Introduction
Lung cancer is one of the leading causes of cancer
death. It is the most widely spread carcinoma with a very poor
prognosis. Although chemotherapy is widely used for treatment of
lung cancer, the survival rate is limited (1). Recent study suggested that herbal
medicines show promise for lung cancer therapy with less toxicity
(2,3). Glycyrrhiza uralensis has been
shown to exhibit different pharmacological effects and antioxidant
activity against oxidative stress and is used for treatment of
various diseases including cancer (4–9).
Previous studies showed Glycyrrhiza uralensis contained
various types of triterpene, saponins, flavonoids and phenolic
compounds (3). However, the major
ingredients of Glycyrrhiza uralensis may have different
biologic activities. In the present study, we report on experiments
designed to test the anticancer activities of the active phenolic
compounds of Glycyrrhiza uralensis in the A549 lung cancer
cell line.
Materials and methods
Materials
All chemicals of analytical grade were purchased
from USB Corporation (Cleveland, OH, USA). The dried root of
Glycyrrhiza uralensis Fisch was obtained from a local
pharmaceutical store. Liquiritin, isoliquiritin and
isoliquiritigenin were purchased from Shanghai Tauto Biotech Co.,
Ltd. (Shanghai, China). HPLC grade acetonitrile (Merck, Whitehouse
Station, NJ, USA), methanol (Fisher, Fair Lawn, NJ, USA) and
trifluoroacetic acid were used for chromatography. Water was
purified by Milli-Q Academic purification system (Millipore,
Billerica, MA, USA) prior to experiment. Preparation of the active
fraction was prepared according to the established method with
modifications using different solvent systems (3).
Fetal boving serum (FBS), penicillin streptomycin
antibiotic solution (PS), Roswell Park Memorial Institute
(RPMI)-1640 media and Trypsin-EDTA were purchased from Invitrogen
(Carlsbad, CA, USA). Kaighn’s modification of Ham’s F-12 Medium
(F-12K) was purchased from Gibco Co. (Carlsbad, CA, USA).
Primary antibodies against phospho-AKT (ser473),
phospho-AKT (thr308), Akt, p-c-Raf (ser259), p-GSK-3β (ser9),
p-Pten (ser380), p-PDK1 (ser241), p53, cleaved caspase-3,
caspase-3, PARP, cleaved PARP, caspase-9, cleaved caspase-9,
caspase-7, cleaved caspase-7 and anti-rabbit lgG were purchased
from Cell Signaling Technology, Inc. (Danvers, MA, USA). p21, Bax,
Bid and Bcl-2 were purchased from Santa Cruz Biotechnology (Santa
Cruz, CA, USA). GAPDH was obtained from Ambion/Applied Biosystems
(Foster City, CA, USA) and anti-mouse from Zymed Laboratories
(Carlsbad, CA, USA).
Cell line and culture
The A549 cell line was obtained from American Type
Culture Collection (ATCC, Rockville, MD, USA) and cultured with
specific medium with 10% fetal bovine serum (FBS) and 1%
penicillin-streptomycin (PS) in a 37°C incubator with 5%
CO2. Subcultures were performed by using 0.25% trypsin
solution.
MTT assay
The inhibition effects of the ethyl acetate extract
were evaluated with the A549 cell line. A549 cells were seeded onto
96-well plate at the concentration of 5×103. After 24-h
incubation, cells were treated with iquiritin, isoliquiritin and
isoliquiritigenin for 24, 48 and 72 h, respectively. MTT (20 μl)
was added to each well and the plate was kept at 37°C for 2 h. The
purple formazan so formed in the well was dissolved by 150 μl of
DMSO. The absorbance of the product was measured at 540 nm. Cells
without treatment served as vehicle control.
LDH assay
The CytoTox 96 Non-Radioactive cytotoxicity kit
(Promega, Madison, WI, USA) was used to evaluate liquiritin,
isoliquiritin and isoliquiritigenin cytotoxicity by assessing the
total release of cytosolic lactate dehydrogenase in the culture
medium. After 24-h incubation, the cell samples were lysed by
adding 10 μl of Lysis 10X Solution [9% (v/v) Triton X-100 in water]
per 100 μl of culture medium, followed by incubation at 37°C for 60
min. The supernatants of the cell sample (50 μl) were mixed with
the reconstituted substrate mix (50 μl). The enzymatic assay was
stopped after 30 min by adding 50 μl/well of the stop solution (1 M
acetic acid). The plate was read at 490 nm using the plate reader.
The amount of LDH release was measured to calculate whole LDH pool
in control cells.
Flow cytometric analysis of apoptosis and
cell cycle
A549 cells were treated with the active fraction
that contained liquiritin, isoliquiritin and isoliquiritigenin for
24 h. Cells were harvested with trypsin and were washed with cold
PBS, followed by staining with FITC-PI or PI for the apoptosis or
cell cycle study. Fluorescence-activated cells were sorted with
10,000 events per sample in triplicate using a BD Biosciences
FACSCanto instrument. The results were analyzed with the FlowJo
software version 7.6.1.
Caspase-3/7 assay
The caspase-3/7 substrate Rhodamine 110,
Bis-(N-CBZ-L-aspartyl-L-valyl-L-aspartic acid amide; Z-DEVD-R110),
is a profluorescent substrate used to study the caspase-3
activities. Cells were treated with the active mixture prepared
according to a previous method (3).
After 24-h incubation, Apo-One® Homogeneous Caspase-3/7
assay was performed according to the Promega Techinical protocol.
The prepared reagent (100 μl) was added to each well (1:1)
including the blank, the control or treated cells in culture plate.
The mixture was placed in a plate shaker at 300 rpm and incubated
at room temperature for 2 h. The fluorescence of each well was
measured at excitation 485 nm and emission 520 nm using Tecan
microplate reader Infinite M200.
Western blot analysis
The cellular proteins were prepared by using lysis
buffer [50 mM Trizma base, 100 mM NaCl, 5 mM EDTA, 67 mM sodium
pyrophosphate and 1% Triton X-100, supplemented with protease
inhibitor cocktail (Roche, Mannheim, Germany) and 0.5 mM sodium
orthovanadate] on ice. The proteins were collected by
centrifugation at 8,000 rpm at 4°C for 30 min. The protein
concentration was determined by the DC Protein assay (Bio-Rad
Laboratories) using BSA as the standard. Proteins (60 μg) were
subjected to 8–15% SDS-PAGE to separate the protein using 50–120
voltages for 2 h, and were transferred to a PVDF membrane for 45
min at 12 voltages (Pall Corp., Ann Arbor, MI, USA). After blocking
the transfer action with non-fat dry milk (5% w/v) in TBST for 1 h
at room temperature, the membrane was incubated with 1:1,000
specific primary antibodies with 5% non-fat milk or BSA in TBST at
4°C overnight. After washing with TBST, the membrane was incubated
with 1:5,000 HPR-conjugated goat anti-rabbit or goat anti-mouse
secondary antibodies for 1 h. Blots were developed using the ECL
chemiluminescence detection reagent (GE Healthcare Life Sciences,
Pittsburgh, PA, USA) according to the manufacturer’s protocol. The
membranes were re-probed with GAPDH as loading reference. The blots
were quantified by densitometry and normalized in order to correct
any error in protein loading. Densitometric analysis was performed
with Gel-Pro 32 software.
Statistical analysis
Statistical analysis of raw data was carried out by
one-way analysis of variance (ANOVA) followed by the Tukey’s Post
Hoc test. The data are expressed as means ± SD (n=3).
Results
Cell viability assay
Table I shows the
effects of liquiritin, isoliquiritin, isoliquiritigenin and the
ethyl acetate extract on the viability of A549 cell lines by MTT
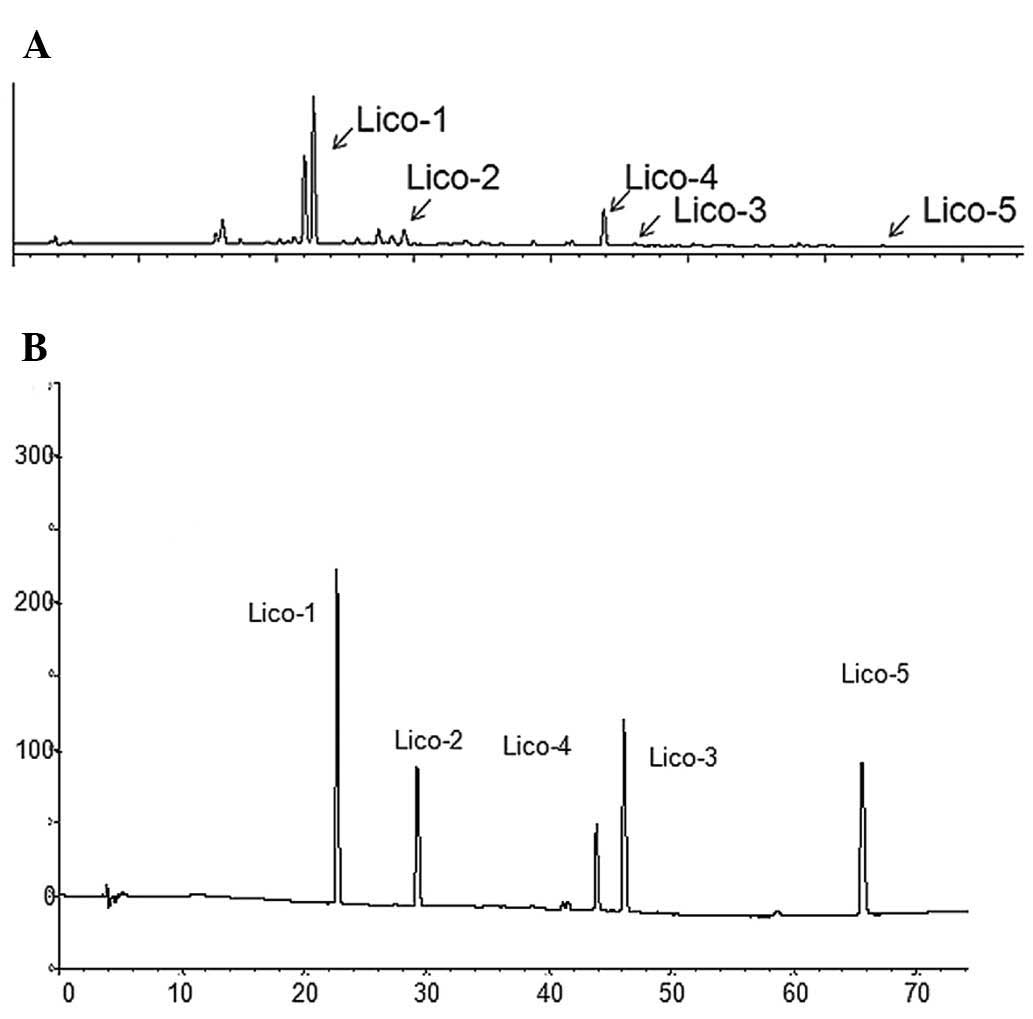
assay. Fig. 1 shows that the active
fraction contained liquiritin, isoliquiritin and isoliquiritigenin
only. The active compounds and the extract inhibited the growth of
A549 cells. MTT results show that liquiritin, isoliquiritin and
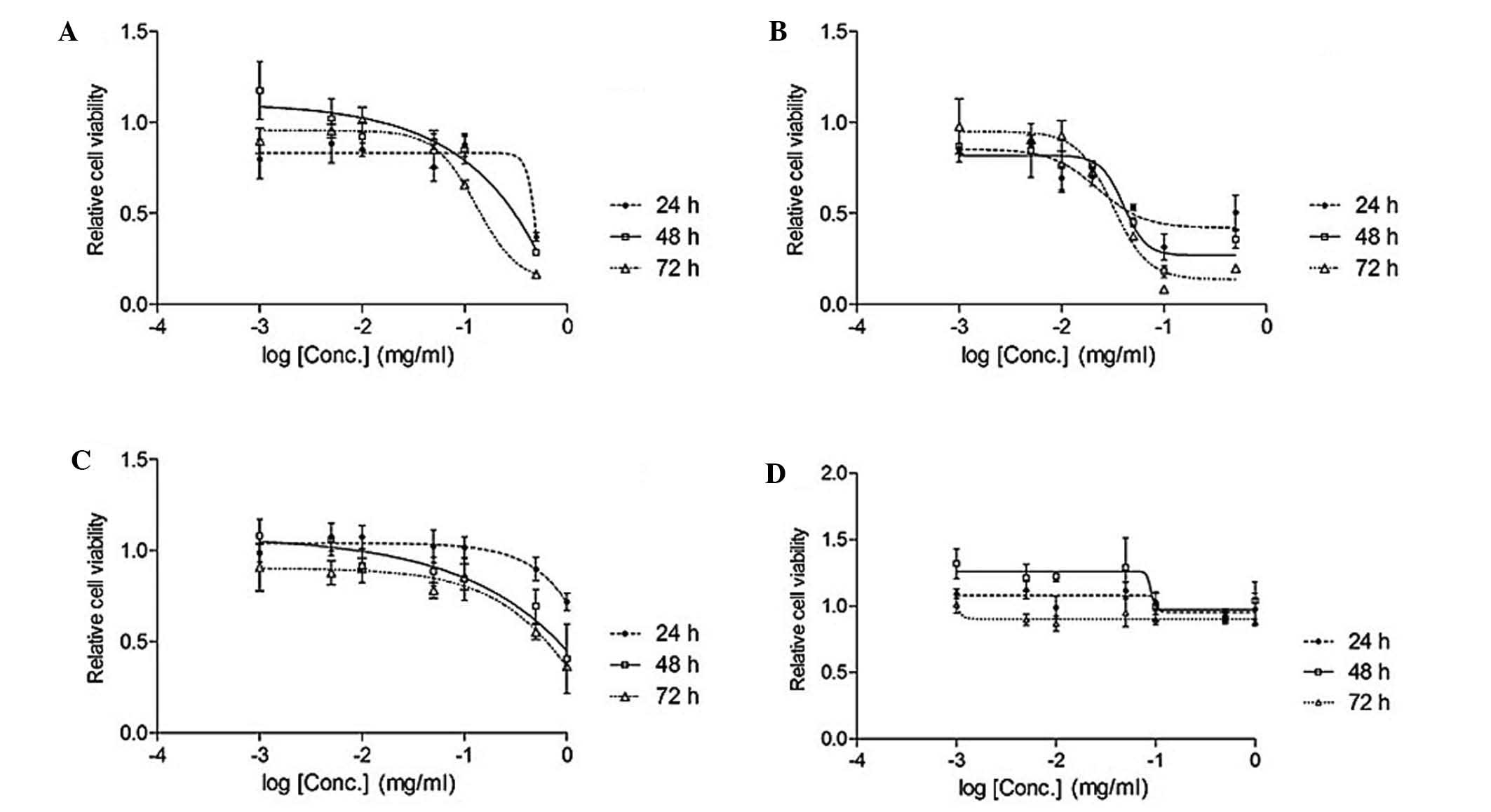
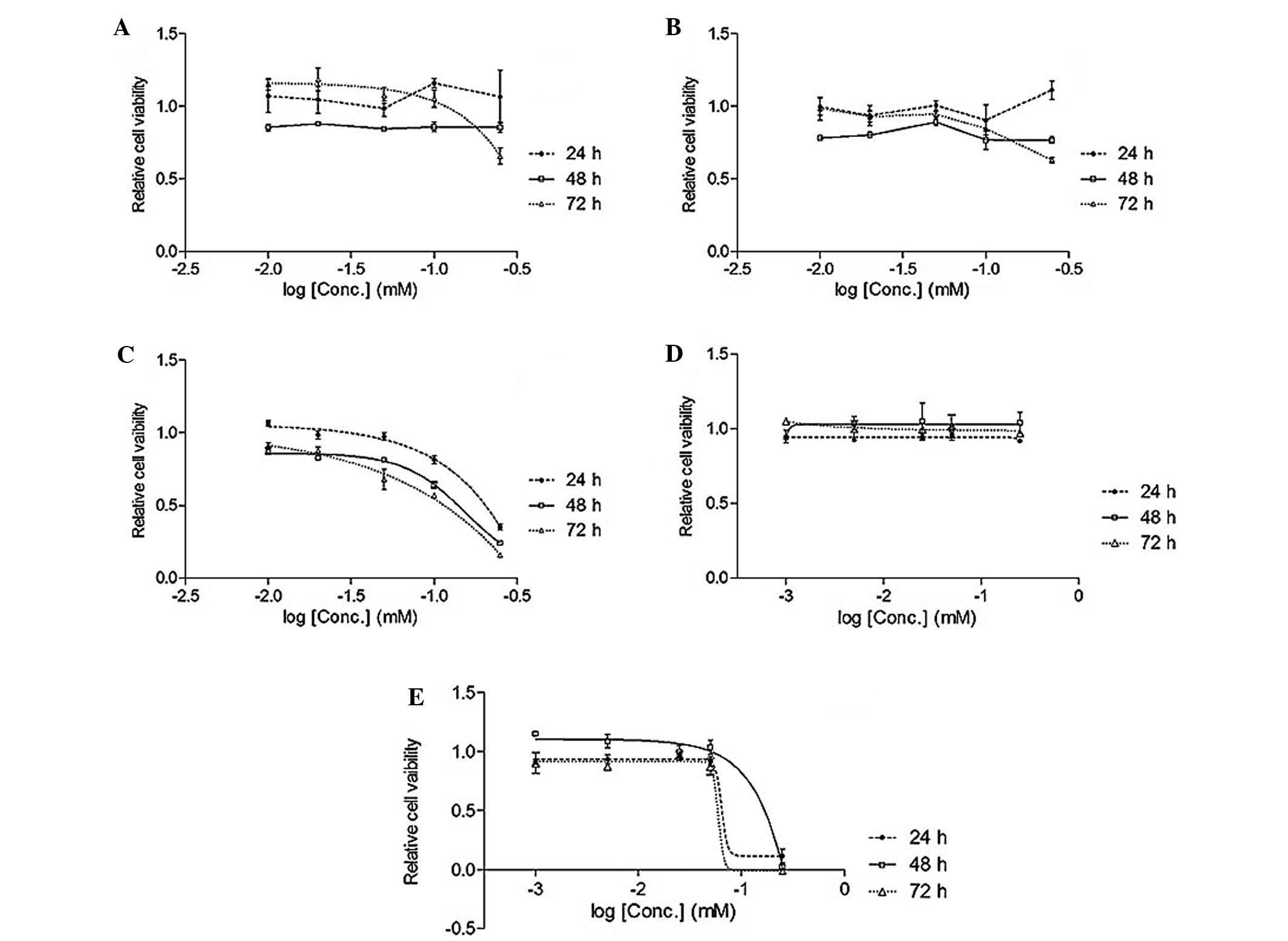
isoliquiritigenin inhibited the lung cancer cell line (Figs. 2 and 3).
 | Table IIC50 of Glycyrrhiza
uralensis extract and standard compounds in A549 lung cancer
cell line. |
Table I
IC50 of Glycyrrhiza
uralensis extract and standard compounds in A549 lung cancer
cell line.
| Fraction | Incubation period (h)
(24/48/72) |
|---|
| EAL (mg/ml) | 0.049 |
| Lico-1 (mM) | ND |
| Lico-2 (mM) | ND |
| Lico-3 (mM) |
0.093/0.065/0.037 |
| Lico-4 (mM) | ND |
| Lico-5 (mM) | 0.14/0.087/0.074 |
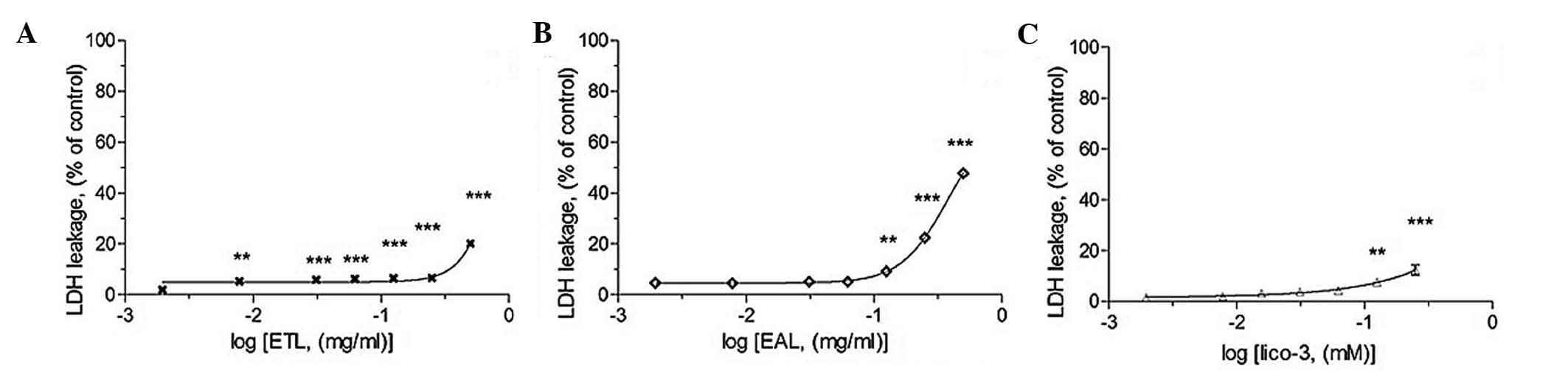
LDH leakage assay
The LDH assay shows the cytotoxic effects of the
active compounds on A549 cells (Fig.
4). During necrosis, lactate dehydrogenase (LDH) would leak
from the inner to the outer of cell since the membrane is broken.
Fig. 4 showed that the active
extract induced necrosis in A549 cells. Since the LDH leakage was
triggered by the noted, a lower concentration range of the active
compounds with relatively low LDH leakage, was chosen to evaluate
its apoptotic potential.
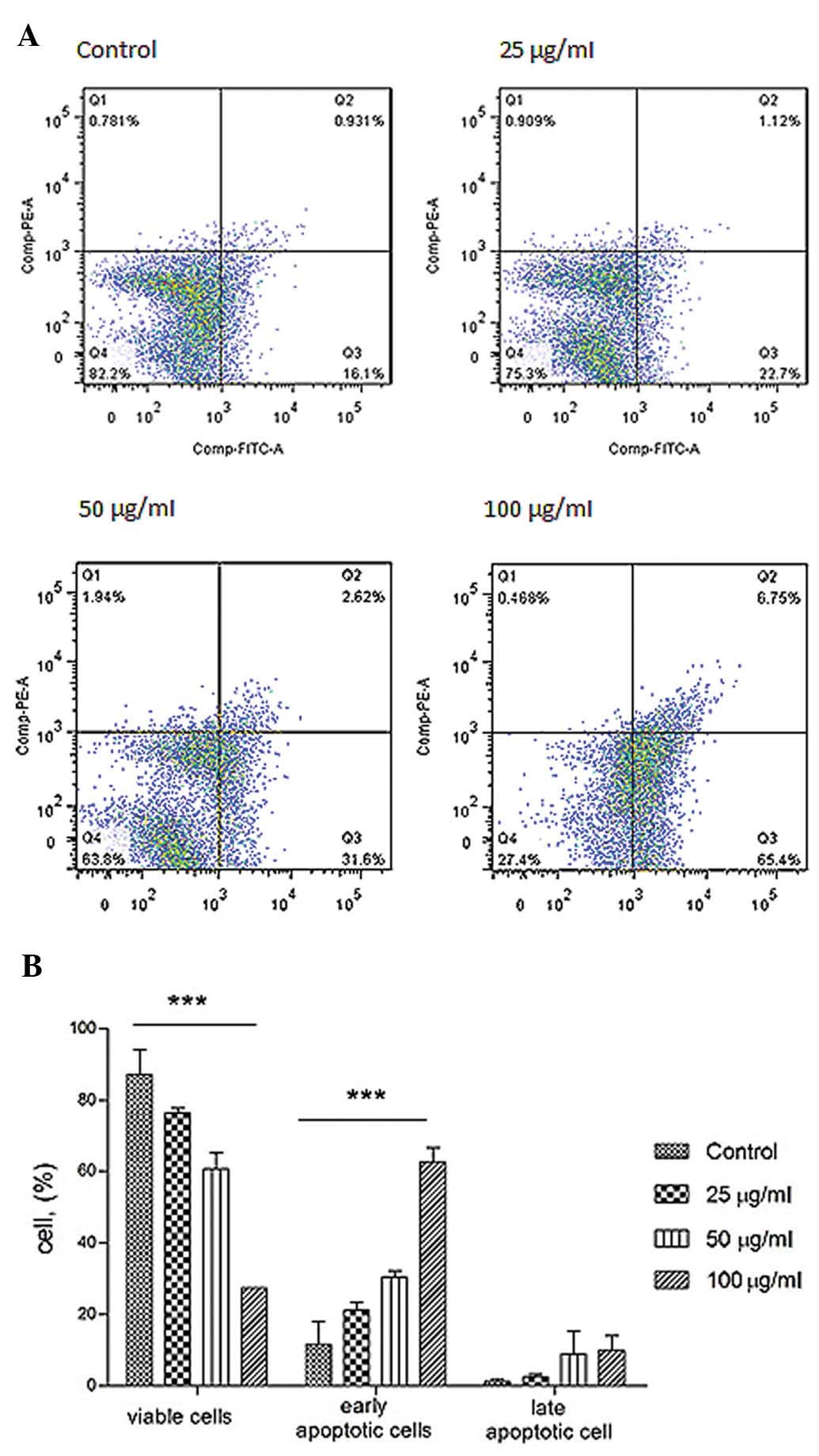
Annexin V and PI staining
The mode of cell death induced was evaluated to
discover how it induced cell death. Annexin V-FITC and propidium
iodide staining followed, cytometric analysis were carried out to
study the cellular responses. Under the assay condition, the
majority of cells were not stained with Annexin V and PI (Fig. 5). During apoptosis, outward
translocation of phosphory-serine binds to Annexin V which was
conjugated with FITC. In contract, infusion of PI into cells during
late apoptosis/necrosis was permitted staining the DNA. The cell
viability after 24-h treatment was increased.
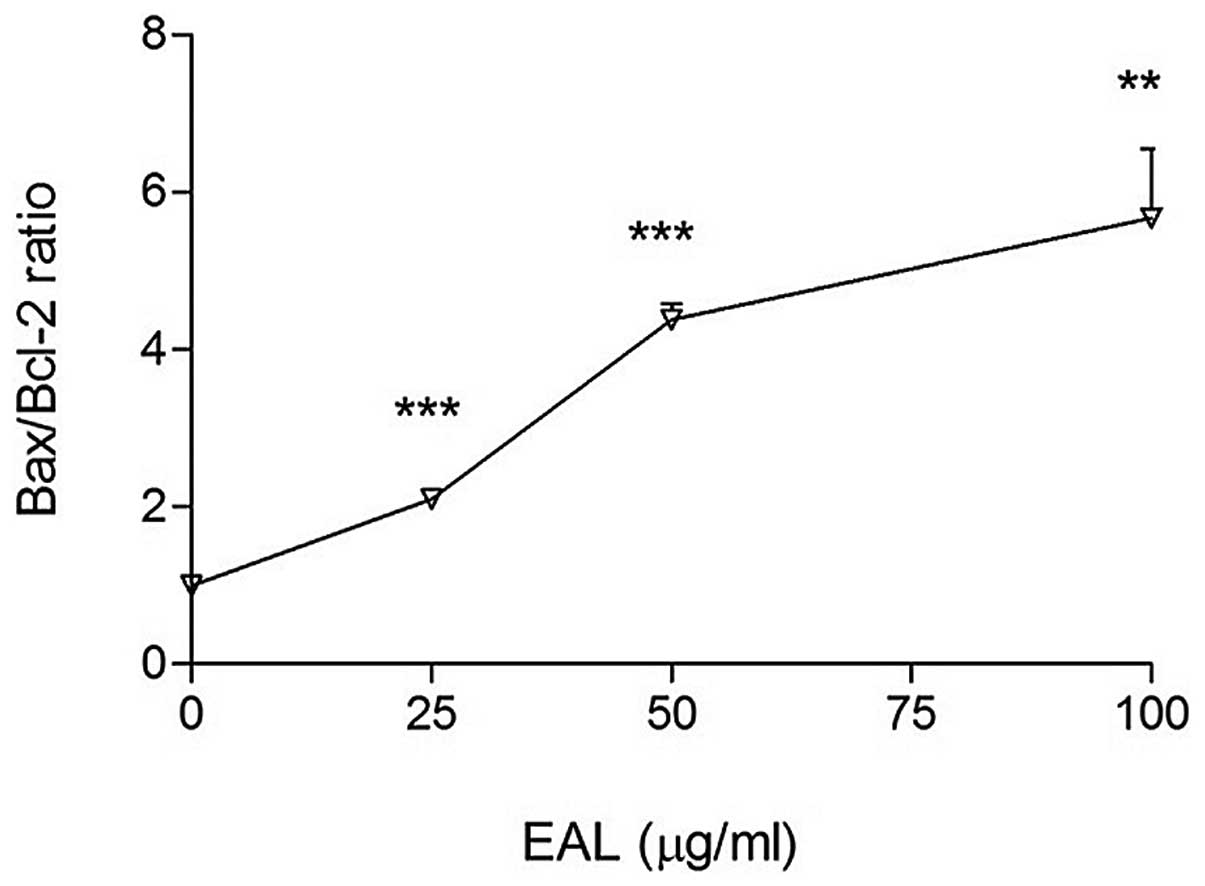
Activation of caspase and Bcl-2
family
Bcl-2 family proteins regulate caspase activation.
The pro-apoptotic and anti-apoptotic protein members in the family
determine the fate of the cell. Bcl-2 family includes pro-apoptotic
proteins such as Bax, Bak, Bad, Bid and anti-apoptotic proteins
such as Bcl-2 and Bcl-XL. The Bcl-2 family regulates apoptosis
through Bcl-2 (inhibitor) or Bax (activator). The ratio of
Bax/Bcl-2 is a key factor in regulation of apoptosis. The
expression levels of Bcl-2 family, Bcl-2, Bax and Bid were measured
with western blot analysis (Figs.
6–8) and the ratio of Bax/Bcl-2
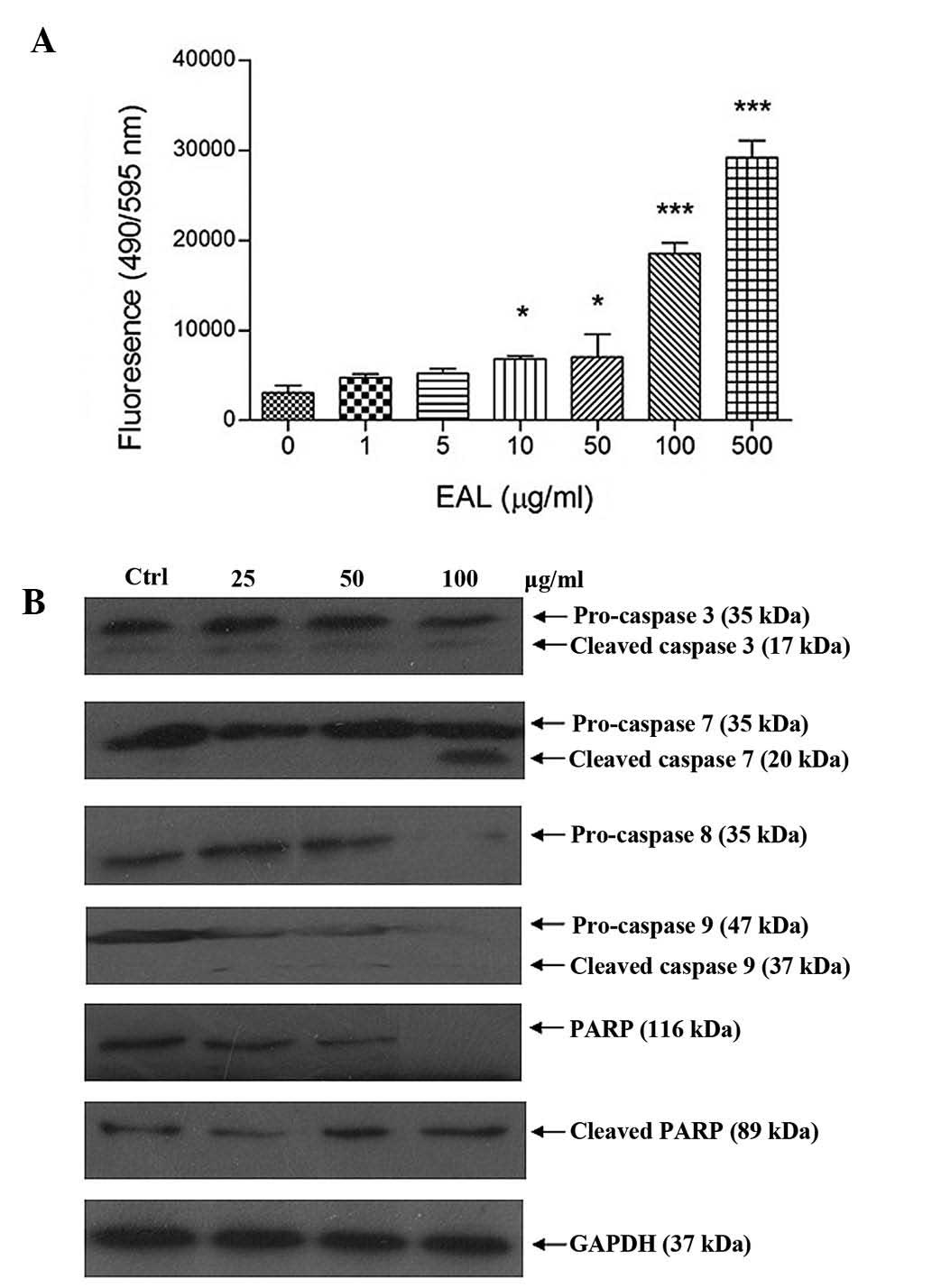
was recorded. Apo-One Homogeneous Caspase-3/7 assay kit was used to
measure caspase-3/7 activities of the A549 cells after treatment.
The assay measured the non-fluorescent caspase substrate
Z-DEVD-R110 that is cleaved by caspase-3/7, producing the
fluorescent Rhodamine 110. Fluorescence generated in the experiment
was proportional to the amount of caspase-3/7 cleavage activity in
the cultured sample. Fig. 8 shows
the increase of fluorescence intensity of caspase-3/7 after
treatment.
Caspase family protein expression level in A549
cells after treatment was analyzed by western blotting. The
intensity of protein was measured using the Gel-Pro software. The
results showed that it caused cleavage of caspase family and
PARP.
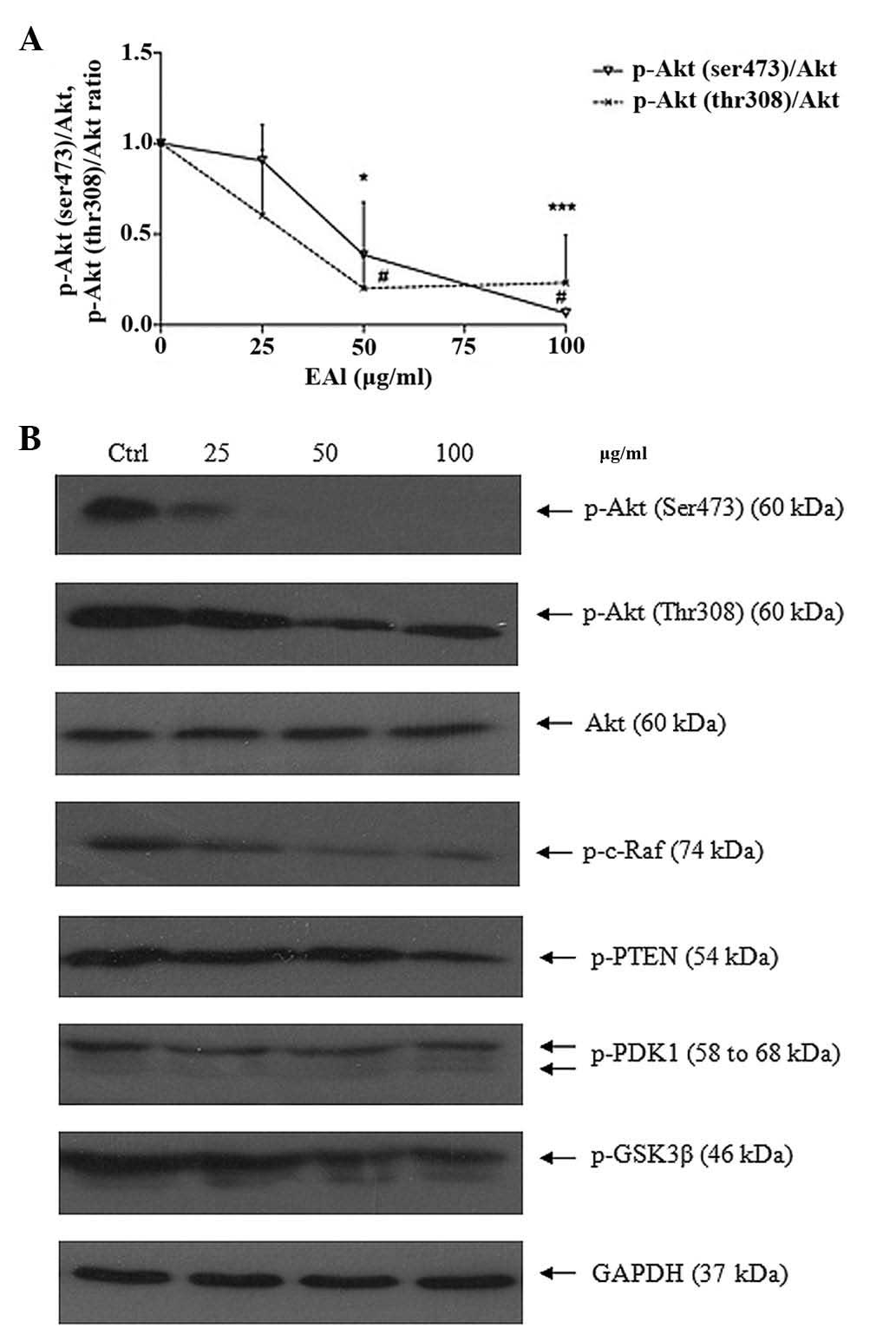
Suppress Akt survival pathway
The serine/threonine protein kinase Akt (or protein
kinase B) activity is related to cancer. Akt inhibits apoptosis by
phosphorylation to initiate multiple additional conversions that
directly mediate the action of cell survival or apoptosis. The
protein expression level of Akt pathway including Akt
phoshorylation forms, p-DK1, GSK-3, PTEN and Raf was studied by
western blotting (Fig. 9). EAL
could inhibit the phosphorylation of Ak, p-PDK1, p-GSK-3β, p-PTEN
and p-c-Raf. The ratio of p-Akt (Ser, Thr) over total Akt was
measured.
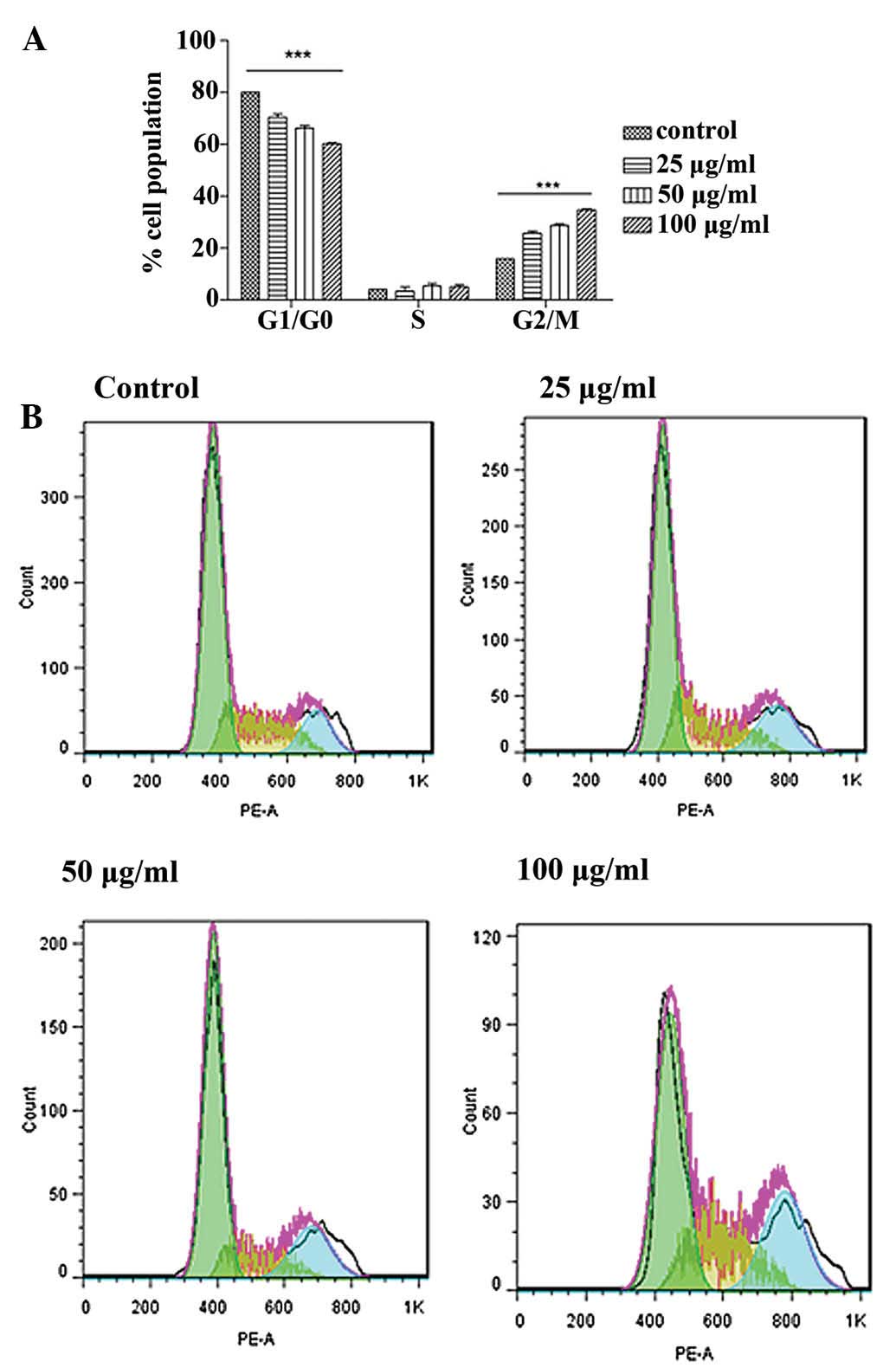
Cell cycle arrest
To identify whether the growth inhibitory effect is
caused by specific perturbation of cell cycle-related events, DNA
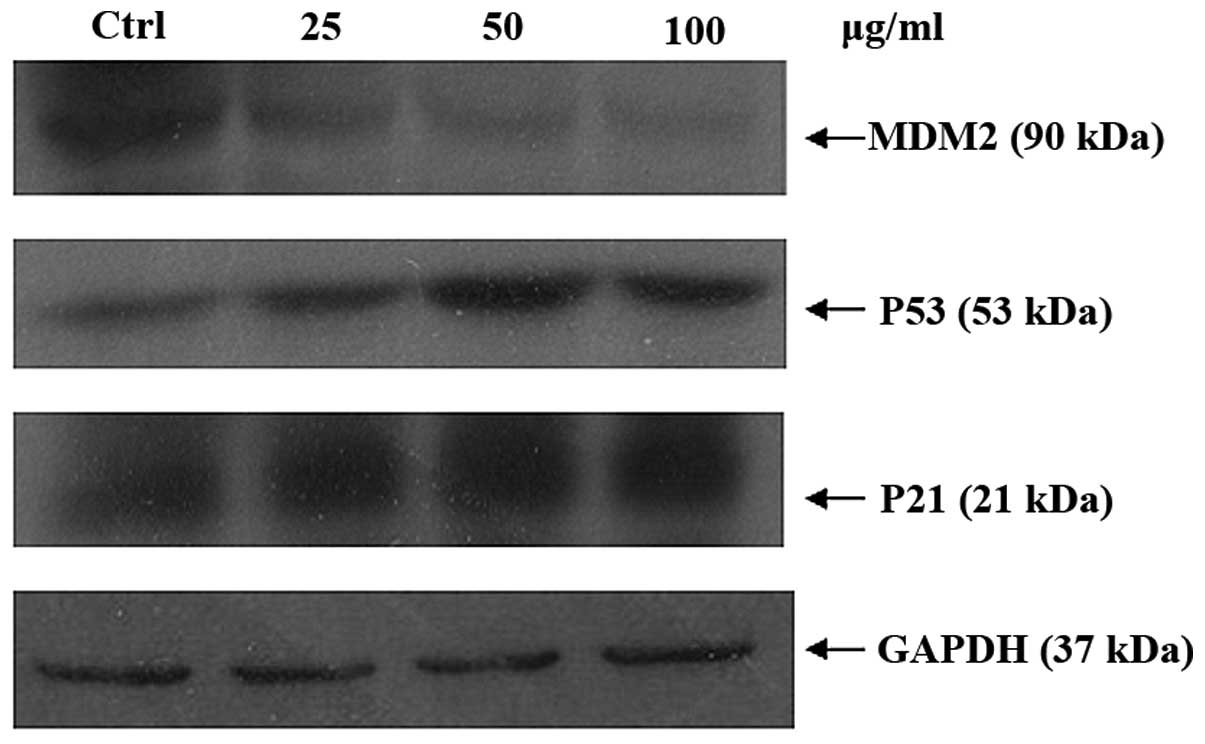
contents of A549 cells were measured by flow cytometry. p21 is an
inhibitor of CDKs which can be regulated by p53. MDM2, which
participates in an auto-regulatory loop with p53, has been shown to
function as a ubiquitin ligase that targets p53 for degradation.
The activation and stabilization of p53 is believed to play a role
in regulating function of MDM2. Any defects in the pathways are
believed to influence MDM2 activity in the tumors. MDM2, p53 and
p21 protein expression levels were examination by western blot
analysis. EAL can upregulate p53 and p21 but downregulated MDM2
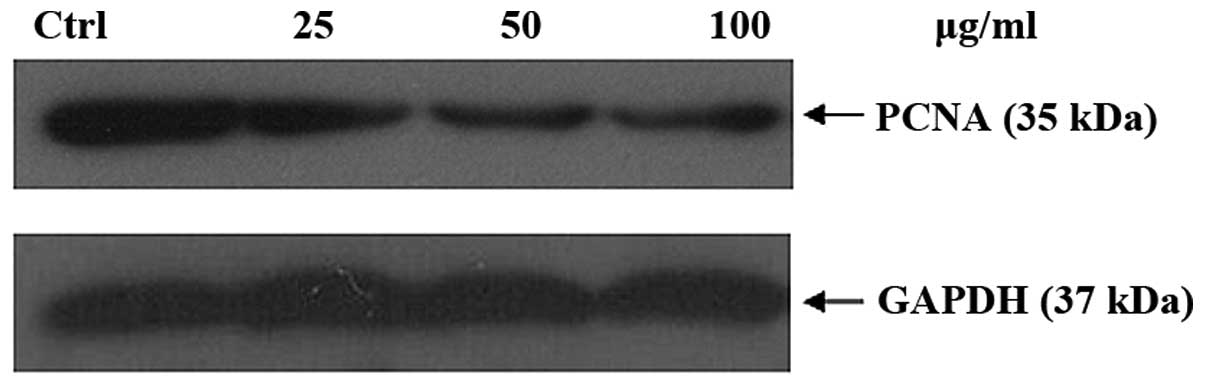
(Fig. 10). Proliferation of cell
nuclear antigen (PCNA) serves as a processivity factor for DNA
polymerase in eukaryotic cells. It played an important role in the
cell cycle process, and was a causative factor in PCNA alteration.
It reduced PCNA the expression. Fig.
11 shows that it can downregulate the PCNA expression
level.
DNA histograms showed that an active fraction
containing liquiritin, isoliquiritin and isoliquiritigenin
increased the population of G2/M cells in a concentration-dependent
manner at 24 h (Fig. 12).
Discussion
The pharmacological activity of active fractions
prepared from different methods can vary. Ethyl acetate extraction
appeared to be more potent and showed relatively higher antitumor
activity in non-small cell lung cancer. The composition analysis of
the ethyl acetate extract showed it contained three polyphenols,
namely, liquiritin, isoliquiritin and isoliquiritigenin. The
bioactivities of individual compounds present in Glycyrrhiza
uralensis also showed anticancer effects on the lung cancer
cell line.
MTT results showed that isoliquiritigenin has the
most potent anticancer activity. The present findings suggest that
isoliquiritigenin was responsible for the cytotoxicity in the A549
cell line. LDH assay and Annexin V staining confirmed that
combination of liquiritin, isoliquiritin and isoliquiritigenin in
the active fraction remarkably induced apoptosis.
The expression of related Bcl-2 family
anti-apoptotic protein Bcl-2, pro-apoptotic members Bax and Bid,
and the cascade activity provide supportive evidence that the Bcl-2
and caspase cascades are involved in apoptosis of the A549 lung
cancer cell line. Our results revealed that the active extract
containing liquiritin, isoliquiritin and isoliquiritigenin
upregulated Bax, Bid protein and downregulated Bcl-2. It increased
the enzymatic activity of initiator caspase-8 and capsase-9.
Subsequently it activated the effector caspase-3 and caspase-7,
leading to the activation of intrinsic caspase cascades. Caspase-3
is known to be one of the key executioners of apoptosis because
caspase-3 activation causes the cleavage or degradation of
downstream PARP (10). The results
suggest the apoptosis of A549 cells is mediated through
caspase-dependent activity.
The present findings showed that the expression
level of Akt pathway is inhibited. A previous study reported that
PI3K/Akt signaling cascade is related to cell survival and is
mediated via the Bcl-2 family members or caspase family proteins
(11). The limitation of full Akt
activity is believed to relate to apoptosis of A549 lung cancer
cells. Inhibition of Akt can downregulate phosphorylation of GSK-3β
at Ser9 (inactive form of GSK-3β) to trigger apoptosis (12). Akt is known to affect the activity
of Erk pathway by silencing c-Raf via phosphorylation at the
inhibitory site of Ser259 (13),
implicating crosstalk among different signaling pathways. The
present findings revealed that deactivation of Akt by
Glycyrrhiza uralensis extract may increase the activity of
caspase-9 resulting in apoptosis; thus, blocking Akt can increase
activities of GSK-3β, c-Raf and caspase-9 in A549 cells.
Phosphorylated PTEN at Ser380 (suppressive residue) was blocked,
suggesting the activation of PTEN through mediation of the Akt
pathway. These polyphenols in Glycyrrhiza uralensis caused
inhibition of the expression level of phosphorylated PDK1 and Akt
resulting in the cell cycle arrest. Altered expression of
regulatory cell cycle protein p53 and p21 was shown to promote G2/M
phase arrest in A549 cells. Upregulation of p53 and p21 and
downregulation of PCNA after treatment suggest it could regulate
cell cycle arrest in A549 cancer cells. The findings suggest
crosstalk between p53, Bcl-2 family, caspase cascades and the Akt
pathway. This could result in apoptosis of cancer cells (14,15).
The results suggest that the combination of liquiritin,
isoliquiritin and isoliquiritigenin was able to mediate multiple
signal pathways resulting in apoptosis.
Acknowledgements
We are grateful to Luck Tissue Mfy Ltd., for their
generosity with a grant no. 6903292.
References
|
1
|
Broker LE and Giaccone G: The role of new
agents in the treatment of non-small cell lung cancer. Eur J
Cancer. 38:2347–2361. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Jeong SJ, Koh W, Kim B and Kim SH: Are
there new therapeutic options for treating lung cancer based on
herbal medicines and their metabolites? J Ethnopharmacol.
138:652–661. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Zhang QY and Ye M: Chemical analysis of
the Chinese Herbal medicine Gan-Cao (licorice). J Chromatogr A.
1216:1954–1969. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Yo YT, Shieh GS, Hsu KF, Wu CL and Shiau
AL: Licorice and Licochalcone-A induce autophagy in LNCaP prostate
cancer cells by suppression of Bcl-2 expression and the mTOR
pathway. J Agric Food Chem. 57:8266–8273. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Dong SJ, Inoue A, Zhu Y, Tanji M and
Kiyama R: Activation of rapid signally pathways and the subsequent
transcriptional regulation for the proliferation of breast cancer
MCF-7 cells by the treatment with an extract of Glycyrrhiza
glabra root. Food Chem Toxicol. 45:2470–2478. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Fu Y, Hsieh ZC, Guo JQ, Kunicki J, Lee
YWT, Darzynkiewicz Z and Wu JM: Licochalcone-A, a novel flavonoid
isolated from licorice root (Glycyrrhiza glabra), causes G2
and late-G1 arrests in androgen-independent PC-3 prostat cancer
cells. Biochem Biophys Res Commun. 322:263–270. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Hsiang CY, Lai IL, Chao DC and Ho TY:
Differential regulation of activator protein 1 activity by
glycyrrhizin. Life Sci. 70:1643–1656. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Satomi Y, Nishino H and Shibata S:
Glycyrrhetinic acid and related compounds induce G1 arrest and
apoptosis in human hepatocellular carcinoma HepG2. Anticancer Res.
25:4043–4047. 2005.PubMed/NCBI
|
|
9
|
Yoon G, Bok YK and Seung HC: Topoisomerase
I inhibition and cytotoxicity of licochalcones A and E from
Glycyrrhiza inflate. Arch Pharm Res. 30:313–316. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Chang HY and Yang X: Proteases for cell
suicide: functions and regulation of caspases. Microbiol Mol Biol
Rev. 64:821–846. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Hsarkar F and Li YW: Targeting multiple
signal pathways by chemopreventive agents for cancer prevention and
therapy. Acta Pharmacol Sin. 28:1305–1315. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Beurel E and Jope RS: The paradoxical pro-
and anti-apoptotic actions of GSK3 in the intrinsic and extrinsic
apoptosis signaling pathways. Prog Neurobiol. 79:173–189. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Rommel C, Clarke BA, Zimmermann S, Nuñez
L, Rossman R, Reid K, Moelling K, Yancopoulos GD and Glass DJ:
Differentiation stage-specific inhibition of the Raf-MEK-ERK
pathway by Akt. Science. 286:1738–1741. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Gottlieb TM, Martinez-Leal JF, Seger R,
Taya Y and Oren M: Cross-talk between Akt, p53 and MDM2: possible
implications for the regulation of apoptosis. Oncogene.
21:1299–1303. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Mayo LD and Donner DB: The PTEN, MDM2, p53
tumor suppressor-oncoprotein network. Trends Biochem Sci.
27:462–467. 2002. View Article : Google Scholar : PubMed/NCBI
|