Methods. 115:61–69. 1988.
View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Pasare C and Medzhitov R: Toll
pathway-dependent blockade of CD4+CD25+ T
cell-mediated suppression by dendritic cells. Science.
299:1033–1036. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Tada T, Ohzeki S, Utsumi K, Takiuchi H,
Muramatsu M, Li XF, Shimizu J, Fujiwara H and Hamaoka T:
Transforming growth factor-β-induced inhibition of T cell functions
and its relevance to immunosuppression in the tumor-bearing state.
J Immunol. 146:1077–1082. 1991.
|
|
38
|
Bellone G, Turletti A, Artusio E, Mareschi
K, Carbone A, Tibaudi D, Robecchi A, Emanuelli G and Rodeck U:
Tumor-associated transforming growth factor-β and interleukin-10
contribute to a systemic Th2 immune phenotype in pancreatic
carcinoma patients. Am J Pathol. 155:537–547. 1999.
|
|
39
|
Mouri H, Sakaguchi K, Sawayama T, Senoh T,
Ohta T, Nishimura M, Fujiwara A, Terao M, Shiratori Y and Tsuji T:
Suppressive effects of transforming growth factor-β1 produced by
hepatocellular carcinoma cell lines on interferon-γ production by
peripheral blood mononuclear cells. Acta Med Okayama. 56:309–315.
2002.
|
|
40
|
Wang T, Niu G, Kortylewski M, Burdelya L,
Shain K, Zhang S, Bhattacharya R, Gabrilovich D, Heller R, Coppola
D, Dalton W, Jove R, Pardoll D and Yu H: Regulation of the innate
and adaptive immune responses by Stat-3 signaling in tumor cells.
Nat Med. 10:48–54. 2004. View
Article : Google Scholar : PubMed/NCBI
|
|
41
|
Gabrilovich D, Ishida T, Oyama T, Ran S,
Kravtsov V, Nadaf S and Carbone DP: Vascular endothelial growth
factor inhibits the development of dendritic cells and dramatically
affects the differentiation of multiple hematopoietic lineages in
vivo. Blood. 92:4150–4166. 1998.
|
|
42
|
Smyth GP, Stapleton PP, Barden CB, Mestre
JR, Freeman TA, Duff MD, Maddali S, Yan Z and Daly JM: Renal cell
carcinoma induces prostaglandin E2 and T-helper type 2 cytokine
production in peripheral blood mononuclear cells. Ann Surg Oncol.
10:455–462. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
McCarter M, Clarke J, Richter D and Wilson
C: Melanoma skews dendritic cells to facilitate a T helper 2
profile. Surgery. 138:321–328. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Fujimoto T, Duda RB, Szilvasi A, Chen X,
Mai M and O’Donnell MA: Streptococcal preparation OK-432 is a
potent inducer of IL-12 and a T helper cell 1 dominant state. J
Immunol. 158:5619–5626. 1997.PubMed/NCBI
|
|
45
|
Fiorentino DF, Zlotnic A, Vieira P,
Mosmann TR, Howard M, Moore KW and Ogarra A: IL-10 acts on the
antigen-presenting cell to inhibit cytokine production by Th1
cells. J Immunol. 146:3444–3451. 1991.PubMed/NCBI
|