Introduction
Pancreatic cancer is one of the most aggressive and
lethal forms of cancer with an overall 5-year survival rate of less
than 5%. It is currently the fourth cause of death from cancer in
the Western world. Despite this fact, there has been little
improvement in its prognosis over the past 20 years (1). The poor prognosis of pancreatic cancer
can be attributed to the lack of early diagnosis, the aggressive
biological behavior of the tumor and the insensitivity to most
therapies including chemotherapy, radiotherapy and immunotherapy
(2). Moreover, few patients have
the chance to undergo radical surgery because of its poor diagnosis
and those patients with unresectable pancreatic cancer usually
survive for no more than 3–5 months, despite systemic treatment
(3). Currently, gemcitabine remains
the most effectvie agent available for the treatment of advanced
pancreatic cancer, but the disease rapidly develops resistance to
the drug. Thus, novel and efficacious strategies including low
toxic agents that can overcome either the effect of gemcitabine or
chemoresistance to drugs are urgently needed for the treatment for
pancreatic cancer.
Non-toxic phytochemicals offer promising options for
effective chemotherapeutic strategies for various types of cancer.
Some studies support the concept that the consumption of bioactive
dietary phytochemicals reduces the risk of pancreatic cancer
(4,5). Additionally, a number of studies have
shown that various herbal extracts and compounds possess antitumor
activities (6). One of these is
Pulsatilla koreana which belongs to the family
Ranunculaceae. The roots of this plant have been widely used in
traditional medicine for the treatment of various diseases,
including malaria and amoebic dysentery (7). This compound has also been reported to
possess anti-inflammatory and anti-parasitic effects (8). This plant includes various effective
components such as saponins, ranunculin, anemonin, protoanemonin
and triterpenes (9). Specifically,
there are 17 saponins in Pulsatilla koreana, among which,
saponin D has been reported to demonstrate cytotoxicity against
lung cancer cells (10,11). Thus, Pulsatilla saponin D
(hereafter designated as SB365) was selected from many types of
saponins isolated from Pulsatilla koreana for further
investigation. In the present study, we assessed the
chemotherapeutic effect of SB365 in pancreatic cancer cells and
conducted in vivo tumor xenograft studies. Our results
showed that treatment with SB365 resulted in inhibition of cell
growth/proliferation, angiogenesis, and induction of apoptosis in
pancreatic cancer.
Materials and methods
Cells and materials
Human pancreatic cancer cell lines PANC-1,
MIAPaCa-2, BXPC-3, AsPC-1 and HPAC were purchased from the American
Type Culture Collection (AATC, Manassas, VA, USA). PANC-1 and
MIAPaCa-2 cells were cultured in Dulbecco’s modified Eagle’s medium
(DMEM). BXPC-3 and AsPC-1 cells were cultured in Roswell Park
Memorial Institute (RPMI)-1640 medium, and HPAC cells were cultured
in DMEM-F12 supplemented with 10% fetal bovine serum (FBS) and 1%
penicillin/streptomycin. FBS and all other agents used in the cell
culture studies were purchased from Invitrogen Life Technologies
(Carlsbad, CA, USA). The cultures were maintained at 37°C in a
CO2 incubator with a controlled humidified atmosphere
composed of 95% air and 5% CO2.
3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyl tetrazolium bromide (MTT)
and proteinase K were purchased from Sigma-Aldrich (St. Louis, MO,
USA).
Preparation of SB365
SB365 was isolated from the roots of Pulsatilla
koreana, as previously described by Kim et al(11). Briefly, the powdered roots (50 g)
were extracted 3 times using 50% aqueous ethanol (500 ml), and the
residue was then suspended in 300 ml of acetone and centrifuged.
The precipitate was suspended in water and filtered to remove the
insoluble portion. Next the fraction was chromatographed using a
Sephadex LH-20 column (200 g, 60×4 cm) and solid phase high
performance liquid chromatography (HPLC; solid phase; RP-C18,
250×10 mm, mobile phase; MeOH:H2O (82:20) as the mobile
phase, 210 nm, 1 ml/min) to yield SB365, a saponin D. To generate a
more exact analysis of the purified SB365, we also used mass
spectrometry and nuclear magnetic resonance (NMR) spectroscopy.
Measurement of cell proliferation
Cell viability was measured using the MTT assay.
Briefly, PANC-1, BXPC-3, MIAPaCa-2, AsPC-1 and HPAC pancreatic
cancer cells were plated at a density of 1–3×104
cells/well in 96-well plates and then incubated for 48 h. The media
were then removed, and the cells were treated with either dimethyl
sulfoxide (DMSO), as a negative control, or various concentrations
of SB365. The final concentration of DMSO in the media was ≤0.1%
(v/v). After the cells were incubated for 48 h, 20 μl of MTT
solution (2 mg/ml) was added to each well, and the cells were
incubated for another 4 h at 37°C. The formed formazan crystals
were dissolved in DMSO (200 μl/well) with constant shaking for 5
min. The plate was then read on a microplate reader at 540 nm.
Three replicate wells were used for each analysis. The median
inhibitory concentration (IC50, defined as the drug
concentration at which cell growth was inhibited by 50%) was
assessed using the resulting dose-response curves.
Immunofluorescence microscopy
Pancreatic cancer cells (PANC-1 and BXPC-3) were
plated on 18-mm cover glasses in DMEM or RPMI-1640 medium and
incubated for 24 h so that ~70% confluence was reached. The cells
were then incubated in the presence or absence of 10 μM SB365,
washed twice with PBS, and fixed in an acetone:ethanol solution
(1:2) for 10 min at −20°C. Next, cells were blocked in 1.5% horse
serum in PBS for 30 min at room temperature, and then incubated
overnight at 4°C with the primary antibody (1:50) in a humidified
chamber. After washing twice with PBS, the cells were incubated
with mouse fluorescein-labeled secondary antibody (1:100; Dianova,
Hanburg, Germany) for 1 h at 37°C. The cells were also stained with
4,6-diamidino-2-phenylindole (DAPI) to visualize the nuclei. The
slides were then washed twice with PBS and covered with DABCO
(Sigma-Aldrich) before being viewed with a confocal laser scanning
microscope (Olympus, Tokyo, Japan).
DAPI staining and terminal
deoxynucleotidyl-transferase-mediated dUTP nick end labeling
(TUNEL) assay
PANC-1 pancreatic cancer cells were plated onto
18-mm cover glasses in DMEM medium and grown to ~70% confluence for
24 h. The cells were then treated with SB365 at a dose of 10 μM for
24 h. They were fixed in ice-cold 1% paraformaldehyde, washed with
PBS and then stained with 2 μg/ml of DAPI for 20 min at 37°C. The
stained cells were examined for fluorescence of nuclear
fragmentation. TUNEL was performed using the TUNEL kit (Millipore,
Billerica, MA, USA).
Measurement of mitochondrial membrane
potential
BXPC-3 pancreatic cancer cells were plated on 18-mm
cover glasses in RPMI-1640 medium and incubated for 24 h so that
~70% confluence was reached. The cells were then incubated in the
presence or absence of SB365 (10 μM) for 6 h after which they were
incubated with 5 μM JC-1 fluorescence dye for 20 min in a
CO2 incubator. The slides were then washed twice with
PBS and covered with DABCO before being viewed with a confocal
laser scanning microscope.
Analysis of cytochrome c
localization
BXPC-3 cells were plated on 18-mm cover glasses in
RPMI-1640 medium and incubated for 24 h so that ~70% confluence was
reached. The cells were then treated with SB365 (10 μM) for 6 h and
washed twice with PBS, and fixed in an acetic acid:ethanol solution
(1:2) for 5 min at room temperature. Cells were incubated overnight
at 4°C with cytochrome c antibody (1:20; Santa Cruz
Biotechnology, Inc., Santa Cruz, CA, USA). After washing twice with
PBS, the cells were incubated with mouse fluorescein-labeled
secondary antibody (1:50) containing 100 nM of a
mitochondrion-specific dye (MitoTracker® Red FM;
Molecular Probes Inc., Eugene, OR, USA) for 45 min at 37°C. The
cells were also stained with DAPI to visualize the nuclei. The
slides were then washed twice with PBS and covered with DABCO
before viewing with a confocal laser scanning microscope.
Western blotting
BXPC-3 cells were washed 3 times with ice-cold PBS
before lysis using a buffer containing 1% Triton X-100, 1% Nonidet
P-40, and the following protease and phosphatase inhibitors:
aprotinin (10 mg/ml), leupeptin (10 mg/ml) (ICN Biomedicals,
Asse-Relegem, Belgium), phenylmethylsulfonyl fluoride (1.72 mM),
NaF (100 mM), NaVO3 (500 mM) and
Na4P2O7 (500 mg/ml)
(Sigma-Aldrich). Equal amounts of protein were separated using 10%
sodium dodecyl sulfate-polyacrylamide gel electrophoresis and
transferred onto nitrocellulose membranes. The protein transfer was
then checked using Ponceau S staining solution (Sigma-Aldrich).
Immunostaining of the blots was performed using the primary
antibodies, followed by the secondary antibody-conjugated to
horseradish peroxidase with detection using enhanced
chemiluminescence reagent (Amersham Biosciences, Piscataway, NJ,
USA). Primary antibodies were purchased as follows: Bcl-2 (Santa
Cruz Biotechnology Inc.), β-actin (Abcam, Cambridge, UK), cleaved
caspase-3 (Cell Signaling Technology, Inc., Beverly, MA, USA). The
secondary antibodies were purchased from Amersham Biosciences. The
bands were visualized with the ECL Plus system (Amersham
Biosciences).
Tumor sphere assay
PANC-1 cells were seeded in a 24-well low adhesion
plate (Corning Inc., Lowell, MA, USA) at a density of
1×104 cells/well and an initial volume of 500 ml as
described by Dontu et al(12). Spheres were grown in DMEM medium
(Gibco-BRL, Carlsbad, CA, USA) supplemented with vitamin B27, 20
ng/ml FGF, and 20 ng/ml epidermal growth factor (EGF; R&D
Systems, Minneapolis, MN, USA) and 4 μg/ml heparin. Samples were
subsequently treated with 10 μM SB365. EGF and FGF were replenished
every 3 days. Aside from the addition of heparin, cells were
agitated daily to minimize clumping. Spheres were counted and
photographed after 14 days of continuous growth factor and
supplemental exposure. Spheres were monitored under the microscope
daily to ensure that they were derived from single cells and that
they did not become confluent during the experiment.
Tumor xenograft study
Male athymic BALB/c nude mice were obtained from
Orient Bio. Animal Inc. (Seoul, Korea). Animal care and all
experimental procedures were conducted in accordance with the
approval and guidelines of the INHA Institutional Animal Care and
Use Committee (INHA IACUC) of the Medical School of Inha University
(approval ID: 090518-5). The animals were fed standard rat chow and
tap water ad libitum, and maintained under a 12 h dark/light
cycle at 21°C. Male nude mice (6 weeks old, weighing 22–26 g) were
randomly divided into 2 groups (control and SB365 30 mg/kg). PANC-1
cells were harvested and mixed with PBS (200 μl/mouse) and then
inoculated into one flank of each nude mouse (5×106 of
PANC-1 cells). When the tumors had reached a volume of ~50–100
mm3, mice were given an intraperitoneal injection of
SB365 (30 mg/kg, treatment group) or the vehicle (200 μl PBS,
control group) for 37 days. The tumor dimensions were measured
twice a week using a digital calliper and the tumor volume (V) was
calculated using the formula: V = length × width2 × 0.5.
At the end of the experiment, mice were sacrificed, and tumors were
excised and weighed.
Immunohistochemistry
After being blocked with normal goat serum (Vector
Laboratories, Burlingame, CA, USA) for 1 h, frozen tissue sections
were incubated for 1 h at room temperature in dilutions of 1:100 of
PCNA, cleaved caspase-3, VEGF and CD34 antibodies. The sections
were visualized by an avidin-biotin peroxidase complex solution
using an ABC kit (Vector Laboratories). The sections were washed in
PBS and developed with a diaminobenzidine tetrahydrochloride
substrate for 15 min and then counterstained with hematoxylin.
Statistical analysis
Data are expressed as the means ± SD. Statistical
analysis was performed using ANOVA. A P≤0.05 was considered to
indicate a statistically significant result. Statistical
calculations were performed using SPSS software for Windows
operating system (version 10.0; SPSS, Inc., Chicago, IL, USA).
Results
Effect of SB365 on the proliferation of
human pancreatic cancer cells
To evaluate the anticancer properties of SB365, we
first compared the cell growth of 5 pancreatic cancer cell lines
(MIAPaCa-2, BXPC-3, PANC-1, AsPC-1 and HPAC cell lines) that had
been treated with SB365. As shown in Fig. 1A, the cells were exposed to various
concentrations (0, 1, 2, 5, 10 and 20 μM) of SB365 for 48 h. The
results revealed that SB365 treatment inhibited cell growth in a
dose-dependent manner. SB365 induced a reduction in cell growth,
starting at a dose of 1 μM in pancreatic cancer cells and strongly
inhibited up to 80% of cell growth at doses of 5 and 10 μM. To
confirm the effect of SB365 on cell growth and proliferation, we
identified expression of Ki-67, a cell proliferation marker using a
fluorescent imaging system. As shown in Fig. 1B, SB365 significantly decreased the
number of Ki-67-positive cells, when compared with the control.
Effects of SB365 on the apoptotic cell
death of pancreatic cancer cells
To identify the pro-apoptotic effect of SB365 on
pancreatic cancer cells, we first conducted DAPI staining and TUNEL
assay. As shown in Fig. 2A, the
cells treated with 10 μM of SB365 showed morphological features of
apoptotic cells, such as bright nuclear condensation and
perinuclear apoptotic bodies, upon DAPI staining. SB365-induced
apoptosis was confirmed by detection of DNA fragmentation using
TUNEL staining. To identify involvement of SB365 in changes in the
mitochondrial membrane potential (ΔΨm), which are correlated with
intrinsic apoptosis, we also performed JC-1 staining. As shown in
Fig. 2B, control cells showed
heterogeneous staining of the cytoplasm with both red and green
fluorescence coexisting in the same cells. Consistent with
mitochondrial localization, red fluorescence (corresponding to high
mitochondrial membrane potential) was primarily found in granular
structures distributed throughout the cytoplasm. Treatment of
PANC-1 and BXPC-3 cells with SB365 decreased the red fluorescence
and led to occurrence of frequent clusters of mitochondria.
Treatment of SB365 induced marked changes in ΔΨm evidenced by the
disappearance of red fluorescence or the increase in green
fluorescence in most cells. In addition, we observed that SB365 led
to increased cytochrome c release along with a concomitant
decrease in the-co-localization of cytochrome c and
mitochondria (Fig. 2C).
Furthermore, SB365 increased the protein expression levels of
cleaved caspase-3 and decreased Bcl-2 expression in BXPC-3
pancreatic cancer cells (Fig. 2D).
Collectively, these results indicate that SB365 induced apoptosis
of pancreatic cancer cells.
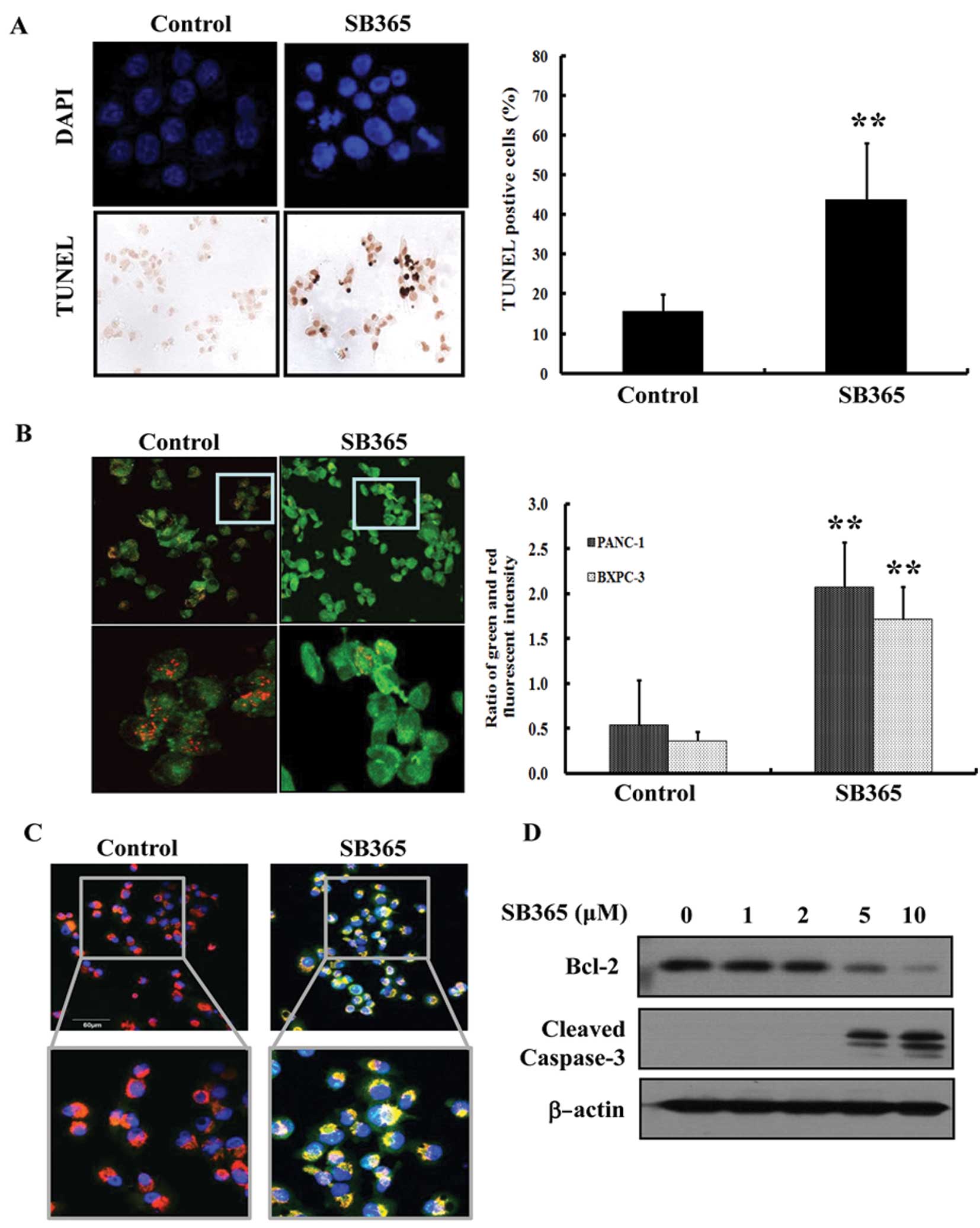 | Figure 2Effect of SB365 on the apoptosis of
pancreatic cancer cells. (A) Induction of apoptosis in PANC-1 cells
by SB365 (10 μM) was determined by 4,6-diamidino-2-phenylindole
(DAPI) and terminal deoxynucleotidyl-transferase-mediated dUTP nick
end labeling (TUNEL) staining, which was photographed at ×400
magnification. Data are expressed as the means ± SD from the 3
experiments. **P<0.01 vs. control. (B) PANC-1 and
BXPC-3 cells were treated with SB365 (10 μM) for 6 h, and the
mitochondrial membrane potential was determined by JC-1 staining
and analyzed using an LS 55 luminescence spectrometer, as well as
photographed at ×400 and ×800 magnification (PANC-1 cells). The
results (green/red ratio) are expressed as the percentage of cells
treated with SB365. Data are expressed as the means ± SD of
triplicate wells. **P<0.01 vs. control. (C) After
treatment with SB365 (10 μM) for 6 h, BXPC-3 cells were stained
with anti-cytochrome c antibody, MitoTracker and DAPI. The
immunostained cells were analyzed under an Olympus confocal laser
scanning microscope at ×400 and ×800 magnification (D) PANC-1 cells
were treated with SB365 (0, 1, 2, 5 and 10 μM) for 48 h. Cell
lysates were prepared and subjected to western blotting for Bcl-2,
cleaved caspase-3 and β-actin. |
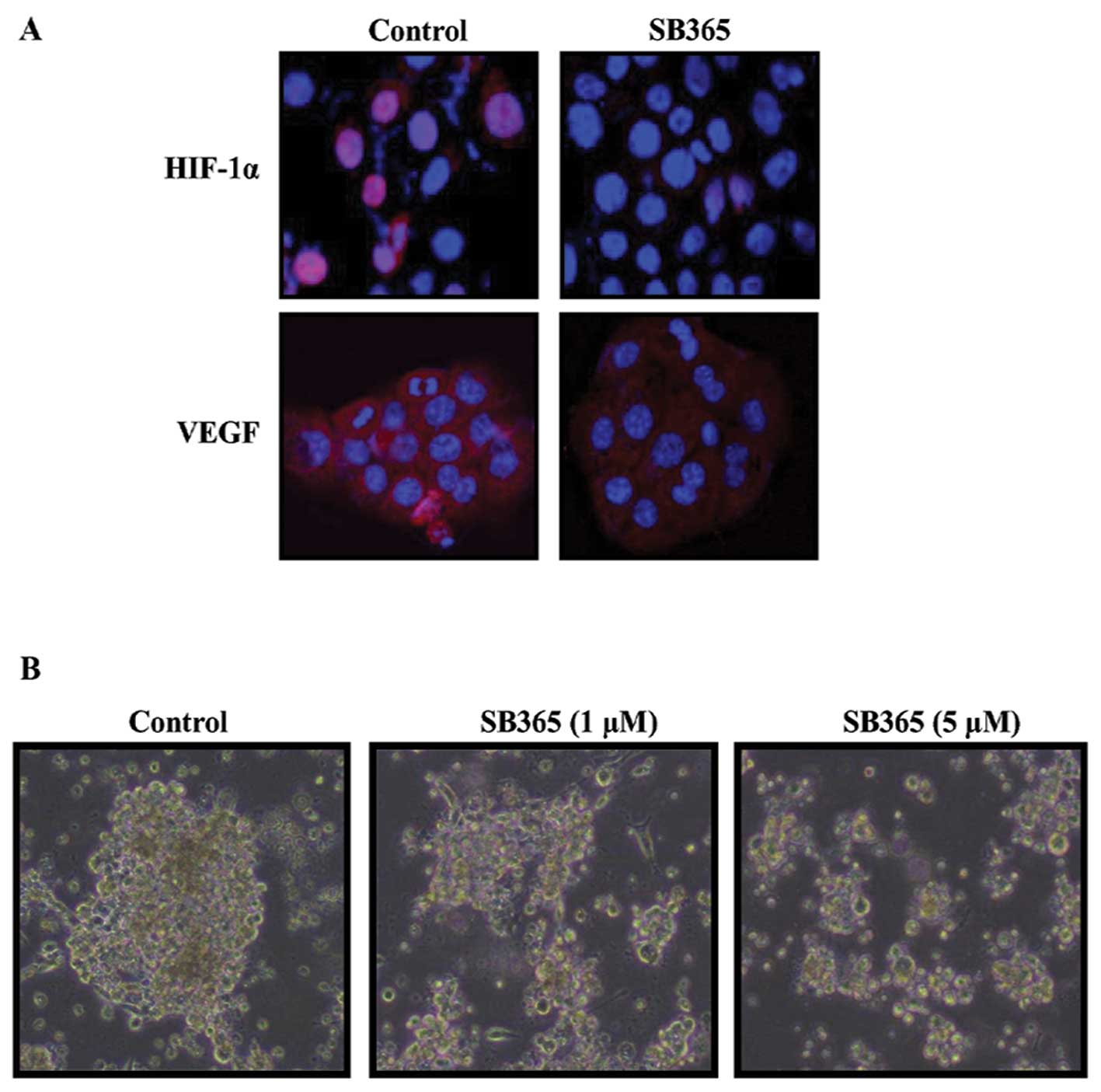
Effects of SB365 on the expression of
HIF-1α/VEGF and tumor sphere formation of pancreatic cancer
cells
Considering the importance of HIF-1α and VEGF in
hypoxia, we examined the effects of SB365 on the expression pattern
of HIF-1α and VEGF in BXPC-3 cells. BXPC-3 pancreatic cancer cells
were treated with SB365 (10 μM) under mimicked hypoxia induced by
treatment with 100 μM CoCl2 for 24 h. As shown in
Fig. 3A, the HIF-1α and VEGF
expression increased under hypoxic condition, whereas SB365
treatment inhibited the hypoxia-induced HIF-1α and VEGF expression.
Moreover, to test for the ability of pancreatic cancer cells to
grow as tumor spheres in culture, PANC-1 cells were plated on
ultra-low attachment plates in defined medium containing EGF, FGF
and B27. After 2 weeks, tumor spheres consisting of ~50–100 cells
were observed, ~50% of which had symmetrical ball-like structures
(Fig. 3B). However, treatment of
SB365 inhibited the formation of tumor spheres of pancreatic cancer
cells in a dose-dependent manner.
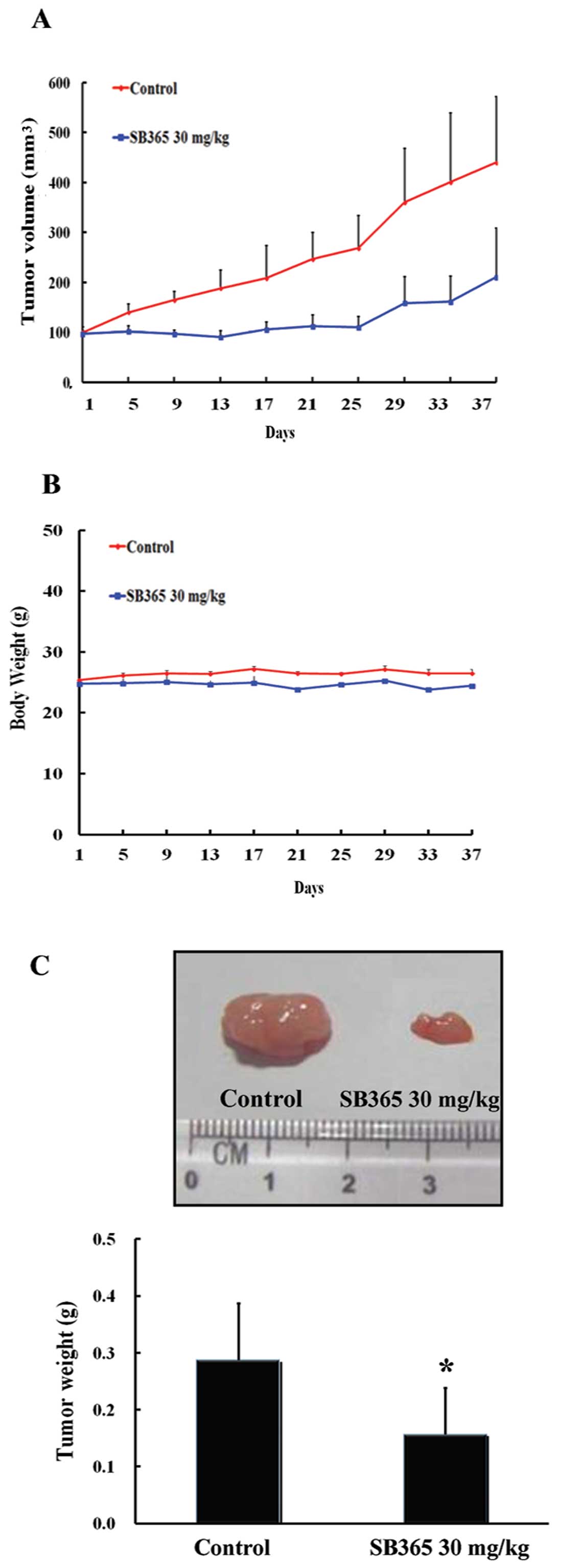
Inhibition of pancreatic tumor growth by
SB365 in a mouse xenograft model
Strong anticancer efficacy of SB365 led us to
investigate the in vivo efficacy of SB365 against xenografts
of PANC-1 pancreatic cancer cells in nude mice. As shown in
Fig. 4A, SB365 suppressed tumor
growth at a dose of 30 mg/kg for 37 days compared with the control
group. SB365 administration resulted in a significant reduction in
tumor volume (55%) in nude mice. In addition, there was no
difference in the body weight in the mouse groups treated with
SB365 when compared with the control group, showing that SB365
exhibited little toxicity to mice at the curative dose (Fig. 4B). Consistent with this observation,
the weight of tumors isolated from the SB365-treated mouse groups
decreased significantly by 51% in the SB365 30 mg/kg group,
compared with the control (Fig. 4C,
P<0.05). Taken together, our results demonstrated that SB365 had
strong antitumor efficacy against pancreatic cancer xenograft
models.
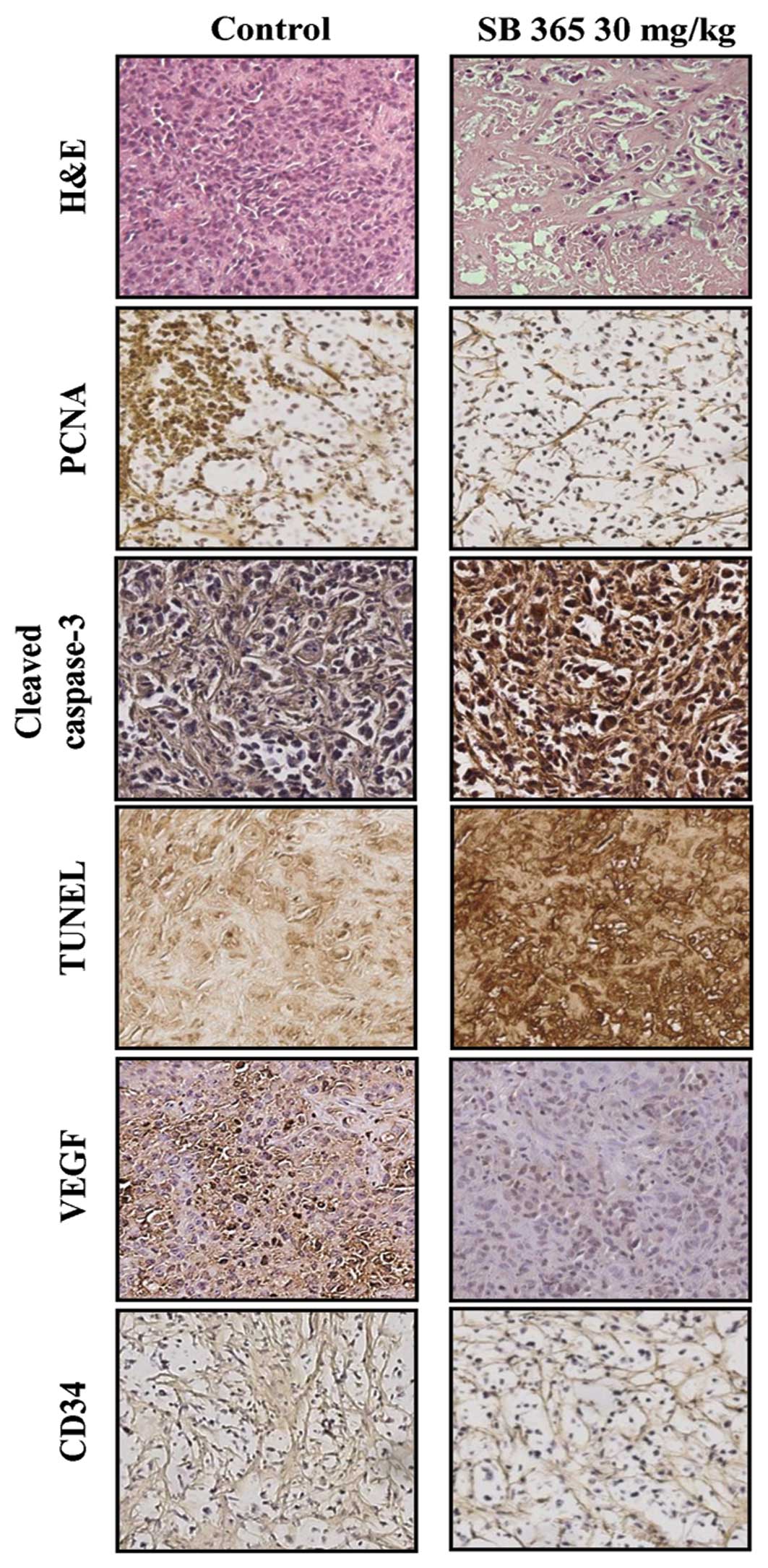
Effects of SB365 on proliferation,
angiogenesis and apoptosis in a xenograft model
Based on histopathological analysis using
hematoxylin and eosin (H&E) staining, we observed a greater
degree of tumor apoptosis and necrosis in the SB365-treated mouse
group when compared to the control groups. Furthermore, SB365
decreased the expression of PCNA, a cell proliferation marker, as
well as both VEGF and CD34 in the SB365-treated group compared with
the control group. In addition, the apoptotic effect of SB365 on
pancreatic cancer tumor tissues was identified by expression of
cleaved caspase-3 and DNA fragments by TUNEL assay. These results
suggest that SB365 had an anti-angiogenic effect on pancreatic
cancer xenografts (Fig. 5).
Discussion
Owing to the advanced stage of the disease and
resistance to cytotoxic chemotherapeutic agents and radiation
therapy, pancreatic cancer remains one of the most intractable
human malignancies. In recent years, bioactive components from
plant origin have obtained considerable attention as promising
options for the development of effective strategies for the
prevention or treatment of cancer. Accordingly, the discovery of
active medicinal compounds from herbal/natural sources has provided
alternative treatment choices for patients (13). Therefore, we isolated SB365, saponin
D from Pulsatilla koreana and conducted a comprehensive
analysis of the effect of SB365 on pancreatic cancer using both
in vivo and in vitro models. In the present study, we
report that SB365 significantly reduced cell growth and
proliferation while inducing apoptosis in pancreatic cancer cells.
This is the first report validating the anticancer properties of
SB365 in both pancreatic cancer cells and a tumor xenograft animal
model.
Loss of cell proliferation and/or induction of
apoptosis are two major mechanisms by which cancer cells are killed
by chemotherapeutic agents (14).
In this study, SB365 inhibited 50–80% of the growth of pancreatic
cancer cells (MIAPaCa-1, BXPC-3, PANC-1, AsPC-1 and HPAC cells) at
concentrations of 2–10 μM, and the level of Ki-67-positive cells
was also significantly decreased by SB365 treatment, indicating
that SB365 has anti-proliferative effect. These results are in line
with those of previous studies, in which many types of saponins
were found to inhibit cell proliferation in various types of cancer
cells (15–18).
While the effect of SB365 on proliferation likely
contributes to its anticancer activity, it appears that pancreatic
cancer cells are more sensitive to apoptosis induced by SB365. In
fact, the IC50 values reflect the overall change in cell
viability, which includes changes in both proliferation and
apoptosis. Indeed, SB365 showed low IC50 values (0.8–2
μM) in 5 pancreatic cancer cell lines. Therefore, we investigated
whether SB365 had an apoptotic effect in pancreatic cancer. In our
study, we observed that SB365 has the capacity to induce pancreatic
cancer cells to undergo apoptosis. DNA fragmentation and nuclear
chromatin condensation were demonstrated in pancreatic cancer cells
treated with SB365. Moreover, the observation of caspase-3
activation and decreased Bcl-2 confirms the promotion of apoptosis
by SB365. In addition, changes in the mitochondrial membrane
potential are induced to create pores of mitochondria, which
dissipates the transmembrane potential and leads to the release of
cytochrome c into the cytoplasm (19,20),
finally resulting in the induction of apoptosis. Therefore, to
identify the effect of SB365 on mitochondrial membrane potential,
we conducted JC-1 and cytochrome c immunofluorescence
staining. SB365 induced significant changes in mitochondrial
membrane potential (ΔΨm) and increased cytochrome c release
along with a concomitant increase in co-localization of cytochrome
c with mitochondria. Collectively, these results indicate
that SB365 induced an apoptotic effect by mitochondrial-mediated
apoptotic molecules against pancreatic cancer cells.
Angiogenesis is a process that plays an important
role in the growth and metastasis of solid tumors (21). The inhibition of angiogenic pathways
is also an alternative for targeting cancer cell proliferation. In
this process, VEGF is a potent inducer of angiogenesis and HIF-1α
is the major regulator of VEGF transcriptional activation (22,23).
In the present study, SB365 obviously decreased the expression of
HIF-1α and VEGF under hypoxia, showing that SB365 inhibited
hypoxia-induced angiogenesis in pancreatic cancer. In our previous
study, SB365 inhibited not only tube formation and migration of
HUVECs but also the formation of neovessels in Matrigel plug
(24). Based on these findings, we
expected that the anti-angiogenic capacity of SB365 would accompany
inhibition of metastasis, which is associated with tumor formation
and migration. Indeed, bursts of angiogenic activity can be
sufficient to initiate sustained tumor angiogenesis and metastasis
(25). To grow beyond a small
sphere, tumor cells must be able to invade across a barrier known
as the basement membrane and into local tissue. At this point, they
can also produce angiogenic factors such as VEGF that recruit new
blood vessel formation (26).
Moreover, when tumor cells are allowed to grow to a large size,
these new vessels support tumor metastasis (27). Based on this knowledge, we evaluated
whether SB365 inhibits pancreatic tumor sphere formation.
Importantly, we found that SB365 obviously inhibited tumor sphere
forming in a population of pancreatic cancer cells with tumor
sphere-forming ability. These findings indicate that SB365
demonstrated potent anti-angiogenic effects along with the
potential for inhibition of metastasis.
Most importantly, our in vitro results were
corroborated in vivo in a pancreatic cancer xenograft model.
Specifically, SB365 inhibited tumor growth in the mouse xenograft
models. These data are in accordance with the decreased
proliferation and angiogenesis documented by PCNA, and CD34 and
VEGF immunostaining, respectively. Additionally, SB365 induced
apoptosis as indicated by increased cleaved caspase-3 and TUNEL
staining in tumor tissues. These in vivo results are similar
to our in vitro study, which further suggests that SB365 may
be an applicable approach to enhance the antitumor activity against
pancreatic cancer.
In conclusion, the present study demonstrated that
SB365 potentiated the anticancer effect against pancreatic cancer
by inducing apoptosis as well as inhibiting cell
growth/proliferation and angiogenesis. Based on our findings, SB365
is a potential therapeutic agent against pancreatic cancer.
However, further investigation to explore possible clinical uses of
SB365 as a natural compound is needed in terms of therapeutic
strategy.
Acknowledgements
This study was supported by the Korean Health
Technology R&D Project (A120266), the Ministry of Health and
Welfare and the National Research Foundation of Korea (NRF) funded
by the Ministry of Education, Science and Technology (NRF
2012-0002988, 2012R1A2A2A01045602, 2012R1A1A2043281) and the Inha
University Grant.
References
|
1
|
Li D, Xie K, Wolff R and Abbruzzese JL:
Pancreatic cancer. Lancet. 363:1049–1057. 2004. View Article : Google Scholar
|
|
2
|
Saif MW: Adjuvant therapy of pancreatic
cancer: beyond gemcitabine. In: Highlights from the ‘2011 ASCO
Gastrointestinal Cancers Symposium’; San Francisco, CA, USA.
January 20–22, 2011; JOP. 12. pp. 106–109. 2011, PubMed/NCBI
|
|
3
|
Theissen G, Pardon B and Wagner R: A
quantitative assessment for transcriptional pausing of
DNA-dependent RNA polymerases in vitro. Anal Biochem. 189:254–261.
1990. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Block G, Patterson B and Subar A: Fruit,
vegetables, and cancer prevention: a review of the epidemiological
evidence. Nutr Cancer. 18:1–29. 1992. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Bueno de Mesquita HB, Maisonneuve P,
Moerman CJ, Runia S and Boyle P: Life-time history of smoking and
exocrine carcinoma of the pancreas: a population-based case-control
study in The Netherlands. Int J Cancer. 49:816–822. 1991.
|
|
6
|
Cassileth BR: Alternative and
complementary medicine. Separating the wheat from the chaff.
Cancer. 86:1900–1902. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Bae K: The Medicinal Plants of Korea.
Kyu-Hak Press; Seoul: pp. 1391999
|
|
8
|
Ye WC, Ou BX, Ji NN, Zhao SX, Ye T,
McKervey MA and Stevenson P: Patensin, a saponin from Pulsatilla
patens varmultifida. Phytochemistry. 39:937–939. 1995.
View Article : Google Scholar
|
|
9
|
Ye WC, Ji NN, Zhao SX, Liu JH, Ye T,
McKervey MA and Stevenson P: Triterpenoids from Pulsatilla
chinensis. Phytochemistry. 42:799–802. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Bang SC, Lee JH, Song GY, Kim DH, Yoon MY
and Ahn BZ: Antitumor activity of Pulsatilla koreana
saponins and their structure-activity relationship. Chem Pharm
Bull. 53:1451–1454. 2005.PubMed/NCBI
|
|
11
|
Kim Y, Bang SC, Lee JH and Ahn BZ:
Pulsatilla saponin D: the antitumor principle from
Pulsatilla koreana. Arch Pharm Res. 27:915–918. 2004.
View Article : Google Scholar
|
|
12
|
Dontu G, Abdallah WM, Foley JM, Jackson
KW, Clarke MF, Kawamura MJ and Wicha MS: In vitro propagation and
transcriptional profiling of human mammary stem/progenitor cells.
Genes Dev. 17:1253–1270. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Smith M and Boon HS: Counseling cancer
patients about herbal medicine. Patient Educ Couns. 38:109–120.
1999. View Article : Google Scholar
|
|
14
|
Li Y, Ahmed F, Ali S, Philip PA, Kucuk O
and Sarkar FH: Inactivation of nuclear factor κB by soy isoflavone
genistein contributes to increased apoptosis induced by
chemotherapeutic agents in human cancer cells. Cancer Res.
65:6934–6942. 2005.
|
|
15
|
Li F, Fernandez PP, Rajendran P, Hui KM
and Sethi G: Diosgenin, a steroidal saponin, inhibits STAT3
signaling pathway leading to suppression of proliferation and
chemosensitization of human hepatocellular carcinoma cells. Cancer
Lett. 292:197–207. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Peng L, Zhou Y, Kong de Y and Zhang WD:
Antitumor activities of dammarane triterpene saponins from
Bacopa monniera. Phytother Res. 24:864–868. 2010.PubMed/NCBI
|
|
17
|
Tin MM, Cho CH, Chan K, James AE and Ko
JK: Astragalus saponins induce growth inhibition and
apoptosis in human colon cancer cells and tumor xenograft.
Carcinogenesis. 28:1347–1355. 2007. View Article : Google Scholar
|
|
18
|
Chen PS, Shih YW, Huang HC and Cheng HW:
Diosgenin, a steroidal saponin, inhibits migration and invasion of
human prostate cancer PC-3 cells by reducing matrix
metalloproteinases expression. PLoS One. 6:e201642011. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Petronilli V, Penzo D, Scorrano L,
Bernardi P and Di Lisa F: The mitochondrial permeability
transition, release of cytochrome c and cell death.
Correlation with the duration of pore openings in situ. J
Biol Chem. 276:12030–12034. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Armstrong JS: Mitochondria: a target for
cancer therapy. Br J Pharmacol. 147:239–248. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Folkman J: Angiogenesis. Annu Rev Med.
57:1–18. 2006. View Article : Google Scholar
|
|
22
|
Xia C, Meng Q, Cao Z, Shi X and Jiang BH:
Regulation of angiogenesis and tumor growth by p110 alpha and AKT1
via VEGF expression. J Cell Physiol. 209:56–66. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Jiang BH, Rue E, Wang GL, Roe R and
Semenza GL: Dimerization, DNA binding, and transactivation
properties of hypoxia-inducible factor 1. J Biol Chem.
271:17771–17778. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Hong SW, Jung KH, Lee HS, Choi MJ, Son MK,
Zheng HM and Hong SS: SB365 inhibits angiogenesis and induces
apoptosis of hepatocellular carcinoma through modulation of
PI3K/Akt/mTOR signaling pathway. Cancer Sci. 103:1929–1937. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Indraccolo S, Stievano L, Minuzzo S,
Tosello V, Esposito G, Piovan E, Zamarchi R, Chieco-Bianchi L and
Amadori A: Interruption of tumor dormancy by a transient angiogenic
burst within the tumor microenvironment. Proc Natl Acad Sci USA.
103:4216–4221. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Hoeben A, Landuyt B, Highley MS, Wildiers
H, Van Oosterom AT and De Bruijn EA: Vascular endothelial growth
factor and angiogenesis. Pharmacol Rev. 56:549–580. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Alitalo A and Detmar M: Interaction of
tumor cells and lymphatic vessels in cancer progression. Oncogene.
31:4499–4508. 2012. View Article : Google Scholar : PubMed/NCBI
|