Introduction
Breast cancer is the most common malignant tumor
noted among women. The incidence rate is consistently increasing
with a trend of younger age at diagnosis (1). The incidence of breast cancer involves
a complex biological process, and the mechanism involves multi-gene
mutations. The current therapeutic strategy for breast cancer
consists of comprehensive treatment modalities that include
surgery, chemotherapy, radiotherapy and endocrine therapy. Yet, the
rates of recurrence and metastasis are still high. Particularly,
patients presenting with advanced stage tumors or those suffering
from poor health condition and with a low indication for surgical
operation, are in need of effective therapeutic methods. Gene
therapy is a promising strategy for the treatment of malignancies.
Yet, single suicide gene therapy has drawbacks. Combination gene
therapy has the added advantages of gene therapy, elevates
therapeutic efficacy and overcomes the shortcomings of single gene
therapy (2,3). Thus, combination gene therapy has
become a new direction for the development of gene therapy.
Cytosine deaminase/5-fluorocytosine (CD/5-FC) and thymidine
kinase/ganciclovir (TK/GCV) are the most common suicide gene
therapy treatment systems (4,5). The
fusion gene CD/TK is a new suicide gene exhibiting better
therapeutic effects (6). Based on
the the overexpression of the vascular endothelial growth factor
(VEGF) in breast cancer cells and the absence of its expression in
normal tissues (7–9), the present study applied the CD/TK
fusion suicide gene system driven by the VEGF promoter to MCF-7
tumor cells. The adenovirus vector efficiently infected and killed
the target cells and induced breast cancer cell apoptosis. The
introduction of the VEGF gene promoter ensured the target of the
treatment. The present study provides an experimental basis for
further application of double suicide gene therapy strategies.
Materials and methods
Recombinant adenovirus vector
The recombinant adenovirus carrying the VEGF
promoter and the CD and TK genes (Ad-VEGFP-CD/TK) was constructed
and preserved by our group in the Department of General Surgery,
Zhujiang Hospital, Southern Medical University, Guangzhou
Guangdong, China. The vector expressing green fluorescence protein
was also used. The titer of the purified recombinant adenoviruses
was as high as 2.0×1011 pfu/ml.
Cells and cell culture
The human breast cancer cell line MCF-7 was obtained
from the American Type Culture Collection (ATCC, Manassas, VA,
USA). Cell culture medium, Dulbecco’s modified Eagle’s medium
(DMEM), fetal bovine serum (FBS), penicillin-streptomycin-glutamine
(x100) and 0.5% trypsin-EDTA solution were purchased from
Invitrogen (Carlsbad, CA, USA). As described previously (10), the MCF-7 cell lines were maintained
in DMEM supplemented with 10% FBS, 50 IU/ml penicillin and 50 μg/ml
streptomycin and were grown at 37°C in a humidified atmosphere with
5% CO2.
Transfection
MCF-7 cells were inoculated in 24-well plates at a
density of 2×105 cells/well. After 12 h, the cells were
infected with Ad-VEGFP-CD/TK at multiplicities of infection (MOI)
of 1, 10, 20, 40, 60, 80, 100 and 200 pfu/cell and then incubated
for an additional 48 h. To estimate the optimum MOI of the
adenoviral vector in MCF-7 cells, GFP fluorescence was observed
under a microscope using a fluorescence light source and a 485-nm
filter (Leica, Germany).
Detection of CD/TK gene expression
Expression of the CD/TK gene in the infected MCF-7
cells was analyzed by RT-PCR. Total RNA from the treated MCF-7
cells was prepared with TRIzol reagent (Invitrogen, La Jolla, CA,
USA) according to the manufacturer’s protocol and digested with
RNase-free DNase to clear residual genomic DNA. RT-PCR was
performed with specific primers from corresponding templates of
CD/TK (sense, 5′-AGGCTAACAGTGTCGAATAACGCT-3′ and antisense,
5′-GTTAGCCTCCCCCATCTC-3′) (a DNA fragment of 2,400 bp). For
internal control, β-actin (sense, 5-CGGAGTCAA CGGATTTGGTGGTAT-3 and
antisense, 5-AGCCTTCTC CATGGTGGTGAAGAC-3) was co-amplified with
CD/TK (a DNA fragment of 306 bp). Before reverse transcription
(RT), residual genomic DNA was removed by treatment with RQ DNase
(Promega, Madison, WI, USA). Total RNA (750 μg) was used to
synthesize cDNA in 9 μl of reaction mixture by standard protocol
with oligo(dT)15 as primer and AMV reverse transcriptase
(Promega). PCR reactions were carried out with 10 μl of RT products
in 40 μl of reaction system using Taq DNA polymerase. After
an initial denaturation at 94°C for 5 min, 30 cycles of
amplification were performed under the following conditions: 94°C
for 40 sec, 57°C for 60 sec and 72°C for 90 sec. PCR products were
run on 1.5% agarose gel (containing 0.5 mg of ethidium
bromide/liter).
Sensitivity of the cells to GCV and
5-FC
Adenoviral-infected and -uninfected parental cells
were plated at a density of 103 cells/well with various
concentrations (0.001, 0.01, 0.1, 1, 10, 100 and 1,000 mg/l) of GCV
or various concentrations of (10, 20, 40, 80, 160, 320 and 640
mg/l) 5-FC (both from Syntex, Palo Alto, CA, USA) or both agents in
96-well flat-bottomed culture plates (Costar, Bethesda, MD, USA).
After incubation for 2 days, the sensitivity of the cells to the
prodrugs was evaluated using the tetrazolium salt
3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT)
(Sigma, St. Louis, MO, USA) conversion assay. The viability of the
cells was determined by comparing the number of viable cells with
and without GCV and 5-FC.
Transmission electron microscopy
MCF-7 cells were cultured with the adenoviral vector
and the prodrugs and incubated for 48 h at 37°C. The cultured cells
were harvested using trypsin and centrifuged for 10 min at 3,500
rpm and room temperature. The pellets were fixed in 4% (v/v)
glutaraldehyde in 0.1 M coccadylate buffer (pH 7.4) for 4 h at 4°C.
The fixed cells were centrifuged and the pellets were blocked in
serum which was later fixed in glutaraldeyde overnight at 4°C. The
specimens were washed in three changes of sodium coccadylate buffer
(pH 7.4) for 10 min each, postfixed in 1% osmium tetraoxide at 4°C.
The specimens were then washed in three changes of sodium
coccadylate buffer (pH 7.4) for 10 min each and dehydrated with a
graded series of acetone (35, 50, 75, 95 and 100%). The cells were
then infiltrated with acetone and resin and embedded with 100%
resin in beam capsule and left to polymerize at 60°C for 48 h. The
area of interest in the embedded cell resin block was chosen using
the toulidine blue staining and later examined using light
microscope. The selected area was cut in ulltrathin sections using
an ultramicrotome. The sections were placed into a grid and stained
with uranyl acetate for 10 min followed by 50% filtered acetone and
finally stained using lead which was then washed twice with
distilled water. The stained samples were then viewed under
transmission electron microscopy (Phillips, Eindhoven, The
Netherlands).
Flow cytometric analysis
The experiments were divided into four groups:
control group, the low concentration group (0.1 mg/l+40 mg/l)
(GCV+5-FC), the medium concentration group (1+80 mg/l) and high
concentration group (10+160 mg/l) for 48 h. To quantitatively
measure the percentage of cells undergoing apoptosis, 100 ml of the
cells (2×105) suspended in 1X binding buffer was
transferred to a 5-ml culture tube. Five microliters of
FITC-conjugated Annexin V and 1 ml of 50 mg/ml PI reagents (Annexin
V-FITC kit; BD Pharmingen, USA) were added and then incubated for
15 min at room temperature in the dark. After the addition of 400
ml 1X binding buffer, flow cytometric analysis was performed within
1 h (FACSCaliber; Becton-Dickinson, USA) according to the
manufacturer’s instructions (11).
Caspase activity
Caspase-3 and -8 activities were measured using
colorimetric assay kits (R&D Systems). After transduction of
the MCF-7 cells with the adenoviral vector and subsequent
incubation with the prodrugs 5-FC and GCV for 48 h, cells were
washed with ice-cold PBS, and caspase-3 and -8 activities were
determined using the kit following the manufacturer’s
recommendations. The control uninfected MCF-7 cells were accepted
the same treatment. Caspase colorimetric substrates DEVD-pNA
(caspase-3) or IETDpNA (caspase-8) were added to the cell lysate,
and assays were performed in a 100-μl volume in 96-well
flat-bottomed plates. Chromophore p-nitroanilide was released as a
result of cleavage of the substrates by caspase activity. The
caspase enzymatic activity in the cell lysate was directly
proportional to the chromophore formation, which was quantified
spectrophotometrically at a wavelength of 405 nm using a microplate
reader. Data were corrected for the background values that had no
substrate or no cell lysate treatment.
Results
Effective infection of the adenoviral
vector in the MCF-7 cells
To determine the infection efficiency of the
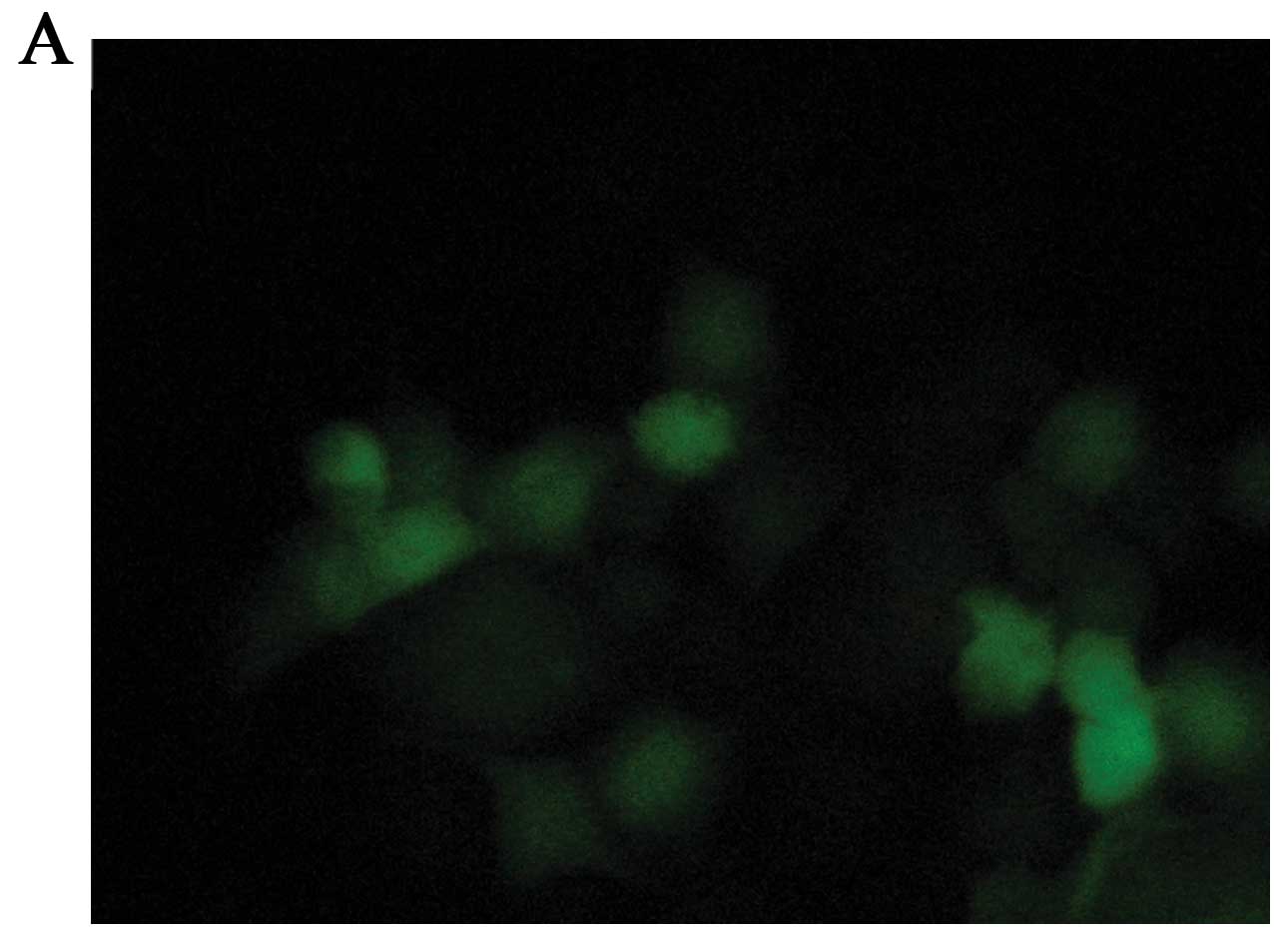
adenoviral vector in MCF-7 cells, we infected the MCF-7 cells with
the Ad-vector at a MOI ranging from 1 to 200 pfu/cell. Fig. 1 shows different transient expression
of the recombinant GFP genes in MCF-7 cells. Preliminary titration
revealed a high transfer efficiency of the recombinant adenovirus
in MCF-7 cells and an optimal expression at 48 h post-infection. At
a MOI of 100 pfu/cell, >95% MCF-7 cells were GFP-positive
without obvious adenoviral toxicity (Fig. 1B). Therefore, we performed most of
our studies at 48 h of incubation using the optimal MOI of 100
pfu/cell.
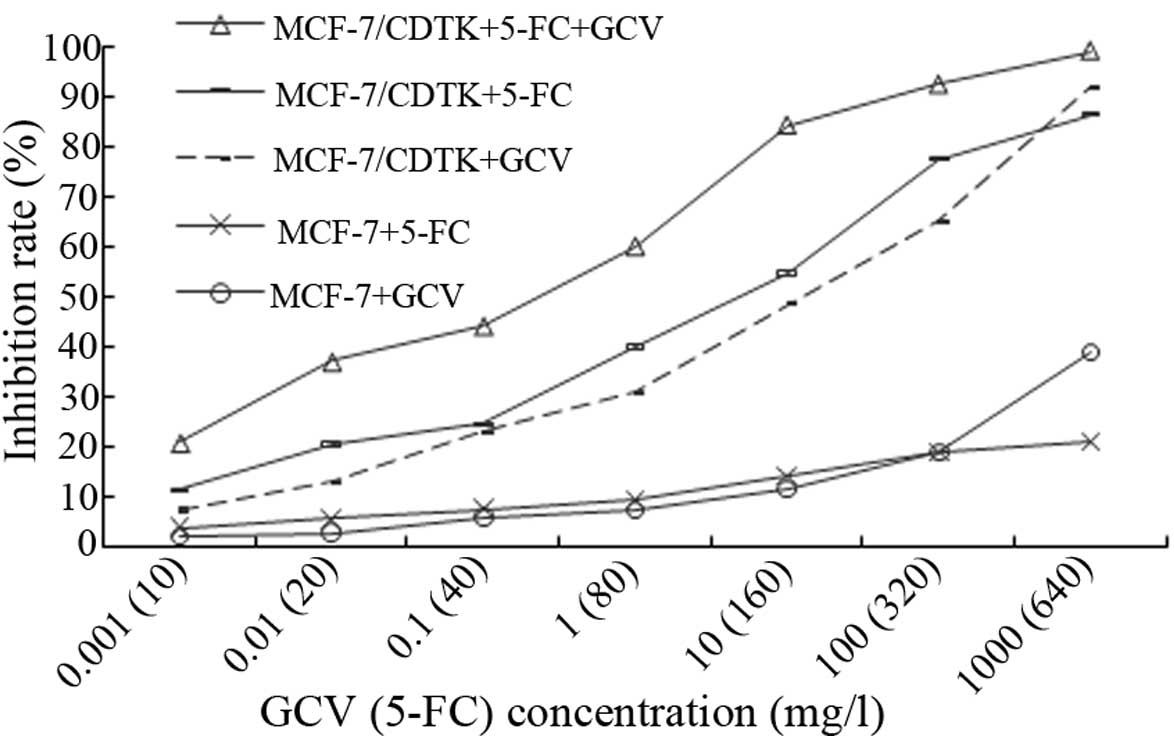
CD/TK gene expression in adv-infected
MCF-7 cells
We used RT-PCR to detect the expression of the CD/TK
gene. A 2,400-bp fragment of the CD/TK gene was observed, which was
consistent with predicted results (Fig.
2). As expected, without CD/TK expression, the growth of the
parental MCF-7 cells was not infected by the adenoviral vector.
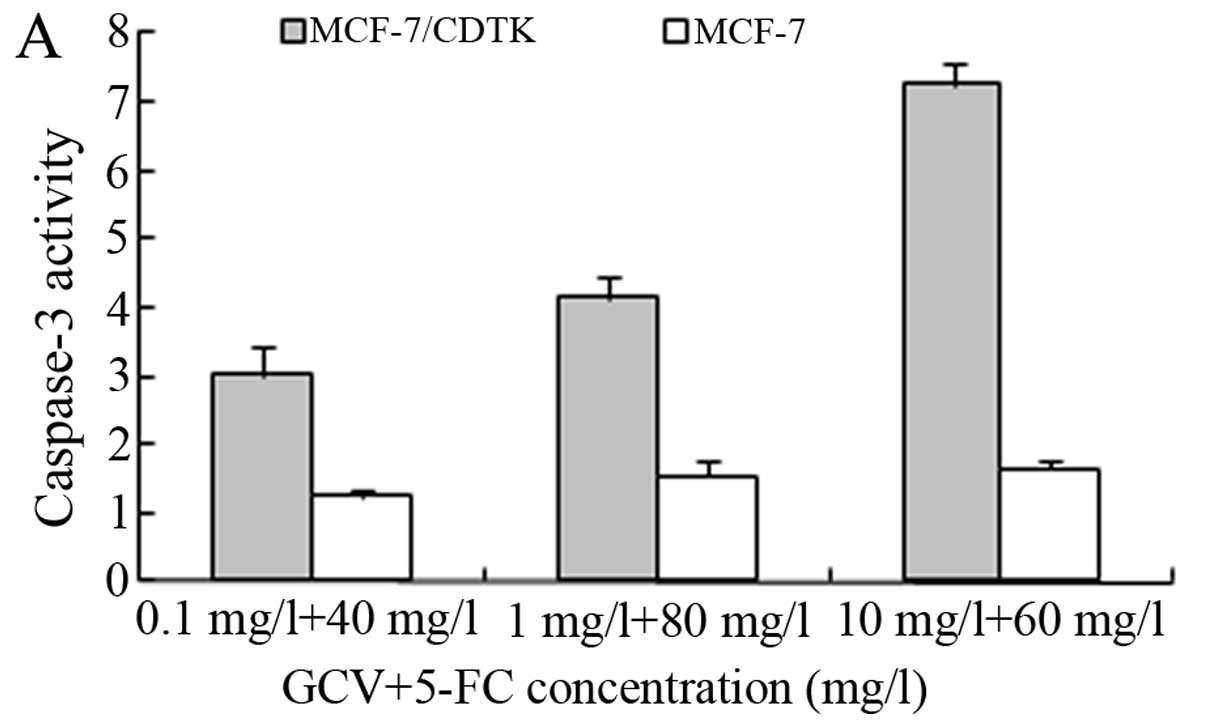
Cytotoxicity of double suicide gene
treatment
To determine whether double prodrugs (5-FC+GCV)
enhance the cytotoxicity of the suicide gene system in
vitro, MCF-7 cells were infected with the recombinant
adenovirus containing the CD/TK fusion gene, and their sensitivity
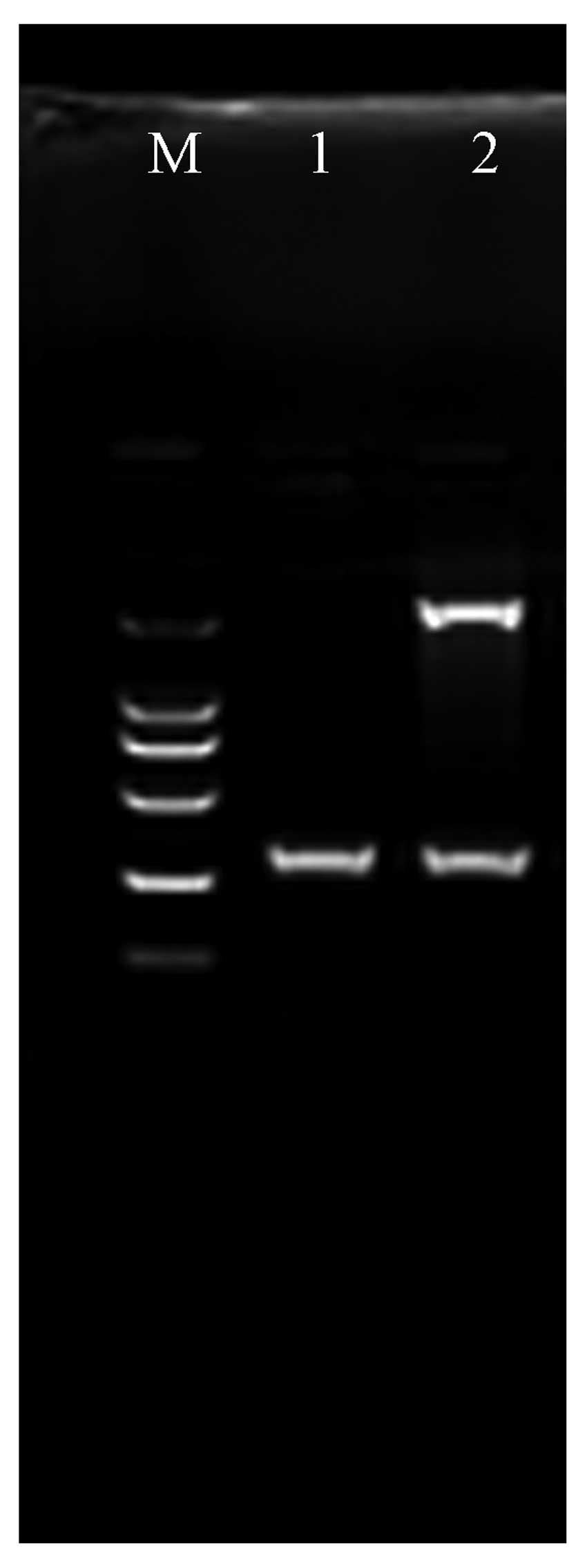
to the prodrugs was compared using MTT assays. As shown in Fig. 3, both prodrugs yielded killing
effects on MCF-7/CDTK cells in a concentration-dependent manner.
The sensitivity of MCF-7/CDTK cells to 5-FC+GCV was greater than
the sensitivity to 5-FC or GCV alone (P<0.01). To achieve an
equal killing effect, lower doses of 5-FC+GCV could be used. These
results demonstrated that double prodrug therapy was superior to a
single prodrug. As shown in Fig. 3,
we found that either GCV or 5-FC had no significant effects on the
non-infected MCF-7 cells when compared to the experimental group
(MCF-7/CDTK+5-FC/GCV) (P<0.01).
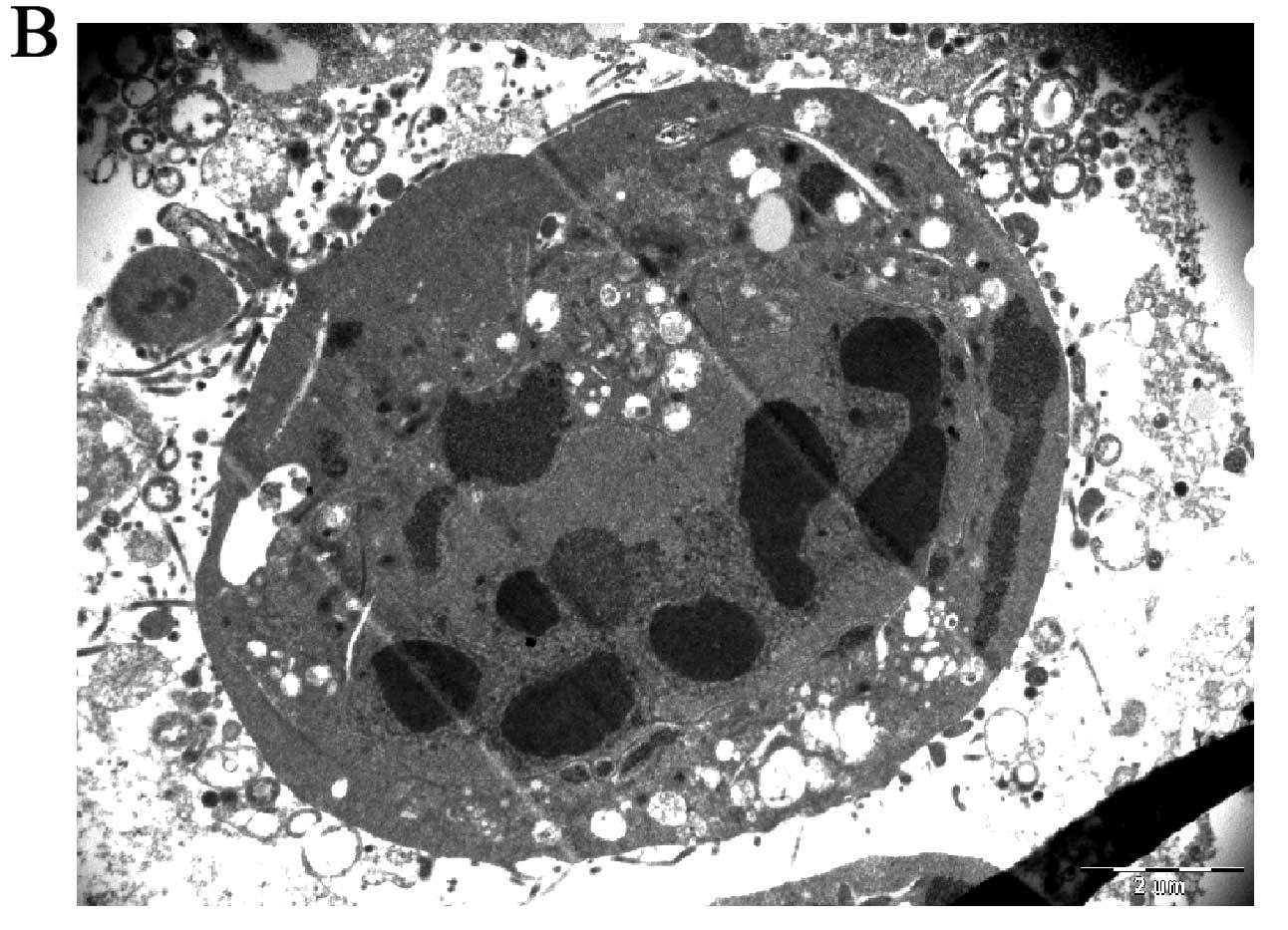
Effects of the prodrugs on MCF-7 using
transmission electron microscopy
Prodrug-treated MCF-7 cells exhibited cell death
corresponding well with the classical features of apoptosis:
nuclear collapse, continuing blebbing and apoptotic body formation
(Fig. 4B). TEM ultrastructure of
untreated MCF-7 cells showed no aberrant structure (Fig. 4A).
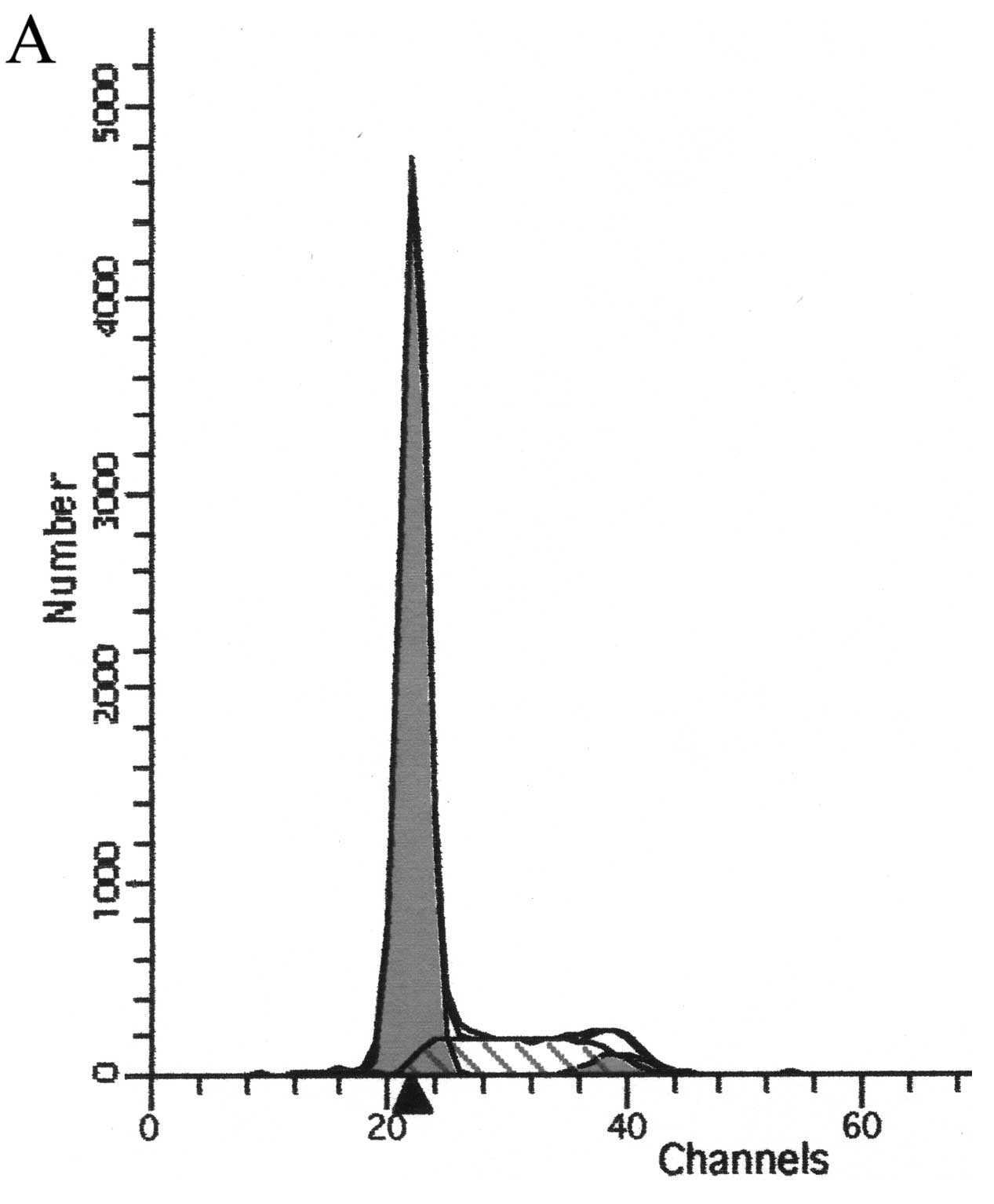
Flow cytometric analysis of the cell
cycle and apoptosis
Analysis of the cell cycle revealed that the
percentage of cells in the G0–G1 phase was
increased and the percentage of cells in the G2-M and S
phase was decreased in the treatment group as detected by flow
cytometry. Apoptotic peak was also shown on the leave side of
G0–G1. The peak became more evident with
increasing concentrations of the prodrugs. As shown in Fig. 5 and Table I, the apoptotic peak and cell
percentage increased with increasing concentrations of the
prodrugs. The effect showed a dose-response relationship.
Statistical analysis revealed a significant difference in the
numbers of apoptotic cells between the two groups (P<0.05).
 | Table IEffects of different concentrations of
the prodrugs on the cell cycle and apoptosis of MCF-7/CDTK
cells. |
Table I
Effects of different concentrations of
the prodrugs on the cell cycle and apoptosis of MCF-7/CDTK
cells.
| Groups | Apoptotic cells |
G0–G1 | S | G2-M |
|---|
| 0 mg/l (control
group) | 1.11±0.47 | 66.91±3.84 | 23.91±1.97 | 9.45±0.87 |
| GCV+5-FC |
| 0.1+40 mg/l | 5.22±0.52a | 68.57±6.97 | 22.46±2.21 | 8.10±0.13a |
| 1+80 mg/l | 12.07±1.47b | 77.51±3.75 | 19.55±1.35a | 1.72±0.28b |
| 10+160 mg/l | 30.00±2.96b | 81.48±5.05a | 17.28±1.07b | 0.83±0.18b |
| F-value | 170.726 | 5.733 | 9.031 | 260.782 |
| P-value | 0.000 | 0.022 | 0.006 | 0.000 |
Caspase-3 and -8 activities
The present study demonstrated that the prodrugs
induced apoptosis in the adv-infected MCF-7 cells, and we aimed to
ascertain whether this involved caspase-3 or -8 activity.
Chromogenic caspase-3 and -8 substrates were used to directly
examine the role of these caspases in the prodrug-treated cells.
The activity of caspase-3 (Fig. 6A)
was clearly increased following the treatment of MCF-7 cells with
GCV and 5-FC. Caspase-3 activity was further significantly
increased following the combination treatment of GCV and 5-FC for
48 h, compared with control alone (P<0.05). These data suggest
that the prodrugs promote double suicide gene-induced apoptosis in
MCF-7 cells via a caspase-3-dependent apoptosis pathway.
Experimental results showed that caspase-8 was not activated in the
prodrug-treated MCF-7 cell lines (P>0.1) compared to the
untreated controls (Fig. 6B).
Discussion
One potential therapeutic strategy for cancer
treatment is the technique of inserting suicide genes that activate
prodrugs to produce cytotoxity in tumor cells. As previously
demonstrated, suicide gene therapy is an effective method for
controlling tumor growth, reducing cell survival and eradicating
tumors such as schwannoma tumor (12), breast cancer (13), ovarian cancer (14) and lung cancer cell lines (15). For many carcinomas, two such
systems, i.e. combination of CD plus 5-FC and herpes simplex virus
(HSV)-TK plus GCV have been widely studied (16,17).
It is evident that expression of a single transgene is unlikely to
be sufficient to eradicate cancer. Numerous studies have
demonstrated that HSV-tk combined with cytokine therapy for the
treatment of breast carcinoma had a high efficacy (18–21).
It has been shown that specific expression of CD or HSV-tk genes in
breast as well as other cancer cells can be achieved using a vector
delivery system containing VEGF promoter sequences (22). Since breast cancer cell lines
exhibit high VEGF expression (23),
we constructed an adenovirus-mediated CD/TK double suicide gene
driven by the VEGF promoter. The aim of the present study was to
enhance targeting of the vector. The results demonstrated that this
vector was capable of directing expression of the CD/TK gene in
VEGF-expressing breast cells. The double suicide gene system
induced the apoptosis of tumor cells infected with the adenovirus
vector and when induced by appropriate concentration of 5-FC in
conjunction with GCV.
We transferred CD-TK fusion genes into human breast
carcinoma cells and investigated the cytotoxic efficacy of GCV
alone, 5-FC alone or the combination of the two prodrugs in
vitro. The results showed that each single prodrug killed tumor
cells, while the prodrugs in combination had a more powerful
killing effect (P<0.01). In addition, the advantage of the
combined treatment with the prodrugs was appreciated by the
reduction of drug dose. Taken together, our study demonstrated that
strong synergism occurred when tumor cells expressing the CD-TK
fusion genes were treated with 5-FC and GCV in combination. Similar
to this, the CD-TK fusion gene system used to treat prostate
carcinoma cells was previously found to present apparent synergism
(24).
Apoptosis is a biological phenomenon that is
involved in processes ranging from embryogenesis to ageing, from
normal tissue homeostasis to many human diseases including cancer.
Apoptotic cells share a number of common features such as cell
shrinkage, nuclear condensation, membrane blebbing, chromatin
cleavage and formation of pyknotic bodies of condensed chromatin
(25,26). These distinctive morphological
features form the basis of some of the most widely used techniques
for the identification and quantification of apoptosis, and thus
morphologic description using light or electron microscopy remains
one of the best manners with which to define apoptosis (27). Programmed cell death (apoptosis)
compared to necrosis is a desired somatic defense mechanism against
cancer cells (28). The double
suicide gene system is a promising treatment that possesses the
potential to induce apoptosis in different cancer cell lines
(29). We found that the CD/TK
genes induced a marked decrease in cell viability (92% in 48 h) in
MCF-7 cells at concentrations of 5-FC (320 mg/l) and GCV (100 mg/l)
through cell death by apoptosis. The present study was intended to
provide evidence of apoptosis in MCF-7 cells induced by the double
suicide gene system.
In the present study, the double suicide gene system
induced cell cycle arrest and apoptosis. The system reduced the
proportion of cells in the G2-M and S phase, and
increased those in the G0–G1 phase.
Therefore, our current findings presented in this study suggest
that the system induced MCF-7 cell apoptosis by modulating cell
cycle progression. Thus, further evaluation of alterations in cell
cycle proteins is warranted.
Apoptosis, also known as programmed cell death,
refers to certain physiological or pathological conditions in which
the end of active cell life is regulated by the activation of a set
of apoptotic factors. In normal cells, apoptosis and proliferation
coexist and maintain a dynamic equilibrium. It has been reported
that HSV-TK/GCV inhibits tumor growth possibly through increased
caspase-3 expression and induction of apoptosis (30,31).
When the CD-TK suicide gene targeting system was delivered into
tumor cells, we found that the system significantly inhibited
breast cancer cell growth, induced apoptosis in MCF-7 cells, and
increased caspase-3 activity (Fig.
6A). Moreover, caspase-8 activity was not obviously altered
(Fig. 6B). These findings indicate
that the CD-TK double suicide gene system inhibited cell
proliferation via the caspase-3 apoptotic pathway. Therefore, our
findings strongly suggest that the VEGF promoter-mediated
tumor-targeting suicide gene therapy system may represent a novel
therapy for breast cancer.
Acknowledgements
This work was supported in part by Shenzhen Council
for Scientific and Technological Innovation grant of 2013, Shenzhen
Nanshan District Science and Technology Project under grant
2012025, and the National High Technology Research and Development
Program (‘863’ Program) of China under grant 2001AA217171.
References
|
1
|
DeSantis C, Siegel R, Bandi P and Jemal A:
Breast cancer statistics, 2011. CA Cancer J Clin. 61:409–418. 2011.
View Article : Google Scholar
|
|
2
|
Liu T, Ye L, He Y, Chen X, Peng J, Zhang
X, Yi H, Peng F and Leng A: Combination gene therapy using
VEGF-shRNA and fusion suicide gene yCDglyTK inhibits gastric
carcinoma growth. Exp Mol Pathol. 91:745–752. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Lee SW, Lee YL, Lee YJ, Park SY, Kim IS,
Choi TH, Ha JH, Ahn BC and Lee J: Enhanced antitumor effects by
combination gene therapy using MDR1 gene shRNA and HSV1-tk in a
xenograft mouse model. Cancer Lett. 291:83–89. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Su GQ, Su G and Huang ZH:
Adenovirus-mediated tissue-targeted expression of the CDglyTk gene
for the treatment of breast cancer. Mol Med Rep. 6:321–329.
2012.PubMed/NCBI
|
|
5
|
Jia W, Mei L, Wang Y, Liu L and Che G:
Double suicide genes selectively kill human umbilical vein
endothelial cells. Virol J. 8:742011. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Niu HX, Du T, Xu ZF, Zhang XK and Wang RG:
Role of wild type p53 and double suicide genes in interventional
therapy of liver cancer in rabbits. Acta Cir Bras. 27:522–528.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Foekens JA, Peters HA, Grebenchtchikov N,
Look MP, Meijer-van Gelder ME, Geurts-Moespot A, van der Kwast TH,
Sweep CG and Klijn JG: High tumor levels of vascular endothelial
growth factor predict poor response to systemic therapy in advanced
breast cancer. Cancer Res. 61:5407–5414. 2001.PubMed/NCBI
|
|
8
|
Konecny GE, Meng YG, Untch M, Wang HJ,
Bauerfeind I, Epstein M, Stieber P, Vernes JM, Gutierrez J, Hong K,
Beryt M, Hepp H, Slamon DJ and Pegram MD: Association between
HER-2/neu and vascular endothelial growth factor expression
predicts clinical outcome in primary breast cancer patients. Clin
Cancer Res. 10:1706–1716. 2004. View Article : Google Scholar
|
|
9
|
Linderholm B, Grankvist K, Wilking N,
Johansson M, Tavelin B and Henriksson R: Correlation of vascular
endothelial growth factor content with recurrences, survival, and
first relapse site in primary node-positive breast carcinoma after
adjuvant treatment. J Clin Oncol. 18:1423–1431. 2000.
|
|
10
|
Nagaiah K, Venkatesham A, Srinivasa Rao R,
Saddanapu V, Yadav JS, Basha SJ, Sarma AV, Sridhar B and Addlagatta
A: Synthesis of new cis-fused tetrahydrochromeno [4,3-b] quinolines
and their antiproliferative activity studies against MDA-MB-231 and
MCF-7 breast cancer cell lines. Bioorg Med Chem Lett. 20:3259–3264.
2010.PubMed/NCBI
|
|
11
|
Kim JE, Chung WY, Chun KS, Lee CK, Park
HJ, Kim WB and Park KK: Pleurospermum kamtschaticum extract
induces apoptosis via mitochondrial pathway and NAG-1 expression in
colon cancer cells. Biosci Biotechnol Biochem. 74:788–792. 2010.
View Article : Google Scholar
|
|
12
|
Mizrak A, Bolukbasi MF, Ozdener GB,
Brenner GJ, Madlener S, Erkan EP, Ströbel T, Breakefield XO and
Saydam O: Genetically engineered microvesicles carrying suicide
mRNA/protein inhibit schwannoma tumor growth. Mol Ther. 21:101–108.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Yi BR, Choi KJ, Kim SU and Choi KC:
Therapeutic potential of stem cells expressing suicide genes that
selectively target human breast cancer cells: Evidence that they
exert tumoricidal effects via tumor tropism (Review). Int J Oncol.
41:798–804. 2012.
|
|
14
|
Hong S, Zhang P, Zhang H, Jia L, Qu X,
Yang Q, Rong F and Kong B: Enforced effect of tk-MCP-1 fusion gene
in ovarian cancer. J Exp Clin Cancer Res. 31:742012. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Cramer F, Christensen CL, Poulsen TT,
Badding MA, Dean DA and Poulsen HS: Insertion of a nuclear factor
kappa B DNA nuclear-targeting sequence potentiates suicide gene
therapy efficacy in lung cancer cell lines. Cancer Gene Ther.
19:675–683. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Luo XR, Li JS, Niu Y and Miao L:
Adenovirus-mediated double suicide gene selectively kills gastric
cancer cells. Asian Pac J Cancer Prev. 13:781–784. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Zarogoulidis P, Chatzaki E,
Hohenforst-Schmidt W, Goldberg EP, Galaktidou G, Kontakiotis T,
Karamanos N and Zarogoulidis K: Management of malignant pleural
effusion by suicide gene therapy in advanced stage lung cancer: a
case series and literature review. Cancer Gene Ther. 19:593–600.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Zhang N, Dong X, Sun Y, Cai X, Zheng C, He
A, Xu K and Zheng X: Cytotoxic effects of adenovirus- and
lentivirus-mediated expression of Drosophila melanogaster
deoxyribonucleoside kinase on Bcap37 breast cancer cells. Oncol
Rep. 29:960–966. 2013.PubMed/NCBI
|
|
19
|
Kang NH, Hwang KA, Yi BR, Lee HJ, Jeung
EB, Kim SU and Choi KC: Human amniotic fluid-derived stem cells
expressing cytosine deaminase and thymidine kinase inhibits the
growth of breast cancer cells in cellular and xenograft mouse
models. Cancer Gene Ther. 19:412–419. 2012. View Article : Google Scholar
|
|
20
|
Li XH, Zhou P, Wang LH, Tian SM, Qian Y,
Chen LR and Zhang P: The targeted gene (KDRP-CD/TK) therapy of
breast cancer mediated by SonoVue and ultrasound irradiation in
vitro. Ultrasonics. 52:186–191. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Joo KM, Park IH, Shin JY, Jin J, Kang BG,
Kim MH, Lee SJ, Jo MY, Kim SU and Nam DH: Human neural stem cells
can target and deliver therapeutic genes to breast cancer brain
metastases. Mol Ther. 17:570–575. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Koshikawa N, Takenaga K, Tagawa M and
Sakiyama S: Therapeutic efficacy of the suicide gene driven by the
promoter of vascular endothelial growth factor gene against hypoxic
tumor cells. Cancer Res. 60:2936–2941. 2000.PubMed/NCBI
|
|
23
|
Radovich M, Hancock BA, Kassem N, Mi D,
Skaar TC and Schneider BP: Resequencing of the vascular endothelial
growth factor promoter reveals haplotype structure and functional
diversity. Angiogenesis. 13:211–218. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Boucher PD, Im MM, Freytag SO and Shewach
DS: A novel mechanism of synergistic cytotoxicity with
5-fluorocytosine and ganciclovir in double suicide gene therapy.
Cancer Res. 66:3230–3237. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Song NM, Jun S, Zang DY, Kim SG, Park HR
and Kang D: Differential susceptibility of gastric cancer cells to
TRAIL-induced apoptosis. Oncol Rep. 29:1224–1230. 2013.PubMed/NCBI
|
|
26
|
Wang T, D’Souza GG, Bedi D, Fagbohun OA,
Potturi LP, Papahadjopoulos-Sternberg B, Petrenko VA and Torchilin
VP: Enhanced binding and killing of target tumor cells by
drug-loaded liposomes modified with tumor-specific phage fusion
coat protein. Nanomedicine. 5:563–574. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Terakawa M, Tsuda H, Ashida H and Sato S:
Assessment of tissue alteration in skin after interaction with
photomechanical waves used for gene transfection. Lasers Surg Med.
42:400–407. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Mandoky L, Szende B, Géczi L, Bodrogi I,
Kásler M and Bak M: Apoptosis regulation and spontaneous apoptosis
index of testicular germ cell tumors are associated with
differentiation and resistance to systemic treatment. Anticancer
Res. 28:1641–1649. 2008.PubMed/NCBI
|
|
29
|
Ortiz R, Prados J, Melguizo C, Rama AR,
Alvarez PJ, Rodríguez-Serrano F, Caba O, Boulaiz H and Aranega A:
Gef gene therapy enhances the therapeutic efficacy of cytotoxics in
colon cancer cells. Biomed Pharmacother. 66:563–567. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Tang W, He Y, Zhou S, Ma Y and Liu G: A
novel Bifidobacterium infantis-mediated TK/GCV suicide gene
therapy system exhibits antitumor activity in a rat model of
bladder cancer. J Exp Clin Cancer Res. 28:1552009.
|
|
31
|
Zhang Z, Lin J, Chu J, Ma Y, Zeng S and
Luo Q: Activation of caspase-3 noninvolved in the bystander effect
of the herpes simplex virus thymidine kinase gene/ganciclovir
(HSV-tk/GCV) system. J Biomed Opt. 13:0312092008. View Article : Google Scholar : PubMed/NCBI
|