Introduction
Conventional anticancer drugs often lack
specificity, resulting in toxicities to healthy tissues and a poor
therapeutic index (1). Increasingly
more research has recently focused on antibody-based drugs
including monoclonal antibodies (mAbs), antibody-drug conjugates
(ADCs), immunotoxins and immunoliposomes (1). These antibody-based targeting drugs
not only inhibit tumor cell proliferation by interfering with
ligand binding or by blocking receptor dimerization (2), but they also maintain the ability to
collaborate with chemotherapeutic drugs (3). Evidence has shown that combinations
comprising antibody-based targeting therapies to several targets
[such as epithelial growth factor receptor (EGFR), HER2]
overexpressed in cancer cells, were more effective than the single
agent (4–6). Integrated combinations comprising
antibody-based drugs, chemotherapy and other agents are promising
for cancer therapy.
As therapeutic targets of cancer, EGFR and HER2, the
subgroup I and II of the ErbB family of receptors closely related
receptor tyrosine kinases, are known to play an essential role in
regulating cell proliferation and differentiation. The
overexpression of EGFR and HER2 observed in many human tumors and
their synergistic interaction in the transformation of cells make
these receptors important targets for the development of new
targeted therapeutics (7,8). Dual inhibition of EGFR and HER2 was
proposed for cancer based on their observed overexpression in
cancer cells (9–11). In fact, various anticancer drugs
that target EGFR have been designed and are used in the adjuvant
chemotherapeutic treatment of metastatic disease (6).
Moreover, it has been reported that matrix
metalloproteinase (MMP)-2 and -9 (also called gelatinases) are
highly expressed in several cancer areas compared to normal areas
(12–14). The expression rate of MMP-2 in the
colorectal tumor border is much higher than in the tumor center
(15), indicating that MMP-2 in
tumor borders has key roles in tumor invasion. The increased levels
of MMP-9 in cancer have also been attributed to cancer invasion and
metastasis (12). MMP-2/9
overexpression is associated with poorer overall and
progression-free survival in patients with colorectal cancer
(13,14). Some biomedical evidence has shown
that MMP-2/9 could be promising therapeutic targeting markers.
We have reported that Ec-LDP-Hr-AE, a ligand-based
(EGFC-loop fragment targeting EGFR) and antibody-based
(VH CDR3 region of anti-HER2 antibody C6.5) bispecific
fusion protein energized with enediyne, showed highly potent
cytotoxicity to a variety of cancer cells in vitro and was
highly effective against carcinoma xenograft in vivo
(16). In addition, a
gelatinase-targeting tandem dFv-based fusion protein dFv-LDP was
generated in this laboratory (17)
(Fig. 1A). Our previous study
showed that these two types of antibody-based fusion proteins had
strong binding abilities to cancer cells and their
enediyne-energized analogues had potent antitumor effects against
cancer xenografts that overexpress EGFR/HER2 or MMP-2/9,
respectively.
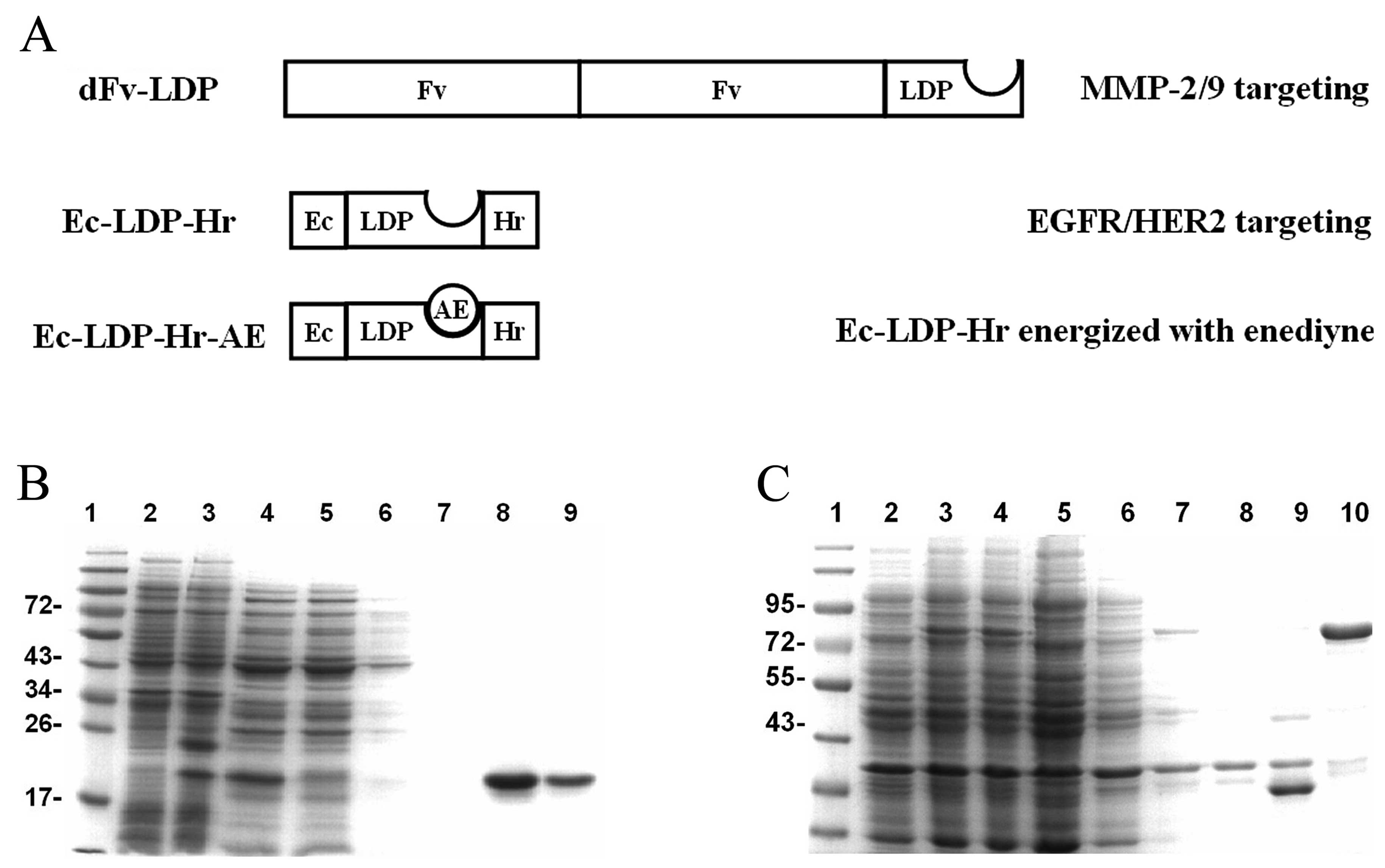 | Figure 1Preparation of fusion proteins dFv-LDP
and Ec-LDP-Hr. (A) Schematic depictions of the tandem scFv-based
fusion protein (dFv-LDP) and bispecific fusion protein Ec-LDP-Hr
and its energized fusion protein (Ec-LDP-Hr-AE) in the present
study. (B) SDS-PAGE analysis of the expression of Ec-LDP-Hr protein
and purification of the protein by Ni2+ affinity
chromatography. 1, Low molecular weight protein markers (kDa); 2,
total cell protein fraction before IPTG induction; 3, total cell
protein fraction after IPTG induction; 4, protein extracted from
E. coli periplasmic space; 5, proteins unbound with
Ni2+ column; 6 and 7, proteins washed with washing
buffer; 8 and 9, Ec-LDP-Hr protein eluted with elution buffer. (C)
SDS-PAGE analysis of the expression of dFv-LDP protein and
purification of the protein by Ni2+ affinity
chromatography. 1, Low molecular weight protein marker (kDa); 2,
total cell protein fraction before IPTG induction; 3 and 4, total
cell protein fraction after IPTG induction; 5, the soluble portion
of recombinant Rosetta(DE3)pLysS pellet after sonication; 6, the
soluble portion of recombinant Rosetta(DE3)pLysS pellet after
sonication washed by 2 M urea; 7, the soluble portion of
recombinant Rosetta(DE3)pLysS pellet after sonication washed by 6 M
urea; 8, proteins unbound with Ni2+ column; 9, proteins
washed with washing buffer; 10, dFv-LDP protein eluted with elution
buffer. |
In addition to targeting therapy, chemotherapy is
also needed to improve the outcome of advanced cancer patients.
Recently, several studies suggested that gemcitabine (GEM) had a
modest activity in cancer patients and may be an option in patients
(18,19). Also, the MMP inhibitor doxycycline
(DOX) could also be an adjuvant therapeutic agent in several types
of cancer (20–22). In the present study, we hypothesized
that an MMP-2/9-oriented drug combination strategy may achieve
enhancement of antitumor efficacy. The present study was performed
with several settings of combinations based on the
MMP-2/9-targeting fusion protein dFv-LDP plus the MMP inhibitor
drug DOX in vivo, and include: i) dFv-LDP plus the
EGFR/HER2-bispecific fusion protein Ec-LDP-Hr, ii) dFv-LDP plus the
EGFR/HER2-bispecific and enediyne-energized fusion protein
Ec-LDP-Hr-AE, iii) dFv-LDP plus Ec-LDP-Hr-AE and DOX, and iv)
dFv-LDP plus GEM and DOX. The results indicated that the
MMP-2/9-targeting fusion protein dFv-LDP acts as a versatile agent
to increase antitumor efficacy against colorectal cancer xenograft
when combined with EGFR/HER2-targeted fusion protein or the
chemotherapeutic GEM. Further enhance efficacy can be achieved by
the addition of the MMP inhibitor DOX.
Materials and methods
Cell culture and materials
The human colorectal cancer HCT-15 and fibrosarcoma
HT1080 cells were cultured in RPMI-1640 (HyClone, Logan, UT, USA).
The media was supplemented with 10% fetal bovine serum (FBS;
HyClone), 100 U/ml penicillin and 100 μg/ml streptomycin. Cells
were routinely grown as monolayer in culture flasks and maintained
in exponential growth to ~90% confluence for experiments. GEM was
purchased from Lilly France S.A. (Lille, France). DOX was purchased
from Shanghai Yuanye Biotechnology Co., Ltd. The primary antibodies
were purchased from Cell Signaling Technology, Inc. (Danvers, MA,
USA). The second antibodies were purchased from Zhongshan Golden
Bridge Biotechnology (Beijing, China). Cell lysis buffer was
purchased from GenStar Biosolutions Co. (Beijing, China).
Western blotting and
coimmunoprecipitation analysis
Cells were lysed for 30 min in RIPA lysis buffer
(GenStar Biosolutions Co). Extracts were clarified by
centrifugation at 10,000 rpm for 20 min at 4°C. Thirty micrograms
of each total protein were applied on a 10% SDS-PAGE and
electroblotted onto polyvinylidene difluoride membranes (Millipore,
Billerica, MA, USA). The membranes were blocked with 5% milk or 5%
bovine serum albumin (BSA) before incubating with primary
antibodies (diluted 1:1,000; Cell Signaling Technology) and then
incubated with goat anti-mouse/rabbit peroxidase-coupled antibody
(diluted 1:4,000). The membranes were washed with Tris-buffered
saline with 0.5% Tween-20 between every step for five times. The
specific bands were visualized with the Immobilon Western
Chemiluminescent HRP Substrate kit (Millipore).
For coimmunoprecipitation assay, cells were lysed in
cell lysis buffer for 30 min at 4°C. One milligram of total protein
from cell lysates was incubated with 100 μg Ec-LDP-Hr or dFv-LDP
protein at 4°C and the mixtures were then incubated overnight with
1 μg anti-EGFR, anti-HER2 or anti-MMP2 antibody at 4°C; complexes
were collected with protein A + G agarose (Beyotime, Beijing,
China) and the precipitates were washed five times with ice-cold
phosphate-buffered saline (PBS). Then, proteins were released by
boiling in sample buffer, followed by western blot analysis as
described above. The fusion protein was detected by incubation of
the membranes with anti-His-tag monoclonal antibody.
MTT assay
3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazolium
bromide (MTT) assay was used to determine cell proliferation. Cells
were seeded in 96-well plates at a density of 3,000–5,000
cells/well, incubated in 37°C for 24 h and then exposed to
different agents for 48 or 72 h. MTT (Sigma) solution (5 mg/ml, 20
μl) was added to each well and incubated for another 4 h at 37°C.
The supernatant was removed and 150 μl DMSO were added to each
well. The absorbance at 570 nm was measured by a microplate reader
(Thermo Fisher Scientific). Growth inhibition was calculated as a
percentage of the non-treated controls.
ELISA examination
Enzyme-linked immunosorbent assay (ELISA) was used
for measuring the binding efficiency of tested proteins to
colorectal cancer cells. Cells were seeded in 96-well plates at a
density of 20,000 cells/well and incubated at 37°C for 24 h and
were then fixed with methanol and blocked with 1% BSA for 1 h at
room temperature. After washing with PBS, cells were supplemented
with 100 μl of 2-fold dilutions of tested proteins ranging from 50
to 0.1 μmol/l and incubated at 37°C for 2 h. A 2,500-fold dilution
of His-Tag (2A8) mouse mAb (M20001L; Abmart, Omaha, NE, USA) and a
2,500-fold diluted horseradish peroxidase (HRP)-conjugated goat
anti-mouse IgG (Zhongshan Golden Bridge Biotechnology) were used as
the primary and the secondary antibodies, respectively. A total of
100 μl of TMB (PA107-01, Tiangen, Beijing, China) was added as
substrate solution, followed by terminating reaction with 2 mol/l
H2SO4. The absorbance at 450 nm was measured
using a microplate reader (Thermo Fisher Scientific, Bremen,
Germany), and the affinity constant was calculated. All assays were
carried out in triplicate.
ELISA was also used to detect the MMP-2 in cell
culture media. The concentration of MMP-2 secreted from cells was
determined by the ELISA kit according to the manufacturer’s
protocol (EK0459; Boster, Wuhan, China).
Cell immunofluorescence and confocal
assays
Cell immunofluorescence was used to measure the
binding efficiency of proteins to colorectal cancer cells. Seeded
at 20,000 cells/well in 24-well plates, the cells were cultured for
24 h. After washing with PBS, the cells were fixed with methanol
and blocked with 1% BSA for 1 h at room temperature and were then
incubated with 50 μmol/l proteins at 4°C overnight. Then, the cells
were incubated with His-Tag (2A8) mouse mAb (1:200; M20001L) at
37°C for 2 h. After washing with PBS, cells were incubated with
tetramethylrhodamine isothiocyanate (TRITC)-conjugated goat
anti-mouse IgG (1:200; Zhongshan Golden Bridge Biotechnology) at
room temperature for 1 h (avoiding light). The cells were then
incubated with 300 nmol/l 4,6-diamidino-2-phenylindole (DAPI) to
stain nuclei for 20 min at room temperature (avoiding light). After
washing with PBS, each sample was analyzed by a fluorescence
microscope and recorded by a camera.
The binding affinity of fusion protein Ec-LDP-Hr to
HER2 on the HCT-15 cell surface was also analyzed by laser scanning
confocal assay. Following the above-mentioned protocol to the step
of incubation with TRITC-conjugated goat anti-mouse IgG, cells were
then washed with PBS and incubated with rabbit anti-HER2 mAb
(#2165; CST, Inc.) at 4°C overnight (avoiding light). Then, cells
were washed with PBS and incubated with the Alexa Fluor
488-conjugated AffiniPure goat anti-rabbit IgG (ZF-0511; Zhongshan
Golden Bridge Biotechnology) at room temperature for 2 h (avoiding
light). Then, the cells were stained with DAPI at room temperature
for 20 min (avoiding light). After washing with PBS, the images
were observed under a laser scanning confocal microscope (Leica TCS
SP2, Germany).
Transwell migration assay
The migration potential of HCT-15 cells was examined
using Transwell inserts fitted with polycarbonate filter (8-μm
available pore size, #3422; Corning Inc., Corning, NY, USA). Cells
in FBS-free medium (5×105 cells/ml) were seeded in the
upper compartment while the lower wells contained 10 or 20% FBS
medium as a chemoattractant. Following 24 or 48 h incubation with
dFv-LDP and DOX, the cells in the upper chamber were removed with a
cotton swab while other cells, which had passed through the filter
on the underside of the membrane, were fixed with methanol, stained
with 0.01% crystal violet and then captured at ×40 magnification
using the camera on invert microscope. Subsequently, the crystal
violet was then dissolved with 33% acetic acid and the absorbance
was measured at 450 nm by using a microplate reader.
Animal experiments
Antitumor experiments were carried out using the
HCT-15 xenograft model of human colorectal carcinoma. Female
athymic mice (BALB/c, nu/nu) were purchased from the Institute for
Experimental Animals, Chinese Academy of Medical Science and Peking
Union Medical College. The experiments were performed according to
the regulation of Good Laboratory Practice for non-clinical
laboratory studies of drugs issued by the National Scientific and
the Technologic Committee of People’s Republic of China.
Animal experiments were performed with athymic mice
weighing 18–20 g. Mice were inoculated subcutaneously in the right
flank with 1×107 HCT-15 cells suspended in 200 μl PBS.
Tumors grown after 3–4 weeks in donor animals were aseptically
dissected and mechanically minced. Pieces of tumor tissue (2 mm in
diameter) were transplanted subcutaneously in the right flank of
mice by a trocar needle. When the tumors were established and
reached ~50–100 mm3 in size, the mice were randomly
divided into groups (n=6). The experimental mice were injected
twice through the tail vein at an interval of 7 days. At the same
time, one group of mice was given physiological saline as control.
Every 2 days, tumor size was measured with a caliper, and tumor
volume was calculated using the following formula: Tumor volume V =
(axb2)/2, where a and b are the long and the
perpendicular short diameters of the tumor, respectively. At the
end of the experiment, all mice were weighted and sacrificed, and
their tumors were excised. Tumors were weighed, and the tumor
growth inhibition was calculated as follows: Tumor growth
inhibition (%) = (1−T/C) × 100%. T is the mean tumor weight of
treated group, whereas C represents the mean tumor weight of the
control group.
Statistical analysis
Data are presented as the means ± SD. Statistical
comparisons between groups were executed by Student’s t-test, and a
significant difference was considered if P<0.05. The correlation
of binding capability among different tested proteins was analyzed
using Pearson’s χ2 test. P-value <0.05 was considered
to indicate a statistically significant difference. Analysis was
performed using the SPSS statistical software package for windows
(version 17.0, SPSS, Inc., Chicago, IL, USA).
Results
Proliferation inhibition of cancer cells
by Hc-LDP-Hr and dFv-LDP
The schematic depictions (Fig. 1A) represent the molecular
constitution of the proteins and enediyne-energized proteins used
in the present study. dFv-LDP is a fusion protein that comprises
LDP and tandem dual Fv segments from the antibody directed against
gelatinase. Ec-LDP-Hr is an EGFR/HER2-bispecific fusion protein in
which Ec is an EGFR-targeting oligopeptide and Hr is a CDR3
sequence from an anti-HER2 antibody. Ec-LDP-Hr-AE, the
enediyne-energized fusion protein were prepared by adding AE to
Ec-LDP-Hr. We prepared the fusion proteins Ec-LDP-Hr and dFv-LDP as
reported (17,18) and analyzed by SDS-PAGE (Fig. 1B and C). The enediyne-energized
fusion protein Ec-LDP-Hr-AE was prepared by integrating the
enediyne AE into the fusion protein Ec-LDP-Hr as previously
described (17).
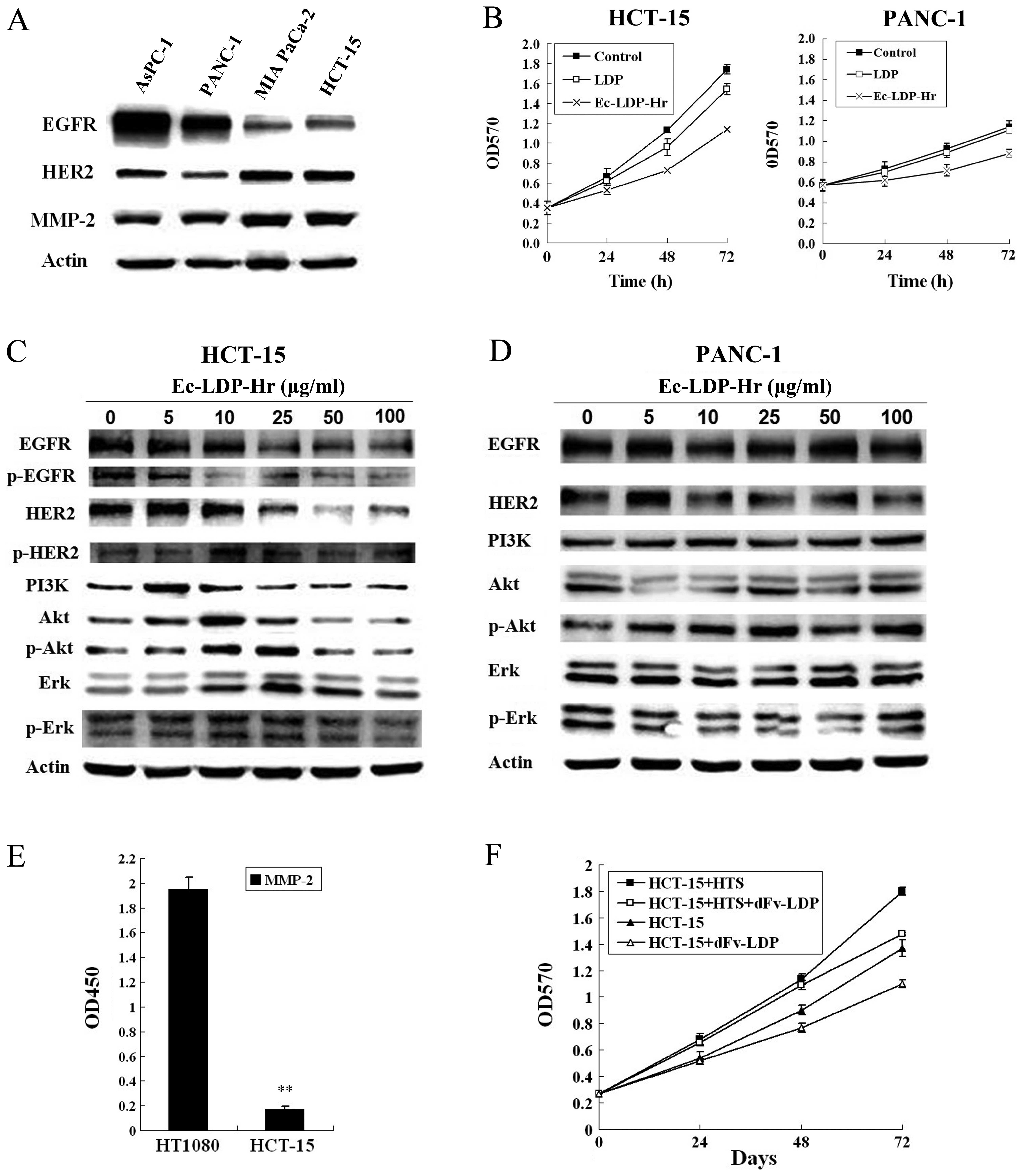
We first detected the expression levels of EGFR,
HER2 and MMP-2 in three pancreatic cancer cell lines (AsPC-1,
PANC-1, MIA PaCa-2) and a colorectal cancer cell (HCT-15) by
western blotting (Fig. 2A). The
results showed that AsPC-1 and PANC-1 cells had higher expression
of EGFR while MIA PaCa-2 and HCT-15 cells had higher expression of
HER2, respectively. However, the expression levels of MMP-2 in MIA
PaCa-2 and HCT-15 were higher than those in AsPC-1 and PANC-1
cells.
Subsequently, we tested the antiproliferation
effects of Ec-LDP-Hr by using MTT assay. The results showed that
Ec-LDP-Hr (50 μg/ml) was more potent than LDP (50 μg/ml) on
inhibitions of HCT-15 and PANC-1 cell proliferation (Fig. 3A). Due to the antibody-based
bispecific fusion protein Ec-LDP-Hr targeting EGFR and HER2,
western blotting was used to analyze the change of EGFR/HER2
signaling pathway. As shown in Fig. 3C
and D, there was a dose-dependent decrease of the expression
and the phosphorylation level of proteins in the EGFR/HER2
signaling pathway in colorectal cancer HCT-15 cells while this was
not observed in PANC-1 cells. These results suggested that the
efficacy of proliferation inhibition by Ec-LDP-Hr was through
EGFR/HER2 signal pathway by HER2 targeting peptide Hr from CDR3
region.
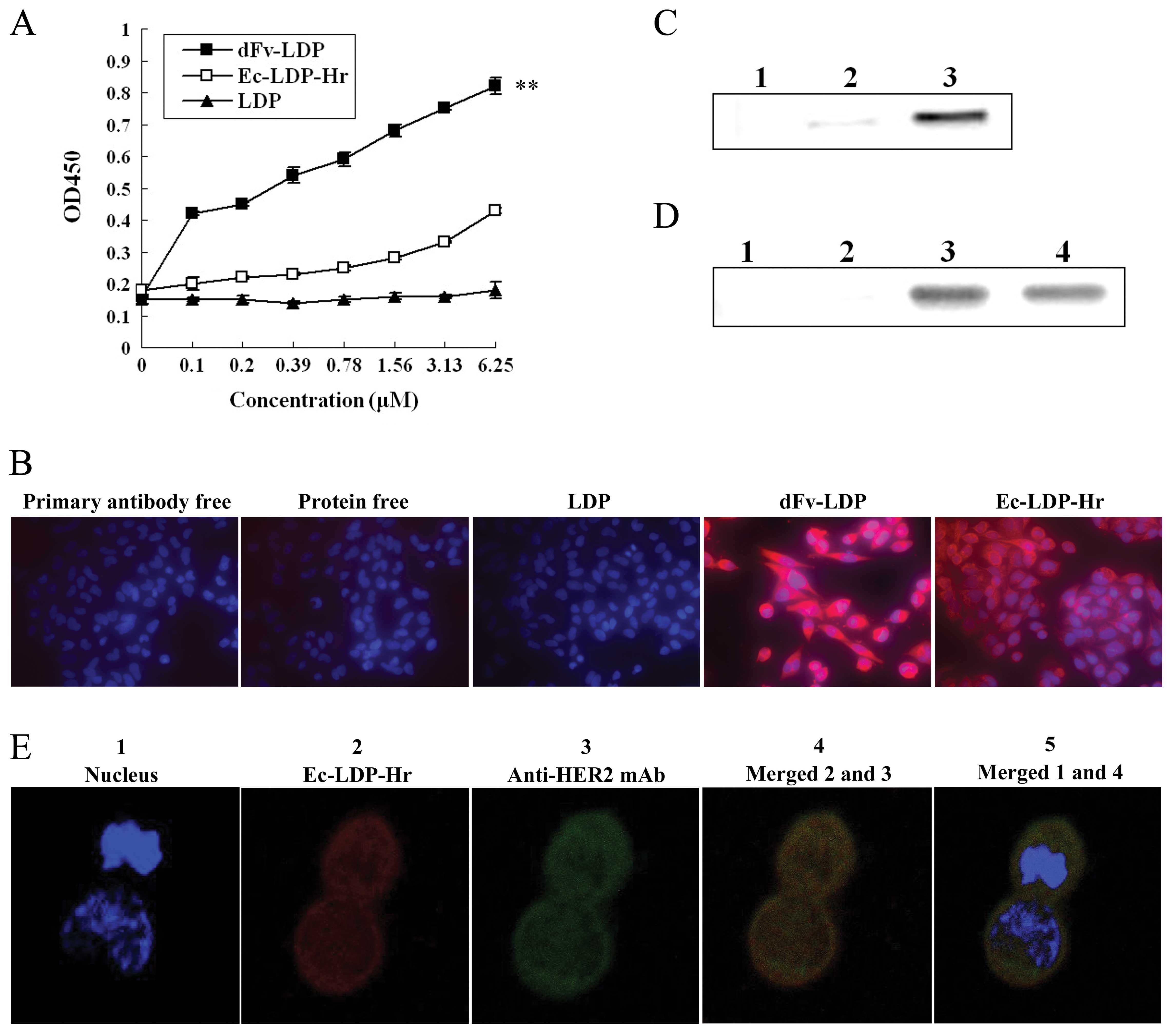 | Figure 3Fusion proteins dFv-LDP and Ec-LDP-Hr
have strong binding ability to human colorectal cancer HCT-15
cells. (A) The binding affinity of dFv-LDP and Ec-LDP-Hr proteins
with HCT-15 cells analyzed by ELISA. **P<0.01
compared with Ec-LDP-Hr and LDP. (B) Binding affinity of dFv-LDP
and LDP with the HCT-15 cells analyzed by immunofluorescent
cytochemical staining at ×400. The red color represents dFv-LDP and
Ec-LDP-Hr by mouse anti-His-Tag antibody and TRITC labeled
anti-mouse IgG. Blue color indicates the nuclei stained with DAPI.
(C and D) The binding specificity of dF-LDP and Ec-LDP-Hr proteins
to MMP-2, EGFR and HER2 were verified by coimmunoprecipitation
assay. Total proteins extracted from HCT-15 cells were incubated
with PBS (lanes C1 and D1) or Ec-LDP-Hr protein (lanes C2–3 and
D2–4). Subsequently, the mixture was incubated with either
preimmune serum (lane C2 and D2), anti-MMP-2 (lane C3), anti-EGFR
(lane D3), anti-HER2 (lane D4). The complexes formed were collected
with protein A + G agarose and were analyzed by SDS-PAGE and
immunoblotting with anti-His-tag monoclonal antibody. (E) Binding
affinity of Ec-LDP-Hr with HER2 on HCT-15 cells analyzed by
confocal microscopy. 1, Blue color indicates the nuclei stained
with DAPI; 2, the red color represents Ec-LDP-Hr by mouse
anti-His-Tag antibody and TRITC labeled anti-mouse IgG; 3, the
green color represents HER2 by mouse anti-HER2 antibody and FITC
labeled anti-mouse IgG; 4, merged 2 and 3, yellow area represents
the co-localization of HER2 and Ec-LDP-Hr; 5, merged 1 and 4. |
Moreover, we found that the MMP-2 secreted from
fibrosarcoma HT1080 cells in their culture supernatant was 6-fold
more than HCT-15 cells analyzed by ELISA (Fig. 2E). After cultivating for 24 h, the
culture of HCT-15 in 96-well plates was replaced with HT1080
culture supernatant adding 10% FBS and exposed to dFv-LDP (100
μg/ml). The result showed that HT1080 culture supernatant promoted
HCT-15 cell proliferation. However, this effect was attenuated by
adding MMP-2/9 targeting protein dFv-LDP (Fig. 2F). The results suggested that
dFv-LDP inhibited the growth of HCT-15 cells by blocking the action
of surrounding MMP-2 on the cancer cells.
Binding capability of dFv-LDP and
Ec-LDP-Hr to human colorectal cancer HCT-15 cells
The binding capability of dFv-LDP and Ec-LDP-Hr to
colorectal cancer cells HCT-15 was examined by ELISA (Fig. 2A) and cell immunofluorescence
(Fig. 2B). The results showed that
dFv-LDP and Ec-LDP-Hr were able to bind to the HCT-15 cells more
markedly compared with LDP. Also, dFv-LDP had stronger binding
ability than Ec-LDP-Hr. To further confirm the ability of dFv-LDP
and Ec-LDP-Hr binding to MMP-2 and EGFR/HER2 respectively,
coimmunoprecipitation assay was carried out. As shown in Fig. 3C and D, the targeting fusion
proteins dFv-LDP and Ec-LDP-Hr were precipitated as part of the
complex with MMP-2 (lane C3), EGFR (lane D3) and HER2 (lane D4). By
laser scanning confocal assay, as shown in the Fig. 2E, Ec-LDP-Hr (red fluorescence) was
found to co-localize with HER2 (green fluorescence) and their merge
(yellow fluorescence) was localized on the cell membrane.
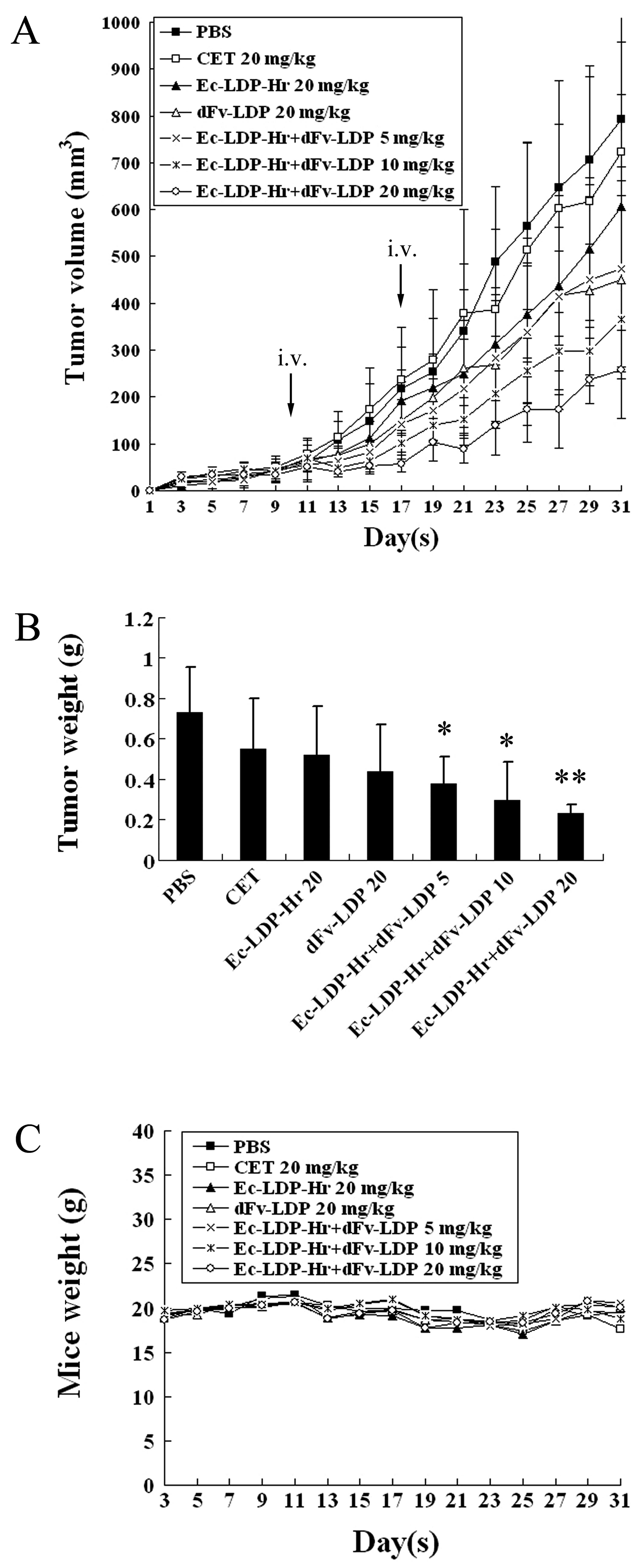
Combinations of dFv-LDP with Ec-LDP-Hr on
HCT-15 xenograft in vivo
The first experiment in vivo was set to
determine the synergistic effect of the fusion proteins dFv-LDP and
Ec-LDP-Hr on colorectal cancer HCT-15 xenograft in nude mice. The
fusion proteins dFv-LDP (20 mg/kg) or Ec-LDP-Hr (20 mg/kg) alone,
and the combination of various dosages including 5, 10, 20 mg/kg
respectively, were tested. Cetuximab at 20 mg/kg was used for
comparison. As evaluated by tumor volume, the inhibitory rates of
Ec-LDP-Hr or dFv-LDP alone were 23.6 and 43.2%, respectively. The
inhibitory rates of the combination of dFv-LDP and Ec-LDP-Hr at
dosages of 5, 10, 20 mg/kg were 40.4, 53.8 and 67.5%, respectively
(Fig. 4A). The groups treated with
a combination of two fusion proteins showed significant statistical
difference from control (P<0.01). The combination treatment with
fusion protein Ec-LDP-Hr (20 mg/kg) and dFv-LDP (20 mg/kg) revealed
a synergistic effect (CDI <0.8). As is known, cetuximab is a
human-mouse chimeric mAb that binds to the extracellular domain of
the EGFR (HER1) molecule and is currently approved for clinical use
to treat colorectal cancer. In this experiment, cetuximab did not
show antitumor efficacy on HCT-15 xenograft in nude mice. At the
end of the experiment, the tumors that were excised from the mice
were weighed. Similar synergistic tumor inhibition by the
combination of dFv-LDP and Ec-LDP-Hr was observed (Fig. 4B). The body weight loss of treated
mice was <10% of the initial weight, considering that the
treatment dosage was tolerated (Fig.
4C).
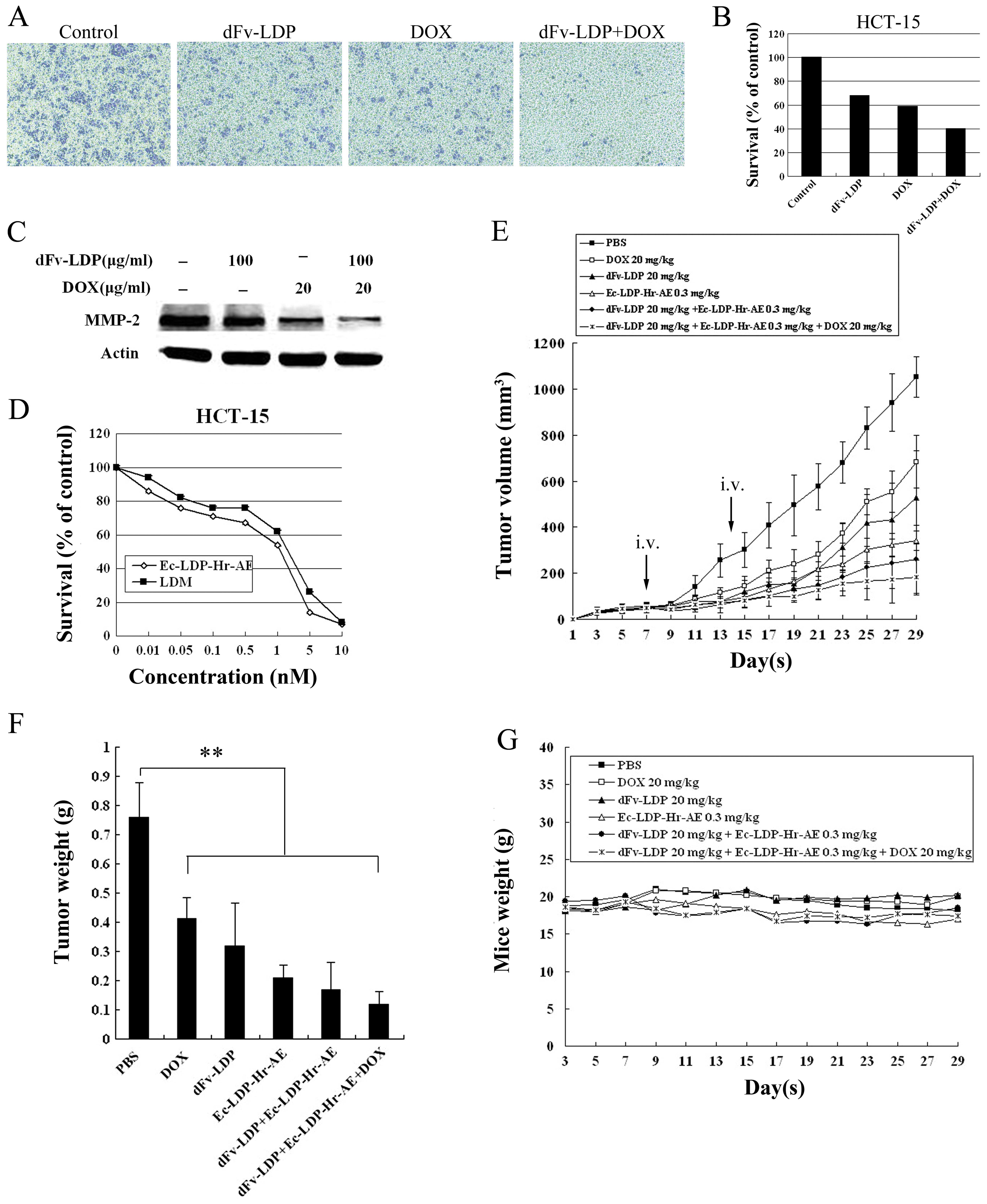
Combinations of dFv-LDP with
enediyne-energized fusion protein Ec-LDP-Hr-AE plus DOX on HCT-15
xenograft in vivo
The cytotoxicity of the antibody-based bispecific,
enediyne-energized fusion protein Ec-LDP-Hr-AE on HCT-15 cell line
was determined by MTT assay. As shown in Fig. 5D, Ec-LDP-Hr-AE displayed highly
potent cytotoxicity to cultured HCT-15 cells and was generally more
active than the lidamycin. For HCT-15 cells, the IC50
value was ~1 nM.
To further improve the antitumor efficacy on
colorectal cancer HCT-15 xenograft in nude mice, another experiment
was set to enhance the antitumor efficacy by a combination strategy
that comprised MMP-2/9-targeting fusion protein dFv-LDP and
EGFR/HER2-bispecific enediyne-energized fusion protein
Ec-LDP-Hr-AE. For even higher antitumor efficacy, DOX was added to
the combination. As shown in Fig.
5E, DOX (20 mg/kg, i.p. administered every other day), dFv-LDP
(20 mg/kg) or Ec-LDP-Hr-AE (0.3 mg/kg) alone displayed antitumor
effects with inhibitory rates of 35, 49.7 and 67.5%, respectively.
The inhibitory rate of combination of dFv-LDP and Ec-LDP-Hr-AE was
75.1%. Furthermore, this combination plus DOX showed stronger
efficacy with an inhibitory rate of 82.7%. Similar antitumor
efficacy was determined by measurement of tumor weights (Fig. 5F). The groups treated with agents in
this experiment showed significant statistical difference from
control (P<0.01). The body weight loss in mice treated with
Ec-LDP-Hr-AE was <10% of the initial body weight, considering
that the treatment dosage was tolerated (Fig. 5G).
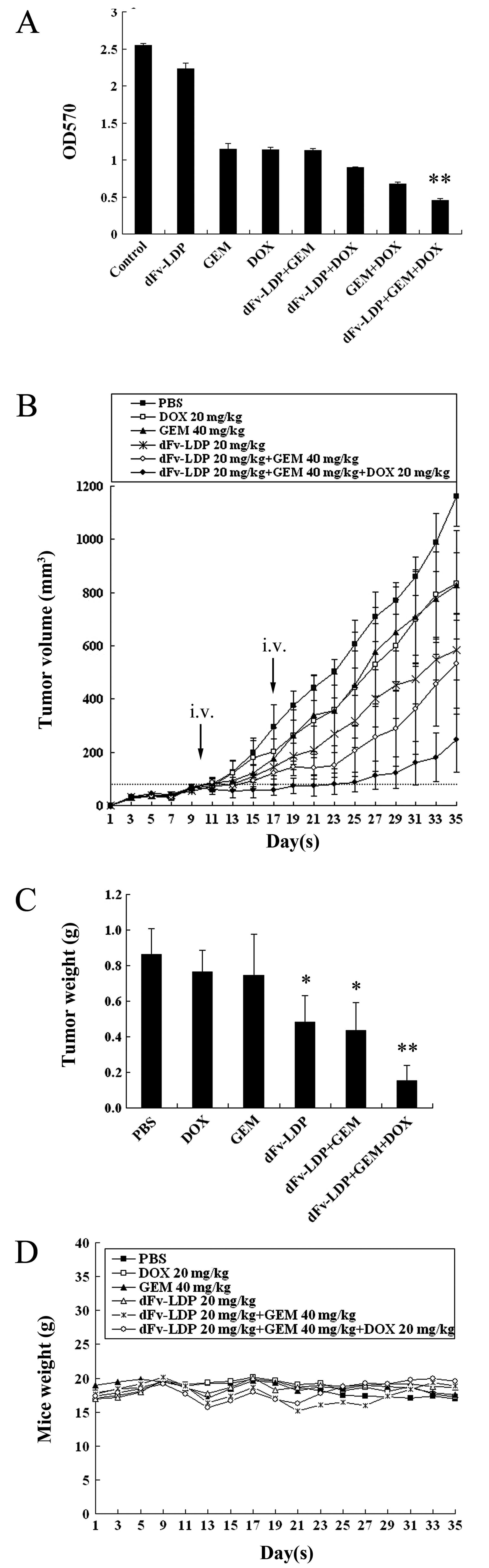
Combinations of dFv-LDP with GEM and DOX
display enhanced antitumor efficacy against HCT-15 xenograft in
vivo
GEM as a chemotherapeutic drug has been used for
several types of cancer. Here, the cytotoxicity of GEM, dFv-LDP,
DOX, and their combinations to HCT-15 cells were determined by MTT
assay (Fig. 6A). The results showed
that the combination of dFv-LDP (200 μg/ml) with GEM (0.05 μM) and
DOX (10 μg/ml) was more potent than any of the two drugs combined
or alone respectively, in cytotoxicity to HCT-15 cells.
The antitumor efficacy by a combination strategy
that comprises dFv-LDP, GEM and DOX was analyzed on HCT-15
xenograft in vivo. As shown in Fig. 6B, dFv-LDP (20 mg/kg), DOX (20 mg/kg)
or GEM (40 mg/kg) alone displayed antitumor effects with inhibitory
rates of 47., 27.2, or 25.3% at day 25, respectively. The
inhibitory rate of the combination of dFv-LDP and GEM was 66.3%.
Furthermore, this combination plus DOX displayed notable antitumor
efficacy, producing initial shrinkage after the first
administration and the tumor inhibitory rate was 85.5% at day 25,
whereas there was no tumor shrinkage observed in any other group.
Similar antitumor efficacy was determined by measurement of tumor
weights (Fig. 6C). The body weight
loss of treated mice was <10% of the initial weight, considering
that the treatment dosage was tolerated (Fig. 6D).
Discussion
At present, the vast majority of biomarker research
has focused on targeted therapies. Efforts are continuing to
identify predictive markers of response or resistance to
chemotherapy (23). One of the most
promising targets for antitumor therapy is the epithelial growth
factor receptor (EGFR) family in the treatment of metastatic cancer
(24,25). Several studies have shown that
agents targeting the EGFR improve outcomes in cancer (26,27).
Currently, two anti-EGFR monoclonal antibodies, panitumumab (an
IgG2 type human antibody directed against the same epitope) and
cetuximab (an IgG3 type chimeric antibody directed against the
extra-cellular portion of the receptor), have been clinically
validated for the treatment of metastatic colorectal cancer.
In addition, matrix metalloproteinases (MMPs) play
an important role in cancer progression and spread such as invading
surrounding tissues, basement membranes and extracellular matrix
(28). In colorectal cancer, MMP-2
immunoexpression associates with advanced disease (29,30),
and high MMP-2 expression in cancer cells and the stroma associates
with poor prognosis (31). MMP-9
correlates with metastatic disease and poor prognosis (32). For the abundance of MMP-2/9
increases in colorectal carcinoma, highly selective MMP inhibitors
should also be potential therapeutics (33). As is well known, MMP-2/9 is
generally overexpressed in highly invasive and metastatic cancer
cells, in cancer associated fibroblast and in proliferating
endothelial cells. Therefore, MMP-2/9 appears to be a highly
attractive, multi-faceted target in cancer therapy. Targeting
MMP-2/9 may actually impose actions on both cancer cells and
stromal cells, leading to cancer cell killing and modulation of the
tumor microenvironment. It is of particular interest to develop a
new strategy for drug combinations on the basis of targeting
MMP-2/9.
Globally, colorectal cancer is the third most
commonly diagnosed cancer in males and the second in females
(34). Approximately one-quarter of
CRC patients have metastases at diagnosis and a further 33–50%
develop metastases over their disease course (35). Chemotherapy has been the standard
care for metastatic colorectal cancer patients for many years and
is based mainly on the use of three agents: 5-fluorouracil,
irinotecan and oxaliplatin. The role of other chemotherapeutic
drugs such as gemcitabine (GEM) has not yet been established
(18). Evidence has shown that GEM
was more potent and had considerably broader antitumor activity
against various murine tumors (myeloma, adenocarcinoma, ovarian
carcinoma, lymphosarcoma and leukemia) and human tumor xenograft
models (breast, colon, lung and pancreatic) (36). In the present study, we set up the
colorectal cancer HCT-15 xenograft in athymic mice to evaluate the
efficacy of various combinations.
In previous studies, we reported two specific
targeted fusion proteins, dFv-LDP and Ec-LDP-Hr (16,17),
which had strong affinity and activity to cancer cells
overexpressing MMP-2/9 and EGFR/HER2, respectively. dFv-LDP is an
MMP-2/9-specific dFv-based fusion protein. Ec-LDP-Hr is an
EGFR/HER2-bispecific fusion protein and energized Ec-LDP-Hr-AE
belongs to its corresponding antibody-drug conjugates (ADCs) in
which the extremely potent cytotoxic enediyne AE serves as
‘warhead’ agent. Here, we demonstrated that the targeting proteins
dFv-LDP and Ec-LDP-Hr had strong binding abilities to colorectal
cancer cells. Both of them had antiproliferation effects of cancer
cells in vitro and therapeutic efficacy in vivo. The
present investigations evaluated several settings of combinations
including i) dFv-LDP plus the EGFR/HER2-bispecific fusion protein
Ec-LDP-Hr, ii) dFv-LDP plus the EGFR/HER2-bispecific and
enediyne-energized fusion protein Ec-LDP-Hr-AE, iii) dFv-LDP plus
Ec-LDP-Hr-AE and DOX, and iv) dFv-LDP plus GEM and DOX. The results
indicated that dFv-LDP enhanced antitumor efficacy against
colorectal cancer xenograft when combined with EGFR/HER2-targeted
fusion protein or the chemotherapeutic GEM. Further enhanced
efficacy can be achieved by the addition of DOX.
In summary, the MMP-2/9-targeting fusion protein
dFv-LDP plus DOX enhances antitumor efficacy in combination
respectively, with EGFR/HER2-bispecific fusion protein, its
corresponding enediyne-energized fusion protein, and the
chemotherapeutic GEM. This MMP-2/9-oriented combination strategy
that uses MMP-2/9-targeting antibody-based fusion protein and the
small molecular inhibitor DOX as the basic composed agents may be
effective in cancer therapy.
Acknowledgements
The authors acknowledge the ‘Significant New Drugs
Development’ Major Science and Technology Projects of China (nos.
2013ZX09102064 and 2012ZX09301002-001-015).
References
|
1
|
Sapra P and Shor B: Monoclonal
antibody-based therapies in cancer: advances and challenges.
Pharmacol Ther. 138:452–469. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Pedersen MW, Jacobsen HJ, Koefoed K, Hey
A, Pyke C, Haurum JS and Kragh M: Sym004: a novel synergistic
anti-epidermal growth factor receptor antibody mixture with
superior anticancer efficacy. Cancer Res. 70:588–597. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Sapra P, Damelin M, Dijoseph J, et al:
Long-term tumor regression induced by an antibody-drug conjugate
that targets 5T4, an oncofetal antigen expressed on
tumor-initiating cells. Mol Cancer Ther. 12:38–47. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Maron R1, Schechter B, Mancini M,
Mahlknecht G, Yarden Y and Sela M: Inhibition of pancreatic
carcinoma by homo- and heterocombinations of antibodies against
EGF-receptor and its kin HER2/ErbB-2. Proc Natl Acad Sci USA.
110:15389–15394. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
McCaffery I, Tudor Y, Deng H, et al:
Putative predictive biomarkers of survival in patients with
metastatic pancreatic adenocarcinoma treated with gemcitabine and
ganitumab, an IGF1R inhibitor. Clin Cancer Res. 19:4282–4289. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Shankaran V, Obel J and Benson AD III:
Predicting response to EGFR inhibitors in metastatic colorectal
cancer: current practice and future directions. Oncologist.
15:157–167. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Normanno N, De Luca A, Bianco C, et al:
Epidermal growth factor receptor (EGFR) signaling in cancer. Gene.
366:2–16. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Hynes NE and Lane HA: ERBB receptors and
cancer: the complexicity of targeted inhibitors. Nat Rev Cancer.
5:341–354. 2005. View
Article : Google Scholar : PubMed/NCBI
|
|
9
|
Harrison S and Benziger H: The molecular
biology of colorectal carcinoma and its implications: a review.
Surgeon. 9:200–210. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Saxby AJ, Nielsen A, Scarlett CJ, Clarkson
A, Morey A, Gill A and Smith RC: Assessment of HER-2 status in
pancreatic adenocarcinoma: correlation of immunohistochemistry,
quantitative real-time RT-PCR, and FISH with aneuploidy and
survival. Am J Surg Pathol. 29:1125–1134. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Yamanaka Y, Friess H, Kobrin MS, Buchler
M, Beger HG and Korc M: Coexpression of epidermal growth factor
receptor and ligands in human pancreatic cancer is associated with
enhanced tumor aggressiveness. Anticancer Res. 13:565–569.
1993.PubMed/NCBI
|
|
12
|
Björklund M and Koivunen E:
Gelatinase-mediated migration and invasion of cancer cells. Biochim
Biophys Acta. 1755:37–69. 2005.PubMed/NCBI
|
|
13
|
Kallakury BV, Karikehalli S, Haholu A,
Sheehan CE, Azumi N and Ross JS: Increased expression of matrix
metalloproteinases 2 and 9 and tissue inhibitors of
metalloproteinases 1 and 2 correlate with poor prognostic variables
in renal cell carcinoma. Clin Cancer Res. 7:3113–3119. 2001.
|
|
14
|
Cho YB, Lee WY, Song SY, Shin HJ, Yun SH
and Chun HK: Matrix metalloproteinase-9 activity is associated with
poor prognosis in T3–T4 node-negative colorectal cancer. Hum
Pathol. 38:1603–1610. 2007.PubMed/NCBI
|
|
15
|
Hong SW, Kang YK, Lee B, et al: Matrix
metalloproteinase-2 and -7 expression in colorectal cancer. J
Korean Soc Coloproctol. 27:133–139. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Guo XF, Zhu XF, Shang Y, Zhang SH and Zhen
YS: A bispecific enediyne-energized fusion protein containing
ligand-based and antibody-based oligopeptides against epidermal
growth factor receptor and human epidermal growth factor receptor 2
shows potent antitumor activity. Clin Cancer Res. 16:2085–2094.
2010. View Article : Google Scholar
|
|
17
|
Zhong G, Zhang S, Li Y, Liu X, Gao R, Miao
Q and Zhen Y: A tandem scFv-based fusion protein and its
enediyne-energized analogue show intensified therapeutic efficacy
against lung carcinoma xenograft in athymic mice. Cancer Lett.
295:124–133. 2010. View Article : Google Scholar
|
|
18
|
Saif MW, Kaley K, Penney R, Hotchkiss S,
Syrigos KN and Strimpakos AS: The efficacy of gemcitabine as
salvage treatment in patients with refractory advanced colorectal
cancer (CRC): a single institution experience. Anticancer Res.
31:2971–2974. 2011.PubMed/NCBI
|
|
19
|
Pelosof L, Yerram SR, Ahujia N, Delmas A,
Danilova L, Herman JG and Azad NS: CHFR silencing or
microsatellite instability is associated with increased antitumor
activity of docetaxel or gemcitabine in colorectal cancer. Int J
Cancer. 134:596–605. 2014. View Article : Google Scholar
|
|
20
|
Shen LC, Chen YK, Lin LM and Shaw SY:
Anti-invasion and anti-tumor growth effect of doxycycline treatment
for human oral squamous-cell carcinoma - in vitro and in vivo
studies. Oral Oncol. 46:178–184. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Guimaraes DA, Rizzi E, Ceron CS, et al:
Doxycycline dose-dependently inhibits MMP-2-mediated vascular
changes in 2K1C hypertension. Basic Clin Pharmacol Toxicol.
108:318–325. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Son K, Fujioka S, Iida T, et al:
Doxycycline induces apoptosis in PANC-1 pancreatic cancer cells.
Anticancer Res. 29:3995–4004. 2009.PubMed/NCBI
|
|
23
|
Heinemann V, Douillard JY, Ducreux M and
Peeters M: Targeted therapy in metastatic colorectal cancer - an
example of personalized medicine in action. Cancer Treat Rev.
39:592–601. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Grünwald V and Hidalgo M: Developing
inhibitors of the epidermal growth factor receptor for cancer
treatment. J Natl Cancer Inst. 95:851–867. 2003.PubMed/NCBI
|
|
25
|
Jones S, Zhang X, Parsons DW, et al: Core
signaling pathways in human pancreatic cancers revealed by global
genomic analyses. Science. 321:1801–1806. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Winder T and Lenz HJ: Vascular endothelial
growth factor and epidermal growth factor signaling pathways as
therapeutic targets for colorectal cancer. Gastroenterology.
138:2163–2176. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Medinger M and Drevs J: Receptor tyrosine
kinases and anticancer therapy. Curr Pharm Des. 11:1139–1149. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Coussens LM and Werb Z: Matrix
metalloproteinases and the development of cancer. Chem Biol.
3:895–904. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Matsuyama Y, Takao S and Aikou T:
Comparison of matrix metalloproteinase expression between primary
tumors with or without liver metastasis in pancreatic and
colorectal carcinomas. J Surg Oncol. 80:105–110. 2002. View Article : Google Scholar
|
|
30
|
Papadopoulou S, Scorilas A, Arnogianaki N,
Papapanayiotou B, Tzimogiani A, Agnantis N and Talieri M:
Expression of gelatinase-A (MMP-2) in human colon cancer and normal
colon mucosa. Tumour Biol. 22:383–389. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Hilska M, Roberts PJ, Collan YU, et al:
Prognostic significance of matrix metalloproteinases-1, -2, -7 and
-13 and tissue inhibitors of metalloproteinases-1, -2, -3 and -4 in
colorectal cancer. Int J Cancer. 121:714–723. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Karakiulakis G, Papanikolaou C, Jankovic
SM, Aletras A, Papakonstantinou E, Vretou E and Mirtsou-Fidani V:
Increased type IV collagen-degrading activity in metastases
originating from primary tumors of the human colon. Invasion
Metastasis. 17:158–168. 1997.PubMed/NCBI
|
|
33
|
Roeb E, Dietrich CG, Winograd R, et al:
Activity and cellular origin of gelatinases in patients with colon
and rectal carcinoma differential activity of matrix
metalloproteinase-9. Cancer. 92:2680–2691. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Jemal A, Bray F, Center MM, Ferlay J, Ward
E and Forman D: Global cancer statistics. CA Cancer J Clin.
61:69–90. 2011. View Article : Google Scholar
|
|
35
|
Garden OJ, Rees M, Poston GJ, et al:
Guidelines for resection of colorectal cancer liver metastases.
Gut. 55(Suppl 3): iii1–iii8. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Gesto DS1, Cerqueira NM, Fernandes PA and
Ramos MJ: Gemcitabine: a critical nucleoside for cancer therapy.
Curr Med Chem. 19:1076–1087. 2012. View Article : Google Scholar : PubMed/NCBI
|