Introduction
Apoptosis is a common morphological form of
programmed cell death that plays a critical role during development
and homeostasis, and in many diseases including cancer, acquired
immunodeficiency syndrome and neurodegenerative disorders (1). This type of apoptosis called
programmed cell death type I can be triggered in tumor cells by
anticancer agents (2,3). Tumor necrosis factor (TNF)-related
apoptosis-inducing ligand (TRAIL) was first identified as a member
of the TNF superfamily and can selectively induce apoptosis in
tumorigenic or transformed cells, yet not in normal cells,
highlighting its potential therapeutic application in cancer
treatment (4).
Iron (Fe) is a useful target as it plays a critical
role in proliferation involved in numerous metabolic pathways such
as ATP generation, oxygen transport and DNA synthesis (5). A few pathways have been proposed in
regards to iron-related carcinogenesis. Studies indicate that
iron-induced oxidative stress may cause DNA, protein and organelle
damage, through production of hydroxyl radicals and hydrogen
peroxide via Haber-Weiss and Fenton-type reactions (6,7). Some
studies suggest that dietary Fe restriction using iron chelators
has been shown to decrease tumor proliferation in a number of in
vitro and in vivo studies (8–10).
Hershko reported that in preliminary studies, deferoxamine (DFO),
an iron chelators, in combination with multidrug chemotherapy was
effective in controlling several tumors (11).
DFO was developed more than 40 years ago and takes
an active part as an iron chelator in the improvement of the
quality of life and overall survival of patients presenting with
iron overload (12). DFO binds iron
tightly, and the iron-DFO complex is excreted in both urine and
stool. DFO has been shown to be effective as an antitumor drug
through several signaling pathways in tumor cells (9,10). DFO
is well known to be used to induce hypoxia-inducible factor-1α
(HIF-1α) as it mimics the hypoxic effects at 21% O2
(13). HIF-1α is a transcriptional
factor composed of α- and β-subunits and is a key regulator of
metabolism to hypoxia (14). HIF-1
is involved in critical aspects of cancer biology such as
angiogenesis, cell survival and invasion and intratumoral hypoxia,
and genetic alterations can lead to HIF-1α overexpression, which
has been related to increased patient mortality in several types of
cancer (15).
Autophagy, ‘the eating of self’, was first coined by
Deter and De Duve several decades ago and is mainly based on the
observed degradation response to starvation or stress whereby
mitochondria and cellular elements are digested by lysosomes
(16). Autophagy, a common
morphological feature in dying cells, appears to be one of the
major functions to keep cells alive under stressful conditions
(17). In cancer cells, autophagy
can promote apoptosis in some cases (18,19).
However, autophagy has a more crucial role in sustaining cell
viability with defects in apoptosis (19–21).
Hu et al suggested that hypoxia-induced autophagy promotes
tumor cell survival (22). In the
present study, we demonstrated that DFO inhibits TRAIL-induced
apoptosis via regulation of autophagy in colon cancer cells.
Materials and methods
Cell culture
The human colon cancer cell line HCT116 was obtained
from the American Type Culture Collection (ATCC; Manassas, VA,
USA). Cells were cultured in RPMI-1640 medium supplemented with 10%
fetal bovine serum (both from Invitrogen-Gibco, Carlsbad, CA, USA),
100 U/ml penicillin, and 0.1 mg/ml gentamycin in a humidified
incubator maintained at 37°C and 5% CO2. Cells were
treated for 24 h with DFO and then exposed for 6 h to 200 ng/ml
TRAIL with or without the autophagy inhibitor chloroquine (10 μM)
(both from Sigma-Aldrich, St. Louis, MO, USA).
Crystal violet assay
Cell morphology was assessed microscopically
(inverted microscope, Nikon), and cell viability was determined by
crystal violet staining, as previously described (23). Briefly, cells were stained for 10
min at room temperature with crystal violet solution (0.5% crystal
violet in 30% ethanol and 3% formaldehyde), washed five times with
water, and then dried. After that, the cells were lysed with 1%
sodium dodecyl sulphate (SDS), and the absorbance was measured at
550 nm. Cell viability was calculated from the relative dye
intensity of the samples compared to the controls.
Trypan blue exclusion assay
The number of viable cells was determined by trypan
blue dye exclusion (Sigma-Aldrich) using a hemocytometer. The
result was expressed as a percentage relative to the
vehicle-treated controls.
Western blot analysis
HCT116 cells were lysed in lysis buffer [25 mM
4-(2-hydroxyethyl)-1-piperazineethanesulfonic acid (HEPES), pH 7.4,
100 mM NaCl, 1 mM ethylene diamine tetraacetic acid (EDTA), 5 mM
MgCl2, 0.1 mM dithiothreitol (DTT) and a protease
inhibitor mixture], and whole cell proteins were
electrophoretically resolved on a 10–15% sodium dodecyl sulfate
polyacrylamide gel and transferred to a nitrocellulose membrane.
Immunoreactivity was detected through sequential incubation with
primary antibodies, horseradish peroxidase-conjugated secondary
antibodies, and enhanced chemiluminescence reagents (Westsave Gold
Detection kit; AbFrontier Inc.). The primary antibodies used for
immunoblotting were anti-human HIF-1α (BD Biosciences), anti-LC3B
(Cell Signaling Technology), anti-P62 (Millipore Corporation),
anti-phospho-AKT (Epitomics, Burlingame, CA, USA) and anti-β-actin
(Sigma-Aldrich). Images were examined using a Fusion FX7 imaging
system (Vilber Lourmat, ZI Sud Torcy, France). The densitometry of
the signal bands was analyzed using Bio-1D software (Vilber
Lourmat, Marne la Vallée, France).
Statistical analysis
The unpaired t-test or Welch’s correction was used
for comparison between the two groups. For multiple comparison, the
one-way ANOVA followed by the Tukey-Kramer test was used. All
statistical analysis was performed using GraphPad Prism software.
Results were considered significant for values p<0.05, p<0.01
or p<0.001.
Results
DFO inhibits TRAIL-induced cell death in
colon cancer cells
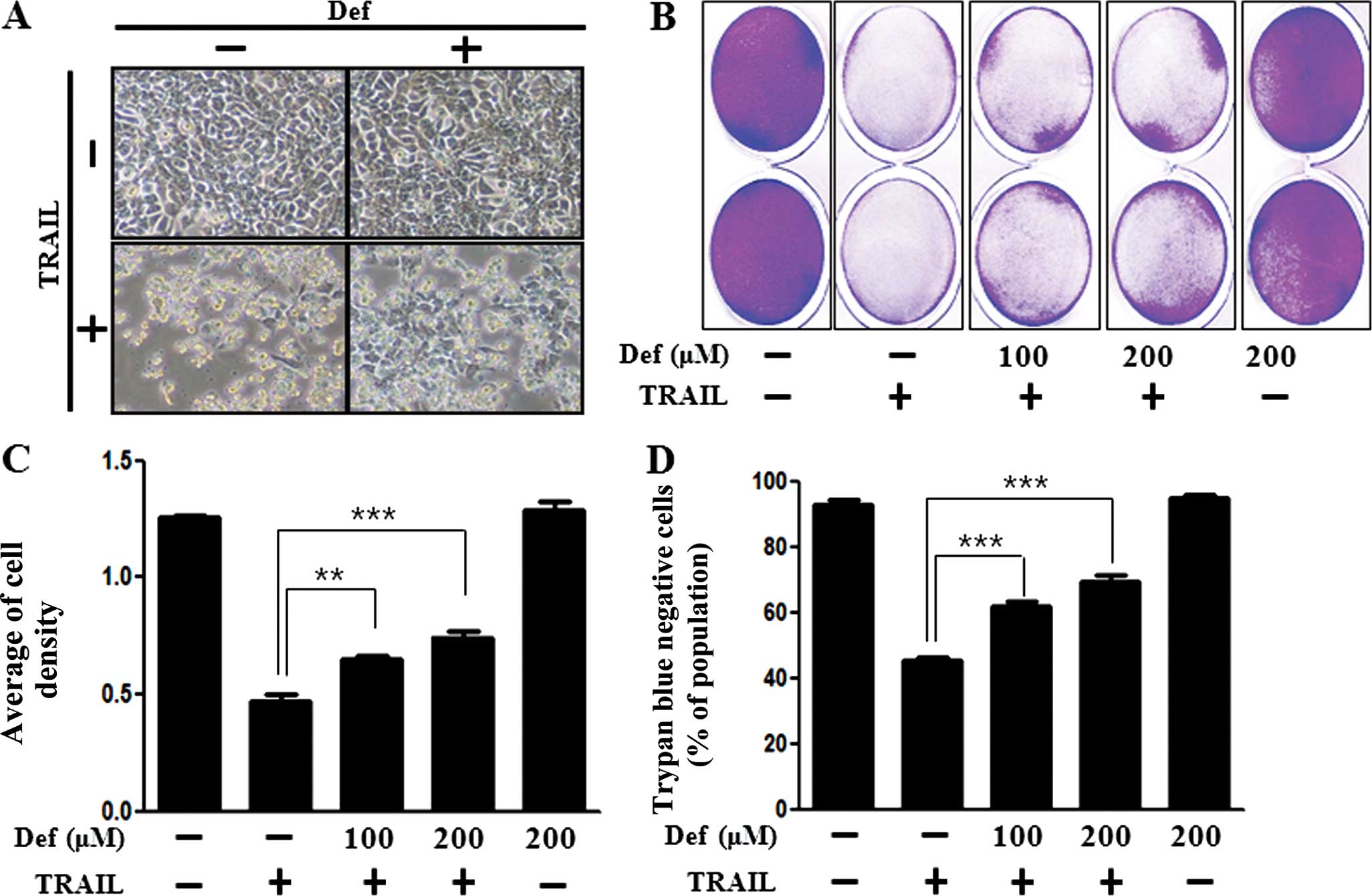
We examined whether DFO promotes or inhibits
TRAIL-induced cell death and whether this effect is associated with
induction of autophagy. We investigated the influence of DFO on
TRAIL-mediated cytotoxicity in HCT116 colon cancer cells by images
and crystal violet assay. HCT116 cells were exposed to DFO with or
without TRAIL. The cell viability of the TRAIL-treated cells was
decreased more than half according to images captured by light
microscopy and crystal violet assay. The cell viability of the
cells treated with DFO only was comparable to that of the untreated
controls. These results revealed that DFO treatment inhibited
TRAIL-induced cytotoxicity in HCT116 colon cancer cells (Fig. 1A–C). Trypan blue exclusion assay was
implemented for cell viability (Fig.
1D).
If cell death is induced by cytotoxicity in cells,
then cells cannot generate exocytosis. Thus, trypan blue particles
can flow in cells easily. For this reason, trypan blue dyed cells
represent dead cells. These comprehensive results exhibited that
DFO treatment inhibited cell death induced by TRAIL treatment
dose-dependently. These results indicate that DFO was effective in
preventing TRAIL-induced cell death in HCT116 colon cancer
cells.
DFO mediates HIF-1α stabilization and Akt
activation
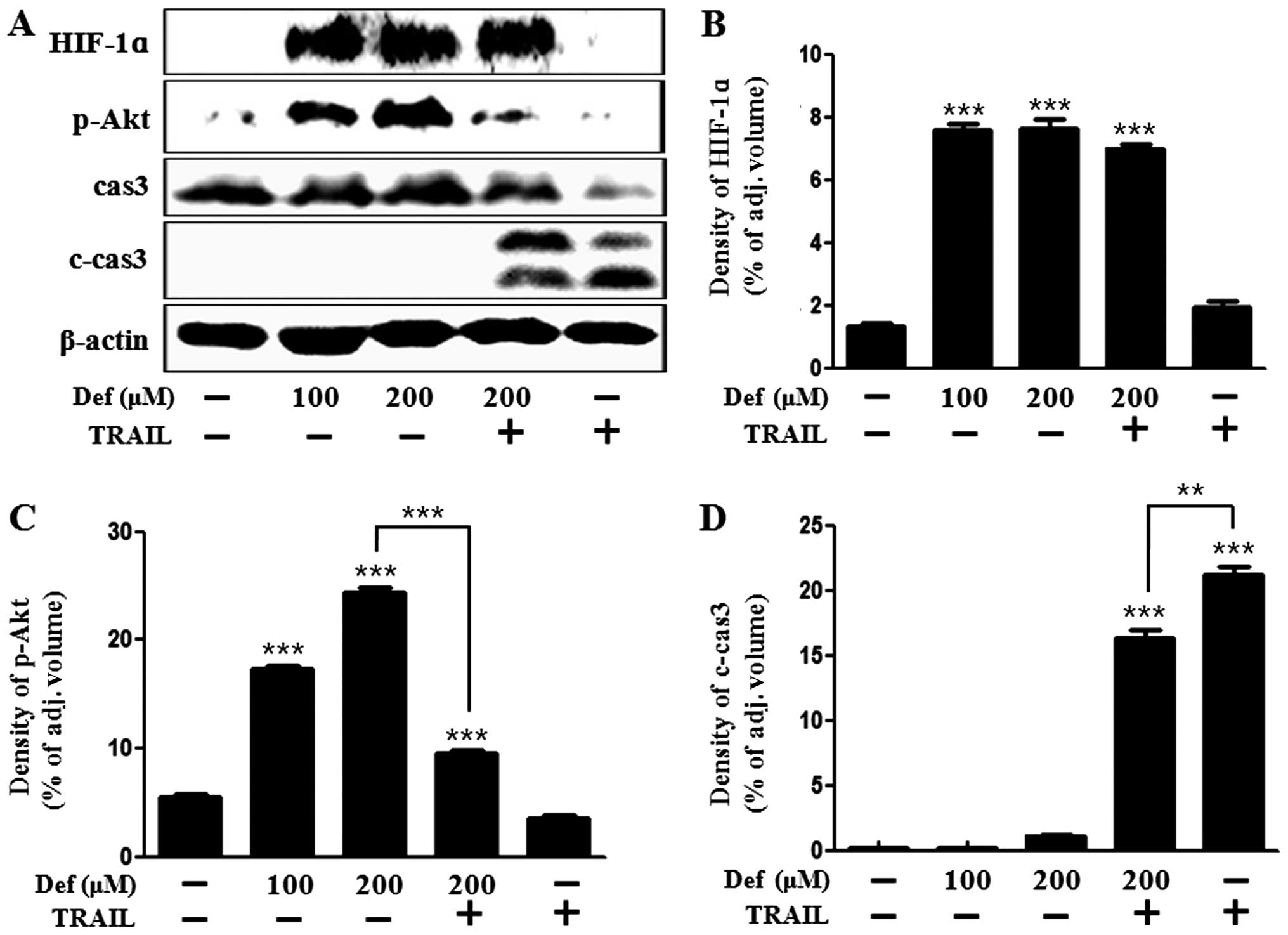
We identified the proliferative and protective
effects of DFO on cancer cells. We hypothesized that DFO would play
a role of an HIF-1α inducer not an iron chelator, since HIF-1α is
involved in critical aspects of cancer biology such as angiogenesis
and cell survival (15). We
investigated whether DFO induces HIF-1α stabilization by western
blot analysis. As shown in Fig. 2A and
B, the level of HIF-1α was increased in the DFO-treated group
in a dose-dependent manner compared with the control group as
detected by western blot analysis and densitometry. These results
suggest that DFO leads to stabilization of HIF-1α. We investigated
whether DFO affects survival and death signals. Akt affects cell
survival and metastasis in many types of cells including colon
cancer cells. Akt acts as a key signal that is associated with
oncogenic receptors to many essential prosurvival cellular
functions in human cancer (24).
Caspase-3 plays a key role in regulating programmed cell death or
apoptosis, a normal process required for maintenance of the
regulation of physiological functions (25). Our data showed that DFO treatment
recovered Akt activation from reduction by TRAIL treatment
(Fig. 2A and C) and decreased the
caspase-3 cleavage induced by TRAIL (Fig. 2A and D). Thus, these results suggest
that DFO confers a protective effect from TRAIL in colon cancer
cells.
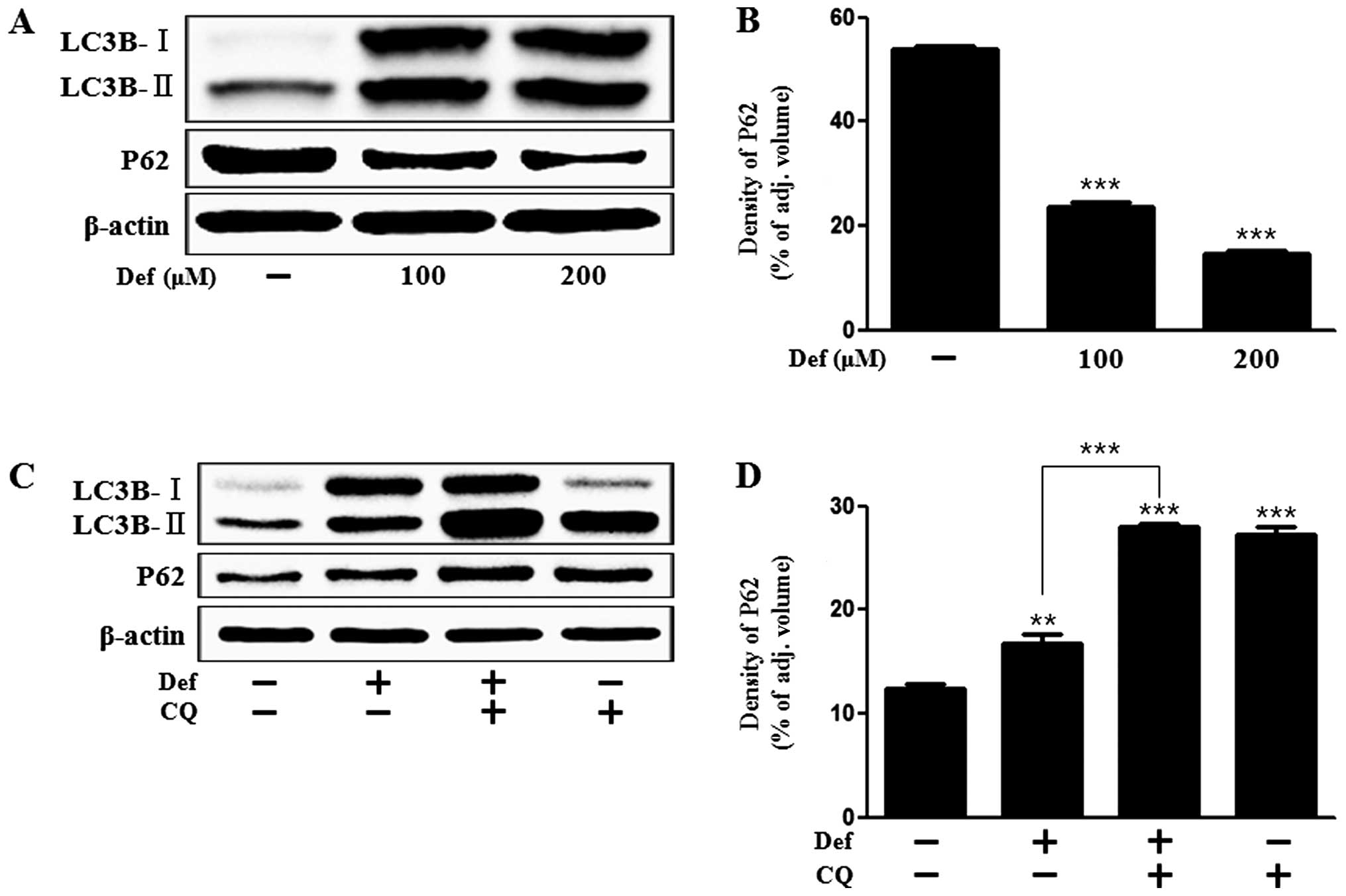
DFO induces autophagic flux
LC3 protein is considered to be an autophagy marker
and is localized and aggregated on autophagosomes. LC3 transforms
from LC3-I to LC3-II during autophagosome formation (26). We examined whether DFO induces
autophagy by assessing LC3 transformation. As shown in Fig. 3, the level of LC3-II was increased
and P62 expression was decreased in the DFO-treated group in a
dose-dependent manner compared with the control group as detected
by western blot analysis and densitometry (Fig. 3A and B). P62 is considered as a
crucial mediator for target protein to the autophagy system in
removal of aggregated proteins. P62 is degraded by itself during
autophagy (27). We used the
autophagy inhibitor, chloroquine (CQ), that is widely used to
inhibit the maturation of autophagosomes into degradative
autolysosomes (18,28). We identified that DFO-induced
upregulation of LC3-II was increased by CQ treatment since CQ
inhibited the fusion of autophagosomes and autolysosomes (Fig. 3C). For this reason, P62 was
increased by CQ treatment (Fig. 3C and
D).
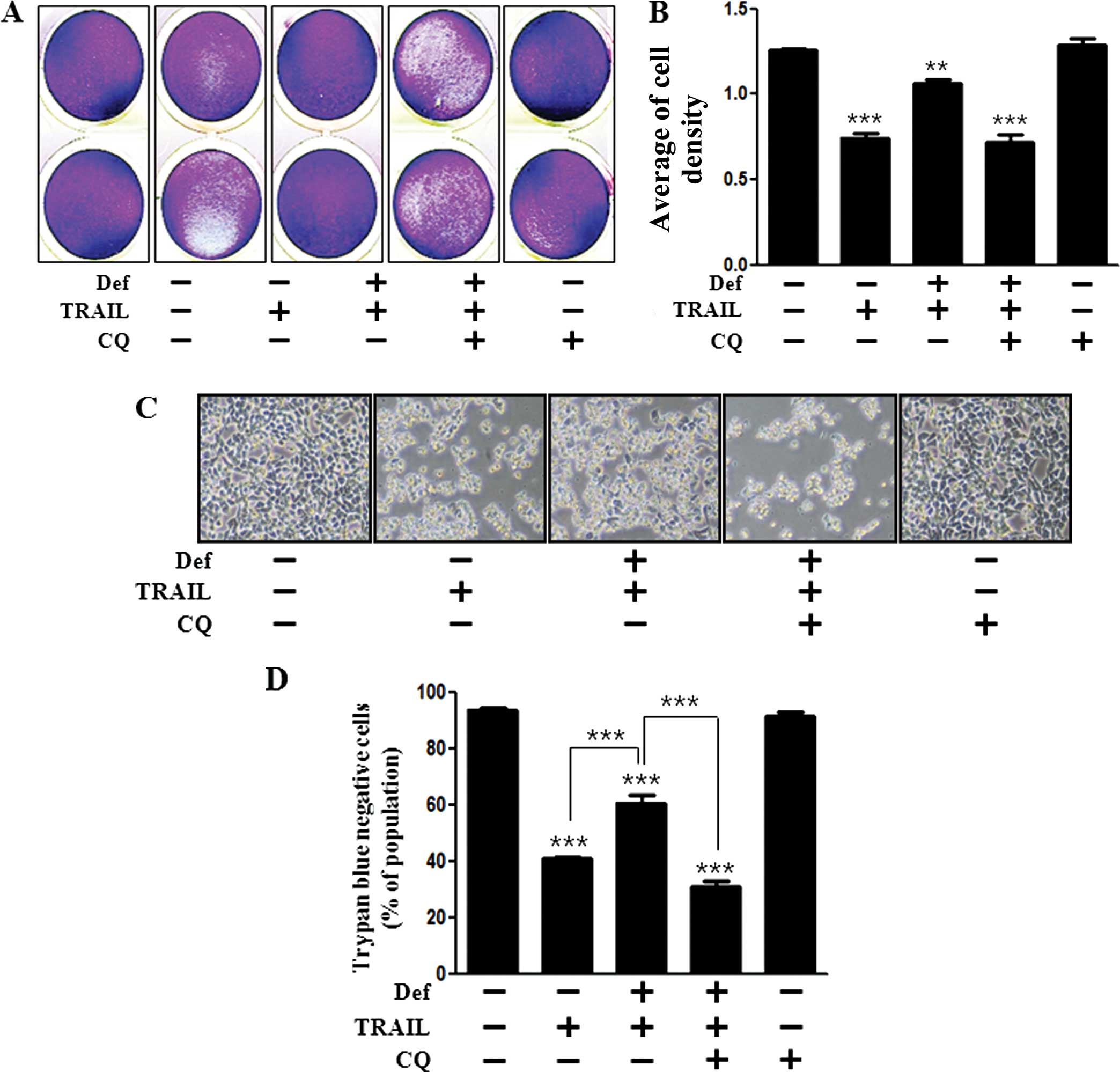
Autophagy inhibitor blocks DFO-mediated
inhibition of TRAIL-induced apoptosis
We analyzed the cell viability using CQ to
investigate the effect of DFO on the activation of autophagy. We
identified that the protective effect of DFO was reversed by CQ in
regards to cell viability by crystal violet assay and graphical
analysis (Fig. 4A and B).
Photographed images and trypan blue exclusion assay were
implemented for cell viability. Inhibition of cell death by DFO
treatment was reversed by CQ pre-treatment (Fig. 4C and D). These results strongly
indicate that DFO treatment inhibits TRAIL-mediated cytotoxicity
through induction of autophagy.
Discussion
Deferoxamine (DFO), an iron chelator, has been
reported to induce hypoxia and HIF-1α expression (29). HIF-1α is a crucial mediator of the
physiological response to hypoxia, and its dysfunction promotes
cancer angiogenesis and metastasis (30). We identified that DFO induced HIF-1α
stabilization. Several studies indicate that HIF-1α induces
autophagy in the cellular response to hypoxia (31–33).
One limitation of our study was that we did not verify whether DFO
induced autophagy through HIF-1α stabilization.
Studies suggest that autophagy is a double-edged
sword, with both beneficial and harmful potential in cancer
(34). Autophagy is the cellular
pathway that mediates lysosomal degradation of intracellular
long-lived macromolecules or organelles for subsequent reuse under
starvation, or stress such as oxidative stress, endoplasmic
reticulum stress, accumulation of abnormal protein and
physiological conditions of differentiation (35,36).
Autophagy inhibitor, chloroquine (CQ), is widely used to inhibit
the maturation of autophagosomes into degradative autolysosomes
(37). We identified that CQ, DFO
and TRAIL-treated cells underwent cell death to a greater extend
than DFO and TRAIL-treated cells (Fig.
4). We suggest that DFO-induced autophagy is a protective
effect against TRAIL in colon cancer cells. CQ-mediated inhibition
of autophagy interrupted the protective effect of autophagy.
Many studies suggest that TRAIL induces autophagy in
several types of cancer cells (38,39).
Yet, our results showed that TRAIL treatment did not mediate
autophagy marker, LC3-II (Fig. 3A).
Rather, TRAIL-treated LC3-II transformation was decreased when
compared with the control. Thus, we suggest that TRAIL is not
associated with autophagy in HCT116 colon cancer cells. Ikeda et
al proposed that DFO promoted Akt activation (40). As shown in Fig. 2A and C, DFO treatment activated Akt
phosphorylation dose-dependently. These findings support our
suggestion that DFO protects against TRAIL-induced cell death
through survival signaling in colon cancer cells.
The crosstalk between autophagy and apoptosis is
intricate and sometimes contradictory; however, it is an important
determinant of the overall fate of the cell. This report is the
first to indicate that DFO-mediated autophagy may play a critical
role in cell protection against TRAIL-induced cytotoxicity in colon
cancer.
The purpose of the present study was to investigate
the role of DFO in TRAIL-induced cell death and the possible
mechanism in human colon cancer cells. The results indicated that
DFO-induced autophagy flux inhibited the TRAIL-mediated anticancer
effect and also suggest that DFO can be a suppressor of anticancer
therapy, particularly in TRAIL-mediated colorectal cancer
therapy.
Acknowledgements
This study was supported by a grant from the
National Research Foundation of Korea (NRF), funded by the Korean
Government (2013R1A1A2063931).
References
|
1
|
Steller H: Mechanisms and genes of
cellular suicide. Science. 267:1445–1449. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Hickman JA: Apoptosis induced by
anticancer drugs. Cancer Metastasis Rev. 11:121–139. 1992.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Zhang JY: Apoptosis-based anticancer
drugs. Nat Rev Drug Discov. 1:101–102. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
MacFarlane M: TRAIL-induced signalling and
apoptosis. Toxicol Lett. 139:89–97. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Lieu PT, Heiskala M, Peterson PA and Yang
Y: The roles of iron in health and disease. Mol Aspects Med.
22:1–87. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Toyokuni S: Iron-induced carcinogenesis:
the role of redox regulation. Free Radic Biol Med. 20:553–566.
1996. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Nelson RL: Dietary iron and colorectal
cancer risk. Free Radic Biol Med. 12:161–168. 1992. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Bedford MR, Ford SJ, Horniblow RD, Iqbal
TH and Tselepis C: Iron chelation in the treatment of cancer: a new
role for deferasirox? J Clin Pharmacol. 53:885–891. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Nurtjahja-Tjendraputra E, Fu D, Phang JM
and Richardson DR: Iron chelation regulates cyclin D1 expression
via the proteasome: a link to iron deficiency-mediated growth
suppression. Blood. 109:4045–4054. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Fu D and Richardson DR: Iron chelation and
regulation of the cell cycle: 2 mechanisms of posttranscriptional
regulation of the universal cyclin-dependent kinase inhibitor
p21CIP1/WAF1 by iron depletion. Blood. 110:752–761.
2007. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Hershko C: Control of disease by selective
iron depletion: a novel therapeutic strategy utilizing iron
chelators. Baillieres Clin Haematol. 7:965–1000. 1994. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Olivieri NF and Brittenham GM:
Iron-chelating therapy and the treatment of thalassemia. Blood.
89:739–761. 1997.PubMed/NCBI
|
|
13
|
Yun Z, Maecker HL, Johnson RS and Giaccia
AJ: Inhibition of PPARγ2 gene expression by the HIF-1-regulated
gene DEC1/Stra13: a mechanism for regulation of adipogenesis by
hypoxia. Dev Cell. 2:331–341. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Nakayama K: Cellular signal transduction
of the hypoxia response. J Biochem. 146:757–765. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Semenza GL: Targeting HIF-1 for cancer
therapy. Nat Rev Cancer. 3:721–732. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Deter RL and De Duve C: Influence of
glucagon, an inducer of cellular autophagy, on some physical
properties of rat liver lysosomes. J Cell Biol. 33:437–449. 1967.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Levine B and Kroemer G: Autophagy in the
pathogenesis of disease. Cell. 132:27–42. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Boya P, González-Polo RA, Casares N, et
al: Inhibition of macroautophagy triggers apoptosis. Mol Cell Biol.
25:1025–1040. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Karantza-Wadsworth V, Patel S, Kravchuk O,
et al: Autophagy mitigates metabolic stress and genome damage in
mammary tumorigenesis. Genes Dev. 21:1621–1635. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Degenhardt K, Mathew R, Beaudoin B, et al:
Autophagy promotes tumor cell survival and restricts necrosis,
inflammation, and tumorigenesis. Cancer Cell. 10:51–64. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Lum JJ, Bauer DE, Kong M, et al: Growth
factor regulation of autophagy and cell survival in the absence of
apoptosis. Cell. 120:237–248. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Hu YL, DeLay M, Jahangiri A, et al:
Hypoxia-induced autophagy promotes tumor cell survival and
adaptation to antiangiogenic treatment in glioblastoma. Cancer Res.
72:1773–1783. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Chaudhari AA, Seol JW, Lee YJ, Seol DW and
Park SY: Hypoxia protects articular chondrocytes from
thapsigargin-induced apoptosis. Biochem Biophys Res Commun.
381:513–517. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Agarwal E, Brattain MG and Chowdhury S:
Cell survival and metastasis regulation by Akt signaling in
colorectal cancer. Cell Signal. 25:1711–1719. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Degterev A, Boyce M and Yuan J: A decade
of caspases. Oncogene. 22:8543–8567. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Rubinsztein DC, Cuervo AM, Ravikumar B, et
al: In search of an ‘autophagomometer’. Autophagy. 5:585–589. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Matsumoto G, Wada K, Okuno M, Kurosawa M
and Nukina N: Serine 403 phosphorylation of p62/SQSTM1 regulates
selective autophagic clearance of ubiquitinated proteins. Mol Cell.
44:279–289. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Kroemer G and Jäättelä M: Lysosomes and
autophagy in cell death control. Nat Rev Cancer. 5:886–897. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Liu Y, Cui Y, Shi M, Zhang Q, Wang Q and
Chen X: Deferoxamine promotes MDA-MB-231 cell migration and
invasion through increased ROS-dependent HIF-1α accumulation. Cell
Physiol Biochem. 33:1036–1046. 2014. View Article : Google Scholar
|
|
30
|
Bertout JA, Patel SA and Simon MC: The
impact of O2 availability on human cancer. Nat Rev
Cancer. 8:967–975. 2008. View
Article : Google Scholar : PubMed/NCBI
|
|
31
|
Mazure NM and Pouysségur J: Atypical
BH3-domains of BNIP3 and BNIP3L lead to autophagy in hypoxia.
Autophagy. 5:868–869. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Gustafsson AB and Gottlieb RA: Autophagy
in ischemic heart disease. Circ Res. 104:150–158. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Wang K, Liu R, Li J, et al: Quercetin
induces protective autophagy in gastric cancer cells: involvement
of Akt-mTOR- and hypoxia-induced factor 1α-mediated signaling.
Autophagy. 7:966–978. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
White E and DiPaola RS: The double-edged
sword of autophagy modulation in cancer. Clin Cancer Res.
15:5308–5316. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Glick D, Barth S and Macleod KF:
Autophagy: cellular and molecular mechanisms. J Pathol. 221:3–12.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Mizushima N: Autophagy: process and
function. Genes Dev. 21:2861–2873. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Mizushima N, Yoshimori T and Levine B:
Methods in mammalian autophagy research. Cell. 140:313–326. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Singh K, Sharma A, Mir MC, et al:
Autophagic flux determines cell death and survival in response to
Apo2L/TRAIL (dulanermin). Mol Cancer. 13:702014. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Park KJ, Lee SH, Kim TI, et al: A human
scFv antibody against TRAIL receptor 2 induces autophagic cell
death in both TRAIL-sensitive and TRAIL-resistant cancer cells.
Cancer Res. 67:7327–7334. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Ikeda Y, Tajima S, Yoshida S, et al:
Deferoxamine promotes angiogenesis via the activation of vascular
endothelial cell function. Atherosclerosis. 215:339–347. 2011.
View Article : Google Scholar : PubMed/NCBI
|