Introduction
Mammary development and breast carcinogenesis are
usually characterized by their hormonal control (1). Among the hormones more frequently
studied, estrogens (E) and progesterone (Pg) regulate several
functions of the normal or tumoral mammary epithelium, such as cell
proliferation and apoptosis. Approximately one-third of breast
cancers maintain E-dependence for growth and the concentration of
estrogen receptors (ERs) in malignant breast tissues is an
indicator of this hormonal dependence (2). E are the main steroid hormones
implicated in the induction of breast cancer, but experimental
evidence in rodents suggests that Pg is also important in its
induction and progression (3).
Prolactin (PRL) has been associated with breast
cancer pathogenesis and progression. Acting at the endocrine and
autocrine/paracrine levels, PRL functions to stimulate the growth
and motility of human breast cancer cells (4). Epidemiologic studies have demonstrated
that hyperprolactinemia is frequently associated with growth,
development and a poor prognosis in mammary cancer (5). Related to PRL actions on mammary
gland, the growth hormone (GH) has also been shown to affect
mammary development and carcinogenesis. In rodents, GH may
potentiate breast cancer since GH- or GH receptor-deficiency is
associated with reduced incidence and aggressiveness of
experimentally induced breast cancer (6).
Other hormonal influences on the mammary gland
involves triiodothyronine (T3) and activation of mammary
thyroid hormone receptors (TRs) inducing differentiation and
lobular growth in an E-like manner. However, there is controversy
concerning the relationship between thyroid disorders and breast
cancer incidence (7). Thyroid
hormones (THs) could participate directly on mammary
carcinogenesis, or indirectly through their action in changes in
the patterns of secretion of other hormones such as E, Pg, PRL
and/or GH, among others. It has been shown that hypothyroidism may
result in reduced incidence of primary breast carcinoma (8), and in vitro studies also
suggest that THs affect cell proliferation and gene expression
(9).
Moreover, THs have important effects on energy
balance, since they influence both energy intake and expenditure.
Abnormal TH levels are frequently associated with changes in body
weight and modifications in the function of adipose tissue. The
adipocyte is a functionally active cell and produces several
biologically active adipokines such as leptin, which could
contribute to an increased breast cancer risk for obese women
(10). Furthermore, estradiol
(E2) is also produced by adipose tissue and it has been
suggested as being responsible for the elevated risk of breast
cancer in these women (10).
Since the epidemiologic and in vitro data
have not provided conclusive evidence of the involvement of THs in
mammary carcinogenesis, we used an in vivo model to assess
the relationship between THs, adipose tissue and breast cancer
development. In the present study, we investigated the effect of
the hormonal milieu induced by hypothyroidism or hyperthyroidism on
mammary carcinogenesis induced by 7,12-dimethylbenz(a)anthracene
(DMBA) in rats.
Materials and methods
Animals
Virgin Sprague-Dawley female rats (180–200 g) bred
in our laboratory were used. The animals were maintained in a
light- (lights on from 06.00 to 20.00 h) and temperature-controlled
room (22–24°C); rat chow (Cargill, Córdoba, Argentina) and tap
water or 6-N-propyl-2-thiouracil (PTU) solution were available
ad libitum.
Animal maintenance and handling were performed
according to the NIH guide for the Care and Use of Laboratory
Animals (NIH publication no. 86-23, revised 1985 and 1991) and the
UK requirements for ethics of animal experimentation (Animal
Scientific Procedures, Act 1986). All the experimental procedures
were approved by the Animal and Ethics Committee of the School of
Medicine of the National University of Cuyo, Mendoza,
Argentina.
Experimental protocols
In order to study the effect of hypothyroidism or
hyperthyroidism on mammary carcinogenesis, the rats were treated
per os with a single dose of DMBA (15 mg/rat) at 55 days of
age and were then divided into two experimental groups and their
respective controls: hypothyroid rats (HypoT, n=26) and untreated
rats (EUT, n=12); hyperthyroid rats (HyperT, n=23) and
vehicle-treated rats (VEH, n=5). The day of DMBA administration was
considered as day 0 of the experiment.
Hypothyroidism was induced by the administration of
PTU (Sigma-Aldrich, Buenos Aires, Argentina) at a concentration of
0.1 g/l in the drinking water, starting on day 3. The respective
control group (EUT) was maintained under standard conditions during
the study period.
Hyperthyroidism was induced by daily subcutaneous
administration of L-thyroxine (T4; Sigma-Aldrich).
T4 was dissolved in saline solution (0.42 N of NaOH) and
injected s.c. daily at a dose of 0.25 mg/ml/kg starting on day 3.
Control age- and weight-matched female rats received VEH s.c.
Since no statistical differences were observed
between the EUT and VEH groups in any of the studied variables, we
grouped and analyzed them as a single control group.
Body weight was monitored weekly. Food intake was
assessed on week 10 of the experiment and expressed as the weight
of rat chow consumed per rat and per day. The animals were assessed
every 72 h for the appearance of the first palpable tumor. The
latency, incidence and progression of tumors were determined in all
groups. The rats were sacrificed by decapitation at 10:00 h on the
day of diestrus when the tumors reached a volume >1,000
mm3 or at the end of the experiment on day 200 when they
did not develop mammary tumors. All the animals were weighed and
their vaginal smears observed before sacrifice. Trunk blood samples
were collected and allowed to clot at room temperature. Serum was
separated and stored at −20°C until assayed for hormone
determinations. Immediately after decapitation, intra-abdominal fat
was removed, weighed and expressed as a percentage of total body
weight. A piece of normal mammary gland and the tumors were removed
for histopathological and immunohistochemical analysis.
Induction of mammary tumors
DMBA (Sigma-Aldrich) was dissolved in sunflower oil
at a concentration of 5 mg/ml and administered at a dose of 15
mg/rat on day 0 by an intragastric probe, 3 h after food and water
deprivation to ensure a complete absorption of the drug. Food and
water were replaced 2 h after DMBA administration. DMBA has been
extensively used at that dose to study the possible mechanisms
responsible for the beginning of the transformation of normal
breast tissue into a tumor (11).
Hormone determinations
Thyroid stimulating hormone (TSH), PRL and GH
concentrations were measured by double antibody radioimmunoassay
using materials generously provided by A.F. Parlow and the National
Hormone and Pituitary Program (NHPP, Harbor-UCLA Medical Center,
Torrance, CA, USA). The hormones were radio-iodinated using the
chloramine T method and purified by passage through Sephadex G75.
The results were expressed in terms of the rat TSH RP-3, rat PRL
RP-3 or rat GH RP-2 standard preparations. Assay sensitivity was
0.5 μg/l serum and the inter- and intra-assay coefficients of
variation were <10% for all hormones.
E2, Pg, T3 and
tetraiodothyronine (T4) concentrations in sera were
measured by radioimmunoassay using commercial kits for total
hormones (TKE 21 and TKPG1 double antibody radioimmunoassay;
Siemens Healthcare Diagnostics, Inc., Los Angeles, CA, USA and
RK-6CT1 and RK-1CT1 double antibody radioimmunoassay; Institute of
Isotopes Ltd., Budapest, Hungary; respectively). Inter- and
intra-assay coefficients of variation were <10%.
Serum leptin concentrations were determined by a
specific radioimmunoassay developed in the Instituto
Multidisciplinario de Biología Celular (IMBICE) and validated for
rat and mouse leptin against a commercial kit (catalog no. RL-83K;
Linco Research, Inc., St. Charles, MO, USA) as previously published
(12). Briefly, synthetic murine
leptin (PrePro Tech, Inc., Rocky Hill, NJ, USA) was used for both
labeled peptide and standard and for the development of anti-leptin
serum. Leptin was radio-iodinated by the chloramine T method and
purified by elution using a Sephacryl S-300 (Sigma-Aldrich) column.
The anti-leptin serum was developed by rabbit immunization with
murine leptin coupled to BSA. The intra- and inter-assay
coefficients of variation were 5–8 and 10–13%, respectively.
Latency, incidence and progression of
tumors
The latency of mammary tumors was considered as the
time between DMBA administration and the appearance of the first
palpable tumor. Incidence was calculated as the percentage of rats
that had tumors within the study period in respect to the total
number of rats per group. We used a caliper to measure the major
(DM) and minor (dm) diameters every 72 h and calculated the tumor
volume (TV = dm2 × DM/2). Tumor progression was assessed
estimating tumor growth rate [GR = TV/(day of sacrifice − day of
appearance of first tumor)].
Tumors and mammary gland histology
Small pieces of the tumor and inguinal mammary gland
(contralateral to the tumor) from each rat were processed for
histopathologic studies by fixing in buffered formalin, dehydrating
in ethanol and embedding in paraffin wax. Sections (3–5 μm) were
cut with a microtome and stained with hematoxylin and eosin
(H&E) to define the histopathological changes in the mammary
glands and to classify tumors according to published criteria
(13). Images were captured with an
Eclipse E200 microscope fitted with a digital still camera
Micrometrics SE Premium (both from Nikon Corp., Japan) under
magnification of ×100, ×400 and ×600.
The quantification of the percentages of stroma,
mostly composed by adipocytes, and epithelial tissue in the mammary
gland was performed by measuring the area occupied by the adipose
tissue or epithelium in 8–10 fields of each preparation from all
rats using the ImageJ 1.42q software available at the NIH site
(http://rsb.info.nih.gov/ij). Each area
was expressed as a percentage of the whole field as previously
published (14).
Apoptotic and mitotic indices
The microscopic analysis was carried out by two
independent observers. The apoptotic and mitotic indices were
calculated by counting the total number of apoptotic bodies and
mitotic figures, respectively, in the histological sections stained
with H&E in 10 ×400 magnification-fields from each animal. The
mitotic/apoptotic ratio (M/A ratio) was calculated by dividing the
mitotic index by the apoptotic index.
Immunohistochemistry (IHC)
Serial sections (3–5 μm) were mounted onto
3-aminopropyltriethoxysilane (Sigma-Aldrich)-coated slides for
subsequent IHC analysis. The primary antibody used in this study
was a monoclonal mouse anti-proliferating cell nuclear antigen
(PCNA; Dako Cytomation, Glostrup, Denmark) at 1:500. An antigen
retrieval protocol using heat was used to unmask the antigens (30
min in citrate buffer, 0.01 M, pH 6.0). Tissue sections were
incubated with the primary antibody overnight at 4°C in humidity
chambers. A commercial kit to detect mouse and rabbit antibodies
was used (Dako EnVision system, horseradish peroxidase,
diaminobenzidine; Dako, Carpinteria, CA, USA). Slides were lightly
counterstained with hematoxylin to reveal the nuclei, examined and
photographed. The percentage of positive nuclei was obtained based
on an average of 700 cells counted per sample, at a ×400
magnification. The immunostaining of the tumor cells was
semi-quantitatively scored as: 0, no staining; 1, nuclear staining
of <10% of tumor cells; 2, staining between 11 and 33% of tumor
cells; 3, staining between 34 and 65% of tumor cells; 4, staining
of >66% of tumor cells. These scores were obtained by two
independent observers blinded regarding the clinical evaluation,
and a few conflicting scores were resolved by consensus.
TUNEL
Apoptosis was measured by a modification of the
TUNEL assay using the ApopTag Plus Apoptosis In Situ Detection kit
(Oncor, Gaitherburg, MD, USA), as reported previously (15). The TUNEL apoptotic index was
calculated as the percentage of positive nuclei, based on an
average of 700 cells counted per case, at ×400 magnification.
Statistical analysis
Values are expressed as means ± SEM of 17–26
animals/group. All statistical analyses were performed using
GraphPad Prism 5.01 software (GraphPad Software, Inc., San Diego,
CA, USA). Differences in the distribution of variables between the
three studied groups (HypoT, HyperT and controls) were assessed
using one-way analysis of variance (ANOVA I) or the Kruskal Wallis
test depending on the normality of the variables as evaluated by
the Kolmogorov-Smirnov test. Two-way analysis of variance (ANOVA
II) was used for analysis of the effects of the PTU or
T4 treatments on body weight over time. Post hoc
comparisons between means were conducted by Bonferroni’s test or
Dunn’s multiple comparison test. Student’s t-test was used when
only two groups were compared. When variances were not homogeneous,
logarithmic transformation of data was applied. Incidence and
percentages of mammary adipose area were analyzed by Chi-square.
Survival curves were compared using the log-rank Mantel-Cox test.
The relation between selected variables was performed by Pearson’s
correlation coefficient. Differences were considered significant
when the probability was 5% or less.
Results
Induction of hypothyroidism and
hyperthyroidism
The chronic thyroid condition of the animals was
evaluated by measuring serum T4, T3 and TSH
concentrations. The rats treated with PTU had low circulating
T4 (20.96±1.10 ng/ml, p<0.001) and T3
(0.42±0.06 ng/ml, p<0.05) concentrations and also markedly
increased levels of TSH (4.98±0.43 ng/ml, p<0.01) compared to
the EUT rats (33.29±2.62, 0.61±0.06 and 0.84±0.06, respectively);
all of them sensitive indicators for hypothyroidism. In contrast,
L-thyroxine administration increased T4 (55.88±6.93
ng/ml, p<0.05) and T3 (0.91±0.10 ng/ml, p<0.05)
and decreased circulating TSH (0.52±0.02 ng/ml, p<0.01)
confirming the state of hyperthyroidism.
Hypothyroidism affects food intake, body
weight and growth of the animals
Chronic treatment with PTU significantly retarded
the growth of the HypoT animals as reflected in the decreased
weight and reduced levels of circulating GH (Table II) in comparison with the HyperT
rats and the untreated controls. HypoT rats had an increase in body
weight similar to HyperT and EUT rats during the first 4 weeks of
treatment with PTU. Afterwards, they stopped gaining weight until
the end of the study showing a significant difference in body
weight when compared with the other two groups (p<0.0001, data
not shown). HyperT rats gained weight steadily similar to the EUT
rats. Daily food intake was significantly lower in HypoT rats
compared to HyperT and EUT rats (12.6±0.2 vs. 23.2±1.2 and 19.1±0.7
g/rat, respectively; p<0.0001).
 | Table IIHormonal profile of rats with
different thyroid states with and without mammary tumors. |
Table II
Hormonal profile of rats with
different thyroid states with and without mammary tumors.
| HypoT | EUT | HyperT |
|---|
|
|
|
|
|---|
| Grouped | Tumor | No tumor | p-value | Grouped | Tumor | No tumor | p-value | Grouped | Tumor | No tumor | p-value |
|---|
| Serum GH
(ng/ml) | 6.4±0.3a | 6.2±0.3 | 6.4±0.3 | 0.77 | 17.0±3.3 | 16.5±3.8 | 19.3±7.4 | 0.76 | 15.0±3.0 | 16.1±4.6 | 13.3±5.9 | 0.79 |
| Serum E2
(pg/ml) | 10.2±2.0a | 18.1±9.8 | 8.7±1.6 | 0.09 | 24.0±3.4 | 22.6±3.6 | 40.4±8.2 | 0.06 | 3.3±0.8a | 5.6±2.1 | 0.4±0.0 | 0.39 |
| Serum PRL
(ng/ml) | 11.6±1.1 | 12.3±1.6 | 11.0±1.2 | 0.69 | 13.9±2.3 | 13.1±2.6 | 15.8±7.6 | 0.67 | 17.6±4.1 | 26.9±9.6 | 10.9±3.3 | 0.38 |
| Serum Pg
(ng/ml) | 10.9±3.4 | 10.2±1.4 | 40.5±15.1 | 0.38 | 15.9±2.3 | 19.6±4.2 | 15.6±9.1 | 0.69 | 12.8±2.0 | 12.4±2.1 | 15.2±8.0 | 0.64 |
| Serum leptin
(ng/ml) | 1.3±0.2a | 1.3±0.4 | 0.6±0.1 | 0.038 | 2.2±0.3 | 1.3±0.7 | 2.7±0.9 | 0.015 | 1.2±0.2 | 1.3±0.2 | 0.7±0.1 | 0.15 |
| Leptin (ng/mg
fat) | 1.2±0.2 | 1.4±0.4 | 0.9±0.2 | 0.29 | 1.9±0.2 | 1.5±0.2 | 2.1±0.1 | 0.1 | 1.9±0.4 | 2.1±0.7 | 0.7±0.1 | 0.3 |
The mass of abdominal fat expressed as a percentage
of the body weight has been previously used as a measure of body
composition in rats (16). No
statistically significant change was observed in this percentage
due to PTU (11.3±1%) or T4 (9.1±0.6%) treatments
compared with controls (11.5±0.7%), although the HyperT rats had a
tendency to show lower values.
Effects of hypothyroidism and
hyperthyroidism on mammary gland histology
The effect of THs on the development of the mammary
gland was evaluated by histological observation and measurement of
the areas occupied by parenchyma or stroma. Table I shows that the mammary glands of
EUT rats had a normal appearance with few ducts surrounded by a
small amount of fibrous connective tissue and abundant adipose
tissue. The mammary glands of HyperT and HypoT rats were also
normal but in a few cases were associated with mammary benign
pathologies such as ductal stasis. Moreover, hyperplasia,
intraductal papilloma and adenosis were observed in the HyperT rats
(Table I).
 | Table ISummary of mammary gland
characteristics and tumors developed in the DMBA-treated rats. |
Table I
Summary of mammary gland
characteristics and tumors developed in the DMBA-treated rats.
| HypoT rats | EUT rats | HyperT rats |
|---|
| No. of rats | 26 | 17 | 23 |
| Mammary glands | | | |
| Normal | 23 | 16 | 18 |
| Benign
lesions | | | |
| Hyperplasia | 0 | 1 | 1 |
| Intraductal
papilloma | 0 | 0 | 1 |
| Adenosis | 0 | 0 | 2 |
| Ductal
ectasis | 3 | 0 | 1 |
| Tumors | | | |
| Rats with
tumors | 3 | 12 | 17 |
| Tumor weight
(g)a | 1.26±0.57 | 1.22±0.17 | 1.32±0.18 |
| Type of tumor | | | |
| Ductal
carcinoma | 3 | 10 | 15 |
| Infiltrating
lobular carcinoma | 0 | 2 | 0 |
| Tubular
carcinoma | 0 | 0 | 2 |
| Ductal tumor
grade | | | |
| I | 1 | 10 | 13 |
| II | 2 | 0 | 2 |
| III | 0 | 0 | 0 |
| Other
characteristics | | | |
| Predominant
component | Invasive | In situ | In situ |
| Inflammatory
response | Low | Moderate | Moderate |
| Necrosis | Moderate | Scarce | Low |
| Rats with
secondary tumors | | | |
| Low-grade ductal
carcinoma | 1 | 4 | 8 |
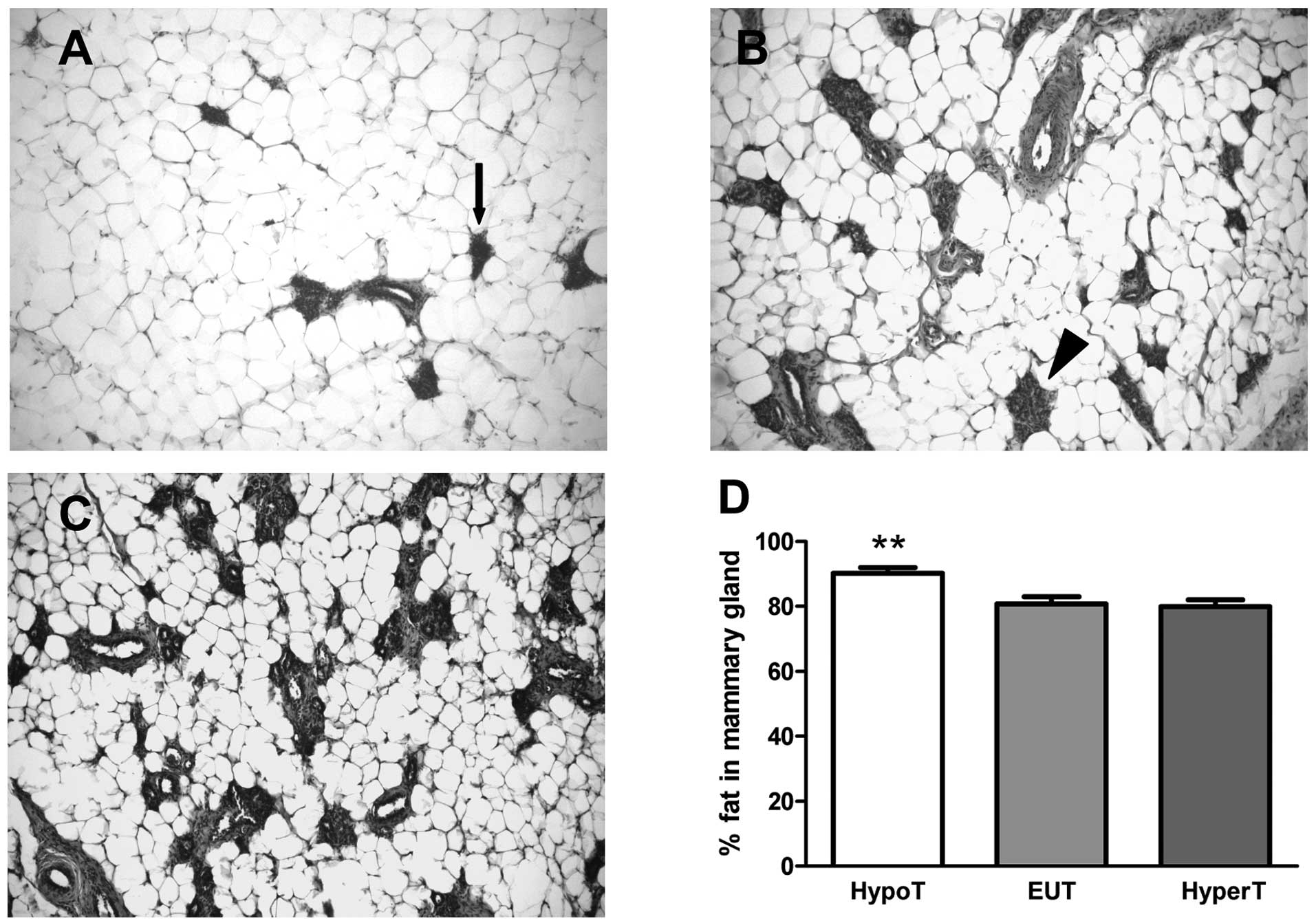
Fig. 1A–C show
representative microphotographs of H&E-stained mammary tissue
from HypoT, EUT and HyperT rats, respectively. Fig. 1D shows the relative percentages of
the adipose area. In EUT and HyperT rats, ~80% of the mammary gland
was composed of stroma, which consisted of fat and connective
tissue. The rest of the gland was represented by the parenchyma
including lobules type 1, lobules type 2 and ductal structures
(Fig. 1B and C). In HypoT rats, a
scarce lobe-alveolar development of the mammary gland (~10%) and an
increased percentage of fat in the stroma (~90%, p<0.001
compared to HyperT and EUT rats) were observed (Fig. 1D), suggesting that hypothyroidism,
but not hyperthyroidism, induced changes in the parenchyma-stroma
ratio.
Effects of hypothyroidism and
hyperthyroidism on mammary gland carcinogenesis
Table I shows the
histopathological characteristics of the tumors in the three
groups. On one hand, tumors of EUT and HyperT rats were all of low
grade considering the characteristics of the nuclei, the presence
of ducts and the number of mitotic figures. They had a predominant
in situ component with a small invasive component. Necrosis
was scarce and they had moderate inflammatory infiltration.
Thirty-three percent of EUT and 47% of HyperT rats developed
secondary tumors. On the other hand, malignant tumors from HypoT
rats were histologically more aggressive with a predominant
invasive component and a small in situ component. Necrosis
was moderate and the host response was lower than in the tumors
from HyperT and EUT rats.
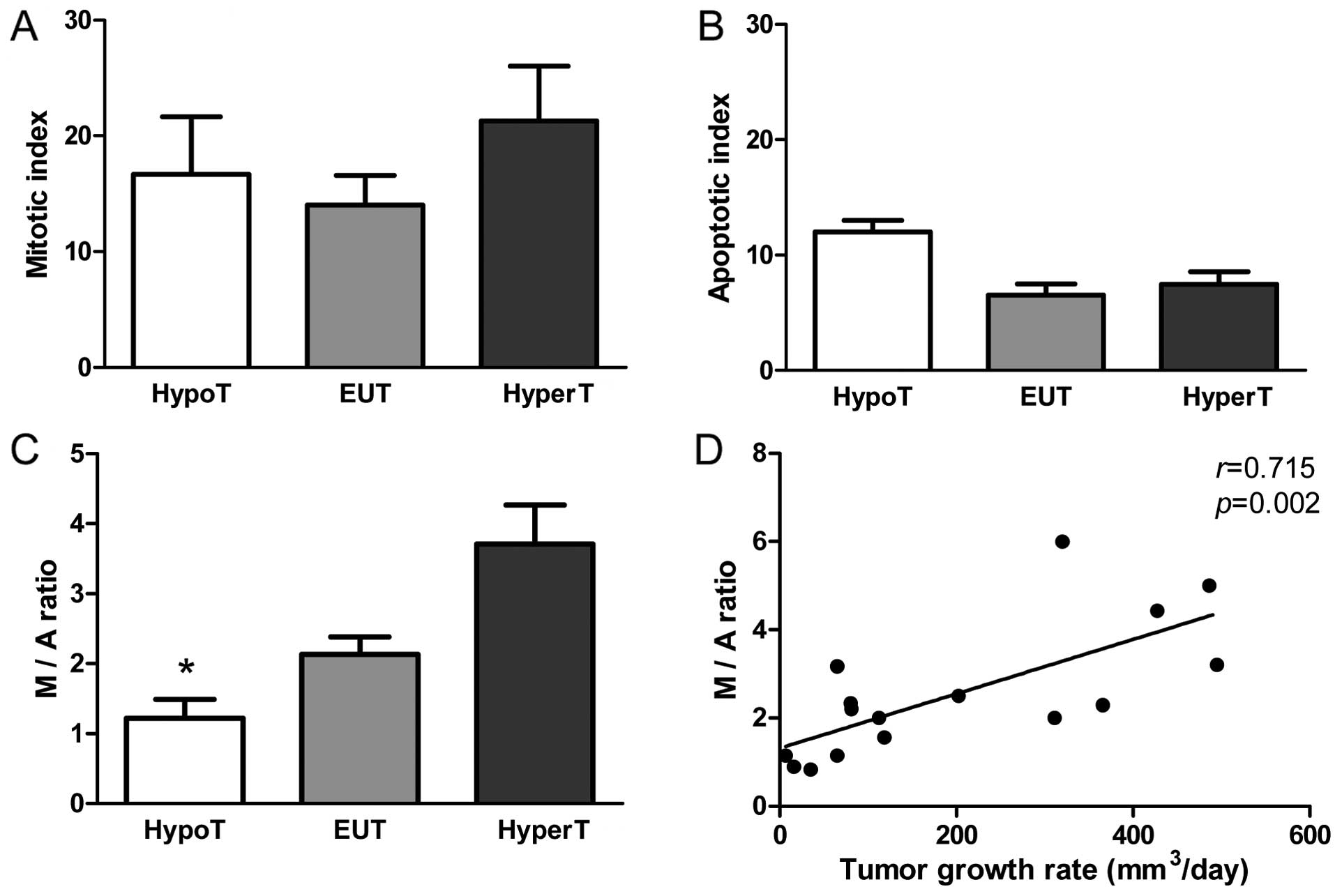
The latency of onset of tumors was longer
(p<0.05) (Fig. 2A) and the
incidence was lower (p<0.0001) (Fig.
2B) in the HypoT rats when compared to the EUT and HyperT rats.
Hypothyroidism retarded tumor growth (p<0.05) (Fig. 2C) and increased tumor-free survival
(p<0.001) (Fig. 2D). No
relationship between tumor growth rate and the histological type of
the neoplasia was apparent in any of the groups.
Hypothyroidism increases apoptosis
without promoting cell proliferation
To assess the effect of THs on tumor progression, we
performed a microscopic analysis of mitosis and apoptosis. The
mitotic index was similar in the three groups (Fig. 3A) while the apoptotic index tended
to increase in tumors of the HypoT rats (p=0.07) (Fig. 3B). In order to investigate if the
balance between mitosis and apoptosis influences tumor development
we calculated the mitotic/apoptotic ratio. This parameter was
significantly lower in HypoT rats when compared to EUT and HyperT
animals (p<0.05) (Fig. 3C), and
was positively correlated with the tumor growth rate (r=0.715;
p=0.002) (Fig. 3D) suggesting that
the difference in the M/A ratio had biological significance.
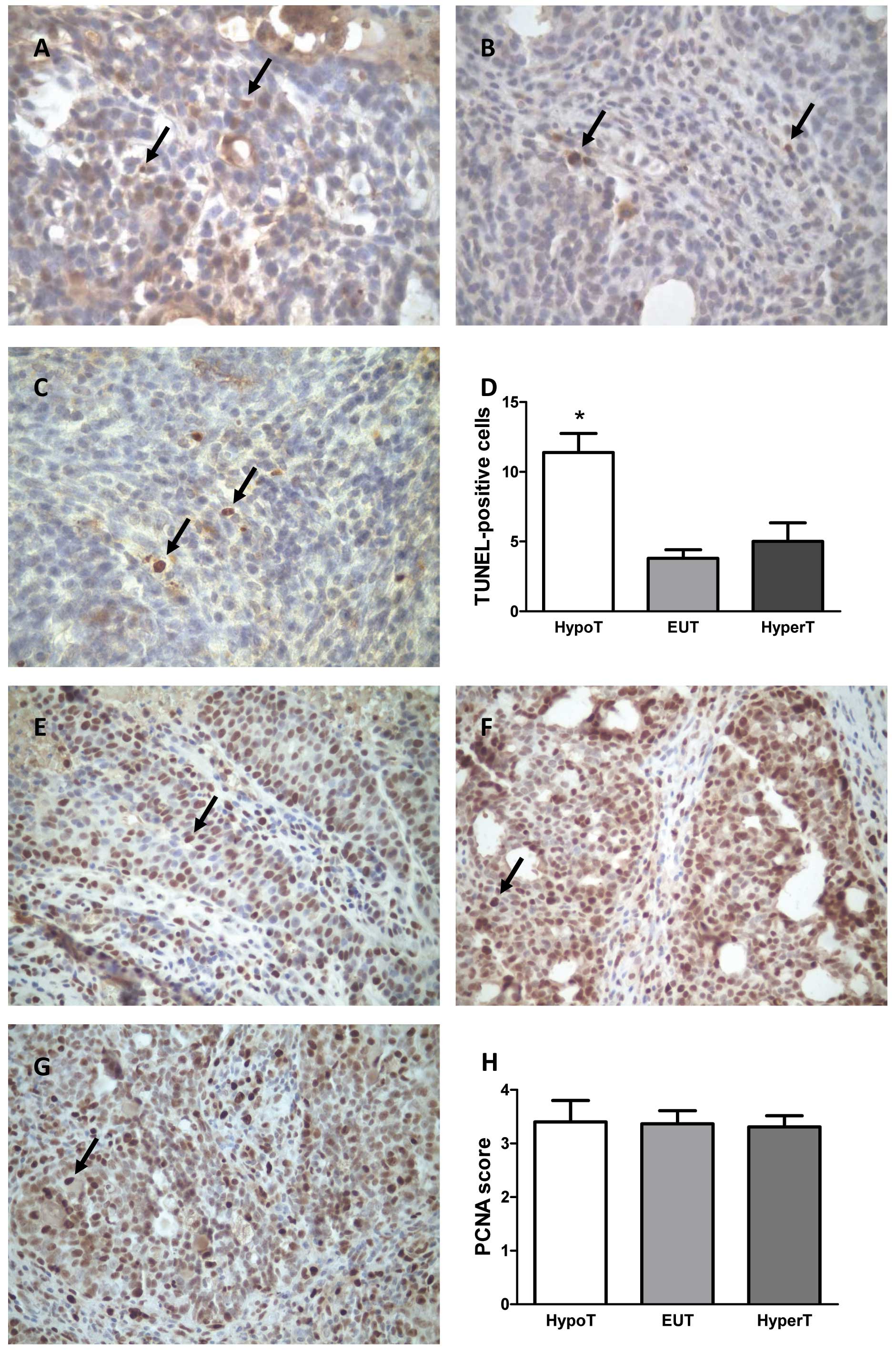
We further studied apoptosis by TUNEL assay, a more
sensitive technique, and we found an increased number of
TUNEL-positive cells in tumors of the HypoT rats compared to the
numbers in the HyperT and EUT animals (p<0.05) (Fig. 4A–D). In order to validate the
results obtained by counting mitotic figures, we additionally
evaluated mitosis by IHC of PCNA in the tumors. No statistically
significant differences between the treatments were observed
(Fig. 4E–H).
Hypothyroidism and hyperthyroidism alter
hormone patterns
The influence of PTU and T4 treatments on
circulating hormone concentrations is shown in Table II. Both treatments decreased
circulating levels of E2 compared to EUT rats
(p<0.0001).
Serum PRL and Pg concentrations were similar between
the three studied groups. No significant differences were observed
in circulating values of GH, PRL, E2 and Pg between
tumor-bearing and tumor-free rats in the HypoT, HyperT and EUT
groups.
Effect of adipose tissue and leptin on
mammary gland carcinogenesis
Leptin concentrations were significantly decreased
in HypoT rats whether they were calculated per milliliter of serum
or per gram of abdominal fat mass (p<0.05, Table II). This suggested that adipocyte
secretion activity was altered in HypoT rats since the body fat
mass was similar in all groups. Interestingly, serum leptin levels
were higher in HypoT rats with tumors than in those without tumors
(1.3±0.4 vs. 0.6±0.1 ng/ml, respectively; p<0.05). No
significant differences were observed in EUT or HyperT rats,
regarding tumor presence.
Discussion
The possible association between thyroid diseases
and breast carcinoma has been debated for decades and remains
controversial (8,13,17,18).
Epidemiological and prospective studies have not been able to
demonstrate a correlation between benign thyroid conditions and the
risk of mammary cancer (13,18,19).
However, other studies have supported a significant association
between primary hypothyroidism and breast cancer (8,20). In
the present study, hypothyroidism prolonged the latency of tumor
appearance, reduced tumor incidence and retarded tumor growth in
rats.
Martinez-Iglesias et al(21) injected MDA-MB-468 cells in
hypothyroid mice and evaluated the growth of the tumors, thus
avoiding the effect of THs on cancer initiation. They found that
hypothyroidism had a dual effect on mammary tumorigenesis, since
tumor growth was slower in hypothyroid mice, but the tumors were
more aggressive and invasive, and the formation of metastasis was
strongly enhanced. Contrarily, in humans, Cristofanilli et
al(8) showed that spontaneous
clinical hypothyroidism was a strong protective factor against
mammary cancer and decreased tumor incidence and aggressiveness.
These authors also found that women with primary hypothyroidism had
a 61% reduction in the risk of invasive breast cancer and they were
more likely to have localized disease and no lymph node involvement
when compared with euthyroid women. Moreover, hypothyroid patients
who developed breast cancer progressed with a more indolent disease
and smaller tumors. On the other hand, Shering et
al(18) and Ito and Maruchi
(17) observed that Japanese women
with Hashimoto’s thyroiditis had a higher incidence of mammary
cancer than women without thyroid disease. Two recent meta-analyses
showed no significant association, except for autoimmune
thyroiditis which increased breast cancer risk; however, the
heterogeneity of the studies analyzed precludes firm conclusions
(7,22).
Our study failed to show a statistically significant
association between hyperthyroidism and breast carcinogenesis. In
contrast, two studies found a correlation between the
administration of THs and increased risk of mammary cancer
(23,24). The results of epidemiological
studies concerning hyperthyroidism in relation to breast cancer are
unclear and seem to depend on the inclusion of pre- or
post-menopausal patients (25). The
authors speculated that subclinical hyperthyroidism in
postmenopausal women contributed to mammary tumor growth as a
result of an E2-like effect through the interactions
between T3/T4 and ER. In vitro
interactions between T3 and ER have been reported in
breast cancer cell lines (19).
Notably, in our present study, circulating E2 was
significantly reduced in HyperT rats even when mammary
carcinogenesis similar to EUT rats was observed. This result
supports the role of TH as an E2-like factor in mammary
tumor growth.
The mechanism whereby the thyroid gland influences
mammary tumorigenesis is unclear. However, our results suggest that
different thyroid states affect breast tumorigenesis by altering
body growth, breast morphology, body composition and adipocytoquine
release, and patterns of secretion of other hormones such as E, Pg,
PRL and/or GH, among others.
Body growth
GH is required for mammary development since it
stimulates ductal growth, proliferation and secretion (26). The effect of GH on mammary
development is in part mediated by insulin-like growth factor 1
(IGF-1), and the action of E2 and Pg are dependent upon
IGF-1 (27). GH stimulates IGF-1
secretion in the liver and in the mammary stroma, therefore a
paracrine role of GH modulating the effects of other hormones on
proliferation and differentiation of mammary epithelium must not be
ruled out (28). The present
results demonstrated that hypothyroidism diminishes the secretion
of GH in the rat, supporting previous studies (29).
Clinical evidence suggests that high circulating
levels of GH/IGF-1 are associated with an increased risk of
subsequently developing breast cancer (30). In studies in vitro, human GH
was found to promote mammary carcinoma cell proliferation in an
autocrine/paracrine manner (31)
and resulted in a phenotypic conversion of human mammary carcinoma
cells into a more invasive phenotype (32). In our in vivo study, we
observed retardation in body growth associated with a significant
decrease in serum GH levels in HypoT rats, which also had a lower
incidence of mammary cancer.
Breast morphology
Breast development occurs through a process of
ductal elongation, branching and sprouting of ductules or alveoli,
a process that requires extensive cell proliferation and
penetration of the ductal epithelium into the stroma (33). Ductal elongation is directed by
E2, GH, IGF-1 and epidermal growth factor (EGF), whereas
ductal branching and alveolar budding are influenced by additional
factors such as Pg, PRL and THs. The stroma of the mammary gland is
a complex structure composed of an extracellular matrix and a
variety of cell types including endothelial cells, inflammatory
cells, fibroblasts, fibroblast-like cells and adipocytes (34). This compartment can also be referred
to as the mammary fat pad (34)
since it mainly contains adipose tissue (35).
The response by the ductal epithelium to various
hormones and growth factors is modulated by epithelial-stromal
interactions, which seem to be bidirectional (36). It is expected that the marked
variations in the epithelial-stroma ratio influence the
bidirectional connection leading to specific modifications in gene
expression that may account for the different susceptibility or
risk to develop breast cancer (33).
In the present study, we observed that
hypothyroidism retarded the growth of the ductal system and induced
an increase in mammary gland fat deposit. In accordance, Vonderhaar
and Greco (37,38) noticed that the gland of primiparous
hypothyroid adult mice retained a primitive ductal appearance, i.e.
that the epithelial component consisted of a sparse ductal system
with few branches filling about one-fourth of the fat pad.
Coincident with our results, they also described that while glands
from hyperthyroid and euthyroid virgin animals preserved a small
degree of ductal branching with primitive alveoli, the glands from
hypothyroid animals showed less ductal branching and were devoid of
alveoli (37). Thus, the decrease
in mammary tumor incidence in our HypoT rats may be due to the
reduced development of the mammary gland and the decrease in the
parenchyma-stroma ratio.
Body composition and adipocytokine
secretion
THs have important effects on energy balance, since
they influence both energy intake and expenditure. Severe
hypothyroidism in the rat causes a sharp drop in food intake and
metabolism, and this consequently suppresses the growth of
DMBA-induced mammary cancer (11).
Accordingly, in the present study, we observed a decreased food
intake and body weight in HypoT rats in comparison with the
controls and the HyperT animals. Calorie restriction was found to
decrease proliferation and angiogenesis and increases apoptosis in
mammary tumors (39).
Many changes related to energy homeostasis, body
weight and food intake are associated with alterations in the
function of adipose tissue. Leptin is the obese gene product
secreted exclusively by adipocytes. Accumulating evidence shows
that leptin is an important pro-angiogenic, pro-inflammatory and
mitogenic factor, whose actions are reinforced through crosstalk
with cytokines/growth factors (40). Increasing leptin levels activate the
thyroid, GH, and gonadal axes (41). Thus, it is conceivable that leptin
may have a role in different states of thyroidal disease. This
relationship has been the subject of several studies, but no
uniform picture has yet emerged from the results. Valcavi et
al(42) reported significantly
reduced serum leptin concentrations before and during replacement
therapy in patients with hypothyroidism. Other authors did not
observe alterations in serum leptin in different hypothyroid
conditions (43) and Leonhardt
et al(44) described
increased leptin concentrations in patients with primary
hypothyroidism or thyroid carcinoma compared with euthyroid
controls.
In hypothyroid Wistar rats, a decrease in
metabolizable energy intake and energy expenditure together with a
shift in lipid and protein partitioning was found after 7 and 15
days of treatment with PTU (11).
Consequently, body lipid percentage significantly increased
compared to euthyroid rats. Our results in Sprague-Dawley rats
showed that, even though the animals treated with PTU had a lower
body weight than the controls, the percentage of body fat was
similar in the three studied groups. However, adipose tissue of the
HypoT rats appeared to be dysfunctional, attending to the lower
amount of leptin expressed per gram of fat and the diminished serum
levels of leptin in these animals. This fact can be one of the
possible mechanisms related to the increased latency in HypoT rats
since leptin has been shown to inhibit apoptosis and to stimulate
growth (45) and also angiogenesis
in breast carcinogenesis (46).
However, in our in vivo study, we did not observe changes in
tumor cell proliferation as evaluated by the mitotic index and by a
more sensitive method, PCNA immunostaining. In turn, in cancer
cells of our HypoT rats apoptosis was augmented in an inverse
relation to serum leptin levels. In support of our results, it has
been described that leptin inhibits apoptosis and this action is
correlated with increased expression of anti-apoptotic protein
Bcl-2 (47).
To summarize, dysfunctional body fat secreting lower
levels of leptin may be another factor responsible for the delayed
development of mammary tumors in HypoT rats. We cannot rule out the
possible role of the stroma in the production of adipokines as
tumor growth factors. Ongoing studies in our laboratory aim to
evaluate the involvement of leptin locally produced by the
peritumoral fat pad in the autocrine/paracrine regulation of tumor
growth.
Hormones targeting the mammary gland
In the present study, we demonstrated that chronic
hypothyroidism induced by PTU treatment modifies certain hormone
patterns. E2, which plays an essential role in mammary
gland development and carcinogenesis, was diminished in HypoT rats.
E2 and Pg are known to promote proliferation and
differentiation in the normal breast. Most breast cancers are
initially hormone dependent and it is well accepted that
E2 plays a crucial role in their development and
progression (48). E2
exerts carcinogenic effects by simultaneously increasing the number
of DNA replication errors by stimulating cell proliferation and
gene expression; and through its oxidative metabolism that forms
DNA damaging species (49).
There is evidence that Pg and its related signaling
pathways are important players in the induction, progression and
maintenance of the neoplastic phenotype in the mammary gland
(50). However, in our in
vivo study we did not find any relationship between circulating
Pg and mammary carcinogenesis in rats with different thyroid
disorders.
On the other hand, PRL secretion during diestrus was
not affected in our experimental thyroid conditions. It has been
previously shown that hypothyroidism can increase serum levels of
PRL during estrous without changing basal secretion on diestrous
day (29). In the present study,
based on the similar values of circulating PRL between the
treatments, we cannot attribute a relevant role of this hormone on
mammary carcinogenesis. Moreover, we did not observe any
significant differences in serum PRL in rats with and without
tumors; further supporting the minor role of PRL in breast cancer
development in HypoT rats.
In conclusion, our results to date show that
hypothyroidism alters animal growth, breast morphology, body
composition and adipocytoquine secretion and serum E2
enhancing apoptosis, consequently retarding mammary carcinogenesis
in rats. Additional studies by us are currently underway to
investigate the involved molecular mechanisms.
Acknowledgements
The present study was partially supported by grants
from Instituto Nacional del Cáncer and from CONICET (Consejo
Nacional de Investigaciones Científicas y Técnicas), Argentina. The
authors are deeply indebted to Mrs. Elina Guiñazú de Di Nasso, Miss
Paola Estalles and Mr. Juan Rosales for their excellent technical
assistance and to Ms. María G. Zubiría and Dr Eduardo Spinedi for
the RIA of leptin.
References
|
1
|
Neville MC, McFadden TB and Forsyth I:
Hormonal regulation of mammary differentiation and milk secretion.
J Mammary Gland Biol Neoplasia. 7:49–66. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Jensen EV, Cheng G, Palmieri C, Saji S,
Mäkelä S, Van Noorden S, Wahlström T, Warner M, Coombes RC and
Gustafsson JA: Estrogen receptors and proliferation markers in
primary and recurrent breast cancer. Proc Natl Acad Sci USA.
98:15197–15202. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Lanari C and Molinolo AA: Progesterone
receptors - animal models and cell signalling in breast cancer.
Diverse activation pathways for the progesterone receptor: possible
implications for breast biology and cancer. Breast Cancer Res.
4:240–243. 2002. View
Article : Google Scholar
|
|
4
|
Clevenger CV, Furth PA, Hankinson SE and
Schuler LA: The role of prolactin in mammary carcinoma. Endocr Rev.
24:1–27. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Frontini L, Lissoni P, Vaghi M, Perego MS,
Pescia S, Ardizzoia A and Gardani G: Enhancement of the efficacy of
weekly low-dose taxotere by the long acting anti-prolactinemic drug
cabergoline in pretreated metastatic breast cancer. Anticancer Res.
24:4223–4226. 2004.PubMed/NCBI
|
|
6
|
Shen Q, Lantvit DD, Lin Q, Li Y, Christov
K, Wang Z, Unterman TG, Mehta RG and Swanson SM: Advanced rat
mammary cancers are growth hormone dependent. Endocrinology.
148:4536–4544. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Angelousi AG, Anagnostou VK, Stamatakos
MK, Georgiopoulos GA and Kontzoglou KC: Mechanisms in
endocrinology: primary HT and risk for breast cancer: a systematic
review and meta-analysis. Eur J Endocrinol. 166:373–381. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Cristofanilli M, Yamamura Y, Kau SW,
Bevers T, Strom S, Patangan M, Hsu L, Krishnamurthy S, Theriault RL
and Hortobagyi GN: Thyroid hormone and breast carcinoma. Primary
hypothyroidism is associated with a reduced incidence of primary
breast carcinoma. Cancer. 103:1122–1128. 2005.PubMed/NCBI
|
|
9
|
Barrera-Hernandez G, Park KS, Dace A, Zhan
Q and Cheng SY: Thyroid hormone-induced cell proliferation in GC
cells is mediated by changes in G1 cyclin/cyclin-dependent kinase
levels and activity. Endocrinology. 140:5267–5274. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Jeong YJ, Bong JG, Park SH, Choi JH and Oh
HK: Expression of leptin, leptin receptor, adiponectin, and
adiponectin receptor in ductal carcinoma in situ and invasive
breast cancer. J Breast Cancer. 14:96–103. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Goodman AD, Hoekstra SJ and Marsh PS:
Effects of hypothyroidism on the induction and growth of mammary
cancer induced by 7,12-dimethylbenz(a)anthracene in the rat. Cancer
Res. 40:2336–2342. 1980.PubMed/NCBI
|
|
12
|
Giovambattista A, Piermaria J, Suescun MO,
Calandra RS, Gaillard RC and Spinedi E: Direct effect of ghrelin on
leptin production by cultured rat white adipocytes. Obesity.
14:19–27. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Russo J and Russo IH: Atlas and histologic
classification of tumors of the rat mammary gland. J Mammary Gland
Biol Neoplasia. 5:187–200. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
López-Fontana CM, Maselli ME, Salicioni AM
and Carón RW: The inhibitory effect of progesterone on lactogenesis
during pregnancy is already evident by mid- to late gestation in
rodents. Reprod Fertil Dev. 24:704–714. 2012.PubMed/NCBI
|
|
15
|
Cuello-Carrión FD and Ciocca DR: Improved
detection of apoptotic cells using a modified in situ TUNEL
technique. J Histochem Cytochem. 47:837–839. 1999.PubMed/NCBI
|
|
16
|
Vazquez-Prieto MA, Renna NF, Diez ER,
Cacciamani V, Lembo C and Miatello RM: Effect of red wine on
adipocytokine expression and vascular alterations in fructose-fed
rats. Am J Hypertens. 24:234–240. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Ito K and Maruchi N: Breast cancer in
patients with Hashimoto’s thyroiditis. Lancet. 2:1119–1121.
1975.
|
|
18
|
Shering SG, Zbar AP, Moriarty M, McDermott
EW, O’Higgins NJ and Smyth PP: Thyroid disorders and breast cancer.
Eur J Cancer Prev. 5:504–506. 1996.
|
|
19
|
Nogueira CR and Brentani MM:
Triiodothyronine mimics the effects of estrogen in breast cancer
cell lines. J Steroid Biochem Mol Biol. 59:271–279. 1996.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Brinton LA, Hoffman DA, Hoover R and
Fraumeni JF Jr: Relationship of thyroid disease and use of thyroid
supplements to breast cancer risk. J Chronic Dis. 37:877–893. 1984.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Martinez-Iglesias O, Garcia-Silva S,
Regadera J and Aranda A: Hypothyroidism enhances tumor invasiveness
and metastasis development. PLoS One. 4:e64282009. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Hardefeldt PJ, Eslick GD and Edirimanne S:
Benign thyroid disease is associated with breast cancer: a
meta-analysis. Breast Cancer Res Treat. 133:1169–1177. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Kapdi CC and Wolfe JN: Breast cancer.
Relationship to thyroid supplements for hypothyroidism. JAMA.
236:1124–1127. 1976. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Mustacchi P and Greenspan F: Thyroid
supplementation for hypothyroidism. An latrogenic cause of breast
cancer? JAMA. 237:1446–1447. 1977. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Saraiva PP, Figueiredo NB, Padovani CR,
Brentani MM and Nogueira CR: Profile of thyroid hormones in breast
cancer patients. Braz J Med Biol Res. 38:761–765. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Raccurt M, Lobie PE, Moudilou E,
Garcia-Caballero T, Frappart L, Morel G and Mertani HC: High
stromal and epithelial human gh gene expression is associated with
proliferative disorders of the mammary gland. J Endocrinol.
175:307–318. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Kleinberg DL, Wood TL, Furth PA and Lee
AV: Growth hormone and insulin-like growth factor-I in the
transition from normal mammary development to preneoplastic mammary
lesions. Endocr Rev. 30:51–74. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Thijssen JH: On the possible role of
mammary-derived growth hormone in human breast cancer. Maturitas.
65(Suppl 1): S13–S16. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Hapon MB, Gamarra-Luques C and Jahn GA:
Short term hypothyroidism affects ovarian function in the cycling
rat. Reprod Biol Endocrinol. 8:142010. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Rinaldi S, Toniolo P, Muti P, Lundin E,
Zeleniuch-Jacquotte A, Arslan A, Micheli A, Lenner P, Dossus L,
Krogh V, Shore RE, Koenig KL, Riboli E, Stattin P, Berrino F,
Hallmans G, Lukanova A and Kaaks R: IGF-I, IGFBP-3 and breast
cancer in young women: a pooled re-analysis of three prospective
studies. Eur J Cancer Prev. 14:493–496. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Kaulsay KK, Mertani HC, Törnell J, Morel
G, Lee KO and Lobie PE: Autocrine stimulation of human mammary
carcinoma cell proliferation by human growth hormone. Exp Cell Res.
250:35–50. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Mukhina S, Mertani HC, Guo K, Lee KO,
Gluckman PD and Lobie PE: Phenotypic conversion of human mammary
carcinoma cells by autocrine human growth hormone. Proc Natl Acad
Sci USA. 101:15166–15171. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Russo J, Lynch H and Russo IH: Mammary
gland architecture as a determining factor in the susceptibility of
the human breast to cancer. Breast J. 7:278–291. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Tan J, Buache E, Chenard MP, Dali-Youcef N
and Rio MC: Adipocyte is a non-trivial, dynamic partner of breast
cancer cells. Int J Dev Biol. 55:851–859. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Neville MC, Medina D, Monks J and Hovey
RC: The mammary fat pad. J Mammary Gland Biol Neoplasia. 3:109–116.
1998. View Article : Google Scholar
|
|
36
|
Petersen OW, Rønnov-Jessen L, Weaver VM
and Bissell MJ: Differentiation and cancer in the mammary gland:
shedding light on an old dichotomy. Adv Cancer Res. 75:135–161.
1998. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Vonderhaar BK and Greco AE: Effect of
thyroid status on development of spontaneous mammary tumors in
primiparous C3H mice. Cancer Res. 42:4553–4561. 1982.PubMed/NCBI
|
|
38
|
Vonderhaar BK and Greco AE:
Lobulo-alveolar development of mouse mammary glands is regulated by
thyroid hormones. Endocrinology. 104:409–418. 1979. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Knight BB, Oprea-Ilies GM, Nagalingam A,
Yang L, Cohen C, Saxena NK and Sharma D: Survivin upregulation,
dependent on leptin-EGFR-Notch1 axis, is essential for
leptin-induced migration of breast carcinoma cells. Endocr Relat
Cancer. 18:413–428. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Zhou W, Guo S and Gonzalez-Perez RR:
Leptin pro-angiogenic signature in breast cancer is linked to IL-1
signalling. Br J Cancer. 104:128–137. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Mantzoros CS: The role of leptin in human
obesity and disease: a review of current evidence. Ann Intern Med.
130:671–680. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Valcavi R, Zini M, Peino R, Casanueva FF
and Dieguez C: Influence of thyroid status on serum immunoreactive
leptin levels. J Clin Endocrinol Metab. 82:1632–1634.
1997.PubMed/NCBI
|
|
43
|
Sreenan S, Caro JF and Refetoff S: Thyroid
dysfunction is not associated with alterations in serum leptin
levels. Thyroid. 7:407–409. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Leonhardt U, Ritzel U, Schäfer G, Becker W
and Ramadori G: Serum leptin levels in hypo- and hyperthyroidism. J
Endocrinol. 157:75–79. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Housa D, Housová J, Vernerová Z and
Haluzik M: Adipocytokines and cancer. Physiol Res. 55:233–244.
2006.
|
|
46
|
Rose DP, Gilhooly EM and Nixon DW: Adverse
effects of obesity on breast cancer prognosis, and the biological
actions of leptin (Review). Int J Oncol. 21:1285–1292.
2002.PubMed/NCBI
|
|
47
|
Artwohl M, Roden M, Hölzenbein T,
Freudenthaler A, Waldhäusl W and Baumgartner-Parzer SM: Modulation
by leptin of proliferation and apoptosis in vascular endothelial
cells. Int J Obes Relat Metab Disord. 26:577–580. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Pasqualini JR: Breast cancer and steroid
metabolizing enzymes: the role of progestogens. Maturitas. 65(Suppl
1): S17–S21. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Bolton JL and Thatcher GR: Potential
mechanisms of estrogen quinone carcinogenesis. Chem Res Toxicol.
21:93–101. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Goepfert TM, McCarthy M, Kittrell FS,
Stephens C, Ullrich RL, Brinkley BR and Medina D: Progesterone
facilitates chromosome instability (aneuploidy) in p53 null normal
mammary epithelial cells. FASEB J. 14:2221–2229. 2000. View Article : Google Scholar : PubMed/NCBI
|